Asthma is characterised by recurrent symptoms associated with variable airflow obstruction and airway hyperresponsiveness, all of which are improved with combination inhaled corticosteroid (ICS)/long-acting β-agonist (LABA) treatment in mild-to-moderate asthma [1]. A proportion of patients however develop fixed airflow obstruction (FAO), despite optimised treatment. FAO is prevalent in up to 60% of patients with severe asthma and is associated with a more rapid decline in lung function and increased symptoms [2]. The underlying mechanisms of FAO in asthma are poorly understood; therefore, development of novel treatment strategies remains a challenge.
Short abstract
Airway smooth muscle cells from severe asthma patients with FAO respond to β2-agonists and corticosteroids in vitro, and at a level similar to mild asthmatics. Intrinsic dysfunction of these signalling pathways is unlikely to contribute to FAO. https://bit.ly/3muvNsW
To the Editor:
Asthma is characterised by recurrent symptoms associated with variable airflow obstruction and airway hyperresponsiveness, all of which are improved with combination inhaled corticosteroid (ICS)/long-acting β-agonist (LABA) treatment in mild-to-moderate asthma [1]. A proportion of patients however develop fixed airflow obstruction (FAO), despite optimised treatment. FAO is prevalent in up to 60% of patients with severe asthma and is associated with a more rapid decline in lung function and increased symptoms [2]. The underlying mechanisms of FAO in asthma are poorly understood; therefore, development of novel treatment strategies remains a challenge.
Airway smooth muscle cells (ASMCs) are the major effector cells of bronchoconstriction in asthma and also contribute to the inflammatory process by secreting pro-inflammatory cytokines and chemokines. Therefore, ASMCs are a major target of both β2-agonist and ICS treatment [3]. Although several studies have suggested that steroid signalling [4] or β2-adrenoceptor (β2AR) signalling may be abnormally regulated in severe asthma [5], it remains unknown whether impaired airway smooth muscle corticosteroid and/or β2-agonist response may contribute to the development of FAO. The aim of this study was to investigate whether primary human ASMCs obtained from severe asthma patients with FAO differ in their response to β2-agonists and corticosteroids compared with asthma patients without FAO and healthy controls. We hypothesised that ASMCs from asthma patients with FAO are less responsive to corticosteroid and β2-agonist treatment than those from patients without FAO.
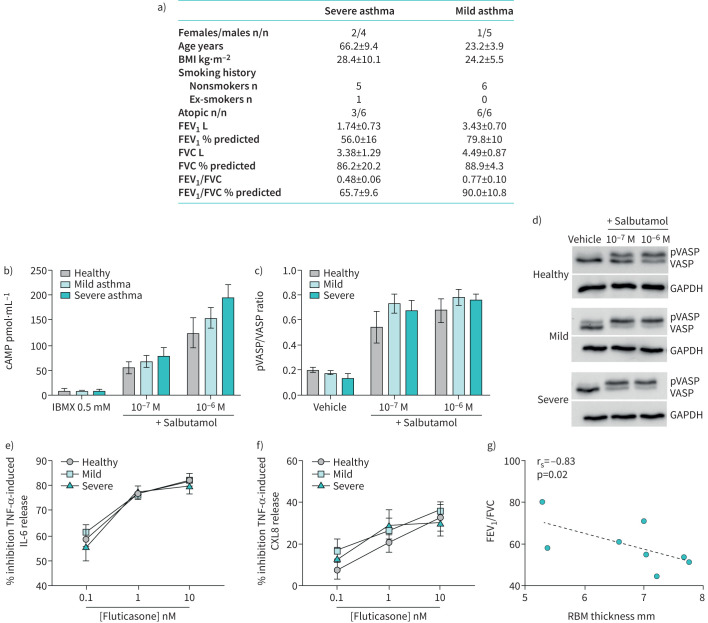
Human ASMCs were obtained from bronchial biopsies of severe asthma patients with FAO (n=6, mean±sd forced expiratory volume in 1 s (FEV1): 56±16% predicted, mean±sd FEV1/forced vital capacity (FVC): 0.48±0.06), who were clinically unresponsive to high dose ICS/LABA treatment [6]; mild asthma patients with reversible airflow obstruction (n=6, mean±sd FEV1: 79.8±10% pred, mean±sd FEV1/FVC: 0.77±0.10), who had clinically improved after ICS/LABA treatment [7]; and from healthy volunteers without any respiratory disease (n=4). Patient demographics are summarised in figure 1a. FAO was defined as <200 mL and 12% improvement in spirometry post-bronchodilator (400 μg inhaled salbutamol) and a post-bronchodilator FEV1/FVC ratio below the lower limit of normal. The ASMCs were isolated and grown in culture as previously described [8]. All experiments were carried out using ASMCs with passage number 4 or 5. Ethical approval for this study was granted by the Ethics Review Committees of Sydney South West Area Health Service (RPA, X02-0137) and Sydney Local Health District (CRGH, HREC/14/CRGH/75).
FIGURE 1.
a) Patient demographics. b–d) Human airway smooth muscle cells (ASMCs) derived from healthy volunteers (n=4), mild asthma patients (n=6) and severe asthma patients with fixed airflow obstruction (n=6) were treated with the β2-agonist salbutamol. b) cAMP (pmol·mL−1) was measured by ELISA in presence of a phosphodiesterase inhibitor, IBMX 0.5 mM. c, d) Vasodilator-stimulated phosphoprotein (VASP) phosphorylation (pVASP) was measured using Western blotting and is expressed as the ratio of total VASP. e, f) ASMCs were also treated with increasing concentrations of fluticasone for 1 h before stimulation with tumour necrosis factor (TNF)-α (10 ng·mL−1) for 24 h. Interleukin (IL)-6 and CXCL8 release was measured using ELISA and data is presented as percent inhibition of TNF-α-induced IL-6 (e) and CXCL8 (f) release. g) Reticular basement membrane (RBM) thickness was measured from endobronchial biopsies of the airway wall of asthma patients (n=8) with a range of airflow obstruction as measured by forced expiratory volume in 1 s (FEV1)/forced vital capacity (FVC) ratio. RBM thickness constituted the measurement of the length of a straight line drawn transversely across the basement membrane perpendicular to the mucosal surface. Six measurements were taken per biopsy using Image J (https://imagej.nih.gov/ij/), of which the average was taken. Increasing RBM thickness was related to decreasing FEV1/FVC ratio, Spearman's rank correlation coefficient=−0.82, p=0.02. BMI: body mass index.
To assess β2AR signalling, the cells were stimulated with the β2-agonist, salbutamol (Sigma-Aldrich, NSW, Australia) and levels of intracellular cAMP and phosphorylated vasodilator-stimulated phosphoprotein (pVASP) were measured as markers of β2AR activation. Activation of β2AR leads to activation of adenylyl cyclase and subsequent cAMP formation. cAMP phosphorylates protein kinase A (PKA), which phosphorylates multiple proteins including VASP, leading to reductions in intracellular calcium, smooth muscle relaxation, and bronchodilation. Maximal cAMP levels (in the presence of a phosphodiesterase (PDE) inhibitor) were measured using an ELISA kit according to the manufacturer's instructions (Cayman Chemical Company, Ann Arbor, MI, USA). In cells with no PDE inhibition, VASP phosphorylation was assessed by Western blotting as previously described [9], using mouse anti-VASP/pVASP (1:1500; BD Biosciences, San Jose, CA, USA) and mouse anti-GAPDH (loading control) (1:5000; MilliporeSigma, Burlington, MA, USA) antibodies.
To assess whether ASMCs from severe asthma patients with FAO are intrinsically more resistant to corticosteroids compared with those from mild asthma patients and healthy controls, cells were treated with increasing concentrations of fluticasone (0.1, 1, 10 nM; Sigma-Aldrich) for 1 h before stimulation with tumour necrosis factor (TNF)-α (10 ng·mL−1) for 24 h. TNF-α is a multipotent proinflammatory mediator, mainly produced by macrophages, and plays a critical role in the immunoregulation of asthma by enhancing inflammation and airway hyperresponsiveness. Release of interleukin (IL)-6 and CXCL8, two pro-inflammatory chemoattractant cytokines that have been implicated in the pathogenesis of severe asthma [10], were measured in the supernatant using sandwich ELISA (BD Biosciences) according to the manufacturer's instructions.
We observed similar salbutamol-induced cAMP release from ASMCs derived from healthy individuals and patients with mild asthma and severe asthma with FAO (figure 1b). There was a trend for greater cAMP production as asthma severity increased. Consistent with this, there was a similar increase in VASP phosphorylation in response to salbutamol in the three groups (figure 1c and d). Furthermore, fluticasone partially reduced IL-6 and CXCL8 release induced by TNF-α (figure 1e and f). At the highest concentration of 10 nM there was an inhibition of ∼80% of TNF-α-induced IL-6 release and 40% of TNF-α-induced CXCL8 release. Again, there were no differences between ASMCs from healthy volunteers or mild versus severe asthma patients.
To our knowledge, our study is the first that has addressed the question of whether ASMCs from severe asthmatic patients with FAO are intrinsically more resistant to β2-agonists and/or steroids compared with cells from responsive asthmatics and healthy subjects. We have previously found both maximal cAMP production and airway smooth muscle-derived cytokine inhibition with corticosteroids to be similar in mild asthma in comparison to non-asthmatic individuals [11, 12], but hypothesised that these would be abnormal in patients with FAO, since clinically these patients are unresponsive to these treatments [6]. We found that ASMCs derived from severe asthmatic patients with FAO respond to β2-agonists and corticosteroids in vitro, and at a level similar to mild asthmatics who are treatment responsive [7], and therefore, reject our hypothesis.
These findings suggest that FAO in asthma is unlikely to be the result of intrinsic dysfunction in β2AR or corticosteroid signalling in airway smooth muscle. Instead, patients with FAO may be refractory to ICS/LABA treatment due to other factors, such as the presence of airway fibrosis limiting the ability of the airways to dilate in response to bronchodilators. We further assessed the relationship between reticular basement membrane (RBM) thickness and FEV1/FVC ratio. We found that greater RBM thickness was related to increased airflow obstruction (decreased FEV1/FVC ratio) (figure 1g), thereby supporting the notion of greater airway fibrosis occurring in patients with FAO. Other factors that could contribute to FAO in asthma include increased smooth muscle layer thickness, which was previously shown to be related to the clinical severity of asthma [13], or changes in the extracellular matrix and its regulators within the airway smooth muscle that may affect airway smooth muscle function [14]. Furthermore, parenchymal changes in patients with asthma and FAO resulting in loss of elastic recoil may also affect the ability of the airways to dilate because of alteration to the airway–parenchymal interdependence [6].
A potential limitation to the present study is that the ASMCs were derived from bronchial biopsies taken from the large airways only. Although both the small and large airways have been implicated in the pathogenesis of asthma, and several studies have reported that structural and functional changes occur in both the small and large airways, it is important to note that the small airways may be more implicated in severe disease [15]. Another potential limitation of our study is that we only measured the biochemical pathway downstream of the β2-receptor. It is possible that either relaxation or contraction are affected independently of the ability to produce cAMP.
Our findings have important implications for the treatment of patients with FAO, and in particular given our observation of no intrinsic β2AR desensitisation or corticosteroid resistance, our data does not support the use of high-dose treatments in these patients if these are used in an attempt to overcome intrinsic abnormalities. Treatment should therefore be titrated according to clinical outcomes. Our data indicate that novel pharmacological interventions targeting β2-receptor or corticosteroid signalling are unlikely to be effective in asthma with FAO and there is a need for treatments that effectively target the underlying causes of FAO and treatment resistance. Unfortunately, mechanisms resulting in FAO in asthma remain poorly understood [2], and until these are discovered FAO will remain difficult to treat.
Footnotes
Data availability: The data sets generated and/or analysed during the study are available from the corresponding author on reasonable request.
Conflict of interest: S. Rutting has nothing to disclose.
Conflict of interest: D. Xenaki has nothing to disclose.
Conflict of interest: K.D. Reddy has nothing to disclose.
Conflict of interest: M. Baraket has nothing to disclose.
Conflict of interest: D.G. Chapman has nothing to disclose.
Conflict of interest: G.G. King reports grants, personal fees and nonfinancial support from AstraZeneca, Boehringer Ingelheim, Chiesi, GlaxoSmithKline, Menarini, MundiPharma and Novartis; and unrestricted research grants from the NHMRC, Boehringer Ingelheim, CycloPharma, GlaxoSmithKline, Menarini, MundiPharma, philanthropic individuals and societies; and nonfinancial and other support from Restech, Italy, during the conduct of the study.
Conflict of interest: B.G. Oliver has nothing to disclose.
Conflict of interest: K.O. Tonga reports a Lung Foundation Australia Ludwig Engel grant-in-aid during the conduct of the study.
Support statement: This study was supported by a bridging grant from the University of Sydney, a philanthropic grant from the Berg Family Foundation and the Lung Foundation Australia Ludwig Engel grant-in-aid. Funding information for this article has been deposited with the Crossref Funder Registry.
References
- 1.McCracken JL, Veeranki SP, Ameredes BT, et al. . Diagnosis and management of asthma in adults: a review. JAMA 2017; 318: 279–290. doi: 10.1001/jama.2017.8372 [DOI] [PubMed] [Google Scholar]
- 2.Guerra S, Martinez FD. Epidemiology of the origins of airflow limitation in asthma. Proc Am Thorac Soc 2009; 6: 707–711. doi: 10.1513/pats.200908-085DP [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Doeing DC, Solway J. Airway smooth muscle in the pathophysiology and treatment of asthma. J Appl Physiol (1985) 2013; 114: 834–843. doi: 10.1152/japplphysiol.00950.2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Barnes PJ. Corticosteroid resistance in patients with asthma and chronic obstructive pulmonary disease. J Allergy Clin Immunol 2013; 131: 636–645. doi: 10.1016/j.jaci.2012.12.1564 [DOI] [PubMed] [Google Scholar]
- 5.Chang PJ, Bhavsar PJ, Michaeloudes C, et al. . Corticosteroid insensitivity of chemokine expression in airway smooth muscle of patients with severe asthma. J Allergy Clin Immunol 2012; 130: 877–885. doi: 10.1016/j.jaci.2012.07.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tonga KO, Chapman DG, Farah CS, et al. . Reduced lung elastic recoil and fixed airflow obstruction in asthma. Respirology 2020; 25: 613–619. doi: 10.1111/resp.13688 [DOI] [PubMed] [Google Scholar]
- 7.Baraket M, Oliver BG, Burgess JK, et al. . Is low dose inhaled corticosteroid therapy as effective for inflammation and remodeling in asthma? A randomized, parallel group study. Respir Res 2012; 13: 11. doi: 10.1186/1465-9921-13-11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Johnson PR, Roth M, Tamm M, et al. . Airway smooth muscle cell proliferation is increased in asthma. Am J Respir Crit Care Med 2001; 164: 474–477. doi: 10.1164/ajrccm.164.3.2010109 [DOI] [PubMed] [Google Scholar]
- 9.Niimi K, Ge Q, Moir LM, et al. . β2-Agonists upregulate PDE4 mRNA but not protein or activity in human airway smooth muscle cells from asthmatic and nonasthmatic volunteers. Am J Physiol Lung Cell Mol Physiol 2012; 302: L334–L342. doi: 10.1152/ajplung.00163.2011 [DOI] [PubMed] [Google Scholar]
- 10.Morjaria JB, Babu KS, Vijayanand P, et al. . Sputum IL-6 concentrations in severe asthma and its relationship with FEV1. Thorax 2011; 66: 537. doi: 10.1136/thx.2010.136523 [DOI] [PubMed] [Google Scholar]
- 11.Trian T, Burgess JK, Niimi K, et al. . β2-Agonist induced cAMP is decreased in asthmatic airway smooth muscle due to increased PDE4D. PLoS One 2011; 6: e20000. doi: 10.1371/journal.pone.0020000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Roth M, Johnson PR, Borger P, et al. . Dysfunctional interaction of C/EBPalpha and the glucocorticoid receptor in asthmatic bronchial smooth-muscle cells. N Engl J Med 2004; 351: 560–574. doi: 10.1056/NEJMoa021660 [DOI] [PubMed] [Google Scholar]
- 13.James AL, Bai TR, Mauad T, et al. . Airway smooth muscle thickness in asthma is related to severity but not duration of asthma. Eur Respir J 2009; 34: 1040–1045. doi: 10.1183/09031936.00181608 [DOI] [PubMed] [Google Scholar]
- 14.Araujo BB, Dolhnikoff M, Silva LFF, et al. . Extracellular matrix components and regulators in the airway smooth muscle in asthma. Eur Respir J 2008; 32: 61–69. doi: 10.1183/09031936.00147807 [DOI] [PubMed] [Google Scholar]
- 15.Postma DS, Brightling C, Baldi S, et al. . Exploring the relevance and extent of small airways dysfunction in asthma (ATLANTIS): baseline data from a prospective cohort study. Lancet Respir Med 2019; 7: 402–416. doi: 10.1016/S2213-2600(19)30049-9 [DOI] [PubMed] [Google Scholar]