Abstract
Traditional cancer therapeutics have been criticized due to various adverse effects and insufficient damage to targeted tumors. The breakthrough of nanoparticles provides a novel approach for upgrading traditional treatments and diagnosis. Actually, nanoparticles can not only solve the shortcomings of traditional cancer diagnosis and treatment, but also create brand-new perspectives and cutting-edge devices for tumor diagnosis and treatment. However, most of the research about nanoparticles stays in vivo and in vitro stage, and only few clinical researches about nanoparticles have been reported. In this review, we first summarize the current applications of nanoparticles in cancer diagnosis and treatment. After that, we propose the challenges that hinder the clinical applications of NPs and provide feasible solutions in combination with the updated literature in the last two years. At the end, we will provide our opinions on the future developments of NPs in tumor diagnosis and treatment.
Keywords: Nanoparticle, Nano-cryosurgery, Targeted delivery, Photothermal therapy, Cancer
Introduction
The incidence and mortality of tumors remain high worldwide. Every year, there are nearly 14 million new cancer patients and 8 million people die of cancer-related diseases [1]. In recent years, traditional tumor treatments, such as chemotherapy, targeted therapy, radiotherapy, surgery, etc., are constantly criticized for being bogged down in progress and for many adverse reactions and unsatisfied treatment outcomes. Because of the shortcomings of traditional tumor therapies, more and more researches have begun to seek new tumor medical methods with targeting ability, effective tumor stem cell killing ability and minor adverse reactions. New tumor treatment methods include, but are not limited to, immunotherapy, targeted therapy, physical ablation, gene therapy, photodynamics therapy (PDT) and photothermal therapy (PTT) which have shown superior efficacy compared to traditional tumor therapy. The treatment methods herein all have a common feature that requires carrier cooperation. Although viruses can be used as carriers, viral vectors have been confirmed to cause insertional mutagenesis and immunogenicity [2]. Therefore, finding a safer and more effective carrier has become a top priority.
Due to nanoparticles’ small size, biosafety, drug loading, and physical properties can assist physical therapy, nanoparticles have been increasingly utilized as carriers in new tumor treatment methods. These nanoparticles-mediated therapies have virtues of multi-function, less adverse reactions and better curative effect [3]. In addition, many medical imaging technologies mediated by nanoparticles also have better clarity and accuracy, which helps accurate tumor diagnosis [4]. With the development of nanotechnology and medical technology, metals and biological materials such as gold, silver, iron, liposomes, etc. have been widely applied in the production of medical nanoparticles (NPs) [5]. At present, many researchers utilize those materials based on their physical, chemical, and/or biological properties to embed drugs, imaging agents and even genes in nanoparticles, expanding the existing field of tumor diagnosis and treatment such as drug targeted delivery, enhanced imaging, cryosurgery, PTT and PDT [6].
In addition, there is a phenomenon that most of the nanoparticles only stay in vivo and in vitro stage. However, there is a lack of literature to summarize the reasons that deter the clinical application of NPs. Therefore, this article aims to not only summarize the application status of nanoparticles in the field of tumor diagnosis and treatment, but also to find the factors that inhibit the entry of nanoparticles into clinical applications and propose feasible solutions.
Preparation and Characterization of Medical Functional Nanoparticles
Nanoparticles commonly used in medicine can be divided into three types: metal nanoparticles, non-metal nanoparticles and composite nanoparticles according to their constituent materials and functions, and their physical and chemical properties are affected by parameters such as size and shape. Therefore, in view of the functional requirements of nanoparticles in different application directions, it is very important to choose a suitable preparation process. All the preparation methods of nanoparticles can be classified into two methods: bottom up approaches and top-down approaches. The bottom-up approach is essentially through basic units (atoms, molecules and even smaller particles can be used as the basis for assembling the required nanostructures) stacked on each other to form nanoparticles, while the top-down approach is essentially a whole solid material begins to decompose into nanoparticles [7]. Table 1 lists some examples of preparing medical nanoparticles.
Table 1.
Typical NPs preparation method
| Synthesis approach | Material | Size (nm) | Method | Features | Ref |
|---|---|---|---|---|---|
| Bottom up approaches | TiO2 | 6–33 | Sol–gel synthesis | Continuous releasing of hydroxyl radicals and superoxide ions when exposed to ultraviolet rays | [8] |
| Fe3O4 | 10 | Co-precipitation | Fe3O4 can be excited by 808 nm infrared light to realize photothermal conversion | [9] | |
| PEG-Fe-PDA NP | 25–43 | Microemulsions | MRI imaging enhancement with pH activation, high photothermal efficiency and excellent biocompatibility | [10] | |
| Magnetite NPs | 39 | Hydrothermal approach | Small size magnetic nanoparticles with biocompatibility and superparamagnetism | [11] | |
| Au NPs | 8–300 | photochemical method | Enhanced medical diagnostic imaging | [12] | |
| Top-down approaches | Cu-Sn oxides NPs | 18–40.5 | electrical wire explosion | Ability to produce reactive oxygen species | [13] |
| Magnetite NPs | 12–20 | Ball milling | Small size magnetic nanoparticles with biocompatibility | [14] |
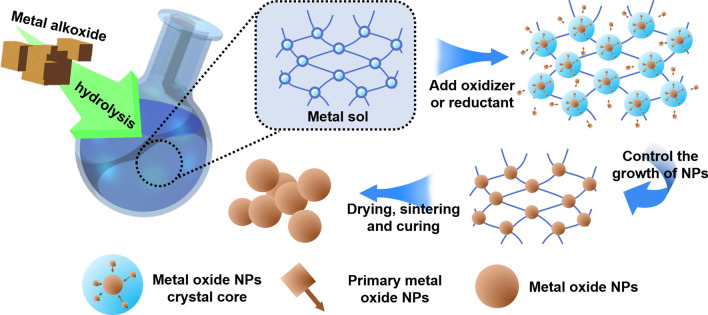
Among the three types of nanoparticles commonly used in medicine, metal nanoparticles are the most widely used. Metal nanoparticle materials include metals and metal oxides. The most commonly used preparation process for metal nanoparticles is the sol–gel (Sol–Gel) process proposed by Japanese scientist Sugimoto et al. in the 1990s, which is often used to prepare monodisperse metal oxide particles in liquid phase. The sol–gel method is a bottom-up preparation process. The main principle of this method for preparing metal nanoparticles is to form a uniformly dispersed sol of metal ions through chemical and physical means, and then form a gel through redox reaction. The metal nanoparticles generated in the gel can controllably nucleate, grow and deposit. As long as the monodispersity of the metal colloid used in the experiment, the concentration relationship of the metal ions and the oxidizing/reducing agent are controlled, the size of the synthesized metal nanoparticles can be controlled. Figure 1 is the schematic diagram of the sol–gel method.
Fig. 1.
Schematic diagram of the sol–gel method
Commonly used bottom-up methods for preparing metal nanoparticles include co-precipitation, hydrothermal approach, and photochemical method. The co-deposition method is a process of nucleation, growth and aggregation in a liquid environment at the same time. When the solution is oversaturated, a large number of small-sized particles insoluble products are obtained [15]. The hydrothermal method is a process performed in a liquid environment to control the morphology of the resulting nanoparticles by controlling the vapor pressure applied to the material in the solution. In addition, there are some top-down methods for preparing metal nanoparticles, such as electrical wire explosion and ball milling. The principle of electrical wire explosion is that in the process of electric explosion, the metal atoms are evaporated and quickly cooled in the electrolyte to form oxide nanoparticles. By controlling the electrolyte composition and current intensity, finer and uniform nanoparticles can be controlled. Ball milling is a method of quickly and large-scale production of nano-particles with controllable size using machining tools such as milling planetary gears by selecting appropriate grinding time and related equipment process parameters. In addition to metal nanoparticles, this preparation method can also be applied to other types of nanoparticles.
The second common type is non-metallic nanoparticles. Non-metallic nanoparticles commonly used in medicine include polymer nanoparticles, biomolecules derived NPs, carbon-based NPs, and silica nanoparticles [16–18]. Among them, silica nanoparticles are the most representative. The silica surface has abundant hydroxyl groups, which facilitates the binding of probes or fluorescent groups on the surface and therefore has more flexible functionality. The commonly used synthesis methods of silica nanoparticles are the sol–gel method and the Stöber method [19, 20]. The classic Stöber method is the simple and efficient preparation of silica nanoparticles through the hydrolysis and condensation of silicate under alkaline conditions.
With the development of nanotechnology, composite nanoparticles have been developed due to their superior functional compatibility. Metal nanoparticles have many characteristics that non-metal nanoparticles do not have, such as plasmon resonance effect (SPR), controllability in a magnetic field, etc., but metal particles are difficult to effectively degrade in the body, and excessive use has certain toxicity to cells [21]. Therefore, combining nanoparticles of different materials into composite nanoparticles through different preparation methods can achieve functional expansion. Wei et al. prepared gold nanorods (Au NRs), and then performed surface-initiated atom transfer radical polymerization (SI-ATRP) of N-isopropylacrylamide (NIPAAM) on Au NRs to synthesize near-infrared response Nano hybrids [22]. This composite nanoparticle that combines metal and polymer materials has both photothermal and near-infrared light corresponding drug release capabilities. The enveloping hydrogel shell makes this nanoparticle have better biocompatibility than single Au nanoparticles. Prakash synthesized composite NPs with Au as the core and SiO2 as the shell through the improved Stöber method. The inert shell of the core–shell nanoparticles is beneficial to reduce the toxicity of metal particles and improve the material stability and drug-carrying capacity of the original single metal NPs [23].
In addition to the traditional preparation methods of nanoparticles mentioned above, with the development of nanotechnology science, new requirements for ecological and environmental protection have been put forward, so new environmentally-friendly nanoparticle synthesis methods have emerged [24]. For the first time, Hajar et al. used Stevia rebaudiana as a biological reducing agent to successfully synthesize ZnS nanoparticles with a particle size ranging from 1 to 40 nm. The ZnS nanoparticles synthesized in this way have good biocompatibility [25]. According to the principles of green chemistry, Miri et al. used P. farcta (A plant belonging to Leguminosae) extract to quickly synthesize CeO2 NPs with a particle size of about 30 nm. This kind of nanoparticles has good biocompatibility [26].
Nanoparticles for Medical Imaging
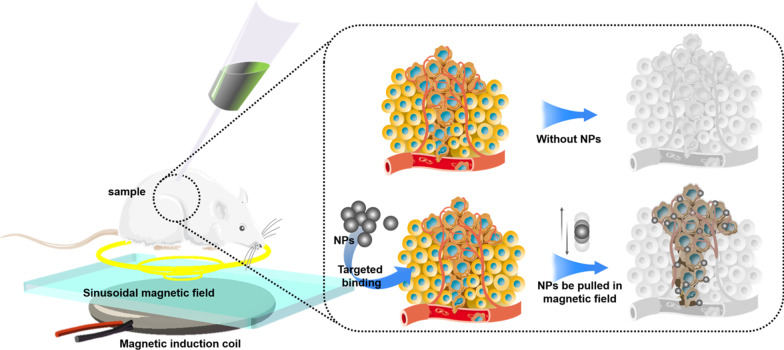
Medical imaging plays an important role in the diagnosis and treatment of tumors. Many nanoparticles, like iron oxide NPs, have optical, magnetic, acoustic, and structural properties that can enhance imaging (Fig. 2). Some studies have shown that introducing NPs into target tissues can improve image contrast and provide better image guidance for tumor surgery and diagnosis [27]. For example, in cryosurgery, NPs can enhance the imaging quality of the tumor and ice ball edges, which helps to cover the ice balls accurately and improve the therapeutic effect [28]. In addition, most of the nanoparticles used in imaging are made of metal. According to the difference of different imaging principles, nanoparticles will also be made of different metal materials. Table 2 lists some recent examples about NPs made by different materials for medical imaging.
Fig. 2.
Diagrammatic illustration of imaging improved of NPs
Table 2.
Typical NPs platforms made by different materials for medical imaging
| NPs | Size (nm) | Targeting material | Cell line | Imaging technology | Ref |
|---|---|---|---|---|---|
| MnO-TETT | 6.7 ± 1.2 | None | C6 glioma cells | Fluorescence/T1-MRI | [29] |
| PLGA-mPEG | 151.1 ± 1.3 | cRGD | SKOV-3 cells | US | [30] |
| USMO@MSNs | 30–50 | Dox | HeLa cells | MRI-guided chemotherapy | [31] |
| OINPs | 300 | Folate | SKOV3 ovarian cancer cells | US/PA | [32] |
| PEG-coated and Gd-loaded fluorescent silica | 125.5 ± 9.9 | YPSMA-1 | LNCaP and PC3 prostate cancer cells | MRI/fluorescence imaging | [33] |
| SPIO/USPIO | 50 | None | 4T1 murine breast cancer cells | MRI/MPI | [34] |
US ultrasound, MSNs mesoporous silica nanoparticles, USMO ultrasmall manganese oxide, GEM Gemcitabine, OINPs oxygen/indocyanine green-loaded lipid nanoparticles, PA photoacoustic, MPI magnetic particle imaging, MRI magnetic resonance imaging, SPIO superparamagnetic iron oxide, USPIO ultra-small SPIO
Optical coherence tomography (OCT) is a non-invasive, micron-level resolution and biomedical imaging technology. OCT is useful in real-time diagnosis and surgical guidance. However, OCT cannot detect inelastic scattered light because this light is not coherent in the incident field [35]. Recently, many researches have proved the motion state of NPs can be able to change the amplitude of OCT, which may deal with this problem. Interfering with the movement of NPs through the magnetic field can cause local changes in light scattering. Some studies have pointed out that placing magnetic NPs in a magnetic field to control its motion can change the optical scattering in the area, so the originally incoherent inelastic scattered light can be detected. This new imaging method is magnetomotive optical coherence tomography (MMOCT) [36].
MRI is one of the most effective noninvasive tumor detection technology. Nevertheless, the lack of MRI signal comparison between biological background and cancer tissue often affects the clinical tumor diagnosis [37]. MRI is a scanning imaging method that measures the magnetization of hydrogen molecules in water molecules. Each anatomical structure presents a different image since the protons of each tissue cause different changes in magnetization. The visibility of images can be improved through applying more contrast agents [38, 39]. The tumor-related EPR effect widely utilized in the early detection of tumors produces great contrast enhancement ability to magnetic NPs [40]. Iron oxide magnetic NPs (IONPs) which are currently the most common MRI nanoprobe contrast agents have certain cell targeting [41]. For example, studies have found that IONPs could enter healthy liver Kupffer cells during the diagnosis of liver cancer by using MRI but will be excluded from cancer cells, resulting in low-signal healthy tissue and high-signal tumor tissue [42]. Based on recent studies, proper particle surface modification and appropriate tumor-specific bio-oligomer embedding of NPs can better fix NPs in tumors to achieve clearer imaging results and can even be used for early micro tumor imaging. For example, studies have found that AuNPs targeted for human transferrin can significantly enhance the imaging effect of brain tumors [43]. Gao et al. equipped with anti-epidermal growth factor receptor monoclonal antibody (mAb) on the basis of paramagnetic NPs probes to achieve imaging of small tumors [44].
Nanoparticles for Targeted Drug Delivery
Although Chemotherapeutic drugs now are the most commonly used treatment for tumors, they still have the problem of poor target enrichment in malignant tumor areas and overaccumulation in healthy tissue [45]. This may cause the inhibition of cells hat divide vigorously, such as bone marrow, hair follicles, gastrointestinal cells and lymphocytes, leading to adverse reactions such as bone marrow suppression, mucositis, hair loss, and even death [46]. Targeted drug delivery which refers to active differentiation between normal cells and cancer cells for drug delivery has better efficacy and fewer adverse reactions than the conventional treatment [45].Many studies have confirmed that NPs can target chemotherapeutic drugs to tumor cells through active or passive targeting [47]. In addition, many experiments have found that NPs also play an important role in the targeted delivery of immune drugs [48].
As shown in Fig. 3, passive targeting often relies on some pathophysiological characteristics of tumor tissue, including abnormal blood vessels, temperature, pH and surface charge of tumor cells [49].For example, due to the enhanced permeability and retention effect (EPR) of blood vessels in the tumor tissue, NPs with a diameter of about 400 nm can be passively transferred to the tumor tissue [50]. However, there are many limitations on the passive targeting approach in terms of physicochemical properties of NPs such as diameter, surface charge, molecular weight, hydrophobicity, or hydrophilicity. Besides, the passive targeting technique underperforms in drug diffusion efficiency and shows insufficient EPR effect in tumor cells [51]. Due to the deficiencies of passive targeting, in recent years, most research about the drug delivery NPs has shifted to active targeting (ligand targeting). Table 3 highlights some recent examples about NPs used in drug delivery.
Fig. 3.
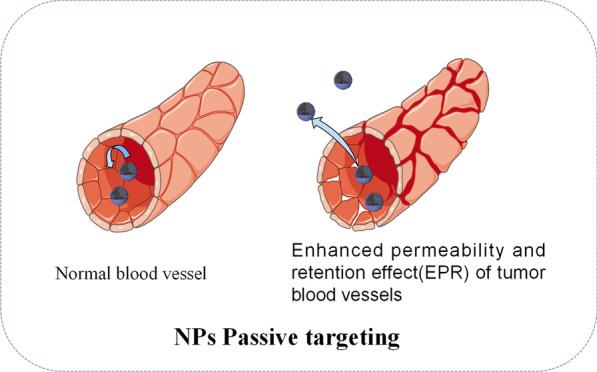
Diagrammatic illustration of passive targeting of NPs
Table 3.
Typical NPs platforms used in drug delivery
| Agent of NPs | Vehicle | Size (nm) | Characters | Effects | Ref |
|---|---|---|---|---|---|
| DNA and RNA | Exosomes | 30–100 | Small size, cellular origin, flexibility to incorporate macromolecules |
Carrier for DNA, RNA and micro-RNA Cross-stringent biological barriers, such as the blood–brain barrier |
[52] |
| DOX | Polymer-lipid encapsulated manganese dioxide | 170 | Bioreactive and multifunctional |
Downregulate TME-associated drug resistance and immunosuppression Enhancing chemotherapeutic efficacy and boosting antitumor immunity |
[53] |
| 5-FU | Au-NPs/chitosan | 100–400 | Natural cationic, biodegradable and biocompatible | Enhance the curative effect for hepatocellular carcinoma cells (HepG2) | [54] |
| Tyrosinase-related protein 2 (Trp2) peptide | Layered double hydroxide (LDH) NPs | 140–150 | Provoking strong cell-mediated immune responses | Adjuvant multiple tumor-associated antigen peptides | [55] |
| Cisplatin; ICG | PLGA | 90–100 |
Folate targeting Controlled drug release |
Promoting the apoptosis of McF-7 tumor cells NIR sensitivity |
[56] |
| IL-2 | PEGylated liposomes with anti-CD137 | 80 | Complete absence of systemic toxicity |
Inducing intratumoural immune responses Initial anti-tumor activity |
[57] |
| FA | Magnetic mesoporous silica | 213 | PH-sensitive drug release | Inhibiting proliferation of HeLa cell lines higher cytotoxicity effect | [58] |
| PTX | Peptide H7K(R2)2-modifiediron oxide NPs | 168.3 ± 2.80 | few side-effects |
Excellent MRI imaging Inhibiting tumor growth |
[59] |
| siRNA | RGDfC-SeNPs | 150 |
No toxicity Multiple tumor targeting |
Carrier for siRNA Inhibiting tumor cells proliferate Promoting the generation of ROS |
[60] |
DOX doxorubicin, 5-FU 5-fluorouracil, FA folic acid, PTX paclitaxel, ROS reactive oxygen species
Active targeting (ligand targeting) NPs often carry some ligands of tumor-specific biomarkers [61]. As shown in Fig. 4 when the ligand contacts to the receptor on the tumor surface, NPs can be internalized by the tumor cells through receptor-mediated endocytosis, and the drugs can be released due to acidic pH and specific enzymes in the intracellular environment [62]. As for targeting ligands, folic acid, transferrin, epidermal growth factor receptor (EGFR) and glycoprotein are generally utilized in current research [62]. For example, Sandoval et al. obeserved significant drug enrichment and evident efficacy in the treatment of mice with breast cancer through EGFR-targeted stearyl NPs equipped with gemcitabine [63]. Pandey et al. found that folic acid-targeted gold NPs carrying berberine hydrochloride (BHC) can effectively deliver drugs to human cervical cancer cells expressing folate receptor [64].
Fig. 4.
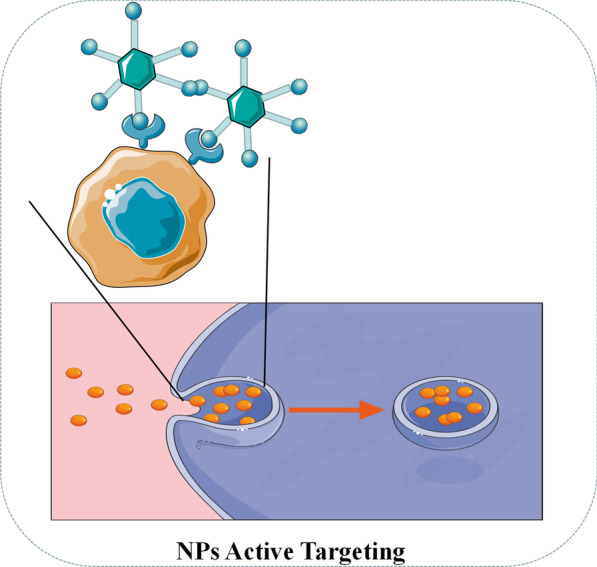
Diagrammatic illustration of active targeting of NPs
In recent years, compared with chemotherapy drugs, short interfering RNA (siRNA)-mediated gene silencing therapy has been regarded as a new prospect for tumor treatment [64]. Although viruses can be used as delivery vehicles for siRNA, viral vectors have been confirmed to cause insertional mutagenesis and immunogenicity [65]. By contrast, selenium NPs are reported to have great potential as siRNA carriers, because the trace element selenium itself can reduce tumor occurrence, lower drug toxicity, and regulate immune function [66]. In addition, the surface of selenium NPs can load various tumor- targeting moieties (such as folate, hyaluronic acid and RGD peptide) to enhance tumor targeting ability [67]. Xia et al. reported that selenium NPs (RGDfC-Se@siRNA) targeted by RGDfC peptide have excellent ability to target HeLa cervical cancer [60]. Meanwhile, because RGDfC can specifically combine with αvβ3 integrin which is highly expressed by a variety of tumor cells, RGDfC-Se@siRNA NPs can be reused for targeted drug delivery for a variety of tumors [68]. In terms of structure, RGDfC-SeNPs with positive charge can tightly package negatively charged siRNA through their electrostatic interaction [69]. Through animal experiments, RGDfC-Se@siRNA NPs show the ability to efficiently enter tumor cells through clathrin-associated endocytosis. In tumor cells, it can quickly release siRNA and efficiently silence related genes and promote the generation of reactive oxygen species (ROS) to inhibit tumor cells proliferate and promote tumor cells apoptosis [69]. Additionally, multiple SeNPs have demonstrated excellent biological safety and have no obvious toxic damage to liver, kidney, heart, lung, spleen and other major organs of mice [60, 70, 71].
At present, although there are many NPs used in targeted drug delivery, most applications still remain in the stage of cell or animal experiments, lacking potent clinical application support. In addition, many NPs are administered intratumorally, which limits the scope of NPs applicable to tumors and lacks special NPs drug delivery tools and other drug delivery methods.
Therefore, exploring a better way to administer NPs may be a direction for the future research of targeted drug delivery NPs. According to the existing academic journals, vascular interventional administration may be a feasible way. In the assumption, first locate the position of tumor-feeding blood vessel with the help of imaging, and then use a guide wire to introduce NPs directly into the tumor-feeding blood vessel and control the movement of the NPs in a small range by applying a magnetic field simultaneously. Therefore NPs can be fixed at the proper position without being influenced by blood flow in the vessel. Otherwise, NPs targeted for drug delivery only have certain limitations. Targeting NPs will affect the systemic distribution of chemotherapeutic drugs and reduce the effect of chemotherapy on free tumor cells and micrometastasis. If they are equipped with targeted drugs, the targeting effect tends to be enhanced whereas the improvement is not evident based on the existing studies.. In addition, anti-tumor drugs are unlikely to eliminate all tumor stem cells by themselves. Nevertheless, physical therapy based on the physical characteristics of NPs tends to be more effective against the tumor stem cells. Therefore, multifunctional NPs targeting drug carriers tend to be advisable in the future, such as cryosurgery, photothermal therapy (PTT) and photodynamics therapy (PDT) etc., to form multi-functional NPs for tumor treatment.
Nanoparticles for Cryosurgery
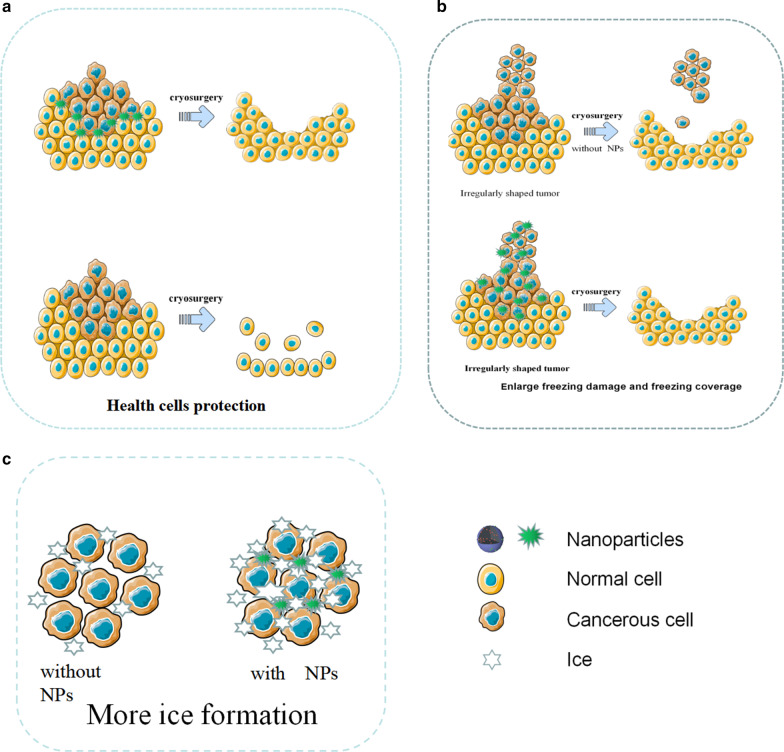
Cryosurgery, the technique of destroying tumor tissue by freezing, has the advantages of low invasiveness, low cost, less intraoperative bleeding and less postoperative complications, but there are still disadvantages such as insufficient freezing efficiency and freezing damage to surrounding tissues [28]. Although protective agents such as antifreeze protein (AFP-1) have been utilized to assist cold ablation, the effect is still not ideal [72]. With the development of nanotechnology, the concept of nano-cryosurgery was proposed. The basic mechanism of nano-cryosurgery is to introduce NPs with specific physical or chemical properties into tumor tissues. By utilizing the properties of NPs, not only can the efficiency and effectiveness of freezing be improved, but the range adjustment and the direction of ice ball formation can be also controlled. Thus, the nano-cryosurgery is capable of killing tumor tissue and preventing surrounding healthy tissue from being frozen simultaneously [73]. The advantages of nano-cryosurgery have shown in Fig. 5.
Fig. 5.
Diagrammatic illustration of NPs for cryosurgery. a NPs protect health cells during cryosurgery. b NPs enhance the freezing damage and control the freezing coverage. c With the help of NPs, more ice has been formed
In cryosurgery, intracellular ice formation is the key to tumor cell damage. Meanwhile, research proves that NPs can effectively induce intracellular ice formation [28]. NPs as external particles can induce heterogeneous nucleation. Studies have found that tissues enriched with NPs freeze faster than conventional tissues and are more prone to heterogeneous nucleation. Under the same freezing conditions, ice formation of tissue with NPs is easier, indicating that NPs can significantly increase the speed and probability of ice formation in cells, which can kill tumor cells more effectively [74]. In addition, NPs with metal oxide will significantly improve the thermal conductivity in tumor tissue. For example, Liu and Deng compared the temperature response curve of pork tissues with and without NPs. They found that the tissues containing NPs cooled rapidly, and the lowest temperature could reach 115 ℃, which was much lower than that of the control group without NPs.
Since tumors are usually irregular in shape, the ice crystals produced by traditional cryosurgery tend not to cover all tumor tissue. Compared with traditional cryosurgery, the nano-cryosurgery can deal with the problem easily. Because NPs can permeate into the intracellular fluid and have good physical properties like thermal conductivity, it is possible to control the growth direction and direction of the ice ball by the distribution of NPs [73].
In cryosurgery, insufficient freezing may not completely destroy tumor tissue, and excessive freezing may damage adjacent healthy tissue. Especially when the tumor is in close contact with fragile organs, its location is deep, or its shape is irregular, the damage to the healthy tissue can be particularly serious. In recent years, phase change materials (PCMs) made from NPs have demonstrated excellent protective potential for surrounding healthy tissues during cryosurgery [75]. For example, Lv et al. microencapsulated phase change NPs with large latent heat and low thermal conductivity through liposomes, and before cryosurgery, injected microencapsulated phase change NPs into healthy tissues around the tumor and found that avoided low temperature damage to healthy tissue [76].
Although NPs have been widely used in cryosurgery, there are still a series of deficiencies. First, it is still unable to control NPs in vitro, which results in uneven distribution of NPs in tumor tissue and unsatisfactory expected function. Secondly, although there are a variety of magnetic nanoparticles, the actual effect of in vitro magnetic field control NPs is still not ideal. In addition, the nano-cryosurgery is lack of clinical experimental research, and many NPs are still in the laboratory stage.
The application of NPs in cold ablation can be generally divided into two types: synergistic effect and protective effect, which are different in terms of the design requirements of NPs and the distribution in vivo. In the future, nano-cryosurgery may be assisted by a variety of NPs, viz, synergistic NPs are distributed inside the tumor while protective NPs are distributed around the tumor. In addition, many nano-positioning devices, such as puncture-designed 3D printed coplanar template (3DPCT) which currently used for tumor positioning before radioactive particle implantation may be used in cryosurgery. Prior to the cryosurgery, protective NPs can be punctured and injected around tumor to protect the surrounding healthy tissue by 3D printing coplanar template (3DPCT) and CT guidance. The NPs are able to assist the cryosurgery ice balls to cover the irregular edge of tumor. Then synergistic NPs will be introduced into the tumor tissue through the preset ablation site puncture or vascular intervention to perform cold ablation. This nano-cryosurgery technique can not only overcome the difficulties of cold ablation of irregular tumors but also increase the effect of cold ablation and reduce the damage to healthy tissue. This method may become the future research direction of nano-cryosurgery. Table 4 highlights some recent examples about NPs used in cryosurgery.
Table 4.
Typical NPs platforms used in cryosurgery
| NPs | Size (nm) | Thermal conductivity (W/m K) | Heat capacity (J/m3 K) | Benefits | Ref |
|---|---|---|---|---|---|
| PCM NPs | 10–20 | 0.35 | 2.56 × 106 | Health tissue protection | [76] |
| Fe3O4 NPs | 8–14 | 7.1 | 3.2 × 106 |
More intracellular ice formation High thermal conductivity |
[77] |
| Au NPs | 3 | 297.7 | 2.2 × 106 |
Good biological compatibility High thermal conductivity |
[78] |
| HCPN-CG NPs | 103.9 ± 1.5 | None | None |
Cold-responsive nanoparticle for controlled drug release NIR-induced photothermal effect |
[79] |
| MgO NPs | 50 | 34.3 | 3.2 × 106 | Nontoxic, biodegradable, and few side-effects | [74] |
HCPN-CG H for hyaluronic acid, C for chitosan, P for PF127, N for PNIPAM-B, C for CPT, and G for ICG, PCM phase change materials
Nanoparticles for PTT and PDT
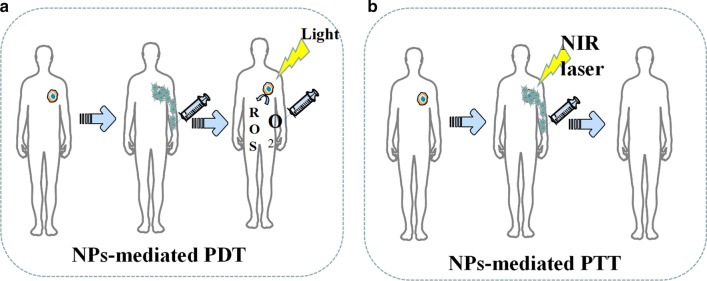
At present, photothermal therapy (PTT) and photodynamic therapy (PDT) based on nanoparticles (NPs) have shown the virtues of strong efficacy, small invasion and mild adverse effects during tumor treatment (Fig. 6) [80]. In addition to killing tumor cells directly, fragments of dead tumor cells produced by PDT and PTT treatment can be used as potential antigens to trigger a continuous immune response, called photothermal and photodynamic immunotherapy [81]. Nanoparticles designed based on the PTT treatment concept are a new type of light-to-heat conversion nanomaterials, which can convert light energy into heat energy to kill cancer cells. Compared with traditional photothermal conversion materials, nanoparticles have many advantages. First, NPs can achieve the effect of tumor targeted aggregation through particle surface modification, which contributes to higher enrichment ability of target tumor [82, 83]. Second, nanoparticles have better imaging capabilities than traditional photothermal materials, which can be accurately positioned by CT, MRI and photoacoustic imaging [84, 85]. Targeted nanoparticles synthesized by Pan et al. can perform PTT under 0.2 W/cm2 NIR to induce tumor cell apoptosis by destroying the tumor cell nuclear DNA and inhibiting the DNA repair process [86]. Table 5 lists some recent examples about NPs used in PDT and PTT.
Fig. 6.
Diagrammatic illustration of NPs-mediated PDT and PTT. a NPs promote the generation of reactive oxygen. b NPs enhance tumor damage during PTT
Table 5.
Typical NPs platforms used in PDT and PTT
| NPs Platform | Photosensitizers | λ (nm) | Size (nm) | Effects | Cell line | Ref |
|---|---|---|---|---|---|---|
| MnO2 | Chlorin e6 | 660 | 3.94 | Upregulating the secretion of IL-12,IFN-γ,TNF-αInducing decomposition of tumor endogenous H2O2 to relieve tumor hypoxia | 4T1 murine breast tumor | [87] |
| Au-liposome | None | 780 | 100 ± 6.5 | The cytotoxicity was enhanced to 90% upon laser irradiation for a duration of 5 min | B16 F10 (melanoma) | [88] |
| Silica-coated TiN | None | 785 | 80 | High nitridation temperatures and long residence times lead to increased NIR light absorption | HeLa cells | [89] |
| Silica | Verteporfin | 425 | 160–168 | Inducing singlet oxygen release 30% reduction in cell growth | SK-MEL 28 (melanoma) | [90] |
| Graphdiyne | None | 808 | 160 |
Higher cancer inhibition rate compared both in vitro and in vivo Biocompatibility and no obvious side effects |
MDA-MB-231 | [91] |
| RCDs | chlorin e6 | 671 | 3.7 |
Multimodal imaging capabilities Activating PTT and PDT at the same time |
MCF-7,4T1 and Hela | [92] |
| CuSe | non-porphyrin containing COF | 808 | 150 |
Activating PTT and PDT at the same time Enhancing therapeutic effect on killing cancer cells and inhibiting the tumor growth |
HeLa | [93] |
| HSA | ICG and chlorin e6 | 808 | 120 |
Preventing the side effects of active Ce6 Activating PTT and PDT at the same time |
PC3 | [94] |
RCDs amino-rich red emissive carbon dots, COF covalent organic framework, ICG indocyanine green, HAS serum albumin
In addition, some studies have found that nanoparticle-mediated PTT can reverse tumor multidrug resistance (MDR). The overexpression of drug transporters, multidrug resistance-associated protein 1 (MRP1), and p-glycoprotein (p-gp) are generally believed to cause MDR in various tumors [95]. For example, multifunctional light-triggered nanoparticles designed by Li et al. can inhibit the expression of MRP1 in PTT, which consequently reverses the drug resistance of A549R cells [96]. Wang et al. reported that both gold nanoparticles and carbon-based nanoparticles can overcome DOX resistance by promoting the expression of heat shock factor trimer in PTT, thereby inhibiting the generation of p-gp [97, 98]. Besides, nanoparticle-mediated PTT can also increase the effectiveness of chemotherapy by destroying the integrity of tumor cell membranes [99].
PDT is a treatment that uses the selective retention of photosensitizing substances (PSs) in tumor tissue under the activation of specific wavelength excitation light and the presence of molecular oxygen to produce singlet oxygen and other reactive oxygen species, which leads to tumor cell apoptosis and necrosis [100]. However, traditional PS has poor tumor targeting, poor solubility, and instability, which is vulnerable to the internal environment [100]. Nanoparticle carriers modified by targeted molecules can not only improve the stability and biocompatibility of PS but also deliver PS to target cells, which improves the efficacy and reduces adverse effects [100]. Additionally, some common nanomaterials, like gold nanorods, have excellent PTT effects themselves. For example, Vankayala et al. found that the exposure of gold nanorods to near infra-red light (915 nm) were able to efficiently induce the generation of singlet oxygen [100].
In recent years, the role of up-conversion (UC) nanoparticles in PDT has attracted much attention. The NPs can convert long-wavelength light excitation into multiple short wavelengths, which enables the UC to replace the traditional ps-dependent short-wavelength excitation light with the near-infrared light with strong tissue penetration ability [101]. For example, Li et al. developed dual-band luminescent lanthanide nanoparticles as a PS carrier. This UC nanoparticles rely on the excitation light wavelength of 808 nm to achieve image-guided PDT without affecting imaging signals [102].
Since most photosensitive materials utilized in the phototherapy are metals, the biocompatibility of NPs designed for inorganic nanomaterials like metal ions still needs to be improved.
NPs-mediated phototherapy is now credited for not only the effectiveness against tumor but also the potential for spare internal space of nanoparticles since the therapy only utilizes the physical properties of NPs skeleton. Therefore, NPs are often multifunctioned by PDT and PTT. In the future, such NPs may be designed as dedicated NPs for tumor stem cells that are not sensitive to chemotherapy. Tumor stem cells are dormant for a long time and have a variety of drug-resistant molecules, so it is difficult to kill them by conventional treatments like chemotherapy, whereas the light therapy is more effective by killing the tumor stem cells physically. In the future, nanophysical therapy may be used with many other techniques, such as the multifunctional NPs for photothermal therapy after cryosurgery. Multifunctional NPs mediated therapy can give full play to its characteristics of low side effects, strong local lethality, and tumor stem cell killing. In addition, because nano-physiotherapy has a local killing effect and can effectively kill tumor stem cells, it may become a treatment method for small metastases.
Nanoparticles for Radiotherapy
Radiotherapy (RT) is a tumor treatment technique that kills local cells by ionizing radiation generated by rays and is currently an effective treatment for many primary and metastatic solid tumors [103]. Experiments prove that radiotherapy can effectively kill tumor stem cells [104].However, how to further improve the efficacy of radiotherapy is still a serious challenge. In recent years, nanoparticles in the field of radiotherapy have demonstrated strong radiosensitization capabilities, tumor-targeted delivery capabilities of radiosensitizing drugs, and imaging guidance enhancement capabilities [105]. At present, the most popular nanoparticles are made by high Z (atomic number) metal materials, which are featured by chemical inertness and strong radiation absorption capacity. They produce various reactions such as photoelectric effect and Compton effect after absorbing radiation, thereby releasing a variety of particles such as optoelectronics, Compton electrons, and Auger electrons. These electrons react with organic molecules or water in tumor cells to generate a large number of free radicals, leading to synergistic chemotherapy [106]. Common chemotherapy-sensitized NPs are currentlycategorized as precious metals, iron oxides, and semiconductors in terms of materials.
Precious metals NPs are made of high atomic number metal materials such as gold, silver, gadolinium, hafnium, platinum, bismuth, etc. [107]. Among them, gold nanoparticles have become the most popular NPs due to their good biocompatibility, chemical stability, and relatively strong photoelectric absorption coefficient [108]. In 2000, Herold et al. discovered the chemosensitizing ability of gold nanoparticles in kilovoltage X-rays. Nowadays, the specific mechanism of chemosensitization of gold nanoparticles is not yet clear, and the mainstream view believes that it depends on the photoelectric absorption capacity of high atomic number [109]. In addition to this, there are studies suggesting that the presence of gold nanoparticles improve the chemical sensitization of DNA to radiation, which increases the DNA damage induced by ionizing radiation (IR). At the same time, gold NPs can catalyze the mechanism of radiotherapy sensitization such as free radical production [105]. For instance, Liu found that AuNPs could significantly increase the production of hydroxyl radicals as well as the killing effect of x-rays and fast carbon ions on cells [110]. The hypothesis of the chemotherapy sensitization mechanism of other precious metals is similar to that of gold nanoparticles. Particularly, platinum NPs have an anti-tumor effect due to the inherent nature. Consequently, platinum NPs are expected to play the role of chemotherapy and radiotherapy simultaneously. However, the number of relevant research reports is insufficient, and the sensitizing effect of platinum NPs is also questionable. For example, Charest et al. reported that liposomal formulation of cisplatin was able to increase the uptake of platinum by tumor cells, and could enhance the killing of F98 glioma cells by γ-rays at the same time [111]. On the contrary, Jawaid et al. reported that platinum NPs would reduce the generation of reactive oxygen species (ROS) and the efficacy of radiotherapy during chemotherapy [112].
Iron oxide nanoparticles (IONs), especially the superparamagnetic magnet Fe3O4, have shown great potential in image-guided tumor radiotherapy because they are capable of enhancing the dose of radiotherapy and MRI imaging, whereas its sensitization mechanism is not clear yet. Its sensitization mechanism is not yet clear. Some studies believe that iron oxide NPs mainly catalyze the generation of ROS through Fenton's reaction and Haber–Weiss reaction. Then the highly reactive ROS will kill tumors [112–115]. Other studies propose that the mechanism depends on the radiation sensitization and synergistic effects of magnetic nanoparticles. As Khoei reported, iron oxide NPs can improve the radiosensitization of prostate cancer cells in vitro [116]. Huang et al. pointed out that cross-linked dextran-coated IONs (CLIONs) could be internalized by HeLa cells and EMT-6 mouse breast cancer cells, which enhances radiation therapy [117]. Although the synergistic effect of iron oxide NPs is obvious, its biological safety still needs to be improved. Many studies have proved that the biocompatibility and chemical stability of iron oxide NPs are questionable, and it has certain toxicity [118].
Semiconductor NPs like silica NPs have also been found to have a synergistic effect on radiotherapy. For instance, Zhang et al. used flow cytometry analysis and MTT experiments to find that mesoporous silica NPs can effectively enhance the radiotherapy of glioblastoma [119]. He et al. reported the mechanism of radioactive enhancement of silica NPs. He found that under X-ray irradiation, silica nanoparticles could produce fine hydroxyl radicals, which can effectively kill tumor cells [120].
At present, although many experiments have confirmed that NPs were able to sensitize radiotherapy, the specific mechanism of sensitization is still unclear, which hinders the development of new sensitized NPs. There are some doctrines like sensitizing chemotherapy that promotes free radical production. Nevertheless, there is a lack of a quantitative relationship among the amount of free radical production, radiation intensity, and physical data of nanoparticles. In addition, most sensitized NPs are made of high atomic number metals. These metals have many disadvantages in human body such as difficulty in self-metabolism and biodegrading. Meanwhile, long-term accumulation of the metals will produce toxicity, which limits the safe use of radiosensitized NPs. Moreover, compared with the radiotherapy sensitization NPs, fewer studies focused on NPs which can prevent the adverse reactions of radiotherapy and protect healthy tissues. The research on radiotherapy protective NPs is short in quantities.
In the future, searching for NPs material that can be metabolized by the kidney, biometabolized, biocompatible, stable in physicochemical properties, and inherently less toxic, or looking for surface modification that can help the body metabolize NPs may become a research direction for sensitized NPs. Moreover, although there have been many NPs studies on multi-function, namely simultaneous sensitization of radiotherapy and chemotherapy, there are still many potentials in this field, which are worthy of focus in the future. The development of protective NPs that can protect normal tissues around radiotherapy and alleviate poor defense against radiotherapy may also become a research direction.
Conclusion
The poor curative effect, inefficient targeting ability, various side effects, and potential biological risk are some of the unfavorable attributes of conventional cancer therapy and diagnosis. In recent years, advanced nanotechnology and molecular cell biology have promoted the applications of NPs in cancer field. Not only metal NPs, but also many lipid, nucleic acid and silicon NPs showed evident outperformance in cancer diagnosis and treatment.. Moreover, new generation of NPs is no longer limited to solo but multiple functions. For example, gold-coated poly(lactic-co-glycolic acid) (PLGA) NPs equipped with PD-1 blockers which were designed by Luo et al. can not only target drug delivery but also mediate PTT therapy [121]. (Pd @ Au) / Fe3O4 Spirulina NPs with doxorubicin created by Wang et al. demonstrated the functions of photothermal therapy, delivery of chemotherapy drugs, and magnetic field control in cell experiments [122]. Multifunctional nanoparticles will become the trend of future research.
At present, we find that most of the nanoparticles only stay in vivo and in vitro stage. According to this review, we think the following reasons hinder the clinical application of NPs.
-
(i)
Lack of injection routes and methods
Most NPs are injected into body via puncture or intravenous injection. Therefore, the blood flow will take away NPs, making NPs difficult to stay in the target area for a long time, which leads to just few NPs that can be uptaked by tumor cells. Low-concentration drugs cannot produce the expected therapeutic effect, and low-concentration NPs also affect the physical killing effects of PDT, PTT, cryosurgery, and radiotherapy. In our opinion, magnetic NPs platform may be a solution. There have been many in vitro and in vivo experiments that have proved the feasibility of using the three-dimensional magnetic field to control the movement of NPs against blood flow [122–125]. However, how to solve the interference of the human body to the magnetic field, how to solve the impact of blood cells colliding with NPs, and how to control a large number of NPs in a group are still in discovery.
-
(ii)
Difficulty in localization of NPs in vivo
Compared with the human body, the size of NPs is too tiny. Even if NPs are loaded with fluorescent proteins, it is still difficult for conventional imaging equipment (CT, X-ray, MRI) to locate the NPs in the human body in real time. To deal with this challenge, photoacoustic computed tomography (PACT) may be a solution. Photoacoustic computed tomography (PACT) has attained high spatiotemporal resolution (125-μm in-plane resolution and 50-μs frame−1 data acquisition), deep penetration (48-mm tissue penetration in vivo), and anatomical and molecular contrasts [126]. Because of excellent performance, PACT has great potential in NPs localization imaging in vivo. The PACT-guided microrobotic system designed by Wu et al. has achieved controlled propulsion and prolonged cargo retention in vivo of NPs with a diameter of 50 μm [127]. Although the current resolution and deep penetration of PACT are still insufficient, it is superior to conventional imaging equipment (CT, X-ray, MRI) in terms of NPs imaging positioning.
-
(iii)
Difficulty of degrading in the human body
Although NPs are made of high biosafety materials, there is still a risk of damages to liver, kidney, and other organs if they stay in the body for a long time and cannot be degraded or excreted The use of materials that will be disintegrated after near-infrared light irradiation to fabricate NPs may be a solution to this problem. Recently, more and more NPs have been produced by these materials. Such NPs mediate PTT while loading drugs, meanwhile, the substances produced by the disintegration of NPs can be rapidly metabolized by the human body. In addition, the use of more biocompatible and degradable materials for nanoparticle preparation is also a solution. For example, the surface of chitosan is positively charged and can be broken down by the colonic flora, which facilitates interaction with specific tissues and can be metabolized by the body. The biocompatibility and degradability of chitosan has been proven to be non-toxic at appropriate drug concentrations [128].
-
(iv)
Difficulty in avoiding mononuclear phagocytic system (MPS)
In biofluids, NPs will adsorb proteins to form a corona layer referred to as “protein corona” in a broader sense giving biological identity to NPs and alters their biological characters, which will attract MPS especially macrophages to uptake NPs [129]. In order to avoid being uptaken by MPS, various polymer coatings such as forpolyether, polybetaine (PB) and polyolhave were investigated to cover NPs. For example, polyglycerol-grafting NPs are able to evade macrophage uptake by reducing protein adsorption [130]. In addition, there are two types of tumor-associated macrophages (TAM), M1 and M2. M1 macrophages inhibit tumor growth while M2 macrophages promote tumor growth. Therefore, no longer avoiding macrophages, but designing NPs targeted by macrophages, by regulating the function of macrophages, and even using macrophages as new drug carriers to exert anti-tumor effects may become a novel solution. At present, common design strategies for such NPs include inhibiting macrophage recruitment, depleting TAM, reprogramming TAMs, and blocking CD47-SIRPα pathway [131]. Among them, following the design concept of reprogramming or blocking CD47-SIRPα pathway, NPs that repolarize M2 macrophages to M1 type have made a breakthrough in vivo experiments [132].
Considering the above difficulties and referencing to advanced researches, we come up with a new possible design of NPs. The NPs skeleton is made of pyrolytic material (spirulina, exosomes, et al.). Then, photothermal materials (Au, Pd, etc.) are deposited on the NPs skeleton through electroless plating. After that the superparamagnetic iron oxide will be loaded on the surface of NPs through the sol–gel method. Then, suitable polymers (polybetaine, polyglycerol, etc.) will coat the NPs. Finally, drug (like doxorubicin) will be loaded on the NPs. Afterwards, under the guidance of PACT, NPs will be injected into the upstream of tumor supplying blood vessel, and the tumor will be irradiated with NIR. At the same time, three-dimensional magnetic field control is given to maximize the accumulation of NPs at the tumor site. Through this design, a large number of NPs will accumulate at the tumor site to ensure the drug concentration and PTT effect. At the same time, most NPs will be decomposed at the tumor site, and only a small number of NPs will circulate in the body.
Nowadays, anti-tumor therapy with NPs as the main body is still in the exploratory stage, and related technologies and equipments need to be invented, so it is unlikely to be clinically used in the short term. However, NPs can change part of the function or structure of many actual technologies. The upgrade of actual technologies is expected to be applied in clinic quickly, which contributes to upgrading the diagnosis and treatment of tumors in consequence. For example, NPs can help to develop electrochemical devices based on the interaction between ions and conductive polymers, such as organic electrochemical transistors (OFETs), electrolyte gated field-effect transistors (FETs), fin field-effect transistor (FinFETs), tunneling field-effect transistors (TFETs), electrochemical lab-on-chips (LOCs) [133]. These electrochemical devices are widely used in various tumor testing and diagnostic equipment. The use of NPs can help improve the accuracy of the equipment and reduce the detecting time. Many studies indicate that medical equipment using electronic components upgraded by NPs have been applied clinically [133–136].
Based on the evidence cited above, future research of NPs may not only focus on NPs themselves but also consider a feasible administration and efficacy assessing platform. In addition, the platform needs to be able to monitor immunotoxicity, the long-term toxicity, and neurotoxicity of NPs. As nanotechnology develops, if these problems were solved, NPs would be an ideal approach to upgrade cancer therapy and diagnosis.
Acknowledgements
None.
Abbreviations
- NPs
Nanoparticles
- PDT
Photodynamics therapy
- PTT
Photothermal therapy
- SPR
Plasmon resonance effect
- Au NRs
Gold nanorods
- SI-ATRP
Surface-initiated atom transfer radical polymerization
- NIPAAM
N-isopropylacrylamide
- US
Ultrasound
- MSNs
Mesoporous silica nanoparticles
- USMO
Ultrasmall manganese oxide
- GEM
Gemcitabine
- OINPs
Oxygen/indocyanine green-loaded lipid nanoparticles
- PA
Photoacoustic
- MPI
Magnetic particle imaging
- MRI
Magnetic resonance imaging
- SPIO
Superparamagnetic iron oxide
- USPIO
Ultra-small SPIO
- OCT
Optical coherence tomography
- MMOCT
Magnetomotive optical coherence tomography
- mAb
Monoclonal antibody
- DOX
Doxorubicin
- 5-FU
5-Fluorouracil
- FA
Folic acid
- PTX
Paclitaxel
- ROS
Reactive oxygen species
- EPR
Enhanced permeability and retention effect
- EGFR
Epidermal growth factor receptor
- BHC
Berberine hydrochloride
- AFP-1
Antifreeze protein
- PCMs
Phase change materials
- 3DPCT
3D printed coplanar template
- RCDs
Amino-rich red emissive carbon dots
- COF
Covalent organic framework
- ICG
Indocyanine green
- HSA
Serum albumin
- MDR
Multidrug resistance
- MRP1
Multidrug resistance-associated protein 1
- p-gp
P-glycoprotein
- PSs
Photosensitizing substances
- UC
Up-conversion
- RT
Radiotherapy
- PLGA
Poly(lactic-co-glycolic acid)
- PACT
Photoacoustic computed tomography
- MPS
Mononuclear phagocytic system
- PB
Polybetaine
- TAM
Tumor-associated macrophages
- OFETs
Organic electrochemical transistors
- FETs
Electrolyte gated field-effect transistors
- FinFETs
Fin field-effect transistor
- TFETs
Tunnelling field-effect transistors
- LOCs
Electrochemical lab-on-chips
Authors’ contributions
ZY: Writing- Original draft preparation. ZY and KC: Performed the Literature Search of the Databases. LG: Writing- Reviewing and Editing. QZ: translation. QL and WZ: investigation. KH: Supervision. All authors read and approved the final manuscript.
Funding
(1) National Key R&D Program of China (2018YFC1705102), which supported by Biological Center of Ministry of Science and Technology of China. (2) Capital Health Development Research Project (CFH2018-1-4201), which supported by Beijing Municipal Science and Technology Commission. (3) New Innovation Project-Yiqilin Leading Talent Project, which supported by Beijing Yizhuang Economic Development Zone Government. The fund doesn’t have code.
Availability of data and materials
All data generated or analysed during this study are included in this published article.
Competing interests
No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Zhongyang Yu and Lei Gao are co-first authors.
References
- 1.Liu Y, Bhattarai P, Dai Z, Chen X. Photothermal therapy and photoacoustic imaging via nanotheranostics in fighting cancer. Chem Soc Rev. 2019;48:2053–2108. doi: 10.1039/C8CS00618K. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Herma R, et al. Carbosilane dendrimers with phosphonium terminal groups are low toxic non-viral transfection vectors for sirna cell delivery. Int J Pharmaceut. 2019;562:51–65. doi: 10.1016/j.ijpharm.2019.03.018. [DOI] [PubMed] [Google Scholar]
- 3.Woodman C, Vundu G, George A, Wilson CM (2021) Applications and strategies in nanodiagnosis and nanotherapy in lung cancer. Semin Cancer Biol 69:349–364 [DOI] [PubMed]
- 4.Israel LL, Galstyan A, Holler E, Ljubimova JY. Magnetic iron oxide nanoparticles for imaging, targeting and treatment of primary and metastatic tumors of the brain. J Control Release. 2020;320:45–62. doi: 10.1016/j.jconrel.2020.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Singh H, Du J, Singh P, Mavlonov GT, Yi T. Development of superparamagnetic iron oxide nanoparticles via direct conjugation with ginsenosides and its in-vitro study. J Photochem Photobiol B. 2018;185:100–110. doi: 10.1016/j.jphotobiol.2018.05.030. [DOI] [PubMed] [Google Scholar]
- 6.Tao Y, Li M, Ren J, Qu X. Metal nanoclusters: novel probes for diagnostic and therapeutic applications. Chem Soc Rev. 2015;44:8636–8663. doi: 10.1039/C5CS00607D. [DOI] [PubMed] [Google Scholar]
- 7.Sajid M, Potka-Wasylka J. Nanoparticles: synthesis, characteristics, and applications in analytical and other sciences. Microchem J. 2020;154:104623. doi: 10.1016/j.microc.2020.104623. [DOI] [Google Scholar]
- 8.Priyanka KP, Sukirtha TH, Balakrishna KM, Varghese T (2016) Microbicidal activity of TiO2 nanoparticles synthesised by sol–gel method. IET Nanobiotechnol 10(2):81–86 [DOI] [PMC free article] [PubMed]
- 9.Qi X, Xiong L, Peng J, Tang D. Near infrared laser-controlled drug release of thermoresponsive microgel encapsulated with Fe3O4 nanoparticles. RSC Adv. 2017;7:19604–19610. doi: 10.1039/C7RA01009E. [DOI] [Google Scholar]
- 10.Liu F, et al. Controllable synthesis of polydopamine nanoparticles in microemulsions with pH-activatable properties for cancer detection and treatment. J Mater Chem B. 2015;3:6731–6739. doi: 10.1039/C5TB01159K. [DOI] [PubMed] [Google Scholar]
- 11.Daou TJ, et al. Hydrothermal synthesis of monodisperse magnetite nanoparticles. Chem Mater. 2006;18:4399–4404. doi: 10.1021/cm060805r. [DOI] [Google Scholar]
- 12.McGilvray KL, Decan MR, Wang D, Scaiano JC. Facile photochemical synthesis of unprotected aqueous gold nanoparticles. J Am Chem Soc. 2006;128:15980–15981. doi: 10.1021/ja066522h. [DOI] [PubMed] [Google Scholar]
- 13.Shahriyari F, Yaarali D, Ahmadi R, et al. (2020) Synthesis and characterization of Cu-Sn oxides nanoparticles via wire explosion method with surfactants, evaluation of in-vitro cytotoxic and antibacterial properties. Adv Powder Technol 31(6):2337–2347
- 14.de Carvalho JF, de Medeiros SN, Morales MA, Dantas AL, Carriço AS. Synthesis of magnetite nanoparticles by high energy ball milling. Appl Surf Sci. 2013;275:84–87. doi: 10.1016/j.apsusc.2013.01.118. [DOI] [Google Scholar]
- 15.Mascolo MC, Pei Y, Ring TA. Room temperature co-precipitation synthesis of magnetite nanoparticles in a large pH window with different bases. Materials (Basel, Switzerland) 2013;6:5549–5567. doi: 10.3390/ma6125549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Seidi F, Jenjob R, Phakkeeree T, Crespy D. Saccharides, oligosaccharides, and polysaccharides nanoparticles for biomedical applications. J Control Release. 2018;284:188–212. doi: 10.1016/j.jconrel.2018.06.026. [DOI] [PubMed] [Google Scholar]
- 17.Chandler M, Afonin K. Smart-responsive nucleic acid nanoparticles (NANPs) with the potential to modulate immune behavior. Nanomaterials. 2019;9:611. doi: 10.3390/nano9040611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Asadian E, Ghalkhani M, Shahrokhian S (2019) Electrochemical sensing based on carbon nanoparticles: a review. Sens Actuators B Chem 293(C):183–209
- 19.Wang J, Shah ZH, Zhang S, Lu R. Silica-based nanocomposites via reverse microemulsions: classifications, preparations, and applications. Nanoscale. 2014;6:4418–4437. doi: 10.1039/c3nr06025j. [DOI] [PubMed] [Google Scholar]
- 20.Xie C, Yang Z, Sun Y. Synthesis and characterization of monodispersed SiO2@Y3Al5O12:Er3+ core–shell particles. J Fluoresc. 2009;19:623–629. doi: 10.1007/s10895-008-0453-8. [DOI] [PubMed] [Google Scholar]
- 21.Yu J, Yang L, Zhang L. Pattern generation and motion control of a vortex-like paramagnetic nanoparticle swarm. Int J Robot Res. 2018;37(8):912–930. doi: 10.1177/0278364918784366. [DOI] [Google Scholar]
- 22.Wei Q, Ji J, Shen J. Synthesis of near-infrared responsive gold nanorod/PNIPAAm core/shell nanohybrids via surface initiated ATRP for smart drug delivery. Macromol Rapid Commun. 2008;29:645–650. doi: 10.1002/marc.200800009. [DOI] [Google Scholar]
- 23.Nallathamby PD, Hopf J, Irimata LE, McGinnity TL, Roeder RK (2016) Preparation of fluorescent Au–SiO2 core–shell nanoparticles and nanorods with tunable silica shell thickness and surface modification for immunotargeting. J Mater Chem B 4(32):5418–5428 [DOI] [PubMed]
- 24.Gour A, Jain NK. Advances in green synthesis of nanoparticles. Artif Cells Nanomed Biotechnol. 2019;47:844–851. doi: 10.1080/21691401.2019.1577878. [DOI] [PubMed] [Google Scholar]
- 25.Alijani HQ, Pourseyedi S, Torkzadeh Mahani M, Khatami M. Green synthesis of zinc sulfide (ZnS) nanoparticles using Stevia rebaudiana Bertoni and evaluation of its cytotoxic properties. J Mol Struct. 2019;1175:214–218. doi: 10.1016/j.molstruc.2018.07.103. [DOI] [Google Scholar]
- 26.Miri A, Sarani M. Biosynthesis, characterization and cytotoxic activity of CeO2 nanoparticles. Ceram Int. 2018;44:12642–12647. doi: 10.1016/j.ceramint.2018.04.063. [DOI] [Google Scholar]
- 27.Smith B, Gambhir SS. Nanomaterials for in vivo imaging. Chem Rev. 2017;117:901–986. doi: 10.1021/acs.chemrev.6b00073. [DOI] [PubMed] [Google Scholar]
- 28.Hou Y, Sun Z, Rao W, Liu J. Nanoparticle-mediated cryosurgery for tumor therapy. Nanomed Nanotechnol Biol Med. 2018;14:493–506. doi: 10.1016/j.nano.2017.11.018. [DOI] [PubMed] [Google Scholar]
- 29.Lai J, et al. MnO nanoparticles with unique excitation-dependent fluorescence for multicolor cellular imaging and MR imaging of brain glioma. Mikrochim Acta. 2018;185:244. doi: 10.1007/s00604-018-2779-5. [DOI] [PubMed] [Google Scholar]
- 30.Huang H, et al. GSH-sensitive Pt(IV) prodrug-loaded phase-transitional nanoparticles with a hybrid lipid-polymer shell for precise theranostics against ovarian cancer. Theranostics. 2019;9:1047–1065. doi: 10.7150/thno.29820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang D, et al. Effective pH-activated theranostic platform for synchronous magnetic resonance imaging diagnosis and chemotherapy. ACS Appl Mater Interfaces. 2018;10:31114–31123. doi: 10.1021/acsami.8b11408. [DOI] [PubMed] [Google Scholar]
- 32.Liu Y, et al. Folate-Targeted and oxygen/indocyanine green-loaded lipid nanoparticles for dual-mode imaging and photo-sonodynamic/photothermal therapy of ovarian cancer in vitro and in vivo. Mol Pharm. 2019;16:4104–4120. doi: 10.1021/acs.molpharmaceut.9b00339. [DOI] [PubMed] [Google Scholar]
- 33.Jiang W, et al. PEG-coated and Gd-loaded fluorescent silica nanoparticles for targeted prostate cancer magnetic resonance imaging and fluorescence imaging. Int J Nanomed. 2019;14:5611–5622. doi: 10.2147/IJN.S207098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Makela AV, Gaudet JM, Schott MA, Sehl OC, Contag CH, Foster PJ (2020) Magnetic particle imaging of macrophages associated with cancer: filling the voids left by iron-based magnetic resonance imaging. Mol Imaging Biol 22(4):958–968 [DOI] [PubMed]
- 35.Huang D, et al. Optical coherence tomography. Science. 1991;254:1178–1181. doi: 10.1126/science.1957169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Odonnell M. Magnetic nanoparticles as contrast agents for molecular imaging in medicine. Phys C. 2018;548:103–106. doi: 10.1016/j.physc.2018.02.031. [DOI] [Google Scholar]
- 37.Gao Z, et al. Small is smarter: nano MRI contrast agents—advantages and recent achievements. Small. 2016;12:556–576. doi: 10.1002/smll.201502309. [DOI] [PubMed] [Google Scholar]
- 38.Estelrich J, Sanchezmartin MJ, Busquets MA. Nanoparticles in magnetic resonance imaging: from simple to dual contrast agents. Int J Nanomed. 2015;10:1727–1741. doi: 10.2147/IJN.S76501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ito A, Shinkai M, Honda H, Kobayashi T. Medical application of functionalized magnetic nanoparticles. J Biosci Bioeng. 2005;100:1–11. doi: 10.1263/jbb.100.1. [DOI] [PubMed] [Google Scholar]
- 40.Albanese A, Tang PS, Chan WCW. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu Rev Biomed Eng. 2012;14:1–16. doi: 10.1146/annurev-bioeng-071811-150124. [DOI] [PubMed] [Google Scholar]
- 41.Oldenburg AL, Toublan FJJ, Suslick KS, Wei A, Boppart SA. Magnetomotive contrast for in vivo optical coherence tomography. Opt Express. 2005;13:6597–6614. doi: 10.1364/OPEX.13.006597. [DOI] [PubMed] [Google Scholar]
- 42.Laconte LEW, Nitin N, Bao G. Magnetic nanoparticle probes. Mater Today. 2005;8:32–38. doi: 10.1016/S1369-7021(05)00893-X. [DOI] [Google Scholar]
- 43.Dixit S, et al. Transferrin receptor-targeted theranostic gold nanoparticles for photosensitizer delivery in brain tumors. Nanoscale. 2015;7:1782–1790. doi: 10.1039/C4NR04853A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Liu C, et al. Magnetic/upconversion fluorescent NaGdF4:Yb, Er nanoparticle-based dual-modal molecular probes for imaging tiny tumors in vivo. ACS Nano. 2013;7:7227–7240. doi: 10.1021/nn4030898. [DOI] [PubMed] [Google Scholar]
- 45.Bahrami B, et al. Nanoparticles and targeted drug delivery in cancer therapy. Immunol Lett. 2017;190:64–83. doi: 10.1016/j.imlet.2017.07.015. [DOI] [PubMed] [Google Scholar]
- 46.Maiyo F, Singh M. Selenium nanoparticles: potential in cancer gene and drug delivery. Nanomedicine-UK. 2017;12:1075–1089. doi: 10.2217/nnm-2017-0024. [DOI] [PubMed] [Google Scholar]
- 47.Zhang J, Sun H, Ma PX. Host−guest interaction mediated polymeric assemblies: multifunctional nanoparticles for drug and gene delivery. ACS Nano. 2010;4:1049–1059. doi: 10.1021/nn901213a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Surendran SP, Moon MJ, Park R, Jeong YY. Bioactive nanoparticles for cancer immunotherapy. Int J Mol Sci. 2018;19:3877. doi: 10.3390/ijms19123877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Prabhu RH, Patravale VB, Joshi MD. Polymeric nanoparticles for targeted treatment in oncology: current insights. Int J Nanomed. 2015;10:1001–1018. doi: 10.2147/IJN.S56932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Smith AM, Duan H, Mohs AM, Nie S. Bioconjugated quantum dots for in vivo molecular and cellular imaging. Adv Drug Deliv Rev. 2008;60:1226–1240. doi: 10.1016/j.addr.2008.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ferreira DDS, Lopes SCDA, Franco MS, De Oliveira MC. PH-sensitive liposomes for drug delivery in cancer treatment. Ther Deliv. 2013;4:1099–1123. doi: 10.4155/tde.13.80. [DOI] [PubMed] [Google Scholar]
- 52.Jiang L, Vader P, Schiffelers RM. Extracellular vesicles for nucleic acid delivery: progress and prospects for safe RNA-based gene therapy. Gene Ther. 2017;24:157–166. doi: 10.1038/gt.2017.8. [DOI] [PubMed] [Google Scholar]
- 53.Amini MA, et al. Combining tumor microenvironment modulating nanoparticles with doxorubicin to enhance chemotherapeutic efficacy and boost antitumor immunity. J Natl Cancer Inst. 2019;111:399–408. doi: 10.1093/jnci/djy131. [DOI] [PubMed] [Google Scholar]
- 54.Salem DS, Sliem MA, Elsesy MS, Shouman SA, Badr Y. Improved chemo-photothermal therapy of hepatocellular carcinoma using chitosan-coated gold nanoparticles. J Photochem Photobiol B. 2018;182:92–99. doi: 10.1016/j.jphotobiol.2018.03.024. [DOI] [PubMed] [Google Scholar]
- 55.Zhang L, Xie X, Liu D, Xu ZP, Liu R. Efficient co-delivery of neo-epitopes using dispersion-stable layered double hydroxide nanoparticles for enhanced melanoma immunotherapy. Biomaterials. 2018;174:54–66. doi: 10.1016/j.biomaterials.2018.05.015. [DOI] [PubMed] [Google Scholar]
- 56.Gu L, et al. Folate-modified, indocyanine green-loaded lipid-polymer hybrid nanoparticles for targeted delivery of cisplatin. J Biomater Sci Polym E. 2017;28:690–702. doi: 10.1080/09205063.2017.1296347. [DOI] [PubMed] [Google Scholar]
- 57.Zhang Y, Li N, Suh H, Irvine DJ. Nanoparticle anchoring targets immune agonists to tumors enabling anti-cancer immunity without systemic toxicity. Nat Commun. 2018;9:6. doi: 10.1038/s41467-017-02251-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Avedian N, Zaaeri F, Daryasari MP, Javar HA, Khoobi M. PH-sensitive biocompatible mesoporous magnetic nanoparticles labeled with folic acid as an efficient carrier for controlled anticancer drug delivery. J Drug Deliv Sci Technol. 2018;44:323–332. doi: 10.1016/j.jddst.2018.01.006. [DOI] [Google Scholar]
- 59.Zheng X, et al. The theranostic efficiency of tumor-specific, ph-responsive, peptide-modified, liposome-containing paclitaxel and superparamagnetic iron oxide nanoparticles. Int J Nanomed. 2018;13:1495–1504. doi: 10.2147/IJN.S157082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Xia Y, et al. Functionalized selenium nanoparticles for targeted sirna delivery silence Derlin1 and promote antitumor efficacy against cervical cancer. Drug Deliv. 2020;27:15–25. doi: 10.1080/10717544.2019.1667452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bae YH, Park K. Targeted drug delivery to tumors: myths, reality and possibility. J Control Release. 2011;153:198–205. doi: 10.1016/j.jconrel.2011.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Farokhzad OC, Langer R. Impact of nanotechnology on drug delivery. ACS Nano. 2009;3:16–20. doi: 10.1021/nn900002m. [DOI] [PubMed] [Google Scholar]
- 63.Sandoval MA, et al. EGFR-targeted stearoyl gemcitabine nanoparticles show enhanced anti-tumor activity. J Control Release. 2012;157:287–296. doi: 10.1016/j.jconrel.2011.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Pandey S, et al. Biogenic gold nanoparticles as fotillas to fire berberine hydrochloride using folic acid as molecular road map. Mater Sci Eng C. 2013;33:3716–3722. doi: 10.1016/j.msec.2013.05.007. [DOI] [PubMed] [Google Scholar]
- 65.Li L, et al. Codelivery of DOX and siRNA by folate-biotin-quaternized starch nanoparticles for promoting synergistic suppression of human lung cancer cells. Drug Deliv. 2019;26:499–508. doi: 10.1080/10717544.2019.1606363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zhou Y, et al. Improving the anticancer efficacy of laminin receptor-specific therapeutic ruthenium nanoparticles (RuBB-loaded EGCG-RuNPs) via ROS-dependent apoptosis in SMMC-7721 cells. ACS Appl Mater Interfaces. 2016;8:15000–15012. doi: 10.1021/acsami.5b02261. [DOI] [PubMed] [Google Scholar]
- 67.Chen Q, et al. Multifunctional selenium nanoparticles: chiral selectivity of delivering MDR-siRNA for reversal of multidrug resistance and real-time biofluorescence imaging. Nanomed Nanotechnol Biol Med. 2015;11:1773–1784. doi: 10.1016/j.nano.2015.04.011. [DOI] [PubMed] [Google Scholar]
- 68.Xia Y, et al. Novel functionalized nanoparticles for tumor-targeting co-delivery of doxorubicin and sirna to enhance cancer therapy. Int J Nanomed. 2017;13:143–159. doi: 10.2147/IJN.S148960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Fu X, et al. RGD peptide-conjugated selenium nanoparticles: antiangiogenesis by suppressing VEGF-VEGFR2-ERK/AKT pathway. Nanomed Nanotechnol Biol Med. 2016;12:1627–1639. doi: 10.1016/j.nano.2016.01.012. [DOI] [PubMed] [Google Scholar]
- 70.Xia Y, et al. Silencing KLK12 expression via RGDfC-decorated selenium nanoparticles for the treatment of colorectal cancer in vitro and in vivo. Mater Sci Eng C. 2020;110:110594. doi: 10.1016/j.msec.2019.110594. [DOI] [PubMed] [Google Scholar]
- 71.Xia Y, et al. Doxorubicin-loaded functionalized selenium nanoparticles for enhanced antitumor efficacy in cervical carcinoma therapy. Mater Sci Eng C. 2020;106:110100. doi: 10.1016/j.msec.2019.110100. [DOI] [PubMed] [Google Scholar]
- 72.Muldrew K, et al. Flounder antifreeze peptides increase the efficacy of cryosurgery. Cryobiology. 2001;42:182–189. doi: 10.1006/cryo.2001.2321. [DOI] [PubMed] [Google Scholar]
- 73.Liu J, Deng Z. Nano-cryosurgery: advances and challenges. J Nanosci Nanotechnol. 2009;9:4521–4542. doi: 10.1166/jnn.2009.1264. [DOI] [PubMed] [Google Scholar]
- 74.Di D, He Z, Sun Z, Liu J. A new nano-cryosurgical modality for tumor treatment using biodegradable MgO nanoparticles. Nanomed Nanotechnol Biol Med. 2012;8:1233–1241. doi: 10.1016/j.nano.2012.02.010. [DOI] [PubMed] [Google Scholar]
- 75.Chua KJ, Chou SK, Ho JC. An analytical study on the thermal effects of cryosurgery on selective cell destruction. J Biomech. 2007;40:100–116. doi: 10.1016/j.jbiomech.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 76.Lv Y, Zou Y, Yang L. Uncertainty and sensitivity analysis of properties of phase change micro/nanoparticles for thermal protection during cryosurgery. Forsch Ingenieurwes. 2012;76:41–50. doi: 10.1007/s10010-012-0153-z. [DOI] [Google Scholar]
- 77.Ye P, et al. Fe3O4 nanoparticles and cryoablation enhance ice crystal formation to improve the efficiency of killing breast cancer cells. Oncotarget. 2017;8:11389–11399. doi: 10.18632/oncotarget.13859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Jelveh S, Chithrani DB. Gold nanostructures as a platform for combinational therapy in future cancer therapeutics. Cancers. 2011;3:1081–1110. doi: 10.3390/cancers3011081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wang H, et al. Enhanced cancer therapy with cold-controlled drug release and photothermal warming enabled by one nanoplatform. Biomaterials. 2018;180:265–278. doi: 10.1016/j.biomaterials.2018.07.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Slovak R, Ludwig JM, Gettinger SN, Herbst RS, Kim HS. Immuno-thermal ablations-boosting the anticancer immune response. J ImmunoTherapy Cancer. 2017;5:78. doi: 10.1186/s40425-017-0284-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Hou X, et al. Nanoparticle-based photothermal and photodynamic immunotherapy for tumor treatment. Int J Cancer. 2018;143:3050–3060. doi: 10.1002/ijc.31717. [DOI] [PubMed] [Google Scholar]
- 82.Zhu X, et al. Folic acid-modified and functionalized CuS nanocrystal-based nanoparticles for combined tumor chemo- and photothermal therapy. J Drug Target. 2017;25:425–435. doi: 10.1080/1061186X.2016.1266651. [DOI] [PubMed] [Google Scholar]
- 83.Zhu H, et al. Targeted delivery of siRNA with pH-responsive hybrid gold nanostars for cancer treatment. Int J Mol Sci. 2017;18:2029. doi: 10.3390/ijms18020444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ju Y, et al. Monodisperse Au-Fe2C Janus nanoparticles: an attractive multifunctional material for triple-modal imaging-guided tumor photothermal therapy. ACS Nano. 2017;11:9239–9248. doi: 10.1021/acsnano.7b04461. [DOI] [PubMed] [Google Scholar]
- 85.Song S, et al. Indocyanine green loaded magnetic carbon nanoparticles for near infrared fluorescence/magnetic resonance dual-modal imaging and photothermal therapy of tumor. ACS Appl Mater Interfaces. 2017;9:9484–9495. doi: 10.1021/acsami.7b00490. [DOI] [PubMed] [Google Scholar]
- 86.Pan L, Liu J, Shi J. Nuclear-targeting gold nanorods for extremely low nir activated photothermal therapy. ACS Appl Mater Interfaces. 2017;9:15952–15961. doi: 10.1021/acsami.7b03017. [DOI] [PubMed] [Google Scholar]
- 87.Yang G, et al. Hollow MnO2 as a tumor-microenvironment-responsive biodegradable nano-platform for combination therapy favoring antitumor immune responses. Nat Commun. 2017;8:902. doi: 10.1038/s41467-017-01050-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Singh SP, et al. NIR triggered liposome gold nanoparticles entrapping curcumin as in situ adjuvant for photothermal treatment of skin cancer. Int J Biol Macromol. 2017;110:375–382. doi: 10.1016/j.ijbiomac.2017.11.163. [DOI] [PubMed] [Google Scholar]
- 89.Gschwend PM, Conti S, Kaech A, Maake C, Pratsinis SE. Silica-coated TiN particles for killing cancer cells. ACS Appl Mater Interfaces. 2019;11:22550–22560. doi: 10.1021/acsami.9b07239. [DOI] [PubMed] [Google Scholar]
- 90.Rizzi M, et al. Verteporfin based silica nanoparticle for in vitro selective inhibition of human highly invasive melanoma cell proliferation. J Photochem Photobiol B. 2017;167:1–6. doi: 10.1016/j.jphotobiol.2016.12.021. [DOI] [PubMed] [Google Scholar]
- 91.Jin J, et al. Graphdiyne nanosheet-based drug delivery platform for photothermal/chemotherapy combination treatment of cancer. ACS Appl Mater Interfaces. 2018;10:8436–8442. doi: 10.1021/acsami.7b17219. [DOI] [PubMed] [Google Scholar]
- 92.Sun S, et al. Ce6-modified carbon dots for multimodal-imaging-guided and single-NIR-laser-triggered photothermal/photodynamic synergistic cancer therapy by reduced irradiation power. ACS Appl Mater Interfaces. 2019;11:5791–5803. doi: 10.1021/acsami.8b19042. [DOI] [PubMed] [Google Scholar]
- 93.Chunling H, Zhixiang Z, Sainan L, Xiangjian L, Maolin P. Monodispersed CuSe sensitized covalent organic framework photosensitizer with an enhanced photodynamic and photothermal effect for cancer therapy. ACS Appl Mater Interfaces. 2019;11:23072–23082. doi: 10.1021/acsami.9b08394. [DOI] [PubMed] [Google Scholar]
- 94.Ji C, et al. Activatable photodynamic therapy for prostate cancer by NIR dye/photosensitizer loaded albumin nanoparticles. J Biomed Nanotechnol. 2019;15:311–318. doi: 10.1166/jbn.2019.2685. [DOI] [PubMed] [Google Scholar]
- 95.Fletcher JI, Williams R, Henderson MJ, Norris MD, Haber M. ABC transporters as mediators of drug resistance and contributors to cancer cell biology. Drug Resist Update. 2016;26:1–9. doi: 10.1016/j.drup.2016.03.001. [DOI] [PubMed] [Google Scholar]
- 96.Li Y, et al. Multipronged design of light-triggered nanoparticles to overcome cisplatin resistance for efficient ablation of resistant tumor. ACS Nano. 2015;9:9626–9637. doi: 10.1021/acsnano.5b05097. [DOI] [PubMed] [Google Scholar]
- 97.Wang L, et al. Novel insights into combating cancer chemotherapy resistance using a plasmonic nanocarrier: enhancing drug sensitiveness and accumulation simultaneously with localized mild photothermal stimulus of femtosecond pulsed laser. Adv Funct Mater. 2014;24:4229–4239. doi: 10.1002/adfm.201400015. [DOI] [Google Scholar]
- 98.Wang L, et al. Using hollow carbon nanospheres as a light-induced free radical generator to overcome chemotherapy resistance. J Am Chem Soc. 2015;137:1947–1955. doi: 10.1021/ja511560b. [DOI] [PubMed] [Google Scholar]
- 99.Fay BL, Melamed JR, Day ES. Nanoshell-mediated photothermal therapy can enhance chemotherapy in inflammatory breast cancer cells. Int J Nanomed. 2015;10:6931–6941. doi: 10.2147/IJN.S93031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Moghissi K, Dixon K. Is Bronchoscopic photodynamic therapy a therapeutic option in lung cancer. Eur Respir J. 2003;22:535–541. doi: 10.1183/09031936.03.00005203. [DOI] [PubMed] [Google Scholar]
- 101.Gu T, et al. Upconversion Composite nanoparticles for tumor hypoxia modulation and enhanced near-infrared-triggered photodynamic therapy. ACS Appl Mater Interfaces. 2018;10:15494–15503. doi: 10.1021/acsami.8b03238. [DOI] [PubMed] [Google Scholar]
- 102.Li Y, et al. A versatile imaging and therapeutic platform based on dual-band luminescent lanthanide nanoparticles toward tumor metastasis inhibition. ACS Nano. 2016;10:2766–2773. doi: 10.1021/acsnano.5b07873. [DOI] [PubMed] [Google Scholar]
- 103.Jin J, Zhao Q. Engineering nanoparticles to reprogram radiotherapy and immunotherapy: recent advances and future challenges. J Nanobiotechnol. 2020;18:17–75. doi: 10.1186/s12951-020-0579-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Baumann M, et al. Radiation oncology in the era of precision medicine. Nat Rev Cancer. 2016;16:234–249. doi: 10.1038/nrc.2016.18. [DOI] [PubMed] [Google Scholar]
- 105.Her S, Jaffray DA, Allen C. Gold nanoparticles for applications in cancer radiotherapy: mechanisms and recent advancements. Adv Drug Deliver Rev. 2017;109:84–101. doi: 10.1016/j.addr.2015.12.012. [DOI] [PubMed] [Google Scholar]
- 106.Kwatra D, Venugopal A, Anant S. Nanoparticles in radiation therapy: a summary of various approaches to enhance radiosensitization in cancer. Transl Cancer Res. 2013;2:330–342. [Google Scholar]
- 107.Liu Y, et al. Metal-based nanoenhancers for future radiotherapy: radiosensitizing and synergistic effects on tumor cells. Theranostics. 2018;8:1824–1849. doi: 10.7150/thno.22172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Yang X, Yang M, Pang B, Vara M, Xia Y. Gold nanomaterials at work in biomedicine. Chem Rev. 2015;115:10410–10488. doi: 10.1021/acs.chemrev.5b00193. [DOI] [PubMed] [Google Scholar]
- 109.Herold DM, Das IJ, Stobbe CC, Iyer RV, Chapman JD. Gold microspheres: a selective technique for producing biologically effective dose enhancement. Int J Radiat Biol. 2000;76:1357–1364. doi: 10.1080/09553000050151637. [DOI] [PubMed] [Google Scholar]
- 110.Liu Y, et al. The dependence of radiation enhancement effect on the concentration of gold nanoparticles exposed to low- and high-LET radiations. Phys Med. 2015;31:210–218. doi: 10.1016/j.ejmp.2015.01.006. [DOI] [PubMed] [Google Scholar]
- 111.Charest G, Paquette B, Fortin D, Mathieu D, Sanche L. Concomitant treatment of F98 glioma cells with new liposomal platinum compounds and ionizing radiation. J Neuro-Oncol. 2010;97:187–193. doi: 10.1007/s11060-009-0011-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Jawaid P, et al. Effects of SOD/catalase mimetic platinum nanoparticles on radiation-induced apoptosis in human lymphoma U937 cells. Apoptosis. 2014;19:1006–1016. doi: 10.1007/s10495-014-0972-5. [DOI] [PubMed] [Google Scholar]
- 113.Ma P, et al. Enhanced cisplatin chemotherapy by iron oxide nanocarrier-mediated generation of highly toxic reactive oxygen species. Nano Lett. 2017;17:928–937. doi: 10.1021/acs.nanolett.6b04269. [DOI] [PubMed] [Google Scholar]
- 114.Hauser AK, et al. Targeted iron oxide nanoparticles for the enhancement of radiation therapy. Biomaterials. 2016;105:127–135. doi: 10.1016/j.biomaterials.2016.07.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Titus D, Samuel EJJ, Roopan SM. Current scenario of biomedical aspect of metal-based nanoparticles on gel dosimetry. Appl Microbiol Biotechnol. 2016;100:4803–4816. doi: 10.1007/s00253-016-7489-5. [DOI] [PubMed] [Google Scholar]
- 116.Khoei S, Mahdavi SR, Fakhimikabir H, Shakerizadeh A, Hashemian A. The role of iron oxide nanoparticles in the radiosensitization of human prostate carcinoma cell line DU145 at megavoltage radiation energies. Int J Radiat Biol. 2014;90:351–356. doi: 10.3109/09553002.2014.888104. [DOI] [PubMed] [Google Scholar]
- 117.Huang F, et al. Enhancement of irradiation effects on cancer cells by cross-linked dextran-coated iron oxide (CLIO) nanoparticles. Phys Med Biol. 2010;55:469–482. doi: 10.1088/0031-9155/55/2/009. [DOI] [PubMed] [Google Scholar]
- 118.Revia RA, Zhang M. Magnetite nanoparticles for cancer diagnosis, treatment, and treatment monitoring: recent advances. Mater Today. 2016;19:157–168. doi: 10.1016/j.mattod.2015.08.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Zhang H, Zhang W, Zhou Y, Jiang Y, Li S. Dual functional mesoporous silicon nanoparticles enhance the radiosensitivity of VPA in glioblastoma. Transl Oncol. 2017;10:229–240. doi: 10.1016/j.tranon.2016.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.He L, Lai H, Chen T. Dual-function nanosystem for synergetic cancer chemo-/radiotherapy through ros-mediated signaling pathways. Biomaterials. 2015;51:30–42. doi: 10.1016/j.biomaterials.2015.01.063. [DOI] [PubMed] [Google Scholar]
- 121.Luo L, et al. Laser immunotherapy in combination with perdurable PD-1 blocking for the treatment of metastatic tumors. ACS Nano. 2018;12:7647–7662. doi: 10.1021/acsnano.8b00204. [DOI] [PubMed] [Google Scholar]
- 122.Wang X, et al. Facile fabrication of magnetic microrobots based on spirulina templates for targeted delivery and synergistic chemo-photothermal therapy. ACS Appl Mater Interfaces. 2019;11:4745–4756. doi: 10.1021/acsami.8b15586. [DOI] [PubMed] [Google Scholar]
- 123.Jia G, et al. NRP-1 targeted and cargo-loaded exosomes facilitate simultaneous imaging and therapy of glioma in vitro and in vivo. Biomaterials. 2018;178:302–316. doi: 10.1016/j.biomaterials.2018.06.029. [DOI] [PubMed] [Google Scholar]
- 124.Hennig TL, Unterweger H, Lyer S, Alexiou C, Cicha I. Magnetic accumulation of SPIONs under arterial flow conditions: effect of serum and red blood cells. Molecules (Basel, Switzerland) 2019;24:2588. doi: 10.3390/molecules24142588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Manshadi MKD, et al. Delivery of magnetic micro/nanoparticles and magnetic-based drug/cargo into arterial flow for targeted therapy. Drug Deliv. 2018;25:1963–1973. doi: 10.1080/10717544.2018.1497106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Li L, et al. Small near-infrared photochromic protein for photoacoustic multi-contrast imaging and detection of protein interactions in vivo. Nat Commun. 2018;9:2734. doi: 10.1038/s41467-018-05231-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Wu Z, et al. A microrobotic system guided by photoacoustic computed tomography for targeted navigation in intestines in vivo. Sci Robot. 2019;4:x613. doi: 10.1126/scirobotics.aax0613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Cosco D, et al. Rutin-loaded chitosan microspheres: characterization and evaluation of the anti-inflammatory activity. Carbohydr Polym. 2016;152:583–591. doi: 10.1016/j.carbpol.2016.06.039. [DOI] [PubMed] [Google Scholar]
- 129.Tavano R, et al. C1q-mediated complement activation and C3 opsonization trigger recognition of stealth poly(2-methyl-2-oxazoline)-coated silica nanoparticles by human phagocytes. ACS Nano. 2018;12:5834–5847. doi: 10.1021/acsnano.8b01806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Zou Y, et al. Polyglycerol grafting shields nanoparticles from protein corona formation to avoid macrophage uptake. ACS Nano. 2020;14:7216–7226. doi: 10.1021/acsnano.0c02289. [DOI] [PubMed] [Google Scholar]
- 131.Xia Y, et al. Engineering macrophages for cancer immunotherapy and drug delivery. Adv Mater. 2020;32:2002054. doi: 10.1002/adma.202002054. [DOI] [PubMed] [Google Scholar]
- 132.Rong L, et al. Iron chelated melanin-like nanoparticles for tumor-associated macrophage repolarization and cancer therapy. Biomaterials. 2019;225:119515. doi: 10.1016/j.biomaterials.2019.119515. [DOI] [PubMed] [Google Scholar]
- 133.Coluccio ML, et al. Emerging designs of electronic devices in biomedicine. Micromachines-Basel. 2020;11:123. doi: 10.3390/mi11020123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Malara N, et al. Superhydrophobic lab-on-chip measures secretome protonation state and provides a personalized risk assessment of sporadic tumour. NPJ Precis Oncol. 2018;2:26. doi: 10.1038/s41698-018-0069-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Xu X, et al. Detection EGFR exon 19 status of lung cancer patients by DNA electrochemical biosensor. Biosens Bioelectron. 2016;80:411–417. doi: 10.1016/j.bios.2016.02.009. [DOI] [PubMed] [Google Scholar]
- 136.Amouzadeh Tabrizi M, Shamsipur M, Farzin L. A high sensitive electrochemical aptasensor for the determination of VEGF165 in serum of lung cancer patient. Biosens Bioelectron. 2015;74:764–769. doi: 10.1016/j.bios.2015.07.032. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analysed during this study are included in this published article.