Abstract
BACKGROUND AND PURPOSE: Placement of a covered stent to control carotid blowout (CB) in malignant tumors of the head and neck has been reported to be an effective treatment. However, it is not uncommon to encounter recurrent hemorrhage. The purpose of this study was to evaluate the follow-up results of patients treated with covered stents.
MATERIALS AND METHODS: We retrospectively reviewed the results of 7 consecutive patients who underwent placement of a covered stent to control CB. Most of them had poor wound healing because of previous irradiation, surgery, or both. The initial procedures were successful in all patients. Their clinical course was reviewed for rebleeding, additional endovascular treatments in recurrent cases, and outcomes.
RESULTS: Recurrence developed in 6 of 7 patients. The interval between the first procedure and the hemorrhagic event was from 3 to 44 days. In 6 patients who had a recurrent CB, 4 had rebleeding from the previous site of the stent, whereas 2 other patients experienced recurrent bleeding in a different area from the site of the stent. Additional endovascular treatments were carried out in all affected patients by another insertion of a covered stent (n = 3), coil embolization (n = 2), or insertion of a covered stent followed by permanent arterial occlusion (n = 1).
CONCLUSION: Placement of a covered stent in patients with head and neck cancer who sustain CB showed frequent rebleeding despite favorable initial rescue results. Recurrent CB at the previous stent site developed frequently in patients with uncontrolled wound infection. Concomitant or short-interval arterial trapping should be considered selectively in those conditions.
Carotid blowout (CB) is a life-threatening event with a high rate of mortality and morbidity. It can be encountered during or after certain treatments in patients with various malignant tumors of the head and neck and occurs more frequently in patients with a previous history of radiation therapy.1–3
Surgical treatment of CB may not be effective, or it may sometimes be impossible because of the nature of the operative wound. Endovascular trapping of the involved carotid artery is very effective in the control of massive hemorrhage.4,5 However, the destructive nature of this procedure is not allowable if the collateral circulation is insufficient.
Placement of a covered stent is a good alternative treatment option. It can decrease the risk of ischemic stroke by preserving the involved artery while the leaking hole can be sealed by the stent at the same time. There have been several reports of the successful application of covered stents for the treatment of a leaking artery.6–13
Our previous experience with covered stents also coincided with other positive reports.14 However, we recently became suspicious of the long-term results of patients treated with covered stents because there have been several cases of recurrent hemorrhage on follow-up, even when initial results were successful. Most of these recurrences were serious and unexpected, such as an initial hemorrhagic event.
The purpose of this study was to evaluate the follow-up results of patients who sustained CB and who were treated by placement of a covered stent.
Materials and Methods
Patients
Seven consecutive patients underwent placement of a covered stent for the management of CB associated with various malignant tumors of the head and neck between September 2003 and April 2006. We retrospectively reviewed their medical records and radiologic findings under the approval of the Institutional Review Board of Asan Medical Center.
There were 6 men and 1 woman with a mean age of 64 years (age range, 55–80 years). They had various kinds of malignant growths of the head and neck. All of these patients had previous histories of irradiation in the area of the CB without exception. The time interval varied between the initiation of radiation therapy and the endovascular therapy (1 month to 30 years; median, 11 months). Six of the 7 patients had histories of various surgical resections. The patients’ clinical characteristics are summarized in the Table.
Summary of clinical and lesion characteristics
| Age/Sex | Clinical History | Treatment | Fistular Formation | Flap Surgery | Infection | Initial Site | Rebleeding | Recurrence Site | Interval between Two Events | Clinical Course |
|---|---|---|---|---|---|---|---|---|---|---|
| 62/M | Esophageal ca, Lt supraclavicular lymphadenopathy | RTx, I & D(2) | Yes | No | Yes | Lt mid CCA | Yes | Proximal end of the stent margin | 10 days | No recurrent hemorrhage on 2 months F/U |
| 50/M | Nasopharyngeal ca | RTx(2), skull base op. w ND (Rt), PMMC flap, free flap, I & D | Yes | Unfavorable result | Yes | Rt carotid bulb | Yes | Proximal end of the stent margin | 44 days | Permanent carotid arterial occlusion for recurrent bloody sputum after the second stent placement, death after 8 months |
| 67/M | Laryngeal ca | RTx, TL w both ND several flap surgery | Yes | Favorable result | Yes, streptococcus | Lt mid CCA | No | N/A | N/A | Death 1 month later, no recurrent hemorrhage |
| 64/M | Laryngeal ca | RTx,(2) TL w both ND | No | No | No | Rt distal CCA | Yes | Rt ECA branch | 3 days | Discharge in stable condition 2 weeks later and lost F/U |
| 69/M | Laryngeal ca | RTx, TL w both ND, PMMC | Yes | Unfavorable result | Yes | Rt distal CCA | Yes | Distal end of the stent margin | 23 days | Discharge for palliative care and lost F/U |
| 55/F | Hypopharyngeal ca | RTx, TLPE w ND(Rt) | Yes | Unfavorable result | Yes, Klebsiella | Lt distal CCA | Yes | Distal end of the stent margin | 11 days | Discharge for palliative care and lost F/U |
| 80/M | Hypopharyngeal ca | RTx(2) | No | No | No | Rt mid CCA | Yes | Lt mid CCA | 40 days | Death from sepsis 10 days after the second stent placement |
Note:—Ca indicates cancer; CCA, common carotid artery; ECA, external carotid artery; F/U, follow-up; I&D, incision and drainage; TLPE, total laryngopharyngoesophagectomy; Lt, left; mid, middle; N/A, not applicable; ND, neck dissection; op, operation; PMMC, pectoralis major myocutaneous; Rt, right; RTx, radiotherapy; TL, total laryngectomy.
Among these patients, 5 (71%) presented with massive oral or wound bleeding, or both, which was impossible to control with anterior or posterior nasal packing or with compression of the local wound. Alternatively, we became aware of the other 2 (29%) patients during conventional angiography because of a suspected abnormality in the common carotid artery, which had been detected incidentally during wound reconstruction surgery. In these 2 patients, we performed placement of a covered stent for prophylactic purposes.
Procedure
After we decided to perform placement of a covered stent, we obtained written informed consent from the patients or their legal representatives confirming the need for this procedure. We also explained the potential risks and benefits of the procedure. After identifying the bleeding focus, which seemed to be a culprit lesion, we exchanged the diagnostic catheter and wire to a 9F introducer sheath and a 0.035-inch stiff exchange guidewire (Terumo, Tokyo, Japan). The covered stent we used was a self-expandable nitinol stent covered with PTFE (polytetrafluoroethylene) (NITI-S Stent; Taewoong Medical, Seoul, South Korea). In our initial experience, we tried to occlude a leaking pseudoaneurysm by placing several coils and a bare metallic stent. Unfortunately, however, this procedure did not offer enough hemostatic effect. We performed additional placement of a covered stent over the bare metallic stent to control the hemorrhage effectively.
After we obtained hemostasis, we administered oral antiplatelet medication with 100 mg of aspirin and 75 mg of clopidogrel (Plavix) daily after loading dose to minimize the risk of embolic ischemia and in-stent thrombosis. When secure hemostasis was doubtful on follow-up clinical inspection, or if oozing developed at the wound site, we stopped any antiplatelet medication to prevent possible rebleeding.
Analysis
We analyzed the initial clinical and angiographic results after placement of the covered stent. The recurrence rate of hemorrhage, the time interval between initial treatment and recurrent hemorrhage, the focus of recurrence, the condition of the local wound including presence of infection, additional methods of endovascular treatment, and clinical follow-up were analyzing points.
Results
The initial clinical and angiographic results of the covered stent placement were successful in the 5 patients who presented with active bleeding (Fig 1) as well as in the other 2 patients with anticipated bleeding (Fig 2).
Fig 1.
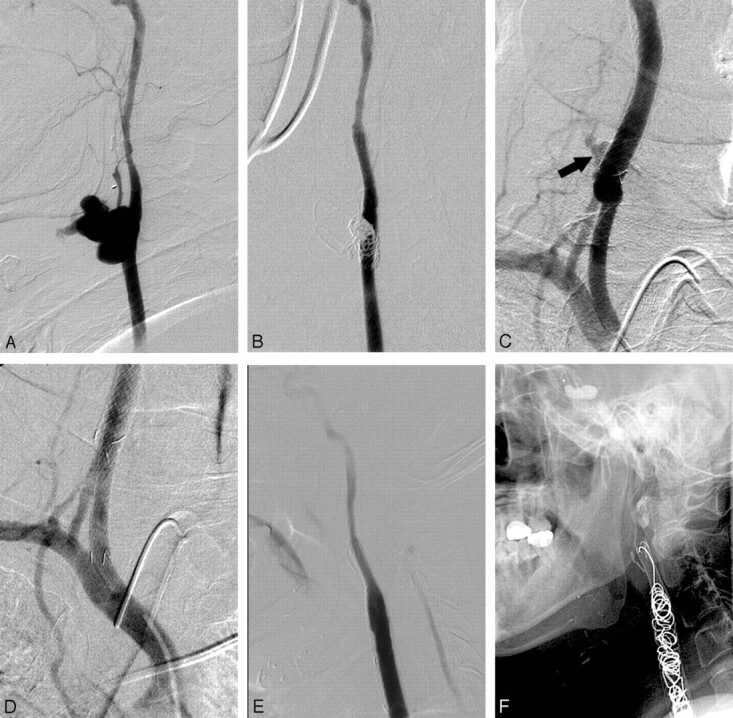
A 50-year-old man with nasopharyngeal cancer presents initially with massive oral bleeding. A, B, A large, lobulated pseudoaneurysm in the right carotid bulb is successfully isolated with use of detachable coils and a bare stent combined with a covered stent. C, D, Recurrent CB develops at the proximal end of the stent (arrow in C); a supplementary covered stent is inserted to obtain control of rebleeding. E, F, Permanent arterial occlusion is performed because of persistent bloody sputum 3 weeks after the second endovascular treatment.
Fig 2.
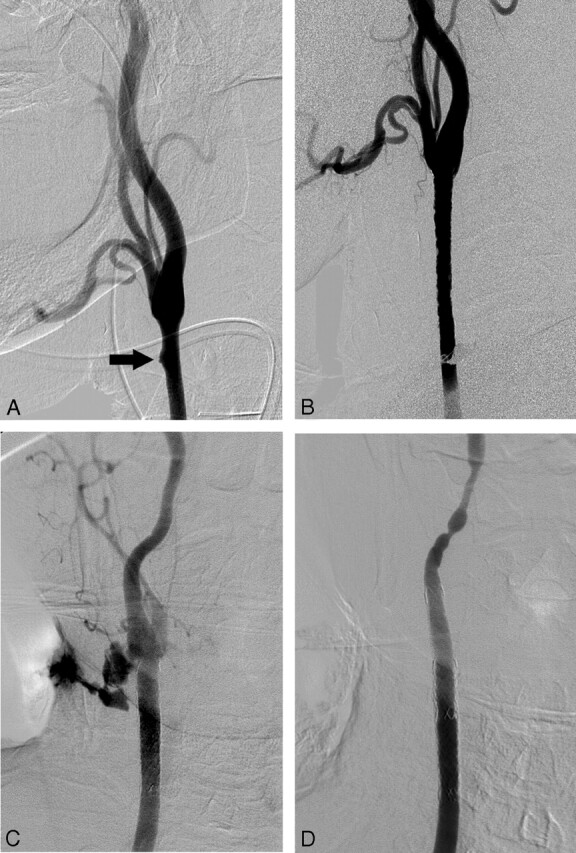
A 69-year-old man with a small, blisterlike lesion of the right common carotid artery, detected incidentally during surgical reconstruction of his wound. A, Conventional angiography shows a small ulcerative lesion (arrow) in the distal portion of the right common carotid artery. B, The lesion disappeared after preventive placement of the covered stent. C, A massive contrast extravasation is noted, near the distal end of the previous inserted stent, on the right common carotid angiogram obtained for the evaluation of the rebleeding, which occurred 3 weeks after the initial procedure. D, Complete control of hemorrhage is achieved by additional insertion of a covered stent.
On angiography, initial lesions were seen in the common carotid artery in all patients except for 1, who experienced leakage at the carotid bulb just above the carotid bifurcation. Placement of a covered stent (diameter, 8–10 mm; length, 40–70 mm) was enough to achieve immediate hemostasis.
Devastating recurrent bleeding was noted in 6 (85%) of 7 patients. The interval between the initial endovascular procedure and the recurrent episode of bleeding was from 3 to 44 days (median, 17 days). Subsequent angiography was immediately performed. Although manifestation of the bleeding episodes was similar to that of the initial events, the lesion sites of recurrent hemorrhages were different in all 6 patients. In 6 patients who sustained recurrent CB, 4 had rebleeding from the site of the previous stent insertion, whereas 2 other patients experienced recurrent bleeding from a completely different site not related to the location of the stent. In the strict sense, the rate of recurrent CB related to failure of stent durability was 57% (4/7). The foci of recurrent bleeding were at the cranial (n = 2) or caudal end (n = 2) of the previously inserted stent margin, a branch of the ipsilateral external carotid artery apart from the stent margin (n = 1), and contralateral common carotid artery (n = 1). We put another covered stent in 4 of these 6 patients. In the other 2 patients, we embolized the bleeding foci by using several metallic coils. An ipsilateral carotid trapping procedure was performed in 1 patient, who showed continuous oozing even after placement of the secondary stent. This patient had 3 endovascular treatments and died 8 months later without any further recurrence of hemorrhage. The successive hemostatic procedures were durable until death from septic cause in 1 patient, or discharge for palliative supportive care in the other 5 patients.
Discussion
CB is one of the most devastating events that both long-standing patients with head and neck cancer and their physicians hope not to face. In several recent studies, endovascular treatment with a covered stent for CB has been reported as a quick and effective method of lower morbidity compared with an emergent surgical procedure or permanent arterial occlusion with detachable balloons and coils.6–14 Moreover, in emergent situations such as profuse and active bleeding, which would be responsible for unstable vital signs, a less time-consuming method would be the preferred treatment option.
The immediate clinical results of the 7 patients who underwent placement of a covered stent for CB were favorable as well as those of several previous reports on the successful application of covered stents.6–14 However, we encountered a relatively high proportion of patients (6/7) who sustained recurrent active bleeding shortly after the successful first treatment. In fact, recurrence directly related to the site of the stent developed in 4 (57%) of 7 patients. However this ratio also was higher than that in previous reports.1,14,15 All of our patients had undergone radiation therapy with a dose of at least 60 Gy, and 3 patients with recurrent cancer had received a further booster dose of radiation. Irradiation can be attributed as a cause of clinically serious injury of the large arteries, including occlusive disease. In addition, injury of the vasa vasorum after radiation therapy may account for an increased incidence of CB.2,3,16–18
Two patients in our study underwent placement of a covered stent as a prophylactic measure. Nevertheless, both patients experienced massive oral or wound bleeding 1 month after the procedure, thereby requiring the second interventional treatment. Contrary to the case presented by Warren et al,13 in which prophylactic placement of a covered stent in the common carotid artery encased by a tumor showed good follow-up results, 2 of our patients did not enjoy such benefits. It is worthwhile to mention that these 2 patients had poor wound healing caused by wound dehiscence after pectoralis major myocutaneous flap surgery for treatment of a postsurgical fistula.
In our experience, infection was an important predisposing factor of arterial rupture. In the neck, infection may be manifested by disrupted skin flaps or a pharyngocutaneous fistula.12,17 The myocutaneous flaps are the optimum solution with their independent vascular pedicles. They provide the best environment for wound healing: diversion of salivary stream, and well-vascularized soft tissue so as to exclude it from the contaminated field and scaffolding effect of the stents.12,13 However, failure of the graft might be followed by infection of the local wound. The carotid artery underneath the graft may lose its external supporting soft tissue because of inevitable wound dehiscence or formation of a cutaneous fistula, or both. In 3 of 4 patients who underwent myocutaneous flap surgery, flap necrosis developed. Five of 7 patients sustained various cutaneous fistulas and wound infections with or without previous flap surgery.
There were some possible causes of frequent rebleeding after successful hemostasis. The patients in our study who exhibited recurrent CB were divided into 2 groups according to the site of the rebleeding. The first group, which included patients with a recurrent CB at the site of the stent insertion (4/6), had a fistula with infection followed by flap surgery or incisional drainage. To the contrary, the other group, who had rebleeding from a site different from that of the stent insertion (2/6), did not represent any possible causes that the adjacent rebleeding group had. Therefore, existence of an uncontrolled ongoing infection at the stent site is an important factor to predict a recurrent CB from a previous stent site.
In addition, continuous irritation by stent struts, especially the bare portion at both ends of the covered stent, was another cause of recurrent CB in the group who had rebleeding from the stent site. This factor could explain the cause of frequent rebleeding at both ends of the stent in this group. In addition to the chronic mechanical irritation, the presence of the stent itself could have acted as a foreign body in an unfavorable environment. To solve this problem, a newly designed covered stent with less irritability and more biologic stability should be developed.
The purpose of this report was not to deny the potential role of a covered stent in the management of a CB. Instead, we tried to emphasize that clinicians be aware of the possibility of a recurrent CB, even when the initial rescue procedure is successful. Of course, careful consideration is necessary for all patients who undergo placement of a covered stent for active hemorrhage or as a preventive measure, but more attention is mandatory for patients who have a contaminated area by a fistula, flap failure, or abscessed cavity as a result of tumor necrosis. In these patients under such emergent situations, placement of a covered stent would be the optimum treatment. Once successful control of bleeding is achieved, successive or short-term additional endovascular trapping of the involved artery can be definitely considered. Moreover, endo-vascular trapping of the involved artery should be the first prophylactic treatment in patients with poor wound healing. Of course, if there is evidence of controlled infection and improvement of the wound, close observation with hold of additional procedures can also be deliberated.
Conclusion
Placement of a covered stent in patients with head and neck cancer who sustain CB showed frequent rebleeding within a short interval despite successful rescue results. Recurrent CB at the previous site of the stent developed frequently in patients with uncontrolled wound infections. After the emergent situation is arrested, if a patient shows tolerant result in temporary occlusion test with a balloon catheter, concomitant or short-interval arterial trapping should be considered selectively in place of covered stent placement.
References
- 1.Chaloupka JC, Roth TC, Putman CM, et al. Recurrent carotid blowout syndrome: diagnostic and therapeutic challenges in a newly recognized subgroup of patients. AJNR Am J Neuroradiol 1999;20:1069–77 [PMC free article] [PubMed] [Google Scholar]
- 2.Katras T, Baltazar U, Colvett K, et al. Radiation-related arterial disease. Am Surg 1999;65:1176–79 [PubMed] [Google Scholar]
- 3.Maran AG, Amin M, Wilson JA. Radical neck dissection: a 19-year experience. J Laryngol Otol 1989;103:760–64 [DOI] [PubMed] [Google Scholar]
- 4.Citardi MJ, Chaloupka JC, Son YH, et al. Management of carotid artery rupture by monitored endovascular therapeutic occlusion (1988–1994). Laryngoscope 1995;105:1086–92 [DOI] [PubMed] [Google Scholar]
- 5.Morrissey DD, Andersen PE, Nesbit GM, et al. Endovascular management of hemorrhage in patients with head and neck cancer. Arch Otolaryngol Head Neck Surg 1997;123:15–19 [DOI] [PubMed] [Google Scholar]
- 6.Auyeung KM, Lui WM, Chow LC, et al. Massive epistaxis related to petrous carotid artery pseudoaneurysm after radiation therapy: emergency treatment with covered stent in two cases. AJNR Am J Neuroradiol 2003;24:1449–52 [PMC free article] [PubMed] [Google Scholar]
- 7.Bates MC, Shamsham FM. Endovascular management of impending carotid rupture in a patient with advanced head and neck cancer. J Endovasc Ther 2003;10:54–57 [DOI] [PubMed] [Google Scholar]
- 8.Cheng KM, Chan CM, Cheung YL, et al. Endovascular treatment of radiation-induced petrous internal carotid artery aneurysm presenting with acute haemorrhage. A report of two cases. Acta Neurochir (Wien) 2001;143:351–55; discussion 355–56 [DOI] [PubMed] [Google Scholar]
- 9.Desuter G, Hammer F, Gardiner Q, et al. Carotid stenting for impending carotid blowout: suitable supportive care for head and neck cancer patients? Palliat Med 2005;19:427–29 [DOI] [PubMed] [Google Scholar]
- 10.Kubaska SM 3rd, Greenberg RK, Clair D, et al. Internal carotid artery pseudoaneurysms: treatment with the Wallgraft endoprosthesis. J Endovasc Ther 2003;10:182–89 [DOI] [PubMed] [Google Scholar]
- 11.Kwok PC, Cheung JY, Tang KW, et al. Re: Endovascular treatment of acute carotid blow-out syndrome. J Vasc Interv Radiol 2001;12:895–96 [DOI] [PubMed] [Google Scholar]
- 12.Upile T, Triaridis S, Kirkland P, et al. The management of carotid artery rupture. Eur Arch Otorhinolaryngol 2005;262:555–60 [DOI] [PubMed] [Google Scholar]
- 13.Warren FM, Cohen JI, Nesbit GM, et al. Management of carotid ‘blowout’ with endovascular stent grafts. Laryngoscope 2002;112:428–33 [DOI] [PubMed] [Google Scholar]
- 14.Kim HS, Lee DH, Kim HJ, et al. Life-threatening common carotid artery blowout: rescue treatment with a newly designed self-expanding covered nitinol stent. Br J Radiol 2006;79:226–31 [DOI] [PubMed] [Google Scholar]
- 15.Lesley WS, Chaloupka JC, Weigele JB, et al. Preliminary experience with endovascular reconstruction for the management of carotid blowout syndrome. AJNR Am J Neuroradiol 2003;24:975–81 [PMC free article] [PubMed] [Google Scholar]
- 16.Martins AN, Johnston JS, Henry JM, et al. Delayed radiation necrosis of the brain. J Neurosurg 1977;47:336–45 [DOI] [PubMed] [Google Scholar]
- 17.McCready RA, Hyde GL, Bivins BA, et al. Radiation-induced arterial injuries. Surgery 1983;93:306–12 [PubMed] [Google Scholar]
- 18.Murros KE, Toole JF. The effect of radiation on carotid arteries. A review article. Arch Neurol 1989;46:449–55 [DOI] [PubMed] [Google Scholar]


