Abstract
For clinical chemistry, or for non-invasive sensing through skin, it is often necessary to obtain quantitative information in highly scattering media. We describe two simple methods for fluorescence lifetime measurements in highly scattering media, and in particular in an intralipid suspension. Lifetimes can be measured using an intensity decay law that accounts for the time delays and pulse-broadening effects of multiple light-scattering events in the intralipid. Alternatively, the phase and modulation measurements can be performed relative.to a reference fluorophore with a known lifetime. These approaches provide reliable lifetime data for a pH-sensitive fluorophore contained within a microeuvette 4 mm under the surface of the intralipid suspension. Fluorescence lifetime-based sensing is now recognized as a valuable methodology in clinical and analytical chemistry, and the possibility of lifetime-based sensing in turbid media has been demonstrated.
Keywords: Clinical chemistry, Fluorescence, Huorescence spectroscopy, Frequency-domain fluorescence, Lifetime-based sensing, Non-invasive sensing, pH sensing, Photon migration, Sensing, Time-resolved fluorescence
1. Introduction
Fluorescence detection is widely used in analytical and clinical chemistry [1–3]. At present, such measurements require some invasive procedures, such as drawing blood or insertion of a catheter, followed by one or more manipulations to prepare the sample for the optical measurements. Hence, it is desirable to extend fluorescence methods to provide non-invasive diagnostics. The possibility of non-invasive measurements is evident from the red light that can be seen penetrating one’s hand in front of a flashlight. The technical feasibility of non-invasive measurements is suggested by the availability of simple and robust red/NIR laser diodes. However, quantitative fluorescence intensity measurements require careful consideration of the instrumentation, sample holder, and clarity of the sample, to avoid the undesired effects of light losses, photobleaching, absorption, and scatter of the incident light by the sample. These difficulties have prevented the use of fluorescence as a non-invasive diagnostic tool.
In recent years it has been recognized that time-resolved measurements may offer advantages in situations where fluorescence intensities are difficult to measure. For instance, it is even difficult to measure the absorption spectra of chromophores in highly scattering media [4]. This problem has been solved in part by the use of frequency-domain or phase-modulation measurements of the time delay due to multiple scattering events [5]. As another example, in fluorescence microscopy, it is not possible to control the local intracellular concentration of fluorophores. Consequently the fluorescence intensity images often lack the required information on intracellular analytes. In contrast, the fluorescence lifetime or decay-time images arc independent of the local probe concentration, and can thus provide quantitative information on the analytes even without wavelength-ratiometric measurements [6–8]. The advantages of time-resolved imaging and sensing have been summarized in several reports [9,10], and lifetime sensors are now known for Ca2+, Mg2+, K+, pH, and glucose [10–16]. Additionally, lifetime-sensing schemes have been developed for use in immunoassays [17,18], flow cytometry [19], and for use with laser diode sources [20].
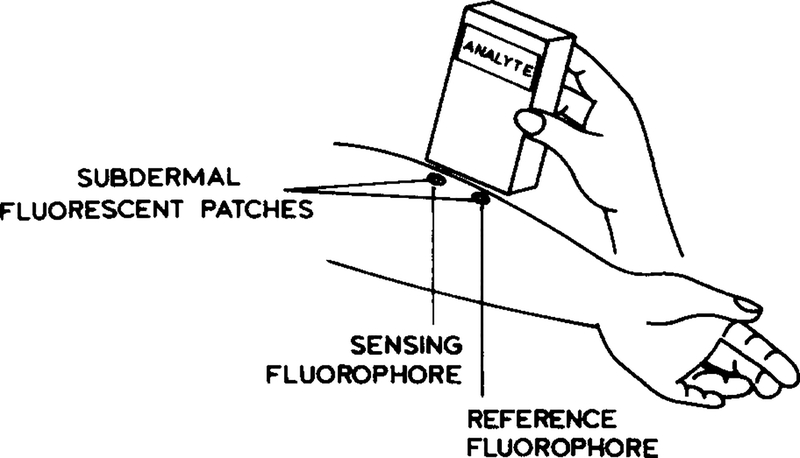
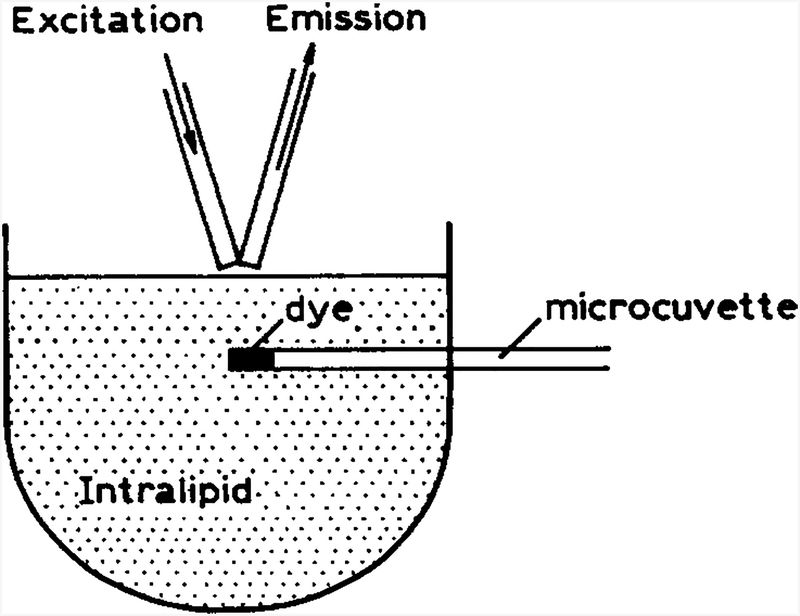
We now describe how time-resolved or lifetime-based sensing can be used in real-world situations, such as quasi non-invasive measurements performed through skin. In these eases we imagine that sensing patches may be embedded below the skin, as suggested previously for glucose [16] (Fig. 1). The possibility of lifetime-based sensing in scattering media is supported by a computational study using the known scattering and absorption properties of tissues [21]. These simulations suggested that properly referenced phase and modulation measurements can be used to recover the lifetime of fluorophores uniformly dispersed in scattering media [21]. In the present report, we consider a sensing fluorophore localized below the surface of the turbid medium. Our experimental configuration is shown in Fig. 2. The sensing fluorophore is contained within a capillary tube below the surface of an intralipid suspension. The microcuvette contains a fluorophore whose lifetime is sensitive to the analyte, which is pH in the present report. Our goal is to measure the pH of the ‘sensor’ embedded in the intralipid using the time-dependent fluorescence that re-emerges through the surface of the intralipid. However, time-dependent fluorescence measurements require a reference optical signal from the same geometrical position as that of the sample. Usually, a scattering solution at low concentration is used to determine the lamp profile in time-domain or the phase angle and modulation of excitation light in frequency-domain measurements. In the case of highly scattering media, it is not possible to observe scattered light from the same region as the embedded sensor. Therefore, the lamp profile for time-domain, or the phase angle and modulation of excitation light for frequency-domain measurements, cannot be determined, since the incident light will re-emerge from an unknown region of the sample. We describe how this difficulty can be circumvented using a reference fluorophore of known decay time and the frequency-domain technique.
Fig. 1.
Intuitive description of transdermal lifetime-based sensing.
Fig. 2.
Experimental geometry for lifetime-based sensing in turbid media.
2. Materials and methods
Fluorescence intensity decays were determined using the frequency-domain (FD) method [22,23] and the gigahertz instrumentation described previously [24]. Excitation was provided by the 3.795 MHz cavity-dumped output of a Rhodamine 6G dye laser at 570 nm, or using the 75.9 MHz mode-locked output of an argon-ion laser at 514 nm, which provides pulse widths near 5 and 100 ps, respectively. The emission was isolated with an interference filter at 620 or 640 nm.
The sample consisted of a suspension of 20% intralipid, diluted 20-fold to 1%. The incident light was directed onto the surface of the intralipid using a 1 mm diameter fiber bundle. The emission or scattered light was collected from the surface with a similar fiber-optic bundle and directed to the photomultiplier tube (Fig. 2).
The fluorescent samples were contained in about 1 nun (inner) diameter capillary tubes of the type used for melting-point determinations, which allowed control of the probe’s environment and knowledge of its lifetime(s). Texas Red Hydrazide (TRH) was used as a pH-insensitive reference fluorophore, and Carboxy SNARF-6 (CS6) was used as the pH lifetime sensor [15]. Phase and modulation measurements were performed relative to scattered excitation from the surface of the intralipid at the excitation wavelength, or from the reference fluorophore at the same depth under the intralipid surface using an interference filter to isolate the emission.
3. Theory
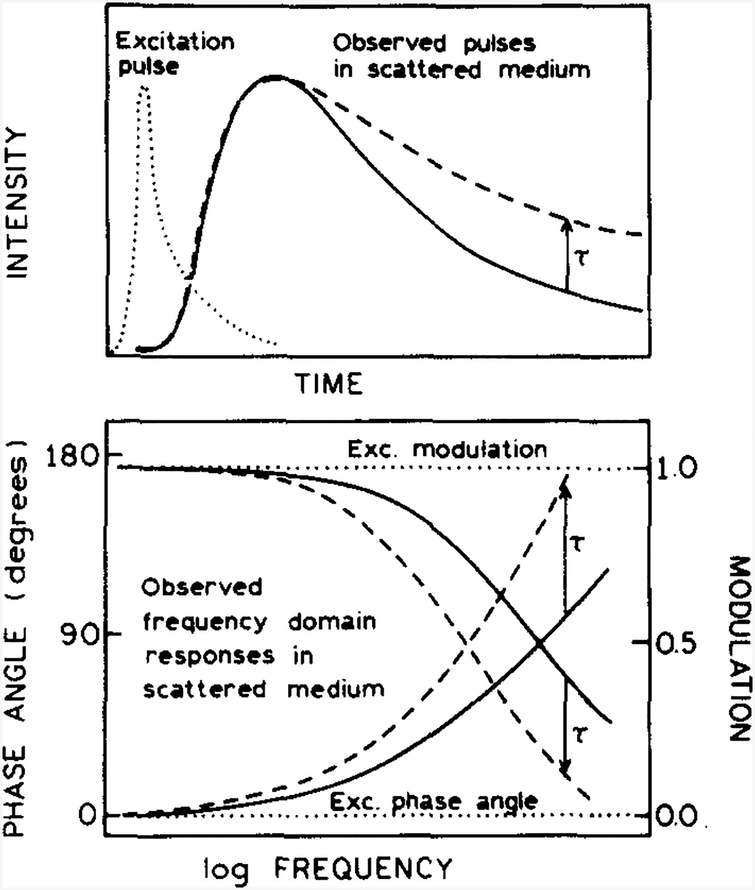
We used the FD method to measure the lifetimes of fluorophores in scattering media. The basis of these measurements can be understood by considering the effect of the scattering medium on the time-domain (TD) data. Assume the sample is exposed to δ-function pulse of light. This light pulse is time delayed and broadened due to multiple scattering events prior to arriving at the fluorescent sensor (Fig. 3, top). The broadened and time-delayed pulse excites the fluorophores, which may then decay by a single or multi-exponential decay law. The emitted light is again time delayed and broadened as it migrates to the detector located at the surface of the sample. At the surface of the scattering medium the re-emerged light (solid curve) is time delayed and broadened relative to the incident light (dotted curve). The fluorescence emission (dashed curve) is further delayed relative to the scattered light because of the lifetime τ of the excited state.
Fig. 3.
Effect of multiple scattering events on time-domain (top) or phase-modulation (bottom) measurements of a fluorescence lifetime. Pulsed or modulated excitation (· · · · ·), time-delayed scattered light (—), and thefluorescence (- - -), which is further delayed by the lifetime τ, arc shown.
A similar effect is seen in the phase-modulation measurements (Fig. 3, bottom). In this case, the re-emergent scattered light (solid curve) is delayed in phase and demodulated relative to the intensity-modulated incident light (dotted curve). The re-emergent fluorescent light is further phase shifted and demodulated due to the fluorescent lifetime (dashed curve). Our goal is to measure the phase (ϕ) and modulation (m) of the emission, independent of the phase shift and demodulation due to multiple scattering events.
3.1. Frequency-domain lifetime measurements
Prior to consideration of the effects of light scattering, we briefly summarize the principle of FD or phase-modulation fluorometers. Suppose a fluorescent sample is illuminated with intensity-modulated light. The emission is delayed in phase by an angle (ϕω) due to the lifetime (τ) of the excited state. The emission is also demodulated by a factor mω relative to the modulation of the incident light. For a single-exponential decay the phase and modulation are related to the decay time τ by
| (1) |
| (2) |
where ω to is the light modulation frequency in rad s−1. If the fluorescence decay is a single exponential, then the same lifetime is recovered independently of the light modulation frequency.
The intensity decay of fluorophores is often multi-exponential.
| (3) |
when τ are the decay times and αi the associated amplitudes. In this case, one measures the phase and modulation over a range of light-modulation frequencies. The values of αi and τi are determined by fitting the measured values (ϕω and mω) to calculated values ( and ) by the procedure of nonlinear least squares [23]. One minimizes the goodness-of-fit parameter
| (4) |
where v is the number of degrees of freedom, and δϕ and δm are the experimental uncertainties in the measured phase and modulation values, respectively. The calculated values of and are determined from
| (5) |
where ω = 2πf is the modulation frequency (in Hz), and
| (6) |
| (7) |
| (8) |
where
| (9) |
3.2. Frequency-domain measurements of time-dependent photon migration
We now consider the effects of scatter on the modulated incident light. The theory of time-dependent photon migration in tissues has been presented by several groups [25–27], and this theory has been extended to FD measurements [28]. These theories describe the effects of sample geometry, extent of scattering and other factors on light migration. However, for the present purpose of non-invasive lifetime measurements, we prefer a phenomenological model [29] that provides an intuitive description of the effect of scattering on the incident light.
When the light pulse enters the scattering medium it is time delayed (tL) due to multiple scattering events, as is shown schematically in Fig. 3. The magnitude of the time-delay tL is expected to increase with increasing depth in the sample. Additionally, the pulse is broadened due to multiple path lengths, which is most evident on the rising side of the pulse. This effect is analogous to a transit-time spread in a photomultiplier tube (PMT), and hence we refer to this time as Δt. Finally the light decays due to sample absorption and/or migration away from the detector. We refer to the decay time as to, not to be confused with a fluorescence decay time (τ or τi).
For the phenomenological model the phase angle of the re-emergent light, without interacting with any fluorophore, is given by
| (10) |
The phase angle due to the time delay (tL) is given by ϕL = ωtL [29], and is therefore strongly dependent on the light-modulation frequency ω (rad s−l). The decay time tD behaves analogously to the fluorescence lifetime (Eq. (1)). The modulation of the re-emergent scattered light is given by
| (11) |
where
| (12) |
| (13) |
Eq. (13) is analogous to the usual relationship between a decay time and demodulation (Eq. (2)). We note that the modulation values are not sensitive to the phase of the scattered light, and hence not sensitive to tL, since tL does not appear in Eqs. (12) and (13). This model was found to account for the FD data, so the parameters tD, tL and Δt are adequate to describe the re-emerged scattered light.
3.3. Lifetime measurements in scattering media
Suppose the fluorophore is excited by the migrating light pulse. In this case the ‘excitation pulse’ is not the original & function, but rather the time-delayed and broadened pulse (Fig. 3). Measurements of the emission, relative to the pulse that arrives at the sensor, will provide the correct decay time of the sensor. However, in highly scattering media it is not experimentally possible to observe the incident light that re-emerges from the same position as the embedded sensor. The detected scattered light will be predominantly due to scattering from the surface of the solution. In contrast, the fluorescence will be excited with the delayed and broadened pulse that has migrated down to the sensor, and the emission will be delayed and broadened as it migrates to the surface. Consequently, the phase and modulation values ( and ), measured relative to light scattered from the surface(s), will contain contributions from both time-dependent photon migration and the lifetime of the fluorophore. For a single-exponential lifetime (τ) these values will be
| (14) |
| (15) |
Eqs. (14) and (15) will strictly correct for measurement of the emission relative to the excitation pulse, as could be observed with a mirror in front of the fibers in place of the scattering solution.
Examination of Eqs. (14) and (15) reveals that the effects of τ or tD are mathematically similar, so that it is not possible to recover both values (τ and tD) from measurement of the emission relative to the light scattered from the surface. The time delay tL appears in a linear manner, and does not contribute to the modulation, so this value can be found from the FD data. Fortunately, the values of the decay time tD are modest for all but the shortest lifetimes. For instance, the value of tD for a position 4 mm deep in the 1% intralipid has been estimated as about 140 ps. Hence, for lifetimes 1 ns and longer use of the values of and provides a good approximation (τapp) to the true lifetime (τ):
| (16) |
| (17) |
In practice the values of tL and Δt are determined from the FD data ( and ) because of their distinct contribution in Eqs. (16) and (17). In particular, we found that the use of τapp, from Eqs. (16) and (17) was adequate to recover a single-exponential decay. However, if the decay was a multi-exponential, we found there was too much correlation between the tD and τi values, which distorted the recovered parameters (αi and τi).
3.4. Lifetime measurements in scattering media using a reference fluorophore
As described above, the scattered light is dominated by light scattered from near the surface, whereas the fluorescence originates from the embedded sensor deeper in the sample. Consequently, the values of and , contain contributions from tD, tL and Δt (Eqs. (14) and (15)). This difficulty can be avoided using a reference fluorophore of known lifetime at the same location as the sensor. In this way, we can use an emission filter to separate the emission of the reference fluorescence from the scattered light.
In the present experiments we replaced the sensor with a reference fluorophore emitting at the same wavelength as the sensor. Alternatively, one could use adjacent sensors and reference fluorophores that emit at different wavelengths, and isolate the desired emission with optical filters. The use of a reference fluorophore corrects for the effects of light scatter while the excitation is traveling to the sensor, and the emission is migrating to the surface. In both cases the effects of scattering are the same, and the only difference in the re-emergent light is due to the different lifetimes of the sample and reference fluorophore. In the present analysis we are ignoring the effects of wavelength, which are expected to be minor for the small wavelength difference between the scattering and emission wavelengths.
The use of a reference fluorophore requires correction for its decay time. Suppose the reference displays a single-exponential decay τR The measured phase angle of the sensor, relative to the reference fluorophore, is smaller than the true value due to the phase of the reference fluorophore (ϕR). Similarly, the modulation of the sensor, measured relative to the reference, is larger than the true value due to the decreased modulation of the emission of the reference compared to the modulation of the excitation. The actual phase (ϕω) and modulation (mω) values can be calculated from the observed values relative to the reference fluorophore using
| (18) |
| (19) |
where tan ϕR= ωτR [30]. In this case, the measured phase and modulation values are corrected for the effects of time-dependent photon migration, and if desired, can be used to recover a multi-exponential decay (Eqs. (3)–(9)).
4. Results
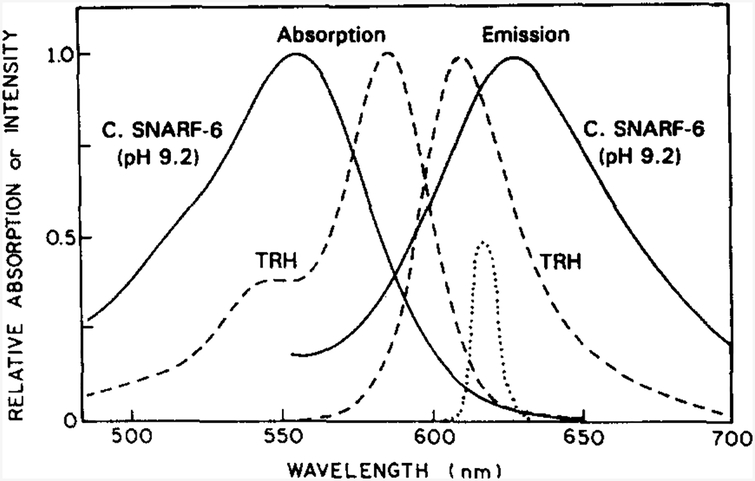
We examined the effects of a scattering medium on the FD measurements, using fluorophores contained in microcuvettes located below the surface of the intralipid (Fig. 2). By placing the fluorophores within the microcuvette, we avoided changes in the lifetime due to possible molecular interactions of the fluorophore with the intraiipid. Two fluorophores were selected for these experiments: Texas Red Hydrazide (TRH) and Carboxy SNARF-6 (CS6). Absorption and emission spectra of these fluorophores are shown in Fig. 4. Both fluorophores can be excited with the 570 nm cavity-dumped output of our R6G dye laser, or by the mode-locked output of the argon-ion laser at 514 nm. CS6 was selected as a pH indicator whose mean lifetime depends on pH [15]. TRH was chosen because its emission could be observed at the same wavelengths as used for CS6. The intensity decay of TRH in the absence of intralipid is essentially a single exponential, and there are no significant effects of pH on its lifetime.
Fig. 4.
Absorption and emission spectra oftbe pH sensor Carboxy SNARF-6 (CF6) and the reference fluorophore Texas Red Hydrazide (TRH). The transmission profile (· · · · ·) of the 620 nm interference filter used to isolate the emission is also shown. Intralipid was not present.
4.1. Effect on intralipid on the phase and modulation data
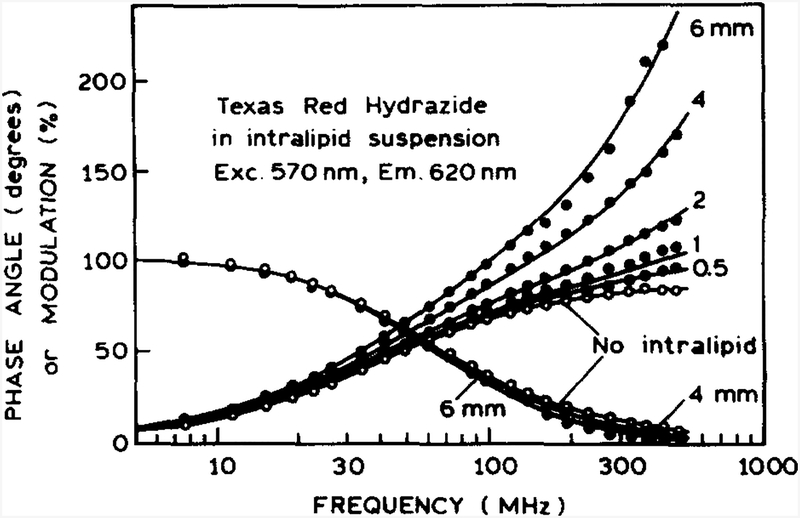
FD intensity decay data of TRH are shown in Fig. 5. These data were obtained for TRH in the microcuvette, located at various depths below the intralipid surface. The phase and modulation values were measured in the usual manner, by comparing the phase and modulation of the emission at 620 nm with the phase and modulation of the scattered light at the excitation wavelength of 570 nm. In the absence of intralipid the phase values show the usual dependence on light modulation frequency; at high light modulation frequencies the phase angle approaches 90°, and the modulation decreases toward zero. In the presence of intralipid, the phase angles exceed 90°, which cannot be explained by the decay of directly excited species [31]. Phase angle in excess of 90° can result only from excited-state reactions or time delays in the detected signal due to some process such as time-dependent photon migration, different path lengths for scattered and fluorescent light, or color effects in the detector. In the present case the rapidly increasing phase angles are due to the effects of the delay time tL, which results in increasingly large phase angles at higher light modulation frequencies (tL on the phase angles prevent direct use of the phase angle for determining the lifetime. In contrast, tL does not affect the modulation, and the effects of Δt are modest. Consequently, the modulation values, measured relative to the surface scatter, can provide a reasonable estimate of the lifetime of the embedded probe.
Fig. 5.
Frequency response of TRH in the microeuvette at various depths below the intralipid surface.
The data in Fig. 5 were analyzed using the model described in Eqs. (16) and (17), where the apparent decay time was taken as representative of the decay time of TRH (Table 1). We also examined the FD data for CS6 at pH 9.2 when the intensity decay is dominated by a component with τ = 1.15 ns. As the depth of the microcuvette is increased, the delay time increases, as does the transit-time spread. The values of the delay time increase approximately linearly with depth, and are consistent with a mean path length about 10 to 15 times larger than the time-of-flight for the geometric distance between the surface of the intralipid (excitation source), the position of the microcuvette, and back to the intralipid surface (emission collection). The transit-time spread also increases with depth, which suggests that the emission is affected by broadening of the pulse prior to its arrival at the sample, and again during migration of the fluorescence to the surface.
Table 1.
Intensity decays of Texas Red Hydrazide (TRH) and Carboxy SNARF-6 (CS6) in capillary tubes at various depths in the intralipid suspensiona
| Depth (mm) | Dye | τapp (ns) | tL (ps) | Δt (ps) | χR2 |
|---|---|---|---|---|---|
| No intralipid | CS6 | 1.09 | −18 | 〈0〉 | 1.3 |
| TRH | 4.10 | − 34 ± 2 | 67 ± 83 | 1.5 | |
| 0.5 | TRH | 3.84 | 59 | 157 ± 36 | 1.9 |
| 1.0 | TRH | 3.94 | 132 | 156 ± 35 | 1.7 |
| 2.0 | CS6 | 1.22 | 208 | 190 ± 10 | 1.0 |
| TRH | 3.99 | 226 | 205 ± 66 | 8.2 | |
| 4.0 | CS6 | 1.33 | 473 | 350 ± 32 | 15.0 |
| TRH | 4.20 | 480 | 376 ± 86 | 18.0 | |
| 6.0 | TRH | 3.89 | 847 | 957 ± 170 | 65.9 |
1.0% intralipid suspension, 20 °C, excitation at 570 nm, emission at 620 nm. The Carboxy SNARF-6 wa in 80 mM tris, pH 9.2.
The decay times (τ) recovered for TRH and CS6 are close to the expected values of 4.1 and 1.15 ns, respectively, for all depths of the microcuvette in the intralipid (Table 1). Nonzero values of tL and Δt in the absence of intralipid indicate that the intensity decays of TRH and CS6 are not pure single exponential (see Table 3). However, the FD data appear to be adequately fit by a single-exponential decay (apparent decay time), as can be seen from the reasonable values of χR2 from 1 to 2. The elevated values of χR2 for sensors embedded deeper in the intralipid (above 2 mm) appear to be the result of a decreased signal-to-noise ratio for the weaker signal. The values of χR2 were not improved by using a multi-exponential decay time (τapp i). These results suggests that for lifetimes longer than 1 ns, the effects of time-dependent photon migration can be satisfactorily described by the time delay tL and transit-time spread Δt. The recovered apparent decay times (τapp) are not significantly altered by photon migration relative to the values in the absence of intralipid (Table 1). The elevated values of χR2 with depth in intralipid suggest that the presented phenomenological model cannot be applied for multi-exponential fluorescence intensity decay analysis.
Table 3.
Multi-exponential intensity decay analysis of Carboxy SNARF-6 (pH 9.2) measured relative to its excitation source and to the fluorescent reference TRH
| Reference/Sample | τi (ns) | αi | fia | χR2 |
|---|---|---|---|---|
| Surface scatter/ | 1.06 | 1 | 1 | 6.7 |
| CS6 without intralipid | 0.46 | 0.188 | 0.084 | |
| 1.15 | 0.812 | 0.916 | 0.6 | |
| TRH/CS6 without intralipid | 1.10 | 1 | 1 | 20.9 |
| 0.16 | 0.243 | 0.040 | ||
| 1.19 | 0.757 | 0.960 | 0.9 | |
| TRH/CS6 under 2 mm of intralipid | 1.03 | 1 | 1 | 38.7 |
| 0.17 | 0.310 | 0.062 | ||
| 1.16 | 0.690 | 0.938 | 1.6 | |
| TRH/CS6 under 4 mm of intralipid | 1.12 | 1 | 1 | 10.4 |
| 0.30 | 0.128 | 0.036 | ||
| 1.18 | 0.872 | 0.964 | 5.0 |
The fractional contribution of each component to the steady-state intensity can be calculated using fi = αi τi / Σjαjτj.
4.2. Lifetime measurements using a reference fluorophore
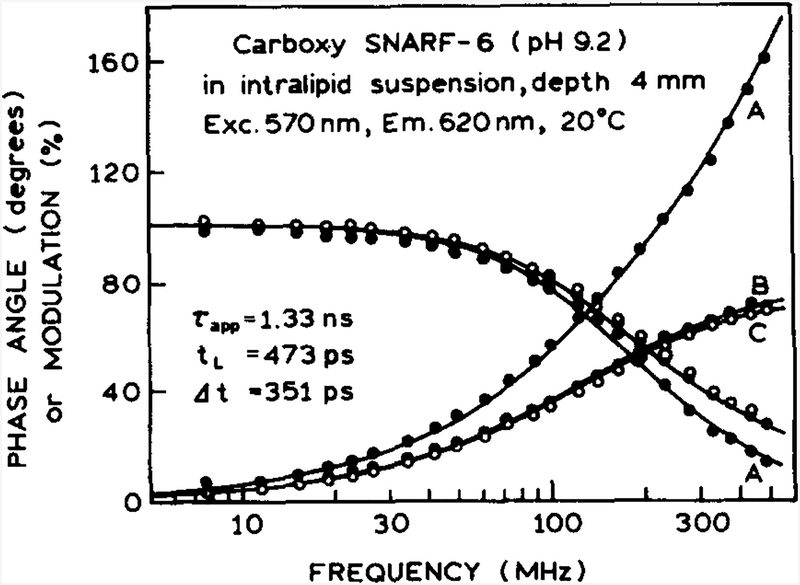
The previous results demonstrated good precision of the decay times using light scattered off the surface as the reference signal. However, it was necessary to allow for time delays and broadening of the incident light. We expected more reliable lifetime measurements, over a wider range of experimental conditions, using a reference fluorophore at the same location as the sensor. Such measurements are shown in Fig. 6, where the sensing fluorophore is CS6 and the reference fluorophore is TRH. For these measurements, the CS6 and the TRH reference were contained in the microcuvettes. When measured relative to the scattered light from the surface, the phase angles and modulations show the usual effects of the time delay and transit-time spread (Fig. 6, A). When measured relative to TRH, the phase angle (B) and modulations are essentially identical to those found in the absence of intralipid (C). These results illustrate that using a reference fluorophore corrects for the effects of time-dependent photon migration.
Fig. 6.
Frequency response of CS6, in the microcuvette 4 nun below the intralipid surface, when measured relative to the scattered light (A), relative to the TRI-I standard (B), and without intralipid (C).
Additional data and analyses using the reference fluorophore are given in Table 2. When measured relative to surface scatter, the analysis using Eqs. (16) and (17) shows that significant values of tL and Δt are required to account for the data. When measured relative to TRH (Eqs. (18) and (19)), the values of tL and Δt are near zero, indicating that it is no longer necessary to consider these effects when using a reference fluorophore. Given the unknown factors in time-resolved studies of tissues, we feel an internal reference will provide the most reliable lifetime measurements of implanted sensors.
Table 2.
Intensity decays of Curboxy SNARF-6 (pH 9.2) measured relative to the excitation source and to the fluorescent reference TRHa
| Scattering condition | τ or τapp (ns) | tL (ps) | Δt (ps) | χR2 |
|---|---|---|---|---|
| Relative to the excitation source | ||||
| τapp (ns) | ||||
| No intralipid | 1.09 | −18 | ≈0 | 1.3 |
| In intralipidb, 2 mm | 1.22 | 208 | 190 | 1.0 |
| In intralipid, 4 mm | 1.33 | 473 | 351 | 15.0 |
| Relative to the fluorescent reference TRH | ||||
| τ (ns) | ||||
| No intralipid | 1.17 | −33 | ≈0 | 1.2 |
| In intralipid, 2 mm | 1.14 | −46 | ≈0 | 2.7 |
| In intralipid, 4 mm | 1.18 | −22 | ≈0 | 7.0 |
Excitation at 570 mm, emission at 620 nm, 20 °C. 1.0% intralipid suspension. The Carboxy SNARF-6 was in 80 mM tris, pH 9.2.
Depth under surface of intralipid suspension.
To illustrate further the usefulness of the reference fluorophore, we attempted a multi-exponential analysis of the intensity decay of CS6 using Eqs. (5))–(9). In the absence of intralipid, the intensity decay is a double exponential (Table 3), as can be seen from the 10-fold decrease in χR2 from 6.7 to 0.6 for the two-exponential fit. However, the amplitude of the shorter 0.16–0.46 ns component is weak and contributes only 4–8% of the total intensity. In spite of this weak contribution, two decay times and amplitudes of CS6 were recovered from 2 and 4 mm of intralipid when measured using the reference fluorophore. This result suggests that even complex intensity decays can be quantitatively recovered from sensors embedded in highly scattering media.
To avoid confusion, we note that double-exponential intensity decays of CS6 were not recovered when using Eqs. (16) and (17) (Table 2). In this case there is a weak contribution of the short-lived component, and it appears that the value of tL can vary and to some extent account for the short-lived component in the decay of CS6. For instance, in the absence of intralipid (Table 2), the value tL = −18 ps is a consequence of the multi-exponential decay of CS6. An advantage of using the reference fluorophore is that the data can be analyzed using Eqs. (5)–(9), without the use of tL and Δt as additional parameters.
4.3. Non-invasive measurements of pH
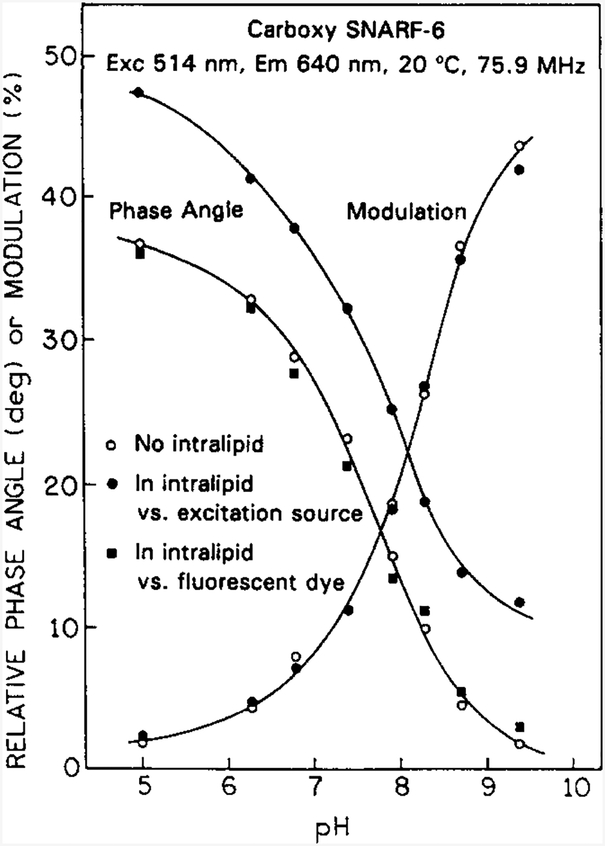
To illustrate the possibility of non-invasive pH measurements, we examined a series of CS6 solutions of various pH values. The experimental conditions were changed to 514 nm excitation and 640 nm emission, which select preferentially for excitation of the acid form and for detection of the base form of CS6 [15], and result in an apparent pKa value near 7.5 as seen from the phase angle and 8 for the modulation. However, wavelength-dependent apparent pKa, values require both forms of the sensor to be fluorescent. The calibration curve for these experimental conditions is shown in Fig. 7 (○). When measured relative to surface scatter, the phase angles (●) are larger than the true values and do not reveal the correct pH value. When measured relative to the TRH standard, the correct phase angles are observed (■), demonstrating the possibility of non-invasive pH sensing in scattering media. It is interesting to notice that for CS6 under these experimental conditions, essentially the same modulation values are measured versus surface scatter (●) as measured without intralipid (○). At this time, we do not know whether the favorable characteristics of the modulation data are transferable to tissues where the absorption coefficient may be higher and different for various depths in the tissues.
Fig. 7.
pH sensing at a 4 mm depth in 0.25% intralipid using CS6. Excitation wa the mode-locked output of an argon-ion laser at 514 nm. The emission was detected at 640 nm. Phase and modulation data were obtained without intralipid (○), in intralipid vs. the scattered fight from the surface (●) and in intralipid vs. the TRH standard (■).
5. Discussion and conclusions
The results described above demonstrate that lifetime-based sensing is possible for sensors embedded in turbid media. However, the measurements were performed using a mode-locked argon-ion laser and a picosecond dye laser light source, which is obviously too complex for clinical applications. Hence, it is natural to ask whether simple instrumentation can be made for use in the doctor’s office, at home, or even as a portable device. In our opinion, medical phase-modulation fluorometers can be constructed using currently available probe and electro-optics technology. Such a device is shown in Fig. 1, which illustrates one possible configuration for non-invasive measurements. The sensing patch may be implanted under the skin, and of course, would be permeable to the analyte of interest. The lifetime of the sensing patch could be measured using the methods described in this report. To support this claim, we note the rapid development of long-wavelength fluorophores [32–37], many of which can be excited with currently available laser diodes [35–37]. It is well known that the optical output of laser diodes can be intensity modulated to gigahertz frequencies, and that wavelengths above 620 nm readily penetrate the skin, so that laser diodes can be the light source for embedded phase-modulated sensors. Laser diodes have already been used for FD lifetime measurements [38], for FD lifetime measurements through fibers [20], and for a fiber-optic pH sensor [39]. Finally, we note that a new class of fluorescence sensors is emerging, which display long decay times over 100 ns [40,41], and that such long-lived fluorophores can be excited with simple modulated light sources such as an electroluminescent device [42]. It has even been possible to develop a sterilizable sensor using these long-lived fluorophores [43]. For these longer lifetimes, the effect of time-dependent photon migration will be less significant, and can possibly be neglected. Based on these developments, we conclude that lifetime-based sensing in tissues is clinically practical with currently available technology.
Acknowledgements
This work was supported by grants from the National Institutes of Health (RR-08119) with support for instrumentation from the NSF (DIR-8710401) and the NIH (RR-07510 and RR-10416). J.R.L. also expresses appreciation for support from the Medical Biotechnology Center at the University of Maryland.
Biographies
Henryk Szmacinski is research assistant professor of biochemistry at the University of Maryland School of Medicine. Dr Szmacinski has introduced and developed the area of lifetime-based sensing and recently has developed fluorescence lifetime imaging microscopy. These developments are resulting in practical applications of time-resolved fluorescence.
Joseph R. Lakowicz is professor of biochemistry and adjunct professor of electrical engineering at the University of Maryland School of Medicine. Dr Lakowicz also directs the Center for Fluorescence Spectroscopy, which provides time-resolved fluorescence measurements to scientists whose research projects can benefit from the measurements. He has published widely in the area of biochemical and biomedical applications of fluorescence and is the Editor-in-Chief of the Journal of Fluorescence and Journal of Biomedical Optics.
References
- [1].Wolfbeis OS (ed.), Proc. 1st Eur. Conf. Optical Chemical Sensors and Biosensors, Graz, Austria, 12–15 April, 1992, Sensors and Actuators B, 11 (1993). [Google Scholar]
- [2].Van Dyke K and Van Dyke R, Luminescence Immunoassay and Molecular Application, CRC Press, Boca Ratun, 1990. [Google Scholar]
- [3].Lakowicz JR and Thompson RB, Advances in Fluorescence Sensing Technology, SPIE Proc, Vol. 1885, 1993. [Google Scholar]
- [4].Durkin AJ, Jakumar S, Ramanujam N and Richards-Kortum R, Relation between fluorescence spectra of dilute and turbid samples, Appl. Opt, 33 (1994) 414–423. [DOI] [PubMed] [Google Scholar]
- [5].Fantini S, Franceschini MA, Flshkin JB, Barbieri B and Granon E, Quantitative determination of the absorption spectra of chromophores in strongly scattering media: a light-emitting-diode based technique, Appl. Opt, 33 (1994) 5204–5213. [DOI] [PubMed] [Google Scholar]
- [6].Lakowicz JR, Szmacinski H, Nowaczyk K, Lederer WJ, Kirby MS and Johnson ML, Fluorescence lifetime imaging of intracellular calcium in COS cells using Quln-2, Cell Calcium, 15 (1994) 7–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Wang XF, Periasamy A, Gordon GW, Wodnicki P and Herman B, Fluorescence lifetime imaging microscopy (FLIM) and its applications, SP1E Proc, Vol. 2137, 1994, pp. 64–76. [Google Scholar]
- [8].Gadella TWJ, Jovin TM and Clegg RM, Fluorescence lifetime imaging microscopy (FLIM): Spatial resolution of microstructures on the nanosecond time scale, Biophys. Chem, 48 (1993) 221–239. [Google Scholar]
- [9].Lippitsch ME and Draxler S, Luminescence decay-time-based optical sensors: Principles and problems, Sensors and Actuators B, 11 (1993) 97–101. [Google Scholar]
- [10].Szmacinski H and Lakowicz JR, Lifetime-based sensing, in Topics in Fluorescence Spectroscopy, Vol. 4, Probe Design and Chemical Sensing, Plenum Press, New York, 1994, pp. 295–334. [Google Scholar]
- [11].Lakowicz JR and Szmacinski H, Fluorescence lifetime-based sensing of pH, Ca2+, K+ and glucose, Sensors and Actuators B, 11 (1993) 133–143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Miyoshi N, Hara K, Kimmura S, Nakanishi K and Fukuda M, A new method of determining intracellular free Ca2+ concentration using Quin-2 fluorescence, Photochem. Photobiol, 53 (1991) 415–418. [DOI] [PubMed] [Google Scholar]
- [13].Hirshfield KM, Toptygin D, Packard BS and Brand L, Dynamic fluorescence measurements of two-state systems: applications to calcium-chelating probes, Anal. Biochem, 209 (1993) 209–218. [DOI] [PubMed] [Google Scholar]
- [14].Lippitsch ME and Draxler S, A family of lifetime sensors for medical purposes, SPIE Proc, Vol. 2388, 1995, pp. 132–137. [Google Scholar]
- [15].Szmacinski H and Lakowicz JR, Optical measurements of pH using fluorescence lifetimes and phase-modulation fluorometry, Anal. Chem, 65 (1993) 1668–1674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Lakowicz LR and Maliwal B, Optical sensing of glucose using phase-modulation fluorimetry, Anal. Chim. Actu, 271 (1993) 155–164. [Google Scholar]
- [17].Ozinskas AJ, Malak H, Joshi J, Szmacinski H, Britz J, Thompson RB, Koen PA and Lakowicz JR, Homogeneous model immunoassay of thyroxine by phase-modulation fluorescence spectroscopy, Anal Biochem, 213 (1993) 264–270. [DOI] [PubMed] [Google Scholar]
- [18].Diamandis EP, Immunoassay with time-resolved fluorescence spectroscopy: principles and applications, Clin. Biochem, 21 (1988) 139–150. [DOI] [PubMed] [Google Scholar]
- [19].Pinsky BG, Ladasky JJ, Lakowicz JR, Berndt K and Hoffman RA, Phase-resolved fluorescence lifetime measurements for flow cytometry, Cytometry, 14 (1993) 123–135. [DOI] [PubMed] [Google Scholar]
- [20].Thompson RB, Frisoli JK and Lakowicz JR, Phase fluorometry using a continuously modulated laser diode, Anal. Chem, 64 (1992) 2075–2078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Burch CL, Lakowicz JR and Sevick-Muraca EM, Fluorescence lifetime-based sensing in tissues: a computational study, Biophys. J, 68 (1995) 1574–1582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Lakowicz JR and Gryczynski I, Frequency-domain spectroscopy, in Topics in Fluorescence Spectroscopy, Vol. 1, Techniques, Plenum Press, New York, 1991, pp. 293–335. [Google Scholar]
- [23].Gratton E, Limkeman M, Lakowicz JR, Maliwal BP, Cherek H and Laczko G, Solution of mixtures of fluorophores using variable-frequency phase and modulation data, Biophys. J, 46 (1984) 479–486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Laczko G, Gryczynski I, Gryczynski Z, Wiczk W, Malak H and Lakowicz JR, A 10 GHz frequency-domain fluorometer, Rev. Sci. lnstrum, 61 (1990) 2331–2337. [Google Scholar]
- [25].Patterson MS, Chance B and Wilson BC, Time-resolved reflectance and transmittance for the non-invasive measurement of tissue optical properties, Appl. Opt, 28 (1989) 2331–2336. [DOI] [PubMed] [Google Scholar]
- [26].Bonner RF, Nossal R, Havlin S and Weiss GH, Model for photon migration in turbid biological media, J. Opt. Soc. Am, 4 (1987) 423–432. [DOI] [PubMed] [Google Scholar]
- [27].Chance B, Nioka S, Kent J, McCully K, Fountain M, Greenfeld R and Holtom G, Time-resolved spectroscopy of hemoglobin and myoglobin in resting and ischemic muscle, Anal. Biochem, 174 (1988) 698–707. [DOI] [PubMed] [Google Scholar]
- [28].Patterson MS, Moulton JD, Wilson BC, Berndt KW and Lakowicz JR, Frequency-domain reflectance for the determination of the scattering and absorption properties of tissue, Appl. Opt, 30 (1991) 4474–4476. [DOI] [PubMed] [Google Scholar]
- [29].Lakowicz JR and Berndt K, Frequency-domain measurements of photon migration in tissues, Chem. Phys. Lett, 166 (1990) 246–252. [Google Scholar]
- [30].Lakowicz JR, Cherek H and Baiter A, Correction of timing errors in photomultiplier tubes used in phase-modulation fluorometry, J. Biochem. Biophys. Methods, 5 (1981) 131–146. [DOI] [PubMed] [Google Scholar]
- [31].Lakowicz JR and Baiter A, Theory of phase-modulation fluorescence spectroscopy for excited-state processes, Biophys. Chem, 16 (1982) 99–115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Le Bris M-T, Mngnier J, Bourson J and Valeur B, Spectral properties of a new fluorescent dye emitting in the red: a benzoxazinone derivative, Chem. Phys. Lett, 106 (1984) 124–127. [Google Scholar]
- [33].Southwick PL, Ernst LA, Tauriello EW, Parker SR, Mujumdar RB, Mujumdar SR, Clever HA and Waggoner AS, Cyanine dye labeling reagents - carbocymethylindocyanine succinimidyl esters, Cytometry, 11 (1990) 418–430. [DOI] [PubMed] [Google Scholar]
- [34].Ishchenko AA, Svidro VA and Dervyanko NA, Characteristics of the fluorescence spectra of asymmetric polymethine dyes, Opt. Spectrosc. (USSR), 71 (1991) 266–269. [Google Scholar]
- [35].Terpetschnig E, Szmacinski H and Lakowicz JR, Synthesis, spectral properties and photostabilities of symmetrical and unsymmetrical squarines; a new class of fluorophores with long-wavelength excitation and emission, Anal. Chim. Acta, 282 (1993) 633–641. [Google Scholar]
- [36].Terpetschnig E, Szmacinski H, Ozinskas A and Lakowicz JR, Synthesis of squaralne-N-hydroxysuccinimide esters and their biological application as long-wavelength fluorescent labels, Anal. Biochem, 217 (1994) 197–204. [DOI] [PubMed] [Google Scholar]
- [37].Boyer AE, Lipowska M, Zen J-M, Patonay G and Tsang VCW, Evaluation of near infrared dyes as labels for immunoassays utilizing laser diode detection: development of near infrared dye immunoassay (NIRDIA), Anal. Lett, 25 (1992) 415–428. [Google Scholar]
- [38].Berndt KW, Gryczynski I and Lakowicz JR, Phase-modulation fluorometry using a frequency-doubled pulsed laser diode light source, Rev. Sci. Instrum, 61 (1990) 1816–1820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Thompson RB and Lakowicz JR, Fiber optic pH sensor based on phase fluorescence lifetimes, Anal. Chem, 65 (1993) 853–856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Lakowicz JR, Terpetschnig E, Szmacinski H and Malak H, Metal-ligand complexes as a new class of long-lived fluorophores for protein hydrodynamics, Biophys. J, 68 (1995) 342–350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Terpetschnig E, Szmacinski H and Lakowicz JR, Fluorescence polarization immunoassay of a high molecular weight antigen based on a long-lifetime Ru-ligand complex, Anal. Biochem, 227 (1995) 140–147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Bemdt KW and Lakowicz JR, Electroluminescent lamp-based phase fluorometer and oxygen sensor, Anal. Biochem, 201 (1992) 319–325. [DOI] [PubMed] [Google Scholar]
- [43].Bambot SB, Holavanahali R, Lakowicz JR, Carter GM and Rao G, Phase fluorometric sterilizable optical oxygen sensor, Biotechnol. Bioeng, 43 (1994) 1139–1145. [DOI] [PubMed] [Google Scholar]