Abstract
Background
SARS-CoV-2 attacks hemoglobin through its structural protein ORF3a, dissociating the iron from the heme, as iron is necessary by cell machinery for virus replication. In this process protoporphyrin (PpIX) is released.
Methods
The decrease in the hemoglobin levels observed in patients with Covid-19 is frequently accompanied by an increase in PpIX levels. This evidence was confirmed by the quantification of PpIX by high-performance liquid chromatography (HPLC). PpIX emission is observed in its two characteristic bands at approximately 635 nm and 705 nm.
Results
This paper searches to understand the role of heme and PpIX inside the cells. Perspectives on the use of PpIX fluorescence as a sensor to monitor the presence of SARS-CoV-2 in the tissue, blood, urine, or feces to map the evolution and severity of the disease or to monitor the response of the Covid-19 treatment modalities were described.
Conclusion
Fluorescence spectroscopy could be adopted as an excellent diagnostic technique for Covid-19, of low cost and high sensitivity. This method can potentially be used as a marker to monitor the response to the treatments. Photodynamic and sonodynamic therapies using the endogenous PpIX increased in the acute phase of the disease, could be employed for Covid-19 treatment.
Keywords: Covid-19, Hemoglobin, Protoporphyrin IX, Fluorescence
1. Introduction
Methods based on fluorescence are those in which the excitation of a molecule is obtained through photon absorption. In this case, the molecule is promoted to an electronic state of higher energy, which returns to the fundamental state accompanied by the emission of electromagnetic radiation [1].
We can mention some important properties of fluorescence as high sensitivity and low detection limits. Fluorescent measurements usually present low background signals. Fluorophores show characteristic wavelengths of excitation or emission that guarantee selectivity. The instrumental setup is simple with low maintenance and analysis costs when compared with other analytical methods [1].
Fluorescence spectroscopy is particularly important for disease diagnosis [[2], [3], [4], [5], [6], [7], [8], [9], [10]]. The fluorescence emission from different natural fluorophores in biological tissues is obtained by exciting them with ultraviolet or visible light. Examples of tissue fluorophores' contribution to autofluorescence are nucleotides and polynucleotides, nicotinamide adenine dinucleotide (NADH), flavin dinucleotide (FAD), elastin, collagen, tryptophan, and porphyrins [11].
The two most important forms of diagnostic testing available for SARS-CoV-2 are molecular and serological tests. Among those, the serum neutralization assay stands out as the gold standard for evaluation of the effectiveness of neutralizing antibodies (NAbs) against viral infections. Some publications have mentioned the fluorescence method to detect Covid-19. The RT-PCR (reverse transcriptase-polymerase chain reaction) determination of the SARs-CoV-2 is possible by applying an immunochromatographic fluorescence assay [12]. Froggatt et al. developed a fluorescence assay based on a GFP-derived protein [13]. Nars et al. used l-Serine doped QDs for fast detection of Covid-19 [14]. Guo et al. applied quantum dot nanobeads and magnetic Fe3O4 nanospheres as a highly sensitive fluorescence-linked immunosorbent assay for the determination of human IgG in serum [15]. Plasmonic biosensing schemes for virus detection were also described [16]. In contrast, our paper is specifically directed to the virus determination via porphyrin fluorescence.
The protoporphyrin IX (PpIX) is composed of four pyrrolic rings linked by methylene bridges. Its absorption spectrum has five bands: the most intense band, the Soret band, is in the region of 400 nm, and four extra bands, known as Q bands, which comprise the region between 450 and 750 nm. The PpIX emission is observed in its two characteristic bands at approximately 635 nm and 705 nm [17]. The PpIX tetrapyrrole structure enables chelation with transition metals, for example, iron and zinc to form metalloporphyrins, which perform a variety of biologic functions. Red blood cell zinc protoporphyrin IX (Zn-PpIX) provides a functional measurement of iron deficiency. When iron supply decreased hemoglobin production is restricted [18]. Incrementing of zinc PpIX formation and its fluorescence peak around 590 nm indicates iron deficiency and the development of anemia [18].
In general, fast proliferating cells may accumulate more porphyrins preferentially. Besides porphyria patients, cancer, and atherosclerosis patients present increased PpIX concentration in tissues and blood, and PpIX fluorescence can be applied for these diseases’ diagnosis [2,3,19].
This review is going to describe the role of hemoglobin, heme, iron, and PpIX inside the cells. We will also evaluate if PpIX fluorescence could be used for the diagnosis of Covid-19 since an abnormal phenomenon related to hemoglobin dysfunction was observed in the patients with this disease. According to the best of our knowledge, there are no further published or ongoing studies about the link between SARS-CoV-2 and PpIX fluorescence that could become a biosensor of SARS-CoV-2 infection. Therapy approaches using PpIX are going to be described.
2. Covid-19 and red blood cells
Covid-19 is a highly transmitted acute infectious disease caused by the SARS-CoV-2 coronavirus. The viruses are unable to replicate themselves, and it is, therefore, essential to parasitize a cell to replicate themselves. Covid-19 patients with induced pneumonia present as symptoms: fever, dry cough, dyspnea, fatigue, headache, loss of taste or smell, sore throat, and tomography revealing ground-glass opacity in the lungs [20]. This pneumonia was first discovered in December 2019 in Hubei Province, China [20]. In April 2021 more than 141 million people have been infected worldwide and more than 3,000,000 have died.
Viral infections initiate with local invasion of an epithelial or mucosal barrier. Once the virus overcomes the early mechanical barriers, such as cilia, mucus, or skin integrity, it infects the target cell. A typical viral life cycle involves the 1) entrance of the virus into the host cell; 2) translation of viral proteins; 3) replication of the viral genome; 4) assembly of viral particles, and 5) release of the mature virions into the extracellular environment. Once attached the virus penetrates the cell or it fuses itself with the cell membrane, receptor-mediated endocytosis, or non-clathrin-mediated endocytosis [21]. During uncoating, replication, and assembly, the viruses attach to the host cell and incorporate their genetic material into it inducing the cell machinery to replicate the exogenous nucleic acids. During the maturation, the created viruses are released from the host cell, either by causing the cell to break apart waiting for the cell to die or by budding off through the cell membrane.
The SARS-CoV-2 is an enveloped virus of approximately 60–140 nm in diameter [22]. The nucleic acid of SARS-CoV-2 is a positive-stranded RNA. Its structural proteins include spike protein (S), an envelope protein (E), membrane protein (M), and nucleocapsid phosphoprotein. Transcribed non-structural proteins include orf1ab, ORF3a, ORF6, ORF7a, ORF10, and ORF8. Spike proteins, ORF8 and ORF3a proteins are significantly different from other known SARS-like coronaviruses.
The spike protein consists of an extracellular N-terminal, a transmembrane domain (TM) anchored in the viral membrane, and a short tail intracellular C-terminal [23]. The spicules are coated with polysaccharide molecules producing a camouflage, which aids it to avoid the host immune system surveillance during the invasion [24]. A positively charged area, a polybasic cleavage site, was recently discovered, which is located at 10 nm from the exact point that the spicule uses to connect to cells [25]. This area of protein allows a strong bond between the virus and the cellular receptors, which are negatively charged.
When S protein binds to the receptor a protease serine 2 (TMPRSS2) located on the host cell membrane promotes the virus invasion into the cell. SARS-CoV-2 attaches itself to the receptor ACE2 and viral entry-associated protease TMPRSS2 is highly expressed in the nasal goblet and ciliated cells. High ACE2 expression was identified in type II alveolar cells (AT2) of the lungs, upper esophagus and stratified epithelial cells, absorptive enterocytes from ileum and colon, myocardial cells, kidney proximal tubule cells, and bladder urothelial cells [26]. Once the virus enters the cell the viral RNA is released into the cytoplasm where it will be translated and replicated, or it will reach the cell nucleus [27].
Viruses depend on host cell survival during replication and the cellular metabolism requires iron as a nutrient [28]. Iron is necessary for many fundamental enzymatic and non-enzymatic reactions and diverse physiological processes, such as mitochondrial function including ATP generation, DNA/RNA synthesis, and repair [27].
Several publications report that SARS-CoV-2 attacks hemoglobin and attacks the normal metabolic pathway of heme and tissue iron overload [[29], [30], [31], [32], [33], [34], [35]]. A study with 1210 Covid-19 patients showed significantly low hemoglobin levels (<100 g/L) [36,37]. The values of serum ferritin index, erythrocyte sedimentation rate, C-reactive protein, albumin, and lactate dehydrogenase of many patients have increased significantly [38]. Decreased heme production dampens repression of aminolevulinic acid synthase, and thereby increases the production of heme precursors. Hence, it leads to porphyrin accumulation. Excess porphyrins in red blood cells can precipitate cell lysis and it can be responsible for hemolytic anemia development [30].
Cavezzi et al. reported pathologic metabolic pathways deriving from hemoglobin denaturation, and iron metabolism dysregulation [33]. This interaction resulted in a hemoglobin functioning quantity decrease, elevated serum ferritin levels, free toxic circulating heme, low level of oxygen in the blood, and systemic hypoxia, reduction of nitric oxide, coagulation activation, ferroptosis with oxidative stress and lipoperoxidation, mitochondrial degeneration, and apoptosis.
Cavezzi et al. suggested that SARS-CoV-2 can have a tropism for blood cells [33]. They proposed that the interaction with hemoglobin molecule occurs through CD147, CD26, and other receptors located on erythrocyte and/or blood cell precursors and that hepcidin-mimetic action of a viral spike protein, inducing ferroportin blockage [33]. SARS-CoV-2 could induce hemolysis and/or form a complex with the released heme, generating a quote of dysfunctional hemoglobin, with reduced oxygen and CO2 transport [33].
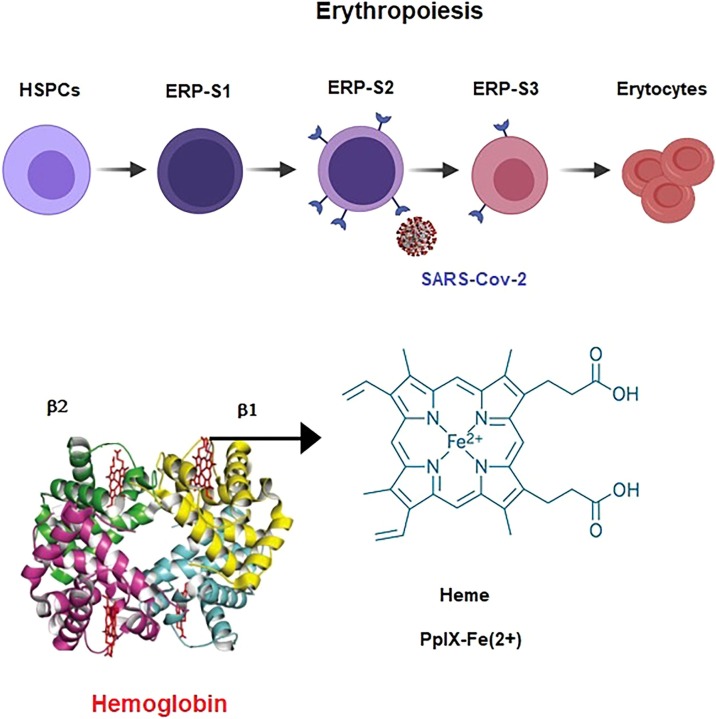
Ropa et al. reported that hematopoietic stem cells (HSCs) from cord blood express ACE2 and the exposure to the S protein can reduce their functionality [39]. Recently Cosic et al., found that the red blood cells (RBC) Band3 surface proteins have the same resonant recognition model characteristics as ACE2 receptors. They proposed that hypoxia in severe cases of COVID-19 is caused by SARS-CoV-2 interacting with the RBC Band3 surface protein and hampering RBC oxygen transport function [40]. Bernards et al. reported an aberrant increase of erythroid progenitors in circulation [41]. Erythrocyte lysis may occur in COVID-19 due to the high oxidative environment [42]. Encabo et al. studied hematopoietic stem/progenitor cell (HSPCs) and different erythroid progenitor populations to assess if they can be infected by SARS-CoV-2 [43] (Fig. 1 ). They show the first evidence of direct infection of specific erythroid progenitors. They reported the high ability of the virus to infect ERP-S2, the most vulnerable erythroid progenitor population and show that the decline in hemoglobin levels coincides with an aberrant increase of nucleated red blood cells in circulation. Shahbaz et al. reported that the nucleated red blood cells correspondent to ERP-S3 cells can be infected by SARS-CoV-2 and that the infection induces the immunosuppressive capacity of these cells [44].
Fig. 1.
Direct infection of specific erythroid progenitors, ERP-S2 and ERP-S3 by SARS-CoV-2 [43]. Hemoglobin structure with the alpha chains (α1, α2), the beta chains (β1, β2), and the four heme groups. On the right the structure of the iron protoporphyrin IX subunit of heme B.
3. Hemoglobin and Iron
Cellular processes such as DNA synthesis and the molecules ATP generation require iron [45]. Viruses depend on iron ions to replicate within living host cells [28]. The uptake of iron pathways free iron and from heme are associated with their proliferation and virulence and, consequently, their pathogenicity [46].
In the human body, iron exists in complex forms of hemoproteins as heme compounds such as hemoglobin or myoglobin, heme enzymes, or non-heme compounds as flavin-iron enzymes, iron-sulfur clusters, transferring, and ferritin [47].
More than two‐thirds of the human body’s iron is localized in the hemoglobin of developing erythroid precursors and mature RBCs. Iron is also stored in liver cells and macrophages bound to ferritin. A smaller fraction is bound to myoglobin in muscle tissue and a variety of enzymes involved in oxidative metabolism and many other cell functions [47].
Iron is important for hemoglobin and myoglobin synthesis, also iron formation depends on enzymes involved in oxidative phosphorylation, which is the metabolic pathway that converts nutrients to energy [47]. Critical enzymes in the DNA metabolism, including multiple DNA repair enzymes and ribonucleotide reductase, take iron as an indispensable cofactor to function [48].
A balance between iron uptake, transport, storage, and utilization is required to maintain iron homeostasis. Non-heme iron compounds are transported across the apical membrane of the intestinal enterocyte by divalent metal transporter 1 (DMT1) and are exported into circulation via ferroportin 1 (FPN1). The iron homeostasis regulator is the liver-derived peptide hepcidin [49]. Fe-transferrin (Tf) complex transports iron to other cells and tissues through the blood circulation in the body. Tf binds to transferrin receptor 1 (TfR1) on the cell membrane, and the TfR1-TfFe complex is then endocytosed by the cell, where the iron content is released. Free iron ion enters the mitochondria for its utilization in metabolic processes, such as the synthesis of hemoglobin. Excess iron is transported out of the cell by iron efflux protein ferroportin 1 (FPN1) located at the cell membrane [50]. When cellular iron concentrations are low the iron regulatory proteins (IRPs) are in their mRNA-binding conformations. The binding of IRPs to the untranslated ferritin mRNA regions (UTRs) blocks the translation ensuring that ferritin is produced when iron storage is not required.
Hemoglobin consists of four subunits, 2-α and 2-β, and each subunit presents a heme as observed in Fig. 1. Heme synthesis occurs in both the cytosol and the mitochondria with the formation of δ- aminolevulinic acid (ALA) [51]. It begins with glycine and succinyl coenzyme A and ends with the production of a PpIX ring. The insertion of Fe2+ into PpIX catalyzed by ferrochelatase in the mitochondria determines the final step of the heme biosynthetic pathway. Heme regulates a wide spectrum of gene expression, cell differentiation, proliferation, and immune stimulation [52]. In non-erythroid cells, the heme synthesis depends on the ALA production, and in erythroid cells, it depends on the availability of iron for ferrochelatase.
Hemoglobin (Hb) is expressed by both erythroid and non-erythroid cells [53]. Hb expression has been observed in different cells including macrophages, alveolar, lungs, hepatocytes, mesangial, retinocytes, endometrium, cervix, vaginal, and neuronal cells [[53], [54], [55]]. In erythroid cells in the blood, Hb carries oxygen from the lungs throughout the body where it is used in aerobic metabolism pathways. In some non-erythroid cells, Hb expression is upregulated in response to hypoxia as compensation for the increased oxygen demand [54]. Hb can recognize pathogens thereby reducing the risk and/or severity of infections, and this is the possible mechanism by which Hb exhibits antimicrobial and antioxidative functions.
Grek et al. discovered that hemoglobin RNA and protein are expressed in several alveolar cell lines. Including A549 and MLE-15 as well as in primary AT2 cells purified from murine and rat lungs. They demonstrated that hemoglobin mRNA is up-regulated during hypoxic exposure in the murine AT2 and MLE-15 cell lines [54].
Heme is a hydrophobic molecule and is insoluble in the aqueous cellular milieu. Free heme is toxic to biological macromolecules. High levels of free heme result in pro-inflammatory and proliferative effects [56]. Free heme causes inflammation and acute lung injury [57]. In pathologies such as hemolytic diseases, sepsis, renal injuries, malaria, and atherosclerosis, large quantities of hemoproteins are released into the plasma.
Hemoglobin is degraded into globin and heme molecules, which occurs when the tetrapyrrole ring of porphyrin is opened by the action of the enzyme heme oxygenase breaking the methine bridge between pyrroles I and II. This reaction releases Fe2+, which is quickly sequestered by ferritin, carbon monoxide (CO), and biliverdin (BV), and further converted into bilirubin (BR) through the enzyme biliverdin reductase (BVR) [52]. CO, BV/BR exert important cytoprotective and anti-inflammatory effects during oxidative stress and inflammation. CO binds to hemoglobin and interferes in the oxygen-carrying capacity of the blood leading to tissue hypoxia [58]. When BR production is increased it accumulates in the blood. BR plays a crucial role in innate immunity affecting the immune system depending on the complement cascade it interrupts the C1 complex to antibodies [59]. The BV function relies on the increasing level of heme oxygenase (HO-1) by BVR in the lungs. The blockage of HO-1 activity results in loss of BV inhibitory effects on lipopolysaccharide LPS-induced lung injury [60].
4. Covid-19 and PpIX
San Juan et al. quantified the total porphyrin content in the serum of 134 Covid-19 patients in the acute phase of the disease (confirmed by positive rt-PCR testing) through high-performance liquid chromatography (HPLC). They reported an abnormal accumulation of porphyrins in association with severe Covid‐19 [61]. Porphyrins possessing free iron, and other ROS/oxidants generated from the ensuing inflammation create a toxic milieu that causes protein aggregation [62]. Therefore, protein aggregation provides a potential mechanism for cell and tissue damages. Thus, this could be causing neurological problems attributed to Covid-19.
The virus’s invasion mechanism via non-structural proteins predicted by Wenzhong et al. could occur via erythroid cells as well as non-erythroid cells. They made a computational analysis to compare the biological roles of specific proteins belonging to the novel coronavirus [63]. The conserved domain analysis showed that the envelope protein (E), nucleocapsid phosphoprotein (N) and ORF3a had heme linked sites, which is Arg134 of ORF3a, Cys44 of E, Ile304 of N were the heme-iron linked site, respectively. ORF3a's role would be the iron heme dissociation to form porphyrin. The docking results showed that orf1ab, ORF10, and ORF3a proteins coordinate the attack of the hemoglobin’s β1 chain and some structural and non-structural viral proteins bind to the porphyrins. According to them, the binding part of the orf1ab protein acts as a clip, that is bounded to the α chain and attacks the β chain. This attack causes configurational changes in their α and β chains. The ORF3 attacks the β chain and exposes heme. ORF10 attaches to the β chain and directly impacts the iron atoms on the heme of the β chain. The heme is dissociated into porphyrin, and orf1ab finally captures the porphyrin. ORF8 and surface glycoproteins could combine themselves with the porphyrin to become a complex. Facilitating the entrance of the virus into the cell.
We hypothesize that SARS-CoV-2, that depends on iron to replicate within living host cells, obtains this iron at the expense of hemoglobin and heme, which once disrupted, release PpIX.
The porphyrin is degraded into bilirubin and excreted from the bile duct. Porphyrins that leave the cells and appear in blood, urine, and feces are mostly oxidized and are fluorescent. To date, there seem to be no further published or ongoing studies about the link between SARS-CoV-2 and PpIX fluorescence.
PpIX could represent not only a sensitive indirect biosensor of SARS-CoV-2 infection but also a specific and low-cost diagnostic test.
5. Covid-19: PpIX diagnostic marker
COVID-19 does lead to increased porphyrin release. Thus, the PpIX fluorescence analysis from blood, urine, and feces samples can yield information about the presence of SARS-CoV-2 in the body. Through the PpIX spectroscopic properties, it is possible to monitor and quantify its concentration [2].
A variety of porphyrins are presented in urine or feces, which could be easily collected at home. In ordinary light, normal urine is amber, or it shows a pinkish amber tinge if concentrated. Normal urine excited with UV light around 400 nm shows a greenish-blue or greenish-yellow or pinkish-yellow fluorescence if concentrated. Urine containing an increased concentration of porphyrins may be pink, red, or brown. When excited around 400 nm, urine containing porphyrins presents red fluorescence [64]. So, patients with Covid-19 must present urine with red fluorescence.
Hennig et al. measured the red blood cell zinc protoporphyrin at ∼589 nm in the microcirculation of the lower lip to established an iron status indicator [18]. The iron deficiency detection sensitivity and specificity were 97 % and 90 %, respectively. They concluded that the fluorescence method potentially provided a rapid, easy-to-use means for point-of-care screening for iron deficiency in resource-limited settings lacking laboratory infrastructure. A similar method could be employed to detect Covid-19.
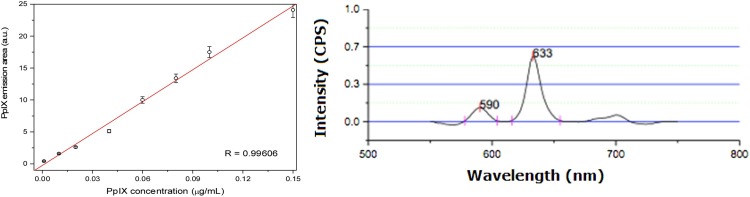
In Fig. 2 a it is shown a calibration curve obtained with synthetic PpIX fluorescence (Sigma). This figure shows that fluorescence spectroscopy can distinguish low PpIX concentrations, and this method should present good sensitivity. For quantitative analysis, to monitor the progression of Covid-19, PpIX could be easily extracted from blood, or feces using acetone (3 parts of acetone/1 part of the analyte), centrifugation, and discarding the cell pellet formed in the bottom of the tube [65,66].
Fig. 2.
a) Calibration curve of synthetic protoporphyrin IX prepared in acetone (Sigma). b) PpIX was extracted from blood samples from thirty people without Covid-19 and blood disorders. PpIX fluorescence was obtained under excitation at 410 nm and the presented spectrum is an average of the thirty spectra.
Fig. 2b shows the average fluorescence spectra of PpIX extracted with acetone from blood samples from 30 people without Covid-19 (Reference values for blood protoporphyrin of healthy people: 0.16 to 0.60 μg/mL or 0.28–1.07 μmol/L). In this spectrum, it is possible to see Zn-PpIX (∼590 nm) and PpIX (∼633 nm) peaks. As an abnormal accumulation of porphyrins is expected in a patient with Covid-19 [62], increased blood PpIX concentration is predictable for contaminated people. The fluorescence method could contribute to monitoring and evaluating the severity or evolution of the disease. Even asymptomatic patients should present abnormal PpIX levels. Covid-19 patients with anemia, more susceptible to death should present an increment in Zn-PpIX/PpIX rate [67].
Mobile platforms allow online and on-site diagnostic transitioning from laboratory-based trials. Since the introduction of smartphones, hundreds of millions of devices possessing high pixel count images operating under low light conditions have been sold. Such cameras can be employed as a sensor to perform functions equivalent to much larger and more expensive laboratory instruments. The smartphone fluorimeter can perform tests with a good detection limit which could be applicable in medical clinics [[68], [69], [70]].
Studies are necessary to determine the method specificity because other diseases as porphyria, cancer, or atherosclerosis can lead to an increase in porphyrin level in blood or body fluids. Several publications demonstrated that highly multiplying cells, like cancer cells [4,5], show an intracellular accumulation of PpIX [71,72]. In these cases, the accumulation of PpIX in cells is affected by multiple biological factors, which include 5-ALA uptake [73], ABC transporters, such as ABCG2, that excrete PpIX from cells, enzymes in the heme biosynthesis pathway, and the intracellular Fe2+ content [71].
So, it is important to point out that the PpIX fluorescence method should be employed preferably in patients with less than 40 years old to avoid false-positive results, but further investigations are necessary to assess this role. However cancer incidence is much less, and it has its well-defined symptoms with no overlap with that of Covid-19 cases.
6. Covid-19: PpIX-treatment perspectives
RNA-type microorganisms are more easily photo-inactivated than their DNA-type counterparts. This suggests that SARS-CoV-2, which is an enveloped RNA-type virus, can be easily neutralized by photodynamic therapy (PDT) [74]. Extracorporeal photoinactivation of coronaviruses and other clinically relevant pathogens using methylene blue (MB)-mediated PDT has been reported [74,75]. PDT has been used for decades in lung cancer treatment and in laryngeal papilloma treatment, which was a success [76]. More recently, Schikora et al. reported successful use of PDT to disinfect oral and nasal cavity of patients in the early stages of COVID-19 infection [77].
Porphyrins are natural sensitizers in photodynamic and sonodynamic therapies (PDT and SDT) and innumerous publications applying these therapies to diseases like cancer or atherosclerosis, or infections were described [[78], [79], [80], [81], [82], [83]]. Both light and ultrasound can excite porphyrins from the ground state (S0) to the excited singlet state (S1). Porphyrins can then either relax back to S0 state emitting light or go to the excited triplet state (T1) by intersystem crossing. The energy of porphyrins in the T1 state can either be relaxed by phosphorescence or be transferred to oxygen molecules through type I and type II reactions. In type I reaction an electron transfer process produces free radicals, which interact with water or oxygen molecules leading to the production of hydroxyl radicals (OH) or superoxide anions (O2 ·−). In the type II reaction, an energy transfer process between the T1 state of porphyrins with 3O2 results in the formation of highly cytotoxic singlet oxygen (1O2). The reactive oxygen species (ROS) can interact with proteins, lipids, and nucleic acids, causing their oxidation and, consequently, damage to cells, tissues, and microorganisms as SARS-CoV-2 [19]. The use of Photofrin ®, a porphyrin derivative, as a photosensitizer could help to mitigate the COVID-19 [19] and prevent severe stages, however, there is no data available on the photodynamic inactivation. Furthermore, porphyrins when excited by light, like sunlight, with sufficient intensity could effectively destroy SARS-CoV-2 by photobiomodulation (PBM). In the case of photobiomodulation, ROS are present at low concentrations, below that required for cytotoxicity, and have a wide range of positive stimulatory effects on the cell. PMB employing low levels of red light could be an alternative to acute lung injury, for example [84]. Nevertheless, to date, no experimental data was supporting the influence of PBM on COVID-19.
PDT is limited to local effects and COVID-19 is a systemic disease. The optical radiation penetration into the biological tissue with wavelengths around 400 nm, which is the maximum porphyrins absorption band, is around 250 μm [85]. As a result, only a superficial effect can be obtained. Nevertheless, an endoscopic fiber could be applied for the delivery of light into deeper tissues. Additionally, sonodynamic therapy (SDT) could be employed to eliminate SARS-CoV-2 in deeper regions as the lungs [86]. SDT involves the exposure of the sensitized tissues with relatively low-intensity ultrasound (US). This suggests that sonodynamic therapy may find wider clinical application particularly for the non-invasive treatment of difficult accessible lesions.
The cavitation effect of ultrasound is responsible for the SDT mechanism [79]. The acoustic cavitation promotes the formation, growth, and collapse of bubbles during US wave propagation in liquids. The bubbles explosion lead to sonoluminescence and endogenous PpIX activation [87]. Recently it was observed that the PpIX was activated from the ground to an excited state induced by methyl aminolevulinate gold nanoparticles (MALA:AuNPs) and exposed to a 1 MHz and 1 W/cm2 ultrasound [88]. On returning to the ground state it released energy, which was transferred to oxygen to produce ROS, such as singlet oxygen and free radicals. So, both nanoparticles and PpIX were responsible for macrophages' total reduction.
Pre-clinical and clinical studies are planned to examine the early feasibility, safety, and efficacy of the proposed approaches. In this future prospective work, we must avoid interference from cancer patients.
7. Conclusions
The early diagnosis and treatment of Covid-19 can drastically improve the prognosis of this disease. SARS-CoV- 2 attacks hemoglobin through its structural protein ORF3a dissociating the iron from the heme molecules since iron is necessary for the virus replication. In this process, PpIX is released. In this context, PpIX fluorescence could be employed on the Covid-19 diagnosis or potentially be used as a marker for the evaluation of the patient's response to the treatment. Furthermore, from our perspective, photobiomodulation, photodynamic and sonodynamic therapies taking advantage of the increased endogenous PpIX concentration in the acute phase of the disease could be employed as an additional option for the treatment of Covid-19 patients. A mechanism to prevent virus replication by interrupting hemoglobin rupture could serve as an effective way to help combat the disease.
Ethical approval
Not required/not applicable.
Funding
Fundação de Amparo à Pesquisa do Estado de São Paulo, FAPESP Grant number 2017/23686-6.
Author’s contribution
L.C.C. conceived the idea presented. All authors discussed the ideas and contributed to the final manuscript.
Declaration of Competing Interest
The authors report no declarations of interest.
References
- 1.Lakowicz J.R. 3rd edition. Springer; New York: 2006. Principles of Fluorescence Spectroscopy. [Google Scholar]
- 2.da Silva M.N., Sicchieri L.B., Silva F.R.D., Andrade M.F., Courrol L.C. Liquid biopsy of atherosclerosis using protoporphyrin IX as a biomarker. Analyst. 2014;139(6):1383–1388. doi: 10.1039/c3an01945d. [DOI] [PubMed] [Google Scholar]
- 3.Gotardelo D.R., Courrol L.C., Bellini M.H., Silva F.R.D., Soares C.R.J. Porphyrins are increased in the faeces of patients with prostate cancer: a case-control study. BMC Cancer. 2018;18 doi: 10.1186/s12885-018-5030-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Masilamani V., Al-Zhrani K., Al-Salhi M., Al-Diab A., Al-Ageily M. Cancer diagnosis by autofluorescence of blood components. J. Lumin. 2004;109(3–4):143–154. [Google Scholar]
- 5.Masilamani V., Alsalhi M.S., Vijmasi T., Govindarajan K., Rathan Rai R., Atif M., Prasad S., Aldwayyan A.S. Fluorescence spectra of blood and urine for cervical cancer detection. J. Biomed. Opt. 2012;17(9) doi: 10.1117/1.JBO.17.9.098001. 98001-1. [DOI] [PubMed] [Google Scholar]
- 6.Bae S.J., Lee D.S., Berezin V., Kang U., Lee K.H. Multispectral autofluorescence imaging for detection of cervical lesions: a preclinical study. J. Obstet. Gynaecol. Res. 2016;42(12):1846–1853. doi: 10.1111/jog.13101. [DOI] [PubMed] [Google Scholar]
- 7.Bao N., Jagadeesan B., Bhunia A.K., Yao Y., Lu C. Quantification of bacterial cells based on autofluorescence on a microfluidic platform. J. Chromatogr. A. 2008;1181(1–2):153–158. doi: 10.1016/j.chroma.2007.12.048. [DOI] [PubMed] [Google Scholar]
- 8.Blanco K.C., Moriyama L.T., Inada N.M., Salvio A.G., Menezes P.F.C., Leite E.J.S., Kurachi C., Bagnato V.S. Fluorescence guided PDT for optimization of the outcome of skin cancer treatment. Front. Phys. 2015;3 [Google Scholar]
- 9.Buchalla W., Lennon A.M., Attin T. Fluorescence spectroscopy of dental calculus. J. Periodont. Res. 2004;39(5):327–332. doi: 10.1111/j.1600-0765.2004.00747.x. [DOI] [PubMed] [Google Scholar]
- 10.Silva F.R.O., Nabeshima C.T., Bellini M.H., Schor N., Vieira N.D., Jr., Courrol L.C. Study of protoporphyrin IX elimination by body excreta: a new noninvasive cancer diagnostic method? J. Fluoresc. 2013;23(1):131–135. doi: 10.1007/s10895-012-1126-1. [DOI] [PubMed] [Google Scholar]
- 11.Bachmann L., Zezell D.M., Ribeiro A.D., Gomes L., Ito A.S. Fluorescence spectroscopy of biological tissues - a review. Appl. Spectrosc. Rev. 2006;41(6):575–590. [Google Scholar]
- 12.Manigandan S., Wu M.T., Ponnusamy V.K., Raghavendra V.B., Pugazhendhi A., Brindhadevi K. A systematic review on recent trends in transmission, diagnosis, prevention and imaging features of COVID-19. Process. Biochem. 2020;98:233–240. doi: 10.1016/j.procbio.2020.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Froggatt H.M., Heaton B.E., Heaton N.S. Development of a fluorescence-based, high-throughput SARS-CoV-2 3CL(p)(ro) reporter assay. J. Virol. 2020;94(22):9. doi: 10.1128/JVI.01265-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nasr S., Hidouri T., Zouidi F. New strategy against COVID-19: L-Serine doped QDs for fast detection of COVID-19 and blocking of S-Protein. Ecs J. Solid State Sci. Technol. 2020;9(10):10. [Google Scholar]
- 15.Guo J.P., Wang Y.M., Niu S.S., Li H.P., Tian Y.M., Yu S.C., Yu F., Wu Y.J., Liu L.E. Highly sensitive fluorescence-linked immunosorbent assay for the determination of human IgG in serum using quantum dot nanobeads and magnetic Fe3O4 nanospheres. ACS Omega. 2020;5(36):23229–23236. doi: 10.1021/acsomega.0c02987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mauriz E. Recent progress in plasmonic biosensing schemes for virus detection. Sensors. 2020;20(17) doi: 10.3390/s20174745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sachar M., Anderson K.E., Ma X.C. Protoporphyrin IX: the good, the bad, and the ugly. J. Pharmacol. Exp. Ther. 2016;356(2):267–275. doi: 10.1124/jpet.115.228130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hennig G., Homann C., Teksan I., Hasbargen U., Hasmuller S., Holdt L.M., Khaled N., Sroka R., Stauch T., Stepp H., Vogeser M., Brittenham G.M. Non-invasive detection of iron deficiency by fluorescence measurement of erythrocyte zinc protoporphyrin in the lip. Nat. Commun. 2016;7 doi: 10.1038/ncomms10776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Almeida A., Faustino M.A.F., Neves M.G.P.M. Antimicrobial photodynamic therapy in the control of COVID-19. Antibiotics Basel (Basel) 2020;9(6) doi: 10.3390/antibiotics9060320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhu N., Zhang D., Wang W., Li X., Yang B., Song J., Zhao X., Huang B., Shi W., Lu R., Niu P., Zhan F., Ma X., Wang D., Xu W., Wu G., Gao G.F., Tan W., C. China Novel A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020;382(8):727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Le Roy C., Wrana J.L. Clathrin- and non-clathrin-mediated endocytic regulation of cell signalling. Nat. Rev. Mol. Cell Biol. 2005;6(2):112–126. doi: 10.1038/nrm1571. [DOI] [PubMed] [Google Scholar]
- 22.Nazarenko Y. Air filtration and SARS-CoV-2. Epidemiol. Health. 2020;42 doi: 10.4178/epih.e2020049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shang J., Wan Y.S., Liu C., Yount B., Gully K., Yang Y., Auerbach A., Peng G.Q., Baric R., Li F. Structure of mouse coronavirus spike protein complexed with receptor reveals mechanism for viral entry. PLoS Pathog. 2020;16(3) doi: 10.1371/journal.ppat.1008392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Huang Y., Yang C., Xu X.F., Xu W., Liu S.W. Structural and functional properties of SARS-CoV-2 spike protein: potential antivirus drug development for COVID-19. Acta Pharmacol. Sin. 2020:9. doi: 10.1038/s41401-020-0485-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wang Y.J., Liu M.Y., Gao J.L. Enhanced receptor binding of SARS-CoV-2 through networks of hydrogen-bonding and interactions. Proc. Natl. Acad. Sci. U. S. A. 2020;117(25):13967–13974. doi: 10.1073/pnas.2008209117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xu H., Zhong L., Deng J., Peng J., Dan H., Zeng X., Li T., Chen Q. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int. J. Oral Sci. 2020;12(1):8. doi: 10.1038/s41368-020-0074-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Liu W., Zhang S.P., Nekhai S., Liu S.J. Depriving Iron supply to the virus represents a promising adjuvant therapeutic against viral survival. Curr. Clin. Microbiol. Rep. 2020;7(2):13–19. doi: 10.1007/s40588-020-00140-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schmidt S.M. The role of iron in viral infections. Front. Biosci. 2020;25:893–911. doi: 10.2741/4839. [DOI] [PubMed] [Google Scholar]
- 29.Asghar M.S., Kazmi S.J.H., Khan N.A., Akram M., Hassan M., Rasheed U., Khan S.A. Poor prognostic biochemical markers predicting fatalities caused by COVID-19: a retrospective observational study from a developing country. Cureus. 2020;12(8) doi: 10.7759/cureus.9575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu X.Q., Zhang R., He G.S. Hematological findings in coronavirus disease 2019: indications of progression of disease. Ann. Hematol. 2020;99(7):1421–1428. doi: 10.1007/s00277-020-04103-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ashokka B., Chakraborty A., Subramanian B.J., Karmakar M.K., Chan V. Reconfiguring the scope and practice of regional anesthesia in a pandemic: the COVID-19 perspective. Reg. Anesth. Pain Med. 2020;45(7):536–543. doi: 10.1136/rapm-2020-101541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bonetti G., Manelli F., Patroni A., Bettinardi A., Borrelli G., Fiordalisi G., Marino A., Menolfi A., Saggini S., Volpi R., Anesi A., Lippi G. Laboratory predictors of death from coronavirus disease 2019 (COVID-19) in the area of Valcamonica, Italy. Clin. Chem. Lab. Med. 2020;58(7):1100–1105. doi: 10.1515/cclm-2020-0459. [DOI] [PubMed] [Google Scholar]
- 33.Cavezzi A., Troiani E., Corrao S. COVID-19: hemoglobin, iron, and hypoxia beyond inflammation. A narrative review. Clin. Pract. 2020;10(2):24–30. doi: 10.4081/cp.2020.1271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ejigu T., Patel N., Sharma A., Vanjarapu J.M.R., Nookala V. Packed red blood cell transfusion as a potential treatment option in COVID-19 patients with hypoxemic respiratory failure: a case report. Cureus. 2020;12(6) doi: 10.7759/cureus.8398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chen N., Zhou M., Dong X., Qu J., Gong F., Han Y., Qiu Y., Wang J., Liu Y., Wei Y., Xia J., Yu T., Zhang X., Zhang L. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lippi G., Mattiuzzi C. Hemoglobin value may be decreased in patients with severe coronavirus disease 2019. Hematol. Transfus. Cell Ther. 2020;42(2):116–117. doi: 10.1016/j.htct.2020.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Schoning V., Liakoni E., Baumgartner C., Exadaktylos A.K., Hautz W.E., Atkinson A., Hammann F. Development and validation of a prognostic COVID-19 severity assessment (COSA) score and machine learning models for patient triage at a tertiary hospital. J. Transl. Med. 2021;19(1) doi: 10.1186/s12967-021-02720-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J., Speciality H.L.H.A. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395(10229):1033–1034. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ropa J., Cooper S., Capitano M.L., Broxmeyer H.E. Sars-cov-2 spike protein induces cellular changes in primitive and mature hematopoietic cells. Blood. 2020;136 [Google Scholar]
- 40.Cosic I., Cosic D., Loncarevic I. RRM prediction of erythrocyte Band3 protein as alternative receptor for SARS-CoV-2 virus. Appl. Sci.-Basel. 2020;10(11) [Google Scholar]
- 41.Bernardes J.P., Mishra N., Tran F., Bahmer T., Best L., Blase J.I., Bordoni D., Franzenburg J., Geisen U., Josephs-Spaulding J., Kohler P., Kunstner A., Rosati E., Aschenbrenner A.C., Bacher P., Baran N., Boysen T., Brandt B., Bruse N., Dorr J., Drager A., Elke G., Ellinghaus D., Fischer J., Forster M., Franke A., Franzenburg S., Frey N., Friedrichs A., Fuss J., Gluck A., Hamm J., Hinrichsen F., Hoeppner M.P., Imm S., Junker R., Kaiser S., Kan Y.H., Knoll R., Lange C., Laue G., Lier C., Lindner M., Marinos G., Markewitz R., Nattermann J., Noth R., Pickkers P., Rabe K.F., Renz A., Rocken C., Rupp J., Schaffarzyk A., Scheffold A., Schulte-Schrepping J., Schunk D., Skowasch D., Ulas T., Wandinger K.P., Wittig M., Zimmermann J., Busch H., Hoyer B.F., Kaleta C., Heyckendorf J., Kox M., Rybniker J., Schreiber S., Schultze J.L., Rosenstiel P., Network H.C.A.L.B., Deutsch C.-O.I. Longitudinal multi-omics analyses identify responses of Megakaryocytes, erythroid cells, and plasmablasts as hallmarks of severe COVID-19. Immunity. 2020;53(6):1296–+. doi: 10.1016/j.immuni.2020.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yagci S., Serin E., Acicbe O., Zeren M.I., Odabasi M.S. The relationship between serum erythropoietin, hepcidin, and haptoglobin levels with disease severity and other biochemical values in patients with COVID-19. Int. J. Lab. Hematol. 2021:10. doi: 10.1111/ijlh.13479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Huerga Encabo H., Grey W., Garcia-Albornoz M., Wood H., Ulferts R., Aramburu I.V., Kulasekararaj A.G., Mufti G., Papayannopoulos V., Beale R., Bonnet D. Human erythroid progenitors are directly infected by SARS-CoV-2: implications for emerging erythropoiesis in severe COVID-19 patients. Stem Cell Reports. 2021;16(3):428–436. doi: 10.1016/j.stemcr.2021.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Shahbaz S. In: Erythroid Precursors and Progenitors Suppress Adaptive Immunity and Get Invaded by SARS-CoV-2. Xu L., Osman M., Sligl W., Shields J., Joyce M., Tyrrell L., Oyegbami O.S., Elahi, editors. BioRxiv; 2020. 2020.08.18.255927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gao G.F., Li J., Zhang Y.T., Chang Y.Z. In: Brain Iron Metabolism and Cns Diseases. Chang Y.Z., editor. Springer-Verlag Singapore Pte Ltd; Singapore: 2019. Cellular iron metabolism and regulation; pp. 21–32. [Google Scholar]
- 46.Chhabra R., Saha A., Chamani A., Schneide N., Shah R., Nanjundan M. Iron pathways and Iron chelation approaches in viral, microbial, and fungal infections. Pharmaceuticals. 2020;13(10):23. doi: 10.3390/ph13100275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Abbaspour N., Hurrell R., Kelishadi R. Review on iron and its importance for human health. J. Res. Med. Sci. 2014;19(2):164–174. [PMC free article] [PubMed] [Google Scholar]
- 48.Puig S., Ramos-Alonso L., Romero A.M., Martinez-Pastor M. The elemental role of iron in DNA synthesis and repair. Metallomics. 2017;9(11):1483–1500. doi: 10.1039/c7mt00116a. [DOI] [PubMed] [Google Scholar]
- 49.Drakesmith H., Prentice A. Viral infection and iron metabolism. Nat. Rev. Microbiol. 2008;6(7):541–552. doi: 10.1038/nrmicro1930. [DOI] [PubMed] [Google Scholar]
- 50.Claiborne A., Yeh J.I., Mallett T.C., Luba J., Crane E.J., Charrier V., Parsonage D. Protein-sulfenic acids: diverse roles for an unlikely player in enzyme catalysis and redox regulation. Biochemistry. 1999;38(47):15407–15416. doi: 10.1021/bi992025k. [DOI] [PubMed] [Google Scholar]
- 51.Ajioka R.S., Phillips J.D., Kushner J.P. Biosynthesis of heme in mammals. Biochim. Biophys. Acta, Mol. Cell Res. 2006;1763(7):723–736. doi: 10.1016/j.bbamcr.2006.05.005. [DOI] [PubMed] [Google Scholar]
- 52.Wagener F., Volk H.D., Willis D., Abraham N.G., Soares M.P., Adema G.J., Figdor C.G. Different faces of the heme-heme oxygenase system in inflammation. Pharmacol. Rev. 2003;55(3):551–571. doi: 10.1124/pr.55.3.5. [DOI] [PubMed] [Google Scholar]
- 53.Saha D., Koli S., Reddy K.V.R. Transcriptional regulation of Hb-alpha and Hb-beta through nuclear factor E2-related factor-2 (Nrf2) activation in human vaginal cells: A novel mechanism of cellular adaptability to oxidative stress. Am. J. Reprod. Immunol. 2017;77(6) doi: 10.1111/aji.12645. [DOI] [PubMed] [Google Scholar]
- 54.Grek C.L., Newton D.A., Spyropoulos D.D., Baatz J.E. Hypoxia up-regulates expression of hemoglobin in alveolar epithelial cells. Am. J. Respir. Cell Mol. Biol. 2011;44(4):439–447. doi: 10.1165/rcmb.2009-0307OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Tezel T.H., Geng L.J., Lato E.B., Schaal S., Liu Y.Q., Dean D., Klein J.B., Kaplan H.J. Synthesis and secretion of hemoglobin by retinal pigment epithelium. Invest. Ophthalmol. Vis. Sci. 2009;50(4):1911–1919. doi: 10.1167/iovs.07-1372. [DOI] [PubMed] [Google Scholar]
- 56.Schaer D.J., Buehler P.W., Alayash A.I., Belcher J.D., Vercellotti G.M. Hemolysis and free hemoglobin revisited: exploring hemoglobin and hemin scavengers as a novel class of therapeutic proteins. Blood. 2013;121(8):1276–1284. doi: 10.1182/blood-2012-11-451229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ghosh S., Adisa O.A., Chappa P., Tan F., Jackson K.A., Archer D.R., Ofori-Acquah S.F. Extracellular hemin crisis triggers acute chest syndrome in sickle mice. J. Clin. Invest. 2013;123(11):4809–4820. doi: 10.1172/JCI64578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ryter S.W., Otterbein L.E. Carbon monoxide in biology and medicine. Bioessays. 2004;26(3):270–280. doi: 10.1002/bies.20005. [DOI] [PubMed] [Google Scholar]
- 59.Basiglio C.L., Arriaga S.M., Pelusa H.F., Almara A.M., Roma M.G., Mottino A.D. Protective role of unconjugated bilirubin on complement-mediated hepatocytolysis. Biochim. Biophys. Acta, Gen. Subj. 2007;1770(7):1003–1010. doi: 10.1016/j.bbagen.2007.03.005. [DOI] [PubMed] [Google Scholar]
- 60.Sarady-Andrews J.K., Liu F., Gallo D., Nakao A., Overhaus M., Ollinger R., Choi A.M., Otterbein L.E. Biliverdin administration protects against endotoxin-induced acute lung injury in rats. Am. J. Physiol. Lung Cell. Mol. Physiol. 2005;289(6):L1131–L1137. doi: 10.1152/ajplung.00458.2004. [DOI] [PubMed] [Google Scholar]
- 61.San Juan I., Bruzzone C., Bizkarguenaga M., Bernardo-Seisdedos G., Laín A., Gil-Redondo R., Diercks T., Gil-Martínez J., Urquiza P., Arana E., Seco M., García de Vicuña A., Embade N., Mato J.M., Millet O. Abnormal concentration of porphyrins in serum from COVID-19 patients. Br. J. Haematol. 2020 doi: 10.1111/bjh.17060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Maitra D., Cunha J.B., Elenbaas J.S., Bonkovsky H.L., Shavit J.A., Omary M.B. Porphyrin-induced protein oxidation and aggregation as a mechanism of porphyria-associated cell injury. Cell. Mol. Gastroenterol. Hepatol. 2019;8(4):535–548. doi: 10.1016/j.jcmgh.2019.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.wenzhong l. In: COVID-19:Attacks the 1-Beta Chain of Hemoglobin and Captures the Porphyrin to Inhibit Human Heme Metabolism. Hualan L., editor. ChemRxiv; 2020. 11938173. [Google Scholar]
- 64.Zeligman I. Red fluorescence of urine in Wood’s light as aid in office diagnosis of porphyria. Arch. Derm. Syphilol. 1950;61(5):853–854. doi: 10.1001/archderm.1950.01530120144014. [DOI] [PubMed] [Google Scholar]
- 65.Courrol L.C., de Oliveira Silva F.R., Bellini M.H., Mansano R.D., Schor N., Vieira N.D., Junior . In: Optical Methods for Tumor Treatment and Detection: Mechanisms and Techniques in Photodynamic Therapy XVI. Kessel D., editor. 2007. Blood porphyrin luminescence and tumor growth correlation - art. no. 64270Y. pp. Y4270-Y4270. [Google Scholar]
- 66.Lippert B.M., Grosse U., Klein M., Kuelkens C., Klahr N., Brossmann P., Teymoortash A., Ney M., Doss M.O., Werner J.A. Excretion measurement of porphyrins and their precursors after topical administration of 5-aminolaevulinic acid for fluorescence endoscopy in head and neck cancer. Res. Commun. Mol. Pathol. Pharmacol. 2003;113:75–85. [PubMed] [Google Scholar]
- 67.Tao Z.Y., Xu J., Chen W., Yang Z.T., Xu X.M., Liu L., Chen R.W., Xie J.Y., Liu M.Y., Wu J.Y., Wang H.M., Liu J.L. Anemia is associated with severe illness in COVID-19: a retrospective cohort study. J. Med. Virol. 2020 doi: 10.1002/jmv.26444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Yu H., Tan Y.F., Cunningham B.T. Smartphone fluorescence spectroscopy. Conference on Frontiers in Biological Detection - From Nanosensors to Systems VII; Spie-Int Soc Optical Engineering, San Francisco, CA; 2015. [Google Scholar]
- 69.Hojeong Y., Yafang T., Cunningham B.T. Smartphone fluorescence spectroscopy. Anal. Chem. 2014;86(17):8805. doi: 10.1021/ac502080t. (Report) [DOI] [PubMed] [Google Scholar]
- 70.Das A.J., Wahi A., Kothari I., Raskar R. Ultra-portable, wireless smartphone spectrometer for rapid, non-destructive testing of fruit ripeness. Sci. Rep. 2016;6:8. doi: 10.1038/srep32504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kitajima Y., Ishii T., Kohda T., Ishizuka M., Yamazaki K., Nishimura Y., Tanaka T., Dan S., Nakajima M. Mechanistic study of PpIX accumulation using the JFCR39 cell panel revealed a role for dynamin 2-mediated exocytosis. Sci. Rep. 2019;9(1):8666. doi: 10.1038/s41598-019-44981-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.de Oliveira Silva F.R., Helena Bellini M., Regina Tristao V., Schor N., Vieira N.D., Jr., Courrol L.C. Intrinsic fluorescence of protoporphyrin IX from blood samples can yield information on the growth of prostate tumours. J. Fluoresc. 2010;20(6):1159–1165. doi: 10.1007/s10895-010-0662-9. [DOI] [PubMed] [Google Scholar]
- 73.Hagiya Y., Endo Y., Yonemura Y., Takahashi K., Ishizuka M., Abe F., Tanaka T., Okura I., Nakajima M., Ishikawa T., Ogura S. Pivotal roles of peptide transporter PEPT1 and ATP-binding cassette (ABC) transporter ABCG2 in 5-aminolevulinic acid (ALA)-based photocytotoxicity of gastric cancer cells in vitro. Photodiagnosis Photodyn. Ther. 2012;9(3):204–214. doi: 10.1016/j.pdpdt.2011.12.004. [DOI] [PubMed] [Google Scholar]
- 74.Sabino C.P., Ball A.R., Baptista M.S., Dai T.H., Hamblin M.R., Ribeiro M.S., Santos A.L., Sellera F.P., Tegos G.P., Wainwright M. Light-based technologies for management of COVID-19 pandemic crisis. J. Photochem. Photobiol. B. 2020;212:8. doi: 10.1016/j.jphotobiol.2020.111999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Eickmann M., Gravemann U., Handke W., Tolksdorf F., Reichenberg S., Mueller T.H., Seltsam A. Inactivation of three emerging viruses - severe acute respiratory syndrome coronavirus, Crimean-Congo haemorrhagic fever virus and Nipah virus - in platelet concentrates by ultraviolet C light and in plasma by methylene blue plus visible light. Vox Sang. 2020;115(3):146–151. doi: 10.1111/vox.12888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Agostinis P., Berg K., Cengel K.A., Foster T.H., Girotti A.W., Gollnick S.O., Hahn S.M., Hamblin M.R., Juzeniene A., Kessel D., Korbelik M., Moan J., Mroz P., Nowis D., Piette J., Wilson B.C., Golab J. Photodynamic therapy of cancer: an update. CA Cancer J. Clin. 2011;61(4):250–281. doi: 10.3322/caac.20114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.S. D . In: Reduction of the Viral Load by non-Invasive Photodynamic Therapy in Early Stages of COVID-19 Infection. Hepburn J P.S.R., editor. 2020. American Journal of Virology & Disease. [Google Scholar]
- 78.Tian J., Huang B.X., Nawaz M.H., Zhang W.A. Recent advances of multi-dimensional porphyrin-based functional materials in photodynamic therapy. Coord. Chem. Rev. 2020;420 [Google Scholar]
- 79.Costley D., Mc Ewan C., Fowley C., McHale A.P., Atchison J., Nomikou N., Callan J.F. Treating cancer with sonodynamic therapy: a review. Int. J. Hyperth. 2015;31(2):107–117. doi: 10.3109/02656736.2014.992484. [DOI] [PubMed] [Google Scholar]
- 80.Giuntini F., Foglietta F., Marucco A.M., Troia A., Dezhkunov N.V., Pozzoli A., Durando G., Fenoglio I., Serpe L., Canaparo R. Insight into ultrasound-mediated reactive oxygen species generation by various metal-porphyrin complexes. Free Radic. Biol. Med. 2018;121:190–201. doi: 10.1016/j.freeradbiomed.2018.05.002. [DOI] [PubMed] [Google Scholar]
- 81.Sadanala K.C., Chaturvedi P.K., Seo Y.M., Kim J.M., Jo Y.S., Lee Y.K., Ahn W.S. Sono-photodynamic combination therapy: a review on sensitizers. Anticancer Res. 2014;34(9):4657–4664. [PubMed] [Google Scholar]
- 82.Zhou Y.M., Liang X.L., Dai Z.F. Porphyrin-loaded nanoparticles for cancer theranostics. Nanoscale. 2016;8(25):12394–12405. doi: 10.1039/c5nr07849k. [DOI] [PubMed] [Google Scholar]
- 83.Cheng J.L., Sun X., Guo S.Y., Cao W., Chen H.B., Jin Y.H., Li B., Li Q.N., Wang H., Wang Z., Zhou Q., Wang P., Zhang Z.G., Cao W.W., Tian Y. Effects of 5-aminolevulinic acid-mediated sonodynamic therapy on macrophages. Int. J. Nanomedicine. 2013;8:669–676. doi: 10.2147/IJN.S39844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.da-Palma-Cruz M., da Silva R.F., Monteiro D., Rehim H.M.M.A., Grabulosa C.C., Ligeiro de Oliveira A.P., Lino-dos-Santos-Franco A. Photobiomodulation modulates the resolution of inflammation during acute lung injury induced by sepsis. Lasers Med. Sci. 2019;34(1):191–199. doi: 10.1007/s10103-018-2688-1. [DOI] [PubMed] [Google Scholar]
- 85.Lee G.H., Moon H., Kim H., Kwon W., Yoo S., Myung D., Yun S.H., Bao Z., Hahn S.K. Multifunctional materials for implantable and wearable photonic healthcare devices. Nat. Rev. Mater. 2020;5(2):149–165. doi: 10.1038/s41578-019-0167-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kipshidze N., Yeo N.I. Photodynamic and sonodynamic therapy of acute hypoxemic respiratory failure in patients with COVID-19. Photodiagnosis Photodyn. Ther. 2020;31:2. doi: 10.1016/j.pdpdt.2020.101961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Li L., Chen Y., Wang X.B., Feng X.L., Wang P., Liu Q.H. Comparison of protoporphyrin IX produced cell proliferation inhibition between human breast cancer MCF-7 and MDA-MB-231 cells. Pharmazie. 2014;69(8):621–628. [PubMed] [Google Scholar]
- 88.Goncalvez K.D., Vieira D.P., Courrol L.C. Study of THP-1 macrophage viability after sonodynamic therapy using methyl ester of 5-aminolevulinic acid gold nanoparticles. Ultrasound Med. Biol. 2018;44(9):2009–2017. doi: 10.1016/j.ultrasmedbio.2018.05.012. [DOI] [PubMed] [Google Scholar]