Abstract
Purpose
Minimally invasive (MIS) approaches in combination with short stems have gained popularity in recent years in total hip arthroplasty (THA). A decreased risk for periprosthetic femoral fractures (PFFs) is reported for cementless short-stem THA, but in contrast to other approaches, the risk factors for PFFs for short-stem THA using MIS anterolateral approach in supine position are not described in literature.
Methods
A single-center consecutive series of 1052 hips in 982 patients, performed between 2014 and 2019 with a short curved stem and a press fit using an MIS anterolateral approach in supine position, was retrospectively screened for inclusion. Fourteen patients were lost to follow-up. Therefore, 1038 THAs in 968 patients were included. Risk factors for intra- and postoperative PFFs within 90 days were analyzed. We investigated for sex, age, body mass index (BMI), diagnosis, and laterality.
Results
In total, 18 PFFs (1.7%) occurred. Intraoperative fracture occurred in ten cases ( 0.9%), with another eight cases (0.8%) occurring postoperatively. Increased American Society of Anesthesiologists (ASA) Score was a significant risk factor for PFF (p = 0.026), whereas sex (p = 0.155), age (p = 0.161), BMI (p = 0.996), and laterality (p = 1.000) were not. Seven PFFs (0.7%) required revision arthroplasty.
Conclusion
Cementless short-stem THA using the MIS anterolateral approach is a procedure with a low number of PFFs within 90 days from index surgery. Fracture rates are comparable to other MIS approaches, and comparable femoral short stems are used. Age, sex, and BMI were not identified as risk factors of PFF, while risk for PFF increased with ASA Score.
Level of Evidence
Level IV
Keywords: Short stem, Total hip arthroplasty, Minimally invasive, Anterolateral approach, Watson-Jones, Periprosthetic fracture
Periprosthetic femoral fracture (PFF) is a major complication in total hip arthroplasty (THA). The overall mortality risk within 12 months of the complication increases up to 11% in patients with PFF [1, 2]. Additionally, PFF often requires complex revision surgery [3, 4], which increases postoperative readmission and functional limitation after revision surgery [2, 5]. The prevalence of PFFs appears to be increasing [5, 6], with an expected increase of 4.6% every decade over the next 30 years [7].
Minimally invasive (MIS) approaches in THA have gained popularity over the last years owing to faster recovery, less pain, and fewer postoperative precautions [8, 9]. In particular, anterior-based MIS approaches are increasingly performed for THA, including the direct anterior approach (DAA) as well as the abductor-sparing MIS Watson-Jones anterolateral approach [10]. With the popularization of MIS approaches, femoral short stems are utilized more frequently in THA as they facilitate soft-tissue sparing implantation [11].
Periprosthetic fractures (PFFs) are a known risk factor in anterior approaches [8–10]. The rate of PFFs in MIS anterolateral approach can be as high as 8.3% within 90 days from index surgery [10]. Especially fractures of the greater trochanter are described in the literature, with a rate of 3.2% in nonobese and up to 9.7% in obese patients [9].
Female sex [12, 13], increasing age [12–15], presence of osteoporosis [15], and rheumatoid arthritis have been associated with increasing rates of PFFs [5]. In particular, cementless femoral components have been associated with a higher risk of intra- and postoperative PFFs [15–18]. In a recent review, cementless femoral implants in general, and especially single-wedge and double-wedge components, have higher rates of PFFs [5]. Cementless short-stem THA shows a reduced risk for PFFs compared with standard cementless straight stems [19]. The rate of PFFs was found to be significantly decreased in short stem in DAA, with 1.6% compared with 6.8% in cementless straight stems [19]. Molli et al. [20] proposed that a short stem could decrease the incidence of intraoperative periprosthetic fracture compared with a standard-length stem because of less load during broaching.
The rate of PFFs in cementless short-stem arthroplasty is not fully evaluated, especially in minimally invasive anterolateral approach. Therefore, we conducted this study to evaluate the rate of early PFFs in cementless short-stem THA using an MIS anterolateral approach in supine position.
Methods
Patients
A retrospective evaluation of consecutive THAs of a single center performed via a minimally invasive anterolateral approach using a cementless, curved short stem (Fitmore stem, ZimmerBiomet, Warsaw, IN, USA) and cementless titanium press-fit cup with or without screws (Allofit/-S, ZimmerBiomet, Warsaw, IN, USA) was carried out. The study was approved by the institutional review board (EK-No.: 1239/2019) in accordance with the World Medical Association Declaration of Helsinki. Because it was a retrospective evaluation of preexisting medical records, informed consent was not required.
A consecutive series of 1052 hips in 982 patients with index surgery between 2014 and 2019 was analyzed, and the medical records until 90 days postoperation were evaluated. Fourteen patients were lost to follow-up. Consequently, 1038 hips in 968 patients were included. All electronically saved and archived medical records were reviewed, including operative reports, postoperative notes, discharge summaries, and postoperative medical records. Age, sex, weight, height, body mass index (BMI), American Society of Anesthesiologists (ASA) Score, preoperative diagnosis, and laterality were documented. All reports of intra- and postoperative fractures within a 90-day postoperative period were collected.
Surgical technique and treatment protocol
Surgical procedures were carried out at the author’s institution by surgeons with different levels of experience including 11 consultants and 7 residents. All consultants perform more than 50 and all senior consultants more than 100 arthroplasties per year. Resident surgeries were done under the guidance of a consultant. In all cases, a minimally invasive anterolateral Watson-Jones approach in supine position on a standard operating table under laminar flow was performed [21]. Extremity preparation was performed with threefold antiseptic scrub with alcohol disinfectant. Draping with sterile adhesive surgical iodine film was used. A skin incision was centered over the greater trochanter. An incision at the border between the tensor fasciae latae and the tractus iliotibialis was performed. Then, the Watson-Jones interval between tensor fasciae latae and gluteus medius was bluntly dissected. A capsulectomy was performed in each case. The standardized peri- and postoperative protocol was identical in all cases, including single-shot antibiotics [cefuroxime 1.5 g intravenous (i.v.), directly preoperatively], weight-bearing as tolerated from the first postoperative day on, indomethacin 75 mg twice daily for the prevention of heterotopic ossification on days 1–4 postoperatively, and 40 mg low-molecular-weight heparin or rivaroxaban 10 mg for 28 days postoperatively as venous thromboembolic event prophylaxis.
In all patients, a cementless, curved short stem (Fitmore stem, ZimmerBiomet, Warsaw, IN, USA) was digitally templated using mediCAD version 5.1 (Hectec GmbH, Altdorf, Germany). Fitmore hip stem is a titanium alloy stem (Ti Al6V4) that has a Porolock Ti-VPS coating in the proximal part to enhance bone ingrowth and is available in four different neck angle options (127°, 129°, 137°, 140°) [22]. A cementless titanium press-fit cup with or without screws (Allofit/-S, ZimmerBiomet, Warsaw, IN, USA) was used in all patients.
In the case of suspected or apparent intraoperative PFF, fluoroscopy was draped and utilized. Fractures of the greater trochanter were treated either nonoperatively or with a cerclage wire, depending on the stability of the fracture and stem. Each patient was mobilized with touch weight-bearing for 6 weeks. In the case of intraoperative fractures of the calcar, medial, or lateral cortex, we performed a reduction around the implanted stem using cerclage wires. In the case of primary stability, Fitmore hip stem was kept in situ. Patients were mobilized with touch weight-bearing for 4 weeks. If primary stability was not achieved, a cementless straight stem (Alloclassic SL, ZimmerBiomet, Warsaw, IN, USA) or a cementless monoblock revision straight stem (Alloclassic SLL, ZimmerBiomet, Warsaw, IN, USA) was used. In the case of stem revision, patients were mobilized with touch weight-bearing for 4 weeks. In the case of intra- or postoperative PFF, patients received a clinical and radiological follow-up prior to permission of full weight-bearing. Patients were then followed clinically and radiologically at our outpatient department 3 months and 1 year after occurrence of PFF.
Statistics
Descriptive statistical analysis was conducted for age, sex, body mass index (BMI), preoperative diagnosis, and laterality. Shapiro–Wilk test was performed for testing for normality distribution. As not all variables were normally distributed, nonparametric testing was performed. Fisher’s exact test was performed for categorical variables (sex, diagnosis, ASA Score, laterality) to evaluate any association between independent variables and likelihood of a fracture. Post-hoc calculations with Bonferroni correction were carried out in the case of significant differences. Wilcoxon Mann–Whitney U test was performed on continuous variables (age and BMI). Binary logistic regression models for dichotomous outcomes were estimated to model the effect of sex, age, BMI, surgeon’s experience, and ASA Score on the likelihood of a fracture. A power analysis was not performed owing to the consecutive recruitment of patients over a longer time period with an observed PFF rate [23]. Statistical analysis was calculated with SPSS version 26 (IBM SPSS statistics, Chicago, IL, USA). A p value < 0.05 was considered as statistically significant.
Results
In total, 1038 hips in 968 patients were included. Eighteen (1.7%) periprosthetic fractures (PFFs) occurred. Of these fractures, ten PFFs (0.9%) occurred intraoperatively, while eight PFFs (0.8%) occurred postoperatively within the first 90 days after index surgery. Thirteen cases (1.25%) of PFFs occurred in female patients, and five in male (0.45%) patients. Average age at operation was 67.24 years (range 24.21–99.02 years), in patients without PFF 67.23 years (range 24.21–94.38 years), and in patients with PFF 71.78 years (range 42.04–99.02 years). Average time to diagnosis of postoperative fracture was 18.75 days (range 4–70 days). Average BMI was 27.92 ± 4.96 kg/m2 (range 16.38–48.93 kg/m2), in patients with PFF 28.0 ± 5.62 kg/m2 (range 17.78–43.44 kg/m2), and in patients without PFF 27.91 ± 4.95 kg/m2 (range 16.38–48.93 kg/m2). Revision rate was 0.7%, with 7 of 18 PFFs needing revision arthroplasty. Full patient demographics are presented in Table 1.
Table 1.
Patient demographics and p values for testing for statistical significance of occurrence in PFF stratified for sex, age, diagnosis, ASA Score, laterality, and BMI
| Patient demographics | p Valuea | |
|---|---|---|
| Sex, n (%) | 0.155 | |
| Male | 472 (45.5%) | |
| Female | 566 (54.5%) | |
| Age (years) | 67.24 ± 11.37 (24.21–99.02) | 0.161 |
| Average age (no fracture) | 67.23 ± 11.32 (24.21–94.38) | |
| Average age (fracture) | 71.78 ± 13.77 (42.04–99.02) | |
| Diagnosis, n (%) | 0.005 | |
| Primary osteoarthritis | 878 (84.6%) | |
| Avascular necrosis | 97 (9.3%) | |
| Hip dysplasia | 47 (4.5%) | |
| Femoral neck fracture | 2 (0.2%) | |
| Posttraumatic osteoarthritis | 14 (1.3%) | |
| ASA Score, n (%) | 0.026 | |
| ASA I | 195 (18.8%) | |
| ASA II | 591 (56.9%) | |
| ASA III | 247 (23.8%) | |
| ASA IV | 5 (0.5%) | |
| Laterality, n (%) | 1.000 | |
| Left | 488 (47.0%) | |
| Right | 550 (53.0%) | |
| BMI (kg/m2) | 27.92 ± 4.96(16.38–48.93) | 0.996 |
| BMI (no fracture) | 27.91 ± 4.95(16.38–48.93) | |
| BMI (fracture) | 28.0 ± 5.62(17.78–43.44) | |
| Surgeon’s experience (n, THAs) | ||
| Consultants | 788 (75.9%) | |
| Residents | 250 (24.1%) | |
| Fractures | ||
| Total | 18 (1.7%) | |
| Female | 13 (1.25%) | |
| Male | 5 (0.45%) | |
| Intraoperative fractures | 10 (0.9%) | |
| Postoperative fractures | 8 (0.8%) | |
| Revisions | 7 (0.7%) | |
| Fractures CCD angle | Fractures/THAs | 0.648 |
| A (140°) | 4/186 (2.2%) | |
| B (137°) | 12/604 (2.0%) | |
| B extended (129°) | 2/244 (0.8%) | |
| C (127°) | 0/18 (0.0%) |
aTesting patients with fracture versus patients without fracture
Intraoperative fractures occurred in ten patients (55.6%), while eight PFFs (44.4%) were diagnosed postoperatively. Type of fracture and time of occurrence/diagnosis of PFF are presented in Table 2. Table 3 presents cause of fracture, time of fracture in days, Vancouver classification of postoperative PFF, and treatment for every PFF in detail. Figures 1, 2 and 3 show different PFFs and their treatment.
Table 2.
Type of PFFs and time of occurrence/diagnosis
| Type (n) | Intraoperatively | Postoperatively | Percentage of all fractures | Percentage of all hips |
|---|---|---|---|---|
| Medial cortex (n = 8) | 5 (27.8%) | 5 (27.8%) | 55.6% | 1.0% |
| Calcar (n = 1) | 1 (5.6%) | 0 (0.0%) | 5.6% | 0.1% |
| Lateral cortex (n = 3) | 2 (11.1%) | 2 (11.1%) | 22.2% | 0.4% |
| Greater trochanter (n = 3) | 1 (5.6%) | 2 (11.1%) | 16.7% | 0.3% |
Table 3.
Cause of fracture, time of fracture, Vancouver classification, and treatment for all PFFs
| Fracture type | Cause of fracture | Time of fracture | Vancouver classification | Treatment |
|---|---|---|---|---|
| Intraoperatively (n = 10) | ||||
| Medial cortex | Implant insertion | One cerclage cable | ||
| Medial cortex | Implant insertion | One cerclage cable | ||
| Calcar | Implant insertion | One cerclage cable | ||
| Lateral cortex | Implant insertion | Two cerclage cables | ||
| Lateral cortex | Implant insertion | Conservatively, no weight-bearing for 6 weeks | ||
| Medial cortex | Broaching | One cerclage cable | ||
| Greater trochanter | Retraction with Hohmann retractor | One cerclage cable | ||
| Medial cortex | Implant insertion | One cerclage cable | ||
| Medial cortex | Implant insertion | Straight stem, two cerclage cables | ||
| Lateral cortex | Failed cerclage of intraoperatively detected fracture | Revision on day 31 | Vc B2 | Revision arthroplasty (monoblock revision stem, three cerclage cables) |
| Postoperatively (n = 8) | ||||
| Greater trochanter | Avulsion | 9d | Vc Ag | Conservatively |
| Medial cortex | No history of fall | 6d | Vc B2 | Revision arthroplasty (straight stem, four cerclage cables) |
| Medial cortex | No history of fall | 4d | Vc B2 | Revision arthroplasty (straight stem, three cerclage cables) |
| Medial cortex | No history of fall | 14d | Vc B2 | Revision arthroplasty (straight stem, three cerclage cables) |
| Medial cortex | Fell while walking | 24d | Vc B2 | Revision arthroplasty (Monoblock revision stem, three cerclage cables) |
| Medial cortex | No history of fall | 14d | Vc B2 | Revision arthroplasty (straight stem, three cerclage cables) |
| Greater trochanter | Avulsion | 5d | Vc Ag | Conservatively |
| Lateral cortex | Fell while walking | 70d | Vc B2 | Revision arthroplasty (monoblock revision stem, two cerclage cables) |
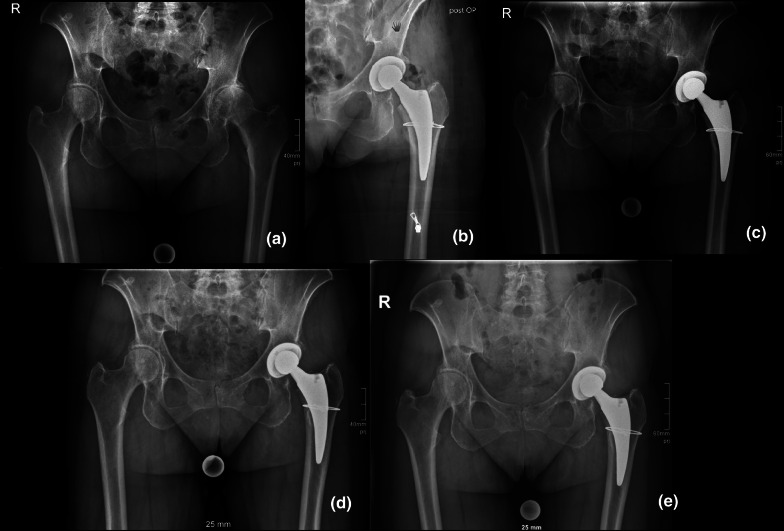
Fig. 1.
Intraoperative fracture of the calcar treated with one cerclage wire: a preoperative; b postoperative; c 6 weeks postoperative; d 3 months postoperative; e 1 year postoperative
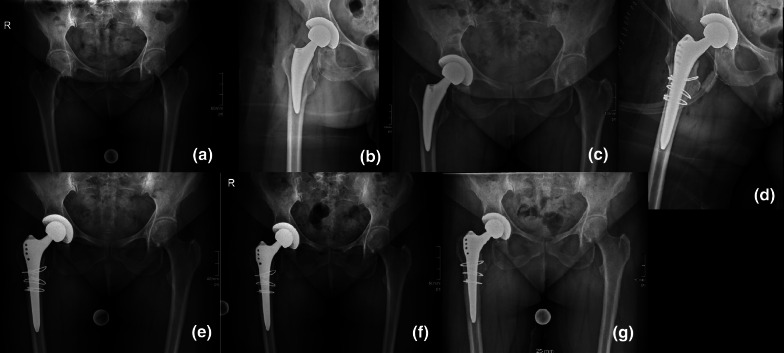
Fig. 2.
Occult fracture of the medial cortex detected on the fourth postoperative day; a preoperative; b postoperative; c fourth postoperative day; d postoperatively after revision (Alloclassic SL (ZimmerBiomet) with three cerclage wires); e 6 weeks after revision; f 3 months after revision; g 1 year after revision
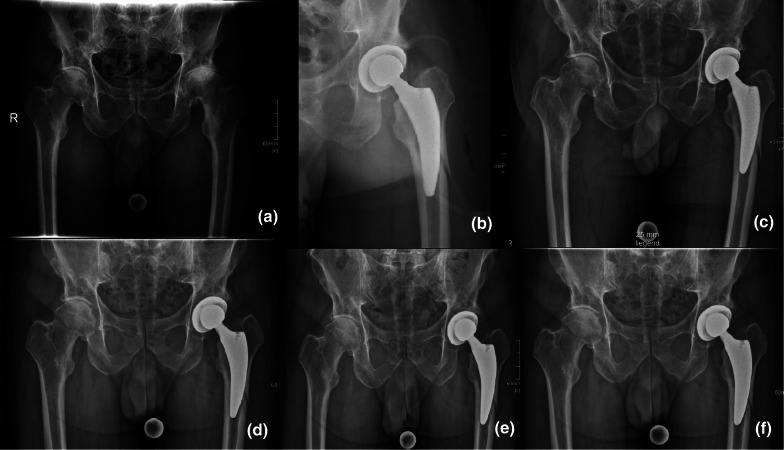
Fig. 3.
Intraoperative fracture treated without revision surgery; detected on day of surgery on postoperative x-ray with patient already outside the operating theater; patient did not give informed consent for revision surgery; conservative treatment with 6 weeks without weight-bearing; a preoperative, b postoperative, c discharge at home; d 6 weeks after surgery; e 3 months after surgery; f 1 year after surgery
Testing and logistic regression
The detailed results for testing are presented in Table 1 and for logistic regression in Table 4. A significance was detected for ASA Score with p = 0.026. Post-hoc calculation showed a significance for group ASA Score III. Testing for influence of diagnosis on PFF showed a p value of 0.005 for osteoarthritis. However, post-hoc calculations did not show any significance. Logistic regression showed an increased risk of PFF occurrence depending on diagnosis. Logistic regression showed an increased risk for PFFs with increasing ASA Score. A statistical significance for age and sex in occurrence of PFFs could not be detected for either variable. Logistic regression did not show any significance or increased risk for age, sex, or BMI in occurrence of PFFs.
Table 4.
Logistic regression for age, sex, BMI, ASA Score, diagnosis, surgeon’s experience, and neck angle; odds ratio, confidence interval (95%), and p value
| Odds ratio | Confidence interval | p Value | |
|---|---|---|---|
| Age | 1.041 | 0.994–1.090 | 0.089 |
| Sex | 0.455 | 0.161–1.287 | 0.138 |
| BMI | 1.003 | 0.914–1.102 | 0.944 |
| ASA Score | 2.476 | 1.195–5.129 | 0.015 |
| Diagnosis | 1.679 | 1.009–2.984 | 0.046 |
| Surgeon’s experience | 1,217 | 0.429–3.447 | 0.712 |
| Neck angle | 0.618 | 0.292–1.308 | 0.208 |
Significant value in bold letters
Discussion
The use of cementless femoral short stems in MIS THA has increased in the last years. The rate of PFFs in cementless THA differs considerably, up to 27.8% [19]. Use of femoral short stems is described as decreasing the risk for PFF [19, 20, 24]. Dietrich et al. [19] investigated the rate of PFFs using DAA with short stems (including Fitmore short stem) in 183 THAs and found a significantly reduced rate of PFFs of 1.6% compared with 6.8% in 457 THAs with conventional straight stems. We report a comparable rate of 1.7% for MIS anterolateral approach. The use of short stems leads to reduction fractures of the greater trochanter [19]. Yu et al. [24] investigating the Tri-Lock stem (Depuy, Warsaw, IN, USA) in 103 patients found no intraoperative periprosthetic fracture in the short-stem group, compared with 8.6% with the conventional stem. For MIS anterolateral approach, the rate of greater trochanter fractures is reported to be between 2.5% and 3.2% in nonobese and up to 9.7% in obese patients [8–10]. Our results show a low rate of 0.3% PFFs of the greater trochanter. Fitmore hip stem can be inserted initially in a more varus oriented position, following a C-shaped path out of varus until it is ultimately oriented with the mechanical axis of the diaphysis [22]. Therefore, we postulate that short curved stems in MIS anterolateral approach reduce the pressure on the greater trochanter, leading to a reduced rate of fractures of the greater trochanter.
Age is also described as a risk factor for PFF, with a risk 2.9 times higher in patients over 70 years for all approaches [25]. Also, in DAA as well as MIS anterolateral approach an increased risk in older patients is described [10, 26, 27]. Berend et al. [26] report age as a risk factor for PFFs in DAA, with the average age of patients with fractures being 72 years compared with 63 years without fractures. Herndon et al. [10] did not find a significance for age (p = 0.13) but postulated age being a risk factor, because of a higher average age in the fracture group, with 69.2 years compared with 66.6 years in the nonfracture group. Gkagkalis et al. [11] report a similar rate for intraoperative fissures in short-stem arthroplasty in elderly patients (over 75 years) compared with a younger control group (< 60 years), with 1.5% and 1.4%, respectively. Our results show an average age in patients with fracture with 71.78 years compared with 67.23 years in patients without fracture. Age in patients with PFF is on average 4.55 years higher. However, we could not find a statistical significance (p = 0.161).
PFF occurred more often in female patients, with 13 PFFs compared with 5 PFFs in male patients. However, testing did not show significance (p = 0.155). Logistic regression found an OR of 0.455 (CI 0.161–1.287; p = 0.138). The odds ratio indicates a lower risk for occurrence of PFFs in male patients, but without statistical significance. Female sex is also associated with higher risk for PFFs [10, 27, 28]. The results in the presented study could not support these findings. We report a consecutive case series with over 1000 implantations with findings contrary to comparable studies [10, 27]. Sheth et al. [28] report an increased OR of 2.74 (CI 1.28–5.89; p = 0.01) for female sex in 5313 primary THAs with a high number of different stem designs and fixation. We report a higher number of PFFs occurring in female patients and a lower OR for male patients but cannot support these findings with statistical significance. Therefore, female sex could not be fully proven as a risk factor for PFF in cementless short-stem THA.
The presented study reports a revision rate of 0.7%, with 38.89% of all PFFs needing revision arthroplasty. Herndon et al. [10] report a revision rate of 2.0%, with 48,3% of all PFFs needing revision arthroplasty. Sheth et al. [28] report a revision rate of 100% of all 32 PFFs that occurred within 90 days after index surgery. Watts et al. [29] report revision arthroplasty in 50% and 76%, respectively, for two different stem designs, while Gromov et al. [30] report revision arthroplasty in 97.0% of all PFFs. In our study, PFFs could be fixed using a standard straight stem or its monoblock revision straight stem derivate in the case of revision arthroplasty. These findings indicate a major advantage of cementless short stems of preserving more bone stock, and a different fracture type in the case of PFF occurrence resulting in easier revision arthroplasty.
The type of fixation is also discussed broadly regarding the occurrence of PFFs. Cemented femoral components are associated with a significantly lower rate of PFFs in direct comparison with cementless stems [5, 28, 31]. However, indication for THA is inhomogeneous. In particular, cementless fixation in femoral neck fracture is associated with an increased rate of PFFs of up to 7.4% [31, 32], but yields better functional scores [31]. Fitmore short stem shows excellent outcome regarding aseptic loosening, with 99.6% survival rate after 8.6 years and excellent functional outcome with an average Harris Hip Score between 96 and 98 points after an average follow-up of 7.7 years [33]. Our results show a low number of PFFs without any statistical significance for age and sex. Therefore, cementless short-stem THA using minimally invasive approaches yields excellent functional outcome combined with a significantly reduced rate of PFFs compared with cementless straight-stem THA.
Some limitations need to be noted. Although this is a retrospective study of consecutive patients, selection bias cannot be excluded. Another limitation is the limited follow-up period of 90 days within index surgery, which is due to the retrospective study design. A longer follow-up period of 1 year might have resulted in a significantly higher number of patients lost to follow-up and might have impaired the number of included patients immensely. Therefore, we favored a significantly higher number of patients and implantations with a shorter follow-up period to prevent a high number of patients being lost to follow-up.
Conclusion
Cementless total hip arthroplasty with a curved short stem using the minimally invasive anterolateral approach shows a low number of intra- and postoperative periprosthetic fractures within 90 days from index surgery. Fracture rates are comparable to other minimally invasive approaches and other comparable femoral short stems. Age and sex are not associated with a higher risk for periprosthetic fractures.
Acknowledgements
Supported by Johannes Kepler Open Access Publishing Fund.
Authors’ contributions
ML: wrote the manuscript, performed the statistical analysis, designed the study, acquired data, and interpreted the data. GH: jointly conceived the study, interpreted the data. CS: involved in the acquisition of and interpretation of the data. BS: revised the manuscript. RH: revised the manuscript. JA: involved in the acquisition and interpretation of the data. TG: revised the manuscript. AK: jointly conceived the study, performed statistical analysis, and edited the manuscript. All authors read and approved the final manuscript.
Funding
The study was conducted without any funding or benefits from a commercial party. Three coauthors have received or will receive benefits for personal or professional use from a commercial party outside the conduction of this study.
Availability of data and materials
Data and materials are available on request.
Declarations
Ethics approval and consent to participate
This study received ethical approval from the local institutional review board of the “Ethikkommission OÖ” of the Johannes Kepler University Linz (JKU Linz) (EK-No.: 1239/2019).
Consent to participate
The study was approved by the institutional review board (EK-No.: 1239/2019) in accordance with the World Medical Association Declaration of Helsinki. Because of the retrospective evaluation of preexisting medical records, informed consent was not required.
Competing interests
One coauthor (R.H) received consultant honoraria of ZimmerBiomet, Europe, outside the submitted work. We report personal fees paid to one coauthor (T.G.) during the conduct of the study from Zimmer Biomet, Europe, and from Depuy Synthes Orthopädie Gmbh, Peter Brehm GmbH, ImplanTec GmbH, outside the submitted work. We report research grants paid to our institution during the conduct of the study from Zimmer Biomet, Europe, Mathys AG Switzerland, Anika Therapeutics outside the submitted work.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Matthias Luger, Email: Matthias.luger@kepleruniklinikum.at.
Günter Hipmair, Email: Guenter.hipmair@kepleruniklinikum.at.
Clemens Schopper, Email: Clemens.schopper@kepleruniklinikum.at.
Bernhard Schauer, Email: Bernhard.schauer@kepleruniklinikum.at.
Rainer Hochgatterer, Email: Rainer.hochgatterer@kepleruniklinikum.at.
Jakob Allerstorfer, Email: Jakob.allerstorfer@kepleruniklinikum.at.
Tobias Gotterbarm, Email: Tobias.gotterbarm@kepleruniklinikum.at.
Antonio Klasan, Email: Klasan.antonio@me.com.
References
- 1.Young SW, Walker CG, Pitto RP. Functional outcome of femoral peri prosthetic fracture and revision hip arthroplasty: a matched-pair study from the New Zealand Registry. Acta Orthop. 2008;79(4):483–488. doi: 10.1080/17453670710015463. [DOI] [PubMed] [Google Scholar]
- 2.Bhattacharyya T, Chang D, Meigs JB, Estok DM, 2nd, Malchau H. Mortality after periprosthetic fracture of the femur. J Bone Joint Surg Am. 2007;89(12):2658–2662. doi: 10.2106/JBJS.F.01538. [DOI] [PubMed] [Google Scholar]
- 3.Vioreanu MH, Parry MC, Haddad FS, Duncan CP. Field testing the Unified Classification System for peri-prosthetic fractures of the pelvis and femur around a total hip replacement : an international collaboration. Bone Joint J. 2014;96(11):1472–1477. doi: 10.1302/0301-620x.96b11.34214. [DOI] [PubMed] [Google Scholar]
- 4.da Assuncao RE, Pollard TC, Hrycaiczuk A, Curry J, Glyn-Jones S, Taylor A. Revision arthroplasty for periprosthetic femoral fracture using an uncemented modular tapered conical stem. Bone Joint J. 2015;97(8):1031–1037. doi: 10.1302/0301-620X.97B8.34431. [DOI] [PubMed] [Google Scholar]
- 5.Carli AV, Negus JJ, Haddad FS. Periprosthetic femoral fractures and trying to avoid them: what is the contribution of femoral component design to the increased risk of periprosthetic femoral fracture? Bone Joint J. 2017;99((1 Supple A)):50–59. doi: 10.1302/0301-620X.99B1.BJJ-2016-0220.R1. [DOI] [PubMed] [Google Scholar]
- 6.Yasen AT, Haddad FS. Periprosthetic fractures: bespoke solutions. Bone Joint J. 2014;96((11 Supple A)):48–55. doi: 10.1302/0301-620X.96B11.34300. [DOI] [PubMed] [Google Scholar]
- 7.Pivec R, Issa K, Kapadia BH, Cherian JJ, Maheshwari AV, Bonutti PM, Mont MA. Incidence and future projections of periprosthetic femoral fracture following primary total hip arthroplasty: an analysis of international registry data. J Long Term Eff Med Implants. 2015;25(4):269–275. doi: 10.1615/jlongtermeffmedimplants.2015012625. [DOI] [PubMed] [Google Scholar]
- 8.Nakai T, Liu N, Fudo K, Mohri T, Kakiuchi M. Early complications of primary total hip arthroplasty in the supine position with a modified Watson-Jones anterolateral approach. J Orthop. 2014;11(4):166–169. doi: 10.1016/j.jor.2014.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Iwata H, Sakata K, Sogo E, Nanno K, Kuroda S, Nakai T. Total hip arthroplasty via an anterolateral supine approach for obese patients increases the risk of greater trochanteric fracture. J Orthop. 2018;15(2):379–383. doi: 10.1016/j.jor.2018.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Herndon CL, Nowell JA, Sarpong NO, Cooper HJ, Shah RP, Geller JA. Risk factors for periprosthetic femur fracture and influence of femoral fixation using the mini-anterolateral approach in primary total hip arthroplasty. J Arthroplasty. 2020;35(3):774–778. doi: 10.1016/j.arth.2019.10.011. [DOI] [PubMed] [Google Scholar]
- 11.Gkagkalis G, Goetti P, Mai S, Meinecke I, Helmy N, Bosson D, Kutzner KP. Cementless short-stem total hip arthroplasty in the elderly patient—is it a safe option?: a prospective multicentre observational study. BMC Geriatr. 2019;19(1):112. doi: 10.1186/s12877-019-1123-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wu CC, Au MK, Wu SS, Lin LC. Risk factors for postoperative femoral fracture in cementless hip arthroplasty. J Formos Med Assoc. 1999;98(3):190–194. [PubMed] [Google Scholar]
- 13.Rogmark C, Leonardsson O. Hip arthroplasty for the treatment of displaced fractures of the femoral neck in elderly patients. Bone Joint J. 2016;98(3):291–297. doi: 10.1302/0301-620X.98B3.36515. [DOI] [PubMed] [Google Scholar]
- 14.Bonnin MP, Neto CC, Aitsiselmi T, Murphy CG, Bossard N, Roche S. Increased incidence of femoral fractures in small femurs and women undergoing uncemented total hip arthroplasty—why? Bone Joint J. 2015;97(6):741–748. doi: 10.1302/0301-620X.97B6.35022. [DOI] [PubMed] [Google Scholar]
- 15.Berend ME, Smith A, Meding JB, Ritter MA, Lynch T, Davis K. Long-term outcome and risk factors of proximal femoral fracture in uncemented and cemented total hip arthroplasty in 2551 hips. J Arthroplasty. 2006;21(6 Suppl 2):53–59. doi: 10.1016/j.arth.2006.05.014. [DOI] [PubMed] [Google Scholar]
- 16.Schwartz JT, Jr, Mayer JG, Engh CA. Femoral fracture during non-cemented total hip arthroplasty. J Bone Joint Surg Am. 1989;71(8):1135–1142. doi: 10.2106/00004623-198971080-00003. [DOI] [PubMed] [Google Scholar]
- 17.Singh JA, Jensen MR, Harmsen SW, Lewallen DG. Are gender, comorbidity, and obesity risk factors for postoperative periprosthetic fractures after primary total hip arthroplasty? J Arthroplasty. 2013;28(1):126–131e121-122. doi: 10.1016/j.arth.2012.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lindahl H. Epidemiology of periprosthetic femur fracture around a total hip arthroplasty. Injury. 2007;38(6):651–654. doi: 10.1016/j.injury.2007.02.048. [DOI] [PubMed] [Google Scholar]
- 19.Dietrich M, Kabelitz M, Dora C, Zingg PO. Perioperative fractures in cementless total hip arthroplasty using the direct anterior minimally invasive approach: reduced risk with short stems. J Arthroplasty. 2018;33(2):548–554. doi: 10.1016/j.arth.2017.09.015. [DOI] [PubMed] [Google Scholar]
- 20.Molli RG, Lombardi AV, Jr, Berend KR, Adams JB, Sneller MA. A short tapered stem reduces intraoperative complications in primary total hip arthroplasty. Clin Orthop Relat Res. 2012;470(2):450–461. doi: 10.1007/s11999-011-2068-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pfeil J. Minimally invasive surgery in total hip arthroplasty. Berlin: Springer; 2010. [Google Scholar]
- 22.Gustke K. Short stems for total hip arthroplasty: initial experience with the Fitmore stem. J Bone Joint Surg Br. 2012;94((11 Suppl A)):47–51. doi: 10.1302/0301-620X.94B11.30677. [DOI] [PubMed] [Google Scholar]
- 23.Hoenig JM, Heisey DM. The abuse of power. Am Stat. 2001;55(1):19–24. doi: 10.1198/000313001300339897. [DOI] [Google Scholar]
- 24.Yu H, Liu H, Jia M, Hu Y, Zhang Y. A comparison of a short versus a conventional femoral cementless stem in total hip arthroplasty in patients 70 years and older. J Orthop Surg Res. 2016;11:33. doi: 10.1186/s13018-016-0367-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cook RE, Jenkins PJ, Walmsley PJ, Patton JT, Robinson CM. Risk factors for periprosthetic fractures of the hip: a survivorship analysis. Clin Orthop Relat Res. 2008;466(7):1652–1656. doi: 10.1007/s11999-008-0289-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Berend KR, Mirza AJ, Morris MJ, Lombardi AV., Jr Risk of periprosthetic fractures with direct anterior primary total hip arthroplasty. J Arthroplasty. 2016;31(10):2295–2298. doi: 10.1016/j.arth.2016.03.007. [DOI] [PubMed] [Google Scholar]
- 27.Hartford JM, Knowles SB. Risk factors for perioperative femoral fractures: cementless femoral implants and the direct anterior approach using a fracture table. J Arthroplasty. 2016;31(9):2013–2018. doi: 10.1016/j.arth.2016.02.045. [DOI] [PubMed] [Google Scholar]
- 28.Sheth NP, Brown NM, Moric M, Berger RA, Della Valle CJ. Operative treatment of early peri-prosthetic femur fractures following primary total hip arthroplasty. J Arthroplasty. 2013;28(2):286–291. doi: 10.1016/j.arth.2012.06.003. [DOI] [PubMed] [Google Scholar]
- 29.Watts CD, Abdel MP, Lewallen DG, Berry DJ, Hanssen AD. Increased risk of periprosthetic femur fractures associated with a unique cementless stem design. Clin Orthop Relat Res. 2015;473(6):2045–2053. doi: 10.1007/s11999-014-4077-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gromov K, Bersang A, Nielsen CS, Kallemose T, Husted H, Troelsen A. Risk factors for post-operative periprosthetic fractures following primary total hip arthroplasty with a proximally coated double-tapered cementless femoral component. Bone Joint J. 2017;99(4):451–457. doi: 10.1302/0301-620x.99b4.bjj-2016-0266.r2. [DOI] [PubMed] [Google Scholar]
- 31.Langslet E, Frihagen F, Opland V, Madsen JE, Nordsletten L, Figved W. Cemented versus uncemented hemiarthroplasty for displaced femoral neck fractures: 5-year followup of a randomized trial. Clin Orthop Relat Res. 2014;472(4):1291–1299. doi: 10.1007/s11999-013-3308-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Inngul C, Blomfeldt R, Ponzer S, Enocson A. Cemented versus uncemented arthroplasty in patients with a displaced fracture of the femoral neck: a randomised controlled trial. Bone Joint J. 2015;97(11):1475–1480. doi: 10.1302/0301-620X.97B11.36248. [DOI] [PubMed] [Google Scholar]
- 33.Innmann MM, Weishorn J, Bruckner T, Streit MR, Walker T, Gotterbarm T, Merle C, Maier MW. Fifty-six percent of proximal femoral cortical hypertrophies 6 to 10 years after Total hip arthroplasty with a short Cementless curved hip stem—a cause for concern? BMC Musculoskelet Disord. 2019;20(1):261. doi: 10.1186/s12891-019-2645-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data and materials are available on request.