Abstract
Mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes (MELAS) syndrome is one of the most common maternally inherited mitochondrial disorders, with no specific treatment available. We report a case of a 34-year-old female in whom symptoms of MELAS were initially misdiagnosed as herpes simplex encephalitis (HSE). Her clinical course was marked by an acute episode of consciousness disturbance with newly developed lesions on brain MRI five years after disease onset and followed by progressive sensorineural hearing loss. Brain imaging sequences throughout the seven years of her illness are presented. The final diagnosis of MELAS syndrome was confirmed by m.3243A>G mitochondrial mutation. In conclusion, understanding the overlapping imaging features between MELAS syndrome and other mimickers, such as HSE or ischemic stroke, is crucial to help establish early diagnosis and initiate appropriate treatment.
Keywords: Mitochondrial Encephalomyopathy, MELAS, Magnetic Resonance Imaging, Herpes Simplex Encephalopathy
Introduction
Mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes (MELAS) syndrome is a multisystem disorder with extensive phenotypic variability. Its clinical manifestations include stroke-like episodes, focal or generalized seizures, recurrent migraine-like headaches, vomiting, short stature, hearing loss, and muscle weakness. The most common mutation associated with MELAS syndrome is the m.3243A>G in the MT-TL1 (mitochondrially encoded tRNA leucine 1) gene [6].
Case report
A 34-year-old right-handed woman was hospitalized three times due to headache, vomiting, abnormal behavior, seizures with no fever. Her birth, child development and early adulthood were unremarkable. She had been a general practitioner with short stature as compared with her siblings and no prior medical conditions.
The patient first presented with symptoms of consciousness disturbance, seizure and vomiting at the age of 27 (2013), she was admitted to a different hospital. During this episode, her lumbar puncture results were lost. Her brain images, including computed tomography (CT) and magnetic resonance imaging (MRI), are shown in Fig. 1 and 2, respectively. She was diagnosed with herpes simplex encephalitis (HSE). After discharge, while other symptoms resolved, her communication became mildly impaired, evidenced by slower reaction speed. She was still independent in activities of daily living and continued work as a doctor.
Fig. 1.
Brain CT scan on first admission. Arrows indicate hyperintense signal involving bilateral basal ganglia, likely calcification.
Fig. 2.
Brain MRI findings. Fluid-attenuated inversion recovery (FLAIR), diffusion weighted imaging (DWI), and apparent diffusion coefficient (ADC) images at three presentations. FLAIR images demonstrate hyperintense edema in the left and then right temporal and occipital lobes. These do not correspond to any vascular territory and appear as local mass effect (arrowheads, 2018) and subsequently old lesions with partial encephalomalacia (green arrow, 2020). DWI shows hyperintense cortical areas of restricted diffusion that are hypointense on ADC (yellow arrow), and subcortical vasogenic edema that is hyperintense on ADC (red arrows). (Color version of figure is available online)
Five years later (2018), at the age of 32, the patient was admitted to our hospital because of similar symptoms. Routine laboratory tests included complete blood count, liver and kidney function tests, electrolytes, glycemia, thyroid function tests, and were all normal. MRI of the brain was obtained (Fig. 2, 3) and revealed new lesions in the right temporal and occipital lobes. Cerebrospinal fluid (CSF) analyses were normal except for increased lactate levels (4 mmol/L, normal range 1.1-2.4 mmol/L). Tests for herpes simplex, cytomegalovirus, tuberculosis, bacteria and fungi in CSF were negative, as were tests for Japanese encephalitis virus, human immunodeficiency virus (HIV) and syphilis. Antinuclear Antibody (ANA) and anti-dsDNA were negative. To investigate whether paraneoplastic or autoimmune encephalitis was possible, CT scan of the chest and abdomen was performed and came back normal. Paraneoplastic antineuronal serum antibodies (Anti-NMDAR antibody, Anti-Yo, Anti-Hu, Anti-Ri, Anti-Amphiphysin, Anti-Ma2 antibody, Anti-CV2 (CRMP5) antibody, anti-GAD antibody) also turned out negative. Serum test for HSV IgG antibody was positive while IgM antibody was absent. An electroencephalography (EEG) study showed slow waves in the right temporal lobe and no epileptiform discharges. Again, she was diagnosed with recurrent HSE and treated with acyclovir and corticosteroid. This hospitalization was followed by 2 years of progressive dementia, progressive hearing loss, but no seizure.
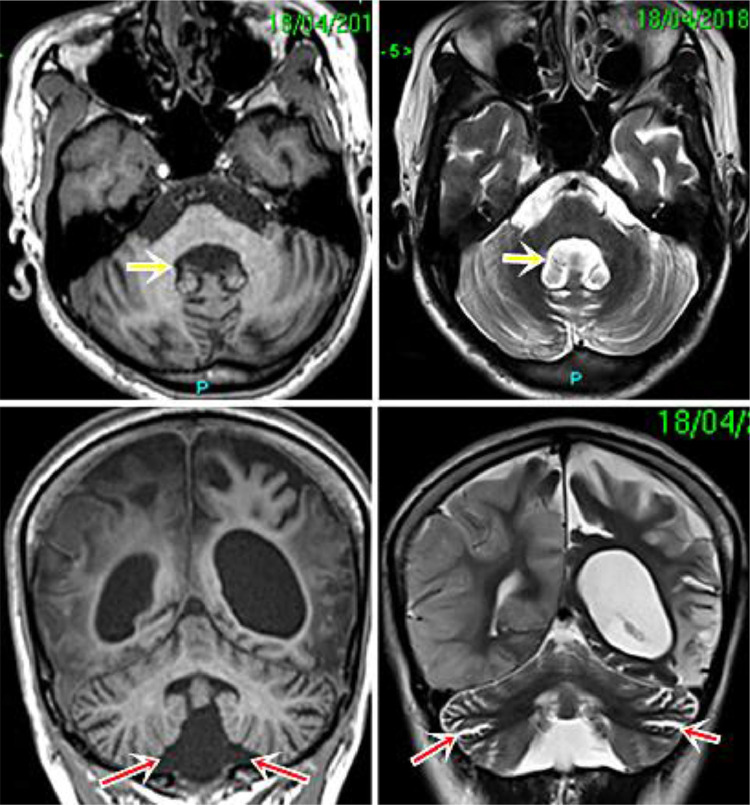
Fig. 3.
Cerebellar atrophy, a common neurological manifestation in mitochondrial disorders. Axial and coronal T1 and T2 images show signs of cerebellar atrophy: reduction in cerebellar volume, enlargement of the fourth ventricle (yellow arrow) and sulci and cisterna magna (red arrows). (Color version of figure is available online)
The patient was admitted to our hospital the third time at the age of 34, when she developed headache and vomiting. Neither fever nor abnormal behavior was present. Neurologic examination revealed substantial cognitive deficits, impaired verbal communication and severe hearing loss. Again, routine laboratory tests, including complete blood count, liver and kidney function tests, electrolytes, glycemia, thyroid function tests, were normal. Plasma lactate was elevated at the level of 3 mmol/L (normal range 0.5-2.2 mmol/L). Echocardiography and electromyography were normal. Fundus photography revealed pigmentary retinopathy. A repeated MRI brain was performed, disclosing no new lesions (Fig. 2). This time, the clinical and imaging features were felt to be suggestive of mitochondrial encephalomyopathy, hence genetic studies were ordered. This revealed an m.3243A>G mutation, a substitution of G for A at base pair 3243 in the mitochondrial genome, which confirmed the diagnosis of MELAS (Fig. 4). The patient was started on coenzyme Q-10, thiamine, folic acid and acetyl L-carnitine. Her condition gradually improved after several months and she was eventually able to communicate well with hearing aids.
Fig. 4.
Analysis of mitochondrial genome. Direct sequencing of MT-TL1 gene from peripheral blood revealed point mutation m.3242A>G.
Discussion
Overall, our patient met the diagnostic criteria of MELAS, which include i) stroke-like episodes before age 40 years, ii) encephalopathy characterized by seizures and/or dementia, and either iii) blood lactic acidosis or the presence of ragged-red fibers on muscle biopsy. The diagnosis is further secured as she also had a normal early development, recurrent headache and vomiting [10], and short stature (weight 36kg, height 155 cm, and body mass index 14,9 kg/m2 at her third hospitalization).
The pathogenesis of MELAS syndrome remains poorly understood. Genetic abnormalities play a key role, with 80% of afflicted individuals found to carry the m.3243A>G mutation in MT-TL1 gene. This gene is located on the p-arm of mitochondrial DNA at position 12 and encodes tRNA leucine 1. Its mutation interrupts mitochondrial protein translation, decreases energy production, eventually leading to multi-organ dysfunction, a manifestation commonly observed in MELAS syndrome. Furthermore, microvasculature angiopathy and nitric oxide (NO) deficiency have also been suggested to contribute to the phenotype and complications of MELAS syndrome [6].
The patient was misdiagnosed with HSE twice, at disease onset and five years later when she first came to our hospital, during which she presented with abrupt consciousness disturbance, heachade, seizures and vomiting. Her brain MRI showed a new lesion involving the right temporal lobe that did not respect vascular territories, which led us to a tentative diagnosis of recurrent HSE.
In retrospect, however, the patient's presentation was not entirely compatible with HSE in several key points. Firstly, her CSF profile was normal - neither lymphocytic pleocytosis nor herpes simplex virus DNA detection by polymerase chain reaction, the gold standard for diagnosing HSE, were found. Secondly, the patient's lesions on brain MRI involved bilateral temporal and occipital lobes, findings that were found to significantly lower the odds of HSE [4,17]. As a result, CT scan of the chest and abdomen, together with serum anti-neuronal antibodies were performed to rule out paraneoplastic and autoimmune encephalitis. As these came back negative, we accepted the original diagnosis of HSE and discharge the patient.
Finally, we were only able to consider the possibility of a metabolic disorder, specifically MELAS syndrome, when the patient subsequently developed recurrent vomiting, headache and remarkable sensorineural hearing loss that was confirmed by audiogram and eventually required hearing aids. These symptoms have not been reported to be common HSE sequelae, which are aphasia and amnesia [18].
Given the rarity and often complexity of its manifestation, diagnosing MELAS syndrome timely is particularly challenging and necessitates understanding of its overlapping features with other conditions, such as HSE, ischemic stroke and posterior reversible encephalopathy syndrome (PRES). Several key differences in imaging studies between MELAS syndrome and HSE are provided in Table 1. Typical findings of MELAS syndrome include stroke-like areas, basal ganglia calcifications and brain atrophy [17,22,23]. Our patient had basal ganglia calcifications (Fig. 1), a feature previously reported by Pauli et al [17], and hyperintense edema in the right temporal and occipital lobes on FLAIR images, which did not correspond to a vascular territory. A retrospective case series reported that the odd of HSE was significantly lowered if lesions outside the temporal lobes are present [4].
Table 1.
Neuroimaging findings of HSE and MELAS.
| MELAS | HSE | ||
|---|---|---|---|
| CT scan | Hyperintense signal and calcification involving bilateral basal ganglia [4,17] | No basal ganglia calcification | |
| MRI | Side | Uni- or bilateral [4] | Unilateral more often than bilateral [4] |
| Location | |||
| Restricted diffusion | Signal intensity on ADC map is usually not reduced or only mildly reduced compared to DWI [1,21] | ||
| Hemorrhage | Infrequent finding | Can present as necrotizing form [5] | |
HSE, herpes simplex encephalitis
MELAS, mitochondrial encephalomyopathy, lactic acidosis and stroke-like episodes
DWI and ADC sequences play a crucial role in distinguishing between ischemic stroke and MELAS syndrome. Ischemic areas are predominantly caused by cytotoxic edema and commonly presented as restricted diffusion and low signal on ADC maps. In contrast, in MELAS syndrome, lesions most often occur due to vasogenic edema, thus the signal intensity on ADC maps is not reduced or only mildly reduced compared to that seen in acute ischemic stroke, and is most likely an indication of reduced cellular energy [7,8,19,20].
Another mimicker of MELAS syndrome is PRES, whose brain images frequently show vasogenic edema in subcortical areas in the occipital and temporal lobes [2,3]. PRES is often preceded by several conditions: hypertensive encephalopathy with acute blood pressure fluctuations, acute or chronic renal diseases, hemolytic and uremic syndrome, eclampsia, or immunosuppressive therapy [14,17,11]. Its reversibility upon removal of precipitated factors have been reported in many cases and often help establish the correct diagnosis.
In conclusion, while magnetic resonance spectroscopy and muscle biopsy were not performed, a stroke-like episode, MRI findings and elevation of lactate in CSF pointed to a diagnosis of MELAS syndrome in our case. The definitive diagnosis was confirmed by molecular genetic testing of mitochondrial DNA. Recognizing the overlapping imaging features between MELAS syndrome and other mimickers, typically HSE, would significantly increase its awareness and help avoid misdiagnosis.
Author's contribution
P. C. Chien and T. M. Trang contributed equally to this article as co-first authors. Each author gave final approval of the version and agreed to be accounted for all aspects of the work in ensuring that questions related to the accuacy of any part of the work.
Footnotes
Acknowledgements: We would like to thank the patient and her family for the permission to publish this report.
Competing Interests: None
Patient Consent: The protocol was reviewed and approved by the Human Research Ethics Committee of the University of Medicine and Pharmacy at Ho Chi Minh City. The study was performed in accordance with the Declaration of Helsinki. The patient wrote informed consent.
REFERENCES
- 1.Abe K., Yoshimura H., Tanaka H, Fujita N, Hikita T, Sakoda S. Comparison of conventional and diffusion-weighted MRI and proton MR spectroscopy in patients with mitochondrial encephalomyopathy, lactic acidosis, and stroke-like events. Neuroradiology. 2004;46(2):113–117. doi: 10.1007/s00234-003-1138-2. [DOI] [PubMed] [Google Scholar]
- 2.Adam G., Ferrier M., Patsoura S, Gramada R, Meluchova Z, Cazzola V. Magnetic resonance imaging of arterial stroke mimics: a pictorial review. Insights Imaging. 2018;9(5):815–831. doi: 10.1007/s13244-018-0637-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brady E., Parikh N.S., Navi BB, Gupta A, Schweitzer AD. The imaging spectrum of posterior reversible encephalopathy syndrome: A pictorial review. Clin Imaging. 2018;47:80–89. doi: 10.1016/j.clinimag.2017.08.008. [DOI] [PubMed] [Google Scholar]
- 4.Chow F.C., Glaser C.A., Sheriff H, Xia D, Messenger S, Whitley R. Use of clinical and neuroimaging characteristics to distinguish temporal lobe herpes simplex encephalitis from its mimics. Clin Infect Dis. 2015;60(9):1377–1383. doi: 10.1093/cid/civ051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Demaerel P., Wilms G., Robberecht W, Johannik K, Van Hecke P, Carton H. MRI of herpes simplex encephalitis. Neuroradiology. 1992;34(6):490–493. doi: 10.1007/BF00598957. [DOI] [PubMed] [Google Scholar]
- 6.El-Hattab A.W., Adesina A.M., Jones J, Scaglia F. MELAS syndrome: Clinical manifestations, pathogenesis, and treatment options. Mol Genet Metab. 2015;116(1–2):4–12. doi: 10.1016/j.ymgme.2015.06.004. [DOI] [PubMed] [Google Scholar]
- 7.Farrar M.A., Lin C.S., Krishnan AV, Park SB, Andrews PI, Kiernan MC. Acute, reversible axonal energy failure during stroke-like episodes in MELAS. Pediatrics. 2010;126(3):e734–e739. doi: 10.1542/peds.2009-2930. [DOI] [PubMed] [Google Scholar]
- 8.Geberhiwot T., Chakrapani A., Hendriksz C. Case 36-2005: a woman with seizure, disturbed gait, and altered mental status. N Engl J Med. 2006;354(10):1096–1097. doi: 10.1056/NEJMc053506. author reply 1096-1097. [DOI] [PubMed] [Google Scholar]
- 9.Gieraerts C., Demaerel P., Van Damme P, Wilms G. Mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes (MELAS) syndrome mimicking herpes simplex encephalitis on imaging studies. J Comput Assist Tomogr. 2013;37(2):279–281. doi: 10.1097/RCT.0b013e3182811170. [DOI] [PubMed] [Google Scholar]
- 10.Hirano M., Ricci E., Koenigsberger MR, Defendini R, Pavlakis SG, DeVivo DC. Melas: an original case and clinical criteria for diagnosis. Neuromuscul Disord. 1992;2(2):125–135. doi: 10.1016/0960-8966(92)90045-8. [DOI] [PubMed] [Google Scholar]
- 11.How J., Blattner M., Fowler S, Wang-Gillam A, Schindler SE. Chemotherapy-associated posterior reversible encephalopathy syndrome: a case report and review of the literature. Neurologist. 2016;21(6):112–117. doi: 10.1097/NRL.0000000000000105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Iizuka T., Sakai F., Suzuki N, Hata T, Tsukahara S, Fukuda M. Neuronal hyperexcitability in stroke-like episodes of MELAS syndrome. Neurology. 2002;59(6):816–824. doi: 10.1212/wnl.59.6.816. [DOI] [PubMed] [Google Scholar]
- 13.Malhotra K., Liebeskind D.S. Imaging of MELAS. Curr Pain Headache Rep. 2016;20(9):54. doi: 10.1007/s11916-016-0583-7. [DOI] [PubMed] [Google Scholar]
- 14.Martin J.N., Jr., Brewer J.M., Blake PG, Owens MY, LaMarca B. PP137. Posterior reversible encephalopathy syndrome (PRES) is a constant component of eclampsia. Pregnancy Hypertens. 2012;2(3):314. doi: 10.1016/j.preghy.2012.04.248. [DOI] [PubMed] [Google Scholar]
- 15.McCabe K., Tyler K., Tanabe J. Diffusion-weighted MRI abnormalities as a clue to the diagnosis of herpes simplex encephalitis. Neurology. 2003;61(7):1015–1016. doi: 10.1212/01.wnl.0000082387.97051.f5. [DOI] [PubMed] [Google Scholar]
- 16.Obeid M., Franklin J., Shrestha S, Johnson L, Quattromani F, Hurst D. Diffusion-weighted imaging findings on MRI as the sole radiographic findings in a child with proven herpes simplex encephalitis. Pediatric radiology. 2007;37(11):1159–1162. doi: 10.1007/s00247-007-0577-z. [DOI] [PubMed] [Google Scholar]
- 17.Pauli W., Zarzycki A., Krzyształowski A, Walecka A. CT and MRI imaging of the brain in MELAS syndrome. Polish journal of radiology. 2013;78(3):61–65. doi: 10.12659/PJR.884010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Riancho J., Delgado-Alvarado M., Sedano MJ, Polo JM, Berciano J. Herpes simplex encephalitis: clinical presentation, neurological sequelae and new prognostic factors. Ten years of experience. Neurol Sci. 2013;34(10):1879–1881. doi: 10.1007/s10072-013-1475-9. [DOI] [PubMed] [Google Scholar]
- 19.Tzoulis C., Bindoff L.A. Serial diffusion imaging in a case of mitochondrial encephalomyopathy, lactic acidosis, and stroke-like episodes. Stroke. 2009;40(2):e15–e17. doi: 10.1161/STROKEAHA.108.523118. [DOI] [PubMed] [Google Scholar]
- 20.Xu W., Wen J., Cao J, Li Y, Geng D. Conventional and diffusional magnetic resonance imaging features of mitochondrial Encephalomyopathy, lactic acidosis, and stroke-like episodes in Chinese patients: a study of 40 cases. J Comput Assist Tomogr. 2018;42(4):510–516. doi: 10.1097/RCT.0000000000000712. [DOI] [PubMed] [Google Scholar]
- 21.Yoneda M., Maeda M., Kimura H, Fujii A, Katayama K, Kuriyama M. Vasogenic edema on MELAS: a serial study with diffusion-weighted MR imaging. Neurology. 1999;53(9):2182–2184. doi: 10.1212/wnl.53.9.2182. [DOI] [PubMed] [Google Scholar]
- 22.Finsterer J., Zarrouk-Mahjoub S. Cerebellar atrophy is common among mitochondrial disorders. Metabolic Brain Disease. 2018;33(4):987–988. doi: 10.1007/s11011-018-0238-y. [DOI] [PubMed] [Google Scholar]
- 23.Rosen L., Phillips S., Enzmann D. Magnetic resonance imaging in MELAS syndrome. Neuroradiology. 1990;32(2):168–171. doi: 10.1007/BF00588572. [DOI] [PubMed] [Google Scholar]