Abstract
Viruses are abiotic obligate parasites utilizing complex mechanisms to hijack cellular machinery and reproduce, causing multiple harmful effects in the process. Viruses represent a growing global health concern; at the time of writing, COVID-19 has killed at least two million people around the world and devastated global economies. Lingering concern regarding the virus' prevalence yet hampers return to normalcy. While catastrophic in and of itself, COVID-19 further heralds in a new era of human-disease interaction characterized by the emergence of novel viruses from natural sources with heretofore unseen frequency. Due to deforestation, population growth, and climate change, we are encountering more viruses that can infect larger groups of people with greater ease and increasingly severe outcomes. The devastation of COVID-19 and forecasts of future human/disease interactions call for a creative reconsideration of global response to infectious disease. There is an urgent need for accessible, cost-effective antiviral (AV) drugs that can be mass-produced and widely distributed to large populations. Development of AV drugs should be informed by a thorough understanding of viral structure and function as well as human biology. To maximize efficacy, minimize cost, and reduce development of drug-resistance, these drugs would ideally operate through a varied set of mechanisms at multiple stages throughout the course of infection. Due to their abundance and diversity, natural compounds are ideal for such comprehensive therapeutic interventions. Promising sources of such drugs are found throughout nature; especially remarkable are the algae, a polyphyletic grouping of phototrophs that produce diverse bioactive compounds. While not much literature has been published on the subject, studies have shown that these compounds exert antiviral effects at different stages of viral pathogenesis. In this review, we follow the course of viral infection in the human body and evaluate the AV effects of algae-derived compounds at each stage. Specifically, we examine the AV activities of algae-derived compounds at the entry of viruses into the body, transport through the body via the lymph and blood, infection of target cells, and immune response. We discuss what is known about algae-derived compounds that may interfere with the infection pathways of SARS-CoV-2; and review which algae are promising sources for AV agents or AV precursors that, with further investigation, may yield life-saving drugs due to their diversity of mechanisms and exceptional pharmaceutical potential.
Keywords: Antiviral, Algae, Seaweed, SARS-CoV-2, COVID-19, Pathogenesis, Viral inhibitors, Prophylactic, Virucidal, Virustatic, Entry inhibitors, Enzyme inhibitors, Immunostimulants, Antioxidants
Graphical abstract
1. Introduction
According to best estimates at the time of writing, at least 2.4 million people have been killed by COVID-19 in >200 countries since January 2020 [1,2]. Owing to a lack of testing and overwhelmed healthcare systems, it is likely that COVID-19's actual death toll is significantly higher, especially in developing nations [3,4]. Global economies have drastically declined, and tens of millions of people are at risk of dropping down past the $1.90 income threshold of extreme deprivation [5,6]. In addition to its catastrophic short-term effects, COVID-19 is a harbinger of a growing global trend in infectious disease.
Widespread viral outbreaks will continue to challenge human civilization [7]. COVID-19 is caused by SARS-CoV-2, a zoonotic virus that originated in an alternative animal host (potentially a horseshoe bat or pangolin) before spreading to humans [8]. It is estimated that 60% of known and 75% of novel, ‘emerging’ human pathogens are zoonotic [9]. Diseases caused by zoonotic viruses are common, examples include tick encephalitis, Dengue Fever, West Nile Virus (WNV), Zika (ZV), and rabies (RBV) [[9], [10], [11]]. Zoonotic viruses are becoming increasingly common with the growth of human populations [11,12]. As the global population swells, humans encroach into previously unsettled areas and thus encounter animals carrying new viruses with ever-increasing frequency, especially in developing nations [7,10,13,14]. Furthermore, rapid growth of industrial livestock farming inherently involves close contact between dense populations of genetically homogenous animals which allows viruses to rapidly reproduce and eventually spread to humans [9,11,14].
Trends towards the emergence of new viruses due to population growth, deforestation, and industrial agriculture are exacerbated by climate change [15,16]. As average temperatures increase, arthropod vectors carrying dozens of known viruses may expand their geographical range and periods of seasonal activity. Of special prominence among these are mosquitoes and ticks, which carry many serious viruses and cause other non-viral illnesses as well [[17], [18], [19], [20]]. Climate change can magnify negative outcomes of viral diseases among especially vulnerable populations. Warming temperatures and pollution may apply bodily homeostatic and immunological stress and decrease standards of hygiene and medical care due to extreme weather events and geopolitical instability [[21], [22], [23]]. The worst outcomes of viral infection will certainly be suffered by the Global South, where populations are expanding rapidly and access to sanitary conditions and medical care remains limited [23,24].
Amidst the devastation of COVID-19, as we consider the social dynamics of emerging diseases, there is an urgent need to reconsider the strategies we use to confront widespread viral outbreaks. The sheer scope of COVID-19's effect and lack of effectively coordinated response indicates that there is a clear need for widely accessible AV therapies. Such therapies may be derived from a variety of sources but should all be grounded in a comprehensive understanding of viral structure/function and infection progression.
Viruses are microscopic particles fundamentally consisting of a genome surrounded by a protein capsid and/or a host membrane derived lipid bilayer often referred to as an “envelope” [25]. Viruses are abiotic obligate parasites, meaning that they depend on infecting living organisms and ‘hijacking’ cellular machinery to reproduce [26,27]. During infection, viruses must enter the human body via epithelial tissues lining body surfaces or direct penetration into the bloodstream. After invading the body, some viruses remain localized and replicate in the tissues that first provided them entry. Others must circulate through the lymph and blood to reach specific target tissues. In order to replicate, viruses must invade particular cells and hijack cellular machinery to produce copies of their genetic material and proteins. Viruses must carry out these steps while evading or subverting the immune system [28,29]. Viruses utilize a vast array of highly specialized mechanisms to infect the body and carry out their replication cycles [26]. These different mechanisms can be targeted by AV compounds at the various stages of viral pathogenesis in order to prevent negative outcomes [30]. One holistic strategy for developing AV therapies is to use an array of compounds that act at different stages of infection in order to maximize drug efficacy and avoid issues of bioavailability, harmful cytotoxic effects, and the development of viral resistance through mutation that are common issues with some AV drugs [[31], [32], [33], [34], [35], [36]].
Such bioactive compounds can be derived from natural or synthetic sources [30,37]. Many naturally occurring AVs are optimal for use because they are geographically widespread, abundant, easily refined, and reliably produced from renewable sources [37,38]. One promising source for AV compounds is algae. The term ‘algae’ refers to a polyphyletic grouping of ubiquitous mostly aquatic phototrophs distributed throughout the world's bodies of salt and freshwater [39]. They are fast growing and require low resource and energy inputs for cultivation. Representing a phylogenetically diverse group of photosynthetic micro- and macroorganisms, algae are highly productive, renewable sources of many bioactive compounds used in medicinal, cosmetic, commercial, and food products as well as an intriguing source for the future generation of fuels [[40], [41], [42]]. Many studies reporting AV activities from algae-derived compounds have been published in recent years.
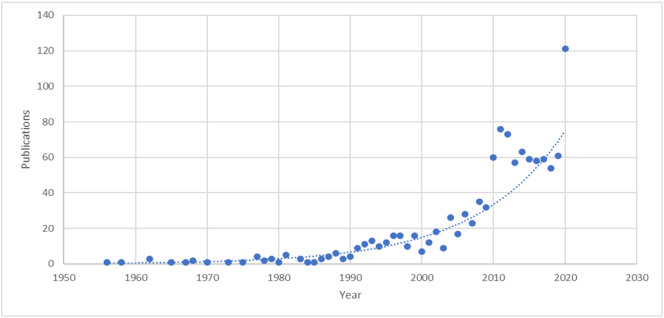
Though the study of AV compounds from algae is still in its fledgling stage, the base of literature nevertheless shows it to be a promising area of inquiry. A Web of Science® database topic search for the string, “algae antiviral” shows that prior to the year 2000, only 165 papers had been published in this research area. 207 papers were published in the years 2000–2010 and 741 from 2010-present (Fig. 1 ) [43]. This positive trend indicates that algae are receiving increased attention as sources of bioactive antiviral compounds and may, with further infrastructure development and research into clinical applications, yield many life-saving compounds.
Fig. 1.
“Algae Antiviral” Publications 1956–2020. Annual publications containing the keywords “algae antiviral” since 1956, determined by Web of Science Database topic search.
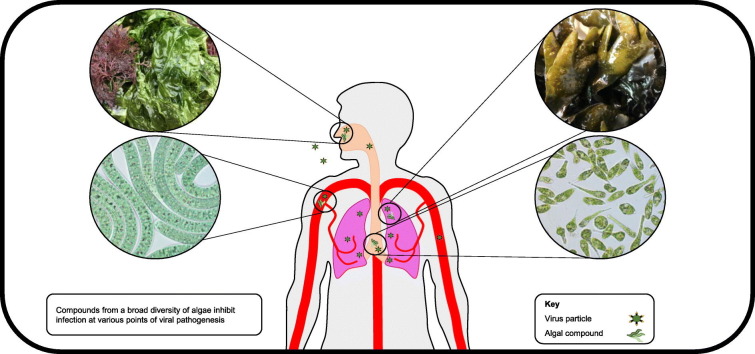
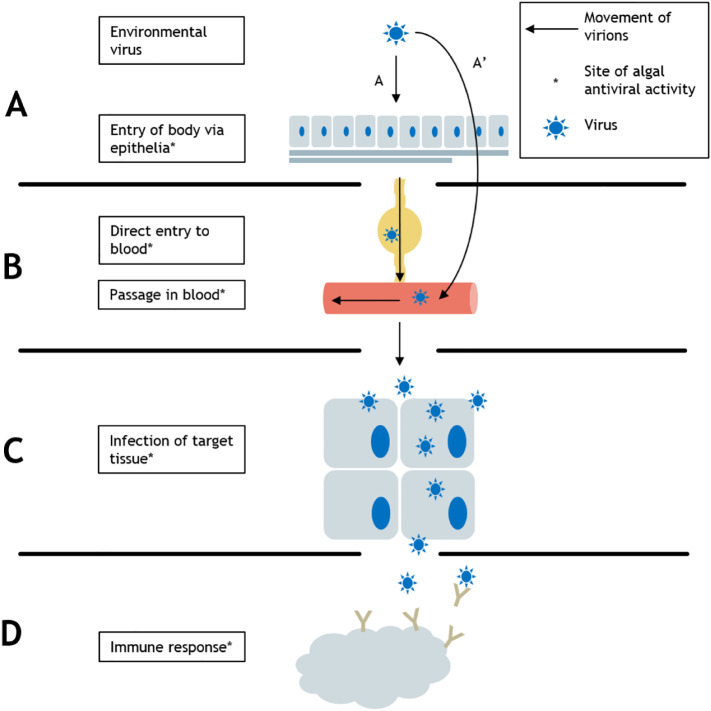
In this review, we follow the steps of viral replication and discuss algae-derived compounds that have demonstrated AV effects at each stage. We begin with the invasion of viruses from the external environment into the human body, proceed to viral transport within the body through the lymph and blood, and conclude by examining the infection of target cells as well as immune response to viral infection (Fig. 2 ). We discuss compounds derived from a broad diversity of eukaryotic micro- and macroalgae as well as cyanobacteria (Fig. 3 ) that inhibit or interfere with the life cycles of numerous types of virus (Table 1 ). We demonstrate how due to their exceptional pharmaceutical potential, algal-derived compounds deserve increased attention as potential sources of widely available AV compounds.
Fig. 2.
Viral pathogenesis. In a generalized infection cycle, a virus must (A) enter the human body via a layer of epithelial tissue or (A′) directly into the bloodstream via a dermal lesion, (B) be transported through the body via the lymph and blood, and (C) infect its target tissue, all while (D) evading or subverting immune response.
Fig. 3.
A broad diversity of algae produces antiviral compounds. Isolated compounds or crude extracts from genera depicted in Fig. 3 have demonstrated antiviral activity and are discussed in this review. These algae represent marine and freshwater species distributed throughout the world.
Table 1.
Algae-derived compounds show inhibitory activity towards a broad diversity of viruses. Table 1 shows the families of viruses and associated diseases that have been discussed in this review. Virus families are sorted by genome and envelope morphologies to indicate the structural diversity of viruses that are inhibited by algae-derived compounds.
| Genome type | DNA viruses | RNA viruses | |||
|---|---|---|---|---|---|
| Envelope | Enveloped | Non-enveloped | Enveloped | Non-enveloped | |
| Genome strands | Double-stranded | Single-stranded | Double-stranded | Single-stranded | |
| Virus and associated disease |
Herpesviridae
|
Papillomaviridae
|
Paramyxoviridae
|
Picornaviridae
|
Birnaviridae
|
Coronaviridae
| |||||
Rhabdoviridae
| |||||
Roniviridae
| |||||
Pneumoviridae
| |||||
Hepadnaviridae
|
Adenoviridae
|
Orthomyxoviridae
|
|||
Retroviridae
| |||||
Flaviviridae
| |||||
2. Viral pathogenesis
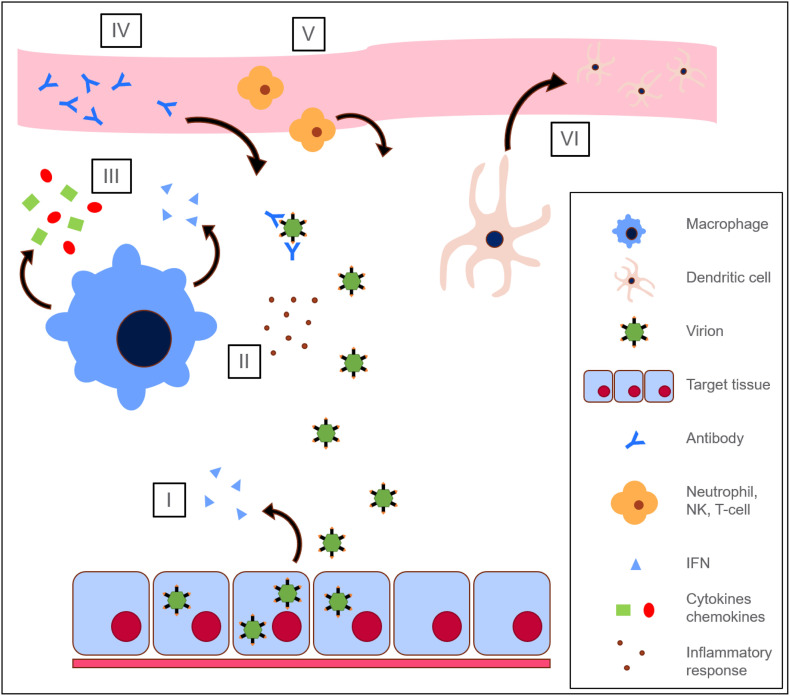
In order to replicate, a virus must hijack the machinery of specific target cells in the human body [25]. However, the process by which a virus infects the body and replicates itself is complex and varies considerably between various groups of viruses. Viruses infect hosts through a complicated series of steps and array of mechanisms. Generally speaking, a virion (virus particle) must first enter the human body (Fig. 2A, B), which is well-protected against invasive foreign pathogens. Many viruses enter the body by infecting layers of epithelial tissue which line the body's borders. Others enter into the bloodstream through disruptions in the normally impenetrable layers of dermal tissue that cover the vast majority of the body's surface [28,29].
Some viruses infect and replicate in the exterior layers of tissue that first provided them entrance to the body. However, others require access to non-epithelial tissues in order to replicate and thus necessitate transport through the body via the lymph and blood, a process termed viremia (Fig. 2B). In the lymph and blood, virions may encounter mobile immune system phagocytes, namely macrophages and neutrophils that are responsible for clearing the body of infectious foreign particles.
Virions that make their way into the blood are spread throughout the body; however, certain types of viruses typically have highly specific surface receptors that only bind certain target cells. Virions that come into contact with their target cells will adhere to, penetrate their targets (Fig. 2C) and release their genetic material for replication therein (using some combination of viral and cellular machinery). Copies of viral proteins and genetic material will then be packaged and released from the cell through budding or apoptosis and will continue to infect more target cells within the host or be released from the host into the environment to begin the cycle anew. Viral infections often cause a host immune response (Fig. 2D), which may eliminate or reduce harmful effects.
While they are complicated and highly variable between virus types, viral pathogeneses provide many different avenues for intervention and deterrence of infection. Algae-derived compounds may be administered at many timepoints throughout the infection cycle to prevent harmful effects by directly interfering with viral replication or boosting the body's natural immune response. Some algae-derived compounds prevent infection from occurring by blocking common avenues into the body. These represent proactive forms of treatment, preventing infection before it occurs.
2.1. Algal compounds prevent viral entry into the body
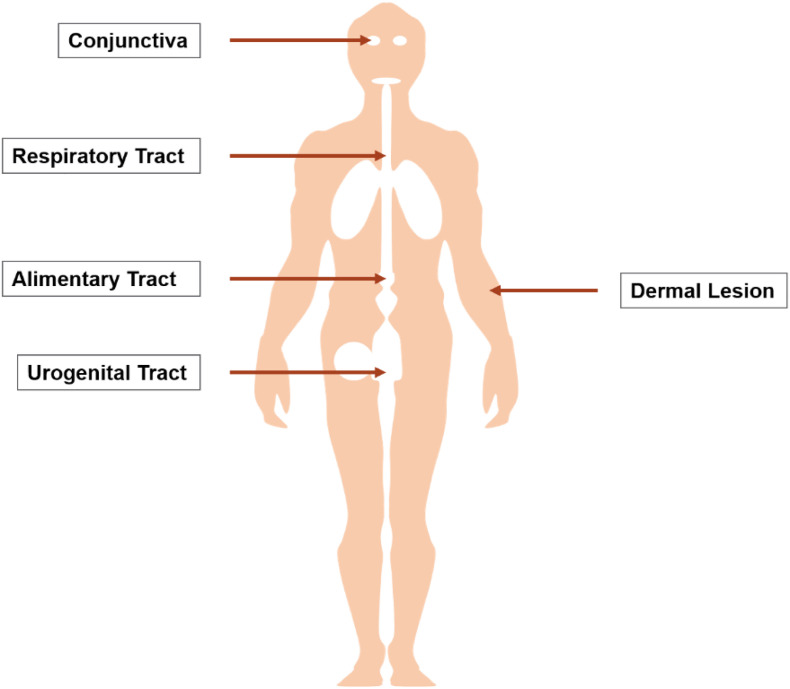
In order to cause infection, a virus must invade the human body. A virus may access the body through different avenues (Fig. 4 ). While the outer layer of dermal tissue is dry, dead, and impenetrable for viruses in its intact form, there are a number of living layers of epithelial tissue that a virus can access through bodily orifices or exposed surfaces. These primarily include the respiratory tract (coronaviruses (CoVs), influenza viruses (IAVs), etc.), the alimentary tract (hepatitis (HV), cytomegaloviruses (CTMVs), etc.), the urogenital tract (human papilloma (HPV), some herpes (HSV), human immunodeficiency viruses (HIV), etc.), and more rarely, the conjunctiva (adenoviruses (ADVs), enteroviruses (ENTVs), etc.) [28,44]. Epithelial tissues represent entry points that viruses can pass through on their way to infecting other bodily tissues. Some viruses do not enter the body through epithelial tissues, but instead by penetrating the skin through bites (rabies, Zika, Dengue Fever viruses, etc.), scrapes (pox, some HSVs, etc.), or injections with contaminated needles (Epstein-Barr (EBV), Ebola viruses (EV), etc.) [28,44].
Fig. 4.
Environmental virus particles commonly enter the body through a layer of epithelial tissue (conjunctiva, respiratory, alimentary, urogenital) or through dermal lesion (bite, scrape, injection). Algae-derived compounds prevent viruses from infecting the body via each of these avenues.
Therapeutic use of algae derived compounds is an example of a step that can be taken to prevent viral entry into the body. Other common preventative measures that lower the chances of internalizing environmental virions include wearing masks and washing hands. Indeed, prevention measures, whether behavioral (i.e., washing hands, sterilizing needles) or technological (i.e., vaccines) are always preferable to treatment as they avert any negative outcomes accompanying infection and medical intervention. Here we discuss viruses that enter the body through different avenues and corresponding preventative algae-derived compounds (Table 2 ).
Table 2.
Algal compounds prevent the entrance of viruses into the body.
| Mode of entry | Compound | Species classification | Species | Model | Virus | Reference |
|---|---|---|---|---|---|---|
| Respiratory epithelia | Carrageenan | Rhodophyta | Not reported | Clinical trials, murine | CoV IAV PIAV MPNV RSV RHV |
[[45], [46], [47], [48]] |
| Fucoidan | Phaeophyceae | Kjellmaniella crassifolia | Murine | IAV-A | [49] | |
| Alimentary epithelia | Fucoidan | Phaeophyceae | Undaria pinnatifida | Murine | IAV-A Avian IAV-A |
[58,59,61] |
| Urogenital epithelia | Carrageenan | Rhodophyta | Not reported | Clinical trial Rhesus macaque |
HPV HIV HSV-2 Simian immunodeficiency virus (SIV) HIV HPV |
[53,54] [55] [56] [57] |
| Lectins | Assorted algae, cyanobacteria Rhodophyta |
Various Originally isolated from Griffithsia sp., produced recombinantly in Nicotiana benthamiana (Solanaceae) |
In vitro In vitro; TZM-bl, HeLa, Vero |
HIV HSV-2, HPV |
[58] [59] |
|
| MGDG | Chlorophyta | Coccomyxa sp. | Murine | HSV-2 | [60] | |
| Dermal lesion | Diterpenes | Phaeophyceae | Canistrocarpus cervicornis | Murine | HSV-1 | [65] |
| Bromophenols | Rhodophyta | Symphyocladia latiuscula | Murine | HSV-1 | [62] | |
| Protein extract | Phaeophyceae | Macrocystis pyrifera, Durvillaea antarctica | Murine | HSV-1, HSV-2 | [63] | |
| PMG | Phaeophyceae | Not reported | Murine | HPV | [64] | |
| Conjunctiva | Carrageenan | Rhodophyceae | Not reported | Feline | FHV-1 | [65] |
2.1.1. Respiratory epithelia
Respiratory viruses are incredibly widespread and the leading cause of disease in humans around the world. They are responsible for roughly one-fifth of all childhood mortality, especially in poor tropical regions [66]. Respiratory viruses can spread rapidly through populations via person-to-person contact. Typically, virions are inhaled from the air or transmitted to the respiratory tract via direct contact (i.e., touching fingers to the mouth or nose). Some compounds derived from algae are able to protect against respiratory disease by preventing virion particles from entering and infecting the respiratory tract epithelia (Table 2).
2.1.1.1. Carrageenan
Carrageenan is a sulfated polysaccharide derived from various macroalgae in the phylum Rhodophyta including Gigartina, Chondrus crispus, Eucheuma, and Hypnea [67,190]. In several recent clinical trials, a carrageenan-based nasal spray demonstrated AV activity against human common cold viruses by preventing viral attachment to epithelial cells in the nasal cavity [[45], [46], [47]] (Table 2). Carrageenan bound to viral glycoproteins and acted as a physical barrier preventing virions from infecting their target cells. By limiting the number of viruses able to access epithelial tissue, fewer viruses were able to replicate leading to a reduction in viral titers and faster resolution of symptoms. Importantly, carrageenan acted as a non-specific physical barrier to several different types of pathogen, including CoV, IAV, parainfluenza (PIAV), rhino (RHV), metapneumonia (MPNV), and respiratory syncytial viruses (RSV). Carrageenan did not itself pass through respiratory mucosa and enter into the circulation. This mode of action is important to highlight, as a host-independent mechanism is unlikely to lead to adverse side-affects. It was later shown that carrageenan can be paired with an additional influenza-targeting drug to achieve a synergistic AV effect and prevent the development of viral resistance [48]. The combination therapy was able to prevent fatality from a lethal influenza strain in a murine model. Because influenza virus infections are often accompanied by other respiratory viruses, targeting with influenza-specific drugs often fails to alleviate symptoms. Combination treatment with carrageenan can be an effective solution by acting as a non-specific barrier to many different viruses that would otherwise cause harmful respiratory disease making. Due to its ability to counter infection from a variety of different viruses, carrageenan is an ideal broad-spectrum preventative agent. Multiple carrageenan-based nasal sprays are currently marketed in Europe and Canada [68,69]; additional research and investment could provide a massive protective effect to vulnerable populations in other countries around the world. A clinical trial evaluating the prophylactic effect of a carrageenan nasal spray against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is currently underway in the United States [70] and similar efforts to develop an anti-SARS-CoV-2 carrageenan nasal spray are being made in the UK [71].
2.1.1.2. Fucoidan
Fucoidan is a sulfated polysaccharide found in many algae within the class Phaeophyceae. Using a murine model, Wang et al. determined the intranasal application of fucoidan derived from Kjellmaniella crassifolia was able to protect against Influenza A virus (IAV) pathology through a series of mechanisms [49] (Table 2). Fucoidan interacts with IAV surface enzyme neuraminidase (NA) in a host-independent manner to form a stable, inert complex that prevents viral entry into cells. Fucoidan may also interact with sialic acid residues on the viral envelope to prevent activation of the epidermal growth factor receptor (EGFR) pathway that would otherwise result in viral endocytosis. Unlike the commercial drugs amantadine and Oseltamivir, fucoidan did not give rise to resistant viral strains. The administration of fucoidan in the nasal cavity prevented IAV infection in mouse models, as lower titers of virus were recovered from the lungs after treatment. Fucoidan's high molecular weight (MW) may prevent it from passing through the respiratory mucosa or being absorbed by epithelial cells and likely limits IAV virion interactions with cellular receptors.
2.1.2. Alimentary epithelia
The alimentary tract consists of a series of organs involved with consumption, digestion, and excretion. These primarily include the mouth, pharynx, stomach, and intestines. The alimentary tract is lined with layers of epithelial tissue for nutrient absorption and chemical secretion. While protected with layers of mucus, bile, stomach acid, and other deterrents, the alimentary tract is nonetheless vulnerable to certain pathogens. Many viruses are ingested and infect the body via the alimentary tract [28,29]. Some, like HSV, EBV, and CTMV invade the cells of the mouth. Many others invade the intestinal tract, including reoviruses, ADVs, Norwalk virus, ENTVs, HVs, and rotaviruses. Algae-derived compounds administered in the alimentary tract via oral consumption have the ability to prevent viruses from infecting alimentary epithelia (Table 2).
2.1.2.1. Fucoidan
Hayashi et al. found that a low molecular weight (LMW) (9 kDa) orally administered fucoidan from the brown alga Undaria pinnatifida was able to significantly decrease the IAV titer in mice lungs, and that this benefit was augmented by combining treatment with the commercial pharmaceutical Oseltamivir (Table 2) [50]. Furthermore, treatment with oral fucoidan was able to entirely prevent mortality. More recently, Richards et al. supported this result, determining that the oral administration of fucoidan from Undaria pinnatifida was able to reduce symptoms and lung pathology after IAV infection [51], potentially by preventing virions from interacting with alimentary epithelia. In addition to infection and replication in the respiratory tract, influenza viruses are known to replicate in the alimentary tract [72]. Due to its binding and inactivation of IAV, orally administered fucoidan in the alimentary tract may prevent harmful interactions between the virus and alimentary epithelia. Richards et al. showed that the administration of fucoidan led to a minimal decrease in viral titers [51]. While the result was not statistically significant, it is possible that a higher dose would demonstrate a more robust discrepancy. It should be noted that this study used a fucoidan sample with some impurities (7.2%) which may have contained trace amounts of other bioactive compounds. Furthermore, the fucoidan sample used exhibited a broad MW range (20–1100 kDa) with a peak value of 72.1 kDa, and so it is possible that some small amount of fucoidan uptake from the intestines occurred. Thus, potentially confounding factors are the interaction between fucoidan and virions in the bloodstream, immunomodulation, or inactivation at the lung epithelia.
Additional research by Synytsya et al. characterized a LMW (9 kDa) fucoidan derived from Undaria pinnatifida called Mekabu fucoidan (MF) [52]. This study showed that the oral administration of MF decreased lung titers of two IAV subtypes by 70–85%. The authors proposed that this reduction was due to both direct interaction between MF and virions (preventing viral adsorption from the alimentary tract into circulation) as well as immunomodulation by MF absorbed into the bloodstream. Due to its direct inactivation of virions and activation of immune system function, it is impossible to determine which mechanism predominated, but it is likely that some AV activity in the alimentary tract decreased the severity of infection.
Significantly, Synytsya et al. and Hayashi et al. both showed that the oral administration of a LMW fucoidan resulted in a significant decrease in lung IAV titer [50,52] (Table 2). Both studies also showed an immunomodulatory effect from orally administered fucoidan. It is likely that limited IAV replication can be attributed to both a direct inactivating effect from fucoidan molecules in the alimentary tract and its immunostimulatory effects. Fucoidan may serve as an effective AV in the alimentary tract and continue to exert further AV activity during later stages of pathogenesis. In following sections, we will further discuss fucoidan's AV activity, including its properties as an immunostimulant. This research provides evidence that AV compounds may exert activity by preventing viral absorption through intestinal epithelia.
2.1.3. Urogenital epithelia
The urogenital system is responsible for the formation and excretion of urine as well as important reproductive functions. The urogenital system is lined with epithelial cells and is susceptible to infection from HIV, HPV, HV, and HSV [28,29]. Compounds derived from algae can be topically applied to female urogenital epithelia to prevent invasion of viruses, typically those associated with sexually transmitted diseases (Table 2).
2.1.3.1. Carrageenan
In several clinical trials, topically applied carrageenan gels have shown effectiveness in preventing the spread of sexually transmitted viruses. In a human trial with female volunteers, the carrageenan-based, topically applied gel Carraguard was associated with a 40% reduced likelihood of contracting HPV compared to control subjects [53] (Table 2). The authors suggest that carrageenan acts as a physical barrier, competing with and preventing HPV from binding to epithelial cell receptors. Increased protection from HPV was associated with adherence to consistent gel use. However, the study was nonetheless limited by inconsistencies in reporting and it is possible that an even higher protection rate could be attained by improving behavioral performance. Better adherence to the protocol by study participants would improve the comparison's power as well.
In a later study, 40 women suffering from HPV infection used a carrageenan-based, vaginally applied gel over the course of four months. At the end of the trial, 60% of women using the carrageenan gel negatively tested for HPV infection, compared to 25% of control group participants [54]. HPV only replicates in the urogenital epithelia, and so by preventing virions from entering epithelial cells, the carrageenan gel was able to reduce and eventually eliminate HPV infection entirely for a subset of study participants.
Another study showed that participants using the commercial carrageenan-based gel Carraguard contracted fewer HIV infections than controls, but the result was not statistically significant [55]. However, this study was also severely limited by the poor adherence to regular gel application before intercourse. It is thus possible that Carraguard may yet be an effective clinical HIV solution. However, carrageenans have been shown to only be effective against HIV at extremely high concentrations [73], and so it may be unlikely that carrageenans prove an effective topically applied anti-HIV compound. Furthermore, this case study shows how even effective solutions might be limited by real-world application factors, an important consideration to keep in mind as the various compounds in this study are discussed. Recent reports using combination therapies showed that an intravaginal ring releasing carrageenan as well as two other synthetic pharmaceuticals limited HIV, HPV, and HSV infection in rhesus macaques [56,57] (Table 2).
2.1.3.2. Lectins
Lectins are non-immunoglobulin carbohydrate-binding proteins that can interact with glycans on viral envelopes to prevent binding with target cells [58]. As topically applied microbicides, they have great potential to limit viral infection. Research performed using rhesus macaques found that the lectin cyanovirin (CV-N) from the cyanobacteria Nostoc ellipsosporum inhibited 63–85% of HIV infection [58] (Table 2). Another demonstrated that griffithsin (GRFT), a lectin from red algae (Rhodophyta), binds both HPV and HSV glycoproteins and inhibits entry into their target cells [59]. Using a murine model, researchers showed that the combination of vaginally applied GRFT and carrageenan protect against HPV and HSV infection. This is especially significant with HSV, as HSV particles can be absorbed into the bloodstream after infecting urogenital epithelia and cause harmful pathologies to other tissue types.
2.1.3.3. Monogalactosyl diacylglycerol (MGDG)
MGDG compounds are fatty acids found in many organisms. Hayashi et al. found that the vaginal administration of an MGDG fraction from the green (phylum Chlorophyta) microalga Coccomyxa sp. exerted a prophylactic effect against HSV-2 infection using a murine model [60] (Table 2). MGDG decreased viral load in the urogenital cavity, reduced lesion formation, and improved the survival rates of infected mice. MGDG was shown to exert a protective effect by binding virion particles and degrading them. The mechanism likely involves the MGDG compounds fusing with and partially disrupting the lipid membrane of the viruses [74]. This mechanism will be further discussed in Section 2.2.1 where we examine virucidal compounds.
2.1.4. Dermal lesions
Dermal tissue covers the vast majority of our body surfaces. Exposed dermis is dead, keratinized tissue that is impenetrable to viruses in their intact state. However, viruses are able to enter through openings in the skin created by abrasion, laceration, injection, or bite. After penetrating the skin, viruses may reproduce and cause damage locally or spread through the blood to other regions of the body [28,44]. Many prevention measures can eliminate the possibility of contracting certain viruses that enter the body directly through disruptions of the skin, such as sterilizing needles, using mosquito repellant, etc. Some algae-derived compounds can prevent viral infection via the skin should its integrity be damaged (Table 2). This protective effect can be achieved through topical or oral administration.
2.1.4.1. Diterpenes
Diterpenes are small molecules composed of four isoprene units and are found throughout nature. A diterpene-based topical ointment from Canistrocarpus cervicornis, a brown alga (Phaeophyceae), was recently evaluated against the spread of HSV-1 via cutaneous lesion [61] (Table 2). In this investigation, mice were scratched, inoculated with HSV-1, and then treated with diterpene ointment. The diterpene ointment protected against lesion injury, paralysis, and death as a result of infection in a manner similar to the synthetic drug acyclovir. Furthermore, the diterpene ointment showed promising activity against an acyclovir-resistant strain of HSV as well. The diterpene ointment did not show harmful effects, alter hepatic or renal function, nor reduce bodyweight. Diterpenes interfere with viral entry into target cells by binding with viral glycoproteins [75], and so while virion-diterpene complexes may have entered into the vasculature, the virions were unable to enter their target cells. Diterpenes do not form a physical barrier in the same manner as some previously discussed polysaccharide compounds. Topical application of diterpenes in this manner may be an effective way to prevent against HSV infection. In practice, one might apply a diterpene-containing ointment to a scrape, sunburn, or otherwise damaged dermal tissue after injury to prevent HSV infection.
2.1.4.2. Bromophenol
Bromophenols are small organic molecules containing at least one bromine atom covalently bonded to a phenolic ring. Research performed by Park et al. determined that the oral administration of tribromophenolic compounds from the red alga Symphyocladia latiuscula reduced the development of skin lesions in mice after HSV-1 infection (Table 2). This is remarkable, as mice were scratched and infected with HSV-1 at the skin level but the oral administration of bromophenol still exerted a protective effect. It seems probable that bromophenols were taken up by the bloodstream and made their way to the dermis where they exert their AV activity, limiting the number of virions that entered the body. In addition to opposing HSV-1 infection of dermal tissue, the topical application of bromophenol reduced yields of virus in the brain, thus indicating that less virus was able to pass through the dermis and enter the bloodstream. The efficacy of bromophenols when applied either orally or topically is a promising indicator of potentially robust AV activity [62].
2.1.4.3. Proteins
Castillo et al. collected semi-refined cytosolic extracts of two brown algae, M. pyrifera and D. antarctica [63] that were mostly proteinous in nature but also included some additional small molecules (Table 2). Two fractions were generated from the extract, one with proteins >10 kDa in size and one with proteins <10 kDa. While the larger protein fraction exerted stronger AV activity against HSV than the smaller, both fractions together yielded the strongest effect in vitro. Using a murine model, the investigators first inoculated skin lesions with HSV and then applied a topical formulation of the protein extract to the skin 24 h later. Due to the time course of experimental steps, virions accumulated in the dorsal root ganglia, showing that the virus effectively infected the mice, but no viral accumulation was shown at the skin. Thus, it is possible that the preemptive topical application of this protein extract would prevent infection of the skin and other vulnerable tissues targeted by HSV that require entrance to the body. This study demonstrates an important point about therapeutic compounds derived from natural sources: in the words of Sharaf et al., “Natural products containing bioactive compounds are sometimes more effective in their natural combination, rather than in a pure concentrated effective compound with identified composition” [76]. Unrefined extracts of natural organisms may contain hundreds or thousands of compounds and it is certainly possible that multiple of those may exert antiviral activity. Due to the great diversity of compounds from natural sources, it is impossible to identify every downstream effect of every compound within the human body. An unrefined sample containing a multitude of compounds may thus improve therapeutic efficacy by eliminating the effects of harmful pathogens at various stages of the infection cycle, reducing the likelihood for developing harmful mutations, and decreasing the cost of treatment preparation [32,77].
2.1.4.4. Polymannuroguluronate (PMG)
PMG is a sulfated polysaccharide derived from seaweed in the class Phaeophyceae. Research performed by Wang et al. found that an LMW (10 kDa) PMG had potent anti-HPV effects in vitro and in vivo using a murine skin-infection model [64] (Table 2). In this study, mice were scarified using an abrasive hand tool and then inoculated with HPV in solution. When applied before or concurrently with infection, PMG significantly limited infection. In vitro analysis showed that PMG inhibits HPV through a series of mechanisms which may prevent drug-resistant virus strains. Cytotoxicity assays also revealed that PMG has an extremely high selectivity index (SI) and low cytotoxicity in three different cell lines. PMG is thus a promising prophylactic treatment against HPV, the most common sexually transmitted disease.
2.1.5. Conjunctiva
The conjunctiva is the mucous membrane covering the eye and inside of the eyelids and is the route of entry into the body used by some ADVs and ENTVs [28,29]. Recent evidence also demonstrated that the SARS-CoV-2 virion can also infect the human body via the conjunctival route [78]. The conjunctiva is more vulnerable to infection than the epidermis but is protected by constant secretions (tears) and wiping by the eyelid. Compounds from algae may be able to protect against infection of the conjunctiva.
2.1.5.1. λ-Carrageenan
Stiles et al. found that λ-carrageenan from seaweed in the phylum Rhodophyta limited feline herpesvirus-1 (FHV-1) infection in vitro. When applied topically to the eyes of cats in vivo, λ-carrageenan reduced the time period of viral shedding, when new virions are produced and released into the environment, but did not alter clinical signs of disease [65] (Table 2). Indeed, it seems that the application of λ-carrageenan in this experimental setup did not prevent adsorption through the conjunctiva. λ-Carrageenan irritated some subjects after topical application. Research in this particular area is especially limited and more investigation is needed to elucidate any potentially harmful effects or benefits of carrageenan application to the conjunctiva.
2.1.6. Conclusion
For infection to take place, virions must bypass the body's external defenses. By taking simple protective measures at this step, harmful and costly infections can be prevented. Mask use, for example, has been shown to prevent against the spread of SARS-CoV-2 as well as other respiratory illnesses; wearing a mask to prevent the contraction or transmission of such diseases is preferable to a potential hospital stay or expensive antiviral therapies [79]. The best solution to a problem is often proactive avoidance. In this section we have shown that compounds derived from algae including carrageenan, fucoidan, and lectins among others have the potential to prevent infection from a variety of harmful viruses at the initial stage of entry into the body (Table 2). In subsequent sections we will demonstrate the antiviral activities of algal compounds after infection has taken place.
2.2. Algal compounds inhibit viral passage through the body
Despite defense efforts, some virions are able to invade past both natural and introduced exterior defenses and pass into the body. Some viruses remain localized in external regions of the body, residing and replicating in epithelial tissue [28,29]. Others are transported via the vasculature to target tissues found throughout the body's organ systems. These include togaviruses (TVs), ENTVs, orbiviruses (ORBVs), HSV, EBV, CMV, and lentiviruses (LNTVs) among others [25]. Viremia is the presence of viruses in the vasculature (lymph and/or bloodstream). Recent results suggest that COVID-19 viremia is associated with critical illness and more severe outcomes [80]. During their transport through the body's vasculature, viruses may be inactivated by introduced or endogenous interventions.
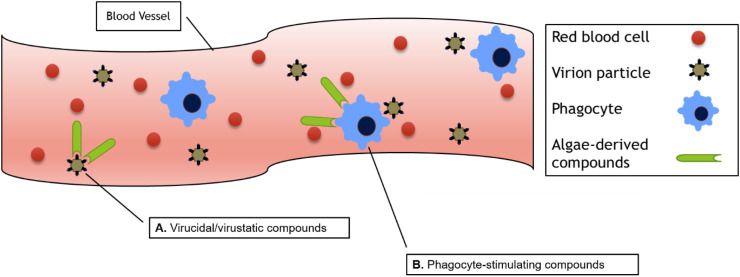
Non-native compounds that directly target virions may achieve a delocalized protective effect when introduced to the vasculature (Fig. 5 ). Generally speaking, compounds that target virions directly are virucidal (degrading, damaging virions) or virustatic (binding virions and holding them inert). Examples of both types of drug have been isolated from algae (Table 3 ).
Fig. 5.
Algae-derived compounds can limit viral spread through the vasculature. A., virucidal/virustatic compounds degrade virions or hold them inert and B., phagocyte-stimulating compounds boost phagocyte activity as they patrol the body looking for invading pathogens.
Table 3.
Algae-derived compounds exert virucidal and virustatic activities.
| Activity | Compound | Species classification | Species | Model (cell line) | Virus | Reference |
|---|---|---|---|---|---|---|
| Virucidal | MC15 | Phaeophyceae | Eisenia bicyclis | In vitro (CHSE-214) | IHNV | [88] |
| MGDG | Chlorophyta | Coccomyxa sp. |
In vitro (Vero) In vivo (Murine) |
HSV-1/2 | [60] | |
| 7-Keto stigmasterol | Chlorophyta | Prasiola crispa | In vitro (RK-13) | EHV-1 | [89] | |
| Phycobiliprotein | Cyanobacteria | Spirulina platensis | In vitro (E. coli) | MS-2 ΦX-174 |
[90] | |
| Virustatic | Cyanovirin-N | Cyanobacteria | Nostoc ellipsosporum | In vitro (PBL, MAC) | HIV | [91] |
| GRFT | Rhodophyta | Griffithsia sp. | In vitro, (Vero, HEK-293, Huh-7, MRC-5) | MERS-CoV | [92,93] | |
| Lectins | Various | Various | Various | HIV | [58] | |
| Κ-Carrageenan oligosaccharide | Rhodophyta | Not reported | In vitro (MDCK) | IAV | [94] | |
| Fucoidan | Phaeophyceae | Fucus evanescens |
In vitro (Vero, MT-4) In vivo (Murine) |
HSV-1/2, HIV, ECHO-1 | [95] | |
| Ulvan | Chlorophyta | Ulva pertusa | In vitro (DF-1) | ALV | [96] | |
| Xylomannan | Rhodophyta | Scinaia hatei | In vitro (Vero) | HSV | [97] | |
| p-KG03 | Dinophyceae | Gyrodinium impudium | In vitro (MDCK) | IAV | [98] | |
| AEX | Chlorophyta | Coccomyxa gloeobotrydiformis | In vitro (Vero) | IBDV | [99] | |
| GFP | Rhodophyta | Grateloupia filicina | In vitro (DF-1) | ALV | [100] | |
| PMG | Phaeophyceae | Not reported | HPV | [64] | ||
| Unknown | Loliolide | Phyllanthaceae (land plant) Phaeophyceae, Rhodophyta, Chlorophyta |
Phyllanthus urinaria Various |
In vitro (Huh-7.5) | HCV | [101] [102] |
| Polyphenol | Phaeophyceae Rhodophyta |
Ecklonia arborea Solieria filiformis |
In vitro (Vero) | MeV | [103] | |
| Pheophorbide | Chlorophyta | Dunaliella primolecta | In vitro (Vero) | HSV-1 | [104] | |
| Diterpenes | Phaeophyceae | Dictyota menstrualis | In vitro (Vero) | ZV | [105] | |
| Arthrospira extract | Cyanobacteria | Arthrospira fusiformis | In vitro (RC-37) | HSV-1 | [76] | |
| Dolastane | Phaeophyceae | Canistrocarpus cervicornis | In vitro (Vero) | [106] |
Endogenous defense is provided by the immune system. Compounds from algae can boost the innate immune system during viral transport through the vasculature, enhancing the activity of patrolling macrophages and neutrophils that are the body's first line of defense against invading pathogens (Table 3), (Fig. 5).
2.2.1. Virucidal and virustatic compounds from algae prevent vascular spread
Virucidal compounds attack virion particles in their free state, disrupting their surface integrity or penetrating inside the capsid to destroy their genome [81,82]. Virustatic compounds bind to the surface molecules of virions and hold them inert, preventing them from binding to cell receptors and initiating infection. Certain compounds derived from algae may exert virucidal or virustatic effects as they travel through the body's vasculature (Table 3). These compounds may represent an ideal form of treatment, as they do not invade the body's cells and may limit the development of viral resistance [83]. They can be injected or consumed orally, thus removing the need for administration by trained medical professionals. Many such chemicals are found in algae. Hudson et al. collected 16 species of Chlorophyte, Rhodophyte, and Phaeophyte macroalgae off the coast of British Columbia and found (but did not distinguish between) virucidal or virustatic activity from all of them [84]. Clearly, there exists a great deal of potential in investigating the virucidal/virustatic activity of algae-derived compounds. In this section, we explore purified compounds from algae that demonstrate virucidal/virustatic activity and note the difficulty in precisely elucidating antiviral mechanisms. While time-of-addition experiments can suggest that a compound interferes with an early stage of the viral replication cycle, such as binding or entry, pre-treating virions with compounds of interest is generally necessary to show that a compound acts on virions and not cells. Pre-treatment assays cannot distinguish between virucidal and virustatic compounds. In order to do so, imaging or centrifugation to isolate virion components is typically required to show virucidal activity. Virustatic action may be inferred by assays where an inactivated virion is treated with a dissociation agent that would then lead to reactivation and reveal the presence of a virustatic compound. Many studies do not carry out the rigorous examinations required for mechanism elucidation; nevertheless, we report on direct inactivating compounds in this section, noting deficits in knowledge where appropriate.
As we discuss various algae-derived antiviral compounds, we present information about their safety and pharmacokinetic profiles. Administration of drugs into the vasculature is dangerous as compounds are able to diffuse throughout the body and interact with different cell types, leading to unforeseeable and potentially negative outcomes. Where applicable, we present information about effective or inhibitory concentrations of antiviral compounds (EC50, IC50), measures of drug potency that indicate the dose of a drug required to inhibit viral replication or activity by 50%. We also report cytotoxicity indices (CT50), the concentration of a compound required to reduce cell viability in vitro by 50%. Wherever possible, we present the SI values of antiviral compounds which are calculated by dividing experimental EC50 or IC50 by CT50 values. Higher SI values demonstrate drug potency without cytotoxicity and suggest specific interactions between drugs and their viral targets. Drugs with a wide range of SI values are currently used in various pharmaceutical applications. For example, the cardiac glycoside digoxin has a therapeutic index of only 2–3 [85], while the opiate anesthetic remifentanil has an SI >30,000 [86]. While drugs with high SI values are safer, even drugs with low SI values can be utilized with proper care.
An additional concern to keep in mind throughout our discussion of AV compounds in the vasculature relates to drug diffusion and bioavailability. Drugs administered intravenously are often inefficiently transported through the circulation due to various properties including size, polarity, and proclivity to form bonds with bond with other molecules [87]. Thus, it is important to keep these various factors in mind when proposing antiviral candidates derived from algae to be administered in the vasculature. We discuss these factors where applicable.
2.2.1.1. MC15
Kamei et al. assayed extracts of 342 species of marine algae against salmonid infectious hematopoietic virus (IHNV), a rhabdovirus related to human rabies virus (Table 3). The extract of the brown alga Eisenia bicyclis showed particularly strong activity in vitro. The active antiviral compound was purified and shown to be MC15, a chlorophyll c2 derivative [88]. Time course experiments showed that MC15 directly inactivated IHNV with a minimum inhibitory concentration of 0.8 μg/mL. Data suggests MC15 may damage lipid membrane of enveloped viruses, as it showed inhibitory activity to other enveloped viruses with both DNA and RNA genomes but did not affect non-enveloped viruses.
Chen & Roca demonstrated that a variety of chlorophyll molecules and their derivatives from three species of Chlorophyta, Phaeophyte, and Rhodophyta macroalgae were bioavailable upon metabolism using an in vitro model [107]. This result suggests that MC15, a derivative of chlorophyll C, would be absorbed into the vasculature following metabolism, but further research using an animal model is necessary to confirm this result.
2.2.1.2. Monogalactosyl diacylglycerol (MGDG)
Hayashi et al. demonstrated that an MGDG fraction from the green microalga Coccomyxa sp. was actively antiviral against HSV-2 (Table 3) [60]. In vitro assays and subsequent microscopy investigation showed that MGDG damaged viral envelopes. The lipophilic MGDG molecule likely fuses with the viral envelope, disrupting its integrity. MGDG EC50 was <13 μg/mL with a SI of ~15. Topical administration of MGDG to the urogenital tract of female mice eliminated pathogenicity of HSV-2 infection, decreasing viral yields and lesion formation, and increasing survival rates.
MGDG may not be a viable candidate for oral administration as an antiviral drug, as research indicates that structurally similar digalactosyl diacylglycerides are degraded in the intestinal tract [108]. Diacylglycerols are important cell-signaling molecules [109], and while they may be regularly cleared from the blood, might nevertheless exert positive nutritional and antiviral effects when administered intravenously [110].
2.2.1.3. 7-Keto-stigmasterol
7-Keto stigmasterol (Table 3), a ketosteroid isolated from the green alga Prasiola crispa, demonstrated inhibitory activity against equine herpesvirus (EHV-1) [89]. Using in vitro assays and time-of-addition experiments, 7-keto stigmasterol demonstrated a direct virucidal effect on EHV-1 virions. Crucially, 7-keto stigmasterol only interacted with the virion and not the cell or complex formed by the virion attached to the cell. This is a promising finding for medicinal application, as it suggests that 7-keto stigmasterol will not damage cell surface integrity. 7-Keto stigmasterol demonstrated an EC50 value of 45 μM and SI from 20 to 47. Administering 100 μM of 7-keto stigmasterol resulted in 100% inhibition of EHV-1. As a lipophilic steroid molecule, the mechanism of virucidal action likely involves fusing with and disrupting the integrity of the viral membrane [89].
7-Keto stigmasterol belongs to the class of chemical compounds known as phytosterols. Phytosterols may play a role in preventing heart disease, although there are uncertainties regarding their safety [111]. Phytosterols are found in many plant oils along with long-chain fatty acids that are an essential part of the human diet. When injected parenterally as nutritional supplements, these long chain fatty acids are accompanied by phytosterols. Phytosterols are not metabolized in the human body and are excreted by the hepatobiliary system. Evidence suggests that the overaccumulation of phytosterols in the blood may lead to liver disease [112]. Thus, research into the use of 7-keto stigmasterol should be performed with caution.
2.2.1.4. Loliolide
Loliolide is a lactone, produced in the degradation of carotenoids, that has been isolated in many species of terrestrial plants and algae and has demonstrated antimicrobial properties [113]. Chung et al. found that loliolide extracted from the medicinal (non-algal) plant Phyllanthus urinaria demonstrated AV activity against hepatitis C virus (HCV) in vitro (Table 3) [101]. Loliolide demonstrated direct inactivating effect on virion particles, but a virucidal mechanism was not observed. Loliolide may interact with the viral envelope or glycoproteins in a virustatic manner, rendering virions unable to bind cell receptors or fuse with the cell membrane. Loliolide inhibited HCV with an EC50 value of 3.1 μM and SI of 62.6. Interestingly, an unpurified fraction from the Phyllanthus extraction demonstrated even stronger viral inactivation than loliolide, indicating that there are other compounds contained in the cytosol of Phyllanthus cells that may be promising avenues for investigation. While the study reported on here did not use loliolide derived from algae, because the same compound has been observed in Phaeophyceae, Rhodophyta, and Chlorophyta algae it supports the use of algae for producing AV compounds, especially as they generally exhibit faster growth than terrestrial plants [114]. Loliolide is found throughout many different organisms and has been incorporated into many folk medicines around the world. It is used in diverse remedies as an anti-bacterial, anti-inflammatory, anti-depressive, and anti-cancer agent. Loliolide is thus likely a safe candidate for antiviral drug development [115].
2.2.1.5. Lectins
Algal lectins are carbohydrate-binding agents that recognize and bind specific residues on viral glycoproteins with a high degree of specificity. Lectins do not covalently alter the viral glycoproteins or demonstrate virucidal properties, but rather, exert virustatic activity by preventing their recognition of cell surface receptors [58]. Several lectins isolated from cyanobacteria and eukaryotic algae have recently shown anti-HIV activity with high efficacy and low cytotoxicity (Table 3) [58]. O'Keefe et al. demonstrated that cyanovirin-N, a lectin extracted from marine cyanobacterium Nostoc ellipsosporum irreversibly inactivates HIV virions in vitro. Cyanovirin-N is a 101 amino acid, 11 kDa protein that displays high affinity for HIV glycoprotein 120 (gp120) [91]. In vitro testing revealed that cyanovirin-N inhibited various HIV laboratory strains and primary isolates with EC50 values <5 nM. Crucially, cyanovirin-N binds viral gp120 in an irreversible manner and prevents viral adhesion with target cells. Treatment with strong denaturing agents could not dissociate the cyanovirin-N-gp120 complex, indicating that cyanovirin-N exerts a powerful virustatic effect on HIV virions. Cyanovirin-N did not interact with cell receptors.
One recent study showed that the lectin GRFT from the red seaweed Griffithsia sp. was able to bind the spike (S) protein of Middle East Respiratory Syndrome Coronavirus (MERS-CoV) and prevent its interaction with target cells in vitro [92]. Other studies have shown how GRFT demonstrates a broad spectrum of activity against other enveloped and non-enveloped viruses, and so while the predominant mechanism of this lectin is likely the direct inactivation of virions, there may yet be other mechanisms at play as well [93].
In addition to their use as therapeutic compounds, lectins may even improve the absorption and bioavailability of other drugs by facilitating mucoadhesion, cytoadhesion, and cytoinvasion [116]. Zhang et al. showed that insulin coupled to three different lectins showed high bioavailability when administered orally [117]. Additional research performed by Pooja et al. indicates that the anticancer drug PTX is more bioavailable upon oral administration when conjugated to a lectin targeting ligand [118].
2.2.1.6. Polyphenol
In a survey of polyphenol-rich extracts seaweed, brown (Phaeophyte) macroalga Ecklonia arborea and red (Rhodophyta) macroalga Solieria filiformis showed inhibitory activity against measles virus (MeV) in vitro (Table 3) [103]. E. arborea and S. filiformis extracts showed IC50 values against MeV of 3 and 0.5 μg/mL respectively. Time-of-addition assays indicate that the extract directly interacts with virions, preventing infection into target cells. Other sulfated polysaccharides, also extracted from seaweed, combined with the polyphenol extract showed even stronger inhibition. No cytotoxicity was detectable in the extracts, with E. arborea and S. filiformis showing SI values of >3750 and >576.9 respectively, indicating strong potential for drug development. This study did not differentiate between virucidal and virustatic activity, however the authors suggest that the extract may be used after infection to prevent viral spread throughout the body.
An additional study provides evidence that polyphenols exhibit a direct inhibitory, virustatic effect on virion particles [119]. In this investigation, administration of the polyphenols eckol, dieckol, and phlorofucofuroeckol at concentrations ranging from 3.8 to 5.4 μM completely blocked porcine epidemic diarrhea virus binding to target cells in vitro. Time course experiments suggested that the binding of the viral S-protein to prevent interaction with surface receptors mediated the activity of these compounds.
Polyphenols are abundant in many commonly consumed plants. They are a regular part of our diet and have recently garnered attention as powerful antioxidant compounds. Polyphenols are typically perceived as safe for consumption, although their bioavailabilities and health effects differ widely among various polyphenol classes [120].
2.2.1.7. Pheophorbide
Ohta et al. found that an extract of Dunaliella primolecta, a green alga, had strong inhibitory activity against HSV-1 and HSV-2 in vitro (Table 3). Purification of the extract revealed that the active AV compounds had novel pheophorbide-like structures [104]. Pheophorbide is a product of chlorophyll degradation. In time-of-addition assays, pheophorbide showed a strong direct inactivation or denaturing of viral activity. Pheophorbide demonstrated an EC50 of 5 μg/mL, 100% inhibition at 10 μg/mL and no observable toxicity at 40 μg/mL. The study did not investigate a potential virucidal vs. virustatic effect, however due to its potency, low toxicity, and ubiquity of chlorophyll across the various groups of algae, pheophorbide might be a promising candidate for drug development. Chen & Roca demonstrated that products of chlorophyll breakdown from Chlorophytes, Rhodophytes, and Phaeophytes are bioavailable upon breakdown. Pheophorbide C was the most absorbable compound surveyed, suggesting high potential for antiviral drug development [107].
2.2.1.8. Diterpenes
Cirne-Santos et al. examined the AV activities of extracts from the brown alga (Phaeophyceae) Dictyota menstrualis against ZV in vitro and found that two extracts containing mainly diterpenes had strong inhibitory potential (Table 3) [105]. An extract fraction (FAc-2) rich in cyclic diterpenes exerted a virucidal pattern of activity against ZV with an EC50 of 0.85 μg/mL and SI of 595.06. A 20 μg/mL treatment inhibited >90% of viral activity. While the investigators speculated that diterpenes in the FAc-2 extract might interfere with the viral capsid or the genome directly, the virucidal assay carried out in this experiment did not implicate a specific mechanism. However, the extract did not interfere with viral absorption, suggesting that a virucidal or virustatic mechanism was at play.
Further research by the same group found anti-Chikungunya virus (CHIKV) and anti-ZV activity from an extract of the brown alga (Phaeophyceae) Canistrocarpus cervicornis in vitro and purified dolastane (a subtype of diterpene) as the actively antiviral compound [106]. While the crude extract was fairly potent against ZV and CHIKV with SI values of 203 and 178 respectively, dolastane was even more potent with experimental SI values of 1246 and 730 respectively. The EC50 values of dolastane against both viruses were <3.5 μg/mL. Dolastane demonstrated a direct inactivating effect on virions, but it is uncertain whether or not this mechanism was due to a virucidal or virustatic effect. However, due to dolastane's low molecular weight, inability to form many hydrogen bonds, remarkable SI, and the relatively high potency of the algal crude extract, this group of algae and dolastane-related compounds deserve more scrutiny as potentially potent, accessible antiviral agents. This is especially relevant in the Global South, the regions of the world most affected by ZV and CHIKV.
The diterpenes discussed here are small molecules, with likely high corresponding bioavailabilities. Research has shown that diterpenes from other natural sources have high bioavailability and low toxicity after oral and intravenous administration. Thus, diterpenes from algae are promising candidates for antiviral drug development [121].
2.2.1.9. Arthrospira extract
Sharaf et al. examined the AV activity of cold water, hot water, and phosphate buffer extracts of the cyanobacterium Arthrospira fusiformis and found direct inactivating effects on HSV virions in vitro (Table 3) [76]. When pre-treated with these extracts, viral replication was inhibited ~90%. No mechanism nor bioactive compound was identified in this experiment; however, the authors postulate that the buffer extract contained allophycocyanin as an active component. This study used Arthrospira extracts from Egyptian and American cyanobacteria and found similar activities in both strains. Importantly, this demonstrates that even crudely extracted compounds from geographically removed locations can exert powerful bioactivities which bodes well for accessible drug production.
Additional studies have shown that phycocyanin, a similar phycobiliprotein from cyanobacteria, is bioavailable and non-toxic when administered orally or via injection and may also have some powerful anti-inflammatory effects [122,123]. Thus, allophycocyanin is a promising virucidal compound from Arthrospira that merits further research.
2.2.1.10. Phycobiliproteins
One experiment examined the AV activity of a Spirulina (Arthrospira) platensis extract against two bacteriophages, MS-2 and ΦX-174 in vitro (Table 3) [90]. MS-2, an ssRNA virus, is commonly used as a model for human poliovirus, hepatitis A virus, and enterovirus. ΦX-174 is a ssDNA virus commonly used as a model for hepatitis B virus, hepatitis C virus, and HIV. An extract containing phycobiliproteins showed powerful virucidal action against both phages, severely altering the morphological characteristic of their capsids and interfering with internalization into E. coli. Unlike some of the other viruses we have previously examined, neither of the phages studied in this experiment have lipid envelopes. Thus, it seems that the phycobiliproteins are interacting with the protein capsid directly to exert virucidal activity. This virucidal effect impaired replication and lowered viral titer. Phycobiliproteins may thus hold some promise as useful therapeutics against non-enveloped viruses such as rhinoviruses, polioviruses, and noroviruses.
Mysliwa-Kurdziel & Solymosi reviewed phycobiliproteins used in medicinal applications, and concluded that phycobiliproteins demonstrate bioavailability without any evidence of cytotoxicity when administered via the oral route or injection [124]. Thus, phycobiliproteins are potentially safe, effective virucidal compounds. The Spirulina cultures were collected from the Nile River in Egypt, supporting the notion that potent antivirals can be derived from accessible sources in diverse environments.
2.2.1.11. Caulerpin
Esteves et al. found that caulerpin, an alkaloid derived from the green alga Caulerpa racemosa showed a strong direct inhibitory pattern of activity against CHIKV in vitro [125] with an EC50 value of 3.1 μg/mL and an SI of 736.62 (Table 3). A concentration of 5 μg/mL caulerpin was able to exert a 100% inhibitory effect in pre-incubation virucidal assays. No investigation was made to differentiate between the virucidal and virustatic mechanisms of caulerpin. However, due to caulerpin's low MW, tendency to engage in minimal hydrogen bonding, and high SI, this compound deserves further investigation as a potentially potent AV compound. Furthermore, caulerpin has recently been proposed as a novel inhibitor of the SARS-CoV-2 main protease enzyme, suggesting that this compound may have multiple promising modes of AV action [126].
2.2.1.12. Polysaccharides
Polysaccharides are long chains of sugar molecules. They are remarkably diverse in their sugar constituents, branching, and size. Polysaccharides are synthesized in cells for many purposes including energy storage, structural stability, and cell signaling. Antiviral polysaccharides frequently have virustatic mechanisms based on negatively charged molecules embedded within their structure interacting with positively charged regions of the viral surface. For brevity's sake and due to their similarities in mechanism, we describe a few of the best characterized polysaccharides from three major eukaryotic algal phyla showing virustatic activity and list others (Table 3). We also describe some challenges associated with using polysaccharides as antiviral drugs and potential solutions.
The sulfated polysaccharide carrageenan has virustatic and potentially virucidal properties [127]. Wang et al. examined a 2 kDa κ-Carrageenan oligosaccharide and found that it exerted a direct inactivating effect on IAV in vitro [94]. The oligosaccharide had an IC50 value of 32.1 μg/mL and an SI of 26.7. In this study, the carrageenan oligosaccharide had a direct inactivating effect on virions, likely mediated by binding of anionic sulfate groups in the oligosaccharide to positively charged regions of the viral surface. The finding that carrageenan was unable to bind the cell surface supports the notion that it exerts a virustatic effect. Interestingly, a different study suggested that carrageenan was able to modify the HSV glycoprotein gB, thus postulating that carrageenan might exert some virucidal as well as virustatic activity [128]. Carrageenan may have elicited a temporary, reversible change in the virion structure and so uncertainty about its precise inactivating mechanism remains. Nevertheless, the oligosaccharide form of carrageenan has potential as an AV drug.
Because carrageenan is generally high MW, it is perhaps not well-suited to transport through the vasculature although some research indicates that carrageenan might be available for a short time in the vasculature after injection [129]. Indeed, this is one common issue frequently encountered in the use of polysaccharides as intravenous drugs [130]. However, the oligosaccharide is two orders of magnitude smaller than typical carrageenan molecules, more easily absorbed, and much more mobile traveling through the body. This study showed that carrageenan oligosaccharide lacked cytotoxicity, but other concerns about carrageenan cytotoxicity have been raised, especially in oligosaccharide form [67]. Thus, this question merits further investigation. Carlucci et al. found that the administration of carrageenan was able to select for a drug-resistant viral mutant, posing an additional concern regarding carrageenan's use as a therapeutic intervention [128].
Another well-characterized polysaccharide showing diverse bioactivities is fucoidan, derived from brown (Phaeophyceae) macroalgae such as kelp and sargassum. Krylova et al. examined the AV effect of two fucoidans from Fucus evanescens for AV activity against HIV, HSV-1, HSV-2, and enterovirus (ECHO-1) in vitro and in vivo [95]. One fucoidan was of high MW (~160 kDa), and one was enzymatically degraded to a low MW (~50 kDa). Assays showed that both fucoidans directly inactivated HSV-1/2 with mean SI values ~19. Both fucoidans showed moderate direct inactivation of HIV with SI values ~8. Direct inactivation of ECHO-1 was minimal (SI ~3). Fucoidan's direct inactivating effects are due to a virustatic effect characterized by interactions between anionic sulfate groups on the fucoidan polysaccharide and positively charged regions of the viral surface. Later in vivo testing showed that both fucoidans had a protective effect against intravaginal HSV-2 infection in a murine model when injected into the intraperitoneal space, indicating that fucoidan is bioavailable upon injection. Protective effects in vivo were also observed in a recent study which found that the oral administration of fucoidan derived from Laminaria japonica protected against murine norovirus infection and increased survival rates in a murine model [131]. In vitro testing showed that pretreatment of virions with fucoidan inhibited infection. An additional study provided evidence that fucoidan interacts directly with the HSV virion. In time-of-addition experiments, Sun et al. found that fucoidan did not have a protective effect against HSV-2 infection when pretreated with the cell. However, when added with the virus during infection, infection was significantly inhibited [132]. In addition to inhibition of HSV, one recent study also showed that fucoidan is able to bind the SARS-CoV-2 S-protein, preventing the virion from binding to its target cells [133].
The sulfated polysaccharide Ulvan is derived from green (Chlorophyta) macroalgae. Sun et al. investigated the inhibitory potential of ulvans with varying molecular weights on avian leukosis virus (ALV) in vitro [96]. At 2 mg/mL, cell viability remained >95% indicating minimal cytotoxicity. In AV assays, ulvan showed an inhibitory effect at the initial stage of viral adhesion but had no effect when pretreated with the cell. Thus, ulvan is assumed to interact with virions directly in a virustatic manner. The results suggest that the negatively charged sulfate groups likely bind to positively charged regions of surface glycoproteins. In this study, ulvans with MWs of ~2–~160 kDa were surveyed for their AV activity. A 4.3 kDa ulvan showed the greatest inhibitory potential. Another study found that ulvan inhibited Newcastle Disease Virus (NDV) spread between infected cells but did not interact with the virion particle, displaying neither virucidal nor virustatic activity [134]. This discrepancy in result is hardly surprising, considering the diversity in viral envelope proteins and natural variability of polysaccharides between individual organisms. More research is needed to investigate the potential for developing directly inactivating AV drugs from ulvan.
Carrageenan, fucoidan, and ulvan are three extensively characterized polysaccharides representing the three large taxa of red, brown, and green macroalgae. Despite the immense diversity of algae, these three polysaccharides largely operate through a similar mechanism: anionic regions of the polysaccharides consisting primarily of sulfate and uronic acids interact with viral glycoproteins to prevent glycoprotein-cell receptor complex formation. This interaction is virustatic, holding virions inert and preventing infection. A similar mechanism has been suggested across many other types of algae-derived polysaccharides, including xylomannan [97], p-KG03 [98], AEX [99], GFP [100], and PMG [64].
Polysaccharides are promising compounds for drug development as they display a wide range of bioactivities. However, using polysaccharides as intravenous drugs is challenging, due to their poor transport through the vasculature, difficulty in diffusing across membranes, anticoagulant activity, and potential cytotoxicity [130]. One potential solution is degradation into smaller oligosaccharides that maintain similar bioactivities without the disadvantages of unwieldy molecular size. Some studies show improved AV activity from low MW polysaccharides produced through oxidative degradation [96,100]. However, concerns over the health effects of degraded polysaccharides necessitate further investigation.
2.2.1.13. Conclusion
The compounds reported on in this section (Table 3) directly inactivate virions by either binding them irreversibly, preventing them from engaging with their target cells, or compromising the virion's structural integrity. If introduced into the vasculature by consumption or injection, these compounds may prevent virions from interacting with their target cells. While some of the investigations examined in this section distinguished between virucidal and virustatic mechanisms, several did not. This necessitates more research, as the compounds in this section represent some of the most auspicious currently known antiviral compounds extracted from algae.
Many of the virucidal and virustatic compounds reported on in this section were studied using in vitro models, which do not fully represent living systems. It is likely that some of the bioactive compounds reported on here would show harmful side effects or lose potency in vivo. However, the compounds described this section did not demonstrate harmful side effects in vitro using a variety of cell lines. Furthermore, many of the compounds preferentially bind virions and do not interact with cell surfaces or receptors at all, suggesting that more research is needed on the clinical applications of these compounds as antiviral drugs. Evidence suggests that the compounds reported here could be administered orally or via injection after infection and may prevent viruses from spreading through the vasculature to reach and invade their target tissues. This is preventative treatment, as inactivating virions before they reach their targets avoids negative cytopathic effects entirely. Due to the diversity of compounds, algae, and viruses (enveloped and non-enveloped) reported on in this section, directly inactivating antiviral drugs from algae should be the subject of further investigation.
2.2.2. Phagocyte-stimulating algal compounds enhance innate immune response
Viruses that invade past layers of epithelial tissue into the vasculature may also be inactivated by the patrolling cells that are components of the body's innate immune response. In the vasculature, viruses encounter a variety of mobile antimicrobial cells, including macrophages and neutrophils, which are the immune system's first line of defense against invading pathogens [28,135]. Macrophages are large, phagocytic cells encountered throughout the body's tissues and circulating in the blood. They are a crucial part of the primary immune response after viral infection [28,29]. Neutrophils circulate in the blood and are the first cells recruited to sites of infection or injury and are responsible for antimicrobial defense via phagocytizing or caging novel pathogens [136]. Algae-derived compounds can boost macrophage and neutrophil activity, which may have an important protective role in preventing viral infection (Table 4 ).
Table 4.
Algae-derived compounds stimulate phagocyte activity.
| Compound | Species classification | Species | Activity | Model | Reference |
|---|---|---|---|---|---|
| Fucoidan | Phaeophyceae | Cladosiphon okamuranus | Macrophage-activating | In vitro (murine macrophage) | [137] |
| Ulvan | Chlorophyta | Ulva rigida | Macrophage and neutrophil-activating | In vitro (Turbot phagocyte) | [138] [139] |
| Laminaran | Phaeophyceae | Laminaria hyperborea | Macrophage-activating | In vitro (Salmo salar macrophages) | [140] |
| Alginate | Phaeophyceae | Laminaria digitata | Neutrophil-activating | In vivo (Oncorhynchus mykiss) | [129] |
| Carrageenan | Rhodophyta Rhodophyta |
Chondrus ocellatus Not reported Eucheuma spinosa |
Macrophage-activating Macrophage-destroying Macrophage-inhibitory |
In vivo (Cyprinus carpio) In vivo (Murine) In vitro (L929) |
[143] [144] [145] |
| Neoagarohexaose | Not reported | Macrophage-activating mediates AV activity | In vitro RAW264.7 | [146] | |
| Red algae extracts | Rhodophyta Rhodophyta |
Porphyra yezoensis Gracilaria verrucosa |
Macrophage-activating and proliferation-stimulating Macrophage stimulation |
In vitro & In vivo (murine macrophages) In vivo (murine macrophages) |
[147,148] [149] |
2.2.2.1. Fucoidan
In addition to its potential as a topically applied viral blocking agent, the polysaccharide fucoidan may also have a positive effect on the function of patrolling macrophage activity. Teruya et al. determined that fucoidan from Laminaria angustata has an activating effect on macrophages in vitro via interactions with the toll-like receptor 4 (TLR4), cluster of differentiation 14 (CD14), and scavenger receptor class-A (SRA) receptors as well as the mitogen-activated protein kinase (MAPK) signaling pathway (Table 4) [137]. Later, the same team determined that fucoidan stimulated production of nitric oxide (NO), tumor necrosis factor-α (TNF- α), and interleukin-6 (IL-6), molecules produced by macrophages that are crucially important to immune response [150]. They showed macrophage activation and demonstrate that fucoidan is an immunostimulant. An additional study suggested that phagocyte-stimulating agents might have an important role in defense against microbial infection. This investigation showed that in addition to stimulating NO, TNF-α, and IL-6 production, fucoidan elevated macrophage phagocytosis and lysosome enzyme activity [151]. Fucoidan was mitogenic (encouraging cell proliferation) in lymphocytes and macrophages. These results were further supported by a recent study showing that fucoidan induced macrophage activation via the MAPK and NF-κB signaling pathways [135]. Significantly, this study demonstrated the immunostimulatory capacity of an LMW fucoidan and emphasized its potential for improved bioactivities and absorption.
2.2.2.2. Ulvan
Ulvan is an acidic, sulfated polysaccharide produced in the cell walls of many green macroalgae (e.g., Ulva spp., Chlorophyta). Castro et al. showed that a crude extract from Ulva rigida stimulated turbot (fish) macrophage and granulocyte activity as indicated by an increase in reactive oxygen species (ROS) (Table 4). An increase in ROS (primarily NO and H2O2) is a crucial component of AV response and is important for recruiting phagocytes to help repel pathogens. This activity was largely attributable to polysaccharides [138]. The compound from Ulva rigida stimulating this ‘respiratory burst’ was later identified as an ulvan [139]. The desulfated ulvan did not show stimulatory activity, indicating the importance of the anionic sulfate groups in this cell signaling pathway. Significantly, small and large polysaccharides both stimulated the macrophage/granulocyte mixture, which is of considerable importance as large polysaccharides are not easily absorbed or transported through the bloodstream. Ulvan probably stimulates phagocyte activity through interaction with cell-surface receptors. One recent study completed a chemical synthesis of Ulvan. The synthetic Ulvan displayed immunomodulatory activities, augmenting the phagocytic activity of macrophages in vitro [152].
2.2.2.3. Laminaran
Laminaran is a polysaccharide storage product derived from brown macroalgae (Phaeophyceae) and diatoms. Dalmo et al. found that laminaran activates the salmonid (fish) non-specific immune system, of which macrophages are a central component (Table 4) [140]. Salmonid macrophages stimulated with laminaran in vitro contained more vacuoles and granules, exhibited increased phagocytotic and pinocytotic activity, generated elevated levels of superoxide, and showed higher activity of lysosomal enzymes when compared to control cells. These are important components of the non-specific response that phagocytes use to destroy invading pathogens, including viruses. Laminaran was an effective immunomodulator of salmonid macrophages. This finding is supported by further research conducted by El-Boshy et al., 2010, which found that laminaran from the brown (Phaeophyte) macroalga Laminaria japonica stimulates the Oreochromis niloticus (tilapia) immune system against bacterial infection [153]. Because it can be absorbed from the intestines into circulation, laminaran has potential as an orally applied immune-boosting drug for both fish (in aquaculture operations) and humans.
2.2.2.4. Alginate
Alginate is an acidic polysaccharide derived from the cell walls of brown macroalgae (Phaeophyceae). Peddie et al. showed that alginate from Laminaria digitata stimulates neutrophil migration and phagocytosis using an in vivo piscine model (Table 4) [142]. In this study, the injection of alginate caused a significant migration of leukocytes into the peritoneal cavity, with an increased proportion of neutrophils when compared to controls. Macrophage count decreased as a result of alginate administration. The change in macrophage vs neutrophil concentrations is striking, as macrophages are typically more concentrated in the piscine peritoneum. Alginate caused an elevated expression of interleukins (IL) 1β and 8 as well as TNF-α. The elevated expression of IL-8 may explain selective neutrophil activation, as IL-8 is a chemoattractant cytokine showing a great deal of specificity for neutrophils. More research is required to elucidate specific interactions between alginate and macrophages compared to neutrophils. In addition to altered migratory patterns and gene expression, more leukocytes exposed to alginate exhibited phagocytic activity. Alginate is thus a promising immunostimulant with special neutrophil modulating properties. This is especially important in viral infection, as neutrophils are the first cell recruited to sites of viral infection to clear pathogens and initiate a proinflammatory response [136].
2.2.2.5. Carrageenan
Shin et al. showed that the peritoneal injection of κ-carrageenan caused the recruitment and migration of phagocytes from the head kidney (analogous to the mammalian adrenal gland) to the peritoneum using an in vivo carp model (Table 4) [143]. These migrated phagocytes (especially macrophages) demonstrated increased levels of phagocytosis and cleared a harmful bacterial infection resulting in higher survival rates compared to controls. While the model was not challenged with a viral infection, phagocytes are also important in clearing viral pathogens, suggesting that κ-carrageenan can recruit the immune system against viruses too.
Ogata et al. showed mixed results following the administration of ι-carrageenan. Carrageenan primed leukocytes for production of TNF, an important component of response to infection, but also destroyed macrophages [144]. An additional study demonstrated that ι-carrageenan is inhibitory to murine macrophage activity [145]. These studies warn of the potential mixed effects of immunomodulating agents. ι- and κ-carrageenan differ slightly in structure [154] and this variation clearly leads to a difference in functional properties. Both carrageenans participate in cell signaling during important phases of immune response, but more research is needed to elucidate the specific mechanisms of each carrageenan's role. A thorough structure-function analysis may elucidate certain types of carrageenan that augment beneficial effects of phagocytes without incurring a serious negative toll.
2.2.2.6. Neoagarohexaose (NA6)
The oligosaccharide NA6 is derived from agarose, a major component of red algae (Rhodophyta) cell walls. Kim et al. examined the effect of NA6 administration on immune activation in vitro using a RAW264.7 cell line (Table 4) [146]. This study determined that NA6 is an agonist for the TLR4 signaling pathway, important in the production of proinflammatory cytokines (TNF, interleukins) and interferons (IFNs). This research demonstrated that NA6 administration primed macrophage anti-norovirus response, mediated by IFN signaling. NA6 boosted induction of IFN-β and upregulated IFN-regulatory factor-1, an IFN-stimulated gene. Oral administration of NA6 activated TLR4 signaling and thereby reduced norovirus loads [146].
2.2.2.7. Red algae extracts
Yoshizawa et al. examined the in vitro and in vivo macrophage-boosting effect of sulfated polysaccharide fractions isolated from the red (Rhodophyta) macroalga Porphyra yezoensis (Table 4) [147]. Two fractions were isolated using water and acid solvents. Carrageenan may have been a principle component of both fractions. The water-soluble fraction activated macrophages as indicated by elevated levels of nitric oxide production. The acid-soluble fraction did not increase nitrite production. The water-soluble fraction increased production of IL-1 and TNF, while the acid-soluble fraction only stimulated production of TNF. In vivo effects of the fractions on phagocytic activity were measured; the acid-soluble fraction stimulated phagocytosis more strongly than the water-soluble fraction. Increases in phagocytosis stimulated by the acid-soluble fraction were due to elevated activity of the phagocytes, as measurements of organ mass revealed that the number of phagocytes remained the same. The water-soluble fraction, in contrast, increased the number of phagocytes but not their relative activity. The differences in activities between the fractions is attributable to variation in content: the water-soluble fraction had a somewhat higher protein concentration, a lower proportion of 3,6-anhydrogalactose, and a higher molecular weight. Each fraction engaged with signaling pathways in slightly different manners, however both demonstrated activating effects of murine phagocytes that may have immunopotentiating significance.
Yoshizawa et al. later expanded this investigation and found that desulfation of the acid-soluble fraction reduced its ability to stimulate macrophage activity in vitro [148]. Increased sulphation of the fraction did not increase activity, indicating the importance of both the sulphation and specific physical conformation of the polysaccharides in their engagement with the cell-signaling pathway. Specifically, researchers identified the sulfate group at position C-6 as essential to macrophage stimulation. The initial molecular weight of the acid-soluble polysaccharide was 400 kDa; digestion into smaller pieces decreased viscosity and increased macrophage stimulating activities. This is an important observation, as bulky compounds often hamper drug delivery. A partially digested oligosaccharide represents a more promising drug.
Yoshizawa et al. performed further research on an enzymatically degraded water-soluble polysaccharide fraction of Gracilaria verrucosa, another type of red (Rhodophyta) macroalga [149]. Researchers determined the principle component of the polysaccharide fraction was a sulfated galactan, potentially carrageenan. Intraperitoneal injection or oral administration of the polysaccharide fraction stimulated macrophage activity in vivo using a murine model. Intraperitoneal injection of the polysaccharide elevated macrophage numbers and increased production of radical oxygen species (ROS). Chemiluminescence assays showed that oral consumption of polysaccharide also stimulated macrophage activity. This is a promising result, as it indicates that oral consumption of the polysaccharide fraction from red macroalgae has an immunostimulatory effect, thus suggesting adequate bioavailability and raising the possibility for easily administered medicinal food products.
2.2.2.8. Conclusion
As viruses travel through the lymph and blood, they encounter mobile defensive cells of the immune system, namely the phagocytes termed macrophages and neutrophils. These cells are a crucial component of the body's innate immune response. In this section, we have shown how algae-derived compounds boost the activities of these cells by stimulating phagocytosis, inflammatory response via the generation of ROS, and further immune system activation via interleukins, cytokines, and other signaling pathways.
The mobile phagocytic cells of our immune system are able to clear the body of many viruses before they initiate large-scale infections. Compounds from algae show the ability to boost the activity of these cells (Table 4), exerting an important protective effect in the continual effort to prevent against infection.
2.2.3. Viral passage through the vasculature–conclusion
After invasion of the body, many viruses must be transported to specific target tissues before they can replicate. Indeed, viruses do not exert their most harmful effects until they reach their target tissues. By intervening to counter viral infection during transport through the lymph and blood, harmful cytopathic effects can be averted. In this section, we have seen how compounds derived from algae can directly inhibit and degrade virions, thus offering promising sources of drugs than can be introduced to the vasculature. We have also seen compounds that boost innate immune response, specifically augmenting the activities of macrophages and neutrophils, cells of the immune system that travel throughout the body searching out and destroying harmful infectious particles. By these two mechanisms, algal-derived compounds can protect against the worst effects of infection that come later when viruses reach their target tissues. In the next section, we will examine algae-derived compounds that can specifically interfere with stages of the viral replication process at the cellular level.
2.3. Algal compounds inhibit the viral replication cycle
A virion that arrives at its target tissue will begin the replication cycle. This involves invading target cells, hijacking cellular machinery, and producing many hundreds or thousands of copies of viral genetic material and accompanying proteins to the detriment of cellular function and survivability (Fig. 5). Often, the accumulation of virions within the cell will cause lysis, thus releasing copies of the virus in the environment to carry out the same process all over again. Other viruses use normal cellular processes to facilitate their release, allowing the cell to remain alive longer. Some viruses cause chronic disease by incorporating their genetic material into the host genome and lying dormant for years before reemerging to continue the infection cycle [27].
The replication process is tremendously variable between different types of virus. This variability is concerning from a healthcare standpoint, as it makes the development of broad-spectrum antiviral drugs difficult. However, the specificity and complexity of a virus' interactions with its given target cell allows for directed interventions that operate at different timepoints during the replication cycle. In this section, we explore the cycle of a generalized viral infection at the cellular level and examine the steps at which algae-derived compounds might be deployed to combat viral infection. It is important to note that each antiviral compound presented in this section faces the same challenges with bioavailability and cytotoxicity as those presented in Section 2.2. Many of the studies discussed here are performed in vitro, which may highlight promising compounds for antiviral drug discovery, but does not offer a comprehensive picture of drug administration, transport, absorption, and delivery. Every compound has a characteristic pharmacokinetic profile which means that drug development must proceed deliberately and meticulously to guarantee drug efficacy and safety.
2.3.1. Adhesion
A virion that comes into contact with its target cell must bind to the cell surface before it is able to be internalized. Surface molecules on viral capsids or envelopes bind to receptors on cell surfaces. Viruses have evolved the ability to bind many human host cell receptors; however, the receptors have other functions besides the binding of virions. For example, the ligand of human RHV, VP1,3, binds the important intracellular adhesion molecule ICAM-1 (the cellular receptor evolved to carry out cell signaling and important to inflammatory response). Some viruses bind multiple receptors on their target cells [28]. Many algae-derived compounds have demonstrated the ability to prevent viral adhesion, thus blocking infection. Preventing adhesion is another ideal point of interference in the viral replication cycle, as treatments preventing adhesion are non-invasive and thus potentially carry reduced risk of interference in important cellular processes. This ‘prevention’ of infection at the cellular level is akin to masking the cell. However, it is important to note that introducing exogenous compounds that that interact with cells may have many unforeseeable, potentially harmful downstream effects.
In Section 2.2.1, we discussed many algae-derived compounds that prevent viral binding by directly interacting with the virion independent of the target tissue, either rendering the virion inert or degrading it. In this section, we discuss compounds that prevent viral adhesion in context of a target cell. Some compounds discussed here compete with the virus for binding cellular receptors and some bind only the complexed virion and surface receptors but not either one independently. Some may interfere with conformational changes of proteins or block signaling pathways essential for viral adhesion (Table 5 ).
Table 5.
Algae-derived compounds prevent viral adhesion to target cells.
| Compound | Species classification | Species | Virus | Model | Reference |
|---|---|---|---|---|---|
| Palmitic acid | Phaeophyceae | Sargassum fusiforme | HIV-1 | In vitro (MΦs & PBL) | [155] |
| Galactan | Rhodophyta |
Gracilaria corticata Schizymenia binderi |
HSV-1/2 HSV-1/2 |
In vitro (Vero) In vitro (Vero) |
[156] [157] |
| Calcium spirulan | Cyanobacteria |
Spirulina (Arthrospira) platensis Spirulina (Arthrospira) platensis |
HSV-1, CTMV, MeV, MuV, IAV, HIV-1 HSV-1 |
In vitro (Vero, MDCK, HeLa, HEL, MT-4) In vitro (Vero) |
[158] [159] |
| Allophycocyanin | Cyanobacteria | Spirulina (Arthrospira) platensis | Enterovirus-71 (EV71) | In vitro (Vero, human rhabdomyosarcoma) | [160] |
| Arthrospira extract | Cyanobacteria | Arthrospira platensis | HSV-1 | In vitro (Vero) | [161] |
| Nostoflan | Cyanobacteria | Nostoc flagelliforme | HSV-1 | In vitro (Vero) | [162] |
| Chlorella polysaccharide extract | Chlorophyta | Chlorella vulgaris | HSV-1 | In vitro (Vero) | [163] |
2.3.1.1. Palmitic acid
Lee et al. isolated a saturated fatty acid known as palmitic acid from the brown (Phaeophyte) macroalgae Sargassum fusiforme and determined that it exerted anti-HIV-1 activity in vitro against CXCR4 and CCR5-tropic viruses (Table 5) [155]. Treatment with 100 μM of palmitic acid reduced infection in both CXCR4 and CCR5 viruses by up to 70%. Treatment with 22 μM of palmitic acid inhibited CXCR4 infection in peripheral blood lymphocytes by 95% and treatment with 100 μM of palmitic acid inhibited CCR5 infection in primary macrophages by 90%, indicating effective broad-spectrum activity. Fluorescence spectroscopy demonstrated that Palmitic acid prevented HIV binding to T-cells by competing for the binding of the CD4 receptor necessary for viral attachment. This study found that the use of an identical molecule with a slightly shorter hydrocarbon chain was unable to prevent HIV infection, emphasizing the specificity of palmitic acid-CD4 receptor binding.
Supporting the proposed anti-adhesion mechanism demonstrated here by palmitic acid, an earlier study found that palmitic acid inhibits bacteriophage Φ6 replication but does not inactivate virions [164]. Palmitic acid only prevented infection at the early stage of infection, suggesting a similar virion-independent inhibition of attachment. Strikingly, the monounsaturated palmitoleic acid, which is otherwise identical to palmitic acid, had a powerful virucidal effect on bacteriophage Φ6 particles. Differences in hydrocarbon structure thus lead to alternate function in compound-virion-host cell receptor interactions.
Palmitic acid also demonstrated anti-HSV activity [165]. One additional study extracted lipophilic fractions from the brown (Phaeophyceae) macroalga Sargassum vulgare and found that the primary bioactive compound was a sulfoquinovosyldiacylglycerol (SQDG) [166]. The SQDG in question was esterified primarily by palmitic acid and displayed potent anti-HSV-1 activity. The authors speculate that this activity may have been in part due to the bioactivities of the abundant palmitic acid, which may compete with HSV particles to bind cellular receptors.
2.3.1.2. Galactan
Mazumder et al. isolated a sulfated galactan polysaccharide from the red (Rhodophyta) macroalga Gracilaria corticata that demonstrated anti-HSV-1/2 activity in vitro (Table 5) [156]. A virucidal assay demonstrated that the isolated galactan did not exert any direct inactivating effect on virions at IC50 concentration, suggesting that galactan inhibited HSV at an alternative point in the replication cycle. However, time of addition experiments indicated that galactan only showed AV activity during the initial adsorption stage of infection, suggesting that it interfered with the interaction of cell receptors and viral glycoproteins in a cell-dependent manner. These results are supported by another study that showed how a galactan isolated from the red (Rhodophyta) macroalgae Schizymenia binderi demonstrated anti-HSV-1/2 activity with extremely high selectivity indices (>1000) in vitro [157]. The authors postulated that the sulfated galactan interferes with interaction between HSV particles and cellular heparan sulfate residues that serve as the primary receptor for viral binding. This result further suggests that galactan interferes with the viral adsorption mechanism without directly inactivating the virus itself.
2.3.1.3. Calcium spirulan
Calcium spirulan, a sulfated polysaccharide from the cyanobacterium Spirulina (Arthrospira) platensis, has demonstrated AV activity against an array of viruses (Table 5) [158,159]. In a study conducted by Hayashi et al., calcium spirulan showed AV activity in vitro against HSV, CTMV, MeV, mumps (MuV), IAV, and HIV with SI values of 8587, 578, 371, 274, 574, and 1261 respectively [158]. No activity against polio or coxsackievirus was observed, indicating selectivity for enveloped viruses. Time-of-addition assays showed AV activity at multiple points during infection, but preincubation with cells showed the most potent AV effect. Calcium spirulan most actively prevented viral adsorption, indicating that calcium spirulan likely interacts with the cell to prevent viral attachment. The broad spectrum of activity, reported longer half-life in murine blood compared to other sulfated polysaccharides [167], as well as the fact that these assays were performed across a variety of cell lines highlight calcium spirulan as a promising medicinal agent.
2.3.1.4. Allophycocyanin
Shih et al. found that allophycocyanin, a 104 kDa pigment protein extracted from Arthrospira platensis effectively prevents enterovirus 71 (EV71) infection in vitro using Vero and human rhabdomyosarcoma cells [160]. When added to cell cultures prior to viral inoculation, allophycocyanin reduced infection with an SI of ~27 as observed in a plaque reduction assay. This effect was reduced when allophycocyanin was added post-infection. This result suggests that allophycocyanin prevents viral adhesion to target cells; however, more research is required to elucidate whether allophycocyanin interacts with host cell receptors, viral glycoproteins, or virus-cell complexes.
2.3.1.5. Arthrospira extracts
Rechter et al. assayed the antiviral activity of semi-refined Arthrospira platensis extracts in vitro [161]. These extracts consisted of varying amounts of polysaccharide and protein, potentially including previously-discussed compounds such as calcium spirulan and allophycocyanin. The extracts showed anti-HSV and CTMV activity that was most pronounced when preincubated with the cells (Table 5). Time-of-addition assays indicate that the compounds were active during the adsorption and penetration stages of viral infection. Virucidal assays showed that preincubation of compounds with virions significantly decreased infection when compared to preincubation of compounds with cells. This suggests that the compounds in the extracts bound cell surface receptors to inhibit viral adhesion and entry.
2.3.1.6. Nostoflan
Kanekiyo et al. assayed AV activity of an acidic polysaccharide, nostoflan, isolated from the cyanobacterium Nostoc flagelliforme against HSV-1 in vitro (Table 5) [162]. Nostoflan was found to interfere with the initial binding stage of infection, but not with independent virions or the subsequent penetration of virions into the cell. Notably, no protective effect was observed when target cells were preincubated with Nostoflan, indicating that nostoflan does not bind to cells or virions independently. This presents the possibility that nostoflan may only bind a virion-surface receptor complex. Furthermore, nostoflan's adhesion mechanism may depend on conformational changes in either or both viral surface glycoprotein receptors and CD4 receptors.
2.3.1.7. Chlorella polysaccharide extract
Santoyo et al. examined the in vitro anti-HSV-1 activities of acetone, ethanol, and water extracts of the green microalga Chlorella vulgaris (Table 5) [163]. Water and ethanol extracts were able to inhibit 70% of infection at concentrations of 75 μg/mL when used to pretreat cells. After promising initial testing of crude extracts, further concentrated polysaccharide fractions inhibited HSV-1 infection to an even greater degree. Pretreatment of cells with semi-refined polysaccharide fractions inhibited 90% of HSV-1 infection. It is possible that some AV activity can be attributed to the presence of phytol, but the majority of activity is likely due to polysaccharides. Due to their high SI value (49.04), polysaccharides from Chlorella are potentially valuable anti-adhesive AV drugs.
2.3.2. Entry
Viruses enter target cells through one of two mechanisms. First, a virus may enter the cell through endocytosis. This mechanism is typical of enveloped and non-enveloped viruses. In this mechanism, the virion-cell receptor complex interacts with the cell surface molecule clathrin and is invaginated by the cell membrane [28,29]. Secondly, a virion (enveloped) adhered to the cell membrane will fuse its own surface envelope with that of the cell membrane, releasing its contents inside of the cell [27]. Certain algal-derived compounds inhibit this step by interfering with vital protein machinery (Table 6 ). In considering anti-entry mechanisms, one should bear in mind how it is difficult to experimentally determine between anti-entry and anti-adhesion modes of drug action. Time-course assays are typically not sensitive enough to distinguish between the two and more advanced experiments are typically required.
Table 6.
Algae-derived compounds prevent viral entry into target cells.
| Compound | Species classification | Species | Virus | Activity | Model | Reference |
|---|---|---|---|---|---|---|
| SPMG | Phaeophyceae | Not reported | HIV-1 | Block interaction between CD4 and gp120 | Surface plasmon resonance with rgp120 and sCD4 | [168,169] |
| KW | Phaeophyceae | Kjellmaniella crassifolia | IAV | Inactivate EGFR pathway-mediated virion internalization | In vitro (MDCK) | [49] |
| PML | Chlorophyta | Monostroma latissimum | EV71 | Inactivate EGFR pathway-mediated virion internalization | In vitro (Vero) | [170] |
| MWS | Chlorophyta | Monostroma nitidum | EV71 | Decrease expression of EGFR protein | In vitro (Vero) | [171] |
2.3.2.1. Sulfated polymannuroguluronate (SPMG)
The low weight sulfated polysaccharide SPMG is extracted from brown (Phaeophyceae) macroalgae [172]. SPMG has displayed anti-HIV effects in vitro which can be attributed to a unique anti-entry mechanism. In order to infect T-cells, HIV virions depend on interactions between the CD4 cell receptor and viral glycoprotein 120 (gp120). The binding of CD4 and gp120 result in the exposure of a previously hidden domain of gp120, the V3 loop. When exposed, the V3 loop binds another chemokine receptor colocalized with CD4 and exposes viral glycoprotein 41 (gp41), which facilitates cell-virus fusion [173]. SPMG is able to interfere with this mechanism, as elucidated by one x-ray crystallography study (Table 6) [169]. SPMG, it seems, binds to both the V3 loop and the CD4 receptor to form a trimolecular complex. Additionally, SPMG may bind to gp120 residues other than the V3 loop in a cell-independent manner. SPMG is potentially able to operate through a few mechanisms, binding to either gp120, CD4, or both to inhibit gp120 attachment and viral entry. A further study characterized SPMG's interaction with lymphocytes and determined that SPMG binds positively charged epitopes on the CD4 cell receptor in a polyanion mediated, highly specific, multivalent manner [168]. Thus, SPMG may represent a novel, promising way to treat HIV infection by interfering with the viral entry mechanism. SPMG entered into clinical trials in China, passing Phase I and entering into Phase II [172].
2.3.2.2. Epidermal growth factor receptor (EGFR) pathway inhibitors
The EGFR pathway is activated in many human tissues and is crucial to epithelial and epidermis cell differentiation and growth [174]. Activation of this pathway also facilitates the entry of some types of virus into cells. Eierhoff et al. showed that EGFR promoted the entry of IAV into the cell [175]. Some algae-derived compounds inhibit viral infection via blocking the EGFR pathway. Interfering with the EGFR pathway is also potentially dangerous due to its importance in normal cell function. This must be considered when developing antiviral drugs that interfere with the EGFR pathway.
In a study conducted by Wang et al., a fucoidan from the brown (Phaeophyceae) macroalgae Kjellmaniella crassifolia inactivated the EGFR pathway in vitro to prevent the internalization of IAV (Table 6) [49]. In another study, the sulfated rhamnan polysaccharide PML from the green (Chlorophyta) macroalgae Monostroma latissimum reduced EV71 infection in vitro and increase mouse survival in vivo. While PML did have some direct inactivating effect on a viral capsid protein, it also targeted the EGFR pathway to prevent internalization of EV71 particles [170,171]. It is important to note that some compounds from algae activate the EGFR pathway as well [176]. While some viruses may use the EGFR to facilitate entry into target cells, it is important to remember that EGFR signaling is required for proper functioning of epidermal tissue, and so many unforeseen consequences may result from the administration of even (seemingly) benign naturally-derived compounds that interfere with this pathway.
2.3.3. Uncoating
Instructions from the viral genome are essential for directing further steps of the replication process. While some transcription and translation steps may take place inside the viral capsid, oftentimes the viral genetic material must be released into the cytosol or cell nucleus. Many RNA viruses replicate in the cytosol and thus require the release of their genetic material outside the nucleus, whereas some DNA viruses require release inside the nucleus [27]. Some algae-derived compounds block viral infection at the uncoating stage (Table 7 ).
Table 7.
Algae-derived compounds prevent viral uncoating.
Fucoidan exerts anti-DENV activity in vitro mediated by interference with the viral internalization/uncoating step (Table 7) [177]. The DENV mechanism of entry and uncoating depends on the EGP glycoprotein, which mediates attachment, endocytosis, and fusion between the viral membrane and endosome to release genetic material into the cytosol. Fucoidan interfered with the action of this glycoprotein, potently inhibiting infection.
Talarico et al. showed that carrageenan prevents DENV uncoating using an elegant set of experiments in vitro (Table 7) [178]. DENV virions that penetrated into the cell via endocytosis were treated with λ-carrageenan which prevented uncoating within the cell. Injecting DENV RNA facilitated infection as usual, indicating that no replication or transcription step was affected by carrageenan. While carrageenan had previously demonstrated anti-DENV activity [179], this was the first demonstration of a specific anti-uncoating mechanism. Due to their wide range of molecular weights and corresponding differences in diffusion properties, it is possible that some LMW carrageenans are able to enter into cells and prevent uncoating of other types of virus as well. Carrageenan can enter into cells to inhibit later steps of the viral replication cycle [180], but more research and specific experimental designs akin to those of Talarico et al. are needed to reveal any anti-uncoating activities.
Indeed, many algae-derived compounds display AV activity post-adhesion and entry but operate by undefined mechanisms. Such compounds include aplysiatoxins from cyanobacteria [181], which inhibit CHIKV activity post-infection in vitro as determined by time-of-addition experiments. However, uncertainty remains concerning the specific replication step that aplysiatoxins inhibit CHIKV. More emphasis will need to be placed on AV research from algae to specifically elucidate the mechanisms of potentially life-saving drugs. In the next sections we examine algae-derived compounds that show AV activity at viral life cycle steps that take place inside the cell and involve replication of viral genetic material or proteins.
2.3.4. Replication
Replication refers to the process by which viral genetic material is ultimately used to produce copies of itself and the protein components essential to viral structure and function. Viral information is encoded in either RNA or DNA which varies in its orientation (positive or negative sense), segmentation (number of overall pieces of RNA or DNA), and number of strands (single or double). DNA viruses must undergo transcription and translation to produce their proteins, akin to living organisms. Positive-sense single stranded RNA virus genomes are essentially mRNA and can be directly translated into proteins once the genome enters the cytoplasm after infection. However, replication of positive-sense single stranded RNA genomes requires a virus-encoded RNA dependent RNA polymerase as host cells do not have this type of enzyme. In contrast, negative-sense RNA viruses must first synthesize the complementary strand of mRNA before viral proteins can be translated. Retroviruses, including HIV, utilize a special enzyme to synthesize double-stranded DNA from an RNA template prior to integration into the host genome [28].
The replication of genetic material and proteins is highly variable between virus types. This complicates antiviral drug development but provides many possible avenues for intervention. Many algae-derived compounds are able to interfere with specific steps of viral replication, with a range of specificity (Table 8 ). In this section, we outline the AV bioactivities of algae-derived compounds against the steps of viral genome replication.
Table 8.
Algae-derived compounds inhibit the viral replication cycle.
| Compound | Species classification | Species | Virus | Activity | Model | Reference |
|---|---|---|---|---|---|---|
| Dolastatin 3 | Cyanobacteria | Lyngbya majuscula | HIV-1 | Anti-HIV-1 integrase | N/A (enzyme assay) | [182] |
| Macroalgae extracts | Phaeophyceae Phaeophyceae Chlorophyta (1 species) Phaeophyceae (8 species) Rhodophyta (6 species) |
Ecklonia cava, Ishige okamurae, Sargassum confusum Sargassum hemiphyllum, Sargassum ringgoldianum Various |
HIV-1 HIV-1 HIV-1 |
Anti-HIV-1 integrase and RT Anti-HIV-1 integrase Anti-HIV-1 RT |
N/A (enzyme assay) N/A (enzyme assay) N/A (enzyme assay) |
[183] [183] [183] |
| 8,8′-Bieckol | Phaeophyceae | Ecklonia cava | HIV-1 | Anti-HIV-1 RT, protease | N/A (enzyme assay) | [184] |
| DEHC | Phaeophyceae | Ishige okamurae | HIV-1 | Anti-HIV-1 integrase and RT | N/A (enzyme assay) | [185] |
| Diterpenes | Phaeophyceae | Dictyota menstrualis | HIV-1 | Anti-HIV-1 DNA synthesis, likely mediated by anti-RT activity | In vitro (PM-1 from Hut78) | [186] |
| Fucan, fucoidan, alginic acid | Phaeophyceae |
Dictyota mertensii Lobophora variegata Spatoglossum schroederi Fucus vesiculosus |
HIV-1 | Anti-HIV-1 RT | N/A (enzyme assay) | [187] |
| Alginate, ‘911’ | Not reported | Not reported | HIV-1 HBV |
Anti-HIV-1 RT Anti-HBV DNA polymerase |
In vitro (MT4) HepG2215 |
[188] Surveyed [173,183] |
| λ-Carrageenan | Rhodophyta | Schizymenia pacifica | HIV | Anti-HIV RT | In vitro (MT-4), enzyme assays | [191] |
| SQDG | Rhodophyta | Gigartina tenella | HIV-1 | Anti-HIV RT | N/A (enzyme assay) | [192] |
| Peyssonol A | Rhodophyta | Peyssonnelia sp. | HIV-1 | Anti-HIV-1/2 RT | N/A (enzyme assay) | [193] |
| Cyanobacterial extracts | Cyanobacteria | Nostoc, Phormidium, Oscillatoria, Chroococcus, Schizothrix, Aphanocapsa, Synechococcus, Aphanothece, Xenococcus | AMV HIV-1 |
Anti-RT | N/A (enzyme assay) | [194] |
| κ-Carrageenan oligosaccharide | Rhodophyta | Not reported | IAV (H1N1) | Inhibit RNA expression | In vitro (MDCK) | [94] |
| Phlorotannins | Phaeophyceae | Ecklonia cava | PEDV | Inhibit RNA/protein expression | In vitro (Vero) | [119] |
| dsRNA | Chlorophyta | Chlamydomonas reinhardtii | YHV | RNAi | In vivo (Penaeus vannamei) | [195] |
2.3.4.1. DNA synthesis
DNA replication is an important part of some viral life cycles. DNA viruses replicate using either cellular or viral polymerases. Polyoma, parvo-, circo-, and anelloviruses require cellular DNA polymerase to replicate while ADVs, poxviruses, and HSVs use their own DNA polymerase. Many viruses integrate their genetic material into host cell chromosomes as part of their life cycle [28]. This viral DNA can immediately produce proteins important for the completion of the viral life cycle or lay dormant, potentially for years, before actively producing viral machinery. Some viruses, most famously retroviruses like HIV, rely on a reverse transcriptase (RT) enzyme that uses an RNA template to synthesize a double-stranded DNA ‘provirus’ that can be incorporated into the host genome. Inhibiting this enzyme is a potentially effective way to prevent infection and many algae-derived compounds have demonstrated the ability to do so. Many algae-derived compounds derive AV activity from inhibition of DNA integration or synthesis processes (Table 8).
Preventing integration of viral DNA via inhibition of integrase enzymes may be one effective way to prevent chronic infection. The cyclic peptide dolastatin 3 was isolated from a marine cyanobacterium, Lyngbya majuscula, and shown to demonstrate anti-HIV integrase activity in vitro, though with a relatively poor IC50 value of 5 mM (Table 8) [182]. Cyanobacterial peptides may be promising AV agents, but little research has been performed into this area. Dolastatins from other sources have shown cytotoxicity [196]; further research is needed to clarify the potential benefits and drawbacks from dolastatin 3 and similar unique compounds derived from cyanobacteria.
In a 2002 study, Ahn et al. screened the methanolic extracts of 47 Korean macroalgae species for anti-HIV-RT and anti-HIV-integrase activity [183]. These enzymes are not indigenous to human host cells and thus are appealing potential targets for AV drugs. 15 of the 47 species across Chlorophyta, Rhodophyta, and phaeophyte groups inhibited HIV-RT (Table 8). Among all the macroalgae tested, only five of the phaeophytes inhibited HIV-integrase. Three species, Ecklonia cava, Ishige okamurae, and Sargassum confusum inhibited HIV-RT and integrase, highlighting these species as particularly worthy of investigation going forward. This study did not determine which specific compounds were bioactive components of the methanolic extracts but eliminated potential contribution from polysaccharides by further fractionation with ethylacetate. Follow-up research from the same team found that phlorotannins extracted from E. cava exerted strong anti-HIV-RT activity in vitro [184]. Four compounds were isolated: eckol, 8,8′-bieckol, 8,4″′-dieckol, and phlorofucofuroeckol A. 8,8′-bieckol showed particularly strong anti-HIV-RT activity and displayed uncompetitive inhibition with a Ki of 0.23 μM. Additional investigation also showed that the phlorotannin diphlorethohydroxycarmalol (DEHC) from I. okamurae showed anti-HIV-RT and integrase activity with IC50 values of 9.1 μM and 25.2 μM respectively [185]. Taken together, these studies seem to indicate that that the bioactive compounds from the initial methanolic extract study were likely phlorotannins. These studies demonstrated that a wide range of algae species across different phyla inhibited HIV-replication and serve as a promising foundation for future AV research.
In a different study, diterpenes from the brown (Phaeophyceae) macroalga Dictyota menstrualis showed anti-HIV activity in vitro using a mechanism involving the inhibition of DNA integration or synthesis (Table 8) [186]. In this study, diterpenes prevented double-stranded DNA synthesis. Inhibition of HIV production as a whole occurred on the same order of magnitude as observed in a specific anti-RT assay, suggesting anti-RT activity as a primary mechanism for AV activity. Diterpenes have been shown elsewhere in vitro and in molecular docking analyses to inhibit HIV-RT [197].
Due to the global prevalence of HIV, potential anti-RT mediated treatments have been researched extensively, including many algae-derived compounds. Compounds from algae that have shown anti-HIV-RT activity include fucan [187], alginate [188,189], λ-carrageenan [191], and sulfoquinovosyldiacylglycerol [192]. The latter also inhibited eukaryotic polymerases and displayed some cytotoxicity. This demonstrates an important point regarding anti-HIV research. RT enzymes are a type of DNA polymerase, and inhibitors of RT may inhibit cellular DNA polymerase as well. It is vital to carefully develop anti-RT drugs that do not interfere with essential cellular functions.
One interesting HIV-RT inhibitor that illustrates this point is peyssonol A, a sesquiterpene hydroquinone derived from the red macroalga Peyssonelia sp. [198]. Loya et al. showed that peyssonols A and B inhibit anti-HIV-RT activity and while peyssonol B showed moderate inhibitory activity against eukaryotic DNA polymerases, peyssonol A showed very little activity against DNA polymerases even at extremely high concentrations (Table 8) [193]. This result is promising, as it suggests a high degree of selectivity for only HIV DNA synthesis and would likely not result in adverse effects against human cells. In recent follow-up research, analogues of peyssonol A were synthesized [199]. These analogues all exhibited anti-RT activity and varying degrees of cytotoxicity in vitro. The authors suggest several compounds that deserve special consideration going forward.
Exploring cyanobacterial compounds is another especially exciting area of research that may reveal many RT inhibitors. Lau et al. surveyed over 900 cyanobacterial extracts for their ability to inhibit AMV and HIV RT enzymes (Table 8) [194]. 18 aqueous extracts showed promising activity and further refining generated fractions that had RT specificity and did not interfere with cellular genetic material.
Hepatitis B virus (HBV), a double-stranded DNA (dsDNA) virus that causes chronic liver disease, uses a viral DNA polymerase to replicate its genome [30,200]. One polysaccharide derived from a brown seaweed, referred to as 911, inhibited HBV replication in vitro with an IC50 value of 17.3 mg/mL and SI of 3.37 (Table 8) [201]. The researchers concluded that the activity of 911 was due to inhibition of the HBV polymerase enzyme. While this is a unique bioactivity that may have significant therapeutic value, the low SI value of this compound is concerning, and more research is needed to either identify similar, more potent compounds or chemically refine this compound for improved antiviral properties.
2.3.4.2. RNA synthesis
RNA synthesis is essential in the life cycle of all viruses as a common step in protein synthesis and for the replication of RNA virus genetic material. In order to replicate, RNA viruses must synthesize copies of their genetic material, often using an RNA-dependent RNA polymerase (RDRP) enzyme. The RDRP enzyme uses an RNA strand to synthesize mRNA or RNA to serve as the viral genome in future generations. Multiple kinds of RDRP exist depending on the virus and the orientation (positive or negative sense) of its genome. Some compounds derived from algae inhibit RNA synthesis, an important step in some way during the life cycle of all viruses (Table 8).
Wang et al. showed that a LMW carrageenan oligosaccharide entered into cells and inhibited IAV mRNA expression in vitro (Table 8) [94]. Time of addition experiments indicated that carrageenan was active after viral internalization and before viral release. Carrageenan did not inhibit entry of virions into the cell, but displayed similar properties to the commercial pharmaceutical Ribavirin, a known mRNA synthesis inhibitor of IAV. Carrageenan thus seems to be an mRNA synthesis inhibitor in its LMW form. This is promising, as we have already illustrated that carrageenan inhibits multiple viral replication steps. The multiple modes of operation hold promise for effective AV activity with reduced likelihood of developing viral resistance.
Many polyphenolic compounds inhibit RNA synthesis. In a study conducted by Ryu et al., two phlorotannins derived from the brown (Phaeophyceae) macroalga E. cava inhibited PEDV RNA and protein synthesis in vitro (Table 8) [119]. One recent study performed molecular docking on plant-derived polyphenols as inhibitors of the SARS-CoV-2 RDRP enzyme [202]. Four compounds showed exceptional promise and were highlighted. While none of the compounds in this study were derived from algae, similar compounds are found throughout the different groups of algae. More research into algae-derived compounds would certainly yield inhibitors of viral RNA synthesis.
2.3.4.3. RNA interference
RNA interference (RNAi) requires the introduction of a specific dsRNA molecule to target, bind and tag viral mRNA for degradation so that viral proteins cannot be produced [203]. Algae have shown promise as biofactories for specific dsRNAs and have already been used to prevent YHV outbreak in aquaculture operations (Table 8) [183]. RNAi has been used effectively against coronaviruses and has been proposed as a potential therapy against SARS-CoV-2 [203].
2.3.5. Processing
After viral transcription has occurred, mRNAs are translated into proteins. Viruses lack ribosomes, and so rely on host cell ribosomal machinery to complete this step. Due to the high likelihood of interfering with cellular protein synthesis, using ribosome inhibitors as AV compounds carries potentially catastrophic consequences. Thus, safe AV compounds inhibiting viral protein synthesis interfere with specific viral enzymes that are not involved in normal cell functions.
After translation, viral proteins that are translated as a polyprotein or a series of connected viral proteins must be cleaved into individual functional proteins by cellular or virus encoded proteases. The viral proteins can then mediate a range of functions including packaging of viral proteins and genetic material into assembled units to be released from the cell as mature virions. Many processing steps occur independently of cellular machinery and can thus be targeted by AV compounds from algae (Table 9 ) [27].
Table 9.
Algae-derived compounds inhibit viral processing after replication.
| Compound | Species classification | Species | Virus | Activity | Model | Reference |
|---|---|---|---|---|---|---|
| 8,8′-Bieckol 8,4″′-Dieckol | Phaeophyceae | Ecklonia cava | HIV-1 | Anti-HIV-1 protease | N/A (enzyme assay) | [184] |
| Dieckol | Phaeophyceae | Ecklonia cava | SARS-CoV | Anti-3CL protease | N/A (enzyme assay) | [204] |
| Ichthyopeptins A/B | Cyanobacteria | Microcystis ichthyoblabe | IAV | Protease inhibition (proposed) | In vitro (MDCK) | [205] |
| β-Sitosterol | Brassicales Rhodophyta |
Isatis indigotica (land plant) Halosaccion ramentaceum, Rhodomela confervoides, Porphyra sp., Polyides rotunda |
SARS-CoV | Anti-3CL protease | In vitro (Vero) | [206] [207] |
| Caulerpin | Chlorophyta Rhodophyta Phaeophyceae |
Caulerpa racemosa, Chondria armata, Sargassum platycarpum | SARS-CoV-2 | Anti-SARS-CoV-2-3CL protease | Molecular docking analysis | [126] |
HIV protease activity is crucial for the development of mature structural and enzymatic proteins. The HIV protease is essential to the viral life cycle but plays no part in human cellular function and makes an excellent antiviral target. In a study conducted by Ahn et al., phlorotannins isolated from the brown macroalga E. cava showed moderate inhibitory activity towards HIV protease in vitro (Table 9) [184]. These phlorotannins showed more potent anti-RT activity than anti-protease activity, suggesting the potential for the development of a multi-mechanism drug.
Recently, the steroid β-sitosterol extracted the land plant Isatis indigotica from was shown to inhibit the SARS-CoV-1 3C-like protease in vitro with an IC50 of 1210 μM (Table 9) [206]. Β-sitosterol is also found in red (Rhodophyta) macroalgae [207], and should be researched along with similar compounds as a potential anti-SARS-CoV-2 drug. The SARS-CoV-1 main (3CL) protease is essential for proteolytic processing [208] and has received attention as a potential target for AV therapy. Abdelrheem et al. screened 10 natural compounds against the SARS-CoV-2-3CL protease and determined that the alkaloid caulerpin derived from green (Chlorophyta) algae in the genus Caulerpa showed the most potent inhibitory activity [126]. Caulerpin satisfies Lipinski's Rule of Five and ADMET properties. These are guidelines for drug development relating to pharmacokinetic properties including molecular size, proclivity to engage in bonding, and bioavailability [209,210]. Compounds that satisfy these guidelines, such as caulerpin, are generally perceived as promising candidates for further development. Further investigation determined that caulerpin could be synthetically modified to increase protease binding affinity [211].
One other study found that eight phlorotannins isolated from the brown macroalga E. cava inhibited the SARS-CoV-1 3CL protease in vitro [204]. In this study, dieckol showed especially potent inhibitory activity (IC50 = 3.3 μM, SI >2.9). This experimental result was further examined by molecular docking analysis, which demonstrated competitive-type inhibition with a high association rate and strong hydrogen bonding. The use of phlorotannins as protease inhibitors may be substantiated by an extensive investigation carried out by Cannell et al. which surveyed the protease inhibitory activity of extracts derived from 500 species of eukaryotic algae and 80 species of cyanobacteria [212]. This investigation determined that 39 of the species in question showed protease activity against at least one of seven enzymes surveyed (Table 9). Significantly, almost all of the protease inhibitory activity came from methanolic extracts where polyphenolic/phlorotannin compounds are concentrated. None of the protease enzymes in this study were specifically derived from viruses, however, the broad range of anti-protease activities observed across different species of algae suggests that many algae may produce anti-protease compounds that effectively inhibit viral replication.
Zainuddin et al. isolated a fraction from the cyanobacterium Microcystis ichthyoblabe containing two ichthyopeptins, cyclic depsipeptides that inhibited IAV with an IC50 of 12.5 μg/mL (Table 9) [205]. The authors speculate that their antiviral activity is due to protease inhibitory activity. Cyanobacteria have been shown to produce many protease-inhibitory compounds [213,214], further research may show useful applications in combatting viral disease.
2.3.6. Budding/release
Virions may be released from the cell by budding through the membrane or by cell lysis. Enveloped viruses are primarily released by budding [215]. These viruses may acquire their envelopes from the nuclear membrane, the Golgi membrane, endoplasmic reticulum, or cell membranes. Budding is mediated by interactions between viral surface molecules and proteins at the cell surface. Algae-derived compounds have shown the ability to prevent viruses from being released into the environment and continuing the infection process by interfering with this machinery (Table 10 ).
Table 10.
Algae-derived compounds inhibit viral release.
| Compound | Species classification | Species | Virus | Activity | Model | Reference |
|---|---|---|---|---|---|---|
| KW Fucoidan | Phaeophyceae | Kjellmaniella crassifolia | IAV | Anti-neuraminidase |
In vitro (MDCK) In vivo (murine) |
[49] |
| Phlorotannins | Phaeophyceae | Ecklonia cava | IAV (H1N1, H3N2, H9N2) | Anti-neuraminidase | In vitro (MDCK) | [216] |
| Naviculan | Bacillariophyta | Navicula directa | HIV-1 | Anti-syncytia formation | In vitro (CD4-expressing HeLa) | [217] |
| Palmitic acid | Phaeophyceae | Sargassum fusiforme | HIV-1 | Anti-syncytia formation | In vitro (MΦs, PBL) | [155] |
| Cyanovirin-N | Cyanobacteria |
Nostoc ellipsosporum Unknown |
HIV-1 HIV-1 |
Anti-syncytia formation Anti-syncytia formation |
In vitro (CD4+ HeLa) In vitro (TZM-bl) |
[91] [218] |
| Microvirin | Cyanobacteria | Microcystis aeruginosa | HIV-1 | Anti-syncytia formation | In vitro (CD4+ T-cells) | [219] |
| Griffithsin | Rhodophyta | Griffithsia sp. | HIV-1 | Anti-syncytia formation | In vitro (CEM) | [220] |
| Ulvan | Chlorophyta | Ulva clathrata | NDV | Block F-protein cleavage, anti-syncytium formation | In vitro (Vero) | [134] |
| Allophycocyanin | Cyanobacteria | Spirulina (Arthrospira) platensis | EV71 | Anti-apoptosis | In vitro (Vero, human rhabdomyosarcoma) | [160] |
| Durvillaea antarctica polysaccharide | Phaeophyceae | Durvillaea antarctica | EV71 | Anti-apoptosis | In vitro (Vero) | [221] |
Neuraminidase is an influenza surface enzyme that facilitates the release of virions from infected cells after replication [222]. Wang et al. showed that fucoidan from the brown macroalga (Phaeophyceae) Kjellmaniella crassifolia inhibits neuraminidase in vitro (Table 10). Furthermore, fucoidan limited IAV infection, increased survival, and decreased viral titers in a murine model in vivo [49]. Phlorotannins isolated from Ecklonia cava also demonstrated neuraminidase-inhibitory activity [216]. Five phlorotannins showed inhibitory activity against neuraminidase, with three of the five compounds showing potent anti-neuraminidase activity (IC50 < 50 μM) against enzymes derived from three different influenza strains (H1N1, H3N2, H9N2) (Table 10). Inhibition of this enzyme was noncompetitive and acted synergistically with the commercial pharmaceutical neuraminidase inhibitor oseltamivir. Because neuraminidase is also involved in the entry of influenza virions into the cell [223], anti-neuraminidase drugs may be an effective way to manage and limit the spread of influenza infection.
HIV assembly and budding depends on the modification of the Gag protein with myristic acid [224], a saturated fatty acid found in some red algae [225]. Lindwasser et al. found that the addition of mono- or polyunsaturated myristic acid derivatives to infected cells interfered with HIV assembly and release (Table 10) [224]. Synthetically modified myristic acids extracted from algae could represent a promising way to reduce the spread of HIV in infected patients.
2.3.6.1. Cell-cell transmission
As an alternative to being released from cells, some viruses induce the cells they have infected to fuse with other neighboring cells and form syncytia, multinucleate aggregations of cells with one continuous cytoplasm that facilitate the spread of a virus with greater efficiency through a given tissue [25,28]. HIV is an example of a virus that causes syncytia to form, directing the fusion of CD4-expressing cells [226]. HSV and Newcastle Disease Virus (NDV) are also spread between cells by syncytia formation. Algae-derived compounds can prevent syncytia formation (Table 10).
2.3.6.2. Naviculan
Lee et al. extracted the sulfated polysaccharide naviculan from the diatom Navicula directa and determined that it prevented syncytium formation using an HIV fusion assay in vitro (Table 10) [217]. Naviculan also interferes with infection at the adsorption/penetration stage as determined by time-of-addition experiment. Thus, naviculan is a potential multi-mechanism anti-HIV agent that could be useful in managing chronic infection. In this study, naviculan also showed antiviral activity against HSV-1, HSV-2, and IAV with SI values of 270, 310, and 32 respectively. Thus, naviculan has potential to be developed into broad spectrum antiviral drug.
2.3.6.3. Palmitic acid
In another study, palmitic acid from the brown alga (Phaeophyceae) Sargassum fusiforme inhibited HIV infection in vitro by preventing the interaction of the viral glycoprotein gp120 and cellular CD4 receptor (Table 10) [155]. Palmitic acid prevented the interaction of gp120 and CD4 by competing with gp120 for CD4 binding, thus reducing syncytia formation by up to 70%. In Section 2.3.1.1, we described how palmitic acid exerts a powerful anti-adhesive affect to inhibit HIV replication. Due to its multi-mechanism anti-HIV activity, palmitic acid may prove an effective treatment for those suffering with chronic infection.
2.3.6.4. Lectins
At least three different lectins bind HIV glycoproteins to prevent syncytia formation. In a study conducted by O'Keefe et al., the lectin cyanovirin-N (CVN) from the cyanobacterium Nostoc ellipsosporum showed high affinity for gp120 and prevented syncytia formation in vitro (Table 10) [91]. Another study supported this result, demonstrating that CVN prevented syncytia formation. However, concentrations required to prevent syncytia formation were ten-fold higher than those required to inhibit cell-free virus [218]. CVN irreversibly bound to gp120, exerting direct virustatic activity, and is thus a potentially useful multi-mechanism anti-HIV agent. In this study and others, CVN did not show cytotoxicity but did induce low levels of T-cell proliferation and cytokine release, suggesting that overuse of CVN might have an inflammatory effect. In a different study, the lectin microvirin (MVN) isolated from the cyanobacterium Microcystis aeruginosa prevented syncytia formation between HIV-1-infected T-cells and virus-free CD4+ T-cells in vitro (Table 10) [219]. While long-term infection did lead to the development of a viral mutant, the mutant was still vulnerable to inhibition by other lectins. Significantly, the cytotoxicity of MVN was >50-fold lower than CVN. The lectin griffithsin (GRFT) derived from the red macroalga Griffithsia also prevents the transmission of HIV via syncytia formation. In a study conducted by Mori et al., GRFT bound gp120, gp41, and gp160, most critically preventing interaction between gp120 and the cellular CD4 receptor [220]. One additional study determined that an aqueous extract of the cyanobacterium Arthrospira platensis had anti-HIV activity and prevented syncytium formation. While this investigation did not isolate a specific bioactive compound, the authors suggest the bioactivity may be due to the presence of a lectin [227].
2.3.6.5. Ulvan
Aguilar-Briseño et al. examined the effect of ulvan from Ulva clathrata on NDV infection in vitro [134]. NDV relies on a fusion (F) protein to infect cells. The cleavage of F into F1 and F2 facilitates the aggregation of target cells into giant syncytia which increases the speed and efficiency of NDV infection. Ulvan prevented F protein cleavage, which prevented syncytia formation downstream. Ulvan did not have any direct virucidal or virustatic action (Table 10).
2.3.6.6. Allophycocyanin
Apoptosis is an endogenous mechanism involving programmed cell death commonly used by the body to slow the spread of viral infection. However, some viruses can take advantage of apoptosis, inducing this mechanism to hasten their own release into the environment [25]. Shih et al. determined that the pigment protein allophycocyanin extracted from the cyanobacterium Spirulina (Arthrospira) platensis prevents apoptosis induced by EV71 in vitro using Vero and human rhabdomyosarcoma cell lines [160]. Due to EV71 infecting and destroys cells of the nervous system that are not easily replenished, allophycocyanin might be an especially important therapeutic agent.
2.3.6.7. Durvillaea antarctica polysaccharide
Xu et al. found that polysaccharides extracted from the brown alga (Phaeophyceae) Durvillaea antarctica prevented EV71-induced apoptosis in vitro using Vero cells [221]. The administration of D. antarctica polysaccharides downregulated the p53 signaling pathway, which EV71 uses to induce apoptosis. Furthermore, D. antarctica polysaccharides upregulated STAT1 and mTOR which are important cell signaling proteins during EV71 infection. These polysaccharides were also able to decrease inflammatory cytokine production, which are involved in signaling to initiate apoptosis. Thus, Durvillaea antarctica polysaccharides may be an important therapeutic agent against EV71.
2.4. Algae produce immunomodulatory compounds
Immune response to viral infection is immensely complicated, involving the recruitment of molecular, cellular, and organismal defense mechanisms [228]. Immune response can be divided into cellular and humoral components. Humoral immune response to viral infection involves the secretion of antibodies that can directly inhibit viruses or summon cells of the immune system to phagocytose virions. Cellular immune response involves the directed killing of infected cells by natural killer or cytotoxic T-lymphocytes [25]. Generally speaking, complex signaling between infected cells and cells of the immune system initiate protective mechanisms to clear viral infection and protect cells from harmful pathologies [25,44]. A comprehensive discussion of immune response to viral infection is beyond the scope of this paper. However, many studies have shown that algae-derived compounds augment both cellular and humoral immune function against viral infection, either through direct immunostimulating effects or modulating inflammatory response (Fig. 6 ). Furthermore, early research has shown that consumption of algae modifies the profile of host gene expression with some potential benefits to immune response [229]. In Section 2.4, we provide an overview of some important immune-activating properties observed in algae-derived compounds (Table 11, Fig. 7 ).
Fig. 6.
Algae-derived compounds prevent viral (I) adhesion, (II) entry and uncoating, (III) replication, (IV) processing, and (V) budding.
Table 11.
Algae-derived compounds modulate immune response.
| Compound | Species classification | Species | Virus | Activity | Model | Reference |
|---|---|---|---|---|---|---|
| Fucoidan | Phaeophyceae |
Undaria pinnatifida Undaria pinnatifida Unknown Holothuria polii (sea cucumber) |
IAV (H1N1) IAV, AIV N/A N/A |
Decrease pathology without affecting viral titer Stimulate antibody production Hematopoietic mobilization Stimulate hematopoiesis |
In vivo (murine) In vivo (murine) In vivo (murine) In vivo (murine) |
[51] [52] [211] [212] |
| Ascophyllan HS | Phaeophyceae | Ascophyllum nodosum | N/A N/A N/A |
Stimulate IL-12 production Upregulate CD40, CD80, CD86, MHCI, MHCII, IL-6, IL-12, TNF-α Promote Th1, Tc1 cell generation, enhance response Induce DC cell maturation Stimulate TNF- α, G-CSF, NO production, upregulate iNOS |
In vivo (murine) In vivo (murine) In vitro (RAW264.7) |
[233] [234] [235] |
| Lipoprotein | Cyanobacteria | Arthrospira platensis | IAV (H1N1) | Decreased histopathology Upregulate IL-1β, TNF-α, (IL)–8, MCP-1, MIP-1, MIP-1, IP-10, COX-2 |
In vivo (murine) In vitro (THP-1) |
[236] [237,238] |
| Paramylon | Euglenozoa | Euglena gracilis | IAV (H1N1) | Increase subject survival rate Lower viral titer Upregulate IL-1 β, IL-6, IL-12 (p70), IFN-γ, and IL-10 |
In vivo (murine) | [239] |
| AEX | Chlorophyta | Coccomyxa gloeobotrydiformis | IBDV | Increase production of NO Upregulate IL-1β, IL-6, TNF-α, and iNOS production Activate PBMCs Promote extracellular antigen presentation Promote splenic lymphocyte and DT40 cell proliferation |
In vivo (chicken) | [99] |
Fig. 7.
Algae-derived compounds stimulate immune response against viral infection. Algae-derived compounds stimulate (I) IFN release, (II) inflammatory response, (III) cytokine/chemokine production, (IV) antibody production, (V) recruitment of immune system actors including neutrophils, natural killer (NK), and T-cells, and (VI) dendritic cell maturation.
2.4.1. Fucoidan
The polysaccharide fucoidan from brown macroalgae has demonstrated immunomodulatory effects in the prevention of viral infection. In a study conducted by Richards et al., an orally administered fucoidan from the brown (Phaeophyceae) macroalga Undaria pinnatifida decreased histopathology after influenza infection in a murine model (Table 11) [51]. Significantly, fucoidan did not decrease the viral titer, indicating that its beneficial effect could primarily be attributed to anti-inflammatory activity or boosting immune response. While we have discussed reports of direct antiviral effects from fucoidans, it is possible that their beneficial effects in vivo do not depend entirely on interaction with the viral replication cycle but rather on protective immunomodulation.
One other study examined the in vivo immunomodulatory and antiviral effects of a LMW fucoidan from U. pinnatifida [52]. Using a murine model, fucoidan decreased viral replication and increased antibody production against two strains of IAV, as well as increasing antibody and mucosal IgA production against a strain of avian IAV (Table 11). This result provides strong evidence that fucoidan serves a role in stimulating the humoral immune response against viral infection.
Fucoidan has shown immunomodulatory effects in other experimental settings. Fucoidan has been investigated as a potential vaccine adjuvant [240,241]. Investigations have shown that fucoidan is able to mobilize hematopoietic progenitor cells [231]. Li et al. showed that fucoidan derived from a sea cucumber increased hematopoiesis in immunosuppressed mice, increasing white blood cell and neutrophil counts (Table 11) [232]. Fucoidan may stimulate the migration of white blood cells and neutrophils from the bone marrow to the bloodstream. These results show that fucoidan stimulates the immune response and plays an important role in protecting against viral infection.
2.4.2. Ascophyllan HS
Ascophyllan HS is a polysaccharide derived from the brown (Phaeophyceae) macroalga Ascophyllum nodosum. It is similar to fucoidan, but contains a higher amount of xylose and uronic acid [233]. In a study conducted by Okimura et al., ascophyllan HS promoted murine survival in a bacterial respiratory infection model [233]. Mice administered ascophyllan HS showed reduced histopathology and increased levels of IL-12, indicating that the improved pathology can be attributed at least in part to stimulation of the immune system (Table 11). Additional research showed that ascophyllan HS induced the activation of dendritic cells in vitro and in vivo by promoting their production of pro-inflammatory cytokines. Ascophyllan increased the number of cells producing IFN, which increases the number of Th1 and Tc1 cells [234]. In this study, ascophyllan facilitated the maturation of dendritic cells, a crucial part of the adaptive immune response during infection that allows the host organism to develop immunity to a specific pathogen.
In another study, ascophyllan activated macrophages in vitro by upregulating levels of iNOS expression, inducing the production of nitric oxide (NO), and increasing production of TNF-α and G-CSF. In this study, ascophyllan showed greater activation effects than fucoidan with less cytotoxicity (Table 11) [235]. More research is needed to clarify the effect of ascophyllan on modulating immune system response against viral infection. Due to the profound immunostimulatory effects against other invading pathogens observed in these studies, ascophyllan may also play a key role in protecting against viral infection.
2.4.3. Lipoprotein
Pugh et al. examined the immunostimulatory properties of Immulina®, a commercial extract primarily composed of lipoproteins derived from the cyanobacterium Arthrospira platensis, and its effect on IAV infection in a murine model (Table 11) [236]. Mice that were fed a diet supplemented with the lipoproteins exhibited reduced weight loss, fewer signs of disease, and improved histopathology scores. Such lipoproteins are immunostimulatory agents and Immulina® has shown the ability to activate the production of immunoglobulin, interleukins, IFN, and TNF-α as well as augmenting the innate response of macrophages and dendritic cells [237,238]. This data suggests that the immunostimulating effects derived from Immulina® are able to protect against viral infection.
2.4.4. Euglena gracilis
Euglenophyceae is a unique class of excavate microalgae. Euglena gracilis is cultivated as a food and as a supplement. Paramylon is a polysaccharide found in Euglena gracilis that is used as a primary means of carbohydrate storage. Nakashima et al. examined the immunoprotective effect of Euglena gracilis and paramylon against IAV in vivo (Table 11) [239]. Paramylon and Euglena consumed as dietary supplements both increased production of interleukins, TNF, and IFNs, increased survival rates in mice, and lowered viral titers. It seems the beneficial effects were mediated by the activation of dendritic cells and induction of CD8+ T-cells and/or natural killer (NK) cells.
2.4.5. Polysaccharide AEX
The acidic polysaccharide AEX, isolated from the green (Chlorophyte) microalga Coccomyxa gloeobotrydiformis, was shown to have immunomodulatory effects (Table 11). Guo et al. examined the effects of AEX on immune response in chickens against infectious bursal disease virus (IBDV) [242]. Investigators found that the administration of AEX upregulated pro-inflammatory and T-helper cell differentiation cytokines as well as increasing production of nitric oxide (NO) in peripheral blood mononuclear cells (PBMCs). These effects are important components of immune response to viral infection [243]. In splenic lymphocytes, AEX was also found to repress major histocompatibility complex I (MHCI). This is an interesting result, as recent research indicates that MHCI can exacerbate viral infections despite its important role in host adaptive immunity [244]. It is important to note that MHCI plays a central role in activating cytotoxic T-lymphocytes (CTLs) which are a crucial player in adaptive immunity [25]. Thus, the use of AEX as an immunostimulant may have negative effects in certain situations. This study also demonstrated that AEX stimulates the differentiation of lymphocytes. It is important to note that the workings of the immune system are immensely complex and not comprehensively understood at this point in time. While it seems that AEX is an immunostimulant, the discovery that AEX represses MHCI function and lowers viral antibody titers suggests that the bodily response to AEX is more nuanced than is currently understood. It seems that AEX has potential as an immunostimulant, vaccine adjuvant, and direct AV drug, but more research is required to clarify AEX's mechanism.
2.5. Algae produce antioxidant compounds
The immune system often undergoes an oxidative response after viral infection. Proinflammatory cytokines are important for mobilization of the immune system against infection. The production of ROS can encourage proliferation of T-cells. Infection activates phagocytes, which release proinflammatory cytokines [245]. However, oxidative response to infection can also be harmful. Some viral replication cycles are promoted by oxidative conditions and overstimulation of the immune system [246,247]. For example, viruses often induce cell death via apoptosis as a component of their life cycles [245]. Antioxidants can limit viral replication by preventing apoptosis. Studies have shown that treatment with antioxidants can alleviate harmful infection pathologies and limit viral replication [248,249].
Many compounds from algae demonstrate antioxidant abilities. Sulfated polysaccharides from algae are scavengers of free radicals [250], so in addition to some of their direct inactivating effects on virions, they may improve clinical outcomes by reducing oxidative imbalance of infected cells. Phlorotannins have also demonstrated antioxidant, radical-scavenging ability in addition to their other antiviral activities [251,252]. Furthermore, algae produce many antioxidant compounds including ascorbate, glutathione, carotenoids (e.g. beta carotene and astaxanthin), amino acids, catechins, gallate, eckol, ascorbic acid, and tocopherols that could be incorporated into antiviral therapies [253,254].
3. Conclusion
On October 22, 2020, roughly 10 months after the first confirmed case of COVID-19 on US shores, the FDA approved the first drug to be used as treatment for COVID-19. This drug, known as remdesivir (RDV) and marketed under the name Veklury is manufactured by Gilead Sciences Inc. [255]. The initial announced cost of RDV will be from $2340–$3120 depending on geographical location and healthcare system provider [256]. RDV potently inhibits the MERS-CoV RNA-dependent RNA-polymerase enzyme, supporting the notion that it may also be a useful therapeutic against SARS-CoV-2 [257]. RDV was shown in one US trial to significantly reduce recovery times by 33.3% compared to placebo but has not demonstrated the ability to reduce mortality among infected COVID-19 patients [258]. In WHO trials, neither RDV nor the other proposed COVID treatments hydroxychloroquine, lopinavir, or IFN-β1a reduced mortality, initiation of ventilation treatment, or hospitalization duration [259].
Clearly there is a need for antiviral compounds that can be quickly deployed to confront pandemics. As we encounter novel pathogens, we need readily accessible sources of medicine that can provide additional protection to prevent some of their most harmful effects. Natural compounds operate through a myriad of different mechanisms and may be cost-effective solutions to this problem. Due to their incredible diversity, algae represent a relatively untapped source of such natural compounds.
In recent studies, algae-derived compounds have shown potential as anti-SARS-CoV-2 agents. These compounds act at different points during SARS-CoV-2 pathogenesis. Clinical trials examining the therapeutic potential of a carrageenan-based anti-SARS-CoV-2 nasal spray are underway in the USA, with similar research taking place in the UK [70,71]. Thus, algae-derived compounds may serve as a prophylactic measure, similar to wearing a mask, to prevent SARS-CoV-2 infection by preventing virions from entering the respiratory tract. In Section 2.2, we discussed algae-derived compounds that can inhibit viruses as they pass through the vasculature. SARS-CoV-2 viremia is associated with severe clinical outcomes [80], but lectins derived from algae show bioavailability when introduced into the blood and are potential direct inactivators of SARS-CoV-2 virions [260]. At the cellular level, several polysaccharides derived from algae have potential to prevent SARS-CoV-2 adhesion to target receptors. Fucoidan, glucuronomannan and sulfated galactofucan have all shown the ability to block interactions between the SARS-CoV-2 S-protein and cell surface receptors [133,261]. Recently, molecular docking analysis showed that polyphenols extracted from land plants are potential inhibitors of SARS-CoV-2 RDRP [202]. Algae are also prolific producers of polyphenols, and so compounds from algae may be able to block this important step in the viral replication cycle. The compound caulerpin from green (Chlorophyte) algae has recently shown promise as an inhibitor of the SARS-CoV-2 3CL protease [126]. By interfering with this enzyme, caulerpin may be a safe, effective blocker of SARS-CoV-2 replication. Furthermore, because SARS-CoV-2 is associated with a severe inflammatory response [262], antioxidant compounds from algae may also serve an important role in alleviating some harmful pathologies during the course of treatment [203]. Accumulating evidence indicates that algae-derived compounds may be able to make up broad-spectrum, multi-mechanism therapies that potently and economically inhibit SARS-CoV-2 infection. Deploying such treatment plans may prove an effective way to manage viral outbreaks on a large scale.
In this review, we examined stages in the viral replication cycle and discussed how compounds derived from algae inhibit viral infection and replication at each stage. We discussed many prophylactic compounds that can be topically applied to prevent viral entry into the body. These compounds are important to emphasize as easily accessible preventative measures that can reduce the need for clinical intervention and all the expenses of intensive medical care. We discussed how some viruses are transported through the body by way of the lymph and blood before they reach their target tissue and discussed a few mechanisms that algae-derived compounds can use to counter infection at this stage. Certain compounds from algae show the ability to directly interact with and degrade virions as they travel through the vasculature or augment the activities of phagocytes, the body's mobile defense system. We further discussed viral replication at the cellular level and mentioned compounds derived from algae that have the ability to limit infection during adhesion, entry, uncoating, replication, processing, and release. Many synthetic drugs operate at individual stages of the replication process; due to the diversity of natural compounds there is exceptional potential for developing multi-target treatment plans that could work throughout the infection cycle to limit viral replication. We further discussed the body's natural immune response to infection. We mentioned an array of algae-derived compounds that have demonstrated the ability to augment this immune response via a series of complex pathways and mechanisms. Intimately related to the immune response is the oxidative response to infection. While a thorough analysis of intra- and extracellular redox states during infection is beyond the scope of this paper, we mentioned algae-derived compounds that can reduce harmful oxidative effects associated with viral infection.
Natural products adapted for medicinal purposes can have powerful downstream health benefits in the human body. Gueven et al. performed research using human subjects and showed that the oral administration of fucoidan from the brown (Phaeophyceae) macroalga Undaria pinnatifida affected the expression of 53 micro-RNAs in the hours and days following consumption [229]. These micro-RNAs have been implicated in diverse functions throughout the human body relating to immune function, cell growth and division, inflammation, and neurological control. Health effects derived from the administration or consumption of algae-based compounds are complex but likely profound.
While it has grown somewhat in recent years, the scope of algae-antiviral literature is quite limited. Nevertheless, we have shown how a diverse array of algae-derived compounds can prevent against viral infection at the various stages of pathogenesis. In this review we demonstrated that species from all the major groups of algae have shown antiviral effects against a similarly broad diversity of viruses. The exceptional potential demonstrated by the existing literature represents the tip of an iceberg. There are certainly many more yet-unobserved compounds that could play a crucial part in controlling harmful widespread diseases through direct antiviral action, augmentation of protective endogenous biological pathways, or further processing and chemical modification to form effective semi-synthetic therapeutics. More research should be performed on antiviral compounds derived from algae and other natural sources, as such investigations might yield many life-saving drugs.
CRediT authorship contribution statement
Daman Reynolds: Conceptualization, Investigation, Writing – Original Draft, Visualization.
Michael Huesemann: Conceptualization, Writing – Review & Editing, Supervision, Project Supervision.
Scott Edmundson: Conceptualization, Writing – Review & Editing, Supervision.
Amy Sims: Writing – Review & Editing.
Brett Hurst: Writing – Review & Editing.
Sherry Cady: Writing – Review & Editing.
Nathan Beirne: Investigation.
Jacob Freeman: Investigation.
Adam Berger: Investigation.
Song Gao: Investigation.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
Acknowledgment
This work was supported in part by the U.S. Department of Energy, Office of Science, Office of Workforce Development for Teachers and Scientists (WDTS) under the Science Undergraduate Laboratory Internships Program (SULI).
A portion of the research described in this paper was conducted under the Laboratory Directed Research and Development Program at Pacific Northwest National Laboratory, a multiprogram national laboratory operated by Battelle for the U.S. Department of Energy.
Informed consent, human/animal rights
No conflicts, informed consent, or human or animal rights are applicable to this study.
References
- 1.Coronavirus Resource Center 2020. https://coronavirus.jhu.edu/map.html
- 2.Beltekian E.O.-O. Hannah Ritchie Diana, Mathieu Edouard, Hasell Joe, Macdonald Bobbie, Giattino Charlie, Roser Max, Yunits Breck, van Woerden Ernst, Gavrilov Daniel, Bergel Matthieu, Ahmad Shahid, Crawford Jason. Coronavirus (COVID-19) Deaths. 2020. https://ourworldindata.org/covid-deaths
- 3.Philip Schellekens D.S. 2020. COVID-19 Mortality in Rich and Poor Countries: A Tale of Two Pandemics? (Policy Res. Work. Pap). [Google Scholar]
- 4.Viglione G. The true toll of the pandemic. Nature. 2020;585 [Google Scholar]
- 5.McKee M., Stuckler D. If the world fails to protect the economy, COVID-19 will damage health not just now but also in the future. Nat. Med. 2020;26:640–642. doi: 10.1038/s41591-020-0863-y. [DOI] [PubMed] [Google Scholar]
- 6.Barrett P., Bluedorn J., Bogmans C., Carton B., Caselli F., Eugster J., Grigoli F., Jaumotte F., Kuan T., Lian W., Lio W., Mohommad A., Pescatori A., Pugacheva E., Sandri D., Tavares M., Valckx N., Voigts S. International Monetary Fund, Publication Services; Washington D.C.: 2020. International Monetary Fund. World Economic Outlook: A Long and Difficult Ascent. [Google Scholar]
- 7.Jones K.E., Patel N.G., Levy M.A., Storeygard A., Balk D., Gittleman J.L., Daszak P. Global trends in emerging infectious diseases. Nature. 2008;451:990–993. doi: 10.1038/nature06536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mackenzie J.S., Smith D.W. COVID-19: a novel zoonotic disease caused by a coronavirus from China: what we know and what we don’t. Microbiol. Aust. 2020;41:45. doi: 10.1071/ma20013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Parvez M.K., Parveen S. Evolution and emergence of pathogenic viruses: past, present, and future. Intervirology. 2017;60:1–7. doi: 10.1159/000478729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hassell J.M., Begon M., Ward M.J., Fèvre E.M. Urbanization and disease emergence: dynamics at the wildlife–livestock–human Interface. Trends Ecol. Evol. 2017;32:55–67. doi: 10.1016/j.tree.2016.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.UNEP . 2016. UNEP Frontiers 2016 Report: Emerging Issues of Environmental Concern. [Google Scholar]
- 12.Taylor L.H., Latham S.M., Woolhouse M.E.J. Risk factors for human disease emergence. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2001;356:983–989. doi: 10.1098/rstb.2001.0888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Daszak P. Emerging infectious diseases of wildlife--threats to biodiversity and human health. Science (80-. ) 2000;287:443–449. doi: 10.1126/science.287.5452.443. [DOI] [PubMed] [Google Scholar]
- 14.Tomley F.M., Shirley M.W. Livestock infectious diseases and zoonoses. Philos. Trans. R. Soc. B. 2009;364:2637–2642. doi: 10.1098/rstb.2009.0133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Epstein P.R. Climate change and emerging infectious diseases. Microbes Infect. 2001;3:747–754. doi: 10.1016/s1286-4579(01)01429-0. [DOI] [PubMed] [Google Scholar]
- 16.Mirsaeidi M., Motahari H., Khamesi M.T., Sharifi A., Campos M., Schraufnagel D.E. Climate change and respiratory infections. Ann. Am. Thorac. Soc. 2016;13:1223–1230. doi: 10.1513/AnnalsATS.201511-729PS. [DOI] [PubMed] [Google Scholar]
- 17.Afrane Y.A., Githeko A.K., Yan G. The ecology of Anopheles mosquitoes under climate change: case studies from the effects of deforestation in East African highlands. Ann. N. Y. Acad. Sci. 2012;1249:204–210. doi: 10.1111/j.1749-6632.2011.06432.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jaenson T.G., Hjertqvist M., Bergström T., Lundkvist Å. Why is tick-borne encephalitis increasing? A review of the key factors causing the increasing incidence of human TBE in Sweden. Parasit. Vectors. 2012;5:184. doi: 10.1186/1756-3305-5-184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Süss J., Klaus C., Gerstengarbe F., Werner P.C. What makes ticks tick? Climate change, ticks, and tick-borne diseases. J. Travel Med. 2008;15:39–45. doi: 10.1111/j.1708-8305.2007.00176.x. [DOI] [PubMed] [Google Scholar]
- 20.Starolis M.W., Perez O., Powell E.A. Clinical features and laboratory diagnosis of emerging arthropod-transmitted viruses: a report from the Pan American Society for Clinical Virology Clinical Practice Committee. J. Clin. Virol. 2020;132 doi: 10.1016/j.jcv.2020.104651. [DOI] [PubMed] [Google Scholar]
- 21.Altizer S., Dobson A., Hosseini P., Hudson P., Pascual M., Rohani P. Seasonality and the dynamics of infectious diseases. Ecol. Lett. 2006;9:467–484. doi: 10.1111/j.1461-0248.2005.00879.x. [DOI] [PubMed] [Google Scholar]
- 22.Barbara Majkowska-Wojciechowska M.L.K. In: Allergens, Air Pollutants, and Immune System Function in the Era of Global Warming. Khare M., editor. 2012. (Air Pollut. Monit. Model. Heal. Control). [DOI] [Google Scholar]
- 23.Gero J.W. Jeremy Kohlitz Anna, Lyons Stephanie, Boisson Sophie, Medlicott Kate. World Health Organization; 2019. Climate, Sanitation, and Health. [Google Scholar]
- 24.Epstein M.J., Bing E.G. Innov. Technol. Governance, Glob. Vol. 6. 2011. Delivering health care to the global poor: solving the accessibility problem; pp. 117–141. [DOI] [Google Scholar]
- 25.Cann A.J., Jill Leonard H.W. 6th ed. Elsevier; 2016. Principles of Molecular Virology. [DOI] [Google Scholar]
- 26.Ryu W.-S. Virus life cycle. Mol. Virol. Hum. Pathog. Viruses. 2017:31–45. doi: 10.1016/B978-0-12-800838-6.00003-5. [DOI] [Google Scholar]
- 27.Schätzl D.F.T. Springer; Berlin, Heidelberg: 2013. Viral Proliferation and Replication; pp. 31–38. (Mol. Virol). [DOI] [Google Scholar]
- 28.White D., Fenner F. 4th ed. Academic Press; 1994. Medical Virology. [Google Scholar]
- 29.Nathanson N., Ahmed R., Biron C.A., Gonzalez-Scarano F., Griffin D.E., Holmes K.V., Murphy F.A., Overbaugh J., Richman D.D., Robertson E.S., Robinson H.L. 2nd edition. Elsevier; 2007. Viral Pathogenesis and Immunity.https://books.google.com/books?hl=en&lr=&id=suFRboHkEUIC&oi=fnd&pg=PP1&dq=viral+pathogenesis&ots=hqope9LuVt&sig=M2suseyr02JDhoDLYBVxN8ll1lc#v=onepage&q=viral%20pathogenesis&f=false [Google Scholar]
- 30.De Clercq E., Li G. Approved antiviral drugs over the past 50 years. Clin. Microbiol. Rev. 2016;29:695–747. doi: 10.1128/cmr.00102-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Antonelli G., Turriziani O. Antiviral therapy: old and current issues. Int. J. Antimicrob. Agents. 2012;40:95–102. doi: 10.1016/j.ijantimicag.2012.04.005. [DOI] [PubMed] [Google Scholar]
- 32.Fitzgerald J.B., Schoeberl B., Nielsen U.B., Sorger P.K. Systems biology and combination therapy in the quest for clinical efficacy. Nat. Chem. Biol. 2006;2:458–466. doi: 10.1038/nchembio817. [DOI] [PubMed] [Google Scholar]
- 33.Joshi R.S., Jagdale S.S., Bansode S.B., Shankar S.S., Tellis M.B., Pandya V.K., Chugh A., Giri A.P., Kulkarni M.J. Discovery of potential multi-target-directed ligands by targeting host-specific SARS-CoV-2 structurally conserved main protease. J. Biomol. Struct. Dyn. 2020:1–16. doi: 10.1080/07391102.2020.1760137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Prasad A., Muthamilarasan M., Prasad M. Synergistic antiviral effects against SARS-CoV-2 by plant-based molecules. Plant Cell Rep. 2020;39:1109–1114. doi: 10.1007/s00299-020-02560-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tan X., Hu L., Luquette L.J., Gao G., Liu Y., Qu H., Xi R., Lu Z.J., Park P.J., Elledge S.J. Systematic identification of synergistic drug pairs targeting HIV. Nat. Biotechnol. 2012;30:1125–1130. doi: 10.1038/nbt.2391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhou Y., Hou Y., Shen J., Huang Y., Martin W., Cheng F. Network-based drug repurposing for novel coronavirus 2019-nCoV/SARS-CoV-2. Cell Discov. 2020;6 doi: 10.1038/s41421-020-0153-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lin L.T., Hsu W.C., Lin C.C. Antiviral natural products and herbal medicines. J. Tradit. Complement. Med. 2014;4:24–35. doi: 10.4103/2225-4110.124335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Newman D.J., Cragg G.M., Snader K.M. The influence of natural products upon drug discovery (Antiquity to late 1999) Nat. Prod. Rep. 2000;17:215–234. doi: 10.1039/a902202c. [DOI] [PubMed] [Google Scholar]
- 39.Lee R.E. Cambridge University Press; Cambridge: 2012. Basic Characteristics of the Algae; pp. 3–30. (Phycology). [DOI] [Google Scholar]
- 40.Burja A.M., Banaigs B., Abou-Mansour E., Grant Burgess J., Wright P.C. Marine cyanobacteria—a prolific source of natural products. Tetrahedron. 2001;57:9347–9377. doi: 10.1016/s0040-4020(01)00931-0. [DOI] [Google Scholar]
- 41.Cardoso P.J.S. Susan M., Carvalho Loic G., Rodrigues Mara S. Bioproducts from seaweeds: a review with special focus on the Iberian Peninsula. Curr. Org. Chem. 2014 doi: 10.2174/138527281807140515154116. [DOI] [Google Scholar]
- 42.Benemann J. Microalgae for biofuels and animal feeds. Energies. 2013;6:5869–5886. doi: 10.3390/en6115869. [DOI] [Google Scholar]
- 43.Web of Science 2021. https://apps.webofknowledge.com/Search.do?product=UA&SID=5ED2x7GR4OpTI5g16Pk&search_mode=GeneralSearch&prID=94092cd4-bd80-4dcd-8001-dc82d6d82612 (2021)
- 44.Taylor M.W. Springer; 2014. Viruses and Man: A History of Interactions. [DOI] [Google Scholar]
- 45.Koenighofer M., Lion T., Bodenteich A., Prieschl-Grassauer E., Grassauer A., Unger H., Mueller C.A., Fazekas T. Carrageenan nasal spray in virus confirmed common cold: individual patient data analysis of two randomized controlled trials. Multidiscip. Respir. Med. 2014;9:57. doi: 10.1186/2049-6958-9-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ludwig M., Enzenhofer E., Schneider S., Rauch M., Bodenteich A., Neumann K., Prieschl-Grassauer E., Grassauer A., Lion T., Mueller C.A. Efficacy of a Carrageenan nasal spray in patients with common cold: a randomized controlled trial. Respir. Res. 2013;14:124. doi: 10.1186/1465-9921-14-124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Eccles R., Meier C., Jawad M., Weinmüllner R., Grassauer A., Prieschl-Grassauer E. Efficacy and safety of an antiviral Iota-Carrageenan nasal spray: a randomized, double-blind, placebo-controlled exploratory study in volunteers with early symptoms of the common cold. Respir. Res. 2010;11:108. doi: 10.1186/1465-9921-11-108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Morokutti-Kurz M., König-Schuster M., Koller C., Graf C., Graf P., Kirchoff N., Reutterer B., Seifert J.-M., Unger H., Grassauer A., Prieschl-Grassauer E., Nakowitsch S. The intranasal application of zanamivir and carrageenan is synergistically active against influenza A virus in the murine model. PLoS One. 2015;10 doi: 10.1371/journal.pone.0128794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wang W., Wu J., Zhang X., Hao C., Zhao X., Jiao G., Shan X., Tai W., Yu G. Inhibition of influenza A virus infection by fucoidan targeting viral neuraminidase and cellular EGFR pathway. Sci. Rep. 2017;7:40760. doi: 10.1038/srep40760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hayashi T., Hayashi K., Kanekiyo K., Ohta Y., Lee J.-B. In: Combat. Threat Pandemic Influ. Drug Discov. Approaches. Ed P.F. Torrence., editor. John Wiley & Sons; Hoboken, NJ, USA: 2007. Promising antiviral glyco-molecules from an edible alga; pp. 166–182. [Google Scholar]
- 51.Richards C., Williams N.A., Fitton J.H., Stringer D.N., Karpiniec S.S., Park A.Y. Oral fucoidan attenuates lung pathology and clinical signs in a severe influenza A mouse model. Mar. Drugs. 2020;18:246. doi: 10.3390/md18050246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Synytsya A., Bleha R., Synytsya A., Pohl R., Hayashi K., Yoshinaga K., Nakano T., Hayashi T. Mekabu fucoidan: structural complexity and defensive effects against avian influenza A viruses. 2014;111:633–644. doi: 10.1016/j.carbpol.2014.05.032. [DOI] [PubMed] [Google Scholar]
- 53.Marais D., Gawarecki D., Allan B., Ahmed K., Altini L., Cassim N., Gopolang F., Hoffman M., Ramjee G., Williamson A.-L. The effectiveness of Carraguard, a vaginal microbicide, in protecting women against high-risk human papillomavirus infection. Antivir. Ther. 2011;16:1219–1226. doi: 10.3851/imp1890. [DOI] [PubMed] [Google Scholar]
- 54.A P.C., Maranto Perino M., De Franciscis P., Marci R., Restivo V., Manzone M., Capra G., Cucinella G., Calagna G. Impact of a new carrageenan-based vaginal microbicide in a female population with genital HPV-infection: first experimental results. Eur. Rev. Med. Pharmacol. Sci. 2019;23:6744–6752. doi: 10.26355/eurrev_201908_18567. [DOI] [PubMed] [Google Scholar]
- 55.Skoler-Karpoff S., Ramjee G., Ahmed K., Altini L., Plagianos M.G., Friedland B., Govender S., De Kock A., Cassim N., Palanee T., Dozier G., Maguire R., Lahteenmaki P. Efficacy of Carraguard for prevention of HIV infection in women in South Africa: a randomised, double-blind, placebo-controlled trial. Lancet. 2008;372:1977–1987. doi: 10.1016/s0140-6736(08)61842-5. [DOI] [PubMed] [Google Scholar]
- 56.Derby N., Aravantinou M., Kenney J., Ugaonkar S.R., Wesenberg A., Wilk J., Kizima L., Rodriguez A., Zhang S., Mizenina O., Levendosky K., Cooney M.L., Seidor S., Gettie A., Grasperge B., Blanchard J., Piatak M., Lifson J.D., Fernández-Romero J., Zydowsky T.M., Robbiani M. An intravaginal ring that releases three antiviral agents and a contraceptive blocks SHIV-RT infection, reduces HSV-2 shedding, and suppresses hormonal cycling in rhesus macaques. Drug Deliv. Transl. Res. 2017;7:840–858. doi: 10.1007/s13346-017-0389-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ugaonkar S.R., Wesenberg A., Wilk J., Seidor S., Mizenina O., Kizima L., Rodriguez A., Zhang S., Levendosky K., Kenney J., Aravantinou M., Derby N., Grasperge B., Gettie A., Blanchard J., Kumar N., Roberts K., Robbiani M., Fernández-Romero J.A., Zydowsky T.M. A novel intravaginal ring to prevent HIV-1, HSV-2, HPV, and unintended pregnancy. 2015;213:57–68. doi: 10.1016/j.jconrel.2015.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Huskens D., Schols D. Algal lectins as potential HIV microbicide candidates. Mar. Drugs. 2012;10:1476–1497. doi: 10.3390/md10071476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Levendosky K., Mizenina O., Martinelli E., Jean-Pierre N., Kizima L., Rodriguez A., Kleinbeck K., Bonnaire T., Robbiani M., Zydowsky T.M., O’Keefe B.R., Fernández-Romero J.A. Vol. 59. 2015. Griffithsin and Carrageenan Combination to Target Herpes Simplex Virus 2 and Human Papillomavirus; pp. 7290–7298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hayashi K., Lee J.-B., Atsumi K., Kanazashi M., Shibayama T., Okamoto K., Kawahara T., Hayashi T. In vitro and in vivo anti-herpes simplex virus activity of monogalactosyl diacylglyceride from Coccomyxa sp. KJ (IPOD FERM BP-22254), a green microalga. PLoS One. 2019;14 doi: 10.1371/journal.pone.0219305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.de Souza Barros C., Garrido V., Melchiades V., Gomes R., Gomes M.W.L., Teixeira V.L., de Palmer Paixão I.C.N. Therapeutic efficacy in BALB/C mice of extract from marine alga Canistrocarpus cervicornis (Phaeophyceae) against herpes simplex virus type 1. J. Appl. Phycol. 2017;29:769–773. doi: 10.1007/s10811-016-0865-9. [DOI] [Google Scholar]
- 62.Park H.-J., Kurokawa M., Shiraki K., Nakamura N., Choi J.-S., Hattori M. Antiviral activity of the marine alga Symphyocladia latiuscula against Herpes Simplex Virus (HSV-1) in vitro and its therapeutic efficacy against HSV-1 infection in mice. Biol. Pharm. Bull. 2005;28:2258–2262. doi: 10.1248/bpb.28.2258. [DOI] [PubMed] [Google Scholar]
- 63.D.L.F. Castillo E., Corrales N., Álvarez D.M., Farías M.A., Henríquez A., Smith P.C., Agurto-Muñoz C., González P.A. Anti-herpetic activity of Macrocystis pyrifera and Durvillaea antarctica algae extracts against HSV-1 and HSV-2. Front. Microbiol. 2020;11 doi: 10.3389/fmicb.2020.02006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wang S., Lu Z., Wang S., Liu W., Gao J., Tian L., Wang L., Zhang X., Zhao X., Wang W., Li C. The inhibitory effects and mechanisms of polymannuroguluronate sulfate against human papillomavirus infection in vitro and in vivo. Carbohydr. Polym. 2020;241:116365. doi: 10.1016/j.carbpol.2020.116365. [DOI] [PubMed] [Google Scholar]
- 65.Stiles J., Guptill-Yoran L., Moore G.E., Pogranichniy R.M. Vol. 49. 2008. Effects of λ-Carrageenan on In Vitro Replication of Feline Herpesvirus and on Experimentally Induced Herpetic Conjunctivitis in Cats; p. 1496. [DOI] [PubMed] [Google Scholar]
- 66.Boncristiani H.F., Criado M.F., Arruda E. Respiratory viruses. Encycl. Microbiol. 2009:500–518. doi: 10.1016/B978-012373944-5.00314-X. [DOI] [Google Scholar]
- 67.Campo V.L., Kawano D.F., da Silva D.B., Carvalho I. Carrageenans: biological properties, chemical modifications and structural analysis – a review. Carbohydr. Polym. 2009;77:167–180. doi: 10.1016/j.carbpol.2009.01.020. [DOI] [Google Scholar]
- 68.BETADINE Cold Defense Nasal Spray, (n.d.). http://sg.betadine.com/en/sg/cold-and-flu/betadine-cold-defence-nasal-spray.
- 69.Instructions for Use: Coldamaris Prophylactic Nasal Spray, (n.d.). http://www.faam-zarin.com/images/Coldamaris prophylactic-GA_Englisch,.pdf.
- 70.Dogliotti A. Efficacy of a Nasal Spray Containing Iota-carrageenan in the Prophylaxis of COVID-19 Disease in Health Personnel Dedicated to Patients Care With COVID-19 Disease (CARR-COV-02) 2020. https://clinicaltrials.gov/ct2/show/study/NCT04521322 [DOI] [PMC free article] [PubMed]
- 71.Moakes R.J.A., Davies S.P., Stamataki Z., Grover L.M. BioRxiv. 2020. Formulation of a composite nasal spray enabling enhanced surface coverage and prophylaxis of SARS-COV-2.http://biorxiv.org/content/early/2020/11/18/2020.11.18.388645.abstract 2020.11.18.388645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Openshaw P.J. Crossing barriers: infections of the lung and the gut. Mucosal Immunol. 2009;2:100–102. doi: 10.1038/mi.2008.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Rodríguez A., Kleinbeck K., Mizenina O., Kizima L., Levendosky K., Jean-Pierre N., Villegas G., Ford B.E., Cooney M.L., Teleshova N., Robbiani M., Herold B.C., Zydowsky T., Romero J.A. Fernández. In vitro and in vivo evaluation of two carrageenan-based formulations to prevent HPV acquisition. 2014;108:88–93. doi: 10.1016/j.antiviral.2014.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Isaacs C.E., Kim K.S., Thormar H. Inactivation of enveloped viruses in human bodily fluids by purified lipids. Ann. N. Y. Acad. Sci. 1994:457–464. doi: 10.1111/j.1749-6632.1994.tb38947.x. [DOI] [PubMed] [Google Scholar]
- 75.Chen J., Li H., Zhao Z., Xia X., Li B., Zhang J., Yan X. Diterpenes from the marine algae of the genus Dictyota. Mar. Drugs. 2018;16:159. doi: 10.3390/md16050159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Sharaf A.A.M., Aboul-Enein A., Helmi S., Ballot A., Astani A., Schnitzler P. Molecular authentication and characterization of the antiherpetic activityof the cyanobacterium Arthrospira fusiformis. Pharmazie. 2009;65 doi: 10.1691/ph.2010.9724. [DOI] [PubMed] [Google Scholar]
- 77.Astani A., Reichling J., Schnitzler P. Comparative study on the antiviral activity of selected monoterpenes derived from essential oils. Phyther. Res. 2010;24:673–679. doi: 10.1002/ptr.2955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Coroneo M.T. The eye as the discrete but defensible portal of coronavirus infection. Ocul. Surf. 2021;19:176–182. doi: 10.1016/j.jtos.2020.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wang J., Pan L., Tang S., Ji J.S., Shi X. Mask use during COVID-19: a risk adjusted strategy. Environ. Pollut. 2020;266:115099. doi: 10.1016/j.envpol.2020.115099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Bermejo-Martin J.F., Gonzalez-Rivera M., Almansa R., Micheloud D., Tedim A., Dominguez-Gil M., Resino S., Martin-Fernansez M., Murua P.R. Viral RNA load in plasma is associated with critical illness and a dysregulated host 2 response in COVID-19. Crit. Care. 2020;24 doi: 10.1186/s13054-020-03398-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Abdul Ahmad S.A., Palanisamy U.D., Tejo B.A., Chew M.F., Tham H.W., Syed Hassan S. Geraniin extracted from the rind of Nephelium lappaceum binds to dengue virus type-2 envelope protein and inhibits early stage of virus replication. Virol. J. 2017;14:1–13. doi: 10.1186/s12985-017-0895-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Galabov A.S. Virucidal agents in the eve of manorapid synergy. GMS Krankenhhyg. Interdiszip. 2007;2 http://www.ncbi.nlm.nih.gov/pubmed/20200679%0Ahttp://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=PMC2831485 Doc18. [PMC free article] [PubMed] [Google Scholar]
- 83.Cagno V., Andreozzi P., D’Alicarnasso M., Silva P.J., Mueller M., Galloux M., Le Goffic R., Jones S.T., Vallino M., Hodek J., Weber J., Sen S., Janecek E.R., Bekdemir A., Sanavio B., Martinelli C., Donalisio M., Welti M.A.R., Eleouet J.F., Han Y., Kaiser L., Vukovic L., Tapparel C., Král P., Krol S., Lembo D., Stellacci F. Broad-spectrum non-toxic antiviral nanoparticles with a virucidal inhibition mechanism. Nat. Mater. 2018;17:195–203. doi: 10.1038/NMAT5053. [DOI] [PubMed] [Google Scholar]
- 84.Hudson J.B., Kim J.H., Lee M.K., Hong Y.K., DeWreede R.E. Multiple antiviral activities in extracts of seaweeds from British Columbia. Pharm. Biol. 1999;37:300–306. doi: 10.1076/phbi.37.4.300.5804. [DOI] [Google Scholar]
- 85.Becker D.E. Drug therapy in dental practice: general principles. Part 1 - pharmacokinetic considerations. Anesth. Prog. 2006;53:19–24. doi: 10.2344/0003-3006(2006)53[140:DTIDPG]2.0.CO;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Stanley T.H. Anesthesia for the 21st century. Baylor Univ. Med. Cent. Proc. 2000;13:7–10. doi: 10.1080/08998280.2000.11927635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Allen T.M., Cullis P.R. Drug delivery systems: entering the mainstream. Science (80-. ) 2004;303:1818–1822. doi: 10.1126/science.1095833. [DOI] [PubMed] [Google Scholar]
- 88.Kamei Y., Aoki M. A chlorophyll c2 analogue from the marine brown alga Eisenia bicyclis inactivates the infectious hematopoietic necrosis virus, a fish rhabdovirus. 2007;152:861–869. doi: 10.1007/s00705-006-0920-y. [DOI] [PubMed] [Google Scholar]
- 89.dos Santos Souza Marinho C.J.B.R. Robson, Leite V.L.T. José Paulo Gagliard, de Palmer Paixão C.A.D.B. Izabel Christina Nunes, Pereira A.M.V.P. Antônio Batista. Antiviral activity of 7-keto-stigmasterol obtained from green Antarctic algae Prasiola crispa against equine herpesvirus 1. J. Appl. Phycol. 2017;29:555–562. [Google Scholar]
- 90.El Hamid M.I.A., El Fatah W.M.A., El Morsi A.A., Draz M.S., Kallakuri S., Bungau S., Endres L., Bardaca C., Hafez E.E. Anti-HIV/HCV activity of cyanobacterial phycobiliproteins by a new standardized method using bacteriophage surrogates. Rev. Chim. 2019;70:3115–3122. [Google Scholar]
- 91.Boyd M.R., Gustafson K.R., McMahon J.B., Shoemaker R.H., O’Keefe B.R., Mori T., Gulakowski R.J., Wu L., Rivera M.I., Laurencot C.M., Currens M.J., J.H.C., Buckheit R.W., Nara P.L., Pannell L.K., Sowder R.C., Henderson L.E. Discovery of cyanovirin-N, a novel human immunodeficiency virus-inactivating protein that binds viral surface envelope glycoprotein gp120: potential applications to microbicide development. Antimicrob. Agents Chemother. 1997;41 doi: 10.1128/aac.41.7.1521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Millet J.K., Séron K., Labitt R.N., Danneels A., Palmer K.E., Whittaker G.R., Dubuisson J., Belouzard S. Middle East respiratory syndrome coronavirus infection is inhibited by griffithsin. Antivir. Res. 2016;133:1–8. doi: 10.1016/j.antiviral.2016.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lee C. Griffithsin, a highly potent broad-spectrum antiviral lectin from red algae: from discovery to clinical application. Mar. Drugs. 2019;17 doi: 10.3390/md17100567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Wang W., Zhang P., Hao C., Zhang X.E., Cui Z.Q., Guan H.S. In vitro inhibitory effect of carrageenan oligosaccharide on influenza A H1N1 virus. Antivir. Res. 2011;92:237–246. doi: 10.1016/j.antiviral.2011.08.010. [DOI] [PubMed] [Google Scholar]
- 95.Krylova N.V., Ermakova S.P., Lavrov V.F., Leneva I.A., Kompanets G.G., Iunikhina O.V., Nosik M.N., Ebralidze L.K., Falynskova I.N., Silchenko A.S., Zaporozhets T.S. The comparative analysis of antiviral activity of native and modified fucoidans from brown algae fucus evanescens in vitro and in vivo. Mar. Drugs. 2020;18:224. doi: 10.3390/md18040224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Sun Y.H., Chen X.L., Song L., Liu S., Yu H.H., Wang X.Q., Qin Y.K., Li P.C. Antiviral activity against avian leucosis virus subgroup J of degraded polysaccharides from Ulva pertusa. Biomed. Res. Int. 2018;2018 doi: 10.1155/2018/9415965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Ray S., Pujol C.A., Damonte E.B., Ray B. Additionally sulfated xylomannan sulfates from Scinaia hatei and their antiviral activities. Carbohydr. Polym. 2015;131:315–321. doi: 10.1016/j.carbpol.2015.06.019. [DOI] [PubMed] [Google Scholar]
- 98.Kim M., Yim J.H., Kim S.-Y., Kim H.S., Lee W.G., Kim S.J., Kang P.-S., Lee C.-K. In vitro inhibition of influenza A virus infection by marine microalga-derived sulfated polysaccharide p-KG03. 2012;93:253–259. doi: 10.1016/j.antiviral.2011.12.006. [DOI] [PubMed] [Google Scholar]
- 99.Guo Q., Shao Q., Xu W., Rui L., Sumi R., Eguchi F., Li Z. Immunomodulatory and anti-IBDV activities of the polysaccharide AEX from Coccomyxa gloeobotrydiformis. Mar. Drugs. 2017;15:1–15. doi: 10.3390/md15020036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Cheng X.C. Yuhao Sun Ziqiang, Liu Song, Yu Huahua, Wang Xueqin, Li Pengcheng. Degradation of polysaccharides from Grateloupia filicina and their antiviral activity to avian leucosis virus subgroup J. Mar. Drugs. 2017;15 doi: 10.3390/md15110345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Chung C.-Y., Liu C.-H., Burnouf T., Wang G.-H., Chang S.-P., Jassey A., Tai C.-J., Tai C.-J., Huang C.-J., Richardson C.D., Yen M.-H., Lin C.-C., Lin L.-T. Activity-based and fraction-guided analysis of Phyllanthus urinaria identifies loliolide as a potent inhibitor of hepatitis C virus entry. 2016;130:58–68. doi: 10.1016/j.antiviral.2016.03.012. [DOI] [PubMed] [Google Scholar]
- 102.Percot A., Yalçin A., Aysel V., Erduǧan H., Dural B., Güven K.C. Loliolide in marine algae. Nat. Prod. Res. 2009;23:460–465. doi: 10.1080/14786410802076069. [DOI] [PubMed] [Google Scholar]
- 103.Morán-Santibañez K., Peña-Hernández M., Cruz-Suárez L., Ricque-Marie D., Skouta R., Vasquez A., Rodríguez-Padilla C., Trejo-Avila L. Virucidal and synergistic activity of polyphenol-rich extracts of seaweeds against measles virus. Viruses. 2018;10:465. doi: 10.3390/v10090465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Shiomi F.O. Souichi Ohta Yasuki, Nakao Teruyuki, Aozasa Osamu, Takatoshi K.K. Nagate Shoji Yamaguchi, Nishi Masatoshi, Miyata Hideaki. Anti-Herpes simplex virus substances produced by the marine green alga, Dunaliella primolecta. J. Appl. Phycol. 1998;10:349–355. [Google Scholar]
- 105.Cirne-Santos C.C., Barros C.D.S., Gomes M.W.L., Gomes R., Cavalcanti D.N., Obando J.M.C., Ramos C.J.B., Villaça R.C., Teixeira V.L., Paixão I.C.N.D.P. In vitro antiviral activity against zika virus from a natural product of the brazilian brown seaweed Dictyota menstrualis. Nat. Prod. Commun. 2019;14 doi: 10.1177/1934578x19859128. 1934578X1985912. [DOI] [Google Scholar]
- 106.Cirne-Santos C.C., De Souza Barros C., De Oliveira M.C., Rabelo V.W.-H., Azevedo R.C., Teixeira V.L., Ferreira D.F., De Palmer Paixão I.C.N. In vitro studies on the inhibition of replication of Zika and Chikungunya viruses by dolastane isolated from seaweed Canistrocarpus cervicornis. Sci. Rep. 2020;10 doi: 10.1038/s41598-020-65357-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Chen K., Roca M. In vitro bioavailability of chlorophyll pigments from edible seaweeds. J. Funct. Foods. 2018;41:25–33. doi: 10.1016/j.jff.2017.12.029. [DOI] [Google Scholar]
- 108.Ohlsson L., Blom M., Bohlinder K., Carlsson A. Vol. 1. 1998. Orally Fed Digalactosyldiacylglycerol Is Degraded During Absorption in Intact and Lymphatic Duct Cannulated Rats; pp. 239–245. [DOI] [PubMed] [Google Scholar]
- 109.Eichmann T.O., Lass A. DAG tales: the multiple faces of diacylglycerol - stereochemistry, metabolism, and signaling. Cell. Mol. Life Sci. 2015;72:3931–3952. doi: 10.1007/s00018-015-1982-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Yasunaga K., Saito S., Zhang Y.L., Hernandez-Ono A., Ginsberg H.N. Effects of triacylglycerol and diacylglycerol oils on blood clearance, tissue uptake, and hepatic apolipoprotein B secretion in mice. J. Lipid Res. 2007;48:1108–1121. doi: 10.1194/jlr.M600524-JLR200. [DOI] [PubMed] [Google Scholar]
- 111.Weingärtner O., Böhm M., Laufs U. Controversial role of plant sterol esters in the management of hypercholesterolaemia. Eur. Heart J. 2009;30:404–409. doi: 10.1093/eurheartj/ehn580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Zaloga G.P. Phytosterols, lipid administration, and liver disease during parenteral nutrition. J. Parenter. Enter. Nutr. 2015;39:39S–60S. doi: 10.1177/0148607115595978. [DOI] [PubMed] [Google Scholar]
- 113.Murata M., Nakai Y., Kawazu K., Ishizaka M., Kajiwara H., Abe H., Takeuchi K., Ichinose Y., Mitsuhara I., Mochizuki A., Seo S. Loliolide, a carotenoid metabolite, is a potential endogenous inducer of herbivore resistance. Plant Physiol. 2019;179:1822–1833. doi: 10.1104/pp.18.00837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Kimura J., Maki N. New loliolide derivatives from the brown alga Undaria pinnatifida. J. Nat. Prod. 2002;65:57–58. doi: 10.1021/np0103057. [DOI] [PubMed] [Google Scholar]
- 115.Grabarczyk M., Wińska K., Mączka W., Potaniec B., Anioł M. Loliolide - the most ubiquitous lactone. Folia Biol. Oecol. 2015;11:1–8. doi: 10.1515/fobio-2015-0001. [DOI] [Google Scholar]
- 116.Gabor F., Bogner E., Weissenboeck A., Wirth M. The lectin-cell interaction and its implications to intestinal lectin-mediated drug delivery. Adv. Drug Deliv. Rev. 2004;56:459–480. doi: 10.1016/j.addr.2003.10.015. [DOI] [PubMed] [Google Scholar]
- 117.Zhang N., Ping Q.N., Huang G.H., Xu W.F. Investigation of lectin-modified insulin liposomes as carriers for oral administration. Int. J. Pharm. 2005;294:247–259. doi: 10.1016/j.ijpharm.2005.01.018. [DOI] [PubMed] [Google Scholar]
- 118.Pooja D., Kulhari H., Kuncha M., Rachamalla S.S., Adams D.J., Bansal V., Sistla R. Improving efficacy, oral bioavailability, and delivery of paclitaxel using protein-grafted solid lipid nanoparticles. Mol. Pharm. 2016;13:3903–3912. doi: 10.1021/acs.molpharmaceut.6b00691. [DOI] [PubMed] [Google Scholar]
- 119.Kwon H., Ryu Y.B., Kim Y., Song N., Kim C.Y., et al. In vitro antiviral activity of phlorotannins isolated from Ecklonia cava against porcine epidemic diarrhea coronavirus infection and hemagglutination. Bioorg. Med. Chem. 2013;21:4706–4713. doi: 10.1016/j.bmc.2013.04.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Manach C., Scalbert A., Morand C., Rémésy C., Jiménez L. Polyphenols: food sources and bioavailability. Am. J. Clin. Nutr. 2004;79:727–747. doi: 10.1093/ajcn/79.5.727. [DOI] [PubMed] [Google Scholar]
- 121.Habtemariam S. The therapeutic potential of rosemary (Rosmarinus officinalis) diterpenes for Alzheimer’s disease, evidence-based complement. Altern. Med. 2016;2016 doi: 10.1155/2016/2680409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Vadiraja B.B., Gaikwad N.W., Madyastha K.M. 20Hepatoprotective effect of C-phycocyanin: protection for carbon tetrachloride and R-(+)-pulegone-mediated hepatotoxicty in rats. Biochem. Biophys. Res. Commun. 1998;249:428–431. doi: 10.1006/bbrc.1998.9149. [DOI] [PubMed] [Google Scholar]
- 123.Remirez D., González A., Merino N., González R., Ancheta O., Romay C., Rodríguez S. Effect of phycocyanin in zymosan-induced arthritis in mice - phycocyanin as an antiarthritic compound. Drug Dev. Res. 1999;48:70–75. doi: 10.1002/(SICI)1098-2299(199910)48:2<70::AID-DDR4>3.0.CO;2-N. [DOI] [Google Scholar]
- 124.B.M.-K., Solymosi K. Phycobilins and phycobiliproteins used in the food industry and medicine. Mini Rev. Med. Chem. 2017;17:1173–1193. doi: 10.2174/1389557516666160912180155. [DOI] [PubMed] [Google Scholar]
- 125.Esteves P.O., de Oliveira M.C., de Souza Barros C., Cirne-Santos C.C., Laneuvlille V.T., Palmer Paixão I.C. Antiviral effect of caulerpin against chikungunya. Nat. Prod. Commun. 2019;14:1–6. doi: 10.1177/1934578X19878295. [DOI] [Google Scholar]
- 126.Abdelrheem D.A., Ahmed S.A., Abd El-Mageed H.R., Mohamed H.S., Rahman A.A., Elsayed K.N.M., Ahmed S.A. The inhibitory effect of some natural bioactive compounds against SARS-CoV-2 main protease: insights from molecular docking analysis and molecular dynamic simulation. J. Environ. Sci. Health A. 2020:1–14. doi: 10.1080/10934529.2020.1826192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Wang W., Wang S.X., Guan H.S. The antiviral activities and mechanisms of marine polysaccharides: an overview. Mar. Drugs. 2012;10:2795–2816. doi: 10.3390/md10122795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Carlucci M.J., Scolaro L.A., Damonte E.B. Herpes simplex virus type 1 variants arising after selection with an antiviral carrageenan: lack of correlation between drug susceptibility and syn phenotype. J. Med. Virol. 2002;68:92–98. doi: 10.1002/jmv.10174. [DOI] [PubMed] [Google Scholar]
- 129.Pujol C.A., Scolaro L.A., Ciancia M., Matulewicz M.C., Cerezo A.S., Damonte E.B. Antiviral activity of a carrageenan from Gigartina skottsbergii against intraperitoneal murine herpes simplex virus infection. Planta Med. 2006;72:121–125. doi: 10.1055/s-2005-373168. [DOI] [PubMed] [Google Scholar]
- 130.Hasui M., Matsuda M., Okutani K., Shigeta S. In vitro antiviral activities of sulfated polysaccharides from a marine microalga (Cochlodinium polykrikoides) against human immunodeficiency virus and other enveloped viruses. 1995;17:293–297. doi: 10.1016/0141-8130(95)98157-t. [DOI] [PubMed] [Google Scholar]
- 131.Kim H., Lim C.Y., Lee D.B., Seok J.H., Kim K.H. Inhibitory effects of Laminaria japonica fucoidans against noroviruses. Viruses. 2020 doi: 10.3390/v12090997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Sun Q.L., Li Y., Ni L.Q., Li Y.X., Cui Y.S., Jiang S.L., Xie E.Y., Du J., Deng F., Dong C.X. Structural characterization and antiviral activity of two fucoidans from the brown algae Sargassum henslowianum. Carbohydr. Polym. 2020;229:10. doi: 10.1016/j.carbpol.2019.115487. [DOI] [PubMed] [Google Scholar]
- 133.Kwon P.S., Oh H., Kwon S.-J., Jin W., Zhang F., Fraser K., Hong J.J., Linhardt R.J., Dordick J.S. Sulfated polysaccharides effectively inhibit SARS-CoV-2 in vitro. Cell Discov. 2020;6 doi: 10.1038/s41421-020-00192-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Aguilar-Briseño J.A., Cruz-Suarez L.E., Sassi J.F., Ricque-Marie D., Zapata-Benavides P., Mendoza-Gamboa E., Rodríguez-Padilla C., Trejo-Avila L.M. Sulphated polysaccharides from Ulva clathrata and Cladosiphon okamuranus seaweeds both inhibit viral attachment/entry and cell-cell fusion, in NDV infection. Mar. Drugs. 2015;13:697–712. doi: 10.3390/md13020697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Bi D., Yu B., Han Q., Lu J., White W.L., Lai Q., Cai N., Luo W., Gu L., Li S., Xu H., Hu Z., Nie S., Xu X. Immune activation of RAW264.7 macrophages by low molecular weight fucoidan extracted from New Zealand Undaria pinnatifida. J. Agric. Food Chem. 2018;66:10721–10728. doi: 10.1021/acs.jafc.8b03698. [DOI] [PubMed] [Google Scholar]
- 136.Galani I.E., Andreakos E. Neutrophils in viral infections: current concepts and caveats. J. Leukoc. Biol. 2015;98:557–564. doi: 10.1189/jlb.4vmr1114-555r. [DOI] [PubMed] [Google Scholar]
- 137.Teruya T., Tatemoto H., Konishi T., Tako M. Structural characteristics and in vitro macrophage activation of acetyl fucoidan from Cladosiphon okamuranus. Glycoconj. J. 2009;26:1019. doi: 10.1007/s10719-008-9221-x. [DOI] [PubMed] [Google Scholar]
- 138.Castro R., Zarra I., Lamas J. Water-soluble seaweed extracts modulate the respiratory burst activity of turbot phagocytes. 2004;229:67–78. doi: 10.1016/s0044-8486(03)00401-0. [DOI] [Google Scholar]
- 139.Castro R., Piazzon M.C., Zarra I., Leiro J., Noya M., Lamas J. Stimulation of turbot phagocytes by Ulva rigida C. Agardh polysaccharides. Aquaculture. 2006;254:9–20. doi: 10.1016/j.aquaculture.2005.10.012. [DOI] [Google Scholar]
- 140.Dalmo R.A., Seljelid R. The immunomodulatory effect of LPS, laminaran and sulfated laminaran beta(1,3)-D-glucan on Atlantic salmon, Salmo-salar L, macrophages in-vitro. J. Fish Dis. 1995;18:175–185. doi: 10.1111/j.1365-2761.1995.tb00275.x. [DOI] [Google Scholar]
- 142.Peddie S., Zou J., Secombes C.J. Immunostimulation in the rainbow trout (Oncorhynchus mykiss) following intraperitoneal administration of Ergosan. 2002;86:101–113. doi: 10.1016/s0165-2427(02)00019-3. [DOI] [PubMed] [Google Scholar]
- 143.Fujiki K., Shin D.-H., Nakao M., Yano T. Effects of K-carrageenan on the non-specific defense system of carp Cyprinus carpio. Fish. Sci. 1997;63:934–938. doi: 10.2331/fishsci.63.934. [DOI] [Google Scholar]
- 144.Ogata M., Matsui T., Kita T., Shigematsu A. Carrageenan primes leukocytes to enhance lipopolysaccharide-induced tumor necrosis factor alpha production. Infect. Immun. 1999;67:3284–3289. doi: 10.1128/iai.67.7.3284-3289.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Schmidt R.J., Chung L.Y., Andrews A.M., Spyratou O., Turner T.D. Biocompatibility of wound management products: a study of the effects of various polysaccharides on murine L929 fibroblast proliferation and macrophage respiratory burst. J. Pharm. Pharmacol. 1993;45:508–513. doi: 10.1111/j.2042-7158.1993.tb05589.x. [DOI] [PubMed] [Google Scholar]
- 146.Kim M., Lee J.E., Cho H., Jung H.G., Lee W., Seo H.Y., Lee S.H., Ahn D.G., Kim S.J., Yu J.W., Oh J.W. Antiviral efficacy of orally delivered neoagarohexaose, a nonconventional TLR4 agonist, against norovirus infection in mice. Biomaterials. 2020;263:120391. doi: 10.1016/j.biomaterials.2020.120391. [DOI] [PubMed] [Google Scholar]
- 147.Yoshizawa Y., Enomoto A., Todoh H., Ametani A., Kaminogawa S. Activation of murine macrophages by polysaccharide fractions from marine algae (Porphyra yezoensis) Biosci. Biotechnol. Biochem. 1993;57:1862–1866. doi: 10.1271/bbb.57.1862. [DOI] [PubMed] [Google Scholar]
- 148.Yoshizawa Y., Ametani A., Tsunehiro J., Nomura K., Itoh M., Fukui F., Kaminogawa S. Macrophage stimulation activity of the polysaccharide fraction from a marine alga (Porphyra yezoensis): structure-function relationships and improved solubility. Biosci. Biotechnol. Biochem. 1995;59:1933–1937. doi: 10.1271/bbb.59.1933. [DOI] [PubMed] [Google Scholar]
- 149.Yoshizawa Y., Tsunehiro J., Nomura K., Itoh M., Fukui F., Ametani A., Kaminogawa S. Vol. 60. 1996. In Vivo Macrophage-stimulation Activity of the Enzyme-degraded Water-soluble Polysaccharide Fraction from a Marine Alga (Gracilaria verrucosa) pp. 1667–1671. [DOI] [PubMed] [Google Scholar]
- 150.Teruya T., Takeda S., Tamaki Y., Tako M. Fucoidan isolated from Laminaria angustata var. longissima induced macrophage activation. Biosci. Biotechnol. Biochem. 2010;74:1960–1962. doi: 10.1271/bbb.100294. [DOI] [PubMed] [Google Scholar]
- 151.YO K.A.J. Choi E.M. Kim, Hwang J.K. Immunomodulating activity of arabinogalactan and fucoidan in vitro. J. Med. Food. 2005;8:446–453. doi: 10.1089/jmf.2005.8.446. [DOI] [PubMed] [Google Scholar]
- 152.Zhang L., Wang X., Hua Q., Wang J., Liu J., Yang Y. Synthesis and immunomodulatory activity of the sulfated tetrasaccharide motif of type B ulvanobiuronic acid 3-sulfate. Org. Biomol. Chem. 2020;18:7932–7935. doi: 10.1039/d0ob01852j. [DOI] [PubMed] [Google Scholar]
- 153.El-Boshy M.E., El-Ashram A.M., AbdelHamid F.M., Gadalla H.A. Immunomodulatory effect of dietary Saccharomyces cerevisiae, β-glucan and laminaran in mercuric chloride treated Nile tilapia (Oreochromis niloticus) and experimentally infected with Aeromonas hydrophila. Fish Shellfish Immunol. 2010;28:802–808. doi: 10.1016/j.fsi.2010.01.017. [DOI] [PubMed] [Google Scholar]
- 154.Lee C. Carrageenans as broad-spectrum microbicides: current status and challenges. Mar. Drugs. 2020;18:1–27. doi: 10.3390/MD18090435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Lee D.Y.W., Lin X., Paskaleva E.E., Liu Y., Puttamadappa S.S., Thornber C., Drake J.R., Habulin M., Shekhtman A., Canki M. Palmitic acid is a novel cd4 fusion inhibitor that blocks HIV entry and infection. AIDS Res. Hum. Retrovir. 2009;25:1231–1241. doi: 10.1089/aid.2009.0019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Mazumder S., Ghosal P.K., Pujol C.A., Carlucci M.J., Damonte E.B., Ray B. Isolation, chemical investigation and antiviral activity of polysaccharides from Gracilaria corticata (Gracilariaceae, Rhodophyta) Int. J. Biol. Macromol. 2002;31:87–95. doi: 10.1016/S0141-8130(02)00070-3. [DOI] [PubMed] [Google Scholar]
- 157.Matsuhiro B., Conte A.F., Damonte E.B., Kolender A.A., Matulewicz M.C., Mejías E.G., Pujol C.A., Zúñiga E.A. Structural analysis and antiviral activity of a sulfated galactan from the red seaweed Schizymenia binderi (Gigartinales, Rhodophyta) 2005;340:2392–2402. doi: 10.1016/j.carres.2005.08.004. [DOI] [PubMed] [Google Scholar]
- 158.Hayashi T., Hayashi K., Maeda M., Kojima I. Calcium spirulan, an inhibitor of enveloped virus replication, from a blue-green alga Spirulina platensis. J. Nat. Prod. 1996;59:83–87. doi: 10.1021/np960017o. [DOI] [PubMed] [Google Scholar]
- 159.Lee J.B., Srisomporn P., Hayashi K., Tanaka T., Sankawa U., Hayashi T. Effects of structural modification of calcium spirulan, a sulfated polysaccharide from Spirulina platensis, on antiviral activity. Chem. Pharm. Bull. 2001;49:108–110. doi: 10.1248/cpb.49.108. [DOI] [PubMed] [Google Scholar]
- 160.Shih S.-R., Tsai K.-N., Li Y.-S., Chueh C.-C., Chan E.-C. Inhibition of enterovirus 71-induced apoptosis by allophycocyanin isolated from a blue-green algaspirulina platensis. 2003;70:119–125. doi: 10.1002/jmv.10363. [DOI] [PubMed] [Google Scholar]
- 161.Rechter S., König T., Auerochs S., Thulke S., Walter H., Dörnenburg H., Walter C., Marschall M. Antiviral activity of Arthrospira-derived spirulan-like substances. 2006;72:197–206. doi: 10.1016/j.antiviral.2006.06.004. [DOI] [PubMed] [Google Scholar]
- 162.Kanekiyo K., Hayashi K., Takenaka H., Lee J.-B., Hayashi T. Anti-herpes simplex virus target of an acidic polysaccharide, nostoflan, from the edible blue-green alga Nostoc flagelliforme. Biol. Pharm. Bull. 2007;30:1573–1575. doi: 10.1248/bpb.30.1573. [DOI] [PubMed] [Google Scholar]
- 163.Santoyo S., Plaza M., Jaime L., Ibañez E., Reglero G., Señorans F.J. Pressurized liquid extraction as an alternative process to obtain antiviral agents from the edible microalga Chlorella vulgaris. J. Agric. Food Chem. 2010;58:8522–8527. doi: 10.1021/jf100369h. [DOI] [PubMed] [Google Scholar]
- 164.Sands J.A. Vol. 12. 1977. Inactivation and Inhibition of Replication of the Enveloped Bacteriophage 6 by Fatty Acids; pp. 523–528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Santoyo S., Jaime L., Plaza M., Herrero M., Rodriguez-Meizoso I., Ibañez E., Reglero G. Antiviral compounds obtained from microalgae commonly used as carotenoid sources. J. Appl. Phycol. 2012;24:731–741. doi: 10.1007/s10811-011-9692-1. [DOI] [Google Scholar]
- 166.Plouguerné E., De Souza L., Sassaki G., Cavalcanti J., Villela Romanos M., Da Gama B., Pereira R., Barreto-Bergter E. Vol. 11. 2013. Antiviral Sulfoquinovosildiacylglycerols (SQDGs) From the Brazilian Brown Seaweed Sargassum vulgare; pp. 4628–4640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Hayashi K., Hayashi T., Kojima I. Calcium spirulan: evaluation of anti-herpes and anti-human immunideficieny virus. AIDS Res. Hum. Retrovir. 1996;12:1463–1471. doi: 10.1089/aid.1996.12.1463. [DOI] [PubMed] [Google Scholar]
- 168.Miao B., Geng M., Li J., Li F., Chen H., Guan H., Ding J. Sulfated polymannuroguluronate, a novel anti-acquired immune deficiency syndrome (AIDS) drug candidate, targeting CD4 in lymphocytes. Biochem. Pharmacol. 2004;68:641–649. doi: 10.1016/j.bcp.2004.04.009. [DOI] [PubMed] [Google Scholar]
- 169.Meiyu G., Fuchuan L., Xianliang X., Jing L., Zuowei Y., Huashi G. The potential molecular targets of marine sulfated polymannuroguluronate interfering with HIV-1 entryInteraction between SPMG and HIV-1 rgp120 and CD4 molecule. Antivir. Res. 2003;59:127–135. doi: 10.1016/s0166-3542(03)00068-8. [DOI] [PubMed] [Google Scholar]
- 170.Wang S., Wang W., Hao C., Yunjia Y., Qin L., He M., Mao W. Antiviral activity against enterovirus 71 of sulfated rhamnan isolated from the green alga Monostroma latissimum. Carbohydr. Polym. 2018;200:43–53. doi: 10.1016/j.carbpol.2018.07.067. [DOI] [PubMed] [Google Scholar]
- 171.Wang S., Wang W., Hou L., Qin L., He M., Li W., Mao W. A sulfated glucuronorhamnan from the green seaweed Monostroma nitidum: characteristics of its structure and antiviral activity. Carbohydr. Polym. 2020;227:115280. doi: 10.1016/j.carbpol.2019.115280. [DOI] [PubMed] [Google Scholar]
- 172.Lu C.X., Li J., Sun Y.X., Qi X., Wang Q.J., Xin X.L., Geng M.Y. Sulfated polymannuroguluronate, a novel anti-AIDS drug candidate, inhibits HIV-1 Tat-induced angiogenesis in Kaposi’s sarcoma cells. Biochem. Pharmacol. 2007;74:1330–1339. doi: 10.1016/j.bcp.2007.06.012. [DOI] [PubMed] [Google Scholar]
- 173.Wilen C.B., Tilton J.C., Doms R.W. HIV: cell binding and entry. Cold Spring Harb. Perspect. Med. 2012;2:1–14. doi: 10.1101/cshperspect.a006866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Oda K., Matsuoka Y., Funahashi A., Kitano H. A comprehensive pathway map of epidermal growth factor receptor signaling. Mol. Syst. Biol. 2005;1 doi: 10.1038/msb4100014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Eierhoff T., Hrincius E.R., Rescher U., Ludwig S., Ehrhardt C. The epidermal growth factor receptor (EGFR) promotes uptake of influenza a viruses (IAV) into host cells. PLoS Pathog. 2010;6 doi: 10.1371/journal.ppat.1001099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Cian R., Drago S., De Medina F., Martínez-Augustin O. Proteins and carbohydrates from red seaweeds: evidence for beneficial effects on gut function and microbiota. Mar. Drugs. 2015;13:5358–5383. doi: 10.3390/md13085358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Hidari K.I.P.J., Takahashi N., Arihara M., Nagaoka M., Morita K., Suzuki T. Structure and anti-dengue virus activity of sulfated polysaccharide from a marine alga. Biochem. Biophys. Res. Commun. 2008;376:91–95. doi: 10.1016/j.bbrc.2008.08.100. [DOI] [PubMed] [Google Scholar]
- 178.Talarico L.B., Damonte E.B. Interference in dengue virus adsorption and uncoating by carrageenans. Virology. 2007;363:473–485. doi: 10.1016/j.virol.2007.01.043. [DOI] [PubMed] [Google Scholar]
- 179.Talarico L.B., Pujol C.A., Zibetti R.G.M., Faría P.C.S., Noseda M.D., Duarte M.E.R., Damonte E.B. The antiviral activity of sulfated polysaccharides against dengue virus is dependent on virus serotype and host cell. Antivir. Res. 2005;66:103–110. doi: 10.1016/j.antiviral.2005.02.001. [DOI] [PubMed] [Google Scholar]
- 180.Shao Q., Guo Q., Xu W.P., Li Z., Zhao T.T. Vol. 10. 2015. Specific Inhibitory Effect of κ-Carrageenan Polysaccharide on Swine Pandemic 2009 H1N1 Influenza Virus. e0126577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Gupta D.K., Kaur P., Leong S.T., Tan L.T., Prinsep M.R., Chu J.J.H. Anti-Chikungunya viral activities of aplysiatoxin-related compounds from the marine cyanobacterium Trichodesmium erythraeum. Mar. Drugs. 2014;12:115–127. doi: 10.3390/md12010115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Mitchell S.S., Faulkner D.J., Rubins K., Bushman F.D. Dolastatin 3 and two novel cyclic peptides from a Palauan collection of Lyngbya majuscula. J. Nat. Prod. 2000;63:279–282. doi: 10.1021/np990353f. [DOI] [PubMed] [Google Scholar]
- 183.Ahn M.-J., Yoon K.-D., Kim C.Y., Min S.-Y., Kim Y.-U., Kim H.J., Kim J.H., Shin C.-G., Lee C.-K., Kim T.G., Kim S.H., Huh H., Kim J. Inhibition of HIV-1 reverse transcriptase and HIV-1 integrase and antiviral activity of Korean seaweed extracts. J. Appl. Phycol. 2002;14:325–329. doi: 10.1023/a:1022192329471. [DOI] [Google Scholar]
- 184.Ahn M.J., et al. Inhibition of HIV-1 reverse transcriptase and protease by phlorotannins from the brown alga Ecklonia cava. Biol. Pharm. Bull. 2004;27:544–547. doi: 10.1248/bpb.27.544. [DOI] [PubMed] [Google Scholar]
- 185.Ahn M.J., Yoon K.D., Kim C.Y., Kim J.H., Shin C.G., Kim J. Inhibitory activity on HIV-1 reverse transcriptase and integrase of a carmalol derivative from a brown alga, Ishige okamurae. Phyther. Res. 2006;20:711–713. doi: 10.1002/ptr.1939. [DOI] [PubMed] [Google Scholar]
- 186.Pereira H.S., Leão-Ferreira L.R., Moussatch’ec N., Teixeirab V.L., Cavalcantib D.N., Costad L.J., Diazd R., Frugulhetti I.C.P.P. Antiviral activity of diterpenes isolated from the Brazilian marine alga Dictyota menstrualis against human immunodeficiency virus type 1 (HIV-1) Antivir. Res. 2004;64:69–76. doi: 10.1016/j.antiviral.2004.06.006. [DOI] [PubMed] [Google Scholar]
- 187.Queiroz K.C.S., Medeiros V.P., Queiroz L.S., Abreu L.R.D., Rocha H.A.O., Ferreira C.V., Jucá M.B., Aoyama H., Leite E.L. Inhibition of reverse transcriptase activity of HIV by polysaccharides of brown algae. 2008;62:303–307. doi: 10.1016/j.biopha.2008.03.006. [DOI] [PubMed] [Google Scholar]
- 188.Xianliang X., Meiyu G., Huashi G., Zelin L. Study on the mechanism of inhibitory action of 911 on replication of HIV-1 in vitro. Zhongguo Hai Yang Yao Wu Chin. J. Mar. Drugs. 2000;19:15–18. http://europepmc.org/abstract/CBA/339491 [Google Scholar]
- 189.Shi Q., Wang A., Lu Z., Qin C., Hu J., Yin J. Overview on the antiviral activities and mechanisms of marine polysaccharides from seaweeds. Carbohydr. Res. 2017;453–454:1–9. doi: 10.1016/j.carres.2017.10.020. [DOI] [PubMed] [Google Scholar]
- 190.Ahmadi A., Zorofchian Moghadamtousi S., Abubakar S., Zandi K. Antiviral potential of algae polysaccharides isolated from marine sources: a review. Biomed. Res. Int. 2015;2015:825203. doi: 10.1155/2015/825203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Nakashima H., Kido Y., Kobayashi N., Motoki Y., Neushul M., Yamamoto N. Purification and characterization of an avian myeloblastosis and human immunodeficiency virus reverse transcriptase inhibitor, sulfated polysaccharides extracted from sea algae. Antimicrob. Agents Chemother. 1987;31:1524–1528. doi: 10.1128/aac.31.10.1524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Ohta K. Ohta, Mizushina Y., Hirata N., Takemura M., Sugawara F., Matsukage A., Yoshida S., Sakaguchi K. Sulfoquinovosyldiacylglycerol, KM043, a new potent inhibitor of eukaryotic DNA polymerases and HIV-reverse transcriptase type 1 from a marine red alga, Gigartina tenella. Chem. Pharm. Bull. (Tokyo) 1998;46:684–686. doi: 10.1248/cpb.46.684. [DOI] [PubMed] [Google Scholar]
- 193.Loya S., Bakhanashvili M., Kashman Y., Hizi A. Vol. 316. 1995. Peyssonols A and B, Two Novel Inhibitors of the Reverse Transcriptases of Human Immunodeficiency Virus Types 1 and 2; pp. 789–796. [DOI] [PubMed] [Google Scholar]
- 194.Lau A.F., Siedlecki J., Anleitner J., Patterson G.M.L., Caplan F.R., Moore R.E. Inhibition of reverse-transcriptase activity by extracts of cultured blue-green-algae (Cyanophyta) Planta Med. 1993;59:148–151. doi: 10.1055/s-2006-959631. [DOI] [PubMed] [Google Scholar]
- 195.Somchai P., Jitrakorn S., Thitamadee S., Meetam M., Saksmerprome V. Use of microalgae Chlamydomonas reinhardtii for production of double-stranded RNA against shrimp virus. Aquac. Rep. 2016;3:178–183. doi: 10.1016/j.aqrep.2016.03.003. [DOI] [Google Scholar]
- 196.Datta D., Nath Talapatra S., Swarnakar S. Bioactive compounds from marine invertebrates for potential medicines – an overview. Int. Lett. Nat. Sci. 2015;34:42–61. doi: 10.18052/www.scipress.com/ilns.34.42. [DOI] [Google Scholar]
- 197.Miceli L., Teixeira V., Castro H., Rodrigues C., Mello J., Albuquerque M., Cabral L., De Brito M., De Souza A. Vol. 11. 2013. Molecular Docking Studies of Marine Diterpenes as Inhibitors of Wild-Type and Mutants HIV-1 Reverse Transcriptase; pp. 4127–4143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Kannan M.G.K., Rengasamy R.R., Stirk Wendy A., Van Staden Johannes. Advances in algal drug research with emphasis on enzyme inhibitors. Biotechnol. Adv. 2014;32:1364–1381. doi: 10.1016/j.biotechadv.2014.08.005. [DOI] [PubMed] [Google Scholar]
- 199.Treitler D.S., Li Z., Krystal M., Meanwell N.A., Snyder S.A. Evaluation of HIV-1 inhibition by stereoisomers and analogues of the sesquiterpenoid hydroquinone peyssonol A. Bioorg. Med. Chem. Lett. 2013;23:2192–2196. doi: 10.1016/j.bmcl.2013.01.098. [DOI] [PubMed] [Google Scholar]
- 200.Tiollais P., Pourcel C., Dejean A. The hepatitis B virus. Nature. 1985;317(10):489–495. doi: 10.1038/317489a0. [DOI] [PubMed] [Google Scholar]
- 201.Jiang B., Xu X., Li L. Study on ‘911’ anti-HBV effect in HepG2.2.15 cells culture. Mod. Prev. Med. 2003;30:517–518. [Google Scholar]
- 202.Singh S., Sk M.F., Sonawane A., Kar P., Sadhukhan S. Plant-derived natural polyphenols as potential antiviral drugs against SARS-CoV-2 via RNA-dependent RNA polymerase (RdRp) inhibition: an in-silico analysis. J. Biomol. Struct. Dyn. 2020;0:1–16. doi: 10.1080/07391102.2020.1796810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Rosales-Mendoza S., García-Silva I., González-Ortega O., Sandoval-Vargas J.M., Malla A., Vimolmangkang S. The potential of algal biotechnology to produce antiviral compounds and biopharmaceuticals. Molecules. 2020;25:1–25. doi: 10.3390/molecules25184049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Park J.-Y., Kim J.H., Kwon J.M., Kwon H.-J., Jeong H.J., Kim Y.M., Kim D., Lee W.S., Ryu Y.B. Dieckol, a SARS-CoV 3CLpro inhibitor, isolated from the edible brown algae Ecklonia cava. Bioorg. Med. Chem. 2013;21:3730–3737. doi: 10.1016/j.bmc.2013.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Zainuddin E.N., Mentel R., Wray V., Jansen R., Nimtz M., Lalk M., Mundt S. Cyclic depsipeptides, ichthyopeptins A and B, from Microcystis ichthyoblabe. J. Nat. Prod. 2007;70:1084–1088. doi: 10.1021/np060303s. [DOI] [PubMed] [Google Scholar]
- 206.Lin C.W., Tsai F.J., Tsai C.H., Lai C.C., Wan L., Ho T.Y., Hsieh C.C., Chao P.D.L. Anti-SARS coronavirus 3C-like protease effects of Isatis indigotica root and plant-derived phenolic compounds. Antivir. Res. 2005;68:36–42. doi: 10.1016/j.antiviral.2005.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Idler D.R., Saito A., Wiseman P. Sterols in red algae (Rhodophyceae) Steroids. 1968;11:465–473. doi: 10.1016/s0039-128x(68)80062-5. [DOI] [PubMed] [Google Scholar]
- 208.Muramatsu T., Takemoto C., Kim Y.T., Wang H., Nishii W., Terada T., Shirouzu M., Yokoyama S. SARS-CoV 3CL protease cleaves its C-terminal autoprocessing site by novel subsite cooperativity. Proc. Natl. Acad. Sci. U. S. A. 2016;113:12997–13002. doi: 10.1073/pnas.1601327113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Lipinski C.A. Lead- and drug-like compounds: the rule-of-five revolution. Drug Discov. Today Technol. 2004;1:337–341. doi: 10.1016/j.ddtec.2004.11.007. [DOI] [PubMed] [Google Scholar]
- 210.Hodgson J. ADMET - turning chemicals into drugs. Rapidly resolving the pharmacokinetic and toxicological properties of drug candidates remains a key challenge for drug developers. Nat. Biotechnol. 2001;19:722–726. doi: 10.1038/90761. [DOI] [PubMed] [Google Scholar]
- 211.S.A. Ahmed, D.A. Abdelrheem, H.R. Abd El-Mageed, H.S. Mohamed, A.A. Rahman, K.N. M Elsayed, S.A. Ahmed, Destabilizing the structural integrity of COVID-19 by caulerpin and its derivatives along with some antiviral drugs: an in silico approaches for a combination therapy, (n.d.). doi: 10.1007/s11224-020-01586-w/Published. [DOI] [PMC free article] [PubMed]
- 212.Cannell R.J.P., Kellam S.J., Owsianka A.M., Walker J.M. Results of a large-scale screen of microalgae for the production of protease inhibitors. Planta Med. 1988:10–14. doi: 10.1055/s-2006-962319. [DOI] [PubMed] [Google Scholar]
- 213.Radau G. Serine protease inhibiting cyanopeptides. Pharmazie. 2000;55:554–559. [PubMed] [Google Scholar]
- 214.Moore R.E. Cyclic peptides and depsipeptides from cyanobacteria: a review. 1996;16:134–143. doi: 10.1007/bf01570074. [DOI] [PubMed] [Google Scholar]
- 215.Eckert D.M., Kim P.S. Mechanisms of viral membrane fusion and its inhibition. Annu. Rev. Biochem. 2001;70:777–810. doi: 10.1146/annurev.biochem.70.1.777. [DOI] [PubMed] [Google Scholar]
- 216.Ryu Y.B., Jeong H.J., Yoon S.Y., Park J.Y., Kim Y.M., Park S.J., Rho M.C., Kim S.J., Lee W.S. Influenza virus neuraminidase inhibitory activity of phlorotannins from the edible brown alga ecklonia cava. J. Agric. Food Chem. 2011;59:6467–6473. doi: 10.1021/jf2007248. [DOI] [PubMed] [Google Scholar]
- 217.Lee J.-B., Hayashi K., Hirata M., Kuroda E., Suzuki E., Kubo Y., Hayashi T. Vol. 29. 2006. Antiviral Sulfated Polysaccharide From Navicula directa, a Diatom Collected From Deep-sea Water in Toyama Bay; pp. 2135–2139. [DOI] [PubMed] [Google Scholar]
- 218.Buffa V., Stieh D., Mamhood N., Hu Q., Fletcher P., Shattock R.J. Cyanovirin-N potently inhibits human immunodeficiency virus type 1 infection in cellular and cervical explant models. J. Gen. Virol. 2009;90:234–243. doi: 10.1099/vir.0.004358-0. [DOI] [PubMed] [Google Scholar]
- 219.Huskens D., Férir G., Vermeire K., Kehr J.C., Balzarini J., Dittmann E., Schols D. Microvirin, a novel α(1,2)-mannose-specific lectin isolated from Microcystis aeruginosa, has anti-HIV-1 activity comparable with that of cyanovirin-N but a much higher safety profile. J. Biol. Chem. 2010;285:24845–24854. doi: 10.1074/jbc.M110.128546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Mori T., O’Keefe B.R., Sowder R.C., Bringans S., Gardella R., Berg S., Cochran P., Turpin J.A., Buckheit R.W., McMahon J.B., Boyd M.R. Isolation and characterization of Griffithsin, a novel HIV-inactivating protein, from the red alga Griffithsia sp. J. Biol. Chem. 2005;280:9345–9353. doi: 10.1074/jbc.M411122200. [DOI] [PubMed] [Google Scholar]
- 221.Xu T., Li Y., Wu H. Lian, Chen H., Wu H., Guo M., Zhao M., Wang C., Lin T., Lin Z., Chen D., Xiang W., Zhu B. The inhibition of enterovirus 71 induced apoptosis by Durvillaea antarctica through P53 and STAT1 signaling pathway. J. Med. Virol. 2020:1–7. doi: 10.1002/jmv.26693. [DOI] [PubMed] [Google Scholar]
- 222.Giurgea L.T., Morens D.M., Taubenberger J.K., Memoli M.J. Influenza neuraminidase: a neglected protein and its potential for a better influenza vaccine. Vaccines. 2020;8:409. doi: 10.3390/vaccines8030409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Ohuchi M., Asaoka N., Sakai T., Ohuchi R. Roles of neuraminidase in the initial stage of influenza virus infection. Microbes Infect. 2006;8:1287–1293. doi: 10.1016/j.micinf.2005.12.008. [DOI] [PubMed] [Google Scholar]
- 224.Lindwasser O.W., Resh M.D. Myristoylation as a target for inhibiting HIV assembly: unsaturated fatty acids block viral budding. Proc. Natl. Acad. Sci. U. S. A. 2002;99:13037–13042. doi: 10.1073/pnas.212409999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Saravanakumar D.E.M., Folb P.I., Campbell B.W., Smith P. Antimycobacterial activity of the red alga Polysiphonia virgata. Pharm. Biol. 2008;46:254–260. doi: 10.1080/13880200701739413. [DOI] [Google Scholar]
- 226.Gruters J.J.N. Rob A., de G. Matthijs Tersmette R.E.Y., Miedema H.G.H. Abraham Tulpt Frank, Ploeght H.L. Interference with HIV -induced syncytium formation and viral infectivity by inhibitors of trimming glucosidase. Nature. 1987;330:74–77. doi: 10.1038/330074a0. [DOI] [PubMed] [Google Scholar]
- 227.Ayehunie S., Belay A., Baba T.W., Ruprecht R.M. Inhibition of HIV-1 replication by an aqueous extract of Spirulina platensis (Arthrospira platensis) J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 1998;18:7–12. doi: 10.1097/00042560-199805010-00002. [DOI] [PubMed] [Google Scholar]
- 228.G.M., Austyn J. First Edit. Wiley-VCH Verlag GmbH & Co; 2012. The Immune System. (Explor. Immunol. Concepts Evid). [Google Scholar]
- 229.Gueven N., Spring K.J., Holmes S., Ahuja K., Eri R., Park A.Y., Fitton J.H. Micro RNA expression after ingestion of fucoidan; a clinical study. Mar. Drugs. 2020;18:143. doi: 10.3390/md18030143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.P.S.F., Weiss L. Sulfated glycans induce rapid hematopoietic progenitor cell mobilization: evidence for selectin-dependent and independent mechanisms. Blood. 2000;96:2460–2468. [PubMed] [Google Scholar]
- 232.Li C., Niu Q., Li S., Zhang X., Liu C., Cai C., Li G., Yu G. Fucoidan from sea cucumber Holothuria polii: structural elucidation and stimulation of hematopoietic activity. Int. J. Biol. Macromol. 2020;154:1123–1131. doi: 10.1016/j.ijbiomac.2019.11.036. [DOI] [PubMed] [Google Scholar]
- 233.Okimura T., Jiang Z., Komatsubara H., Hirasaka K., Oda T. Therapeutic effects of an orally administered edible seaweed-derived polysaccharide preparation, ascophyllan HS, on a Streptococcus pneumoniae infection mouse model. Int. J. Biol. Macromol. 2020;154:1116–1122. doi: 10.1016/j.ijbiomac.2019.11.053. [DOI] [PubMed] [Google Scholar]
- 234.Zhang W., Du J.-Y., Jiang Z., Okimura T., Oda T., Yu Q., Jin J.-O. Ascophyllan purified from Ascophyllum nodosum induces Th1 and Tc1 immune responses by promoting dendritic cell maturation. Mar. Drugs. 2014;12:4148–4164. doi: 10.3390/md12074148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Jiang Z., Okimura T., Yamaguchi K., Oda T. The potent activity of sulfated polysaccharide, ascophyllan, isolated from Ascophyllum nodosum to induce nitric oxide and cytokine production from mouse macrophage RAW264.7 cells: Comparison between ascophyllan and fucoidan. Nitric Oxide. 2011;25:407–415. doi: 10.1016/j.niox.2011.10.001. [DOI] [PubMed] [Google Scholar]
- 236.Pugh N.D., Edwall D., Lindmark L., Kousoulas K.G., Iyer A.V., Haron M.H., Pasco D.S. Oral administration of a Spirulina extract enriched for Braun-type lipoproteins protects mice against influenza A (H1N1) virus infection. Phytomedicine. 2015;22:271–276. doi: 10.1016/j.phymed.2014.12.006. [DOI] [PubMed] [Google Scholar]
- 237.Pugh N., Ross S.A., Elsohly H.N., Elsohly M.A., Pasco D.S. 2001. Lated With Enhanced Granulocyte-Macrophage Progenitor Cells in Mice Infected With; pp. 737–742. [Google Scholar]
- 238.Grzanna R., Polotsky A., Phan P.V., Pugh N., Pasco D., Frondoza C.G. Immolina, a high-molecular-weight polysaccharide fraction of spirulina, enhances chemokine expression in human monocytic THP-1 cells. J. Altern. Complement. Med. 2006;12:429–435. doi: 10.1089/acm.2006.12.429. [DOI] [PubMed] [Google Scholar]
- 239.Nakashima A., Suzuki K., Asayama Y., Konno M., Saito K., Yamazaki N., Takimoto H. Oral administration of Euglena gracilis Z and its carbohydrate storage substance provides survival protection against influenza virus infection in mice. Biochem. Biophys. Res. Commun. 2017;494:379–383. doi: 10.1016/j.bbrc.2017.09.167. [DOI] [PubMed] [Google Scholar]
- 240.Tsai M.-H., Chuang C.-C., Chen C.-C., Yen H.-J., Cheng K.-M., Chen X.-A., Shyu H.-F., Lee C.-Y., Young J.-J., Kau J.-H. Nanoparticles assembled from fucoidan and trimethylchitosan as anthrax vaccine adjuvant: in vitro and in vivo efficacy in comparison to CpG. Carbohydr. Polym. 2020;236:116041. doi: 10.1016/j.carbpol.2020.116041. [DOI] [PubMed] [Google Scholar]
- 241.Kuznetsova T.A., Persiyanova E.V., Ermakova S.P., Khotimchenko M.Y., Besednova N.N. The sulfated polysaccharides of brown algae and products of their enzymatic transformation as potential vaccine adjuvants. Nat. Prod. Commun. 2018;13:1083–1095. [Google Scholar]
- 242.Guo Q., Shao Q., Xu W., Rui L., Sumi R., Eguchi F., Li Z. Vol. 15. 2017. Immunomodulatory and Anti-IBDV Activities of the Polysaccharide AEX From Coccomyxa gloeobotrydiformis; p. 36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Akaike T., Maeda H. Nitric oxide and virus infection. Immunology. 2000;101:300–308. doi: 10.1046/j.1365-2567.2000.00142.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Xia S., Tao Y., Cui L., Yu Y., Xu S. MHC class I molecules exacerbate viral infection by disrupting type I interferon signaling. J Immunol Res. 2019;2019:5370706. doi: 10.1155/2019/5370706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Sansone C., Brunet C., Noonan D.M., Albini A. Marine algal antioxidants as potential vectors for controlling viral diseases. Antioxidants. 2020;9:392. doi: 10.3390/antiox9050392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Gullberg R.C., Jordan Steel J., Moon S.L., Soltani E., Geiss B.J. Oxidative stress influences positive strand RNA virus genome synthesis and capping. Virology. 2015;475:219–229. doi: 10.1016/j.virol.2014.10.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Heinrich R.B. Andreas Hensel Michael, Spiegler Verena, Kayser Oliver, Hempel Georg, Kraft Karin. Challenges at the time of COVID-19: opportunities and innovationsin antivirals from nature. Planta Med. 2020;86:659–664. doi: 10.1055/a-1177-4396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Fedoreyev S.A., Krylova N.V., Mishchenko N.P., Vasileva E.A., Pislyagin E.A., Iunikhina O.V., Lavrov V.F., Svitich O.A., Ebralidze L.K., Leonova G.N. Antiviral and antioxidant properties of echinochrome A. Mar. Drugs. 2018;16:1–10. doi: 10.3390/md16120509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Uchide N., Toyoda H. Antioxidant therapy as a potential approach to severe influenza-associated complications. Molecules. 2011;16:2032–2052. doi: 10.3390/molecules16032032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Wang L., Wang X., Wu H., Liu R. 2014. Overview on Biological Activities and Molecular Characteristics of Sulfated Polysaccharides From Marine Green Algae in Recent Years. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Kang S.-C.K. Soo-Jin Heo Sung-Myung, Cha Seon-Heui, Lee Seung-Hong, Kang Do-Hyung, Oha A.A. Won-Kyo Jung Chulhong, Jeon You-Jin. Inhibitory effect of diphlorethohydroxycarmalol on melanogenesis and itsprotective effect against UV-B radiation-induced cell damage. Food Chem. Toxicol. 2010;48:1355–1361. doi: 10.1016/j.fct.2010.03.001. [DOI] [PubMed] [Google Scholar]
- 252.Heo S.J., Jeon Y.J. Evaluation of diphlorethohydroxycarmalol isolated from Ishige okamurae for radical scavenging activity and its protective effect against H2O2-induced cell damage. Process Biochem. 2009;44:412–418. doi: 10.1016/j.procbio.2008.12.005. [DOI] [Google Scholar]
- 253.Mariya V., Ravindran V.S. Biomedical and pharmacological significance of marine macro algae-review. Indian J. Mar. Sci. 2013;42:527–537. [Google Scholar]
- 254.Kobayashi M., Kakizono T., Nishio N., Nagai S., Kurimura Y., Tsuji Y. Antioxidant role of astaxanthin in the green alga Haematococcus pluvialis. Appl. Microbiol. Biotechnol. 1997;48:351–356. doi: 10.1007/s002530051061. [DOI] [Google Scholar]
- 255.FDA Approves First Treatment for COVID-19 | FDA, (n.d.). https://www.fda.gov/news-events/press-announcements/fda-approves-first-treatment-covid-19 (accessed November 17, 2020).
- 256.Herper M. Gilead Announces Long-awaited Price for Covid-19 Drug Remdesivir. 2020. https://www.statnews.com/2020/06/29/gilead-announces-remdesivir-price-covid-19/
- 257.Gordon C.J., Tchesnokov E.P., Feng J.Y., Porter D.P., Götte M. The antiviral compound remdesivir potently inhibits RNAdependent RNA polymerase from Middle East respiratory syndrome coronavirus. J. Biol. Chem. 2020;295:4773–4779. doi: 10.1074/jbc.AC120.013056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Beigel J.H., Tomashek K.M., Dodd L.E., Mehta A.K., Zingman B.S., Kalil A.C., Hohmann E., Chu H.Y., Luetkemeyer A., Kline S., de Castilla D. Lopez, Finberg R.W., Dierberg K., Tapson V., Hsieh L., Patterson T.F., Paredes R., Sweeney D.A., Short W.R., Touloumi G., Lye D.C., Ohmagari N., Oh M., Ruiz-Palacios G.M., Benfield T., Fätkenheuer G., Kortepeter M.G., Atmar R.L., Creech C.B., Lundgren J., Babiker A.G., Pett S., Neaton J.D., Burgess T.H., Bonnett T., Green M., Makowski M., Osinusi A., Nayak S., Lane H.C. Remdesivir for the treatment of Covid-19 — final report. N. Engl. J. Med. 2020;383:1813–1826. doi: 10.1056/nejmoa2007764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Pan H., Peto R., Karim Q.A., Alejandria M., Henao-Restrepo A.M., García C.H., Kieny M.-P., Malekzadeh R., Murthy S., Preziosi M.-P., Reddy S., Periago M.R., Sathiyamoorthy V., Røttingen J.-A., Swaminathan S. MedRxiv. 2020. Repurposed antiviral drugs for COVID-19 –interim WHO SOLIDARITY trial results.http://medrxiv.org/content/early/2020/10/15/2020.10.15.20209817.abstract 2020.10.15.20209817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Barre A., Damme E.J.M.V., Simplicien M., Benoist H., Rougé P. Man-specific, GalNAc/T/Tn-specific and Neu5Ac-specific seaweed lectins as glycan probes for the SARS-CoV-2 (COVID-19) coronavirus. Mar. Drugs. 2020;18 doi: 10.3390/md18110543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Jin W., Zhang W., Mitra D., Mccandless M.G., Sharma P. The structure-activity relationship of the interactions of SARS-CoV-2 spike glycoproteins with glucuronomannan and sulfated galactofucan from Saccharina japonica. Int. J. Biol. Macromol. 2020;163:1649–1658. doi: 10.1016/j.ijbiomac.2020.09.184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Mehta P., McAuley D.F., Brown M., Sanchez E., Tattersall R.S., Manson J.J. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033–1034. doi: 10.1016/s0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]