Despite the advance in targeted therapy, lung adenocarcinoma (LUAD) remains one of the leading causes of death in patients with cancer. CD155, a member of poliovirus receptor–related (PRR) family, has been identified as the ligand of the Ig-like receptors tactile (CD96) and DNAM-1 (CD226) on T cells. The interaction of CD155 with CD96 transmits an inhibitory signal and suppresses immune response. The interaction of CD155 with CD226 enhances the immune response.1–3 CD155 expression has been shown to be elevated in many types of cancers4,5 and is involved with immune suppression in melanoma.6 However, how CD155/CD96 is involved in immune response in the tumor microenvironment of LUAD remains unknown.
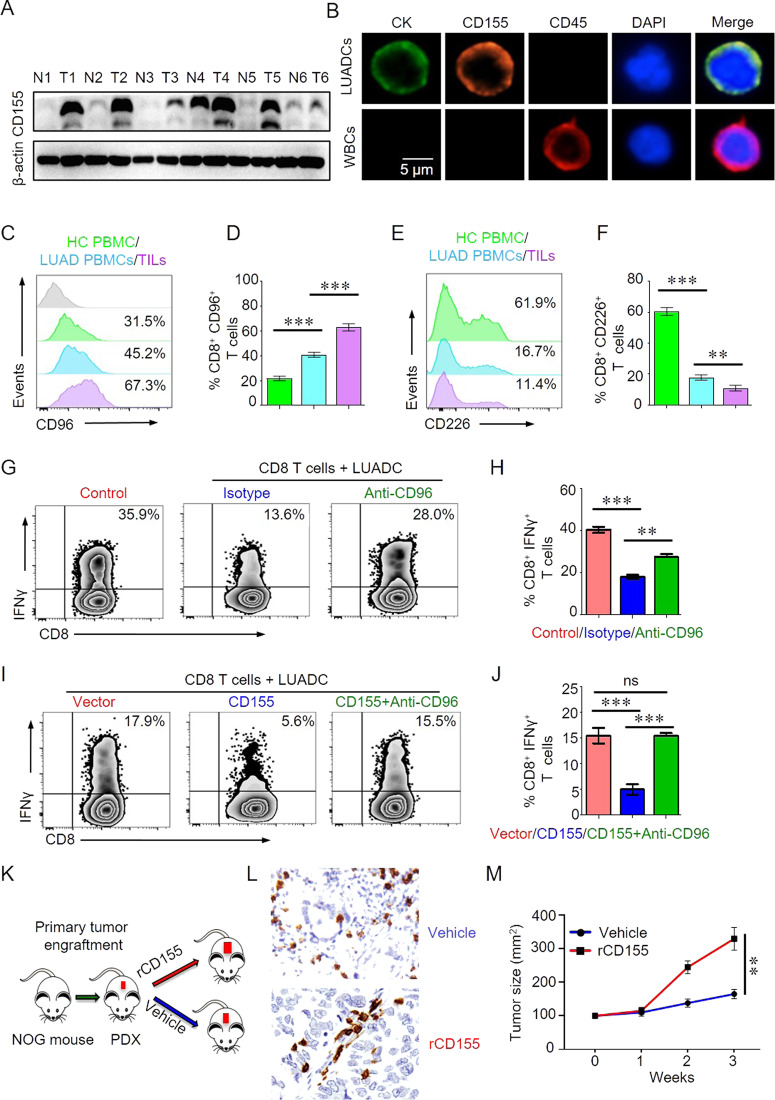
To investigate the expression of CD155 in LUAD tumor, we first collected tumor tissues from LUAD patients and para-tumor tissue were used as control. Western blot result showed that CD155 was increased significantly in LUAD. To further detail CD155 expression in LUAD cells (LUADCs), LUADCs were isolated from tumor tissues of LUAD patients using NanoVelcro as described previously.7 Immunofluorescence staining demonstrated that CD155 was highly expressed by LUADCs (Fig. 1b). The increased expression of CD155 by LUADCs suggested CD155 might be involved with immune response in tumor microenvironment.
Fig. 1.
CD96/CD155 mediated immune suppression and promoted tumor growth in lung adenocarcinoma patients. a CD155 expression in tumor or para-tumor lung tissues from patients with LUAD was measured by western blot. Representative bands were shown. b Primary lung adenocarcinoma (LUAD) cancer cells were isolated and expanded from circulating tumor cells. CD155 expression on primary lung LUAD cells was detected by confocal immunofluorescence. Representative images were shown. c–f CD96 or CD226 expression on CD8 T cells from PBMC of patients with lung adenocarcinoma (LAC) or healthy controls (HC) or tumor infiltrating lymphocytes (TILs) was analyzed by flow cytometry. Representative histograms were shown. Percentages of CD8+CD96+ or CD8+CD226+ cells in PBMC (n = 24) or TILs (n = 18) were summarized and shown as bar graphs. g, h CD8 T cells isolated from HC were stimulated with anti-CD3/CD28 beads and cocultured with or without LUAD cells for 3 days. Anti-CD96 antibody (5 μg/ml) was added in some experiments. IFNγ production in CD8 T cells were measured by flow cytometry. Representatives were shown. Percentage of IFNγ+ CD8 T cells was summarized from 6 independent samples. i, j CD155 or empty vector was transfected into LUADC. CD8 T cells isolated from HC were stimulated with anti-CD3/CD28 beads and cocultured with LUADC-CD155 or LUADC-vector for 3 days. Anti-CD96 antibody (5μg/ml) was added in some experiments. IFNγ production in CD8 T cells were measured by flow cytometry. Representatives were shown. Percentage of IFNγ+ CD8 T cells was summarized from 6 independent samples. k Scheme of the patient-derived xenograft (PDX) experiment. Fresh patient tumor samples were collected and engrafted into NOG mice subcutaneously. One week later, PDX mice were treated with recombinant CD155 (rCD155, 200 μg) or vehicle. l The number of CD8 T cells in tumor microenvironment was measured by immunohistochemistry. Representative images were shown (200×). m Mice were treated with recombinant CD155 or vehicle and tumor growth were monitored. Data were shown as mean ± SEM, **p < 0.01 and ***p < 0.001
CD96, as a co-inhibitory molecule, competes with the co-stimulatory molecule of CD226 for binding to CD155. The lost balance between these two molecules might lead to a hypo- or hyper-immune response. We found that CD8 T cells in PBMC from LUAD patients expressed higher levels of CD96 compared with HC. CD96 expression was even higher in TILs than in T cells from the circulation of LUAD patients (Fig. 1c, d). CD226 expression was substantially lower in CD8 T cells from PBMC of LUAD patients than that in HC. CD226 expression in TILs was further decreased compared with that from the circulation of LUAD patients (Fig. 1e, f). These data indicated that the imbalance of CD155/CD96 and CD155/CD226 signals in LUAD patients could lead to hypo-immune response.
The tumor microenvironment is characterized by increased inhibitory signals and T cells from tumor microenvironment are exhausted. To investigated how LUAD cells affect T cell function in the microenvironment, we performed a T cell-tumor cell culture experiment. We found that IFNγ production in CD8 T cells was decreased dramatically when cocultured with LUAD cells. Blocking CD96 in the T cell-tumor cell co-culture system increased IFNγ production in CD8 T cells (Fig. 1g, h). In addition, IFNγ production in CD8 T cells was further decreased when cocultured with LUAD cells overexpressed CD155. CD96 blockade could neutralize the inhibition mediated by CD155 expression (Fig. 1i, j). Thus, LUAD cells suppress the T cell response through CD155/CD96 signaling in the tumor microenvironment.
To better duplicate the tumor microenvironment similar to LUAD patients, we used a PDX mouse model as described to study the immune response in tumor microenvironment. Previous study shows that CD155/96 is important for NK cells in antitumor immune response. To investigate whether CD155/CD96 affects immune reaction and tumor growth, PDX mice were treated with rCD155 or vehicle (Fig. 1k). The infiltration of CD8 T cells in the tumor microenvironment was decreased to some extent by rCD155 treatment as measured by IHC (Fig. 1l). Importantly, rCD155 treatment promoted tumor growth in the PDX mice (Fig. 1m). The tumor microenvironment has played a critical role in shaping the immune response.8 We showed that blocking CD96 restored CD8 T cell function as inhibited by LUAD cells. LUAD cells impaired CD8 T cell function, which was further inhibited by overexpressing CD155 in cancer cells of LUAD. These data demonstrated a mechanism in which LUAD cells suppress immune response in the tumor microenvironment through CD155/CD96.
Taken together, we demonstrated that LUAD patients with high CD155 expression suffer from immune suppression and experience a poor prognosis, which coincides with up-regulated CD96 expression. Moreover, the inhibition effect can be reversed by CD96 blocking antibody. Blocking CD96 restored IFNγ production in CD8 T cells and neutralized the inhibition of IFNγ production in CD8 T cells mediated by CD155. Animal experiments showed that CD155-mediated LUAD growth might depend on its suppression on antitumor immune response in the tumor microenvironment in PDX mice. Our results suggest that direct interaction between CD155 and CD96 promotes immunosuppression in lung adenocarcinoma (LUAD). Targeting CD155/CD96 to unleash CD8 T cells in the tumor microenvironment could be a novel therapeutic alternative for LUAD patients.
Supplementary information
Acknowledgements
We thank all patients enrolled in this study.
Author contributions
Conception and design: ZK, HZ, and YC. Funding support: ZK. Collection and assembly of data: QL, YL, JZ, WJ, QH, JZ, ZZ, and YS. Data analysis and interpretation: ZK and HZ. Manuscript writing: ZK and HZ. Final approval of manuscript: All authors.
Funding
This work was supported by grants from YFC (2017YFC1308800), National Natural Science Foundation of China to Zunfu Ke (30900650, 81372501, 81572260, 81773299, 81701834, 81502327, 81172232, and 31430030), and Guangdong Natural Science Foundation (2011B031800025, S2012010008378, S2012010008270, S2013010015327, 2013B021800126, 20090171120070, 9451008901002146, 2013B021800126, 2014A030313052, 2014J4100132, 2015A020214010, 2016A020215055, 201704020094, 2013B021800259, 2017B070705002, 16ykjc08, and 2015ykzd07).
Competing interests
The authors declare no competing interests.
Footnotes
These authors contributed equally: Hui Zhang, Qianwen Liu, Yiyan Lei
Supplementary information
The online version of this article (10.1038/s41423-020-00538-y) contains supplementary material.
References
- 1.Shibuya A, et al. DNAM-1, a novel adhesion molecule involved in the cytolytic function of T lymphocytes. Immunity. 1996;4:573–581. doi: 10.1016/S1074-7613(00)70060-4. [DOI] [PubMed] [Google Scholar]
- 2.Bottino C, et al. Identification of PVR (CD155) and Nectin-2 (CD112) as cell surface ligands for the human DNAM-1 (CD226) activating molecule. J. Exp. Med. 2003;198:557–567. doi: 10.1084/jem.20030788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yu X, et al. The surface protein TIGIT suppresses T cell activation by promoting the generation of mature immunoregulatory dendritic cells. Nat. Immunol. 2009;10:48–57. doi: 10.1038/ni.1674. [DOI] [PubMed] [Google Scholar]
- 4.Iguchi-Manaka A, et al. Increased soluble CD155 in the serum of cancer patients. PloS ONE. 2016;11:e0152982. doi: 10.1371/journal.pone.0152982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brooks, J. et al. Perioperative, spatiotemporally coordinated activation of T and NK cells prevents recurrence of pancreatic cancer. Cancer Res. 78, 475–488 (2017). [DOI] [PubMed]
- 6.Inozume T, et al. Melanoma cells control antimelanoma CTL responses via interaction between TIGIT and CD155 in the effector phase. J. investigative Dermatol. 2016;136:255–263. doi: 10.1038/JID.2015.404. [DOI] [PubMed] [Google Scholar]
- 7.Lin M, et al. Nanostructure embedded microchips for detection, isolation, and characterization of circulating tumor cells. Acc. Chem. Res. 2014;47:2941–2950. doi: 10.1021/ar5001617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Whiteside TL. The tumor microenvironment and its role in promoting tumor growth. Oncogene. 2008;27:5904–5912. doi: 10.1038/onc.2008.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.