Graphical abstract
The coronavirus disease (COVID-19) caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) has been declared a pandemic. Global research updates confirm that the infected patients manifest a range of clinical symptoms and sometimes remain entirely asymptomatic, posing a greater threat to the people coming in contact. Despite several case reports coming up every day, our knowledge about the neurotropic mechanism of the SARS-CoV-2, immunological responses, and the mode of disease progression and mechanism of cross-talk between the central nervous system (CNS), heart, lungs, and other major organs is not complete. Report of anosmia, ataxia, dysgeusia, and altered psychological status of the infected COVID-19 patients offers some clue to the possible route of viral entry and multiplication. In this review, we have critically assessed the involvement of CNS dysregulation in COVID-19 patients. The probable mechanism of immunological responses, the impairment of the coagulation pathway, the onset of cytokine storm, its interplay with the HPA axis, and hypoxia are discussed in detail here. Based on the latest research findings and some case reports of hospitalized COVID-19 patients, it is evident that the CNS involvement in disease progression is alarming. Accurate and timely detection of viral load in CNS is necessary to allow prompt and effective treatment modalities.
Possible entry sites of SARS-CoV-2 to the central nervous system of human being and the downstream manifestations.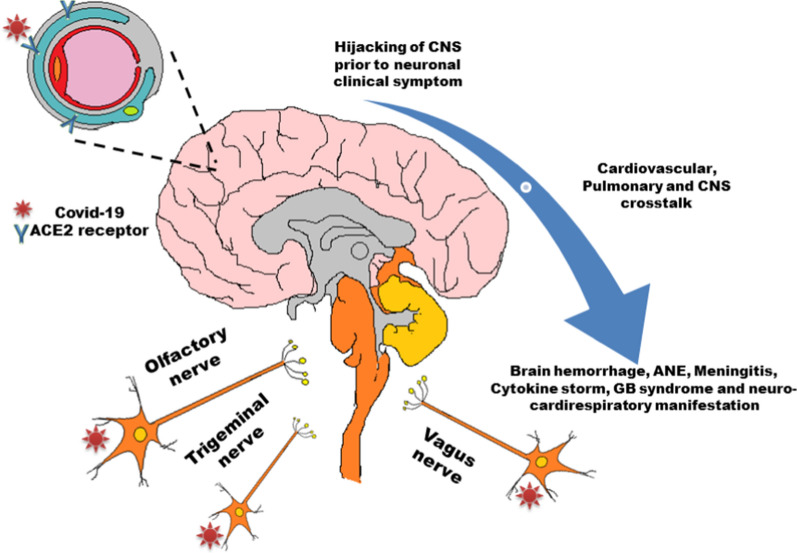
Keywords: COVID-19, central nervous system, neurotropic mechanism, ACE2 receptor, cytokine storm, coagulation pathway
Introduction
The battle against global pandemic severe acute respiratory syndrome (SARS) was not over past 2003, and we are now again in an all-inclusive threat by a novel coronavirus COVID-19 or SARS-CoV-2 since its first outbreak in December 2019 in Wuhan, China (Zhou et al. 2020a, b). The death toll has crossed a few million worldwide and about one-third of the patients are reported to have serious neurological complications (Mao et al. 2020). The real concern about the disease spread is the extremely contagious nature of SARS-CoV-2 compared to the previous strains, viz., SARS-CoV and the Middle East respiratory syndrome (MERS-CoV) (Sahin et al. 2020). Despite the resolute efforts of the researchers, there is still no clue to arrest the pandemic situation. In December 2019, during the initial commencement of COVID-19, it was not considered beyond a severe respiratory disease mainly affecting the pulmonary system with general symptoms of flu including high fever, cough, even pneumonia, but as of April 2020, approximately 34% of hospitalized patients along with sporadic cases were reported with neurological manifestations (Poyiadji et al. 2020; Natoli et al. 2020). With the progression of the epidemic, cases have come to notice where patients did not show breathing or any other respiration related problem instead developed a far severe illness to the central nervous system (CNS) (Moriguchi et al. 2020). The growing evidences of brain hemorrhage in COVID-19 patients question the neurotropic potential of SARS-CoV-2 and a systematic susceptibility of this novel coronavirus (Sharifi-Razavi et al. 2020). From case studies, it has also been observed that the invasion of SARS-CoV-2 into the brain may remain asymptomatic for quite a long time (Moriguchi et al. 2020). Interestingly, the investigators have suggested not ignoring any simple symptoms like loss of taste and/or smell at a very early stage of the COVID-19 attack. At this learning phase of host-COVID-19 interaction, even a minute sign of anosmia and hypogeusia can appear to be proof of viral entry in the olfactory bulb (Baig 2020). The rigorous research for the knowledge on the host receptors of SARS-CoV-2 revealed the similarity with that of SARS-CoV, namely angiotensin-converting enzyme-2 (ACE2), with more high-affinity binding for SARS-CoV-2 (Wrapp et al. 2020). Further, the distribution studies of the ACE2 receptor evidenced the multi-organ infection capability of novel coronavirus (Li et al. 2020b). Starting from a mild fever with a headache, the virus can abode the respiratory system, CNS, peripheral nervous system along with the gastrointestinal tract and moving far to the renal system, it can hijack the cardiovascular system resulting in multi-organ failure leading to death (Guan et al. 2020). Thus, COVID-19 reports noted to date projected the synergistic disruption of body homeostasis combining pulmonary, circulation, digestive, renal, and cardiac system along with neurological dysfunction. Importantly, Baig et al. argued that the leading cerebral edema caused by SARS-CoV-2 infection could end up with mortality alone prior to a systemic homeostasis imbalance (Baig et al. 2020). This implies the importance of noticing any neurological manifestation in patients with the first line of priority to combat the systemic breakdown.
Here in this review, we summarize the intense impact of COVID-19 to CNS for the betterment of basal level diagnosis of patients and finding CNS drugs to be effective against the virus. We also discuss the probable mechanism of viral entry and multiplication in different parts of the brain, immunological response to evaluate an accurate CNS infection status by COVID-19. Moreover, this may enlighten the systemic and chronic treatment of this novel respiratory severe syndrome.
COVID-19 and pathological manifestations
SARS-CoV-2 mainly belongs to the Betacoronavirus genus of the family Coronaviridae and has four different genera identified so far, viz., alpha, beta, gamma, and delta-coronavirus. Genome sequence analysis of this novel coronavirus revealed that they have single-stranded RNA (ssRNA) of 29.8 kb to 29.9 kb comprising 14 open reading frames (ORFs) encoding for 27 proteins. At the 5′-end, the genome comprises of ORF1 and ORF2 encoding for 15 important non-structural proteins for replication of virus while the 3′-end consists of structural proteins like a spike (S), envelope (E), membrane (M), and nucleocapsid (N) proteins along with eight accessory proteins (Malik et al. 2020; Wu et al. 2020a). A comparative analysis of SARS-CoV-2 and its earlier strains MERS-CoV and SARS-CoV is presented in table 1. In the light of genome analysis, it has also been highlighted that substitution of 380 amino acid between these identical CoVs are accumulated in the non-structural protein genes, whereas 27 mutations were mostly located in S protein genes required for entry as well as receptor binding of the cell (Wu et al. 2020a; Benvenuto et al. 2020).
Table 1.
Phylogenetic, epidemiological, and pathogenetic characteristics of SARS-CoV-2, MERS-CoV, and SARS-CoV
| Characteristics | SARS-CoV-2 | MERS-CoV | SARS-CoV |
|---|---|---|---|
| Phylogeny | Clade 1, cluster 2a | Clade 2 | Clade 1, cluster 2b |
| Reproductive number (R0) | 2–2.5 | 0.7 | 1.7–1.9 |
| Primary host | Bats | Bats | Bats |
| Intermediate host | Unknown (pangolins) | Camels | Palm civets |
| Receptor | Angiotensin converting enzyme 2 (ACE2) | Aminopeptidase-N; Dipeptidyl peptidase 4 (DPP4) | Angiotensin converting enzyme 2 (ACE2) |
The COVID-19 is mainly characterized by the broad spectrum of clinical manifestations ranging from respiratory syndrome to upper respiratory tract distress, sepsis, and finally causing acute respiratory distress syndrome (ARDS) (Chen et al. 2020a, b; Wang et al. 2020a; Liu et al. 2020). This new species of CoVs, namely SARS-CoV-2, undergoes rapid and frequent mutations and recombination and thereby readily crosses the species barrier causing frequent novel cross-species infections (Su et al. 2016). SARS-CoV-2 is transmitted between humans through direct and indirect contacts from both symptomatic and asymptomatic patients (Wu et al. 2020b). In fact, the asymptomatic viral propagation in the host body is of more concern for the pathological implications of COVID-19 (Wang et al. 2020c).
Host receptor-virus interaction: The gateways of SARS-CoV-2 entry
Based on the current research outcomes, it has been proved that SARS-CoV-2 and structurally identical SARS-CoV mainly exploit the same binding receptor ACE2 to enter into the host cell, unlike MERS-CoV that uses aminopeptidase-N and dipeptidyl peptidase-4 (DPP4) for entry to the host cells (Shang et al. 2020; Wan et al. 2020). The novel coronavirus binds compactly with the four residues from 482 to 485 present in the ridge of ACE2, thereby enhancing the binding intensity of novel coronavirus towards ACE in comparison to SARS-CoV. In addition, the two hot spots, namely, hot spot-31 and hot spot-353 of the ACE2, are found to be remarkably stabilized by the receptor-binding domain (RBD) of SARS-CoV-2 in comparison to previous CoVs. However, no substitution for an amino acid in RBD was noticed for SARS-CoV-2, which may undergo direct interaction with ACE2 compared to SARS-CoV, though six mutations were observed in other places of RBD (Baig et al. 2020). On the other hand, receptor affinity analysis indicated that this nCoV more efficiently binds with ACE2 compared to the other strains of SARS-CoV known earlier (Wan et al. 2020). ACE2, an ectoenzyme, mostly remains attached to the cellular plasma membrane of several tissues, such as heart, kidney, gastrointestinal tract, and lower respiratory tract (Imai et al. 2010). In brain tissue, the ACE2 mRNA is expressed in the following order of abundance: nucleus accumbens of ventral striatum > posterior hypothalamus > anterior hypothalamus > cortex > hippocampus > cerebellum > spinal cord > medulla oblongata (Harmer et al. 2002). Abundant ACE2 receptors have been detected over glial cells and neurons. Interestingly, newly discovered single-cell RNA sequencing technology empowered us to study the expression of ACE2 with quantitative details at a single-cell resolution (Zhao et al. 2020). Recent RNA expression profiling of ACE2 receptors in the human lungs predicted the distribution of ACE2 receptors over a small population of type-II alveolar cells (AT2) (Zhao et al. 2020). Moreover, the targeted population of AT2 cells has been hijacked cleverly by notorious SARS-CoV-2 for the process of reproduction and transmission. Furthermore, the distribution of ACE2 expressing cell receptors and its specific number in various tissues could be a potential tool for identifying the susceptibility of different organs towards SARS-CoV-2 shortly (Zou et al. 2020). To maintain collinearity, several studies explored the heterogeneity of expression of ACE2 receptors in the specific tissue at a single-cell level (Lin et al. 2020). However, all these profound researches only analyzed the mRNA level of ACE2 receptors. Indeed, the complex composition of tissue and the obtained result from single-cell RNA sequencing also cannot reflect the abundance of ACE2 receptors within tissues. Search is on to find out whether the putative ACE2 receptors of novel coronavirus can successfully edify the true abundance of these receptors within human tissue. Besides, ACE2 receptors host serine protease TMPRSS2 are actively involved in the invasion and immune response of SARS-CoV-2. TMPRSS2 plays a pivotal role in SARS-CoV-2 pathogenesis and is actively expressed in the aerodigestive tract (Xu et al. 2020). Interestingly, differences in the expression pattern of TMPRSS2 in lung cells may vary across different populations, the element implying the susceptibility towards SARS-CoV-2 infection. Thus, a more revealing fact is the expression of TMPRSS2 protein in the lung may vary between men and women and, therefore, could be an important element in understanding the differential susceptibility to SARS-CoV-2 infection. In accordance with ACE2 and serine protease TMPRSS2 receptors, recent evidences suggest that neuropilin-1 (NRP-1) and basigin may also act as a putative alternative receptor that could be potentially involved in the entry of SARS-CoV-2 and, therefore, contribute to tropism (Wang et al. 2020b). NRP-1 receptors are dimeric in structure, contributing thoroughly to neurogenesis and angiogenesis (Yasuhara et al. 2004). They generally bind to ligands containing a C-terminal polybasic motif following the C-end-Rule (CendR) (Teesalu et al. 2009). SARS-CoV-2 contains a furin cleavage site with the capability to produce a solvent-exposed C-terminus containing the CendR R/KXXR/K sequence capable of binding to the coagulation b1 domain of NRP (Teesalu et al. 2009). This observation led to the hypothesis that neuropilins could serve as co-receptor for entry of SARS-CoV-2 to the cell and contributes profusely to tropism as evidenced from previously reported viruses (Lambert et al. 2009). Daly et al. (2020) corroborated NRP-1 is strongly reduced in the case of SARS-CoV-2 infection expressed Hela cells and attributed to the fact that this co-receptor NRP-1 alone was not sufficient as cells that do not express ACE2 are not infected by SARS-CoV-2 (Daly et al. 2020). Shreds of evidence also support the involvement of basigin or CD147 towards the entry of CoV-2 in the cells. It is ubiquitously expressed, and mRNA levels are higher than ACE2 receptors in the lung (Su et al. 2004). Regardless of the fact co-morbidities associated with COVID-19 severity, CD147 is remarkably increased in the respiratory mucosa of smokers and in patients with chronic obstructive pulmonary disease (COPD) (Aguiar et al. 2020; Jouneau et al. 2011). This, in turn, signifies that CD147 plays a vital role in SARS-CoV infection. In conclusion, notable preliminary observations by patient treatment argue for comprehensive additional mechanistic studies to determine all these host receptors’ involvement in virus entry and thereby define novel tropism.
Possible neurotropic mechanism of SARS-CoV-2 and its analogy with similar viruses
Since the outbreak of COVID-19, reports evidenced the high probability of pulmonary invasion of the virus through the nasopharyngeal route (Moriguchi et al. 2020). Interestingly, the dual invasion routes of the virus to the brain, viz., host receptor binding in pericytes of the blood–brain barrier (BBB) and direct entry via intranasal pathway supports the idea of viral neurotropism. Several studies have depicted the neuroinvasive potential of Coronaviruses (CoVs), i.e., due to replication of virus the ability to transmit from the respiratory tract to CNS to trigger various neurological disorders (Desforges et al. 2019). From previous reports, other strains of SARS-CoV also have been evidenced to infect different cells of CNS, especially neuronal and glial cells (Arbour et al. 2000). Studies on the patient samples with MERS and SARS also visualized the exclusive association of SARS-CoV in neurons. Moreover, SARS was detected in the patient’s cerebrospinal fluid (CSF), indicating its potential to infringe on the extremely rigid BBB (Li et al. 2020a, b). Pathogenic viruses probably gain access to the CNS by four different means, such as- (1) Trojan Horse model where perhaps infected monocytes and CD4+ lymphocytes act as vehicles for transportation of the virus from the blood to the CNS and induce more inflammatory cells there, (2) by direct infection of BBB endothelial cells and astrocytes and subsequently these infected BBB cells either infect CNS parenchymal cells or release the free virus into the CNS, (3) transcytosis model involves internalization of the virus by endothelial cells of astrocytes which in turn transfer the virus to CNS, and (4) non-specific entry through disrupted BBB (Eliseo and Joan 2011). Apart from these four entry routes into CNS, the transynaptic transmission through the olfactory nerve via piriform complex may emerge as a major entry path for SARS-CoV-2 into the brain (Netland et al. 2008). During experimental studies by Netland et al. using transgenic mice model for hACE2 expressed under cytokeratin-18 promoter (K18-hACE2) revealed that the neurons are susceptible towards SARS‐CoV. However, experiments on transgenic mice further revealed that intranasal administration of either SARS‐CoV or MERS‐CoV could get an entry in the brain employing olfactory nerves and instantly spread to the brainstem and thalamus (Netland et al. 2008; Li et al. 2020a, b). The association of hyposmia and other neurological manifestations with that of the COVID-19 patients also explains the probable mechanism of entry of SARS-CoV-2 through olfactory nerves to neurons. Analysis of a SARS-infected deceased person showed that the brainstem was massively infected (Li et al. 2020a, b). Following these aforementioned experimental evidences of the viral entry through olfactory receptor neurons and trigeminal nerve of the nasal cavity or the vagus nerve directed towards the brain justifies their robust transportation to the CNS by the neuronal retrograde pathway (Baig et al. 2020; Zhou et al. 2020a, b). It was also documented that SARS-CoV-2 could infect endothelial cells of lymph to the nasal cavity from cervical lymph nodes to reach the CNS (Varga et al. 2020). In the case of retrograde or anterograde dissemination, viruses can transmit through the major protein, viz., kinesins, and synein leading to demyelination (Swanson and McGavern 2015). Some hospitalized COVID patients suffer from acute hypoxia, which may indirectly cause further nervous system injury (Guo et al. 2020). As a consequence of neuroinvasive nature of SARS-CoV-2 an increased risk of neurological disorders may also result.
Based on researches so far, it seems SARS-CoV-2 might follow any of the hematogenous, transcribial, and neuronal retrograde dissemination pathways (Baig et al. 2020; Li et al. 2020a, b; Zhou et al. 2020a, b). The hematogenous pathway depicts that the virus mainly passes the BBB by transcytosis through pericytes and microvascular endothelial cells of the brain. They also pass across the BBB by directly infecting the epithelial or endothelial cells present in the ventricular choroid plexus. Sluggish movement of the virus by microcirculation through brain tissues helps them interact with ACE2 receptors through anchoring, cellular entry, and multiplication (Baig et al. 2020). Moreover, the virus could be intracellularly transported by leukocytes in a concealed manner.
Effect of SARS-CoV-2 on vasculature, blood–brain barrier (BBB), and pericytes: Role of ACE2
Determination of COVID-19 effect on the vascular system along with BBB and pericytes has similarity with the riddle ‘which came first, the chicken or the egg?’ As in the case of the COVID-19 outbreak until April 2020, most of the patients showed clinical symptoms of SARS, the neurological severity of COVID-19 was not focused. Based on the cases that came afterward, neurobiologists argued for the vigilance of the four primary mechanisms of neurological injuries by COVID-19 (Wilner 2020). To date, the cause-effect correlation of the CNS capture and acute systemic illness has been approved but yet to be deciphered with clinical shreds of evidences (Baig et al. 2020). The structural commonality of vascular pericytes in the cardiac system and BBB, along with the omnipresence of ACE2 receptors, may enlighten the downstream effects of the SARS-CoV-2 on the cardio-respiratory system through CNS or vice-versa (Chen et al. 2020a, b). The pericytes control the microcirculation around the endothelial cells and thus restrict blood-borne pathogens (Zhang et al. 2020). SARS-CoV-2 exploits these systems through host ACE2 receptor-viral spike protein interaction just like SARS-CoV-1. The presence of ACE2 on pericytes of the cardiovascular system as well as BBB-associated pericytes indicates the brain invasion functionality of the virus via neuro-cardiovascular pathways. Whether SARS-CoV-2 enters the CNS through the retrograde cardio-respiratory route or others, its life-treating severity has been exposed through acute necrotizing encephalopathy (ANE) (Das et al. 2020). According to the previous reports, SARS-CoV can attack the CNS by breaching the BBB. This clinical sign of ANE indicates the pathological dissemination of BBB by SARS-CoV-2 (Li et al. 2020a, b).
Neuroplasticity impairment and neuropsychological challenges imposed by COVID-19
Neuroplasticity refers to the brain’s instantaneous response leading to continuous changes in emotion, cognition, and behavior of an individual throughout one’s life. Taking lessons from other neurotropic viruses, e.g., rabies, Herpes simplex virus (HSV), Epstein-Barr virus, and other scientists are considering the behavioral changes in COVID-19 patients (Atluri et al. 2015). A female airline worker was reported with a complaint of altered mental status and diagnosed as SARS-CoV positive later on (Lee et al. 2007). As explained by Netland et al., SARS-CoV can spread through specific neurotransmitter pathways in non-neuronal routes and hijack the normal synaptic vesicle functioning involving the neurotransmitters that lead to the impairment of neuroplasticity as well as a remarkable change in cognition and behavior (Netland et al. 2008). Apart from the direct destruction of the neuronal cells due to the multiplication of viruses, the neuroinvasive nature of SARS-CoV-2 may increase the risk of neurodegenerative diseases like Parkinson’s disease, Alzheimer’s disease, and multiple sclerosis in the long run (Toljan 2020). The chance of getting infected with SARS-CoV-2 is much higher in patients with neurodegeneration than the normal elder one (Das et al. 2020). Besides the transmission of viral load in neurotransmitter pathways, the dissemination of glutamate, acetylcholine, and serotonin or other neurotransmitters was evidenced, leading to depression in SARS-CoV-affected individuals (Lee et al. 2007). The home-quarantine condition adds another dimension to this psychological condition. Moreover, these patients had to have psychological counseling for several years past their survival for well-being (Lee et al. 2007).
Common central nervous system (CNS) drugs interacting with repurposed drugs to treat COVID-19
The cytochrome P-450 (CYP) enzymes are the essential drug-metabolizing enzyme found in the endoplasmic reticulum membranes of the hepatocyte and other tissues. CNS depressants like phenobarbitone, thiopentone, carbamazepine, phenytoin, and primidone can induce CYP-3A4 (Mora et al. 2015). Lopinavir/ritonavir and many other antiviral drugs are also subjected to CYP-3A mediated biotransformation in human liver microsomes (Katzung 2018). Thus, plasma concentrations of potential antiviral drugs against COVID-19 like remdesivir, lopinavir/ritonavir, and atazanavir are usually decreased when some antiepileptic drugs like carbamazepine, phenobarbitone, phenytoin, and primidone are co-administered with these drugs (Katzung 2018). Carbamazepine is also responsible for decreasing chloroquine phosphate and hydroxychloroquine concentrations and should not be taken together (Liverpool Drug Interaction Group 2020). Antipsychotic agents are no significant inhibitors of CYPs, and the concentration of the antiviral drugs does not interfere with the antipsychotic agents (Brunton 2018). However, plasma concentrations of some antipsychotics like haloperidol, pimozide, quetiapine, and ziprasidone are increased when administered with lopinavir /ritonavir and atazanavir due to inhibition of CYP-3A by ritonavir (Katzung 2018). Plasma chloroquine and hydroxychloroquine concentrations are triggered when co-administered with zuclopenthixol and thioridazine (Liverpool Drug Interaction Group 2020). Therefore, a judicious quotient of these antiviral, antiepileptic, and antipsychotic drugs is needed to be prescribed in any emergency outbreak of a COVID-19 patient.
Recent case reports of COVID-19 and its impact on the central nervous system (CNS)
Neurological symptoms such as delirium, impaired consciousness, anosmia, headache, and dysgeusia are the common symptoms that point out the entry of SARS-CoV-2 in the CNS. A first case was recently evidenced by meningitis or encephalitis and its association with COVID-19 (Moriguchi et al. 2020). Interestingly, the case was represented with paranasal sinusitis and sudden convulsion accompanied by generalized fatigue and fever. The patient was prescribed laninamivir and antipyretic agents from day 2 to 5. On day nine, the patient was admitted to the hospital due to unconsciousness and was evidenced by a transient generalized seizure that lasted about a minute. Although, SARS-CoV-2 RNA was absent in the nasopharyngeal swab but was showed in CSF. Besides, brain magnetic resonance imaging (MRI) depicted hyper-intensity along the right lateral ventricular wall, and remarkable changes of signal in the hippocampus and in the right mesial temporal lobe evidenced the probability of SARS-CoV-2 meningitis. The other encephalitis case was presented with common respiratory manifestations like fever, myalgia, and shortness of breath (Ye et al. 2020). However, the condition deteriorated with consciousness suddenly progressed to confusion, and the patient has undergone treatment with arbidol as well as oxygen therapy. However, no remarkable improvement in consciousness was noted. Moreover, the CSF specimen was negative for SARS-CoV-2, and patients neither suffered from bacterial nor tubercular infection. Interestingly, no immunoglobulin-M (IgM) antibody against HSV-1 and varicella-zoster was also found. Therefore, after intense observation, SARS-CoV-2 encephalitis was concluded.
As with symptoms of meningitis or encephalitis, patients contracted with COVID-19 also corroborated the necrotizing hemorrhagic encephalopathy symptoms (Poyiadji et al. 2020). This viral disease is mainly characterized by multifocal symmetric lesions with invariable involvement of the thalamus, brain stem, cerebral white matter, and cerebellum. Specifically, SARS-CoV-2 patients may exhibit ANE. Images of brain MRI revealed T2 and FLAIR hyper-intensities with evidence of hemorrhage indicated by a hypo-intense signal on gradient-echo or susceptibility-weighted images and rim enhancement post-contrast study (Poyiadji et al. 2020).
The other case of COVID-19 reported with neurological manifestations was a retrospective, observational case series in Wuhan, China (Mao et al. 2020). The case evidenced the involvement of the nervous system with the characteristic neurological manifestations of SARS-CoV-2. In the case series, 78 out of 214 patients were diagnosed with COVID-19, where neurological symptoms were observed in 36.4% of patients and common in 45.5% of patients with severe infection. In addition, the main neurological outcomes of the patients were categorized under three categories such as (1) manifestations of the central nervous system with dizziness, ataxia, headache, and seizure, (2) manifestations of the peripheral nervous system with smell, taste, and vision impairment, and (3) manifestations of injury of skeletal muscle. In addition to this case series, cases of Guillain-Barre Syndrome (GBS) have also been reported for COVID-19 patients. A case study of a 71-year-old male patient with severe paresthesia at limb extremities as well as distal weakness with rapidly developing tetraparesis was evidenced (Alberti et al. 2020). While undergoing neurological examination, the patient exhibited normal consciousness, no cranial nerve deficit, and normal plantar response. Brain computed tomography (CT) was normal, while the chest CT demonstrated multiple bilateral ground-glass opacities as well as pneumonia. SARS-CoV-2 was positive in the nasopharyngeal swab, while in the case of CSF, it was negative. Overall, all these possible findings were predicted as acute polyradiculoneuritis with prominent demyelination. In this context, the diagnosis was made according to GBS in association with COVID-19. Therefore, all these evidence-based case reports bringing the view that more autopsies of the patients, as well as isolation of SARS-CoV-2 from the glial cells, CSF, and neuronal tissue, may clarify the neurological outcomes and its impact on COVID-19 in the ongoing pandemic outbreak.
Possible immunological response after SARS-CoV-2 infection: Interplay between cytokine storm and coagulation pathway
Initially, the viral infected microglia, macrophages, and astrocytes may activate glial cells and induce local pro-inflammatory cytokines (Li et al. 2004). These activated immune cells and T-cells eventually induce other immune cells, leading to neuronal damage, apoptosis, and demyelination (Alberti et al. 2020; Li et al. 2004). The term ‘cytokine storm’ is a condition characterized by a potent activation of the immune system leading to overproduction of many active components, viz., interferons (IFN), chemokines, interleukins (IL), and tumor necrosis factor-alpha (TNF-α). The release of large amounts of pro-inflammatory cytokines, viz., IFN-α, IFN-γ, IL-1β, IL-6, IL-12, IL-18, IL-33, TNF-α, TGF-β, and several chemokines, viz., CXCL-8, 10, CCL-2, 3, and 5 at a time causes an aberrant systemic inflammatory response that attacks the body, which subsequently causes ARDS and multiple organ failure (Meduri et al. 1995; José et al. 2014). These hyperactive immune responses are also referred to as cytokine release syndrome or macrophage activation syndrome. According to previous reports, neurological insults like ANE involve an intense intracranial cytokine storm resulting in BBB disruption (Rossi 2008). Growing pieces of evidence involving brain-CT scans and MRI reports have projected a sign of cytokine storm syndrome in a subgroup of COVID-19 patients (Mehta et al. 2020; Wong et al. 2006). This cytokine storm in ANE was reported to be initiated mainly by the helper T-cells, which secrete granulocyte-macrophage colony-stimulating factor (GM-CSF) to induce IL-6 producing macrophages (Mehta et al. 2020; Toljan 2020). It was also noticed that the activation of coagulation pathways occurs simultaneously during the overproduction of pro-inflammatory cytokines worsening the negative effect of immunological response in COVID-19 patients (Tang et al. 2020). During the cytokine storm, the coagulation pathway is also impaired (Jose and Manuel 2020). Thrombin promotes clot formation by activating platelets, and the process is regulated by a negative feedback mechanism. It is the key element augmenting the inflammation (Jose and Manuel 2020). McGonagle and colleagues further described macrophage activation syndrome (secondary hemophagocytic lymphohistiocytosis) that entailed systemic hyper-inflammation in COVID-19 patients (McGonagle et al. 2020). Adding to the queue, Quin et al. reported an alteration in lymphocytes in a cohort of 452 COVID-19 patients (Quin et al. 2020). Very recently, Alberti et al. reported GBS related to COVID-19 infection, explaining acute dysimmune neuropathy involving the peripheral nervous system before the onset of pneumonia (Alberti et al. 2020). Also, Frontera and co-workers reported hospitalized COVID-19 patients having brain inflammation encephalitis with seizures caused due to hyper-reaction of the sympathetic nervous system. Some of them also lose consciousness, and others have strokes and lack a sense of smell (Dahm et al. 2016). Moreover, SARS-CoV-2 infection and a subsequent immunological debilitation might hamper the brain stem reflex that causes oxygen starvation leading to more worsening conditions (Dahm et al. 2016). All these evidences indicate a synergistic role of immunological elements that need to be deciphered to construct a fruitful treatment methodology against COVID-19.
Cytokine storm versus severe immunosuppression: The new debate
Considering the cytokine storm in SARS-CoV-2 related pathophysiology, many researchers, clinicians, and medical practitioners have recommended anti-inflammatory drugs, steroids, selective inhibitory blockade, and Janus kinase (JAK) inhibition for possible therapeutics (Mehta et al. 2020; Richardson et al. 2020). Significantly, having experience from previous pandemics,, viz., SARS and MERS corticosteroids have not been routinely prescribed in COVID-19 related lung infection (Russell et al. 2020). However, immunosuppression may emerge beneficial in hyper-inflammation. Cavagna et al. reported the effect of immunosuppressant drug calcineurin-inhibitor on the COVID patients who were suffering from systemic rheumatic disorders or went on organ transplantation (Cavagna et al. 2020). Nevertheless, they did not exclude a direct antiviral activity of calcineurin-inhibitor but observe a positive outcome of this immunosuppressant drug on COVID patients who were already prescribed this drug following an organ transplant or rheumatic disorders. The prevention of massive alveolar macrophage activation and subsequent release of pro-inflammatory cytokines due to long-term immunosuppression might appear as the reason behind this positive outcome. Recently a randomized controlled trial of IL-6 receptor blockade, viz., tocilizumab has also been approved in COVID patients with increased IL-6 level in China (Mehta et al. 2020). On the contrary, Remy and colleagues reported that despite having an elevated level of IL-6, approximately 25% of COVID patients did not show an increase in other typical pro-inflammatory markers (Remy et al. 2020). Several studies showed more concern about the lymphopenia associated with COVID-19 than the episodic cytokine storm (Huang et al. 2020; Wang et al. 2020a, b, c). Primary lymphocyte cells, including CD4+ and CD8+ T-cells, NK cells, are lost in SARS-CoV-2 infection (Zheng et al. 2020). These findings collectively support the hypothesis of the suppressed condition of adaptive and innate immunity in COVID-19 patients, which is contrary to the theory of hyper-inflammation. To find a solution to this debate, Remy et al. proposed a functional diagnosis of COVID-19 patient’s immune status via enzyme-linked immunosorbent spot (ELISpot) assay (Remy et al. 2020).
Hypothalamus-pituitary-adrenal axis and hypoxia: Decisive factors of immunological state
The immunological state of a COVID patient is also crucial for the psychological well-being of individuals. With increasing age, immunosenescence causes dysregulation of the immune system and maintains a low-grade chronic inflammatory condition, a predisposing factor to SARS-CoV-2 severity (Grolli et al. 2020). The incidence of cytokine storm can eventually amplify this predisposing factor (Grolli et al. 2020). Moreover, the decisive factors of the severe inflammatory condition include the hypothalamus–pituitary–adrenal (HPA) axis (Waszkiewicz 2020). The HPA axis functions in a negative feedback mechanism and holds homeostasis between the beneficial and harmful effects of pro-inflammatory cytokines. Following activation of the HPA axis, the hypothalamic hormone stimulates the release of adrenocorticotrophic hormone from the pituitary, which subsequently stimulates the adrenal glands to release corticosteroids (Chen et al. 2017). In a normal situation, the corticosteroid triggers negative feedback after reaching the brain and balances the stimulation of the pituitary and adrenal gland, and releases the corticosteroid itself (Chen et al. 2017). However, during the hyper-inflammation of COVID-19, the prolonged activation of the HPA axis by increased pro-inflammatory cytokines leads to the excessive release of corticosteroids (Steenblock et al. 2020). This excess amount of corticosteroids not only contributes to immune dysfunction but also influence to maintain an elevated viral load (Deek 2020; Waszkiewicz 2020).
On the other hand, hypoxia associated with COVID-19 is a significant risk factor for venous thromboembolism (Algahtani et al. 2020). Also, prolonged hypoxia following SARS-CoV-2 infection may worsen the immunothrombosis initiated by the virus (Thachil 2020). Some hospitalized COVID patients suffer from acute hypoxia, which may indirectly cause further nervous system injury (Guo et al. 2020). Jaunmuktane et al. demonstrated that SARS-CoV-2 related neurological complications resulted from the thromboembolism or thrombus formation within the brain (Jaunmuktane et al. 2020). Also, the immune response to the virus leading to damage in the brain’s blood vessel wall has been shown clearly by Jaunmuktane et al. (2020). Moreover, a few autopsy reports confirmed the neuropathological manifestations due to hypoxia and subsequent thromboembolism in COVID patients’ brains (Kantonen et al. 2020).
Altogether, these findings have proven interplay between several factors, including HPA axis, hypoxia, and immunological responses leading to a severe neuropathological condition in COVID patients.
Conclusion and future direction
Scientists and physicians have already admitted that we are just seeing the tip of the iceberg while searching for the clear clinical manifestations of COVID-19. Several intriguing questions about why some serious COVID patients do not gasp for breath despite deficient blood oxygen level or losing the sense of smell has clinicians worried. The neurotropic effect of the SARS-CoV-2 might be even more acute than is recorded. Most critically ill hospitalized patients remain either in ventilation or sedation; thus, the symptoms are not visible. Thus, an actual number of CNS infected patients might be way more than recorded. The thought-provoking fact is that the parenchyma-rich central nervous system, having decreased permeability, favors viral retention. Once the coronavirus gains access to the CNS after crossing several physiological barriers, it is tough to remove. The nerve cells also lack proteins of major histocompatibility complex, and viral clearance is only assisted by cytotoxic T-cells (Reinhold and Rittner 2017). Thus, a more accurate neurological investigation and attempts to isolate traces of viral RNA or coat proteins from glial and neuronal tissues and CSF are required to understand the mode of neuronal invasion by the virus and its effect on brain and other organs. Neuroprotective therapies, specific antivirals, and immunomodulators may help limit cytokine storm and prevent viral entry to the brain. Small molecule anticoagulants also may be beneficial to some patients. Indian traditional medicine, like herbal and Ayurvedic neuroprotective therapies, may act as a non-specific prophylactic strategy. However, to deepen the knowledge of the silent proliferation of SARS-CoV-2 in the brain, assessing the multi-organ interaction post COVID-19 infection may provide some ray of hope along with the combinatorial therapies.
Footnotes
This article is part of the Topical Collection: COVID-19: Disease Biology and Intervention.
Corresponding editor: Aurnab Ghose
References
- Aguiar JA, Tremblay BJ, Mansfield MJ, Woody O, Lobb B, Banerjee A, Chandiramohan A, Tiessen N, et al. Gene expression and in situ protein profiling of candidate SARS-CoV-2 receptors in human airway epithelial cells and lung tissue. Eur. Respir. j. 2020;56:2001123. doi: 10.1183/13993003.01123-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alberti P, Beretta S, Piatti M, Karantzoulis A, Piatti ML, Santoro P, Viganò M, Giovannelli G, et al. Guillain-Barré syndrome related to COVID-19 infection. Neurol. Neuroimmunol. Neuroinflamm. 2020;7:e741. doi: 10.1212/NXI.0000000000000741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Algahtani FH, AlQahtany FS, Al-Shehri A, Abdelgader AM. Features and incidence of thromboembolic disease: A comparative study between high and low altitude dwellers in Saudi Arabia. Saudi J. Biol. Sci. 2020;27:1632–1636. doi: 10.1016/j.sjbs.2020.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arbour N, Day R, Newcombe J, Talbot PJ. Neuroinvasion by human respiratory coronaviruses. J. Virol. 2000;74:8913–8921. doi: 10.1128/JVI.74.19.8913-8921.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atluri VS, Hidalgo M, Samikkannu T, Kurapati KR and Nair M 2015 Synaptic Plasticity and Neurological Disorders in Neurotropic Viral Infections. Neural plasticity2015 138979. [DOI] [PMC free article] [PubMed]
- Baig AM. Neurological manifestations in COVID-19 caused by SARS-CoV-2. CNS Neurosci. Ther. 2020;26:499–501. doi: 10.1111/cns.13372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baig AM, Khaleeq A, Ali U, Syeda H. Evidence of the COVID-19 Virus Targeting the CNS: Tissue Distribution, Host-Virus Interaction, and Proposed Neurotropic Mechanisms. ACS Chem. Neurosci. 2020;11:995–998. doi: 10.1021/acschemneuro.0c00122. [DOI] [PubMed] [Google Scholar]
- Benvenuto D, Giovanetti M, Ciccozzi A, Spoto S, Angeletti S, Ciccozzi M. The 2019-new coronavirus epidemic: Evidence for virus evolution. J. Med. Virol. 2020;92:455–459. doi: 10.1002/jmv.25688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunton LL 2018 Goodman & Gilman’s the pharmacological basis of therapeutics (McGraw-Hill Education)
- Cavagna L, Seminari E, Zanframundo G, Gregorini M, Matteo AD, Rampino T, Montecucco C, Pelenghi S, et al. Calcineurin Inhibitor-Based Immunosuppression and COVID-19: Results from a Multidisciplinary Cohort of Patients in Northern Italy. Microorganisms. 2020;8:977. doi: 10.3390/microorganisms8070977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L, Deng H, Cui H, Fang J, Zuo Z, Deng J, Li Y, Wang X, et al. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 2017;9:7204–7218. doi: 10.18632/oncotarget.23208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L, Li X, Chen M, Feng Y, Xiong C. The ACE2 expression in human heart indicates new potential mechanism of heart injury among patients infected with SARS-CoV-2. Cardiovascular Research. 2020;116:1097–1100. doi: 10.1093/cvr/cvaa078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dahm T, Rudolph H, Schwerk C, Schroten H, and Tenenbaum T 2016 Neuroinvasion and inflammation in viral central nervous system infections. Mediators Inflamm. 2016 8562805-16 [DOI] [PMC free article] [PubMed]
- Das G, Mukherjee N, Ghosh S. Neurological Insights of COVID-19 Pandemic. ACS Chem. Neurosci. 2020;11:1206–1209. doi: 10.1021/acschemneuro.0c00201. [DOI] [PubMed] [Google Scholar]
- Daly JL, Simonetti B, Klein K, Chen KE, Williamson MK, Antón-Plágaro C, Shoemark DK, Simón-Gracia L, et al. Neuropilin-1 is a host factor for SARS-CoV-2 infection. Science. 2020;370:861–865. doi: 10.1126/science.abd3072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deek SA 2020 Chronic exposure to air pollution implications on COVID-19 severity. Medical Hypotheses145 110303 [DOI] [PMC free article] [PubMed]
- Desforges M, Le Coupanec A, Dubeau P, Bourgouin A, Lajoie L, Dubé M, Talbot PJ. Human Coronaviruses and Other Respiratory Viruses: Underestimated Opportunistic Pathogens of the Central Nervous System? Viruses. 2019;12:14. doi: 10.3390/v12010014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eliseo AE and Joan WB 2011 Mechanisms of viral and cell entry into the central nervous system (Oxford University Press)
- Grolli RE, Mingoti ME, Bertollo AG, Luzardo AR, Quevedo J, Réus GZ and Maria Z 2020 Impact of COVID-19 in the mental health in elderly: psychological and biological updates. [DOI] [PMC free article] [PubMed]
- Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H et al. China Medical Treatment Expert Group for Covid-19 2020 Clinical Characteristics of Coronavirus Disease 2019 in China. New Eng. J. Med.382 1708–1720 [DOI] [PMC free article] [PubMed]
- Guo YR, Cao QD, Hong ZS, Tan YY, Chen SD, Jin HJ, Tan KS, Wang DY, et al. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak-an update on the status. Military Med. Res. 2020;7:11. doi: 10.1186/s40779-020-00240-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harmer D, Gilbert M, Borman R, Clark KL. Quantitative mRNA expression profiling of ACE 2, a novel homologue of angiotensin converting enzyme. FEBS Letters. 2002;532:107–110. doi: 10.1016/S0014-5793(02)03640-2. [DOI] [PubMed] [Google Scholar]
- Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imai Y, Kuba K, Ohto-Nakanishi T and Penninger JM 2010 Angiotensin-converting enzyme 2 (ACE2) in disease pathogenesis. Circ J74 405–410 [DOI] [PubMed]
- Jaunmuktane Z, Mahadeva U, Green A, Sekhawat V, Barrett NA, Childs L, Shankar Hari M, Thom M, et al. Microvascular injury and hypoxic damage: emerging neuropathological signatures in COVID-19. Acta Neuropathologica. 2020;140:397–400. doi: 10.1007/s00401-020-02190-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jose RJ and Manuel A 2020 COVID-19 cytokine storm: the interplay between inflammation and coagulation. Lancet Respir. Med. e46-e47 [DOI] [PMC free article] [PubMed]
- José RJ, Williams AE, Chambers RC. Proteinase-activated receptors in fibroproliferative lung disease. Thorax. 2014;69:190–192. doi: 10.1136/thoraxjnl-2013-204367. [DOI] [PubMed] [Google Scholar]
- Jouneau S, Khorasani N, Souza DE, P, Macedo P, Zhu J, Bhavsar PK and Chung KF, EMMPRIN (CD147) regulation of MMP-9 in bronchial epithelial cells in COPD. Respirology. 2011;16:705–712. doi: 10.1111/j.1440-1843.2011.01960.x. [DOI] [PubMed] [Google Scholar]
- Kantonen J, Mahzabin S, Mäyränpää MI, Tynninen O, Paetau A, Andersson N, Sajantila A, Vapalahti O et al. 2020 Neuropathologic features of four autopsied COVID-19 patients. Brain Pathology 1–5 [DOI] [PMC free article] [PubMed]
- Katzung BG 2018 Basic & Clinical Pharmacology (McGraw-Hill Education)
- Lambert S, Bouttier M, Vassy R, Seigneuret M, Petrow-Sadowski C, Janvier S, Heveker N, Ruscetti FW, et al. HTLV-1 uses HSPG and neuropilin-1 for entry by molecular mimicry of VEGF165. Blood. 2009;113:5176–5185. doi: 10.1182/blood-2008-04-150342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee AM, Wong JG, McAlonan GM, Cheung V, Cheung C, Sham PC, Chu CM, Wong PC, et al. Stress and psychological distress among SARS survivors 1 year after the outbreak. Can. J. Psychiatry. 2007;52:233–240. doi: 10.1177/070674370705200405. [DOI] [PubMed] [Google Scholar]
- Li YC, Bai WZ, Hashikawa T. Response to Commentary on ‘The neuroinvasive potential of SARS-CoV-2 may play a role in the respiratory failure of COVID-19 patients. J. Med. Virol. 2020;92:707–709. doi: 10.1002/jmv.25824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y, Fu L, Gonzales DM, Lavi E. Coronavirus neurovirulence correlates with the ability of the virus to induce proinflammatory cytokine signals from astrocytes and microglia. J. Virol. 2004;78:3398–3406. doi: 10.1128/JVI.78.7.3398-3406.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li MY, Li L, Zhang Y, Wang XS. Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues. Infect Dis. Poverty. 2020;9:45. doi: 10.1186/s40249-020-00662-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin W, Hu L, Zhang Y, Ooi J, Meng T and Jin P 2020 Single-cell analysis of ACE2 expression in human kidneys and bladders reveals a potential route of 2019-nCoV infection. bioRxiv [DOI] [PMC free article] [PubMed]
- Liu K, Fang YY, Deng Y, Liu W, Wang MF, Ma JP, Xiao W, Wang YN, et al. Clinical characteristics of novel coronavirus cases in tertiary hospitals in Hubei Province. Chin. Med. J. 2020;133:1025–1031. doi: 10.1097/CM9.0000000000000744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malik YS, Sircar S, Bhat S, Khan S, Dhama K, Dadar M, Tiwary R, Chaicumpa W. Emerging novel coronavirus (2019-nCoV) current scenario, evolutionary perspective based on genome analysis and recent developments. Vet Q. 2020;40:68–76. doi: 10.1080/01652176.2020.1727993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao L, Jin H, Wang M, Hu Y, Chen S, He Q, Chang J, Hong C et al. 2020 Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol.77 683–690 [DOI] [PMC free article] [PubMed]
- McGonagle D, Sharif K, Regan A and Bridgewood C 2020 Interleukin-6useinCOVID-19pneumonia related macrophage activation syndrome. Autoimmun. Rev.19 102537 [DOI] [PMC free article] [PubMed]
- Meduri GU, Kohler G, Headley S, Stentz F, Postlethwaite A. Inflammatory cytokines in the BAL of patients with ARDS. Persistent elevation over time predicts poor outcome. Chest. 1995;108:1303–1314. doi: 10.1378/chest.108.5.1303. [DOI] [PubMed] [Google Scholar]
- Mehta P, McAuley DF, Brown M, Sanchez E, Tattersall RS, Manson JJ. COVID-19: consider cytokine storm syndromes and immunosuppression. Lancet. 2020;395:1033. doi: 10.1016/S0140-6736(20)30628-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mora F, Molina JD, Zubillaga E, López-Muñoz F, Cecilio Á. CYP450 and its implications in the clinical use of antipsychotic drugs. Clin. Exp. Pharmacol. 2015;5:3. [Google Scholar]
- Moriguchi T, Harii N, Goto J, Harada D, Sugawara H, Takamino J, Ueno M, Sakata H, et al. A first case of meningitis/encephalitis associated with SARS-coronavirus-2. Int. J. Infectious Diseases. 2020;94:55–58. doi: 10.1016/j.ijid.2020.03.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Natoli S, Oliveira V, Calabresi P, Maia LF, Pisani A. Does SARS-Cov-2 invade the brain? Translational lessons from animal models. Eur. J. Neurol. 2020;27:1764–1773. doi: 10.1111/ene.14277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Netland J, Meyerholz DK, Moore S, Cassell M, Perlman S. Severe acute respiratory syndrome coronavirus infection causes neuronal death in the absence of encephalitis in mice transgenic for human ACE2. J. Virol. 2008;82:7264–7275. doi: 10.1128/JVI.00737-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poyiadji N, Shahin G, Noujaim D, Stome M, Patel S, Griffith B. COVID-19-associated acute hemorrhagic necrotizing encephalopathy: CT and MRI features. Radiology. 2020;296:E119–E120. doi: 10.1148/radiol.2020201187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quin C, Zhou L, Hu Z, Zhang S, Peng L, Wang HH, Luo M, Chen L, et al. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin. Infect. Dis. 2020;71:762–768. doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reinhold AK, Rittner HL. Barrier function in the peripheral and central nervous system-a review. Pflugers Arch. 2017;469:123–134. doi: 10.1007/s00424-016-1920-8. [DOI] [PubMed] [Google Scholar]
- Remy KE, Mazer M, Striker DA, Ellebedy AH, Walton AH, Unsinger J, Blood TM, Mudd PA et al. 2020 Severe immunosuppression and not a cytokine storm characterizes COVID-19 infections. JCI Insight5 e140329 [DOI] [PMC free article] [PubMed]
- Richardson P, Griffin I, Tucker C, Smith D, Oechsle O, Phelan A, Rawling M, Savory E, et al. Baricitinib as potential treatment for 2019-nCoV acute respiratory disease. Lancet. 2020;395:e30–31. doi: 10.1016/S0140-6736(20)30304-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossi A. Imaging of acute disseminated encephalomyelitis. Neuroimaging Clinics. 2008;18:149–161. doi: 10.1016/j.nic.2007.12.007. [DOI] [PubMed] [Google Scholar]
- Russell CD, Millar JE, Baillie JK. Clinical evidence does not support corticosteroid treatment for 2019-nCoV lung injury. Lancet. 2020;395:473–475. doi: 10.1016/S0140-6736(20)30317-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahin AR, Erdogan A, Agaoglu PM, Dineri Y, Cakirci AY, Senel ME, Okyay RA, Tasdogan AM. 2019 novel coronavirus (COVID-19) outbreak: A review of the current literature. Eurasian J. Med. Oncol. 2020;4:1–7. [Google Scholar]
- Shang J, Ye G, Shi K, Wan Y, Luo C, Aihara H, Geng Q, Auerbach A, et al. Structural basis of receptor recognition by SARS-CoV-2. Nature. 2020;581:221–224. doi: 10.1038/s41586-020-2179-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharifi-Razavi A, Karimi N and Rouhani N 2020 COVID-19 and intracerebral haemorrhage: causative or coincidental? New Microbes New Infect.35 100669 [DOI] [PMC free article] [PubMed]
- Steenblock C, Todorov V, Kanczkowski W, Eisenhofer G, Schedl A, Wong ML, Licinio J, Bauer M, et al. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and the neuroendocrine stress axis. Mol. Psychiatry. 2020;25:1611–1617. doi: 10.1038/s41380-020-0758-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su AI, Wiltshire T, Batalov S, Lapp H, Ching KA, Block D, Zhang J, Soden R, et al. A gene atlas of the mouse and human protein-encoding transcriptomes. Proc. Natl. Acad. Sci. USA. 2004;101:6062–6067. doi: 10.1073/pnas.0400782101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su S, Wong G, Shi W, Liu J, Lai ACK, Zhou J, Liu W, Bi Y, et al. Epidemiology, genetic recombination, and pathogenesis of coronaviruses. Trends Microbiol. 2016;24:490–502. doi: 10.1016/j.tim.2016.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swanson PA, McGavern DB. Viral diseases of the central nervous system. Curr. Opin. Virol. 2015;11:44–54. doi: 10.1016/j.coviro.2014.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang N, Li D, Wang X, Ziyong S. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020;18:844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teesalu T, Sugahara KN, Kotamraju VR, Ruoslahti E. C-end rule peptides mediate neuropilin-1-dependent cell, vascular, and tissue penetration. Proc. Natl. Acad. Sci. USA. 2009;106:16157–16162. doi: 10.1073/pnas.0908201106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thachil J. Hypoxia—An overlooked trigger for thrombosis in COVID-19 and other critically ill patients. J. Thromb. Haemost. 2020;18:3109–3110. doi: 10.1111/jth.15029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toljan K. Letter to the editor regarding the viewpoint ‘Evidence of the COVID-19 virus targeting the CNS: Tissue distribution, host−virus interaction, and proposed neurotropic mechanism. ACS Chem. Neurosci. 2020;11:1192–1194. doi: 10.1021/acschemneuro.0c00174. [DOI] [PubMed] [Google Scholar]
- Varga Z, Flammer AJ, Steiger P, Haberecker M, Andermatt R, Zinkernagel AS, Mehra MR, Schuepbach RA, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan Y, Shang J, Graham R, Baric RS, Li F. Receptor Recognition by the Novel Coronavirus from Wuhan: an Analysis Based on Decade-Long Structural Studies of SARS Coronavirus. J. Virol. 2020;94:e00127–e220. doi: 10.1128/JVI.00127-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 2020;323:1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang K, Chen W, Zhang Z, Deng Y, Lian JQ, Du P, Wei D, Zhang Y, et al. CD147-spike protein is a novel route for SARS-CoV-2 infection to host cells. Signal Transduction and Targeted Therapy. 2020;5:283. doi: 10.1038/s41392-020-00426-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Kang H, Liu X, Tong Z. Asymptomatic cases with SARS-CoV-2 infection. J. Med. Virol. 2020;92:1401–1403. doi: 10.1002/jmv.25990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waszkiewicz N 2020 Possible special needs for mental online support in female and male health care workers during the COVID-19. J. Med. Virol. [DOI] [PMC free article] [PubMed]
- Wilner AN 2020 How COVID-19 affects the brain. https://www.medscape.com/viewarticle/928903
- Wong AM, Simon EM, Zimmerman RA, Wang HS, Toh CH, Ng SH. Acute necrotizing encephalopathy of childhood: correlation of MR findings and clinical outcome. AJNR Am. J. Neuroradiol. 2006;27:1919–1923. [PMC free article] [PubMed] [Google Scholar]
- Wrapp D, Wang N, Corbett KS, Goldsmith JA, Hsieh CL, Abiona O, Graham BS, McLellan JS. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science. 2020;367:1260–1263. doi: 10.1126/science.abb2507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu A, Peng Y, Huang B, Ding X, Wang X, Niu P, Meng J, Zhu Z, et al. Genome Composition and Divergence of the Novel Coronavirus (2019-nCoV) Originating in China. Cell Host Microbe. 2020;27:325–328. doi: 10.1016/j.chom.2020.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu F, Zhao S, Yu B, Chen YM, Wang W, Song ZG, Hu Y, Tao ZW, et al. A new coronavirus associated with human respiratory disease in China. Nature. 2020;579:265–269. doi: 10.1038/s41586-020-2008-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu H, Zhong L, Deng J, Peng J, Dan H, Zeng X, Li T, Chen Q. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int. J. Oral Sci. 2020;12:8. doi: 10.1038/s41368-020-0074-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yasuhara T, Shingo T, Date I. The potential role of vascular endothelial growth factor in the central nervous system. Rev. Neurosci. 2004;15:293–307. doi: 10.1515/REVNEURO.2004.15.4.293. [DOI] [PubMed] [Google Scholar]
- Ye M, Ren Y, Lv T. Encephalitis as a clinical manifestation of COVID-19. Brain Behav. Immun. 2020;88:945–946. doi: 10.1016/j.bbi.2020.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang ZS, Zhou HN, He SS, Xue MY, Li T, Liu LM. Research advances in pericyte function and their roles in diseases. Chin. J. Traumatol. 2020;23:89–95. doi: 10.1016/j.cjtee.2020.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Y, Zhao Z, Wang Y, Zhou Y, Ma Y, Zuo W. Single-cell RNA expression profiling of ACE2, the receptor of SARS-CoV-2. Am. J. Respir. Crit. Care Med. 2020;202:756–759. doi: 10.1164/rccm.202001-0179LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng M, Gao Y, Wang G, Song G, Liu S, Sun D, Xu Y, Tian Z. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell Mol. Immunol. 2020;17:533–535. doi: 10.1038/s41423-020-0402-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou L, Zhang M, Wang J and Gao J 2020 Sars-CoV-2: Underestimated damage to nervous system. Travel Med. Infect. Dis.36 101642 [DOI] [PMC free article] [PubMed]
- Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zou X, Chen K, Zou J, Han P, Hao J, Han Z. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front. Med. 2020;14:185–192. doi: 10.1007/s11684-020-0754-0. [DOI] [PMC free article] [PubMed] [Google Scholar]


