Abstract
Introduction
Type 2 diabetes (T2D) and non-alcoholic fatty liver disease (NAFLD) show a rapidly increasing incidence worldwide. Although both diseases often occur in the same patient population, their mutual influence is not fully understood. We therefore aimed at analyzing the impact of T2D on the incidence of NAFLD in a large cohort of outpatients in Germany.
Research design and methods
32 201 patients with T2D diagnosed between 2012 and 2018 were identified in the IQVIA Disease Analyzer database. Probability of NAFLD was analyzed using Cox regression models.
Results
The cumulative incidence of NAFLD within the 7-year observation period was 4.3%. The probability of NAFLD was significantly higher among patients with T2D with increased body mass index but not hemoglobin A1c. Prescriptions of sodium-glucose cotransporter-2 inhibitors (HR: 0.54, 95% CI 0.45 to 0.64), glucagon-like peptide-1 receptor antagonists (HR: 0.65, 95% CI 0.52 to 0.81), and insulin (HR: 0.72, 95% CI 0.62 to 0.8) were significantly associated with lower incidence of NAFLD.
Conclusion
Our data from a large population-based cohort of patients with T2D identified sociodemographic and therapeutic parameters associated with NAFLD incidence in patients with T2D which should be taken into account for novel therapeutic concepts.
Keywords: NAFLD, insulin, metformin
Significance of this study.
What is already known about this subject?
Type 2 diabetes and non-alcoholic fatty liver disease often occur in the same patient population; however, their mutual influence is not fully understood.
What are the new findings?
The probability of non-alcoholic fatty liver disease is significantly higher among patients with type 2 diabetes.
Increased body mass index but not hemoglobin A1c might represent additional risk factors for the development of non-alcoholic fatty liver disease in patients with type 2 diabetes.
Prescriptions of sodium-glucose cotransporter-2 inhibitors, glucagon-like peptide-1 receptor antagonists, and insulin are significantly associated with lower incidence of non-alcoholic fatty liver disease.
How might these results change the focus of research or clinical practice?
Our data suggest that patients with type 2 diabetes should be included in surveillance strategies for early detection of non-alcoholic fatty liver disease.
Introduction
Non-alcoholic fatty liver disease (NAFLD) has been defined as ‘the accumulation of hepatic steatosis in ≥5% of hepatocytes in the absence of excessive alcohol consumption’. For many years, the incidence of NAFLD has constantly risen. NAFLD currently affects about one in four people in the Western world, adding up to nearly 80 million patients in the USA and 25 million patients in Germany.1–3 While many patients have a benign course of NAFLD, increasingly severe courses with progression to NASH, cirrhosis, and ultimately hepatocellular carcinoma (HCC) are observed. Currently, NAFLD already represents one of the most common conditions leading to liver transplantation in many developed countries.4 5 Due to its initially asymptomatic course, NAFLD tends to be underdiagnosed. Patients are usually diagnosed at advanced stages and specific therapies by specialists are often initiated late.6 In this setting, effective risk stratification of patients could help to both increase the referral of patients at high risk and decrease the referral of those at low risk to liver specialists, thereby improving healthcare access and resource allocation to those who need it the most.7
Type 2 diabetes (T2D) represents one of the most important risk factors for the development of NAFLD as well as for the progression of NAFLD into cirrhosis and HCC.8 Moreover, the presence of T2D was identified as an independent risk factor for mortality in patients with NAFLD.9–12 Although both diseases often occur in the same patient population, data on their mutual influence, especially in the context of therapeutic interventions, are not fully understood. We therefore aimed at identifying patient-related and therapeutic factors which are associated with an increased incidence of NAFLD in different subgroups of patients with T2D.
Methods
Database
This study was based on data from the Disease Analyzer database (IQVIA), which compiles drug prescriptions, diagnoses, and basic medical and demographic data obtained directly and in anonymous format from computer systems used in the practices of general practitioners and specialists.13 The database covers approximately 3% of all outpatient practices in Germany. Diagnoses (according to International Classification of Diseases, 10th Revision (ICD-10)), prescriptions (according to the Anatomical Therapeutic Chemical classification system), and the quality of reported data are being monitored by IQVIA. In Germany, the sampling methods used to select physicians’ practices are appropriate for obtaining a representative database of general and specialized practices.13 Finally, this database has already been used in previous studies focusing on diabetes14 as well as NAFLD.15
Study population
This retrospective cohort study included adult patients (≥18 years) with an initial diagnosis of T2D (index date) and at least one antihyperglycemic prescription between January 2012 and December 2018. Further inclusion criteria were availability of at least one body mass index (BMI) and at least one hemoglobin A1c (HbA1c) value between the index date and the NAFLD diagnosis or at the end of follow-up (when no NAFLD diagnosis). Patients with NAFLD diagnosis prior to or on the index date were excluded (figure 1). The ‘Disease Analyzer’ database, used for analyses, contains anonymized electronic patient records. Patient data were analyzed in aggregated form without individual data being available. An individual consent form was not obtained following national and European legislation.
Figure 1.
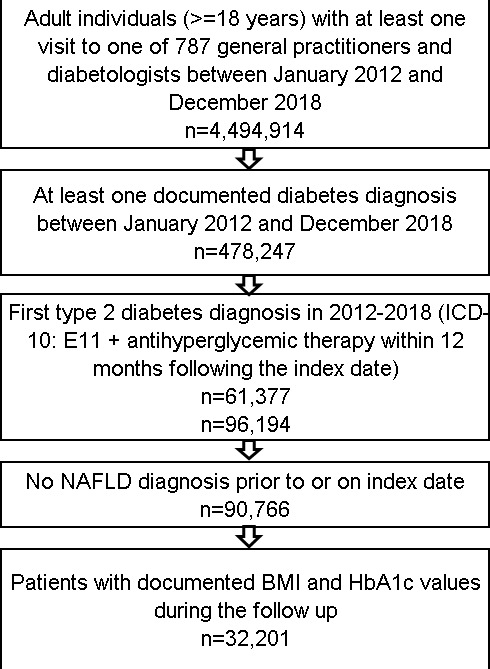
Selection of study patients. BMI, body mass index; HbA1c, hemoglobin A1c; ICD-10, International Classification of Diseases, 10th Revision; NAFLD, non-alcoholic fatty liver disease.
Study outcomes and statistical analyses
The main study outcome was the incidence of NAFLD within up to 7 years after the index date. A Cox proportional hazards regression model was used to estimate the relationship between predefined variables and NAFLD. These variables included age groups (18–50, 51–60, 61–70, 71–80, >80 years), sex (male, female), practice specialty (general practice, diabetologist practice), comorbidities documented prior to NAFLD diagnosis or end of follow-up when no NAFLD was diagnosed (diabetic renal complications (ICD-10: E11.2, N18, N19), diabetic neuropathy (ICD-10: N11.4), lipid metabolism disorders (ICD-10: E78), hypertension (ICD-10: I10), chronic coronary heart disease (ICD-10: I24, I25 excluding I25.2), myocardial infarction (ICD-10: I21–I23, I25.2), stroke/transient ischemic attack (TIA) (ICD-10: I64, I64, G45), peripheral vascular disease (ICD-10: E11.5, I70.2, I73.9)), last HbA1c value (<6.5%, 6.5–7.4%, 7.5%–8.4%, 8.5%–9.4%, ≥9.5%), last BMI value (<25.0, 25.0–29.9, 30.0–34.9, ≥35 kg/m2), and antihyperglycemic therapy prescribed prior to NAFLD diagnosis or end of follow-up (metformin, sulfonylureas, dipeptidyl peptidase-4 (DPP-4) inhibitors, sodium-glucose cotransporter-2 (SGLT2) inhibitors, glucagon-like peptide-1 (GLP-1) receptor agonists, other oral antihyperglycemic drugs (glinides, acarbose, glitazones), and insulin). Additionally, to the main model, each antihyperglycemic drug class was compared with metformin monotherapy as the reference group, and a sensitivity analysis was done including the last prescribed antihyperglycemic drugs, also comparing with metformin monotherapy. As 27 variables were included in the model, a Bonferroni correction for p value was performed, and a p value of <0.05 was considered statistically significant. All analyses were carried out using SAS V.9.4.
Results
Basic characteristics of the study sample
The present study included 32 201 patients with T2D from 729 general and 58 diabetologist practices. The characteristics of study subjects are summarized in table 1. The mean age (SD) was 63.4 years (13.23 years), and 45.6% of patients were female and 44.4% were male. The majority of patients with T2D (66.2%) were treated in general practices, while 33.8% were treated in diabetologist practices. The average HbA1c was 7.2% (SD: 1.4%). The mean BMI was 30.9 kg/m2 (SD: 6.0 kg/m2). Hypertension (76.9%), lipid metabolism disorders (49.8%), and chronic coronary heart disease (24.7%) were the most frequently diagnosed comorbidities, while less than 10% of the study sample had a history of myocardial infarction (8.3%) or stroke/TIA (8.4%) prior to the diagnosis of NAFLD (table 1). In terms of antidiabetic treatment, metformin was the most frequently prescribed drug (79.1%), followed by insulin (41.5%) and DPP-4 inhibitors (39.2%). SGLT2 inhibitors (19.1%), sulfonylureas (10.5%), and GLP-1 receptor agonists (8.2%) were prescribed less frequently (table 1).
Table 1.
Basic characteristics of the study sample
| Variable | Proportion affected among patients with diabetes, n (%) (N=32 201) |
| Age, mean (SD) | 63.4 (13.3) |
| 18–50 | 5520 (17.1) |
| 51–60 | 7595 (23.6) |
| 61–70 | 8330 (23.9) |
| 71–80 | 7836 (24.3) |
| >80 | 2920 (9.1) |
| Women | 14 669 (45.6) |
| Men | 17 532 (54.4) |
| General practice | 21 310 (66.2) |
| Specialized diabetes practices | 10 891 (33.8) |
| Codiagnoses documented prior to NAFLD diagnosis or end of follow-up | |
| Diabetic renal complications | 6177 (19.2) |
| Diabetic neuropathy | 3582 (11.1) |
| Lipid metabolism disorders | 16 047 (49.8) |
| Hypertension | 24 749 (76.9) |
| Chronic coronary heart disease | 7965 (24.7) |
| History of myocardial infarction | 2681 (8.3) |
| History of stroke/TIA | 2707 (8.4) |
| Peripheral vascular disease | 4484 (13.9) |
| HbA1c, mean (SD) | 7.2 (1.4) |
| <6.5 | 10 682 (33.2) |
| 6.5–7.4 | 11 168 (34.7) |
| 7.5–8.4 | 5642 (17.5) |
| 8.5–9.4 | 2481 (7.7) |
| ≥9.5 | 2228 (6.9) |
| BMI, mean (SD) | 30.9 (6.0) |
| <25.0 | 4906 (15.2) |
| 25.0–29.9 | 10 416 (32.4) |
| 30.0–34.9 | 9283 (28.8) |
| ≥35 | 7596 (23.6) |
| Antihyperglycemic drugs prescribed prior to NAFLD diagnosis or end of follow-up | |
| Metformin | 25 459 (79.1) |
| Sulfonylureas | 3370 (10.5) |
| DPP-4 inhibitors | 12 620 (39.2) |
| SGLT2 inhibitors | 6163 (19.1) |
| GLP-1 receptor agonists | 2647 (8.2) |
| Insulin | 13 366 (41.5) |
| Other drugs | 1195 (3.7) |
Proportions of patients given in %, unless otherwise indicated.
BMI, body mass index; DPP-4, dipeptidyl peptidase-4; GLP-1, glucagon-like peptide-1; HbA1c, hemoglobin A1c; NAFLD, non-alcoholic fatty liver disease; SGLT2, sodium-glucose cotransporter-2; TIA, transient ischemic attack.
Evaluation of factors associated with a higher incidence of NAFLD in patients with T2D
During the 7-year observation period, a total of 1377 (4.3%) patients with T2D had their first diagnosis of NAFLD. The mean duration until diagnosis of NAFLD was 3.1 (SD: 2.0) years. The probability of NAFLD was significantly higher in patients with BMI 30.0–34.9 kg/m2 (HR: 1.60, 95% CI 1.43 to 1.79), BMI ≥35 kg/m2 (HR: 1.57, 95% CI 1.40 to 1.76), and BMI 25.0–34.9 kg/m2 (HR: 1.27, 95% CI 1.13 to 1.42) as compared with BMI <25 kg/m2. In contrast, the probability of NAFLD was not increased in patients with T2D with increased HbA1c values compared with patients with an HbA1c <6.5%. Interestingly, we observed a distinctive impact on antidiabetics on the occurrence of NAFLD. As such, prescriptions of SGLT2 inhibitors (HR: 0.54, 95% CI 0.45 to 0.64), GLP-1 receptor antagonists (HR: 0.65, 95% CI 0.52 to 0.81), and insulin (HR: 0.72, 95% CI 0.62 to 0.83) were significantly associated with a lower incidence of NAFLD compared with metformin monotherapy (table 2). These associations were confirmed in the second model including the last antihyperglycemic therapy. Finally, pre-existing comorbidities were not significantly associated with NAFLD, although lipid metabolism disorders showed a trend toward a higher probability of NAFLD (table 2).
Table 2.
Association between defined variables and NAFLD in patients with diabetes followed in general and diabetologist practices in Germany (Cox regression models)
| Variable | HR (95% CI) | P value |
| Age 18–50 | 1.90 (1.38 to 2.62) | <0.001 |
| Age 51–60 | 1.87 (1.37 to 2.54) | <0.001 |
| Age 61–70 | 1.71 (1.26 to 2.31) | <0.001 |
| Age 71–80 | 1.20 (0.88 to 1.64) | 0.251 |
| Age >80 | Reference | |
| Women | Reference | |
| Men | 1.00 (0.90 to 1.11) | 0.977 |
| General practice | Reference | |
| Diabetologist practice | 0.99 (0.87 to 1.12) | 0.745 |
| Codiagnoses documented prior to NAFLD diagnosis or end of follow-up | ||
| Diabetic renal complications | 1.04 (0.79 to 1.38) | 0.762 |
| Diabetic neuropathy | 0.99 (0.71 to 1.37) | 0.928 |
| Lipid metabolism disorders | 1.16 (1.03 to 1.31) | 0.012 |
| Hypertension | 1.03 (0.92 to 1.16) | 0.606 |
| Chronic ischemic heart disease | 0.89 (0.73 to 1.07) | 0.215 |
| History of myocardial infarction | 1.17 (0.88 to 1.67) | 0.278 |
| History of stroke/TIA | 0.73 (0.51 to 1.04) | 0.082 |
| Peripheral vascular disease | 0.79 (0.58 to 1.07) | 0.126 |
| HbA1c <6.5 | Reference | |
| HbA1c 6.5–7.4 | 1.02 (0.90 to 1.16) | 0.761 |
| HbA1c 7.5–8.4 | 1.10 (0.94 to 1.30) | 0.244 |
| HbA1c 8.5–9.4 | 1.14 (0.91 to 1.42) | 0.264 |
| HbA1c ≥9.5 | 1.07 (0.84 to 1.36) | 0.595 |
| BMI <25.0 | Reference | |
| BMI 25.0–29.9 | 1.27 (1.13 to 1.42) | <0.001 |
| BMI 30.0–34.9 | 1.60 (1.43 to 1.79) | <0.001 |
| BMI ≥35 | 1.57 (1.40 to 1.76) | <0.001 |
| Antihyperglycemic drugs prescribed prior to NAFLD diagnosis or end of follow-up | ||
| Metformin monotherapy (no further therapy) | Reference | |
| Sulfonylureas | 0.91 (0.62 to 1.35) | 0.651 |
| DPP-4 inhibitors | 1.07 (0.94 to 1.24) | 0.313 |
| SGLT2 inhibitors | 0.54 (0.45 to 0.64) | <0.001 |
| GLP-1 receptor agonists | 0.65 (0.52 to 0.81) | <0.001 |
| Insulin | 0.72 (0.62 to 0.83) | <0.001 |
| Other drugs | 0.81 (0.58 to 1.12) | 0.201 |
Significant values are in bold.
BMI, body mass index; DPP-4, dipeptidyl peptidase-4; GLP-1, glucagon-like peptide-1; HbA1c, hemoglobin A1c; NAFLD, non-alcoholic fatty liver disease; SGLT2, sodium-glucose cotransporter-2; TIA, transient ischemic attack.
Discussion
By analyzing a total of 32 201 patients with T2D diagnosed between 2012 and 2018, we demonstrate that the probability of NAFLD was significantly higher among patients with T2D with increased BMI compared with those with normal or low BMI. Strikingly, SGLT2 inhibitors, GLP-1 receptor antagonists, and insulin therapy significantly reduced the probability of NAFLD in our cohort, while DPP-4 inhibitors had no influence on the incidence of NAFLD in patients with T2D.
T2D is a major risk factor for the development of NAFLD as well as for the progression of NAFLD to cirrhosis and HCC.8 However, data on their mutual influence, especially in the context of therapeutic interventions, are not fully understood. Both diseases are referred to as manifestations of metabolic syndrome, which show a dramatically increasing incidence in Western countries.16 We found a cumulative incidence of NAFLD of 4.3% within the 7-year observation period. Notably, our data regarding a higher incidence of NAFLD in patients with T2D with elevated BMI were in line with recent analyses, for example, by Bhatt et al17 suggesting that BMI was significantly higher in patients with NAFLD compared with a control group without NAFLD. Moreover, recent meta-analyses demonstrated that the prevalence of NAFLD in patients with T2D is influenced by total cholesterol concentrations, triglycerides, BMI, HbA1c, high-density lipoprotein, low-density lipoprotein, aspartate transaminase, and alanine transaminase.18 These data further support the hypothesis that patients with T2D might not only demonstrate higher incidence of NAFLD but also develop more aggressive disease courses with occurrence of NASH, cirrhosis and HCC.19 Since our database does not feature laboratory values or results from histological specimen, we were unable to validate these previous data in our cohort. Nevertheless, our data clearly argue for an elevated risk of NAFLD in patients with T2D and therefore should trigger a particular awareness for NAFLD in the population with diabetes.
T2D represents a risk factor for various diseases from the gastroenterological, cardiological and neurological field. Surprisingly, when codiagnoses documented prior to NAFLD diagnosis or end of follow-up were analyzed, no specific clustering of individual diseases (hypertension, chronic ischemic heart disease, stroke/TIA, peripheral vascular disease) could be identified, further highlighting that T2D represents a strong and independent risk factor for the development of NAFLD. The strong association between T2D and NAFLD would argue for a specific treatment of all patients with T2D by specialized physicians. However, when comparing ‘risks’ for being diagnosed with NAFLD, no differences between general or specialized practices were found.
T2D is further associated with overweight and obesity in many cases. Both have increasingly been associated with chronic inflammation, oxidative stress, and the upregulation of hepatotoxic cytokines, all mechanisms implicated in the pathophysiology of NAFLD. Just recently different treatment options directly interfering with these pathophysiological mechanisms have been proposed. GLP-1 and SGLT2, which induce a pronounced weight loss, seem to be protective for NAFLD in our analysis. Surprisingly, insulin as an ‘anabolic hormone’ also reduced the risk of NAFLD. This could indicate that in T2D without coexisting obesity, the decisive factor for the incidence of NAFLD is how well the diabetes (and not the overweight) is controlled. Conversely, for the development of NAFLD in obese T2D, the high BMI is decisive for NAFLD incidence, and in this setting presumably only the weight-reducing antidiabetic drugs show a benefit. However, the negative association of insulin with NAFLD may also be explained with metformin as a reference group in the regression analysis. Metformin might have hepatotoxic effects on injured livers and therefore promote NAFLD, which could potentially explain that a weight loss-inducing agent is associated with a higher incidence of NAFLD.20 Nevertheless, data regarding the effect of different antidiabetics on the development of NAFLD are conflicting and the pathophysiology leading to NAFLD in T2D is complex and only poorly understood. While our analysis cannot elucidate molecular mechanism, our data should trigger further functional studies in this context.
Our study has numerous limitations, but these are due to the study design and thus cannot be avoided.21 In brief, the German Disease Analyzer database does not contain larger panels of laboratory values or histological parameters and thus analyses correlating NAFLD stages or grades with the effect size are not possible. Similarly, data on socioeconomic status (eg, education and income of patients) as well as lifestyle-related risk factors (eg, smoking, alcohol consumption, and physical activity) are also lacking. Moreover, we were unable to perform analyses based on specific laboratory values for NAFLD. Additionally, the individual duration of T2D is missing within the database. Finally, we cannot exclude selection bias in our study for those with diagnosis of T2D, meaning that patients who have an established diagnosis of T2D may be more likely to be examined for NAFLD. Nevertheless, it is important to note that the German Disease Analyzer database has been extensively validated in different studies and was shown to be representative of the German outpatient sector.13 22
In summary, we present data from a large German primary care provider database showing that T2D is associated with an increased incidence of NAFLD, irrespective of other comorbidities or patients’ characteristics. Thus, along with previous data, our study suggests that the clinical management of patients with T2D should include a careful and structured work-up of NAFLD in order to improve long-term outcomes in these patients. As an example, all patients with NAFLD might be presented in a ‘metabolic board’ and discussed with dedicated hepatologists in order to recognize NAFLD as early as possible.
Footnotes
Contributors: SHL, CR, KK, and TL designed the study. KK performed the statistical analyses and generated the figures and tables. SHL, CR, and KK wrote the manuscript. TL, AK, ML, and MJ provided intellectual input and corrected the manuscript. All authors agreed to the final version of the manuscript.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement
All data relevant to the study are included in the article. Data will be made available upon meaningful request.
References
- 1.Wong W-K, Chan W-K. Nonalcoholic fatty liver disease: a global perspective. Clin Ther 2021;43:473–99. 10.1016/j.clinthera.2021.01.007 [DOI] [PubMed] [Google Scholar]
- 2.European Association for the Study of the Liver (EASL), European Association for the Study of Diabetes (EASD), European Association for the Study of Obesity (EASO) . EASL-EASD-EASO clinical practice guidelines for the management of non-alcoholic fatty liver disease. J Hepatol 2016;64:1388–402. 10.1016/j.jhep.2015.11.004 [DOI] [PubMed] [Google Scholar]
- 3.Roeb E, Steffen HM, Bantel H. S2k-Leitlinie nicht alkoholische Fettlebererkrankungen AWMF register NR. 021-025 version Januar 2015, Erstauflage. Z Gastroenterol 2015;53:668–723. [DOI] [PubMed] [Google Scholar]
- 4.Gadiparthi C, Spatz M, Greenberg S, et al. Nafld epidemiology, emerging pharmacotherapy, liver transplantation implications and the trends in the United States. J Clin Transl Hepatol 2020;8:215–21. 10.14218/JCTH.2020.00014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Murag S, Ahmed A, Kim D. Recent epidemiology of nonalcoholic fatty liver disease. Gut Liver 2021;15:206–16. 10.5009/gnl20127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Blais P, Husain N, Kramer JR, et al. Nonalcoholic fatty liver disease is underrecognized in the primary care setting. Am J Gastroenterol 2015;110:10–14. 10.1038/ajg.2014.134 [DOI] [PubMed] [Google Scholar]
- 7.Vieira Barbosa J, Lai M. Nonalcoholic fatty liver disease screening in type 2 diabetes mellitus patients in the primary care setting. Hepatol Commun 2021;5:158–67. 10.1002/hep4.1618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Singh A, Garg R, Lopez R, et al. Diabetes liver fibrosis score to detect advanced fibrosis in diabetics with nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol 2021;38. 10.1016/j.cgh.2021.01.010 [DOI] [PubMed] [Google Scholar]
- 9.Hossain N, Afendy A, Stepanova M, et al. Independent predictors of fibrosis in patients with nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol 2009;7:1224–9. 10.1016/j.cgh.2009.06.007 [DOI] [PubMed] [Google Scholar]
- 10.Stepanova M, Rafiq N, Makhlouf H, et al. Predictors of all-cause mortality and liver-related mortality in patients with non-alcoholic fatty liver disease (NAFLD). Dig Dis Sci 2013;58:3017–23. 10.1007/s10620-013-2743-5 [DOI] [PubMed] [Google Scholar]
- 11.Younossi ZM, Otgonsuren M, Henry L, et al. Association of nonalcoholic fatty liver disease (NAFLD) with hepatocellular carcinoma (HCC) in the United States from 2004 to 2009. Hepatology 2015;62:1723–30. 10.1002/hep.28123 [DOI] [PubMed] [Google Scholar]
- 12.Younossi ZM, Gramlich T, Matteoni CA, et al. Nonalcoholic fatty liver disease in patients with type 2 diabetes. Clin Gastroenterol Hepatol 2004;2:262–5. 10.1016/S1542-3565(04)00014-X [DOI] [PubMed] [Google Scholar]
- 13.Rathmann W, Bongaerts B, Carius H-J, et al. Basic characteristics and representativeness of the German disease analyzer database. Int J Clin Pharmacol Ther 2018;56:459–66. 10.5414/CP203320 [DOI] [PubMed] [Google Scholar]
- 14.Zingel R, Bohlken J, Riedel-Heller S, et al. Association between low-density lipoprotein cholesterol levels, statin use, and dementia in patients followed in German general practices. J Alzheimers Dis 2021;79:37–46. 10.3233/JAD-201176 [DOI] [PubMed] [Google Scholar]
- 15.Bach L, Kostev K, Schiffmann L, et al. Association between thyroid gland diseases and breast cancer: a case-control study. Breast Cancer Res Treat 2020;182:207–13. 10.1007/s10549-020-05675-6 [DOI] [PubMed] [Google Scholar]
- 16.Bang KB, Cho YK. Comorbidities and metabolic derangement of NAFLD. J Lifestyle Med 2015;5:7–13. 10.15280/jlm.2015.5.1.7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bhatt K, Pranav V, Dipika Y, et al. Prevalence of nonalcoholic fatty liver disease in type 2 diabetes mellitus and its relation with insulin resistance in South Gujarat region. J Mahatma Gandhi Inst Med Sci 2017;22:8. 10.4103/0971-9903.202001 [DOI] [Google Scholar]
- 18.Dharmalingam M, Yamasandhi PG. Nonalcoholic fatty liver disease and type 2 diabetes mellitus. Indian J Endocrinol Metab 2018;22:421. 10.4103/ijem.IJEM_585_17 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Younossi ZM, Gramlich T, Matteoni CA, et al. Nonalcoholic fatty liver disease in patients with type 2 diabetes. Clin Gastroenterol Hepatol 2004;2:262–5. 10.1016/s1542-3565(04)00014-x [DOI] [PubMed] [Google Scholar]
- 20.Deng Z, Bi S, Jiang M. Endogenous H2S-activated orthogonal second near-infrared emissive nanoprobe for in situ ratiometric fluorescence imaging of metformin-induced liver injury. ACS Nano 2021;15:3201–11. 10.1021/acsnano.0c09799 [DOI] [PubMed] [Google Scholar]
- 21.Labenz C, Huber Y, Michel M, et al. Nonalcoholic fatty liver disease increases the risk of anxiety and depression. Hepatol Commun 2020;4:1293–301. 10.1002/hep4.1541 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Becher H, Kostev K, Schröder-Bernhardi D. Validity and representativeness of the "Disease Analyzer" patient database for use in pharmacoepidemiological and pharmacoeconomic studies. Int J Clin Pharmacol Ther 2009;47:617–26. 10.5414/cpp47617 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data relevant to the study are included in the article. Data will be made available upon meaningful request.


