Abstract
Primary Sjögren’s syndrome (pSS) is considered to be a multifactorial disease, where underlying genetic predisposition, epigenetic mechanisms and environmental factors contribute to disease development. In the last 5 years, the first genome-wide association studies in pSS have been completed. The strongest signal of association lies within the HLA genes, whereas the non-HLA genes IRF5 and STAT4 show consistent associations in multiple ethnicities but with a smaller effect size. The majority of the genetic risk variants are found at intergenic regions and their functional impact has in most cases not been elucidated. Epigenetic mechanisms such as DNA methylation, histone modifications and non-coding RNAs play a role in the pathogenesis of pSS by their modulating effects on gene expression and may constitute a dynamic link between the genome and phenotypic manifestations. This article reviews the hitherto published genetic studies and our current understanding of epigenetic mechanisms in pSS.
Keywords: primary Sjögren’s syndrome, genetics, GWAS, HLA, IRF5, STAT4, epigenetics, DNA methylation, histone modification, non-coding RNA
Rheumatology key messages
The strongest signal of genetic association with primary Sjögren’s syndrome is within the HLA genes.
Genes relevant to the innate and adaptive immune systems are associated with modest effect sizes.
DNA hypomethylation at interferon-induced genes is an epigenetic modification in different cells and tissues.
Introduction
Primary Sjögren's syndrome (pSS) is a complex disease where genetic, epigenetic and environmental factors are thought to contribute to disease pathogenesis. The overwhelmingly strongest genetic predisposition to pSS is attributed to the HLA genes, where early studies reported different HLA class II gene haplotypes associated with pSS in different ethnic groups [1]. Initial attempts to identify non-HLA susceptibility genes employed a candidate gene approach, later followed by genome-wide association studies (GWASs) as the technology for large-scale genotyping advanced. Genetic risk variants in pSS are predominantly found at intergenic regions and deciphering the functional impact of identified variants is a challenge. Epigenetic modifications have emerged as important mechanisms for understanding how the interaction of genetic predisposition and environmental factors may give rise to chronic autoimmunity, including pSS (Fig. 1) [2]. In this review, current studies of genetic variants and epigenetic alterations in pSS and their implications for disease pathogenesis will be discussed.
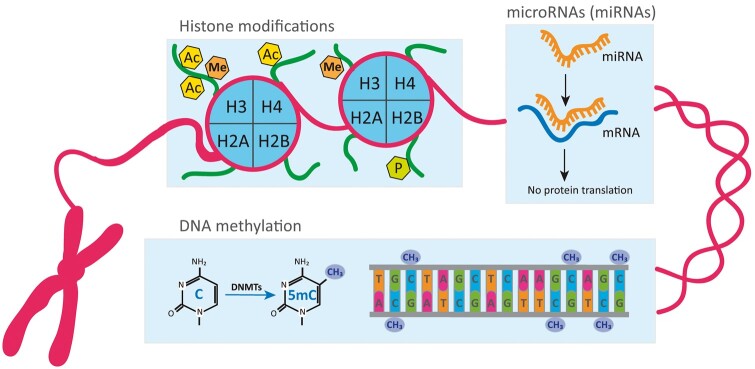
Fig. 1.
Schematic representation of fundamental epigenetic mechanisms involved in the regulation of gene expression
Histone modifications are covalent post-translational modifications within the N-terminal tail of histone proteins. The most studied histone marks include Ac, Me and P of lysine residues at histones H3 and H4. DNA methylation refers to the covalent modification of C residues to 5mC by DNMTs typically in the context of CpG dinucleotides. Histone modifications and DNA methylation modulate chromatin structure and transcriptional accessibility of the DNA, thereby altering gene expression. At the post-transcriptional level, miRNAs, a class of small ncRNA, can bind to mRNA of target genes interfering with their expression. 5mC: 5-methyl-cytosine; Ac: acetylation; C: cytosine; DNMTs: DNA methyltransferases; Me: methylation; P: phosphorylation.
Genetics
The genetics of pSS are largely understudied compared with other systemic autoimmune diseases. Initial evidence supporting an important role of inherited factors in pSS came from reports of familial aggregation, increased concordance rates among monozygotic twins and increased prevalence of other autoimmune diseases amongst relatives of pSS patients [3–5]. The estimated odds of female sibling concordance (λs) for pSS ranges from 8 to 30 and dizygotic twin concordance rates from 2 to 4% [6]. The genetic contribution to pSS has been investigated by studies of X chromosome aneuploidy, candidate genes and GWASs in populations of European and Asian ancestry.
X chromosome
The single most important risk factor for pSS is being female. Given the remarkable gender disparity (14 female:1 male), the X chromosome has been a particularly promising candidate for genetic studies [7]. Harris et al. [8] showed that men with Klinefelter’s syndrome (47,XXY) had a similar risk of pSS as 46,XX women. Furthermore, the coexistence of Turner syndrome (45,X) with pSS is very rare, whereas the estimated prevalence of pSS in women with 47,XXX is ∼2.9 times higher than that in women with 46,XX [9]. The identification of structural chromosomal aberrations resulting in partial triplications of the X chromosome (Xp11.4::pter) in three patients with pSS (a mother–daughter pair and an unrelated female) suggests that dosage-sensitive risk genes may be located within this chromosomal interval [10]. The gene(s) that mediates this effect remains undefined.
MHC
Like in many autoimmune diseases, the strongest pSS genetic associations are within the MHC, and risk variants vary by serological status and among different ethnic groups (Table 1). The most consistent associations to date have been with DR2 and DR3 alleles at the DRB1 locus in Caucasian populations [20, 21]. Very strong associations with anti-Ro/SSA and/or anti-La/SSB were identified in patients that carry DRB1*03 and DQB1*02 alleles or were heterozygous for DQw1 and DQw2 [22, 23]. A meta-analysis of 1166 cases and 6470 controls of diverse ethnic backgrounds derived from 23 studies confirmed that significant risk was associated with pSS and HLA class II alleles DRB1*03:01, DQA1*05:01 and DQB1*02:01 [24].
Table 1.
Established and suggestive primary Sjögren’s syndrome risk loci based on GWASs
| Gene | SNP | ORa (95% CI) | P-value | Population | References |
|---|---|---|---|---|---|
| Risk loci associated at genome-wide significance level (P < 5.0E-08) | |||||
| HLA-DQB1 | rs115575857 | 3.53 (3.03, 4.11) | 7.65E-114 | European | Lessard et al. [11] |
| HLA-DQA1 | rs116232857 | 2.53 (2.24, 2.86) | 1.33E-96 | European | Lessard et al. [11] |
| HLA-DRB1, HLA-DQA1 | rs9271573 | 2.02 (1.82, 2.23) | 3.00E-42 | Multi-ethnicb | Taylor et al. [12] |
| rs9271573 | 2.29 (2.01, 2.62) | 3.00E-34 | European | Taylor et al. [12] | |
| rs9271588 | 0.57 (0.53, 0.63) | 8.52E-37 | Han Chinese | Li et al. [13] | |
| HLA-DQA1, HLA-DQB1 | rs3021302 | 2.24 (1.97, 2.54) | 2.00E-35 | Multi-ethnicb | Taylor et al. [12] |
| HLA-DQB1, HLA-DQA2 | rs9275572 | 2.28 (1.99, 2.61) | 7.00E-33 | European | Taylor et al. [12] |
| HLA-DPB1, COL11A2 | rs4282438 | 1.58 (1.45, 1.72) | 8.77E-25 | Han Chinese | Li et al. [13] |
| MICA | MICA*008 | 1.90 (1.56, 2.31) | 9.37E-09 | European | Carapito et al. [14] |
| IRF5-TNPO3 | rs3757387 | 1.44 (1.29, 1.62) | 2.73E-19 | European | Lessard et al. [11] |
| rs17339836 | 1.58 (1.36, 1.84) | 2.43E-16 | European | Lessard et al. [11] | |
| rs3823536 | 1.49 (1.34, 1.65) | 3.00E-14 | Multi-ethnicb | Taylor et al. [12] | |
| rs3823536 | 1.54 (1.36, 1.76) | 7.00E-11 | European | Taylor et al. [12] | |
| rs59110799 | 1.72 (1.49, 1.99) | 3.00E-13 | Multi-ethnicb | Taylor et al. [12] | |
| rs3807306 | 1.50 (1.32, 1.71) | 6.00E-10 | European | Taylor et al. [12] | |
| STAT4 | rs10553577 | 1.43 (1.26, 1.62) | 6.80E-15 | European | Lessard et al. [11] |
| rs10168266 | 1.44 (1.32, 1.57) | 1.77E-17 | Han Chinese | Li et al. [13] | |
| rs11889341 | 1.40 (1.26, 1.56) | 9.00E-10 | Multi-ethnicb | Taylor et al. [12] | |
| rs7574865 | 1.51 (1.31, 1.75) | 2.00E-08 | European | Taylor et al. [12] | |
| IL12A | rs485497 | 1.30 (1.16, 1.46) | 1.17E-10 | European | Lessard et al. [11] |
| BLK | rs2736345 | 1.30 (1.16, 1.47) | 4.97E-10 | European | Lessard et al. [11] |
| CXCR5 | rs7119038 | 0.74 (0.64, 0.86) | 1.10E-08 | European | Lessard et al. [11] |
| TNIP1 | rs6579837 | 1.43 (1.20, 1.71) | 3.30E-08 | European | Lessard et al. [11] |
| OAS1 | rs10774671 | 0.75 (0.66, 0.86) | 2.59E-09 | European | Li et al. [15] |
| TNFAIP3 | rs5029939 | 1.67 (1.40, 1.99) | 7.75E-09 | Han Chinese | Li et al. [13] |
| GTF2IRD1-GTF2I-NCF1 | rs117026326 | 2.20 (1.99, 2.43) | 1.31E-53 | Han Chinese | Li et al. [13] |
| rs117026326 | 1.98 (1.67, 2.35) | 1.10E-15 | Han Chinese females | Song et al. [16] | |
| rs117026326 | 2.03 (1.06, 2.57) | 4.60E-09 | Chinese | Zhao et al. [17] | |
| IKZF1 | rs4917129 | 0.70 (0.61, 0.79) | 4.24E-08 | Han Chinese | Qu et al. [18] |
| Suggestive risk loci associated at significance level P > 5.0E-08 | |||||
| HLA-DPB1 | rs9277554 | 1.65 (1.37, 2.00) | 3.00E-07 | Asian | Taylor et al. [12] |
| HLA-DPB1/COL11A2 | rs3117221 | 1.47 (1.26, 1.71) | 9.52E-07 | Han Chinese | Li et al. [13] |
| HLA-DQA1 | rs9405117 | 0.54 (0.42, 0.69) | 9.83E-07 | Han Chinese | Li et al. [13] |
| HLA-DQB1 | rs6928482 | 1.43 (1.23, 1.66) | 2.52E-06 | Han Chinese | Li et al. [13] |
| HLA-DMB/PSMB9 | rs11756897 | 1.57 (1.32, 1.87) | 2.76E-07 | Han Chinese | Li et al. [13] |
| LOC105370283-PTMAP5 | rs17074492 | 1.53 (1.31, 1.79) | 6.00E-08 | European | Taylor et al. [12] |
| TNFAIP3 | rs6933404 | 1.29 (1.13, 1.47) | 6.53E-08 | European | Lessard et al. [11] |
| PTTG1 | rs2431098 | 0.81 (0.73, 0.91) | 2.28E-07 | European | Lessard et al. [11] |
| RELN | rs7341475 | 1.39 (1.23, 1.57) | 3.00E-07 | Multi-ethnicb | Taylor et al. [12] |
| PDRM1/ATG5 | rs548234 | 1.52 (1.29, 1.78) | 3.61E-07 | Han Chinese | Li et al. [13] |
| STAT4 | rs3821236 | 1.47 (1.27, 1.71) | 2.92E-07 | Han Chinese | Li et al. [13] |
| KLRG1 | rs1805673 | 0.62 (0.51, 0.74) | 6.00E-07 | Asian | Taylor et al. [12] |
| MIS18BP1/LINC00871 | rs1957173 | 0.61 (0.50, 0.74) | 7.00E-07 | Multi-ethnicb | Taylor et al. [12] |
| IRAK1BP1 | rs1507153 | 1.26 (1.11, 1.43) | 7.09E-07 | European | Lessard et al. [11] |
| HTR2A/LINC00562 | rs7999279 | 1.42 (1.23, 1.63) | 1.00E-06 | Multi-ethnicb | Taylor et al. [12] |
| SHISA9 | rs9938751 | 0.59 (0.48, 0.73) | 1.00E-06 | European | Taylor et al. [12] |
| ZNF208 | rs10416159 | 1.49 (1.26, 1.75) | 1.54E-06 | Han Chinese | Li et al. [13] |
| PRCC/SH2D2A | rs16837677 | 1.54 (1.29, 1.84) | 2.00E-06 | Multi-ethnicb | Taylor et al. [12] |
| NFAT5 | rs7192380 | 1.28 (1.16, 1.42) | 2.00E-06 | Multi-ethnicb | Taylor et al. [12] |
| ITSN2 | rs1545257 | 0.81 (0.71, 0.91) | 2.47E-06 | European | Lessard et al. [11] |
| LINC00648/RPS29 | rs67617551 | 0.62 (0.50, 0.76) | 2.73E-06 | Han Chinese | Li et al. [13] |
| EGLN3/SPTSSA | rs712299 | 1.48 (1.25, 1.74) | 2.84E-06 | Han Chinese | Li et al. [13] |
| PRDM1 | rs526531 | 1.22 (1.09, 1.38) | 2.93E-06 | European | Lessard et al. [11] |
| PDE8B | rs181851 | 0.67 (0.56, 0.79) | 3.00E-06 | European | Taylor et al. [12] |
| NACC2 | rs4842091 | 1.39 (1.21, 1.61) | 5.00E-06 | European | Taylor et al. [12] |
| GRIP2/CCDC174 | rs79407237 | 0.61 (0.49, 0.75) | 5.00E-06 | European | Taylor et al. [12] |
| ZNF43/ZNF208 | rs2522092 | 1.45 (1.24, 1.70) | 5.86E-06 | Han Chinese | Li et al. [13] |
| PHIP | rs10943608 | 1.23 (1.08, 1.40) | 6.22E-06 | European | Lessard et al. [11] |
| C7orf72, IKZF1 | rs4917014 | 0.68 (0.57, 0.81) | 9.86E-06 | Han Chinese | Li et al. [13] |
| RBMS3 | rs13079920 | 1.35 (1.17, 1.55) | 2.90E-05 | Han Chinese | Song et al. [16] |
| GTF2IRD1-GTF2I-NCF1 | rs117026326 | 3.12 (1.36, 7.17) | 7.30E-03 | European American | Zhao et al. [17] |
The OR for each variant indicates the disease risk conferred by the minor allele.
Multi-ethnic cohort: complete SICCA cohort [19].
A large-scale study by Lessard et al. [11] replicated previously identified associations in the MHC, which represent the strongest genetic risk factors in pSS [odds ratio (OR) ∼3.5]. Two highly significant associations were found. The first was with an extended 5 Mb haplotype peaking at HLA-DQB1 that carries multiple loci with expression levels that are altered by risk genotypes. A second independent effect was comprised of variants within a narrow region of association peaking at HLA class II, within HLA-DQA1. These loci were also associated with pSS in a study of Europeans by Taylor et al. [12]. Li et al., in their GWAS of Han Chinese pSS patients, obtained the strongest associations with HLA-DRB1/HLA-DQA1 and HLA-DPB1 [13], while Taylor et al. [12] observed much weaker effects in their Asian subpopulation (HLA-DPB1 and HLA-DRB9). A non-canonical MHC-linked but HLA-independent risk locus for pSS is MHC class I polypeptide-related sequence A (MICA). The MICA gene is highly polymorphic and a strong association of pSS with the MICA*008 allele has been shown in two European cohorts [14].
Non-MHC genes
Beyond the associations with the MHC and X chromosome, the majority of the early studies of pSS genetics focused on candidate genes previously described as implicated in immune function or other autoimmune diseases. Larger genetic studies of pSS using unbiased, genome-wide approaches were first completed in 2013, one in patients of European ancestry and the other in a Han Chinese population [11, 13]. Genetic associations have now been established for pSS at levels reaching a stringent genome-wide significance threshold (P < 5.0E-8) and most of these findings have been replicated in independent studies [11–13, 15–18]. Additional candidate genes with suggestive evidence of association are summarized in Table 2. In general, the locations of peak association lie outside the protein coding regions and the functional consequences are not precisely known, but they are thought to involve regulation of gene expression and function. Candidate genes for these associated regions involve both innate and adaptive immune functions and offer interesting biological clues to the potential pathogenesis of pSS.
Table 2.
Primary Sjögren’s syndrome risk loci derived from candidate gene studies
| Gene | SNP | ORa (95% CI) | P-value | Function | References |
|---|---|---|---|---|---|
| IRF5-TNPO3 | rs2004640 | 1.93 (1.15, 3.42) | 0.01 | Type I IFN and TLR signalling, cytokine production (e.g. IL-6, IL-12), apoptosis | Miceli-Richard et al. [25] |
| rs10488631 | 1.57 (1.23, 1.99) | 3.23E-04 | Nordmark et al. [26] | ||
| CGGGGindel | 2.00 (1.50, 2.70) | 6.60E-06 | Miceli-Richard et al. [27] | ||
| 1.49 (1.24, 1.79) | 2.41E-05 | Nordmark et al. [26] | |||
| rs13246321 | 1.70 (1.35, 2.13) | 5.50E-06 | Nordmark et al. [28] | ||
| STAT4 | rs7574865 | 1.46 (1.09, 1.97) | 0.01 | Type I and II IFN pathways, NOD2 signalling | Korman et al. [29] |
| 1.40 (1.21, 1.62) | 4.70E-06 | Palomino-Morales et al. [30] | |||
| rs7582694 | 1.41 (1.14, 1.73) | 1.45E-03 | Nordmark et al. [26] | ||
| 1.40 (1.15, 1.70) | 7.00E-04 | Nordmark et al. [28] | |||
| BLK-FAM167A | rs12549796 | 1.37 (1.15, 1.63) | 4.70E-04 | B cell signalling and development, thymopoiesis, insulin secretion and synthesis, NF-κB signalling | Nordmark et al. [28] |
| EBF1 | rs3843489 | 1.68 (1.29, 2.18) | 9.90E-05 | Enhancer of transcriptional activity during B cell development | Nordmark et al. [28] |
| TNSF4/OX40L | rs1234315 | 1.34 (1.14, 1.64) | 7.40E-04 | Co-stimulation, T cell proliferation, cytokine production | Nordmark et al. [28] |
| BAFF/TNFSF13B | 5′ haplotypeb | 2.60 (1.70, 4.10) | 4.00E-05 | B cell maturation, proliferation and survival, epithelial cell survival | Nossent et al. [31] |
| 5′ haplotypec (TATT, GTTC) | – | <0.05 | Nezos et al. [32] | ||
| NCR3/NKp30 | rs11575837 | 0.48 | 0.0039 | Regulation of dendritic and NK cell communication, regulation of Th1 (IL-12 and IFN-γ) cytokines | Rusakiewicz et al. [33] |
| rs2736191 | 0.56 | 0.0019 | Rusakiewicz et al. [33] | ||
| PTPN22 | rs2476601 | 2.42 (1.24, 4.75) | 0.01 | T cell activation | Gomez et al. [34] |
| TNIP1 | rs3792783 | 1.33 (1.16, 1.52) | 3.40E-05 | NF-κB signalling, EGF/ERK signalling, TNF-induced apoptosis | Nordmark et al. [35] |
| rs7708392 | 1.21 (1.08, 1.36) | 1.30E-03 | Nordmark et al. [35] | ||
| TNFAIP3 | rs2230926 | 3.26 (1.31, 8.12) | 0.011 | NF-κB signalling (repressor), TNF-induced apoptosis, TLR4 signalling, cytokine production (e.g. IL-1B) | Nocturne et al. [36] |
| BAFF-R | His159Tyr | 4.13 (1.19, 14.3) | 0.01 | NF-κB2 signalling | Papageorgiou et al. [37] |
| LTA/LTB/TNF | rs1800629 | 2.00 (1.61, 2.49) | 2.48E-10 | Lymphoid organogenesis, tertiary lymphoid tissue maintenance | Bolstad et al. [38] |
| rs909253 | 1.59 (1.34, 1.89) | 1.25E-07 | |||
| MECP2 | rs17435 | 1.33 (1.12, 1.59) | 0.0016 | Methylation-mediated transcriptional silencing | Cobb et al. [39] |
| CHRM3 | rs7548522 | 1.93 (1.24, 3.01) | 0.0033 | Muscarinic receptor | Appel et al. [40] |
The OR for each variant indicates the disease risk conferred by the minor allele.
The haplotype in the 5′ regulatory region of the BAFF gene is constituted by the SNPs rs9514827:-2841T>C, rs3759467:-2704T>C, rs1041569:-2701T>A and rs9514828:871C>T.
Nezos et al. [32] analysed the 5′ regulatory region BAFF haplotype and also identified risk haplotypes for lymphoma.
EGF/ERK: epidermal growth factor/extracellular signal-regulated kinase; NOD2: nitric oxide synthetase 2.
Genetic susceptibility to dysregulation of innate immunity is exemplified by associations of pSS with IFN regulatory factor 5 (IRF5), signal transducer and activator of transcription 4 (STAT4), IL-12A (IL12A) and 2′–5′ oligoadenylate synthetase 1 (OAS1). All are involved in type I IFN signalling and overexpression of IFN-induced genes, the so-called IFN signature, which is predominanatly seen in pSS patients positive for anti-Ro/SSA and/or anti-La/SSB [11, 41, 42].
IRF5 is a transcription factor that acts downstream of the toll-like receptors (TLRs) and type I IFN receptor to promote the expression of numerous antiviral and pro-inflammatory proteins [43, 44]. IRF5 is associated with pSS in patients of all investigated ancestries, the most significant association being a CGGGG insertion/deletion (indel) polymorphism in the promoter region [12, 13, 25–27]. An additional risk haplotype that is present only in European ancestry results from variants spanning both IRF5 and TNPO3 [28]. A recent model suggests that within this haplotype, the risk allele increases binding of the transcription factor ZBTB3 to the IRF5 promoter, resulting in upregulated IRF5 expression as the potential pathogenic mechanism [45].
STAT4 is a transcription factor, activated by type I IFN, IL-12 and IL-23, that is also associated with SLE and RA [26, 29, 30, 46]. Interestingly, genetic models that support additive effects between the major risk alleles in IRF5 and STAT4 have been described [26, 46].
IL-12 is an immunomodulatory cytokine primarily secreted by monocytes and dendritic cells that acts upstream of STAT4 and plays a critical role in the differentiation of Th1 cells and production of IFN-γ by T cells and NK cells [47]. The IL12A gene encodes the p35 subunit that forms the IL-12 heterodimer together with the p40 subunit encoded by IL12B [48]. Variants in the 3′ end of IL12A have been reported for primary biliary cirrhosis, while 5′ effects have been described for celiac disease [49–51]; however, the effect in the region of IL12A in pSS appears to be distinct from those previously reported and is stronger in Europeans than in Asians [12].
OAS 1 is a type I IFN-induced gene often seen among the overexpressed genes in autoimmune disease. The pSS risk allele gives rise to a splicing shift of the p46 isoform to alternative transcripts that lack translational response to type I IFN stimulation. At the protein level, the risk variant of OAS1 is associated with decreased OAS1 enzymatic activity and viral clearance [15].
NK cells are a critical cytotoxic lymphocyte type that plays a major role in innate immune responses. Based on the potential role of NK cells in animal models of sialadenitis, a candidate gene case–control study of NCR3/NKp30 showed that two promoter single nucleotide polymorphisms (SNPs) are protective from pSS, suggesting that NK cells may promote an NKp30-dependent inflammatory state in salivary glands [33].
The pathogenesis of pSS also involves dysregulation of adaptive immune responses. Several genes associated with pSS are primarily involved in B cell function. Variants in a region comprising two genes, FAM167A and the BLK (B lymphocyte kinase) locus, were first found to be associated with pSS in a candidate gene study of Swedish and Norwegian patients, and later replicated in other European and Asian cohorts [11, 28, 52]. FAM167A and BLK are transcribed in opposite directions, possibly from common promoter elements, and expression levels are inversely correlated. FAM167A encodes a novel protein, DIORA-1, which is highly expressed in the lung, suggesting a potential role in pulmonary involvement [53]. BLK is involved in B cell signalling that results in activation of multiple nuclear transcription factors. Reduced expression of BLK is hypothesized to lead to a breakdown in tolerance by allowing autoreactive cells to escape depletion [28].
Significantly increased levels of B cell activating factor (BAFF/BLyS), a member of the TNF family, have been demonstrated in serum of pSS patients compared with healthy controls and directly correlate with the degree of clinical activity and titre of circulating autoantibodies [31, 54–58]. Disease susceptibility for anti-Ro/SSA- and anti-La/SSB-positive pSS has been associated with a haplotype of four SNPs located in the 5′ regulatory region of the BAFF gene [31], where Nezos et al. [32] further characterized a different haplotype associated with pSS-lymphoma. A novel association in Lessard et al. [11] was attributed to a SNP located ∼16 kb 5′ of the coding region of CXCR5. CXCR5 associations have been reported in multiple sclerosis and primary biliary cirrhosis [59, 60], and studies in pSS have found dysregulated CXCR5 gene expression in B cells in both the periphery and in salivary gland tissues [61, 62]. Carrying the CXCR5 pSS risk allele correlates with downregulated CXCR5 gene expression in peripheral blood B cells with concomitant low numbers of CXCR5+ peripheral blood B cells and homing of CXCR5+ B cells in the minor salivary glands [63]. Targeting the CXCR5–CXCL13 axis might be a novel therapeutic approach in pSS.
Associations of pSS with risk variants in TNFAIP3 and TNIP1 that are important in NF-κB signalling may affect multiple cell types through a variety of mechanisms, including regulation of TNF-induced apoptosis, TLR4 activation and cytokine production [36]. The variants in TNIP1 identified by Lessard et al. [11] have so far not been replicated in Asian GWASs. The exact function of TNIP1 has not yet been defined, but it binds to TNFAIP3, which in turn suppresses TLR-induced apoptosis by negatively regulating NF-κB [64, 65].
Several additional associations with pSS that appear to be exclusive to Asian ancestry have been reported. The GWAS of Han Chinese pSS patients by Li et al. [13] identified an association with the novel GTF2I; the strongest associations were with SNPs extending from GTF2I to GTF2IRD1-GTF2I. A GWAS of Taiwanese Han Chinese pSS females replicated the association, which is also strongly associated with SLE in Asians, and further identified a suggestive variant in RBMS3, a modulator of acinar apoptosis and TGF-β signalling in the exocrine system [16, 17]. Taylor et al. [12] found an Asian-only association with KLRG1 and IKZF1; the latter was replicated in Han Chinese pSS patients [18]. IKZF1 is a transcription factor implicated in chromatin remodelling that regulates lymphocyte differentiation and shares a common interaction with members of the histone deacetylase families with GTF2I [18]. Zhao et al. [17] fine-mapped the region spanning GTF2IRD1-GTF2I and determined that the pSS risk allele tags the p.Arg90His variant in the neighbouring NCF1 gene, which is associated with pSS in Chinese.
In summary, apart from the HLA genes, the effect sizes of identified genetic associations are generally small and the functional impact of identified genetic variants has in most cases not been elucidated. Studying the interaction of the genetic risk loci with epigenetic alterations may increase our understanding of pSS genetic risk.
Epigenetics
Epigenetic modifications comprise mitotically heritable changes that can potentially influence the phenotype without involving any alteration to the DNA sequence itself. Epigenetic marks are relatively stable over time, facilitating the maintenance of cell identity, yet dynamic enough to change in response to various external and internal stimuli [66]. Epigenetic regulation is primarily accomplished by DNA methylation, histone modifications and non-coding RNAs (ncRNAs).
Role of DNA methylation in pSS
The most widely studied epigenetic mark is DNA methylation, depicted in Fig. 1. DNA methylation can modulate gene expression through chromatin rearrangements, leading to altered accessibility of gene regulatory regions for binding of transcription factor complexes [67]. In addition, a considerable number of Cytosine-phosphate-Guanine dinulceotide (CpG) sites are located in intergenic regions, indicating their contribution to long-distance regulatory processes.
Early investigations in pSS have assessed methylation at specific CpG sites in candidate genes or determined global methylation without single CpG site resolution in different cell and tissue types [68–74]. More recently, epigenome-wide association studies (EWASs) have applied the Infinium HumanMethylation450 (HM450k) BeadChip array [96], summarized in Table 3.
Table 3.
Epigenetic studies in primary Sjögren’s syndrome
| Genome-wide studies of DNA methylation using HM450k array technology | |||
|---|---|---|---|
| Cells/tissues | Number of cases/controls | Main outcome | References |
| Naïve CD4+ T cells | 11/11 | Hypomethylation at LTA and IFN-induced genes | Altorok et al. [75] |
| CD4+ T cells, CD19+ B cells | 26/22 | Larger methylation differences in B cells than in T cells | Miceli-Richard et al. [76] |
| Whole blood, CD19+ B cells, minor salivary glands | 100/400, 24/47, 15/13 | Hypomethylation at IFN-induced genes; meQTL effects at pSS GWAS risk loci | Imgenberg-Kreuz et al. [77] |
| Whole blood | 24/24 (case–case) | Hypomethylation at a ncRNA in high fatigue | Braekke-Norheim et al. [78] |
| Minor salivary glands | 13/13 | Enrichment for differential methylation in promoters | Cole et al. [79] |
| SGECs | 8/4 | Hypomethylation at IFN-induced genes | Charras et al. [80] |
| Studies of differential miRNA expression | |||
|---|---|---|---|
| Method | Cells/tissues | Main outcome | References |
| RT-qPCR | PBMCs | Dysregulation of miR-146a/b, miR-155, miR-223, miR-483-5p | Pauley et al. [81], Kapsogeorgou et al. [82], Zilahi et al. [83], Shi et al. [84], Gourzi et al. [85], Chen et al. [86] |
| SGECs |
|
||
| Minor salivary glands | Dysregulation of miR-16 and miR-181a | Gourzi et al. [85], Wang et al. [88] | |
| RT-qPCR OpenArray | Serum | Correlation of miRNA expression with clinical parameters | Lopes et al. [89] |
| Microarray | PBMCs | Upregulation of miR-181a | Peng et al. [90] |
| CD14+ monocytes | Dysregulation of miRNAs in TGF-β pathway | Williams et al. [91] | |
| CD4+ T cells, CD19+ B cells | Inverse correlation of miR-30b-5p and BAFF expression in B cells | Wang-Renault et al. [92] | |
| Minor salivary glands | Inverse correlation of miR-768-3p and miR-574 with focus score | Alevizos et al. [93] | |
| Next-generation sequencing | PBMCs | Downregulation of miR-105-5p | Chen et al. [94] |
| Minor salivary glands | Upregulation of miR-5100 | Tandon et al. [95] | |
AB: autoantibody; meQTL: methylation quantitative trait loci; RT-qPCR: reverse transcriptase quantitative PCR; SGECs: salivary gland epithelial cells.
EWASs in peripheral blood cells
The first EWAS in pSS was published by Altorok et al. [75], where differentially methylated CpG sites in naïve CD4+ T cells were investigated. A main finding was hypomethylation at multiple sites of lymphotoxin (LT)-α. Signalling of LT-α/-β expressed on immune cells through the LT-β receptor (LTβR) expressed on epithelial cells is essential for the formation of ectopic lymphoid tissue, and upregulated expression of LT-β mRNA in pSS minor salivary glands has been described previously [38, 62]. Further results included hypomethylation at IFN-induced genes such as STAT1, IFI44L and IFITM1 and genes encoding members of solute carrier proteins. This study benefits from the analysis of a single cell type of naïve CD4+ T cells, which allows assessment of DNA methylation changes preceding immune activation.
In a later EWAS, Miceli-Richard et al. [76] interrogated methylation patterns in peripheral CD19+ B cells and CD4+ T cells. A more pronounced differential methylation was seen in B cells compared with T cells, which may indicate aberrant gene regulation in pSS B cells as an important disease mechanism. Hypomethylated CpG sites in pSS B cells were found at IFN-induced genes, more prominently when only patients positive for anti-Ro/SSA and/or La/SSB antibodies were included. Patients with high disease activity also presented with more abundant differential methylation [97]. However, the relatively low number of study subjects precludes any firm conclusion regarding the impact of methylation changes on disease activity.
A larger EWAS was performed by Imgenberg-Kreuz et al. [77]. In whole blood, the most significantly hypomethylated sites were found at type I IFN-induced genes such as MX1, IFI44L, PARP9 and IFITM1, where the difference in methylation levels was more pronounced when only anti-Ro/SSA and/or anti-La/SSB antibody–positive patients were included, confirming results by Miceli-Richard et al. [76]. A larger difference in methylation levels at IFN-regulated genes was detected when analysing peripheral blood B cells rather than whole blood, suggesting that single cell type analyses may be advantageous. This study also investigated gene expression patterns in B cells, which showed increased mRNA expression of IFN-induced genes in pSS, corroborating the IFN signature both at the methylation and gene expression level.
Epigenetic signatures are cell type specific and whole blood cell composition differs between patients with pSS and controls [77]. This can be partly overcome by reference-based methods for statistical deconvolution of major blood cell types, although the results may still be confounded by cellular subtypes. In order to fully dissect epigenetic changes in different immune cell types in pSS, analyses of fractionated cell populations are warranted. Given the central role for B cells in pSS pathogenesis, longitudinal studies of DNA methylation patterns of peripheral naïve B cells and activated B cell subtypes would be of great interest to decipher epigenetic changes during immune activation. Further, studying plasmacytoid dendritic cells (pDCs), the main IFN-producing cell type, would be of importance, but these cells are infrequent in the circulation and epigenetic analysis may prove difficult.
To summarize the results from the three EWASs, hypomethylation at IFN-induced genes is observed in different cell types and is more pronounced in patients with anti-Ro/SSA and/or anti-La/SSB antibodies. This is in concordance with previous findings of a gene expression IFN signature in whole blood, different peripheral blood mononuclear cells (PBMCs) and salivary gland tissue from patients with pSS, preferentially seen in antibody-positive patients with pSS [41, 62, 98–100].
EWASs in salivary glands and salivary gland epithelial cells
In the study by Imgenberg-Kreuz et al. [77], analysis of minor salivary gland biopsies identified the most significantly hypomethylated CpG site at the IFN-induced gene OAS2. Cole et al. [79] also performed an EWAS on minor salivary glands; among genes enriched for promoter differential methylation were transcription factors, long ncRNAs, miRNAs and the cell surface marker genes CXCR5 and TNFRSF13B (TACI). A limitation of both studies is the fact that minor salivary gland tissue consists of multiple cell types, where inflammatory cells are only sparsely present in control glands, and therefore the results must be interpreted with caution.
To overcome the effects of cellular heterogeneity, Charras et al. [80] studied long-term cultured salivary gland epithelial cells derived from minor salivary glands of patients with pSS and controls. IFN-induced genes were among differentially methylated genes, corroborating the results obtained with whole minor salivary glands. These findings emphasize that immunologically active epithelial cells display similar signalling pathways as blood cells, supporting the concept of pSS as autoimmune epithelitis [101]. Analyses of immune cells dissected from patient salivary glands may be another possibility. Comparing DNA methylation levels in, for example, peripheral naïve and infiltrating activated B cells from the same patient could yield important insights into gene regulation during B cell homing and activation, providing additional information to the results of epigenetic analyses of peripheral blood cells only.
DNA methylation patterns of clinical subphenotypes
DNA methylation reflects the epigenetic status at a certain time point that might correlate with disease activity or specific disease manifestations. In a case–case EWAS, Braekke Norheim et al. [78] investigated differential DNA methylation in whole blood samples from pSS patients with either high or low fatigue, assessed by the fatigue visual analogue scale. Hypomethylation of a ncRNA associated with high fatigue was the main finding. Limitations of this study are the relatively small sample size and the study design analysing fatigue as a categorical variable.
Genetic regulation of DNA methylation
Genetic risk variants in pSS may potentially have functional consequences by influencing DNA methylation patterns of target genes, thereby modulating transcriptional accessibility. In the study by Imgenberg-Kreuz et al. [77], genetic variants at seven pSS GWAS loci were tested for association with methylation levels in whole blood from control individuals in a methylation quantitative trait loci analysis [11]. The results indicate that all tested pSS GWAS alleles have the potential to affect DNA methylation levels at nearby genes, most notably within the HLA region and at the IRF5-TNPO3 locus (Fig. 2), pointing to a connection between pSS susceptibility loci and epigenetic regulation. However, studies investigating methylation quantitative trait loci effects in fractionated cells and additional tissues types from patients with pSS are still lacking.
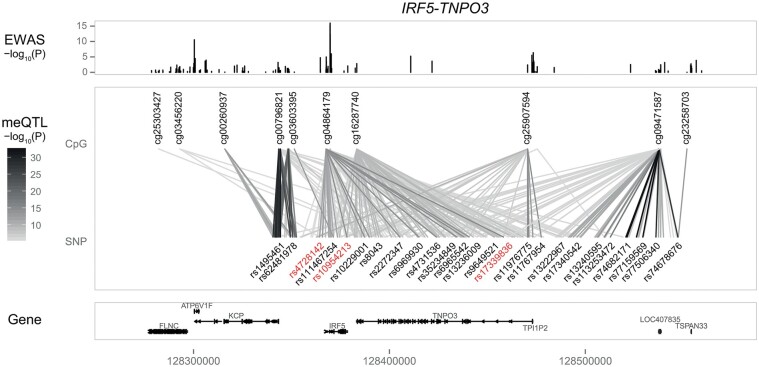
Fig. 2.
Illustration of genetic regulation of DNA methylation at the IRF5-TNPO3 pSS susceptibility locus
Data presented here are derived from meQTL analysis by Imgenberg-Kreuz et al. [73]. Top panel: Results (as –log10 P-values) of the pSS case–control EWAS of 100 pSS patients and 400 controls, with cg04864179 being the top differentially methylated CpG site within this locus. Middle panel: Significant meQTLs analysed in 382 controls, genotyped on the ImmunoChip, with the lines connecting the CpG site and corresponding SNPs in darker grey representing a stronger significance of the meQTL. GWAS index SNPs are indicated in red. Bottom panel: The RefSeq genes in the region. meQTL: methylation quantitative trait loci.
Histone modifications
The N-terminal tails of histones protrude out of the nucleosome and are subject to a variety of covalent post-translational modifications with acetylation and methylation of lysine (K) residues at histones H3 and H4 being the most studied modifications (Fig. 1) [102]. Briefly, acetylation leads to a relaxed chromatin conformation permissive to transcription, while deacetylation results in transcriptional repression by inducing chromatin compaction [103]. Konsta et al. [104] found an association of genetic risk variants with marks for promoters and enhancers in reference B cells and with enhancers in reference monocytes. In the study by Imgenberg-Kreuz et al. [77], hypomethylated sites in pSS were enriched for enhancer regions in reference B and T cells, whereas hypermethylated sites predominantly overlapped with a histone mark that indicates actively transcribed genes. For now, there are no studies available analysing histone marks directly in primary cells from patients with pSS. However, attempts at integration of multiple layers of genome-wide data such as SNP genotyping, DNA methylation and histone modifications hold great promise to provide further insights into the functional roles of pSS-associated variants.
ncRNAs
ncRNAs comprise a large family of different RNAs, with miRNAs being the most extensively studied. miRNAs are a class of evolutionary highly conserved small single-stranded RNA molecules with a length of 19–25 nucleotides that can bind to target mRNA transcripts and interfere with translation (Fig. 1). Aberrant expression of miRNAs has been demonstrated in numerous autoimmune diseases [105].
In pSS, studies in peripheral blood cells, minor salivary gland tissue and salivary gland epithelial cells have identified a number of differentially expressed miRNAs, summarized in [81–95]. However, the target genes and the functional role of these miRNAs remain elusive in many cases. General limitations of these studies are small sample sizes, heterogeneous clinical parameters and potential confounding of the results by variation in cell type distribution. A consistent finding of several studies is upregulated expression of miR-146a/b in PBMCs from patients with pSS [81, 83, 84, 86]. A proposed mechanism for miR-146-dependent regulation of immune responses is via negative feedback mechanisms targeting TLR signalling [106, 107]; dysregulated miR-146 expression may promote excess inflammation, leading to autoimmune responses.
The development of expression microarray and next-generation sequencing technologies has facilitated simultaneous analysis of expression patterns of a larger number of miRNAs. Peng et al. [90] identified overexpression of miR-181a in PBMCs in a Chinese pSS cohort compared with controls. In contrast, a study by Wang et al. [88] found miR-181a, along with miR-16 expression, to be downregulated in labial salivary gland tissue in pSS. Additional studies investigating miR-181a and miR-16 expression in different tissue types in larger, clinically well-characterized cohorts are needed to ascertain the role of these miRNAs in pSS pathogenesis. In a recent study, Wang-Renault et al. [92] analysed miRNA expression patterns in peripheral T cells and B cells from patients with pSS and healthy controls. When only anti-Ro/SSA-positive patients were included in the analysis increased differential miRNA expression was seen in B cells, but not in T cells.
A few studies have correlated clinical manifestations with miRNA expression. Lopes et al. [89] performed unsupervised hierarchical clustering based on serum expression levels of nine small ncRNAs and found correlations with a number of laboratory parameters. A recent study by Kapsogeorgou et al. [87] reported that downregulated miR200b-5p expression in minor salivary gland biopsies preceded lymphoma development, suggesting miR200-5p levels as a predictive biomarker for lymphoma. Additional studies comprising larger sample sizes are required to replicate these intriguing findings and to further explore the role of miRNA expression for lymphomagenesis in pSS.
In summary, dysregulated miRNAs are likely to promote excess inflammation and autoimmune responses in pSS. The studies point to aberrant miRNA expression preferentially affecting B cell activation in antibody-positive pSS, a pattern also observed for pSS-related differential DNA methylation.
Conclusion
As of today, genetic studies in pSS on a genome-wide scale comprising large cohorts have been successful in establishing convincing associations, albeit restricted to individuals of European and Asian ancestry. The most robust association has been determined for HLA class II genes, reflecting that antigen presentation to CD4+ T cells and subsequent immune activation is an important disease mechanism. The consistent genetic associations with IRF5 and STAT4 in the type I IFN system are in agreement with DNA hypomethylation and overexpression of IFN-induced genes present in different pSS cell types [41, 42, 61, 98, 99, 108]. A schematic summary of genetic and epigenetic mechanisms associated with pSS susceptibility is depicted in Fig. 3.
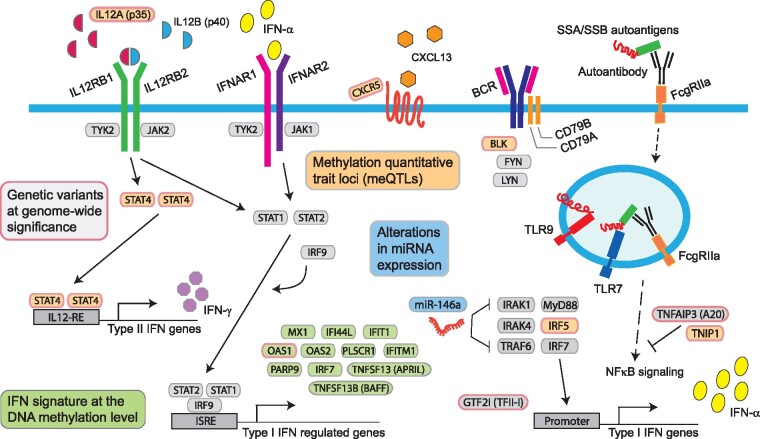
Fig. 3.
Schematic summary of genetic and epigenetic mechanisms associated with pSS susceptibility
Boxes with pink borders represent genes with SNPs associated with pSS susceptibility at genome-wide significance outside of the HLA region and include BLK, CXCR5, GTF2I, IL12A, IRF5, OAS1, STAT4, TNFAIP3 and TNIP1. Green boxes: IFN-induced genes such as MX1, IFI44L, OAS1, OAS2, TNFSF13B and IRF7, with hypomethylated CpG sites in pSS. Orange boxes: meQTLs, referring to association between a genetic variant (SNP) and the methylation level at a nearby CpG site demonstrated at BLK, CXCR5, IL12A, IRF5-TNPO3, STAT4 and TNIP1. Blue boxes: Alterations in miRNA expression, including miRNA-146a, have been identified in pSS. meQTL: methylation quantitative trait loci.
Genetic and epigenetic studies continue to reveal the complexity of pSS. To advance the field further, next-generation whole-genome sequencing of large cohorts of clinically well-characterized pSS patients are warranted, intersected with genome-wide transcriptional profiling and epigenetic data. Application of advanced analytical approaches, such as imputing genotypes across the genome to increase density, and hence information content of SNP data, will further aid in genetic studies of pSS. Continuing to increase sample sizes will be critical to enhancing statistical power for discovery of additional novel genes and identifying patient subsets based on molecular classifications. Analysis of patient subphenotypes will advance our understanding of the disease mechanisms underlying the diverse clinical manifestations and add novel insights into biomarkers and potential therapeutic targets.
Acknowledgments
The authors thank Jonas Carlsson Almlöf for performing analysis and plotting of methylation quantitative trait loci.
Funding: This study was supported by grants from the Swedish Research Council for Medicine and Health (2016-01982 to GN), an AstraZeneca–Science for Life Laboratory research collaboration grant (GN and JIK) and the National Institutes of Health [grants 5R01 AR50782 (KLS), P50 AR0608040 (KLS), 5U19 AI 082714 (KLS), 5U01DE017593 (KLS and AR) and 5R01 DE018209 (KLS and AR)]. The contents are the sole responsibility of the authors and do not necessarily represent the official views of the National Institutes of Health. Additional funding was obtained from the Phileona Foundation (KLS) and the Oklahoma Medical Research Foundation (KLS and AR).
Disclosure statement: The authors have declared no conflicts of interest.
References
- 1. Kang HI, Fei HM, Saito I. et al. Comparison of HLA class II genes in Caucasoid, Chinese, and Japanese patients with primary Sjögren’s syndrome. J Immunol 1993;150:3615–23. [PubMed] [Google Scholar]
- 2. Ballestar E, Li T.. New insights into the epigenetics of inflammatory rheumatic diseases. Nat Rev Rheumatol 2017;13:593–605. [DOI] [PubMed] [Google Scholar]
- 3. Bolstad AI, Haga HJ, Wassmuth R. et al. Monozygotic twins with primary Sjögren’s syndrome. J Rheumatol 2000;27:2264–6. [PubMed] [Google Scholar]
- 4. Houghton KM, Cabral DA, Petty RE, Tucker LB.. Primary Sjögren’s syndrome in dizygotic adolescent twins: one case with lymphocytic interstitial pneumonia. J Rheumatol 2005;32:1603–6. [PubMed] [Google Scholar]
- 5. Scofield RH, Kurien BT, Reichlin M.. Immunologically restricted and inhibitory anti-Ro/SSA in monozygotic twins. Lupus 1997;6:395–8. [DOI] [PubMed] [Google Scholar]
- 6. Anaya JM, Gomez L, Castiblanco J.. Is there a common genetic basis for autoimmune diseases? Clin Dev Immunol 2006;13:185–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Brito-Zeron P, Acar-Denizli N, Zeher M. et al. Influence of geolocation and ethnicity on the phenotypic expression of primary Sjögren’s syndrome at diagnosis in 8310 patients: a cross-sectional study from the Big Data Sjögren Project Consortium. Ann Rheum Dis 2017;76:1042–50. [DOI] [PubMed] [Google Scholar]
- 8. Harris VM, Sharma R, Cavett J. et al. Klinefelter’s syndrome (47,XXY) is in excess among men with Sjögren’s syndrome. Clin Immunol 2016;168:25–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Liu K, Kurien BT, Zimmerman SL. et al. X chromosome dose and sex bias in autoimmune diseases. Arthritis Rheumatol 2016;68:1290–300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Sharma R, Harris VM, Cavett J. et al. Rare X chromosome abnormalities in systemic lupus erythematosus and Sjögren’s syndrome. Arthritis Rheumatol 2017;69:2187–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Lessard CJ, Li H, Adrianto I. et al. Variants at multiple loci implicated in both innate and adaptive immune responses are associated with Sjögren’s syndrome. Nat Genet 2013;45:1284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Taylor KE, Wong Q, Levine DM. et al. Genome-wide association analysis reveals genetic heterogeneity of Sjögren’s syndrome according to ancestry. Arthritis Rheumatol 2017;69:1294–305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Li Y, Zhang K, Chen H. et al. A genome-wide association study in Han Chinese identifies a susceptibility locus for primary Sjögren’s syndrome at 7q11.23. Nat Genet 2013;45:1361–5. [DOI] [PubMed] [Google Scholar]
- 14. Carapito R, Gottenberg J-E, Kotova I. et al. A new MHC-linked susceptibility locus for primary Sjögren’s syndrome: mICA. Hum Mol Genet 2017;26:2565–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Li H, Reksten TR, Ice JA. et al. Identification of a Sjögren’s syndrome susceptibility locus at OAS1 that influences isoform switching, protein expression, and responsiveness to type I interferons. PLoS Genet 2017;13:e1006820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Song I-W, Chen H-C, Lin Y-F. et al. Identification of susceptibility gene associated with female primary Sjögren’s syndrome in Han Chinese by genome-wide association study. Hum Genet 2016;135:1287–94. [DOI] [PubMed] [Google Scholar]
- 17. Zhao J, Ma J, Deng Y. et al. A missense variant in NCF1 is associated with susceptibility to multiple autoimmune diseases. Nat Genet 2017;49:433–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Qu S, Du Y, Chang S. et al. Common variants near IKZF1 are associated with primary Sjögren’s syndrome in Han Chinese. PLoS One 2017;12:e0177320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Shiboski SC, Shiboski CH, Criswell LA. et al. American College of Rheumatology classification criteria for Sjögren’s syndrome: a data-driven, expert consensus approach in the Sjögren’s International Collaborative Clinical Alliance cohort. Arthritis Care Res (Hoboken) 2012;64:475–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Cobb BL, Lessard CJ, Harley JB. et al. Genes and Sjögren’s syndrome. Rheum Dis Clin North Am 2008;34:847–68, vii. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Harley JB, Reichlin M, Arnett FC. et al. Gene interaction at HLA-DQ enhances autoantibody production in primary Sjögren’s syndrome. Science 1986;232:1145–7. [DOI] [PubMed] [Google Scholar]
- 22. Gottenberg J-E, Busson M, Loiseau P. et al. Association of transforming growth factor beta1 and tumor necrosis factor alpha polymorphisms with anti-SSB/La antibody secretion in patients with primary Sjögren’s syndrome. Arthritis Rheum 2004;50:570–80. [DOI] [PubMed] [Google Scholar]
- 23. Gottenberg J-E, Busson M, Loiseau P. et al. In primary Sjögren’s syndrome, HLA class II is associated exclusively with autoantibody production and spreading of the autoimmune response. Arthritis Rheum 2003;48:2240–5. [DOI] [PubMed] [Google Scholar]
- 24. Cruz-Tapias P, Rojas-Villarraga A, Maier-Moore S. et al. HLA and Sjögren’s syndrome susceptibility. A meta-analysis of worldwide studies. Autoimmun Rev 2012;11:281–7. [DOI] [PubMed] [Google Scholar]
- 25. Miceli-Richard C, Comets E, Loiseau P. et al. Association of an IRF5 gene functional polymorphism with Sjögren’s syndrome. Arthritis Rheum 2007;56:3989–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Nordmark G, Kristjansdottir G, Theander E. et al. Additive effects of the major risk alleles of IRF5 and STAT4 in primary Sjögren’s syndrome. Genes Immun 2009;10:68–76. [DOI] [PubMed] [Google Scholar]
- 27. Miceli-Richard C, Gestermann N, Ittah M. et al. The CGGGG insertion/deletion polymorphism of the IRF5 promoter is a strong risk factor for primary Sjögren’s syndrome. Arthritis Rheum 2009;60:1991–7. [DOI] [PubMed] [Google Scholar]
- 28. Nordmark G, Kristjansdottir G, Theander E. et al. Association of EBF1, FAM167A(C8orf13)-BLK and TNFSF4 gene variants with primary Sjögren’s syndrome. Genes Immun 2011;12:100–9. [DOI] [PubMed] [Google Scholar]
- 29. Korman BD, Alba MI, Le JM. et al. Variant form of STAT4 is associated with primary Sjögren’s syndrome. Genes Immun 2008;9:267–70. [DOI] [PubMed] [Google Scholar]
- 30. Palomino-Morales RJ, Diaz-Gallo L-M, Witte T. et al. Influence of STAT4 polymorphism in primary Sjögren’s syndrome. J Rheumatol 2010;37:1016–9. [DOI] [PubMed] [Google Scholar]
- 31. Nossent JC, Lester S, Zahra D. et al. Polymorphism in the 5′ regulatory region of the B-lymphocyte activating factor gene is associated with the Ro/La autoantibody response and serum BAFF levels in primary Sjögren’s syndrome. Rheumatology (Oxford) 2008;47:1311–6. [DOI] [PubMed] [Google Scholar]
- 32. Nezos A, Papageorgiou A, Fragoulis G. et al. B-cell activating factor genetic variants in lymphomagenesis associated with primary Sjögren’s syndrome. J Autoimmun 2014;51:89–98. [DOI] [PubMed] [Google Scholar]
- 33. Rusakiewicz S, Nocturne G, Lazure T. et al. NCR3/NKp30 contributes to pathogenesis in primary Sjögren’s syndrome. Sci Transl Med 2013;5:195ra96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Gomez LM, Anaya J-M, Gonzalez CI. et al. PTPN22 C1858T polymorphism in Colombian patients with autoimmune diseases. Genes Immun 2005;6:628–31. [DOI] [PubMed] [Google Scholar]
- 35. Nordmark G, Wang C, Vasaitis L. et al. Association of genes in the NF-κB pathway with antibody-positive primary Sjögren’s syndrome. Scand J Immunol 2013;78:447–54. [DOI] [PubMed] [Google Scholar]
- 36. Nocturne G, Boudaoud S, Miceli-Richard C. et al. Germline and somatic genetic variations of TNFAIP3 in lymphoma complicating primary Sjögren’s syndrome. Blood 2013;122:4068–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Papageorgiou A, Mavragani CP, Nezos A. et al. A BAFF receptor His159Tyr mutation in Sjögren’s syndrome-related lymphoproliferation. Arthritis Rheumatol 2015;67:2732–41. [DOI] [PubMed] [Google Scholar]
- 38. Bolstad AI, Le Hellard S, Kristjansdottir G. et al. Association between genetic variants in the tumour necrosis factor/lymphotoxin alpha/lymphotoxin beta locus and primary Sjögren’s syndrome in Scandinavian samples. Ann Rheum Dis 2012;71:981–8. [DOI] [PubMed] [Google Scholar]
- 39. Cobb BL, Fei Y, Jonsson R. et al. Genetic association between methyl-CpG binding protein 2 (MECP2) and primary Sjögren’s syndrome. Ann Rheum Dis 2010;69:1731–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Appel S, Le Hellard S, Bruland O. et al. Potential association of muscarinic receptor 3 gene variants with primary Sjögren’s syndrome. Ann Rheum Dis 2011;70:1327–9. [DOI] [PubMed] [Google Scholar]
- 41. Emamian ES, Leon JM, Lessard CJ. et al. Peripheral blood gene expression profiling in Sjögren’s syndrome. Genes Immun 2009;10:285–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Brkic Z, Maria NI, van Helden-Meeuwsen CG. et al. Prevalence of interferon type I signature in CD14 monocytes of patients with Sjögren’s syndrome and association with disease activity and BAFF gene expression. Ann Rheum Dis 2013;72:728–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Takaoka A, Yanai H, Kondo S. et al. Integral role of IRF-5 in the gene induction programme activated by Toll-like receptors. Nature 2005;434:243–9. [DOI] [PubMed] [Google Scholar]
- 44. Taniguchi T, Ogasawara K, Takaoka A. et al. IRF family of transcription factors as regulators of host defense. Annu Rev Immunol 2001;19:623–55. [DOI] [PubMed] [Google Scholar]
- 45. Kottyan LC, Zoller EE, Bene J. et al. The IRF5-TNPO3 association with systemic lupus erythematosus has two components that other autoimmune disorders variably share. Hum Mol Genet 2015;24:582–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Gestermann N, Mekinian A, Comets E. et al. STAT4 is a confirmed genetic risk factor for Sjögren’s syndrome and could be involved in type 1 interferon pathway signaling. Genes Immun 2010;11:432–8. [DOI] [PubMed] [Google Scholar]
- 47. Xu M, Mizoguchi I, Morishima N. et al. Regulation of antitumor immune responses by the IL-12 family cytokines, IL-12, IL-23, and IL-27. Clin Dev Immunol 2010;2010:832454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Watford WT, Hissong BD, Bream JH. et al. Signaling by IL-12 and IL-23 and the immunoregulatory roles of STAT4. Immunol Rev 2004;202:139–56. [DOI] [PubMed] [Google Scholar]
- 49. Lessard CJ, Adrianto I, Ice JA. et al. Identification of IRF8, TMEM39A, and IKZF3-ZPBP2 as susceptibility loci for systemic lupus erythematosus in a large-scale multiracial replication study. Am J Hum Genet 2012;90:648–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Hirschfield GM, Liu X, Xu C. et al. Primary biliary cirrhosis associated with HLA, IL12A, and IL12RB2 variants. N Engl J Med 2009;360:2544–2555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Hunt KA, Zhernakova A, Turner G. et al. Newly identified genetic risk variants for celiac disease related to the immune response. Nat Genet 2008;40:395–402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Sun F, Li P, Chen H. et al. Association studies of TNFSF4, TNFAIP3 and FAM167A-BLK polymorphisms with primary Sjögren’s syndrome in Han Chinese. J Hum Genet 2013;58:475–9. [DOI] [PubMed] [Google Scholar]
- 53. Mentlein L, Thorlacius GE, Meneghel L. et al. The rheumatic disease-associated FAM167A-BLK locus encodes DIORA-1, a novel disordered protein expressed highly in bronchial epithelium and alveolar macrophages. Clin Exp Immunol 2018;193:167–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Mariette X, Roux S, Zhang J. et al. The level of BLyS (BAFF) correlates with the titre of autoantibodies in human Sjögren’s syndrome. Ann Rheum Dis 2003;62:168–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Pers J-O, Daridon C, Devauchelle V. et al. BAFF overexpression is associated with autoantibody production in autoimmune diseases. Ann N Y Acad Sci 2005;1050:34–9. [DOI] [PubMed] [Google Scholar]
- 56. Schneider P, MacKay F, Steiner V. et al. BAFF, a novel ligand of the tumor necrosis factor family, stimulates B cell growth. J Exp Med 1999;189:1747–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Groom J, Kalled SL, Cutler AH. et al. Association of BAFF/BLyS overexpression and altered B cell differentiation with Sjögren’s syndrome. J Clin Invest 2002;109:59–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Mackay F, Browning JL.. BAFF: a fundamental survival factor for B cells. Nat Rev Immunol 2002;2:465–75. [DOI] [PubMed] [Google Scholar]
- 59.International Multiple Sclerosis Genetics Consortium, Wellcome Trust Case Control Consortium 2, Sawcer S, Hellenthal G, Pirinen M. et al. Genetic risk and a primary role for cell-mediated immune mechanisms in multiple sclerosis. Nature 2011;476:214–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Mells GF, Floyd JAB, Morley KI. et al. Genome-wide association study identifies 12 new susceptibility loci for primary biliary cirrhosis. Nat Genet 2011;43:329–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Imgenberg-Kreuz J, Sandling JK, Björk A. et al. Transcription profiling of peripheral B cells in antibody-positive primary Sjögren’s syndrome reveals upregulated expression of CX3CR1 and a type I and type II interferon signature. Scand J Immunol 2018;87:e12662. [DOI] [PubMed] [Google Scholar]
- 62. Hjelmervik TOR, Petersen K, Jonassen I. et al. Gene expression profiling of minor salivary glands clearly distinguishes primary Sjögren’s syndrome patients from healthy control subjects. Arthritis Rheum 2005;52:1534–44. [DOI] [PubMed] [Google Scholar]
- 63. Aqrawi LA, Ivanchenko M, Björk A. et al. Diminished CXCR5 expression in peripheral blood of patients with Sjögren’s syndrome may relate to both genotype and salivary gland homing. Clin Exp Immunol 2018;192:259–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Adrianto I, Wen F, Templeton A. et al. Association of a functional variant downstream of TNFAIP3 with systemic lupus erythematosus. Nat Genet 2011;43:253–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Beyaert R, Heyninck K, Van Huffel S.. A20 and A20-binding proteins as cellular inhibitors of nuclear factor-κB-dependent gene expression and apoptosis. Biochem Pharmacol 2000;60:1143–51. [DOI] [PubMed] [Google Scholar]
- 66. Schubeler D. Function and information content of DNA methylation. Nature 2015;517:321–6. [DOI] [PubMed] [Google Scholar]
- 67. Du J, Johnson LM, Jacobsen SE. et al. DNA methylation pathways and their crosstalk with histone methylation. Nat Rev Mol Cell Biol 2015;16:519–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Yin H, Zhao M, Wu X. et al. Hypomethylation and overexpression of CD70 (TNFSF7) in CD4+ T cells of patients with primary Sjögren’s syndrome. J Dermatol Sci 2010;59:198–203. [DOI] [PubMed] [Google Scholar]
- 69. Yu X, Liang G, Yin H. et al. DNA hypermethylation leads to lower FOXP3 expression in CD4+ T cells of patients with primary Sjögren’s syndrome. Clin Immunol 2013;148:254–7. [DOI] [PubMed] [Google Scholar]
- 70. Gestermann N, Koutero M, Belkhir R. et al. Methylation profile of the promoter region of IRF5 in primary Sjögren’s syndrome. Eur Cytokine Netw 2012;23:166–72. [DOI] [PubMed] [Google Scholar]
- 71. Thabet Y, Le Dantec C, Ghedira I. et al. Epigenetic dysregulation in salivary glands from patients with primary Sjögren’s syndrome may be ascribed to infiltrating B cells. J Autoimmun 2013;41:175–81. [DOI] [PubMed] [Google Scholar]
- 72. Konsta OD, Le Dantec C, Charras A. et al. Defective DNA methylation in salivary gland epithelial acini from patients with Sjögren’s syndrome is associated with SSB gene expression, anti-SSB/LA detection, and lymphocyte infiltration. J Autoimmun 2016;68:30–8. [DOI] [PubMed] [Google Scholar]
- 73. Mavragani CP, Sagalovskiy I, Guo Q. et al. Expression of long interspersed nuclear element 1 retroelements and induction of type I interferon in patients with systemic autoimmune disease. Arthritis Rheumatol 2016;68:2686–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. González S, Aguilera S, Alliende C. et al. Alterations in type I hemidesmosome components suggestive of epigenetic control in the salivary glands of patients with Sjögren’s syndrome. Arthritis Rheum 2011;63:1106–15. [DOI] [PubMed] [Google Scholar]
- 75. Altorok N, Coit P, Hughes T. et al. Genome-wide DNA methylation patterns in naive CD4+ T cells from patients with primary Sjögren’s syndrome. Arthritis Rheumatol 2014;66:731–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Miceli-Richard C, Wang-Renault S-F, Boudaoud S. et al. Overlap between differentially methylated DNA regions in blood B lymphocytes and genetic at-risk loci in primary Sjögren’s syndrome. Ann Rheum Dis 2016;75:933–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Imgenberg-Kreuz J, Sandling JK, Almlöf JC. et al. Genome-wide DNA methylation analysis in multiple tissues in primary Sjögren’s syndrome reveals regulatory effects at interferon-induced genes. Ann Rheum Dis 2016;75:2029–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Brække Norheim K, Imgenberg-Kreuz J, Jonsdottir K. et al. Epigenome-wide DNA methylation patterns associated with fatigue in primary Sjögren’s syndrome. Rheumatology (Oxford) 2016;55:1074–82. [DOI] [PubMed] [Google Scholar]
- 79. Cole MB, Quach H, Quach D. et al. Epigenetic signatures of salivary gland inflammation in Sjögren’s syndrome. Arthritis Rheumatol 2016;68:2936–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Charras A, Konsta OD, Le Dantec C. et al. Cell-specific epigenome-wide DNA methylation profile in long-term cultured minor salivary gland epithelial cells from patients with Sjögren’s syndrome. Ann Rheum Dis 2017;76:625–8. [DOI] [PubMed] [Google Scholar]
- 81. Pauley KM, Stewart CM, Gauna AE. et al. Altered miR-146a expression in Sjögren’s syndrome and its functional role in innate immunity. Eur J Immunol 2011;41:2029–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Kapsogeorgou EK, Gourzi VC, Manoussakis MN. et al. Cellular microRNAs (miRNAs) and Sjögren’s syndrome: candidate regulators of autoimmune response and autoantigen expression. J Autoimmun 2011;37:129–35. [DOI] [PubMed] [Google Scholar]
- 83. Zilahi E, Tarr T, Papp G. et al. Increased microRNA-146a/b, TRAF6 gene and decreased IRAK1 gene expressions in the peripheral mononuclear cells of patients with Sjögren’s syndrome. Immunol Lett 2012;141:165–8. [DOI] [PubMed] [Google Scholar]
- 84. Shi H, Zheng L-Y, Zhang P. et al. miR-146a and miR-155 expression in PBMCs from patients with Sjögren’s syndrome. J Oral Pathol Med 2014;43:792–7. [DOI] [PubMed] [Google Scholar]
- 85. Gourzi VC, Kapsogeorgou EK, Kyriakidis NC. et al. Study of microRNAs (miRNAs) that are predicted to target the autoantigens Ro/SSA and La/SSB in primary Sjögren’s syndrome. Clin Exp Immunol 2015;182:14–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Chen J-Q, Zilahi E, Papp G. et al. Simultaneously increased expression of microRNA-155 and suppressor of cytokine signaling 1 (SOCS1) gene in the peripheral blood mononuclear cells of patients with primary Sjögren’s syndrome. Int J Rheum Dis 2017;20:609–13. [DOI] [PubMed] [Google Scholar]
- 87. Kapsogeorgou EK, Papageorgiou A, Protogerou AD. et al. Low miR200b-5p levels in minor salivary glands: a novel molecular marker predicting lymphoma development in patients with Sjögren’s syndrome. Ann Rheum Dis 2018;77:1200–7. [DOI] [PubMed] [Google Scholar]
- 88. Wang Y, Zhang G, Zhang L. et al. Decreased microRNA-181a and -16 expression levels in the labial salivary glands of Sjögren syndrome patients. Exp Ther Med 2018;15:426–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Lopes AP, Hillen MR, Chouri E. et al. Circulating small non-coding RNAs reflect IFN status and B cell hyperactivity in patients with primary Sjögren’s syndrome. PLoS One 2018;13:e0193157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Peng L, Ma W, Yi F. et al. MicroRNA profiling in Chinese patients with primary Sjögren syndrome reveals elevated miRNA-181a in peripheral blood mononuclear cells. J Rheumatol 2014;41:2208–13. [DOI] [PubMed] [Google Scholar]
- 91. Williams AEG, Choi K, Chan AL. et al. Sjögren’s syndrome-associated microRNAs in CD14+ monocytes unveils targeted TGFβ signaling. Arthritis Res Ther 2016;18:95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Wang-Renault S-F, Boudaoud S, Nocturne G. et al. Deregulation of microRNA expression in purified T and B lymphocytes from patients with primary Sjögren’s syndrome. Ann Rheum Dis 2018;77:133–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Alevizos I, Alexander S, Turner RJ. et al. MicroRNA expression profiles as biomarkers of minor salivary gland inflammation and dysfunction in Sjögren’s syndrome. Arthritis Rheum 2011;63:535–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Chen J-Q, Papp G, Póliska S. et al. MicroRNA expression profiles identify disease-specific alterations in systemic lupus erythematosus and primary Sjögren’s syndrome. PLoS One 2017;12: e0174585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Tandon M, Gallo A, Jang S-I. et al. Deep sequencing of short RNAs reveals novel microRNAs in minor salivary glands of patients with Sjögren’s syndrome. Oral Dis 2012;18:127–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Bibikova M, Barnes B, Tsan C. et al. High density DNA methylation array with single CpG site resolution. Genomics 2011;98:288–95. [DOI] [PubMed] [Google Scholar]
- 97. Seror R, Ravaud P, Bowman SJ. et al. EULAR Sjögren’s syndrome disease activity index: development of a consensus systemic disease activity index for primary Sjögren’s syndrome. Ann Rheum Dis 2010;69:1103–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Wildenberg ME, van Helden‐Meeuwsen CG, van de Merwe JP. et al. Systemic increase in type I interferon activity in Sjögren’s syndrome: a putative role for plasmacytoid dendritic cells. Eur J Immunol 2008;38:2024–33. [DOI] [PubMed] [Google Scholar]
- 99. Maria NI, Steenwijk EC, Ijpma AS. et al. Contrasting expression pattern of RNA-sensing receptors TLR7, RIG-I and MDA5 in interferon-positive and interferon-negative patients with primary Sjögren’s syndrome. Ann Rheum Dis 2017;76:721–30. [DOI] [PubMed] [Google Scholar]
- 100. Brauner S, Folkersen L, Kvarnström M. et al. H1N1 vaccination in Sjögren’s syndrome triggers polyclonal B cell activation and promotes autoantibody production. Ann Rheum Dis 2017;76:1755–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Moutsopoulos HM. Sjögren’s syndrome: autoimmune epithelitis. Clin Immunol Immunopathol 1994;72:162–5. [DOI] [PubMed] [Google Scholar]
- 102. Smolle M, Workman JL.. Transcription-associated histone modifications and cryptic transcription. Biochim Biophys Acta 2013;1829:84–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Choudhary C, Weinert BT, Nishida Y. et al. The growing landscape of lysine acetylation links metabolism and cell signalling. Nat Rev Mol Cell Biol 2014;15:536–50. [DOI] [PubMed] [Google Scholar]
- 104. Konsta OD, Le Dantec C, Charras A. et al. An in silico approach reveals associations between genetic and epigenetic factors within regulatory elements in B cells from primary Sjögren’s syndrome patients. Front Immunol 2015;6:437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Chen J-Q, Papp G, Szodoray P. et al. The role of microRNAs in the pathogenesis of autoimmune diseases. Autoimmun Rev 2016;15:1171–80. [DOI] [PubMed] [Google Scholar]
- 106. Taganov KD, Boldin MP, Chang K-J. et al. NF-κB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc Natl Acad Sci USA 2006;103:12481–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Li L, Chen XP, Li YJ.. MicroRNA-146a and human disease. Scand J Immunol 2010;71:227–31. [DOI] [PubMed] [Google Scholar]
- 108. Bodewes ILA, Al-Ali S, van Helden-Meeuwsen CG. et al. Systemic interferon type I and type II signatures in primary Sjögren’s syndrome reveal differences in biological disease activity. Rheumatology (Oxford) 2018;57:921–30. [DOI] [PubMed] [Google Scholar]