Abstract
β-lactam resistance represents a worldwide problem and a serious challenge for antimicrobial treatment. Hence this research was conducted to recognize several mechanisms mediating β-lactam resistance in E. coli and K. pneumoniae clinical isolates collected from Mansoura University hospitals, Egypt. A total of 80 isolates, 45 E. coli and 35 K. pneumoniae isolates, were collected and their antibiotic susceptibility was determined by the Disc diffusion method followed by phenotypic and genotypic detection of extended-spectrum β-lactamases (ESBLs), AmpC β-lactamase, carbapenemase enzymes. The outer membrane protein porins of all isolates were analyzed and their genes were examined using gene amplification and sequencing. Also, the resistance to complement-mediated serum killing was estimated. A significant percentage of isolates (93.8%) were multidrug resistance and showed an elevated resistance to β-lactam antibiotics. The presence of either ESBL or AmpC enzymes was high among isolates (83.75%). Also, 60% of the isolated strains were carbapenemase producers. The most frequently detected gene of ESBL among all tested isolates was blaCTX-M-15 (86.3%) followed by blaTEM-1 (81.3%) and blaSHV-1 (35%) while the Amp-C gene was present in 83.75%. For carbapenemase-producing isolates, blaNDM1 was the most common (60%) followed by blaVIM-1 (35%) and blaOXA-48 (13.8%). Besides, 73.3% and 40% of E. coli and K. pneumoniae isolates respectively were serum resistant. Outer membrane protein analysis showed that 93.3% of E. coli and 95.7% of K. pneumoniae isolates lost their porins or showed modified porins. Furthermore, sequence analysis of tested porin genes in some isolates revealed the presence of frameshift mutations that produced truncated proteins of smaller size. β-lactam resistance in K. pneumoniae and E. coli isolates in our hospitals is due to a combination of β-lactamase activity and porin loss/alteration. Hence more restrictions should be applied on β-lactams usage to decrease the emergence of resistant strains.
1. Introduction
In the 21stcentury, the emergence and dissemination of resistant bacteria to antimicrobial agents is considered a challenging and threat to global public health as antibiotic resistance leads to higher medical costs, longer hospital stays, and increased mortality [1, 2]. β-lactam antibiotics are among the most commonly prescribed antibiotics due to their minimal side effects and broad antibacterial spectrum. However, various mechanisms are responsible for resistance to β-lactam compounds, such as the production of degrading enzymes, alteration of the drug target (modification of penicillin-binding proteins), decreased membrane permeability, and drug efflux pump [3, 4].
β-lactam compounds resistance is a rising problem and the production of different β-lactamases was documented to be the main cause of β-lactam resistance especially among gram-negative bacilli. Carbapenems group is often used as β-lactam of last resort in treating infections caused by multi-drug resistant bacteria especially those belonging to Enterobacteriaceae. However, in this family reduced susceptibility to carbapenem group can be frequently associated with carbapenemases production, membrane impermeability coupled with elevated expression of other β-lactamases, or a combination of these mechanisms [5–7].
The gram-negative bacteria outer membrane permits the passive diffusion of small hydrophilic solutes and important antibiotics (β-lactams and fluoroquinolones) by channel-forming proteins [8]. Porins are considered one class of these proteins that are present in gamma-proteobacteria members such as E. coli, Salmonella, Shigella, and others [9–12]. Porins also maintain the envelope integrity of the cells and act as receptors for bacteriocins and bacteriophages. Additionally, they participate in bacterial pathogenesis such as invasion, serum resistance, and adherence [13]. In gram-negative bacteria, different porins types have been identified and classified according to their functional structure, their activity, and their regulation and expression [10].
From the gram-negative bacteria, E. coli and K. pneumoniae which are responsible for the majority of nosocomial and community-acquired infections. The three major porins that have been identified in E. coli include OmpF, OmpC, and PhoE which differ from one another according to charge and size of solutes [10, 14, 15]. Also, K. pneumoniae contains two main porins, Ompk35, and Ompk36, through which hydrophilic solutes gain access to bacterial-cell [10]. Loss of membrane permeability may be due to mutation in porin that renders it non-functional or alteration in expression level or may be due to complete loss of the porin proteins [16]. Many studies illustrated that loss of Ompk35 and/or Ompk36 in K. pneumoniae, contribute to their resistance to cephalosporins and carbapenems [17–19]. Also, mutations in the OmpC or OmpF genes that result in subsequent porin loss have been reported in the resistance of Enterobacter spp., E. coli, and S. marcescens, for carbapenems [20, 21]. This work aimed to investigate the prevalence of porin alteration mediated resistance to β- lactam antibiotics in β-lactamase producing and non-producing strains of multidrug-resistant clinical isolates of E. coli and K. pneumoniae.
2. Methods
2.1. Bacterial isolates
A total of 80 clinical isolates were separated from urine and sputum samples which were collected from microbiological laboratories in Mansoura University hospitals, Dakahlia governorate, Egypt. These isolates were identified as 45 isolates of E. coli and 35 isolates of K. pneumoniae using biochemical standard assay methods [22, 23]. This work was done after approval of the administrative authorities (Research Ethics Committee) in the faculty of pharmacy. Mansoura University, Egypt.
2.2. Antimicrobial susceptibility testing for isolates
The susceptibility of all isolates was determined using the disk diffusion method to different antimicrobials including cefotaxime (30 μg), ceftriaxone (30μg), ceftazidime (30 μg), cefepime (30 μg), piperacillin-tazobactam (36 μg), amoxicillin-clavulanic acid(30μg), imipenem (10 μg), amikacin (30 μg), trimethoprim/sulfamethoxazole (25 μg), Ofloxacin (5 μg), and nitrofurantoin (100 μg) on Mueller-Hinton agar plates [24]. All antibiotic discs were obtained from Oxoid, United Kingdom. According to zones of inhibitions, bacterial strains were classified as resistant, intermediate, or susceptible using Clinical and Laboratory Standard Institute guidelines [25].
2.3. Phenotypic screening for ESBLs enzymes production
The production of ESBL by clinical isolates was tested by Modified Double Disc Synergy Test (MDDST) using amoxicillin-clavulanic acid disc (20/10 μg) along with three third-generation cephalosporins discs {ceftazidime(30μg), ceftriaxone (30 μg) and cefotaxime (30 μg)} and one fourth-generation cephalosporin disc (cefepime 30 μg) [26]. The amoxicillin-clavulanate disc was centered on Mueller-Hinton agar plate lawned with test organism suspension equivalent to 0.5 McFarland standards. The discs of cephalosporins were placed around the amoxicillin-clavulanate disc with a distance of 15mm (for third-generation cephalosporins discs) and 20mm (for fourth-generation cephalosporin disc), and then the plates were incubated at 37°C. Positive results for ESBL production are considered by any increase in the inhibition zone around any of these cephalosporin discs towards the disc of amoxicillin-clavulanate.
2.4. Phenotypic screening for AmpC enzyme production
AmpC enzyme production for all isolated strains was determined by three- dimensional enzyme extract method [27]. Mueller Hinton agar plates were inoculated by E. coli DH5α and cefoxitin discs (30 μg) were centered on them. The extraction of crude enzymes from all isolates was done as mentioned by Livermore et al., 1984 [28]. A circular well and a linear slit were made in the agar plates as described before [29]. The enzyme extracts were added to the wells then the plates were kept upright for 5–10 min and incubated overnight at 37°C. Clear distortion of cefoxitin disc inhibition zone was shown in isolates which were AmpC β-lactamase producers while AmpC β-lactamase non-producers gave no distortion.
2.5. Phenotypic screening for carbapenemases enzymes production
All of the tested isolates were screened for carbapenemase production on Mueller-Hinton agar plates using the Modified Hodge test (MHT) as described by [30] using E. coli ATCC 25922 as an indicator and a meropenem disc (10μg).
2.6. PCR screening for β-lactamase genes and porin encoding genes
The presence of ESBL encoding genes (blaTEM-1, blaSHV-1, blaCTX-M-15), Amp-c gene, carbapenemase- encoding genes (blaNDM-1, blaVIM-1, and blaOXA-48) among all E. coli and K. pneumoniae isolates and also porin encoding genes (ompK35 for K. pneumoniae isolates and ompC and ompF for E. coli isolates) were examined by PCR utilizing the primers listed in Table 1. Genomic DNA was prepared from heating one-two colonies in 100 μl of distilled water at 95°C for 10 min. A reaction mixture contained 1μl of each primer (10 μM), 12.5 μl Dream Taq Green PCR Master Mix (2x) (Thermo Fisher Scientific Inc, USA), 1μl of total DNA, and 9.5 μl nuclease-free water. The PCR program was done as follows: initial denaturation at 94°C for 10 min; 35 cycles of DNA denaturation at 94°C for 30 s, annealing at a temperature specified for each primer as listed in Table 1 for 30 S and extension at 72°C for 1 min followed by final elongation step at 72°C for 7 min. The PCR products were visualized by electrophoresis in 1% agarose gels stained with ethidium bromide.
Table 1. Primers sequences used for screening tested genes.
| Gene Type | Primer | Nucleotide sequence | Annealing Temp | Amplicon size (bp) | reference | |
|---|---|---|---|---|---|---|
| Carbapenemase genes primers | VIM-1 | F | 5`–GAGCTCTTCTATCCTGGTG– 3` | 52°C | 103 | [29] |
| R | 5`–CTTGACAACTCATGAACGG– 3` | |||||
| NDM-1 | F | 5`–ACTTCCTATCTCGACATGC– 3` | 52°C | 133 | ||
| R | 5`–TGATCCAGTTGAGGATCTG– 3` | |||||
| OXA-48 | F | 5`–TTGGTGGCATCGATTATCGG– 3` | 55°C | 743 | ||
| R | 5`–GAGCACTTCTTTTGTGATGGC– 3` | |||||
| AmpC genes primers | AmpC | F | 5’-ACACGAGTTTGCATCGCCTG-3’ | 60°C | 254 | [27] |
| AmpC | R | 5’-CTGAACTTACCGCTAAACAGTGGAAT-3’ | ||||
| ESBL genes primers | SHV | F | 5’-ACTATCGCCAGCAGGATC-3’ | 53°C | 356 | [32] |
| SHV | R | 5’-ATCGTCCACCATCCACTG-3’ | ||||
| TEM | F | 5’-GATCTCAACAGCGGTAAG-3’ | 50°C | 786 | ||
| TEM | R | 5’-CAGTGAGGCACCTATCTC-3’ | ||||
| CTX-M-15 | F | 5’-GTGATACCACTTCACCTC-3’ | 49°C | 255 | ||
| CTX-M-15 | R | 5’-AGTAAGTGACCAGAATCAG-3’ | ||||
| Porin primers | OmpC | F | TAG GTG CTT ATT TCG CCA TTC | 56°C | 1443 | This study |
| OmpC | R | GTA CGT GAT TAT CCT CAT GCG | ||||
| OmpF | F | AGC ACT TTC ACG GTA GCG AAA | 54°C | 1341 | ||
| OmpF | R | AGG CTG TTT TTG CAA GAC GTG | ||||
| OmpK35 | F | CGC TTT GGT GTA ATC GTT GTC | 56°C | 1128 | ||
| OmpK35 | R | GAC ACC AAA CTG TCA TCA ATG | ||||
F, forward; R, reverse; bp, base pair.
2.7. Isolation and characterization of the outer-membrane proteins
The outer-membrane proteins (OMPs) of all E.coli and K. pneumoniae isolates were analyzed by SDS-PAGE [31] and compared to standard E.coli ATCC 25922 and K. pneumoniae ATCC 33495. Barwa and Shaaban procedure was used for the preparation of OMPs from all tested isolates [32]. Then OMPs were analyzed by SDS-PAGE (10%) and gels were visualized by staining with Coomassie blue.
2.8. Serum bactericidal assay
The resistance of all isolates to complement-mediated serum killing was estimated; bacteria were cultivated in nutrient broth until mid-logarithmic phase, centrifuged, washed twice with barbital buffer saline (BBS), and resuspended to a final concentration of 106 CFU mL-1 in BBS. 40% normal human serum (NHS) in BBS was inoculated with the bacteria for 2 hrs at 37°C with shaking. Heat-inactivated NHS at 56°C for 30 min (HI-NHS) was considered as control. Samples which were taken at 0 and 2 hrs were serially diluted and then cell survival was determined by plating them onto nutrient agar plates followed by overnight incubation at 37°C. Each isolate was tested three times. The killing of bacteria by serum was estimated by measuring the decrease in the viable count over time [33].
2.9. Sequencing analysis of OMP genes
OmpF gene of three E. coli isolates (E7, E27, and E36), ompC gene of one E. coli isolate (E38) and ompK35 gene of two K. pneumoniae isolates (K5 and K 23) were sequenced as previously described [34]. In brief, the tested outer membrane protein genes were amplified using PCR, each reaction mixture (50ul) contained 1 ng of DNA, 0.5 μm of each primer, 200 μm of dNTPs, 2mM MgCl2,10 μl of 5x Q5 buffer and 1 U of Q5 High-Fidelity DNA polymerase (NEB, UK). The QIA quick Gel Extraction kit (Qiagen, USA) was employed to purify PCR products according to the supplier’s protocol. The purified amplicons were subsequently sequenced in both directions with the Sanger method using the BigDye® Terminator v3.1 Cycle Sequencing Kit and Applied Biosystems Genetic Analyzer 3500 (Thermo Fisher Scientific Inc, USA). The obtained sequence for each strain was analyzed and aligned with ompF, ompC of E. coli K12 reference gene sequences, and ompK35 reference gene sequence (GenBank Accession no JX310553.1) using Blast program of NCBI. To identify non-synonymous point mutations, DNA sequences were translated to amino acids using the BLAST software.
2.10. Statistical analysis
Data were fed to the computer and analyzed using IBM SPSS Corp. Released 2013. IBM SPSS Statistics for Windows, Version 22.0. Armonk, NY: IBM Corp. Qualitative data were described using number and percent. Significance of the obtained results was judged at the (0.05) level. Graph pad prism version 6.01 was used for figure design. Chi-Square test for comparison of 2 or more groups.
3. Results
3.1. Identification and antimicrobial susceptibility testing of bacterial strains
In this study, 80 clinical isolates of Enterobacteriaceae were collected from several microbiological laboratories in Mansoura University Hospitals. Of these, 45 isolates were identified as E. coli and 35 isolates were identified as K. pneumoniae using the standard biotyping method. Twenty- six E. coli isolates and 16 K. pneumoniae isolates were obtained from urine samples collected from the Urology and Nephrology Center whereas 5 and 6 isolates of K. pneumoniae were separated from urine samples collected from Emergency Hospital and Children Hospital respectively. Additionally, 19 E. coli isolates and 8 isolates of K. pneumoniae were obtained from sputum samples which were collected from Emergency Hospital and Children Hospital.
According to the breakpoints which were indicated in CLSI guidelines [25], the antibiotic susceptibility patterns for all isolates were analyzed. Resistance was observed to amoxicillin-clavulanic acid in 93.3% of E. coli isolates followed by 91.1% were resistant to ceftazidime, ceftriaxone, cefotaxime, ofloxacin, and trimethoprim-sulfamethoxazole in E. coli isolates. Also, E. coli strains showed a high resistance level to piperacillin-tazobactam (88.9%), cefepime (68.9℅), and imipenem (60%). In contrast, resistance to nitrofurantoin was seen only in 35.6% of E. coli isolates, and susceptibility to amikacin was retained by most of E. coli isolates (86.7%).
Furthermore, the highest resistance rate was recorded among K. pneumoniae isolates against ceftazidime (91.4%), ceftriaxone, amoxicillin-clavulanic acid, and trimethoprim-sulfamethoxazole (88.6℅), cefotaxime, and ofloxacin. (85.7℅), piperacillin-tazobactam and nitrofurantoin (82.9%), cefepime (74.3%), amikacin (67.7℅), and 23 isolates (65.7%) were resistant to imipenem.
3.2. Analysis of ESBLs and AmpC enzymes production
ESBLs and AmpC enzymes were both phenotypically (Fig 1) and genetically detected (Fig 2). The Determination of ESBL production by the MDDST method showed that 38 isolates of E. coli (84.4%) and 29 isolates of K. pneumoniae (82.9%) were considered positive for ESBL production (Figs 1A and 3). On the other hand, AmpC production which was detected by the three-dimensional extract method indicated that 38 E. coli isolates and 29 K. pneumoniae isolates were AmpC producers (Figs 1B and 3B). Moreover, phenotypic tests showed that 3 E. coli isolates and 6 K. pneumoniae isolates were considered ESBL and AmpC non-producers besides, 4 E. coli isolates produced only ESBL, and 4 E. coli isolates had AmpC activity only among all tested isolates while the remainder 34 E. coli produced both ESBL and AmpC enzymes.
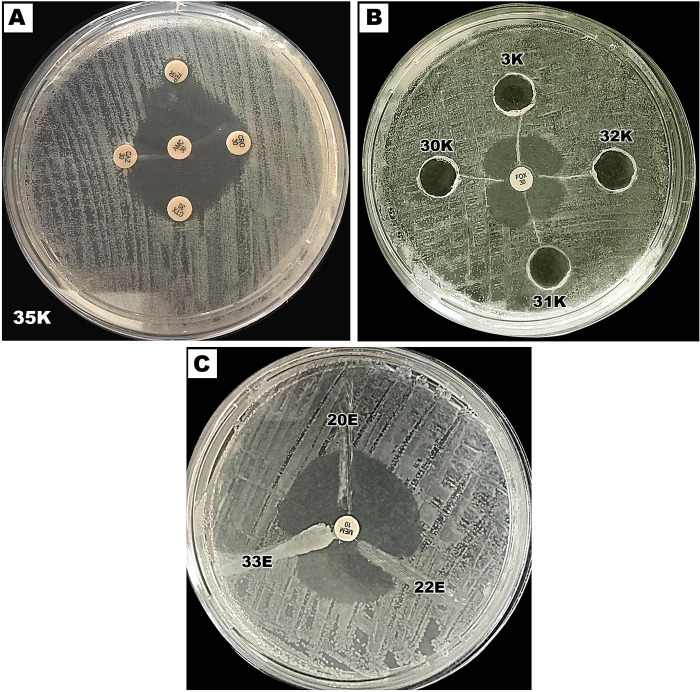
Fig 1. Phenotypic detection of β-lactamase production.
A. Modified-double disc synergy test for determination of ESBL production. B. The 3-dimensional enzyme extract test for detection of AmpC production. C. Modified Hodge test for detection of carbapenemase production.
Fig 2. PCR assay for β-lactamase enzyme genes.
Lane M was 100bp DNA marker and lane C was a negative control. A. Agarose gel electrophoresis of AmpC gene amplicons (254 bp). B. Agarose gel electrophoresis of blaCTX-M-15 gene amplicons (255 bp). C. Agarose gel electrophoresis of blaSHV-1 gene amplicons (356 bp). D. Agarose gel electrophoresis of blaTEM-1 gene amplicons (786 bp).
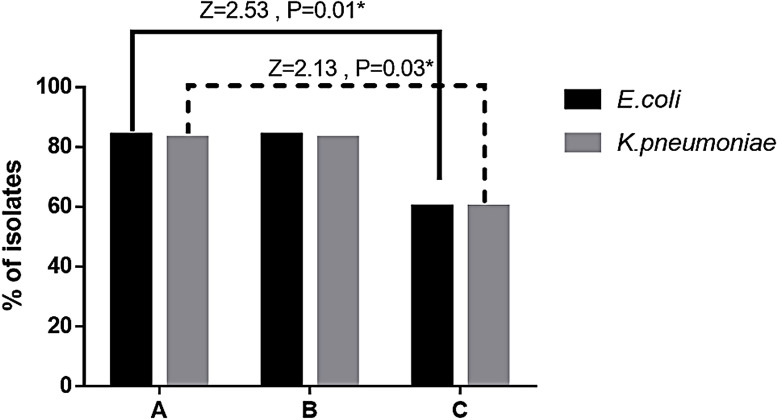
Fig 3. Frequency of β-lactamase producing tested isolates by phenotypic analysis.
A. ESBL producers, B. AmpC producers, C. Carbapenemase producers. A statistically significant difference was detected between ESBL producers and carbapenemase producers among K. pneumoniae and similarly among E. coli without a significant difference between ESBL producers and AmpC producers in either type. Results were analyzed with Z-test for proportion with p<0.05*.
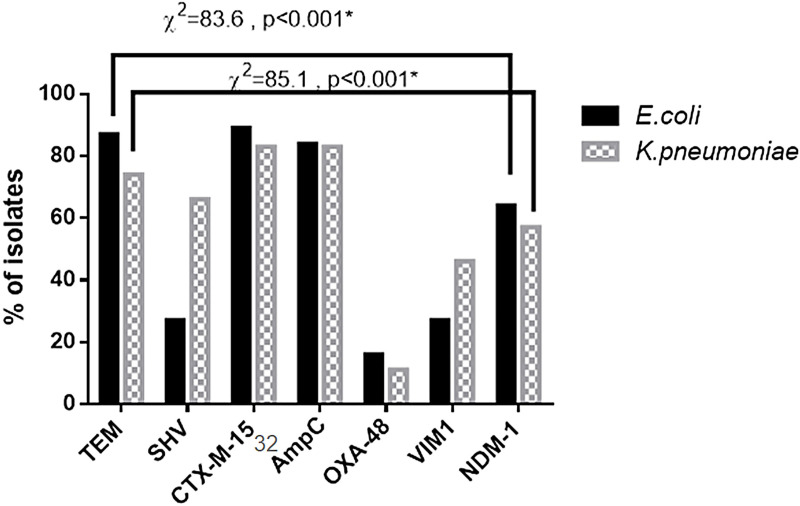
Considering PCR analysis, 95.6% of E. coli isolates (43/45) and 85.7% of K. pneumoniae (30/35) had at least one of the tested ESBL genes. The distribution of tested ESBL genes among the clinical isolates revealed that blaTEM-1and blaCTX-M-15were predominant among E. coli isolates (87% and 89% respectively). In contrast, blaSHV-1 was detected in 27% of E. coli isolates. Moreover, blaSHV-1, blaTEM-1, and blaCTX-M-15 genes were predominantly carried by K. pneumoniae isolates whereas they were detected in 66%, 74%, and 86% (Fig 4). Also, AmpC gene detection by PCR revealed that it was harbored by 38 E. coli isolates and 29 K. pneumoniae isolates (Fig 4). The two E. coli isolates and also two K. pneumoniae which found to be sensitive to all tested antimicrobials showed that they did not harbor any of the tested β-lactamase genes.
Fig 4. Percentage of ESBL, AmpC β-lactamase, and carbapenemase genes among the tested isolates.
There is a statistically significant difference between different genes studied among K. pneumoniae and also among E. coli (p<0.001*). Results were analyzed with the Chi-Square test.
3.3. Analysis of carbapenemases presence
The presence of carbapenemases was studied through the MHT test whereas 27 isolates of E. coli (60%) and 21 K. pneumoniae isolates (60%) had a positive Hodge test hence, they produced a distorted or clover-leaf shaped inhibition zone (Fig 1C). Also, it was found that 25 E. coli isolates and 21 K. pneumoniae isolates were positive for both ESBL and carbapenemase.
Also, PCR indicated that 29 E. coli strains and 19 K. pneumoniae strains harbored the blaNDM-1 gene, and 12 E. coli isolates and 16 K. pneumoniae isolates contained the blaVIM-1 gene (Fig 5). However, blaOXA-48 was present in only 7 isolates of E. coli and 4 isolates of K. pneumoniae (Fig 4). The results showed that the coexistence of the ESBL encoding gene and carbapenemase encoding gene in the same strain was found among all tested E. coli isolates. Also, 21 K. pneumoniae isolates were found to carry any of the tested carbapenemase genes but in association with ESBL and AmpC presence.
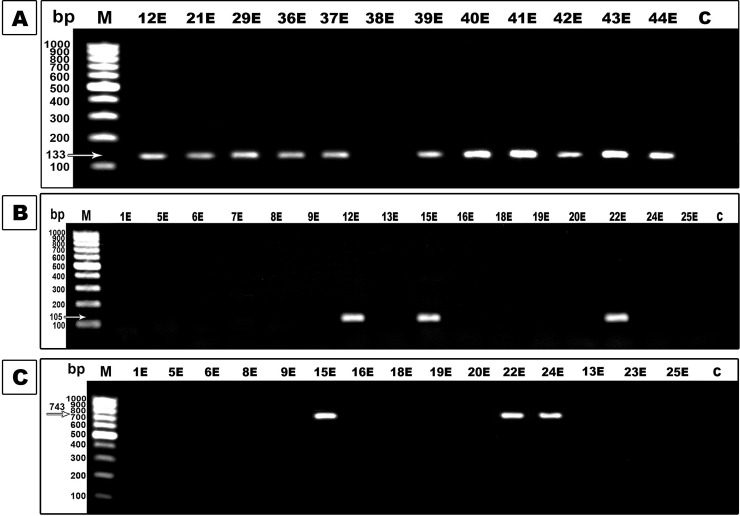
Fig 5. PCR assay for carbapenemases enzymes genes.
Lane M was 100bp DNA marker and lane C was a negative control. A. Agarose gel electrophoresis of the blaNDM-1 gene amplicons (133 bp). B. Agarose gel electrophoresis of blaVIM-1 gene amplicons (105 bp). C. Agarose gel electrophoresis of blaOXA-48 gene amplicons (743 bp).
3.4. Analysis of outer-membrane proteins (porins)
SDS-PAGE analysis of porins showed that 93.3% of E. coli isolates (42/45) and 85.7% of K. pneumoniae isolates (30/35) lost their porins or showed a modification in their electrophoretic migration pattern by comparison to the standard strain (Fig 6). From the results of porins analysis, it was found that 34 E. coli isolates that exhibited a loss in their porins had β-lactamase producing capability except 14 isolates had no carbapenemase-producing capability, 4 isolates did not produce AmpC enzyme and 4 isolates had no ESBL producing capability. Also, E. coli isolates with altered porin pattern had β- lactamase producing capability except for 6 isolates that could not produce carbapenemase enzymes, and 4 isolates were negative for ESBL enzymes but harbored one or more of the tested β-lactamase genes.
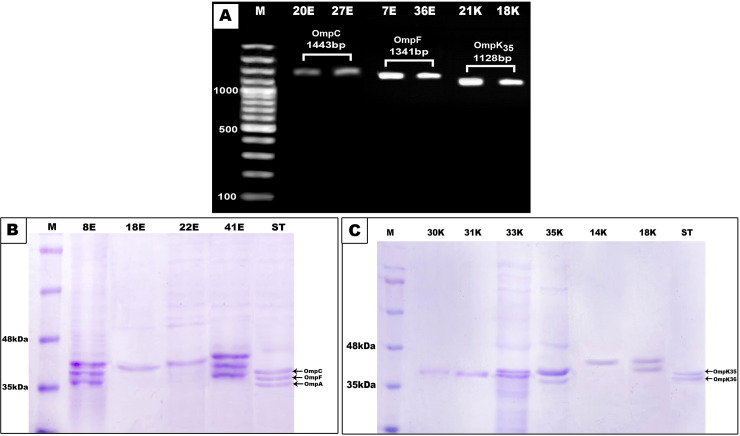
Fig 6. Outer-membrane porin protein and gene analyses.
A. PCR detection of outer membrane porin encoding genes. Lane M: 100 bp plus DNA marker; lanes 1 and 2: amplified product of OmpC gene (1443 bp); lanes 3 and 4: amplified product of OmpF gene (1341 bp) which are porin genes in E. coli; lanes 5 and 6: amplified product of OmpK35 gene (1128bp) which is porin gene in K. pneumoniae. B. SDS-PAGE analysis of outer membrane protein of some E. coli isolates. C. SDS-PAGE analysis of outer membrane protein of some K. pneumoniae isolates. Lane M: marker protein; lane ST: Standard strain used as control.
Regarding K. pneumoniae, 30 isolates that exhibited a loss in their porins or modification in porin pattern, had β -lactamase producing capability except for 8 isolates (no. 6, 15, 16, 20, 22, 25, 26, and 34) had no carbapenemases producing capability. Interestingly, 5 K. pneumoniae isolates that had unaltered porin pattern (isolates no. 2, 3, 13, 17, and 19) could not produce any of the tested β-lactamase enzymes.
Taking into account porin genes PCR analysis, among 45 E. coli isolates, 35 isolates exhibited the OmpC gene and 32 isolates had the OmpF gene. They had β-lactamase producing capability except 4 isolates did not produce AmpC or ESBL, 10 isolates did not produce carbapenemase, and 2 sensitive isolates which had no β-lactamase producing capability. On the other hand, 19 K. pneumoniae isolates harbored the OmpK35 gene, as 14 of them produced ESBL and AmpC enzyme and 9 of them are considered also carbapenemase producers.
3.5. Serum bactericidal assay
The survival of bacteria in 40% NHS was measured for a period of 2 hrs. For E. coli isolates, 73.3% (33/45) were serum resistant and 90.9% (30/33) of these isolates were capable of producing at least one type of the tested β-lactamases (ESBL, AmpC and carbapenemases). On the other hand, 40% (14/35) of K. pneumoniae isolates were serum resistant from which 92.8% (13/14) were positive for all 3 tested β-lactamases. Furthermore, 28.5% (6/21) of serum sensitive K. pneumoniae isolates did not produce any type of the tested β-lactamases. Interestingly, we observed that 90.9% (30/33) of serum resistant E. coli isolates and 100% (14/14) of serum resistant K. pneumoniae isolates showed either loss or alteration in their porin pattern.
3.6. Sequencing analysis of OMP genes
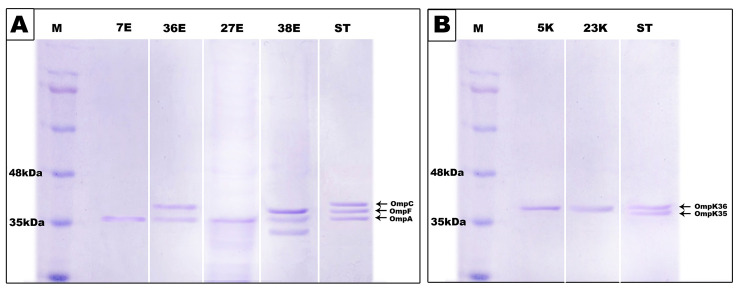
The sequence analysis of ompF, ompC, and ompK35 genes of representative group of isolates (nos. 38E, 7E, 27E, 36E, 5K and 23K) demonstrated that most of these tested genes contained a frameshift mutation and all of them had a stop codon at different positions (Table 2). These mutations resulted in the production of truncated proteins of a smaller size than the wild type proteins. Hence, SDS-PAGE analysis of these isolates showed that they exhibited a modified porin pattern (Fig 7). E. coli isolate no.38 had a frameshift mutation and a stop codon at amino acid position 298 in OmpC porin and accordingly, a band of lower molecular mass (33 Kda) was observed (Fig 7)
Table 2. Detected mutations in tested OmpF, OmpC and OmpK35 OMP genes.
| Strain | Gene | Point mutation | Insertion or deletion of aa | Frameshift mutation |
|---|---|---|---|---|
| 38E | OmpC | • Fourteen missense mutations: D49S, V50E, M57V, S85T, A86S, E90K, N91E, G138D, A274G, N275G, K276E, A277K, Q278H, N279K. • Seven silent mutation • Stop codon aa 298 |
1 insertion: 88D | aa 280 (Deletion of one nucleotide at position 839–840 bp) |
| 1 deletion: 173G | ||||
| 7E | OmpF | • One missense mutations: K28Q • One silent mutation • Stop codon aa 67 |
None | aa 51 (Deletion of one nucleotide at position 150–151 bp) |
| 27E | • Two missense mutations: N26H, N51D • Two silent mutation • Stop codon aa 110 |
1 deletion: 2aa | aa 102 (insertion of one nucleotide at position 305 bp) | |
| 36E | • Nine missense mutations: G48D, E51V, M60K, T61N, Y62L, A63C, R64P, L65S, G66W • Four silent mutation • Stop codon aa 68 |
None | None | |
| 5K | OmpK35 | • One silent mutation • Stop codon aa 63 |
None | aa 62 (Deletion of one nucleotide at position 184–185 bp) |
| 23K | • Stop codon aa 16 | None | aa 14 (Deletion of one nucleotide at position 39–40 bp) |
Fig 7. SDS-PAGE analysis of outer membrane proteins of sequenced genes.
A. SDS-PAGE analysis of outer membrane protein of E. coli isolates 7E, 36E, 27E and 38E. B. SDS-PAGE analysis of outer membrane protein of K. pneumoniae isolates 5K and 23K. Lane M: marker protein; Lane ST: Standard strain used as control.
4. Discussion
The widespread use of antibiotics leads to the emergence of many resistant microorganisms which is increasing worldwide and becoming one of the major serious health care problems [35, 36]. Our results showed a greater prevalence of resistance to common antibiotics and this agreed with that of Hassan et al. [37]. Indeed, the antibiotic susceptibility pattern of either isolated E. coli strains or K. pneumoniae strains revealed a high resistance rate (greater than 80%) which was recorded with ceftazidime, ceftriaxone, cefotaxime, amoxicillin/ clavulanic acid, trimethoprim/ sulfamethoxazole, piperacillin/ tazobactam, and ofloxacin, and these results are in agreement with other reports that showed higher resistance to sulfamethoxazole /trimethoprim (85.7%) and ciprofloxacin (72%) [38]. In contrast, Zaki et al. showed high susceptibility to sulfamethoxazole /trimethoprim and ciprofloxacin for E. coli isolates [39]. Here, it was found that 68.9% of E. coli isolates and 74.3% of K. pneumoniae isolates were resistant to cefepime and these findings were similar to those found in previous studies on antibiotic resistance in E. coli and K. pneumoniae [40, 41]. Carbapenems are considered the last shelter for gram-negative bacteria treatment but the development of carbapenem resistance is increasing in the Middle East. As the existing study demonstrated high resistance to imipenem in the tested E. coli isolates (60%) and K. pneumoniae (65.7%) and these findings were concordant with other studies that were performed in Egypt [39, 42, 43]. While high susceptibility of E. coli isolates was recorded with nitrofurantoin (64.4%) and amikacin (86.7%) but these antibiotics are less effective on K. pneumoniae isolates and this result was found to be similar to other studies [39, 42, 44].
β-lactam antibiotics are among the most widely prescribed drugs for bacterial infection treatment and resistance to them is an increasing problem [45]. The most prevalent mechanism of β-lactam resistance is the production of β-lactamase especially in Enterobacteriaceae [46]. Their continuous mutation leads to various β-lactamases enzymes [47]. Among them, ESBLs are of great interest and have been reported worldwide especially in Enterobacteriaceae [26, 48]. At present, more than 300 different ESBL variants have been identified [24] but the most common types which were reported in Enterobacteriaceae in several areas of the world are CTX-M, SHV, and TEM [49, 50]. Also, AmpC β-lactamases have been found all over the world and contribute to the problem of third generation cephalosporin resistance [51, 52]. Egypt is considered among the countries with the highest rate of ESBL production among Enterobacteriaceae [53], whereas previous studies in Egypt showed an elevated rate of ESBL producing isolates [39, 44, 54, 55]. This coincides with our results as ESBL production was detected in 38 isolates of E. coli (84.4%) and 29 isolates of K. pneumoniae (82.9%), while a low rate of ESBL production was observed by Abdelmegeed et al., in E. coli isolates [56]. As well as higher proportions were found in other countries including India (˃80%) and China (˃60%) [57]. PCR was used for confirmation of phenotypic outcomes and discrimination of different types of genes encoding ESBL enzymes among tested isolates. Many studies in Egypt and other countries showed that the most commonly detected β-lactamase–encoding gene among the ESBL-producing Enterobacteriaceae was blaCTX-M [55, 58] whereas, the blaCTX-M gene was able to be horizontally transferred by several mobile genetic elements [59]. Our data showed that blaCTX-M-15 was the most frequently observed ESBL-encoding gene (found among 89% of E. coli isolates and 86% of K. pneumoniae isolates) and this finding was consistent with the results of other studies performed in Egypt which indicated that blaCTX-M-15 was the most common ESBL encoding gene among ESBL-producing Enterobacteriaceae [55, 60, 61]. Besides, the next most frequently detected gene was blaTEM-1 which was found in 87% of E. coli isolates and 74% of K. pneumoniae isolates. Additionally, SHV was detected in 66% of K. pneumoniae isolates and a small percentage of E. coli isolates (27%) and these results are in agreement with a study of Abdallah et al., who indicated that CTX-M was the predominant detected ESBL gene followed by TEM then by SHV [55]. In contrast to our results, Yazdi et al., and Bajpai et al., reported that the most prevalent β-lactamase-encoding gene was blaTEM followed by blaSHV [62, 63] and in Zaki et al., study SHV was the most commonly detected gene (61.22%) [39]. The wider distribution of ESBL enzymes may be due to the mobilization of their genes on a genetic element as was reported by Shahid et al. [64].
Regarding AmpC enzyme production and the prevalence of its encoding gene, it was found in a higher percentage of E. coli and K. pneumoniae isolates (84.4% and 82.9% respectively). So the resistance to amoxicillin/clavulanate was high in tested isolates as AmpC enzymes are poorly inhibited by clavulanic acid [65]. In contrast, previous studies from Egypt showed a lower prevalence of the AmpC enzyme among Enterobacteriaceae [30, 56, 66]. The possible reason for the high prevalence of ESBL and AmpC producing organisms may be due to excessive usage of extended-spectrum cephalosporin in the treatment of gram-negative infection [67]. An important observation in this study is that many AmpC positive isolates were also ESBL-producer (67/80) and this result was equivocal with a study carried by Park et al., who indicated that the organism which was AmpC-producer is frequently associated with a high co-presence rate of ESBLs [67].
Carbapenems are the preferred therapy for ESBL producing Enterobacteriaceae [68]. However, the development of carbapenemases producing Enterobacteriaceae becomes a public health threat and leaves few therapeutic choices [69]. Many reports that have been published worldwide including Middle East countries on carbapenemases production among Enterobacteriaceae spp. showed a high presence of gram-negative bacteria which exhibited carbapenem resistance in different areas in Egypt [43, 70, 71]. Similarly in the current study, 60% of E. coli isolates and 60% of K. pneumoniae isolates were found to be carbapenemase producers and harbored carbapenemase encoding genes; blaNDM-1, blaVIM-1, and blaOXA-48. This result is consistent with the high rate of carbapenemase production (69.8%) which was reported in Mahmoud et al. study [72] which may be due to widespread abuse of carbapenem in Egyptian hospitals [54, 73]. In contrast, Amjad et al. illustrated that 38% of E. coli isolates and 17% of K. pneumoniae were carbapenemase production positive [74], and Zaki et al., reported that 34.1% of E. coli isolates were carbapenemase producers [39]. Among the many types of carbapenemase encoding genes, the widespread of NDM type among Enterobacteriaceae has been reported in many countries [75–77]. The same was observed in our results which revealed that blaNDM-1 gene was the dominant carbapenemase encoding gene in tested isolates since it was present in all carbapenemase-producing isolates except E25 and E29 which did not contain any of the tested genes and K14 and K27 which contained blaVIM-1 and blaOXA-48 only respectively. These findings confirm previous reports which indicated that blaNDM-1 gene was common in Egypt and the Middle East. [43]. However, a low prevalence of blaNDM-1 gene was found in previous studies [66, 78] and no NDM-1 producing isolates were found in a study in China [79]. Besides this, blaVIM-1 was detected in 35% of the isolated strains while blaOXA-48 was detected in 13.8% of the tested isolates, whereas OXA-48 producing Enterobacteriaceae recently detected in Egypt [32, 39, 80]. Moreover, OXA-48 was commonly identified among Enterobacteriaceae in other countries including Saudi Arabia [81, 82]. North Africa, and Turkey [83]. Results of various studies on K. pneumoniae in Egypt are coincident with the current study as they reported that the NDM gene was the most predominant detectable carbapenemase gene followed by VIM [32, 84]. On the other hand, El kholy et al found that blaOXA-48 dominated (40.6%) followed by blaNDM1 (23.7%) among K. pneumoniae isolates and a small percentage of E. coli harbored blaNDM-1 [61]. It was reported that the NDM1 gene is harbored by diverse plasmids that also carry multiple resistance genes to macrolide, rifampin, carbapenem, cephalosporin, and Sulfamethoxazole and few treatment options are available for those strains which carried this gene [85]. So it was found in our study that the strains which harbored NDM-1 showed elevated variability resistance to various tested antibiotics. The current study showed excessive coexistence of various resistance genes in tested isolate and this leads to elevated variability in resistance and this agreed with that of Martin and Bachman [86].
The β-lactam resistance in gram-negative bacteria has been attributed not only to the presence of the hydrolyzing enzymes but also to the modification in the permeability of the outer membrane as well as to upregulation of multidrug efflux pump and alteration of antibiotic target proteins [11, 87]. Porin loss or mutation of the porin-coding sequence decreases membrane permeability and leads to impairment of antibiotic entry [88], hence loss of porins have been associated with a carbapenem and extended-spectrum cephalosporin resistance [89]. The mutational loss or alteration of porins in Enterobacteriaceae has been reported to be responsible for decreased susceptibility to cephalosporin and carbapenem [90, 91]. The increased resistance of ESBL producer E. coli and K. pneumoniae to cefotaxime and oxyimino β-lactam due to loss of porins was reported in many studies [92, 93]. Also, Domenech-Sanchez et al., found that the combination of decreased outer membrane permeability and the presence of SHV-1 and TEM-1 β-lactamase elevated cefotaxime MIC [94]. Therefore, we examined the porins as an additional resistance mechanism. Our results demonstrated that all the studied isolates showed porin loss or alteration in porin structure except three isolates of E. coli and 5 isolates of K. pneumoniae. And these results are in agreement with that obtained by Kitchel and coauthors who illustrated that the absence of OmpK35 was in 80% of K. pneumoniae isolates [95]. As well as it was observed that all of them harbored one ESBL and/or AmpC enzyme and this agreed with that of Wozniak et al. [88], additionally, this finding is consistent with that ESBL producing K. pneumoniae isolates don’t express OmpK35 and both OmpK35 and Ompk36 porins were expressed in K. pneumoniae isolate lacking ESBL which was reported in other studies [94, 96]. The loss of porins participates in the antimicrobial resistance of ESBL producing bacteria [16]. Besides, previous studies indicated that the absence or low expression of OmpK35in K. pneumoniae and OmpC and OmpF porins in E. cloacae has been associated with carbapenem resistance [34, 97]. The present data confirm this conclusion as almost all imipenem resistant isolates showed loss or alteration in tested porins and also the carbapenemase production was detected in them except 2 out of 23 strains of K. pneumoniae and 9 out of 27 strains of E. coli did not produce carbapenemase and this finding was concordant with the results of Barwa and Shaaban who found that all tested carbapenem-resistant isolates lost OmpK35 except one strain [32]. Besides that, it was reported that porin loss increases the resistance of ESBL-producing organisms to other non- β-lactam drugs such as fluoroquinolones [98, 99] and this explains the reason for the decreased susceptibility to ofloxacin by the tested organisms.
Since serum resistance is an important virulence trait for extraintestinal pathogenic E. coli, we reported that 73.3% of E. coli isolates collected from urine or sputum were serum resistant. However, the ability of serum sensitive isolates (26.7%) to cause extraintestinal infection may be due to the presence of antibodies that inhibited complement-mediated killing as indicated by Coggon et al. [100] In the current study, the association between serum resistance and production of different β-lactamases was clear among E. coli and K. pneumoniae isolates where the possession of β-lactamases was beneficial for some strains regarding serum survival and competitive fitness as reported elsewhere [101–104]. Several studies reported that certain porins in E. coli and K. pneumonia play a role in complement system activation as they promote the deposition of C1q and thus induce antibody-dependent classical pathway bactericidal activity. Therefore, loss or mutation of these immunogenic porins results in elevated resistance to complement-mediated killing [105–107]. This agreed with our results where 93.6% (44/47) of serum resistant E. coli and K. pneumoniae isolates showed either porin loss or alteration in their porin electrophoretic migration pattern.
Mutations in ompK35 gene of tested K. pneumoniae isolates K5 and K23, ompF gene of E. coli isolates E7, E27, and E36 and ompC gene of E. coli isolate E38 involved a stop codon that formed truncated porins lacking the C-terminal phenylalanine residue which is essential for membrane anchoring [108] thus producing nonfunctional protein and porin cannot be inserted in the outer membrane. The same results were observed by Wozniak et al., who found a frameshift mutation and a stop codon at position 144 in the tested ompK35 genes [88]. In addition, Wozniak et al., reported that OmpK36 was the preferred porin and was thus conserved by K. pneumonia because of its smaller pore than OmpK35 and was, therefore, more restrictive for antibiotic entrance. Hence, in our study, the band observed in the SDS-PAGE analysis of 5K and 23K (Fig 7) corresponds most likely to OmpK36.
The exceeding emergence of β-lactam resistance in E. coli and K. pneumoniae strains in Mansoura, Egypt is disturbing. Moreover, our results demonstrated that β-lactam resistance in E. coli and K. pneumoniae isolates were mediated by β-lactamases plus porin loss or mutation of the porin-coding sequence. Hence, the recommendation must be taken during the administration of β-lactam and carbapenem in our hospitals to decrease the spread of β -lactam resistant isolates.
Supporting information
(DOCX)
(DOCX)
(PDF)
Acknowledgments
All thanks and appreciations are given to the clinical laboratories of the Mansoura University Hospitals for providing the clinical isolates which are used in the following study.
Data Availability
All relevant data are within the manuscript and its Supporting Information files.
Funding Statement
The authors received no specific funding for this work.
References
- 1.World; Health Organization (WHO). Antimicrobial resistance: global report on surveillance. Geneva: WHO; 2014. https://www.who.int/drugresistance/documents/surveillancereport/en/. [Google Scholar]
- 2.Cassell Gail H. ASM task force urges a broad program of antimicrobial resistance. ASM News. 1995; 61. [Google Scholar]
- 3.Nikaido H. Prevention of drug access to bacterial targets: permeability barriers and active efflux. Science. 1994. April 15; 264 (5157): 382–388. 10.1126/science.8153625 [DOI] [PubMed] [Google Scholar]
- 4.Spratt BG. Resistance to antibiotics mediated by target alterations. Science. 1994. April 15; 264 (5157): 388–393. 10.1126/science.8153626 [DOI] [PubMed] [Google Scholar]
- 5.Raimondi A, Traverso A, Nikaido H. Imipenem-and meropenem-resistant mutants of Enterobacter cloacae and Proteus Rettgeri lack porins. Antimicrobial agents and chemotherapy. 1991; 35(6): 1174–1180. 10.1128/aac.35.6.1174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cornaglia G, Russell K, Satta G, Fontana R. Relative importance of outer membrane permeability and group 1 beta-lactamase as determinants of meropenem and imipenem activities against Enterobacter cloacae. Antimicrobial agents and chemotherapy. 1995; 39(2): 350–355. 10.1128/aac.39.2.350 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Temkin E, Adler A, Lerner A, Carmeli Y. Carbapenem-resistant Enterobacteriaceae: biology, epidemiology, and management. Ann. N. Y. Acad. Sci. 2014; 1323:22–42 10.1111/nyas.12537 [DOI] [PubMed] [Google Scholar]
- 8.Achouak W, Heulin T, Pagès J.-M. Multiple facets of bacterial porins. FEMS microbiology letter. 2001; 199(1): 1–7. 10.1111/j.1574-6968.2001.tb10642.x [DOI] [PubMed] [Google Scholar]
- 9.Koebnik R, Locher KP, Van Gelder P. Structure and function of bacterial outer membrane proteins: barrels in a nutshell. Molecular microbiology. 2000; 37(2): 239–253. 10.1046/j.1365-2958.2000.01983.x [DOI] [PubMed] [Google Scholar]
- 10.Schulz GE. The structure of bacterial outer membrane proteins. Biochimica et Biophysica Acta (BBA)-Biomembranes. 2002. October 11; 1565(2):308–317. 10.1016/s0005-2736(02)00577-1 [DOI] [PubMed] [Google Scholar]
- 11.Ghai I, Ghai S. Exploring bacterial outer membrane barrier to combat bad bugs. Infection and drug resistance. 2017; 10: 261. 10.2147/IDR.S144299 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ghai I, Ghai S. Understanding antibiotic resistance via outer membrane permeability.Infection and drug resistance. 2018; 11: 523. 10.2147/IDR.S156995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Buchanan SK. β-Barrel proteins from bacterial outer membranes: structure, function and refolding. Current opinion in structural biology. 1999; 9(4):455–461. 10.1016/S0959-440X(99)80064-5 [DOI] [PubMed] [Google Scholar]
- 14.Delcour AH. Solute uptake through general porins. Front. Biosci, 2003. May 1; 8:1055–1071. 10.2741/1132 [DOI] [PubMed] [Google Scholar]
- 15.Nikaido H. Molecular basis of bacterial outer membrane permeability revisited. Microbiology and molecular biology reviews. 2003. December 1; 67(4):593–656. 10.1128/mmbr.67.4.593-656.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Doménech-Sánchez A, Martínez-Martínez L, Hernández-Allés S, del Carmen Conejo M, Pascual Á, Tomás JM, et al. Role of Klebsiella pneumoniae OmpK35 porin in antimicrobial resistance. Antimicrobial agents and chemotherapy. 2003; 47(10):3332–3335. 10.1128/aac.47.10.3332-3335.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kaczmarek FM, Dib-Hajj F, Shang W, Gootz TD. High-level carbapenem resistance in a Klebsiella pneumoniae clinical isolate is due to the combination of blaACT-1 β-lactamase production, porin OmpK35/36 insertional inactivation, and down-regulation of the phosphate transport porin PhoE. Antimicrobial agents and chemotherapy. 2006; 50(10):3396–3406. 10.1128/AAC.00285-06 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang XD, Cai JC, Zhou HW, Zhang R, Chen G-X. Reduced susceptibility to carbapenems in Klebsiella pneumoniae clinical isolates associated with plasmid-mediated β-lactamase production and OmpK36 porin deficiency. Journal of medical microbiology. 2009; 58(9):1196–1202. 10.1099/jmm.0.008094-0 [DOI] [PubMed] [Google Scholar]
- 19.Shi W, Li K, Ji Y, Jiang Q, Wang Y, Shi M, et al. Carbapenem and cefoxitin resistance of Klebsiella pneumoniae strains associated with porin OmpK36 loss and DHA-1 β-lactamase production. Brazilian Journal of Microbiology. 2013; 44(2): 435–442. 10.1590/S1517-83822013000200015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hutsul J.-A., Worobec E. Molecular characterization of the Serratia marcescens OmpF porin, and analysis of S. Marcescens OmpF and OmpC osmoregulation. Microbiology. 1997; 143(8): 2797–2806. [DOI] [PubMed] [Google Scholar]
- 21.Oteo J, Delgado-Iribarren A, Vega D, Bautista V, Rodríguez M C, Velasco M, et al. Emergence of imipenem resistance in clinical Escherichia coli during therapy. International journal of antimicrobial agents. 2008; 32(6):534–537. 10.1016/j.ijantimicag.2008.06.012 [DOI] [PubMed] [Google Scholar]
- 22.Crichton PB. Enterobacteriaceae: Escherichia, Klebsiella, proteus, and other genera. In: Mackie and McCartney practical medical microbiology. 14th ed. Churchill Livingstone: New York. 1996; 14:361–4. [Google Scholar]
- 23.Vandepitte J, Verhaegen J, Engbaek K, Rohner P, Piot P, Heuck C, et al. Basic laboratory procedures in clinical bacteriology. 2ed. Geneva: World Health Organization. 2003. [Google Scholar]
- 24.Bayer A, Kirby W, Sherris J, Turck M. Antibiotic susceptibility testing by a standardized single disc method. Am J Clin Pathol. 1966; 45(4):493–496. [PubMed] [Google Scholar]
- 25.Clinical and Laboratory Standards Institute (CLSI). Performance standards for antimicrobial susceptibility testing: twenty-fifth informational supplement, M100-S25. Wayne, PA, USA: CLSI; 2015; Vol. 33. [Google Scholar]
- 26.Paterson DL, Bonomo RA. Extended-spectrum β-lactamases: a clinical update. Clinical microbiology reviews. 2005; 18(4):657–686. 10.1128/CMR.18.4.657-686.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Manchanda V, Singh NP. Occurrence and detection of AmpC β-lactamases among Gram-negative clinical isolates using a modified three-dimensional test at Guru Tegh Bahadur Hospital, Delhi, India. Journal of Antimicrobial Chemotherapy. 2003; 51(2):415–418. [DOI] [PubMed] [Google Scholar]
- 28.Livermore DM, Maskell JP, Williams JD. Detection of PSE-2 beta-lactamase in enterobacteria. Antimicrobial agents and chemotherapy. 1984; 25(2):268–272. 10.1128/aac.25.2.268 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Barwa R, Abdelmegeed E, Abd El Galil K. Occurrence and detection of AmpC-lactamases among some clinical isolates of Enterobacteriaceae obtained from Mansoura University Hospitals, Egypt. African Journal of Microbiology Research. 2012; 6(41):6924–6930. [Google Scholar]
- 30.Kamel NA, El-Tayeb WN, El-Ansary MR, Mansour MT, Aboshanab KM. Phenotypic screening and molecular characterization of carbapenemase-producing Gram-negative bacilli recovered from febrile neutropenic pediatric cancer patients in Egypt. PLoS One. 2018; 13(8), p.e0202119. 10.1371/journal.pone.0202119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hernández-Allés S, Albertí S, Álvarez D, Doménech-Sánchez A, Martínez-Martínez L, Gil J, et al. Porin expression in clinical isolates of Klebsiella pneumoniae. Microbiology. 1999;145(3):673–679. [DOI] [PubMed] [Google Scholar]
- 32.Barwa R, Shaaban M. Molecular characterization of Klebsiella pneumoniae clinical isolates with elevated resistance to carbapenems. The Open Microbiology Journal.2017; 11:152. 10.2174/1874285801711010152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Abd El-Aziz AM, Elgaml A, Ali YM. Bacteriophage therapy increases complement-mediated lysis of bacteria and enhances bacterial clearance after acute lung infection with multidrug-resistant Pseudomonas aeruginosa. The Journal of Infectious Diseases. 2019; 219(9):1439–1447. 10.1093/infdis/jiy678 [DOI] [PubMed] [Google Scholar]
- 34.Doumith M, Ellington MJ, Livermore DM, Woodford N. Molecular mechanisms disrupting porin expression in ertapenem-resistant Klebsiella and Enterobacter spp. clinical isolates from the UK. Journal of Antimicrobial Chemotherapy. 2009; 63(4):659–667. 10.1093/jac/dkp029 [DOI] [PubMed] [Google Scholar]
- 35.Gharrah MM, El-Mahdy AM, Barwa R. Association between virulence factors and extended-spectrum beta-lactamase-producing Klebsiella pneumoniae compared to nonproducing isolates. Interdisciplinary Perspectives on Infectious Diseases. 2017; 10.1155/2017/7279830 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Panteleev PV, Bolosov IA, Balandin SV, Ovchinnikova TV. Design of antimicrobial peptide arenicin analogs with improved therapeutic indices. Journal of Peptide Science. 2015; 21(2): 105–113. 10.1002/psc.2732 [DOI] [PubMed] [Google Scholar]
- 37.Hassan R, Barwa R, Shehata HR. Antimicrobial resistance genes and some virulence factors in Escherichia coli and Streptococcus pyogenes isolated from Mansoura University Hospitals. The Egyptian Journal of Medical Microbiology. 2010; 19(1):27–40. [Google Scholar]
- 38.Camara M, Mane MT, Ba-Diallo A, Dieng A, Diop-Ndiaye H, Karam F, et al. Extended-spectrum beta-lactamase-and carbapenemase-producing Enterobacteriaceae clinical isolates in a Senegalese teaching hospital: A cross-sectional study. African Journal of Microbiology Research. 2017;11(44):1600–1605. [Google Scholar]
- 39.Zaki M, El-Halaby H, Elmansoury E, Zeid M, Khaled K, Nomir M. Genetic study of extended-spectrum Beta-Lactamase and carbapenemase-producing Escherichia coli causing sepsis among Egyptian children. The Open Microbiology Journal. 2019; 13(1): 128–137. [Google Scholar]
- 40.Senad K, Musaddique M. In vitro efficacy testing of EDTA in combination with antibiotics against prominent pathogens. Biosci Biotech Res Comm. 2011; 4:41–46. [Google Scholar]
- 41.El-Sokkary MMA, Abdelmegeed ES. Antibacterial resistance pattern among Escherichia coli strains isolated from Mansoura hospitals in Egypt with a special reference to quinolones. African Journal of Microbiology Research. 2015; 9(9):662–670. [Google Scholar]
- 42.Gawad WE, Helmy OM, Tawakkol WM, Hashem AM. Effect of EDTA on biofilm formation and antibiotic susceptibility of multidrug-resistant uropathogenic Escherichia coli clinical isolates in Egypt. African Journal of Microbiology Research. 2017; 11 (38):1445–1458. [Google Scholar]
- 43.Khalifa HO, Soliman AM, Ahmed AM, Shimamoto T, Hara T, Ikeda M, et al. High carbapenem resistance in clinical gram-negative pathogens isolated in Egypt. Microbial Drug Resistance. 2017;23(7):838–844. 10.1089/mdr.2015.0339 [DOI] [PubMed] [Google Scholar]
- 44.Elsawy E, Elkady I, El-Masry S, Ahmed Y. Extended-spectrum β- lactamase producing Escherichia coli and Klebsiella pneumonia in urinary tract infections. Egypt. J. Exp. Biol. (Bot.). 2011; 7(1):135–141. [Google Scholar]
- 45.Piscitelli SC, Rodvold KA, Pai MP. Drug interactions in infectious diseases. Humana Press. 2011; http://doi: 10.1007/978-1-61779-213-7. [Google Scholar]
- 46.Livermore DM, Woodford N. The β-lactamase threat in Enterobacteriaceae, Pseudomonas, and Acinetobacter. Trends in microbiology. 2006; 14(9): 413–420. 10.1016/j.tim.2006.07.008 [DOI] [PubMed] [Google Scholar]
- 47.Poole K. Resistance to β-lactam antibiotics. Cellular and Molecular Life Sciences CMLS. 2004; 61(17): 2200–2223. 10.1007/s00018-004-4060-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bradford PA. Extended-spectrum β-lactamases in the 21st century: characterization, epidemiology, and detection of this important resistance threat. Clinical microbiology reviews. 2001; 14(4): 933–951. 10.1128/CMR.14.4.933-951.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Paterson DL, Hujer KM, Hujer AM, Yeiser B, Bonomo MD, Rice LB, et al. Extended-spectrum β-lactamases in Klebsiella pneumoniae bloodstream isolates from seven countries: dominance and widespread prevalence of SHV-and CTX-M-type β-lactamases. Antimicrobial agents and chemotherapy. 2003; 47(11):3554–3560. 10.1128/aac.47.11.3554-3560.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Coque T, Baquero F, Canton R. Increasing prevalence of ESBL-producing Enterobacteriaceae in Europe. Eurosurveillance. 2008; 13(47):19044. [PubMed] [Google Scholar]
- 51.Philippon A, Arlet G, Jacoby GA. Plasmid-determined AmpC-type β-lactamases. Antimicrobial agents and chemotherapy. 2002; 46(1): 1–11. 10.1128/aac.46.1.1-11.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Jacoby GA. AmpC β-lactamases. Clinical microbiology reviews. 2009; 22(1):161–182. 10.1128/CMR.00036-08 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bouchillon S, Johnson B, Hoban D, Johnson J, Dowzicky M, Wu D, et al. Determining incidence of extended-spectrum β-lactamase producing Enterobacteriaceae, vancomycin-resistant Enterococcus faecium and methicillin-resistant Staphylococcus aureus in 38 centers from 17 countries: the PEARLS study 2001–2002. International journal of antimicrobial agents. 2004; 24(2):119–124. 10.1016/j.ijantimicag.2004.01.010 [DOI] [PubMed] [Google Scholar]
- 54.Saied T, Elkholy A, Hafez S F, Basim H, Wasfy MO, El-Shoubary W, et al. Antimicrobial resistance in pathogens causing nosocomial bloodstream infections in university hospitals in Egypt. American journal of infection control. 2011; 39(9):e61–e65. 10.1016/j.ajic.2011.04.009 [DOI] [PubMed] [Google Scholar]
- 55.Abdallah H, Wintermans B, Reuland E, Koek A, Al Naiemi N, Ammar A, et al. Extended-spectrum β-lactamase-and carbapenemase-producing Enterobacteriaceae isolated from Egyptian patients with suspected bloodstream infection. PLoS One. 2015; 10(5): p. e0128120. 10.1371/journal.pone.0128120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Abdelmegeed ES, Barwa R, Abd El Galil K H. Comparative study on prevalence and association of some virulence factors with extended-spectrum beta-lactamases and AmpC producing Escherichia coli. African Journal of Microbiology Research. 2015;9(17): 165–1174. [Google Scholar]
- 57.Molton JS, Tambyah PA, Ang BS, Ling ML, Fisher DA. The global spread of healthcare-associated multidrug-resistant bacteria: a perspective from Asia. Clinical infectious diseases. 2013; 56(9):1310–1318. 10.1093/cid/cit020 [DOI] [PubMed] [Google Scholar]
- 58.Cantón R, González-Alba JM,Galán J.C. CTX-M enzymes: origin and diffusion. Frontiers in microbiology. 2012;3:110. 10.3389/fmicb.2012.00110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhao W.-H, Hu Z.-Q. Epidemiology and genetics of CTX-M extended-spectrum β-lactamases in Gram-negative bacteria. Critical reviews in microbiology. 2013; 39(1):79–101. 10.3109/1040841X.2012.691460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ahmed OB, Omar AO, Asghar AH, Elhassan MM, Al-Munawwarah A.-M. Prevalence of TEM, SHV, and CTX-M genes in Escherichia coli and Klebsiella spp Urinary Isolates from Sudan with confirmed ESBL phenotype. Life Sci J. 2013; 10(2):191–195. [Google Scholar]
- 61.El-Kholy AA, Girgis SA, Shetta MA, Abdel-Hamid DH, Elmanakhly AR. Molecular characterization of multidrug-resistant Gram-negative pathogens in three tertiary hospitals in Cairo, Egypt. European Journal of Clinical Microbiology & Infectious Diseases. 2020; 39: 987–992. 10.1007/s10096-020-03812-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Yazdi M, Nazemi A, Mirinargasi M, Jafarpour M, Sharifi S, Branch T. Genotypic versus Phenotypic methods to detect extended-spectrum beta-lactamases (ESBLs) in uropathogenic Escherichia coli. Ann Biol Res. 2012; 3(5):2454–8. [Google Scholar]
- 63.Bajpai T, Pandey M, Varma M, Bhatambare G. Prevalence of TEM, SHV, and CTX-M Beta-Lactamase genes in the urinary isolates of a tertiary care hospital. Avicenna journal of medicine. 2017; 7(1):12. 10.4103/2231-0770.197508 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Shahid M, Singh A, Sobia F, Rashid M, Malik A, Shukla I, et al. blaCTX-M, blaTEM, and blaSHV in Enterobacteriaceae from North-Indian tertiary hospital: high occurrence of combination genes. Asian Pacific Journal of Tropical Medicine. 2011; 4(2):101–105. 10.1016/S1995-7645(11)60046-1 [DOI] [PubMed] [Google Scholar]
- 65.Decré D, Verdet C, Raskine L, Blanchard H, Burghoffer B, Philippon A, et al. Characterization of CMY-type β-lactamases in clinical strains of Proteus mirabilis and Klebsiella pneumoniae isolated in four hospitals in the Paris area. Journal of Antimicrobial Chemotherapy. 2002; 50(5):681–688. 10.1093/jac/dkf193 [DOI] [PubMed] [Google Scholar]
- 66.Fam N, Gamal D, El Said M, Aboul-Fadl L, El Dabei E, El Attar S, et al. Detection of plasmid-mediated AmpC beta-lactamases in clinically significant bacterial isolates in a research institute hospital in Egypt. Life Science Journal. 2013; 10(2):2294–2304. [Google Scholar]
- 67.Park YS, Yoo S, Seo M-R, Kim JY, Cho YK, Pai H. Risk factors and clinical features of infections caused by plasmid-mediated AmpC β-lactamase-producing Enterobacteriaceae. International journal of antimicrobial agents. 2009; 34(1): p. 38–43. 10.1016/j.ijantimicag.2009.01.009 [DOI] [PubMed] [Google Scholar]
- 68.Aslan AT, Akova M. Extended spectrum β-lactamase producing enterobacteriaceae: Carbapenem sparing options. Expert review of anti-infective therapy, 2019. December 2; 17(12):969–981. 10.1080/14787210.2019.1693258 [DOI] [PubMed] [Google Scholar]
- 69.Tzouvelekis L, Markogiannakis A, Psichogiou M, Tassios P, Daikos G. Carbapenemases in Klebsiella pneumoniae and other Enterobacteriaceae: an evolving crisis of global dimensions. Clinical microbiology reviews. 2012; 25(4):682–707. 10.1128/CMR.05035-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Abdelaziz MO, Bonura C, Aleo A, Fasciana T, Mammina C. NDM-1-and OXA-163-producing Klebsiella pneumoniae isolates in Cairo, Egypt, 2012. Journal of Global Antimicrobial Resistance. 2013;1(4):213–215. 10.1016/j.jgar.2013.06.003 [DOI] [PubMed] [Google Scholar]
- 71.Talaat M, El-Shokry M, El-Kholy J, Ismail G, Kotb S, Hafez S, et al. National surveillance of health-care-associated infections in Egypt: developing a sustainable program in a resource-limited country. American journal of infection control. 2016; 44(1 1):1296–1301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Mahmoud AB, Eissa NA, El-Raghy NA, El-Sebaey HM, Melake NA, Awad ET, et al. Phenotypic and molecular characterization of multidrug-resistant Enterobacteriaceae clinical isolates from intensive care units at Menoufia University hospitals. Menoufia Medical Journal. 2016; 29(4):835. [Google Scholar]
- 73.Talaat M, Saied T, Kandeel A, El-Ata GAA, El-Kholy A, Hafez S, et al. A point prevalence survey of antibiotic use in 18 hospitals in Egypt. Antibiotics. 2014; 3(3):450–460. 10.3390/antibiotics3030450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Amjad A, Mirza IA, Abbasi S, Farwa U, Malik N, Zia F. Modified Hodge test: A simple and effective test for detection of carbapenemase production. Iranian journal of microbiology. 2011; 3(4):189. [PMC free article] [PubMed] [Google Scholar]
- 75.Kumarasamy K.K., Toleman MA, Walsh TR, Bagaria J, Butt F, Balakrishnan R, et al. Emergence of a new antibiotic resistance mechanism in India, Pakistan, and the UK: a molecular, biological, and epidemiological study. The Lancet infectious diseases. 2010; 10(9):597–602. 10.1016/S1473-3099(10)70143-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Poirel L, Benouda A, Hays C, Nordmann P. Emergence of NDM-1-producing Klebsiella pneumoniae in Morocco. Journal of Antimicrobial Chemotherapy. 2011;66(12):2781–2783. 10.1093/jac/dkr384 [DOI] [PubMed] [Google Scholar]
- 77.Sonnevend A, Al Baloushi A, Ghazawi A, Hashmey R, Girgis S, Hamadeh MB, et al. Emergence and spread of NDM-1 producer Enterobacteriaceae with contribution of IncX3 plasmids in the United Arab Emirates. Journal of medical microbiology. 2013; 62(7): 1044–1050. 10.1099/jmm.0.059014-0 [DOI] [PubMed] [Google Scholar]
- 78.Kamel NA, El-Tayeb WN, El-Ansary MR, Mansour MT, Aboshanab KM. Phenotypic screening and molecular characterization of carbapenemase-producing Gram-negative bacilli recovered from febrile neutropenic pediatric cancer patients in Egypt. PloS one. 2018; 13(8):e0202119. 10.1371/journal.pone.0202119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Liao K, Chen Y, Huang H, Guo P, Chen D, Liu M, et al. Molecular characteristics of carbapenem-resistant Enterobacteriaceae isolates from a Chinese Tertiary hospital in Guangdong. Journal of Microbiology & Infectious Diseases. 2014; 4(4): 145–151. [Google Scholar]
- 80.Khalil MA, Elgaml A, El-Mowafy M. Emergence of multidrug-resistant New Delhi Metallo-β-lactamase-1-producing Klebsiella pneumoniae in Egypt. Microbial Drug Resistance. 2017; 23(4): 480–487. 10.1089/mdr.2016.0003 [DOI] [PubMed] [Google Scholar]
- 81.Zaman TU, Aldrees M, Al Johani SM, Alrodayyan M, Aldughashem FA, Balkhy HH. Multi-drug carbapenem-resistant Klebsiella pneumoniae infection carrying the OXA-48 gene and showing variations in outer membrane protein 36 causing an outbreak in a tertiary care hospital in Riyadh, Saudi Arabia. International Journal of Infectious Diseases. 2014; 28:186–192. 10.1016/j.ijid.2014.05.021 [DOI] [PubMed] [Google Scholar]
- 82.Memish ZA, Assiri A, Almasri M, Roshdy H, Hathout H, Kaase M, et al. Molecular characterization of carbapenemase production among gram-negative bacteria in Saudi Arabia. Microbial Drug Resistance. 2015; 21(3):307–314. 10.1089/mdr.2014.0121 [DOI] [PubMed] [Google Scholar]
- 83.Nordmann P, Poirel L, Carrër A, Toleman MA, Walsh TR. How to detect NDM-1 producers. Journal of clinical microbiology. 2011; 49(2):718–721. 10.1128/JCM.01773-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Elwakil BH, Ali SM, Hafez SF, Bekhit AA, El-Naggar MY, Olama Z A. Resistance prevalence profile of Klebsiella pneumoniae in the Intensive Care Units of Al-Shatby Pediatric Hospital, Alexandria, Egypt. Novel Research in Microbiology Journal. 2019; 3(6): 535–545. [Google Scholar]
- 85.Nordmann P, Naas T, Poirel L. Global spread of carbapenemase-producing Enterobacteriaceae. Emerging infectious diseases. 2011; 17(10): 1791. 10.3201/eid1710.110655 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Martin RM, Bachman MA. Colonization, infection, and the accessory genome of Klebsiella pneumoniae. Frontiers in cellular and infection microbiology. 2018; 8:4. 10.3389/fcimb.2018.00004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ghai I, Bajaj H, Bafna JA, Hussein HA, Winterhalter M, Wagner R. Ampicillin permeation across OmpF, the major outer-membrane channel in Escherichia coli. Journal of Biological Chemistry. 2018. May 4;293(18):7030–7037. 10.1074/jbc.RA117.000705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Wozniak A, Villagra NA, Undabarrena A, Gallardo N, Keller N, Moraga M, et al. Porin alterations present in non-carbapenemase-producing Enterobacteriaceae with high and intermediate levels of carbapenem resistance in Chile. Journal of medical microbiology. 2012; 61(9):1270–1279. 10.1099/jmm.0.045799-0 [DOI] [PubMed] [Google Scholar]
- 89.Perez F, Endimiani A, Hujer KM, Bonomo RA. The continuing challenge of ESBLs. Current opinion in pharmacology. 2007; 7(5):459–469. 10.1016/j.coph.2007.08.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Phan K, Ferenci T. The fitness costs and trade-off shapes associated with the exclusion of nine antibiotics by OmpF porin channels. The ISME journal. 2017. June;11(6):1472–82. 10.1038/ismej.2016.202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Masi M, Réfregiers M, Pos KM, Pagès JM. Mechanisms of envelope permeability and antibiotic influx and efflux in Gram-negative bacteria. Nature microbiology. 2017. February 22;2(3):1–7. 10.1038/nmicrobiol.2017.1 [DOI] [PubMed] [Google Scholar]
- 92.Wu TL, Siu L, Su L-H, Lauderdale T, Lin F, Leu H-S, et al. Outer membrane protein change combined with co-existing TEM-1 and SHV-1 β-lactamases lead to false identification of ESBL-producing Klebsiella pneumoniae. Journal of Antimicrobial Chemotherapy. 2001; 47(6):755–761. 10.1093/jac/47.6.755 [DOI] [PubMed] [Google Scholar]
- 93.Arora B, Jagdale T. Altered membrane permeability in multidrug-resistant Escherichia coli isolated from extra-intestinal infections. African Journal of Biotechnology. 2009; 8(21): 5995–5999. [Google Scholar]
- 94.Doménech-Sánchez AA, Hernández-Allés S, Martínez-Martínez L, Benedí VJ, Albertí S. Identification and characterization of a new porin gene of Klebsiella pneumoniae: its role in β-lactam antibiotic resistance. Journal of bacteriology. 1999; 181(9): 2726–2732. 10.1128/JB.181.9.2726-2732.1999 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Kitchel B, Rasheed JK, Endimiani A, Hujer AM. Anderson KF, Bonomo RA, et al. Genetic factors associated with elevated carbapenem resistance in KPC-producing Klebsiella pneumoniae. Antimicrobial agents and chemotherapy. 2010; 54(10): 4201–4207. 10.1128/AAC.00008-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Martínez-Martínez L, Pascual A, del Carmen Conejo M, García I, Joyanes P, Doménech-Sánchez A, et al. Energy-dependent accumulation of norfloxacin and porin expression in clinical isolates of Klebsiella pneumoniae and relationship to extended-spectrum β-lactamase production. Antimicrobial agents and chemotherapy. 2002;46(12):3926–3932. 10.1128/aac.46.12.3926-3932.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Sugawara E, Kojima S, Nikaido H. Klebsiella pneumoniae major porins OmpK35 and OmpK36 allow more efficient diffusion of β-lactams than their Escherichia coli homologs OmpF and OmpC. Journal of bacteriology. 2016; 198(23): 3200–3208. 10.1128/JB.00590-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Jacoby GA, Han P. Detection of extended-spectrum beta-lactamases in clinical isolates of Klebsiella pneumoniae and Escherichia coli. Journal of clinical microbiology. 1996; 34(4): 908–911. 10.1128/JCM.34.4.908-911.1996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Livermore D. Beta-lactamase-mediated resistance and opportunities for its control. The Journal of antimicrobial chemotherapy. 1998; 41Suppl 4:25–41. 10.1093/jac/41.suppl_4.25 [DOI] [PubMed] [Google Scholar]
- 100.Coggon CF, Jiang A, Goh KG, et al. A novel method of serum resistance by Escherichia coli that causes urosepsis. MBio. 2018; 9:3. 10.1128/mBio.00920-18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Hughes C, Phillips R, Roberts A. Serum resistance among Escherichia coli strains causing urinary tract infection in relation to O type and the carriage of hemolysin, colicin, and antibiotic resistance determinants. Infection and Immunity. 1982; 35(1):270–275. 10.1128/IAI.35.1.270-275.1982 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Sahly H, Aucken H, Benedi V, Forestier C, Fussing V, Hansen D, et al. Increased serum resistance in Klebsiella pneumoniae strains producing extended-spectrum β-lactamases. Antimicrobial agents and chemotherapy. 2004; 48(9): 3477–3482. 10.1128/AAC.48.9.3477-3482.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ranjan A., Scholz J., Semmler T., Wieler L. H., Ewers C., Müller S., et al. , ESBL-plasmid carriage in E. coli enhances in vitro bacterial competition fitness and serum resistance in some strains of pandemic sequence types without overall fitness cost. Gut pathogens. 2018; 10(1):1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Hussain A, Shaik S, Ranjan A, Suresh A, Sarker N., Semmler T., et al., Genomic and functional characterization of poultry Escherichia coli from India revealed diverse Extended-spectrum β-lactamase-producing lineages with shared virulence profiles. Frontiers in microbiology. 2019;10: 2766. 10.3389/fmicb.2019.02766 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Albertí S, Rodríquez-Quiñones F, Schirmer T, Rummel G, Tomás JM, Rosenbusch JP, et al. A porin from Klebsiella pneumoniae: sequence homology, three-dimensional model, and complement binding. Infection and immunity. 1995; 63(3): 903–910. 10.1128/IAI.63.3.903-910.1995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Liu Y-F, Yan J-J, Lei H-Y, Teng C-H, Wang M-C, Tseng C-C, et al. Loss of outer membrane protein C in Escherichia coli contributes to both antibiotic resistance and escaping antibody-dependent bactericidal activity. Infection and Immunity. 2012; 80(5):1815–1822. 10.1128/IAI.06395-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Miajlovic H, Smith SG. Bacterial self-defense: how Escherichia coli evades serum killing. FEMS microbiology letters. 2014; 354(1):1–9. 10.1111/1574-6968.12419 [DOI] [PubMed] [Google Scholar]
- 108.Struyve´ M, Moons M, Tommassen J. Carboxy-terminal phenylalanine is essential for the correct assembly of a bacterial outer membrane protein. J Mol Biol. 1991; 218: 141–148. 10.1016/0022-2836(91)90880-f [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX)
(DOCX)
(PDF)
Data Availability Statement
All relevant data are within the manuscript and its Supporting Information files.