Abstract
Objective and design:
The omega-3 polyunsaturated fatty acid docosahexaenoic acid (DHA) has been reported to suppress inflammation. Pulmonary inflammation can be directly linked to exposure of various occupational and man-made particles leading to pulmonary diseases. Therapeutic treatments are lacking for particle-induced pulmonary inflammation. These studies evaluated DHA as a therapeutic treatment for semi-acute and chronic particle-induced pulmonary inflammation.
Methods:
Balb/c mice were oropharyngeal instilled with hydrophobic multi-walled carbon nanotube (MWCNT) or hydrophilic crystalline silica (SiO2) either as one instillation (semi-acute) or once a week for 4 weeks (chronic). One week later, the mice were placed on either a control or 1% DHA-containing diet for 3 weeks (semi-acute) or 12 weeks (chronic). Mice were assessed for inflammatory signaling within the lung lavage fluid, impact on phagolysosomal membrane permeability, shifts of macrophage phenotype gene expression (M1, M2a, M2b, and M2c), and pulmonary histopathology.
Results:
DHA increased pulmonary inflammatory markers and lung pathology when mice were exposed to SiO2. There were trending decreases of inflammatory markers for MWCNT-exposed mice with DHA treatment, however, mostly not statistically significant.
Conclusion:
The anti-inflammatory benefits of DHA treatment depend upon the type of inflammatory particle, magnitude of inflammation, and duration of treatment.
Keywords: pulmonary inflammation, crystalline silica, multi-walled carbon nanotube, docosahexaenoic acid, macrophage phenotype, phagolysosomal membrane damage
1. Introduction
Two important families of polyunsaturated fatty acids (PUFA) are omega-6 and omega-3 which encompass the essential fatty acids of linoleic and α-linolenic acids; respectively. These essential fatty acids are converted into other fatty acids within each PUFA family such as arachidonic (ARA) and docosahexaenoic (DHA) acids through various desaturation and elongation steps [1]. Unfortunately, in most Western diets there is too much omega-6; in part due to high levels of consumption of red meat, and not enough of omega-3 which can be improved with consumption of fatty fish. This diet leads to an omega-6/−3 ratio of 10–20/1 where most adults intake less than 0.2 g/day of DHA [1, 2]. Increased ARA content has been linked to inflammation while exposure to omega-3 PUFAs; specifically DHA, has been reported to have various anti-inflammatory capabilities [1, 3]. Recent research has suggested that omega-3 PUFAs suppress particle-induced pulmonary and systemic inflammation as well [4, 5].
Respiratory diseases make up five of the thirty most common causes of death [6]. It is known that exposure to airborne toxicants can cause acute and chronic inflammation within the lungs which can lead to serious inflammatory diseases; such as lung fibrosis, and contribute to autoimmune diseases [7]. One inhalable particle that is of concern is a common engineered nanomaterial known as a multi-walled carbon nanotube (MWCNT). The use of MWCNTs have been increasing in various commercial products such as in electronics and medical applications, however, MWCNT-exposure has been linked to lung fibrosis and granuloma formation [8]. The surface of MWCNT is very hydrophobic suggesting possible association with nonpolar regions of membranes and has been reported to cause increased inflammatory responses [9]. Another particle of concern is micron-sized crystalline silica (SiO2). SiO2 exposure primarily occurs in occupational settings such as mining and manufacturing and is known to cause silicosis and autoimmune disorders [8, 10]. In contrast to MWCNT, the surface of SiO2 is hydrophilic with nearly free surface silanols associated with membrane phospholipid headgroups which contribute to driving SiO2-induced inflammation [11].
Understanding the mechanisms of lung inflammation and the development of therapeutic treatments are lacking within the field of particle-induced pulmonary inflammation. Evidence has suggested that DHA blocked SiO2-triggered autoimmunity in autoimmune disease-prone NZBWF1 mice and that omega-3 PUFAs decreased inflammation in both C57BL/6 wild-type mice; which are Th1-prone, and a transgenic Fat-1 murine model with a C57BL/6 background [4, 5, 12]. Balb/c mice are Th2-prone and commonly used to assess pulmonary/allergic inflammatory diseases since they have a more sensitive airway inflammatory response than C57/BL6 mice [12–14]. However, assessing the effects of DHA treatment for particle-induced inflammation within a Balb/c murine model is currently limited.
The objectives of the current studies were to determine whether DHA could be used as a prospective therapeutic treatment within both semi-acute and chronic particle exposure models. Most research on DHA treatment is assessing it as a potential prophylactic treatment, but research of DHA as a possible therapeutic is scarce. In reality, treatment to inflammatory diseases typically occurs after inflammation has ensued. Therefore, it is crucial to address this gap of knowledge in order to understand if DHA treatment could be used as a therapeutic for particle-induced inflammation. The hypothesis of these studies is that DHA will decrease pulmonary inflammatory markers in response to particle-induced inflammation. The findings are considered with respect to the differential physical characteristics of the two particles as well as the types of inflammation that the particles cause.
2. Materials and Methods
2.1. Particle preparations
Preparations and characteristics of a highly inflammatory multi-walled carbon nanotube (MWCNT; provided by the National Toxicology Program at NIEHS; referred to as FA21 [15]) and acid-washed crystalline SiO2 (SiO2; Min-U-Sil-5, Pennsylvania Glass Sand Corp.; Pittsburgh, PA, USA) are as previously described [15, 16]. Briefly, both particles were prepared fresh in dispersion media vehicle (DM; 0.6 mg/mL mouse serum albumin [Sigma-Aldrich; St. Louis, MO, USA] and 0.01 mg/mL 1,2-dipalmitoyl-sn-glycero-3-phosphocholine [Sigma-Aldrich] in PBS). The particle suspensions were sonicated (550 watts, 20 kHz, 5 minutes, 35% amplitude) in a cup-horn sonicator.
2.2. Mice and diets
Both male and female wild type Balb/c mice (bred in-house at the University of Montana), 6–8 weeks old, were used in equal numbers. The number of mice used for each experiment are depicted within each figure legend. Please note that sex comparisons could not be performed due to too low of numbers for each individual sex separately; therefore, male and female mice were combined within each group to evaluate statistical analysis. The animal room is programmed on a 12-hour light/dark cycle at approximately 18–26°C with food and water provided ad libitum. The animals were monitored on a daily basis and weighed once a week to track food consumption. Please refer to the Ethics approval section below.
Formulations of experimental diets were summarized in Online Resource 1. Briefly, modified American Institute of Nutrition (AIN)-93G powdered diet (Dyets, Inc.; Bethlehem, PA, USA) containing 70 g/kg fat was prepared with 10 g/kg food-grade corn oil (Wesson Pure 100% Natural Corn Oil, Amazon; Seattle, WA, USA) to provide essential fatty acids. The control diet contained 60 g/kg food-grade oleic safflower oil (Oleico Natural Safflower Oil, Walmart; Bentonville, AR, USA). The oleic safflower oil was replaced with 25 g/kg microalgal oil containing 40% DHA (DHASCO™, Martek Biosciences Corp., provided by Dr. Pestka at Michigan State University; East Lansing, MI, USA) which corresponded to 1% (w/w) DHA. This dosage was chosen to represent a relevant human dietary intake of 5 g/day and was conducted in collaboration with Dr. Pestka in a similar manner as previously described [4]. The European Food Safety Authority deemed supplements containing 5 g/day omega-3 PUFA from microalgal oil safe for human consumption [17]. All diets were changed every 48 hours to prevent oxidation. Confirmation of fatty acid composition within red blood cells was sent for analysis (semi-acute; Online Resource 2, and chronic; Online Resource 3, OmegaQuant; Sioux Falls, SD, USA) which confirmed a significant increase in DHA content within the DHA-fed mice compared to the control diet for both exposure models (Online Resource 4).
2.3. Experimental design
The overall study designs are depicted in Online Resource 5 (semi-acute; A and chronic; B). Briefly, mice were anesthetized with isoflurane (5%, inhaled) before instillation so as not to use any restraints or cause distress and then oropharyngeal instilled once with either DM-only vehicle (50 μl/mouse), MWCNT (50 μg/mouse), or SiO2 (1 mg/mouse) at week 0 (semi-acute) or once a week at weeks 0, 1, 2, 3 (chronic). The route and doses used for these studies were selected based on the inflammatory activity established in prior work [18]. The mice were kept on the normal mouse chow during this time for each study. One week after the last particle instillation (week 1; semi-acute, or week 4; chronic) the mice were placed on either a control or 1% DHA-containing diet (25 g/kg) for 3 weeks (semi-acute) or 12 weeks (chronic). The mice were euthanized by a lethal dose of sodium pentobarbital (150 mg/kg, Euthasol™) either 4 weeks (semi-acute) or 13 weeks (chronic) after the last particle instillation by intraperitoneal injection; a method consistent with the American Veterinary Medical Association guidelines. This method of euthanasia causes minimal distress to the mice along with ensuring no injury to the lungs.
2.4. Isolation of pulmonary cells
Cells within the lungs were isolated by performing lung lavages and assessed as previously described [19, 20]. Briefly, the lungs were removed from the body and lavaged with ice-cold PBS to obtain optimal cell yields. The first pull (instillation of 0.5 mL of ice-cold PBS, withdrawn, then the same fluid repeated twice more) consisted of the most concentrated lung lavage fluid (LLF). Lung lavaged cells were isolated by centrifugation (400 x g, 5 min, 4°C) and cell counts were obtained via Coulter Z2 particle counter (Beckman Coulter; Brea, CA, USA). Cells were stained for differential analysis via Wright-Geimsa stain in a Hematek 2000 autostainer (Miles-Bayer-Siemens Diagnostics; Deerfield, IL, USA). Alveolar macrophages (AM) within the LLF were isolated and cultured ex vivo in RPMI-1640 media (Corning; Corning, NY, USA) supplemented with 10% heat-inactivated fetal bovine serum (VWR; Radnor, PA, USA), 1% penicillin-streptomycin (Corning) and 1% sodium pyruvate (Corning) for 24 hours at 37°C with LPS for priming (20 ng/mL) to assess IL-1β levels within the supernatant.
2.5. Analysis of cell damage and inflammatory mediators
Cell damage and inflammatory markers were assessed within LLF of the mice. Cell damage was determined by release of lactate dehydrogenase (LDH; CytoTox 96 cytotoxicity assay, Promega; Madison, WI, USA) and protein concentration (Pierce BCA protein assay, Thermo Fisher Scientific; Waltham, MA, USA) according to manufacturer’s instructions. Cytokines were assessed by using a customized MesoScale Discovery U-PLEX assay platform (IFNγ, IL-1β, TNFα, IL-33, IL-6, IL-10, IL-13; Meso Scale Diagnostics LLC; Rockville, MD, USA) according to manufacturer’s instructions. Phagolysosomal membrane permeability (LMP) analysis was determined by total cathepsin (CTS; Z-LR-AMC fluorogenic peptide substrate, R&D Systems; Minneapolis, MN, USA) and cathepsin B (cathepsin B inhibitor II - Calbiochem, Millipore Sigma; Burlington, MA, USA) release within the LLF and IL-1β release (mouse IL-1β DuoSet ELISA, R&D Systems) within the supernatants of isolated ex vivo AM.
2.6. Macrophage phenotype analysis
Macrophage phenotype was assessed in lung and spleen tissues as previously described [21]. Briefly, sections of lung and spleen tissues were snap-frozen on the day of harvest and RNA was isolated (TRIzol®, Thermo Fisher Scientific; Waltham, MA, USA) according to manufacturer’s instructions. All RNA was converted to cDNA (iScript RT Supermix, Bio-Rad; Hercules, CA, USA) and qPCR analysis was performed (SSoAdvanced Univ. SYBR Green Supermix, Bio-Rad) according to manufacturer’s instructions to determine macrophage phenotype: M1 (CXCL10, IL-1β, TNFα, iNos), M2a (FN1, CD206, Fizz1, YM1), M2b (CCL1, LIGHT, CXCL3, SPHK1), and M2c (CD163, CXCL13, TIMP1, TLR8). All qPCR primers with associated positive controls and reference genes used were validated PrimePCR SYBR Green assays from Bio-Rad. All signals were collected via 384-well CFX Maestro (Bio-Rad, supplied by Dr. Patel at FYR Diagnostics, Missoula, MT, USA and Dr. Kreitinger at Dermaxon, Missoula, MT, USA) and levels were normalized to reference gene β−2 microglobulin (B2m; ΔCq). Relative gene expressions of MWCNT and SiO2 of control-fed mice were normalized to DM-only of control-fed mice (ΔΔCq) to assess particle effect. The relative gene expressions of DM-only, MWCNT, and SiO2 of the 1% DHA-fed mice were normalized to DM-only, MWCNT, and SiO2 of the control-fed mice; respectively, (ΔΔCq) to assess diet effect. The ΔΔCq values were analyzed within a heat-map. A dominant phenotype shift was indicated by an upregulation of gene expression of at least three of the four genes within each phenotype or a trending increase indicated by upregulation of two of the four genes.
2.7. Histopathology
Lungs from each mouse were inflation-fixed through the trachea with 3% paraformaldehyde-PBS and placed in this same fixative overnight at 4°C. The lungs were washed with ice-cold PBS, dehydrated with 70% EtOH, and paraffin-embedded. Tissue sections (5 μm) were stained with Masson’s trichrome for histological analysis. Mouse lung sections were imaged at 10x using Nikon E 800 epi-fluorescent/transmitted light microscope with Olympus DP71 camera and CellSens software in the Molecular Histology and Fluorescence Imaging Core at the University of Montana.
The lung tissues were scored in a blinded fashion by two observers separately with three mice per treatment group. The degree of lung disease visible within the lung sections were scored using a 5-point scale system (0, 1, 2, 3, 4) for the percentage of total pulmonary tissue effected with (0) being no effect, (1) minimal (<10 %), (2) slight (10 – 25 %), (3) moderate (26 – 50 %), and (4) severe (51 – 75 %) lung pathology by assessing collagen deposition, extracellularity, inflammation, lesion formation, particle encapsulation, and airway thickening. Cronbach’s-α was used to assess reliability between scorers where inter-rater reliability was significant at 0.956 indicating agreement between the scores of the two observers. The values shown are the median of the two scorers’ values. Lung airway thickness was measured on the iCys Laser Scanning Cytometer (ThorLabs; Newton, NJ, USA) in the Fluorescence Cytometry Core at the University of Montana. Six airways with four measurements per airway in duplicate lung sections per mouse were measured with three mice per treatment group.
2.8. Statistical analysis
Depending on the data type, data had to undergo a log-transformation in order to equalize the variance followed by a parametric one- or two-way analysis of variance (ANOVA) and post hoc mean comparison (Holm-Sidak). Data was expressed as mean ± SEM with 0.05 significance level. Subjective histopathology scoring is ordinal level data; therefore, raw scores were rank-transformed prior to ANOVA and post hoc analysis. Graphics and analyses were performed on PRISM 8.3.1 (GraphPad; San Diego, CA, USA).
3. Results
3.1. Effect of DHA on pulmonary cells and lung injury
A semi-acute model was used to assess the effects of DHA treatment following delivery of a short-term particle exposure and a chronic model to simulate a prolonged DHA treatment following a more substantial particle exposure model. After termination of the two scenarios, Balb/c mice were extensively evaluated to determine particle effects with and without DHA. Whole lung lavages were performed in order to collect pulmonary immune cells and assess inflammatory responses. The semi-acute model showed no significant differences between the diets for total cell count regardless of exposure; however, there was a significant increase in the cell count of SiO2-exposed mice compared to DM mice while on the DHA diet (Fig. 1a). When assessing the cell differentials (Fig. 1b), the mice had mostly alveolar macrophages (AM) present regardless of diet and exposure. Mice exposed to SiO2 also had neutrophils and eosinophils present which were significantly increased when compared to the DM-only mice within the same diets. Cellular toxicity was determined by assessing LDH levels (indicative of overall pulmonary cell damage; Fig. 1c) and protein concentration (marker for epithelial cell damage; Fig. 1d), within the lung lavage fluid (LLF). There were no statistically significant outcomes when assessing cellular toxicity, however, there were various trends. Since there was a mixture of results between cellular differentials and toxicity, both inflammatory and pro-resolving cytokines were assessed within the LLF to determine the effects of particles and DHA treatment on signaling responses (Fig. 2). Mice exposed to MWCNT with DHA treatment had a significant decrease in TNFα levels. Mice exposed to SiO2 with DHA treatment had significantly increased IFN-γ and IL-1β levels compared to SiO2-exposed mice on the control diet. (Fig. 2). The SiO2-exposed mice on the DHA diet also had a significant increase of all the inflammatory cytokine levels within the LLF when compared to the DM-exposed mice on the DHA diet. These results showed that mice with a semi-acute particle exposure to MWCNT while on a therapeutic 1% DHA diet had less lung injury compared to that of SiO2-exposed mice while on the same DHA diet.
Fig. 1. Pulmonary cells and lung injury analysis in semi-acute model.
Balb/c mice were instilled once with either DM-only, MWCNT, or SiO2 at week 0, kept on the normal mouse chow for 1 week, and then put on either a control or 1% DHA diet for 3 weeks. (a) Total cell counts and (b) percentage of cell types via cytospins of cell differentials were assessed. (c) LDH and (d) protein concentration were assessed in the LLF for lung damage. Data presented as mean ± SEM, n=4–6. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001 compared to DM, + compared to different diet within the same exposure group. Analyses were done with log-transformed data
Fig. 2. Cytokine analysis in semi-acute model.
Balb/c mice were instilled once with either DM-only, MWCNT, or SiO2 at week 0, kept on the normal mouse chow for 1 week, and then put on either a control or 1% DHA diet for 3 weeks. Inflammatory cytokines; IFN-γ, IL-1β, TNFα, IL-33, pro-resolving cytokines; IL-10, and IL-13, and IL-6 were analyzed in the LLF. Data presented as mean ± SEM, n=4–6. *P<0.05, **P<0.01, ****P<0.0001 compared to DM, + compared to different diet within the same exposure group. Analyses were done with log-transformed data
In the chronic exposure model, there were no significant differences between the diets for total cell count regardless of exposure; however, there was a significant increase in the cell count of SiO2-exposed mice compared to DM mice within both the control and DHA diets (Fig. 3a). The cell differentials showed mostly AM present regardless of exposure group as well as increases in lymphocytes (Fig. 3b). There were no significant differences between the diets for mice exposed to MWCNT when assessing cytotoxicity within the LLF, however, there was a significant decrease of TNFα levels for MWCNT-exposed mice on the DHA diet compared to MWCNT-exposed mice on the control diet (Fig. 4). Exposure to SiO2 significantly increased neutrophils and eosinophils when compared to the DM-exposed mice in addition to AM and lymphocytes for both diets (Fig. 3b). This corresponded to increased cellular toxicity as indicated by a significant increase in LDH levels (Fig. 3c) within the LLF of mice on the DHA diet compared to control diet mice. SiO2-exposed mice had a significant increase in LDH levels and protein concentration when compared to the DM-exposed mice regardless of diet (Fig. 3c,d). Mice exposed to SiO2 and treated with DHA also significantly increased IFN-γ and IL-1β levels and significantly decreased IL-10 levels within the LLF when compared to SiO2-exposed mice on the control diet (Figure 4). These results indicated that mice on the therapeutic 1% DHA diet with a chronic particle exposure to SiO2 was not protective.
Fig. 3. Pulmonary cells and lung injury analysis in chronic model.
Balb/c mice were instilled with either DM-only, MWCNT, or SiO2 once a week at weeks 0, 1, 2, 3 while on the normal mouse chow and at week 4 were put on a control or 1% DHA diet for 12 weeks. (a) Total cell counts and (b) percentage of cell types via cytospins of cell differentials were assessed. (c) LDH and (d) protein concentration were assessed in the LLF for lung damage. Data presented as mean ± SEM, n=4–7. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001 compared to DM, + compared to different diet within the same exposure group. Analyses were done with log-transformed data
Fig. 4. Cytokine analysis in chronic model.
Balb/c mice were instilled with either DM-only, MWCNT, or SiO2 once a week at weeks 0, 1, 2, 3 while on the normal mouse chow and at week 4 were put on a control or 1% DHA diet for 12 weeks. Inflammatory cytokines; IFN-γ, IL-1β, TNFα, IL-33, pro-resolving cytokines; IL-10, and IL-13, and IL-6 were analyzed in the LLF. Data presented as mean ± SEM, n=4–7. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001 compared to DM, + compared to different diet within the same exposure group. Analyses were done with log-transformed data
3.2. Alteration of LMP due to DHA
As described above, AM were the dominant cell type present within the LLF of the mice; regardless of exposure group, for both the semi-acute and chronic exposure models. Pulmonary macrophages are responsible for maintenance of homeostatic conditions within the lungs. This includes regulating immune responses to inhaled foreign materials; such as exposure to MWCNT or SiO2, by phagocytosing these foreign substances and processing them for elimination. However, these environmental stimuli can cause lysosomal damage which can lead to phagolysosomal membrane permeability (LMP) where various lysosome-specific proteases; such as cathepsins, are released into the cytosol. Activation of the NLRP3 inflammasome by these proteases, cathepsin B in particular, can occur and ultimately IL-1β release ensues which results in various inflammatory cascades [22, 23]. Therefore, the effects of DHA on LMP were assessed in order to determine if DHA had impacted the stability of the phagolysosmal membrane. AM were isolated from the LLF and cultured ex vivo in order to assess IL-1β release specifically from AM, unlike above where it was measured within the total LLF. Total cathepsin and cathepsin B levels; lysosome-specific proteases, were measured within the LLF as well.
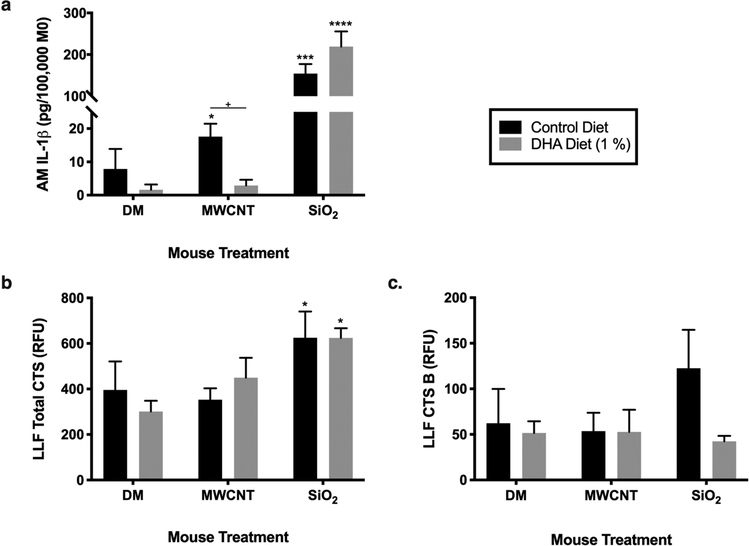
In the semi-acute exposure model, mice exposed to MWCNT while on the DHA diet had a significant decrease of IL-1β release ex vivo from AM (Fig. 5a); however, there was no significant difference between diets for the total cathepsin levels (Fig. 5b) an no change between diets for cathepsin B release (Fig. 5c) within the LLF. SiO2-exposed mice had a significant increase of both IL-1β release ex vivo from AM and total cathepsin levels in the LLF regardless of diet when compared to the DM-only mice (Fig. 5a,b), however, no significant differences between the diets.
Fig. 5. Assessment of LMP in semi-acute model.
Balb/c mice were instilled once with either DM-only, MWCNT, or SiO2 at week 0, kept on the normal mouse chow for 1 week, and then put on either a control or 1% DHA diet for 3 weeks. LMP was assessed through detection of (a) IL-1β release directly from AMs ex vivo, (b) cathepsin release within the LLF, and (c) cathepsin B release within the LLF. Data presented as mean ± SEM, n=3–6. *P<0.05, ***P<0.001 ****P<0.0001 compared to DM, + compared to different diet within the same exposure group. Analyses were done with log-transformed data. CTS; cathepsin
Results from the chronic exposure model did not have significant differences between the diets for MWCNT-exposed mice (Fig. 6). SiO2-exposed mice with DHA treatment had a significant decrease of IL-1β release ex vivo from AM when compared to control diet-fed mice (Fig. 6a). The SiO2-exposed mice had a significant increase in both total cathepsin levels and cathepsin B levels when compared to the DM-exposed mice regardless of diet (Fig. 6b,c). These results indicated that mice exposed to SiO2 had decreased phagolysosomal membrane stability; however, DHA was protective by decreasing AM inflammatory signaling as well.
Fig. 6. Assessment of LMP in chronic model.
Balb/c mice were instilled with either DM-only, MWCNT, or SiO2 once a week at weeks 0, 1, 2, 3 while on the normal mouse chow and at week 4 were put on a control or 1% DHA diet for 12 weeks. LMP was assessed through detection of (a) IL-1β release directly from AMs ex vivo, (b) cathepsin release within the LLF, and (c) cathepsin B release within the LLF. Data presented as mean ± SEM, n=4–7. *P<0.05, **P<0.01, ***P<0.001, ****P<0.0001 compared to DM, + compared to different diet within the same exposure group. Analyses were done with log-transformed data. CTS; cathepsin
3.3. DHA shifts macrophage phenotype
A recent in vivo study within our laboratory demonstrated that a 1% DHA diet used as a prophylactic treatment for a 24-hour MWCNT exposure shifted macrophages towards a pro-resolving phenotype in the lung tissue [21]. Various signaling factors; such as cytokines and immune complexes, generate different macrophage phenotypes that are broadly defined as either classically activated, “pro-inflammatory” M1 or alternatively activated, “anti-inflammatory” M2. However, a spectrum of macrophage phenotypes is always present which could be representing hybrid phenotypes rather than individually-defined phenotypes. Extracellular and intracellular signaling factors determine which macrophage phenotype is dominant to regulate the overall response. Depending on the overall environment, macrophages could either be aiding in inflammation or suppressing inflammation through phenotype shifts [24–26]. It is important to understand that this is a snap-shot in time to assess the shifts in phenotypes between particle exposure as well as DHA treatment groups at these particular timepoints. Assessing the effects of DHA on macrophage phenotype when exposed to various particles is lacking in the current literature, especially when analyzing the M2b and M2c subsets since the main focus is usually on the M1 and M2a subsets. Therefore, macrophage phenotypes were assessed within the lung and spleen tissues using sixteen known gene markers in order to determine if exposure to particles caused phenotype shifts and if DHA treatment impacted these alterations. Spleen tissue was included for evaluation in this portion of the study since our laboratory has recently shown trafficking of MWCNT into the spleen resulting in changes of immune cell populations.
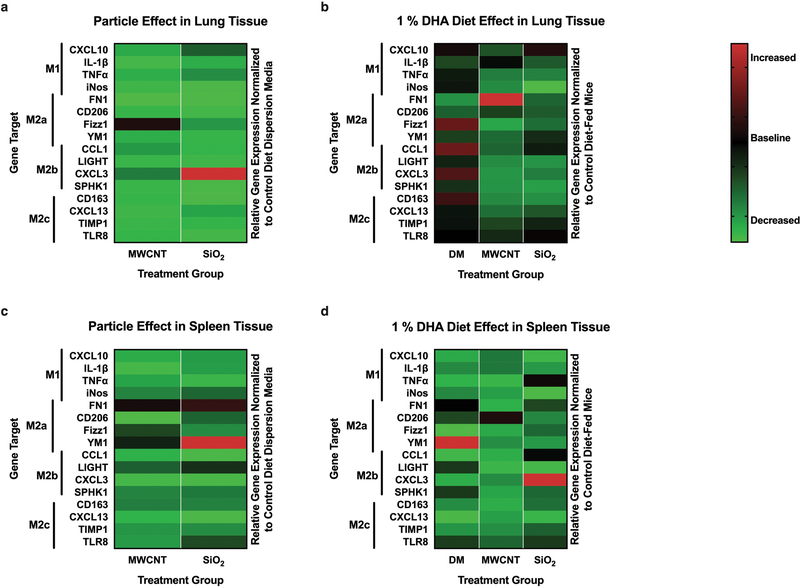
In the lung tissue of the semi-acute exposure model, there was a dominant downregulation of overall gene expression for macrophage phenotype markers for both MWCNT and SiO2-exposed mice when the effect of particle-only exposure was assessed compared to DM-only exposed mice (Fig. 7a). The DHA diet altered this gene expression within the lung tissue by bringing most of those same genes closer to baseline levels (Fig. 7b). Mice exposed to DM-only had a trending upregulation of the M2b phenotype as indicated by an upregulation of two of the four genes with DHA treatment (Fig. 7b). There were dominant baseline gene expression levels of the M2c phenotype for mice exposed to DM-only while on the DHA diet which indicated that these gene expressions were similar for both control- and DHA-fed mice (Fig. 7b). However, this was not the case for mice exposed to either particle upon DHA treatment which were mostly downregulated changes in gene expression and some baseline values (Fig. 7b). Gene expression within the spleen tissue had a dominant downregulation of overall macrophage phenotype markers for MWCNT-exposed mice (Fig. 7c). There was a trending upregulation in the M2a markers for SiO2-exposed mice; however, it was not enough to describe it as a dominant macrophage phenotype shift (Fig. 7c). Upon DHA treatment within the spleen tissue, there were minor gene expression shifts; however, the DHA diet did not alter the overall gene expression of macrophage phenotype markers within the spleen tissue when compared to the particle-only effect (Fig. 7d). DHA treatment did downregulate the trending M2a gene expression of the particle-only effect observed for the SiO2-exposed mice in the spleen tissue (Fig. 7d). These results indicated that particle-exposed mice on a therapeutic 1% DHA diet did not have dominant macrophage phenotype effects in a semi-acute model; however, DHA treatment brought gene expression levels closer to baseline within the lung tissue.
Fig. 7. Impact of particle exposure and DHA treatment on macrophage phenotype in semi-acute model.
Balb/c mice were instilled once with either DM-only, MWCNT, or SiO2 at week 0, kept on the normal mouse chow for 1 week, and then put on either a control or 1% DHA diet for 3 weeks. Relative gene expression of control diet MWCNT or SiO2 mice were normalized to control diet DM-only mice for assessment of particle effect in (a) lung and (c) spleen tissue. Relative gene expression of 1% DHA diet mice were normalized to corresponding control diet mice for assessment of diet effect in (b) lung and (d) spleen tissue. Each bar within the heat maps consist of 5–7 mice for lung tissue and 4–5 mice for spleen tissue
The particle-only effect in the lung tissue of the chronic particle exposure model had an overall downregulation of gene expression within most of the macrophage phenotype markers, regardless of particle exposure (Fig. 8a). Upon DHA diet treatment, there was a general downregulation of gene expression throughout the macrophage phenotype markers regardless of treatment group for the lung tissue (Fig. 8b). There was a trending upregulation of the M2b phenotype with SiO2-exposed mice within the lung tissue with DHA treatment; however, not enough to indicate it as a dominant phenotype shift (Fig. 8b). The spleen tissue assessment of the particle-only effect showed a dominant upregulation of the M2c phenotype for both MWCNT- and SiO2-exposed mice when compared to DM-only mice as indicated by upregulation of at least three of the four genes (Fig. 8c). DHA treatment altered these responses within the spleen tissue via a dominant downregulation of gene expression levels of almost all of the macrophage phenotype markers regardless of particle group (Fig. 8d). These results indicated that a therapeutic 1% DHA diet for chronic particle-exposed mice did not have a dominant macrophage phenotype shift within the lung tissue; however, DHA downregulated gene expression within the spleen tissue.
Fig. 8. Impact of particle exposure and DHA treatment on macrophage phenotype in chronic model.
Balb/c mice were instilled with either DM-only, MWCNT, or SiO2 once a week at weeks 0, 1, 2, 3 while on the normal mouse chow and at week 4 were put on a control or 1% DHA diet for 12 weeks. Relative gene expression of control diet MWCNT or SiO2 mice were normalized to control diet DM-only mice for assessment of particle effect in (a) lung and (c) spleen tissue. Relative gene expression of 1% DHA diet mice were normalized to corresponding control diet mice for assessment of diet effect in (b) lung and (d) spleen tissue. Each bar within the heat maps consist of 5–7 mice for lung tissue and 4–6 mice for spleen tissue
3.4. Effect of DHA on lung histopathology
The therapeutic 1% DHA treatment affected inflammatory signaling cascades for both the semi-acute and chronic particle exposure models. Therefore, investigation of the impact of DHA on particle-induced inflammation for lung pathology was assessed. Lung tissue sections were scored for histological analysis and airway thickness was examined since recent studies in our laboratory using 1% DHA as a prophylactic treatment indicated particle-induced airway thickening.
In the semi-acute model, both the MWCNT- and SiO2-exposed mice had minor differences for both the pathology scoring and airway thickness analyses. SiO2-exposed mice did have a significant increase in airway thickness for both diets when compared to the DM-only exposed mice (Fig. 9b). Representative photomicrographs of these findings are shown in Fig. 9c. These results suggested that a therapeutic 1% DHA treatment had limited effects on particle-induced inflammation and overall lung pathology for a semi-acute particle exposure model.
Fig. 9. Histopathology analysis of lung tissue in semi-acute model.
Balb/c mice were instilled once with either DM-only, MWCNT, or SiO2 at week 0, kept on the normal mouse chow for 1 week, and then put on either a control or 1% DHA diet for 3 weeks. (a) Pathology was scored blinded by two observers; data presented as median ± SEM. (b) Airway thickness was measured blinded; data presented as mean ± SEM. (c) Representative images (10x) of Masson’s trichrome-stained lung airway sections. n=3; **P<0.01 compared to DM. Arrows indicate particle encapsulation, blue staining indicates collagen deposition
For the chronic exposure model, mice exposed to MWCNT and treated with DHA had no significant difference for either pulmonary inflammation or airway thickness when compared to control diet-fed mice (Fig. 10a,b). There was no difference between diets for the pathology scores of SiO2-exposed mice, but there was significantly increased airway thickness for mice on the DHA diet of these mice (Fig. 10b). There appears to be an increase of collagen deposition regardless of particle exposure and treatment group as indicated by the blue staining which could be indicative of pulmonary fibrosis. Representative photomicrographs of these findings are shown in Fig. 10c. These results indicated that a therapeutic 1% DHA treatment was not beneficial for lung histopathology for these chronic particle exposures.
Fig. 10. Histopathology analysis of lung tissue in chronic model.
Balb/c mice were instilled with either DM-only, MWCNT, or SiO2 once a week at weeks 0, 1, 2, 3 while on the normal mouse chow and at week 4 were put on a control or 1% DHA diet for 12 weeks. (a) Pathology was scored blinded by two observers; data presented as median ± SEM. (b) Airway thickness was measured blinded; data presented as mean ± SEM. (c) Representative images (10x) of Masson’s trichrome-stained lung airway sections. n=3; *P<0.05, **P<0.01, ***P<0.001 compared to DM, + compared to different diet within the same exposure group. Arrows indicate particle encapsulation, arrow head indicates increased lung airway thickness, and blue staining indicates collagen deposition
4. Discussion
Research of how DHA; an omega-3 PUFA reported to have anti-inflammatory capabilities, affects particle-induced lung inflammation is currently limited. Our laboratory had previously assessed if DHA could be used as a potential prophylactic treatment for particle-induced inflammation. Those results showed that DHA treatment was more beneficial for a nano-sized MWCNT exposure than a micron-sized SiO2 exposure which may be linked to phagolysosomal membrane stability and macrophage phenotype shifts. Most research is assessing the impacts of DHA as a potential prophylactic treatment; however, therapeutic treatments are lacking for particle-induced inflammatory diseases as well. The goal of these studies was to assess if DHA could potentially assist with treatment, specifically pulmonary inflammation induced by MWCNT or SiO2 in semi-acute and chronic exposure models. Therefore, inflammatory signaling within the LLF, lung histopathology analysis, the occurrence of LMP, and macrophage phenotype shifts were analyzed to address this issue.
Current analysis of inflammatory mediators in the LLF demonstrated that a therapeutic 1% DHA treatment had varying results between the particle exposure models. This continual variance of DHA’s impact between the two different types of particles could be linked to various physiochemical particle characteristics. These variables could be due to particle morphology, size, surface charge, or composition. MWCNT are hydrophobic man-made rolled graphene sheets in the size range of 1–100 nm in at least one dimension with a high surface area per mass [27]. SiO2, on the other hand, is hydrophilic and commonly found in nature as quartz which can be fractured to a respirable size of <10 μm [10, 28]. The current results comparing the DHA and control diets were mostly not statistically significant; however, it appeared that DHA treatment was more protective after the MWCNT exposure rather than the SiO2 exposure. The SiO2-exposed mice on the 1% DHA diet had more of a significant increase of the various inflammatory markers when compared to the DM-exposed mice on the DHA diet; unlike that of the MWCNT-exposed mice on the DHA diet. The MWCNT-exposed mice while on the DHA diet had many trending decreases of various inflammatory markers when compared to the MWCNT-exposed mice on the control diet. When assessing toxicity and cytokine production in the LLF, LDH levels as well as IFNγ and TNFα levels were consistently decreased upon DHA treatment for MWCNT-exposed mice. This trend continued when LMP markers were assessed where mice exposed to MWCNT while on the DHA diet had a decrease in IL-1β release ex vivo for both studies. Furthermore, there was a trending decrease in pulmonary inflammation within the chronic exposure model upon DHA treatment with mice exposed to MWCNT. The length of the DHA treatment appeared to contribute to the varying particle effects within the Balb/c mice as well.
There was a consistency throughout the studies where prolonged treatment of DHA was more promising for the exposure groups than the shorter treatment. Both the basal health levels; as indicated by the DM-only exposed, and the MWCNT-exposed mice had more trending decreased levels of inflammatory cytokines in the LLF upon DHA treatment for the chronic exposure model. The differences seen between the length of DHA treatment for particle effects may partially be due to the time it takes for DHA to equilibrate into the phospholipids. Therefore, if DHA is used as a therapeutic treatment the particle-induced inflammation could persist for a few weeks before beneficial effects of DHA treatment would be observed. Consequently the current semi-acute model may have required a longer timepoint for DHA treatment as a therapeutic in order to see more beneficial effects. The extended incorporation of DHA into phospholipids could alter lipid packing and membrane raft structure in lysosomal membranes which could affect membrane stability and LMP [29, 30]. This observation continued when macrophage phenotype was assessed which indicated a downregulation of gene expression in the spleen tissue with DHA treatment in the chronic particle-exposure study. These results demonstrated that prolonged treatment of DHA; especially when exposed to MWCNT, had the best outcome rather than the semi-acute model which supported the hypothesis. The protective mechanisms of DHA as a therapeutic treatment were still unclear; however, future assessments of DHA’s impact on macrophage metabolism and autophagy should be done in order to determine these potential capabilities.
Various key functions of macrophages are related to singular metabolic signatures [31]. There is an association between PM2.5 exposure and increased incidence of metabolic syndrome-related diseases [32]. The metabolic pathways are different between the macrophage phenotypes. M1 macrophages are associated with glycolysis, disrupted Krebs cycle, and ROS production while M2a macrophages are linked to fatty acid oxidation, lipolysis, and glutaminolysis [31]. Macrophage deletion of Ascl1; which is an enzyme involved in fatty acid synthesis, is associated with decreased IL-1β and TNFα [31]. In the present chronic particle-exposure model, there were decreased levels of both of those cytokines within the LLF after DHA treatment. The NLRP3 inflammasome regulates glycolysis and M1 macrophages have increased glycolysis [33]. The current results showed an increase in various M1-associated markers for chronic particle exposure which were downregulated upon DHA treatment. However; it has been speculated that DHA could be exerting anti-inflammatory effects through preventing macrophage activation [34] which correlated with the current semi-acute particle-exposure model where there was minimal macrophage phenotype shifts. This suggested that longer exposure of DHA treatment as a therapeutic may be more beneficial for particle-induced inflammation.
It has been suggested that M2 macrophage activation may be associated with increased fatty acid oxidation and mitochondrial biogenesis [35]. One study reported that a fish oil diet improved mitochondrial function [36]. Benmoussa et al. showed that the removal of damaged mitochondria via autophagy as well as the inhibition of fatty acid synthase decreased NLRP3 inflammasome activation and IL-1β secretion [35]. However, it has been suggested that impaired lysosome function blocks autophagy [37]. The current results showed that DM-only exposed mice with DHA treatment had trending decreased total cathepsin, cathepsin B, and IL-1β levels which indicates increased stability of the phagolysosomal membrane. Exposure to particles slightly altered these results which could imply impaired lysosome function; however, the particle-exposed mice were slightly protected upon DHA treatment. One study reported that DHA increased autophagosome formation within bone marrow-derived macrophages [38] while another study suggested that DHA-induced activation of p38 MAPK in U937 cells was related to autophagy [39]. It has been shown that SiO2-exposed autophagy-deficient mice had increased lung pathology [40]. It would be interesting to assess the impacts of DHA on autophagy within a particle-exposure model.
Taken together, the current results implied that if assessing a semi-acute vs. chronic exposure timepoint that DHA was more protective as a therapeutic treatment in a chronic particle-exposure model. There were some potentially beneficial outcomes with regard to DHA treatment for MWCNT-exposed mice even though they were mostly not statistically significant when comparing between the two diets. This could be linked to the time it takes for DHA to become incorporated into phospholipids in order to stabilize the phagolysosomal membrane as well as shifts in macrophage phenotype formation with alterations of macrophage metabolism or autophagy. Exposure to SiO2 with DHA treatment appeared to be inflammatory throughout the exposure studies which is most likely due to the particle’s mechanistic properties. DHA could potentially be used as a therapeutic treatment for MWCNT-induced inflammation, but needs to be researched further to assess the impacts on autophagy and metabolism since these results are not fully understood.
Supplementary Material
Acknowledgments
Paige Fletcher was supported by the Ruth L. Kirschstein NRSA Pre-doctoral Fellowship from the National Institute of Environmental Health Sciences under F31 ES028100. This research was supported by the National Institute of Environmental Health Sciences under Grants R01 ES023209 and R01 ES027353 and by the National Institute of General Medical Sciences under Grant P30 GM103338. The content within is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Paige Fletcher was awarded one of QIAGEN’s featured young scientists of the month (November 2018) where she received QIAGEN products that contributed to this research. The authors would like to thank the technical support from the CEHS Core Facilities: Inhalation and Pulmonary Physiology Core, Molecular Histology and Fluorescence Imaging Core, and the Fluorescence Cytometry Core. A special thank you to: Dr. Joanna Kreitinger at Dermaxon and Dr. Sarjubhai Patel at FYR Diagnostics for use of their 384-well CFX Maestro’s; Lou Herritt, and Pamela Shaw within the CEHS Core facilities; UM’s Laboratory Animal Resources technicians and facility; and Iheanyi Amadi for help with lung airway thickness analysis.
5.1. Funding
Paige Fletcher was supported by the Ruth L. Kirschstein NRSA Pre-doctoral Fellowship from the National Institute of Environmental Health Sciences (F31 ES028100). This research was supported by grants from the National Institute of Environmental Health Sciences (R01 ES023209 and R01 ES027353) and National Institute of General Medical Sciences (P30 GM103338). The content within is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Paige Fletcher was awarded one of QIAGEN’s featured young scientists of the month (November 2018) where she received QIAGEN products that contributed to this research.
Footnotes
5.2 Conflicts of interest/Competing interests
The authors have no conflicts of interest or competing interests to declare.
5. Declarations
5.3 Ethics approval
The animal use protocol (035–16AHCEHS-062816) was approved by the University of Montana Institutional Animal Care and Use Committee for all mouse studies described within this manuscript. The mice are maintained in microisolation containers within the BSL-2 Laboratory Animal Resources facility at the University of Montana in the accordance with the Guide for the Care and Use of Laboratory Animals. The animal care facility at the University of Montana is staffed with full-time veterinarians that are AAALAC accredited. Mice were monitored on a daily basis along with during/after exposure to particles. Mice were anesthetized with isoflurane before particle or vehicle control instillations so as not to use any restraints or cause distress. All procedures within these studies caused minimal discomfort to the mice; however, in any cases where it was deemed that the mice were in pain or distress (adverse body weight, abnormal activity, poor grooming, abnormal posture) the animal was humanely euthanized.
5.4 Consent to participate
Not applicable.
5.5 Consent for publication
Not applicable.
5.6 Availability of data and material
Data and material will be provided by the authors if requested.
5.7 Code Availability
Not applicable.
Publisher's Disclaimer: This Author Accepted Manuscript is a PDF file of an unedited peer-reviewed manuscript that has been accepted for publication but has not been copyedited or corrected. The official version of record that is published in the journal is kept up to date and so may therefore differ from this version.
References
- [1].Calder PC Omega-3 fatty acids and inflammatory processes: From molecules to man. Biochemical Society Transactions. 2017;45:1105–1115. 10.1042/BST20160474 [DOI] [PubMed] [Google Scholar]
- [2].Molendi-Coste O, Legry V, & Leclercq IA Why and How Meet n-3 PUFA Dietary Recommendations? Gastroenterology Research and Practice. 2011;364040. 10.1155/2011/364040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Swanson D, Block R, & Mousa SA Omega-3 Fatty Acids EPA and DHA: Health Benefits Throughout Life. Advances in Nutrition. 2012;3:1–7. 10.3945/an.111.000893 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Bates MA, Brandenberger C, Langohr II, Kumagai K, Lock AL, Harkema JR, Holian A, & Pestka JJ Silica-Triggered Autoimmunity in Lupus-Prone Mice Blocked by Docosahexaenoic Acid Consumption. PLOS ONE. 2016;11(8):e0160622. 10.1371/journal.pone.0160622 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Li X-Y, Hao L, Liu Y-H, Chen C-Y, Pai VJ, & Kang JX Protection against fine particle-induced pulmonary and systemic inflammation by omega-3 polyunsaturated fatty acids. Biochimica Et Biophysica Acta. General Subjects. 2017;1861:577–584. 10.1016/j.bbagen.2016.12.018 [DOI] [PubMed] [Google Scholar]
- [6].Forum of International Respiratory Societies. The global impact of respiratory disease. 2nd ed. Sheffield, European Respiratory Society; 2017. [Google Scholar]
- [7].Wong J, Magun BE, & Wood LJ Lung inflammation caused by inhaled toxicants: A review. International Journal of Chronic Obstructive Pulmonary Disease. 2016;11:1391–1401. 10.2147/COPD.S106009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Lam C, James JT, McCluskey R, Arepalli S, & Hunter RL A Review of Carbon Nanotube Toxicity and Assessment of Potential Occupational and Environmental Health Risks. Critical Reviews in Toxicology. 2006;36:189–217. 10.1080/10408440600570233 [DOI] [PubMed] [Google Scholar]
- [9].Ray JL and Holian A Sex differences in the inflammatory immune response to multi-walled carbon nanotubes and crystalline silica. Inhal. Toxicol 2019;31(7):285–297. 10.1080/08958378.2019.1669743 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Pollard KM Silica, Silicosis, and Autoimmunity. Frontiers in Immunology. 2016;7(97). 10.3389/fimmu.2016.00097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Pavan C, Santalucia R, Leinardi R, Fabbiani M, Yakoub Y, …, & Fubini B Nearly free surface silanols are the critical molecular moieties that initiate the toxicity of silica particles. Proc. Natl. Acad. Sci 2020;117(45):27836–27846. 10.1073/pnas.2008006117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Watanabe H, Numata K, Ito T, Takagi K, & Matsukawa A Innate immune response in TH1- and TH2-dominant mouse strains. Shock. 2004;22(5):460–466. 10.1097/01.shk.0000142249.08135.e9 [DOI] [PubMed] [Google Scholar]
- [13].Limjunyawong N, Craig JM, Lagasse HAD, Scott AL, & Mitzner W Experimental progressive emphysema in BALB/cJ mice as a model for chronic alveolar destruction in humans. American Journal of Physiology-Lung Cellular and Molecular Physiology. 2015;309:L662–L676. 10.1152/ajplung.00214.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Sahu N, Morales JL, Fowell D, & August A Modeling Susceptibility versus Resistance in Allergic Airway Disease Reveals Regulation by Tec Kinase Itk. PLoS ONE. 2010;5(6):e11348. 10.1371/journal.pone.0011348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Hamilton RF, Buford M, Xiang C, Wu N, & Holian A NLRP3 inflammasome activation in murine alveolar macrophages and related lung pathology is associated with MWCNT nickel contamination. Inhalation Toxicology. 2012;24(14):995–1008. 10.3109/08958378.2012.745633 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Thakur SA, Hamilton R, Pikkarainen T, & Holian A Differential Binding of Inorganic Particles to MARCO. Toxicological Sciences. 2009;107(1):238–246. 10.1093/toxsci/kfn210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific Opinion on the extension of use for DHA and EPA-rich algal oil from Schizochytrium sp. as a novel food ingredient. European Food Safety Authority Journal. 2014;12(10):3843. 10.2903/j.efsa.2014.3843 [DOI] [Google Scholar]
- [18].Jessop F, Hamilton RF, Rhoderick JF, Fletcher P, & Holian A Phagolysosome acidification is required for silica and engineered nanoparticle-induced lysosome membrane permeabilization and resultant NLRP3 inflammasome activity. Toxicology and Applied Pharmacology. 2017;318:58–68. 10.1016/j.taap.2017.01.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Jessop F, & Holian A Extracellular HMGB1 regulates multi-walled carbon nanotube-induced inflammation in vivo. Nanotoxicology. 2015;9(3):365–372. 10.3109/17435390.2014.933904 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Burmeister R, Rhoderick JF, & Holian A Prevention of crystalline silica-induced inflammation by the antimalarial hydroxychloroquine. Inhalation Toxicology. 2019;31(7):274–284. 10.1080/08958378.2019.1668091 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Fletcher P, Hamilton RF, Rhoderick JF, Pestka JJ, & Holian A Docosahexaenoic acid impacts macrophage phenotype subsets and phagolysosomal membrane permeability with particle exposure. Journal of Toxicology and Environmental Health, Part A. 2020;1–21. 10.1080/15287394.2020.1842826 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Schroder K and Tschopp J The inflammasomes. Cell. 2010;140:821–832. 10.1016/j.cell.2010.01.040 [DOI] [PubMed] [Google Scholar]
- [23].Strowig T, Henao-Mejia J, Elinav E, & Flavell R Inflammasomes in health and disease. Nature. 2012;481:278–286. 10.1038/nature10759 [DOI] [PubMed] [Google Scholar]
- [24].Labonte AC, Tosello-Trampont A-C, & Hahn YS The role of macrophage polarization in infectious and inflammatory diseases. Molecules and Cells. 2014;37(4):275–285. 10.14348/molcells.2014.2374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Italiani P, & Boraschi D From Monocytes to M1/M2 Macrophages: Phenotypical vs. Functional Differentiation. Frontiers in Immunology. 2014;5(514). 10.3389/fimmu.2014.00514 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Byrne AJ, Mathie SA, Gregory LG, & Lloyd CM Pulmonary macrophages: Key players in the innate defence of the airways. Thorax. 2015;70:1189–1196. 10.1136/thoraxjnl-2015-207020 [DOI] [PubMed] [Google Scholar]
- [27].Saifuddin N, Raziah AZ, & Junizah AR Carbon Nanotubes: A Review on Structure and Their Interaction with Proteins. Journal of Chemistry. 2013;676815. 10.1155/2013/676815 [DOI] [Google Scholar]
- [28].Dyachenko AG, Borysenko MV, & Pakhovchyshyn SV Hydrophilic/Hydrophobic Properties of Silica Surfaces Modified with Metal Oxides and Polydimethylsiloxane. Adsorption Science & Technology. 2004;22(6):511–516. 10.1260/0263617042879546 [DOI] [Google Scholar]
- [29].Turk HF, & Chapkin RS Membrane lipid raft organization is uniquely modified by n-3 polyunsaturated fatty acids. Prostaglandins, Leukotrienes and Essential Fatty Acids. 2013;88(1):43–47. 10.1016/j.plefa.2012.03.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Wassall SR, Leng X, Canner SW, Pennington ER, Kinnun JJ, Cavazos AT, Dadoo S, Johnson D, Heberle FA, Katsaras J, & Shaikh SR Docosahexaenoic acid regulates the formation of lipid rafts: A unified view from experiment and simulation. Biochimica et Biophysica Acta (BBA) - Biomembranes. 2018; 1860(10): 1985–1993. 10.1016/j.bbamem.2018.04.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Stunault MI, Bories G, Guinamard RR, & Ivanov S Metabolism Plays a Key Role during Macrophage Activation. Mediators of Inflammation. 2018;2426138. 10.1155/2018/2426138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Li R, Qiu X, Xu F, Lin Y, Fang Y, & Zhu T Macrophage-Mediated Effects of Airborne Fine Particulate Matter (PM 2.5 ) on Hepatocyte Insulin Resistance in Vitro. ACS Omega. 2016;1:736–743. 10.1021/acsomega.6b00135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Finucane OM, Sugrue J, Rubio-Araiz A, Guillot-Sestier M-V, & Lynch MA The NLRP3 inflammasome modulates glycolysis by increasing PFKFB3 in an IL-1β-dependent manner in macrophages. Scientific Reports. 2019;9:4034. 10.1038/s41598-019-40619-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Ali M, Heyob K, & Rogers LK DHA Suppresses Primary Macrophage Inflammatory Responses via Notch 1/ Jagged 1 Signaling. Scientific Reports. 2016;6:22276. 10.1038/srep22276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Benmoussa K, Garaude J, & Acín-Pérez R How Mitochondrial Metabolism Contributes to Macrophage Phenotype and Functions. Journal of Molecular Biology. 2018;430:3906–3921. 10.1016/j.jmb.2018.07.003 [DOI] [PubMed] [Google Scholar]
- [36].Shen L, Yang Y, Ou T, Key C-CC, Tong SH, … & Zhu X Dietary PUFAs attenuate NLRP3 inflammasome activation via enhancing macrophage autophagy. Journal of Lipid Research. 2017;58:1808–1821. 10.1194/jlr.M075879 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Saitoh T, Fujita N, Jang MH, Uematsu S, Yang B-G, …, & Akira S Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1β production. Nature. 2008;456:264–268. 10.1038/nature07383 [DOI] [PubMed] [Google Scholar]
- [38].Williams-Bey Y, Boularan C, Vural A, Huang N-N, Hwang I-Y, Shan-Shi C, & Kehrl JH Omega-3 Free Fatty Acids Suppress Macrophage Inflammasome Activation by Inhibiting NF-κB Activation and Enhancing Autophagy. PLoS ONE. 2014;9(6):e97957. 10.1371/journal.pone.0097957 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Kawano A, Ariyoshi W, Yoshioka Y, Hikiji H, Nishihara T, & Okinaga T Docosahexaenoic acid enhances M2 macrophage polarization via the p38 signaling pathway and autophagy. Journal of Cellular Biochemistry. 2019;120:12604–12617. 10.1002/jcb.28527 [DOI] [PubMed] [Google Scholar]
- [40].Jessop F, Hamilton RF, Rhoderick JF, Shaw PK, & Holian A Autophagy deficiency in macrophages enhances NLRP3 inflammasome activity and chronic lung disease following silica exposure. Toxicology and Applied Pharmacology. 2016;309:101–110. 10.1016/j.taap.2016.08.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.