Abstract
Monopolar spindle 1 (MPS1), which plays a critical role in somatic mitosis, has also been revealed to be essential for meiosis I in oocytes. Spermatogenesis is an important process involving successive mitosis and meiosis, but the function of MPS1 in spermatogenesis remains unclear. Here, we generated Mps1 conditional knockout mice and found that Ddx4-cre-driven loss of Mps1 in male mice resulted in depletion of undifferentiated spermatogonial cells and subsequently of differentiated spermatogonia and spermatocytes. In addition, Stra8-cre-driven ablation of Mps1 in male mice led to germ cell loss and fertility reduction. Spermatocytes lacking Mps1 have blocked at the zygotene-to-pachytene transition in the prophase of meiosis I, which may be due to decreased H2B ubiquitination level mediated by MDM2. And the expression of many meiotic genes was decreased, while that of apoptotic genes was increased. Moreover, we also detected increased apoptosis in spermatocytes with Mps1 knockout, which may have been the reason why germ cells were lost. Taken together, our findings indicate that MPS1 is required for mitosis of gonocytes and spermatogonia, differentiation of undifferentiated spermatogonia, and progression of meiosis I in spermatocytes.
Subject terms: Cell division, Spermatogenesis
Introduction
Spermatogenesis occurs within the testis seminiferous epithelium and consists of the following stages in mice: mitosis of spermatogonia, meiosis of spermatocytes, and maturation of haploid spermatozoa1. Initially, spermatogonial stem cells undergo a series of mitosis events and then differentiate into B-type spermatogonia. They then enter meiosis I, becoming primary spermatocytes, as indicated by STRA8 expression2–4. Meiosis I involves the stages leptotene, zygotene, pachytene, diplotene, and diakinesis, which can be distinguished by different expression patterns of SYCP35,6. Moreover, at the leptotene stage, double-strand breaks occur and activate γH2AX and SPO117,8, while at zygonema, RAD51 is highly enriched at the break sites in the chromosome strands9. During the pachytene stage, meiotic homologous chromosomes combine and form the synaptonemal complex. SYCP3 localizes along the lateral chromosome axis, and SYCP1 loads onto the center to connect the two homologous chromosomes5,6,9. Subsequently, diplonema occurs, which features sex bodies marked by γH2AX10, and is followed by diakinesis.
Monopolar spindle 1 (MPS1) is a kinase that is conserved from yeast to mammals that plays a critical role in preventing erroneous chromosome segregation through the spindle assembly checkpoint (SAC) complex11–13. During mitosis, MPS1 directly binds to unattached kinetochores through CDC80, after which it phosphorylates and recruits other SAC proteins, such as BUB3, BUBR1, and MAD2, to the kinetochore14–17. This process inhibits anaphase-promoting complex/cyclosome (APC/C) activation until all microtubules stably attach to the kinetochores at anaphase onset18. MPS1 has important functions not only in mitosis but also in meiosis19. In previous research, a mutant mouse strain was generated that expresses a form of ΔNMPS1 with a 107-amino acid deletion at the N-terminus. The N-terminus is involved in binding to the kinetochore20,21. Without MPS1 localization, precocious APC/C activation leads to the missegregation or nondisjunction events observed in mouse oocytes due to failure of SAC control and chromosome alignment in meiosis I20. Furthermore, MPS1 kinase activation is essential for cohesin protection in a manner dependent on the localization of SGO2 to the centromere in mouse oocytes22. However, whether Mps1 plays an essential role in mouse spermatogenesis, especially in mitosis and male meiosis, is still unknown.
To investigate the function of MPS1 in the regulation of spermatogenesis, we generated two conditional Mps1 knockout strains and found that Mps1 is required for mitosis of gonocytes and spermatogonia, differentiation of undifferentiated spermatogonia, and progression of meiosis I in spermatocytes.
Results
Mps1 was required for postnatal mitotic divisions of male germ cells
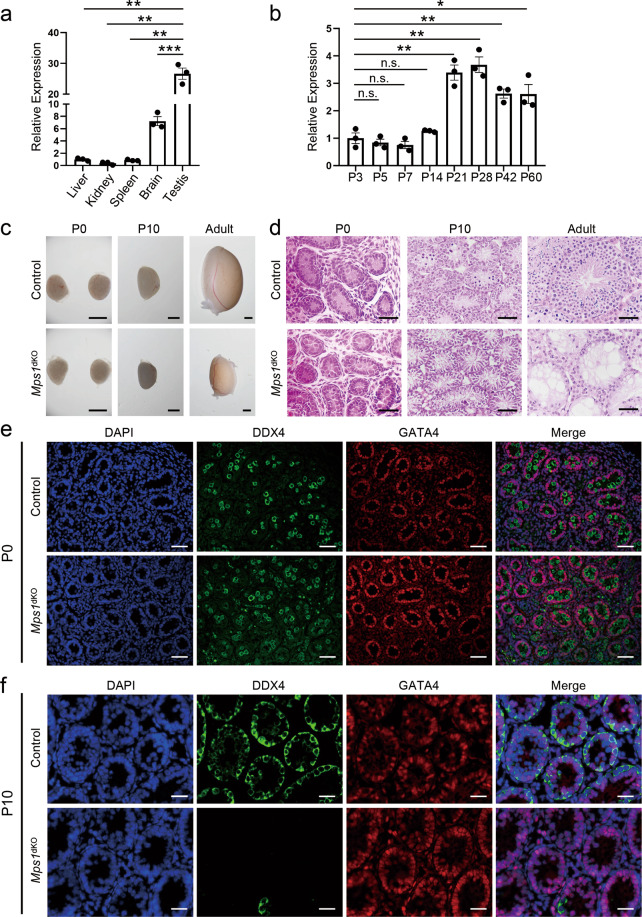
To explore Mps1’s function in spermatogenesis, we first characterized the spatiotemporal expression pattern of Mps1 in mice. Quantitative polymerase chain reaction (qPCR) analysis of different adult tissues showed higher Mps1 expression levels in testes than in other tissues (Fig. 1a). In testes, Mps1 expression levels in the mouse testes aged from P3 (postnatal day 3) to P60 showed a dramatic increase from P21, when the male mice started to sexually mature (Fig. 1b). Using the published testes RNA-seq dataset from Soumillon et al.23, we further found that Mps1 is especially enriched in spermatocytes (Fig. S1a). All these data suggested that Mps1 may have important roles in spermatogenesis.
Fig. 1. Ablation of Mps1 in gonocytes and spermatogonia led to the depletion of germ cells.
a qPCR analysis of Mps1 expression level of liver, kidney, spleen, brain, and testis of wild-type mice at 2 month of age. n = 3; Student’s t test; **P < 0.01, ***P < 0.001. b qPCR analysis of Mps1 expression level of wildtype mouse testes from P3 to P60. n = 3; Student’s t test; n.s. no significance, *P < 0.05, **P < 0.01. c Images of control and Mps1dKO testes at P0, at P10, and during adulthood. Scale bar = 1 mm. d H&E staining of control and Mps1dKO testes at P0, at P10, and during adulthood. Scale bar = 50 µm. e Immunofluorescence staining of DDX4 and GATA4 in control and Mps1dKO testes at P0. Scale bar = 50 µm. f Immunofluorescence staining of DDX4 and GATA4 in control and Mps1dKO testes at P10. Scale bar = 50 µm.
For studying the role of Mps1, we generated a conditional Mps1-knockout mouse strain. Specifically, we first generated Mps1fl/fl mice by inserting two LoxP sites in intron 5 and intron 8 of the Mps1 gene, which caused deletion of exons 6–8 and a frameshift mutation driven by Cre recombinase (Fig. S1b). Then, by mating Mps1 floxed mice with Ddx4-Cre mice, which specifically express Cre recombinase in germ cells as early as embryonic day 15.5 (E15.5)24, we obtained germ cell-specific Mps1-knockout (Mps1dKO) mice. Successful knockout was verified by PCR (Fig. S1c, d).
The Mps1dKO mice grew normally like the control mice, and we focused on the spermatogenesis of male mice. First, we examined the morphology of testes from newborn (P0) to adult mice (Fig. 1c). There was no difference between Mps1dKO and control mice in terms of testis size at P0. At P10, the testes of Mps1dKO and control mice started to become different in size, with the testes of Mps1dKO mice being slightly smaller than those of control mice. The difference became much more significant in adult mice. Then, to determine the reason, we assessed the composition of cell types in seminiferous tubules, which are the sites of spermatogenesis (where germ cells develop into spermatozoa). Using hematoxylin–eosin (H&E) staining, we were able to distinguish two different types of cells on the basis of morphology, i.e., triangle-like Sertoli cells and round germ cells. At P0, both types of cells were visible in Mps1dKO and control seminiferous tubules (Fig. 1d). However, there were few germ cells (round-shaped) left in the seminiferous tubules at P10, and no germ cells were present in adult Mps1dKO mice; only triangle-like Sertoli cells remained (Fig. 1d). Moreover, many empty seminiferous tubules existed in adult Mps1dKO mice. Consistently, no mature sperm were found in the cauda epididymides of Mps1dKO mice (Fig. S1e).
To further identify the cell types that disappeared, we employed immunostaining on seminiferous tubule sections of P0 and P10 mice. We used two kinds of marker genes to distinguish cell types: GATA4 for Sertoli cells and DDX4 for germ cells. Consistent with the H&E staining results, DDX4-positive germ cells and GATA4-positive Sertoli cells were present in both control and Mps1dKO P0 mice (Fig. 1e). However, almost no DDX4-positive germ cells were found in the Mps1dKO P10 testes (Fig. 1f). Nonetheless, Sertoli cells (GATA4-positive) remained unaffected in the Mps1dKO testes, as expected. Taken together, the results indicate that Mps1 knockout in early-stage male germ cells results in severe germ cell loss. Previous studies have revealed the important roles of MPS1 in mitosis in somatic cells. Given that germ cells are generated via mitosis of gonocytes and spermatogonia, the findings suggest that the male germ cell depletion in Mps1dKO mice might be due to disruption of postnatal mitosis.
Postnatal disruption of Mps1 led to severe defects in spermatogenesis and significantly reduced male fertility
Since knocking out Mps1 in Mps1dKO mouse testes triggered such dramatic germ cell loss that almost no spermatocytes could be observed, the role of MPS1 in the meiosis of spermatocytes remained unclear. To investigate the function of MPS1 in meiosis, we knocked out the Mps1 gene in germ cells by mating Stra8-cre mice, which specifically express Cre recombinase at P3 (early spermatogonia) and exhibit increased expression until P7 (preleptotene spermatocytes) in testes25. This strain (Mps1sKO) was verified by PCR (Fig. S2a). The knockout efficiency of Mps1 was confirmed by using Western blotting, which revealed that the protein levels of MPS1 were significantly reduced in the testes of the Mps1sKO mice (Fig. S2b).
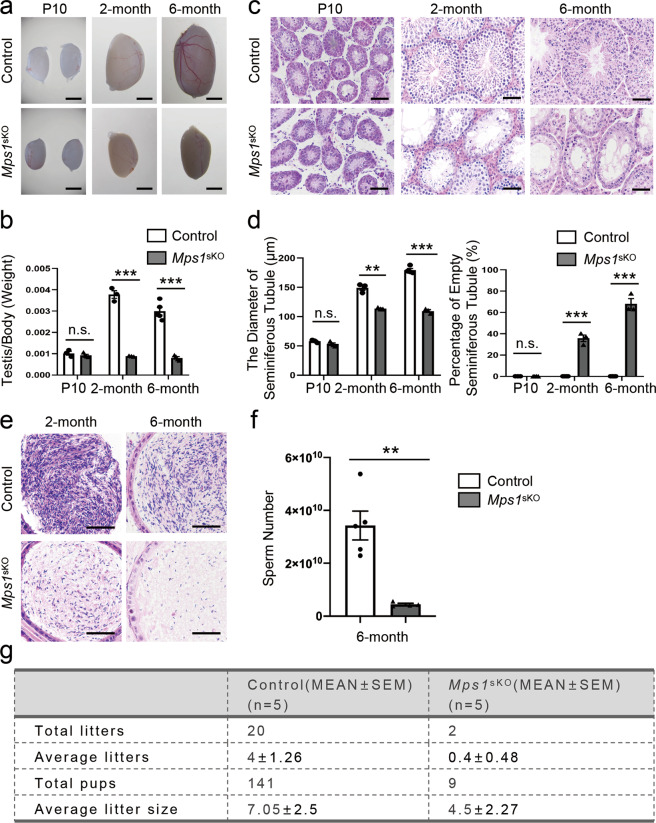
As we observed, the Mps1sKO mice were mostly normal, but their testes were different from those of the control mice. The testes of Mps1sKO and control mice were similar in size and mass at P10, but the testes of Mps1sKO mice became smaller and lighter than those of control mice at 2 months and 6 months (Fig. 2a, b). Specifically, the testis weight/body weight ratio was significantly reduced from 0.37% in control mice to 0.12% in Mps1sKO mice at 2 month (32.4%) and from 0.30% to 0.08% at 6 month (28.3%) (Fig. 2b). In addition, the epididymides were detectably smaller in Mps1sKO mice than in control mice (Fig. S2c). Histologically, the seminiferous tubules of Mps1sKO mice displayed decreased diameters, reduced cell numbers, and increased empty seminiferous tubule percentages at 2 and 6 months (Fig. 2c, d). The average diameter of Mps1sKO seminiferous tubules was reduced to 76% and 61% of that of the control mice at 2 and 6 months, respectively (Fig. 2d). At 2 months and 6 months, 35% and 68% of Mps1sKO seminiferous tubules were degenerated with aberrant spermatogenesis, respectively, while degenerated tubules were hardly detected in the control mice in the time window we examined (Fig. 2d).
Fig. 2. Postnatal disruption of Mps1 led to severe defects in spermatogenesis and significantly reduced male fertility.
a Image of control and Mps1sKO testes at P10, 2 months of age, and 6 months of age. Scale bar = 2 mm. b Ratios of testis weight to body weight for control and Mps1sKO mice at P10, 2 months of age, and 6 months of age. n ≥ 3; Student’s t test; n.s. no significance; ***P < 0.001. c H&E staining of control and Mps1sKO testes at P10, 2 months of age and 6 months of age. Scale bar = 50 µm. d Statistical results for the seminiferous tubule diameters and empty seminiferous tubule percentages of control and Mps1sKO testes at P10, 2 months of age, and 6 months of age. At least 100 tubules were counted from at least 3 different mice; Student’s t test; **P < 0.01, ***P < 0.001. e H&E staining of control and Mps1sKO cauda epididymides from 2 and 6-month-old mice. Scale bar = 50 µm. f Sperm counts from the cauda epididymides of at least four different mice at 6-month of age. Student’s t test; **P < 0.01. g Fertility test of male control and Mps1sKO mice. In each group, five 3-month-old male mice were individually mated with two wild-type female mice for 3 continuous months.
Next, we examined whether Stra8-Cre-mediated Mps1 ablation affected sperm production and male fertility. H&E staining of the cauda epididymis, which is the primary storage site for mature sperm, showed that the density of mature sperm was largely reduced in adult (2 months and 6 months) Mps1sKO mice compared to control mice (Fig. 2e). To validate this finding, we released and counted all mature sperm from the cauda epididymides of 6-month-old mice. Strikingly, the average sperm number of Mps1sKO mice was decreased to approximately one-tenth of that of control mice (Fig. 2f). We then assessed the fertility of male Mps1sKO and control mice. Three-month-old male control and Mps1sKO mice were bred with two adult wild-type females for 3 continuous months (n = 5). Whereas the control males produced litters of normal size over this period, the Mps1sKO males showed significantly reduced fertility. The total number of litters produced by the control mice was 20 (average number of litters: 4 ± 1.26), and the total number of pups was 141 (average litter size: 7.05 ± 2.5). However, 3 of 5 Mps1sKO mice were infertile during this time period, and another two Mps1sKO mice each produced only one litter (average number of litters: 0.4 ± 0.49); the total pup number dropped to 9 (average litter size: 4.5 ± 2.27) (Fig. 2g). Thus, postnatal deletion of Mps1 led to lower productivity of mature sperm and significantly reduced male fertility, suggesting that the reductions in average litter number and size might have been caused by reductions in sperm numbers in the testes of Mps1sKO males.
Deletion of Mps1 within differentiating spermatogonia via Stra8-Cre revealed a role for Mps1 in spermatogonial differentiation
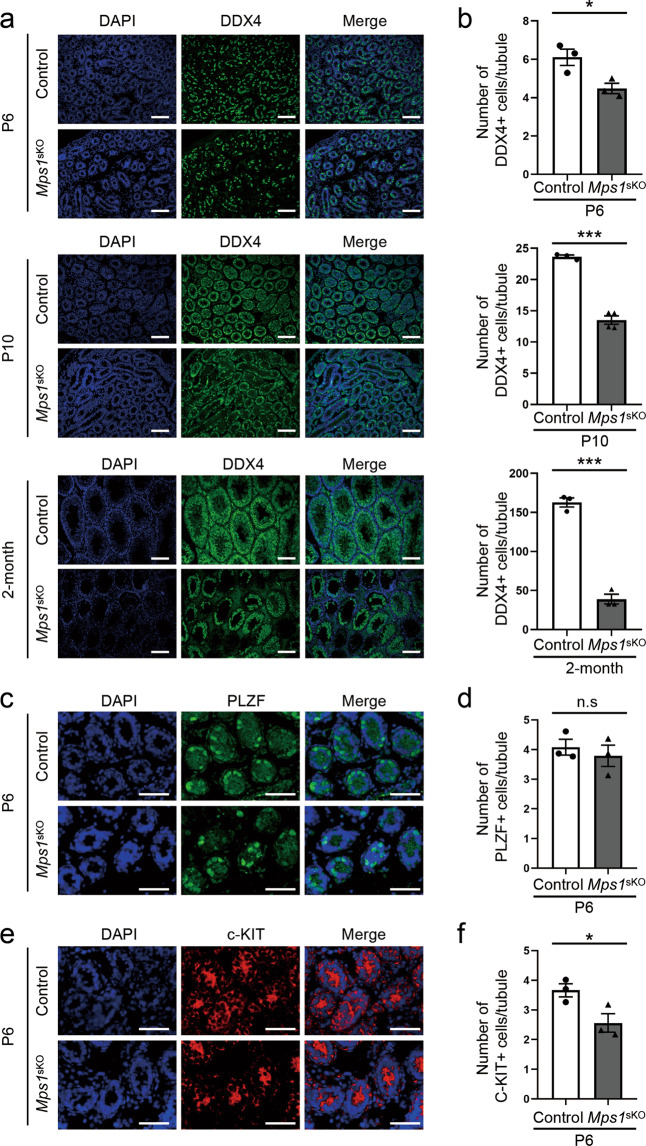
To determine the mechanism of severe cell loss and fertility reduction in Mps1sKO mice, it was necessary to investigate which cell types were lost and when they began to disappear during spermatogenesis. We examined the expression of germ cell markers in testes of mice at P6, P10, and 2 months by immunostaining analysis. The DDX4 staining data showed that the number of Mps1sKO mouse germ cells per seminiferous tubule had decreased to 66% and 57% of that in control mice by P6 and P10, respectively (Fig. 3a, b), although the testis weight did not differ, as previously mentioned (Fig. 2b). The impact was greater in 2-month-old Mps1sKO mice, which retained only 24% of the germ cells in control mice. On the other hand, the number of PLZF-positive cells per tubule, representing undifferentiated spermatogonia, was the same between Mps1sKO mice and controls at different ages, as expected (Fig. 3c, d; Fig. S3a, b). We further investigated the fates of Mps1sKO spermatogonia via immunostaining for c-KIT, a marker of differentiated spermatogonia. The results indicated that the c-KIT-positive cell number in Mps1sKO testes was reduced to 70% of that in control testes at P6, which suggested that spermatogonial differentiation may have been affected in Mps1sKO mice (Fig. 3e, f). In summary, postnatal deletion of Mps1 via Stra8-Cre does not affect the number of undifferentiated spermatogonia but plays a role in spermatogonial differentiation during spermatogenesis.
Fig. 3. Deletion of Mps1 reduced spermatogonial differentiation during spermatogenesis.
a Immunofluorescence staining of DDX4 in testes of control and Mps1sKO mice at P6, P10, and 2 months of age. Scale bar = 50 µm. b Statistical results for the number of DDX4-positive cells per tubule at P6, P10, and 2 months of age. n ≥ 3; Student’s t test; *P < 0.05, ***P < 0.001. c Immunofluorescence staining of PLZF in control and Mps1sKO testes at P6. Scale bar = 50 µm. d Statistical results for the number of PLZF-positive cells per tubule at P6. n ≥ 3; Student’s t test; n.s. no significance. e Immunofluorescence staining of c-KIT in control and Mps1sKO testes at P6. Scale bar = 50 µm. f Statistical results for the number of c-KIT-positive cells per tubule at P6. n ≥ 3; Student’s t test; *P < 0.05.
Loss of Mps1 compromised the progression of meiosis I and delayed the transition from zygotene to pachytene
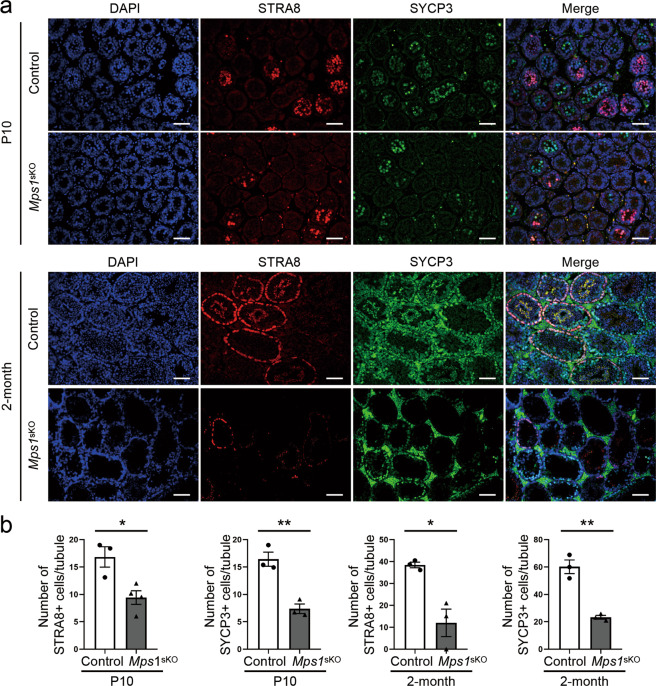
Meiosis is a key feature of spermatogenesis that begins at approximately P10. STRA8 and SYCP3 are a marker of meiosis initiation and a synaptonemal complex component, respectively. Immunostaining analysis showed that the numbers of STRA8-expressing and SYCP3-expressing cells per seminiferous tubule in Mps1sKO mice were significantly reduced to approximately 56% and 45% of those in control mice at P10. Similarly, the staining results at 2 month revealed that Mps1sKO mice had only 30% of the STRA8-positive and 38% of the SYCP3-positive cells of control mice (Fig. 4a, b).
Fig. 4. Loss of Mps1 caused meiosis defects during spermatogenesis.
a Immunofluorescence staining of STRA8 and SYCP3 in control and Mps1sKO testes at P10 and 2 months of age. Scale bar = 50 µm. b Statistical results for the number of STRA8-positive and SYCP3-positive cells per tubule in testes from control and Mps1sKO mice at P10 and 2 months of age. n ≥ 3; *P < 0.05, **P < 0.01.
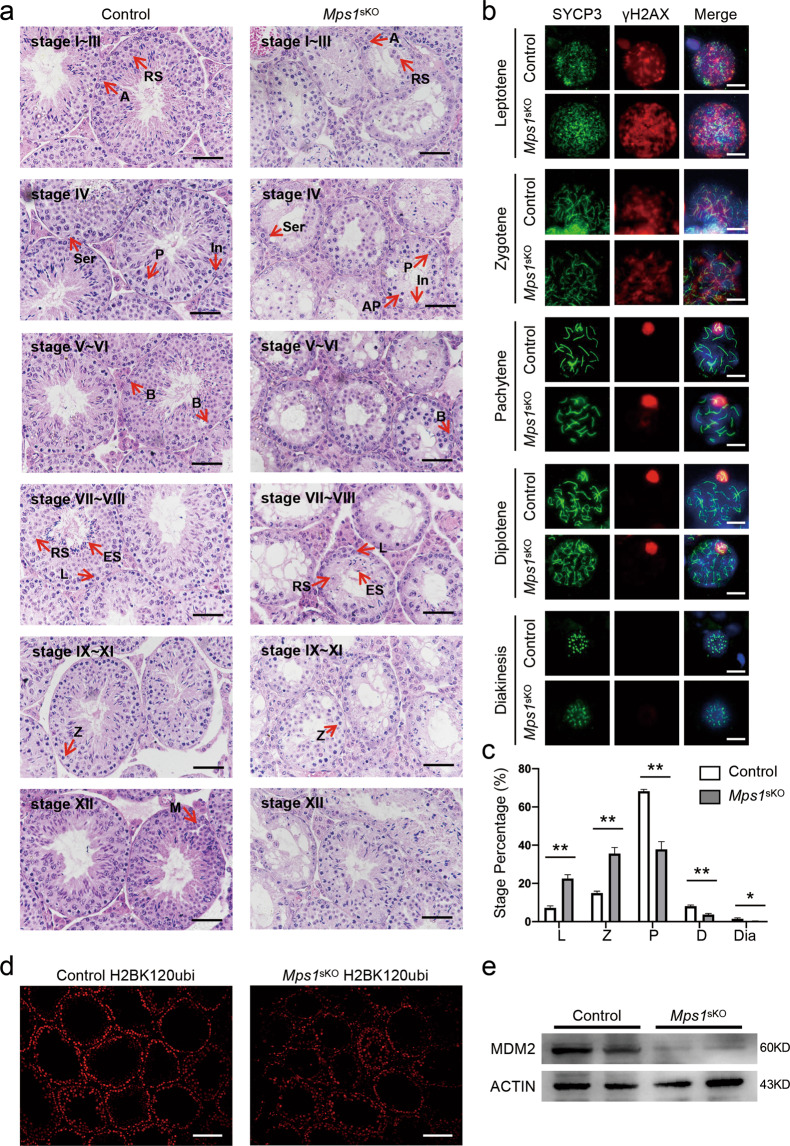
According to the spermatocyte composition in the seminiferous tubules, spermatid development in mice has been divided into 12 stages26,27. To determine the exact stage of meiosis that was affected by Mps1 knockout, we performed H&E staining on testes of 6-month-old mice to trace the seminiferous epithelium stages of the testicular tubules. We found that spermatogenesis seemed delayed at stage IV, during which pachytene spermatocytes should appear and localize inside of the seminiferous lumen. There were fewer pachytene spermatocytes (marked as P) in Mps1-deletion testes than in control testes. Apoptotic pachytenes (marked as AP) were also observed (Fig. 5a). Although some spermatocytes survived in stage IV, fewer round and elongated spermatids formed in Mps1sKO testes than in control testes. In addition, meiotic spermatocytes (marked as M) were rarely observed in stage XII Mps1sKO testes.
Fig. 5. In Mps1sKO mouse testes, the zygotene-to-pachytene transition was delayed, and the progression of meiosis I was compromised.
a Representative H&E staining of control and Mps1sKO seminiferous tubules. A type A spermatogonia, In intermediate spermatogonia, B type B spermatogonia, P pachytene spermatocytes, L leptotene spermatocytes, Z zygotene spermatocytes, RS round spermatids, ES elongated spermatids, M meiotic divisions, AP apoptosis pachytene, Ser Sertoli. Scale bar = 50 µm. b Meiotic spermatocyte spreads for control and Mps1sKO mice at 1-month of age. Co-staining was performed with SYCP3 and γH2AX antibodies. Scale bar = 10 µm. c Percentage of meiotic spermatocytes in each stage. L leptotene, Z zygotene, P pachytene, D diplotene, Dia diakinesis. A total of 650–1000 spermatocytes were counted from 3 different mice. Student’s t test; *P < 0.05, **P < 0.01. d Immunofluorescence staining of H2BK120ubi in control and Mps1sKO testes at 2 months of age. Scale bar = 100 µm. e Western blot of MDM2 in control and Mps1sKO testes at 2 months of age.
To further identify the delayed stages in the prophase of meiosis, meiotic stage progression was assessed from chromosomal spreads of spermatocytes that were prepared from testes of 1-month male mice and immunostained with anti-SYCP3 and anti-γH2AX antibodies (Fig. 5b, c). In both the Mps1sKO and control groups, all five stages from leptotene to diakinesis were observed based on the immunostaining patterns of SYCP3 and γH2AX in spermatocytes. The proportion of pachytene spermatocytes was reduced from 68.25% in control mice to 37.85% in Mps1sKO mice, while the proportions of spermatocytes in diplotene and diakinesis were reduced from 8.08% in control mice to 3.70% in Mps1sKO mice and from 1.56% in control mice to 0.28% in Mps1sKO mice, respectively. On the other hand, spermatocytes of Mps1sKO mice accumulated in leptotene (control mice: 7.24%; Mps1sKO mice: 22.55%) and in zygotene (control mice: 14.97%; Mps1sKO mice: 35.61%). These data indicated that the transition of zygotene spermatocytes to pachytene spermatocytes was impaired. This finding was investigated further via examination of H3 core histone phosphorylation at serine 10 (H3pSer10). This type of phosphorylation is coincident with metaphase progression, and the signal then becomes more intense throughout chromatin during the diakinesis phase and the first meiotic metaphase (MI) in spermatocytes28. We found that the numbers of H3pSer10-positive germ cells were dramatically reduced in Mps1sKO testes (Fig. S4a, b) and that the percentages of H3pSer10-positive metaphase and anaphase spermatocytes were significantly lower in tubule sections from Mps1sKO mice than in those from control mice (Fig. S4b, right). This finding suggested that loss of Mps1 compromised the progression of meiosis I.
Collectively, the evidence indicated that deletion of Mps1 during meiosis resulted in a dramatic decrease in the number of germ cells, which may have been caused by defects in the meiosis process. In particular, the transition from the zygotene to the pachytene stage might have been delayed or partially disrupted in the spermatocytes of Mps1sKO mice.
Previously, Xu et al.6 found H2B ubiquitination regulates the progression of meiosis I by promoting chromatin relaxation, and its E3 ligase Rnf20 knockout mice had similar phenotypes, such as the smaller size of testes, loss of germ cells and spermatocytes arrested at the prophase of meiosis I. We examined the levels of H2BK120 mono-ubiquitination in the testes of control and Mps1sKO mice and noticed that it decreased in Mps1sKO testes at 2 months of age compared with that in control testes (Figs. 5d and S4c). MPS1 has been reported to regulate the level of H2BK120ubi mediated by MDM2, another E3 ubiquitin ligase promoting the ubiquitination of H2B28,29, and knockdown of Mps1 by RNAi diminished MDM2 expression in U2OS cells30. As expected, the expression level of MDM2 was downregulated in Mps1sKO testes, compared to that in control testes (Fig. 5e). In a word, loss of Mps1 compromised the prophase of meiosis I progress which may be due to decreased H2B ubiquitination level mediated by MDM2.
The expression of meiosis-related genes and apoptosis-related genes was altered in the testes of Mps1sKO mice
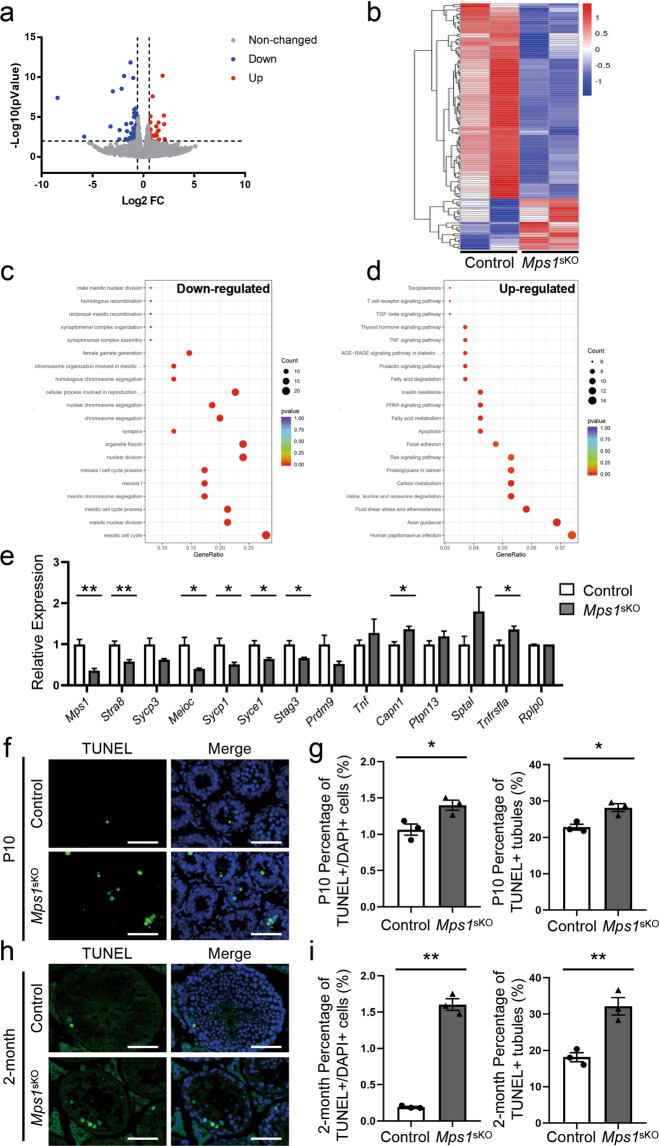
To investigated the changes in gene expression after Mps1 deletion, we performed RNA sequencing of P10 testes from control and Mps1sKO mice. We found that there were more downregulated genes than upregulated genes in Mps1sKO mice (Fig. 6a and Table S2). Using cut-offs of a fold change greater than 1.5 and a P value less than 0.01, 92 genes were identified as downregulated, and 24 genes were identified as upregulated (Fig. 6b). We then performed Gene Ontology (GO) analysis to identify the enriched biological processes for each group of differentially expressed genes (DEGs). Notably, for the downregulated genes, the top ten enriched terms were all related to cell division. More importantly, seven of them were meiosis-related, including the meiotic cell cycle, synapsis, and chromosome segregation terms, among others. The most abundant term, the meiotic cell cycle, was enriched for 21 (22.8%) of the 92 downregulated genes (Fig. 6c and Table S3). To fully discover the biological pathways affected, we relaxed the cut-off for upregulated genes to 1.2-fold, and another 475 genes were identified as upregulated. Of these, 8 out of 189 genes were associated with the apoptosis term. Unsurprisingly, apoptosis stood out as one of the enriched Kyoto Encyclopedia of Genes and Genomes (KEGG) terms for the upregulated genes (Fig. 6d and Table S4). To validate the expression changes in these genes, we examined 13 of them (Mps1, Stra8, Sycp3, Meioc, Sycp1, Syce1, Stag3, Prdm9, Tnf, Capn1, Ptpn13, Sptal, and Tnfrsfla) by qPCR and detected consistent changes in Mps1sKO testes (Fig. 6e). Therefore, the transcriptomic data indicated that widespread impacts on meiosis and apoptosis resulted from Mps1 deletion.
Fig. 6. Loss of Mps1 altered the expression of genes involved in meiosis and caused abnormal apoptosis in seminiferous tubules.
a Gene expression pattern in Mps1sKO testes compared with control testes at P10. An FC > 1.5 and a P value < 0.01 were used as the cut-offs. b Heatmap of the DEGs between control and Mps1sKO testes at P10. In total, 92 genes were downregulated, and 24 genes were upregulated. An FC > 1.5 and a P value < 0.01 were used as the cut-offs. c Top 20 GO terms for the downregulated DEGs. An FC > 1.5 and a P value < 0.01 were used as the cut-offs. d Top 20 KEGG terms for the upregulated DEGs. An FC > 1.2 and a P value < 0.05 were used as the cut-offs. e qPCR analysis of DEGs involved in meiotic biological processes and apoptosis in Mps1sKO testes at P10. Student’s t test; n = 3; *P < 0.05. f Immunofluorescence staining of TUNEL in control and Mps1sKO testes at P10. g Statistical results for the percentage of TUNEL-positive cells among DAPI-positive cells per tubule and the percentage of TUNEL-positive seminiferous tubules in control and Mps1sKO testes at P10. n ≥ 3; *P < 0.05. h Immunofluorescence staining of TUNEL in control and Mps1sKO testes at 2 months of age. i Statistical results for the percentage of TUNEL-positive cells among DAPI-positive cells per tubule and the percentage of TUNEL-positive seminiferous tubules in control and Mps1sKO testes at 2 months of age. n ≥ 3; **P < 0.01.
Mps1 deletion elevated apoptosis in testes
In order to determine the fates of germ cells with Mps1 loss, we conducted a TUNEL assay to investigate whether there were more cells undergoing apoptosis in Mps1sKO testes than in control testes. On the one hand, there were more seminiferous tubules with TUNEL-positive cells in Mps1sKO testes than in control testes (Fig. 6f–i). The percentage of TUNEL-positive tubules increased from 22.8% in control mice to 28.1% in Mps1sKO mice at P10 and from 18.1% to 32.1% at 2 months. On the other hand, the percentage of TUNEL-positive cells per seminiferous tubule was also increased in the Mps1sKO testes (Fig. 6f–i). At P10, on average, 1.06% of the cells in control seminiferous tubules were TUNEL positive, while 1.39% of the cells in Mps1sKO tubules were TUNEL positive. At 2-month of age, the average percentage of TUNEL-positive cells was 0.19% in control tubules, while 1.60% in Mps1sKO tubules. Overall, the TUNEL assay results showed that many more germ cells in seminiferous tubules underwent apoptosis in Mps1sKO mice than in control mice, which indicated that the significant reductions in male germ cell numbers in Mps1sKO mice were partly due to increased apoptosis.
Discussion
MPS1, also known as TTK protein kinase, has been well studied in the context of mitosis in somatic cells14,31 and has also been reported to function in meiosis I during oogenesis20. Nevertheless, it remains unexplored whether Mps1 plays roles in male spermatogenesis. Here, we explored MPS1 function in mouse spermatogenesis for the first time by utilizing conditional Mps1 knockout in DDX4-positive and STRA8-positive germ cells and demonstrated that MPS1 is critical for spermatogonial differentiation and for the progression of meiosis I spermatocytes. We also revealed that Mps1 may play its role partially by promoting male germ cell apoptosis.
STRA8 expression begins at P3 in the early stage of spermatogonia and lasts through the preleptotene stage25. Stra8-Cre mice have been used to investigate gene functions during male meiosis. In our study, we deleted Mps1 in STRA8-positive male germ cells and revealed its function in the meiotic cell cycle. However, we also detected germ cell loss at P6 in Mps1sKO seminiferous tubules. Germ cells at this stage included predominantly undifferentiated and differentiated spermatogonial cells that had not entered the meiotic process. Therefore, Stra8-Cre Mps1 knockout affected not only meiosis but also the differentiation of spermatogonial cells. These results suggest that a spermatocyte-specific Cre knock-in mouse line may be more suitable than the knockout mouse line in the current study for research focusing exclusively on meiosis.
Apoptosis is a major form of programmed cell death critical for development and the damage response32. Activation of the TNF pathway can induce distinct cellular outcomes, including cell death, inflammation, and cell survival, in a context-dependent manner33. In our study, the TNF signaling pathway was upregulated in Mps1sKO testes, which might have contributed to the increased apoptosis in Mps1sKO testes. Given that loss of Mps1 compromised the progression of meiosis I and increased apoptosis in seminiferous tubules, it seems likely that some germ cells that fail to complete meiosis I may undergo cell death in the absence of MPS1 expression.
Previous studies on Mps1 function in cell division have mainly illustrated its function in the SAC. Hached and his colleagues revealed that Mps1 functions in SAC control and chromosome segregation in oocyte meiosis I20. Our data demonstrate Mps1’s role in the prophase of the meiotic cell cycle, showing that Mps1 ablation in this stage results in blockade of the zygotene-to-pachytene transition, which may be due to decreased H2B ubiquitination level mediated by MDM2. We also found that the percentages of H3pSer10-positive metaphase and anaphase spermatocytes were significantly decreased in tubule sections from Mps1sKO mice, which suggests that Mps1 may play a role in the progression from prophase to metaphase and anaphase. Further studies investigating the underlying mechanisms by which MPS1 plays roles in SAC control and chromosome segregation during male meiosis I are needed to complete our knowledge of MPS1 function in male germ cells.
Materials and methods
Animals
Mps1 floxed conditional knockout mice were generated by targeted homologous recombination (Shanghai Model Organisms). Homology arms, LoxP sites, and selection markers were introduced into the targeting vector via a recombineering-based method. BAC DNA was purchased from the Sanger Institute. Then, the linearized vector was electroporated into ESCs and screened with G418 and Ganc. The correct clones were then injected into C57BL/6J blastocysts. After mating and identification by PCR with the primers listed below, we obtained Mps1-floxed mice. All mice and samples were selected randomly and proceeded in arbitrary order. No formal randomization techniques were used. All mice used and samples used in IF, H&E, sequencing analysis were only labeled with mouse ID numbers and no genotypes or treatments were indicated. Genotypes and treatments were labeled after the completion of data acquisition and analysis. The mice were handled according to the university guidelines, and all animal procedures were approved by the Tongji University Institutional Animal Care and Use Program Advisory Committee (protocol number: IACUC-017-006).
Genotyping primers
The primers used to identify the Mps1 floxed genotypes were Mps1-For (forward primer) and Mps1-Re (reverse primer). The floxed band was 353 bp, and the wild-type band was 167 bp. The primers used to identify the Mps1 deletion alleles were Mps1-min-For and Mps1-Re, which produced a 319 bp band. The band for Ddx4 was 240 bp, and that for Stra8-Cre was 180 bp. The primers for qPCR are listed in Table S1.
Mps1-For: TTGCTTGGTAGTTCTGTGGACT
Mps1-Re: AGCAGCTGTAAGTGCAGGAG
Mps1-min-For: CTGCGCTAACCTTGTGTTGG
Stra8-For: GTGCAAGCTGAACAACAGGA
Stra8-Re: AGGGACACAGCATTGGAGTC
Ddx4-For: CACGTGCAGCCGTTTAAGCCGCGT
Ddx4-Re: TTCCCATTCTAAACAACACCCTGAA.
Histology and immunofluorescence
For paraffin sectioning, testes were fixed in Bouin’s trichrome at 4 °C overnight. After washing in phosphate-buffered saline (PBS), the testes were dehydrated with gradient concentrations of ethanol and xylene, embedded in paraffin, and cut into 5 μm-thick sections. H&E staining was performed and was followed by deparaffinization and rehydration. The slides were imaged with a Nikon microscope.
For frozen sectioning, testes were fixed in 4% PFA solution at 4 °C overnight and then dehydrated with 30% sucrose solution for another 2 days. The testes were dipped in O.C.T. compound and then quickly frozen in liquid nitrogen. Frozen sections with a thickness of 7 μm were subjected to heat-induced antigen retrieval (sodium citrate, pH = 6) in a water bath for 20 min at 98 °C. The sections were cooled down slowly in retrieval buffer until they reached room temperature. Then, they were blocked and permeated in 5% bovine serum albumin with 0.2% Triton in PBS for 1 h before being incubated with primary antibodies overnight at 4 °C. After washing in PBS three times, the sections were incubated with secondary antibodies at room temperature for 2 h. After staining with DAPI (Beyotime) for 5 min, the sections were mounted with a fluorescent mounting medium (Beyotime). All steps were performed with protection from light. Images were obtained with an Olympus microscope and processed with ImageJ and Photoshop software.
Spermatocyte surface spreading
Testes from 1-month-old mice were subjected to a spermatocyte surface spreading assay as described by the previous research34. Briefly, the tunica albuginea was removed, and the tubules were placed into the hypotonic buffer (30 mM Tris, 50 mM sucrose, 17 mM trisodium citrate dihydrate, 5 mM EDTA, 0.5 mM DTT, and 0.5 mM PMSF, pH = 8.2) for 30–60 min. Subsequently, the tubules were torn into pieces in sucrose buffer (100 mM sucrose, pH = 8.2) and pipetted repeatedly to make a suspension. The cell suspension was smeared onto glass slides containing 1% PFA and 0.15% Triton X-100, pH = 9.2. The slides were dried overnight in a closed box with high humidity. Finally, the slides were washed twice for 2 min in 0.4% Photoflo (Kodak) and dried at room temperature. The slides were kept at −20 °C.
Antibody
The antibody used in immunofluorescence staining were as follows: anti-DDX4 (ab13840, Abcam), anti-GATA4 (ab124265 Abcam), anti-c-KIT (25-1171-82, eBioscience) anti-SYCP3 (ab97672, Abcam), anti-STRA8 (ab49602, Abcam), anti-γH2AX (ab2893, Abcam), anti-H3pSer10 (ab5176, Abcam), anti-MPS1 (ab11108, Abcam), anti-H2BK120ubi (#5546, CST), anti-MDM2 (sc-965, Santa Cruz), anti H2B (ab1790, Abcam), and anti-ACTIN (sc-47778, Santa Cruz). All fluorophore-conjugated secondary antibodies were obtained from Jackson Immuno Research.
RNA-Seq and analysis
Total RNA was extracted from whole testes at P10. RNA-Seq libraries were constructed from 1 μg of RNA using a NEBNext UltraTM RNA Library Prep Kit (NEB, USA) following the manufacturer’s instructions. Sequencing was performed on an Illumina NovaSeq platform with 150 bp paired-end reads. DEG analysis was performed with the DESeq2 R package. GO and KEGG enrichment analysis of the DEGs was implemented with the clusterProfiler R package. Libraries constructing, sequencing, and data analyzing were performed by Novogene Co., Ltd. (Tianjin, China).
Sperm counting
Cauda epididymides from 6-month-old mice were removed and cut in 500 μl of DMEM. The medium was incubated at 37 °C for 7 min to release sperm. After diluting them 50 times, the sperm were analyzed with a cell counter (Countstar).
Assessment of fertility
To test fertility, 3-month-old conditional knockout and control males were paired with two random adult wild-type females for at least three months. The numbers of offspring from each pregnancy were recorded.
TUNEL assay
A TUNEL assay was performed following the instructions of an Apoptosis Detection Kit (Millipore). Sections after deparaffinization and rehydration were incubated with proteinase K (20 μg/ml) for 15 min and washed 2 times in PBS. Equilibration buffer was immediately applied and kept on the sections for at least 15 s. The excess liquid was gently tapped off, and the working-strength TDT enzyme was pipetted onto each section. The sections were incubated in a humidified chamber at 37 °C for 1 h and then placed in a stop buffer for 10 min. After washing 3 times in PBS, the sections were incubated with an anti-digoxigenin conjugate for 30 min at room temperature while protected from light and were washed in PBS 3 times again. After staining with DAPI (Beyotime) for 5 min, the sections were mounted with a fluorescent mounting medium. The slides were viewed with a Leica fluorescence microscope.
Statistical analysis
All statistical data were analyzed by using GraphPad Prism. Sample sizes were described in figure legends. The variances of the two groups were compared by one-way ANOVA, and the P value was determined with a two-tailed unpaired Student’s t test or with Welch correction. Results were shown as mean ± s.e.m. Significance was shown as *P < 0.05, **P < 0.01, ***P < 0.001.
Supplementary information
Acknowledgements
We would like to thank present and past members of the laboratory for their contribution to this study. This research is funded by the National Key R&D Program of China (2017YFA0103301), and the Fundamental Research Funds for the Central Universities (Tongji University, Grant No. 22120180521).
Author contributions
Feng Sun and Xiaomei Yang designed the experiments; Fang Qiang performed most of the experiments; Xuelin Chen and Lei Zhang performed part of the experiments. Yabin Li, Chenxin Yang, and Tianzeng Sun performed part of the immunofluorescence experiments. Jianfeng Chang contributed to the data analysis. Feng Sun, Fang Qiang, and Xiaomei Yang wrote the paper. All authors reviewed and approved the paper.
Data availability
All sequencing data that support the findings of this study have been deposited in the NCBI Gene Expression Omnibus (GEO) under accession number GSE165143.
Ethics approval and consent to participate
All animal procedures were approved by the Tongji University Institutional Animal Care and Use Program Advisory Committee (Protocol number: IACUC-017-006).
Conflict of interest
The authors declare no competing interests.
Footnotes
Edited by A. Stephanou
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Xiao-Mei Yang, Email: yxm411@tongji.edu.cn.
Feng Sun, Email: fengsun@outlook.com.
Supplementary information
The online version contains supplementary material available at 10.1038/s41419-021-03815-4.
References
- 1.La HM, Hobbs RM. Mechanisms regulating mammalian spermatogenesis and fertility recovery following germ cell depletion. Cell Mol. Life Sci. 2019;76:4071–4102. doi: 10.1007/s00018-019-03201-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Anderson EL, et al. Stra8 and its inducer, retinoic acid, regulate meiotic initiation in both spermatogenesis and oogenesis in mice. Proc. Natl Acad. Sci. USA. 2008;105:14976–14980. doi: 10.1073/pnas.0807297105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Feng CW, Bowles J, Koopman P. Control of mammalian germ cell entry into meiosis. Mol. Cell Endocrinol. 2014;382:488–497. doi: 10.1016/j.mce.2013.09.026. [DOI] [PubMed] [Google Scholar]
- 4.Li X, et al. The roles of retinoic acid in the differentiation of spermatogonia and spermatogenic disorders. Clin. Chim. Acta. 2019;497:54–60. doi: 10.1016/j.cca.2019.07.013. [DOI] [PubMed] [Google Scholar]
- 5.Xu K, et al. Mettl3-mediated m(6)A regulates spermatogonial differentiation and meiosis initiation. Cell Res. 2017;27:1100–1114. doi: 10.1038/cr.2017.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Xu Z, et al. H2B ubiquitination regulates meiotic recombination by promoting chromatin relaxation. Nucleic Acids Res. 2016;44:9681–9697. doi: 10.1093/nar/gkw183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Keeney S, Giroux CN, Kleckner N. Meiosis-specific DNA double-strand breaks are catalyzed by Spo11, a member of a widely conserved protein family. Cell. 1997;88:375–384. doi: 10.1016/S0092-8674(00)81876-0. [DOI] [PubMed] [Google Scholar]
- 8.Jiang H, et al. MOF influences meiotic expansion of H2AX phosphorylation and spermatogenesis in mice. PLoS Genet. 2018;14:e1007300. doi: 10.1371/journal.pgen.1007300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Baudat F, Imai Y, de Massy B. Meiotic recombination in mammals: localization and regulation. Nat. Rev. Genet. 2013;14:794–806. doi: 10.1038/nrg3573. [DOI] [PubMed] [Google Scholar]
- 10.Fernandez-Capetillo O, et al. H2AX is required for chromatin remodeling and inactivation of sex chromosomes in male mouse meiosis. Dev. Cell. 2003;4:497–508. doi: 10.1016/S1534-5807(03)00093-5. [DOI] [PubMed] [Google Scholar]
- 11.Liu X, Winey M. The MPS1 family of protein kinases. Annu. Rev. Biochem. 2012;81:561–585. doi: 10.1146/annurev-biochem-061611-090435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Manic G, Corradi F, Sistigu A, Siteni S, Vitale I. Molecular regulation of the spindle assembly checkpoint by kinases and phosphatases. Int. Rev. Cell Mol. Biol. 2017;328:105–161. doi: 10.1016/bs.ircmb.2016.08.004. [DOI] [PubMed] [Google Scholar]
- 13.Pachis S. T. & Kops G. Leader of the SAC: molecular mechanisms of Mps1/TTK regulation in mitosis. Open Biol. 8, 180109 10.1098/rsob.180109 (2018). [DOI] [PMC free article] [PubMed]
- 14.Hiruma Y, et al. Competition between MPS1 and microtubules at kinetochores regulates spindle checkpoint signaling. Science. 2015;348:1264–1267. doi: 10.1126/science.aaa4055. [DOI] [PubMed] [Google Scholar]
- 15.Ji Z, Gao H, Yu H. CELL DIVISION CYCLE. Kinetochore attachment sensed by competitive Mps1 and microtubule binding to Ndc80C. Science. 2015;348:1260–1264. doi: 10.1126/science.aaa4029. [DOI] [PubMed] [Google Scholar]
- 16.Sun T, et al. Cellular abundance of Mps1 and the role of its carboxyl terminal tail in substrate recruitment. J. Biol. Chem. 2010;285:38730–38739. doi: 10.1074/jbc.M110.177642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sudakin V, Chan GK, Yen TJ. Checkpoint inhibition of the APC/C in HeLa cells is mediated by a complex of BUBR1, BUB3, CDC20, and MAD2. J. Cell Biol. 2001;154:925–936. doi: 10.1083/jcb.200102093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Izawa D, Pines J. Mad2 and the APC/C compete for the same site on Cdc20 to ensure proper chromosome segregation. J. Cell Biol. 2012;199:27–37. doi: 10.1083/jcb.201205170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Marston AL, Wassmann K. Multiple duties for spindle assembly checkpoint kinases in meiosis. Front. Cell Dev. Biol. 2017;5:109. doi: 10.3389/fcell.2017.00109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hached K, et al. Mps1 at kinetochores is essential for female mouse meiosis I. Development. 2011;138:2261–2271. doi: 10.1242/dev.061317. [DOI] [PubMed] [Google Scholar]
- 21.Wang W, et al. Structural and mechanistic insights into Mps1 kinase activation. J. Cell Mol. Med. 2009;13:1679–1694. doi: 10.1111/j.1582-4934.2008.00605.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.El Yakoubi W, et al. Mps1 kinase-dependent Sgo2 centromere localisation mediates cohesin protection in mouse oocyte meiosis I. Nat. Commun. 2017;8:694. doi: 10.1038/s41467-017-00774-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Soumillon M, et al. Cellular source and mechanisms of high transcriptome complexity in the mammalian testis. Cell Rep. 2013;3:2179–2190. doi: 10.1016/j.celrep.2013.05.031. [DOI] [PubMed] [Google Scholar]
- 24.Gallardo T, Shirley L, John GB, Castrillon DH. Generation of a germ cell-specific mouse transgenic Cre line, Vasa-Cre. Genesis. 2007;45:413–417. doi: 10.1002/dvg.20310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sadate-Ngatchou PI, Payne CJ, Dearth AT, Braun RE. Cre recombinase activity specific to postnatal, premeiotic male germ cells in transgenic mice. Genesis. 2008;46:738–742. doi: 10.1002/dvg.20437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ahmed EA, de Rooij DG. Staging of mouse seminiferous tubule cross-sections. Methods Mol. Biol. 2009;558:263–277. doi: 10.1007/978-1-60761-103-5_16. [DOI] [PubMed] [Google Scholar]
- 27.Meistrich ML, Hess RA. Assessment of spermatogenesis through staging of seminiferous tubules. Methods Mol. Biol. 2013;927:299–307. doi: 10.1007/978-1-62703-038-0_27. [DOI] [PubMed] [Google Scholar]
- 28.Minsky N, Oren MJMC. The RING domain of Mdm2 mediates histone ubiquitylation and transcriptional repression. Mol. Cell. 2004;16:631–639. doi: 10.1016/j.molcel.2004.10.016. [DOI] [PubMed] [Google Scholar]
- 29.Yu ZC, Huang YF, Shieh SY. Requirement for human Mps1/TTK in oxidative DNA damage repair and cell survival through MDM2 phosphorylation. Nucleic Acids Res. 2016;44:1133–1150. doi: 10.1093/nar/gkv1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Huang YF, Chang MD, Shieh SY. TTK/hMps1 mediates the p53-dependent postmitotic checkpoint by phosphorylating p53 at Thr18. Mol. Cell Biol. 2009;29:2935–2944. doi: 10.1128/MCB.01837-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ji Z, Gao H, Yu H. Kinetochore attachment sensed by competitive Mps1 and microtubule binding to Ndc80C. Science. 2015;348:1260–1264. doi: 10.1126/science.aaa4029. [DOI] [PubMed] [Google Scholar]
- 32.Fuchs Y, Steller H. Programmed cell death in animal development and disease. Cell. 2011;147:742–758. doi: 10.1016/j.cell.2011.10.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Li M, Sun S, Priest J, Bi X, Fan Y. Characterization of TNF-induced cell death in Drosophila reveals caspase- and JNK-dependent necrosis and its role in tumor suppression. Cell Death Dis. 2019;10:613. doi: 10.1038/s41419-019-1862-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Peters AH, Plug AW, van Vugt MJ, de Boer P. A drying-down technique for the spreading of mammalian meiocytes from the male and female germline. Chromosome Res. 1997;5:66–68. doi: 10.1023/A:1018445520117. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All sequencing data that support the findings of this study have been deposited in the NCBI Gene Expression Omnibus (GEO) under accession number GSE165143.