Abstract
Background
The therapeutic management of carbapenem-resistant Acinetobacter baumannii (CR-AB) represents a serious challenge to the public health sector because these pathogens are resistant to a wide range of antibiotics, resulting in limited treatment options. The present study was planned to investigate the clonal spread of CR-AB in a clinical setting.
Methodology
A total of 174 A. baumannii clinical isolates were collected from a tertiary care hospitals in Lahore, Pakistan. The isolates were confirmed by VITEK 2 compact system and molecular identification of recA and blaOXA-51. Antimicrobial profile and the screening of carbapenem-resistant genes were carried out using VITEK 2 system and PCR, respectively. The molecular typing of the isolates was performed according to the Pasteur scheme.
Results
Of the 174 A. baumannii isolates collected, the majority were isolated from sputum samples (46.5%) and in the intensive care unit (ICU, 75%). Among these, 113/174 (64.9%) were identified as CR-AB, and 49.5% and 24.7% harbored blaOXA-23 and blaNDM-1, respectively. A total of 11 (9.7%) isolates co-harbored blaOXA-51, blaNDM-1, and blaOXA-23. Interestingly, 46.9% of the CR-AB belonged to sequence type 2 (ST2; CC1), whereas 15.9% belonged to ST1 (CC1). All of the CR-AB isolates showed extensive resistance to clinically relevant antibiotics, except colistin.
Conclusion
The study concluded CR-AB ST2 clone harboring blaOXA-23 and blaNDM-1 are widely distributed in Pakistan’s clinical settings, which could result in increased mortality. Strict compliance with the National Action Plan on Antimicrobial Resistance is necessary to reduce the impacts of these strains.
Keywords: Acinetobacter baumannii, blaOXA, blaNDM, MIC, MLST
Introduction
The development and spread of carbapenem-resistant Acinetobacter baumannii (CR-AB) represents a serious global public health dilemma.1 Extensively drug resistant A. baumannii produce resistance to all or almost all medically approved antibiotics.2 A. baumannii can cause severe infections, such as septicemia, urinary tract infections, wound infections, and ventilator-associated pneumonia, and A. baumannii has been associated with nosocomial infections.3 A. baumannii has been designated as a global priority pathogen by the World Health Organization (WHO)4 and is listed as an ESKAPE pathogen, which effectively resist to various antibacterial drugs, and restricting therapeutic options.5 Every year, over 0.7 million people die from infections due to antimicrobial-resistant (AMR) bacteria globally. By 2050, an individual is estimated to die from infection with an AMR pathogen every 3 seconds, and the global costs associated with AMR infections are expected to reach $100 trillion.6 The Centers for Disease Control and Prevention has reported that CR-AB is responsible for 700 mortality, and $281 million are spent on the therapeutic management of CR-AB infections annually.7 CR-AB develop resistance through numerous mechanisms, such as the production of β-lactam enzymes, porin proteins loss, and efflux pump overexpression.8 Infections caused by CR-AB pathogens are difficult to treat due to the acquisition of various resistant genes such as class D oxacillinase (OXA) and metallo-β-lactamase (MBL).9 The New Delhi metallo-β-lactamases (NDM) belong to a superfamily of class B MBL-producing bacteria,10 which was first identified in a Swedish patient who sought medical attention in New Delhi, India, in 2009.11 NDM pathogens are resistant to β-lactams and other antibiotic classes, including aminoglycosides and fluoroquinolones, and are only susceptible to the potentially toxic colistin and polymyxin B antibiotics.12 However, resistance to colistin has also been reported at the global level.13 The chimeric gene blaNDM-1 is derived from the fusion of aminoglycoside phosphotransferase (aphA6) and an existing blaMBL gene,14 and the promoter for ISAba125, which is encoded upstream, drives the expression of blaNDM-1. The genes blaOXA-23, blaOXA-40, and blaOXA-58 encode the most frequently identified OXAs, whereas, blaOXA-51 is intrinsic to resistant A. baumannii and can be used to identify the Acinetobacter spp.15 These pathogens continue to represent critical issues in many healthcare facilities worldwide, despite the implementation of infection control practices.16 Currently, little evidence has supported the presence of CR-AB in Pakistan. However, a recent study documented the prevalence of NDM-1-producing A. baumannii at a tertiary care hospital in Lahore.11 The aim of the present study was to determine the antimicrobial susceptibility testing, identify the most prevalent sequence types (STs), and investigation of resistant mechanism of CR-AB clinical isolates in Pakistan.
Methodology
Ethical Considerations
Before initiating this research study, ethical approval in accordance with the Declaration of Helsinki was obtained from the Ethical Review Committee, Government College University, Faisalabad. Additionally, prior to collecting the clinical samples, informed consent was obtained from each study participant. Informed consent was read to the person in the language they understand and signed appropriately. They were willing to provide a sample and utilize the isolates for the study. They were assured that the samples would be used solely for research purposes and that personal information would be kept confidential.
Clinical Isolates
A convenience sample consisting of 174 non-duplicate A. baumannii clinical isolates were collected from a tertiary care hospital in Lahore between September 2020 to December 2020. Only a single strain was collected from each case irrespective of the infection site and preserved at −80°C in Luria Bertani broth containing 16% glycerol. A. baumannii strains were primarily acquired from sputum (n=81), pus (n=42), blood (n=19), urine (n=18), throat swabs (n=7), cerebrospinal fluid (CSF) (n=3), nasal swab (n=2), seminal fluid (n=1), and high vaginal swab (n=1). The collection and transport of the clinical strains was done aseptically.
Isolate Identification by VITEK 2 System
Isolates were sub-cultured on blood and MacConkey agar (Oxoid, UK), and the plates were incubated at 37°C overnight under aerobic conditions. Isolates were identified by Gram staining and culture morphology and were confirmed using Gram-negative identification (GN) cards in the VITEK 2 compact system (bioMérieux, France).
Molecular Confirmation of A. baumannii Strains
A. baumannii were further confirmed by the detection of the intrinsic blaOXA-51 and recA genes using specific primers, as described previously. The amplicons were subjected to gel electrophoresis, followed by examination under ultraviolet (UV) light using a gel documentation instrument (Bio-Rad, UK).
Minimum Inhibitory Concentration (MIC)
An antibiogram was generated using various classes of antibiotics, such as mezlocillin, piperacillin, piperacillin/tazobactam, ampicillin/sulbactam, cefepime, cefotaxime, ceftazidime, imipenem, meropenem, gentamicin, amikacin, ciprofloxacin levofloxacin, tetracycline, tobramycin, trimethoprim/sulfamethoxazole, and colistin, in the VITEK 2 compact system. The MIC for colistin was determined using the microbroth dilution method. Interpretation of the antimicrobial susceptibility testing was carried out as per CLSI 2019 guidelines.17
Detection of Carbapenemase and MBL Phenotypes
The modified Hodge test (MHT) was performed to detect carbapenemase, as described previously.18 A 0.5 McFarland Escherichia coli ATCC 25922 turbidity standard was prepared and diluted 1:10 with sterile normal saline. The bacterial suspension was lawned on Mueller-Hinton agar (MHA) plates, and a meropenem (10 µg) disk was placed in the center. Isolates were streaked from the periphery of the disk to the edge of the plates. The MHT was considered positive if the isolates displayed clover leaf-like indentation.
MBL detection was performed using the double-disk synergy test (DDST), as described previously.19 Briefly, a 0.5 McFarland bacterial suspension was lawned on MHA plates, and 2 meropenem and 2 ertapenem disks were placed at a distance of 25 mm, and 10 µL of 5 M ethylenediaminetetraacetic acid (EDTA) solution was added to one of each meropenem (10 µg) and ertapenem (10 µg) disk. MBL production was considered positive if the EDTA disks showed a zone of inhibition 5 mm larger than those for the non-EDTA disks.
Detection of the blaNDM and blaOXA Genotypes
DNA from the clinical isolates was extracted using a commercially available bacterial genomic DNA kit (Thermo Scientific, UK). DNA purity was checked at 260 nm and 280 nm (NanoDrop Spectrophotometer, Thermo Scientific, UK). The DNA integrity was determined by electrophoresis (Bio-Rad, UK). These were following primers; blaNDM-F-5`ATGGAATTGCCCAATATTATGCAC-3` blaNDM-R-5` TCAGCGCAGCTTGTCGGC-3`, NDMV-F- 5` TGGCTTTTGAAACTGTCGCACC-3` NDMV-R-5` CTGTCACATCGAAATCGCGCGA-3`, blaOXA-23-like-F 5`GATCGGATTGGAGAACCAGA-3′ blaOXA-23-like-R-5`ATTTCTGACCGCATTTCCAT-3′, blaOXA-24- like-F-5`GGTTAGTTGGCCCCCTTAAA-3′ blaOXA-24-like-R −5`AGTTGAGCGAAAAGGGGATT-3′, blaOXA-58-like-F-5`AAGTATTGGGGCTTGTGCTG-3′, blaOXA-58-like-R-5`CCCCTCTGCGCTCTACATAC-3′, blaOXA-51-like-F-5` TAATGCTTTGATCGGCCTTG-3′ blaOXA-51-like-R-5`TGGATTGCACTTCATCTTGG-3′, blaOXA-143-F-5′ TGGCACTTTCAGCAGTTCCT-3′ blaOXA-143-R-5′-TAATCTTGAGGGGGCCAACC-3′, and recA-F-5′ACAATGACATTGCAAGCAATTG-3′ recA-R-5′CCAATTTTCATACGAATCTGG-3′. The presence of blaNDM was detected under the following conditions: initial denaturation at 95°C for 1 min, secondary denaturation at 95°C for 45 sec, annealing at 58°C for 45 sec, primary extension at 72°C for 1 min, and a final extension at 72°C for 5 min. To determine the specific genotypes, blaNDM was re-amplified using another set of primers for sequencing. Multiplex PCR was performed to sequence blaOXA-51, blaOXA-23, blaOXA-24, and blaOXA-143 under the following conditions: initial denaturation at 94°C for 5 min, secondary denaturation at 95°C for 30 sec, annealing at 52°C for 40 sec, primary extension at 72°C for 50 sec, and a final extension at 72°C for 6 min.
Multilocus Sequence Typing (MLST)
Multilocus sequence typing (MLST) was performed to determine the genetic diversity of carbapenem-resistant isolates according to the Pasteur scheme. The DNA sequences of 7 housekeeping genes (cpn60, fusA, gltA, pyrG, recA, rplB, and rpoB) were amplified, followed by purification and sequencing. The sequences were analyzed using the PubMLST database https://pubmlst.org/bigsdb?db=pubmlst_abaumannii_seqdef.
Results
Clinical Information of the Isolates
Of the 174 A. baumannii clinical isolates obtained in this study, the majority were recovered from male (64.9%) patients compared to female (35.1%) patients, with an age range from 1 year to 70 years (mean age: 35.8 years). Isolates were primarily recovered from sputum (n = 81; 46.5%), followed by pus (n = 42; 24%) and blood (n = 19; 10.9%) samples. There were 3 (1.7%) strains of A. baumannii isolates isolated from cerebrospinal fluid samples. A significant proportion of the clinical samples (n = 131; 75.2%) were obtained from patients who were admitted to the intensive care unit (ICU) followed by the male surgical ward (MSW; n = 13; 7.4%) and the female medical ward (FMW; n = 11; 6.3%). The clinical information for the recovered samples is provided in Table 1.
Table 1.
Clinical Information for the Sources of A. baumannii Isolates (n = 174)
| Clinical Information | n (%) |
|---|---|
| Sex | |
| Male | 113 (64.9) |
| Female | 61 (35) |
| Age (years) | |
| Range | 1–70 |
| Mean | 35.8 |
| Clinical samples | |
| Sputum | 81 (46.5) |
| Pus swabs | 42 (24.1) |
| Blood | 19 (10.9) |
| Urine | 18 (10.3) |
| Throat swabs | 7 (4) |
| Cerebrospinal fluid | 3 (1.7) |
| Nasal swabs | 2 (1.1) |
| Seminal fluid | 1 (0.6) |
| High vaginal swabs | 1 (0.6) |
| Hospital wards | |
| Intensive care unit | 131 (75.2) |
| Male surgical ward | 13 (7.4) |
| Female medical ward | 11 (6.3) |
| Operation theater | 8 (4.6) |
| Male medical ward | 6 (3.4) |
| Female surgical ward | 5 (2.8) |
Antimicrobial Resistance Phenotypes
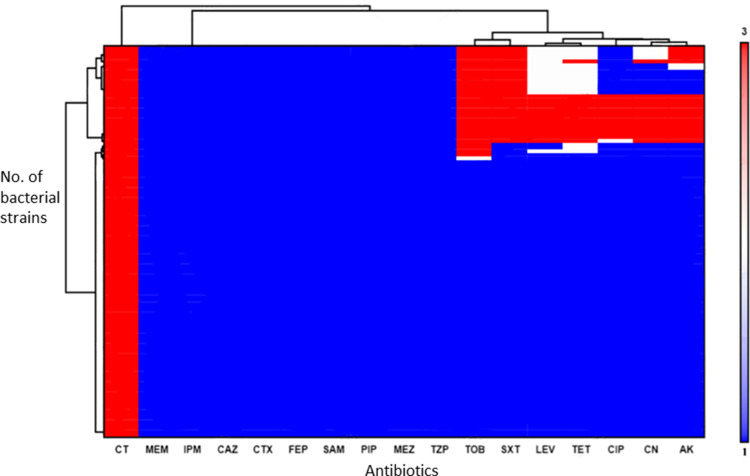
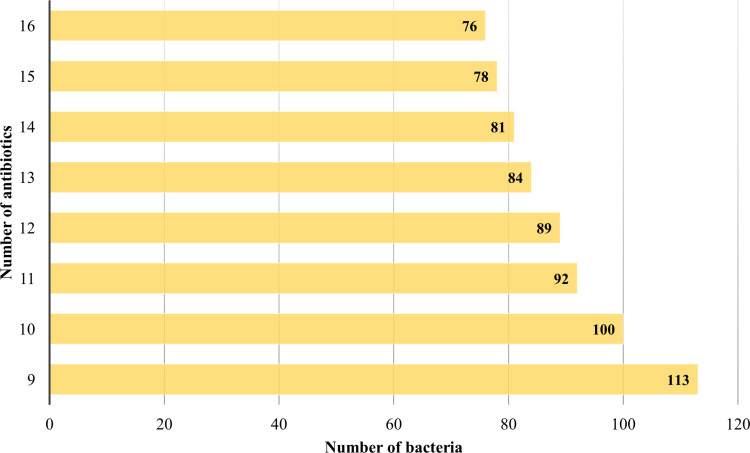
The antimicrobial susceptibility testing for 113 (64.9%) CR-AB isolates displayed 100% resistance to cephalosporins [cefepime (≥32 µg/mL), ceftazidime (≥32 µg/mL), cefotaxime (≥64 µg/mL)], β-lactam inhibitors [piperacillin/tazobactam (≥128/4 µg/mL), ampicillin/sulbactam (≥32/16 µg/mL)], and carbapenems (imipenem and meropenem), with MIC breakpoints ≥8 µg/mL. In addition, 87.6% isolates were resistant to fluoroquinolones (ciprofloxacin), 75.2% to tetracycline (doxycycline), 81.4% to amikacin (aminoglycoside), and 75.2% to co-trimoxazole. However, all of the strains were susceptible to colistin (Figure 1). The WHO AWaRe categorization assigns medically relevant antibiotics into Access (first- and second-line antibiotics), Watch (critical antibiotics), and Reserve (last-resort antibiotics) groups.20 Interestingly, 76 A. baumannii isolates displayed resistance against 16 different AWaRe antibiotics, 78 were resistant to 15 antibiotics, 81 to 14 antibiotics, and 113 to 9 antibiotics (Figure 2).
Figure 1.
The hierarchical clustering of carbapenem-resistant Acinetobacter baumannii. The antibiotics are shown on the x-axis, aligned with the bacterial strains on the y-axis. The scale on the y-axis shows bacterial resistance, intermediate resistance, and sensitivity from 1 to 3, respectively. The dendrogram shows the relationship among the different antibacterial drugs.
Abbreviations: CT, colistin; MEM, meropenem; IPM, imipenem; CAZ, ceftazidime; CTX, cefotaxime; FEP, cefepime; SAM, ampicillin/sulbactam; PIP, piperacillin; MEZ, mezlocillin; TZP, piperacillin/tazobactam; TOB, tobramycin; SXT, trimethoprim/sulfamethoxazole; LEV, levofloxacin; TET, tetracycline; CIP, ciprofloxacin; CN, gentamicin; AK, amikacin.
Figure 2.
Carbapenem-resistant A. baumannii shows simultaneous resistance to multiple classes of antibiotics.
Phenotypic Detection of Carbapenemase and MBL
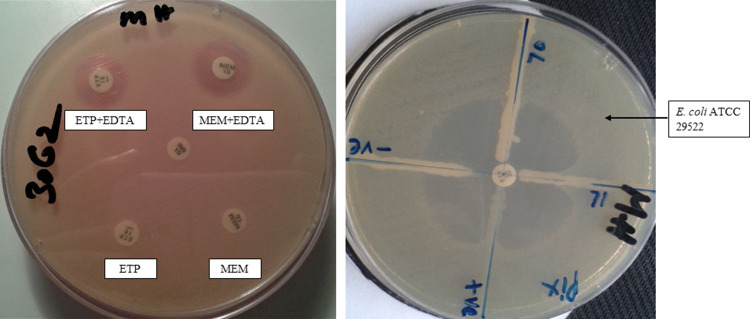
Of the 113 CR-AB isolates identified, 89 (78.7%) were positive for both carbapenemase and MBL production (Figure 3). A significant proportion (n = 64; 71.9%) of the MBL-producing CR-AB isolates were detected in ICU patients, followed by 14 (15.7%) in the MSW, 8 (9%) in the operation theater (OT), 2 (2.2%) in the male medical ward (MMW), 6 (6.7%) in the FMW, and 2 (2.2%) in the female surgical ward (FSW).
Figure 3.
Carbapenem-resistant phenotypes. The left photograph shows the DDST for MBL production, and the right photograph displays the MHT for carbapenemase production.
Abbreviations: ETP, ertapenem; MEM, meropenem; EDTA, ethylenediaminetetraacetic acid; MHT, modified Hodge's test; DDST, double disk susceptibility test; +ve, positive control; -ve, negative control; T-70, 71, tested organisms.
Sequence Typing of Carbapenem Resistant A. baumannii
The MLST results for the 113 CR-AB clinical isolates revealed 7 different STs across the various samples and hospital wards (Table 2). A total of 53 (46.9%) CR-AB isolates belonged to ST2, which was the most predominant ST identified in this study, corresponding to clonal complex 2 (CC2), followed by 18 (15.9%) isolates that belonged to ST1 (CC1), 14 (12.3%) to ST589 (CC1), and 11 (9.7%) to ST7 (CC1). ST2 strains were primarily isolated from sputum samples (n = 27; 50.9%) and were more prevalent in patients admitted to the ICU (n = 21), whereas 13 (24.5%) ST2 isolates were detected in pus samples. Furthermore, 13 (72.3%) ST1 isolates were identified in sputum samples, most of which were found in the ICU (n = 12) cases. In addition, 5 (35.7%) ST589 isolates were also obtained from sputum collected from ICU cases.
Table 2.
Prevalence of Carbapenem-Resistant Acinetobacter baumannii Sequence Types in Different Clinical Wards
| ST (n = 113) | CC | Allelic Profile | Clinical Infections and Hospital Wards | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sputum (n = 60) | Pus (n = 29) | Blood (n = 7) | Throat (n = 5) | Urine (n = 9) | CSF (n = 1) | Nasal (n = 1) | HVS (n = 1) | |||
| 2 (n = 53, 46.9%) | 2 | 2,2,2,2,2,2,2 | 27 (ICU:21, MMW:3, FMW:2, OT:1) | 13 (ICU:7, MSW:3, FSW:1, OT:2) | 4 (ICU:3, MMW: 1) | 3 (ICU) | 5 (FMW:2, ICU:3) | 0 | 0 | 1 (FMW) |
| 1 (n = 18, 15.9%) | 1 | 1, 1, 1, 1, 5, 1, 1 | 13 (ICU:12, FMW:1) | 3 (FSW:1, ICU:1, OT:1) | 0 | 1 (ICU) | 0 | 1 (OT) | 0 | 0 |
| 589 (n = 14, 12.3%) | 1 | 1,1,2,1,9,1,1 | 5 (ICU:3, FMW:1, OT:1) | 4 (ICU:2, MSW:2) | 1 (ICU) | 1 (ICU) | 2 (ICU) | 0 | 1 (MSW) | 0 |
| 7 (n = 11, 9.7%) | 1 | 1,1,1,2,5,1,1 | 5 (ICU) | 4 (MSW:2, FMW:1, ICU:1) | 1 (ICU) | 0 | 1 (ICU) | 0 | 0 | 0 |
| 158 (n = 10, 8.8%) | 158 | 41, 42, 13, 1, 5, 4, 14 | 6 (ICU) | 2 (ICU:1, MSW:1) | 1 (ICU) | 0 | 1 (ICU) | 0 | 0 | 0 |
| 23 (n = 5, 4.4%) | 23 | 1, 3, 10, 1, 4, 4, 4 | 3 (ICU:2, FMW:1) | 2 (ICU) | 0 | 0 | 0 | 0 | 0 | 0 |
| 25 (n = 2, 1.7%) | 25 | 3, 3, 2, 4, 7, 2, 4 | 1 (FMW) | 1 (FSW) | 0 | 0 | 0 | 0 | 0 | 0 |
Note: The ST profiles consist of 7 allele numbers, corresponding to cpn60, fusA, gltA, pyrG, recA, rplB, and rpoB, respectively (https://pubmlst.org/abaumannii/).
Abbreviations: ST, sequence type; CC, clonal complex; CSF, cerebral spinal fluid; HVS, high vaginal swab; ICU, intensive care unit; MMW, male medical ward; FMW, female medical ward; MSW, male surgical ward; FSW, female surgical ward; OT, operation theater.
Genotypic Detection of Carbapenem-Resistant Genes
The CR-AB isolates (n = 113) were further screened for the presence of carbapenem-resistant genes. All CR-AB isolates harbored the intrinsic blaOXA-51 gene, with a high prevalence of blaOXA-23 (n = 56; 49.5%), followed by blaNDM-1 (n = 28; 24.7%), blaOXA-58 (n = 22; 19.4%), and blaOXA-143 (n = 3; 2.6%). In addition, 28 (50%) isolates harbored blaOXA-23, 14 (50%) harbored blaNDM-1, 12 (54.5%) harbored blaOXA-58, and 8 (80%) harbored blaOXA-24, all of which belonged to ST2. Interestingly, 11 (9.7%) isolates harbored blaOXA-51, blaNDM-1, and blaOXA23 together, 3 (2.6%) isolates harbored blaOXA-51, blaOXA-23, blaOXA-24, and blaNDM-1, and 7 (6.2%) harbored blaOXA-51, blaOXA-23, blaOXA-58, and blaNDM-1. In addition, 6 (5.3%) isolates harbored both blaOXA-51 and blaOXA-23, and 17 (15%) harbored blaOXA-24, blaOXA-51, blaOXA-23, and blaOXA-58 (Table 3).
Table 3.
The Co-Detection and Distribution of Carbapenem-Resistant Genes in Different CR-AB Alleles
| ST of CR-AB (n = 113) | blaOXA-23 (n = 56) | blaOXA-24 (n = 10) | blaOXA-58 (n = 22) | blaOXA-143 (n = 3) | blaNDM-1 (n = 28) |
|---|---|---|---|---|---|
| 1 (n = 18) | 6 (10.5%) | 1 (10%) | 2 (9%) | 1 (33.4%) | 2 (7.1%) |
| 2 (n = 53) | 28 (50%) | 8 (80%) | 12 (54.5%) | 2 (66.6%) | 14 (50%) |
| 7 (n = 11) | 4 (7.1%) | 1 (10%) | 2 (9%) | 0 | 4 (14.2%) |
| 23 (n = 5) | 3 (5.3%) | 0 | 2 (9%) | 0 | 1 (3.5%) |
| 25 (n = 2) | 2 (3.57%) | 0 | 0 | 0 | 1 (3.5%) |
| 158 (n = 10) | 4 (7.1%) | 0 | 0 | 0 | 2 (7%) |
| 589 (n = 14) | 9 (16%) | 0 | 4 (18%) | 0 | 4 (14.2%) |
Abbreviations: ST, sequence type; CR-AB, carbapenem-resistant Acinetobacter baumannii.
Discussion
CR-AB infections are difficult to treat and represent a persistent threat to Pakistan’s fragile healthcare system.21 These highly resistant pathogens are associated with extended hospital stays, increased mortality and morbidity, and added financial burden.22 In this study, the intrinsic blaOXA-51 was detected in all A. baumannii isolates, stressing the importance of using the blaOXA-51 gene as an alternative marker for Acinetobacter spp., identification, as described previously.23,24 A. baumannii is considered among the major nosocomial pathogens and has been identified in various areas of the hospital environment, including on medical devices and intravenous catheters.25 In the present study, A. baumannii were recovered from numerous clinical specimens, primarily from sputum (46%) and pus (24%) samples, and were prevalent among patients admitted to the ICU (75%). Previously, multiple studies have indicated that the prevalence of A. baumannii in the ICU was becoming a serious nosocomial problem.26–28 The CR-AB isolates in this study displayed resistance to all available β-lactam drugs, including carbapenems, which are considered to be the ultimate therapy for the infections produced by multidrug-resistant bacteria; although colistin remains the utmost effective drug, colistin is associated with considerable side effects. Among the identified isolates, 76 and 78 isolates showed simultaneous resistance to 16 and 15 medically relevant antibiotics, respectively, which have been categorized in the WHO AWaRe system. Previous data from Pakistan and other parts of the world have documented a similar scenario, in which the primary methods used to treat infections are no longer able to fight these pathogens.11,21,29–31 AMR is primarily caused by the acquisition of resistant determinants, which increase AMR phenotypes. The OXA-type-β-lactamase is the most widely distributed class D enzyme found in gram-negative bacteria.32 In this study, blaOXA-23 was the predominant resistance determinant. OXAs are encoded by plasmid-mediated genes that can easily be transferred by conjugation. The main reservoir for the blaOXA-23 gene exists in Asiatic countries, such as Pakistan, India, China, and Nepal. In addition, an extensive study conducted by the SENTRY Antimicrobial Surveillance Program in various Asian-Pacific counties also revealed the heavy presence of blaOXA-23 in A. baumannii.33 We also found blaOXA-24, blaOXA-58, and blaOXA-143 genes, which had not been widely reported in Pakistan. The A. baumannii harboring blaOXA-58 are primarily prevalent in China,34 whereas A. baumannii harboring blaOXA-143 are prevalent in Brazil35 and Mexico.36 The blaNDM-1 gene encodes the most powerful class B MBL enzyme, which can hydrolyze a wide range of medically relevant antibiotics. The Indian subcontinent is considered to represent the primary reservoir for the emergence and spread of the blaNDM-1 gene globally.6,11,37 This study documented the prevalence of blaNDM-1 in clinical A. baumannii isolates (24.7%) and found that this gene could be detected in isolates that simultaneously harbor the class D genes blaOXA-23, blaOXA-24, and blaOXA-58. Thus far, various studies have provided evidence to support the spread of blaNDM-1 in Enterobacteriaceae in Pakistan rather than in non-fermenting bacteria. However, recently published data have focused on the blaNDM-1-producing A. baumannii isolates in Pakistan.11,38 The previously published data have also described the global detection of blaOXA-23 and blaNDM-1 genes in isolates that also harbor the blaOXA gene. The NDM gene can be readily transferred to A. baumannii via Tn125 by conjugation, and ISAba125 acts as a powerful promoter of the blaNDM-1 gene.15,39,40
MLST is a promising technique for studying molecular epidemiology and investigating bacterial strain outbreaks in clinical and environmental settings. In this study, the A. baumannii isolates belonged to 7 different STs: ST1, ST2, ST7, ST23, ST25, ST158, and ST589. Of these, the majority of the A. baumannii isolates (46.9%) belonged to ST2 (CC2). This clone was especially prevalent among critical care patients and was detected in isolate that also harbored the genes blaOXA-23, blaOXA-58, and blaNDM-1. The international clones, CC1 and CC2, were primarily associated with the enzymes blaOXA-23, blaOXA-24, and blaOXA-158. Mediterranean counties have documented the emergence of A. baumannii belonging to ST2, with fewer identified isolates belonging to ST1.41,42 A Lebanon study reported the detection of ST2 A. baumannii harboring NDM-1.43 Several studies from Pakistan have described the detection of A. baumannii ST2 clones.29,39 ST2 (CC2) can survive in the clinical settings for longer times, allowing for the acquisition of AMR genes, which can complicate treatment. This report represents the first detection of the ST7 strain in Pakistan to date and need further comprehensive study to determine its diversification. This study was a compressive study examining the genes harbored by various CR-AB isolates that fills a gap by providing previously unavailable data regarding the coexistence of blaNDM-1 and blaOXA genes in A. baumannii ST1, ST23, ST25, and ST158 isolates at the national level.
Conclusion
This study highlights the significance of the intrinsic gene blaOXA-51, which can be used to rapidly detect A. baumannii. All strains were observed to be resistant to medically relevant antibacterials, with few treatment options available for infections associated with these isolates. Therefore, active monitoring and urgent management are necessary for clinical settings. The international clone ST2 was the most widespread isolates identified in public hospitals, simultaneously harboring blaOXA-23, blaOXA-24, blaOXA-58, and blaNDM-1, indicating that the circulation of these clones occurs at the international level, and this report represents the first reported detection of ST7 in Pakistan. Due to the impending threat associated with AMR bacteria, Pakistan’s Ministry of Health has recently developed a National Action Plan (NAP) on Antimicrobial Resistance. Stakeholders must implement the NAP rapidly and effectively to halt the spread of these super-bacteria.
Acknowledgments
The authors extend their appreciation to the Deputyship for Research & Innovation, Ministry of Education in Saudi Arabia, for funding this work through the grant number “375213500”. The authors would like to extend their sincere appreciation to the central laboratory at Jouf University for supporting this study.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
All authors declared no conflicts of interest.
References
- 1.Hsu L-Y, Apisarnthanarak A, Khan E, Suwantarat N, Ghafur A, Tambyah PA. Carbapenem-resistant Acinetobacter baumannii and Enterobacteriaceae in south and southeast Asia. Clin Microbiol Rev. 2017;30(1):1–22. doi: 10.1128/cmr.00042-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Magiorakos AP, Srinivasan A, Carey RB, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268–281. doi: 10.1111/j.1469-0691.2011.03570.x [DOI] [PubMed] [Google Scholar]
- 3.Khan HA, Baig FK, Mehboob R. Nosocomial infections: epidemiology, prevention, control and surveillance. Asian Pac J Trop Biomed. 2017;7(5):478–482. doi: 10.1016/j.apjtb.2017.01.019 [DOI] [Google Scholar]
- 4.Talebi Bezmin Abadi A, Rizvanov AA, Haertlé T, Blatt NL. World Health Organization report: current crisis of antibiotic resistance. BioNanoScience. 2019;9(4):778–788. doi: 10.1007/s12668-019-00658-4 [DOI] [Google Scholar]
- 5.Mulani MS, Kamble EE, Kumkar SN, Tawre MS, Pardesi KR. Emerging strategies to combat ESKAPE pathogens in the era of antimicrobial resistance: a review. Front Microbiol. 2019;10:539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Qamar M, Lopes B, Hassan B, et al. The present danger of New Delhi metallo-β-lactamase: a threat to public health. Future Microbiol. 2020;15(18):1759–1778. doi: 10.2217/fmb-2020-0069 [DOI] [PubMed] [Google Scholar]
- 7.CDC. Antibiotic resistance threats in the United States, 2019. Report. 2019.
- 8.Aslam B, Wang W, Arshad MI, et al. Antibiotic resistance: a rundown of a global crisis. Infect Drug Resist. 2018;11:1645–1658. doi: 10.2147/IDR.S173867 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Khurshid M, Rasool MH, Siddique MH, et al. Molecular mechanisms of antibiotic co-resistance among carbapenem resistant Acinetobacter baumannii. J Infect Dev Ctries. 2019;13(10):899–905. doi: 10.3855/jidc.11410 [DOI] [PubMed] [Google Scholar]
- 10.Qamar MU, Nahid F, Walsh TR, Kamran R, Zahra R. Prevalence and clinical burden of NDM-1 positive infections in pediatric and neonatal patients in Pakistan. Pediatr Infect Dis J. 2015;34(4):452–454. doi: 10.1097/INF.0000000000000582 [DOI] [PubMed] [Google Scholar]
- 11.Qamar MU, Walsh TR, Toleman MA, et al. Dissemination of genetically diverse NDM-1, −5, −7 producing-gram-negative pathogens isolated from pediatric patients in Pakistan. Future Microbiol. 2019;14(8):691–704. doi: 10.2217/fmb-2019-0012 [DOI] [PubMed] [Google Scholar]
- 12.Qamar MU, Saleem S, Toleman MA, et al. In vitro and in vivo activity of Manuka honey against NDM-1-producing Klebsiella pneumoniae ST11. Future Microbiol. 2018;13(1):13–26. doi: 10.2217/fmb-2017-0119 [DOI] [PubMed] [Google Scholar]
- 13.Elbediwi M, Li Y, Paudyal N, et al. Global burden of colistin-resistant bacteria: Mobilized Colistin Resistance Genes Study (1980–2018). Microorganisms. 2019;7(10):461. doi: 10.3390/microorganisms7100461 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Qamar MU, Ejaz H, Walsh TR, et al. Clonal relatedness and plasmid profiling of extensively drug-resistant New Delhi metallo-β-lactamase-producing Klebsiella pneumoniae clinical isolates. Future Microbiol. 2021;16(4):229–239. doi: 10.2217/fmb-2020-0315 [DOI] [PubMed] [Google Scholar]
- 15.Joshi PR, Acharya M, Kakshapati T, Leungtongkam U, Thummeepak R, Sitthisak S. Co-existence of blaOXA-23 and blaNDM-1 genes of Acinetobacter baumannii isolated from Nepal: antimicrobial resistance and clinical significance. Antimicrob Resist Infect Control. 2017;6(1):21. doi: 10.1186/s13756-017-0180-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Qamar MU, Saleem S, Arshad U, et al. Antibacterial efficacy of manuka honey against New Delhi metallo-β-lactamase producing gram negative bacteria isolated from blood cultures. Pak J Zool. 2017;49(6):1997–2003. [Google Scholar]
- 17.Weinstein MP, Patel JB, Campeau S, et al. Performance Standarad for Antimicrobial Suceptibility Testing. 28th ed. Clinical and Laboratory Standards Institute;2018. [Google Scholar]
- 18.Qamar MU, Hannan A, Arshad MU, Arshad M. Metallo-β-lactamase producing Enterobacter cloacae: an emerging threat in neonates. Afr J Microbiol Res. 2014;8(6):525–528. doi: 10.5897/AJMR2013.6348 [DOI] [Google Scholar]
- 19.Javed H, Ejaz H, Zafar A, Rathore AW. Metallo-beta-lactamase producing Escherichia coli and Klebsiella pneumoniae: a rising threat for hospitalized children. J Pak Med Assoc. 2016;66(9):1068–1072. [PubMed] [Google Scholar]
- 20.Hsia Y, Lee BR, Versporten A, et al. Use of the WHO access, watch, and reserve classification to define patterns of hospital antibiotic use (AWaRe): an analysis of paediatric survey data from 56 countries. Lancet Glob Health. 2019;7(7):e861–e871. doi: 10.1016/S2214-109X(19)30071-3 [DOI] [PubMed] [Google Scholar]
- 21.Hasan B, Perveen K, Olsen B, Zahra R. Emergence of carbapenem-resistant Acinetobacter baumannii in hospitals in Pakistan. J Med Microbiol. 2014;63(1):50–55. doi: 10.1099/jmm.0.063925-0 [DOI] [PubMed] [Google Scholar]
- 22.Dadgostar P. Antimicrobial resistance: implications and costs. Infect Drug Resist. 2019;12:3903–3910. doi: 10.2147/IDR.S234610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.El-Kazzaz W, Metwally L, Yahia R, Al-Harbi N, El-Taher A, Hetta HF. Antibiogram, prevalence of OXA carbapenemase encoding genes, and RAPD-genotyping of multidrug-resistant acinetobacter baumannii incriminated in hidden community-acquired infections. Antibiotics. 2020;9(9):603. doi: 10.3390/antibiotics9090603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rafei R, Pailhoriès H, Hamze M, et al. Molecular epidemiology of Acinetobacter baumannii in different hospitals in Tripoli, Lebanon using blaOXA-51-like sequence based typing. BMC Microbiol. 2015;15(1):103. doi: 10.1186/s12866-015-0441-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Almasaudi SB. Acinetobacter spp. as nosocomial pathogens: epidemiology and resistance features. Saudi J Biol Sci. 2018;25(3):586–596. doi: 10.1016/j.sjbs.2016.02.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ayobami O, Willrich N, Harder T, Okeke IN, Eckmanns T, Markwart R. The incidence and prevalence of hospital-acquired (carbapenem-resistant) Acinetobacter baumannii in Europe, Eastern Mediterranean and Africa: a systematic review and meta-analysis. Emerg Microbes Infect. 2019;8(1):1747–1759. doi: 10.1080/22221751.2019.1698273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sileem AE, Said AM, Meleha MS. Acinetobacter baumannii in ICU patients: a prospective study highlighting their incidence, antibiotic sensitivity pattern and impact on ICU stay and mortality. Egypt J Chest Dis Tuberc. 2017;66(4):693–698. doi: 10.1016/j.ejcdt.2017.01.003 [DOI] [Google Scholar]
- 28.Begum S, Hasan F, Hussain S, Ali Shah A. Prevalence of multi drug resistant Acinetobacter baumannii in the clinical samples from Tertiary Care Hospital in Islamabad, Pakistan. Pak J Med Sci. 2013;29(5):1253–1258. doi: 10.12669/pjms.295.3695 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hadjadj L, Syed MA, Abbasi SA, Rolain J-M, Jamil B. Diversity of carbapenem resistance mechanisms in clinical gram-negative bacteria in Pakistan. Microb Drug Resist. 2020. doi: 10.1089/mdr.2019.0387 [DOI] [PubMed] [Google Scholar]
- 30.Braun SD, Jamil B, Syed MA, et al. Prevalence of carbapenemase-producing organisms at the Kidney Center of Rawalpindi (Pakistan) and evaluation of an advanced molecular microarray-based carbapenemase assay. Future Microbiol. 2018;13(11):1225–1246. doi: 10.2217/fmb-2018-0082 [DOI] [PubMed] [Google Scholar]
- 31.Abouelfetouh A, Torky AS, Aboulmagd E. Phenotypic and genotypic characterization of carbapenem-resistant Acinetobacter baumannii isolates from Egypt. Antimicrob Resist Infect Control. 2019;8(1):185. doi: 10.1186/s13756-019-0611-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Antunes NT, Fisher JF. Acquired class D β-lactamases. Antibiotics (Basel). 2014;3(3):398–434. doi: 10.3390/antibiotics3030398 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mendes RE, Bell JM, Turnidge JD, Castanheira M, Jones RN. Emergence and widespread dissemination of OXA-23, −24/40 and −58 carbapenemases among Acinetobacter spp. in Asia-Pacific nations: report from the SENTRY surveillance program. J Antimicrob Chemother. 2008;63(1):55–59. doi: 10.1093/jac/dkn434 [DOI] [PubMed] [Google Scholar]
- 34.Chen Y, Guo P, Huang H, Huang Y, Wu Z, Liao K. Detection of co-harboring OXA-58 and NDM-1 carbapenemase producing genes resided on a same plasmid from an Acinetobacter pittii clinical isolate in China. Iran J Basic Med Sci. 2019;22(1):106–111. doi: 10.22038/ijbms.2018.28934.6994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Werneck JS, Picão RC, Girardello R, et al. Low prevalence of blaOXA-143 in private hospitals in Brazil. Antimicrob Agents Chemother. 2011;55(9):4494–4495. doi: 10.1128/AAC.00295-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pérez-Oseguera Á, Castro-Jaimes S, Salgado-Camargo AD, et al. Complete genome sequence of a bla(OXA-58)-producing Acinetobacter baumannii strain isolated from a Mexican hospital. Genome Announc. 2017;5(36):e00949–17. doi: 10.1128/genomeA.00949-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yong D, Toleman MA, Giske CG, et al. Characterization of a new metallo-beta-lactamase gene, bla(NDM-1), and a novel erythromycin esterase gene carried on a unique genetic structure in Klebsiella pneumoniae sequence type 14 from India. Antimicrob Agents Chemother. 2009;53(12):5046–5054. doi: 10.1128/AAC.00774-09 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sartor AL, Raza MW, Abbasi SA, et al. Molecular epidemiology of NDM-1-producing Enterobacteriaceae and Acinetobacter baumannii isolates from Pakistan. Antimicrob Agents Chemother. 2014;58(9):5589–5593. doi: 10.1128/AAC.02425-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Karah N, Khalid F, Wai SN, Uhlin BE, Ahmad I. Molecular epidemiology and antimicrobial resistance features of Acinetobacter baumannii clinical isolates from Pakistan. Ann Clin Microbiol Antimicrob. 2020;19(1):2. doi: 10.1186/s12941-019-0344-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ramoul A, Loucif L, Bakour S, Amiri S, Dekhil M, Rolain JM. Co-occurrence of blaNDM-1 with blaOXA-23 or blaOXA-58 in clinical multidrug-resistant Acinetobacter baumannii isolates in Algeria. J Glob Antimicrob Resist. 2016;6:136–141. doi: 10.1016/j.jgar.2016.05.003 [DOI] [PubMed] [Google Scholar]
- 41.Thirapanmethee K, Srisiri-A-Nun T, Houngsaitong J, Montakantikul P, Khuntayaporn P, Chomnawang MT. Prevalence of OXA-type β-lactamase genes among carbapenem-resistant acinetobacter baumannii clinical isolates in Thailand. Antibiotics (Basel). 2020;9(12):864. doi: 10.3390/antibiotics9120864 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kuo S-C, Huang W-C, Huang T-W, et al. Molecular epidemiology of emerging bla(OXA-23-like)- and bla(OXA-24-like)-carrying Acinetobacter baumannii in Taiwan. Antimicrob Agents Chemother. 2018;62(3):e01215–17. doi: 10.1128/AAC.01215-17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Nawfal Dagher T, Al-Bayssari C, Chabou S, et al. Investigation of multidrug-resistant ST2 Acinetobacter baumannii isolated from Saint George hospital in Lebanon. BMC Microbiol. 2019;19(1):29. doi: 10.1186/s12866-019-1401-2 [DOI] [PMC free article] [PubMed] [Google Scholar]