Abstract
Eosinophilic disorders encompass a large spectrum of heterogeneous diseases sharing the presence of elevated numbers of eosinophils in blood and/or tissues. Among these disorders, the role of eosinophils can vary widely, ranging from a modest participation in the disease process to the predominant perpetrator of tissue damage. In many cases, eosinophilic expansion is polyclonal, driven by enhanced production of interleukin-5, mainly by type 2 helper cells (Th2 cells) with a possible contribution of type 2 innate lymphoid cells (ILC2s). Among the key steps implicated in the establishment of type 2 immune responses, leukocyte recruitment toward inflamed tissues is particularly relevant. Herein, the contribution of the chemo-attractant molecule thymus and activation-regulated chemokine (TARC/CCL17) to type 2 immunity will be reviewed. The clinical relevance of this chemokine and its target, C-C chemokine receptor 4 (CCR4), will be illustrated in the setting of various eosinophilic disorders. Special emphasis will be put on the potential diagnostic, prognostic, and therapeutic implications related to activation of the TARC/CCL17-CCR4 axis.
Keywords: Thymus and activation-regulated chemokine (TARC), CCL17, C-C chemokine receptor 4 (CCR4), Eosinophils, Eosinophilic disorders, Hypereosinophilic syndromes
Introduction
A multitude of conditions are associated with the increased presence of eosinophils in blood and/or tissues, ranging from widespread—and generally benign—disorders such as allergic asthma (AA) or atopic dermatitis (AD) to rare but severe diseases such as myeloproliferative hypereosinophilic syndrome variants (M-HES). The relative contribution of eosinophils to pathogenesis of these disorders is variable, partnering with other immune cell types in the setting of complex interactions (e.g., bullous pemphigoid) or acting as key central effector cells contributing to tissue damage like in PDGFRA/FIP1L1-positive M-HES [1, 2]. Among the factors playing a role in the emergence of eosinophilia, interleukin (IL)-5 is a key cytokine with many effects on this cell throughout its life-span [3]. Sources of IL-5 include type 2 helper T cells (Th2 cells), type 2 innate lymphoid cells (ILC2s), or malignantly transformed cells, all of which can potentially be involved in the pathophysiology of eosinophilic inflammation and associated diseases [3].
Thymus and activation-regulated chemokine (TARC), also named CCL17, is a CC-chemokine commonly associated with type 2 immune responses [4]. By binding to C-C chemokine receptor type 4 (CCR4), which is, inter alia, expressed by Th2 cells [5], TARC/CCL17 participates in trafficking of Th2 cells in eosinophil-associated disorders including AA and AD and presumably of neoplastic cells in certain T cell lymphomas (e.g., angioimmunoblastic T cell lymphoma (AITL), mycosis fungoides (MF), and Sezary syndrome (SS)) [6]. Thus, elevated serum and/or tissue levels of TARC/CCL17 and cellular CCR4 expression observed in these disorders may serve as biomarkers correlating with disease severity (e.g., AD [7]) and/or be targeted for therapeutic purposes [8].
Herein, current knowledge on the sources, properties, and functions of TARC/CCL17 and its receptor CCR4 will be reviewed extensively, after a brief overview of type 2 immune responses, eosinophil biology, and the definition and classification of eosinophilic disorders. Their participation in eosinophilic inflammation will be illustrated through preclinical models and clinical findings. Finally, we will discuss to which extent the TARC/CCL17-CCR4 axis can serve as a diagnostic or prognostic marker and/or as a therapeutic target in human eosinophilic diseases.
General considerations about type 2 immune responses and eosinophil biology
Type 2 immune cells typically participate in host defense against helminths and are the hallmark of the so-called allergic reaction in which genetically predisposed individuals develop immediate hypersensitivity in response to an antigen, called an allergen, after repeated exposures [9]. As a consequence of decreased epithelial barrier integrity, for example following direct trauma, viral infection, or a genetic defect, the immune system may encounter environmental allergens (e.g., peptides derived from pollen or house dust mite (HDM)) [9]. By secreting alarmins, including IL-25, IL-33, and thymic stromal lymphopoietin (TSLP), damaged epithelial cells activate ILC2s, the innate non antigen-receptor-expressing counterpart of Th2 [10]. These cells act as a primary source of type 2 cytokines through expression of the transcription factor GATA-3 [10], thereby initiating type 2 responses by recruiting other innate cells (including eosinophils) and promoting Th2 differentiation [11].
Activation and differentiation of naïve CD4 T cells into IL-4, IL-5, and IL-13 producing Th2 cells is a key step in the generation of type 2 immune responses. The underlying mechanisms are complex, mainly involving IL-4-dependent activation of signal transducer and activator of transcription(STAT)6 that leads to the expression of GATA-3 which in turn collaborates with STAT5 to drive the expression of IL-4 from the shared IL4-IL13 gene within the T cell itself [12, 13]. Once activated, Th2 cells migrate to sites of antigen/allergen exposure and accomplish their effector functions. This recruitment to inflamed tissue involves selective expression of integrins, selectins, and chemokine receptors depending on their state of activation and differentiation [4]. For example, circulating CCR8+ CD4 T cells from healthy humans produce more IL-5 (a characteristic of highly differentiated Th2 cells) than CCR4+ CD4 T cells in which IL-4 is predominant [14]. Other chemoattractant factors for human and murine Th2 cells include IL-33, CCL17, CCL18, CCL20, CCL22, CXCL10, CX3CL1, leukotriene B4, and prostaglandin D2 (PGD2) [4, 15].
The effector functions of Th2 cells in tissue are largely mediated by the canonical type 2 cytokines IL-4, IL-5, and IL-13 which orchestrate the early and late phases of allergic disease. Interleukin-4 favors isotype class-switching within allergen-specific B cells, leading to production of IgE-type immunoglobulins. By binding to the FcεRI expressed on mast cells and basophils, allergen-specific IgE induces degranulation and release of an array of mediators that account for the typical manifestations of allergic reactions [9]. Interleukin-13 induces goblet cell hyperplasia, mucus secretion, smooth muscle contraction, and subepithelial fibrosis [16].
Interleukin-5 is critically involved in the constitution of eosinophilic inflammation. In healthy humans, eosinophils account for 3–5% of blood leucocytes and are easily recognizable by their bilobed nucleus and their cytoplasmic eosin-avid granules. They originate in the bone marrow (BM), where common myeloid progenitors give rise to eosinophilic progenitors (hEoP) characterized by surface expression of the IL-5 receptor alpha chain (IL-5Rα, CD125) that will be preserved throughout their life-cycle [17]. Human EoP will become mature eosinophils through mechanisms relying on the expression of several transcription factors (GATA-1, C/EBPα, C/EBPε, and PU-1) and growth factors including IL-5, IL-3, and granulocyte macrophage colony stimulating factor (GM-CSF) [18, 19].
Eosinophils are the predominant cell type in humans and mice expressing the IL-5 receptor at high levels, explaining the high specificity of IL-5 for this cell-type. Interleukin-5 forms homodimers that bind to the IL-5Rα chain which is coupled with the signal-transducing common beta chain [3]. Effects of IL-5 include eosinophil development through proliferation, differentiation, and maturation of hEoPs; egress of mature eosinophils from bone marrow; homing and activation in inflamed tissue; and inhibition of peripheral apoptosis [3]. ILC2s represent an important source of IL-5 in homeostatic conditions, supporting for example the colonization of the small intestine and visceral adipose tissue by eosinophils in mice [20, 21]. In pathological situations however, IL-5 derives from Th2 cells and mast cells, in addition to ILC2s [3].
Eosinophil trafficking can be independent of IL-5 as demonstrated by the presence of eosinophils in tissues from IL-5-deficient mice [22]. Several chemokines collectively called eotaxins (eotaxin-1 (CCL11), eotaxin-2 (CCL24), and eotaxin-3 (CCL26)) bind to eosinophil-expressed CCR3 and are key factors in eosinophil chemotaxis, both in homeostatic (CCL11) [23] and inflammatory (CCL24 and CCL26) conditions [24]. Cellular sources of eotaxins include epithelial cells, fibroblasts, smooth muscle cells, endothelial cells, chondrocytes, and macrophages, and their synthesis is dependent on IL-4 and IL-13 [25, 26]. VCAM-1/VLA4 [27], PGD2/chemoattractant receptor-homologous molecule expressed on Th2 cells (CRTH2) [28–30], and TSLP/TSLPR [31] interactions are also involved in eosinophil recruitment. The contribution of the TARC/CCL17-CCR4 axis in eosinophil trafficking remains debated, as CCR4 expression by blood and/or lung/bronchoalveolar lavage fluid (BALF) eosinophils has been observed in mice and humans in some [32, 33] but not all studies [34–38].
When engaged in an inflammatory response, eosinophils display a series of effector functions that are largely mediated by pre-formed mediators localized in so-called primary and specific (or crystalloid) granules and in lipid bodies. These mediators, which have been extensively described elsewhere [39], together with reactive oxygen species and IgE antibody–dependent cellular cytotoxicity (ADCC) contribute to host defense against helminths and ectoparasites, even if in vivo data are scarce in humans and divergent in mouse [19]. Furthermore, these effector functions account for eosinophil-mediated cytotoxicity and fibrosis, pro- and anti-inflammatory effects, and antiviral activity to name a few [40] and may cause significant damage in surrounding tissue.
Eosinophilic disease
Eosinophilic disorders encompass a wide range of diseases, from frequent and benign to rare and severe, which are characterized by increased blood and/or tissue eosinophilia associated with variable degrees of eosinophil-mediated damage. Indeed, eosinophil activation and degranulation can result in major, potentially irreversible or lethal organ dysfunction and damage. The archetype of eosinophil-induced toxicity is endomyocardial inflammation favoring formation of mural thrombi and subendocardial fibrosis that may progress to restrictive heart failure. Other deleterious consequences of sustained eosinophilia can occur in all organs including most commonly the skin, lungs, central and peripheral nervous systems, digestive tract, and connective tissue [41].
The definition and classification of eosinophilic disorders were revisited in 2011 by the “International Cooperative Working Group on Eosinophil Disorders” (ICOG-EO), more than 35 years after the first formal elaboration of criteria defining the hypereosinophilic syndrome (HES) [42, 43]. Eosinophilia is defined as an absolute blood eosinophil count (AEC) above 0.5×109/L, while the term hypereosinophilia (HE) applies when an AEC above 1.5×109/L is observed at least twice, with an interval of at least 1 month. In tissue, HE is present when (1) the percentage of eosinophils in BM exceeds 20% of all nucleated cells and/or (2) a pathologist considers that tissue infiltration by eosinophils is excessive and/or (3) marked deposition of eosinophil granule proteins is found. The term HES is reserved for patients fulfilling the criteria for blood and/or tissue HE and presenting with organ damage and/or dysfunction attributable to eosinophils, after exclusion of other disorders or conditions as potential cause(s) of the observed organ damage [42].
Hypereosinophilia can be further classified into variants [42]: proliferation of eosinophils may be clonal and is qualified as neoplastic (HEN), whereas polyclonal expansion of eosinophils driven by enhanced production of growth factors (mainly IL-5) is qualified as reactive (HER). When the mechanism underlying increased eosinophilopoiesis is unknown and no organ dysfunction or symptoms are present, the term HE of undetermined significance (HEUS) applies. Rarely, HE can be detected in several members of a same family and is inherited, defining familial HE.
The first step in the diagnostic approach to eosinophilia or HE is to rule out a reactive/secondary cause [44], such as allergic disease (e.g., severe eosinophilic asthma), parasitosis (e.g., helminths, scabies), adverse drug reactions (e.g., anticonvulsants), and cancer (e.g., certain adenocarcinomas, Hodgkin’s or T cell lymphomas). The second step is to assess for potential eosinophil-induced organ damage. If present, diagnosis of HES must be considered, and further evaluation for HES disease variants is warranted. Neoplastic (HESN, or primary, clonal, myeloproliferative) HES is associated in approximately 80% of cases with a deletion on chromosome 4q12, creating the Fip 1-like 1 (FIP1L1)/platelet-derived growth factor receptor alpha (PDGFRA) fusion gene, which encodes a constitutively active tyrosine kinase (TK) [45]. Reactive (HESR, or secondary) HES describes situations where reactive HE causes organ damage and dysfunction. Besides classical causes of secondary HE (see above), this entity encompasses lymphocytic variant HES (L-HES) where HE is caused by enhanced IL-5 production by a clonal T cell subset with an abnormal surface phenotype [46]. Finally, the term idiopathic HES (I-HES) is used when the diagnostic work-up fails to identify a known etiology for eosinophilic expansion. This last category accounts for more than half of patients presenting with HES in expert centers [47].
The biology of thymus and activation-regulated chemokine
TARC/CCL17: early discoveries, cellular expression, and mechanisms of synthesis
With the discovery of TARC/CCL17 in 1996, Imai et al. were the first to describe a CC chemokine with selective activity for lymphocytes [48]. Its name reflects the observed constitutive expression in the human thymus and its induction in peripheral blood mononuclear cells (PBMCs) following activation by phytohemagglutinin [48]. One year later, the same team identified CCR4 as the main receptor for TARC/CCL17 and showed that CCR4 mRNA was expressed in CD4+ T cells [49].
Human TARC/CCL17 is an 8-kDa protein composed of 71 amino acids and is encoded on chromosome 16q13 [48, 50]. Murine studies have shown that steady-state TARC/CCL17 synthesis occurs in various tissues including the thymus, lymph nodes (LNs), gut, and bronchi but not in the spleen. The cellular sources of this chemokine were mainly Langerhans cells (LCs) and mature myeloid dendritic cells (DCs) [51]. In humans, monocyte-derived DCs were shown to synthetize TARC/CCL17 in response to IL-3 and tumor necrosis factor alpha (TNF-α) in presence of IL-4 in in vitro cultures [5]. Subsequently, several immune and non-immune cellular sources of TARC/CCL17 were identified, as detailed in Table 1.
Table 1.
Sources and mechanisms of TARC/CCL17 synthesis
| Cellular sources of TARC/CCL17 | Species | Inducer(s) | Inhibitors(s) | Potential intracellular pathways involved | Ref(s) |
|---|---|---|---|---|---|
| Monocytes | Human | IL-4, IL-3, GM-CSF | IFN-γ, IL-10 | JAK1/JAK3, STAT6, JMJD3, IRF4 | [5, 52, 53] |
| Monocyte-derived DCs | Human | IL-4, GM-CSF | No effect of IFN-γ | [5] | |
| Macrophages | Mouse | IL-4, GM-CSF | IFN-γ | [52, 53, 158] | |
| M2 macrophages | Human |
IL-4, IL-13, RANK engagement |
Pan-PI3K inhibitor, no effect of glucocorticoids | PI3K | [103a, 159] |
| Myeloid DCs | Human, CD11c+ DCs |
TSLP, TSLP + RANK or CD40 engagement |
/ | / | [160, 161] |
| Mouse, CD11c+ DCs | TSLP | / | / | [162] | |
| IL-33 | / | ERK1/2, JNK, p38 MAPK, | [163] | ||
| IL-4, IL-13 | / | STAT6 | [54] | ||
| Langerhans cells (skin) | Mouse | TNF-α, IL-4, IL-13 | IFN-γ, GM-CSF | / | [164] |
| Monocyte-derived LCs | Human, AD patients | S. aureus extract (PGN) | Histamine H4 receptor antagonist | p38 MAPK | [165] |
| Eosinophils | Human, allergic volunteers | IL-4, IL-4+TNFα | / | STAT6, NFκB, MEK, JNK | [32] |
| CD3+ T cells | Human, HC | IL-4 | / | STAT6 | [55] |
| CD4+CD45RA+ T cells | Human, AA patients | PBMC stimulation with Der f allergen OR with anti-CD3 + CD28 Abs | Anti-CD80 + CD86 Abs | / | [104] |
| Keratinocytes | Human | TNF-α + IFN-γb |
IL-4, IL-13, TGF-β, Casuarinin, Spinasterol Glc, Allopurinol, Roxithromycin |
p38 MAPK, Raf1, JAK2, STAT1, NFκB | [56, 60, 166–171] |
| HDM extract, IL-22 | / | STAT3, ERK, AKT | [79] | ||
| Human, Mouse | GITR engagement | / | NFκB | [172] | |
| Bronchial epithelial cells | Human |
TNF-α + IFN-γ +/- IL-1αb TNF-α + IL-4 + IFN-γb IL-4 alone: weak or no effect |
Glucocorticoids, Long-acting β-2 adrenergic agonist |
NFκB | [57, 58, 173] |
|
Der p alone IL-4 + TGF-β Der p + IL-4 + TGF-β (highest response) IL-4 alone: no effect |
/ |
ADAM-dependent EGFR phosphorylation, p38 MAPK, ERK1/2, NFκB |
[101] | ||
| TLR3 ligand poly(I:C)c | / | / | [174] | ||
| TWEAK + TGF-β1 during EMT | / | / | [175] | ||
| Alveolar epithelial cells | Human |
TNF-α or IL-1α alone TNF-α or IL-1α or IFN-γ + IL-4 or IL-13b RSV + IL-4 or IL-13b IL-4/IL-13 alone: no effect |
Glucocorticoids | NFκB, STAT6 | [57, 58, 176] |
| Nasal epithelial cells | Human, allergic volunteers and HC | TNF-α + IL-4 or IL-13 | / | / | [177] |
| Airway smooth muscle cells | Human |
IL-4 or IL-13 + TNF-α IL-4/IL-13 alone: no effect |
β-adrenergic agonist (isoproterenol), no effect of glucocorticoids | / | [178] |
| Dermal endothelial cells | Human | IL-4 or IL-13 + TNF-αIL-4/IL-13 alone: no effect | No effect of glucocorticoids | p38 MAPK, NFκB | [179] |
| Dermal fibroblasts | Human |
IL-4 or IL-13 + TNF-α (enhanced by IFN-γ) IL-4/IL-13 alone: no effect |
No effect of glucocorticoids | / | [166, 179] |
Abs antibodies, AD atopic dermatitis, ADAM a disintegrin and metalloproteinase, CD40L CD40 ligand, DC dendritic cells, Der f/p dermatophagoides farinae/pteronyssinus, dsRNA double-stranded ribonucleic acid, EGFR epidermal growth factor receptor, EMT epitheliomesenchymal transition, ERK extracellular signal regulated kinase, GITR(L) glucocorticoid-induced TNFR-related protein (ligand), GM-CSF granulocyte monocyte colony stimulating factor, HC healthy controls, HDM house dust mite, IFN interferon, IL interleukin, IRF interferon regulatory factor, JMJD Jumonji C domain-containing proteins, JNK Janus kinase, LC Langerhans cell, LPS lipopolysaccharide, MAPK mitogen-activated protein kinases, MEK mitogen-activated protein/extracellular signal-regulated kinase kinase, NFKB nuclear factor-kappa B, PGN peptidoglycans, PI3K phosphoinositide 3-kinase, PBMC peripheral blood mononuclear cell, RANK(L) receptor activator of nuclear factor kappa-B (ligand), RSV respiratory syncytial virus, STAT signal transducers and activators of transcription, TGF transforming growth factor, TLR toll-like receptor, TNF tumor necrosis factor, TSLP thymic stromal lymphopoietin, TWEAK tumor necrosis factor (TNF)-like weak inducer of apoptosis
aIn this study, macrophages demonstrated a partial M2 phenotype
bA synergistic effect appears when cells are co-stimulated with these agents
cA synthetic analog of viral dsRNA
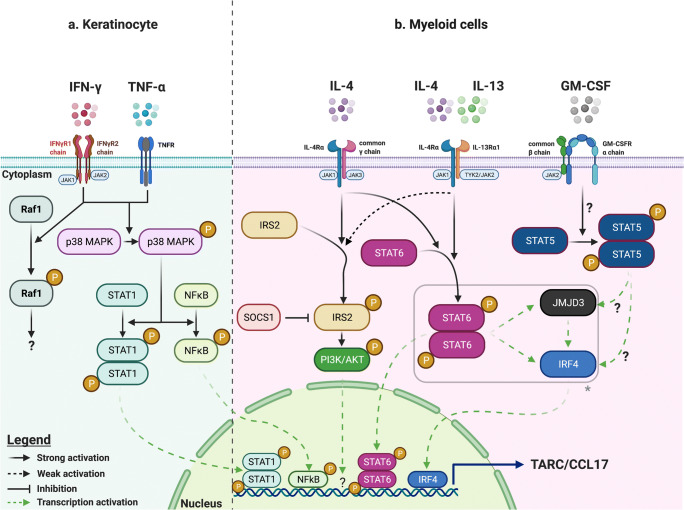
Molecular mechanisms underlying TARC/CCL17 synthesis and secretion are variable depending on the nature of the cell and the stimuli. In immune cells, IL-4 stimulates TARC/CCL17 synthesis, synergizing with other cytokines depending on the cell type [5, 52–54]. STAT6 activation is a key step in IL-4-induced TARC/CCL17 synthesis by binding directly to the CCL17 gene promoter via two binding sites (Fig. 1) [55]. In keratinocytes and bronchial and alveolar epithelial cells, TARC/CCL17 synthesis is triggered by TNF-α and interferon (IFN)-γ that act synergistically in a nuclear factor-kappa B (NFκB)-dependent manner [56–58], consistent with the presence of a NFκB binding site in the CCL17 promoter [59]. The effect of IL-4 varies, with an inhibitory effect observed in keratinocytes [56, 60] while a co-stimulatory effect applies to other cell types (Table 1).
Fig. 1.
Identified molecular mechanisms underlying TARC/CCL17 synthesis in selected cell types. Mechanisms involved in induction of TARC/CCL17 synthesis are shown schematically for a human keratinocyte cell lines (HaCaT) and b human monocytes and murine bone marrow–derived macrophages. In HaCaT cells, TNF-α and IFN-γ induce JAK2, p38 MAPK, and Raf-1 activation by phosphorylation after ligation to their dedicated receptors [168, 170]. Subsequently, activated p38 MAPK phosphorylates STAT1 and NFκB, inducing their activation and translocation into the nucleus to trigger TARC/CCL17 synthesis [169]. b In human monocytes and murine macrophages, IL-4 and IL-13-induced phosphorylation and homodimerization of STAT6 (following engagement of both types of IL-4 receptors) triggers TARC/CCL17 expression directly by binding the TARC gene promoter [53, 206]. In addition, activated STAT6 increases transcription of IRF4 and JMJD3, and the latter induces the demethylation of repressive H3K27me3 in the IRF4 promoter region, resulting in enhanced expression of the transcription factor IRF4 that binds directly to the TARC/CCL17 promoter (*the latter mechanism is demonstrated after IL-4 but not IL-13-induction of STAT6) [53]. A similar pathway is involved in GM-CSF-induced TARC/CCL17 transcription, probably through STAT5 activation [52]. Engagement of the type 1 IL-4Rα/common-γ chain heterodimeric receptor by IL-4 also recruits IRS2, inducing its phosphorylation and activation. In turn, IRS2 phosphorylates/activates the PI3K/AKT pathway, ultimately leading to TARC/CCL17 transcription [206]. SOCS1 expression is also induced by IL-4 in healthy human monocytes and has been shown to interact directly with IRS2 and downregulate its activity, through ubiquitination and proteasomal degradation [206]. This figure was created with BioRender.com. IFN interferon, IL interleukin, IRF interferon regulatory factor, IRS insulin receptor substrate, JAK Janus kinase, JMJD3 Jumonji domain-containing protein D3, MAPK mitogen-activated protein kinases, NFκB nuclear factor-kappa B, PI3K phosphatidylinositol 3-kinase, STAT signal transducer and activator of transcription, SOCS suppressor of cytokine signaling protein, TNF tumor necrosis factor, TYK tyrosine kinase
TARC/CCL17 selectively binds to CCR4
CCR4 belongs to the G protein-coupled receptor (GPCR) family and is thus composed of seven transmembrane domains [61]. In addition to TARC/CCL17, CCR4 binds macrophage-derived chemokine (MDC/CCL22) which shares 37% homology in its amino acid sequence [6]. MDC/CCL22 is also expressed in the human thymus [62] and produced by DC, macrophages, and monocytes [63–65]. MDC/CCL22 exhibits 2- to 3-times higher affinity for CCR4 compared with TARC/CCL17 [62] and is more potent in promoting integrin-dependent arrest of lymphocytes on VCAM-1 [66] as well as inducing CCR4 desensitization and internalization [67]. These differences may be explained by different CCR4 conformations. In human T cells, the major R1 form binds both chemokines while the minor R2 forms only bind MDC/CCL22. When all R1 receptors are occupied, MDC/CCL22 is still able to increase chemotaxis through R2 receptors whereas an additive effect of TARC/CCL17 is not possible [68]. Furthermore, GPCR-induced chemoattraction by MDC/CCL22 of in vitro differentiated murine Th2 cells was shown to rely not only on the phosphatidylinositol 3-kinase (PI3K) signaling pathway (shared with TARC/CCL17) but also on beta-arrestin-2, enhancing chemotaxis [69]. Whether the activation of this pathway by MDC/CCL22 is linked to a distinct CCR4 conformation is not known.
Functional characterization of CCR4-expressing CD4+ T cells supports their Th2 differentiation as they spontaneously synthetize IL-4 and IL-5 but no IFN-γ in in vitro cultures [5]. These early discoveries suggested a potential role of TARC/CCL17 and CCR4-expressing cells in type 2 immune responses as discussed below. Besides Th2 cells, several other cell types involved in type 2 immune responses express CCR4, including ILC2s [70]. Other populations reported to be CCR4-positive are listed in Table 2.
Table 2.
Human cells and tissues with demonstrated CCR4 expression
| Cells expressing CCR4 in humans | Reference(s) |
|---|---|
| Type 2 helper cells (Th2) | [5, 180, 181] |
| CLA+ T cells | [77, 182] |
| Type 2 polarized CD8+ T cells (Tc2) | [183, 184] |
| Regulatory T cells (Tregs) | [80, 185] |
| T helper 17 cells (Th17) | [186] |
| T helper 22 cells (Th22) | [187] |
| Type 2 innate lymphoid cells (ILC2s) | [70, 188, 189] |
| Airway eosinophils (AA patients) | [32] |
| Airway mast cells (AA patients) | [190] |
| Plasmacytoid DCs (AA patients) | [191] |
| Conventional DCs (AA patients) | [192] |
| Airway epithelial cells (BEAS-2B, A549 cell lines) | [193] |
TARC and its relevance in eosinophilic diseases
Several studies have shown that TARC/CCL17 may be increased in serum and/or tissues in various eosinophilic conditions. These findings are summarized in Table 3 and described in more detail below.
Table 3.
Eosinophilic disorders with enhanced TARC/CCL17 production
| Disorder | Site/magnitudea of sTARC/CCL17 increase | Principal cellular source of TARC/CCL17 | Correlation(s) of sTARC with other biomarkers/clinical featuresb | Potential clinical implications | Refs |
|---|---|---|---|---|---|
| Atopic dermatitis |
Skin Serum Median 1733 pg/mL (IQR 696–4742) |
Epidermal keratinocytes Dermal fibroblasts Endothelial cells Langerhans cells CD1a+ DCs |
(+) Score AD index (SCORAD) (+) Six sign AD (SASSAD) (+) Blood eosinophils (+) Serum soluble E-selectin (+) Serum sCD25 |
Assessment and monitoring of disease severity Prediction (in most cases) of severe disease course at diagnosis |
[7, 56, 74, 165] |
| DRESS |
Skin Serum Mean 31,259 pg/ml (SEM ±6374) |
CD11c+ DCs |
(+) Blood eosinophils (+) Serum sCD25 (+) Skin lesions severity |
Differentiation with other drug-induced skin reactions (SJS, maculopapular erythema) Association btw higher sTARC/CCL17 and HHV-6 reactivation |
[82, 83] |
| Bullous pemphigoid |
Skin Serum Mean 1151 pg/mL (range, 91–3981) |
Epidermal keratinocytes |
(+) Blood eosinophils (+) BP Disease Area Index score (+) Urticaria/erythema scores |
Disease activity monitoring (earlier elevation than anti-BP180 autoantibodies) | [86, 138] |
| Senile erythroderma |
Serum Median 6872 pg/ml (IQR: 4303–25,683) (idiopathic group) |
Not assessed | (+) Serum IgE | sTARC/IgE ratio higher in chronic idiopathic erythroderma than in AD in elderly > differential diagnosis | [88, 89] |
| Allergic asthma |
Bronchial epithelium BALF Sputum Serum Mean 271 pg/mL (range 50–1000 pg/mL) |
Bronchial epithelial cells M2 macrophages CD11C+ DCs CD4+CD45RA+ T cells Eosinophils |
(+) serum MDC & serum eotaxin (+) Serum IgE (inconstant finding) (-) PEFR in children with exacerbated AA |
Potential therapeutic target through inhibition of Th2 cells and potentially ILC2 accumulation in airways Combination of sTARC, sCCL26, FeNO and AEC identifies type 2-high asthmatic populationc (PPV 100%, NPV 87%) |
[32, 194–197] |
| EGPA |
Sinus Serum Mean 1122 pg/mL (SEM ±422.7) |
Not assessed (perivascular inflammatory infiltrate) |
(+) Serum IgE (+) Blood eosinophils |
Correlates with disease severity but does not predict relapse | [115, 116] |
|
ABPA (associated with CF) |
Serum Median 589 pg/mL (IQR 465–673) |
Not assessed |
(+) A. fumigatus-specific IgE (+) Recombinant A. fumigatus allergen f4-IgE (–) FEV1 |
Differentiation btw ABPA and aspergillus sensitization or colonization in CF Earlier detection of disease onset compared with total IgE kinetics |
[112, 140] |
| Acute eosinophilic pneumonia |
BALF Serum Median 17,907 pg/mL (range 15,533–32,731) |
Alveolar DCs Macrophages |
(+) TARC/CCL17 in BALF (+) TARC/CCL17 in BALF and CCR4+ CD4 T cells in BALF No correlation with AEC |
Highly sensitive and specific for AEP among patients with ALI or ARDS | [119–122] |
| Mycosis fungoides and Sézary syndrome |
Skin Serum Mean 2889 pg/mL (SEM 725.5) (MF, all stages included) |
Keratinocytes (MF) |
(+) LDH (MF) (+) Serum IgE (MF) (+) Serum sCD25 (MF) (+) MDC (MF) |
Assessment of disease progression in MF Targeted treatment: Mogamulizumab, approved for relapsed/refractory MF or SS (improves PFS compared to vorinostat) |
[127, 154, 198] |
| Adult T cell leukemia-lymphoma |
Skin Serum – |
Not assessed | – |
High (> 285 pg/mL) sTARC/CCL17 associated with unfavorable prognosis Targeted treatment: Mogamulizumab approved for use as add-on to intensive chemotherapy (improves PFS and OS) |
[131, 142, 156, 199] |
| L-HES |
Skin, Serum Median10,855 pg/mL (range 3466–422,787) |
DCs (in vitro) | No correlation with blood CD3-CD4+ T cell count |
Markedly elevated sTARC/CCL17 suggestive of L-HES among patients with F/P- HES sTARC > 3000 pg/ml: 100% sensitivity and 75% specificity (p<0.0001) for CD3-CD4+ L-HES among patients with F/P- HES Predictive of OCS-responsiveness, and suboptimal biological response to mepolizumab |
[136, 137, 143] |
AA allergic asthma, ABPA allergic broncho-pulmonary aspergillosis, AD atopic dermatitis, AEC absolute eosinophil count, AEP acute eosinophilic pneumonia, ALI acute lung injury, ARDS acute respiratory distress syndrome, BALF bronchoalveolar lavage fluid, btw between, CCL26 CC chemokine 6, CCR4 CC chemokine receptor 4, CF cystic fibrosis, DC dendritic cell, DRESS drug reaction eosinophilia and systemic symptoms, EGPA eosinophilic granulomatosis with polyangiitis, FeNO fractional exhaled nitric oxide, FEV1 forced expiratory volume in one second, F/P Fip 1-like 1 (FIP1L1)/platelet-derived growth factor receptor alpha (PDGFRA) fusion gene, HES hypereosinophilic syndrome, HHV-6 human herpes virus 6, IgE ILC2s, type 2 innate lymphoid cells, immunoglobulin E, IQR interquartile range, LDH lactate dehydrogenase, L-HES lymphoid variant HES, MDC macrophage-derived chemokine, MF mycosis fungoides, OCS oral corticosteroid therapy, OS overall survival, PEFR peak expiratory flow rate, PFS progression free survival, Refs references, SEM standard error of the mean, SJS Steven-Johnson syndrome, SS Sezary syndrome, (s)TARC (serum)thymus and activation-regulated chemokine, Th2 cells type 2 helper T cells, sCD25 serum soluble IL-2Ralpha
asTARC in healthy controls: median 243 pg/ml (range 34-973from 143); mean: 220,6 pg/mL (± SEM 27.9 pg/ml)[115]
b(+): positive correlation, (−) negative correlation
cType 2-high status was defined as the mucosal expression of CCL26 gene, periostin gene and a multigene IL-13 in vitro signature
Skin disorders
Atopic dermatitis (AD) is a chronic inflammatory disease characterized by upregulation of Th2 and Th22 cytokines in the acute phase, while a Th1 and Th17 profile has been demonstrated by gene expression studies in chronic lesions [71]. A type 2 immune response is central in the pathogenesis of AD, with production of allergen-specific IgE-type antibodies [71]. Eosinophilia is common in blood and skin in this disorder, but is not likely to play a central role in established lesions, as suggested by the lack of clear-cut efficacy of the anti-IL-5 monoclonal antibody (MoAb) mepolizumab in clinical trials [72, 73].
TARC/CCL17 is expressed in both acute and chronic lesions of AD by epidermal keratinocytes, dermal-infiltrating cells (CD3+ T cells and CD1a+ DC), and endothelial cells, while it is absent in normal skin [74]. Consequently, higher levels of serum (s)TARC/CCL17 are observed in AD compared to healthy controls (HC) [74]. Their levels correlate with AEC and weakly with sIL-2R-alpha (sCD25) levels, a known biomarker of T cell activation in vivo [75, 76]. T cells expressing CCR4 are detectable in lesional skin at the dermoepidermal junction, and the proportion of circulating CD4+CD45RO+ cells expressing CCR4 is higher in patients with AD compared to HC [75]. Of note, in AD, most of the circulating CCR4+ T cells were also positive for cutaneous lymphocyte antigen (CLA) in two other studies [77, 78].
The contribution of the TARC/CCL17-CCR4 axis to AD pathogenesis involves several mechanisms. We previously mentioned the importance of TNF-α and IFN-γ in TARC/CCL17 synthesis by the keratinocyte cell line HaCaT and its repression by IL-4. Furthermore, in vitro cultured peripheral T cells from HC produce IL-22, TNF-α, and IFN-γ in response to HDM extracts, whereas HaCaT cells upregulate IL-22Rα at their surface. Activation of the IL-22/IL-22Rα axis leads to production of TARC/CCL17, IL-1α, and IL-6 by HaCaT cells and recruitment of CCR4+ T cells [79]. Finally, regulatory CD4+CD25+ T cells (Treg) are known to express CCR4 and display higher expression levels in patients with severe AD compared to HC combined with a reduced ability to secrete transforming growth factor (TGF)-β and IL-10 and to suppress autologous effector T cells in vitro, indicating the probable recruitment of functionally impaired Tregs into AD skin [80].
Drug rash with eosinophilia and systemic symptoms (DRESS) is a severe drug reaction associating a disseminated rash, fever, eosinophilia, atypical circulating lymphocytes, lymphadenopathy, and organ dysfunction [81]. Serum TARC/CCL17 levels may be extremely elevated in this disorder, and CD11c+ DC have been shown to be the main source in lesional skin [82]. The level of sTARC/CCL17 correlates positively with the severity of skin manifestations as well as AEC and sCD25 [82] and is significantly higher in patients with demonstrated HHV-6 reactivation [83], although the causal link between viral reactivation and TARC/CCL17 over-expression remains elusive. Some argue that increased TARC/CCL17 could attract Tregs and alter antiviral responses leading to HHV-6 reactivation or, alternatively, TARC/CCL17 could directly induce HHV-6 activation through the chemokine receptor homologues of HHV-6 [83, 84].
Bullous pemphigoid (BP) is an autoimmune blistering disease characterized by autoantibodies targeting hemidesmosomes and is often accompanied by blood eosinophilia, elevated serum IgE levels, and a dermal infiltrate mainly composed of lymphocytes and eosinophils [1]. Several lymphocyte subsets seem implicated in BP including Th2 and Th17 cells [1]. Eosinophils are thought to play a pathogenic role since their degranulation is induced by FcεRI engagement by anti-basement membrane IgE, leading to blister formation in a humanized mouse model of BP [85]. Elevated TARC/CCL17 levels have been found in blister fluid and serum from patients with BP, and this chemokine was detected by immunohistochemistry (IHC) in basal epidermal keratinocytes from lesional skin of BP patients while CCR4 was expressed by dermal CD4+ and peripheral blood CD4+CD45RO+ T cells [86].
Senile erythroderma
Erythroderma is a debilitating skin disease defined as more than 90% of skin surface affected by erythema and scaling. More than a quarter of patients with erythroderma have peripheral eosinophilia [87]. Erythroderma may be idiopathic or secondary, occurring in the setting of disorders such as psoriasis, atopy, drug hypersensitivity (including DRESS), MF, or SS, most of which are associated with elevated sTARC/CCL17. In one series of 68 patients with erythroderma aged 65 years or older, of which 53% had a detectable secondary cause, sTARC/CCL17, serum IgE, and the level of blood and tissue eosinophilia did not differ between idiopathic and secondary subgroups [88]. Interestingly, patients with senile erythroderma (predominantly male in this study) had lower IgE levels and a lower ratio of antigen-specific IgE/total IgE but higher levels of sTARC/CCL17 than patients with AD suggesting that IgE synthesis in this disorder is the consequence of a Th2 shift independent of specific allergens [88, 89].
Other skin disorders
Elevated sTARC/CCL17 levels have been reported sporadically in several other dermatological disorders, namely, eosinophilic pustular folliculitis [90], chronic spontaneous urticaria [91], maculopapular exanthema [92], and Stevens-Johnson syndrome/toxic epidermal necrolysis (although levels are significantly lower than in DRESS) [93]. Patients with non-episodic angioedema with eosinophilia were shown to have elevated sTARC/CCL17 levels at presentation that regressed in parallel with AEC in response to corticosteroid (CS) therapy in one study [94]. In episodic angioedema, serum TARC levels are also elevated and cycle with disease activity as discussed below [95].
Pulmonary disorders
Allergic asthma (AA) is a chronic respiratory disease often characterized by eosinophilic inflammation of the airways together with increased mucus secretion and bronchial hyperreactivity. The underlying type 2 immune response combines ILC2s that represent an early source of type 2 cytokines after allergen challenge and Th2 cells [96]. Consistent with the key role of eosinophils in a subgroup of patients, biologics targeting IL-5 and its receptor reduce exacerbations and improve lung function in severe eosinophilic asthma and have been approved as add-on therapies in this indication [97].
The TARC/CCL17-CCR4 axis has been well studied in asthma, since it contributes at least partially to Th2 cell recruitment to the lung. Ex vivo allergen challenge of human bronchial explants has been shown to induce synthesis of functionally active TARC/CCL17 in patients with asthma but not HC [98]. Furthermore, several studies in asthmatic patients have reported induction of TARC/CCL17 in bronchoalveolar lavage fluid (BALF) after allergen challenge [99, 100]. Cellular sources of TARC/CCL17 in this context include not only bronchial and alveolar epithelial cells [57, 58, 101] but also CD11c+ DCs [102], M2 macrophages [103], and CD4+CD45RA+ T cells [104]. Eosinophils themselves are a potential source of TARC/CCL17 and MDC/CCL22 as demonstrated in a mouse model of allergic airway inflammation [105] and after in vitro stimulation with different cytokines in humans [106]. Altogether, these data indicate that TARC/CCL17 may contribute to effector T cell chemotaxis to lungs in asthma. Although it has been shown that CCR4+ T cells are the main source of type 2 cytokines in asthmatic patients [98], the functional relevance of the TARC/CCL17-CCR4 axis in AA remains to be elucidated, as targeting this pathway in animal models has produced conflicting results with regard to the consequences on airway hyperresponsiveness and airway inflammation [102, 107–110].
Other airway disorders
Patients with allergic rhinitis and chronic rhinosinusitis with nasal polyps (CRSwNP) were reported to have higher TARC/CCL17 levels in nasal secretions than patients with non-allergic, non-infectious rhinitis and chronic rhinosinusitis without nasal polyps, and higher sTARC/CCL17 than HC [111]. Elevated sTARC/CCL17 levels have also been observed in patients with allergic bronchopulmonary aspergillosis (ABPA) both in the setting of cystic fibrosis (CF) and AA [112]. In patients with CF, a positive correlation was observed with A. fumigatus-specific IgE [112].
Eosinophilic granulomatosis with polyangiitis (EGPA, formerly Churg-Strauss syndrome) is a rare granulomatous, eosinophil-rich, necrotizing vasculitis affecting small- and medium-sized vessels and associated with late-onset asthma and eosinophilia [113]. In 30–40% of cases, EGPA is associated with antineutrophil-cytoplasmic-antibodies (ANCAs) [114]. The pathogenesis of EGPA is complex, involving type 2 immunity, B cell activation with antibody production, and Th17 cells [114]. Patients with EGPA often have a history of allergic disease and high serum IgE levels [114]. TARC/CCL17 is expressed in active EGPA lesions in association with eosinophilic infiltrates, colocalizes with CRTH2+ T cells, and elevated serum levels have been reported in several studies [115–117]. sTARC/CCL17 has been shown to correlate with disease activity, AEC, and serum IgE levels in this disease [115].
Acute and chronic eosinophilic pneumonia (AEP and CEP) are characterized by eosinophilic infiltration of the lung parenchyma, and the former may progress to an acute respiratory distress syndrome in some cases [118]. Blood eosinophil counts are generally within normal ranges at diagnosis of AEP while they are increased in 80% of patients with CEP [118]. Elevated levels of type 2 cytokines and TARC/CCL17 in BALF have been reported in both disorders [119]. There was a tendency toward higher TARC/CCL17 levels in AEP, and a positive correlation was observed with IL-5 and IL-13 in this disorder [119]. Similarly, sTARC/CCL17 levels were significantly higher in AEP than in sarcoidosis, hypersensitivity pneumonitis, and interstitial pulmonary fibrosis [119]. A challenge with the suspected trigger of AEP in two patients was followed by a rise in sTARC/CCL17 within 16 h after provocation [120]. Cellular sources of TARC/CCL17 identified in AEP comprise alveolar DC and macrophages [121]. CCR4-positive CD4+ T cells were significantly higher in BALF than in blood in patients with AEP and CEP and were not observed in BALF from HC or patients with sarcoidosis [122]; their numbers correlated positively with BALF TARC/CCL17, MDC/CCL22, and IL-5 [122]. Ultimately, transendothelial migration of eosinophils in response to BALF from patients with AEP, assessed in vitro using human pulmonary microvascular cells, was not abrogated by a CCR4 antagonist in vitro [123]. Together, these findings highlight the increased presence and probable role of the CCR4/CCL17 axis in T cell chemotaxis to the lungs in AEP, but other factors may contribute to eosinophil accumulation.
Lymphoproliferative malignancies
Mycosis fungoides (MF) and Sezary syndrome (SS) belong to the spectrum of cutaneous T cell lymphoma and are characterized by clonal proliferation of mature T cells in the skin [124]. Disease course in MF is progressive while SS is more aggressive and generally combines circulating neoplastic T cells, erythroderma, and lymphadenopathy [125]. As disease progresses, the cytokine profile in MF evolves from type 1 to type 2, while SS typically displays only a type 2 profile where cytokine levels correlate with blood eosinophilia and serum IgE [125]. The atypical lymphoid cells that characterize this disease spectrum have hyperchromatic cerebriform nuclei, they can be detected in peripheral blood, and their distribution within tissue depends on the disease stage [125, 126]. These cells typically express CLA and CCR4 at their surface, while TARC/CCL17 is present within keratinocytes in affected skin [74]. sTARC/CCL17 levels are elevated in all disease stages but are significantly higher in advanced (tumor) stage MF and in SS [127, 128].
Expression of CCR4 may be observed in other T cell malignancies as well, such as angioimmunoblastic T cell lymphoma (AITL), unspecified peripheral T cell lymphoma (PTCL-U), and adult T cell leukemia/lymphoma (ATLL) [129]. TARC/CCL17 was detected by IHC in lymph nodes from patients with AITL and PTCL-U within cells with dendritic morphology, and its expression level correlated with eosinophilic infiltration in lymphomatous tissue [130]. Elevated TARC/CCL17, MDC/CCL22, and CCR4 mRNA expression was reported in skin from patients with ATLL compared with HC [131]. In vitro chemotaxis assays showing that the CCR4+ malignant T cells isolated from peripheral blood of ATL patients respond strongly to TARC/CCL17 and MDC/CCL22 indicate that this axis plays a functional role in pathogenesis of this disorder [131].
Hypereosinophilic syndromes
Lymphocytic variant HES (L-HES) is an indolent T cell lymphoproliferative disorder in which the clonal cells display an abnormal surface phenotype (most often CD3-CD4+TCRα/β-) and produce type 2 cytokines including IL-5 [132], explaining its classification as HESR. Common clinical manifestations include skin lesions, angioedema, lymphadenopathy, and rheumatological involvement [133, 134]. In a study investigating chemokine receptor expression on these naturally occurring human Th2 cells, our group showed that CD3−CD4+ cells expressed CCR5, CXCR4, and CCR4, although the latter was observed only when cells were left in autologous serum-free milieu suggesting that CCR4 was internalized in vivo [135]. Measurement of its ligands TARC/CCL17 and MDC/CCL22 in serum from subjects with CD3-CD4+ L-HES confirmed that sTARC/CCL17, but not sMDC/CCL22, levels were markedly elevated compared to controls, a finding that was subsequently observed in L-HES patients with other phenotypically aberrant T cell subsets as well [135, 136]. Cellular sources of TARC/CCL17 have not yet been explored in vivo, but it was shown in vitro that IL-4 issued from CD3−CD4+ cells can stimulate its production by DC, but not by eosinophils or T cells [135]. A subset of patients with CD3-CD4+ L-HES present clinically with Gleich’s syndrome, also known as episodic angioedema with eosinophilia. One study has shown that serum IL-5 and sTARC/CCL17 peak prior to blood eosinophilia and symptoms in such patients, suggesting an early role for this chemokine in the cascade of events leading to a flare [95].
Besides those with well-documented L-HES, a subset of patients with I-HES have higher sTARC/CCL17 levels than HC. One study showed that PBMC isolated from these patients display some degree of spontaneous IL-5 production in vitro, contrasting with I-HES patients with normal sTARC/CCL17 levels [136]. The proportion of I-HES patients with above-normal sTARC/CCL17 levels reached 36% in a large retrospective multicentric study, although the geometric mean was lower than in patients with L-HES (3406 vs 12,979 pg/mL respectively, p=0.02) [137].
Clinical implications
Activation of the TARC/CCL17-CCR4 axis, as reflected by elevated sTARC/CCL17 levels, may provide clues to the differential diagnosis of certain inflammatory disorders, predict disease severity, and/or help monitor disease activity (Table 3). In certain instances, this axis plays a pathogenic role, either because TARC/CCL17 is a key factor eliciting inflammation or because aberrant cells that drive pathogenesis express CCR4, and therefore represents a potential therapeutic target.
TARC as a biomarker for diagnosis, disease activity, and prediction of disease severity and treatment responses
Atopic dermatitis
In AD patients, an early study showed that sTARC/CCL17 correlated with disease activity (assessed by Scoring AD index, SCORAD) and AEC [75]. A prospective study conducted on a large cohort (n=320) of adults with AD highlighted the accuracy of sTARC/CCL17 to monitor disease severity given the positive relationship between this chemokine and clinical skin scores [7]. Moreover, elevated sTARC/CCL17 at presentation could also predict a more severe course, although this remains to be firmly established given some observed heterogeneity in results so far [7].
DRESS
In patients with severe drug reactions, elevated sTARC/CCL17 has been shown to be a discriminating factor for diagnosis of DRESS rather than Steven-Johnson syndrome and maculopapular erythema [83]. In patients with DRESS, sTARC/CCL17 correlates with the severity of skin manifestations at onset and decreases together with skin healing and normalization of serum IL-5 [82].
Bullous pemphigoid
A positive correlation has been observed between AEC and sTARC/CCL17 as well as disease activity, indirectly suggesting that sTARC/CCL17 could also correlate with disease activity [86]. In this line, sTARC/CCL17 levels were shown to correlate with the BP Disease Area Index score as well as urticaria/erythema scores in a series of 20 BP patients [138]. Serum TARC/CCL17 may actually be a better marker of disease activity than anti-BP180 autoantibodies, as fluctuations occurred earlier in patients experiencing disease flares in a recent study [138].
Senile erythroderma
Although a positive correlation has been observed between IgE and sTARC/CCL17 in patients with both idiopathic and secondary senile erythroderma [88], sTARC/CCL17 was more markedly elevated in patients with chronic idiopathic erythroderma (predominantly male) in one study, leading the authors to propose the use of a sTARC/IgE ratio to distinguish this patient sub-group from elderly patients with AD, showing a sensitivity of 80% and a specificity of 95% when the ratio is superior to 7.24 [89].
Chronic obstructive pulmonary disease
TARC/CCL17 was an independent predictive biomarker for the rapid decline in forced expiratory volume in one second (FEV1) in stable patients with COPD [139].
EGPA
sTARC/CCL17 is elevated in EGPA and correlates with disease activity [115]. Unfortunately, neither eotaxin-3/CCL26 nor TARC/CCL17 had sufficient accuracy for relapse-prediction in previously treated patients [116]. In addition, neither chemokine was useful to distinguish patients with ANCA-negative EGPA from those with HES presenting with a history of asthma and sinusitis [117].
ABPA
sTARC/CCL17 was shown to be more reliable than total IgE and A. fumigatus-specific IgE in serum for the diagnosis of ABPA in patients with CF and helped discriminate this condition from simple colonization or sensitization to A. fumigatus where levels are normal [112]. In addition, the rise in sTARC/CCL17 precedes that of IgE during disease development, offering the potential for early detection and management of this debilitating condition [140]. Higher sTARC/CCL17 levels may predict more severe disease as the levels of this chemokine correlated negatively with lung function in CF patients with ABPA [112].
Acute eosinophilic pneumonia
In patients with acute lung injury (ALI), sTARC/CCL17 levels accurately differentiate patients with severe forms of AEP from those with acute interstitial pneumonia, pneumonia-associated ALI/ARDS, and patients with alveolar hemorrhage. In fact, among several candidate biomarkers (including eotaxin-1/CCL11, Krebs von den Lungen-6 (KL-6) and surfactant protein-D), sTARC/CCL17 had the largest AUC (1.00, 95% CI 1.00 to 1.00) with a concentration threshold from 6259 to 7040 pg/mL [120]. Furthermore, sTARC/CCL17 levels correlated with those in BALF during active disease and decreased in parallel with regression of symptoms [120].
T cell lymphoma
In MF and SS, CCR4+ cell numbers increase in parallel with disease progression [128], and higher expression of CCR3 and CCR4 by lymphomatous cells in skin samples is associated with poor survival [141]. In MF, sTARC/CCL17 were significantly higher in tumor stage than in patch or plaque stage [127]. CCR4 expression by malignant cells is also associated with a poor prognosis in patients with ATLL and PTCL-U [129, 142].
Hypereosinophilic syndrome
In patients presenting with persistent unexplained HE, markedly elevated sTARC/CCL17 levels are associated with L-HES whereas normal values are observed in patients with no evidence for underlying Th2-driven pathogenesis [136]. In a recent study on a large cohort of HES patients, our group determined that a threshold value of 3000 pg/ml should raise suspicion of L-HES [143] although similarly elevated levels were also observed in patients with I-HES presenting clinically as eosinophilic dermatitis. A rise in sTARC/CCL17 could also be an early marker heralding a disease flare in patients with episodic angioedema with eosinophilia associated with CD3-CD4+ T cells [95].
Furthermore, sTARC/CCL17 levels may help predict treatment responses in HES. In a large multi-center retrospective study, the geometric mean sTARC/CCL17 level was significantly higher in CS-responsive patients compared to non-responders (979 vs 242 pg/mL, p=0.01) [137]. In another study evaluating the efficacy of mepolizumab in patients with FIP1L1-PDGFRA-negative HES, a suboptimal hematological response to mepolizumab was observed in patients with elevated sTARC/CCL17 levels, whether or not they had L-HES [144].
The TARC/CCL17-CCR4 pathway as a therapeutic target in eosinophil-associated disorders
To date, two main approaches have been employed to target CCR4-positive cells and/or antagonize the actions of TARC/CCL17 and MDC/CCL22. The first one is represented by a MoAb specifically targeting the extracellular portion of CCR4, namely, mogamulizumab (KW0761). It is a defucosylated humanized IgG1 kappa MoAb that destroys CCR4-positive cells through ADCC and is approved for the treatment of certain T cell neoplasms [8, 145]. Of note, mogamulizumab administration can be associated with occurrence of severe skin reactions (e.g., SJS), probably as a consequence of CCR4+ Treg depletion [146]. Other MoAbs targeting different regions and presumably different conformations of CCR4 have also been designed with the potential to specifically interfere with TARC/CCL17 or MDC/CCL22 activities [68]. The second approach consists in small-molecule CCR4 antagonists [61]. Despite encouraging data from pre-clinical models, none of these small molecules have been registered to date [145].
Studies investigating the functional and clinical impact of targeting the TARC/CCL17-CCR4 axis have been conducted in murine models and in humans for several of the aforementioned eosinophil-associated diseases (Table 4).
Table 4.
Clinical trials evaluating anti-CCR4 agents in healthy subjects, non-malignant and malignant disorders
| NCT number | Subjects/disease | Agent/drug class | Phase | Status | Results’ summarya |
|---|---|---|---|---|---|
| Non-malignant disorders | |||||
| NCT01371812 | HC | GSK2239633/small molecule | Phase I | Terminated | Low CCR4 blockade with the highest dose, not considered to be sufficiently effective for further development [200] |
| NCT01514981 | AA | AMG 761/MoAb (mogamulizumab) | Phase I | Terminated | Prematurely terminated, frequent cutaneous side effects (not severe) (8 events in 18 subjects) |
| NCT04271514 | AD and HC | RPT193/small molecule | Phase I | Recruiting | – |
| Malignant disorders | |||||
| NCT00888927 | Previously treated CTCL |
KW-0761/MoAb (mogamulizumab) |
Phase I/II | Terminated |
No dose-limiting toxicity was observed. Among 38 evaluable patients, ORR was 36.8% [201] |
| NCT01728805 | Relapsed or refractory CTCL | KW-0761/MoAb (mogamulizumab) | Phase III | Terminated |
Higher PFS in KW-0761 vs vorinostat group (median 7.7 months [95% CI 5.7–10.3] vs 3.1 months [2.9–4.1] respectively) [154] |
| NCT00355472 | Relapsed ATLL or PTCL | KW-0761/MoAb (mogamulizumab) | Phase I | Terminated | No dose-limiting toxicity was observed. |
| NCT01192984 | Relapsed or refractory peripheral T/NK-cell Lymphoma | KW-0761/MoAb (mogamulizumab) | Phase II | Terminated | ORR was 35% (95% CI, 20% to 53%), median PFS was 3.0 months (95% CI, 1.6 to 4.9 months) [202] |
| NCT01611142 | Relapsed or refractory PTCL | KW-0761/MoAb (mogamulizumab) | Phase II | Terminated | ORR was 11.4% (95% CI: 3.2–26.7%), disease control (SD or better) rate was 45.7% [203] |
| NCT00920790 | Relapsed ATLL | KW-0761/MoAb (mogamulizumab) | Phase II | Terminated | ORR was 50% (95% CI, 30–70%), median PFS was 5.2 months and median OS 13.7 months [204] |
| NCT01626664 | Relapsed or refractory ATLL | KW-0761/MoAb (mogamulizumab) | Phase II | Terminated | ORR was 14.9% in the KW-0761 group vs 0% with IC regimen [205] |
| NCT03602157 | Relapsed or refractory CD30+ Hodgkin Lymphoma and Cutaneous T-Cell Lymphoma | CAR-T Cells Expressing CD30, CAR and CCR4 | Phase I | Recruiting | – |
AA allergic asthma, AD atopic dermatitis, ATLL adult T cell leukemia/lymphoma, CAR chimeric antigen receptor, CTCL cutaneous T cell lymphoma, HC healthy controls, IC investigator choice, MoAb monoclonal antibody, ORR overall response rate, PTCL peripheral T cell lymphoma, SD stable disease
aIf no citation indicated, data were collected from clinicaltrials.gov
Atopic dermatitis
In an ovalbumin-sensitized mouse model, the CCR4 antagonist compound 22 reduced AD-like lesions as well as CCR4+ T cell infiltrates in the skin [147]. In a canine model of AD however, another antagonist (AZ445) was unable to significantly reduce skin lesions compared to CS, although CCR4+ T cell numbers were locally reduced [148]. Similar antagonists are in development for humans [149]. Of note, the histamine H4 receptor antagonist (ZPL-3893787) which allegedly reduces TARC/CCL17 synthesis [150] was tested in subjects with AD in a randomized, double-blind, placebo-controlled trial and was shown to reduce the SCORAD score and eczema area and severity index (EASI), but not pruritus [151].
Allergic asthma
To date, no therapy targeting TARC/CCL17 or CCR4 has been reported effective in human AA. In fact, among CCR4 antagonists shown to reduce allergic inflammation in mice, the only one tested in a phase I study in humans (GSK2239633) failed to induce sufficient CCR4 blockade at the highest dosing regimen [145]. Another phase I trial (NCT01514981) conducted with mogamulizumab was terminated prematurely due to drug-related adverse events. Finally, concerns have been raised regarding CCR4 blockade in asthma, as Tregs also express this receptor and are reported to colonize lung tissue and to be functional in the effector phase (“recall”) of allergic inflammation in murine models and in humans following segmental allergen challenge [152, 153].
T cell lymphoma
Mogamulizumab was evaluated in a phase III randomized controlled trial in comparison with vorinostat in relapsed/refractory MF and SS. Patients in the mogamulizumab arm had significantly longer progression-free survival (PFS) compared to vorinostat (median PFS of 7.7 months versus 3.1 months respectively, HR 0.53 with 95% CI 0.41–0.59) [154]. Mogamulizumab has also shown efficacy in ATLL when combined with intensive chemotherapy [155]. Indeed, its addition to background treatment resulted in improved PFS and overall survival, and it is now approved by Japanese authorities in newly diagnosed aggressive ATLL in combination with intensive chemotherapy [156].
L-HES
Ledoult and colleagues recently reported that circulating CD3-CD4+ cells bear a Th2 chemokine receptor phenotype ex vivo defined as CCR4+CCR6− in twenty patients with L-HES [157]. This phenotype was also expressed by 6 to 35% of CD3+CD4+ T cells from these patients and was not altered by CS therapy. These results have potential therapeutic implications as mogamulizumab could destroy the clonal IL-5-producing T cells that drive the disease.
Conclusion and perspectives
Both pre-clinical data and the clinical observations described herein firmly establish the intimate link between the TARC/CCL17-CCR4 axis, type 2 immunity, and eosinophilic inflammation. As such, TARC/CCL17 represents a useful biomarker for diagnosis and assessment of disease activity for several allegedly T cell-driven eosinophilic disorders and may also help predict more severe disease forms and/or treatment responses. Furthermore, overexpression/activation of this axis in these disorders makes it an appealing therapeutic target, as illustrated by the successful use of anti-CCR4 MoAb in certain T cell malignancies. Unfortunately, this approach has not yet produced results in the more common type 2 disorders such as AD and AA, and the potential impact on CCR4-expressing Tregs is a subject of concern. Future studies focusing on the precise role played by TARC/CCL17 in various eosinophilic conditions, mechanisms involved in its overexpression, CCR4 isoforms, and downstream signaling pathways will help determine whether the TARC/CCL17-CCR4 axis represents an interesting therapeutic target in non-malignant disorders.
AA allergic asthma, CLA cutaneous lymphocyte antigen, DC dendritic cell
Code availability
Not applicable.
Abbreviations
- AA
Allergic asthma
- ABPA
Allergic bronchopulmonary aspergillosis
- ADCC
Antibody-dependent cell cytotoxicity
- AEC
Absolute eosinophil count
- AEP
Acute eosinophilic pneumonia
- AITL
Angioimmunoblastic T cell lymphoma
- ALI
Acute lung injury
- ANCA
Anti-neutrophil cytoplasmic antibody
- ARDS
Acute respiratory distress syndrome
- ATLL
Adult T cell leukemia/lymphoma
- AUC
Area under the curve
- BALF
Bronchoalveolar lavage fluid
- BM
Bone marrow
- BP
Bullous pemphigoid
- CC
CC-chemokine
- CCR
CC-chemokine receptor
- CEP
Chronic eosinophilic pneumonia
- CF
Cystic fibrosis
- CLA
Cutaneous lymphocyte antigen
- CRSwNP
Chronic rhinosinusitis with nasal polyps
- CRTH2
Chemoattractant receptor-homologous molecule expressed on Th2 cells
- CS
Corticosteroid
- DC
Dendritic cell
- DRESS
Drug rash with eosinophilia and systemic symptoms
- EGPA
Eosinophilic granulomatosis with polyangiitis
- FEV1
Forced expired volume in one second
- FIP1L1
Fip 1-like 1
- GPCR
G protein-coupled receptor
- HC
Healthy control
- HDM
House dust mite
- HE
Hypereosinophilia
- hEoP
Human eosinophil progenitor
- HES
Hypereosinophilic syndrome
- HHV
Human herpes virus
- ICOG-Eo
International Cooperative Working Group on Eosinophil Disorders
- IFN
Interferon
- IHC
Immunohistochemistry
- IL
Interleukin
- ILC2
Type 2 innate lymphoid cell
- LC
Langerhans cell
- LN
Lymph node
- MDC
Macrophage-derived chemokine
- MF
Mycosis fungoides
- MoAb
Monoclonal antibody
- NFkB
Nuclear factor-kappa B
- PBMC
Peripheral blood mononuclear cell
- PDGFRA
Platelet-derived growth factor receptor A
- PFS
Progression free survival
- PGD2
Prostaglandin D2
- PTCL
Peripheral T cell lymphoma
- PI3K
Phosphatidylinositol 3-kinase
- SCORAD
Scoring atopic dermatitis
- SJS
Stevens-Johnson syndrome
- SS
Sézary syndrome
- STAT
Signal transducer and activator of transcription
- TARC
Thymus and activation-regulated chemokine
- TGF
Transforming growth factor
- TK
Tyrosine kinase
- TNF
Tumor necrosis factor
- Treg
Regulatory T cell
- TSLP
Thymic stromal lymphopoietin
- VCAM
Vascular cell adhesion molecule
Funding
FNRS (National Fund for Scientific Research) grant n°1/A/094/21f (JC) and F 5/4/150/5 (FR)
Data Availability
Not applicable.
Declarations
Competing interests
FR has received consultancy fees from GlaxoSmithKline and AstraZeneca, and royalties from UpToDate.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Genovese G, Di Zenzo G, Cozzani E, Berti E, Cugno M, Marzano AV. New insights into the pathogenesis of bullous pemphigoid: 2019 Update. Front Immunol. 2019;10:1506. doi: 10.3389/fimmu.2019.01506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yamada Y, Rothenberg ME, Lee AW, Akei HS, Brandt EB, Williams DA, Cancelas JA. The FIP1L1-PDGFRA fusion gene cooperates with IL-5 to induce murine hypereosinophilic syndrome (HES)/chronic eosinophilic leukemia (CEL)–like disease. Blood. 2006;107:4071–4079. doi: 10.1182/blood-2005-08-3153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Molfino NA, Gossage D, Kolbeck R, Parker JM, Geba GP. Molecular and clinical rationale for therapeutic targeting of interleukin-5 and its receptor. Clin Exp Allergy. 2012;42:712–737. doi: 10.1111/j.1365-2222.2011.03854.x. [DOI] [PubMed] [Google Scholar]
- 4.Islam SA, Luster AD. T cell homing to epithelial barriers in allergic disease. Nat Med. 2012;18:705–715. doi: 10.1038/nm.2760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Imai T, Nagira M, Takagi S, Kakizaki M, Nishimura M, Wang J, Gray PW, Matsushima K, Yoshie O. Selective recruitment of CCR4-bearing Th2 cells toward antigen-presenting cells by the CC chemokines thymus and activation-regulated chemokine and macrophage-derived chemokine. Int Immunol. 1999;11:81–88. doi: 10.1093/intimm/11.1.81. [DOI] [PubMed] [Google Scholar]
- 6.Yoshie O, Matsushima K. CCR4 and its ligands: from bench to bedside. Int Immunol. 2015;27:11–20. doi: 10.1093/intimm/dxu079. [DOI] [PubMed] [Google Scholar]
- 7.Landheer J, de Bruin-Weller M, Boonacker C, Hijnen D, Bruijnzeel-Koomen C, Röckmann H. Utility of serum thymus and activation-regulated chemokine as a biomarker for monitoring of atopic dermatitis severity. J Am Acad Dermatol. 2014;71:1160–1166. doi: 10.1016/j.jaad.2014.07.031. [DOI] [PubMed] [Google Scholar]
- 8.Ollila TA, Sahin I, Olszewski AJ. Mogamulizumab: a new tool for management of cutaneous T-cell lymphoma. OncoTargets Ther. 2019;12:1085–1094. doi: 10.2147/OTT.S165615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Abbas A, Lichtman A, Pillai S. Cell. Mol. Immunol. 9. Philadelphia: Elsevier; 2018. Chapter 20: allergy; pp. 437–456. [Google Scholar]
- 10.Hazenberg MD, Spits H. Human innate lymphoid cells. Blood. 2014;124:700–709. doi: 10.1182/blood-2013-11-427781. [DOI] [PubMed] [Google Scholar]
- 11.Gurram RK, Zhu J. Orchestration between ILC2s and Th2 cells in shaping type 2 immune responses. Cell Mol Immunol. 2019;16:225–235. doi: 10.1038/s41423-019-0210-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Paul WE, Zhu J. How are TH2-type immune responses initiated and amplified? Nat Rev Immunol. 2010;10:225–235. doi: 10.1038/nri2735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Walker JA, McKenzie ANJ. TH2 cell development and function. Nat Rev Immunol. 2018;18:121–133. doi: 10.1038/nri.2017.118. [DOI] [PubMed] [Google Scholar]
- 14.Islam SA, Chang DS, Colvin RA, Byrne MH, McCully ML, Moser B, Lira SA, Charo IF, Luster AD. Mouse CCL8, a CCR8 agonist, promotes atopic dermatitis by recruiting IL-5+ TH2 cells. Nat Immunol. 2011;12:167–177. doi: 10.1038/ni.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Komai-Koma M, Xu D, Li Y, McKenzie ANJ, McInnes IB, Liew FY. IL-33 is a chemoattractant for human Th2 cells. Eur J Immunol. 2007;37:2779–2786. doi: 10.1002/eji.200737547. [DOI] [PubMed] [Google Scholar]
- 16.Galli SJ, Tsai M, Piliponsky AM. The development of allergic inflammation. Nature. 2008;454:445–454. doi: 10.1038/nature07204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mori Y, Iwasaki H, Kohno K, et al. Identification of the human eosinophil lineage-committed progenitor: revision of phenotypic definition of the human common myeloid progenitor. J Exp Med. 2009;206:183–193. doi: 10.1084/jem.20081756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fulkerson PC. Transcription factors in eosinophil development and as therapeutic targets. Front Med. 2017;4:115. doi: 10.3389/fmed.2017.00115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Klion AD, Ackerman SJ, Bochner BS. Contributions of eosinophils to human health and disease. Annu Rev Pathol Mech Dis. 2020;15:179–209. doi: 10.1146/annurev-pathmechdis-012419-032756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nussbaum JC, Van Dyken SJ, von Moltke J, et al. Type 2 innate lymphoid cells control eosinophil homeostasis. Nature. 2013;502:245–248. doi: 10.1038/nature12526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Molofsky AB, Nussbaum JC, Liang H-E, Van Dyken SJ, Cheng LE, Mohapatra A, Chawla A, Locksley RM. Innate lymphoid type 2 cells sustain visceral adipose tissue eosinophils and alternatively activated macrophages. J Exp Med. 2013;210:535–549. doi: 10.1084/jem.20121964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kopf M, Brombacher F, Hodgkin PD, et al. IL-5-deficient mice have a developmental defect in CD5+ B-1 cells and lack eosinophilia but have normal antibody and cytotoxic T cell responses. Immunity. 1996;4:15–24. doi: 10.1016/S1074-7613(00)80294-0. [DOI] [PubMed] [Google Scholar]
- 23.Marichal T, Mesnil C, Bureau F (2017) Homeostatic eosinophils: characteristics and functions. Front Med. 10.3389/fmed.2017.00101 [DOI] [PMC free article] [PubMed]
- 24.Ravensberg AJ, Ricciardolo FLM, van Schadewijk A, Rabe KF, Sterk PJ, Hiemstra PS, Mauad T. Eotaxin-2 and eotaxin-3 expression is associated with persistent eosinophilic bronchial inflammation in patients with asthma after allergen challenge. J Allergy Clin Immunol. 2005;115:779–785. doi: 10.1016/j.jaci.2004.11.045. [DOI] [PubMed] [Google Scholar]
- 25.Conroy DM, Williams TJ. Eotaxin and the attraction of eosinophils to the asthmatic lung. Respir Res. 2001;2:150–156. doi: 10.1186/rr52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Alblowi J, Tian C, Siqueira MF, Kayal RA, McKenzie E, Behl Y, Gerstenfeld L, Einhorn TA, Graves DT. Chemokine expression is upregulated in chondrocytes in diabetic fracture healing. Bone. 2013;53:294–300. doi: 10.1016/j.bone.2012.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cheng LE, Sullivan BM, Retana LE, Allen CDC, Liang H-E, Locksley RM. IgE-activated basophils regulate eosinophil tissue entry by modulating endothelial function. J Exp Med. 2015;212:513–524. doi: 10.1084/jem.20141671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Singh D, Ravi A, Southworth T. CRTH2 antagonists in asthma: current perspectives. Clin Pharmacol Adv Appl. 2017;9:165–173. doi: 10.2147/CPAA.S119295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Peinhaupt M, Sturm EM, Heinemann A. Prostaglandins and their receptors in eosinophil function and as therapeutic Targets. Front Med. 2017;4:104. doi: 10.3389/fmed.2017.00104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fujishima H, Fukagawa K, Okada N, Takano Y, Tsubota K, Hirai H, Nagata K, Matsumoto K, Saito H. Prostaglandin D2 induces chemotaxis in eosinophils via its receptor CRTH2 and eosinophils may cause severe ocular inflammation in patients with allergic conjunctivitis. Cornea. 2005;24:S66–S70. doi: 10.1097/01.ico.0000178733.42921.4c. [DOI] [PubMed] [Google Scholar]
- 31.Wong CK, Hu S, Cheung PFY, Lam CWK. Thymic stromal lymphopoietin induces chemotactic and prosurvival effects in eosinophils: implications in allergic inflammation. Am J Respir Cell Mol Biol. 2010;43:305–315. doi: 10.1165/rcmb.2009-0168OC. [DOI] [PubMed] [Google Scholar]
- 32.Liu LY, Jarjour NN, Busse WW, Kelly EAB. Chemokine receptor expression on human eosinophils from peripheral blood and bronchoalveolar lavage fluid after segmental antigen challenge. J Allergy Clin Immunol. 2003;112:556–562. doi: 10.1016/S0091-6749(03)01798-6. [DOI] [PubMed] [Google Scholar]
- 33.Yi S, Zhai J, Niu R, et al. Eosinophil recruitment is dynamically regulated by interplay among lung dendritic cell subsets after allergen challenge. Nat Commun. 2018;9:3879. doi: 10.1038/s41467-018-06316-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nagase H, Kudo K, Izumi S, Ohta K, Kobayashi N, Yamaguchi M, Matsushima K, Morita Y, Yamamoto K, Hirai K. Chemokine receptor expression profile of eosinophils at inflamed tissue sites: decreased CCR3 and increased CXCR4 expression by lung eosinophils. J Allergy Clin Immunol. 2001;108:563–569. doi: 10.1067/mai.2001.118292. [DOI] [PubMed] [Google Scholar]
- 35.Borchers MT, Ansay T, DeSalle R, Daugherty BL, Shen H, Metzger M, Lee NA, Lee JJ. In vitro assessment of chemokine receptor-ligand interactions mediating mouse eosinophil migration. J Leukoc Biol. 2002;71:1033–1041. doi: 10.1189/jlb.71.6.1033. [DOI] [PubMed] [Google Scholar]
- 36.Bochner BS, Bickel CA, Taylor ML, MacGlashan DW, Gray PW, Raport CJ, Godiska R. Macrophage-derived chemokine induces human eosinophil chemotaxis in a CC chemokine receptor 3– and CC chemokine receptor 4–independent manner. J Allergy Clin Immunol. 1999;103:527–532. doi: 10.1016/S0091-6749(99)70481-1. [DOI] [PubMed] [Google Scholar]
- 37.Wilkerson EM, Johansson MW, Hebert AS, Westphall MS, Mathur SK, Jarjour NN, Schwantes EA, Mosher DF, Coon JJ. The peripheral blood eosinophil proteome. J Proteome Res. 2016;15:1524–1533. doi: 10.1021/acs.jproteome.6b00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Larose M-C, Archambault A-S, Provost V, Laviolette M, Flamand N (2017) Regulation of eosinophil and group 2 innate lymphoid cell trafficking in asthma. Front Med 4:136 [DOI] [PMC free article] [PubMed]
- 39.Lee J, Rosenberg H (2013) Eosinophil secretory functions. In: Eosinophils Health Dis. Elsevier, pp 229–275
- 40.Ramirez GA, Yacoub M-R, Ripa M, Mannina D, Cariddi A, Saporiti N, Ciceri F, Castagna A, Colombo G, Dagna L (2018) Eosinophils from physiology to disease: a comprehensive review. Biomed Res Int 2018:1–28 [DOI] [PMC free article] [PubMed]
- 41.Curtis C, Ogbogu P. Hypereosinophilic syndrome. Clin Rev Allergy Immunol. 2016;50:240–251. doi: 10.1007/s12016-015-8506-7. [DOI] [PubMed] [Google Scholar]
- 42.Valent P, Klion AD, Horny H-P, et al. Contemporary consensus proposal on criteria and classification of eosinophilic disorders and related syndromes. J Allergy Clin Immunol. 2012;130:607–612.e9. doi: 10.1016/j.jaci.2012.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chusid M, Dale D, West B, Wolff S. The hypereosinophilic syndrome: analysis of fourteen cases with review of the literature. Medicine (Baltimore) 1975;54(1):1–27. doi: 10.1097/00005792-197501000-00001. [DOI] [PubMed] [Google Scholar]
- 44.Roufosse F, Weller PF. Practical approach to the patient with hypereosinophilia. J Allergy Clin Immunol. 2010;126:39–44. doi: 10.1016/j.jaci.2010.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cools J, DeAngelo DJ, Gotlib J, et al. A tyrosine kinase created by fusion of the PDGFRA and FIP1L1 genes as a therapeutic target of imatinib in idiopathic hypereosinophilic syndrome. N Engl J Med. 2003;348:1201–1214. doi: 10.1056/NEJMoa025217. [DOI] [PubMed] [Google Scholar]
- 46.Cogan E, Schandené L, Crusiaux A, Cochaux P, Velu T, Goldman M. Brief report: clonal proliferation of type 2 helper T cells in a man with the hypereosinophilic syndrome. N Engl J Med. 1994;330:535–538. doi: 10.1056/NEJM199402243300804. [DOI] [PubMed] [Google Scholar]
- 47.Klion AD. How I treat hypereosinophilic syndromes. Blood. 2015;126:1069–1077. doi: 10.1182/blood-2014-11-551614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Imai T, Yoshida T, Baba M, Nishimura M, Kakizaki M, Yoshie O. Molecular cloning of a novel T cell-directed CC chemokine expressed in thymus by signal sequence trap using Epstein-Barr virus vector. J Biol Chem. 1996;271:21514–21521. doi: 10.1074/jbc.271.35.21514. [DOI] [PubMed] [Google Scholar]
- 49.Imai T, Baba M, Nishimura M, Kakizaki M, Takagi S, Yoshie O. The T cell-directed CC chemokine TARC is a highly specific biological ligand for CC chemokine receptor 4. J Biol Chem. 1997;272:15036–15042. doi: 10.1074/jbc.272.23.15036. [DOI] [PubMed] [Google Scholar]
- 50.Nomiyama H, Imai T, Kusuda J, Miura R, Callen DF, Yoshie O. Assignment of the human CC chemokine gene TARC (SCYA17) to chromosome 16q13. Genomics. 1997;40:211–213. doi: 10.1006/geno.1996.4552. [DOI] [PubMed] [Google Scholar]
- 51.Alferink J, Lieberam I, Reindl W, et al. Compartmentalized production of CCL17 in vivo. J Exp Med. 2003;197:585–599. doi: 10.1084/jem.20021859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Achuthan A, Cook AD, Lee M-C, et al. Granulocyte macrophage colony-stimulating factor induces CCL17 production via IRF4 to mediate inflammation. J Clin Invest. 2016;126:3453–3466. doi: 10.1172/JCI87828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hsu AT, Lupancu TJ, Lee M-C, Fleetwood AJ, Cook AD, Hamilton JA, Achuthan A. Epigenetic and transcriptional regulation of IL4-induced CCL17 production in human monocytes and murine macrophages. J Biol Chem. 2018;293:11415–11423. doi: 10.1074/jbc.RA118.002416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Medoff BD, Seung E, Hong S, Thomas SY, Sandall BP, Duffield JS, Kuperman DA, Erle DJ, Luster AD. CD11b+ myeloid cells are the key mediators of Th2 cell homing into the airway in allergic inflammation. J Immunol. 2009;182:623–635. doi: 10.4049/jimmunol.182.1.623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wirnsberger G, Hebenstreit D, Posselt G, Horejs-Hoeck J, Duschl A. IL-4 induces expression of TARC/CCL17via two STAT6 binding sites. Eur J Immunol. 2006;36:1882–1891. doi: 10.1002/eji.200635972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kakinuma T, Nakamura K, Wakugawa M, Yano S, Saeki H, Torii H, Komine M, Asahina A, Tamaki K. IL-4, but not IL-13, modulates TARC (thymus and activation-regulated chemokine)/CCL17 and IP-10 (interferon-induced protein of 10kDa)/CXCL10 release by TNF-α and IFN-γ in HaCaT cell line. Cytokine. 2002;20:1–6. doi: 10.1006/cyto.2002.1965. [DOI] [PubMed] [Google Scholar]
- 57.Sekiya T, Miyamasu M, Imanishi M, et al. Inducible expression of a Th2-type CC chemokine thymus- and activation-regulated chemokine by human bronchial epithelial cells. J Immunol Baltim Md 1950. 2000;165:2205–2213. doi: 10.4049/jimmunol.165.4.2205. [DOI] [PubMed] [Google Scholar]
- 58.Berin MC, Eckmann L, Broide DH, Kagnoff MF. Regulated production of the T helper 2–Type T-cell chemoattractant TARC by human bronchial epithelial cells in vitro and in human lung xenografts. Am J Respir Cell Mol Biol. 2001;24:382–389. doi: 10.1165/ajrcmb.24.4.4360. [DOI] [PubMed] [Google Scholar]
- 59.Nakayama T, Hieshima K, Nagakubo D, Sato E, Nakayama M, Kawa K, Yoshie O. Selective induction of Th2-attracting chemokines CCL17 and CCL22 in human B cells by latent membrane protein 1 of Epstein-Barr virus. J Virol. 2004;78:1665–1674. doi: 10.1128/JVI.78.4.1665-1674.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Fujii-Maeda S, Kajiwara K, Ikizawa K, Shinazawa M, Yu B, Koga T, Furue M, Yanagihara Y. Reciprocal regulation of thymus and activation-regulated chemokine/macrophage-derived chemokine production by interleukin (IL)-4/IL-13 and interferon-γ in HaCaT keratinocytes is mediated by alternations in E-cadherin distribution. J Invest Dermatol. 2004;122:20–28. doi: 10.1046/j.0022-202X.2003.22103.x. [DOI] [PubMed] [Google Scholar]
- 61.Solari R, Pease JE. Targeting chemokine receptors in disease – a case study of CCR4. Eur J Pharmacol. 2015;763:169–177. doi: 10.1016/j.ejphar.2015.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Imai T, Chantry D, Raport CJ, Wood CL, Nishimura M, Godiska R, Yoshie O, Gray PW. Macrophage-derived chemokine is a functional ligand for the cc chemokine receptor 4. J Biol Chem. 1998;273:1764–1768. doi: 10.1074/jbc.273.3.1764. [DOI] [PubMed] [Google Scholar]
- 63.Vulcano M, Albanesi C, Stoppacciaro A, et al. Dendritic cells as a major source of macrophage-derived chemokine/CCL22 in vitro and in vivo. Eur J Immunol. 2001;31:812–822. doi: 10.1002/1521-4141(200103)31:3<812::AID-IMMU812>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- 64.Yamashita U, Kuroda E. Regulation of macrophage-derived chemokine (MDC, CCL22) production. Crit Rev Immunol. 2002;22:105–114. doi: 10.1615/CritRevImmunol.v22.i2.10. [DOI] [PubMed] [Google Scholar]
- 65.Hashimoto S, Nakamura K, Oyama N, Kaneko F, Tsunemi Y, Saeki H, Tamaki K. Macrophage-derived chemokine (MDC)/CCL22 produced by monocyte derived dendritic cells reflects the disease activity in patients with atopic dermatitis. J Dermatol Sci. 2006;44:93–99. doi: 10.1016/j.jdermsci.2006.08.004. [DOI] [PubMed] [Google Scholar]
- 66.D’Ambrosio D, Albanesi C, Lang R, Girolomoni G, Sinigaglia F, Laudanna C. Quantitative differences in chemokine receptor engagement generate diversity in integrin-dependent lymphocyte adhesion. J Immunol. 2002;169:2303–2312. doi: 10.4049/jimmunol.169.5.2303. [DOI] [PubMed] [Google Scholar]
- 67.Mariani M, Lang R, Binda E, Panina-Bordignon P, D’Ambrosio D. Dominance of CCL22 over CCL17 in induction of chemokine receptor CCR4 desensitization and internalization on human Th2 cells. Eur J Immunol. 2004;34:231–240. doi: 10.1002/eji.200324429. [DOI] [PubMed] [Google Scholar]
- 68.Viney JM, Andrew DP, Phillips RM, Meiser A, Patel P, Lennartz-Walker M, Cousins DJ, Barton NP, Hall DA, Pease JE. Distinct conformations of the chemokine receptor CCR4 with implications for its targeting in allergy. J Immunol. 2014;192:3419–3427. doi: 10.4049/jimmunol.1300232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Lin R, Choi Y, Zidar DA, Walker JKL. β-arrestin-2–dependent signaling promotes CCR4–mediated chemotaxis of murine T-helper type 2 cells. Am J Respir Cell Mol Biol. 2018;58:745–755. doi: 10.1165/rcmb.2017-0240OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Weston CA, Rana BMJ, Cousins DJ. Differential expression of functional chemokine receptors on human blood and lung group 2 innate lymphoid cells. J Allergy Clin Immunol. 2019;143:410–413.e9. doi: 10.1016/j.jaci.2018.08.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Weidinger S, Beck LA, Bieber T, Kabashima K, Irvine AD. Atopic dermatitis. Nat Rev Dis Primer. 2018;4:1. doi: 10.1038/s41572-018-0001-z. [DOI] [PubMed] [Google Scholar]
- 72.Oldhoff JM, Darsow U, Werfel T, et al. Anti-IL-5 recombinant humanized monoclonal antibody (mepolizumab) for the treatment of atopic dermatitis. Allergy. 2005;60:693–696. doi: 10.1111/j.1398-9995.2005.00791.x. [DOI] [PubMed] [Google Scholar]
- 73.Kang EG, Narayana PK, Pouliquen IJ, Lopez MC, Ferreira-Cornwell MC, Getsy JA. Efficacy and safety of mepolizumab administered subcutaneously for moderate to severe atopic dermatitis. Allergy. 2020;75:950–953. doi: 10.1111/all.14050. [DOI] [PubMed] [Google Scholar]
- 74.Saeki H, Tamaki K. Thymus and activation regulated chemokine (TARC)/CCL17 and skin diseases. J Dermatol Sci. 2006;43:75–84. doi: 10.1016/j.jdermsci.2006.06.002. [DOI] [PubMed] [Google Scholar]
- 75.Kakinuma T, Nakamura K, Wakugawa M, et al. Thymus and activation-regulated chemokine in atopic dermatitis: serum thymus and activation-regulated chemokine level is closely related with disease activity. J Allergy Clin Immunol. 2001;107:535–541. doi: 10.1067/mai.2001.113237. [DOI] [PubMed] [Google Scholar]
- 76.Rubin LA, Nelson DL. The soluble interleukin-2 receptor: biology, function, and clinical application. Ann Intern Med. 1990;113:619–627. doi: 10.7326/0003-4819-113-8-619. [DOI] [PubMed] [Google Scholar]
- 77.Vestergaard C, Bang K, Gesser B, Yoneyama H, Matsushima K, Larsen CG. A Th2 chemokine, TARC, produced by keratinocytes may recruit CLA+CCR4+ lymphocytes into lesional atopic dermatitis skin. J Invest Dermatol. 2000;115:640–646. doi: 10.1046/j.1523-1747.2000.00115.x. [DOI] [PubMed] [Google Scholar]
- 78.Wakugawa M, Nakamura K, Kakinuma T, Onai N, Matsushima K, Tamaki K. CC chemokine receptor 4 expression on peripheral blood CD4+ T cells reflects disease activity of atopic dermatitis. J Invest Dermatol. 2001;117:188–196. doi: 10.1046/j.0022-202x.2001.01430.x. [DOI] [PubMed] [Google Scholar]
- 79.Jang M, Kim H, Kim Y, Choi J, Jeon J, Hwang Y, Kang JS, Lee WJ. The crucial role of IL-22 and its receptor in thymus and activation regulated chemokine production and T-cell migration by house dust mite extract. Exp Dermatol. 2016;25:598–603. doi: 10.1111/exd.12988. [DOI] [PubMed] [Google Scholar]
- 80.Zhang Y-Y, Wang A-X, Xu L, Shen N, Zhu J, Tu C-X. Characteristics of peripheral blood CD4+CD25+ regulatory T cells and related cytokines in severe atopic dermatitis. Eur J Dermatol. 2016;26:240–246. doi: 10.1684/ejd.2015.2709. [DOI] [PubMed] [Google Scholar]
- 81.Husain Z, Reddy BY, Schwartz RA. DRESS syndrome. J Am Acad Dermatol. 2013;68:693.e1–693.e14. doi: 10.1016/j.jaad.2013.01.033. [DOI] [PubMed] [Google Scholar]
- 82.Ogawa K, Morito H, Hasegawa A, et al. Identification of thymus and activation-regulated chemokine (TARC/CCL17) as a potential marker for early indication of disease and prediction of disease activity in drug-induced hypersensitivity syndrome (DIHS)/drug rash with eosinophilia and systemic symptoms (DRESS) J Dermatol Sci. 2013;69:38–43. doi: 10.1016/j.jdermsci.2012.10.002. [DOI] [PubMed] [Google Scholar]
- 83.Ogawa K, Morito H, Hasegawa A, et al. Elevated serum thymus and activation-regulated chemokine (TARC/CCL17) relates to reactivation of human herpesvirus 6 in drug reaction with eosinophilia and systemic symptoms (DRESS)/drug-induced hypersensitivity syndrome (DIHS) Br J Dermatol. 2014;171:425–427. doi: 10.1111/bjd.12948. [DOI] [PubMed] [Google Scholar]
- 84.Watanabe H. Recent advances in drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms. J Immunol Res. 2018;2018:1–10. doi: 10.1155/2018/5163129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Lin L, Hwang B-J, Culton DA, et al. Eosinophils mediate tissue injury in the autoimmune skin disease bullous pemphigoid. J Invest Dermatol. 2018;138:1032–1043. doi: 10.1016/j.jid.2017.11.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kakinuma T, Wakugawa M, Nakamura K, Hino H, Matsushima K, Tamaki K. High level of thymus and activation-regulated chemokine in blister fluid and sera of patients with bullous pemphigoid. Br J Dermatol. 2003;148:203–210. doi: 10.1046/j.1365-2133.2003.05066.x. [DOI] [PubMed] [Google Scholar]
- 87.Miyashiro D, Sanches JA. Erythroderma: a prospective study of 309 patients followed for 12 years in a tertiary center. Sci Rep. 2020;10:9774. doi: 10.1038/s41598-020-66040-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Nakano-Tahara M, Terao M, Nishioka M, Kitaba S, Murota H, Katayama I. T Helper 2 polarization in senile erythroderma with elevated levels of TARC and IgE. Dermatology. 2015;230:62–69. doi: 10.1159/000366502. [DOI] [PubMed] [Google Scholar]
- 89.Ohga Y, Bayaraa B, Imafuku S. Chronic idiopathic erythroderma of elderly men is an independent entity that has a distinct TARC/IgE profile from adult atopic dermatitis. Int J Dermatol. 2018;57:670–674. doi: 10.1111/ijd.13976. [DOI] [PubMed] [Google Scholar]
- 90.Murayama T, Nakamura K, Tsuchida T. Eosinophilic pustular folliculitis with extensive distribution: correlation of serum TARC levels and peripheral blood eosinophil numbers. Int J Dermatol. 2015;54:1071–1074. doi: 10.1111/ijd.12281. [DOI] [PubMed] [Google Scholar]
- 91.Zhang L, Qi R, Yang Y, Gao X, Chen H, Xiao T. Serum miR-125a-5p and CCL17 upregulated in chronic spontaneous urticaria and correlated with treatment response. Acta Derm Venereol. 2019;99:571–578. doi: 10.2340/00015555-3149. [DOI] [PubMed] [Google Scholar]
- 92.Tapia B, Morel E, Martín-Díaz M-A, Díaz R, Alves-Ferreira J, Rubio P, Padial A, Bellón T. Up-regulation of CCL17, CCL22 and CCR4 in drug-induced maculopapular exanthema. Clin Exp Allergy J Br Soc Allergy Clin Immunol. 2007;37:704–713. doi: 10.1111/j.1365-2222.2007.02699.x. [DOI] [PubMed] [Google Scholar]
- 93.Quaglino P, Caproni M, Antiga E, et al. Serum levels of the Th1 promoter IL-12 and the Th2 chemokine TARC are elevated in erythema multiforme and Stevens-Johnson syndrome/toxic epidermal necrolysis and correlate with soluble Fas ligand expression. An immunoenzymatic study from the Italian Group of Immunopathology. Dermatology. 2007;214:296–304. doi: 10.1159/000100880. [DOI] [PubMed] [Google Scholar]
- 94.Teraki Y, Taguchi R. Serum TARC levels correlate with disease activity in patients with non-episodic angioedema with eosinophilia. J Dermatol. 2014;41:858–860. doi: 10.1111/1346-8138.12596. [DOI] [PubMed] [Google Scholar]
- 95.Khoury P, Herold J, Alpaugh A, et al. Episodic angioedema with eosinophilia (Gleich syndrome) is a multilineage cell cycling disorder. Haematologica. 2015;100:300–307. doi: 10.3324/haematol.2013.091264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Chen R, Smith SG, Salter B, El-Gammal A, Oliveria JP, Obminski C, Watson R, O’Byrne PM, Gauvreau GM, Sehmi R. Allergen-induced increases in sputum levels of group 2 innate lymphoid cells in subjects with asthma. Am J Respir Crit Care Med. 2017;196:700–712. doi: 10.1164/rccm.201612-2427OC. [DOI] [PubMed] [Google Scholar]
- 97.Emma R, Morjaria JB, Fuochi V, Polosa R, Caruso M. Mepolizumab in the management of severe eosinophilic asthma in adults: current evidence and practical experience. Ther Adv Respir Dis. 2018;12:1–12. doi: 10.1177/1753466618808490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Vijayanand P, Durkin K, Hartmann G, et al. Chemokine receptor 4 plays a key role in T cell recruitment into the airways of asthmatic patients. J Immunol Baltim Md 1950. 2010;184:4568–4574. doi: 10.4049/jimmunol.0901342. [DOI] [PubMed] [Google Scholar]
- 99.Bochner BS, Hudson SA, Xiao HQ, Liu MC. Release of both CCR4-active and CXCR3-active chemokines during human allergic pulmonary late-phase reactions. J Allergy Clin Immunol. 2003;112:930–934. doi: 10.1016/j.jaci.2003.08.012. [DOI] [PubMed] [Google Scholar]
- 100.Pilette C, Francis JN, Till SJ, Durham SR. CCR4 ligands are up-regulated in the airways of atopic asthmatics after segmental allergen challenge. Eur Respir J. 2004;23:876–884. doi: 10.1183/09031936.04.00102504. [DOI] [PubMed] [Google Scholar]
- 101.Heijink IH, Marcel Kies P, van Oosterhout AJM, Postma DS, Kauffman HF, Vellenga E. Der p, IL-4, and TGF-β cooperatively induce EGFR-dependent TARC expression in airway epithelium. Am J Respir Cell Mol Biol. 2007;36:351–359. doi: 10.1165/rcmb.2006-0160OC. [DOI] [PubMed] [Google Scholar]
- 102.Perros F, Hoogsteden HC, Coyle AJ, Lambrecht BN, Hammad H. Blockade of CCR4 in a humanized model of asthma reveals a critical role for DC-derived CCL17 and CCL22 in attracting Th2 cells and inducing airway inflammation. Allergy. 2009;64:995–1002. doi: 10.1111/j.1398-9995.2009.02095.x. [DOI] [PubMed] [Google Scholar]
- 103.Staples KJ, Hinks TSC, Ward JA, Gunn V, Smith C, Djukanović R. Phenotypic characterization of lung macrophages in asthmatic patients: overexpression of CCL17. J Allergy Clin Immunol. 2012;130:1404–1412.e7. doi: 10.1016/j.jaci.2012.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Hirata H, Arima M, Cheng G, Honda K, Fukushima F, Yoshida N, Eda F, Fukuda T. Production of TARC and MDC by naive T cells in asthmatic patients. J Clin Immunol. 2002;23:12. doi: 10.1023/a:1021948214742. [DOI] [PubMed] [Google Scholar]
- 105.Jacobsen EA, Ochkur SI, Pero RS, Taranova AG, Protheroe CA, Colbert DC, Lee NA, Lee JJ. Allergic pulmonary inflammation in mice is dependent on eosinophil-induced recruitment of effector T cells. J Exp Med. 2008;205:699–710. doi: 10.1084/jem.20071840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Liu LY, Bates ME, Jarjour NN, Busse WW, Bertics PJ, Kelly EAB. Generation of Th1 and Th2 chemokines by human eosinophils: evidence for a critical role of TNF-α. J Immunol. 2007;179:4840–4848. doi: 10.4049/jimmunol.179.7.4840. [DOI] [PubMed] [Google Scholar]
- 107.Chvatchko Y, Hoogewerf AJ, Meyer A, Alouani S, Juillard P, Buser R, Conquet F, Proudfoot AEI, Wells TNC, Power CA. A key role for CC chemokine receptor 4 in lipopolysaccharide-induced endotoxic shock. J Exp Med. 2000;191:1755–1763. doi: 10.1084/jem.191.10.1755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Kawasaki S, Takizawa H, Yoneyama H, et al. Intervention of thymus and activation-regulated chemokine attenuates the development of allergic airway inflammation and hyperresponsiveness in mice. J Immunol. 2001;166:2055–2062. doi: 10.4049/jimmunol.166.3.2055. [DOI] [PubMed] [Google Scholar]
- 109.Conroy DM, Jopling LA, Lloyd CM, Hodge MR, Andrew DP, Williams TJ, Pease JE, Sabroe I. CCR4 blockade does not inhibit allergic airways inflammation. J Leukoc Biol. 2003;74:558–563. doi: 10.1189/jlb.0103030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Honjo A, Ogawa H, Azuma M, Tezuka T, Sone S, Biragyn A, Nishioka Y. Targeted reduction of CCR4+ cells is sufficient to suppress allergic airway inflammation. Respir Investig. 2013;51:241–249. doi: 10.1016/j.resinv.2013.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Tsybikov NN, Fefelova EV. Biomarker assessment in chronic rhinitis and chronic rhinosinusitis: endothelin-1, TARC/CCL17, neopterin, and. Allergy Asthma Proc. 2016;37:35–42. doi: 10.2500/aap.2016.37.3899. [DOI] [PubMed] [Google Scholar]
- 112.Hartl D, Latzin P, Zissel G, Krane M, Krauss-Etschmann S, Griese M. Chemokines indicate allergic bronchopulmonary aspergillosis in patients with cystic fibrosis. Am J Respir Crit Care Med. 2006;173:1370–1376. doi: 10.1164/rccm.200508-1271OC. [DOI] [PubMed] [Google Scholar]
- 113.Jennette JC, Falk RJ, Bacon PA, et al. 2012 Revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013;65:1–11. doi: 10.1002/art.37715. [DOI] [PubMed] [Google Scholar]
- 114.Gioffredi A, Maritati F, Oliva E, Buzio C. Eosinophilic granulomatosis with polyangiitis: an overview. Front Immunol. 2014;5:549. doi: 10.3389/fimmu.2014.00549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Dallos T, Heiland GR, Strehl J, et al. CCL17/thymus and activation-related chemokine in Churg-Strauss syndrome. Arthritis Rheum. 2010;62:3496–3503. doi: 10.1002/art.27678. [DOI] [PubMed] [Google Scholar]
- 116.Dejaco C, Oppl B, Monach P, et al. Serum biomarkers in patients with relapsing eosinophilic granulomatosis with polyangiitis (Churg-Strauss) PLoS One. 2015;10:e0121737. doi: 10.1371/journal.pone.0121737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Khoury P, Zagallo P, Talar-Williams C, Santos CS, Dinerman E, Holland NC, Klion AD. Serum biomarkers are similar in Churg-Strauss syndrome and hypereosinophilic syndrome. Allergy. 2012;67:1149–1156. doi: 10.1111/j.1398-9995.2012.02873.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Suzuki Y, Suda T. Eosinophilic pneumonia: a review of the previous literature, causes, diagnosis, and management. Allergol Int. 2019;68:413–419. doi: 10.1016/j.alit.2019.05.006. [DOI] [PubMed] [Google Scholar]
- 119.Miyazaki E, Nureki S, Fukami T, Shigenaga T, Ando M, Ito K, Ando H, Sugisaki K, Kumamoto T, Tsuda T. Elevated levels of thymus- and activation-regulated chemokine in bronchoalveolar lavage fluid from patients with eosinophilic pneumonia. Am J Respir Crit Care Med. 2002;165:1125–1131. doi: 10.1164/ajrccm.165.8.2106110. [DOI] [PubMed] [Google Scholar]
- 120.Miyazaki E, Nureki S, Ono E, Ando M, Matsuno O, Fukami T, Ueno T, Kumamoto T. Circulating thymus- and activation-regulated chemokine/CCL17 is a useful biomarker for discriminating acute eosinophilic pneumonia from other causes of acute lung injury. Chest. 2007;131:1726–1734. doi: 10.1378/chest.06-2596. [DOI] [PubMed] [Google Scholar]
- 121.Nureki S, Miyazaki E, Ando M, Kumamoto T, Tsuda T. CC chemokine receptor 4 ligand production by bronchoalveolar lavage fluid cells in cigarette-smoke-associated acute eosinophilic pneumonia. Clin Immunol. 2005;116:83–93. doi: 10.1016/j.clim.2005.03.001. [DOI] [PubMed] [Google Scholar]
- 122.Katoh S, Fukushima K, Matsumoto N, Matsumoto K, Abe K, Onai N, Matsushima K, Matsukura S. Accumulation of CCR4-expressing CD4+ T cells and high concentration of its ligands (TARC and MDC) in bronchoalveolar lavage fluid of patients with eosinophilic pneumonia. Allergy. 2003;58:518–523. doi: 10.1034/j.1398-9995.2003.00149.x. [DOI] [PubMed] [Google Scholar]
- 123.Nakagome K, Shoda H, Shirai T, Nishihara F, Soma T, Uchida Y, Sakamoto Y, Nagata M. Eosinophil transendothelial migration induced by the bronchoalveolar lavage fluid of acute eosinophilic pneumonia: eosinophil accumulation in AEP. Respirology. 2017;22:913–921. doi: 10.1111/resp.12982. [DOI] [PubMed] [Google Scholar]
- 124.Hristov AC, Tejasvi T, Wilcox RA. Mycosis fungoides and Sézary syndrome: 2019 update on diagnosis, risk-stratification, and management. Am J Hematol. 2019;94:1027–1041. doi: 10.1002/ajh.25577. [DOI] [PubMed] [Google Scholar]
- 125.Jawed SI, Myskowski PL, Horwitz S, Moskowitz A, Querfeld C. Primary cutaneous T-cell lymphoma (mycosis fungoides and Sézary syndrome) J Am Acad Dermatol. 2014;70:205.e1–205.e16. doi: 10.1016/j.jaad.2013.07.049. [DOI] [PubMed] [Google Scholar]
- 126.Tamaki K, Kakinuma T, Saeki H, et al. Serum levels of CCL17/TARC in various skin diseases. J Dermatol. 2006;33:300–302. doi: 10.1111/j.1346-8138.2006.00072.x. [DOI] [PubMed] [Google Scholar]
- 127.Kakinuma T, Sugaya M, Nakamura K, Kaneko F, Wakugawa M, Matsushima K, Tamaki K. Thymus and activation-regulated chemokine (TARC/CCL17) in mycosis fungoides: serum TARC levels reflect the disease activity of mycosis fungoides. J Am Acad Dermatol. 2003;48:23–30. doi: 10.1067/mjd.2003.132. [DOI] [PubMed] [Google Scholar]
- 128.Sugaya M, Morimura S, Suga H, Kawaguchi M, Miyagaki T, Ohmatsu H, Fujita H, Sato S. CCR4 is expressed on infiltrating cells in lesional skin of early mycosis fungoides and atopic dermatitis. J Dermatol. 2015;42:613–615. doi: 10.1111/1346-8138.12852. [DOI] [PubMed] [Google Scholar]
- 129.Ishida T, Inagaki H, Utsunomiya A, Takatsuka Y, Komatsu H, Iida S, Takeuchi G, Eimoto T, Nakamura S, Ueda R. CXC chemokine receptoR 3 and CC chemokine receptor 4 expression in T- cell and NK-cell lymphomas with special reference to clinicopathological significance for peripheral T-cell lymphoma, unspecified. Clin Cancer Res. 2004;10:5494–5500. doi: 10.1158/1078-0432.CCR-04-0371. [DOI] [PubMed] [Google Scholar]
- 130.Thielen C, Radermacher V, Trimeche M, Roufosse F, Goldman M, Boniver J, de Leval L. TARC and IL-5 expression correlates with tissue eosinophilia in peripheral T-cell lymphomas. Leuk Res. 2008;32:1431–1438. doi: 10.1016/j.leukres.2008.02.016. [DOI] [PubMed] [Google Scholar]
- 131.Yoshie O, Fujisawa R, Nakayama T, et al. Frequent expression of CCR4 in adult T-cell leukemia and human T-cell leukemia virus type 1–transformed T cells. Blood. 2002;99:1505–1511. doi: 10.1182/blood.V99.5.1505. [DOI] [PubMed] [Google Scholar]
- 132.Roufosse F, Cogan E, Goldman M. Lymphocytic variant hypereosinophilic syndromes. Immunol Allergy Clin N Am. 2007;27:389–413. doi: 10.1016/j.iac.2007.07.002. [DOI] [PubMed] [Google Scholar]
- 133.Lefèvre G, Copin M-C, Staumont-Sallé D, et al. The lymphoid variant of hypereosinophilic syndrome: study of 21 patients with CD3-CD4+ aberrant T-cell phenotype. Medicine (Baltimore) 2014;93:255–266. doi: 10.1097/MD.0000000000000088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Carpentier C, Verbanck S, Schandené L, Heimann P, Trépant A-L, Cogan E, Roufosse F. Eosinophilia associated with CD3−CD4+ T cells: characterization and outcome of a single-center cohort of 26 patients. Front Immunol. 2020;11:1765. doi: 10.3389/fimmu.2020.01765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.de Lavareille A, Roufosse F, Schandené L, Stordeur P, Cogan E, Goldman M. Clonal Th2 cells associated with chronic hypereosinophilia: TARC-induced CCR4 down-regulation in vivo. Eur J Immunol. 2001;31:1037–1046. doi: 10.1002/1521-4141(200104)31:4<1037::AID-IMMU1037>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]
- 136.de Lavareille A, Roufosse F, Schmid-Grendelmeier P, Roumier A-S, Schandené L, Cogan E, Simon H-U, Goldman M. High serum thymus and activation-regulated chemokine levels in the lymphocytic variant of the hypereosinophilic syndrome. J Allergy Clin Immunol. 2002;110:476–479. doi: 10.1067/mai.2002.127003. [DOI] [PubMed] [Google Scholar]
- 137.Ogbogu PU, Bochner BS, Butterfield JH, et al. Hypereosinophilic syndrome: a multicenter, retrospective analysis of clinical characteristics and response to therapy. J Allergy Clin Immunol. 2009;124:1319–1325.e3. doi: 10.1016/j.jaci.2009.09.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Suzuki M, Yamaguchi Y, Nakamura K, Kanaoka M, Matsukura S, Takahashi K, Takahashi Y, Kambara T, Aihara M (2020) Serum thymus and activation-regulated chemokine (TARC/CCL17) may be useful to predict the disease activity in patients with bullous pemphigoid. J Eur Acad Dermatol Venereol. 10.1111/jdv.16851 [DOI] [PubMed]
- 139.Machida H, Inoue S, Shibata Y, et al. Thymus and activation-regulated chemokine (TARC/CCL17) predicts decline of pulmonary function in patients with chronic obstructive pulmonary disease. Allergol Int. 2021;70:81–88. doi: 10.1016/j.alit.2020.04.004. [DOI] [PubMed] [Google Scholar]
- 140.Latzin P, Hartl D, Regamey N, Frey U, Schoeni MH, Casaulta C. Comparison of serum markers for allergic bronchopulmonary aspergillosis in cystic fibrosis. Eur Respir J. 2008;31:36–42. doi: 10.1183/09031936.00078107. [DOI] [PubMed] [Google Scholar]
- 141.Shono Y, Suga H, Kamijo H, Fujii H, Oka T, Miyagaki T, Shishido-Takahashi N, Sugaya M, Sato S. Expression of CCR3 and CCR4 suggests a poor prognosis in mycosis Fungoides and Sézary syndrome. Acta Derm Venereol. 2019;99:809–812. doi: 10.2340/00015555-3207. [DOI] [PubMed] [Google Scholar]
- 142.Ishida T, Utsunomiya A, Iida S, et al. Clinical significance of CCR4 expression in adult T-cell leukemia/ lymphoma: its close association with skin involvement and unfavorable outcome. Cancer Res. 2003;9:3625–3634. [PubMed] [Google Scholar]
- 143.Carpentier C, Schandené L, Dewispelaere L, Heimann P, Cogan E, Roufosse F (2021) CD3−CD4+ Lymphocytic variant hypereosinophilic syndrome: diagnostic tools revisited. J Allergy Clin Immunol Pract. 10.1016/j.jaip.2021.01.030 [DOI] [PubMed]
- 144.Roufosse F, de Lavareille A, Schandené L, Cogan E, Georgelas A, Wagner L, Xi L, Raffeld M, Goldman M, Gleich GJ. Mepolizumab as a corticosteroid-sparing agent in lymphocytic variant hypereosinophilic syndrome. J Allergy Clin Immunol. 2010;126:828–835.e3. doi: 10.1016/j.jaci.2010.06.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Pease JE, Horuk R. Recent progress in the development of antagonists to the chemokine receptors CCR3 and CCR4. Expert Opin Drug Discovery. 2014;9:467–483. doi: 10.1517/17460441.2014.897324. [DOI] [PubMed] [Google Scholar]
- 146.Ishida T, Ito A, Sato F, Kusumoto S, Iida S, Inagaki H, Morita A, Akinaga S, Ueda R. Stevens-Johnson syndrome associated with mogamulizumab treatment of adult T-cell leukemia/lymphoma. Cancer Sci. 2013;104:647–650. doi: 10.1111/cas.12116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Matsuo K, Nagakubo D, Komori Y, et al. CCR4 is critically involved in skin allergic inflammation of balb/c mice. J Invest Dermatol. 2018;138:1764–1773. doi: 10.1016/j.jid.2018.02.027. [DOI] [PubMed] [Google Scholar]
- 148.Murray C, Ahrens K, Devalaraja M, et al. Use of a canine model of atopic dermatitis to investigate the efficacy of a CCR4 antagonist in allergen-induced skin inflammation in a randomized study. J Invest Dermatol. 2016;136:665–671. doi: 10.1016/j.jid.2015.11.001. [DOI] [PubMed] [Google Scholar]
- 149.Cheng LE, Jorapur A, Jacobson S, et al. 561 Development and first-in-human characterization of a potent oral CCR4 antagonist for the treatment of atopic dermatitis. J Invest Dermatol. 2020;140:S77. doi: 10.1016/j.jid.2020.03.571. [DOI] [Google Scholar]
- 150.Ohsawa Y, Hirasawa N. The role of histamine H1 and H4 receptors in atopic dermatitis: from basic research to clinical studY. Allergol Int. 2014;63:533–542. doi: 10.2332/allergolint.13-RA-0675. [DOI] [PubMed] [Google Scholar]
- 151.Werfel T, Layton G, Yeadon M, et al. Efficacy and safety of the histamine H4 receptor antagonist ZPL-3893787 in patients with atopic dermatitis. J Allergy Clin Immunol. 2019;143:1830–1837.e4. doi: 10.1016/j.jaci.2018.07.047. [DOI] [PubMed] [Google Scholar]
- 152.Faustino L, da Fonseca DM, Takenaka MC, Mirotti L, Florsheim EB, Guereschi MG, Silva JS, Basso AS, Russo M. Regulatory T cells migrate to airways via CCR4 and attenuate the severity of airway allergic inflammation. J Immunol Baltim Md 1950. 2013;190:2614–2621. doi: 10.4049/jimmunol.1202354. [DOI] [PubMed] [Google Scholar]
- 153.Afshar R, Strassner JP, Seung E, Causton B, Cho JL, Harris RS, Hamilos DL, Medoff BD, Luster AD. Compartmentalized chemokine-dependent regulatory T-cell inhibition of allergic pulmonary inflammation. J Allergy Clin Immunol. 2013;131:1644–1652. doi: 10.1016/j.jaci.2013.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Kim YH, Bagot M, Pinter-Brown L, et al. Mogamulizumab versus vorinostat in previously treated cutaneous T-cell lymphoma (MAVORIC): an international, open-label, randomised, controlled phase 3 trial. Lancet Oncol. 2018;19:1192–1204. doi: 10.1016/S1470-2045(18)30379-6. [DOI] [PubMed] [Google Scholar]
- 155.Ishida T, Jo T, Takemoto S, et al. Dose-intensified chemotherapy alone or in combination with mogamulizumab in newly diagnosed aggressive adult T-cell leukaemia-lymphoma: a randomized phase II study. Br J Haematol. 2015;169:672–682. doi: 10.1111/bjh.13338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Cook LB, Fuji S, Hermine O, et al. Revised Adult T-Cell Leukemia-Lymphoma International Consensus Meeting Report. J Clin Oncol. 2019;37:677–687. doi: 10.1200/JCO.18.00501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Ledoult E, Groh M, Kahn J-E, et al. Assessment of T-cell polarization on the basis of surface marker expression: diagnosis and potential therapeutic implications in lymphocytic variant hypereosinophilic syndrome. J Allergy Clin Immunol Pract. 2020;8:1110–1114.e2. doi: 10.1016/j.jaip.2019.08.049. [DOI] [PubMed] [Google Scholar]
- 158.Liddiard K, Welch JS, Lozach J, Heinz S, Glass CK, Greaves DR. Interleukin-4 induction of the CC chemokine TARC (CCL17) in murine macrophages is mediated by multiple STAT6 sites in the TARC gene promoter. BMC Mol Biol. 2006;7:45. doi: 10.1186/1471-2199-7-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Fujimura T, Kambayashi Y, Furudate S, Asano M, Kakizaki A, Aiba S. Receptor activator of NF-κB ligand promotes the production of CCL17 from RANK+ M2 macrophages. J Invest Dermatol. 2015;135:2884–2887. doi: 10.1038/jid.2015.209. [DOI] [PubMed] [Google Scholar]
- 160.Soumelis V, Reche PA, Kanzler H, et al. Human epithelial cells trigger dendritic cell–mediated allergic inflammation by producing TSLP. Nat Immunol. 2002;3:673–680. doi: 10.1038/ni805. [DOI] [PubMed] [Google Scholar]
- 161.Nakanishi T, Inaba M, Inagaki-Katashiba N, Tanaka A, Vien PTX, Kibata K, Ito T, Nomura S. Platelet-derived RANK ligand enhances CCL17 secretion from dendritic cells mediated by thymic stromal lymphopoietin. Platelets. 2015;26:425–431. doi: 10.3109/09537104.2014.920081. [DOI] [PubMed] [Google Scholar]
- 162.Zhou B, Comeau MR, Smedt TD, Liggitt HD, Dahl ME, Lewis DB, Gyarmati D, Aye T, Campbell DJ, Ziegler SF. Thymic stromal lymphopoietin as a key initiator of allergic airway inflammation in mice. Nat Immunol. 2005;6:1047–1053. doi: 10.1038/ni1247. [DOI] [PubMed] [Google Scholar]
- 163.Kurokawa M, Matsukura S, Kawaguchi M, Ieki K, Suzuki S, Watanabe S, Homma T, Yamaguchi M, Takeuchi H, Adachi M. Interleukin-33-activated dendritic cells induce the production of thymus and activation- regulated chemokine and macrophage-derived chemokine. Int Arch Allergy Immunol. 2013;161(suppl 2):52–57. doi: 10.1159/000350363. [DOI] [PubMed] [Google Scholar]
- 164.Xiao T, Fujita H, Saeki H, et al. Thymus and activation-regulated chemokine (TARC/CCL17) produced by mouse epidermal Langerhans cells is upregulated by TNF-α and IL-4 and downregulated by IFN-γ. Cytokine. 2003;23:126–132. doi: 10.1016/S1043-4666(03)00221-7. [DOI] [PubMed] [Google Scholar]
- 165.Miyano K, Matsushita S, Tsuchida T, Nakamura K. Inhibitory effect of a histamine 4 receptor antagonist on CCL17 and CCL22 production by monocyte-derived Langerhans cells in patients with atopic dermatitis. J Dermatol. 2016;43:1024–1029. doi: 10.1111/1346-8138.13294. [DOI] [PubMed] [Google Scholar]
- 166.Yu B, Koga T, Urabe K, Moroi Y, Maeda S, Yanagihara Y, Furue M. Differential regulation of thymus- and activation-regulated chemokine induced by IL-4, IL-13, TNF-a and IFN-g in human keratinocyte and fibroblast. J Dermatol Sci. 2002;30:29–36. doi: 10.1016/S0923-1811(02)00046-4. [DOI] [PubMed] [Google Scholar]
- 167.Zheng X, Nakamura K, Tojo M, Oyama N, Nishibu A, Satoh M, Kakinuma T, Wakugawa M, Tamaki K, Kaneko F. TGF-b1-mediated regulation of thymus and activation- regulated chemokine (TARC/CCL17) synthesis and secretion by HaCaT cells co-stimulated with TNF-a and IFN-g. J Dermatol Sci. 2002;30:154–160. doi: 10.1016/S0923-1811(02)00071-3. [DOI] [PubMed] [Google Scholar]
- 168.Komine M, Kakinuma T, Kagami S, Hanakawa Y, Hashimoto K, Tamaki K. Mechanism of thymus- and activation-regulated chemokine (TARC)/CCL17 production and its modulation by roxithromycin. J Invest Dermatol. 2005;125:491–498. doi: 10.1111/j.0022-202X.2005.23840.x. [DOI] [PubMed] [Google Scholar]
- 169.Kwon D-J, Bae Y-S, Ju SM, Goh AR, Youn GS, Choi SY, Park J. Casuarinin suppresses TARC/CCL17 and MDC/CCL22 production via blockade of NF-κB and STAT1 activation in HaCaT cells. Biochem Biophys Res Commun. 2012;417:1254–1259. doi: 10.1016/j.bbrc.2011.12.119. [DOI] [PubMed] [Google Scholar]
- 170.Jung M, Lee TH, Bang M-H, Kim H, Son Y, Chung DK, Kim J. Suppression of thymus- and activation-regulated chemokine (TARC/CCL17) production by 3-O-β-d-glucopyanosylspinasterol via blocking NF-κB and STAT1 signaling pathways in TNF-α and IFN-γ-induced HaCaT keratinocytes. Biochem Biophys Res Commun. 2012;427:236–241. doi: 10.1016/j.bbrc.2012.08.087. [DOI] [PubMed] [Google Scholar]
- 171.Osabe M, Tajika T, Tohkin M. Allopurinol suppresses expression of the regulatory T-cell migration factors TARC/CCL17 and MDC/CCL22 in HaCaT keratinocytes via restriction of nuclear factor-κB activation. J Appl Toxicol JAT. 2018;38:274–283. doi: 10.1002/jat.3522. [DOI] [PubMed] [Google Scholar]
- 172.Byrne AM. Identification of glucocorticoid-induced TNF receptor-related protein ligand on keratinocytes: ligation by GITR induces keratinocyte chemokine production and augments T-cell proliferation. J Invest Dermatol. 2009;129:2784–2794. doi: 10.1038/jid.2009.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Hung C-H, Chu Y-T, Hua Y-M, Hsu S-H, Lin C-S, Chang H-C, Lee M-S, Jong Y-J. Effects of formoterol and salmeterol on the production of Th1- and Th2-related chemokines by monocytes and bronchial epithelial cells. Eur Respir J. 2008;31:9. doi: 10.1183/09031936.00121406. [DOI] [PubMed] [Google Scholar]
- 174.Ritter M, Mennerich D, Weith A, Seither P. Characterization of Toll-like receptors in primary lung epithelial cells: strong impact of the TLR3 ligand poly(I:C) on the regulation of Toll-like receptors, adaptor proteins and inflammatory response. J Inflamm. 2005;2:16. doi: 10.1186/1476-9255-2-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Matsuno K, Harada N, Harada S, et al. Combination of TWEAK and TGF-β1 induces the production of TSLP, RANTES, and TARC in BEAS-2B human bronchial epithelial cells during epithelial-mesenchymal transition. Exp Lung Res. 2018;44:332–343. doi: 10.1080/01902148.2018.1522558. [DOI] [PubMed] [Google Scholar]
- 176.Monick MM, Powers LS, Hassan I, Groskreutz D, Yarovinsky TO. Barrett CW, Castilow EM, Tifrea D, Varga SM, Hunninghake GW. Respiratory syncytial virus synergizes with Th2 cytokines to induce optimal levels of TARC/CCL17. J Immunol. 2007;179:1648–1658. doi: 10.4049/jimmunol.179.3.1648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Terada N, Nomura T, Kim WJ, et al. Expression of C-C chemokine TARC in human nasal mucosa and its regulation by cytokines. Clin Htmlent Glyphamp Asciiamp Exp Allergy. 2001;31:1923–1931. doi: 10.1046/j.1365-2222.2001.01152.x. [DOI] [PubMed] [Google Scholar]
- 178.Faffe DS, Whitehead T, Moore PE, Baraldo S, Flynt L, Bourgeois K, Panettieri RA, Shore SA. IL-13 and IL-4 promote TARC release in human airway smooth muscle cells: role of IL-4 receptor genotype. Am J Physiol-Lung Cell Mol Physiol. 2003;285:L907–L914. doi: 10.1152/ajplung.00120.2003. [DOI] [PubMed] [Google Scholar]
- 179.Shoda T, Futamura K, Kobayashi F, Saito H, Matsumoto K, Matsuda A. Expression of thymus and activation-regulated chemokine (TARC) by human dermal cells, but not epidermal keratinocytes. J Dermatol Sci. 2014;76:90–95. doi: 10.1016/j.jdermsci.2014.08.009. [DOI] [PubMed] [Google Scholar]
- 180.Morgan AJ, Symon FA, Berry MA, Pavord ID, Corrigan CJ, Wardlaw AJ. IL-4-expressing bronchoalveolar T cells from asthmatic and healthy subjects preferentially express CCR 3 and CCR 4. J Allergy Clin Immunol. 2005;116:594–600. doi: 10.1016/j.jaci.2005.03.052. [DOI] [PubMed] [Google Scholar]
- 181.Blom LH, Juel-Berg N, Larsen LF, Hansen KS, Poulsen LK. Circulating allergen-specific TH2 lymphocytes: CCR4+ rather than CLA+ is the predominant phenotype in peanut-allergic subjects. J Allergy Clin Immunol. 2018;141:1498–1501.e5. doi: 10.1016/j.jaci.2017.10.037. [DOI] [PubMed] [Google Scholar]
- 182.Biedermann T, Schwärzler C, Lametschwandtner G, Thoma G, Carballido-Perrig N, Kund J, de Vries JE, Rot A, Carballido JM. Targeting CLA/E-selectin interactions prevents CCR4-mediated recruitment of human Th2 memory cells to human skin in vivo. Eur J Immunol. 2002;32:3171–3180. doi: 10.1002/1521-4141(200211)32:11<3171::AID-IMMU3171>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 183.Geginat J, Lanzavecchia A, Sallusto F. Proliferation and differentiation potential of human CD8+ memory T-cell subsets in response to antigen or homeostatic cytokines. Blood. 2003;101:4260–4266. doi: 10.1182/blood-2002-11-3577. [DOI] [PubMed] [Google Scholar]
- 184.Inaoki M, Sato S, Shirasaki F, Mukaida N, Takehara K. The frequency of type 2 CD8+ T cells is increased in peripheral blood from patients with psoriasis vulgaris. J Clin Immunol. 2003;23:269–278. doi: 10.1023/A:1024584732614. [DOI] [PubMed] [Google Scholar]
- 185.Ahern D, Lloyd CM, Robinson DS. Chemokine responsiveness of CD4+ CD25+ regulatory and CD4+ CD25- T cells from atopic and nonatopic donors. Allergy. 2009;64:1121–1129. doi: 10.1111/j.1398-9995.2008.01962.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Lim HW, Lee J, Hillsamer P, Kim CH. Human Th17 cells share major trafficking receptors with both polarized effector T cells and FOXP3 + regulatory T cells. J Immunol. 2008;180:122–129. doi: 10.4049/jimmunol.180.1.122. [DOI] [PubMed] [Google Scholar]
- 187.Trifari S, Kaplan CD, Tran EH, Crellin NK, Spits H. Identification of a human helper T cell population that has abundant production of interleukin 22 and is distinct from TH-17, TH1 and TH2 cells. Nat Immunol. 2009;10:864–871. doi: 10.1038/ni.1770. [DOI] [PubMed] [Google Scholar]
- 188.Mjösberg JM, Trifari S, Crellin NK, Peters CP, van Drunen CM, Piet B, Fokkens WJ, Cupedo T, Spits H. Human IL-25- and IL-33-responsive type 2 innate lymphoid cells are defined by expression of CRTH2 and CD161. Nat Immunol. 2011;12:1055–1062. doi: 10.1038/ni.2104. [DOI] [PubMed] [Google Scholar]
- 189.Salimi M, Barlow JL, Saunders SP, et al. A role for IL-25 and IL-33–driven type-2 innate lymphoid cells in atopic dermatitis. J Exp Med. 2013;210:2939–2950. doi: 10.1084/jem.20130351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Amin K, Janson C, Harvima I, Venge P, Nilsson G. CC chemokine receptors CCR1 and CCR4 are expressed on airway mast cells in allergic asthma. J Allergy Clin Immunol. 2005;116:1383–1386. doi: 10.1016/j.jaci.2005.08.053. [DOI] [PubMed] [Google Scholar]
- 191.Bratke K, Prieschenk C, Garbe K, Kuepper M, Lommatzsch M, Virchow JC. Plasmacytoid dendritic cells in allergic asthma and the role of inhaled corticosteroid treatment. Clin Exp Allergy. 2013;43:312–321. doi: 10.1111/cea.12064. [DOI] [PubMed] [Google Scholar]
- 192.Vittorakis S, Samitas K, Tousa S, Zervas E, Aggelakopoulou M, Semitekolou M, Panoutsakopoulou V, Xanthou G, Gaga M. Circulating conventional and plasmacytoid dendritic cell subsets display distinct kinetics during in vivo repeated allergen skin challenges in atopic subjects. Biomed Res Int. 2014;2014:231036. doi: 10.1155/2014/231036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Bonner K, Pease JE, Corrigan CJ, Clark P, Kay AB. CCL17/thymus and activation-regulated chemokine induces calcitonin gene-related peptide in human airway epithelial cells through CCR4. J Allergy Clin Immunol. 2013;132:942–950.e1–3. doi: 10.1016/j.jaci.2013.04.015. [DOI] [PubMed] [Google Scholar]
- 194.Hartl D, Lee CG, Da Silva CA, Chupp GL, Elias JA. Novel biomarkers in asthma: chemokines and chitinase-like proteins. Curr Opin Allergy Clin Immunol. 2009;9:60–66. doi: 10.1097/ACI.0b013e32831f8ee0. [DOI] [PubMed] [Google Scholar]
- 195.Sekiya T, Yamada H, Yamaguchi M, Yamamoto K, Ishii A, Yoshie O, Sano Y, Morita A, Matsushima K, Hirai K. Increased levels of a TH2-type CC chemokine thymus and activation-regulated chemokine (TARC) in serum and induced sputum of asthmatics. Allergy. 2002;57:173–177. doi: 10.1034/j.1398-9995.2002.5720256.x. [DOI] [PubMed] [Google Scholar]
- 196.Leung T, Wong C, Chan IHS, Ip W, Lam CWK, Wong GWK. Plasma concentration of thymus and activation-regulated chemokine is elevated in childhood asthma. J Allergy Clin Immunol. 2002;110:404–409. doi: 10.1067/mai.2002.126378. [DOI] [PubMed] [Google Scholar]
- 197.Leung TF, Wong CK, Lam CWK, Li AM, Ip WK, Wong GWK, Fok TF. Plasma TARC concentration may be a useful marker for asthmatic exacerbation in children. Eur Respir J. 2003;21:616–620. doi: 10.1183/09031936.03.00083303. [DOI] [PubMed] [Google Scholar]
- 198.Abou El-Ela M, El-Rifae AE-A, Fawzi M, Abdel Hay R, Gohary Y, Shaker O. Thymus and activation-regulated chemokine in different stages of mycosis fungoides: tissue and serum levels: TARC in mycosis fungoides. Australas J Dermatol. 2011;52:167–171. doi: 10.1111/j.1440-0960.2010.00688.x. [DOI] [PubMed] [Google Scholar]
- 199.Inagaki A, Ishida T, Yano H, et al. Clinical significance of serum TARC/CCL17 in ATLL patients: high TARC/CCL17 level is a significant unfavorable prognostic factor. Blood. 2008;112:2821–2821. doi: 10.1182/blood.V112.11.2821.2821. [DOI] [Google Scholar]
- 200.Cahn A, Hodgson S, Wilson R, et al. Safety, tolerability, pharmacokinetics and pharmacodynamics of GSK2239633, a CC-chemokine receptor 4 antagonist, in healthy male subjects: results from an open-label and from a randomised study. BMC Pharmacol Toxicol. 2013;14:14. doi: 10.1186/2050-6511-14-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Duvic M, Pinter-Brown LC, Foss FM, et al. Phase 1/2 study of mogamulizumab, a defucosylated anti-CCR4 antibody, in previously treated patients with cutaneous T-cell lymphoma. Blood. 2015;125:1883–1889. doi: 10.1182/blood-2014-09-600924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Ogura M, Ishida T, Hatake K, et al. Multicenter phase II study of mogamulizumab (KW-0761), a defucosylated anti-CC chemokine receptor 4 antibody, in patients with relapsed peripheral T-cell lymphoma and cutaneous T-cell lymphomA. J Clin Oncol. 2014;32:1157–1164. doi: 10.1200/JCO.2013.52.0924. [DOI] [PubMed] [Google Scholar]
- 203.Zinzani PL, Karlin L, Radford J, et al. European phase II study of mogamulizumab, an anti-CCR4 monoclonal antibody, in relapsed/refractory peripheral T-cell lymphoma. Haematologica. 2016;101:e407–e410. doi: 10.3324/haematol.2016.146977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Ishida T, Joh T, Uike N, et al. Defucosylated anti-CCR4 monoclonal antibody (KW-0761) for relapsed adult T-cell leukemia-lymphoma: a multicenter phase II study. J Clin Oncol Off J Am Soc Clin Oncol. 2012;30:837–842. doi: 10.1200/JCO.2011.37.3472. [DOI] [PubMed] [Google Scholar]
- 205.Phillips AA, Fields P, Hermine O, et al. A prospective, multicenter, randomized study of anti-CCR4 monoclonal antibody mogamulizumab (moga) vs investigator’s choice (IC) in the treatment of patients (pts) with relapsed/refractory (R/R) adult T-cell leukemia-lymphoma (ATL) J Clin Oncol. 2016;34:7501–7501. doi: 10.1200/JCO.2016.34.15_suppl.7501. [DOI] [Google Scholar]
- 206.McCormick SM, Gowda N, Fang JX, Heller NM. Suppressor of cytokine signaling (SOCS)1 regulates iNTERLEUKIN-4 (IL-4)-activated insulin receptor substrate (IRS)-2 tyrosine phosphorylation in monocytes and macrophages via the proteasome. J Biol Chem. 2016;291:20574–20587. doi: 10.1074/jbc.M116.746164. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.