Abstract
Severe COVID-19 disease is associated with an increase in pro-inflammatory markers, such as IL-1, IL-6, and tumor necrosis alpha, less CD4 interferon-gamma expression, and fewer CD4 and CD8 cells, which increase the susceptibility to bacterial and fungal infections. One such opportunistic fungal infection is mucormycosis. Initially, it was debated whether a person taking immunosuppressants, such as corticosteroids, and monoclonal antibodies will be at higher risk for COVID-19 or whether the immunosuppresive state would cause a more severe COVID-19 disease. However, immunosuppressants are currently continued unless the patients are at greater risk of severe COVID-19 infection or are on high-dose corticosteroids therapy. As understood so far, COVID-19 infection may induce significant and persistent lymphopenia, which in turn increases the risk of opportunistic infections. It is also noted that 85% of the COVID-19 patients’ laboratory findings showed lymphopenia. This means that patients with severe COVID-19 have markedly lower absolute number of T lymphocytes, CD4+T and CD8+ T cells and, since the lymphocytes play a major role in maintaining the immune homeostasis, the patients with COVID-19 are highly susceptible to fungal co-infections. This report is intended to raise awareness of the importance of early detection and treatment of mucormycosis and other fungal diseases, such as candidiasis, SARS-CoV-2-associated pulmonary aspergillosis, pneumocystis pneumonia and cryptococcal disease, in COVID-19 patients, to reduce the risk of mortality.
Keywords: SARS-CoV-2, COVID-19, mucormycosis, fungal infection, candida auris, aspergillosis, pneumocystis pneumonia, cryptococcus neoformans, coinfection.
SUMMARY
1. Introduction
2. COVID-19 and immunosuppression
3. Fungal co-infections in COVID-19
3.1 Mucormycosis
3.2 Candidiasis (Candida auris)
3.3 SARS-CoV-2–assoc. pulmonary aspergillosis
3.4 Pneumocystis pneumonia
3.5 Cryptococcal disease (C. neoformans)
4. Current treatment options for fungal infections in COVID-19 patients
4.1 Mucormycosis
4.2 Candidiasis (Candida auris)
4.3 SARS-CoV-2–assoc. pulmonary aspergillosis
4.4 Pneumocystis pneumonia
4.5 Cryptococcal disease (C. neoformans)
5. Discussion
6. Conclusion
1. Introduction
Towards the end of 2019, an outbreak of a pneumonia-like infection occurred in Wuhan, China, and rapidly spread across the globe. With the utilization of genome sequencing technology, the disease was identified as coronavirus disease or more commonly known as coronavirus disease 2019 (COVID-19), and the etiological agent was identified as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)1. As of March 2021, there are 93.3 million infected cases, with 2 million confirmed deaths reported worldwide. This respiratory illness has significantly impacted all sectors of life, primarily healthcare and the economy, and as a result, on March 11, 2020, the WHO officially recognized COVID-19 disease as a pandemic. The coronaviruses are enveloped, positive-sense single-stranded viruses that utilize the human angiotensin-converting enzyme 2 (ACE2) receptors located on cells in many organs/tissues, including the lung, heart, kidney, bladder, eyes, nasal and oral cavities, brain, thyroid, liver, gallbladder, stomach, pancreas, intestine, reproductive system of males and females, and skin, to gain entry and trigger a range of clinical manifestations2. Evaluating the data available from various parts of the world has demonstrated that the SARS-CoV-2 virus follows an exponential growth, and the mean basic reproduction number, which indicates the number of new infections that can arise from a single case of COVID-19 infection, is estimated to be 1.4-41.
The COVID-19 disease is associated with a range of clinical manifestations, particularly due to SARS-CoV-2’s high human-to-human transmission rate via respiratory droplets1. However, the presence of the binding receptors in different tissues determines the range of clinical manifestations. The clinical manifestations range from a self-limiting illness with general symptoms, such as fever, cough, and headache, to more severe complications, including acute respiratory distress syndrome, sudden cardiac death, liver dysfunction, and multi-organ failure. An analysis of 13 studies on laboratory-confirmed COVID-19 cases conducted in China demonstrated the prevalence of COVID-19-associated co- infections with bacteria, viruses, and fungi3. Severe COVID-19 disease is associated with an increase in pro-inflammatory markers, such as IL-1, IL-6, IL-6, and tumor necrosis alpha, less CD4 interferon-gamma expression, and fewer CD4 and CD8 cells; this, therefore, increases the susceptibility to bacterial and fungal infections4. One such opportunistic fungal infection involved is mucormycosis. Mucormycosis is an opportunistic fungal infection that belongs to the zygomycete family and are ubiquitous in the environment. The major route of infection is via inhalations of spores, which then spread to the paranasal sinuses and lungs. Mucormycosis is non-pathologic in immunocompetent individuals as a result of the presence of an intact immunity via neutrophils which hence permit the elimination of these spores5. However, in immunocompromised patients such as those with uncontrolled diabetes mellitus, diabetic ketoacidosis, presence of an open wound, HIV/ AIDS, cancer, and organ transplant, mucormycosis can result in a severe invasive fungal infection6.
Infection with mucormycosis is associated with high mortality primarily due to complications such as cavernous sinus thrombosis, disseminated infection, osteomyelitis, and death. Diagnosis is made via routine blood work, biopsy, and radiological imaging. The standard protocol for the management is primarily reversal of risk factors, surgical debridement as well as intravenous antifungal medication such as Amphotericin B6. Infection with SARS-CoV-2 drastically impacts the immune system via induction of an inflammatory storm, an increase in neutrophil count as well a decrease in lymphocyte count, specifically CD4+ and CD8+ T cells. Neutrophils ensure the immunocompetence of individuals, so one would expect an even more increased immune response against mucormycosis fungi when reading that neutrophil numbers are rising during SARS-CoV-2. Consequently, these patients are at increased susceptibility of developing opportunistic infections such as mucormycosis, due to decreasing lymphocyte cells5. CD4+ and CD8+ T cells serve a prominent role against infection with mucormycosis via recruiting cytokines, such as IL-4, IL-10, IL-17, and IFN-γ5.
2. COVID-19 and immunosuppression
Initially it was debated whether a person taking immunosuppresants be at higher risk of getting COVID-19 or whether the immunosuppresive state would cause more severe COVID-19 infection7. However, currently the immunosuppressants are continued unless the patients are at greater risk of severe COVID-19 infection or are on high-dose corticosteroids therapy8. As understood so far, COVID-19 infection may induce significant and persistent lymphopenia which in turn increases the risk of opportunistic infections9. It is also noted that 85% of the COVID-19 patients’ laboratory findings showed lymphopenia10. This means patients with severe COVID-19 have markedley lower absolute number of T lymphocytes, CD4+T and CD8+ T cells11. And since the lymphocytes play a major role in maintaining the immune homeostasis10, the patients with COVID-19 are highly susceptible to fungal co-infections8. Moreover, the aggressive disease course of the SARS-CoV-2 virus damages the lung tissues and alveolo-interstitial lesions which puts the COVID-19 infected person prone to getting invasive fungal infections, specifically those that are airborne or with primarily pulmonary entry. These include infections like pneumocystis and mucormycosis11. Mucormycosis is a rare fungal infection which is often life-threatening8. It is characterized by vascular invasion by the fungal hyphae which leads to thrombosis and necrosis8. There has been a surge of mucormycosis co-infection in COVID-19 patients, many cases being reported worldwide.
3. Fungal co-infections in COVID-19
3.1 Mucormycosis
Mucormycosis, previously known as zygomycosis, is a severe fungal infection caused by a group of molds called mucoromycetes. It can affect the sinuses, brain or lungs and therefore can be quite common in people suffering or recovering from COVID-19. The common symptoms linked to mucormycosis are swelling in one side of the face, fever, headache, nasal or sinus congestion, black lesions on nasal bridge or upper inside of mouth12. Fungi that most commonly cause mucormycosis are: Rhizopus species, Mucor species, Rhizomucor species, Syncephalastrum species, Cunninghamella Bertholletia, Apophysomyces species, and Lichtheimia species. Mucormycosis can present in many forms such as, gastrointestinal mucormycosis (more common in young children), rhinocerebral mucormycosis (more common in people with uncontrolled diabetes and in people who had a kidney transplant), disseminated mucormycosis, pulmonary mucormycosis (more common in people with cancer and in people who have had an organ transplant or a stem cell transplant) and cutaneous mucormycosis13.
Symptoms of some fungal diseases can be similar to those of COVID-19, including fever, cough, and shortness of breath.Laboratory testing is necessary to determine if a person has a fungal infection or COVID-19. Some patients can have COVID-19 and a fungal infection at the same time. People with severe COVID-19, such as those in an intensive care unit (ICU), are particularly vulnerable to bacterial and fungal infections. Mucormycosis and orbital compartment syndrome in addition to COVID-19, has been diagnosed in a previously healthy young female14. Other authors have reported cases of acute invasive fungal rhino-orbital mucormycosis15 and cavitary pulmonary mucormycosis5,16 in patients with COVID-19. Patients hospitalized for COVID-19 are at risk for healthcare-associated infections17, including candidemia, or bloodstream infections caused by Candida and other fungal diseases.
3.2 Candidiasis (Candida auris)
Candida auris (C. auris) is an emerging fungus that can cause outbreaks of severe infections in healthcare facilities and invasive candidiasis are some of the common presentations in COVID 19 patients. Clinicians should consider fungal pneumonias as a possible cause of respiratory illness, particularly if COVID-19 testing is negative. A total of 1,518 confirmed cases and 30 probable C. Auris cases, were reported by U.S. States as of October 202018. A fungal infection commonly seen during COVID-19 is C. Auris. The main concern regarding this fungal infection is related to C. auris being a multi-drug resistant, being extremely hard to identify with standard laboratory methods and, C. auris having caused various outbreaks in healthcare settings. Some studies have suggested an increased risk for Candida species in COVID-19 patients resulting in poor outcomes19,20. The C. auris fungal infection is known to spread quickly in long-term care facilities. However, during the pandemic there have been an increasing number of reports of C. auris in COVID-19 acute care units. Researchers suspect that these outbreaks may be related to changes in routine infection control practices due to the health crisis. For example, there is a limited availability of gloves and gowns and there is a possibility of faulty cleaning and disinfection practices21,22. According to this the US Center for Disease Control (CDC) report, among 67 patients admitted to the COVID-19 unit and screened during point prevalence surveys, 35 (52%) received positive test results. Mean age of colonized patients was 69 years (range = 38–101 years) and 60% were male. Six (17%) colonized patients later had clinical cultures that grew C. auris. Among patients screened who had available medical records (17), two (10%) were admitted directly from a long-term care facility and eight (40%) died within 30 days of screening, but whether C. auris contributed to death is unknown23. A recent study showed that mortality among patients with COVID-19 and C. auris candidemia was of 83.3% even when the right antifungal treatment was used. All investigated isolates shown resitance to the amphotericin B24.
3.3 SARS-CoV-2–associated pulmonary asper-gillosis (CAPA)
Aspergillus genera, most frequently Aspergillus fumigatus, can be found almost everywhere in the environment and can be responsible for many infections in humans, including for the invasive pulmonary aspergillosis (IPA), chronic pulmonary aspergillosis (CAPA), allergic bronchopulmonary aspergillosis (ABPA), fungal asthma, aspergillus bronchitis or chronic rhinosinusitis. Invasive pulmonary aspergillosis, which is the most severe form of disease from Aspergillus, is known to be associated with high mortality rates. It is also a prominent complication seen in severely immunosuppressed individuals, including the indiviudals with hematopoietic transplantation, as well as those with structural lung damage treated with systemic corticosteroids for their underlying condition (e.g.chronic obstructive pulmonary diseases (COPD) patients). Once they occur, these super infections are associated with high mortality rates and may prolong the acute phase of COVID-1925. Among fungal co-infections in France, the incidence of putative invasive pulmonary aspergillosis was high (30%)24,26. To date, >100 cases of CAPA have been reported from many countries in Europe, Asia, Australia, and South America27-29, often occurring in patients with no other risk factor than COVID-19-associated ARDS27, and multiple of them proven by autopsy28-30. According to Hoenigl’s study, fungal diseases, and particularly CAPA, add insult to injury in a significant proportion of critically ill COVID-19 patients and are associated with high mortality rates, which may be reduced by early diagnosis and initiation of appropriate antifungal therapy. In the absence of antifungal prophylaxis, screening of COVID-19 ARDS patients for CAPA and other fungal diseases is essential31. CAPA should be suspected in severe COVID-19 patients who have worsening respiratory function or sepsis18.
When looking at incidences of CAPA in critically ill patients (e.g., patients on ICUs), rates vary substantially from 4% to as high as 35%. The challenges in diagnosing fungal infections, and particularly CAPA, are thought to explain this variety in incidence rates. Clinical symptoms and abnormalities observed when the chest is imaged are non-specific. Positive sputum or tracheal aspirates do not differentiate between colonization and infection, and to perform invasive diagnostic procedures (e.g. bronchoscopy and bronchoalveolar lavage or lung biopsy) is often not feasible due to the clinical state of the patient. Moreover, these procedures are risky due to the possible airborne transmission of SARS-CoV-232.
Some studies show that as many as one third of the COVID-19 patients with severe disease that require intensive care may also be fighting another life-threatening infection: for example, invasive aspergillosis, a deadly fungal superinfection caused by Aspergillus mold33. During the retrospective observational study of COVID-19 patients, bacterial and fungal co-infections occurred in <5% of all cases. However, this is of significant concern due to their occurrence in the most vulnerable patients34.
3.4 Pneumocystis pneumonia
Pneumocystispneumonia also shares similar symptoms of COVID-19 such as fever, cough, difficulty breathing, chest pain, chills, fatigue, and coinfection with Pneumocystis jirovecii may not be appreciated in patients with severe SARS-CoV-2 infection. There have been reported cases of these two co-infections35. In another study, which included almost exclusively immunocompromised patients has reported an unexpectedly high proportion of critically ill COVID-19 patients detected with P. jirovecii (10/108 patients; 9.3%). COVID-19 patients mostly exhibited marked lymphopenia and alterations in lymphocyte functions, likely explaining the high-rate of P. jirovecii detection36. This association has been reported even in young severely ill patients37. Differentiating COVID-19 from pneumocystis jirovecii pneumonia is not usually possible from signs and symptoms. Sputum culture, RT-PCR, CT chest are recommended to diagnose on time and do a differential between COVID-19 and P. jirovecii38.
3.5 Cryptococcal disease (C. neoformans)
Disseminated Cryptococcus neoformans (C. neoformans) infection is a serious infection that can occur in immunocompromised patients. There have been reported cases of C. neoformans and COVID -19 patients. The case reported by Mohamad Y and colleges, highlights the importance of early suspicion of C. neoformans infection and other opportunistic infections in immunocompromised patients, putting in mind that patients with Cryptococci have a high risk of mortality within 30 days, which warrants the use of corticosteroid and immunomodulatory drugs in a critically ill patient with COVID‐19. In the current scenario, the use of immunosuppressive therapy should be justified and to be alert for opportunistic infection like C. neoformans infection, which can lead to sepsis and mortality39.
4. Current treatment options for fungal infections in COVID-19
4.1 Mucormycosis
Global guidelines have demonstrated detailed and different approaches for the diagnosis and management of mucormycosis in 2019 by the European Confederation of Medical Mycology (ECMM) and Mycoses Study Group Education and Research Consortium40. It states an immediate and complete surgical intervention should be taken in the first place. In addition to it, systemic antifungals should be added to first-line management. There is a strong recommendation of high dose liposomal amphotericin B along with the adequate dosage of intravenous isavuconazole and posaconazole. Both triazoles can be given as salvage treatment as well. In high-risk patients such as neutropenic patients who have graft versus host disease, prophylaxis with posaconazole can be given. There is not enough data on the usage of combinations of other antifungal drugs. Limited choice of therapy is creating significant pressure on patients with low incomes. There is an excellent margin for uncertainty, and so much research work can be on it40.
4.2 Candidiasis (Candia Auris)
Patients suspected or confirmed with C. auris should be treated with echinocandins (caspofungin, micafunging, and anidulafungin), azoles (fluconazole, voriconazaole, itraconazole), and Amophotericin B and its liposomes. Therapeutic drug monitoring for optimizing efficacy and limiting toxicity of azoles should be considered40,41. As this is often a multidrug-resistant candida infection, some strains may be resistant to all antifungals. A fungus highly likely to cause hospital setting outbreaks, requires urgent identification and treatment initiation with contact precautions in place40,42.
4.3 SARS-CoV-2–associated pulmonary aspergillosis (CAPA)
Drugs recommended for the treatment of CAPA are divided in two categories. For allergic aspergillosis and prevention, CDC recommends triazoles (itraconazole, voriconazole, posiconzaole, esaconzaole) with corticosteroids. However, for invasive aspergillosis, like the one seen in COVID-19, CDC recommends voriconazole, lipid amphotericin B formulations, posaconazole, isavuconazole, itraconazole, and echinococcus (micafungin or caspofungin). Triazoles can be a safe choice for patients, but therapeutic drug monitoring (TDM) is highly recommended, and the interaction between azoles drugs should be observed diligently to observe for side effects. If the patient develops aspergilloma, surgery may be required40,43.
4.4 Pneumocystis pneumonia
The standard treatment for the pneumocystis pneumonia is a combination of trimethoprim and sulfamethoxazole. Corticosteroids can also be used for the moderate to severe pneumonia with low oxygen levels35-38.
4.5 Cryptococcal disease (C. neoformans)
For asymptomatic patients and patients with mild-to-moderate pulmonary infections, CDC recommends fluconazole. For people with severe lung infections or infections in the central nervous system (brain and spinal cord), CDC recommended initial treatment with Amphotericin B in combination with flucytosine. Patient is then switched to fluconazole for an extended period of time until infection is clear. Dose and duration of the antifungal treatment may differ for pregnant women, children, and people with limited resources. Some patients may require surgery for removal of fungal growth, known as cryptococcomas40,44.
5. Discussion
Mucormycosis is associated with invasion of blood vessels, which results in ischemic necrosis. Mucormycosis has the potential to invade various systems in the body resulting in a myriad of clinical symptoms that progress rapidly. Based on the anatomic site involved, mucormycosis can be classified into the following forms: rhino-cerebral, pulmonary, gastrointestinal, cutaneous, and disseminated8. As a result of the harmful complications associated with opportunistic co-infections such as invasive fungal infections in COVID-19 disease, it is essential that health care workers, mainly physicians, are vigilant and aware of the possibility of infection and take the necessary precautions45.
Monte Junior et al. (2020) reported a case of an 86-year-old male patient with history of arterial hypertension, admitted to the emergency room with symptoms of acute diarrhea, cough, dyspnea, and fever that started 5 days ago with a confirmed COVID-19 diagnosis. Within 5 days of admission, patient’s hemoglobin level of 14.3 mg/dL dropped to level of 5.6 mg/dL, which required infusion of three units of red blood cells. Esophago-gastroduodenoscopy to rule out cause of internal bleeding revealed two giant gastric ulcers with necrotic debris, confirmed to be mucormycosis upon biopsy. This patient did not have any other typical comorbidities to suggest suspicion of mucormycosis, such as diabetes. A rare disease of gastrointestinal mucormycosis was contributed to immune dysregulation caused by COVID-19. Despite intensive care, this patient died within 36 hours of the procedure. When the anticipated gastrointestinal (GI) mucormycosis symptoms such as fever, nausea, abdominal pain, GI bleeding and perforation are missing, establishing a correct diagnosis early on is challenging. Since the GI mucormycosis is known to have up to 85% fatality rate, early diagnosis and treatment becomes imperative in patient’s survival8.
Mehta et al. (2020) presented a case of a 60-year-old male patient with a past medical history of >10 years of diabetes with a three-day history of severe breathlessness, pyrexia, tachypnea and generalized malaise. While admitted for COVID-19 management, on day 10, patient developed bilateral lid edema with right eye prominence. MRI of the brain showed soft tissue swelling of orbits and paranasal sinuses with mucosal thickening. Patient was initially diagnosed with orbital cellulitis and ophthalmic consultation was obtained the next day. Upon examination, the ophthalmologist suspected a diagnosis of invasive fungal infection like mucormycosis, which was confirmed with a nasal swab on a sabourauds dextrose agar culture. Since diabetes is one of the most common comorbidities of mucormycosis, it is possible that the patient either had a previous undiagnosed mucor infection or it may have been aggravated with further dysregulation of immunity secondary to COVID-19. Use of steroids and monoclonal antibodies to treat COVID-19 may lead to exacerbation of opportunistic infections like mucormycosis as well45.
Pasero et al. (2020) discussed a case of a 66-year-old male patient with a past medical history of arterial hypertension, admitted to ICU in Italy with COVID-19. The patient was intubated due to COVID-19 complications. Over the course of 2 week, the patient had multiple organ dysfunction with sequential organ failure assessment (SOFA) score of 14, and his respiratory parameters continued worsening. Patient was started on empiric antibiotics therapy. After 2 weeks of empirical therapy a bronchial aspirate was repeated to confirm SARS-CoV-2 and to detect other coinfections. The bronchial aspirate confirmed coinfection of Rhizopus spp. and treated appropriately, yet the patient did not survive and died at day 62 of refractory shock and liver failure. The researchers here also suggested altered and impaired immune system were the cause of higher risk of opportunistic infection and negative outcome5.
Gago and Ibrahim (2021) represented a case of 53-year-old male patient with past medical history of obesity and depression was diagnosed with secondary acute myeloid leukemia (AML) in January 2020 and transferred to the hospital for further treatment. After five weeks of treatment the patient developed sore throat, paraguesia, dysosmia and fever and was diagnosed with SARS-CoV-2 confirmed with nasal RT-PCR at day 54 after induction of chemotherapy. On day 8 after SARS-CoV-2 diagnosis, patient required intubation, and was extubated on day 18 as he stabilized. However, shortly after he developed fever up to 39.5 °C with negative blood culture, acridine-orange leukocyte cytospin test, viral PCRs, and fungal biomarkers. Due to rapid respiratory deterioration patient required reintubation on day 22 with a positive SARS-CoV-2 nasopharyngeal swab and negative bronchoalveolar lavage. Due to worsening hemodynamic situation, the patient died on day 24. A full autopsy was performed and revealed invasive Rhizopus microspores from lung tissue. Researchers suggested chemotherapy combined with corticosteroids resulted in a prolonged neutropenic phase leading to opportunistic infection like mucormycosis. Due to negative testing of fungal infections the patient was not treated with any other azoles except voriconazole, an antifungal used a prophylaxis in such patients. This report was also intended to raise awareness for early detection and treatment of mucormycosis46.
Hanley et al. (2020) reported a post-mortem case series of nine patients with confirmed COVID-19 at premortem between March 1 and April 30, 2020. The median age at death of this cohort was 73 years (IQR 52-79). At least one major organ, predominantly the lung showed thrombotic features at full autopsies and diffuse alveolar damage was the most consistent lung finding in all ten patients. The series supported several novel autopsy findings, including secondary disseminated mucormycosis, requiring additional investigation to understand the role of this opportunistic infection in COVID-19 patients47.
Sen et al. (2021) conducted a retrospective, interventional study on 6 COVID-19 patients who developed rhino-orbital mucormycosis and were managed at a tertiary ophthalmic center in India between August 1 and December 15, 2020. All patients were men with mean age of 60.5 ± 12 with type 2 diabetes. All except one patient received systemic corticosteroids treatment for COVID-19. The researchers reported mean duration of 15.6 ± 9.6 days between diagnosis of COVID-19 and development of symptoms of mucor. All patients had undergone endoscopic sinus debridement and only two required orbital exenteration, however all six patients were alive at the last follow up. The researchers put much emphasis on high index of suspicion, early diagnosis, and appropriate management for patient survival48.
Kanwar et al. (2021) also reported a fatal case of 56-year-old man who was hospitalized for COVID-19. This patient had pre-existing end-stage renal disease on hemodialysis and ultimately developed mucormycosis during his hospitalization. Patient has a positive SARS-CoV-2 RT-PCR while asymptomatic, until four days later, he was hospitalized for fatigue and shortness of breath, at which time he received methylprednisolone, tocilizumab and single dose of convalescent plasma. Upon admission, blood cultures were negative for bacterial and fungal microorganisms. Patient was discharged home 7 days later, readmitted five days after discharge with generalized fatigue, shortness of breath and hemoptysis. Patient was started on empiric antibiotics therapy (IV vancomycin and piperacillin-tazobactam) for suspected healthcare-associated pneumonia. His chest x-ray showed increasing airspace density in both lungs with pleural effusion. On Day 3, a repeat sputum revealed filamentous fungus and empiric treatment of liposomal amphotericin B was started. Despite persistent drainage of pleural effusion with pigtail catheter over the next few days, repeat chest CT was unchanged. On repeated sample analysis, Rhizopus azygosporous was also diagnosed, and patient was put on appropriate treatment. Despite all efforts patient developed cardiac arrest and died on hospitalization day 17. Researchers recommended severe COVID-19 should be considered as a risk factor for invasive fungal infections, particularly those who receive immunosuppressive medications like steroids and IL-6 inhibitors (tocilizumab). Because there are no non-invasive tests available for invasive fungal infections, researchers believe cases of mucormycosis cases may be higher in numbers than what is reported49.
Moorthy et al. (2021) completed multi-centric retrospective study in Bangalore, India in 18 patients with diabetes mellitus (DM) with positive SARS-CoV-2 infections. 15 of 18 patients had confirmed uncontrolled DM and all received corticosteroid for COVID-19 treatment. Surprisingly, 12 of 18 patients had complained of vision loss, 7 of whom then underwent orbital exenteration. The results showed 16 cases of mucormycosis, 1 of aspergillosis and 1 case of mixed fungal infection. Six of these patients died, 11 survived and 1 was lost to follow up. Researchers confirmed significantly higher incidence of fungal infections (p = 0.03) amongst diabetic patients and suspect strong association with immunosuppression related to corticosteroid administration50.
Karimi-Galougahi et al. (2021) reported a case of 61-year-old woman with no past medical history and was hospitalized for COVID-19 infection for 2 weeks. During her admission, she received remdesivir, interferon-alpha, and systemic corticosteroid. Patient did not require intubation and mechanical ventilation. Patient developed right hemifacial pain with no other sinonasal symptoms, hemifacial numbness, decreased visual acuity, and chemosis one week after her discharge, which prompted her second hospitalization. The non-contrast CT of paranasal sinuses, MRI, and diagnostic sinonasal endoscopy suggested and confirmed invasive fungal infection of mucormycosis. In this case, although the patient was healthy, researchers indicated that the corticosteroid treatment induced diabetes and immunosuppression in addition to immune dysregulation caused by COVID-19 caused invasive mucormycosis, encouraging prompt diagnosis by physicians51.
Several other Rhino-orbital mucormycosis cases have been reported. Waizel-Haiat et al. (2021) reported a case of a 24-year-old female in Mexico City, with past medical history of obesity, who tested positive for COVID-19. She was brought to the emergency room, where she complained of left midface pain at least six days prior to admission, and within two days showed signs of left lid swelling and maxillary hypoesthesia. When oral amoxicillin-clavulanate did not provide relief, rhinoscopy was completed with contrast-enhanced CT of head and chest, revealing invasive fungal infection. This patient developed multiple other complications of COVID-19, including metabolic acidosis combined with pulmonary insult and acute kidney injury due to disseminated intravascular coagulopathy, leading to death due to multi-organic failure due to septic shock. The researchers concluded this patient had immunosuppressive state secondary to diabetic ketoacidosis, making her susceptible to COVID-19 and mucormycosis coinfections. Her late diagnosis and delay in treatment further contributed to this unfortunate outcome52.
Currently, there is only one ongoing clinical trial [NCT04368221] at Rennes University Hospital in France to assess prevalence of opportunistic fungal co-infections in COVID-19 infected and mechanically ventilated patients in ICU. Researchers feel ICU patients with ARDS are not systematically screened for the respiratory fungal infections. The study will allow to determine median time between entry in ICU and beginning of ARDS and colonization of fungal infections such as Aspergillus, Pneumocystis jirovecii and mucoromycetes. The study will also evaluate time between diagnosis and targeted treatment at ICU discharge, up to 1 month and further help develop preventive strategies53.
6. Conclusion
This report is intended to raise awareness for early detection and treatment of various fungal infections, particularly high mortality mucormycosis, in COVID-19 patients to reduce the risk of mortality. Due to association with a very high mortality rate, the researchers put much emphasis on high index of suspicion, early diagnosis, and appropriate management for patient survival. It is essential to assess the risk factors, the types of invasive mycosis to provide appropriate individualized treatment. Additional investigation is needed to understand the role of opportunistic infections in COVID-19 patients. Finally, we provide a flow diagram (Figure 1) to assist the clinicians and laboratory experts in the management of aspergillosis, candidiasis, mucormycosis, or cryptococcosis, as comorbidities in COVID-19 patients.
Figure 1. Diagnostic and therapeutic pathway for invasive fungal co-infections in COVID-19 patients.
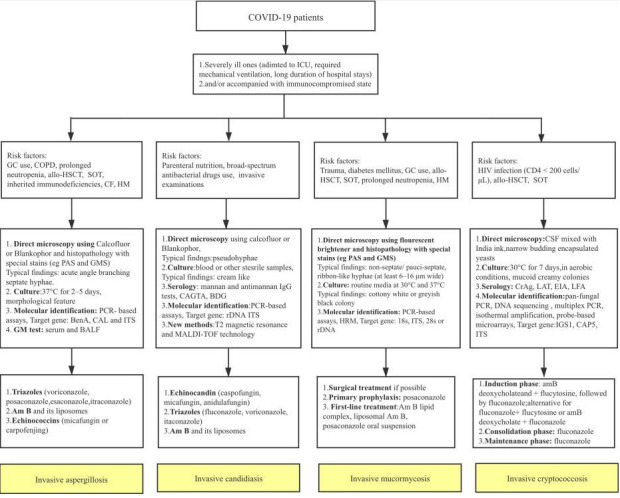
Reproduced with permission from reference40.
Acknowledgments
The author thank to their host institutions for support.
Footnotes
Conflict of interests: The authors declare no conflicts of interest.
Angiotensin-converting enzyme 2 (ACE 2); Allergic bronchopulmonary aspergillosis (ABPA); Acquired immune deficiency syndrome (AIDS); Acute myeloid leukemia (AML); Acute respiratory distress syndrome (ARDS); Candida Auris (C. Auris); COVID-19 associated pulmonary aspergillosis (CAPA) Cluster of differentiation (CD); Centers for Disease Control and Prevention (CDC); Chronic obstructive pulmonary diseases (COPD); Coronavirus disease of 2019 (COVID-19); Chronic pulmonary aspergillosis (CPA); Cryptococcus Neoformans (C. Neoformans); Deciliter (dL); Human immunodeficiency virus (HIV); ICU (Intensive care unit); Interferon gamma (IFN-γ); Interleukin (IL); Interquartile range (IQR); Milligram (mg); Pneumocystis jirovecii (P. jirovecii); Reverse transcription polymerase chain reaction (RT-PCR); Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2); Sequential organ failure assessment (SOFA); World Health Organization (WHO).
DISCOVERIES is a peer-reviewed, open access, online, multidisciplinary and integrative journal, publishing high impact and innovative manuscripts from all areas related to MEDICINE, BIOLOGY and CHEMISTRY
References
- 1.COVID-19: A Global Challenge with Old History, Epidemiology and Progress So Far. Khan Mujeeb, Adil Syed F., Alkhathlan Hamad Z., Tahir Muhammad N., Saif Sadia, Khan Merajuddin, Khan Shams T. Molecules. 2020;26(1):39. doi: 10.3390/molecules26010039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Virology, Epidemiology, Pathogenesis, and Control of COVID-19. Jin Yuefei, Yang Haiyan, Ji Wangquan, Wu Weidong, Chen Shuaiyin, Zhang Weiguo, Duan Guangcai. Viruses. 2020;12(4) doi: 10.3390/v12040372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Co-infections among patients with COVID-19: The need for combination therapy with non-anti-SARS-CoV-2 agents? Lai Chih-Cheng, Wang Cheng-Yi, Hsueh Po-Ren. Journal of microbiology, immunology, and infection = Wei mian yu gan ran za zhi. 2020;53(4):505–512. doi: 10.1016/j.jmii.2020.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fungal co-infection in COVID-19 patients: Should we be concerned? Pemán Javier, Ruiz-Gaitán Alba, García-Vidal Carolina, Salavert Miguel, Ramírez Paula, Puchades Francesc, García-Hita Marta, Alastruey-Izquierdo Ana, Quindós Guillermo. Revista iberoamericana de micologia. 2020;37(2):41–46. doi: 10.1016/j.riam.2020.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.A challenging complication following SARS-CoV-2 infection: a case of pulmonary mucormycosis. Pasero Daniela, Sanna Silvana, Liperi Corrado, Piredda Davide, Branca Gian Pietro, Casadio Lorenzo, Simeo Raffaella, Buselli Alice, Rizzo Davide, Bussu Francesco, Rubino Salvatore, Terragni Pierpaolo. Infection. 2020 doi: 10.1007/s15010-020-01561-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hernández Jorge L., Buckley. Clifford J. Mucormycosis. StatPearls [Internet]; 2020. [Google Scholar]
- 7.COVID-19 and immunosuppression: a review of current clinical experiences and implications for ophthalmology patients taking immunosuppressive drugs. Thng Zheng Xian, De Smet Marc D, Lee Cecilia S, Gupta Vishali, Smith Justine R, McCluskey Peter J, Thorne Jennifer E, Kempen John H, Zierhut Manfred, Nguyen Quan Dong, Pavesio Carlos, Agrawal Rupesh. British Journal of Ophthalmology. 2020;105(3):306-310. doi: 10.1136/bjophthalmol-2020-316586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rare and Fatal Gastrointestinal Mucormycosis (Zygomycosis) in a COVID-19 Patient: A Case Report. Monte Junior Epifanio Silvino do, Santos Marcos Eduardo Lera dos, Ribeiro Igor Braga, Luz Gustavo de Oliveira, Baba Elisa Ryoka, Hirsch Bruno Salomão, Funari Mateus Pereira, de Moura Eduardo Guimarães Hourneaux. Clinical Endoscopy. 2020;53(6):746-749. doi: 10.5946/ce.2020.180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.SARS-CoV-2 and COVID-19: A Threat to Global Health. Saha Otun, Rakhi Nadira Naznin, Sultana Afroza, Rahman Md. Mahbubur, Rahaman Md. Mizanur. Discoveries Reports. 2020;3:e13. [Google Scholar]
- 10.Opportunistic Fungal Infections in the Epidemic Area of COVID-19: A Clinical and Diagnostic Perspective from Iran. Salehi Mohammadreza, Ahmadikia Kazem, Badali Hamid, Khodavaisy Sadegh. Mycopathologia. 2020;185(4):607–611. doi: 10.1007/s11046-020-00472-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Invasive fungal diseases during COVID-19: We should be prepared. Gangneux J-P, Bougnoux M-E, Dannaoui E, Cornet M, Zahar J R. Journal de mycologie medicale. 2020;30(2):100971. doi: 10.1016/j.mycmed.2020.100971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Coronavirus: severe fungal infection cases linked to COVID-19? ETimes. 2020. https://timesofindia.indiatimes.com/life-style/health-fitness/health-news/coronavirus-severe-fungal-infection-cases-linked-to-covid-19-heres-what-we-know/photostory/79836584.cms?picid=79836589 https://timesofindia.indiatimes.com/life-style/health-fitness/health-news/coronavirus-severe-fungal-infection-cases-linked-to-covid-19-heres-what-we-know/photostory/79836584.cms?picid=79836589
- 13.Goyal Shikha. Mucormycosis, a rare Fungal Infection linked to COVID-19: Causes, Symptoms, Types, Prevention and Treatment. jagranjosh.com. 2020. https://www.jagranjosh.com/general-knowledge/mucormycosis-a-rare-fungal-infection-1608646233-1 https://www.jagranjosh.com/general-knowledge/mucormycosis-a-rare-fungal-infection-1608646233-1
- 14.Mucormycosis with orbital compartment syndrome in a patient with COVID-19. Werthman-Ehrenreich Amanda. The American journal of emergency medicine. 2021;42:264.e5–264.e8. doi: 10.1016/j.ajem.2020.09.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Acute Invasive Rhino-Orbital Mucormycosis in a Patient With COVID-19-Associated Acute Respiratory Distress Syndrome. Mekonnen Zesemayat K, Ashraf Davin C, Jankowski Tyler, Grob Seanna R, Vagefi M Reza, Kersten Robert C, Simko Jeffry P, Winn Bryan J. Ophthalmic plastic and reconstructive surgery. 2021;37(2):e40–e80. doi: 10.1097/IOP.0000000000001889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.A case report of COVID-19 associated pulmonary mucormycosis. Khan Nariman, Gutierrez Christina G., Martinez David Villafuerte, Proud Kevin C. Archive of Clinical Cases. 2020;07(03):46-51. doi: 10.22551/2020.28.0703.10172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Drug Repurposing for Prevention and Treatment of COVID-19: A Clinical Landscape. Hossain Md. Shahadat, Hami Ithmam, Sawrav Md. Sad Salabi, Rabbi Md. Fazley, Saha Otun, Bahadur Newaz Mohammed, Rahaman Md. Mizanur. Discoveries. 2020;8(4):e121. doi: 10.15190/d.2020.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Invasive Candidiasis. Centers for Disease Control and Prevention. 2020. https://www.cdc.gov/fungal/diseases/candidiasis/invasive/index.html https://www.cdc.gov/fungal/diseases/candidiasis/invasive/index.html
- 19.COVID-19 associated invasive candidiasis. Al-Hatmi Abdullah M.S., Mohsin Jalila, Al-Huraizi Aisha, Khamis Faryal. Journal of Infection. 2021;82(2):e45-e46. doi: 10.1016/j.jinf.2020.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Candida auris —the growing menace to global health. Lone Shabir A., Ahmad Aijaz. Mycoses. 2019;62(8):620-637. doi: 10.1111/myc.12904. [DOI] [PubMed] [Google Scholar]
- 21.Joe JH, Koh P. The Rise of Fungal Infections in COVID-19 Patients. BNF. 2021. https://www.askbnf.com/post/the-rise-of-fungal-infections-in-covid-19-patients https://www.askbnf.com/post/the-rise-of-fungal-infections-in-covid-19-patients
- 22.Candida auris Outbreak in a COVID-19 Specialty Care Unit — Florida, July–August 2020. Prestel Christopher, Anderson Erica, Forsberg Kaitlin, Lyman Meghan, de Perio Marie A., Kuhar David, Edwards Kendra, Rivera Maria, Shugart Alicia, Walters Maroya, Dotson Nychie Q. MMWR. Morbidity and Mortality Weekly Report. 2021;70(2):56-57. doi: 10.15585/mmwr.mm7002e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Outbreak of Candida auris infection in a COVID-19 hospital in Mexico. Villanueva-Lozano Hiram, Treviño-Rangel Rogelio de J., González Gloria M., Ramírez-Elizondo María Teresa, Lara-Medrano Reynaldo, Aleman-Bocanegra Mary Cruz, Guajardo-Lara Claudia E., Gaona-Chávez Natalia, Castilleja-Leal Fernando, Torre-Amione Guillermo, Martínez-Reséndez Michel F. Clinical Microbiology and Infection. 2021;27(5):813-816. doi: 10.1016/j.cmi.2020.12.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.COVID-19 Associated Pulmonary Aspergillosis (CAPA)-From Immunology to Treatment. Arastehfar Amir, Carvalho Agostinho, van de Veerdonk Frank L, Jenks Jeffrey D, Koehler Philipp, Krause Robert, Cornely Oliver A, S Perlin David, Lass-Flörl Cornelia, Hoenigl Martin. Journal of fungi (Basel, Switzerland) 2020;6(2) doi: 10.3390/jof6020091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Prevalence of putative invasive pulmonary aspergillosis in critically ill patients with COVID-19. Alanio Alexandre, Dellière Sarah, Fodil Sofiane, Bretagne Stéphane, Mégarbane Bruno. The Lancet. Respiratory medicine. 2020;8(6):e48–e49. doi: 10.1016/S2213-2600(20)30237-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Surveillance for COVID-19-associated pulmonary aspergillosis. Brown Li-An K, Ellis Jayne, Gorton Rebecca, De Surjo, Stone Neil. The Lancet. Microbe. 2020;1(4):e152. doi: 10.1016/S2666-5247(20)30091-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Invasive aspergillosis in patients with severe COVID-19 pneumonia. Helleberg Marie, Steensen Morten, Arendrup Maiken Cavling. Clinical microbiology and infection : the official publication of the European Society of Clinical Microbiology and Infectious Diseases. 2021;27(1):147–148. doi: 10.1016/j.cmi.2020.07.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Confirmed Invasive Pulmonary Aspergillosis and COVID-19: the value of postmortem findings to support antemortem management. Santana Monique Freire, Pivoto Guilherme, Alexandre Márcia A Araujo, Baía-da-Silva Djane Clarys, Borba Mayla Gabriela da Silva, Val Fernando Almeida, Brito-Sousa Jose Diego, Melo Gisely Cardoso, Monteiro Wuelton Marcelo, Souza João Vicente Braga, Pinheiro Silviane Bezerra, Ferreira Luiz Carlos Lima, Naveca Felipe Gomes, Nascimento Valdinete Alves, Corado André Lima Guerra, Hajjar Ludhmila Abrahão, Silva Neto João Ricardo, Siva George Alan Villarouco, Pasqualotto Alessandro C, Lacerda Marcus Vinícius Guimarães. Revista da Sociedade Brasileira de Medicina Tropical. 2020;53:e20200401. doi: 10.1590/0037-8682-0401-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.COVID-19-associated invasive pulmonary aspergillosis. Rutsaert Lynn, Steinfort Nicky, Van Hunsel Tine, Bomans Peter, Naesens Reinout, Mertes Helena, Dits Hilde, Van Regenmortel Niels. Annals of intensive care. 2020;10(1):71. doi: 10.1186/s13613-020-00686-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Invasive pulmonary aspergillosis complicating SARS-CoV-2 pneumonia: A diagnostic challenge. Antinori Spinello, Rech Roberto, Galimberti Laura, Castelli Antonio, Angeli Elena, Fossali Tommaso, Bernasconi Davide, Covizzi Alice, Bonazzetti Cecilia, Torre Alessandro, Carsana Luca, Tonello Cristina, Zerbi Pietro, Nebuloni Manuela. Travel medicine and infectious disease. 2020;38:101752. doi: 10.1016/j.tmaid.2020.101752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Invasive Fungal Disease complicating COVID-19: when it rains it pours. Hoenigl Martin. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2020 doi: 10.1093/cid/ciaa1342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.COVID-19 and Aspergillus Co-infections. MRC Centre for Medical Mycology, University of Exeter; Accessed on: March 29, 2021. 2020. https://www.exeter.ac.uk/medicalmycology/news/articles/covid-19andaspergillusco-.html https://www.exeter.ac.uk/medicalmycology/news/articles/covid-19andaspergillusco-.html
- 33.COVID-19 and Fungal Superinfections: The Deadly, Perfect Storm. Xconomy. 2020. https://xconomy.com/san-diego/2020/05/05/covid-19-and-fungal-superinfections-the-deadly-perfect-storm/ https://xconomy.com/san-diego/2020/05/05/covid-19-and-fungal-superinfections-the-deadly-perfect-storm/
- 34.Bacterial and fungal coinfections in COVID-19 patients hospitalized during the New York City pandemic surge. Nori Priya, Cowman Kelsie, Chen Victor, Bartash Rachel, Szymczak Wendy, Madaline Theresa, Punjabi Katiyar Chitra, Jain Ruchika, Aldrich Margaret, Weston Gregory, Gialanella Philip, Corpuz Marilou, Gendlina Inessa, Guo Yi. Infection control and hospital epidemiology. 2021;42(1):84–88. doi: 10.1017/ice.2020.368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.A Case of COVID-19 and Pneumocystis jirovecii Coinfection. Menon Aravind A, Berg David D, Brea Elliot J, Deutsch Aaron J, Kidia Khameer K, Thurber Emilia G, Polsky Sylvie B, Yeh Tiffany, Duskin Jonathan A, Holliday Alison M, Gay Elizabeth B, Fredenburgh Laura E. American journal of respiratory and critical care medicine. 2020;202(1):136–138. doi: 10.1164/rccm.202003-0766LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.The presence of Pneumocystis jirovecii in critically ill patients with COVID-19. Alanio Alexandre, Dellière Sarah, Voicu Sebastian, Bretagne Stéphane, Mégarbane Bruno. Journal of Infection. 2021;82(4):84-123. doi: 10.1016/j.jinf.2020.10.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Concurrent COVID-19 and Pneumocystis jirovecii pneumonia in a severely immunocompromised 25-year-old patient. Bhat Pavan, Noval Mandee, Doub James B, Heil Emily. International journal of infectious diseases : IJID : official publication of the International Society for Infectious Diseases. 2020;99:119–121. doi: 10.1016/j.ijid.2020.07.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.COVID-19 and Pneumocystis jirovecii pneumonia: Back to the basics. Mouren D, Goyard C, Catherinot E, Givel C, Chabrol A, Tcherakian C, Longchampt E, Vargaftig J, Farfour E, Legal A, Couderc L-J, Salvator H. Respiratory medicine and research. 2021;79:100814. doi: 10.1016/j.resmer.2021.100814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cryptococcemia in a patient with COVID-19: A case report. Khatib Mohamad Y, Ahmed Amna A, Shaat Said B, Mohamed Ahmed S, Nashwan Abdulqadir J. Clinical case reports. 2021;9(2):853–855. doi: 10.1002/ccr3.3668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fungal Co-infections Associated with Global COVID-19 Pandemic: A Clinical and Diagnostic Perspective from China. Song Ge, Liang Guanzhao, Liu Weida. Mycopathologia. 2020;185(4):599–606. doi: 10.1007/s11046-020-00462-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Management of Patients with Candida auris Fungemia at Community Hospital, Brooklyn, New York, USA, 2016–20181. Park Jenny YeiSol, Bradley Nicole, Brooks Steven, Burney Sibte, Wassner Chanie. Emerging Infectious Diseases. 2019;25(3):601-602. doi: 10.3201/eid2503.180927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Treatment for C. neoformans Infection. Centers for Disease Control and Prevention; Accessed: 2021 March 28. 2020. https://www.cdc.gov/fungal/diseases/cryptococcosis-neoformans/treatment.html https://www.cdc.gov/fungal/diseases/cryptococcosis-neoformans/treatment.html
- 43.Candida auris. Centers for Disease Control and Prevention; Accessed: 2021 March 28. 2021. https://www.cdc.gov/fungal/candida-auris/index.html https://www.cdc.gov/fungal/candida-auris/index.html
- 44.Treatment for Aspergillosis. Centers for Disease Control and Prevention; Accessed: 2021 March 28. 2021. https://www.cdc.gov/fungal/diseases/aspergillosis/treatment.html https://www.cdc.gov/fungal/diseases/aspergillosis/treatment.html
- 45.Rhino-Orbital Mucormycosis Associated With COVID-19. Mehta Salil, Pandey Abha. Cureus. 2020;12(9):e10726. doi: 10.7759/cureus.10726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Autopsy Proven Pulmonary Mucormycosis Due to Rhizopus microsporus in a Critically Ill COVID-19 Patient with Underlying Hematological Malignancy. Zurl Christoph, Hoenigl Martin, Schulz Eduard, Hatzl Stefan, Gorkiewicz Gregor, Krause Robert, Eller Philipp, Prattes Juergen. Journal of fungi (Basel, Switzerland) 2021;7(2) doi: 10.3390/jof7020088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Histopathological findings and viral tropism in UK patients with severe fatal COVID-19: a post-mortem study. Hanley Brian, Naresh Kikkeri N, Roufosse Candice, Nicholson Andrew G, Weir Justin, Cooke Graham S, Thursz Mark, Manousou Pinelopi, Corbett Richard, Goldin Robert, Al-Sarraj Safa, Abdolrasouli Alireza, Swann Olivia C, Baillon Laury, Penn Rebecca, Barclay Wendy S, Viola Patrizia, Osborn Michael. The Lancet. Microbe. 2020;1(6):e245–e253. doi: 10.1016/S2666-5247(20)30115-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mucor in a Viral Land: A Tale of Two Pathogens. Sen Mrittika, Lahane Sumeet, Lahane Tatyarao P, Parekh Ragini, Honavar Santosh G. Indian journal of ophthalmology. 2021;69(2):244–252. doi: 10.4103/ijo.IJO_3774_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.A Fatal Case of Rhizopus azygosporus Pneumonia Following COVID-19. Kanwar Anubhav, Jordan Alex, Olewiler Scott, Wehberg Kurt, Cortes Michael, Jackson Brendan R. Journal of fungi (Basel, Switzerland) 2021;7(3) doi: 10.3390/jof7030174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.SARS-CoV-2, Uncontrolled Diabetes and Corticosteroids-An Unholy Trinity in Invasive Fungal Infections of the Maxillofacial Region? A Retrospective, Multi-centric Analysis. Moorthy Aditya, Gaikwad Rohith, Krishna Shreya, Hegde Raghuraj, Tripathi K K, Kale Preeti G, Rao P Subramanya, Haldipur Deepak, Bonanthaya Krishnamurthy. Journal of maxillofacial and oral surgery. 2021:1–8. doi: 10.1007/s12663-021-01532-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fulminant mucormycosis complicating coronavirus disease 2019 (COVID-19). Karimi-Galougahi Mahboobeh, Arastou Shima, Haseli Sara. International forum of allergy & rhinology. 2021 doi: 10.1002/alr.22785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.A Case of Fatal Rhino-Orbital Mucormycosis Associated With New Onset Diabetic Ketoacidosis and COVID-19. Waizel-Haiat Salomon, Guerrero-Paz Jose Alberto, Sanchez-Hurtado Luis, Calleja-Alarcon Salvador, Romero-Gutierrez Laura. Cureus. 2021;13(2):e13163. doi: 10.7759/cureus.13163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Characterization of Fungal Infections in COVID-19 Infected and Mechanically Ventilated Patients in ICU (MY-CO-VID) US National Library of Medicine; clnicaltrials.gov; Accessed on March 28, 2021. 2020. www.clinicaltrials.gov/ct2/show/NCT04368221 www.clinicaltrials.gov/ct2/show/NCT04368221