Abstract
SUMMARY: Hemiballism with corresponding striatal T1 hyperintensity on MR imaging has occasionally been reported in patients with nonketotic hyperglycemia. However, the subthalamic nucleus lesion, which is believed to be pathogenetically related to hemiballism, is rarely documented in a living patient with nonketotic hyperglycemia. We describe a patient with nonketotic hyperglycemia-induced hemiballism, whose responsible lesion (ie, the subthalamus) was demonstrated by MR imaging.
Hemiballism-hemichorea (HB-HC) is an involuntary, irregular, wide-amplitude, and poorly patterned movement disorder. HB-HC can mostly result from a focal vascular lesion in the contralateral basal ganglia.1 In the absence of an identifiable focal vascular lesion, metabolic derangements (eg, nonketotic hyperglycemia or hyperthyroidism), brain neoplasm, and infectious diseases of the central nervous system (eg, human immunodeficiency virus infection) are considered to be the more common causes.1 Recent studies have shown that striatal hyperintensity, seen on the T1-weighted MR imaging of the brain, can occur in the acute stage of HB-HC, caused by nonketotic hyperglycemia.2 These radiologic images provided the clue for the early diagnosis of this dyskinesia. Animal studies and subsequent human studies supported the finding that the subthalamus is strongly related to hemiballism.3 There are only scanty reports documenting the subthalamic involvement of nonketotic hyperglycemia in a living patient, which prompted us to report a case of nonketotic hyperglycemia-induced hemiballism, with the responsible subthalamic lesion confirmed by MR imaging.
Case Report
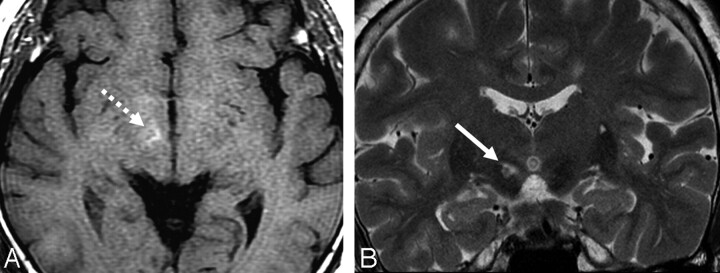
A 53-year-old woman was admitted for sudden onset of abnormal movement on the left side. This jerky irregular involuntary movement was first noted on the left leg 3 days before admission. Her symptoms worsened during the last 3 days. She had no history of hypertension, diabetes, headache, parkinsonism, or other neurologic diseases. At admission, her initial blood glucose level was 242 mg/dL and her hemoglobin A1c concentration was 11.7% (normal values are 70–110 mg/dL and 4.7–6.4%, respectively). The urine specimen was negative for ketones, indicating nonketotic hyperglycemia. A neurologic examination showed her sensorium to be normal. Deep tendon reflexes were symmetrically hypoactive. The remaining results of the examinations were unremarkable, except for left hemiballism. Brain CT, performed on the first hospital day, revealed no abnormalities. The MR image, obtained 4 days after her initial presentation, showed high signal intensities in the right subthalamic nucleus (STN) on T1-weighted and T2-weighted images (Fig 1) and subtle high signal intensities on diffusion-weighted images (DWIs, not shown). Gradient-echo images and B0 images of DWI showed no susceptibility artifact in the right STN. Contrast-enhanced MR images showed no additional findings. The caudate and putamen appeared normal in all sequences of MR images. Hemiballism was improved with clonazepam (6 mg/day) after 14 days. The serum glucose level was normalized with insulin administration.
Fig 1.
The T1-weighted axial image (A) shows increased signal intensity (broken arrow), whereas the T2-weighted coronal image clearly demonstrates a high-signal-intensity lesion (arrow) in the right STN.
Discussion
Hyperglycemia-associated chorea and ballismus have been reported predominantly in older patients with diabetes and nonketotic hyperglycemia.4 The T1-weighted MR images have shown unilateral hyperintensity in the putamen and caudate, but the findings on the T2-weighted MR images have varied.5 Although a significant association between hyperglycemia and hyperintensity on T1-weighted MR images has been reported, the mechanism underlying the imaging findings has been unknown. Protein dissociation in wallerian degeneration secondary to acute putaminal dysfunction has been proposed as one of the plausible mechanisms behind the imaging findings.6 Increased viscosity, the presence of myelin-breakdown products, the presence of blood products, or deposition of calcium and other minerals have been suggested as possible explanations as well.7–9
As to the MR signal intensities alone, our imaging findings confirmed results from previous reports. In this case, however, MR imaging revealed the sole involvement of the contralateral STN without a putaminal lesion. Although subthalamic involvement has never been reported in hyperglycemia-induced hemiballism, acute hemiballism was reported in a small cavernous angioma or infarction confined to the STN.10,11
The STN serves as the main excitatory input to the medial part of the globus pallidus, which inhibits, by using gamma amino butyric acid, the activity of the ventromedial thalamus. The destruction of the STN causes a decrease of this inhibitory function and increases thalamic excitation of the motor and premotor cortex, resulting in involuntary movement.12 This mechanism is supported by findings during deep brain stimulation of the subthalamus in Parkinson disease.13
Possible differential diagnosis in this case includes acute infarction by vascular insult. Unlike T1 hyperintensity due to nonketotic hyperglycemia, acute infarction of 3 days duration shows bright signal intensity on DWIs and low signal intensity on T1-weighted images. When T1 hyperintensity is noted in acute infarction at 3 days, it should be hemorrhagic transformation and should, therefore, show dark signal intensity on T2*-weighted images.14 Acute hemorrhage is also unlikely because there is no evidence of magnetic susceptibility artifacts on T2*-weighted images as well as on DWIs with a b-value of 0 s/mm2.
In conclusion, unilateral subthalamic involvement can be the sole imaging finding in nonketotic hyperglycemia-induced hemiballism. This case suggests a pathophysiologic explanation for hemiballism with its strategic localization.
Footnotes
This work was supported by the Second-Phase of BK (Brain Korea) 21 Project in 2007.
References
- 1.Dewey B, Jankovic J. Hemiballism-hemichorea: clinical and pharmacologic findings in 21 patients. Arch Neurol 1989;46:862–67 [DOI] [PubMed] [Google Scholar]
- 2.Yahikozawa H, Hanyu N, Yamamoto K, et al. Hemiballism with striatal hyperintensity on T1- weighted MRI in diabetes patients; a unique syndrome. J Neurol Sci 1994;124:208–14 [DOI] [PubMed] [Google Scholar]
- 3.Postuma RB, Lang AE. Hemiballism: revisiting a classic disorder. Lancet Neurol 2003;2:661–68 [DOI] [PubMed] [Google Scholar]
- 4.Ifergane G, Masalha R, Herishanu YO. Transient hemichorea/hemiballismus associated with new onset hyperglycemia. Can J Neurol Sci 2001;28:365–68 [DOI] [PubMed] [Google Scholar]
- 5.Lee EJ, Choi JY, Lee SH, et al. Hemichorea-hemiballism in primary diabetic patients: MR correlation. J Comput Assist Tomogr 2002;26:905–11 [DOI] [PubMed] [Google Scholar]
- 6.Wintermark M, Fischbein NJ, Mukherjee P, et al. Unilateral putaminal CT, MR, and diffusion abnormalities secondary to nonketotic hyperglycemia in the setting of acute neurologic symptoms mimicking stroke. AJNR Am J Neuroradiol 2004;25:975–76 [PMC free article] [PubMed] [Google Scholar]
- 7.Chu K, Kang DW, Kim ED, et al. Diffusion-weighted and gradient echo magnetic resonance findings of hemichorea-hemiballismus associated with diabetic hyperglycemia. Arch Neurol 2002;59:448–52 [DOI] [PubMed] [Google Scholar]
- 8.Lin JJ, Chang MK. Hemiballism-hemichorea and nonketotic hyperglycemia. J Neurol Neurosurg Psychiatry 1994;57:748–50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.McCall AL. The impact of diabetes on the CNS. Diabetes 1992;41:557–70 [DOI] [PubMed] [Google Scholar]
- 10.Absher JR, Vogt BA, Clark DG, et al. Hypersexuality and hemiballism due to subthalamic infarction. Neuropsychiatry Neuropsychol Behav Neurol 2000;13:220–29 [PubMed] [Google Scholar]
- 11.Linn JJ, Seelos KC, Botzel K. Hemiballism caused by a small cavernoma in the subthalamic nucleus. Mov Disord 2006;21:2266–67 [DOI] [PubMed] [Google Scholar]
- 12.Guisado R, Arieff A. Neurologic manifestations of diabetic comas: correlation with biochemical alterations in the brain. Metabolism 1975;24:665–79 [DOI] [PubMed] [Google Scholar]
- 13.Doshi P, Bhatt M. Hemiballism during subthalamic nucleus lesioning. Mov Disord 2002;17:848–49 [DOI] [PubMed] [Google Scholar]
- 14.Chang MH, Chiang HT, Lai PH, et al. Putaminal petechial haemorrhage as the cause of chorea: a neuroimaging study. J Neurol Neurosurg Psychiatry 1997;63:300–03 [DOI] [PMC free article] [PubMed] [Google Scholar]