Abstract
Streams of action potentials or long depolarizations evoke a massive exocytosis of transmitters and peptides from the surface of dendrites, axons and cell bodies of different neuron types. Such mode of exocytosis is known as extrasynaptic for occurring without utilization of synaptic structures. Most transmitters and all peptides can be released extrasynaptically. Neurons may discharge their contents with relative independence from the axon, soma and dendrites. Extrasynaptic exocytosis takes fractions of a second in varicosities or minutes in the soma or dendrites, but its effects last from seconds to hours. Unlike synaptic exocytosis, which is well localized, extrasynaptic exocytosis is diffuse and affects neuronal circuits, glia and blood vessels. Molecules that are liberated may reach extrasynaptic receptors microns away. The coupling between excitation and exocytosis follows a multistep mechanism, different from that at synapses, but similar to that for the release of hormones. The steps from excitation to exocytosis have been studied step by step for the vital transmitter serotonin in leech Retzius neurons. The events leading to serotonin exocytosis occur similarly for the release of other transmitters and peptides in central and peripheral neurons. Extrasynaptic exocytosis occurs commonly onto glial cells, which react by releasing the same or other transmitters. In the last section, we discuss how illumination of the retina evokes extrasynaptic release of dopamine and ATP. Dopamine contributes to light-adaptation; ATP activates glia, which mediates an increase in blood flow and oxygenation. A proper understanding of the workings of the nervous system requires the understanding of extrasynaptic communication.
Keywords: serotonin, extrasynaptic release, modulation, nerve cell communication, somatic exocytosis, glia
Introduction
Our view of the workings of the nervous system have been dominated by four threads of fundamental evidence: First, Cajal defined nerve circuits as networks of neurons connected in stereotyped manner, forming transmission lines for specific information processing. Second, physiologists such as Helmholtz, Hodgkin and Huxley showed that nerve impulses spread along axons at ~300 km/h. Third, Sherrington, Katz, Kuffler and Eccles demonstrated that synapses transmit information in ~0.5 ms. Fourth, plasticity adapts synaptic transmission to variations in the ongoing pattern of electrical activity. Such conceptual framework explains how a table tennis player detects the trajectory and velocity of a ball approaching at 50 Km/h and in ~0.3 ms and coordinates his whole-body motion to send it back to an opposite corner of the table. In the games, such cycles may occur twice per second!
This review article deals with a parallel form of communication: streams of electrical impulses or long depolarizations promote massive liberation of signaling molecules from certain neurons. Release occurs without use of synaptic structures, therefore, it is named extrasynaptic. Molecules that are released extrasynaptically from the soma, dendrites and axon modulate the responses of entire neuronal circuits from seconds to days (Trueta and De-Miguel, 2012). Such form of communication may explain why a table tennis player is defeated after being left by his fiancée. His reduced concentration, motivation and attention make him react poorly during the game. A hypothesis gaining increasing support is that ranges of physiological concentrations of extracellular signaling molecules modulate the responses of whole neuronal circuits; concentrations below or above produce pathologies (Calabresi et al., 2015; Del-Bel and De-Miguel, 2018; Pál, 2018; Quentin et al., 2018).
Our focus here is exclusively on molecules that are released by exocytosis. Other substances such as nitric oxide or cannabinoids are released by diffusion across the plasma membrane (Del-Bel and De-Miguel, 2018); nucleic acids and proteins are released encapsulated inside vesicles that flow extracellularly (Colombo et al., 2014; Mendolesi, 2018). Those forms of release also follow increases in electrical activity.
A good example as to how extrasynaptic exocytosis exerts its effects comes from studies of aggression in lobsters by Kravitz and his colleagues (Kravitz, 1988; Huber et al., 1997). An encounter between two lobsters triggers aggression. Lobsters approximate to each other displaying their powerful claws and urinating on each other. The episodic encounters, initially lasting seconds, decrease their strength and duration as one lobster becomes dominant and the other submissive. The aggressive posture is evoked by a systemic injection of serotonin; an injection of octopamine reproduces the submissive posture (Livingstone et al., 1980).
The serotonergic A1 neurons in the abdominal ganglia of lobsters innervate the ganglia and project branches to the circulation. Command neurons that evoke tail flipping during aggression evoke serotonin release from the A1 neurons. Serotonin that is released in the ganglia lowers the firing threshold of central neurons; the serotonin discharged to the circulation increases motoneuron transmission and strengthens muscle contractions and heart beat (Glusman and Kravitz, 1982; Hörner et al., 1997; Hernández-Falcón et al., 2005). In experiments in which serotonergic neurons were depleted of serotonin by systemic injection of 5–7 dihydroxytryptamine, aggression still occurred. However, the strength and duration of the encounters lacked modulation. Therefore, serotonin “sets the gain” of the circuitry for aggression by acting all along the neuronal circuit.
Cellular Basis of Extrasynaptic Exocytosis
Extrasynaptic exocytosis has been studied in central and peripheral neurons. The similitude among the mechanism that links excitation with exocytosis suggests a widely conserved mechanism, similar to that in gland cells but remarkably different from that for synaptic release (Sun and Poo, 1987; Huang et al., 2012; Hirasawa et al., 2015; Ludwig and Stern, 2015; Hökfelt et al., 2018; Quentin et al., 2018). Most transmitters and all peptides have been shown to be released extrasynaptically (Trueta and De-Miguel, 2012), and neurons may release more than one type of substance (Burnstock, 2012; Nusbaum et al., 2017; Hökfelt et al., 2018).
Extrasynaptic Exocytosis From Different Neuronal Compartments
An evolutionary feature shared by neurons that release extrasynaptically is that small numbers innervate the nervous system extensively, and produce a wide variety of effects. For example, ~235,000 serotonergic neurons in humans (Baker et al., 1990) project from the raphe nuclei to the entire central nervous system. Neurons releasing catecholamines, acetylcholine or peptides exist in similar small numbers (Zetler, 1970; Mouton et al., 1994; Nair-Roberts et al., 2008; Li et al., 2018). Such extensive innervation is complemented by the neuronal capability to release differentially from the soma, dendrites and axon. A well-known example is the release of the peptides vasopressin or oxytocin from magnocellular hypothalamic neurons (Ludwig and Stern, 2015).
The axons of magnocellular neurons bear rosaries of varicosities that release extrasynaptically on the spread of action potentials; their terminals discharge peptide onto the blood flow (Acher and Chauvet, 1954; Du Vigneaud, 1954). During lactation, suckling evokes oxytocin axonal release but dendritic release is delayed. However, dendritic release is locally evoked on activation of extrasynaptic NMDA receptors (de Kock et al., 2004; Tobin et al., 2012), as it also happens in dendrites of raphe neurons (Colgan et al., 2012).
Discovery of Extrasynaptic Communication
Serotonin that had been released extrasynaptically was discovered by Dalstrom and Fuxe in the 60s using the Falck-Hillarp technique, by which exposure to formaldehyde vapors transforms the monoamines serotonin, dopamine or adrenaline into fluorescent derivatives (Fuxe et al., 2007; Borroto-Escuela et al., 2015). Brain sections of raphe nuclei contained serotonin-derived fluorescence surrounding the fluorescent cell bodies, distantly from the axonal release sites. Similar observations made in dopaminergic neurons, plus the fact that peptides can be released far away from their receptors led to the concept of volume transmission by Fuxe and his colleagues, meaning that molecules act on receptors located distantly from the release sites (Borroto-Escuela et al., 2015). It was later shown that axons of neurons releasing monoamines, acetylcholine, ATP and peptides bear vesicle arrangements but scarce presynaptic active zones. Therefore, most exocytosis occurs extrasynaptically (Hökfelt, 1968; Umbriaco et al., 1995; Contant et al., 1996; Descarries et al., 1996; Descarries and Mechawar, 2000; Burnstock, 2012).
Somatic Release of Serotonin
The vast diversity and distribution of serotonin functions, the small numbers of serotonergic neurons and synapses, and the extraordinary chemical properties of serotonin explain why extrasynaptic serotonergic communication has been widely studied. Serotonin that is released from the cell body and dendrites of raphe neurons has been detected distantly by voltammetry, based on its redox properties (Bunin and Wightman, 1998). Moreover, serotonin exocytosis has been detected by amperometric electrodes apposed onto the soma of leech Retzius neurons (Bruns et al., 2000), or in the soma and dendrites of raphe neurons by multiphoton excitation (Kaushalya et al., 2008; Colgan et al., 2012; Sarkar et al., 2012; Maity and Maiti, 2018).
The mechanism for somatic exocytosis of serotonin has been studied step by step in Retzius neurons (De-Miguel et al., 2012). Their large (60–80 μm) soma contains “astronomic” numbers of dense-core vesicles loaded with serotonin (Coggeshall, 1972). Most vesicles rest distantly from the plasma membrane. However, electron microscopy and fluorescence of FM dyes, which stain the intravesicular membrane during exo-endocytosis (Hoopmann et al., 2012), indicate that vesicles move massively to the plasma membrane following trains of action potentials but not individual impulses (Trueta et al., 2003). The formation of fluorescent spots beneath the soma surface indicates that fusion of dense-core vesicles occurs in preferential sites. The development of FM fluorescent spots reflects the kinetics of release by clusters of vesicles; the number of fluorescent spots is a measure of the amount of release. Such experiments gave unexpected results: First, exocytosis starts seconds after the end of stimulation. Second, exocytosis lasts hundreds of seconds (Trueta et al., 2003), as hormone release from gland cells does (Thorn et al., 2016). Similar results have been obtained from cholinergic, dopaminergic, noradrenergic and peptidergic neurons (Sun and Poo, 1987; Huang and Neher, 1996; Jaffe et al., 1998; Puopolo et al., 2001; Bao et al., 2003; Kaushalya et al., 2008; Huang et al., 2012; Ludwig and Stern, 2015).
In Retzius neurons and magnocellular neurons it is the frequency of the action potentials, not their number, what determines the amount of release. The maximum release occurs at 20 impulses per second (Dreifuss et al., 1971; Leon-Pinzon et al., 2014), but may be enhanced by alternate periods of stimulation and rest (Dutton and Dyball, 1979).
Vesicle Transport
The delay between stimulation and exocytosis reflects the vesicle transport to the plasma membrane (De-Miguel et al., 2012). The large calcium transient that develops upon the stimulation train is essential for the vesicle transport. In electron micrographs, vesicles remain at rest upon stimulation with extracellular magnesium substituting for calcium to block calcium entry. Moreover, experimental perturbations of the tubulin-kinesin or actin-myosin transport systems prevent somatic exocytosis from Retzius and magnocellular neurons (Tobin and Ludwig, 2007; De-Miguel et al., 2012; Noguez et al., 2019).
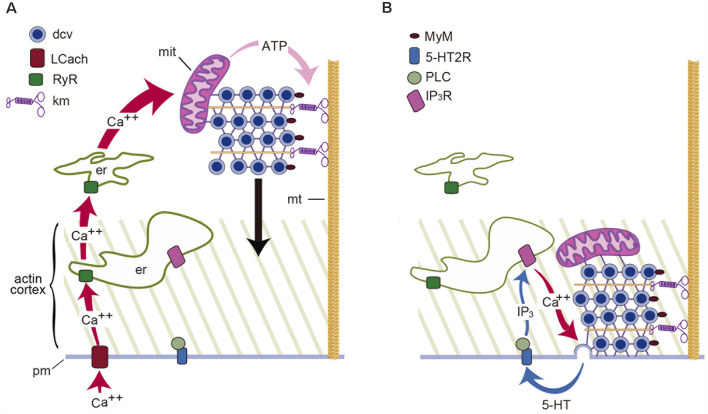
Stimulation and the activation of the transport are linked by a chain of events: first, stimulation promotes calcium entry through L-type channels (Trueta et al., 2003), which are advantageous, for their slow inactivation sustains calcium entry along trains of impulses or long depolarizations. Second, increasing the stimulation frequency increases the amplitude of the intracellular calcium transient (Leon-Pinzon et al., 2014). Third, calcium activates ryanodine receptors, inducing calcium-dependent calcium release (Trueta et al., 2004; Leon-Pinzon et al., 2014), as in substantia nigra of dopaminergic and magnocellular neurons (Ludwig et al., 2002; Patel et al., 2009). Fourth, a calcium tsunami floods the cell body and invades the mitochondria, which respond by producing ATP. Fifth, ATP sets in motion the kinesin- and myosin-dependent vesicle transport (Figure 1; De-Miguel et al., 2012).
Figure 1.
Schematic representation of the mechanism for somatic exocytosis of serotonin in Retzius neurons. (A) Electrical activity sets in motion the transport of dense core vesicles (dcv) to the plasma membrane. In response to a train of action potentials, L-type calcium channels (LCach) open. Calcium entry activates ryanodine receptors (RyR) in the endoplasmic reticulum (er) and produces calcium-induced calcium release. The amplified calcium wave invades the soma; in the mitochondria (mit), calcium stimulates the synthesis of ATP, which activates kinesin motors (km) and vesicle transport along microtubules (mt). (B) Vesicles enter the actin cortex and myosin motors (MyM) carried by the vesicles couple to actin filaments and contribute to the transport. Release is maintained by a positive feedback loop in which the serotonin that is released activates 5-HT2 receptors (5-HT2R). Activation of phospholipase C (PLC) produces IP3 which acts on its receptors (IP3R) to activate calcium release. Such calcium maintains exocytosis going on until the last vesicles fuse (Adapted from Leon-Pinzon et al., 2014).
Energy Cost of the Vesicle Transport
Application of thermodynamic theory to the kinetics of exocytosis predicts that three variables determine the latency from stimulation to the onset of exocytosis (De-Miguel et al., 2012 ): the traveling distance to the plasma membrane (0.2–6.0 μm), the velocity of the transport (15–90 nm/s) and the number of vesicles carried per cluster (90 to >1,000). Upon arrival at the plasma membrane, vesicles fuse at a 0.5–4.0 s−1 rate, which reflects the transport velocity. For example, exocytosis from 1,000 vesicles at a 4 s−1 rate lasts 250 s. The energy expenses of the transport, calculated from the work of the motors, range from 10–200 ATP molecules per vesicle fused, depending on the same variables (De-Miguel et al., 2012).
How thermodynamically-efficient is the use of ATP during the vesicle transport? An answer has been obtained also from the application of thermodynamic theory (Noguez et al., 2019). Surprisingly, the largest thermodynamic efficiency value is 6.2%, which is lower than the 20% efficiency of a contemporary car running on a highway. The remaining energy is dissipated as heat along the path, owing to friction forces. The origin of such friction was predicted from the distribution of efficiency values along the traveling pathway. The lowest values correlate with the penetration of vesicles to the actin cortex and their passage between endoplasmic reticulum layers. Both essential contributors to the transport increase the energy cost by being frictive obstacles. Such a phenomenon adds energy cost to the modulation of neuronal circuits.
Calcium and Exocytosis
Measurements of the intracellular calcium dynamics with fluorescent dyes unveiled that by the time vesicles arrive at the plasma membrane, the intracellular calcium concentration has returned to resting levels except in the soma shell (Leon-Pinzon et al., 2014). Such peripheral calcium elevation drives the fusion of vesicles as they arrive at the plasma membrane. Voltage clamp measurements failed to detect any transmembrane calcium flow following the train of impulses. Instead, the peripheral calcium transient was reproduced by iontophoretic serotonin application to activate membrane receptors. Conclusive evidence that the peripheral calcium transient depends on serotonin that had been released came from experiments in which both the peripheral calcium transient and somatic exocytosis were prevented by blocking serotonergic 5HT2 receptors with methysergide, or by blocking the activation of phospholipase C (PLC) with U-73122 before stimulation of somatic exocytosis (Leon-Pinzon et al., 2014). Therefore, the calcium that sustains exocytosis is released by peripheral endoplasmic reticulum upon a serotonin-mediated IP3 production and activation of IP3 receptors. Similar observations made in peptidergic dorsal root ganglion and magnocellular neurons (Bao et al., 2003; Ludwig and Stern, 2015) point to another general principle: the long-lasting exocytosis is sustained by a feedback loop. Transmitter that is liberated activates autoreceptors coupled to phospholipase C; IP3 production evokes intracellular calcium release; calcium promotes exocytosis; the released substance maintains the cycle. Termination of the loop follows the fusion of the last vesicles in the clusters (Figure 1).
Molecules Catalyzing Exocytosis
Synapses contain a calcium-sensitive molecular complex that drives vesicle fusion upon local calcium elevations (Südhof, 2013). In the soma and dendrites of dopaminergic neurons, antibodies recognize isoforms of the fusion complex components VAMP-2, SNAP25, and syntaxin, which are unusual at synapses (Witkovsky et al., 2009). Moreover, the synaptic calcium sensors synaptotagmins 1 and 2 are substituted by the more appropriate high affinity synaptotagmins 4 and 7 (Mendez et al., 2011), since the fusion of dense-core vesicles depends on distant calcium sources.
Dense-Core Vesicle Recycling
The vesicle recycling has been studied by adding the marker peroxidase to the extracellular fluid before stimulation (Trueta et al., 2012). Sections for electron microscopy incubated with anti-peroxidase antibody coupled to gold particles, showed peroxidase inside dense-core vesicles, newly-formed small (~40 nm) clear vesicles, and inside multivesicular bodies containing both vesicle types (Figure 1). Multivesicular bodies are transported retrogradely and their content is recycled in the perikaryon to form new vesicles.
Synaptic vs. Extrasynaptic Exocytosis
Studies in Retzius neurons permit a comparison of the amounts of transmitter liberated from synaptic and extrasynaptic vesicle pools. The formation of specific synapses between identified leech neurons in culture allowed John Nicholls and his colleagues (Nicholls and Kuffler, 1990) to examine the fine mechanisms of transmission. At synapses, the fusion of clear vesicles is calcium-dependent. Amperometric records show that a vesicle liberates ~4,700 serotonin molecules (Bruns and Jahn, 1995). Synapses display short-term plasticity (Stewart et al., 1989); for example, 10 impulses at 20-Hz evoke rapid facilitation followed by depression, along which ~60 quanta get released.
A common form of extrasynaptic exocytosis of transmitters and peptides occurs from dense-core vesicles surrounding synaptic active zones (Hökfelt et al., 2018). The differences between synaptic and perisynaptic exocytosis are schematized in Figure 2. The presynaptic boutons of cultured Retzius neurons contain clear synaptic vesicles and dense-core perisynaptic vesicles all filled with serotonin (Kuffler et al., 1987; Bruns and Jahn, 1995). Perisynaptic release increases along stimulation trains and produces large quantal amperometric spikes upon release of ~90,000 serotonin molecules (Bruns and Jahn, 1995). Amazingly, three dense-core vesicles release about the same number of serotonin molecules as the 60 synaptic vesicles that fuse along a 20-Hz train.
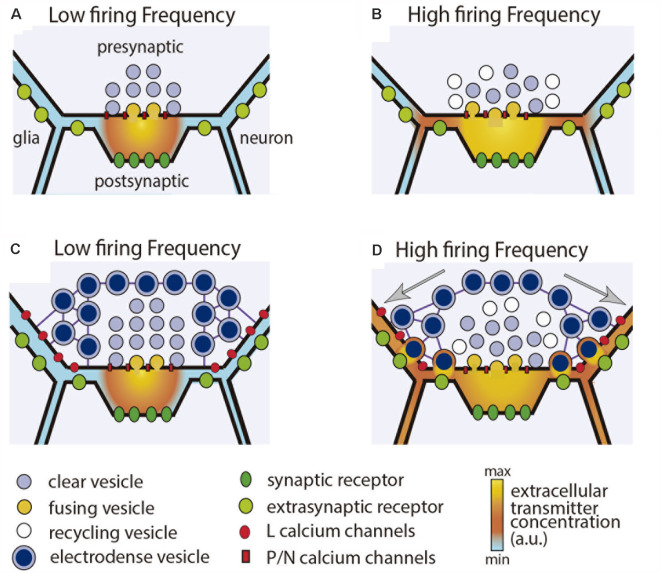
Figure 2.
Synaptic and perisynaptic exocytosis. (A) Low frequency stimulation activates presynaptic P or N type calcium channels and evokes fusion of synaptic vesicles in the active zone. Transmitter liberated into the synaptic cleft activates post-synaptic receptors. (B) Increasing the stimulation frequency increases synaptic exocytosis and produces transmitter spillover, which acts on extrasynaptic receptors in adjacent glia and neuronal processes. (C) In the presence of perisynaptic dense-core vesicles, a low stimulation frequency evokes mostly synaptic release. (D) Increasing the stimulation frequency produces summation of calcium currents flowing through L type channels. Dense core vesicles are transported to the plasma membrane and fuse in presynaptic regions of the terminal. Transmitter that has been released acts on extrasynaptic receptors in the pre-and post-synaptic terminals, and also in adjacent glial and neuronal processes. Clear and dense core vesicles may have the same or different transmitters. Dense core vesicles also may contain peptides.
The difference between synaptic and somatic exocytosis is more drastic. Electron microscopy and FM dye staining of vesicles indicate that a 20-Hz train evokes fusion of ~40,000 vesicles from ~80 release sites, each vesicle cluster carrying on an average 500 vesicles (Del-Bel and De-Miguel, 2018). By assuming that ~90,000 serotonin molecules integrate a quantum, a 10-impulse train at 20-Hz would trigger release of ~3.6 billion molecules. Moreover, the long thick axon discharges serotonin from clear and dense-core vesicles in undetermined amounts. It is predictable that the huge amount of serotonin being released from a pair of Retzius cells in each ganglion suffices to modulate behavior (Willard, 1981).
Transmitter Spillover
Glutamate and GABA, the conventional transmitters at synapses, act extrasynaptically upon spillover from the synaptic cleft when synaptic release increases (Isaacson et al., 1993; Rusakov and Kullmann, 1998; DiGregorio et al., 2002). Spillover-mediated transmission occurs through activation of low-affinity extrasynaptic receptors in neighboring cells (Pál, 2018). In addition, astroglia and microglia sense and release glutamate (Pál, 2018).
Glia as Mediator of Extrasynaptic Communication
Glia are common counterparts for extrasynaptic exocytosis. Glial cell membranes respond to transmitters and transport many of them (Marcaggi and Attwell, 2004; Káradóttir et al., 2005; Verkhratsky et al., 2009). In response to transmitters such as glutamate, serotonin and ATP, networks of electrically-coupled astrocytes propagate calcium transients (Munsch and Deitmer, 1992; Metea and Newman, 2006; Verkhratsky et al., 2009). In return, glia releases the same or other transmitters, peptides and proteins (Billups and Attwell, 1996; Henneberger et al., 2010; Igelhorst et al., 2015). Observations like these have led to the hypothesis of tripartite synapses, in which glia reacts to transmitters that spillover and in return modulate synaptic activity (Perea et al., 2009; Corkrum et al., 2020).
Extrasynaptic Integration of Retinal Responses to Light
The retina provides a clear example of the integrative roles of extrasynaptic communication at the cellular level. A light spot shone onto a dark-adapted retina, evokes visual processing and extrasynaptic release of transmitters from amacrine cells (Hirasawa et al., 2015; Newman, 2015). Dopamine contributes to light adaptation by uncoupling electrical synapses and by acting directly on neurons at every level of the visual processing (Piccolino et al., 1984; Witkovsky, 2004; Zhang et al., 2011). Solid evidence about glia as mediator between extrasynaptic exocytosis and the regulation of blood flow has been contributed by Newman and his colleagues (Newman, 2015). ATP released from Amacrine cells activates Muller cells, the main type of retinal glia. In response, Muller cells synthesize and release factors that increase blood flow and oxygenation of the illuminated area. By extrapolation, the magnetic resonance images may be a product of extrasynaptic communication.
Conclusions
-
1.
Extrasynaptic exocytosis is common in the nervous system. It may occur differentially from the soma, dendrites and axon, allowing neurons to produce multiple effects.
-
2.
Synaptic and extrasynaptic exocytosis coexist in the same neurons.
-
3.
Neurotransmitters and peptides are released extrasynaptically.
-
4.
Synaptic transmission is punctual; extrasynaptic transmission is diffuse. Substances released extrasynaptically act via volume transmission at variable distances and with different time courses.
-
5.
Extrasynaptic communication integrates the activity of neurons, glia and blood vessels.
-
6.
Other forms of extrasynaptic neurotransmission occur upon diffusive release of molecules, such as gases and cannabinoids; vesicles are released loaded with cocktails of molecules.
-
7.
Understanding the functioning of the nervous system requires understanding of its modulation by extrasynaptic communication.
Author Contributions
FD-M wrote the first version of the manuscript. All authors contributed to the article and approved the submitted version.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Footnotes
Funding. This work was supported by Human Frontier Science Program (HFSP; Ref RGP0060/2019); Consejo Nacional de Ciencia y Tecnología (CONACyT; grant 130031); and Dirección General de Asuntos del Personal Académico (DGAPA; grant IN210317) from the Universidad Nacional Autónoma de México to FD-M.
References
- Acher R., Chauvet J. (1954). The structure of bovine vasopressin. Biochim. Biophys. Acta. 14, 421–429. 10.1016/0006-3002(54)90202-4 [DOI] [PubMed] [Google Scholar]
- Baker K. G., Halliday G. M., Törk I. (1990). Cytoarchitecture of the human dorsal raphe nucleus. J. Comp. Neurol. 8, 147–161. 10.1002/cne.903010202 [DOI] [PubMed] [Google Scholar]
- Bao L., Jin S. X., Zhang C., Wang L. H., Xu Z. Z., Zhang F. X., et al. (2003). Activation of delta opioid receptors induces receptor insertion and neuropeptide secretion. Neuron 37, 121–133. 10.1016/s0896-6273(02)01103-0 [DOI] [PubMed] [Google Scholar]
- Billups B., Attwell D. (1996). Modulation of non-vesicular glutamate release by pH. Nature 379, 171–174. 10.1038/379171a0 [DOI] [PubMed] [Google Scholar]
- Borroto-Escuela D. O., Agnati L. F., Bechter K., Jansson A., Tarakanov A. O., Fuxe K. (2015). The role of transmitter diffusion and flow versus extracellular vesicles in volume transmission in the brain neural-glial networks. Philos. Trans. R. Soc. Lond. B Biol. Sci. 370:20140183. 10.1098/rstb.2014.0183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruns D., Jahn R. (1995). Real-time measurement of transmitter release from single synaptic vesicles. Nature 377, 62–65. 10.1038/377062a0 [DOI] [PubMed] [Google Scholar]
- Bruns D., Riedel D., Klingauf J., Jahn R. (2000). Quantal release of serotonin. Neuron 28, 205–220. 10.1016/s0896-6273(00)00097-0 [DOI] [PubMed] [Google Scholar]
- Bunin M. A., Wightman R. M. (1998). Quantitative evaluation of 5-hydroxytryptamine (serotonin). neuronal release and uptake: an investigation of extrasynaptic transmission. J. Neurosci. 18, 4854–4860. 10.1523/JNEUROSCI.18-13-04854.1998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnstock G. (2012). Purinergic signalling: its unpopular beginning, its acceptance and its exciting future. Bioessays 34, 218–225. 10.1002/bies.201100130 [DOI] [PubMed] [Google Scholar]
- Calabresi P., Ghiglieri V., Mazzocchetti P., Corbelli I., Picconi B. (2015). Levodopa-induced plasticity: a double-edged sword in Parkinson’s disease. Philos. Trans R Soc. Lond B Biol. Sci. 370:20140184. 10.1098/rstb.2014.0184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coggeshall R. E. (1972). Autoradiographic and chemical localization of 5-hydroxytryptamine in identified neurons in the leech. Anat. Rec. 172, 489–498. 10.1002/ar.1091720303 [DOI] [PubMed] [Google Scholar]
- Colgan L. A., Cavolo S. L., Commons K. G., Levitan E. S. (2012). Action potential-independent and pharmacologically unique vesicular serotonin release from dendrites. J. Neurosci. 32, 15737–15746. 10.1523/JNEUROSCI.0020-12.2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colombo M., Raposo G., Théry C. (2014). Biogenesis, secretion and intercellular interactions of exosomes and other extracellular vesicles. Annu. Rev. Cell Dev. Biol. 30, 255–289. 10.1146/annurev-cellbio-101512-122326 [DOI] [PubMed] [Google Scholar]
- Contant C., Umbriaco D., Garcia S., Watkins K. C., Descarries L. (1996). Ultrastructural characterization of the acetylcholine innervation in adult rat neostriatum. Neuroscience 71, 937–947. 10.1016/0306-4522(95)00507-2 [DOI] [PubMed] [Google Scholar]
- Corkrum M., Covelo A., Lines J., Bellocchio L., Pisansky M., Loke K., et al. (2020). Dopamine-evoked synaptic regulation in the nucleus accumbens requires astrocyte activity. Neuron 105, e5.1036–e5.1047. 10.1016/j.neuron.2019.12.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Kock C. P. J., Burnashev N., Lodder J. C., Mansvelder H. D., Brussaard A. B. (2004). NMDA receptors induce somatodendritic secretion in hypothalamic neurones of lactating female rats. J. Physiol. 561, 53–64. 10.1113/jphysiol.2004.069005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Del-Bel E., De-Miguel F. F. (2018). Extrasynaptic neurotransmission mediated by exocytosis and diffusive release of transmitter substances. Front. Synaptic Neurosci. 10:13. 10.3389/fnsyn.2018.00013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- De-Miguel F. F., Santamaría-Holek I., Noguez P., Bustos C., Hernández-Lemus E., Rubí J. M. (2012). Biophysics of active vesicle transport, an intermediate step that couples excitation and exocytosis of serotonin in the neuronal soma. PLoS One 7:e45454. 10.1371/journal.pone.0045454 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Descarries L., Mechawar N. (2000). Ultrastructural evidence for diffuse transmission by monoamine and acetylcholine neurons of the central nervous system. Prog. Brain Res. 125, 27–47. 10.1016/S0079-6123(00)25005-X [DOI] [PubMed] [Google Scholar]
- Descarries L., Watkins K. C., Garcia S., Bosler O., Doucet G. (1996). Dual character, asynaptic and synaptic, of the dopamine innervation in adult rat neostriatum: a quantitative autoradiographic and immunocytochemical analysis. J. Comp. Neurol. 375, 167–186. [DOI] [PubMed] [Google Scholar]
- DiGregorio D. A., Nusser Z., Silver R. A. (2002). Spillover of glutamate onto synaptic AMPA receptors enhances fast transmission at a cerebellar synapse. Neuron 35, 521–533. [DOI] [PubMed] [Google Scholar]
- Dreifuss J. J., Kalnins I., Kelly J. S., Ruf K. B. (1971). Action potentials and release of neurohypophysial hormones in vitro. J. Physiol. 215, 805–817. 10.1113/jphysiol.1971.sp009499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du Vigneaud V. (1954). Hormones of the posterior pituitary gland: oxytocin and vasopressin. Harvey Lect. 50, 1–26. [PubMed] [Google Scholar]
- Dutton A., Dyball R. E. (1979). Phasic firing enhances vasopressin release from the rat neurohypophysis. J. Physiol. 290, 433–440. 10.1113/jphysiol.1979.sp012781 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuxe K., Dahlström A., Höistad M., Marcellino D., Jansson A., Rivera A., et al. (2007). From the Golgi-Cajal mapping to the transmitter-based characterization of the neuronal networks leading to two modes of brain communication: wiring and volume transmission. Brain Res. Rev. 55, 17–54. 10.1016/j.brainresrev.2007.02.009 [DOI] [PubMed] [Google Scholar]
- Glusman S., Kravitz E. A. (1982). The action of serotonin on excitatory nerve terminals in lobster nerve-muscle preparations. J. Physiol. 325, 223–241. 10.1113/jphysiol.1982.sp014147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hökfelt T. (1968). In vitro studies on central and peripheral monoamine neurons at the ultrastructural level. Z Zellforsch Mikrosk Anat. 91, 1–74. 10.1007/BF00336984 [DOI] [PubMed] [Google Scholar]
- Hökfelt T., Barde S., Xu Z. D., Kuteeva E., Rüegg J., Le Maitre E., et al. (2018). Neuropeptide and small transmitter coexistence: fundamental studies and relevance to mental illness. Front. Neural Circuits 12:106. 10.3389/fncir.2018.00106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hörner M., Weiger W. A., Edwards D. H., Kravitz E. A. (1997). Excitation of identified serotonergic neurons by escape command neurons in lobsters. J. Exp. Biol. 200, 2017–2033. [DOI] [PubMed] [Google Scholar]
- Henneberger C., Papouin T., Oliet S. H., Rusakov D. A. (2010). Long-term potentiation depends on release of D-serine from astrocytes. Nature 463, 232–236. 10.1038/nature08673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hernández-Falcón J., Basu A. C., Govindasamy S., Kravitz E. A. (2005). Changes in heart rate associated with contest outcome in agonistic encounters in lobsters. Cell Mol. Neurobiol. 25, 329–343. 10.1007/s10571-005-3063-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirasawa H., Contini M., Raviola E. (2015). Extrasynaptic release of GABA and dopamine by retinal dopaminergic neurons. Philos Trans. R Soc. Lond B Biol. Sci. 370:20140186. 10.1098/rstb.2014.0186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoopmann P., Rizzoli S. O., Betz W. J. I. (2012). Imaging synaptic vesicle recycling by staining and destaining vesicles with FM dyes. Cold Spring Harb. Protoc. 2012, 77–83. 10.1101/pdb.prot067603 [DOI] [PubMed] [Google Scholar]
- Huang L. Y., Neher E. (1996). Ca2+-dependent exocytosis in the somata of dorsal root ganglion neurons. Neuron 17, 135–145. 10.1016/s0896-6273(00)80287-1 [DOI] [PubMed] [Google Scholar]
- Huang H. P., Zhu F. P., Chen X. W., Xu Z. Q., Zhang C. X., Zhou Z. (2012). Physiology of quantal norepinephrine release from somatodendritic sites of neurons in locus coeruleus. Front. Mol. Neurosci 5:29. 10.3389/fnmol.2012.00029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huber R., Orzeszyna M., Pokorny N., Kravitz E. A. (1997). Biogenic amines and aggression: experimental approaches in crustaceans. Brain Behav. Evol. 50, 60–68. 10.1159/000113355 [DOI] [PubMed] [Google Scholar]
- Igelhorst B. A., Niederkinkhaus V., Karus C., Lange M. D., Dietzel I. D. (2015). Regulation of neuronal excitability by release of proteins from glial cells. Philos Trans. R Soc. Lond B Biol. Sci. 370:20140194. 10.1098/rstb.2014.0194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isaacson J. S., Solís J. M., Nicoll R. A. (1993). Local and diffuse synaptic actions of GABA in the hippocampus. Neuron 10, 165–175. 10.1016/0896-6273(93)90308-e [DOI] [PubMed] [Google Scholar]
- Jaffe E. H., Marty A., Schulte A., Chow R. H. (1998). Extrasynaptic vesicular transmitter release from the somata of substantia nigra neurons in rat midbrain slices. J. Neurosci. 18, 3548–3553. 10.1523/JNEUROSCI.18-10-03548.1998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Káradóttir R., Cavelier P., Bergersen L. H., Attwell D. (2005). NMDA receptors are expressed in oligodendrocytes and activated in ischaemia. Nature 438, 1162–1166. 10.1038/nature04302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaushalya S. K., Desai R., Arumugam S., Ghosh H., Balaji J., Maiti S. (2008). Three-photon microscopy shows that somatic release can be a quantitatively significant component of serotonergic neurotransmission in the mammalian brain. J. Neurosci. Res. 86, 3469–3480. 10.1002/jnr.21794 [DOI] [PubMed] [Google Scholar]
- Kravitz E. A. (1988). Hormonal control of behavior: amines and the biasing of behavioral output in lobsters. Science 241, 1775–1781. 10.1126/science.2902685 [DOI] [PubMed] [Google Scholar]
- Kuffler D. P., Nicholls J., Drapeau P. (1987). Transmitter localization and vesicle turnover at a serotoninergic synapse between identified leech neurons in culture. J. Comp. Neurol. 256, 516–526. 10.1002/cne.902560404 [DOI] [PubMed] [Google Scholar]
- Leon-Pinzon C., Cercós M. G., Noguez P., Trueta C., De-Miguel F. F. (2014). Exocytosis of serotonin from the neuronal soma is sustained by a serotonin and calcium-dependent feedback loop. Front. Cell. Neurosci. 8:169. 10.3389/fncel.2014.00169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X., Yu B., Sun Q., Zhang Y., Ren M., Zhang X., et al. (2018). Generation of a whole-brain atlas for the cholinergic system and mesoscopic projectome analysis of basal forebrain cholinergic neurons. Proc. Natl. Acad. Sci. U S A 115, 415–420. 10.1073/pnas.1703601115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livingstone M. S., Harris-Warrick R. M., Kravitz E. A. (1980). Serotonin and octopamine produce opposite postures in lobsters. Science 208, 76–79. 10.1126/science.208.4439.76 [DOI] [PubMed] [Google Scholar]
- Ludwig M., Sabatier N., Bull P. M., Landgraf R., Dayanithi G., Leng G. (2002). Intracellular calcium stores regulate activity-dependent neuropeptide release from dendrites. Nature 418, 85–89. 10.1038/nature00822 [DOI] [PubMed] [Google Scholar]
- Ludwig M., Stern J. (2015). Multiple signalling modalities mediated by dendritic exocytosis of oxytocin and vasopressin. Philos Trans. R Soc. Lond B Biol. Sci. 370:20140182. 10.1098/rstb.2014.0182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maity B. K., Maiti S. (2018). Label-free imaging of neurotransmitters in live brain tissue by multi-photon ultraviolet microscopy. Neuronal Signal 2:NS20180132. 10.1042/NS20180132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcaggi P., Attwell D. (2004). Role of glial amino acid transporters in synaptic transmission and brain energetics. Glia 47, 217–225. 10.1002/glia.20027 [DOI] [PubMed] [Google Scholar]
- Mendez J. A., Bourque M. J., Fasano C., Kortleven C., Trudeau L. E. (2011). Somatodendritic dopamine release requires synaptotagmin 4 and 7 and the participation of voltage-gated calcium channels. J. Biol. Chem. 286, 23928–23937. 10.1074/jbc.M111.218032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mendolesi J. (2018). Exosomes and ectosomes in intercellular communication. Curr. Biol. 28, R435–R444. 10.1016/j.cub.2018.01.059 [DOI] [PubMed] [Google Scholar]
- Metea M. R., Newman E. A. (2006). Calcium signaling in specialized glial cells. Glia 54, 650–655. 10.1002/glia.20352 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mouton P. R., Pakkenberg B., Gundersen H. J., Price D. L. (1994). Absolute number and size of pigmented locus coeruleus neurons in young and aged individuals. J. Chem. Neuroanat. 7, 185–190. 10.1016/0891-0618(94)90028-0 [DOI] [PubMed] [Google Scholar]
- Munsch T., Deitmer J. W. (1992). Calcium transients in identified leech glial cells in situ evoked by high potassium concentrations and 5-hydroxytryptamine. J. Exp. Biol. 167, 251–265. [DOI] [PubMed] [Google Scholar]
- Nair-Roberts R. G., Chatelain-Badie S. D., Benson E., White-Cooper H., Bolam J. P., Ungless M. A., et al. (2008). Stereological estimates of dopaminergic, GABAergic and glutamatergic neurons in the ventral tegmental area, substantia nigra and retrorubral field in the rat. Neuroscience 152, 1024–1031. 10.1016/j.neuroscience.2008.01.046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman E. A. (2015). Glial cell regulation of neuronal activity and blood flow in the retina by release of gliotransmitters. Philos Trans R Soc. Lond B Biol. Sci. 370:20140195. 10.1098/rstb.2014.0195 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicholls J. G., Kuffler D. P. (1990). Quantal release of serotonin from presynaptic nerve terminals. Neurochem. Int. 17, 157–163. 10.1016/0197-0186(90)90138-j [DOI] [PubMed] [Google Scholar]
- Noguez P., Rubí J. M., De-Miguel F. F. (2019). Thermodynamic efficiency of somatic exocytosis of serotonin. Front. Physiol. 10:473. 10.3389/fphys.2019.00473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nusbaum M. P., Blitz D. M., Marder E. (2017). Functional consequences of neuropeptide and small-molecule co-transmission. Nat. Rev. Neurosci. 18, 389–403. 10.1038/nrn.2017.56 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pál B. (2018). Involvement of extrasynaptic glutamate in physiological and pathophysiological changes of neuronal excitability. Cell Mol. Life Sci. 75, 2917–2949. 10.1007/s00018-018-2837-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel J. C., Witkovsky P., Avshalumov M. V., Rice M. E. (2009). Mobilization of calcium from intracellular stores facilitates somatodendritic dopamine release. J. Neurosci. 29, 6568–6579. 10.1523/JNEUROSCI.0181-09.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perea G., Navarrete M., Araque A. (2009). Tripartite synapses: astrocytes process and control synaptic information. Trends Neurosci. 32, 421–431. 10.1016/j.tins.2009.05.001 [DOI] [PubMed] [Google Scholar]
- Piccolino M., Neyton J., Gerschenfeld H. M. (1984). Decrease of gap junction permeability induced by dopamine and cyclic adenosine 3’5’-monophosphate in horizontal cells of turtle retina. J. Neurosci. 4, 2477–2488. 10.1523/JNEUROSCI.04-10-02477.1984 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puopolo M., Hochstetler S. E., Gustincich S., Wightman R. M., Raviola E. (2001). Extrasynaptic release of dopamine in a retinal neuron: activity dependence and transmitter modulation. Neuron 30, 211–225. 10.1016/s0896-6273(01)00274-4 [DOI] [PubMed] [Google Scholar]
- Quentin E., Belmer A., Maroteaux L. (2018). Somato-Dendritic regulation of raphe serotonin neurons; a key to antidepressant action. Front. Neurosci. 12:982. 10.3389/fnins.2018.00982 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rusakov D. A., Kullmann D. M. (1998). Extrasynaptic glutamate diffusion in the hippocampus: ultrastructural constraints, uptake and receptor activation. J. Neurosci. 18, 3158–3170. 10.1523/JNEUROSCI.18-09-03158.1998 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Südhof T. C. (2013). Neurotransmitter release: the last millisecond in the life of a synaptic vesicle. Neuron 80, 675–690. 10.1016/j.neuron.2013.10.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarkar B., Das A. K., Arumugam S., Kaushalya S. K., Bandyopadhyay A., Balaji J., et al. (2012). The dynamics of somatic exocytosis in monoaminergic neurons. Front. Physiol. 3:414. 10.3389/fphys.2012.00414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart R. R., Adams W. B., Nicholls J. G. (1989). Presynaptic calcium currents and facilitation of serotonin release at synapses between cultured leech neurones. J. Exp. Biol. 144, 1–12. [DOI] [PubMed] [Google Scholar]
- Sun Y., Poo M. -M. (1987). Evoked release of acetylcholine from the growing embryonic neurons. Proc. Natl. Acad. Sci. U S A 84, 2540–2544. 10.1073/pnas.84.8.2540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorn P., Zorec R., Rettig J., Keating D. J. (2016). Exocytosis in non-neuronal cells. J. Neurochem. 137, 849–859. 10.1111/jnc.13602 [DOI] [PubMed] [Google Scholar]
- Tobin V., Leng G., Ludwig M. (2012). The involvement of actin, calcium channels and exocytosis proteins in somato-dendritic oxytocin and vasopressin release. Front. Physiol. 3:261. 10.3389/fphys.2012.00261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tobin V. A., Ludwig M. (2007). The role of the actin cytoskeleton in oxytocin and vasopressin release from rat supraoptic nucleus neurons. J. Physiol. 582, 1337–1348. 10.1113/jphysiol.2007.132639 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trueta C., De-Miguel F. F. (2012). Extrasynaptic exocytosis and its mechanisms: a source of molecules mediating volume transmission in the nervous system. Front. Physiol. 3:319. 10.3389/fphys.2012.00319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trueta C., Kuffler D. P., De-Miguel F. F. (2012). Cycling of dense core vesicles involved in somatic exocytosis of serotonin by leech neurons. Front. Physiol. 3:175. 10.3389/fphys.2012.00175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trueta C., Méndez B., De-Miguel F. F. (2003). Somatic exocytosis of serotonin mediated by L-type calcium channels in cultured leech neurones. J. Physiol. 547, 405–416. 10.1113/jphysiol.2002.030684 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trueta C., Sánchez-Armass S., Morales M. A., De-Miguel F. F. (2004). Calcium-induced calcium release contributes to somatic secretion of serotonin in leech Retzius neurons. J. Neurobiol. 61, 309–316. 10.1002/neu.20055 [DOI] [PubMed] [Google Scholar]
- Umbriaco D., Garcia S., Beaulieu C., Descarries L. (1995). Relational features of acetylcholine, noradrenaline, serotonin and GABA axon terminals in the stratum radiatum of adult rat hippocampus (CA1). Hippocampus 5, 605–620. 10.1002/hipo.450050611 [DOI] [PubMed] [Google Scholar]
- Verkhratsky A., Krishtal O. A., Burnstock G. (2009). Purinoceptors on neuroglia. Mol. Neurobiol. 39, 190–208. 10.1007/s12035-009-8063-2 [DOI] [PubMed] [Google Scholar]
- Willard A. L. (1981). Effects of serotonin on the generation of the motor program for swimming by the medicinal leech. J. Neurosci. 1, 936–944. 10.1523/JNEUROSCI.01-09-00936.1981 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witkovsky P., Patel J. C., Lee C. R., Rice M. E. (2009). Immunocytochemical identification of proteins involved in dopamine release from the somatodendritic compartment of nigral dopaminergic neurons. Neuroscience 164, 488–496. 10.1016/j.neuroscience.2009.08.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witkovsky P. (2004). Dopamine and retinal function. Doc. Ophthalmol. 108, 17–40. 10.1023/b:doop.0000019487.88486.0a [DOI] [PubMed] [Google Scholar]
- Zetler G. (1970. “Distribution of peptidergic neurons in mammalian brain,” in Aspects of Neuroendocrinology, eds Bargmann W., Scharrer B., 287–295. 10.1007/978-3-642-46207-8_30 [DOI] [Google Scholar]
- Zhang A. J., Jacoby R., Wu S. M. (2011). Light- and dopamine-regulated receptive field plasticity in primate horizontal cells. J. Comp. Neurol. 519, 2125–2134. 10.1002/cne.22604 [DOI] [PMC free article] [PubMed] [Google Scholar]