Abstract
Whilst the entire world is battling the second wave of COVID-19, a substantial proportion of patients who have suffered from the condition in the past months are reporting symptoms that last for months after recovery, i. e., long-term COVID-19 symptoms. We aimed to assess the current evidence on the long-term symptoms in COVID-19 patients. We did a systematic review on PubMed, Web of Science, EMBASE, and Google Scholar from database inception to February 15, 2021, for studies on long-term COVID-19 symptoms. We included all type of papers that reported at least one long-term COVID-19 symptom. We screened studies using a standardized data collection form and pooled data from published studies. Cohort cross-sectional, case-report, cases-series, case-control studies, and review were graded using specific quality assessment tools. Of 11,361 publications found following our initial search we assessed 218 full-text articles, of which 145 met all selection criteria. We found that 20.70% of reports on long-term COVID-19 symptoms were on abnormal lung functions, 24.13% on neurologic complaints and olfactory dysfunctions, and 55.17% on specific widespread symptoms, mainly chronic fatigue, and pain. Despite the relatively high heterogeneity of the reviewed studies, our findings highlighted that a noteworthy proportion of patients who have suffered from SARS-CoV-2 infection present a “post-COVID syndrome.” The multifaceted understanding of all aspects of the COVID-19 pandemic, including these long-term symptoms, will allow us to respond to all the global health challenges, thus paving the way to a stronger public health.
Keywords: COVID-19, long-term symptoms, persistent symptoms, long-term sequalae, virus
Introduction
As of March 2021, about 117 million people worldwide have been diagnosed with COVID-19, with more than 2.6 million deaths (1). COVID-19 is caused by the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a heterogeneous virus that manifests itself with a wide spectrum of symptoms, from asymptomatic to life-threatening and fatal disease (2–7). Interstitial pneumonia is one of the most common features of SARS-CoV-2 and can be complicated by acute respiratory distress syndrome (ARDS), a disease related with high mortality, particularly in elderly people with multiple comorbidities (2, 3). As the pandemic of COVID-19 continues, numerous additional symptoms, such as fever, dry cough, shortness of breath, fatigue, myalgias, nausea/vomiting or diarrhea, headache, weakness, rhinorrhea, anosmia/ageusia, and many laboratory abnormalities, i.e., lymphopenia and elevated inflammatory markers (e.g., erythrocyte sedimentation rate, C-reactive protein, ferritin, tumor necrosis factor-α, IL-1, and IL-6) have been reported (2, 3). Other critical and severe complications of COVID-19 can include impaired function of the heart, brain, lung, liver, kidney, and coagulation system (4–7).
Most of the infected patients completely recovered after COVID-19 infection. However, a substantial proportion of patients who have been infected with SARS-CoV-2 continue to have symptoms long past the time that they recovered from the initial phases of COVID-19 disease. Clinicians worldwide called these long-term effects of COVID-19 “Long-Haul COVID-19” or “Long-term COVID-19” (8–11). In detail, “long-term COVID-19” defines those individuals who have had SARS-CoV-2 infection but do not recover completely over a period of a few weeks (commonly 2–3 weeks) (8–11). Based on the COVID-19 Symptom Study, a study carried-out on more than 4 million people in the US, UK, and Sweden, in which people enter their ongoing symptoms on a smartphone app, around 10% of patients who have tested positive for SARS-CoV-2 virus remain unwell beyond 3 weeks, and a smaller proportion for months (8). Thus, it is becoming clear, that some people who had a SARS-CoV-2 infection, even those described as “mild,” continue to suffer from persisting or cyclical symptoms. However, because COVID-19 is a novel disease, to date, there is not yet consensus on the definition of post-COVID-19 symptoms. Since long-term symptoms and complications have been described for other highly homologous human coronaviruses, i.e., Middle East Respiratory Syndrome (MERS) and Severe Acute Respiratory Syndrome (SARS), to date, it is unknown whether lessons from MERS and SARS are applicable to COVID-19 and the critical question is: “Do persistent symptoms at the post-viral stage of the disease constitute a post-COVID-19 syndrome (long-term COVID-19) and what are the main persistent symptoms in patients that might cause such a syndrome?” (12–14). The obvious answer is in research, but to date we do not know what to tell patients when they are asking about the course and prognosis of their ongoing complaints and potential long-term symptoms. Finding a concrete answer to these questions would also provide more information on the COVID-19 disease and enable comprehensive and targeted care to be given to survivors through the development of preventive and effective treatments. Although we are aware that it is too early to completely answer these questions, we believe that some general predictions are now possible, and would help to implement the right public health measures in particular after the pandemic has subsided. Thus, to give a complete overview on the persistent symptoms at the post-viral stage of COVID-19, we carried out a systematic review of the current data considering all types of papers evaluating individual persistent symptoms in mild, moderate, and severe/critical COVID-19 patients. Realizing the long-term sequelae of COVID-19 is imperative for understanding the complete history of disease, truly predicting the growing effect of the disease beyond hospitalization and mortality and defining whether inpatient or post-discharge-specific rehabilitation should be evaluated.
Methods
Eligibility Criteria
The PICO model was used to formulate the questions for this study: (1) studies that considered patients with long-term COVID-19 symptoms (Population), (2) studies where the primary aim was to evaluate long-term COVID-19 symptoms in mild, moderate, severe, and critical patients that have a follow-up of at least 14 days (Interventions), (3) studies with or without a control group (Comparisons), (4) studies that reported the long-term COVID-19 symptoms (Outcomes). Studies conducted up to February 15, 2021 were included in this review if they met the PICO criteria.
Search Strategies
Our systematic review involved a search conducted on February 15, 2021. We performed the review according to PRISMA statement (15). The search was carried out on PubMed, Web of Science, EMBASE, and Google Scholar databases to identify all type of papers on the long-term symptoms of COVID-19. The search was conducted combining the terms COVID-19, persistent symptoms, long-term symptoms, chronic symptoms, enduring symptoms, permanent symptoms. The combination of free-vocabulary and/or MeSH terms for the identification of studies in PubMed, Web of Science, EMBASE, and Google Scholar were reported in Table 1. Reference lists of relevant articles were searched for other potentially appropriate publications.
Table 1.
Cohort (perspective and retrospective), cross-sectional, case-report, cases-series and case-control studies on long-term lungs symptoms, long-term neurological and olfactory symptoms, and widespread long-term symptoms.
| References | Study type | Study country and period | Patients characteristics (numbers, gender, age) | COVID-19 severity | Hospitalization | ICU admission | Baseline COVID-19 symptoms | Method of evaluating long-term COVID-19 symptoms | Follow-up | Long-term COVID-19 symptoms |
|---|---|---|---|---|---|---|---|---|---|---|
| Persistent lungs symptoms and dysfunctions | ||||||||||
| Bellan et al. (18) | Prospective | •Novara, Italy •March 1 and June 29, 2020 |
•238 patients: •96 females •142 males •Mean age: 61 (50–71) |
Mild to-severe | Yes | 28 patients | Fever, cough, dyspnea, ageusia, anosmia, diarrhea, arthralgia, myalgia | DLCO, score for posttraumatic stress symptoms and for functional impairment | 120 days | DLCO reduced to less than 80% of the estimated value in 113 patients and less than 60% in 34 patients. Functional impairmentin 53 patients |
| Chun et al. (19) | Retrospective | New Haven, CT | •61 patients •44% females 56% males •Mean age: 53 (43–62) |
•13 mild •30 non-critical •18 critical |
30 patients | 18 patients | Dyspnea and cough | Pulmonary function tests, plasma biomarker profiling | 45–67 days | Dyspnea (69%), cough (58%). Pulmonary function declined as acute COVID-19 severity increased and not correlate with symptoms. LCN2, MMP-7, HGF were higher in ICU subjects and inversely correlated with pulmonary function |
| Daher et al. (20) | Prospective | •Aachen, Germany •February–May 2020 |
•33 patients •11 females •22 males •Mean age: 64 ± 3 |
Severe | Yes | No | •Increased D-dimer •LDH activity and CRP, ferritin, and IL-6 |
Body plethysmography, DLCO, blood gas analysis (ABG), 6-min walk test (6MWT), echocardiography, laboratory tests | 45 days | Reduced DLCO and 6MWT, and persistent fatigue and dyspnea in most patients. |
| Ding et al. (21) | Retrospective | •Wuhan, China •February–March 2020 |
•112 patients: •61 females •51 males •Mean age: 55.8 |
NR | Yes | NR | Fever, dry cough, fatigue, chest distress, dyspnea, myalgia | CT scan | 28 days | Abnormalities in 98.1 % of lungs CT scans (ground-glass opacities, crazy-paving pattern, consolidation and linear opacities) |
| Frija-Masson et al. (22) | Retrospective | •Paris, France •4 March−1 April 2020 |
•50 patients: •22 females •28 males •Age ≤ 85 |
•12 mild •17 moderate •16 severe •5 not classified |
Yes | 8 patients | Respiratory symptoms | Spirometry, functional residual capacity, total lung capacity, DLCO (single breath real-time CO/NH4) | 30 days | Impaired lung function in 54% of patients (restriction and/or altered DLCO), with a mix of restrictive and low diffusion patterns |
| Hall et al. (23) | Retrospective | •London, UK •May 2020 |
•200 patients: •38.5 females •61.5% males •Mean age: 54.8 ± 15.0 |
Moderate to- severe | 89 patients | 77 patients | NR | Dual energy CT or high-resolution CT, ventilation-perfusion scanning, spirometry, echocardiography and ECG | 30–45 days | 40% of patients with cardiorespiratory cause of breathlessness, i.e. persistent parenchymal abnormality pulmonary embolism, cardiac complications |
| Han et al. (24) | Prospective | •Hubei, China •December 25, 2019 - February 20, 2020 |
•114 patients: •34 females •80 males •Mean age: 54 ± 12 |
Severe | Yes | NR | Pneumonia | CT scan | 175 ± 20 days | Lung fibrotic-like changes in 35% patients, while in 65% patients complete radiological resolution (38%) or residual ground-glass opacification or interstitial thickening (27%) |
| Heiss et al. (25) | Case-reports | Erlangen, Germany | •1 male • 60-year-old |
Severe | Yes | No | Peripheral, multilobar areas of ground-glass Opacity (GGO) | CT scan MRI | 90 days | Residual pulmonary changes with patchy, peripheral GGOs, and consolidations |
| Hu et al. (26) | Retrospective | •Wuhan, China •1 January 2020–28 February 2020 |
•46 patients: •19 females •27 males •Mean age: 39.17 |
•36 mild/moderate •10 severe |
Yes | NR | Fever, cough, myalgia, fatigue, vomiting, or diarrhea | CT scan | 31 days | Lung lesions completely absorbed only in 28.57 % of patients |
| Latronico et al. (27) | Prospective | •Brescia, Italy •February–June 2020 |
•59 patients •Median age: 54–64 |
Critical | Yes | Yes | Acute respiratory distress syndrome | X-ray, spirometry | 90–180 days | Chest X-ray and pulmonary function altered in 70% of patients at 3 months; few patients had persisting respiratory symptoms at 6 months |
| Liang et al. (28) | Prospective | Wuhan, China | •76 patients: •55 females •21 males •Mean age 41.3 ± 13.8 |
•69 mild/general •7 severe/critical |
Yes | 9 patients | NR | Standard questionnaire; pulmonary function tests (total lung capacity -TLC, DLCO, carbon monoxide diffusion constant (DLCO/VA) | 90 days | 42% of patients with pulmonary function abnormalities |
| Liao et al. (29) | Retrospective | •Guangzhou, China •January 22,–April 10, 2020 |
•158 patients: •22 females •88 males •Mean age: 48.0 ± 17.7 |
•14 mild •110 moderate •34 severe |
Yes | 3 patients | fever, fatigue, diarrhea, polypnea, anorexia | Peripheral blood analyses (inflammatory cytokines expression), CT scan | 60 days | Persistent elevation of IL-6 associated with persistent pulmonary lesions |
| Manckoundia et al. (30) | Case-report | Dijon, France | 49-year-old man | Mild | No | No | Asthenia, fever, dry cough, dysgeusia, headache | General practitioner consult | 90 days | Non-inflammatory tracheal hypersecretion |
| Mo et al. (31) | Cross-sectional | •Guangzhou, China •February–March 2020 |
•110 patients: •55 females •55 males •Mean age: 49.10 |
•24 mild •67 moderate •19 severe |
Yes | NR | NR | Spirometry, DLCO | 20 ± 6 days in mild cases; 29 ± 8 days in moderate cases; 34 ± 7 days in severe cases | DLCO anomalies in 47.2% of patients, total lung capacity in 25.0%, forced expiratory volume in 1 s (FEV1) % in 13.6%, forced vital capacity (FVC) % in 9.1%, FEV1/FVC in 4.5% and small airway function in 7.3% of patients |
| Moreno-Perez et al. (32) | Prospective | •Alicante, Spain •February–April 2020 |
•277 patients: •47.3% females •52.7% males •Median age: 62.0 |
•34.3% mild •65.7% severe |
182 | NR | NR | Spirometry, chest radiology | 70 - 98 days | Spirometry alterations present in 9.3% patients, while in radiographs in 18.9% |
| Ramakrishnan et al. (33) | Retrospective | •Atlanta, USA •April, 2020 |
•107 patients: •26 males •81 females •Mean age: 55 |
NR | NR | NR | Fever, cough, smell or taste alteration | Lung auscultation, ECG | 30 days | 10% of with dyspnea and fatigue |
| Shah et al. (34) | Prospective | •Vancouver, Canada •March-May 2020 |
•60 patients: •32% females •68% males •Median age: 67 |
NR | Yes | NR | Dyspnoea, cough | Pulmonary function testing (PFT), 6 min walk test (6MWT), high-resolution CT of the chest | 90 days | More than half of patients with lung function and chest imaging abnormalities |
| Sonnweber et al. (35) | Prospective | Innsbruck, Austria | •109 patients: •44 females •65 males •Mean age: 58 |
•22 mild •34 moderate •35 severe •18 critical |
87 patients | 18 patients | NR | CT scan, serum biomarkers | 60 days | Iron deficiency in 30% of patients, anemia in 9%. Increased inflammation markers levels, such as IL-6 and C-reactive protein in anemic patients. 38% of patients with hyperferritinemia associated with severe lung pathologies |
| Tabatabaei et al. (36) | Retrospective | Kashan, Iran | •52 patients: •20 females •32 males •Mean age: 50.17 ± 13.1 |
Severe/critical | Yes | 11 patients | Fever, fatigue, dyspnea, GGO, consolidation, and mixed pattern | CT scan, serum biomarkers | 90 days | 42.3% with residual pulmonary disease. General poor health status in the domains of functional impairment (64%), fatigue (69%), QoL (72%) |
| Trinkmann et al. (37) | Prespective | •Heidelberg, Germany •March–June 2020 |
246 patients: Mean age: 48 ± 15 | Mild to- severe | 20 patients | 2 patients | Olfactory loss, cough, pyrexia, dyspnoea, sore throat, rhinitis, thoracic pain, limb pain, cephalgia, fatigue | Spirometry and body-plethysmography | 68 ± 16 days | Lower lung function even in younger SARS-CoV-2 convalescents with few comorbidities |
| Truffaut et al. (38) | Retrospective | •Brussels, Belgium •March–June 2020 |
•22 patients: •6 females •16 males •Mean age: 54.6 ± 10.9 |
Severe | Yes | Yes | NR | Pulmonary function test (PFT), 6-min walking distance test (6MWDT), dyspnoea (modified Medical Research Council (mMRC) | 90 days | 55% of patients with restrictive pattern ± altered DLCO. 65% with a 6MWDT below 80% and 52% were free from exertional dyspnoea according to mMRC scale |
| van den Borst et al. (39) | Prospective | •Nijmegen, The Netherlands •23 April - 15 July 2020 |
•124 patients •50 females •74 males •Mean age: 59 ± 14 |
•27 mild •51 moderate •26 severe •20 critical |
Yes | Yes | NR | CT scan Clinical Frailty Scale (CFS) Pulmonary function tests (DLCO, TLC) | 90 days | 90% of patients with residual pulmonary parenchymal abnormalities |
| van Gassel et al. (40) | Retrospective | March–May 2020 | 48 patients | Severe | Yes | Yes | Severe pneumonia | Pulmonary function testing (PFT), i.e.spirometry, lung volumes, DLCO adjusted for Hb, chest high-resolution CT (HRCT)imaging, and 6-minute-walk test (6-MWT) | 90 days | Reduced total lung capacity and diffusion capacity in 23 and 36 participants, respectively, but no airway obstruction on PFT. Ground-glass opacities in 89% of cases. Signs of reticulation, bronchiectasis, bronchiolectasis in 67% of cases |
| Weerahandi et al. (41) | Prospective | •New York, USA •April 15, 2020, |
•152 patients: •57 females •95 males •Mean age: 62 |
Severe | Yes | 101 | NR | Patient-Reported Outcomes Measurement Information System (PROMIS®) Dyspnea Characteristics instrument | 30–40 days | Shortness of breath in 74% of patients; 35.1% patients require home oxygen after hospital discharge |
| Yao et al. (42) | Case-report | •China •January 27, 2020 |
•1 female • 78-year-old |
Mild | Yes | No | Multiple patchy shadows in both lungs | Lungs biopsy | 14 days | Diffuse alveolar damage, extensive desquamation of proliferative type II alveolar epithelial cells, exudative monocytes and macrophages |
| Zhao et al. (43) | Retrospective | •3 tertiary hospitals of Henan Province, China •20 January−24 February 2020 |
•55 patients: •23 females •32 males •Mean age: 47.74 |
•4 mild •47 moderate •4 severe |
Yes | NR | Gastrointestinal symptoms, headache, fatigue, dyspnea, cough, sputum, olfactory, and gustatory dysfunctions | CT scan, pulmonary function test | 90 days | Abnormalities of pulmonary function and chest radiography in three quarters of patients. Higher D-dimer level at admission predict impaired DLCO 3 months after discharge |
| Zhu et al. (44) | Case-report | •Hubei, China •January 2020 |
30-year-old male | Severe | Yes | NR | Dry cough, fever, emphysema in both upper lungs, with ground glass density at the edge | Chest CT, laboratory examination results, lung function examination, sleep monitoring, sex hormones, sperm morphology and activity | 11 months | Abnormal airway function, cough, chest pain, chest tightness, and shortness of breath, unstructured sleep apnea hypopnea syndrome, and nocturnal sleep hypoxemia |
| Persistent neurological symptoms and olfactory dysfunctions | ||||||||||
| Boscolo-Rizzo (45) | Prospective | Treviso, Italy | 183 patients | Mild | NR | No | Fever, dry cough or coughing up mucus, loss of appetite, felt tired, altered sense of smell or taste | Interviews | 60 days | 18.6% of patients with altered sense of smell or taste |
| Caronna et al. (46) | Prospective | •Barcelona, Spain •28 March−22 April 2020 |
•130 patients: •66 females •64 males •Mean age: 53.9 |
Mild-to-severe | 80% | 8.5% | Headache, fever, malaise, myalgia, dizziness, cough, dyspnea, chest pain, expectoration, odynophagia, loss of smell/taste, diarrhea | Neurological assessment | 45 days | 74.6 % of patients had headache. At follow-up 37.8% of these had persistent headache (50% with no previous headache history) |
| D'Ascanio et al. (47) | Case-control | •Santa Croce Hospital AORMN, Fano-Pesaro, Italy •1 February–April 24, • 2020 |
•43 COVID-19 patients •25 healthy controls |
Mild | 20 patients | No | Anosmia, hyposmia, headache | A 7-question survey instrument, subjective olfactory dysfunction | 30 days | Resolution of anosmia or hyposmia in ~85% of patients |
| Dani et al. (48) | Case-series | London, UK | •6 female patients •Age: 26–50 years |
NR | No | No | Gastrointestinal symptoms, upper respiratory tract symptoms, chesty cough, flu-like symptoms | Echocardiogram | 21 days | Orthostatic intolerance syndromes (orthostatic hypotension, vasovagal syncope, postural orthostatic tachycardia syndrome) |
| Fjaeldstad et al. (49) | Retrospective | •Denmark •22 April−4 May 2020 |
•109 patients: •79 females •30 males •Mean age: 39.4 |
Mild | No | No | Fever, headache, fatigue, dyspnea, cough sputum, olfactory, gustatory loss | Subjective chemosensory function | > 30 days | 28% and 20% of patients not experienced improvement respectively of their olfactory and gustatory function, whereas 44% and 50% fully recovered olfactory and gustatory loss respectively |
| Galal et al. (50) | Cross-sectional | •Aswan, Egypt •18 July−31 August 2020 |
•430 patients: •274 females •156 males •Mean age: 37.4 ± 12.6 |
Mild-to-critical | 103 patients | 20 patients | Myalgia, fever, restriction of daily activities, memory loss | A 4-point Likert scale | 30 days | Myalgia (60.0%), arthralgia (57.2%), restriction of daily activities (57.0%), sleeping troubles (50.9%), nervousness and hopelessness (53.3%), anorexia (42.6%), chest pain (32.6%), gastritis (32.3%), cough (29.3%) and dyspnea (29.1%) |
| Gallus et al. (51) | Retrospective | •Sassari, Italy •April-May 2020 |
•48 patients: •37 females •11 males •37 (77%) Mean age: 45 |
Mild | No | No | Fever, dyspnea, cough, thoracic pain, asthenia, myalgia, diarrhea, conjuntivitis, general malaise, sore throat, headache, cutaneous rash, hypo-anosmia, hypo-ageusia | Tonal pure tone audiometry, a vHIT and SHIMP test | 14 days from the second negative swab | 8.3% patients reported hearing loss, 4.2% tinnitus, 8.3% dizziness, 2% spinning vertigo, 2% dynamic imbalance, 6.3 static imbalance |
| Guedj et al. (52) | Case-report | Marseill, France | •54-year-old man •62-year-old man |
•1 severe •1 moderate |
Yes | Yes | Acute respiratory distress syndrome, anosmia or ageusia | Whole-body18F-FDG PET | 30 days | Hypometabolism of the olfactory/rectus gyrus on the two patients |
| Hellmuth et al. (53) | Case-report | San Francisco, CA, USA | •33-year-old woman •56-year-old woman |
Mild | No | No | Neck pain, fatigue, fever, cough, myalgias, and non-migrainous headaches, cognitive symptoms | Cerebrospinal fluid and blood analyses, MRI | •149 days •72 days |
•Deficits in working memory and digit span backwards with high average attentional skills •Word finding difficulties, inefficient learning, and decreased organization leading to missed deadlines |
| Lim et al. (54) | Case-report | UK | 55-year-old woman | Mild | Yes | No | Fever, myalgia, cough, breathlessness, anosmia, ageusia, headache | CT scan, MRI, Addenbrooke's Cognitive Examination-III | 52 days | Persistent psychotic symptoms |
| Lu et al. (55) | Prospective | •Fuyang No.2 People‘s Hospital, China •January -February 2020 |
•60 patients: •26 females •34 males •Mean age: 44.10 •39 age and •sex-matched non COVID-19 controls |
•47 mild •12 severe •1 critical |
Yes | NR | Fever, cough, gastrointestinal symptoms, neurological symptoms | Diffusion tensor imaging (DTI), 3D high-resolution T1WI sequences | 90 days | 68.33% of patients with disruption to micro-structural and functional brain integrity during infection and 55% of them maintain the same symptoms after 90 days |
| Mendez et al. (56) | Prospective | •Valencia, Spain •March -April |
•179 patients •74 females •105 males •Mean rage: 22–81 |
Mild-to severe | Yes | 34 patients | NR | Standardized instruments evaluating neurocognitive function, psychiatric morbidity, and QoL | 60 days | 58.7% presented at least moderate neurocognitive decline, 39.1% psychiatric morbidity, and ~40% had poor QoL |
| Moein et al. (57) | Prospective | •Tehran, Iran •21 March−3 May, 2020 |
•82 patients: •28 females •45 males •Mean age: 45.53 |
•58 mild •30 moderate •12 severe |
Yes | No | Fever, cough, breathlessness, headache, myalgia, shivering, sweating, gastrointestinal symptoms, malaise, tinnitus, bloody sputum | 40-item University of Pennsylvania Smell Identification Test (UPSIT) | 40–60 days | 96% of patients with smell loss during infection. At follow-up, the test scores of 63% of the retested patients were normal. However, the mean UPSIT score at that time continued to remain below that of age- and sex matched healthy controls |
| Negrini et al. (58) | Case-series | •Milan, Italy •3 March−8 April, 2020 |
•9 patients: •3 females •6 males •Mean age: 60 |
•4 mild/moderate •5 severe |
Yes | 5 patients | NR | Mini-Mental State Examination (MMSE) test | 30 days | General cognitive decay in 33.3% of patients, with a specific decline in attention, memory, language, and praxis abilities. The cognitive decay appears to be associated with the length of stay (in days) in ICU |
| Novak et al. (59) | Case-report | Boston, USA | 64-year-old woman | NR | No | No | Cough, dyspnea | CT scan | 20 days | Probable orthostatic hypoperfusion syndrome and painful small fiber neuropathy in post- COVID disease. |
| Panda et al. (60) | Prospective | •New Delhi, India •23 April−29 June 2020 |
•225 patients: •63 females •159 males •3 transgenders •Mean age: 34.96 |
•145 mild •80 asymptomatic |
No | No | Otolaryngologic symptoms, fever, cough, dyspnea, gastrointestinal symptoms | Ear, Nose and Throat (ENT) symptoms evaluation | 28 days | 96% of the patients regaining ENT function at follow-up |
| Pilotto et al. (61) | Retrospective | •Brescia, Italy •February–April 2020 |
•165 patients: •50 females •115 males •Mean age: 64.8 ± 12.6 |
Moderate-to severe | Yes | NR | NR | Montreal Cognitive Assessment (MoCA) score | 180 days | Fatigue (34%), memory/attention (31%), sleep disorders (30%). 37.4% of patients with neurological abnormalities, i.e. cognitive deficits (17.5%), hyposmia (15.7%), postural tremor (13.8%) |
| Pritza et al. (62) | Retrospective | •Thessaloniki, Greece •March–April 2020 |
•90 patients: •37 females •53 males •Mean age: 55.8 •± 17.3 |
•45 mild •35 moderate •10 severe |
Yes | 10 patients | Olfactory and gustatory dysfunction | Questionnaires | 61 days | 8.57 % patients with persistent hyposmia |
| Raahimi et al. (63) | Case-report | Portsmouth, UK | 46-year-old man | Severe | Yes | Yes | Sensory loss in his feet, progressing to gait unsteadiness and distal lower limb weakness | Cerebrospinal fluid analysis, ECG, CT scan, MRI, spirometry | 90–150 days | At 90 days intermittent neuropathic pain and paraesthesia in distal limbs were present. At 150 days improvement in nerve function, with normalizing distal motor latencies |
| Sampaio Rocha-Filho et al. (64) | Case-report | Recife, Brazil | 40-year-old woman | Mild | No | No | Diarrhea, cough, fatigue, myalgia, anosmia, facial pain, headache | MRI, intracranial magnetic resonance angiography | 85 days | Persistent anosmia and headaches |
| Tobechukwu et al. (65) | Case-report | Red Bank, USA | 46-year-old woman | Mild | Yes | No | Fever, chest pain, vomiting, cough, confusion | X-ray, CT scan. MRI | 90 days | Delirium and allucinations |
| Ugurlu et al. (66) | Retrospective | •Çorum, Turkey •March–June 2020 |
•42 patients: •23 female 19 male Mean age: 41.2 ± 14.6 |
Mild | No | No | Fever, cough, dyspnea, diarrhea, sore throat, nasal drip, nasal obstruction, headache | Brief smell identification test | 90 days | Full recovery in 85.7% of patients. Olfactory dysfunction persisted in 14,3% of patients |
| Vaira et al. (67) | Prospective | •University •Hospital of Sassari, San Paolo Hospital in Milan, and Bellaria-Maggiore Hospital in •Bologna, Italy |
•138 patients: •70 females •68 males •Mean age: 51.2 |
Mild-to-severe | Yes | NR | Chemosensitive dysfunction | Self-administered olfactory and gustatory psychophysical tests in outpatients, Connecticut Chemosensory Clinical Research Center orthonasal olfaction test in hospitalized patients | 60 days | 5.8 % with moderate to severe olfactory dysfunction, 4.3 % with significant taste disorder. Four patients with combined chemosensitive dysfunctions, 4 patients with isolated smell impairments and two patients with isolated taste disorders |
| Yan et al. (68) | Cross-sectional | •California, USA •9 March–April 29, 2020 |
46 patients | NR | NR | NR | NR | 10-point scale score for sense of smell | 16 days | Olfactory dysfunction reported by 23 patients (17 reported no loss, 5 were unreachable, 1 died). At follow up 78% of patients with chemosensory dysfunction |
| Widespread persistent symptoms | ||||||||||
| Abdallah et al. (69) | Case-report | •Philadelphia, USA •March 2020 |
30-year-old man | Mild | No | No | Chest pain, fever, anosmia | X-ray, CT scan | 8 months | Chest pain, dyspnoea, and fatigue, intercostal neuralgia |
| Arnold et al. (70) | Prospective | •Southmead Way, Bristol •30 March and 3 June 2020 |
110 patients | 27 mild65 moderate18: severe | Yes | No | NR | Chest radiograph, spirometry, exercise test, bloods, and health-related quality of life (HRQoL) questionnaires | 83 days | Most (74%) patients with persistent symptoms (notably breathlessness and excessive fatigue) with reduced HRQoL |
| Buonsenso et al. (71) | Cross-sectional | •Rome, Italy •March–November 2020 |
•129 children: •62 females •67 males •Mean age: 11 ± 4.4 |
Mild-to severe | 6 patients | 3 patients | NR | Questionnaire | 162.5 ± 113.7 days | 35.7% had 1 or 2 symptoms and 22.5% had 3 or more. 52.7% had at least one symptom 120 days or more after diagnosis. Fatigue, muscle and joint pain, headache, insomnia, respiratory problems and palpitations are the main reported symptoms |
| Buselli et al. (72) | Case-report | Azienda Ospedaliero-Universitaria Pisana, Pisa, Italy | 50-year-old woman | Mild | No | No | Dry cough, asthenia, myalgia, diarrhea, fever, dyspnea, headache, fatigue, dysphonia | Pneumology examination, CT scan, neurological examination with brain scan, cardiology examination with echocardiograph, pulmonary ultrasound and ENT specialist examination | ≥ ys | Persistent fatigue and dysphonia |
| Carfi et al. (73) | Retrospective | •Fondazione Policlinico Universitario •Agostino Gemelli IRCCS, Rome, Italy •21 April – 29 May, 2020 |
•143 patients: •53 females •90 males •Mean age: 56.5 |
•21 mild •104 moderate •18 severe |
Yes | 18 patients | Fatigue, dyspnea, joint pain, chest pain, cough, anosmia, sicca syndrome, rhinitis, red eyes, dysgeusia, headache, sputum production, lack of appetite, sore throat, vertigo, myalgia, diarrhea | EuroQol visual analog scale | 60 days | At follow-up, only 12.6% of patients with no COVID-19 related symptom, while 32% had 1 or 2 symptoms and 55%had 3 or more. Main persistent symptoms were fatigue (53.1%), dyspnea (43.4%), joint pain, (27.3%) and chest pain (21.7%). |
| Carvalho-Schneider et al. (74) | Prospective | •Tours University Hospital, France •17 March−3 June, 2020 |
•150 patients: •84 females •66 males •Mean age: 49 |
Non-critical | Yes | No | Dyspnea, fever, weight loss, chest pain, headache, asthenia, myalgia, gastrointestinal symptoms, anosmia, ageusia | Clinical algorithm | 30 and 60 days | At 30 days 68% of patients with at least one symptom and 66% at 60 days. Anosmia/ageusia: 28% at 30 days, 23% at 60 days. Dyspnea: 36.7% at 30 days, 30% at 60 days. Asthenia: 50% at 30 days, 40%) at 60 days. Persistent symptoms at 60 days significantly associated with age 40–60, hospital admission and abnormal auscultation at symptom onset |
| Chen et al. (75) | Cross-sectional | •12 Hospitals in Wenzhou, Zhejiang, China •17 January−20 March, 2020 |
•361 patients: •175 females •186 males •Mean age: 47.22 |
•327 mild •34 severe |
Yes | NR | NR | Chinese version of Short-Form 36-item questionnaire (SF-36) | 30 days | Health-related quality of life (HRQoL) was poor among COVID-19 patients at follow-up |
| Cirulli et al. (76) | Prospective | •Nevada, USA •April–September 2020 |
233 patients | Mild | 8 patients | No | Fever, headache, asthenia, fatigue, diarrhea, ageusia, dry cough, chest pain, bone and joint pain, red eyes, dizziness, anorexia | Self-reported short and long-term symptoms | 30 and 90 days | 43.4% of patients with symptoms longer than 30 days, 24.1% with at least one symptom after 90 days. Long-term symptoms were anosmia, ageusia, difficulty concentrating, fatigue, dyspnea, memory loss, confusion, headache, heart palpitations, chest pain, pain with deep breaths, dizziness, and tachycardia |
| D'Cruz et al. (77) | Prospective | June–July 2020 | •119 patients: •45 females •74 males •Mean age: 58.7 ± 14.4 |
Severe | Yes | Yes | Pneumonia | X-ray, CT scan, clinical outcomes, symptom questionnaires, mental health screening, physiologic (4MGS and STS) al testing | 51–67 days | Persistent fatigue (68%), sleep disturbance (57%) and breathlessness (32%), post-traumatic stress disorder (25%), anxiety (22%) and depression (18%). 4MGS was slow in 38% and 35% desaturated by ≥4% during the STS test |
| Erçalik et al. (78) | Retrospective | •Istanbul, Turkey •March–May 2020 |
•206 patients: •105 females •101 males •Mean age: 56.24 ± 16.99 |
•153 mild •48 moderate •5 severe |
Yes | Yes | Fever, cough, dyspnea, runny nose | Pain assessment using a numeric rating scale | 45.99 ± 14.64 days | 40.7% of the patients had chronic pain for at least 3 months before COVID, and this rate increased to 82.5% during COVID and to 55.1% after COVID |
| Galván-Tejada et al. (79) | Case-control | •Zacatecas Mexico •July–September!!!!2020 |
•219 patients: •141 recovered •78 controls •51% females •49% males •Mean age female: 39.14 Mean age male: 39.01 |
NR | Yes | NR | NR | Questionnaire | 60 days | Chills, dyspnea, anosmia or dysgeusia, nausea or vomiting cough, red eyes as persistent symptoms in COVID-19 patients |
| Hosseini et al. (80) | Case-report | Qom, Iran | 48-year-old man | Mild | No | No | Fever, chills, weakness, lethargy, myalgia | Laboratory Tests, CT scan, ECG | 30 days | Persisten advanced atrioventricular block |
| Huang et al. (81) | Prospective | Wuhan, China between January−7 May 2020 | •1733 patients •48% females •52% males •Median age: 57.0 |
Mild to-severe | Yes | 76 patients | NR | Questionnaires, physical examination, blood tests, CT scan, 6-min walking test | 186 days | Fatigue or muscle weakness (63%), sleep difficulties (26%) were the most common symptoms. Anxiety or depression was reported among 23% of patients |
| Isoldi et al. (82) | Prospective | •Latina, Italy •April–June 2020 |
•15 children: •7 females •8 males •Median age: 12.2 |
Mild | No | No | Fever, hyperemia of the pharynx (53.3%), abdominal swelling, tender to the touch (33.3%), active conjunctival injection (6.7%) | Laboratory (blood, urine, feces) tests, ECG | 180 days | Two patients with hyperfiltration exhibited high blood pressure levels at diagnosis, and persistence of a prehypertension at 6-month follow-up |
| Iqbal et al. (83) | Cross-sectional | •Karachi, PAK •September–December!!!! 2020 |
•158 patients: •87 females •71 males •Mean age: 32.10 ± 12.42 |
•112 mild •33 moderate •13 severe |
Yes | 13 patients | NR | Questionnaire | 20–90 days | Fatigue (82.9%), poor sleep quality (56.3%), anxiety (53.2%), dyspnea (50%), joint pain (47.5%) were the most prevalent post-discharge manifestation |
| Jacobs et al. (84) | Prospective | •New Jersey, USA •22 March–April 16 |
•183 patients •38.5% females •61.5% males •Mean age: 57 •61.5% male |
•160 mild •23 severe |
Yes | 23 | Fatigue, shortness of breath, cough, lack of taste, muscular pain, diarrhea, lack of smell, production of phlegm, headache | PROMIS® instruments to identify symptoms and quality of life parameters | 35 days | Fatigue (55.0%), dyspnea (45.3%), muscular pain (51%), lower odds rating general health (41.5%), quality of life (39.8%), physical health (38.7%), mental health (43.7%) and social active role (38.7%) |
| Khalaf et al. (85) | Cross-sectional | •Assiut, Egypt •August–October 2020, |
•538 patients •Mean age: 41.17 ± 14.84 •45.9 females •54.1% males |
•Mild-to-severe •(61.3% mild, 31% moderate, 7.6% severe) |
51.3% of patients | 6.5% of patients | NR | Online questionnaire | 83 days | Fatigue (59.1%), sense of fever (46.5%), anorexia (24.3%), diarrhea (24.3%), loss of taste and smell (22.3%), headache (21.4%), cough (20.8), dyspnea (21%) |
| Ludvigsson et al. (86) | Case-report | •Stockholm, Sweden •October 2020 |
•5 children •Mean age: 12 •4 girls •1 boy |
Mild | No | No | Fever, dyspnea, abdominal pain, upper respiratory symptoms, dizziness, extreme fatigue, cough, lost taste and smell, headache, abdominal pain, diarrhea, nausea, norexia | NR | 6–8 months | Fatigue, dyspnoea, heart palpitations or chest pain, headaches, difficulties concentrating, muscle weakness, dizziness, sore throats |
| Mandal et al. (87) | Cross-sectional | London, UK | •384 patients •Mean age: 59.9 •38% females •62% males |
Mild-to-critical | Yes | 54 patients | NR | CT scan, blood tests, 11-point (0–10) scale score | 54 days | Persistent breathlessness (53%), cough (34%) fatigue (69%), depression (14.6%), elevated d-dimer (30.1%) and C reactive protein (9.5%), abnormal chest radiographs (38%) |
| Mahmud et al. (88) | Prospective | •Dhaka, •Bangladesh •June–August 2020 |
•355 patients •148 females •207 males •Mean age: 39.8 |
•221 mild •93 moderate 41 severe |
Yes | Yes | Fever, cough, respiratory distress, anosmia, anorexia headache, lethargy | Telephonic interview | At least 30 days | 46% of patients developed long-term symptoms. Post-viral fatigue (70%) was the most prevalent symptom. Post-COVID features are significantly higher among female |
| Martin et al. (89) | Retrospective | •USA •March–September!!!! 2020 |
9,989 patients | Mild- to severe | Yes | NR | NR | Electronic health records | 90–180 days | Persistent neuropsychiatric, pulmonary, metabolic, and coagulopathic phenotypes |
| Pellaud et al. (90) | Retrospective | •Fribourg, Switzerland •March–April 2020 |
•196 patients: •77 females •119 males •Mean age: 70 |
•Mild •Moderate •Severe/Critical |
Yes | 49 patients | NR | Data collected by electronic health records or by telephone | 30 days | Among 117 patients discharged from hospital within 30 days after the beginning of symptoms, 63% reported persistent symptoms. The main persistent symptoms are asthenia (67%), respiratory symptoms (56%), anosmia/dysgeusia (10%) |
| Petersen et al. (91) | Retrospective | •Tórshavn, Faroe Islands •22 April−16 August 2020 |
•180 patients •Mean age: 39.9 ± 19.4 •98 females •82 males |
Mild-to-moderate | 8 patients | No | Fatigue, fever, headache, chills, and loss of smell and taste | Questionnaire | 125 days | 53.1% reported persistence of at least one symptom, 33.3% reported one or two symptoms and 19.4% three or more symptoms. Most prevalent persistent symptoms: fatigue, loss of smell and taste, arthralgias |
| Raman et al. (92) | Prospective | Oxford, UKMarch–May 2020 | •58 patients: •24 females •34 males •Mean age: 55 ± 13 |
Moderate to- severe | Yes | 21 patients | Fever, malaise, shortness of breath, cough, dysgeusia, anosmia, diarrhea, chest pain, headache, vomiting | MRI of the brain, lungs, heart, liver, kidneys, 6-minute walk (6MWT) test, spirometry, cardiopulmonary exercise test (CPET), questionnaires, blood tests | 60–90 days | •64% of patients experienced breathlessness and 55% fatigue. MRI, abnormalities in lungs (60%), heart (26%), liver (10%), and kidneys (29%). •Impaired cognitive performance and reduced six-minute walk distance |
| Rosales-Castillo et al. (93) | Retrospective | March–May 2020 | •118 patients: •44.1 females •55.9% male •Mean age: 60.16 |
Mild to-severe | Yes | 7.6% of patients | Fever, cough, dyspnoea, diarrhea, ageusia, myalgia, anosmia, chest pain, headache, expectoration | Physician consultation | 50 days | 62.5% of patients reported persistence of symptoms: dyspnoea (31.4%), asthenia (30.5%), myalgia (13%), cough (5%), anosmia (1.7%), and ageusia (1%) |
| Saeed et al. (94) | Case-report | Lahore | •48-years-old- woman •42-years-old- woman •32-years-old- woman •37-years-old- woman |
Mild | No | No | Dry cough, fever, abdominal discomfort and diarrhea | Dermatological consulting | 60–90 days | Hair shedding: telogen effluvium |
| Saiful Islam et al. (95) | Cross-sectional | •Bangladesh •September–October 2020 |
•1,002 patients: •422 females •580 male •mean age = 34.7 ± 13.9 |
Mild to- severe | 208 patients | NR | Fever and fatigue | Online questionnaire | 30 days | 20% of patients reported persistent symptoms. The most reported persistent symptoms were diarrhea (12.7%) and fatigue (11.5%). 48% of participants had moderate to severe depression |
| Smane et al. (96) | Retrospective | •Riga, Latvia •July 2020 |
•30 children: •13 females •17 male •Mean age: 9.2 |
•5 asymptomatic •24 mild •1 moderate |
No | No | Fever, rhinorrhoea, cough | Physician assessment | 101 days | 70% patients completely free of any COVID-19-related symptoms, while 30% had at least one symptom (fever, joint pain, headache, anosmia, ageusia, microhaematuria) |
| Sofian et al. (97) | Case-series | •Arak, Iran •February–April 2020 |
•10 patients •9 females •1 male |
Mild-to moderate | No | No | Fever, dry cough, nasal congestion, weakness, high diaphoresis, loss of smell, fatigue | CT scan | 60 days | Dry cough, headache, severe sweating, shivering, loss of smell, mild on/off fever, and diarrhea, weight loss |
| Stavem et al. (98) | Cross-sectional | •Lørenskog, Norway •Until 1 June 2020 |
•451 patients: •253 females •198 males •Mean age: 49.7 |
Mild | No | No | Fever, loss of smell, headache, dry cough, myalgia, chills, dyspnea, sore throat, gastrointestinal manifestations | Mixed-mode survey | 117 days | 53 % of woman and 67 % of men with no persistent symptoms. Fatigue and dyspnoea are common about 60 days |
| Sykes et al. (99) | Retrospective | Hull, UK | •134 patients: •46 females •88 males •Median age: 58 |
Mild to-severe | Yes | 20% patients | Breathlessness, myalgia anxiety, fatigue, low mood, sleep disturbance | X-ray, standardized clinical assessment, questionnaires for dyspnea, and quality of life | 113 days | 86% of patients reported at least one residual symptom: breathlessness (60%), anxiety (47.8%), extreme fatigue (39.6%), lowmood (37.3%), and sleep disturbance (35.1%). Females reported most residual symptoms including anxiety, fatigue, and myalgia |
| Taboada et al. (100) | Prospective | •Santiago, Spain •March–April 2020 |
•91 patients: •32 females •59 males •Mean age: 65.5 |
Critical | Yes | All patients | Myalgia, asthenia, insomni, arthralgi, cough, anosmia, chest pain | Questionnaire | 180 days | Decrease in quality of life in 67% of patients (56% mobility, 37% usual activities, 13% self-care, 48% pain/discomfort, 46% anxiety/depression). Dyspnoea on exertion (57%), asthaenia (37%), myalgia (37%), and arthralgia (29%). Only 16% of patients were completely free of persistent symptoms |
| Townsend et al. (101) | Retrospective | Dublin, Ireland | •128 patients •54% females •46% males •Mean age: 49.5 ± 15 |
Mild-to critical | 71 patients | 18 patients | Fatigue | Chalder Fatigue Score (CFQ-11), markers of peripheral immune activation and circulating pro-inflammatory cytokines | 72 days | 52.3% of patients reported persistent fatigue. No association between fatigue and COVID-19 severity, laboratory markers of inflammation, pro-inflammatory molecules |
| Townsend et al. (102) | Cross-sectional | •Dublin, Ireland •March–May 2020 |
•153 patients: •57 females •96 males •Median age: 48 |
Mild-Critical | Yes | 19 patients | NR | X-ray | 75 days | Persistent abnormal x-rays of either persistent infiltrate or atelectasis in 19% of patients. 62% patients had not returned to full health, while 47% met the case definition for fatigue |
| Varghese et al. (103) | Retrospective | •Münster, Germany •June–September 2020 |
•116 patients •17 females •99 males •Mean age: 41 |
NR | 10 patients | No | Cough, anosmia, fatigue, fever, myalgia, headache | Laboratory measurements, attending physicians document symptoms | 22–102 days | •At 3 months of follow-up persisting symptoms were fatigue (54%), dyspnea (29%), and anosmia (25%), lymphopenia (12%) •Lymphopenia in the later follow-up range of 80–102 days |
| Ya-Wen An et al. (104) | Cross-sectional | •Guangdong, China •February–May 2020 |
•46 patients: •20 females •26 males •Mean age: 46.8 ± 15.3 |
•36 non-severe •10 severe |
Yes | Yes | Fever, weak blocked or watery nose haryngeal symptoms muscle or joint pain chest distress dizziness or headache gastrointestinal symptom | Blood routine, blood biochemistry, urine routine, stool routine, and chest CT scans | 60 days | Extremely low outlier ratio of total protein, albumin, and globulin |
Inclusion and Exclusion Criteria
Papers of any design evaluating individual persistent symptoms in mild, moderate, severe, and critical COVID-19 patients that have a follow-up of at least 14 days were included in this review.
Exclusion criteria included: unpublished reports, unspecified date/location of the study or suspicion of duplicate reporting, coronavirus strains other than COVID-19, unreported long-term COVID-19 symptoms, and studies that only hypothesize post-COVID-19 sequelae.
Study Selection and Data Extraction
Possible relevant articles were screened using the title and abstract by one reviewer (FS) and articles that did not meet the inclusion criteria were excluded. After screening the title and abstract, articles were submitted to a public reference manager (Mendeley v.1.17.9) to eliminate duplicates. Subsequently, the remaining full-text articles were examined by two reviewers (FS and FV). Any disagreement was resolved through discussion until a consensus was reached, or with the involvement of a third reviewer (MF).
The following items were extracted from each cohort study, cross-sectional, case-report, cases-series, case-control studies, if available: author, study type, study country, and period, patient characteristics (numbers, gender, age), COVID-19 severity (mild, moderate, severe, and critical), hospitalization, ICU admission, baseline COVID-19 symptoms, method of evaluating long-term COVID-19 symptoms, follow-up, and long-term COVID-19 symptoms.
Risk of Bias Assessment
Two reviewers (FS and FV) independently assessed the methodological quality of cohort, cross-sectional, case-reports, case-control, case-series studies, and reviews. Disagreements regarding the methodological quality of the studies were discussed between the two reviewers. If consensus was not reached, a third reviewer (MF) arbitrated. Cohort and Cross-Sectional Studies were assessed by Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies from the National Heart, Lung, and Blood Institute (NIH) (16). Case-control studies were assessed by the quality assessment criteria of The Quality Assessment Tool for Case-Control Studies from NIH (16). The methodological quality of case-series and case-reports were assessed by the quality assessment tool proposed by Murad et al. (17). Finally, reviews were assessed by the Quality Assessment Tool for Systematic Reviews and Meta-Analyses from NIH (16). No bias evaluation was performed for letters, commentary, editorial, news articles, survey, practice, communications, and medical hypothesis.
Results
Study Selection and Characteristics
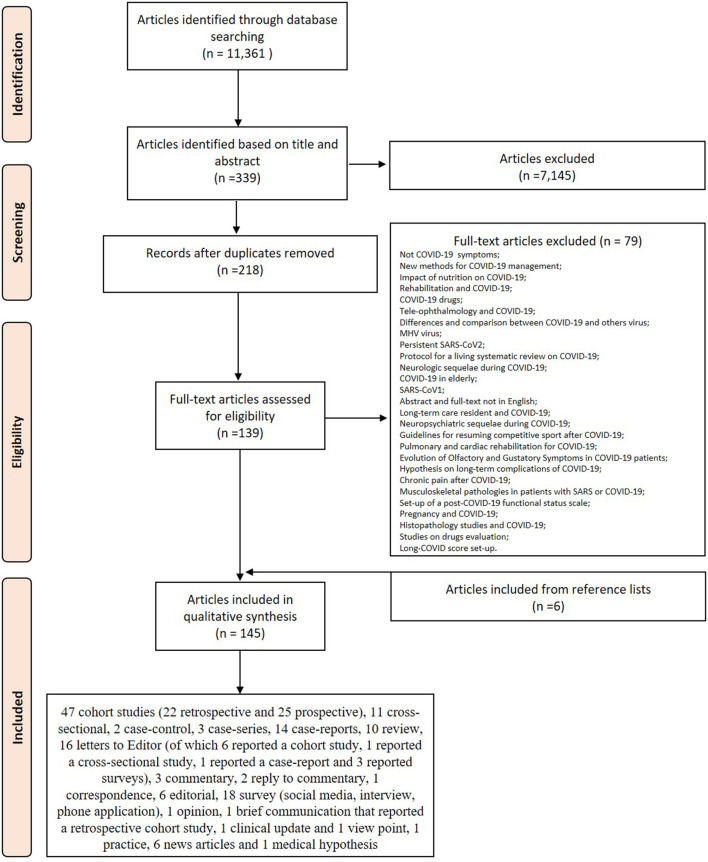
The initial literature search retrieved 11,361 studies. Of those, 3,132 studies were identified using PubMed, 2,776 using Web of Science, 2,073 using EMBASE and 3,380 using Google Scholar. After screening the title and abstract 315 articles were run through Mendeley to eliminate duplicate articles. The resulting 218 full-text articles were then reviewed to establish whether the publication met the inclusion criteria and 139 were considered eligible. From the reference lists of the selected articles 6 additional publications were found. Of the 145 articles eligible for this review 47 were cohort studies (22 retrospective and 25 prospective), 11 cross-sectional, 2 case-control, 3 case-series, 14 case-reports, 10 review, 16 letters to Editor (of which 6 reported a cohort study, 1 reported a cross-sectional study, 1 reported a case-report, and 3 reported surveys), 3 commentary, 2 reply to commentary, 1 correspondence, 6 editorial, 18 survey (social media, interview, phone application), 1 opinion, 1 brief communication that reported a retrospective cohort study, 1 clinical update and 1 view point, 1 practice, 6 news articles, and 1 medical hypothesis. Search strategy and study inclusion and exclusion criteria are detailed in Figure 1.
Figure 1.
PRISMA flowchart for the study selection.
Risk of Bias Assessment
Of the 145 articles eligible for the review, we found 54 cohort studies (28 prospective, 26 retrospective), six of which were published as letters and one as brief communication, and 12 cross-sectional studies, one of which was published as letters. Using the NIH Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies (16), we rated three prospective studies, one ambidirectional cohort study and two cross-sectional studies at a “good” quality rating and 60 studies at a “fair” quality rating (Supplementary Material 1). For the 60 cohort and cross-sectional studies at a “fair” quality rating, the principal missing quality assessment criteria were sample size justification, blinded assessors to the exposure of participants, and missing data on key potential confounding variables measured and adjusted statistically for their impact on the relationship between exposure(s) and outcome(s). Concerning the two case-control studies found, one was at a “good” quality rating and it did not specify only if outcome assessors did not know whether participants were exposed/unexposed, while the other was at a “fair” quality rating (Supplementary Material 1). For the case-control at “fair” quality rating, data on sample size justification, random selection of cases and controls, measures of exposure/risk across all study participants and on blinded assessors of exposure/risk were not reported. The methodological quality of the three case-series and of the 15 case-reports, one of which was published as letters, assessed by the tool proposed by Murad et al. (17), showed that 17/18 studies were at a “good” quality rating (Supplementary Material 1). For the two case-reports rated at a “fair” quality rating, the missing quality assessments criteria were the not adequately ascertained outcome, the lack of alternative causes that may explain the observation, and the absence of sufficient and specific details to describe the case. The quality assessment of reviews showed that 1/10 reviews was at a “good” quality rating while all the others were at a “poor” quality rating (Supplementary Material 1). The “poor” quality rating was because none of them include a comprehensive search of potentially relevant articles and did not use explicit criteria in the selection of articles. The research designs and study characteristics were not appraised, data were not synthesized, and results were not interpreted using a predefined systematic approach.
Long-Term Symptoms of COVID-19
Of the 145 eligible papers, 30 were on persistent lung symptoms (20.70%), 35 were on persistent neurological and olfactory dysfunctions (24.13%), and 80 were on widespread persistent symptoms (55.17%) (Table 1). Table 1 was split-up based on long-term lung symptoms, long-term neurological and olfactory symptoms, and widespread long-term symptoms.
Persistent Lung Symptoms and Dysfunctions
While SARS-CoV-2 was detected in many organ systems, the lungs seem to be the main organs affected by the virus (105–107). Abnormal lung functions and structural changes were reported up to 6 months after hospitalization in mild-to-critical COVID-19 patients (25, 27, 28, 32, 34, 36, 39, 41), also with diffuse alveolar damage, desquamation of alveolar epithelial type II cells, fibrine exudation, hyaline membranes, scattered interstitial inflammation, monocytes, and macrophages (23, 24, 34). Several authors reported that these persistent lung symptoms and dysfunctions correlated with prior COVID-19 severity (19, 20, 24, 25, 27, 36, 38, 41). In this context, Han et al. in a prospective study evaluating 114 severe COVID-19 patients showed lung fibrotic-like changes in 35% patients up to 6 months after infection (24). Differently, Latronico et al. showed that since residual abnormal chest-X ray findings were detected in about 70% of critically ill COVID-19 patients at 3 months, very few of them (~12%) had persisting respiratory symptoms at 6 months (27). An anecdotal
study by Zhu et al. also reported long-term abnormal airway function for up to 11 months in a severe COVID-19 patient (44). However, because is a single case, this research does not provide conclusive evidence. A small cohort of critically ill COVID-19 patients also showed alteration in the diffusing capacity of the lung for carbon monoxide (DLCO) for up to 3 months (20, 27, 38, 40). Persistent DLCO impairment was also detected in non-critical COVID-19 patients that also presented shortness of breath and dyspnea up to 4 months after infection (18, 22, 33, 43). Unlike the above cited studies which analyzed exclusively critically ill patients and two case-reports that analyzed solely mild COVID-19 patients (30, 42) all other studies analyzed heterogeneous cohorts of patients, i.e., from mild to severe. Lower lung functions were detected in 246 mild-to-severe SARS-CoV-2 convalescents patients with few comorbidities up to 2 months after infection (37). Widespread lung damages in mild-to-severe COVID-19 patients were further confirmed by numerous papers and by an Editorial where it was underlined that “months after infection with SARS-CoV-2, some people are still battling lung damage” (108–110), with more than one-third of them that having pulmonary tissue death and visible scars up to 6 months after symptoms onset (18, 21, 26, 35, 109, 110). In a news feature article it was reported that these lung damages lessened with time, 88% of patients had visible damage up to 6 weeks after infection, but 2 months after symptom onset this number had fallen to 56% (109). By examining retrospectively a cohort of 158 mild-to-severe COVID-19 patients, it was shown that these persistent pulmonary damages were also associated with a persistent elevation of IL-6 up to 2 months after infection (29). At the same follow-up, Chun et al., evaluating 61 prevalently non-critical COVID-19 patients, highlighted also higher levels of Lipocalin 2, suggesting that COVID-19 patients may have an ongoing neutrophil activation that could be amenable to targeted therapy (19). Sonnweber et al., evaluating a cohort of 109 patients with mild-to-critical COVID-19, showed that severe lung pathologies were also significantly associated with persisting hyperferritinemia that was present in ~38% of patients (35). Other authors evaluated the lung abnormalities by CT scans at different stages of SARS-CoV-2 infection (21, 26). Ding et al., analyzing retrospectively a cohort of 112 COVID-19 patients at different stages of the disease, showed that the frequency of crazy-paving pattern, consolidation, and linear opacities peaked at 10–14 days (62.7%), 15–21 days (75.0%), and at 22–28 days (83.1%) and decreased thereafter (21). However, at more than 28 days of follow-up 98.1% of CT scans still showed abnormalities. Similarly, Hu et al., evaluating 46 patients with mild-to-severe COVID-19 who had an isolated pulmonary lesion on the first positive CT, highlighted the presence of reticular patterns from the 14 days after symptoms onset in 45% of patients. At 22–31 days, the lesions were completely absorbed only in 28.57% (26). Mo et al. also noted pulmonary anomalies in a cohort of 110 discharged COVID-19 cases, 24 mild cases, 67 cases of pneumonia and 19 cases of severe pneumonia (31). The duration from onset of disease to pulmonary function test was 20 ± 6 days in mild cases, 29 ± 8 days in pneumonia cases and 34 ± 7 days in cases with severe pneumonia (110). Anomalies were noted in DLCO (47.2%), total lung capacity (25.0%), forced expiratory volume in 1 s (FEV1) (13.6%), forced vital capacity (FVC) (9.1%), FEV1/FVC (4.5%), and small airway function (7.3%) (31).
Persistent Neurological Symptoms and Olfactory Dysfunctions
Despite SARS-CoV-2 primarily affecting lungs, numerous data supported the neuro-invading potential of SARS-CoV-2 and, according to the first-hand evidence by Mao et al., ~36.4% of COVID-19 patients presented neurological symptoms (5, 111). Additionally, conditions such as hypoxia, encephalitis, and stroke, all present in severe COVID-19 patients, can produce both long-term neurological symptoms and permanent neurocognitive impairment (52, 58, 112, 113). In fact, a case-series by Negrini et al. associated the long-term neurological symptoms and general cognitive decay to the length of stay in the ICU (58). Despite the long-term neurological symptoms and the general cognitive decay being associated to severe/critical COVID-19 patients, in this review we did not find any studies based solely on critically/severely ill patients. On the other hand, we found a retrospective study and several case reports on mild COVID-19 patients (51, 53, 54, 65). Gallus et al., evaluating retrospectively 48 mild COVID-19 patients, underlined that 8.3% patients reported hearing loss, 4.2% tinnitus, 8.3% dizziness, 2% spinning vertigo, 2% dynamic imbalance, and 6.3% static imbalance at about 1 month of follow-up (51). Several anecdotal reports in mild COVID-19 patients also detected persistent deficits in memory and psychotic symptoms during up to 5 months of follow-up (53, 54, 65). In addition to these studies, all the others found in this review analyzed heterogenic populations of patients with COVID-19, from mild to severe. In this context, a recent editorial and a systematic review provided a detailed overview into the spectrum of mental disorders that can occur during the intermediate and long-term phases of COVID-19 in mild-to-critical patients (114, 115). The most frequent neurological long-term symptoms in these patients were myalgia, arthralgia, sleeping troubles, and headache (46, 50, 61, 116). Additionally, a general cognitive decay, i.e., deficit in attention and calculation, short-term memory, constructional apraxia, and written language, was also observed during up to 6 months of follow-up (61). At 2 months of follow-up 58.7% of 179 mild-to-severe COVID-19 patients presented a moderate neurocognitive decline while 39.1% of patients also showed psychiatric morbidity (56). At a longer follow-up of 6 months, Pilotto et al., analyzing retrospectively 165 moderate-to-severe COVID-19 patients, showed that these long-term symptoms persisted in about 37% of patients (61). Also, symptoms consistent with orthostatic hypoperfusion syndrome and painful small fiber neuropathy were reported at short (3 weeks) and long (up to 3 months) follow-ups in two case-reports and in a small case-series (48, 59, 63). In a “Long-Haul COVID” communication, Nath et al., summarizing symptoms reported after mild-to-severe COVID-19, also highlighted persistent symptoms that overlapped with those patients with myalgic encephalomyelitis/chronic fatigue syndrome (117). In addition to the long-term neurological symptoms Lu et al. prospectively examined the presence of brain micro-structural changes in 60 mild-to-critical COVID-19 patients reporting presence of alterations in 50% of recovered patients after 3 months (55). Anecdotal evidence also showed the presence of long-term impairment of the brain structures in two COVID-19 patients highlighting hypometabolism of the olfactory/rectus gyrus in both patients (52).
Since SARS-CoV-2 can affect neuronal cells by both direct and indirect mechanisms, this can lead to various neurological manifestations also including anosmia and hypogeusia. Anosmia and hypogeusia are present both in mild/moderate cases and in severe cases of COVID-19 (45, 47, 49, 57, 60, 62, 64, 66–68, 118–122). As long-term COVID-19 symptoms, anosmia, and hypogeusia were evaluated in severe COVID-19 patients only in one protective study (67). The study evaluated 138 COVID-19 patients at 2 months of follow-up showing that 5.8% of patients had moderate to severe olfactory dysfunction, while 4.3% had a significant taste disorder (67). A greater number of studies evaluated olfactory and gustatory disfunctions in mild COVID-19 patients (45, 47, 49, 64, 66, 68, 118). Using a retrospective questionnaire Fjaeldstad evaluated olfactory and gustatory loss in 109 mild COVID-19 patients (49). At ~1 month after symptoms onset since the chemosensory loss, participants reported relatively low recovery and improvement rates. For participants with olfactory loss, only 44% were fully recovered, whereas 28% had not yet experienced any improvement of symptoms (49). After gustatory loss, 50% had fully recovered, whereas 20% had not yet experienced any improvement. At a longer follow-up of 2 months after symptoms onset, Otte et al. evaluating through a questionnaire 91 mild COVID-19 patients for olfactory function, showed that 45.1% of patients were hyposmic while 53.8% showed an olfactory performance within the normal range (118). In the same way, at 2 months of follow-up, Boscolo-Rizzo et al. evaluated prospectively 183 mildly symptomatic COVID-19 patients showing that 18.6% presented altered sense of smell or taste (45). Interestingly, Ugurlu et al. in a cohort of mild COVID-19 patients showed persistent olfactory dysfunction in 14.3% of patients up to 3 months after symptoms onset (66). At the same follow-up, a long-term anosmia was also reported in a case-report of a 40-year-old woman with a mild COVID-19 diagnosis (64). Differently, other studies analyzing mild and asymptomatic COVID-19 patients for smell and taste disturbance reported resolution of anosmia up to 1 month after diagnosis (47, 60). Comparable results were also reported at the same follow-up by Konstantinidis et al. evaluating mild/moderate COVID-19 patients (119). Finally, Paolo et al., analyzing 75 mild-to moderate COVID-19 patients through a questionnaire reported olfactory and dysgeusia recovery within an average of 17 days, also finding a significantly decrease in viral load (120).
Finally, other studies evaluating heterogenous populations of mild-to-severe COVID-19 patients further confirmed persistent loss of smell up to 6 months after symptom onset (57, 61, 62, 64, 121, 122). Moein et al. in a prospective study on 82 mild-to-severe COVID-19 patients showed smell loss in ~37% of patients at 2 months of follow-up (57). At the same follow-up, a prospective study on 138 patients and a retrospective study on 90 mild-to-severe patients showed persistent hyposmia in 5–8% of patients (57, 67). Lastly, Pilotto et al., by examining retrospectively 165 patients detected the presence of hyposmia in ~15% of patients at up to 6 months of follow-up (61).
Widespread Persistent Symptoms
Numerous research groups reported widespread persisting symptoms in COVID-19 patients for up to 6 months after SARS-CoV-2 infection (70, 75, 123–142). They also described practice on the management of post-acute COVID-19 and performed comprehensive analyses of health-related quality of life (70, 75, 123). Furthermore, numerous editorials, reviews, news articles, clinical updates, narrative interviews, and focus groups have been published to explore what it is like to live with long-term COVID-19, also trying to emphasize the putative pathophysiology, risk factors, and treatments (124–142). Two cohort studies on severe/critical COVID-19 patients reported persistent physiological impairment and decrease in quality of life in more than half of the patients at up to 6 months of follow up (77, 100). Taboada et al. showed that at 6 months of follow-up only 16% of patients were completely free of persistent symptoms (100). However, in a Multistate Health Care Systems Network, Tenforde et al. reported that among 270 interviews conducted on COVID-19 patients, also among persons with milder outpatient illness, 14–21 days after symptoms onset, the 35% of patients had not returned to their usual state of health (143). In this context, Pellaud et al., examining the outcomes of 196 consecutively mild-moderate COVID-19 patients, 1 month after onset of symptoms, showed that among the 60% of patients that returned home, 63% reported persistent symptoms (90). Two months after symptom onset, evaluating 150 mild/moderate COVID-19 patients, Carvalho-Schneider et al. showed that about 66% of patients presented at least one symptom (74). Similarly, evaluating the long-term COVID-19 symptoms in 233 mild COVID-19 patients Cirulli et al. highlighted that ~24% of patients had at least one symptom also after 3 months (76). These results were also confirmed by an online survey of doctors conducted by the British Medical Association (144). They reported that of 3,729 doctors who answered a question about patients' persistent symptoms after COVID-19, a third said that they had seen or treated patients with long-term COVID-19 symptoms (144). Davido et al. also reported that since mid-May they evaluated an average of 30 individuals per week for whom COVID-19 symptoms have not completely subsided, essentially young women (sex ratio 4:1) around 40 years old with no relevant medical history (145–147). Additionally, it was reported that female sex (mean age 47.22) is also a risk factor for poor health-related quality of life in Chinese COVID-19 patients (75). Also, Sudre et al. analyzing 4,182 incident cases of non-severe COVID-19 who logged their symptoms prospectively in the COVID-19 Symptom Study App showed that women aged 50–60 were at greatest risk of developing “long-COVID” (148). Patients described symptoms in every part of the body which were sometimes severe or fluctuating (149, 150). Paul Garner, a professor at Liverpool School of Tropical Medicine and Co-ordinating Editor of the Cochrane Infectious Diseases Group, wrote on the 95th day after symptoms onset in the British Medical Journal Opinion (151). He said “I am unable to be out of bed for more than three hours at a stretch…I have ringing in the ears, intermittent brain fog, palpitations, and dramatic mood swings” (151). Other people also described similar complaints in the same journal (152–154). The science journalist Linda Geddes also discussed data from the Irish Centre for Vascular Biology in Dublin that reported COVID-19 patients being discharged from hospital, only to return several weeks later not only with widespread symptoms but also with deep vein thrombosis or blood clots on the lungs (155).
The main widespread reported long-term symptoms in COVID-19 patients were chronic fatigue, dyspnea, shortness of breath, chest pains, headache, loss of smell/taste, muscle, and joint pain, followed by depression, anxiety, insomnia, and itchy body, heart palpitations, tachycardia, anorexia, tingling fingertips, and brain fog (69, 70, 72, 77, 84, 85, 87, 91, 97, 98, 101, 103, 123, 138, 145–147, 150, 156–159). However, it was reported that the number of widespread long-term symptoms were higher for COVID-19 patients who were initially more ill (77, 100). D'Cruz et al. and Taboada et al., analyzing prospectively two cohorts of 119 and 91 severe/critical COVID-19 patients, respectively, showed the presence of dyspnoea on exertion (57%), asthaenia (37%), myalgia (37%), and arthralgia (29%) up to 2 months after symptoms onset and a general decrease in quality of life (mobility, usual activities, self-care, pain/discomfort, anxiety/depression) in 67% of patients at up to 6 months of follow-up (77, 100). However, these widespread long-term symptoms were not only present in severe COVID-19 patients, but also in patients who had mild and moderate disease (72, 76, 80, 94, 98, 146, 159). Carvalho-Schneider et al., in a prospective study on 150 mild/moderate COVID-19 patients at 2 months of follow-up, highlighted dyspnea and asthenia, respectively, in 30 and 40% of patients (74, 98). Similar results were also obtained in a cross-sectional study on 451 mild COVID-19 patients (98). In addition to these symptoms in a prospective study by Cirulli et al. symptoms such as difficulty concentrating, fatigue, memory loss, confusion, headache, heart palpitations, chest pain, pain with deep breaths, dizziness, and tachycardia were detected at 3 months of follow up (76). Fatigue, dyspnea, and heart dysfunctions in mild COVID-19 patients were also reported in several case-reports (69, 72, 80) at up to 8 months of follow-up. In addition, a case-report on three women also reported telogen effluvium, temporary hair shedding, as a long-term COVID-19 symptom 3 months after getting the infection (94). Several studies analyzing all together mild-to-severe COVID-19 patients also confirmed these long-term widespread symptoms (73, 78–81, 83, 88, 89, 92, 93, 95, 99, 102–104, 160–162). In a large cohort of 355 mild-to-severe COVID-19 patients Mahmud et al. detected that about 46% of patients developed long-term symptoms at 1 month of follow-up and that post-COVID features were significantly higher among the female gender with fatigue as the main long-term symptom (88). Similarly, persistent fatigue was also reported in about 12% of patients by examining a cohort of 1,002 mild-to-severe COVID-19 patients (95). At a longer follow-up, Rosales-Castillo et al. and Townsend et al. confirmed persistence of fatigue as the main long-term symptom in a cohort of mild-to-severe COVID-19 patients (93, 102). Banda et al., analyzing 150 tweets from moderate-to-severe COVID-19 patients, reported that the 10 most commonly long-term symptoms after COVID-19 were chronic fatigue (62%), dyspnea (19%), tachycardia/palpitations (13%), chest pain (13%), sleep disorders (10%), cough (9%), headache (7%), and joint pain, fever, and unspecified pain by 6% each (160). This analysis also matches clinician-collected data reported by an Italian study (73). The study followed 143 hospitalized mild-to-severe patients who were discharged from the hospital after COVID-19 and that had two negative test results for SARS-CoV-2 (73). At an average of 2 months after initial onset of symptoms, “only 12.6% were completely free of any COVID-19-related symptom, while 32% had 1 or 2 symptoms and 55% had 3 or more” (73). Also, in this case the most common symptoms were chronic fatigue (53.1%), dyspnea (43.4%), joint pain (27.3%), and chest pain (21.7%) (73). Authors also observed that individuals who had an initial symptom of dyspnea were more likely to develop long-term symptoms (73). These results were also confirmed by a case-control study that examined 141 mild-to-moderate COVID-19 patients and 78 controls at 2 months of follow-up (79). At the same follow-up a retrospective study on 206 mild-to-moderate COVID-19 patients also detected chronic pain in ~40% of the patients (78). A particular cross-sectional study on 46 mild-to-severe COVID-19 patients also described an extremely low outlier ratio of total protein, albumin, and globulin at 2 months of follow-up, underlying a persistent abnormal liver function (104). At the same follow-up lymphopenia, elevated D-dimer, and C reactive protein were also detected and associated to persistent fatigue, dyspnea, and anosmia (87, 103). Fatigue and dyspnea were also the two most prevalent persistent symptoms 3 months after a SARS-CoV-2 infection in hospitalized and non-hospitalized patients (83, 92, 103, 161). Furthermore, at the same follow-up, Raman et al. also reported abnormalities in heart (26%), liver (10%), and kidneys (29%) (92). Dyspnea (42%), associated with chronic fatigue (55%), loss of memory (34%), concentration and sleep disorders (28 and 30.8%, respectively), was likewise reported in 120 COVID-19 patients (relatively non-severe) analyzed by questionnaire, 100 days after initial symptoms onset (162). In was also shown that these long-term symptoms persisted for up to 6 months, with fatigue or muscle weakness and sleep difficulties as the most common symptoms (81, 99). At 6 months, by examining 9,989 mild-to-severe COVID-19 patients, persistent neuropsychiatric, pulmonary, metabolic, and coagulopathic phenotypes were also reported (89).
Recent data reported several of these widespread long-term symptoms, i.e., fatigue, dyspnea, chest pains, muscle and joint pain, headache, insomnia, and palpitations, also in children and adolescent up to 6–8 months of follow-up (71, 86, 96). At 6 months of follow-up high blood pressure levels and persistence of a prehypertension were also detected in ~13% of mild COVID-19 children (82). Examining a larger cohort of children, it was also described that ~53–70% of these patients had at least one symptom 100 days or more after COVID-19 diagnosis (71, 82, 96, 163). Given these emerging data, recently, Hertting et al. in an editorial on Acta Paediatrica underlined the need to have more research and studies on the long-term effects of COVID-19 in children and adolescents (164).
Discussion
Although we are aware that there are no long-term data on large numbers of COVID-19 patients with persistent symptoms and with comparison groups, and that an analysis in a field as engaging as COVID-19 can never be updated, this review allowed us to outline that a noteworthy number of patients present long-lasting sequelae, up to 6 months, in the post-COVID time. These long-term symptoms are not only present in severe COVID-19, but also in mild and moderate patients. In addition, recent preliminary data also underlined the presence of long-term COVID-19 symptoms on children and adolescents. Some clinical studies and survey questionnaires also highlighted a potential high-risk factor for long-term COVID-19 in the female gender; women's risk of developing long-term COVID-19 seems to be double that of men among patients aged between 40 and 50. After the age of 60 the risk level of long-term COVID between male and female should become similar. This pattern appears to be like that of autoimmune diseases that are more common in female through menopause to become similar between male and female after age 60 (165). Thus, it is possible that these gender differences, as well as other aspects of the disease, may be due to a different immune system response during and after COVID-19. However, currently, it is not yet clear whether this data reflects the population of people with long-term COVID-19 and which is the full spectrum of the duration and severity of long-term symptoms in these patients.
What emerges from this review is that the most common reported symptoms after COVID-19 are abnormal lung functions prevalently with persistent dyspnea, general neurological decay, smell and taste disturbances, and chronic fatigue. Other common symptoms include joint pain and chest pain. These symptoms may linger or recur for weeks or months following initial recovery. In detail, for patients with mild-to-moderate COVID-19 the more common long-term symptoms are chronic fatigue, anosmia/ageusia, dyspnea, but also difficulty in concentration, memory loss, and confusion. These symptoms seem to be present in a higher percentage of patients who were initially more ill. In critical-to-severe COVID-19 patients' supplementary long-term symptoms are lung fibrotic-like changes up to 6 months after infection and a high reduction in diffusing capacity of the lung for DLCO that frequently required oxygen uses also after hospital discharge. Likewise, the general cognitive decay, despite also being present in mild-to-moderate COVID-19 patients, also appears to be more closely related to critical-to-severe forms of COVID-19. Considering the whole overview of widespread long-term symptoms reported in this review the one undeniably most prevalent in mild-to-critical COVID-19 patients is chronic fatigue. This is in line with past research that highlighted high levels of post-infectious fatigue for survivors of epidemics such as SARS and Ebolavirus (166, 167). Moreover, fatigue has been related with infections, such as mononucleosis, that occur outside of an epidemic or pandemic scale (167). Currently, it is not clear why chronic fatigue and the other long-term complications persist in some COVID-19 patients. However, most researchers and clinicians agree that the long-term COVID-19 symptoms are associated with the coronavirus' ability to trigger a massive inflammatory response. Thus, it will be mandatory to analyze cytokine networks in patients who recover from COVID-19 to evaluate whether the “cytokine storm” present during the disease persists and contributes to these long-term complications.
The main strength of this study is that it highlights multiple long-term symptoms which may hinder return to pre-COVID-19 infection functional status. However, despite this finding a weakness of our review was that while some studies included in this review focused on a single population of infected COVID-19 patients, i.e., mild/moderate and severe/critical, numerous studies included heterogeneous populations, from mild to critical, not taking into account disease severity as well as preexisting co-morbidities, treatment regimens, mean ages, gender, and other aspects. This bias can lead to alterations in the data evaluation and analysis, which potentially affect the results. Data from prospective designs, developed by evaluating homogeneous populations of COVID-19 patients able to consider their characteristics prior to and during infection, might provide new and detailed information into predisposing factors that lead to long-term COVID-19 symptoms. Another bias that should be considered is that despite the fact that in some studies the long-term COVID-19 sequelae were evaluated through clinical visits and/or specific instrumental analyzes, many others have used self-administered questionnaires and scores, telephone/online interview, and phone applications. This is because, to date, assessing the patient in the hospital is difficult due to the entry restrictions into the COVID-19 departments. On the other hand, checking and evaluating them at home presents almost insurmountable logistical problems during an emergency health situation like the one we are facing. However, this type of self-assessment highlights bias in the detection of symptoms as patients may have psychological and emotional involvement due to the disease itself.
At this stage, a detailed analysis and understanding of all the aspects associated with long-term COVID-19 are mandatory to mitigate against the potential persistent symptoms identified in the current review. Future studies should assess: (1) the full range of disorders associated with COVID-19 and their long-term manifestations; (2) the underlying associations between viral spread, associated pro-inflammatory changes, and long-term disease pathogenesis; (3) the duration and extent of long-term symptoms in relation to the resolution of the disease; (4) the association between disease severity and long-term dysfunctions; (5) the effect of specific antiviral therapies and/or interventions on long-term symptoms; (6) why symptoms persist or recur; and (7) the potential late effects of COVID-19 on children/adolescents. Another important point that should be assessed is SARS-CoV-2 levels (detection, load) in patients and how this relates to long-term symptoms. To date, it is not clear whether the initial viral load, per se, may meaningfully impact long-term symptoms, particularly in mild-to-moderate COVID-19 patients. Information relating to SARS-CoV-2 detection and viral load at different time points of infection will help the clinical interpretation of long-term symptoms of COVID-19. Similarly, there is a need for further studies to provide robust data on the association between viral shedding and long-term COVID-19. Despite has reports that the median duration of viral shedding goes from 12-to-20 days, there is evidence that ongoing viral shedding in SARS-CoV-2 may be prolonged in the feces compared to respiratory secretions (168–170). The persistent fragments of viral genes, though not infectious, may still be triggering a violent immune overreaction that could explain the symptoms persistence in COVID-19-free patients. Alternatively, even if the virus is cleared, the immune system could continue to be overactive or perturbed, analogous to the long-term debilitation after glandular fever (165). A greater understanding of these last points could improve the knowledge not only of the causes of long-term symptoms but also on the immune system involvement and on transmission risk.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author/s.
Author Contributions
FS and MF designed the review. FS and FV performed the literature search and collected and assembled the data. FS, FV, and MF analyzed the obtained articles. FS, FV, ML, and MF wrote the paper. MF, LM, and ML revised the manuscript critically. All authors have read and agreed to the published version of the manuscript.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Footnotes
Funding. This work was supported by IRCCS Istituto Ortopedico Rizzoli (Ricerca Corrente).
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2021.653516/full#supplementary-material
References
- 1.COVID-19 Dashboard by the Center for Systems Science Engineering (CSSE) at Johns Hopkins University (JHU). Available online at: https://coronavirus.jhu.edu/map.html (accessed December 17, 2020).
- 2.Mao R, Qiu Y, He JS, Tan JY, Li XH, Liang J, et al. Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. (2020) 5:667–78. 10.1016/S2468-1253(20)30126-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Levi M, Thachil J, Iba T, Levy JH. Coagulation abnormalities and thrombosis in patients with COVID-19. Lancet Haematol. (2020) 7:438–40. 10.1016/S2352-3026(20)30145-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Long B, Brady WJ, Koyfman A, Gottlieb M. Cardiovascular complications in COVID-19. Am J Emerg Med. (2020) 38:1504–7. 10.1016/j.ajem.2020.04.048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mao L, Jin H, Wang M, Hu Y, Chen S, He Q, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. (2020) 77:1–9. 10.1001/jamaneurol.2020.1127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Middeldorp S, Coppens M, van Haaps TF, Foppen M, Vlaar AP, Müller MCA, et al. Incidence of venous thromboembolism in hospitalized patients with COVID-19. J Thromb Haemost. (2020) 18:1995–2002. 10.1111/jth.14888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chen YT, Shao SC, Hsu CK, Wu IW, Hung MJ, Chen YC. Incidence of acute kidney injury in COVID-19 infection: a systematic review and meta-analysis. Crit Care. (2020) 24:346. 10.1186/s13054-020-03009-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.COVID Symptom Study . How Long Does COVID-19 Last? Available online at: https://covid19.joinzoe.com/post/covid-long-term?fbclid=IwAR1RxIcmmdL-EFjh_aI- (accessed December 17, 2020).
- 9.Correia AO, Feitosa PWG, Moreira JLS, Nogueira SÁR, Fonseca RB, Nobre MEP. Neurological manifestations of COVID-19 and other coronaviruses: a systematic review. Neurol Psychiatry Brain Res. (2020) 37:27–32. 10.1016/j.npbr.2020.05.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.NICE . Covid-19 Rapid Guideline: Managing the Long-Term Effects of Covid-19. Available online at: https://www.nice.org.uk/guidance/ng188/chapter/4-Planning-care (accessed February 15, 2021).
- 11.Hui DS, Joynt GM, Wong KT, Gomersall CD, Li TS, Antonio G, et al. Impact of severe acute respiratory syndrome (SARS) on pulmonary function, functional capacity and quality of life in a cohort of survivors. Thorax. (2005) 60:401–9. 10.1136/thx.2004.030205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hui DS, Wong KT, Ko FW, Tam LS, Chan DP, Woo J, et al. The 1-year impact of severe acute respiratory syndrome on pulmonary function, exercise capacity, and quality of life in a cohort of survivors. Chest. (2005) 128:2247–61. 10.1378/chest.128.4.2247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ngai JC, Ko FW, Ng SS, To KW, Tong M, Hui DS. The long-term impact of severe acute respiratory syndrome on pulmonary function, exercise capacity and health status. Respirology. (2010) 15:543–50. 10.1111/j.1440-1843.2010.01720.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Park WB, Jun KI, Kim G, Choi JP, Rhee JY, Cheon S, et al. Correlation between pneumonia severity and pulmonary complications in Middle East respiratory syndrome. J Korean Med Sci. (2018) 33:169. 10.3346/jkms.2018.33.e169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. (2009). 6:1000097. 10.1371/journal.pmed.1000097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Study Quality Assessment Tools. Available online at: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed December 17, 2020).
- 17.Murad MH, Sultan S, Haffar S, Bazerbachi F. Methodological quality and synthesis of case series and case reports. BMJ Evid Based Med. (2018) 23:60–3. 10.1136/bmjebm-2017-110853 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bellan M, Soddu D, Balbo PE, Baricich A, Zeppegno P, Avanzi GC, et al. Respiratory and psychophysical sequelae among patients with COVID-19 four months after hospital discharge. JAMA Netw Open. (2021) 4:e2036142. 10.1001/jamanetworkopen.2020.36142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chun HJ, Coutavas E, Pine A, Lee AI, Yu V, Shallow M, et al. Immuno-fibrotic drivers of impaired lung function in post-COVID-19 syndrome. medRxiv [Preprint]. (2021). 10.1101/2021.01.31.21250870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Daher A, Balfanz P, Cornelissen C, Müller A, Bergs I, Marx N, et al. Follow up of patients with severe coronavirus disease 2019 (COVID-19): pulmonary and extrapulmonary disease sequelae. Respir Med. (2020). 174:106197. 10.1016/j.rmed.2020.106197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ding X, Xu J, Zhou J, Long Q. Chest CT findings of COVID-19 pneumonia by duration of symptoms. Eur J Radiol. (2020) 127:109009. 10.1016/j.ejrad.2020.109009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Frija-Masson J, Debray MP, Gilbert M, Lescure FX, Travert F, Borie R, et al. Functional characteristics of patients with SARS-CoV-2 pneumonia at 30 days post-infection. Eur Respir J. (2020) 56:2001754. 10.1183/13993003.01754-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hall J, Myall K, Lam JL, Mason T, Mukherjee B, West A, et al. Identifying patients at risk of post-discharge complications related to COVID-19 infection. Thorax. (2021). 10.1136/thoraxjnl-2020-215861. [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 24.Han X, Fan Y, Alwalid O, Li N, Jia X, Yuan M, et al. Six-month follow-up chest CT findings after severe COVID-19 pneumonia. Radiology. (2021) 299:203153. 10.1148/radiol.2021203153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Heiss R, Grodzki DM, Horger W, Uder M, Nagel AM, Bickelhaupt S. High-performance low field MRI enables visualization of persistent pulmonary damage after COVID-19. Magn Reson Imaging. (2020) 76:49–51. 10.1016/j.mri.2020.11.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hu Q, Guan H, Sun Z, Huang L, Chen C, Ai T, et al. Early CT features and temporal lung changes in COVID-19 pneumonia in Wuhan, China. Eur J Radiol. (2020) 128:109017. 10.1016/j.ejrad.2020.109017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Latronico N, Peli E, Rodella F, Novelli MP, Rasulo FA, Piva S, et al. Six-Month Outcome in Survivors of COVID-19 Associated Acute Respiratory Distress Syndrome. SSRN. Available online at: https://ssrn.com/abstract=3756865 (accessed March, 2021).
- 28.Liang L, Yang B, Jiang N, Fu W, He X, Zhou Y, et al. Three-month follow-up study of survivors of coronavirus disease 2019 after discharge. J Korean Med Sci. (2019) 35:e418. 10.3346/jkms.2020.35.e418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liao B, Liu Z, Tang L, Li L, Gan Q, Shi H, et al. Longitudinal clinical and radiographic evaluation reveals interleukin-6 as an indicator of persistent pulmonary injury in COVID-19. Int J Med Sci. (2021) 18:29–41. 10.7150/ijms.49728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Manckoundia P, Franon E. Is persistent thick copious mucus a long-term symptom of COVID-19? Eur J Case Rep Intern Med. (2020) 7:002145. 10.12890/2020_002145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mo X, Jian W, Su Z, Chen M, Peng H, Peng P, et al. Abnormal pulmonary function in COVID-19 patients at time of hospital discharge. Eur Respir J. (2020) 55:2001217. 10.1183/13993003.01217-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Moreno-Pérez O, Merino E, Leon-Ramirez JM, Andres M, Ramos JM, Arenas-Jiménez J, et al. Post-acute COVID-19 syndrome. Incidence and risk factors: a Mediterranean cohort study. J Infect. (2021) 82:378–83. 10.1016/j.jinf.2021.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ramakrishnan A, Zreloff J, Moore MA, Bergquist SH, Cellai M, Higdon J, et al. Prolonged symptoms after COVID-19 infection in outpatients. Open Forum Infect Dis. (2021) 8:ofab060. 10.1093/ofid/ofab060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Shah AS, Wong AW, Hague CJ, Murphy DT, Johnston JC, Ryerson CJ, et al. A prospective study of 12-week respiratory outcomes in COVID-19-related hospitalisations. Thorax. (2020) 3. 10.1136/thoraxjnl-2020-216308 [DOI] [PubMed] [Google Scholar]
- 35.Sonnweber T, Boehm A, Sahanic S, Pizzini A, Aichner M, Sonnweber B, et al. Persisting alterations of iron homeostasis in COVID-19 are associated with non-resolving lung pathologies and poor patients' performance: a prospective observational cohort study. Respir Res. (2020) 21:276. 10.1186/s12931-020-01546-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tabatabaei SMH, Rajebi H, Moghaddas F, Ghasemiadl M, Talari H. Chest CT in COVID-19 pneumonia: what are the findings in mid-term follow-up? Emerg Radiol. (2020) 27:711–719. 10.1007/s10140-020-01869-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Trinkmann F, Müller M, Reif A, Kahn N, Kreuter M, Trudzinski F, et al. Residual symptoms and lower lung function in patients recovering from SARS-CoV-2 infection. Eur Respir J. (2021). 57:2003002. 10.1183/13993003.03002-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Truffaut L, Demey L, Bruyneel AV, Roman A, Alard S, De Vos N, et al. Post-discharge critical COVID-19 lung function related to severity of radiologic lung involvement at admission. Respir Res. (2021) 22:29. 10.1186/s12931-021-01625-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.van den Borst B, Peters JB, Brink M, Schoon Y, Bleeker-Rovers CP, Schers H, et al. Comprehensive health assessment three months after recovery from acute COVID-19. Clin Infect Dis. (2020) 21:ciaa1750. 10.1093/cid/ciaa1750 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.van Gassel RJJ, Bels JLM, Raafs A, van Bussel BCT, van de Poll MCG, Simons SO, et al. High prevalence of pulmonary sequelae at 3 months after hospital discharge in mechanically ventilated survivors of COVID-19. Am J Respir Crit Care Med. (2021) 203:371–4. 10.1164/rccm.202010-3823LE [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Weerahandi H, Hochman KA, Simon E, Blaum C, Chodosh J, Duan E, et al. Post-discharge health status and symptoms in patients with severe COVID-19. J Gen Intern Med. (2021) 36:738–45. 10.1007/s11606-020-06338-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yao XH, He ZC, Li TY, Zhang HR, Wang Y, Mou H, et al. Pathological evidence for residual SARS-CoV-2 in pulmonary tissues of a ready-for-discharge patient. Cell Res. (2020) 30:541–3. 10.1038/s41422-020-0318-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhao YM, Shang YM, Song WB, Li QQ, Xie H, Xu QF, et al. Follow-up study of the pulmonary function and related physiological characteristics of COVID-19 survivors three months after recovery. EClinicalMedicine. (2020) 25:100463. 10.1016/j.eclinm.2020.100463 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhu M, Chen D, Zhu Y, Xiong X, Ding Y, Guo F, et al. Long-term sero-positivity for IgG, sequelae of respiratory symptoms, and abundance of malformed sperms in a patient recovered from severe COVID-19. Eur J Clin Microbiol Infect Dis. (2021). 10.1007/s10096-021-04178-6. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Boscolo-Rizzo P, Polesel J, Spinato G, Menegaldo A, Fabbris C, Calvanese L, et al. Predominance of an altered sense of smell or taste among long-lasting symptoms in patients with mildly symptomatic COVID-19. Rhinology. (2020) 58:524–5. 10.4193/Rhin20.263 [DOI] [PubMed] [Google Scholar]
- 46.Caronna E, Ballvé A, Llauradó A, Gallardo VJ, Ariton DM, Lallana S, et al. Headache: a striking prodromal and persistent symptom, predictive of COVID-19 clinical evolution. Cephalalgia. (2020) 40:1410–21 10.1177/0333102420965157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.D'Ascanio L, Pandolfini M, Cingolani C, Latini G, Gradoni P, Capalbo M, et al. Olfactory dysfunction in COVID-19 patients: prevalence and prognosis for recovering sense of smell. Otolaryngol Head Neck Surg. (2020) 164:82–6. 10.1177/0194599820943530 [DOI] [PubMed] [Google Scholar]
- 48.Dani M, Dirksen A, Taraborrelli P, Torocastro M, Panagopoulos D, Sutton R, et al. Autonomic dysfunction in 'long COVID': rationale, physiology and management strategies. Clin Med. (2020) 21:e63–7. 10.7861/clinmed.2020-0896 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Fjaeldstad AW. Prolonged complaints of chemosensory loss after COVID-19. Dan Med J. (2020) 67:A05200340. [PubMed] [Google Scholar]
- 50.Galal I, Hussein AAR, Amin MT, Saad MM, Zayan HEE, Abdelsayed MZ, et al. Determinants of persistent post COVID-19 symptoms: value of a novel COVID-19 symptoms score. Egypt J Bronchol. (2021) 15:10. 10.1101/2020.11.11.20230052 [DOI] [Google Scholar]
- 51.Gallus R, Melis A, Rizzo D, Piras A, De Luca LM, Tramaloni P, et al. Audiovestibular symptoms and sequelae in COVID-19 patients. J Vestib Res. (2021). 10.3233/VES-201505. [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 52.Guedj E, Million M, Dudouet P, Tissot-Dupont H, Bregeon F, Cammilleri S, et al. (18)F-FDG brain PET hypometabolism in post-SARS-CoV-2 infection: substrate for persistent/delayed disorders? Eur J Nucl Med Mol Imaging. (2020). 30:1–4. 10.21203/rs.3.rs-40021/v1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hellmuth J, Barnett TA, Asken BM, Kelly JD, Torres L, Stephens ML, et al. Persistent COVID-19-associated neurocognitive symptoms in non-hospitalized patients. J Neurovirol. (2021) 2:1–5. 10.1007/s13365-021-00954-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Lim ST, Janaway B, Costello H, Trip A, Price G. Persistent psychotic symptoms following COVID-19 infection. BJPsych Open. (2020) 6:105. 10.1192/bjo.2020.76 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lu Y, Li X, Geng D, Wu PY, Huang CC, Jia T, et al. Cerebral micro-structural changes in COVID-19 patients - an MRI-based 3-month follow-up study. EClinicalMedicine. (2020) 25:100484. 10.1016/j.eclinm.2020.100484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Méndez R, Balanzá-Martínez V, Luperdi SC, Estrada I, Latorre A, González-Jiménez P, et al. Short-term neuropsychiatric outcomes and quality of life in COVID-19 survivors. J Intern Med. (2021). 10.1111/joim.13262. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Moein ST, Hashemian SM, Tabarsi P, Doty RL. Prevalence and reversibility of smell dysfunction measured psychophysically in a cohort of COVID-19 patients. Int Forum Allergy Rhinol. (2020) 10:1127–35. 10.1002/alr.22680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Negrini F, Ferrario I, Mazziotti D, Berchicci M, Bonazzi M, de Sire A, et al. Neuropsychological features of severe hospitalized coronavirus disease 2019 Patients at clinical stability and clues for postacute rehabilitation. Arch Phys Med Rehabil. (2020) 102:155–8. 10.1016/j.apmr.2020.09.376 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Novak P. Post COVID-19 syndrome associated with orthostatic cerebral hypoperfusion syndrome, small fiber neuropathy and benefit of immunotherapy: a case report. eNeurologicalSci. (2020) 21:100276. 10.1016/j.ensci.2020.100276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Panda S, Mohamed A, Sikka K, Kanodia A, Sakthivel P, Thakar A, et al. Otolaryngologic manifestation and long-term outcome in mild COVID-19: experience from a Tertiary Care Centre in India. Indian J Otolaryngol Head Neck Surg. (2020) 14:1–6. 10.1007/s12070-020-02217-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Pilotto A, Cristillo V, Cotti Piccinelli S, Zoppi N, Bonzi G, Sattin D, et al. COVID-19 severity impacts on long-term neurological manifestation after hospitalisation. medRxiv [Preprint]. (2020). 10.1101/2020.12.27.20248903 [DOI] [Google Scholar]
- 62.Printza A, Katotomichelakis M, Metallidis S, Panagopoulos P, Sarafidou A, Petrakis V, et al. The clinical course of smell and taste loss in COVID-19 hospitalized patients. Hippokratia. (2020) 24:66–71. [PMC free article] [PubMed] [Google Scholar]
- 63.Raahimi MM, Kane A, Moore CE, Alareed AW. Late onset of Guillain-Barré syndrome following SARS-CoV-2 infection: part of 'long COVID-19 syndrome'? BMJ Case Rep. (2021) 14:e240178. 10.1136/bcr-2020-240178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sampaio Rocha-Filho PA, Voss L. Persistent headache and persistent anosmia associated with COVID-19. Headache. (2020) 60:1797–9. 10.1111/head.13941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Clouden TA. Persistent hallucinations in a 46-year-old woman after COVID-19 infection: a case report. Cureus. (2020) 12:e11993. 10.7759/cureus.11993 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Ugurlu BN, Akdogan O, Yilmaz YA, Yapar D, Aktar Ugurlu G, Yerlikaya HS, et al. Quantitative evaluation and progress of olfactory dysfunction in COVID-19. Eur Arch Otorhinolaryngol. (2021). 10.1007/s00405-020-06516-4. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Vaira LA, Hopkins C, Petrocelli M, Lechien JR, Chiesa-Estomba CM, Salzano G, et al. Smell and taste recovery in coronavirus disease 2019 patients: a 60-day objective and prospective study. J Laryngol Otol. (2020) 134:703–9. 10.1017/S0022215120001826 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Yan CH, Prajapati DP, Ritter ML, DeConde AS. Persistent smell loss following undetectable SARS-CoV-2. Otolaryngol Head Neck Surg. (2020) 163:923–5. 10.1177/0194599820934769 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Abdallah H, Porterfield F, Fajgenbaum D. Symptomatic relapse and long-term sequelae of COVID-19 in a previously healthy 30-year-old man. BMJ Case Rep. (2020) 13:e239825. 10.1136/bcr-2020-239825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Arnold DT, Hamilton FW, Milne A. Patient outcomes after hospitalisation with COVID-19 and implications for follow-up: results from a prospective UK cohort. Thorax. (2020). 10.1101/2020.08.12.20173526. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Buonsenso D, Munblit D, De Rose C, Sinatti D, Ricchiuto A, Carfi A, et al. Preliminary evidence on long COVID in children. medRxiv [Preprint]. (2021). 10.1101/2021.01.23.21250375 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Buselli R, Corsi M, Necciari G, Pistolesi P, Baldanzi S, Chiumiento M, et al. Sudden and persistent dysphonia within the framework of COVID-19: The case report of a nurse. Brain Behav Immun Health. (2020) 9:100160. 10.1016/j.bbih.2020.100160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Carfì A, Bernabei R, Landi F. Gemelli against COVID-19 post-acute care study group. JAMA. (2020) 324:603–5. 10.1001/jama.2020.12603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Carvalho-Schneider C, Laurent E, Lemaignen A, Beaufils E, Bourbao-Tournois C, Laribi S, et al. Follow-up of adults with noncritical COVID-19 two months after symptom onset. Clin Microbiol Infect. (2020) 27:258–63. 10.1016/j.cmi.2020.09.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Chen KY, Li T, Gong FH, Zhang JS, Li XK. Predictors of health-related quality of life and influencing factors for COVID-19 patients, a follow-up at one month. Front Psychiatry. (2020) 11:668. 10.3389/fpsyt.2020.00668 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Cirulli E, Schiabor Barrett KM, Riffle S, Bolze A, Neveux I, Dabe S, et al. Long-term COVID-19 symptoms in a large unselected population. medRxiv [Preprint]. (2020). 10.1101/2020.10.07.20208702 [DOI] [Google Scholar]
- 77.D'Cruz RF, Waller MD, Perrin F, Periselneris J, Norton S, Smith LJ, et al. Chest radiography is a poor predictor of respiratory symptoms and functional impairment in survivors of severe COVID-19 pneumonia. ERJ Open Res. (2021) 7:00655–2020. 10.1183/23120541.00655-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Erçalik T, Ayyildiz A, Gencer-Atalay K, Akgün C, Özdemir HM, Kuran B. Pain symptoms in COVID-19. Am J Phys Med Rehabil. (2021). 10.1097/PHM.0000000000001699. [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 79.Galván-Tejada CE, Herrera-García CF, Godina-González S, Villagrana-Bañuelos KE, Amaro JDL, Herrera-García K, et al. Persistence of COVID-19 symptoms after recovery in Mexican population. Int J Environ Res Public Health. (2020) 17:9367. 10.3390/ijerph17249367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Hosseini Z, Ghodsi S, Hejazi SF. Persistent complete heart block in a patient with COVID-19 infection: a case report. SN Compr Clin Med. (2021) 6:1–4. 10.1007/s42399-020-00712-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Huang C, Huang L, Wang Y, Li X, Ren L, Gu X, et al. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet. (2021). 397:220–32. 10.1016/S0140-6736(20)32656-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Soldi S, Mallardo S, Marcellino A, Bloise S, Dilillo A, Iorfida D, et al. The comprehensive clinic, laboratory, and instrumental evaluation of children with COVID-19: a 6-months prospective study. J Med Virol. (2021) 93:3122–32. 10.1002/jmv.26871 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Iqbal A, Iqbal K, Arshad Ali S, Azim D, Farid E, Baig MD, Bin Arif T, Raza M. The COVID-19 sequelae: a cross-sectional evaluation of post-recovery symptoms and the need for rehabilitation of COVID-19 survivors. Cureus. (2021) 13:e13080. 10.7759/cureus.13080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Jacobs LG, Gourna Paleoudis E, Lesky-Di Bari D, Nyirenda T, Friedman T, Gupta A, et al. Persistence of symptoms and quality of life at 35 days after hospitalization for COVID-19 infection. PLoS ONE. (2020) 15:e0243882. 10.1371/journal.pone.0243882 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Khalaf M, Bazeed SE, Abdel-Gawad M, Abu-Elfatth A, Abdelhamed W, Zaghloul M, et al. Prevalence and Predictors of Persistent Symptoms after Clearance of SARS-CoV-2 Infection: A Report from Egypt. Available online at: https://papers.ssrn.com/sol3/papers.cfm?abstract_id=3727954 (accessed December 3, 2020).
- 86.Ludvigsson JF. Case report and systematic review suggest that children may experience similar long-term effects to adults after clinical COVID-19. Acta Paediatr. (2020) 110:914–21. 10.1111/apa.15673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Mandal S, Barnett J, Brill SE, Brown JS, Denneny EK, Hare SS, et al. ‘Long-COVID': a cross-sectional study of persisting symptoms, biomarker and imaging abnormalities following hospitalisation for COVID-19. Thorax. (2020). 10.1136/thoraxjnl-2020-215818. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Mahmud R, Rahman M, Rassel MA, Monayem FB, Sayeed SJB. Post COVID syndrome among symptomatic COVID-19 patients: a prospective study in a Tertiary Care Center in Bangladesh. SSRN. (2021). 10.2139/ssrn.3759687. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Martin JT, Akama-Garren E, Puranik A, Liukasemsarn S, Venkatakrishnan AJ, O'Horo JC, et al. Augmented curation of clinical notes of COVID-19 and influenza patients reveals that long-term neuropsychiatric and coagulopathic symptoms are more associated with COVID-19. medRxiv [Preprint]. (2021). 10.1101/2021.01.03.20248997 [DOI] [Google Scholar]
- 90.Pellaud C, Grandmaison G, Pham Huu Thien HP, Baumberger M, Carrel G, Ksouri H, et al. Characteristics, comorbidities, 30-day outcome and in-hospital mortality of patients hospitalised with COVID-19 in a Swiss area - a retrospective cohort study. Swiss Med Wkly. (2020) 150:w20314. 10.4414/smw.2020.20314 [DOI] [PubMed] [Google Scholar]
- 91.Petersen MS, Kristiansen MF, Hanusson KD, Danielsen ME, Á Steig B, Gaini S, Strøm M, et al. Long COVID in the Faroe Islands - a longitudinal study among non-hospitalized patients. Clin Infect Dis. (2020). 10.1093/cid/ciaa1792. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Raman B, Cassar MP, Tunnicliffe EM, Filippini N, Griffanti L, Alfaro-Almagro F, et al. Medium-term effects of SARS-CoV-2 infection on multiple vital organs, exercise capacity, cognition, quality of life and mental health, post-hospital discharge. EClinicalMedicine. (2021) 31:100683. 10.1016/j.eclinm.2020.100683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Rosales-Castillo A, García de Los Ríos C, Mediavilla García JD. Persistent symptoms after acute COVID-19 infection: importance of follow-up. Med Clin. (2021) 156:35–36. 10.1016/j.medcle.2020.08.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Saeed W, Hussain I, Altaf F. Telogen effluvium: long term Covid-19 symptom. J Pakistan Assoc Dermatol. (2020) 30:700–3. [Google Scholar]
- 95.Islam MS, Ferdous MZ, Islam US, Mosaddek ASM, Potenza MN, Pardhan S. Treatment, persistent symptoms, and depression in people infected with COVID-19 in Bangladesh. Int J Environ Res Public Health. (2021) 18:1453. 10.3390/ijerph18041453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Smane L, Stars I, Pucuka Z, Roge I, Pavare J. Persistent clinical features in paediatric patients after SARS-CoV-2 virological recovery: a retrospective population-based cohort study from a single centre in Latvia. BMJ Paediatr Open. (2020). 4:e000905. 10.1136/bmjpo-2020-000905 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Sofian M, Velayati AA, Banifazl M, Fotouhi F, Sadat Larijani M, Afzali N, et al. SARS-CoV-2, a virus with many faces: a series of cases with prolonged persistence of COVID-19 symptoms. Wien Med Wochenschr. (2020) 171:3–6. 10.1007/s10354-020-00793-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Stavem K, Ghanima W, Olsen MK, Gilboe HM, Einvik G. Persistent symptoms 1.5-6 months after COVID-19 in non-hospitalised subjects: a population-based cohort study. Thorax. (2020). 10.1136/thoraxjnl-2020-216377. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Sykes DL, Holdsworth L, Jawad N, Gunasekera P, Morice AH, Crooks MG. Post-COVID-19 symptom burden: what is long-COVID and how should we manage it? Lung. (2021) 11:1–7. 10.1007/s00408-021-00423-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Taboada M, Moreno E, Cariñena A, Rey T, Pita-Romero R, Leal S, et al. Quality of life, functional status, and persistent symptoms after intensive care of COVID-19 patients. Br J Anaesth. (2021) 126:e110–3. 10.1016/j.bja.2020.12.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Townsend L, Dyer AH, Jones K, Dunne J, Mooney A, Gaffney F, et al. Persistent fatigue following SARS-CoV-2 infection is common and independent of severity of initial infection. PLoS ONE. (2020) 15:e0240784. 10.1371/journal.pone.0240784 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Townsend L, Dowds J, O'Brien K, Sheill G, Dyer AH, O'Kelly B, et al. Persistent poor health post-COVID-19 is not associated with respiratory complications or initial disease severity. Ann Am Thorac Soc. (2021). 10.1513/AnnalsATS.202009-1175OC. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Varghese J, Sandmann S, Vollenberg R, Ochs K, Schrempf I, Froemmel C, et al. Follow Up of COVID-19 Features in Recovered Adults without Comorbidities- Persistent Symptoms and Lab-Abnormalities. Res Square [Preprint]. 10.21203/rs.3.rs-116030/v1 [DOI] [Google Scholar]
- 104.An YW, Song S, Li WX, Chen YX, Hu XP, Zhao J, et al. Liver function recovery of COVID-19 patients after discharge, a follow-up study. Int J Med Sci. (2021) 18:176–86. 10.7150/ijms.50691 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. (2020). 382:1708-1720. 10.1056/NEJMoa2002032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Yang X, Yu Y, Xu J, Shu H, Xia J, Liu H, et al. Clinical course and outcomes of critically ill patients with SARS1456 CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. (2020) 8:475–81. 10.1016/S2213-2600(20)30079-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. (2020) 8:420–2. 10.1016/S2213-2600(20)30076-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Fraser E. Long term respiratory complications of covid-19. BMJ. (2020) 370:m3001. 10.1136/bmj.m3001 [DOI] [PubMed] [Google Scholar]
- 109.Marshall M. The lasting misery of coronavirus long-haulers. Nature. (2020) 585:339–341. 10.1038/d41586-020-02598-6 [DOI] [PubMed] [Google Scholar]
- 110.Shaw B, Daskareh M, Gholamrezanezhad A. The lingering manifestations of COVID-19 during and after convalescence: update on long-term pulmonary consequences of coronavirus disease 2019 (COVID-19). Radiol Med. (2020) 1:1–7. 10.1007/s11547-020-01295-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Ahmad I, Rathore FA. Neurological manifestations and complications of COVID-19: A literature review. J Clin Neurosci. (2020) 77:8–12. 10.1016/j.jocn.2020.05.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Bougakov D, Podell K, Goldberg E. Multiple neuroinvasive pathways in COVID-19. Mol Neurobiol. (2020) 29:1–12. 10.1007/s12035-020-02152-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Candan SA, Elibol N, Abdullahi A. Consideration of prevention and management of long-term consequences of post-acute respiratory distress syndrome in patients with COVID-19. Physiother Theory Pract. (2020) 36:663–8. 10.1080/09593985.2020.1766181 [DOI] [PubMed] [Google Scholar]
- 114.Wijeratne T, Gillard Crewther S, Sales C, Karimi L. COVID-19 pathophysiology predicts that ischemic stroke occurrence is an expectation, not an exception-a systematic review. Front Neurol. (2021) 11:607221. 10.3389/fneur.2020.607221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Fischer PR, Renemane R. Mental disorders following COVID-19 infection: a systematic review of acute and long-term psychiatric manifestations and associated brain changes. Proc. Latvian Acad. Sci., Section B. (2020) 74:347–57. 10.2478/prolas-2020-0053 [DOI] [Google Scholar]
- 116.Hundreds of ‘Long Haulers' Present with Neurologic Complaints at Post-COVID-19 Clinics . Available online at: https://journals.lww.com/neurotodayonline/Fulltext/2020/11050/Hundreds_of__Long_Haulers__Present_with_Neurologic.3.aspx (accessed November 5, 2020).
- 117.Nath A. Long-haul COVID. Neurology. (2020) 95:559–560. 10.1212/WNL.0000000000010640 [DOI] [PubMed] [Google Scholar]
- 118.Otte MS, Eckel HNC, Poluschkin L, Klussmann JP, Luers JC. Olfactory dysfunction in patients after recovering from COVID-19. Acta Otolaryngol. (2020) 140:1032–5. 10.1080/00016489.2020.1811999 [DOI] [PubMed] [Google Scholar]
- 119.Konstantinidis I, Delides A, Tsakiropoulou E, Maragoudakis P, Sapounas S, Tsiodras S. Short-term follow-up of self-isolated COVID-19 patients with smell and taste dysfunction in Greece: two phenotypes of recovery. ORL J Otorhinolaryngol Relat Spec. (2020) 82:295–303. 10.1159/000511436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Paolo G. Does COVID-19 cause permanent damage to olfactory and gustatory function? Med Hypotheses. (2020) 143:110086. 10.1016/j.mehy.2020.110086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Hopkins C, Burges Watson DL, Kelly C, Leary V, Smith BC. Managing long covid: don't overlook olfactory dysfunction. BMJ. (2020) 370:m3736. 10.1136/bmj.m3736 [DOI] [PubMed] [Google Scholar]
- 122.Hopkins C, Surda P, Vaira LA, Lechien JR, Safarian M, Saussez S, et al. Six month follow-up of self-reported loss of smell during the COVID-19 pandemic. Rhinology. (2021) 59:26–31. 10.4193/Rhin20.544 [DOI] [PubMed] [Google Scholar]
- 123.Greenhalgh T, Knight M, A'Court C, Buxton M, Husain L. Management of post-acute covid-19 in primary care. BMJ. (2020) 370:m3026. 10.1136/bmj.m3026 [DOI] [PubMed] [Google Scholar]
- 124.Kamal M, Abo Omirah M, Hussein A, Saeed H. Assessment and characterisation of post-COVID-19 manifestations. Int J Clin Pract. (2020). 10.1111/ijcp.13746. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Amenta EM, Spallone A, Rodriguez-Barradas MC, El Sahly HM, Atmar RL, Kulkarni PA. Postacute COVID-19: an overview and approach to classification. Open Forum Infect Dis. (2020) 7:ofaa509. 10.1093/ofid/ofaa509 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Del Rio C, Collins LF, Malani P. Long-term health consequences of COVID-19. JAMA. (2020) 324:1723–24. 10.1001/jama.2020.19719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Hayes JP. Considering the long-term respiratory effects of Covid-19. Occup Med. (2021) Jan 22:kqaa224. 10.1093/occmed/kqaa224 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Higgins V, Sohaei D, Diamandis EP, Prassas I. COVID-19: from an acute to chronic disease? Potential long-term health consequences. Crit Rev Clin Lab Sci. (2020). 10.1080/10408363.2020.1860895. [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 129.Iacobucci G. Long covid: damage to multiple organs presents in young, low risk patients. BMJ. (2020) 371:m4470. 10.1136/bmj.m4470 [DOI] [Google Scholar]
- 130.Ladds E, Rushforth A, Wieringa S, Taylor S, Rayner C, Husain L, et al. Developing services for long COVID: lessons from a study of wounded healers. Clin Med. (2021) 21:59–65. 10.7861/clinmed.2020-0962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Leth S, Gunst JD, Mathiasen VD, Hansen KS, Søgaard OS, Østergaard L, Jensen-Fangel S, et al. Persistent symptoms in hospitalized patients recovering from COVID-19 in Denmark. Open Forum Infectious Diseases. (2021) ofab042. 10.1093/ofid/ofab042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Lopez-Leon S, Wegman-Ostrosky T, Perelman C, Sepulveda R, Rebolledo PA, Cuapio A, et al. More than 50 long-term effects of COVID-19: a systematic review and meta-analysis. medRxiv [Preprint]. (2021). 10.21203/rs.3.rs-266574/v1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Manolis AS, Manolis TA. Long COVID: an emerging puzzle. Rhythmos. (2021). 16:89–94. [Google Scholar]
- 134.Mendelson M, Nel J, Blumberg L, Madhi SA, Dryden M, Stevens W, et al. Long-COVID: an evolving problem with an extensive impact. S Afr Med J. (2020) 111:10–12. 10.7196/SAMJ.2020.v111i11.15433 [DOI] [PubMed] [Google Scholar]
- 135.Nikhra V. Living with ‘Long COVID-19': the long-term complications and sequelae. Int J Clin Virol. (2021) 5:011–7. [Google Scholar]
- 136.Norton A, Olliaro P, Sigfrid L, Carson G, Hastie C, Kaushic C, et al. Long COVID: tackling a multifaceted condition requires a multidisciplinary approach. Lancet Infect Dis. (2021). 10.1016/S1473-3099(21)00043-8. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Outhoff K. Sick and tired of COVID-19: long haulers and post viral (fatigue) syndromes. SAGP. (2020) 1:132–4. 10.36303/SAGP.2020.1.4.0041 [DOI] [Google Scholar]
- 138.Pearmain L, Avram C, Yioe V, Webb P, Margaritopoulos GA, Rivera-Ortega P, et al. P168 patient symptoms following discharge from hospital after COVID-19 pneumonia. Thorax. (2021). 10.1136/thorax-2020-BTSabstracts.313. [Epub ahead of print]. [DOI] [Google Scholar]
- 139.Saigal A, Naidu SB, Shah AJ, Brill SE, Jarvis H, Goldring JG, et al. S54 ‘long-COVID': the need for multi-disciplinary working. Thorax. (2021) 76(Suppl. 1) A33–4. 10.1136/thorax-2020-BTSabstracts.59 [DOI] [Google Scholar]
- 140.Simani L, Ramezani M, Darazam IA, Sagharichi M, Aalipour MA, Ghorbani F, et al. Prevalence and correlates of chronic fatigue syndrome and post-traumatic stress disorder after the outbreak of the COVID-19. J Neurovirol. (2021) 27:154–9. 10.1007/s13365-021-00949-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Yong SJ. Long-haul COVID-19: putative pathophysiology, risk factors, and treatments. Preprints. (2020) 2020120242. 10.20944/preprints202012.0242.v1 [DOI] [Google Scholar]
- 142.Zapatero DC, Hanquet G, Van Den Heede K. Epidemiology of Long Covid: A Pragmatic Review of the Literature. Available online at: https://kce.fgov.be/sites/default/files/atoms/files/2020-04HSR_LongCOVID_COVID%20Contributions_01022021.pdf (accessed March, 2021).
- 143.Tenforde MW, Kim SS, Lindsell CJ, Billig Rose E, Shapiro NI, Files DC, et al. Symptom duration and risk factors for delayed return to usual health among outpatients with COVID-19 in a Multistate Health Care Systems Network - United States, March-June 2020. MMWR Morb Mortal Wkly Rep. (2020). 69:993–8. 10.15585/mmwr.mm6930e1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Rimmer A. Covid-19: Impact of long-term symptoms will be profound, warns BMA. BMJ. (2020) 370:m3218. 10.1136/bmj.m3218 [DOI] [PubMed] [Google Scholar]
- 145.Davido B, Seang S, Tubiana R, de Truchis P. Post-COVID-19 chronic symptoms: a postinfectious entity? Clin Microbiol Infect. (2020) 26:1448–1449. 10.1016/j.cmi.2020.07.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Davido B, Seang S, Barizien N, Tubiana R, de Truchis P. 'Post-COVID-19 chronic symptoms' - Author's reply. Clin Microbiol Infect. (2020) 27:495–6. 10.1016/j.cmi.2020.09.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Miglis MG, Goodman BP, Chémali KR, Stiles L. Re: 'Post-COVID-19 chronic symptoms' by Davido et al. Clin Microbiol Infect. (2020). 27:494. 10.1016/j.cmi.2020.08.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Sudre CH, Murray B, Varsavsky T, Graham MS, Penfold RS, Bowyer RC, et al. Attributes and predictors of long-COVID: analysis of COVID cases and their symptoms collected by the Covid Symptoms Study App. medRxiv [Preprint]. (2020). 10.1101/2020.10.19.20214494 [DOI] [Google Scholar]
- 149.Ladds E, Rushforth A, Wieringa S, Taylor S, Rayner C, Husain L, et al. Persistent symptoms after Covid-19: qualitative study of 114 “long Covid” patients and draft quality criteria for services. medRxiv [Preprint]. (2020). 10.1101/2020.10.13.20211854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Yelin D, Wirtheim E, Vetter P, Kalil AC, Bruchfeld J, Runold M, et al. Long-term consequences of COVID-19: research needs. Lancet Infect Dis. (2020) 20:1115–7. 10.1016/S1473-3099(20)30701-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Garner P. Covid-19 at 14 Weeks—Phantom Speed Cameras, Unknown Limits, and Harsh Penalties. Available online at: https://blogs.bmj.com/bmj/2020/06/23/paul-garner-covid-19-at-14-weeks-phantom-speed-cameras-unknown-limits-and-harsh-penalties/ (accessed November 25, 2020).
- 152.Rayner C, Lokugamage AU, Molokhia M. COVID-19: Prolonged and Relapsing Course of Illness Has Implications for Returning Workers. Available online at: https://blogs.bmj.com/bmj/2020/06/23/covid-19-prolonged-and-relapsing-course-of-illness-has-implications-for-returning-workers/ (accessed November 25, 2020).
- 153.Alwan NA, Attree E, Blair JM, Bogaert D, Bowen MA, Boyle J, et al. From doctors as patients: a manifesto for tackling persisting symptoms of covid-19. BMJ. (2020) 370:m3565. 10.1136/bmj.m3565 [DOI] [PubMed] [Google Scholar]
- 154.Kingstone T, Taylor AK, O'Donnell CA, Atherton H, Blane DN, Chew-Graham CA. Finding the 'right' GP: a qualitative study of the experiences of people with long-COVID. BJGP Open. (2020) 4:bjgpopen20X101143. 10.3399/bjgpopen20X101143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Geddes L. The enduring grip of covid-19. New Sci. (2020) 246:34–8. 10.1016/S0262-4079(20)31141-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Williams FMK, Muirhead N, Pariante C. Covid-19 and chronic fatigue. BMJ. (2020) 370:m2922. 10.1136/bmj.m2922 [DOI] [PubMed] [Google Scholar]
- 157.Nehme M, Braillard O, Alcoba G, Aebischer Perone S, Courvoisier D, Chappuis F, et al. COVID-19 symptoms: longitudinal evolution and persistence in outpatient settings. Ann Intern Med. (2020) 8:M20–5926. 10.7326/M20-5926 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Zuin M, Rigatelli G, Zuliani G, Roncon L. Fatigue as long-term consequence of ARDS in COVID-19 patients. Anaesth Crit Care Pain Med. (2020) 26:100787. 10.1016/j.accpm.2020.10.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Bliddal S, Banasik K, Pedersen OB, Nissen I, Cantwell L, Schwinn M, et al. Acute and persistent symptoms in non-hospitalized PCR-confirmed COVID-19 patients. medRxiv [Preprint]. (2021). 10.1101/2021.01.22.21249945 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Banda JM, Singh GV, Alser O, Prieto-Alhambra D. Long-term patient-reported symptoms of COVID-19: an analysis of social media data. medRxiv [Preprint]. (2020). 10.1101/2020.07.29.20164418 [DOI] [Google Scholar]
- 161.Goërtz YMJ, Van Herck M, Delbressine JM, Vaes AW, Meys R, Machado FVC, et al. Persistent symptoms 3 months after a SARS-CoV-2 infection: the post-COVID-19 syndrome? ERJ Open Res. (2020) 6:00542-2020. 10.1183/23120541.00542-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Garrigues E, Janvier P, Kherabi Y, Le Bot A, Hamon A, Gouze H, et al. Post-discharge persistent symptoms and health-related quality of life after hospitalization for COVID-19. J Infect. (2020) 81:e4–e6. 10.1016/j.jinf.2020.08.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Ludvigsson JF. Reporting suspicions of long COVID in children is justified during this global emergency. Acta Paediatr. (2021). 10.1111/apa.15762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Hertting O. More research is needed on the long-term effects of COVID-19 on children and adolescents. Acta Paediatr. (2021) 110:744–5. 10.1111/apa.15731 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.British Society for Immunology . Long-Term Immunological Health Consequences of COVID-19. Available online at: https://www.immunology.org/sites/default/files/BSI_Briefing_Note_August_2020_FINAL.pdf (accessed August 13, 2020).
- 166.Moldofsky H, Patcai J. Chronic widespread musculoskeletal pain, fatigue, depression and disordered sleep in chronic post-SARS syndrome; a case-controlled study. BMC Neurol. (2011) 11:37. 10.1186/1471-2377-11-37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Wilson HW, Amo-Addae M, Kenu E, Ilesanmi OS, Ameme DK, Sackey SO. Post-ebola syndrome among Ebola virus disease survivors in Montserrado County, Liberia (2016). Biomed Res Int. (2018) 2018:1909410. 10.1155/2018/1909410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Zheng S, Fan J, Yu F, Feng B, Lou B, Zou Q. Viral load dynamics and disease severity in patients infected with SARS-CoV-2 in Zhejiang province, China, January-March 2020: retrospective cohort study. BMJ. (2020) 369:m1443. 10.1136/bmj.m1443 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Widders A, Broom A, Broomc J. SARS-CoV-2: the viral shedding vs. infectivity dilemma. Infect Dis Health. (2020) 25:210–5. 10.1016/j.idh.2020.05.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Katz BZ, Collin SM, Murphy G, Moss-Morris R, Wyller VB, Wensaas KA, et al. The international collaborative on fatigue following infection (COFFI). Fatigue. (2018) 6:106–21. 10.1080/21641846.2018.1426086 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author/s.