Abstract
BACKGROUND AND PURPOSE: The higher relaxivity of gadobenate dimeglumine compared with gadodiamide is potentially advantageous for contrast-enhanced brain MR imaging. This study intraindividually compared 0.1-mmol/kg doses of these agents for qualitative and quantitative lesion enhancement.
MATERIALS AND METHODS: Adult patients with suggested or known brain lesions underwent 2 identical MR imaging examinations at 1.5T, one with gadobenate dimeglumine and the other with gadodiamide. The agents were administered in randomized order separated by 3–14 days. Imaging sequences and postinjection acquisition timing were identical for the 2 examinations. Three blinded readers evaluated images qualitatively for diagnostic information (lesion extent, delineation, morphology, enhancement, and global preference) and quantitatively for contrast-to-noise ratio (CNR).
RESULTS: One hundred thirteen of 138 enrolled patients successfully underwent both examinations. Final diagnoses were intra-axial tumor, metastasis, extra-axial tumor, or other (47, 27, 18, and 21 subjects, respectively). Readers 1, 2, and 3 demonstrated global preference for gadobenate dimeglumine in 63 (55.8%), 77 (68.1%), and 73 (64.6%) patients, respectively, compared with 3, 2, and 3 patients for gadodiamide (P < .0001, all readers). Highly significant (P < .0001, all readers) preference for gadobenate dimeglumine was demonstrated for all qualitative end points and for CNR (increases of 23.3%–34.7% and 42.4%–48.9% [spin-echo and gradient-refocused echo sequences, respectively] for gadobenate dimeglumine compared with gadodiamide). Inter-reader agreement was good for all evaluations (κ = 0.47–0.69). Significant preference for gadobenate dimeglumine was demonstrated for all lesion subgroup analyses.
CONCLUSION: Significantly greater diagnostic information and lesion enhancement are achieved on brain MR imaging with 0.1-mmol/kg gadobenate dimeglumine compared with gadodiamide at an equivalent dose.
Effective management of patients with tumors of the central nervous system (CNS) depends on accurate detection and characterization of enhancing lesions. Enhancement of tumors on gadolinium-enhanced MR imaging is a key imaging feature used in guiding surgical resection, delineating appropriate radiosurgical target volumes, and following patients for disease recurrence.1,2
Five gadolinium-based contrast agents are currently approved by the US Food and Drug Administration for MR imaging of the CNS. Although these agents have different molecular structures and physicochemical properties, they share the ability to transiently accumulate in areas with an abnormal blood-brain barrier, leading to faster T1 relaxation times and demonstrable contrast enhancement. Of these agents, gadobenate dimeglumine (Gd-BOPTA, MultiHance; Bracco, Milan, Italy) exhibits the highest R1 and R2 relaxivity in vivo.3,4 The increased relaxivity derives from weak and transient interactions of the Gd-BOPTA contrast-effective molecule with serum albumin5,6 and leads to increased signal-intensity (SI) enhancement relative to that obtained with other agents at an equivalent dose. Recent comparative studies in patients with brain or spinal tumors7–12 have shown that this increased SI enhancement translates into significantly greater diagnostic performance compared with gadopentetate dimeglumine (Gd-DTPA, Magnevist; Bayer Healthcare, Leverskusen, Germany)7–9 and an agent approved solely in Europe (gadoterate meglumine, Gd-DOTA; Dotarem; Guerbet, Aulnay-sous-Bois, France).10
On the basis of R1 relaxivity values alone,3,4 one might expect similar diagnostic superiority for gadobenate dimeglumine over gadodiamide (Gd-DTPA-BMA, Omniscan; GE Healthcare, Waukesha, Wis) when these agents are compared at equivalent doses, particularly given the similar diagnostic performance of gadodiamide and gadopentetate dimeglumine in patients with CNS disease.13,14 However, whereas early interindividual parallel-group studies to compare gadobenate dimeglumine and gadodiamide revealed equivalence for a double (0.2 mmol/kg of body weight [BW]) dose (or a 1.5-fold dose) of gadobenate dimeglumine compared with a triple (0.3 mmol/kg BW) dose of gadodiamide, little if any differences were noted between single (0.1 mmol/kg BW) doses of these agents.15,16 The aim of this study (MR EvaluatioN of MultiHance And OmniscaN for Contrast Enhancement [the MR-ENHANCE study]) was, therefore, to compare more accurately these 2 agents by using a controlled multicenter double-blind randomized intraindividual crossover study design in which each patient received 0.1-mmol/kg doses of both these agents in 2 identical MR imaging examinations.
Materials and Methods
The study was Health Insurance Portability and Accountability Act–compliant and was conducted according to Good Clinical Practice standards. All patients signed an approved informed-consent form before enrollment. The lead author (H.A.R.) had complete access to the results of the study, and all authors had control of the data and statistical results included in this article.
Patients
A total of 138 patients (79 men, 59 women) referred for contrast-enhanced MR imaging for known or suggested brain tumors were screened and enrolled in a consecutive manner at each of 16 participating centers between May 2006 and February 2007. Patients were ineligible for enrollment if they had received any investigational drug within 30 days before administration of either study agent. Subjects were also excluded if they were to receive any treatment that could change the visualization of CNS lesions before or between the 2 examinations (eg, whole-brain fractionated radiation therapy, investigational drugs, steroids, or chemotherapy). Patients were also ineligible if they were pregnant or nursing or had impaired renal function, congestive heart failure, claustrophobia, gadolinium allergy, a cardiac pacemaker, or other contraindications to MR imaging.
Two patients withdrew from the study after signing the informed consent form but before administration of either contrast agent. The remaining 136 eligible patients (78 men, 58 women; mean age, 54.1 ± 14.1 years; age range, 18–80 years) were prospectively randomized to 2 study groups (groups A and B) to receive contrast agent according to 1 of 2 administration orders. Group A (n = 67) received gadobenate dimeglumine for the first examination and gadodiamide for the second examination and group B (n = 69) received the agents in the opposite order.
MR Imaging
MR imaging was performed on 1.5 T systems from several vendors (Siemens, Erlangen, Germany; GE Healthcare; Philips Medical Systems, Best, The Netherlands; and Toshiba Medical Systems, Zoetermeer, the Netherlands) by using a standard head coil. A controlled imaging protocol comprising T1-weighted spin-echo (T1SE) and T2-weighted fast SE acquisitions before contrast injection and T1SE and T1-weighted gradient-recalled echo (T1GRE) acquisitions after injection ensured protocol uniformity across sites and within individual patients. The parameters for the imaging sequences varied between investigational centers because of the different imaging systems in use at these centers. However, the same MR imaging scanner, imaging planes, section prescriptions, and sequence parameters were used for both examinations in each patient. The range of parameters for the T1SE sequence was as follows: TR, 350–635 ms; TE, 9–20 ms; NEX, 1–3; section thickness, 3–5 mm; intersection gap, 0–1.5 mm; FOV, 20–35 cm; acquisition matrix, 168–320 × 144–512. The parameters for the T1GRE sequence ranged as follows: TR, 6–2150 ms; TE, 1.6–17 ms; flip angle, 8°-90°; NEX, 1–4; section thickness, 3–5 mm; intersection gap, 0–1.5 mm; FOV, 21–38 cm; matrix, 169–512 × 128–1024.
Contrast agent administration was performed intravenously in an identical manner in both examinations by using either a manual bolus injection (n = 113) or a power injector (n = 23) at 2 mL/s at an identical 0.1-mmol/kg BW dose and volume (0.2 mL/kg BW of a 0.5-mol/L formulation). Each contrast agent was administered in a blinded fashion in the order determined by a randomization list. Acquisition of the postcontrast T1-weighted images began at a fixed time point, which was mandated to occur between 3 and 10 minutes after injection but could vary within this range, depending on the site-specific protocol. However, the postcontrast scannings were controlled to be identical in terms of timing and sequence order for both examinations in each patient. The interval between the 2 MR imaging examinations was >48 hours in all patients to avoid any carryover effect, but <14 days to minimize the chance of measurable disease progression or lesion evolution.
Image Evaluation
All images were evaluated by 3 independent experienced neuroradiologists (J.R., S.B., C.C.), who were unaffiliated with the study centers and blinded to the contrast agent used, all patient clinical and radiologic information, and all interpretations by on-site investigators. Each reader evaluated the patient images separately and independently.
All images from each patient were evaluated in global matched-pairs fashion. Images were presented for review on a multimonitor imaging workstation. For each randomized patient number, all images from the first examination (examination 1) were displayed simultaneously with the corresponding images from the second examination (examination 2). Each reader was able to perform all routine interactive image-manipulation functions (eg, window/level, zoom, pan) on both image sets. If the postinjection images from either examination were considered technically inadequate by any of the 3 readers (eg, if artifacts compromised interpretability), no further assessment was performed for that patient by that reader. Once the readers’ assessments were recorded and signed off on an electronic case report form (e-CRF), the data base for that reading was automatically locked.
Qualitative Assessment of Diagnostic Information
Technically adequate images were evaluated qualitatively for diagnostic information and scored in terms of the following: 1) lesion border delineation, 2) disease extent, 3) visualization of lesion internal morphology, and 4) lesion contrast enhancement compared with surrounding normal tissue. All assessments were performed by using 3-point scales from −1 (examination 1 superior) through 0 (both examinations equal) to +1 (examination 2 superior). For the various end points, superiority was recorded for 1 of the examinations if it allowed better separation of 1 or more lesions from surrounding tissue, structures, or edema; better definition of lesion extent; clearer depiction of intralesion features; better difference in SI between lesions and surrounding normal tissue; or depiction of 1 or more lesions only after that examination.
The readers also expressed whether they had an overall diagnostic preference for 1 examination versus the other. For cases in which a reader had an overall diagnostic preference for 1 examination over the other, the reader then selected 1 or more of the following 6 reasons for this preference: superior contrast enhancement, better delineation of normal structure, better delineation of at least 1 lesion, better visualization of lesion internal structure, more lesions identified, or greater diagnostic confidence. If diagnostic confidence was selected as a reason for global preference, we required 1 or more of the following specifications: detection of lesions, characterization of disease, presumptive grading of disease (ie, high- or low-grade in the case of gliomas), definition of disease extent, or other reasons (to be specified on the e-CRF).
Quantitative Assessment
Quantitative evaluation of ≤3 enhancing lesions per patient was performed by each reader independently by using a simultaneous matched-pairs approach. Measurements of SI were made at regions of interest positioned on areas of normal brain parenchyma and on ≤3 lesions identified on postcontrast images from both examinations. Additional SI measurements were made at regions of interest placed in selected areas external to the brain to determine the background noise. To ensure that regions of interest of equal size were positioned at identical coordinates on all corresponding image sets, we ensured that each region of interest placed on the selected postinjection image from 1 examination appear simultaneously on the corresponding image from the other examination, always taking care to avoid inclusion of vessels. If multiple lesions were present in a given patient, regions of interest were placed on ≤3 of the largest most conspicuous lesions. All SI measurements were made by using Analyze software, Version 4.0 (Mayo Foundation, Rochester, Minn) and were subsequently used to calculate the contrast-to-noise ratio (CNR) for both T1SE and T1GRE acquisitions. CNR was calculated as described previously.12
Safety Assessments
Monitoring for adverse events for all patients (n = 136) was performed from the moment the patient signed the informed consent form until 24 hours after administration of the first study agent and then again from the moment the second study agent was administered until 24 hours after administration of the second agent. Adverse events were classified by the principal investigator at each center as either serious (ie, death, life-threatening, requiring or prolonging hospitalization) or not serious, and any perceived relationship to the agent was recorded.
Statistical Analysis
Power determination was based on the primary efficacy assumption that a 0.1-mmol/kg BW dose of gadobenate dimeglumine is superior to an equivalent dose of gadodiamide according to reader preference. On the basis of the results of a recent study8 and assuming a patient drop-out rate of 20%, we needed enrollment of approximately 136 subjects to demonstrate superiority with 90% of power (nQuery Advisor, Version 5.0; Statistical Solutions, Cork, Ireland).
Analysis of blinded reader evaluations was performed by using the statistical software package SAS, Version 8.2 (SAS Institute, Cary, NC). The distribution of reader preferences in diagnostic-information end points was tested statistically by using the Wilcoxon signed rank test. Separate analyses were performed for all evaluable patients together (n = 113) as well as for the largest subsets of patients (ie, patients with gliomas [n = 47], brain metastases [n = 27], and extra-axial lesions [n = 18]). Inter-reader agreement for diagnostic findings was presented as percentage agreement for the 3 readers and assessed by using generalized κ statistics.
Differences between gadobenate dimeglumine and gadodiamide in terms of quantitative study-agent effect were analyzed by using a mixed-effect model. The change from predose was the response variable, and factors included in the model were patient, period, sequence, study agent, and predose score, for which a patient nested within the sequence was the random effect. All statistical tests were conducted at a significance level of P < .05.
Results
Patients
Of the 136 patients randomized and evaluated for safety, 21 (15.4%) prematurely terminated the study after the first contrast agent administration (11 after gadobenate dimeglumines, 10 after gadodiamide) and were excluded from subsequent efficacy evaluations. The reasons for discontinuation were withdrawal of consent (n = 12; 5 after gadobenate dimeglumine, 7 after gadodiamide), initiation of therapy (surgery or radiation therapy, n = 5), deterioration of patient's condition resulting in hospitalization (n = 2), bronchitis that prevented the patient from returning for the second examination (n = 1), and an unrelated adverse event (pulmonary embolism, n = 1). Two further patients were excluded from evaluation due to technical failure (image retrieval) of the postcontrast acquisitions of 1 of their examinations. A total of 113 patients were, therefore, evaluated for diagnostic efficacy by the 3 off-site blinded readers.
The evaluable population comprised 65 men and 48 women, ranging in age from 18 to 80 years (median, 54.4 years). Half were randomized to receive gadobenate dimeglumine on the first examination (n = 56) and the other half (n = 57) received gadodiamide first. No differences in demographic features were apparent between the groups. The diagnoses of these 113 patients were primary glial tumor in 47 (42%) patients (glioma [n = 16], glioblastoma multiforme [n = 16], oligodendroglioma [n = 10], astrocytoma [n = 5]); secondary metastases in 27 (24%) patients (from primary lung cancer [n = 11], breast cancer [n = 5], colon cancer [n = 2], salivary gland cancer [n = 1], sarcoma [n = 1], unknown cancer [n = 7]); extra-axial lesions in 18 (16%) patients (meningioma [n = 14], acoustic neuroma/schwannoma [n = 4]); and other or unspecified diagnosis in 17 (15%) patients (cavernous angioma/hemangioma [n = 3], postoperative scar/fibrosis [n = 3], pineocytoma/neurocytoma [n = 3], lymphoma/demyelinization [n = 1], multiple sclerosis [n = 1], arachnoid cyst [n = 1], unspecified/unknown mass lesion [n = 5]). In the remaining 4/113 (3%) patients, no lesions were detected on either examination.
Qualitative Image Assessment
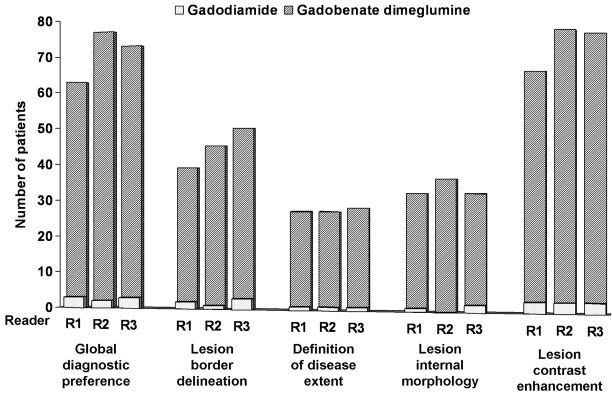
All of the image sets from each of the 113 evaluable patients were technically adequate for assessment. The findings of the 3 blinded readers for global diagnostic preference and each of the individual diagnostic information end points are shown in Fig 1. Readers 1, 2, and 3 demonstrated global diagnostic preference for gadobenate dimeglumine in 63 (55.8%), 77 (68.1%), and 73 (64.6%) patients, respectively, compared with 3 (2.7%), 2 (1.8%), and 3 (2.7%) patients, respectively, for gadodiamide (P < .0001, all readers). Similar highly significant preference was demonstrated for each individual diagnostic information end point (P < .0001, all evaluations).
Fig 1.
Reader preference for gadobenate dimeglumine or gadodiamide based on blinded qualitative evaluation. Each reader expresses a highly significant (P < .0001) preference for gadobenate dimeglumine for each parameter. Note that the number of patients for whom equality is expressed is not shown.
Subset analyses of patients with primary glial tumors (Figs 2 and 3), metastases (Fig 4), and extra-axial lesions revealed similar highly significant preferences for gadobenate dimeglumine for all diagnostic information end points in the case of glial tumors and metastases and for global diagnostic preference and lesion contrast enhancement in the case of extra-axial lesions (Table 1). When a contrast agent preference was expressed in patients with extra-axial lesions, this preference always favored gadobenate dimeglumine; however, due to the small number of patients in this group, reader preferences for gadobenate dimeglumine over gadodiamide rarely achieved statistical significance. Although subgroup analyses were not performed of nontumoral lesions, readers 1, 2, and 3 each preferred gadobenate dimeglumine whenever a preference was expressed.
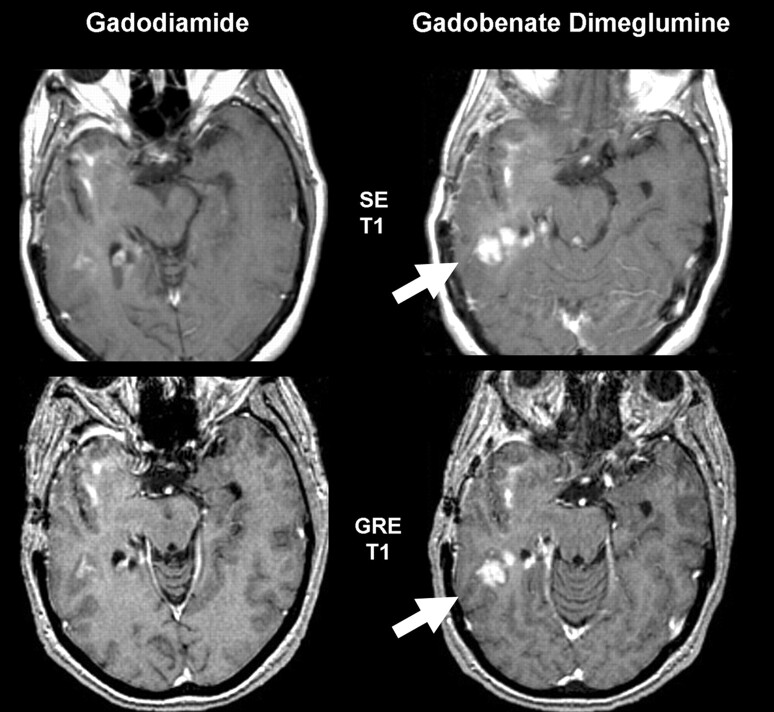
Fig 2.
Glioblastoma with recurrent disease. This 55-year-old woman shows more conspicuous enhancement (arrows) in the right posterior temporal lobe with gadobenate dimeglumine than gadodiamide for both SE and GRE sequences. Although there are differences in angulation, changes were confirmed by review of all adjacent sections.
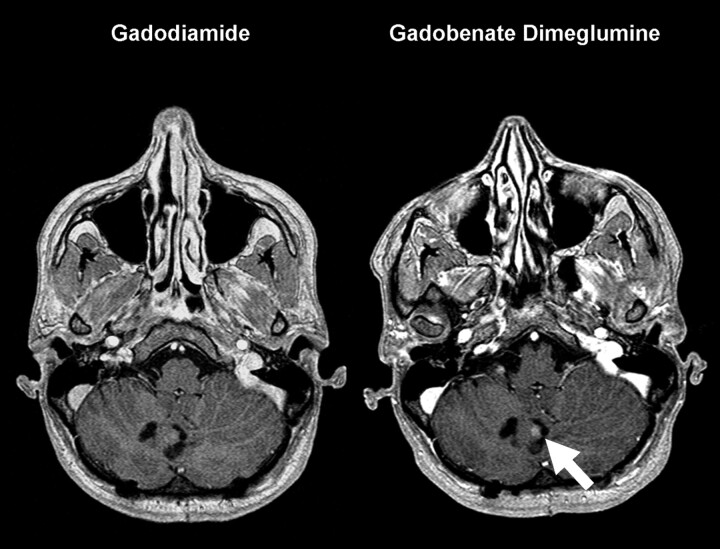
Fig 3.
A 32-year-old woman with primary cerebellar glioma, which had previously been resected. A solid nodule of enhancement (arrow) is seen convincingly on the gadobenate dimeglumine–enhanced image. The clear visualization of contrast enhancement was important in postoperative decision making.
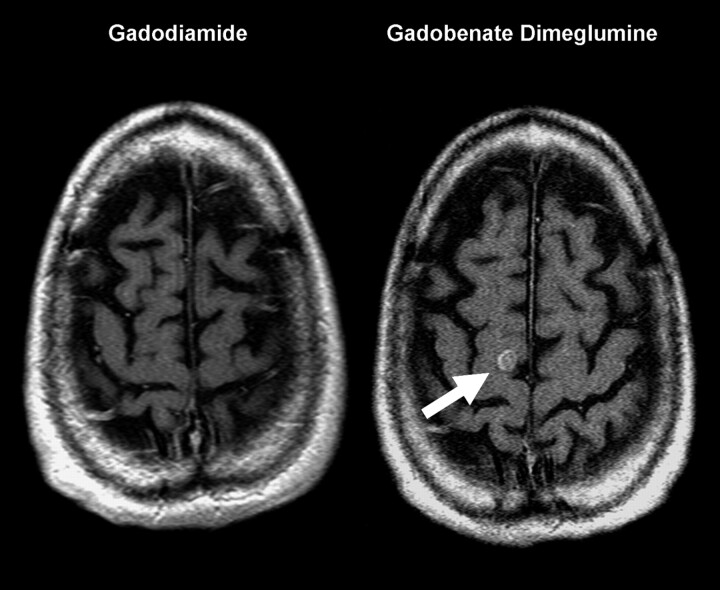
Fig 4.
A 46-year-old man with a primary anaplastic carcinoma of the small bowel who developed sensory changes in the left upper extremity. The solitary metastasis (arrow) in the right superior frontal gyrus is only visualized on the gadobenate dimeglumine–enhanced image.
Table 1:
Qualitative assessments of patients with glial tumors, metastases, and extra-axial lesions
| Diagnostic Information End Point | Reader | Glial Tumors (n = 47) |
Metastases (n = 27) |
Extra-Axial Lesions (n = 18) |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Gadobenate Dimeglumine Preferred (%) | Gadodiamide Preferred (%) | P | Gadobenate Dimeglumine Preferred (%) | Gadodiamide Preferred (%) | P | Gadobenate Dimeglumine Preferred (%) | Gadodiamide Preferred | P | ||
| Global diagnostic preference | 1 | 22 (46.8%) | 1 (2.1) | <.0001 | 20 (74.1) | 2 (7.4) | <.0001 | 14 (77.8) | 0 | .0001 |
| 2 | 35 (74.5) | 1 (2.1) | <.0001 | 18 (66.7) | 1 (3.7) | <.0001 | 13 (72.2) | 0 | .0002 | |
| 3 | 25 (53.2) | 1 (2.1) | <.0001 | 19 (70.4) | 2 (7.4) | <.0001 | 16 (88.9) | 0 | <.0001 | |
| Lesion border delineation | 1 | 15 (31.9) | 1 (2.1) | .0005 | 18 (66.7) | 1 (3.7) | <.0001 | 1 (5.6) | 0 | 1.00 |
| 2 | 18 (38.3) | 1 (2.1) | <.0001 | 17 (63.0) | 0 | <.0001 | 3 (16.7) | 0 | .25 | |
| 3 | 16 (34.0) | 1 (2.1) | .0003 | 17 (63.0) | 2 (7.4) | .0007 | 7 (38.9) | 0 | .0156 | |
| Definition of disease extent | 1 | 8 (17.0) | 0 | .0078 | 13 (48.1) | 1 (3.7) | .0018 | 1 (5.6) | 0 | 1.00 |
| 2 | 7 (14.9) | 0 | .0156 | 13 (48.1) | 1 (3.7) | .0018 | 2 (11.1) | 0 | .50 | |
| 3 | 7 (14.9) | 0 | .0156 | 16 (59.3) | 1 (3.7) | .0003 | 1 (5.6) | 0 | 1.00 | |
| Visualization of lesion internal morphology | 1 | 10 (21.3) | 0 | .002 | 13 (48.1) | 1 (3.7) | .0018 | 2 (11.1) | 0 | .50 |
| 2 | 14 (29.8) | 0 | .0001 | 13 (48.1) | 0 | .0002 | 3 (16.7) | 0 | .25 | |
| 3 | 8 (17.0) | 0 | .0078 | 14 (51.9) | 2 (7.4) | .0042 | 4 (22.2%) | 0 | .125 | |
| Lesion contrast enhancement | 1 | 23 (48.9) | 1 (2.1) | <.0001 | 20 (74.1) | 2 (7.4) | <.0001 | 15 (83.3) | 0 | .0001 |
| 2 | 35 (74.5) | 2 (4.3) | <.0001 | 17 (63.0) | 1 (3.7) | 0.0001 | 14 (77.8) | 0 | .0001 | |
| 3 | 2 (55.3) | 1 (2.1) | <.0001 | 20 (74.1) | 2 (7.4) | <.0001 | 16 (88.9) | 0 | <.0001 | |
Further evaluation of patients for whom a preference for one or the other agent was expressed revealed that in most cases, this was due to a combination of superior contrast enhancement and better delineation of lesions and/or lesion internal structures (Table 2). When greater diagnostic confidence for 1 agent was expressed, the reason was almost exclusively superior definition of disease extent. Although the focus of the study was on comparing the agents for diagnostic information and quantitative lesion enhancement rather than lesion detection, differences in lesion number were nevertheless noted for 5 of 27 patients with metastases by 1 or more readers. In 3 of these 5 patients, the patient had ≥12 lesions on both examinations, and thus the detection of additional lesions with gadobenate dimeglumine in 2 patients (in 1 patient by all 3 readers and in 1 patient by 1 reader only) and gadodiamide in the other patient (by all 3 readers) was of little clinical relevance. The other 2 cases comprised a patient for whom 2 readers detected 2 lesions with gadodiamide but 3 lesions with gadobenate dimeglumine and a patient for whom 1 reader detected 3 lesions with gadodiamide but 4 lesions with gadobenate dimeglumine.
Table 2:
Reasons for global diagnostic preference as expressed by expert blinded neuroradiologists
| Specifications of Global Diagnostic Preference | Reader 1 (n = 66) |
Preference Expressed Reader 2 (n = 79) |
Reader 3 (n= 76) |
|||
|---|---|---|---|---|---|---|
| Gadobenate Dimeglumine (n= 63) (%) | Gadodiamide (n= 3) (%) | Gadobenate Dimeglumine (n = 77) (%) | Gadodiamide (n = 2) (%) | Gadobenate Dimeglumine (n= 73) (%) | Gadodiamide (n = 3) (%) | |
| Superior contrast enhancement | 63 (95.5) | 2 (3.0) | 74 (93.7) | 2 (2.5) | 73 (96.1) | 3 (3.9) |
| Better delineation of normal structures | 0 | 0 | 7 (8.9) | 0 | 3 (3.9) | 0 |
| Better delineation of at least 1 lesion | 33 (50.0) | 1 (1.5) | 39 (49.4) | 0 | 44 (57.9) | 3 (3.9) |
| Better visualization of lesion internal structure | 10 (15.2) | 0 | 30 (38.0) | 0 | 19 (25.0) | 0 |
| Detection of more lesions | 3 (4.8) | 1 (1.5) | 3 (3.8) | 1 (1.3) | 2 (2.6) | 1 (1.3) |
| Greater diagnostic confidence | 10 (15.2) | 0 | 10 (12.7) | 1 (1.3) | 10 (13.2%) | 0 |
Analysis of 3-reader agreement revealed generalized weighted κ values ranging from κ = 0.48 for global diagnostic preference and lesion-border delineation to κ = 0.66 for definition of disease extent when all 113 patients were considered together. All 3 blinded readers agreed completely in their assessments for 61%–81% of the patients, depending on the diagnostic information end point under consideration. Three-reader agreement for the evaluated patient subsets revealed similarly high levels of agreement for glial tumors (κ = 0.43–0.57) and metastases (κ = 0.56–0.63). Lower levels of agreement (κ = −0.14–0.47) were noted in the case of the 18 extra-axial lesions, reflecting the comparatively small number and widely heterogeneous character of the lesions in this subset.
Quantitative Evaluation
Readers 1, 2, and 3 recorded lesion SI measurements relative to normal brain parenchyma and background noise for 80, 85, and 82 lesions, respectively, on T1SE images and for 73, 80, and 74 lesions, respectively, on T1GRE images. Subsequent determinations of CNR for both sequences revealed highly significant (P < .0001, all readers) increases in quantitative enhancement with gadobenate dimeglumine relative to gadodiamide when all lesions were considered together (Table 3). Although the magnitude of SI enhancement was in all cases lower on GRE acquisitions compared with SE acquisitions, the corresponding increase in CNR for gadobenate dimeglumine relative to gadodiamide was greater (23.3%–34.7% for T1SE sequences, 42.4%–48.9% for T1GRE sequences).
Table 3:
Comparison of CNR values on T1SE and T1GRE sequences after administration of gadobenate dimeglumine and gadodiamide for all evaluated lesions and for lesion subsets
| Lesion Type | Reader | Postdose SE Sequences |
Postdose GRE Sequences |
||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean CNR Values |
% Increase in CNR with Gadobenate Dimeglumine (P) | Mean CNR Values |
% Increase in CNR with Gadobenate Dimeglumine (P) | ||||||
| Gadobenate Dimeglumine | Gadodiamide | Gadobenate Dimeglumine | Gadodiamide | ||||||
| All lesions | 1 | 61.07 | 46.65 | 30.9 | <.0001 | 32.26 | 22.65 | 42.4 | <.0001 |
| 2 | 46.46 | 37.67 | 23.3 | <.0001 | 23.59 | 16.19 | 45.7 | <.0001 | |
| 3 | 54.71 | 40.62 | 34.7 | <.0001 | 26.56 | 17.84 | 48.9 | <.0001 | |
| Glial tumors | 1 | 47.08 | 35.28 | 33.5 | .0002 | 30.57 | 21.67 | 41.1 | .0068 |
| 2 | 39.95 | 32.97 | 21.2 | .0125 | 22.80 | 15.81 | 44.2 | .0002 | |
| 3 | 43.84 | 36.82 | 19.1 | .0993 | 25.08 | 20.01 | 25.3 | .0466 | |
| Metastases | 1 | 60.97 | 46.52 | 31.1 | .0162 | 27.41 | 20.63 | 32.9 | .0261 |
| 2 | 46.23 | 37.32 | 23.9 | .0119 | 24.14 | 16.02 | 50.7 | <.0001 | |
| 3 | 51.27 | 37.63 | 36.2 | .0098 | 24.81 | 15.46 | 60.5 | .0004 | |
| Extra-axial lesions | 1 | 80.57 | 67.17 | 19.9 | .0046 | 41.44 | 28.83 | 43.7 | .0021 |
| 2 | 62.61 | 49.21 | 27.2 | .0388 | 27.30 | 20.19 | 35.2 | .0031 | |
| 3 | 77.12 | 54.18 | 42.3 | <.0001 | 33.97 | 21.10 | 61.0 | <.0001 | |
Note:—SE indicates spin-echo; GRE, gradient recalled-echo; CNR, contrast-to-noise ratio.
Subset analyses of quantitative enhancement were performed for 25, 35, and 30 glial tumors; 29, 26, and 26 metastases; and 17, 16, and 17 extra-axial lesions (readers 1, 2, and 3, respectively) on T1SE images; and for 22, 32, and 26 glial tumors; 27, 25, and 23 metastases; and 16, 15, and 16 extra-axial lesions, respectively, on T1GRE images. Highly significant increases in CNR with gadobenate dimeglumine relative to gadodiamide were noted by each reader for each lesion type (Table 3). Again, the magnitude of CNR increases with gadobenate dimeglumine was greater for SI measurements made on T1GRE acquisitions, though the absolute CNR values were higher on T1SE acquisitions.
Safety
Of the 136 patients who received at least 1 contrast agent, 126 received gadobenate dimeglumine and 125 received gadodiamide. No clinically meaningful differences were noted in terms of the incidence of adverse events: 4/126 (3.2%) patients reported 1 or more nonserious adverse events after gadobenate dimeglumine compared with 1/125 (0.8%) patients after gadodiamide. All contrast-related adverse events reported by these patients were mild and self-resolving and comprised minor gastrointestinal symptoms, headache, confusion after gadobenate dimeglumine, and oral hypoesthesia after gadodiamide.
Discussion
This blinded intraindividual crossover comparison demonstrates that gadobenate dimeglumine provides significantly better qualitative and quantitative enhancement of brain lesions compared with gadodiamide when both agents are administered at an equivalent dose of 0.1 mmol/kg. All 3 blinded readers reported highly significant (P < .0001) preference for gadobenate dimeglumine for clinically relevant determinations of lesion border delineation, definition of disease extent, visualization of lesion internal morphology, and lesion contrast enhancement. They also preferred gadobenate dimeglumine globally in more than half (63 [55.8%], 77 [68.1%], and 73 [64.6%] patients; readers 1, 2, and 3, respectively) of the patients evaluated. Concerning quantitative enhancement, the increases in CNR with gadobenate dimeglumine compared with gadodiamide on T1SE images were generally between 20% and 35%, which correspond to the magnitude of increase typically observed with a double dose of gadolinium contrast agent compared with a single dose.17,18
Our results are similar to those of a recent intraindividual crossover comparison of 0.1-mmol/kg BW gadobenate dimeglumine with an identical dose of gadopentetate dimeglumine8 and may be attributed to the roughly twofold higher R1 relaxivity of gadobenate dimeglumine in blood.3–6 This twofold higher R1 relaxivity, which is independent of the field strength of the MR magnet,3 is due to weak and transient interactions of the Gd-BOPTA molecule with serum albumin,5,6 which slow the tumbling rate of the Gd-BOPTA complex in blood. On standard 1.5T imaging systems used in this study, R1 relaxivity values of 6.3–7.9 L·mmol−1·second–1 have been determined for gadobenate dimeglumine in plasma (37°C) compared with values of 3.9–4.1 L · mmol−1·second–1 for gadopentetate dimeglumine3,4 and 4.3 L·mmol−1 ·second–1 for gadodiamide.4 Numerous studies have shown that the greater R1 relaxivity of gadobenate dimeglumine translates into improved SI enhancement for a variety of MR imaging applications.19–22
Compared with the results of previous parallel-group (interindividual) comparisons,15,16 which concluded that gadobenate dimeglumine and gadodiamide provide similar diagnostic information when administered at an equivalent dose of 0.1 mmol/kg, the results of this study highlight the value of an intraindividual approach in demonstrating significant differences in diagnostically relevant contrast-enhancement characteristics.
These results have clinically important implications, particularly for the evaluation and follow-up of gliomas and metastases. In the case of glial tumors, it is well-established that macroscopically complete surgical removal is associated with improved prognosis and longer patient survival.1,23 Because glial tumors often extend beyond the contrast-enhancing and T2 signal intensity margins,24 frequently only partial tumor resection is achieved at surgery, resulting in residual tumor on follow-up imaging. For these lesions, the superiority of gadobenate dimeglumine may lie in better defining the extent and internal morphology of lesions,7–10 thereby potentially improving patient management and surgical planning. In addition, improved detection of residual tumor on early postoperative MR imaging may also improve patient prognosis.23,25
Whereas defining the surgical target volume is important for glial tumors, for patients with intracranial metastases, the need is to accurately define the precise number, size, and location of lesions to select the most appropriate treatment option.26,27 A possible advantage of gadobenate dimeglumine in this setting is the detection of additional lesions or the improved depiction of small or poorly enhancing lesions that might otherwise go undetected with a standard dose of gadodiamide or another conventional gadolinium agent. In this study, 2 of 27 patients with brain metastases who had 2 or 3 lesions detected after gadodiamide, each had an additional lesion detected after gadobenate dimeglumine. In these patients, the additional lesions detected with gadobenate dimeglumine might have altered the approach to patient management from one involving aggressive surgery to one based on radiation therapy.
In the case of extra-axial lesions (meningiomas and schwannomas), the greater SI enhancement achievable with gadobenate dimeglumine improves lesion conspicuity and visibility but may have only minimal impact on patient-management decisions.
Another setting in which the increased R1 relaxivity of gadobenate dimeglumine may be of benefit is in intraoperative imaging for the real-time assessment of surgical resection. Although 1.5T systems are increasingly available for intraoperative applications,28 many interventional systems operate at lower (0.2–0.5T) field strengths.29 These systems have comparatively low SI-to-noise ratio (SNR) and may benefit from the increased SI provided by gadobenate dimeglumine.
Our findings have potentially important clinical implications for interpretation of serial brain tumor MR examinations when these are done with different gadolinium agents. Because gadobenate dimeglumine often shows visibly greater enhancement and lesion definition, interpretation of progression or regression of tumor compared with earlier or later scans with gadodiamide would need to take this factor into account. Our results suggest that it may be preferable to use gadobenate dimeglumine consistently to provide the greatest diagnostic information and avoid the pitfall of cross-agent variability.
We found no difference in acute adverse events reported for the 2 agents in this series. However, an additional potential advantage of gadobenate dimeglumine, not studied here, is its apparently favorable risk profile regarding nephrogenic systemic fibrosis (NSF), compared with other gadolinium contrast agents. Reports on NSF have not always identified the contrast agent used, but when a specific agent was identified, the most common agent was gadodiamide, followed by gadopentetate dimeglumine and gadoversetamide.30 The improved enhancement documented here with gadobenate dimeglumine may facilitate the use of lower doses in patients at risk for NSF. Because the risk of NSF appears to be related to cumulative gadolinium dose and may vary by contrast agent, the possibility of using gadobenate dimeglumine at a lower dose may be of interest to investigating radiologists.30–32
A potential limitation of the study and a bias against gadobenate dimeglumine are that standard imaging sequences developed for use with conventional gadolinium agents were used rather than sequences optimized for use with higher relaxivity contrast agents. It is possible that sequences tailored for use with gadobenate dimeglumine would improve diagnostic performance still further. In this regard, a slightly increased flip angle and/or a slightly shortened TR when using gadobenate dimeglumine leads to increased SNR and CNR and thus improved lesion conspicuity.32,33 Further work is warranted to compare gadobenate dimeglumine with gadodiamide and other conventional agents by using individually optimized sequence parameters and to determine if this advantage holds at a 3T field strength. Preliminary work in an animal model suggests that this is the case.34
A second limitation of this study is that the clinical impact of gadobenate dimeglumine on patient management and outcome was not directly evaluated. Although the greater diagnostic information available with gadobenate dimeglumine would be expected to benefit therapeutic procedures by better defining resection margins and radiosurgical target volumes, a dedicated study would be required to prove this point conclusively.9
Conclusion
In conclusion, 3 blinded neuroradiologists in this study unanimously preferred gadobenate dimeglumine over gadodiamide for MR imaging of brain lesions when equivalent 0.1-mmol/kg doses of these agents were compared under identical imaging conditions. Significant (P < .0001) preference was noted by each reader for both qualitative and quantitative lesion-enhancement end points.
Footnotes
This work was supported by Bracco Diagnostics (Princeton, NJ) through a multicenter clinical trial grant.
References
- 1.Albert FK, Forsting M, Sartor K, et al. Early postoperative magnetic resonance imaging after resection of malignant glioma: objective evaluation of residual tumor and its influence on regrowth and prognosis. Neurosurgery 1994;34:45–60 [DOI] [PubMed] [Google Scholar]
- 2.Kondziolka D, Patel A, Lunsford LD, et al. Stereotactic radiosurgery plus whole brain radiotherapy versus radiotherapy alone for patients with multiple brain metastases. Int J Radiat Oncol Biol Phys 1999;45:427–34 [DOI] [PubMed] [Google Scholar]
- 3.Pintaske J, Martirosian P, Graf H, et al. Relaxivity of gadopentetate dimeglumine (Magnevist), gadobutrol (Gadovist), and gadobenate dimeglumine (MultiHance) in human blood plasma at 0.2, 1.5, and 3 Tesla. Invest Radiol 2006;41:213–21. Erratum in Invest Radiol 2006;41:859 [DOI] [PubMed] [Google Scholar]
- 4.Rohrer M, Bauer H, Mintorovitch J, et al. Comparison of magnetic properties of MRI contrast media solutions at different magnetic field strengths. Invest Radiol 2005;40:715–24 [DOI] [PubMed] [Google Scholar]
- 5.Cavagna FM, Maggioni F, Castelli PM, et al. Gadolinium chelates with weak binding to serum proteins: a new class of high-efficiency, general purpose contrast agents for magnetic resonance imaging. Invest Radiol 1997;32:780–96 [DOI] [PubMed] [Google Scholar]
- 6.Giesel FL, von Tengg-Kobligk H, Wilkinson ID, et al. Influence of human serum albumin on longitudinal and transverse relaxation rates (R1 and R2) of magnetic resonance contrast agents. Invest Radiol 2006;41:222–28 [DOI] [PubMed] [Google Scholar]
- 7.Knopp MV, Runge VM, Essig M, et al. Primary and secondary brain tumors at MR imaging: bicentric intraindividual crossover comparison of gadobenate dimeglumine and gadopentetate dimeglumine. Radiology 2004;230:55–64 [DOI] [PubMed] [Google Scholar]
- 8.Maravilla KR, Maldjian JA, Schmalfuss IM, et al. Contrast enhancement of central nervous system lesions: multicenter intraindividual crossover comparative study of two MR contrast agents. Radiology 2006;240:389–400 [DOI] [PubMed] [Google Scholar]
- 9.Kuhn MJ, Picozzi P, Maldjian JA, et al. Evaluation of intraaxial enhancing brain tumors on magnetic resonance imaging: intraindividual crossover comparison of gadobenate dimeglumine and gadopentetate dimeglumine for visualization and assessment, and implications for surgical intervention. J Neurosurg 2007;106:557–66 [DOI] [PubMed] [Google Scholar]
- 10.Colosimo C, Knopp MV, Barreau X, et al. A comparison of Gd-BOPTA and Gd-DOTA for contrast-enhanced MRI of intracranial tumours. Neuroradiology 2004;46:655–65. Epub 2004 Jun 15 [DOI] [PubMed] [Google Scholar]
- 11.Colosimo C, Ruscalleda J, Korves M, et al. Detection of intracranial metastases: a multi-center, intra-patient comparison of gadobenate dimeglumine-enhanced MRI with routinely used contrast agents at equal dose. Invest Radiol 2001;36:72–81 [DOI] [PubMed] [Google Scholar]
- 12.Essig M, Tartaro A, Tartaglione T, et al. Enhancing lesions of the brain: intra-individual crossover comparison of contrast enhancement after gadobenate dimeglumine versus established gadolinium comparators. Acad Radiol 2006;13:744–51 [DOI] [PubMed] [Google Scholar]
- 13.Valk J, Algra PR, Hazenberg CJ, et al. A double-blind, comparative study of gadodiamide injection and gadopentetate dimeglumine in MRI of the central nervous system. Neuroradiology 1993;35:173–77 [DOI] [PubMed] [Google Scholar]
- 14.Åkeson P, Jonsson E, Haugen I, et al. Contrast-enhanced MRI of the central nervous system: comparison between gadodiamide injection and gadolinium-DTPA. Neuroradiology 1995;37:229–33 [DOI] [PubMed] [Google Scholar]
- 15.Runge VM, Armstrong MR, Barr RG, et al. A clinical comparison of the safety and efficacy of MultiHance (gadobenate dimeglumine) and Omniscan (gadodiamide) in magnetic resonance imaging in patients with central nervous system pathology. Invest Radiol 2001;36:65–71 [DOI] [PubMed] [Google Scholar]
- 16.Runge V, Parker JR, Donovan M, et al. Double-blind, efficacy evaluation of gadobenate dimeglumine, a gadolinium chelate with enhanced relaxivity, in malignant lesions of the brain. Invest Radiol 2002;37:269–80 [DOI] [PubMed] [Google Scholar]
- 17.Yuh WT, Fisher DJ, Engelken JD, et al. MR evaluation of CNS tumors: dose comparison study with gadopentetate dimeglumine and gadoteridol. Radiology 1991;180:485–91 [DOI] [PubMed] [Google Scholar]
- 18.Schneider G, Kirchin MA, Pirovano G, et al. Gadobenate dimeglumine-enhanced magnetic resonance imaging of intracranial metastases: effect of dose on lesion detection and delineation. J Magn Reson Imaging 2001;14:525–39 [DOI] [PubMed] [Google Scholar]
- 19.Knopp MV, Giesel FL, von Tengg-Kobligk H, et al. Contrast-enhanced MR angiography of the run-off vasculature: intraindividual comparison of gadobenate dimeglumine with gadopentetate dimeglumine. J Magn Reson Imaging 2003;17:694–702 [DOI] [PubMed] [Google Scholar]
- 20.Balci NC, Inan N, Anik Y, et al. Low-dose gadobenate dimeglumine versus standard-dose gadopentate dimeglumine for delayed contrast-enhanced cardiac magnetic resonance imaging. Acad Radiol. 2006;13:833–89 [DOI] [PubMed] [Google Scholar]
- 21.Pediconi F, Catalano C, Occhiato R, et al. Breast lesion detection and characterization at contrast-enhanced MR mammography: gadobenate dimeglumine versus gadopentetate dimeglumine. Radiology 2005;237:45–56 [DOI] [PubMed] [Google Scholar]
- 22.Schneider G, Maas R, Schultze Kool L, et al. Low-dose gadobenate dimeglumine versus standard dose gadopentetate dimeglumine for contrast-enhanced magnetic resonance imaging of the liver: an intra-individual crossover comparison. Invest Radiol 2003;38:85–94 [DOI] [PubMed] [Google Scholar]
- 23.Warmuth-Metz M. Postoperative imaging after brain tumor resection. Acta Neurochir Suppl 2003;88:13–20 [DOI] [PubMed] [Google Scholar]
- 24.Ciric I, Vick NA, Mikhael MA, et al. Aggressive surgery for malignant supratentorial gliomas. Clin Neurosurg 1990;36:375–83 [PubMed] [Google Scholar]
- 25.Ekinci G, Akpinar IN, Baltacioglu F, et al. Early-postoperative magnetic resonance imaging in glial tumors: prediction of tumor regrowth and recurrence. Eur J Radiol 2003;45:99–107 [DOI] [PubMed] [Google Scholar]
- 26.Ewend MG, Elbabaa S, Carey LA. Current treatment paradigms for the management of patients with brain metastases. Neurosurgery 2005;57 (suppl 5):S66–77 [DOI] [PubMed] [Google Scholar]
- 27.Kaal EC, Niel CG, Vecht CJ. Therapeutic management of brain metastasis. Lancet Neurol 2005;4:289–98 [DOI] [PubMed] [Google Scholar]
- 28.Nimsky C, Ganslandt O, Fahlbusch R. 1.5 T: intraoperative imaging beyond standard anatomic imaging. Neurosurg Clin N Am 2005;16:185–200 [DOI] [PubMed] [Google Scholar]
- 29.Oh DS, Black PM. A low-field intraoperative MRI system for glioma surgery: is it worthwhile? Neurosurg Clin N Am 2005;16:135–41 [DOI] [PubMed] [Google Scholar]
- 30.Penfield JG, Reilly RF Jr. What nephrologists need to know about gadolinium. Nat Clin Pract Nephrol 2007;3:654–68 [DOI] [PubMed] [Google Scholar]
- 31.Marckmann P, Skov L, Rossen K, et al. Case-control study of gadodiamide-related nephrogenic systemic fibrosis. Nephrol Dial Transplant 2007;22:3174–78 [DOI] [PubMed] [Google Scholar]
- 32.Bleicher AG, Kanal E. A serial dilution study of gadolinium-based MR imaging contrast agents. AJNR Am J Neuroradiol. 2008;29:668–73 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yrjänä SK, Vaara T, Karttunen A, et al. Pulse repetition time and contrast enhancement: simulation study of Gd-BOPTA and conventional contrast agent at different field strengths. Invest Radiol 2008;43:267–75 [DOI] [PubMed] [Google Scholar]
- 34.Runge VM, Biswas J, Wintersperger BJ, et al. The efficacy of gadobenate dimeglumine (Gd-BOPTA) at 3 Tesla in brain magnetic resonance imaging: comparison to 1.5 Tesla and a standard gadolinium chelate using a rat brain tumor model. Invest Radiol. 2006;41:244–48 [DOI] [PubMed] [Google Scholar]