Abstract
Background and aims
Initially, novel severe acute respiratory syndrome coronavirus (SARS-CoV-2) was considered primarily a respiratory pathogen. However, with time it has behaved as a virus with the potential to cause multi-system involvement, including neurological manifestations. Cerebral venous sinus thrombosis (CVT) has increasingly been reported in association with coronavirus infectious disease of 2019 (COVID-19). Here, we have shed light upon CVT and its possible mechanisms in the backdrop of the ongoing COVID-19 pandemic.
Methods
In this review, data were collected from PubMed, EMBASE and Web of Science, until March 30, 2021, using pre-specified searching strategies. The search strategy consisted of a variation of keywords of relevant medical subject headings and keywords, including “COVID-19”, “SARS-CoV-2”, “coronavirus”, and “cerebral venous sinus thrombosis”.
Results
COVID-19 has a causal association with a plethora of neurological, neuropsychiatric and psychological effects. CVT has gained particular importance in this regard. The known hypercoagulable state in SARS-CoV-2 infection is thought to be the main mechanism in COVID-19 related CVT. Other plausible mechanisms may include vascular endothelial dysfunction and altered flow dynamics.
Conclusions
Although there are no specific clinical characteristics, insidious or acute onset headache, seizures, stroke-like, or encephalopathy symptoms in a patient with, or who has suffered COVID-19, should prompt the attending physician to investigate for CVT. The treatment of COVID-19 associated CVT does not differ radically from the therapy of CVT without the infection, i.e. urgent initiation of parenteral unfractionated heparin or low molecular weight heparin followed by conventional or mostly newer oral anticoagulants.
Keywords: COVID-19, SARS-CoV-2, Cerebral venous thrombosis, Neuro-COVID
1. Background and Aims
Although novel severe acute respiratory syndrome coronavirus (SARS-CoV-2) was primarily considered a respiratory pathogen, it has revealed to have the enormous capability in giving rise to umpteen numbers of neurological manifestations [1]. It has affected all parts of the cranio-spinal and neuromuscular axes [[1], [2], [3], [4], [5]]. SARS-CoV-2 infection has immense potential to predispose infected individuals to develop arterial and venous thrombosis anywhere in the body due to endothelial dysfunction [1,6,7], hyperinflammatory state [6,8], platelet activation [6,9], and vascular stasis [6,10] i.e., activation of the Virchow's triad [11].
Complete or partial occlusion of either the main sinus/sinuses or the feeding cortical veins is referred to as cerebral venous sinus thrombosis (CVT), which can lead to secondary effects of vascular congestion and focal or generalized neurological deficits [12]. Manifestations of CVT are heterogeneous including headache, altered consciousness, behavioural abnormality, seizures, speech difficulties and motor paresis [12,13].
Activation of Virchow's triad can consequently lead to the generation of thrombi in venous channels draining the brain parenchyma [11,12]. One of the pivotal risk factors for the development of CVT has been a hypercoagulable state [[14], [15], [16], [17], [18]]. Mutations in genes encoding coagulation factors [16], increased basal production of these factors in certain physiological, pathological states and certain malignancies, as well as autoimmune disorders, lead to an imbalance between prothrombic and antithrombotic factors [[14], [15], [16], [17], [18]]. Infection has always been a risk factor for the development of CVT particularly in Asian populations [19,20]. Infection itself or infection-related alteration in the immune and coagulation system may be at the root of this [14,16,19,20].
Amongst various neurological manifestations of coronavirus infectious disease of 2019 (COVID-19), CVT has gained recent importance. Multiple reports of SARS-CoV-2 associated with CVT have come to surface and continue to do so [[21], [22], [23], [24], [25], [26], [27], [28], [29], [30], [31]]. Whether COVID-19 is truly a causative factor or just a mere association remains elusive [32].
In this review article, the authors will try to put some light upon the ‘missing links’ between SARS-CoV-2 infection and CVT.
2. Methods
In this review, data were collected from PubMed, EMBASE and Web of Science, until March 30, 2021, using pre-specified searching strategies. The search strategy consisted of a variation of keywords of relevant medical subject headings and keywords, including “COVID-19”, “SARS-CoV-2”, “coronavirus”, and “cerebral venous sinus thrombosis”.
3. Results
3.1. Infectious diseases and CVT
The prevalence of infectious disease associated with CVT has declined over the years, mainly due to empirical use of sensitive antibiotics, early detection with various newer techniques and prompt management [20]. In developing and underdeveloped countries, CVT is still found to be primarily or secondarily associated with focal or systemic infection [19,[33], [34], [35], [36], [37], [38]]. However, the associated morbidity and mortality have decreased substantially [29]. Various organisms, e.g., viruses [[39], [40], [41], [42], [43], [44], [45], [46], [47], [48], [49]], tubercular [50,51], bacteria [[52], [53], [54], [55], [56], [57], [58], [59]], protozoa and other parasitic infections [[60], [61], [62], [63]] have led to the development of CVT. Infection, when considered to be a direct cause of CVT, can spread from structures either adjacent to dura matter (e.g., paranasal sinuses, nose, mastoid and orbit) or from a distant locus. Besides, and perhaps on most occasions, infection triggers an inflammatory response in the system and secondarily leads to the generation of CVT. Heller et al. [64] concluded that CVT was a multifactorial disease, in which the majority of the cases result from a combination of prothrombotic risk factors and underlying clinical conditions, which may be an infection.
3.2. COVID-19 and CVT
As previously discussed, the association of CVT with COVID-19 has attracted interest from neurologists. Table 1 summarizes the clinical characteristics of the CVT cases associated with CVT that have been reported [[21], [22], [23], [24], [25], [26], [27],32,[65], [66], [67], [68], [69], [70], [71], [72], [73], [74], [75]]. Hughes et al. [21] reported the first case of CVT in an apparently healthy individual, without inherited or acquired risk factors other than the COVID-19 infection itself, who recovered fully with standard therapy with low molecular weight heparin and apixaban. It was concluded that this index case of CVT (presented with headache and later motor weakness and dysphasia) resulted from a hypercoagulable milieu generated by SARS-CoV-2 infection [21]. Klein et al. [24] also put forward a COVID-19 induced hypercoagulable state as the culprit for the development of hemorrhagic CVT in a young female with thalassemia trait who presented with seizures (and later developed mixed aphasia and bilateral abducens nerve palsy) and who responded well to anticoagulants and antiepileptics. Despite this case having a beta/delta thalassemia trait, raised IgM-anti-cardiolipin antibody titer and iron deficiency anemia (which can give rise to hypercoagulable state), led to the clinical diagnosis of COVID-19 induced CVT [24]. Cavalcanti et al. [25] reported three cases of COVID-19 associated with CVT with worst outcome. All these three patients were young (less than 41 years old) and the interval from infection to the thrombotic event was 2–7 days. Although the authors of the series put blame on the COVID-19 infection, there remained windows of doubt as one of the patients had a history of using oral contraceptives and none of them were screened for genetic thrombophilia profile [25]. The report by Bolaji et al. [26] described a similar case of a 63-year-old previously healthy man with no established risk factors for CVT manifesting with hemiparesis, seizures, and coma early in the course of the disease. This individual recovered well with anticoagulants, antiepileptics, invasive ventilation and rehabilitation therapy [26]. A Spanish report delineated a similar case of COVID-associated CVT with a good outcome who presented with motor paresis and focal seizures [27]. A similar report from Iran described a case of acute onset seizure as a presenting manifestation of COVID-19 induced CVT [22]. The authors ascribed the phenomenon to SARS-CoV-2 induced changes in coagulation cascade and clot formation [22,76,77].
Table 1.
Clinical characteristics of the central venous thrombosis cases in COVID-19.
| Authors | Age/Gender | Comorbidities | Time to central venous thrombosis diagnosis after COVID-19 onset | Treatment | Outcome |
|---|---|---|---|---|---|
| Hughes et al. [21] | 59/M | Non-insulin dependent diabetes mellitus and arterial hypertension | Four days | Low molecular weight heparin | Recovery |
| Hemasian & Ansari [22] | 65/M | – | Four days | Anticoagulant, levetiracetam, hydroxychloroquine and amoxicillin/clavulanic acid | Recovery after 10 days |
| Essajee et al. [23] | Two years and seven months/F | Tuberculosis | Eight days | Aspirin | Recovering with physiotherapy and occupational therapy |
| Klein et al. [24] | 29/F | – | One week | Intravenous heparin, enoxaparin and antiepileptics | Improved clinically |
| Cavalcanti et al. [25] | 38/M | Mild autism spectrum disorder | One week | Enoxaparin and venous thrombectomy | Deceased |
| 41/F | – | Not mentioned | Heparin infusion | Deceased | |
| 23/M | Diabetes ketoacidosis | Not mentioned | Deceased | ||
| Bolaji et al. [26] | 63/M | – | One week | Low molecular weight heparin and levetiracetam | Improved |
| Dahl-Cruz et al. [27] | 53/F | – | One week | Low molecular weight heparin and antiepileptic therapy | Clinically stable, discharged after 10 days |
| Poillon et al. [32] | 62/F | Morbid obesity | 15 days | – | Not reported |
| 54/F | Breast cancer in remission on hormone therapy | Two weeks | – | Not reported | |
| Abouhashem et al. [65] | 22/M | – | One week | Deceased | |
| 28/M | – | Not mentioned | Low molecular weight heparin and levetiracetam | Deceased | |
| Dakay et al. [66] | 17/M | Obesity | One week | Anticoagulant | Improved |
| 72/F | Breast cancer on remission | Three days | Deceased | ||
| 26/M | – | Two weeks | Hemiparesis | ||
| Garaci et al. [67] | 44/F | – | Two weeks | Tocilizumab and nadroparin | Not reported |
| Hoelscher et al. [68] | 54/M | – | 15 days | – | Condition continued to decline |
| Sugiyama et al. [69] | 56/M | – | Eight days | Unfractionated heparin and edoxaban | Improved |
| Chougar et al. [70] | 72/M | – | Eight days | Anticoagulation and antiepileptic therapy | Deceased |
| Aghayari et al. [71] | 43/F | – | Eight days | Levetiracetam, hydroxychloroquine, low molecular weight heparin, oseltamivir and ceftriaxone | Improved |
| Khacha et al. [72] | 55/M | – | A few days | Infused heparin and acenocoumarol | Improved |
| Mowla et al. [73] | 13 patients | – | A few days | Anticoagulant therapy | Six patients discharged with good outcomes and three died |
| Nwajei et al. [74] | 68/F | – | 18 days | Unfractionated heparin | Improved |
| 79/F | Arterial hypertension | Three days | Low molecular weight heparin | Improved | |
| 25/F | Steroid/intravenous immunoglobulin refractory idiopathic thrombocytopenic purpura, lupus anticoagulant, von-Willebrand disease and Evans syndrome | Four months | Unfractionated heparin | Improved | |
| Hussain et al. [75] | 30/M | – | Eight days | Levetiracetam, low molecular weight heparin and rivaroxaban | Stable throughout admission |
3.3. Possible pathogenetic mechanisms of CVT in COVID-19
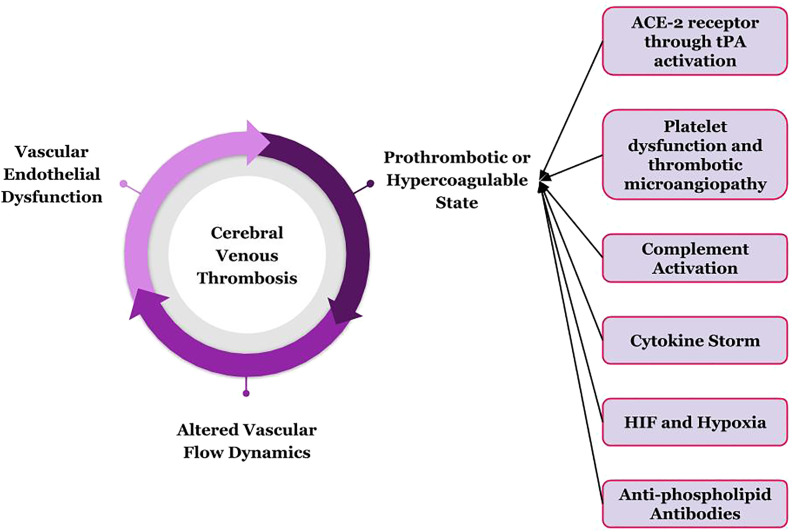
COVID-19 is appropriately being considered a prothrombotic disease [78], though the exact and detailed pathogenesis is yet to be elucidated. Endothelial damage, altered blood flow pattern, hypercoagulable state and hyper-inflammation lead to venous and arterial thromboses in COVID-19 (Fig. 1 ) The authors have hereby tried to unfurl the principles of thrombosis in COVID-19 infection.
-
A)
Vascular endothelial dysfunction in backdrop of COVID-19
Fig. 1.
Possible mechanisms through which SARS-CoV-2 can lead to cerebral venous thrombosis.
Tissue plasminogen activators (tPA) are secreted by vascular endothelium, which hinders the initiation of coagulation cascade by preventing fusion of platelets [79,80]. History reveals that in the previous SARS-CoV infection pandemic, both the virion and the ACE-2 receptor was found in endothelial cells [81,82]. Similarly, recent electron microscopic studies have found SARS-CoV-2 virion-like particle in endothelium and endothelial damage [83,84]. Thus, the dysfunction of the fibrinolytic system results from dysfunctional endothelium, predisposing the vessels for thrombus formation [85]. Besides, in SARS-CoV-2 infection, unopposed emission of von-Willebrand factor occurs, which is otherwise stored in endothelial cells [86]. While these factors initiate thrombosis, propagation of the thrombus is aided by inflammation, sometimes even cytokine storm, in the backdrop of COVID-19 [87]. Another notable postulated mechanism of endothelial damage is through immune complex-mediated vasculitis, but evidence is lacking to support this theory [88]. Again, activation of coagulation stairs can result in disseminated intravascular coagulation and sepsis-induced coagulopathy [89].
-
B)
Altered flow dynamics in vessels of SARS-CoV-2 infected patients
Hyperviscosity, being a potent thrombogenic factor itself, can also induce endothelial damage and dysfunction, and has been an established complication of COVID-19 [90,91]. High fibrinogen levels, one of the major determinants of plasma viscosity, have been associated with SARS-CoV-2 infection [76,92,93]. Impaired microcirculation remains a hallmark of COVID-19 infection and this occurs due to the non-laminar type of flow in these areas together with micro-thrombi occluding newly formed vessels [94]. Turbulent flow in the pre-existing aneurysms on top of a thrombophilic milieu of COVID-19 can lead to aneurysmal rupture and catastrophe [95,96].
-
C)
Hypercoagulable/Prothrombotic state in the backdrop of COVID-19
3.3.1. Role of ACE-2 receptors
ACE-2 promotes the thrombolysis by activation of tPA, whereas angiotensin-II/AT-1R activation leads to genesis and release of plasminogen activator inhibitor-1 (PAI-1) from vascular smooth muscle cells and endothelium [97,98]. And thus, internalization and lysosomal degradation of ACE-2 receptors in COVID-19 infection evidently reset PAI-1/tPA balance to a prothrombotic state [99,100]. Besides, activities of the renin-angiotensin-aldosterone-system are negatively controlled by the ACE-2 via activation of anti-thrombogenic ACE-2-angiotensin-(1–7)-Mas receptor axis [101]. Thus, the absence of ACE-2 receptors can lead to excess thrombus formation [101].
3.3.2. Dysfunction of platelets
The activation of the angiotensin-AT-1R pathway and suppression of the Mas-receptor pathway lead to platelet activation and release of PAI-1 from platelets, increased platelet adherence and aggregation, and decreased nitric oxide release, leading to thrombotic microangiopathy [[102], [103], [104], [105]]. Again, loss of protective ACE-2 can indirectly result in activation of platelets via the bradykinin system [106]. Certainly, platelet dysfunction has a role to play in COVID-19 induced thrombotic manifestations, i.e. CVT.
3.3.3. Activation of complement system and the cytokine storm
The complement system activation is a double-edged sword, as, on one hand, the innate immune response must combat the viral infection, and on the other hand, an unrestrained activation results in inflammation and cell death. Indeed, the terminal products of the activated complement cascade induce prostaglandin and leukotriene synthesis, adding to the increased production of pro-inflammatory cytokines [107]. Again, the complement system and the coagulation cascade are closely interlinked. In skin biopsies of several patients, complement activation has been demonstrated, which can be linked to micro-thrombosis [108]. The hyper-immune response in COVID-19 itself can lead to activation of complements, coagulation cascade, platelet activation and lead to widespread thrombosis by culminating into disseminated intravascular coagulation and sepsis-induced coagulopathy or macrophage activation-like syndrome [[109], [110], [111]]. Eculizumab has shown some efficacy in treating selected patients of COVID-19 infection [112].
3.3.4. Generation of hypoxia inducible factors (HIF) in background of hypoxia
SARS-CoV-2 being primarily a respiratory pathogen, induces fatal hypoxemia in the most severe of the cases that again may result in vasoconstriction, reduced blood flow, vascular stasis and vessel occlusion [109]. Another feasible theory lies that this hypoxemia prompts the activation of several hypoxemia inducible factors (HIF), mainly HIF-2α, which is known to induce or inhibit several important genes of the normal haemostatic mechanisms [109].
3.3.5. Generation of anti-phospholipid antibodies
Genesis of anti-phospholipid antibodies has been reported in COVID-19 infection. But whether this has predisposing role in development of thrombosis remain unanswered [[113], [114], [115]].
3.4. Insights for primary care physicians from the pandemic: authors’ perspective
COVID-19 infection heralds a hypercoagulable and prothrombotic state, which may give rise to widespread thrombosis and thromboembolism involving vessels of all calibres. The virus itself, the resulting hypercytokinemia, changes in flow dynamics in microcirculatory levels, endothelial and platelet dysfunction, and induction of changes at genetic and molecular levels are responsible for genesis and perpetuation of thrombus.
Regarding clinical manifestations, COVID-19 related CVT showed no special characteristics worth mentioning. Clinical spectrum included either encephalopathy symptoms (altered consciousness, widespread neurological signs and coma), symptoms of isolated intracranial hypertension (headache, visual symptoms and papilloedema) or focal deficits (seizures, aphasia and motor weakness, among others). It is, however, worth mentioning that when there is clinical suspicion, appropriate investigations, especially neuroimaging must be done to pinpoint the diagnosis and initiate therapy accordingly. Given that the symptoms in both COVID-19 and CVT are similar, it could complicate things further for the clinicians in the emergency department. There is no specific neurologic symptom of CVT and primary care professionals should be trained adequately for managing these cases with consultation from specialists on as-needed basis. Insidious or acute onset headache, seizures, stroke-like symptoms or encephalopathy should prompt the attending physician to investigate for CVT. From personal experience as frontline COVID-19 healthcare workers from a developing country, we wish to share our concern regarding lack of scope for appropriate and timely investigations and interventions for patients who are having critical non-respiratory illness due to or associated with SARS-CoV-2 infection. Primary care frontiers should take any acute onset neurological symptom as a “red flag” in the backdrop of the ongoing pandemic and they must be provided with a battery of investigations and advanced neuroimaging techniques apart from routine blood parameters and computed tomography scans.
Regarding neuroimaging features of patients with SARS-CoV-2 related CVT, most displayed hemorrhagic venous infarcts as the first imaging feature. Although it is quite a common finding even in other non-COVID-19-CVT cases, the data is too scant to make any strong statement regarding it. For primary care frontiers, a hemorrhagic stroke in an atypical location without a clear cause should raise the suspicion of CVT and should guide the attending physician for further investigations.
The treatment of COVID-19 associated CVT does not differ radically from therapy of CVT without the infection, at least at this point of time. Large multicentre controlled trials are needed to build up a consensus guideline regarding treatment plan of CVT associated with SARS-CoV-2 infection. For now, urgent initiation of parenteral unfractionated heparin or low molecular weight heparin should be administered in higher doses followed by conventional or mostly newer oral anticoagulants for varied duration. Associated morbidities like paresis, aphasia and seizures should be taken care of with help of specific drugs and rehabilitation. It has been observed that those individuals who did not have one or more inherited or acquired risk factor(s) for CVT, outcome with therapy was better compared to others whose genetic and pre-existing acquired thrombophilia profile were unknown. Whether associated genetic predisposition for thrombophilia or infection associated complement activation need specific therapy is beyond making comment at such an early date. While treating patients of severe COVID-19 with parenteral anticoagulants, vigilant watch for development of heparin induced thrombocytopenia must be also kept in mind.
4. Conclusions
COVID-19 manifests in various neurological complications so far. CVT has gained particular importance in this regard. Multiple reports of SARS-CoV-2 associated with CVT have come to surface. Regarding pathophysiology, mechanisms can be attributed to ACE-2 receptors, platelet dysfunction, hypoxia and HIF, and the derangement of the complement system. However, whether COVID-19 is truly the causative factor participating through these mechanistic pathways or just a mere association remains elusive. Although there are no specific clinical characteristics, insidious or acute onset headache, seizures, stroke-like, or encephalopathy symptoms in a patient with, or who has suffered COVID-19, should prompt the attending physician to investigate for CVT. The treatment of COVID-19 associated CVT does not differ radically from the therapy of CVT without the infection, i.e. urgent initiation of parenteral unfractionated heparin or low molecular weight heparin followed by conventional or mostly newer oral anticoagulants.
Disclosures
R. Ghosh reports no disclosures relevant to the manuscript.
D. Roy reports no disclosuresrelevant to themanuscript
A. Mandal reports no disclosures relevant to the manuscript.
S. K. Pal reports no disclosures relevant to the manuscript.
B. C. Swaika reports no disclosures relevant to the manuscript.
D. Naga reports no disclosures relevant to the manuscript.
A. Pandit reports no disclosures relevant to the manuscript.
B. K. Ray reports no disclosures relevant to the manuscript.
J. Benito-León reports no disclosures relevant to the manuscript.
Declaration of competing interest
The authors declare no conflicts of interest.
References
- 1.Roy D., Ghosh R., Dubey S., Dubey M.J., Benito-León J., Kanti Ray B. Neurological and neuropsychiatric impacts of COVID-19 pandemic. Can J Neurol Sci. 2021;48(1):9–24. doi: 10.1017/cjn.2020.173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ghosh R., Dubey S., Kanti Ray B., Chatterjee S., Benito-León J. COVID-19 presenting with thalamic hemorrhage unmasking moyamoya angiopathy. Can J Neurol Sci. 2020;47(6):849–851. doi: 10.1017/cjn.2020.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ghosh R., Lahiri D., Dubey S., Ray B.K., Benito-León J. Hallucinatory palinopsia in COVID-19 induced posterior reversible encephalopathy syndrome. J Neuroophthalmol. 2020;40(4):523–526. doi: 10.1097/WNO.0000000000001135. [DOI] [PubMed] [Google Scholar]
- 4.Ghosh R., Roy D., Sengupta S., Benito-León J. Autonomic dysfunction heralding acute motor axonal neuropathy in COVID-19. J Neurovirol. 2020 Dec;26(6):964–966. doi: 10.1007/s13365-020-00908-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ghosh R., Dubey S., Finsterer J., Chatterjee S., Ray B.K. SARS-CoV-2-Associated acute hemorrhagic, necrotizing encephalitis (ahne) presenting with cognitive impairment in a 44-year-old woman without comorbidities: a case report. Am J Case Rep. 2020;21:e925641. doi: 10.12659/AJCR.925641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bikdeli B., Madhavan M.V., Jimenez D., Chuich T., Dreyfus I., Driggin E., et al. Global COVID-19 thrombosis collaborative group. J Am Coll Cardiol. 2020 Jun 16;75(23):2950–2973. doi: 10.1016/j.jacc.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nagashima S., Mendes M.C., Camargo Martins A.P., Borges N.H., Godoy T.M., Afrds M., et al. Endothelial dysfunction and thrombosis in patients with COVID-19-brief report. Arterioscler Thromb Vasc Biol. 2020;Oct;40(10):2404–2407. doi: 10.1161/ATVBAHA.120.314860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ye Q., Wang B., Mao J. The pathogenesis and treatment of the ‘Cytokine Storm’ in COVID-19. J Infect. 2020;80(6):607–613. doi: 10.1016/j.jinf.2020.03.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hottz E.D., Azevedo-Quintanilha I.G., Palhinha L., Teixeira L., Barreto E.A., Pão C.R.R., et al. Platelet activation and platelet-monocyte aggregate formation trigger tissue factor expression in patients with severe COVID-19. Blood. 2020 Sep 10;136(11):1330–1341. doi: 10.1182/blood.2020007252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Manolis A.S., Manolis T.A., Manolis A.A., Papatheou D., Melita H. COVID-19 infection: viral macro- and micro-vascular coagulopathy and thromboembolism/prophylactic and therapeutic management. J Cardiovasc Pharmacol Ther. 2021;26(1):12–24. doi: 10.1177/1074248420958973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kushner A., West W.P. 2020. Triad PLSV. [Google Scholar]
- 12.Capecchi M., Abbattista M., Martinelli I. Cerebral venous sinus thrombosis. J Thromb Haemost. 2018;16(10):1918–1931. doi: 10.1111/jth.14210. [DOI] [PubMed] [Google Scholar]
- 13.Kumral E., Polat F., Uzunköprü C., Callı C., Kitiş Ö. The clinical spectrum of intracerebral hematoma, hemorrhagic infarct, non-hemorrhagic infarct, and non-lesional venous stroke in patients with cerebral sinus-venous thrombosis. Eur J Neurol. 2012;Apr;19(4):537–543. doi: 10.1111/j.1468-1331.2011.03562.x. [DOI] [PubMed] [Google Scholar]
- 14.Ferro J.M., Sousa D. Cerebral venous thrombosis: an update. Curr Neurol Neurosci Rep. 2019;19(10):74. doi: 10.1007/s11910-019-0988-x. [DOI] [PubMed] [Google Scholar]
- 15.Silvis S.M., Middeldorp S., Zuurbier S.M., Cannegieter S.C., Coutinho J.M. Risk factors for cerebral venous thrombosis. Semin Thromb Hemost. 2016;Sep;42(6):622–631. doi: 10.1055/s-0036-1584132. [DOI] [PubMed] [Google Scholar]
- 16.Green M., Styles T., Russell T., Sada C., Jallow E., Stewart J., et al. Non-genetic and genetic risk factors for adult cerebral venous thrombosis. Thromb Res. 2018;169:15–22. doi: 10.1016/j.thromres.2018.07.005. [DOI] [PubMed] [Google Scholar]
- 17.Hisada Y., Mackman N. Cancer-associated pathways and biomarkers of venous thrombosis. Blood. 2017;28(13):1499–1506. doi: 10.1182/blood-2017-03-743211. 130, doi:10.1182-2017-03–743211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schlück E., Rodier G., Derouiche F., Bronner P., Boulay C., Courtois S., et al. Thrombophilies et thromboses veineuses cérébrales [Thrombophilias associated with cerebral venous thrombosis] Rev Neurol (Paris) 2002;158(5 Pt 1):543–552. [PubMed] [Google Scholar]
- 19.Wasay M., Kaul S., Menon B., Dai A.I., Saadatnia M., Malik A., et al. Asian study of cerebral venous thrombosis. J Stroke Cerebrovasc Dis. 2019;Oct;28(10) doi: 10.1016/j.jstrokecerebrovasdis.2019.06.005. [DOI] [PubMed] [Google Scholar]
- 20.Khatri I.A., Wasay M. Septic cerebral venous sinus thrombosis. J Neurol Sci. 2016;362:221–227. doi: 10.1016/j.jns.2016.01.035. [DOI] [PubMed] [Google Scholar]
- 21.Hughes C., Nichols T., Pike M., Subbe C., Elghenzai S. Cerebral venous sinus thrombosis as a presentation of COVID-19. Eur J Case Rep Intern Med. 2020;29(5) doi: 10.12890/2020_001691. 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hemasian H., Ansari B. First case of Covid-19 presented with cerebral venous thrombosis: a rare and dreaded case. Rev Neurol Paris. 2020;Jun;176(6):521–523. doi: 10.1016/j.neurol.2020.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Essajee F., Solomons R., Goussard P., Van Toorn R. Child with tuberculous meningitis and COVID-19 coinfection complicated by extensive cerebral sinus venous thrombosis. BMJ Case Rep. 2020;14(9) doi: 10.1136/bcr-2020-238597. 13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Klein D.E., Libman R., Kirsch C., Arora R. Cerebral venous thrombosis: a typical presentation of COVID-19 in the young. J Stroke Cerebrovasc Dis. 2020;Aug;29(8):104989. doi: 10.1016/j.jstrokecerebrovasdis.2020.104989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cavalcanti D.D., Raz E., Shapiro M., Dehkharghani S., Yaghi S., Lillemoe K., et al. Cerebral venous thrombosis associated with COVID-19. AJNR Am J Neuroradiol. 2020;Aug;41(8):1370–1376. doi: 10.3174/ajnr.A6644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bolaji P., Kukoyi B., Ahmad N., Wharton C. Extensive cerebral venous sinus thrombosis: a potential complication in a patient with COVID-19 disease. BMJ Case Rep. 2020;13(8):e236820. doi: 10.1136/bcr-2020-236820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dahl-Cruz F., Guevara-Dalrymple N., López-Hernández N. Trombosis venosa cerebral e infección por SARS-CoV-2 [Cerebral venous thrombosis and SARS-CoV-2 infection] Rev Neurol. 2020;70(10):391–392. doi: 10.33588/rn.7010.2020204. [DOI] [PubMed] [Google Scholar]
- 28.Sweid A., Hammoud B., Bekelis K., Missios S., Tjoumakaris S.I., Gooch M.R., et al. Cerebral ischemic and hemorrhagic complications of coronavirus disease 2019. Int J Stroke. 2020;15(7):733–742. doi: 10.1177/1747493020937189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kadono Y., Nakamura Y., Ogawa Y., Yamamoto S., Kajikawa R., Nakajima Y., et al. A case of COVID-19 infection presenting with a seizure following severe brain edema. Seizure. 2020;80:53–55. doi: 10.1016/j.seizure.2020.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Warkentin T.E., Kaatz S. COVID-19 versus HIT hypercoagulability. Thromb Res. 2020;196:38–51. doi: 10.1016/j.thromres.2020.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shakibajahromi B., Borhani-Haghighi A., Haseli S., Mowla A. Cerebral venous sinus thrombosis might be under-diagnosed in the COVID-19 era. eNeurologicalSci. 2020;20:100256. doi: 10.1016/j.ensci.2020.100256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Poillon G., Obadia M., Perrin M., Savatovsky J., Lecler A. Cerebral venous thrombosis associated with COVID-19 infection: causality or coincidence? J Neuroradiol. 2020;11(20):S0150–S9861. doi: 10.1016/j.neurad.2020.05.003. 30167-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kalita J., Chandra S., Kumar B., Bansal V., Misra U.K. Cerebral venous sinus thrombosis from a tertiary care teaching hospital in India. Neurologist. 2016;21(3):35–38. doi: 10.1097/NRL.0000000000000079. [DOI] [PubMed] [Google Scholar]
- 34.Javed I., Sultan T., Rehman Z.U., Yaseen M.R. Clinical spectrum and outcome of cerebral venous sinus thrombosis in children. J Coll Physicians Surg Pak. 2018;May;28(5):390–393. doi: 10.29271/jcpsp.2018.05.390. [DOI] [PubMed] [Google Scholar]
- 35.Bhat S.J., Kashyap P.V. Spectrum of cerebral venous thrombosis in uttarakhand. J Assoc Phys India. 2018;66(7):43–47. Jul. PMID:31325261. [PubMed] [Google Scholar]
- 36.Dhadke V.N., Dhadke S.V., Kulkarni A. Clinical profile of cerebral venous sinus thrombosis. J Assoc Phys India. 2020;Mar;68(3):33–35. PMID:32138480. [PubMed] [Google Scholar]
- 37.Coutinho G., Júlio S., Matos R., Santos M., Spratley J. Otogenic cerebral venous thrombosis in children: a review of 16 consecutive cases. Int J Pediatr Otorhinolaryngol. 2018;113:177–181. doi: 10.1016/j.ijporl.2018.07.050. [DOI] [PubMed] [Google Scholar]
- 38.Kojan S., Al-Jumah M. Infection related cerebral venous thrombosis. J Pak Med Assoc. 2006;56(11):494–497. PMID: 17183974. [PubMed] [Google Scholar]
- 39.Siddiqi S.A., Nishat S., Kanwar D., Ali F., Azeemuddin M., Wasay M. Cerebral venous sinus thrombosis: association with primary varicella zoster virus infection. J Stroke Cerebrovasc Dis. 2012;21(8) doi: 10.1016/j.jstrokecerebrovasdis.2012.04.013. 917.e1-4. [DOI] [PubMed] [Google Scholar]
- 40.Leite J., Ribeiro A., Gonçalves D., Sargento-Freitas J., Trindade L., Duque V. Cerebral venous thrombosis as rare presentation of herpes simplex virus encephalitis. Case Rep Infect Dis. 2019;2019:7835420. doi: 10.1155/2019/7835420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Meyohas M.C., Roullet E., Rouzioux C., Aymard A., Pelosse B., Eliasceiwicz M., et al. Cerebral venous thrombosis and dual primary infection with human immunodeficiency virus and cytomegalovirus. J Neurol Neurosurg Psychiatry. 1989;52(8):1010–1011. doi: 10.1136/jnnp.52.8.1010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bialasiewicz S., Hart G., Oliver K., Agnihotri S.P., Koralnik I.J., Viscidi R., et al. A difficult decision: atypical JC polyomavirus encephalopathy in a kidney transplant recipient. Transplantation. 2017;Jun;101(6):1461–1467. doi: 10.1097/TP.0000000000001275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Shan F., Gao C., Long Y., Huang L., Zheng Y., Chen M., et al. Cerebral venous sinus thrombosis may be associated with hepatitis B virus infection: a preliminary finding. Neurol Res. 2015;Jun;37(6):510–513. doi: 10.1179/1743132815Y.0000000003. [DOI] [PubMed] [Google Scholar]
- 44.Mokkappan S., Basheer A., Iqbal N., Chidambaram S. Bilateral thalamic bleed and cerebral venous sinus thrombosis in Japanese encephalitis. BMJ Case Rep. 2015;2015 doi: 10.1136/bcr-2014-207957. bcr2014207957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Neo R.J. Japanese Encephalitis presenting with cerebral venous sinus thrombosis: a case report. Med J Malaysia. 2019;Dec;74(6):537–539. PMID:31929482. [PubMed] [Google Scholar]
- 46.Sardana V., Mittal L.C., Meena S.R., S D., K G. Acute venous sinus thrombosis after chickenpox infection. J Assoc Phys India. 2014;Aug;62(8):741–743. PMID:25856952. [PubMed] [Google Scholar]
- 47.Lal A., Dhamne M.C., Hui A.C., Ahmad A. Herpes simplex virus (HSV) encephalitis in a young man: an unusual course. BMJ Case Rep. 2018;2018 doi: 10.1136/bcr-2017-222499. bcr2017222499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Modi M., Singla V., Ahluwalia J., Sharma A., Prabhakar S., Khandelwal N., et al. HIV infection presenting as cerebral venous sinus thrombosis. Am J Emerg Med. 2012;30(3) doi: 10.1016/j.ajem.2011.01.022. 517.e1-3. [DOI] [PubMed] [Google Scholar]
- 49.Sherman S., Justo D., Engel T., Yossepowitch O., Bregman N., Gadoth A., et al. Cytomegalovirus-associated cerebral sinus vein thrombosis. J Med Virol. 2012;Dec;84(12):1934–1936. doi: 10.1002/jmv.23424. [DOI] [PubMed] [Google Scholar]
- 50.Guenifi W., Boukhrissa H., Gasmi A., Rais M., Ouyahia A., Hachani A., et al. Thromboses veineuses cérébrales au cours de la méningo-encéphalite tuberculeuse [Cerebral venous thrombosis during tuberculous meningoencephalitis] J Mal Vasc. 2016;41(3):210–214. doi: 10.1016/j.jmv.2016.03.002. [DOI] [PubMed] [Google Scholar]
- 51.Sharawat I.K., Bhattacharya D., Saini L., Singh P. Multiple cerebral sinus venous thrombosis and venous infarct: rare complication of tuberculous meningitis in a child. BMJ Case Rep. 2019;12(7):e231419. doi: 10.1136/bcr-2019-231419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wardle M., Mu A., Tong S.Y.C. Streptococcus gallolyticus subsp. pasteurianus meningitis complicated by venous sinus thrombosis: a case report. Int J Infect Dis. 2018;71:30–32. doi: 10.1016/j.ijid.2018.04.005. [DOI] [PubMed] [Google Scholar]
- 53.Pineda M.C., Lopinto-Khoury C. Cerebral venous sinus thrombosis secondary to typhoid fever: a case report and brief summary of the literature. Neurol. 2012;Jul;18(4):202–203. doi: 10.1097/NRL.0b013e31825cf3e1. [DOI] [PubMed] [Google Scholar]
- 54.Bozzola E., Bozzola M., Colafati G.S., Calcaterra V., Vittucci A., Luciani M., et al. Multiple cerebral sinus thromboses complicating meningococcal meningitis: a pediatric case report. BMC Pediatr. 2014;14:147. doi: 10.1186/1471-2431-14-147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Farstad H., Gaustad P., Kristiansen P., Perminov G., Abrahamsen T.G. Cerebral venous thrombosis and Escherichia coli infection in neonates. Acta Paediatr. 2003;92(2):254–257. doi: 10.1111/j.1651-2227.2003.tb00537.x. [DOI] [PubMed] [Google Scholar]
- 56.Mekala S., Delhi Kumar C.G., Gulati R. Antiphospholipid syndrome complicating pneumococcal meningitis. Indian Pediatr. 2018;15(5):429–431. 55, PMID:29845960. [PubMed] [Google Scholar]
- 57.Bodur H., Colpan A., Gozukucuk R., Akinci E., Cevik M.A., Balaban N. Venous sinus thrombosis after Proteus vulgaris meningitis and concomitant Clostridium abscess formation. Scand J Infect Dis. 2002;34:694–696. doi: 10.1080/00365540210147886. [DOI] [PubMed] [Google Scholar]
- 58.Jena S.S., Mathew A., Sanjith A., Ajith S., Nair B.R., Prakash J. Cerebral venous sinus thrombosis presentation in severe scrub typhus infection: a rare entity. Neurol India. 2014;May-Jun;62(3):308–310. doi: 10.4103/0028-3886.136991. [DOI] [PubMed] [Google Scholar]
- 59.Ibrahimagić O.Ć., Smajlović D., Dostović Z., Iljazović A., Kojić B., Zonić L. Neurobrucellosis and cerebral venous sinus thrombosis: a case report. Acta Clin Belg. 2017;72(5):343–345. doi: 10.1080/17843286.2016.1251385. [DOI] [PubMed] [Google Scholar]
- 60.Luvira V., Chamnanchanunt S., Thanachartwet V., Phumratanaprapin W., Viriyavejakul A. Cerebral venous sinus thrombosis in severe malaria. Southeast Asian J Trop Med Publ Health. 2009;Sep;40(5):893–897. PMID: 19842369. [PubMed] [Google Scholar]
- 61.Pinzón M.A., Pineda J.C., Rosso F., Shinchi M., Bonilla-Abadía F. Plasmodium vivax cerebral malaria complicated with venous sinus thrombosis in Colombia. Asian Pac J Trop Med. 2013;13(5):413–415. doi: 10.1016/S1995-7645(13)60050-4. 6. [DOI] [PubMed] [Google Scholar]
- 62.Kersten M.J., Ettekoven C.N., Heijink D.M. Unexpected neurologic complications following a novel lymphoma treatment “expected” to give rise to neurologic toxicity. BMJ Case Rep. 2019;12(11) doi: 10.1136/bcr-2019-229946. 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wu J.Y., Zhang B.R., Zhao G.H. Cerebral infarction and cranial venous sinus thrombosis caused by paragonimiasis. CNS Neurosci Ther. 2013;19(9):734–736. doi: 10.1111/cns.12146. Sep. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Heller C., Heinecke A., Junker R., Knöfler R., Kosch A., Kurnik K., et al. Cerebral venous thrombosis in children: a multifactorial origin. Circulation. 2003;108(11):1362–1367. doi: 10.1161/01.CIR.0000087598.05977.45. [DOI] [PubMed] [Google Scholar]
- 65.Abouhashem S., Eldawoody H., Taha M.M. Cerebral venous sinus thrombosis in patients with COVID-19 infection. Interdiscip Neurosurg. 2021;24:101091. doi: 10.1016/j.inat.2021.101091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Dakay K., Cooper J., Bloomfield J., Overby P., Mayer S.A., Nuoman R., et al. Cerebral venous sinus thrombosis in COVID-19 infection: a case series and review of the literature. J Stroke Cerebrovasc Dis. 2021;30(1):105434. doi: 10.1016/j.jstrokecerebrovasdis.2020.105434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Garaci F., Di Giuliano F., Picchi E., Da Ros V., Floris R. Venous cerebral thrombosis in COVID-19 patient. J Neurol Sci. 2020;414:116871. doi: 10.1016/j.jns.2020.116871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Hoelscher C., Sweid A., Ghosh R., Al Saiegh F., Keppetipola K.M., Farrell C.J., et al. Cerebral deep venous thrombosis and COVID-19: case report. J Neurosurg. 2020 Sep 4:1–4. doi: 10.3171/2020.5.JNS201542. [DOI] [PubMed] [Google Scholar]
- 69.Sugiyama Y., Tsuchiya T., Tanaka R. Cerebral venous thrombosis in COVID-19-associated coagulopathy: a case report. J Clin Neurosci. 2020;79:30–32. doi: 10.1016/j.jocn.2020.07.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Chougar L., Mathon B., Weiss N., Degos V., Shor N. Atypical deep cerebral vein thrombosis with hemorrhagic venous infarction in a patient positive for COVID-19. AJNR Am J Neuroradiol. 2020;41:1377–1379. doi: 10.3174/ajnr.A6642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Aghayari Sheikh Neshin S., Basirjafari S., Saberi A., Shahhosseini B., Zarei M. Liver abnormality may develop cerebral vein thrombosis in COVID-19. J Neurol Sci. 2020;417:117076. doi: 10.1016/j.jns.2020.117076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Khacha A., Bouchal S., Ettabyaoui A., Haloua M., Lamrani Y.A., Boubbou M., et al. Cavernous sinus thrombosis in a COVID-19 patient: a case report. Radiol Case Rep. 2021;16:480–482. doi: 10.1016/j.radcr.2020.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Mowla A., Shakibajahromi B., Shahjouei S., Borhani-Haghighi A., Rahimian N., Baharvahdat H., et al. Cerebral venous sinus thrombosis associated with SARS-CoV-2; a multinational case series. J Neurol Sci. 2020;419:117183. doi: 10.1016/j.jns.2020.117183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Nwajei F., Anand P., Abdalkader M., Andreu Arasa V.C., Aparicio H.J., Behbahani S., et al. Cerebral venous sinus thromboses in patients with SARS-CoV-2 infection: three cases and a review of the literature. J Stroke Cerebrovasc Dis. 2020;29(12):105412. doi: 10.1016/j.jstrokecerebrovasdis.2020.105412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Hussain S., Vattoth S., Haroon K.H., Muhammad A. A Case of Coronavirus Disease 2019 presenting with seizures secondary to cerebral venous sinus thrombosis. Case Rep Neurol. 2020;12:260–265. doi: 10.1159/000509505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Han H., Yang L., Liu R., Liu F., Wu K.L., Li J., et al. Prominent changes in blood coagulation of patients with SARS-CoV-2 infection. Clin Chem Lab Med. 2020;58(7):1116–1120. doi: 10.1515/cclm-2020-0188. [DOI] [PubMed] [Google Scholar]
- 77.Tang N., Li D., Wang X., Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemostasis. 2020;Apr;18(4):844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ahmed S., Zimba O., Gasparyan A.Y. Thrombosis in Coronavirus disease 2019 (COVID-19) through the prism of Virchow’s triad. Clin Rheumatol. 2020;39(9):2529–2543. doi: 10.1007/s10067-020-05275-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Urano T., Suzuki Y. Accelerated fibrinolysis and its propagation on vascular endothelial cells by secreted and retained tPA. J Biomed Biotechnol. 2012;2012:208108. doi: 10.1155/2012/208108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Teuwen L.A., Geldhof V., Pasut A., Carmeliet P. COVID-19: the vasculature unleashed. Nat Rev Immunol. 2020;Jul;20(7):389–391. doi: 10.1038/s41577-020-0343-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ye J., Zhang B., Xu J., Chang Q., McNutt M.A., Korteweg C., et al. Molecular pathology in the lungs of severe acute respiratory syndrome patients. Am J Pathol. 2007;Feb;170(2):538–545. doi: 10.2353/ajpath.2007.060469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hamming I., Timens W., Bulthuis M.L., Lely A.T., Navis G., Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;Jun;203(2):631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Varga Z., Flammer A.J., Steiger P., Haberecker M., Andermatt R., Zinkernagel A.S., et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;2(10234):1417–1418. doi: 10.1016/S0140-6736(20)30937-5. 395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ackermann M., Verleden S.E., Kuehnel M., Haverich A., Welte T., Laenger F., et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med. 2020 Jul 9;383(2):120–128. doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Suzuki Y., Yasui H., Brzoska T., Mogami H., Urano T. Surface-retained tPA is essential for effective fibrinolysis on vascular endothelial cells. Blood. 2011;15(11):3182–3185. doi: 10.1182/blood-2011-05-353912. 118. [DOI] [PubMed] [Google Scholar]
- 86.Escher R., Breakey N., Lämmle B. Severe COVID-19 infection associated with endothelial activation. Thromb Res. 2020;190:62. doi: 10.1016/j.thromres.2020.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.McGonagle D., Sharif K., O'Regan A., Bridgewood C. The role of cytokines including interleukin-6 in COVID-19 induced pneumonia and macrophage activation syndrome-like disease. Autoimmun Rev. 2020;Jun;19(6) doi: 10.1016/j.autrev.2020.102537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Roncati L., Ligabue G., Fabbiani L., Malagoli C., Gallo G., Lusenti B., et al. Type 3 hypersensitivity in COVID-19 vasculitis. Clin Immunol. 2020;217:108487. doi: 10.1016/j.clim.2020.108487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.McGonagle D., O'Donnell J.S., Sharif K., Emery P., Bridgewood C. Immune mechanisms of pulmonary intravascular coagulopathy in COVID-19 pneumonia. Lancet Rheumatol. 2020;Jul;2(7):e437–e445. doi: 10.1016/S2665-9913(20)30121-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Maier C.L., Truong A.D., Auld S.C., Polly D.M., Tanksley C.L., Duncan A. COVID-19-associated hyperviscosity: a link between inflammation and thrombophilia? Lancet. 2020;6(10239):1758–1759. doi: 10.1016/S0140-6736(20)30479-2. 395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Forconi S., Pieragalli D., Guerrini M., Galigani C., Cappelli R. Primary and secondary blood hyperviscosity syndromes, and syndromes associated with blood hyperviscosity. Drugs. 1987;33 Suppl 2:19–26. doi: 10.2165/00003495-198700332-00006. [DOI] [PubMed] [Google Scholar]
- 92.Ranucci M., Ballotta A., Di Dedda U., Bayshnikova E., Dei Poli M., Resta M., et al. The procoagulant pattern of patients with COVID-19 acute respiratory distress syndrome. J Thromb Haemost. 2020;18(7):1747–1751. doi: 10.1111/jth.14854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Bi X., Su Z., Yan H., Du J., Wang J., Chen L., et al. Prediction of severe illness due to COVID-19 based on an analysis of initial Fibrinogen to Albumin Ratio and Platelet count. Platelets. 2020;3(5):674–679. doi: 10.1080/09537104.2020.1760230. 31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Bray M.A., Sartain S.A., Gollamudi J., Rumbaut R.E. Microvascular thrombosis: experimental and clinical implications. Transl Res. 2020;225:105–130. doi: 10.1016/j.trsl.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Powezka K., Khan T., Narlawar R., Antoniou G.A. Ruptured popliteal artery aneurysm complicated with acute respiratory distress syndrome secondary to SARS-CoV-2 infection. Ann Vasc Surg. 2020;66:24–27. doi: 10.1016/j.avsg.2020.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Shih M., Swearingen B., Rhee R. Ruptured abdominal aortic aneurysm treated with endovascular repair in a patient with active COVID-19 infection during the pandemic. Ann Vasc Surg. 2020;66:14–17. doi: 10.1016/j.avsg.2020.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.van Leeuwen R.T., Kol A., Andreotti F., Kluft C., Maseri A., Sperti G. Angiotensin II increases plasminogen activator inhibitor type 1 and tissue-type plasminogen activator messenger RNA in cultured rat aortic smooth muscle cells. Circulation. 1994 Jul;90(1):362–368. doi: 10.1161/01.cir.90.1.362. [DOI] [PubMed] [Google Scholar]
- 98.Nishimura H., Tsuji H., Masuda H., Nakagawa K., Nakahara Y., Kitamura H., et al. Angiotensin II increases plasminogen activator inhibitor-1 and tissue factor mRNA expression without changing that of tissue type plasminogen activator or tissue factor pathway inhibitor in cultured rat aortic endothelial cells. Thromb Haemost. 1997;77(6):1189–1195. PMID: 9241756. [PubMed] [Google Scholar]
- 99.Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271–280.e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Wright F.L., Vogler T.O., Moore E.E., Moore H.B., Wohlauer M.V., Urban S., et al. Fibrinolysis shutdown correlation with thromboembolic events in severe COVID-19 infection. J Am Coll Surg. 2020;231(2):193–203. doi: 10.1016/j.jamcollsurg.2020.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Fraga-Silva R.A., Da Silva D.G., Montecucco F., Mach F., Stergiopulos N., Silva R.F., et al. The angiotensin-converting enzyme 2/angiotensin-(1-7)/Mas receptor axis: a potential target for treating thrombotic diseases. Thromb Haemostasis. 2012;Dec;108(6):1089–1096. doi: 10.1160/TH12-06-0396. [DOI] [PubMed] [Google Scholar]
- 102.Yamamoto K., Takeshita K., Kojima T., Takamatsu J., Saito H. Aging and plasminogen activator inhibitor-1 (PAI-1) regulation: implication in the pathogenesis of thrombotic disorders in the elderly. Cardiovasc Res. 2005;1(2):276–285. doi: 10.1016/j.cardiores.2004.11.013. 66. [DOI] [PubMed] [Google Scholar]
- 103.Fraga-Silva R.A., Pinheiro S.V., Gonçalves A.C., Alenina N., Bader M., Santos R.A. The antithrombotic effect of angiotensin-(1-7) involves mas-mediated NO release from platelets. Mol Med. 2008;Jan-Feb;14(1–2):28–35. doi: 10.2119/2007-00073.Fraga-Silva. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Becker R.C. COVID-19 update: covid-19-associated coagulopathy. J Thromb Thrombolysis. 2020;50(1):54–67. doi: 10.1007/s11239-020-02134-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Nagai M., Terao S., Vital S.A., Rodrigues S.F., Yilmaz G., Granger D.N. Role of blood cell-associated angiotensin II type 1 receptors in the cerebral microvascular response to ischemic stroke during angiotensin-induced hypertension. Exp Transl Stroke Med. 2011;3:15. doi: 10.1186/2040-7378-3-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Sodhi C.P., Wohlford-Lenane C., Yamaguchi Y., Prindle T., Fulton W.B., Wang S., et al. Attenuation of pulmonary ACE2 activity impairs inactivation of des-Arg9 bradykinin/BKB1R axis and facilitates LPS-induced neutrophil infiltration. Am J Physiol Lung Cell Mol Physiol. 2018;314(1):L17–L31. doi: 10.1152/ajplung.00498.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Jiang Y., Li J., Teng Y., Sun H., Tian G., He L., et al. Complement receptor C5aR1 inhibition reduces pyroptosis in hDPP4-transgenic mice infected with MERS-CoV. Viruses. 2019;11(1):39. doi: 10.3390/v11010039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Magro C., Mulvey J.J., Berlin D., Nuovo G., Salvatore S., Harp J., et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. Transl Res. 2020;220:1–13. doi: 10.1016/j.trsl.2020.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Klok F.A., Kruip M.J.H.A., van der Meer N.J.M., Arbous M.S., Gommers D., Kant K.M., et al. Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: an updated analysis. Thromb Res. 2020;191:148–150. doi: 10.1016/j.thromres.2020.04.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Iba T., Levi M., Levy J.H. Sepsis-induced coagulopathy and disseminated intravascular coagulation. Semin Thromb Hemost. 2020;46(1):89–95. doi: 10.1055/s-0039-1694995. [DOI] [PubMed] [Google Scholar]
- 111.Karakike E., Giamarellos-Bourboulis E.J. Macrophage activation-like syndrome: a distinct entity leading to early death in sepsis. Front Immunol. 2019;10:55. doi: 10.3389/fimmu.2019.00055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Diurno F., Numis F.G., Porta G., Cirillo F., Maddaluno S., Ragozzino A., et al. Eculizumab treatment in patients with COVID-19: preliminary results from real life ASL Napoli 2 Nord experience. Eur Rev Med Pharmacol Sci. 2020;Apr;24(7):4040–4047. doi: 10.26355/eurrev_202004_20875. [DOI] [PubMed] [Google Scholar]
- 113.Zhang Y., Xiao M., Zhang S., Xia P., Cao W., Jiang W., et al. Coagulopathy and antiphospholipid antibodies in patients with covid-19. N Engl J Med. 2020;382:e38. doi: 10.1056/NEJMc2007575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Galeano-Valle F., Oblitas C.M., Ferreiro-Mazón M.M., Alonso-Muñoz J., Toro-Cervera J., Natale M., et al. Antiphospholipid antibodies are not elevated in patients with severe COVID-19 pneumonia and venous thromboembolism. Thromb Res. 2020;192:113–115. doi: 10.1016/j.thromres.2020.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Bertin D., Brodovitch A., Beziane A., Hug S., Bouamri A., Mege J.L., et al. Anti-cardiolipin IgG autoantibodies are an independent risk factor of COVID-19 severity. Arthritis Rheumatol. 2020;72(11):1953–1955. doi: 10.1002/art.41409. [DOI] [PMC free article] [PubMed] [Google Scholar]