Abstract
The novel coronavirus named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which is at the origin of the current pandemic, predominantly manifests with severe respiratory symptoms and a heightened immune response. One characteristic of SARS-CoV-2 is its capacity to induce cytokine storm leading to acute respiratory distress syndrome. Consequently, agents with the ability to regulate the immune response, such as vitamin D, could become tools either for the prevention or the attenuation of the most severe consequences of the coronavirus disease 2019 (COVID-19). Vitamin D has shown antimicrobial as well as anti-inflammatory properties. While SARS-CoV-2 promotes the release of proinflammatory cytokines, vitamin D attenuates the release of at least some of these same molecules. Inflammatory cytokines have been associated with the clinical phenomena of COVID-19 and in particular with its most dangerous complications. Therefore, the goals of this article are as follows: first, present the numerous roles vitamin D plays in modulating the immune response; second, gather data currently available on COVID-19 clinical presentation and its relation to cytokines and similar molecules; third, expose what it is known about how coronaviruses elicit an inflammatory reaction; and fourth, discuss the potential contribution of vitamin D in reducing the risk and severity of COVID-19.
Keywords: ARDS, calcidiol, calcitriol, cytokine storm, pandemic, inflammation, immune response, SARS-CoV-2
Introduction
On 11 March 2020, the World Health Organization (WHO) officially declared the coronavirus disease 2019 (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a global pandemic.1 The SARS-CoV-2 was originally identified in Wuhan, China as the cause of a cluster of pneumonia cases that quickly spread worldwide.2 As of 13 January 2021, over 3.5 million cases have been reported with almost 250,000 deaths around the world.3 Many efforts from the medical and scientific communities are being made to understand, characterize, and combat this alarming infectious disease. Currently, there is a need for treatments against COVID-19 as well as for the identification of measures to prevent the infection or at least attenuate disease escalation. Agents involved in regulating the immune response to infective agents have, at least in theory, the potential to play a role in the containment of COVID-19. One inexpensive and low-risk molecule capable of regulating the immune response is vitamin D.
Vitamin D is a fat-soluble vitamin that is produced through dermal synthesis when ultraviolet (UV) rays from the sun strike the skin. Vitamin D can also be obtained in leafy vegetables, dairy products, fish, and artificially fortified foods. Whether obtained endogenously or in the diet, vitamin D requires further modification in order to become biologically active (Figure 1). First, vitamin D is converted in the liver to calcidiol, or 25-hydroxyvitamin D (25(OH)D) by the enzyme 25-hydroxylase. Then, calcidiol travels to the kidneys and through the enzyme 1α-hydroxylase becomes calcitriol, also known as 1,25-dihydroxyvitamin D (1,25(OH)2D).4 Once metabolically active, vitamin D has many roles in the body such as homeostasis of serum calcium and phosphate levels, bone metabolism, and regulation of the immune system.4–7 Not surprisingly, studies have correlated vitamin D deficiency with a number of autoimmune diseases such as rheumatoid arthritis, type 1 diabetes, systemic lupus erythematosus, and inflammatory bowel disease.8–13 Even further, when assessing the total mortality of the general population, vitamin D deficiency is sometimes considered an independent risk factor.14 It is therefore possible that vitamin D’s influence over the immune system could be involved in how at least some individuals respond to SARS-Cov-2. In order to explore this possibility, here we present the following: (1) the numerous roles vitamin D plays in modulating the immune response; (2) data currently available on COVID-19’s clinical presentation and its relation to cytokines and similar molecules; (3) what it is known about how coronaviruses elicit an inflammatory reaction; and (4) the potential contribution vitamin D may assume in reducing the risk and severity of COVID-19.
Figure 1.
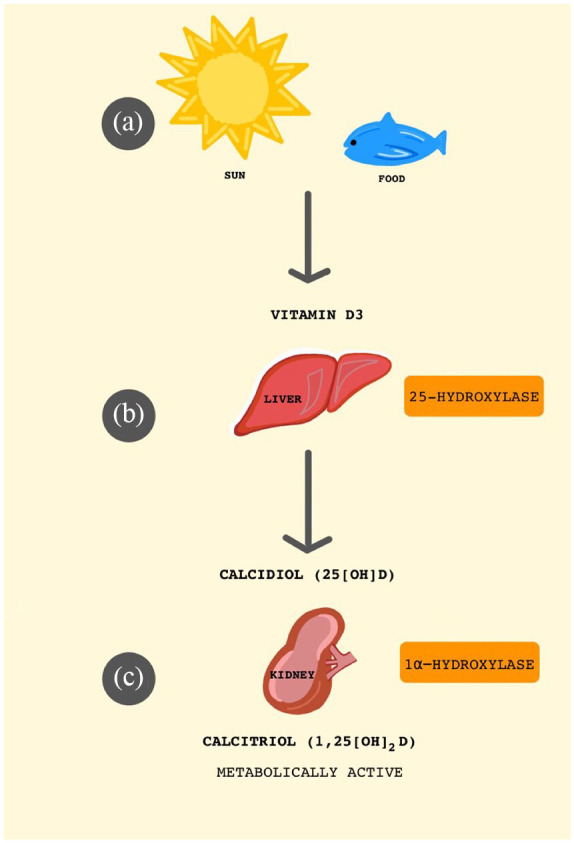
Synthesis of metabolically active vitamin D (1,25(OH)2D). (a) Vitamin D is produced when ultraviolet (UV) rays from the sun strike the skin and can also be obtained from leafy vegetables, dairy products, fish, and vitamin D fortified foods. (b) Vitamin D is converted in the liver to calcidiol, or 25-hydroxyvitamin D (25(OH)D). (c) Then, calcidiol travels to the kidneys and becomes enzymatically converted to calcitriol, also known as 1,25-dihdrydroxyvitamin D (1,25(OH)2D).
Methods
A review of current literature was conducted via MEDLINE and Google Scholar using search terms such vitamin D, immune system, immune response, COVID-19, SARS, and SARS-Cov-2. The search was limited to studies published in English. Special emphasis was placed on finding publications in which vitamin D was used in the treatment of COVID-19 or in which vitamin D levels were associated with inflammatory markers. For each of the scientific papers identified relating to vitamin D, COVID-19, and inflammatory markers, its references were thoroughly inspected with the goal to retrieve further publications on this topic.
Results
Role of vitamin D in modulating the immune response
Vitamin D has a signaling role in modulating both the adaptive and innate branches of the immune system. Vitamin D enacts these changes in its calcitriol form by interacting with nuclear vitamin D receptors (nVDR) expressed on B and T lymphocytes, neutrophils, monocytes, and dendritic cells (DC).9 In addition, these same immune and inflammatory cells have the ability to convert calcidiol to calcitriol through upregulation of enzyme 1-α-hydroxylase (CYP27B1), which hydroxylates calcidiol into its active form, calcitriol.9,11 Vitamin D also induces production of antimicrobial peptides such as defensins β2 and β4 and cathelicidin antimicrobial peptide (CAMP) by macrophages, monocytes, keratinocytes, epithelial, intestinal, lung, and corneal cells.8,9 In macrophages and monocytes, this enhances chemotaxis, autophagy, and phagolysosomal fusion of immune cells. In keratinocytes and epithelial, intestinal, lung, and corneal cells, this reinforces the physical barrier function. Collectively, these anti-microbial effects increase the body’s defense against microbes.8,11
Vitamin D also has effects on the adaptive immune system, which include switching between cell-mediated (Th1) and humoral (Th2) immunity.10,11 Calcitriol downregulates Th1 immune responses by inhibiting the production of type 1 proinflammatory cytokines such as IL-12, IFN-γ, IL-6, IL-8 TNF-α, and IL-9. In contrast, vitamin D upregulates the production of type 2 anti-inflammatory cytokines such as IL-4, IL-5, and IL-10.8,9 This cytokine regulation is mainly mediated by blocking NF-kB p65 activation via the upregulation of the NF-kB inhibitory protein IkBa.9 In addition, calcitriol upregulates Th2 cells and tolerogenic DC, inhibits Th1 cell proliferation, and induces T regulatory (Treg) cells. All of which results in the upregulation of anti-inflammatory cytokines and decreased production of inflammatory cytokines.10 Moreover, vitamin D induces the shift of macrophage polarization from proinflammatory M1 phenotype toward the anti-inflammatory M2 phenotype.15 Vitamin D also inhibits DC differentiation by decreasing its expression of MHC class II molecules, co-stimulatory molecules, and IL-12.9 Inhibition of differentiation and maturation of DC prevents autoimmunity and promotes self-tolerance. This is because antigen presentation by mature DC induces an immune response, while antigen presentation by immature DC induces tolerance.9 In immunoglobulin-producing B cells, calcitriol inhibits B cell differentiation, proliferation, and promotes apoptosis. This has clinical importance for potential autoimmune diseases where antibodies play a major role.9,10 The overall effect of vitamin D is anti-inflammatory and helps to prevent autoimmunity.8 Taken together, it is evident that vitamin D can regulate at least some immune responses and that its role is mostly anti-inflammatory.
Clinical presentation of COVID-19
As mentioned previously, the SARS-CoV-2 (COVID-19) pandemic has affected the lives of many worldwide. In December 2019, the first cases of an unknown respiratory illness in three hospitalized patients caused by a novel virus were reported to the WHO and by 11 March 2020, WHO declared the novel coronavirus outbreak a pandemic.16,17 When SARS-CoV-2 was declared a pandemic, it had already spread to 118 countries, via human to human transmission, and already had over 118,000 cases.16 To determine the contagiousness of a disease, the basic reproduction rate (R0) can be calculated using three main considerations: (1) the potential infectious period; (2) the likelihood of infection between the infected versus the non-infected; and (3) the contact rate.18 An endemic is likely for any disease process that has an R0 value greater than 1. The SARS-CoV-2 virus has an estimated R0 of 2.2 to 3.58, meaning that each infected patient with CoV-2 can potentially spread the disease to an average of 2–4 people.19,20
Coronaviruses are a family of enveloped positive sense RNA viruses that are grouped into four types: alpha, beta, gamma, and delta coronaviruses.21 Coronaviruses more commonly cause diseases in animals, but can also infect humans, most often causing the common cold. However, in recent years, they have also caused more serious diseases in humans, such as SARS-CoV in 2003 and Middle East respiratory syndrome (MERS-CoV) in 2012.21 Shortly after the emergence of SARS-CoV, it was hypothesized that cytokine storms played a large role in the severity of lung inflammation. A 2005 article studied the effects of IFN-γ on human lung epithelial and fibroblast cell lines and found that the alveolar epithelial cells were more susceptible to IFN-γ than the fibroblast cell lines, with IFN-γ enhancing Fas-mediated apoptosis and inhibiting proliferation.22 Moreover, both cell lines were capable of T-cell target cytokine secretion, with the cytokine profile similar to those detected in SARS patients. It was proposed that IFN-γ may be culpable for acute lung injury during the late phase of SARS infection. Cytokine profiles in patients with MERS-CoV infection demonstrated a Th1 and Th17 response with high levels of pro-inflammatory cytokine secretions such as IFN-γ, TNF-α, IL-15 and IL-17 when compared to a control group.23 Both SARS-CoV and MERS-CoV infections involve a heightened pro-inflammatory cytokine response by the immune system that is thought to play a role in mediating disease severity.
The novel coronavirus is of the beta subtype, which also encompasses SARS.19 The CoV-2 virus also shares 85% of its genomic identity with SARS-CoV suggesting similarities in the diseases, though they are not the same.24 Coronavirus transmission is zoonotic, believed to have originated in bats which serve as host reservoirs.25 The virus is then transmitted from bats to intermediate host animals such as the palm civet or dromedary camels, which can then be spread to humans through close contact or consumption of milk/urine, or uncooked meat, as it was the case with SARS and MERS, respectively.25,26
The most common symptoms of COVID-19 infection include fever, shortness of breath, and dry cough, which normally presents after 5.2 days of incubation.27 In 41 patients admitted for COVID in a Wuhan hospital, 32% had an underlying disease (diabetes, hypertension, and cardiovascular disease).28 Laboratory findings showed leukopenia (white blood less < 4 × 109/L) in 25% of patients and lymphopenia (lymphocyte count < 1.0 × 109/L) in 63%.28 Plasma concentrations of inflammatory markers between patients treated in the intensive care unit (ICU) versus non-ICU patients showed higher amounts of IL2, IL7, IL10, GCSF, IP10, MCP1, MIPIA, and TNF in ICU patients.28 A retrospective study of 191 COVID patients from two Wuhan hospitals found that IL-6 was also elevated among COVID patients, with non-survivors having almost double the amount of IL-6 compared to the COVID survivors.29 The authors of this study also observed that the most common complication was sepsis (59%) followed by respiratory failure (54%) and ARDS (31%).29 Non-survivor patients experienced a drastic increase in complications compared to survivors. For example, non-survivors had a 93% rate of ARDS, while only a 7% occurrence rate was seen in survivors.29 Other findings seen in the 191 patient cohort was that increased levels of cardiac troponin I, lactate dehydrogenase, and lymphopenia was observed in more severe COVID-19 cases.29 Chest computed tomography (CT) findings in patients with COVID normally demonstrates bilateral ground glass opacities that do not spare the sub-pleural lining.30,31 Some other findings in a retrospective study of 138 hospitalized patients with COVID included atypical presentations such as diarrhea and nausea, older age, and underlying comorbidities in patients with severe disease, and possible complications of ARDS, arrhythmia, and shock.31 In a study looking at 34 SARS-CoV-2 patients in an ICU in France, the authors observed increased thrombotic events, likely due to the heightened inflammatory response as well as immobilization, reporting 27 patients with confirmed deep venous thrombosis (DVT).32 In another study comprising 150 patients with ARDS due to COVID significantly more cases of thrombotic events, mainly pulmonary embolisms were observed when compared to patients with ARDS not caused by COVID (11.7% vs 2.1%, p < 0.008). Patients in both studies also had elevated fibrinogen and D dimer levels.32,33
Treatment of COVID-19 remains under intense study. Current standard care for mild cases is mainly supportive and focused on relieving clinical symptoms and avoiding complications. Corticosteroids and remdesivir limit disease progression and mortality in moderate and severe cases of COVID-19.34 A recent meta-analysis on the effectiveness of remdesivir described similar results.35 Other treatment approaches that appear to lower mortality in critically ill patients are intravenous immunoglobulin, ivermectin, and tocilizumab.34 This evidence, however, remains inconclusive. However, patients who do not require ICU admission and are treated with either interferon-alpha, itolizumab, sofosbuvir plus daclatasvir, anakinra, or convalescent plasma experience a reduction in mortality.34 Convalescent plasma has also shown to decrease viral clearance.34
The potential therapeutic role of vitamin D in COVID-19 was recently tested by a Spanish team.36 In this pilot study, the authors found that oral 25(OH)D given to patients with confirmed cases of COVID-19 admitted to the hospital prevented the need for ICU treatment. Moreover, 2 of the 25 patients not treated with vitamin D died, while no deaths occurred on any of the 50 treated individuals. While the authors acknowledged that further research is needed in this area, they also suggest that vitamin D reduces the severity of COVID-19. Due to the lack of established treatment alternatives, high expectations have been placed in the effectiveness and safety of the vaccines currently in distribution.37
COVID-19 and the immune response
SARS-CoV-2 is primarily spread through respiratory droplets and binds to angiotensin-converting enzyme 2 (ACE2) which is found on type II alveolar cells within the lungs.38–40 ACE2 is the cellular receptor for both SARS-CoV and the SARS-CoV-2.38,39 Once the virus enters the host cell, it will shed its viral RNA and begin replication.39 In order for the virus to be detected by the host immune system, pathogen-associated molecular patterns (PAMPs) or uncapped RNA is detected by pattern recognition receptors (PRRs) such as endosomal RNA receptors, cytosolic RNA sensors, toll-like receptor 3 and 7 (TLR3, TLR7), retinoic acid-inducible gene 1 (RIG1), or melanoma differentiation-associated protein 5 (MDA5).38–40 Once the PRRs have been activated, a series of downstream signaling occurs inducing the activation of NF-κB and type I interferon which will lead to the production of pro-inflammatory cytokines and antiviral proteins respectively.38–40 For example, TLR7 activates myeloid differentiation factor-88 (MyD88) in turn stimulating kinase complexes (IKK/IKK) which induces NF-κB to increase the transcription of proinflammatory cytokines (TNF, IL-6, IL-12).41 MyD88 also promotes antiviral properties through the production of IFN-α.41 The downstream signaling activated by the PRRs detecting viral RNA is an important step in the innate immune response. Activation of interferon aids in ceasing viral replication during the early phases of infection; however, highly pathogenic coronavirus, such as MERS and SARS, appear to inhibit interferon activation thereby blocking the innate immune system.
Another proposed mechanism by which coronaviruses inhibit the host innate immune response is via non-specific proteins (NSP) 1-16.42,43 NSP1 is of particular interest as it is only found in alpha and beta coronaviruses, which are the main groups of coronaviruses that infect humans.43 In SARS-CoV, NSP1 inhibits the expression of INF-β likely by binding to the host 40S ribosomal subunit and causing translation inhibition as well as endonucleolytic mRNA cleavage.43,44
In addition to its well-known effects on vasoconstriction and blood pressure, the renin-angiotensin system (RAS) is also involved in inflammation.45,46 Once RAS is activated, angiotensin II levels increase, eliciting inflammation via NF-κB activation which in turns stimulates proinflammatory cytokines such as TNF, IL-6, and IL-12.41,45 Under normal circumstances, ACE2 halts RAS activation but infection with SARS-CoV-2 leads to ACE2 endocytosis.45 Lower levels of ACE2 caused by SARS-CoV-2 maintain a proinflammatory cycle as it promotes angiotensin II accumulation that translates into acute lung damage and potentially ARDS.45,47 Under some circumstances, vitamin D can lower renin one of the molecules responsible for RAS activation.45,48,49 By decreasing renin, vitamin D can revert the proinflammatory cycle.
SARS-CoV is also known to cause lung damage by augmenting levels of IFN-γ, IL-1, IL-6, IL-8, IL-12 IP-10, and MCP-1 in more severely ill patients.50 Similarly, over activation of the Th1-mediated immune response and subsequent increase in natural killer (NK) cells and polymorphonuclear neutrophils (PMN) lead to lung injury in patients with SARS-CoV.50 This heightened inflammatory response elicited by SARS-CoV closely corresponds to what is currently known about SARS-CoV-2. Patients with more severe cases of SARS-CoV-2 presented higher levels of IL2, IL6, IL7, IL10, GCSF, IP10, MCP1, MIPIA, and TNF.28,29 This influx of cytokines contributes to heightened inflammatory responses and consequently parenchymal damage with eventual progression to ARDS.
Discussion
The COVID-19 pandemic has caused more than 20 million infections worldwide and close to 800,000 deaths as of August 2020. Unfortunately, at the present time, treatment alternatives remain mostly exploratory, including for those individuals with severe cases of COVID-19.
The best treatment approach, as with any other illness, is prevention. High expectations have been placed in the vaccines against SARS-CoV-2 currently in the early stages of distribution. But, are there any other steps that could be taken in parallel to prevent COVID-19 or at least attenuate its most severe complications? This question prompted us as well as other researchers to explore vitamin D’s effects in detail.
Vitamin D’s appeal as a potential treatment alternative comes from its safety profile and because there are data indicating vitamin D supplementation can prevent as well as improve outcomes in respiratory illnesses (for a review, see Grant et al.51). In addition, a recent literature review examined the relationship between vitamin D supplementation, Parkinson’s disease, and COVID-19.52 This study concluded that daily supplementation of vitamin D3 has the potential to slow the progression of Parkinson’s disease while potentially offer protection against COVID-19.52 Not surprisingly, several authors have proposed vitamin D supplementation as a potential alternative to prevent or at least attenuate complications from COVID-19.45,51–53 Direct evidence for the use of vitamin D in COVID-19 is now available from a pilot, randomized, open-label, double-blind, clinical trial developed on 75 patients. This study reported that oral 25(OH)D reduced the number of admissions to the ICU and even the number of fatalities in patients with confirmed infections by SARS-CoV-2.36 Another recent exploratory study in elderly nursing-home residents found that an oral bolus of 80,000 IU of vitamin D3 during the previous month or right after a diagnosis of COVID-19 significantly lowers the mortality rate.54 A bolus of vitamin D also reduced the severity of COVID-19. Later, this same research team expanded its findings by comparing whether monthly or every two or three months oral boluses of vitamin D3 would improve COVID-19 outcomes and severity.55 Elderly patients who received regular boluses of vitamin D3 had better survival rates and less severe COVID-19 than patients prescribed only a single bolus or not taking vitamin D3 at all. High doses of vitamin D3 have also shown benefit in younger populations. Adults between 36 and 51 years old with vitamin D3 deficiency who tested positive for SARS-CoV-2 were randomized to either receive 60,000 IU of oral cholecalciferol daily for 7 days or placebo. More than 60% of individuals receiving vitamin D3 supplementation became SARS-CoV-2 negative after 21 days while only 20.8% showed negative results in the control group.56 In addition, oral cholecalciferol significantly decreased fibrinogen levels but other inflammatory markers remained unchanged such as D-dimer, procalcitonin and C reactive protein.
A different line of evidence suggesting vitamin D is a relevant factor in the treatment of COVID-19 comes from association studies. Several independent research teams have found that low serum levels of vitamin D are associated with more severe cases of COVID-19.57–59 While replicated data indicates mortality is higher in patients with vitamin D deficiency.58–60 In fact, a meta-analysis of 27 published articles concluded that even though vitamin D deficiency is not associated with increased risk of infection by SARS-CoV-2, low vitamin D increases mortality from COVID-19.61
Also associated with vitamin D deficiency is an increased inflammatory reaction in the presence of SARS-CoV-2. Inflammatory markers such as IL-6, TNFα and ferritin have been found to be higher in patients with COVID-19 with serum levels of 25(OH)D less than 20 ng/mL than in individuals with COVID-19 and serum levels of 25(OH)D greater than 20 ng/mL.58 Another study reported that non-survivors from COVID-19 had higher levels of high-sensitivity C-reactive protein, ferritin, IL-6, D-dimer, fibrinogen, and procalcitonin together with lower levels of 25(OH)D when compared with survivors admitted to the same institution.60 This is not surprising if considered that the more serious complications of COVID-19 appear to be related to a heightened immune response mediated at least in part by elevations in proinflammatory cytokines such IFN-γ, IL-2, IL-6, IL-8, IL-10 IL-12, and TNF. The immunomodulating properties of vitamin D could prove useful under these circumstances as there are reports indicating vitamin D can downregulate all these cytokines (Table 1). Moreover, binding of SARS-CoV-2 to ACE2 is another proposed trigger of inflammation related to acute lung damage.45 Clinical data suggest vitamin D can counter dysregulations in the RAS and therefore halt inflammation caused by lower levels of ACE2.45,48,49
Table 1.
Comparison of cytokines increased by SARS-CoV and cytokines downregulated by vitamin D.
| Cytokines increased by SARS-CoV | References | Cytokines downregulated by vitamin D | References |
|---|---|---|---|
| IFN-γ | Wong et al., 200450
Theron et al., 200522 |
IFN-γ | Sassi et al., 20188
Colotta et al., 20179 |
| IL-8 | Wong et al., 200450
Theron et al., 200522 |
IL-8 | Colotta et al., 20179 |
| IL-6 | Wong et al., 200450
Zhou et al., 202029 |
IL-6 | Sassi et al., 20188
Colotta et al., 20179 |
| IL-12 | Wong et al., 200450 | IL-12 | Colotta et al., 20179 |
| IL-1 | Wong et al., 200450 | IL-17 | Sassi et al., 20188
Colotta et al., 20179 |
| TNF | Huang et al., 202028 | TNF-a | Sassi et al., 20188
Colotta et al., 20179 |
| IL-2 | Huang et al., 202028 | IL-2 | Sassi et al., 20188 |
| IL-18 | Theron et al., 200522 | IL-9 | Colotta et al., 20179 |
| MIG | Theron et al., 200522 | IL-4 | Colotta et al., 20179 |
| IL-10 | Wong et al., 200450
Theron et al., 200522 |
IL-10 | Colotta et al., 20179 |
| MCP-1 | Wong et al., 200450
Theron et al., 200522 |
IL-5 | Colotta et al., 20179 |
SARS-CoV: severe acute respiratory coronavirus.
While there is preliminary evidence about the potential usefulness of vitamin D in COVID-19, more extensive clinically controlled studies are needed to draw any definite conclusions.36 Many other questions remain, including how vitamin D supplementation should proceed. It seems reasonable to suggest that vitamin D should be indicated for those with vitamin D deficiency. Vitamin D deficiency is defined by the Institute of Medicine as a 25(OH)D level of less than 20 ng/mL.62 According to the U.S. Institute of Medicine’s 2011 guidelines, it is recommended that people younger than 70 years supplement with 600 IU daily, and people older than 70 years old supplement with 800 IU daily to reach a level of serum 25(OH)D of 20 ng/mL or higher.62
What clinicians need to consider is that even though vitamin D intoxication is rare, it can occur when serum 25(OH)D reaches levels higher than 150–200 ng/mL.63 The resulting side effects could present as hypercalcemia, hypercalciuria, and/or vascular calcification.63,64 Currently, there is no consensus on the dose of vitamin D to be used for either prevention or treatment of COVID-19. Instead, what most experts recommend is vitamin D supplementation for which several strategies have been proposed.65–67 Prescribing 4000 IU per day, which is sometimes needed, or following other approaches such as supplementation of up to 100,000 IU monthly for 3 years should be monitored by a specialist.66,67
Limitations
There is still no direct evidence about the immunomodulatory effects of vitamin D in patients with COVID-19. Here, we rely on association studies in patients infected with SARS-CoV-2 as well as data coming from in vitro and rodent experiments. The clinical trials that have tested vitamin D as part of the treatment approach for COVID-19 are small, recruiting only between 40 and 77 patients. In addition, the doses of vitamin D used varied significantly among studies.
Conclusion
The search for effective treatment approaches to combat COVID-19 remains. Agents capable of modulating the immune response triggered by SARS-CoV-2 are promising alternatives. Thus, the immunomodulatory capacity of vitamin D should be further explored considering there is already preliminary evidence of its effectiveness against COVID-19.
Acknowledgments
The authors would like to thank the Ling and Esther Tan Early Career Professorship endowment given to AB. They are also grateful for the thorough editorial comments provided by Professor Andrew Francis.
Footnotes
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD: Alfredo Bellon  https://orcid.org/0000-0003-2669-084X
https://orcid.org/0000-0003-2669-084X
References
- 1. World Health Organization. Coronavirus (COVID-19) events as they happen, https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen (2020, accessed 26 August 2020).
- 2. World Health Organization. WHO director-general’s remarks at the media briefing on 2019-nCoV on 11 February 2020, https://www.who.int/dg/speeches/detail/who-director-general-s-remarks-at-the-media-briefing-on-2019-ncov-on-11-february-2020 (2020, accessed 11 February 2020).
- 3. World Health Organization. WHO coronavirus disease (COVID-19) dashboard, https://covid19.who.int/ (2021, accessed 13 January 2021). [PubMed]
- 4. Pazirandeh S, Burns D. Overview of Vitamin D. UpToDate, https://www-uptodate-com.medjournal.hmc.psu.edu:2200/contents/overview-of-vitamin-d?search=Overview%20of%20Vitamin%20D&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1. (2019, accessed 26 August 2020).
- 5. Charoenngam N, Holick MF. Immunologic effects of Vitamin D on human health and disease. Nutrients 2020; 12: 2097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Caprio M, Infante M, Calanchini M, et al. Vitamin D: not just the bone. Evidence for beneficial pleiotropic extraskeletal effects. Eat Weight Disord 2017; 22: 27–41. [DOI] [PubMed] [Google Scholar]
- 7. Fabbri A, Infante M, Ricordi C. Editorial—Vitamin D status: a key modulator of innate immunity and natural defense from acute viral respiratory infections. Eur Rev Med Pharmacol Sci 2020; 24(7): 4048–4052. [DOI] [PubMed] [Google Scholar]
- 8. Sassi F, Tamone C, D’Amelio P. Vitamin D: nutrient, hormone, and immunomodulator. Nutrients 2018; 10: 1656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Colotta F, Jansson B, Bonelli F. Modulation of inflammatory and immune responses by vitamin D. J Autoimmun 2017; 85: 78–97. [DOI] [PubMed] [Google Scholar]
- 10. Gombart AF, Pierre A, Maggini S. A review of micronutrients and the immune system-working in harmony to reduce the risk of infection. Nutrients 2020; 12: 236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Zmijewski MA. Vitamin D and human health. Int J Mol Sci 2019; 20: 145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Infante M, Ricordi C, Sanchez J, et al. Influence of Vitamin D on islet autoimmunity and Beta-cell function in Type 1 diabetes. Nutrients 2019; 11: 2185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Ricordi C, Clare-Salzler M, Infante M, et al. Vitamin D and omega 3 field study on progression of Type 1 diabetes. Cellr4 Repair Replace Regen Reprogram 2019; 7: e2737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Nair R, Maseeh A. Vitamin D: the “sunshine” vitamin. J Pharmacol Pharmacother 2012; 3: 118–126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Zhang X, Zhou M, Guo Y, et al. 1,25-Dihydroxyvitamin D3 promotes high glucose-Induced M1 macrophage switching to M2 via the VDR-PPARγ signaling pathway. Biomed Res Int 2015; 2015: 157834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Cascella M, Rajnik M, Cuomo A, et al. Features, evaluation, and treatment of coronavirus (COVID-19). Treasure Island, FL: Statpearls, 2020. [PubMed] [Google Scholar]
- 17. Cucinotta D, Vanelli M. WHO Declares COVID-19 a Pandemic. Acta Biomed 2020; 91: 157–160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Delamater PL, Street EJ, Leslie TF, et al. Complexity of the basic reproduction number (R0). Emerg Infect Dis 2019; 25: 171901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Li Q, Guan X, Wu P, et al. Early transmission dynamics in Wuhan, China, of novel coronavirus-infected pneumonia. N Engl J Med 2020; 382: 1199–1207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Zhao S, Lin Q, Ran J, et al. Preliminary estimation of the basic reproduction number of novel coronavirus (2019-nCoV) in China, from2019 to 2020: a data-driven analysis in the early phase of the outbreak. Int J Infect Dis 2020; 92: 214–217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Weiss SR, Leibowitz JL. Coronavirus pathogenesis. Adv Virus Res 2011; 81: 85–164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Theron M, Huang KJ, Chen YW, et al. A probable role for IFN-gamma in the development of a lung immunopathology in SARS. Cytokine 2005; 32: 30–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Mahallawi WH, Khabour OF, Zhang Q, et al. MERS-CoV infection in humans is associated with a pro-inflammatory Th1 and Th17 cytokine profile. Cytokine 2018; 104: 8–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med 2020; 382: 727–733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Yin Y, Wunderink RG. MERS, SARS and other coronaviruses as causes of pneumonia. Respirology 2018; 23(2): 130–137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Song Z, Xu Y, Bao L, et al. From SARS to MERS, Thrusting Coronaviruses into the Spotlight. Viruses 2019; 11: 59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Rothan HA, Byrareddy SN. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J Autoimmun 2020; 109: 102433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020; 395: 497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020; 395: 1054–1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lei J, Li J, Li X, et al. CT imaging of the 2019 novel coronavirus (2019-nCoV) pneumonia. Radiology 2020; 295: 18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA 2020; 323: 1061–1069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Nahum J, Morichau-Beauchant T, Daviaud F, et al. Venous Thrombosis Among Critically Ill Patients With Coronavirus Disease 2019 (COVID-19). JAMA Netw Open 2020; 3: e2010478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Helms J, Tacquard C, Severac F, et al. High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study. Intensive Care Med 2020; 46(6): 1089–1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Kim MS, An MH, Kim WJ, et al. Comparative efficacy and safety of pharmacological interventions for the treatment of COVID-19: a systematic review and network meta-analysis. PLoS Med 2020; 17(12): e1003501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Rezagholizadeh A, Khiali S, Sarbakhsh P, et al. Remdesivir for treatment of COVID-19; an updated systematic review and meta-analysis. Eur J Pharmacol 2021; 897: 173926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Entrenas Castillo M, Entrenas Costa LM, Vaquero Barrios JM, et al. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: a pilot randomized clinical study. J Steroid Biochem Mol Biol 2020; 203: 105751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. National Library of Medicine (US). Identifier NCT04283461 safety and immunogenicity study of 2019-NCoV vaccine (mRNA-1273) for Prophylaxis of SARS-CoV-2 infection (COVID-19), https://clinicaltrials.gov/ct2/show/NCT04283461?term=NCT04283461&draw=2&rank=1 (2020, accessed 3 June 2020).
- 38. Prompetchara E, Ketloy C, Palaga T. Immune responses in COVID-19 and potential vaccines: lessons learned from SARS and MERS epidemic. Asian Pac J Allergy Immunol 2020; 38(1): 1–9. [DOI] [PubMed] [Google Scholar]
- 39. de Wit E, van Doremalen N, Falzarano D, et al. SARS and MERS: recent insights into emerging coronaviruses. Nat Rev Microbiol 2016; 14(8): 523–534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Ahmadpoor P, Rostaing L. Why the immune system fails to mount an adaptive immune response to a COVID-19 infection. Transpl Int 2020; 33(7): 824–825. [DOI] [PubMed] [Google Scholar]
- 41. Zhou Y, He C, Wang L, et al. Post-translational regulation of antiviral innate signaling. Eur J Immunol 2017; 47(9): 1414–1426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Fung SY, Yuen KS, Ye ZW, et al. A tug-of-war between severe acute respiratory syndrome coronavirus 2 and host antiviral defence: lessons from other pathogenic viruses. Emerg Microbes Infect 2020; 9(1): 558–570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Narayanan K, Ramirez SI, Lokugamage KG, et al. Coronavirus nonstructural protein 1: common and distinct functions in the regulation of host and viral gene expression. Virus Res 2015; 202: 89–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Kamitani W, Narayanan K, Huang C, et al. Severe acute respiratory syndrome coronavirus nsp1 protein suppresses host gene expression by promoting host mRNA degradation. Proc Natl Acad Sci U S A 2006; 103: 12885–12890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Biesalski HK. Vitamin D deficiency and Co-morbidities in COVID-19 patients-a fatal relationship? NFS J 2020; 20: 10–21. [Google Scholar]
- 46. Marchesi C, Paradis P, Schiffrin EL. Role of the renin-angiotensin system in vascular inflammation. Trends Pharmacol Sci 2008; 29: 367–374. [DOI] [PubMed] [Google Scholar]
- 47. Hanff TC, Harhay MO, Brown TS, et al. Is there an association between COVID-19 Mortality and the Renin-Angiotensin system? A call for epidemiologic investigations. Clin Infect Dis 2020; 71: 870–874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Burgess ED, Hawkins RG, Watanabe M. Interaction of 1,25-dihydroxyvitamin D and plasma renin activity in high renin essential hypertension. Am J Hypertens 1990; 3(12 Pt. 1): 903–905. [DOI] [PubMed] [Google Scholar]
- 49. Schroten NF, Ruifrok WP, Kleijn L, et al. Short-term vitamin D3 supplementation lowers plasma renin activity in patients with stable chronic heart failure: an open-label, blinded end point, randomized prospective trial (VitD-CHF trial). Am Heart J 2013; 166(2): 357–364. [DOI] [PubMed] [Google Scholar]
- 50. Wong CK, Lam CW, Wu AK, et al. Plasma inflammatory cytokines and chemokines in severe acute respiratory syndrome. Clin Exp Immunol 2004; 136(1): 95–103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Grant WB, Lahore H, McDonnell SL, et al. Evidence that Vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients 2020; 12: 988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Hribar CA, Cobbold PH, Church FC. Potential role of Vitamin D in the elderly to resist COVID-19 and to slow progression of Parkinson’s disease. Brain Sci 2020; 10: 284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Laird E, Rhodes J, Kenny RA. Vitamin D and inflammation: potential implications for severity of Covid-19. Ir Med J 2020; 113: 81. [PubMed] [Google Scholar]
- 54. Annweiler C, Hanotte B, Grandin de, l’Eprevier C, et al. Vitamin D and survival in COVID-19 patients: a quasi-experimental study. J Steroid Biochem Mol Biol 2020; 204: 105771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Annweiler G, Corvaisier M, Gautier J, et al. Vitamin D supplementation associated to better survival in hospitalized Frail elderly COVID-19 patients: the GERIA-COVID quasi-experimental study. Nutrients 2020; 12: 3377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Rastogi A, Bhansali A, Khare N, et al. Short term, high-dose vitamin D supplementation for COVID-19 disease: a randomised, placebo-controlled, study (SHADE study). Postgrad Med J 2020; 1: 1–4. [DOI] [PubMed] [Google Scholar]
- 57. Ye K, Tang F, Liao X, et al. Does serum Vitamin D level affect COVID-19 infection and its severity?-A case-control study. J Am Coll Nutr. Epub ahead of print 22 July 2020. DOI: 10.1080/07315724.2020.1826005. [DOI] [PubMed] [Google Scholar]
- 58. Jain A, Chaurasia R, Sengar NS, et al. Analysis of vitamin D level among asymptomatic and critically ill COVID-19 patients and its correlation with inflammatory markers. Sci Rep 2020; 10: 20191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Gavioli EM, Miyashita H, Hassaneen O, et al. An evaluation of serum 25-Hydroxy Vitamin D levels in patients with COVID-19 in New York City. J Am Coll Nutr. Epub ahead of print 19 February 2021. DOI: 10.1080/07315724.2020.1869626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Infante M, Buoso A, Pieri M, et al. Low Vitamin D status at admission as a risk factor for poor survival in hospitalized patients with COVID-19: an Italian retrospective study. J Am Coll Nutr. Epub ahead of print 18 February 2021. DOI: 10.1080/07315724.2021.1877580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Pereira M, Dantas Damascena A, Galvão Azevedo LM, et al. Vitamin D deficiency aggravates COVID-19: systematic review and meta-analysis. Crit Rev Food Sci Nutr. Epub ahead of print 4 November 2020. DOI: 10.1080/10408398.2020.1841090. [DOI] [PubMed] [Google Scholar]
- 62. Ross AC, Manson JE, Abrams SA, et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab 2011; 96(1): 53–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Marcinowska-Suchowierska E, Kupisz-Urbańska M, Łukaszkiewicz J, et al. Vitamin D toxicity: a clinical perspective. Front Endocrinol 2018; 9: 550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Marcinowska-Suchowierska E, Pludowski P, Witamina Z. Vitamin D toxicity. Post N Med 2016; 29(10): 756–759. [Google Scholar]
- 65. Fassio A, Adami G, Rossini M, et al. Pharmacokinetics of oral cholecalciferol in healthy subjects with Vitamin D deficiency: a randomized open-label study. Nutrients 2020; 12: 1553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2011; 96(7): 1911–1930. [DOI] [PubMed] [Google Scholar]
- 67. Malihi Z, Lawes CMM, Wu Z, et al. Monthly high-dose vitamin D supplementation does not increase kidney stone risk or serum calcium: results from a randomized controlled trial. Am J Clin Nutr 2019; 109: 1578–1587. [DOI] [PubMed] [Google Scholar]


