Abstract
Metastasis and drug resistance are intertwined processes that are responsible for the vast majority of patient deaths from breast cancer. The underlying mechanisms remain incompletely understood. We previously demonstrated that KLF8 activates CXCR4 transcription in metastatic breast cancer. Here, we report a novel role of KLF8-CXCR4 signaling for converting single organ metastasis into multiple organ metastasis associated with chemotherapeutic resistance. We show that KLF8 expression in metastatic breast cancer cells can be over-induced by chemotherapeutic drugs. Analysis of data from large-cohorts of patients indicates that post-chemotherapy there is a close correlation between the aberrant high levels of KLF8 and CXCR4 and that this correlation is well associated with drug resistance, metastasis, and poor prognosis. To mimic their aberrant high levels, we overexpressed KLF8 or CXCR4 in a human breast cancer cell line known to metastasize only to the lungs after intravenous injection in nude mice. As expected, these cells become more resistant to chemotherapeutic drugs. Surprisingly, these KLF8 or CXCR4 overexpressing cells, even implanted orthotopically, metastasized extensively to multiple organs particularly the CXCL12-rich organs. Tube formation assay, Ki67 staining and Western blotting revealed that KLF8 or CXCR4 overexpression enhanced angiogenesis involving increased expression and secretion of VEGF protein. We also found that KLF8 or CXCR4 overexpression strongly enhanced formation of filopodium-like protrusions and proliferation via CXCR4 stimulation in a 3D culture model mimicking the colonization step of metastasis. Taken together, these results suggest that the chemo-induction of KLF8 upregulation be critical for drug resistance and systemic metastasis through enhanced tumor angiogenesis and colonization via CXCR4 over-activation and that KLF8-CXCR4 signaling axis may be a new therapeutic target for drug-resistant breast cancer metastasis.
Keywords: KLF8, CXCR4, drug resistance, metastasis, breast cancer
Introduction
Breast cancer is the most commonly diagnosed cancer and the second leading cause of cancer-associated mortality among women. In 2020, an estimated 276,480 new cases of invasive breast cancer will be diagnosed in the U.S. only [1]. Well-confined primary tumors are curable through chemotherapy or surgery. However, extensive metastasis associated with drug resistance contributes to more than 90% of mortality from breast cancer [2,3]. Understanding the mechanisms behind the multi-organ metastasis will be helpful for the treatment development.
Both angiogenesis and colonization are rate-limiting steps of metastasis [4]. Angiogenesis involves the growth and sprouting of new vascular vessels in both primary and secondary tumors. Vascular endothelial growth factor (VEGF) is a critical proangiogenic growth factor for angiogenesis [5,6]. Colonization is a process that turns micrometastasis to macrometastasis at the secondary sites. Formation of filopodium-like-protrusions (FLPs) is believed to be essential for colonization by inciting the aggressive outgrowth of secondary tumors [7-10]. Recent studies suggest that both of the metastatic steps seem to be involved in multi-organ metastatic progression [11-13].
Krüppel-like factor 8 (KLF8) is a transcription factor well known for promoting many aspects of tumor progression including metastasis [14] by regulating expression of various tumor-related target genes including C-X-C chemokine receptor type 4 (CXCR4) [15-20], a receptor specific for the chemokine known as CXCL12 or the stromal-derived-factor-1 (SDF-1) [21-24]. Aberrant overexpression of CXCR4 in cancer cells tends to metastasize to the organs with high levels of CXCL12 by hijacking the physiological homing of immune cells [21]. Indeed, silencing KLF8 expression in LM2, the lung-prone variant of the human metastatic breast cancer cell line MDA-MB-231 [25,26], inhibits the lung metastasis of the cells that can be rescued by overexpressing CXCR4 in the cells [18]. This rescue efforts involves activation of focal adhesion kinase known to be important for FLP formation during colonization [9,10]. CXCR4 is also known for its role for tumor angiogenesis [24,27,28] and VEGF overexpression and autocrine secretion in metastatic breast cancer cells [27,29].
In this report, we demonstrate that KLF8 can be over-induced by treatment with chemotherapeutic drugs in the LM2 cells and ectopic overexpression of KLF8 or CXCR4 in the cells can trigger extensive metastasis into multiple organs with dramatic enhancement of VEGF secretion, angiogenesis, FLP formation and colonization.
Materials and methods
Antibodies and reagents
The antibodies for HA-probe (F-7), VEGFA, and GFP (B-2) were purchased from Santa Cruz Biotechnology Inc. Antibody for Ki67 (8D5) was purchased from Cell Signaling Technology Inc. Recombinant human CXCL12 (300-28A) was purchased from Peprotech (Rocky Hill, CT, USA). Alexa Fluor® 568 Phalloidin (A12380) was purchased from Thermo Fisher Scientific.
Plasmid construction, cell line generation, cell culture and transfection
The lentiviral vectors pLVPZ, pLVPZ-HA-KLF8 and pLVPZ-HA-CXCR4 were constructed as previously described [16]. The LM2 cells [25,26] harboring a GFP-luciferase fusion reporter were infected with these lentiviruses and selected by puromycin to obtain cell line expressing empty vector (LM2-Vector), KLF8 (LM2-KLF8) or CXCR4 (LM2-CXCR4). Cell culture was described previously [18].
Western blotting and quantitative real-time PCR (qRT-PCR)
Antibodies and cell lines are described above. The Western blotting and qRT-PCR were carried out as previously described [18,20,30-33].
Angiogenic tube formation assay
Tube formation assay was operated according to the manufacturer’s instructions (BD BioCoat™ Angiogenesis Systems, Endothelial cell tube formation). Briefly, the HUVEC cells were labelled with CytoTracker™ (Cell Biolabs, Green fluorescence). Then, the HUVEC cells (3 × 105 cells/ml) were seeded to Matrigel in 100 μl of conditional medium collected from the LM2 cell lines. CXCL12 was added to the medium as designed. The software, WimTube, was used to analyze and quantify the tube number.
In vitro matrigel-on top (MoT) assay
The MoT assay was used to assess FLP formation and colonization potential as described [9]. The LM2-Vector, LM2-KLF8 and LM2-CXCR4 cells were cultured under the 3D MoT culture conditions in which 400 cells were plated above a layer of undiluted Matrigel (Corning #356234) (40 μl) and then covered with Matrigel diluted to 1:50 in culture medium into 8-well chamber slides. Additionally, cells were exposed by PBS or CXCL12. After 48 hour culture, the 3D Matrigel inserts were fixed with 4% paraformaldehyde and stained with Alexa Fluor 568 phalloidin to for 3D imaging of the cell clusters (or colonies) as well as FLP formed by Ultraview confocal microscopy. The number of FLP positive clusters and the number of FLPs formed in each colony were counted. Colonization potential was assessed by the cell proliferation under the 3D MoT culture conditions with the cell number in each colony counted after cultured for 2, 5 and 10 days.
Immunofluorescence (IF) staining
After 48-hour 3D MoT culture, the cell colonies were fixed in 4% paraformaldehyde. Then the cell colonies were processed in permeabilizing solution of 0.1% Triton X-100 in PBS and blocking solution of 1% BSA in PBS. The cell colonies were then incubated with primary antibody (Ki67, CST, #9949S, 1:800) overnight. On the second day, the extra primary antibody was washed off and secondary antibody was added and incubated for one more hour. The slides were mounted with ProLong Gold antifade supplemented with DAPI and a coverslip on top. The fluorescent imaging was carried out with the Ultraview confocal microscope.
Analysis of tumor growth and metastasis in vivo
The 4-6-week-old female nude mice (six per group) were tested for orthotopic tumor growth and metastasis with injection of the cancer cell lines into the mammary fad pad. The injections were performed with 50 μl of cell suspension containing 106 cells into an insulin syringe into the mammary fat pads of female nude mice under anesthesia condition as previously described [16,18,30,32,34,35]. The mice were housed and maintained in specific pathogen-free conditions in the UCF vivarium approved by the American Association for Accreditation of Laboratory Animal Care and in accordance with current regulations and standards of the United States Department of Agriculture, United States Department of Health and Human Services, and the National Institute of Health. The animal experiments were guided by the university-approved IACUC protocol with thorough consideration of humane care of the mice.
Bioluminescent imaging (BLI)
Tumor growth and metastasis were monitored and quantitatively analyzed by BLI using Bruker In-vivo Xtreme II. After tumor cell injection, weekly whole body BLI was performed to monitor the tumor progression for 6-8 weeks as described previously [16,18,30,32,34,35]. Briefly, mice were anaesthetized and injected intraperitoneally with 150 mg/kg of D-luciferin (15 mg/ml in PBS). Three minutes after injection, BLI was performed with a Bruker In-vivo Xtreme II imager. After the last whole body BLI, mice were sacrificed and immediately tissue specific BLI was performed within 5 minutes of D-luciferin injection to visualize the organ-specific metastasis.
Immunohistochemical (IHC) staining
The tissue collection and processes and the staining procedures were previously described [16,18,30,32,34,35]. The antibodies specific for GFP and VEGFA were described above.
Statistical analysis
Mean ± standard deviation is presented with a minimum of three observations per group. Student’s t-test, or one-way ANOVA, unpaired, paired or single sample, with the Bonferroni correction for the multiple comparisons was applied as appropriate. Kaplan-Meier Plots were used for assessing patient survival. The alpha level of 0.05 was used to determine difference with statistical significance.
Results
Chemo-induction of KLF8-CXCR4 signaling may be responsible for drug-resistant metastasis and poor survival
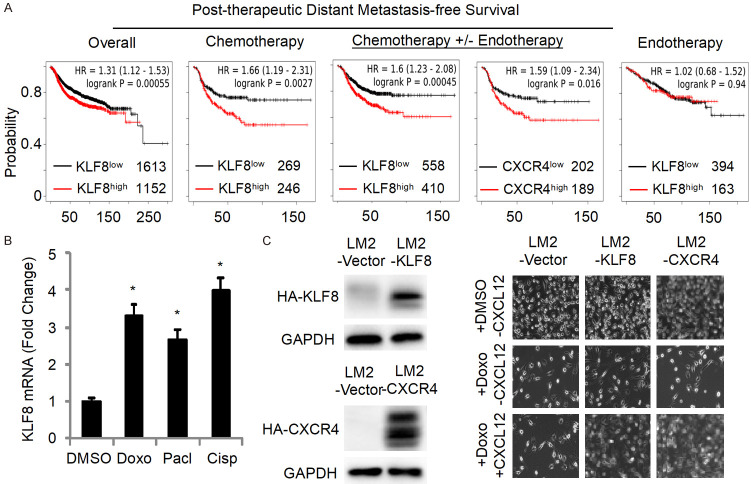
We have previously demonstrated that silencing KLF8 expression in the human breast cancer cell line LM2 inhibits its lung metastasis and this inhibition can be reversed by overexpressing CXCR4 in the cell [18]. To gain further insights into the impact of the KLF8-CXCR4 signaling in breast cancer metastasis, we first investigated potential correlation between levels of KLF8 and CXCR4 with metastatic rate using the public database containing gene expression profiles in tumors forever growing number of breast cancer patients [36]. We performed Kaplan-Meier survival analysis of breast cancer patients of different cohorts and compared the metastasis-free survival rate between patients with tumors expressing high levels of KLF8/CXCR4 and patients with tumors expressing low levels of KLF8/CXCR4. Typically, the tumor mRNA samples are prepared from the surgically removed tumor tissues after pre-operational adjuvant chemotherapy. The analysis results indicate that the patients with the aberrantly high levels of KLF8 or CXCR4 show much worse rate of death from metastasis compared with patients with lower levels of these two genes (Figure 1A) after the patients have received systemic chemotherapy alone or in combination with hormonal therapy after surgical removal of the breast tumors. However, patients receiving hormonal therapy alone do not show a difference regardless of KLF8 expression levels. This suggests that during the post-operational chemotherapy, levels of KLF8 could be further upregulated or over-induced leading to subsequent overexpression of its downstream target CXCR4 and consequently drug resistance associated poor survival. This result also suggests that the patients, like patients with ER-negative tumors, did not respond to hormonal therapy and hormonal therapy does not over-induce KLF8 expression.
Figure 1.
Chemotherapeutic Induction of KLF8-CXCR4 overexpression is related to therapeutic resistant metastasis. A. Aberrant high levels of KLF8 and CXCR4 are correlated with drug resistance and metastasis associated with poor survival. Kaplan-Meier Plots, illustrating the correlation, are based on the public database of cancer patient samples (Oncomine). B. Chemotherapeutic drugs induce an increase in KLF8 expression in LM2 cells. LM2 cells in culture were treated with the chemotherapeutic drugs doxorubicin (Doxo; 0.5 µM), paclitaxol (Pacl; 2.5 µM), cisplatin (Cisp; 3 µM), or DMSO vehicle for 5 days followed by quantitative real-time PCR analysis of KLF8 expression in the cells. C. Overexpression of KLF8 grants drug resistance via CXCR4. LM2 cells overexpressing HA-KLF8 (LM2-KLF8), HA-CXCR4 (LM2-CXCR4) or empty vector (LM2-Vector) were generated. Expression were confirmed by anti-HA blotting. Sensitivity of the cells to doxorubicin treatment were examined in the presence or absence of CXCL12 in the medium.
To test the influence of chemotherapy on KLF8 expression, we treated the LM2 cells, which are ER-negative, with various chemotherapeutic drugs and examined changes in KLF8 message expression. Indeed, KLF8 levels is over-induced in the LM2 cells when treated with these drugs (Figure 1B). To mimic the over-induced KLF8/CXCR4 expression, ectopic overexpression of KLF8 or CXCR4 in the LM2 cells was carried out. And it is evident that these KLF8 or CXCR4-overexpressing LM2 cells have gained strong resistance to the chemotherapeutic drug doxorubicin that depends upon CXCR4 engagement by its ligand CXCL12 interaction (Figure 1C).
KLF8 overexpression triggers metastatic expansion to multiple organs via CXCR4
The LM2 cells are well known to be a lung-prone variant that was established from the human metastatic breast cancer cell line MDA-MB-231 [25,26]. The cells hardly metastasize to tissues other than the lungs even given directly to the circulation via tail vein injection. To determine if overexpressing KLF8 or CXCR4 in the LM2 cells can enhance metastatic potential of the cells, we implanted the LM2-KLF8, LM2-CXCR4 or LM2-Vector cells orthotopically into the mammary fat pad of immunocompromised nude mice. After seven weeks, metastasis of the cells into multiple tissues were examined by tissue-specific bioluminescence imaging (BLI). Strikingly, while the LM2-Vector cells’ metastasis remained restricted in the lungs, both the LM2-KLF8 and LM2-CXCR4 cells metastasized way beyond the lungs (Figure 2A) with particularly high intensities (Figure 2B) and rates (Figure 2C) to the CXCL12-rich tissues such as the liver and bone marrow compared to CXCL12-free tissues such as the kidneys and spleen. These results indicate that the LM2 cells with KLF8 or CXCR4 overexpression gained more aggressive metastatic potential to spread more broadly in addition to the lungs and suggest that this extensive metastatic pattern could be what happens to the patients when KLF8-CXCR4 signaling is over-induced after receiving post-operational systemic chemotherapy.
Figure 2.
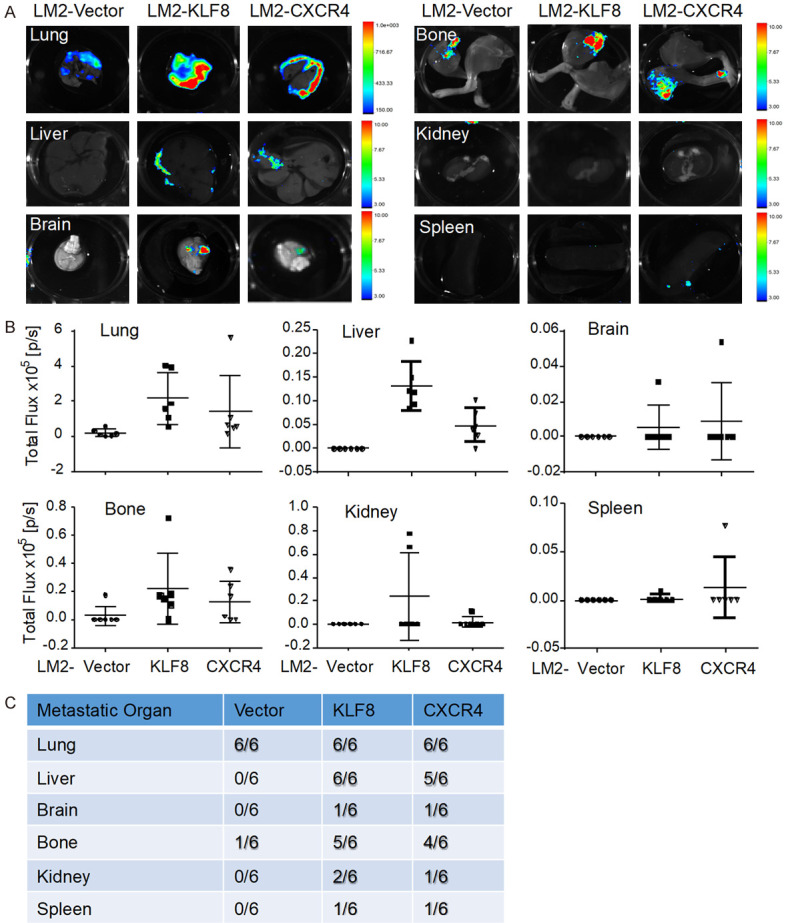
Overexpression of KLF8 drives massive metastasis to multiple organs through CXCR4 signaling. (A) The results of tissue specific BLI imaging 7 weeks after orthotopic injection of the LM-KLF8, LM2-CXCR4 or LM2-Vector cells showing multiple organic metastasis, especially to CXCL12 rich organs. (B) Quantitative analysis of the intensity of tissue specific BLI is shown in (A). (C) Differential metastatic rates to different organs. Animals injected with KLF8 and CXCR4 overexpression cells exhibit bioluminescent evidence of metastasis to more secondary organs compared to the control group. The BLI imaging was carried out as described in the Materials and methods.
KLF8 overexpression enhances endothelial cell proliferation, tube formation and VEGF expression via CXCR4
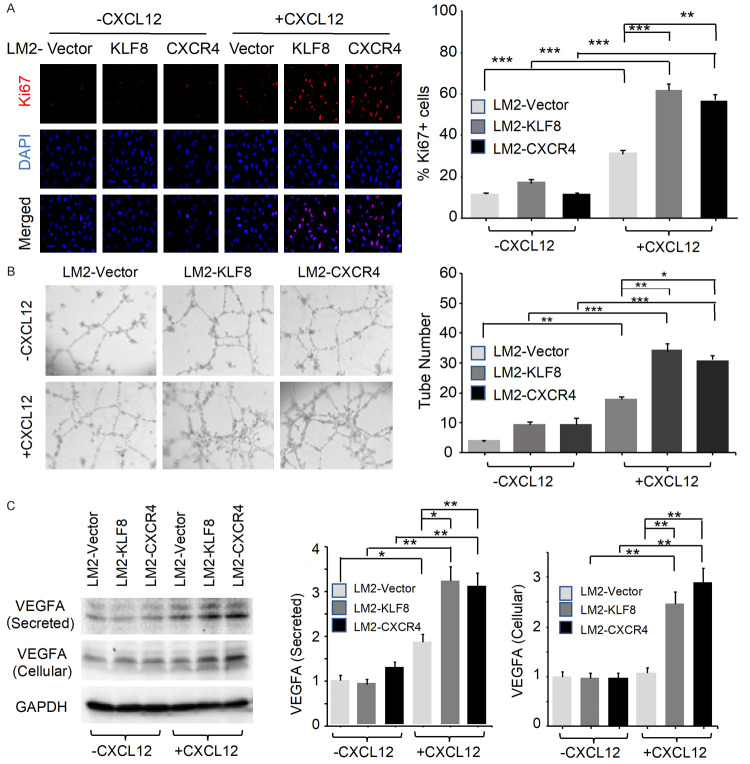
To determine the mechanisms underlying the extensive multi-organ metastasis of the LM2-KLF8 and LM2-CXCR4 cells, we first examined their effects on angiogenesis in vitro. Conditional medium collected from the LM2-KLF8 or LM2-CXCR4 (but not that from LM2-Vector) culture dramatically enhances the HUVEC endothelial cell proliferation as indicated by the expression of the Ki67 proliferation maker, and this enhancement relies on the presence of the ligand CXCL12 (Figure 3A). Next, we performed in vitro tube formation assay which measures the ability of endothelial cells to form capillary-like structures. In the presence of CXCL12, the HUVEC endothelial cells formed significantly more tubes cultured with the condition medium derived from the LM2-KLF8 or LM2-CXCR4 cells, but not the LM2-Vector control cells (Figure 3B). We then looked into the expression of the key angiogenic factor VEGF both inside the cells and in their conditional medium by Western blotting. We found that in the presence of CXCL12, both the LM2-KLF8 and LM2-CXCR4 cells express as well as secret more VEGF than the LM2-Vector cells do (Figure 3C). These results suggest that one of the mechanisms behind the multi-organ metastasis could be enhanced angiogenesis associated with the increase in VEGF expression and secretion caused by the overexpressed KLF8-CXCR4 signaling in the cancer cells.
Figure 3.
KLF8 overexpression enhances angiogenesis in vitro. A. KLF8 overexpression promotes endothelial cell proliferation through CXCR4. HUVEC cells treated by condition medium from LM2-vector, LM2-KLF8, LM2-CXCR4 with or without supplement of CXCL12. Two days after the treatment, Ki67 immunofluorescence staining (Red) was used to test endothelial cell proliferation. B. KLF8 overexpression enhances endothelial tube formation via CXCR4. Representative images of HUVEC tubes (CytoTracker™-labeled) under culture with conditional medium collected from LM2-Vector, LM2-KLF8 and LM2-CXCR4 cells in the presence or absence of CXCL12. C. KLF8 overexpression upregulates VEGF expression and secretion via CXCR4. VEGFA protein levels in cancer cell supernatant and whole cell lysis were examined by Western blotting and quantified with Bio-Rad Imager normalized to GAPDH value. *P<0.05; **P<0.01; ***P<0.001.
KLF8 overexpression enhances metastatic outgrowth through CXCR4-CXCL12 pathway
Next, we examined the colonization capability of the LM2-KLF8 and LM2-CXCR4 cells using the in vitro 3D Matrigel-on-top (MoT) assay that was developed and used for assessing FLP formation as described [9] (Figure 4A). In the presence of CXCL12 in the culture, the LM2-KLF8 and LM2-CXCR4 cells, but not the LM2-Vector cells, formed significantly more FLPs observable as early as a period of two days (Figures 4B and 2C).
Figure 4.
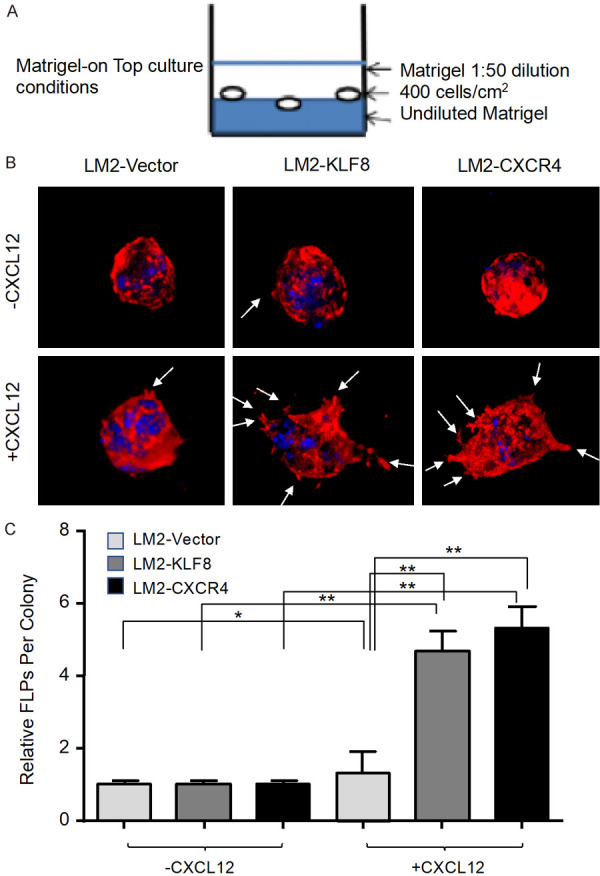
KLF8 overexpression triggers FLP formation through CXCR4-CXCL12 pathway. A. Illustration of the Matrigel On Top (MoT) culture. B, C. The LM2-Vector, LM2-KLF8 and LM2-CXCR4 cells were grown in the MoT culture in the absence or presence of CXCL12 for 48 hours before co-stained with phalloidin (red) and DAPI (blue). The white arrows point to FLPs that were counted in five colonies per treatment group. *P<0.05. **P<0.01.
The MoT assay can also be used to assess potential of colonization, namely, outgrowth of micrometastatic clusters into macrometastatic tumor masses [9]. Thus, we performed the MoT assay with long-term culture up to ten days and examined the proliferation rate of the LM2 cell lines (Figure 5). Clearly, the LM2-KLF8 and LM2-CXCR4 colonies grew much faster than the LM2-Vector colonies leading to significantly more cells per colony (Figure 5A and 5B). The proliferation status of the cells was verified by the positivity of the proliferation marker protein Ki67 in the LM2-KLF8 and LM2-CXCR4 colonies but not the LM2-Vector colonies (Figure 5C).
Figure 5.
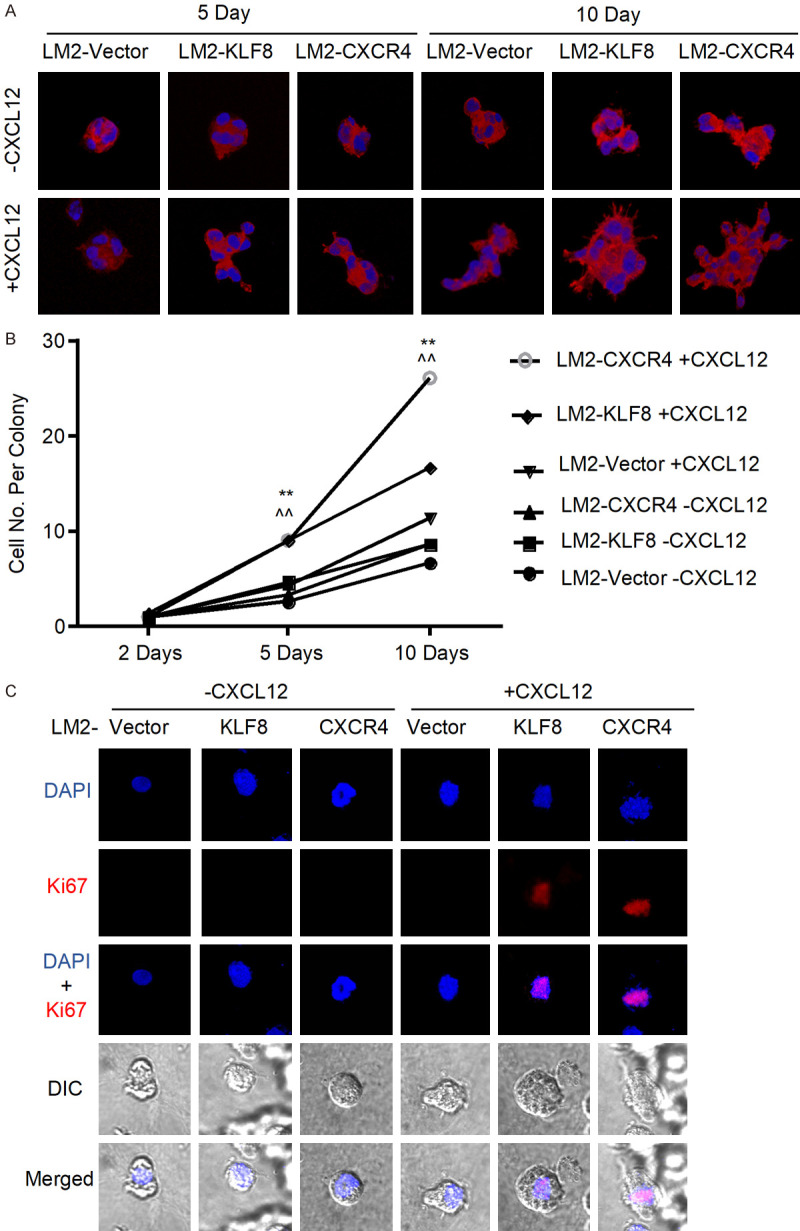
KLF8 overexpression promotes metastatic proliferation through CXCR4-CXCL12 pathway. A and B. Metastatic proliferation rate is measured by counting cell number in each colony over a 10-day period of MoT culture in the absence or presence of CXCL12. Indicated cells were stained as described in Figure 4. Cell number per colony was counted in five colonies for each treatment group at the three different time points shown. **P<0.0001 compared to the -CXCL12 group; ^^P<0.0001 compared to LM2-Vector +CXCL12 group. C. The MoT cells on day 2 were subject to co-immunofluorescent staining with an antibody for the proliferation marker Ki67 (red) and DAPI (blue) followed by fluorescent as well as differential interference contrast (DIC) microscopies.
Notably, the enhanced FLP formation and cell proliferation both rely on the presence of the ligand CXCL12. These MoT assay results indicate that overexpression of KLF8 promotes FLP formation as well as growth rate of the cancer cells in a CXCR4 dependent manner. These results also suggest that enhanced colonization potential caused by elevated KLF8-CXCR4 signaling could be another important mechanism in addition to angiogenesis behind the widespread metastasis of breast cancer in the chemotherapy-resistant patients. To assess this possibility, we quantitatively analyzed the macrometastases in the lungs formed by the LM2 cell lines implanted directly into the mammary fat pad (Figure 6). Indeed, the LM2-KLF8 and LM2-CXCR4 cells formed dramatically more metastatic nodules (Figure 6A) and expressed higher levels of VEGF (Figure 6B) than the LM2-Vector cells did.
Figure 6.
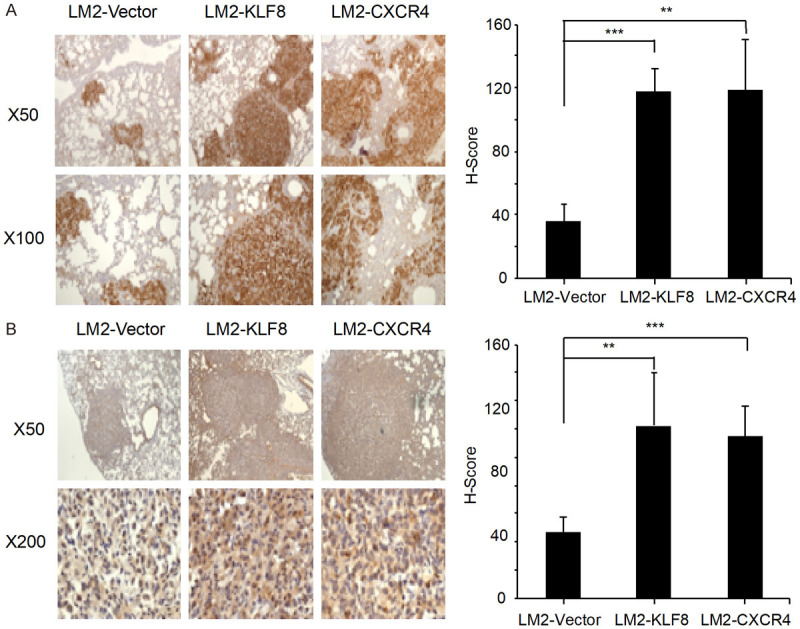
KLF8 overexpression enhances colonization and angiogenesis in the lung via CXCR4. A. The LM2-Vector, LM2-KLF8 or LM2-CXCR4 cells were injected into the mammary fat pad. The lung metastases were visualized immunohistochemical staining of GFP reporter expressed in the tumor cells. B. VEGFA expression in the lung tissues were examined by immunohistochemical staining. The percentage contributions of high positive, positive, low positive, and negative, respectively were analyzed with ImageJ plus IHC Profiler plugin in each of 5 images per group. The histoscore (H-score) was then calculated and graphed for quantitative comparison for statistical significance using One-way ANOVA. **P<0.01; ***P<0.001.
Discussion
In this study, we identified a novel role of KLF8 for drug-resistant multi-organ metastasis. This function of KLF8 is achieved through enhancing at least the tumor angiogenesis and colonization processes of metastasis after its expression is over-elevated by chemotherapeutic treatment. The downstream signaling target CXCR4 plays a major role in mediating this function of KLF8 as it relies on the presence of CXCL12 ligand. And the major mediating role of CXCR4 is also reflected by the fact that the systemic metastasis affects primarily the organs or tissues that express high levels of CXCL12 exactly as does the LM2 cell overexpressing CXCR4. This is also supported by our previous report showing that KLF8 activates CXCR4 transcription in LM2 cells and KLF8 silencing inhibits LM2 lung metastasis [18] and FLP formation [submitted elsewhere] which can be rescued by re-expression of CXCR4.
The LM2 variant is known to metastasize the lungs only even given directly to the blood circulation [25,26] and the higher-than-normal expression of KLF8 and CXCR4 in the cell plays a critical role in the lung metastasis [18]. We demonstrate here that expression of KLF8 in the LM2 cell can be further upregulated by treatment with chemotherapeutic drugs, and that overexpressing KLF8 in LM2 cell mimicking this over-induction of KLF8 expression by chemotherapeutic drugs drove its metastasis extensive to multiple organs and tissues that is way beyond the lungs. This suggests that systemic chemotherapy after adjuvant chemotherapy followed by surgical removal of breast cancer tumors from the patients could cause further upregulation of KLF8 expression in the residual cancer cells, resulting in extensive and systemic metastasis of the drug-resistant cancer cells. ER status of breast cancer does not appear to play a role in the over-induction of KLF8 upregulation and subsequent drug resistant metastasis in the breast cancer patients given that our analysis of the patient cohorts did not indicate a significant impact of hormonal therapy on the patient survival.
We previously demonstrated that the KLF8-CXCR4 signaling plays a critical role in transendothelial migration [18] suggesting that intravasation and extravasation in addition to angiogenesis and colonization may also be enhanced by KLF8-CXCR4 over-induction by chemotherapeutic treatment. These results together make it exciting to think about targeting CXCR4 to prevent drug resistance and multi-organic metastasis of breast cancer overexpressing KLF8.
However, unlike KLF8 that is rarely detectable in normal tissues, CXCR4 is well expressed in the hematopoietic stem cells (HSCs) and many types of immune cells such as monocytes, macrophages, dendritic cells, and B- and T-lymphocytes [21,37-40]. This helps the normal CXCR4+ cells to home to their functional tissues that are rich in the ligand CXCL12, such as the lungs, bone marrow, liver and brain [21,37,41-45]. Clinically, systemic delivery of the CXCR4 antagonist drug AMD3100 to disrupt the interaction between CXCR4 and CXCL12 was approved by the FDA to mobilize HSCs from bone marrow away to the plasma [46,47] for treating hematopoietic malignancies. The successful mobilization and enrichment of HSCs in the blood proves that the CXCR4-CXCL12 interaction is essential for retaining the CXCR4+ cells in their physiological “homes”, i.e., the CXCL12-rich tissues [21,37,41-45].
Indeed, recent studies have drawn increasing attention to the aberrant high levels of CXCR4 expressed in metastatic cancer cells that spread preferentially to the same CXCL12-rich tissues [21,37,41-45,48], suggesting that such CXCR4-high cancer cells take advantage of the ligand-guided immunocyte homing and retaining mechanism to establish metastasis [21,37,41-45]. Inhibition of CXCR4-CXCL12 interaction has led to reduction in experimental breast cancer metastasis in immunocompromised mice [49-51]. Despite these results in the mice and the aberrant high levels of CXCR4 found in patient tumors [42,43,52,53], however, there has been no report of a successful clinical trial of AMD3100 or other antagonists for any CXCR4 overexpressing solid tumor type.
Notably, the LM2-KLF8 cells also metastasize to other organs such as kidney and spleen in addition to the CXCL12-rich organs or tissues. Moreover, proteins other than CXCR4 also play an important role in mediating KLF8 function in promoting angiogenesis [submitted elsewhere]. These results suggest that multiple downstream factors likely work together to contribute to KLF8 function in drug resistant metastasis.
Taken together, our study reported here suggests that KLF8 may serve as a potentially better molecular target for intervening in the drug resistant massive metastasis of breast cancer where KLF8-CXCR4 signaling axis is aberrantly over-induced by chemotherapy.
Acknowledgements
This work was supported by an NIH grant (CA132977), a Florida Breast Cancer Foundation grant, the LIFE@UCF Richard Tucker Grant and an institutional grant to J.Z.
Disclosure of conflict of interest
None.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020;70:7–30. doi: 10.3322/caac.21590. [DOI] [PubMed] [Google Scholar]
- 2.Gonzalez-Angulo AM, Morales-Vasquez F, Hortobagyi GN. Overview of resistance to systemic therapy in patients with breast cancer. Adv Exp Med Biol. 2007;608:1–22. doi: 10.1007/978-0-387-74039-3_1. [DOI] [PubMed] [Google Scholar]
- 3.Jones SE. Metastatic breast cancer: the treatment challenge. Clin Breast Cancer. 2008;8:224–233. doi: 10.3816/CBC.2008.n.025. [DOI] [PubMed] [Google Scholar]
- 4.Guan X. Cancer metastases: challenges and opportunities. Acta Pharm Sin B. 2015;5:402–418. doi: 10.1016/j.apsb.2015.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jackson JK, Gleave ME, Gleave J, Burt HM. The inhibition of angiogenesis by antisense oligonucleotides to clusterin. Angiogenesis. 2005;8:229–238. doi: 10.1007/s10456-005-9018-5. [DOI] [PubMed] [Google Scholar]
- 6.Salvucci O, Yao L, Villalba S, Sajewicz A, Pittaluga S, Tosato G. Regulation of endothelial cell branching morphogenesis by endogenous chemokine stromal-derived factor-1. Blood. 2002;99:2703–2711. doi: 10.1182/blood.v99.8.2703. [DOI] [PubMed] [Google Scholar]
- 7.Barkan D, Kleinman H, Simmons JL, Asmussen H, Kamaraju AK, Hoenorhoff MJ, Liu ZY, Costes SV, Cho EH, Lockett S, Khanna C, Chambers AF, Green JE. Inhibition of metastatic outgrowth from single dormant tumor cells by targeting the cytoskeleton. Cancer Res. 2008;68:6241–6250. doi: 10.1158/0008-5472.CAN-07-6849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fidler IJ. The pathogenesis of cancer metastasis: the ‘seed and soil’ hypothesis revisited. Nat Rev Cancer. 2003;3:453–458. doi: 10.1038/nrc1098. [DOI] [PubMed] [Google Scholar]
- 9.Shibue T, Brooks MW, Inan MF, Reinhardt F, Weinberg RA. The outgrowth of micrometastases is enabled by the formation of filopodium-like protrusions. Cancer Discov. 2012;2:706–721. doi: 10.1158/2159-8290.CD-11-0239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shibue T, Brooks MW, Weinberg RA. An integrin-linked machinery of cytoskeletal regulation that enables experimental tumor initiation and metastatic colonization. Cancer Cell. 2013;24:481–498. doi: 10.1016/j.ccr.2013.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Seiffert K, Schmalfeldt B, Muller V. Current status of targeted treatment in breast cancer. Dtsch Med Wochenschr. 2017;142:1669–1675. doi: 10.1055/s-0043-108468. [DOI] [PubMed] [Google Scholar]
- 12.Sun C, Li J, Wang B, Shangguan J, Figini M, Shang N, Pan L, Zhang Z. Tumor angiogenesis and bone metastasis - correlation in invasive breast carcinoma. J Immunol Methods. 2018;452:46–52. doi: 10.1016/j.jim.2017.10.006. [DOI] [PubMed] [Google Scholar]
- 13.Das DK, Naidoo MK, Ilboudo A, DuBois P, Durojaiye V, Liu C, Ogunwobi OO. Isolation and propagation of circulating tumor cells from a mouse cancer model. J Vis Exp. 2015;9:52861. doi: 10.3791/52861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lahiri SK, Zhao J. Kruppel-like factor 8 emerges as an important regulator of cancer. Am J Transl Res. 2012;4:357–363. [PMC free article] [PubMed] [Google Scholar]
- 15.Lu H, Hu L, Li T, Lahiri S, Shen C, Wason MS, Mukherjee D, Xie H, Yu L, Zhao J. A novel role of Kruppel-like factor 8 in DNA repair in breast cancer cells. J Biol Chem. 2012;287:43720–43729. doi: 10.1074/jbc.M112.418053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lu H, Hu L, Yu L, Wang X, Urvalek AM, Li T, Shen C, Mukherjee D, Lahiri SK, Wason MS, Zhao J. KLF8 and FAK cooperatively enrich the active MMP14 on the cell surface required for the metastatic progression of breast cancer. Oncogene. 2014;33:2909–2917. doi: 10.1038/onc.2013.247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lu H, Wang X, Li T, Urvalek AM, Yu L, Li J, Zhu J, Lin Q, Peng X, Zhao J. Identification of poly (ADP-ribose) polymerase-1 (PARP-1) as a novel Kruppel-like factor 8-interacting and -regulating protein. J Biol Chem. 2011;286:20335–20344. doi: 10.1074/jbc.M110.215632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mukherjee D, Lu H, Yu L, He C, Lahiri SK, Li T, Zhao J. Kruppel-like factor 8 activates the transcription of C-X-C cytokine receptor type 4 to promote breast cancer cell invasion, transendothelial migration and metastasis. Oncotarget. 2016;7:23552–23568. doi: 10.18632/oncotarget.8083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang X, Lu H, Li T, Yu L, Liu G, Peng X, Zhao J. Kruppel-like factor 8 promotes tumorigenic mammary stem cell induction by targeting miR-146a. Am J Cancer Res. 2013;3:356–373. [PMC free article] [PubMed] [Google Scholar]
- 20.Wang X, Zheng M, Liu G, Xia W, McKeown-Longo PJ, Hung MC, Zhao J. Kruppel-like factor 8 induces epithelial to mesenchymal transition and epithelial cell invasion. Cancer Res. 2007;67:7184–7193. doi: 10.1158/0008-5472.CAN-06-4729. [DOI] [PubMed] [Google Scholar]
- 21.Mukherjee D, Zhao J. The role of chemokine receptor CXCR4 in breast cancer metastasis. Am J Cancer Res. 2013;3:46–57. [PMC free article] [PubMed] [Google Scholar]
- 22.Donzella GA, Schols D, Lin SW, Este JA, Nagashima KA, Maddon PJ, Allaway GP, Sakmar TP, Henson G, De Clercq E, Moore JP. AMD3100, a small molecule inhibitor of HIV-1 entry via the CXCR4 co-receptor. Nat Med. 1998;4:72–77. doi: 10.1038/nm0198-072. [DOI] [PubMed] [Google Scholar]
- 23.Walenkamp AME, Lapa C, Herrmann K, Wester HJ. CXCR4 ligands: the next big hit? J Nucl Med. 2017;58(Suppl 2):77s–82s. doi: 10.2967/jnumed.116.186874. [DOI] [PubMed] [Google Scholar]
- 24.Vandercappellen J, Van Damme J, Struyf S. The role of CXC chemokines and their receptors in cancer. Cancer Lett. 2008;267:226–244. doi: 10.1016/j.canlet.2008.04.050. [DOI] [PubMed] [Google Scholar]
- 25.Minn AJ, Gupta GP, Siegel PM, Bos PD, Shu W, Giri DD, Viale A, Olshen AB, Gerald WL, Massague J. Genes that mediate breast cancer metastasis to lung. Nature. 2005;436:518–524. doi: 10.1038/nature03799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Minn AJ, Kang Y, Serganova I, Gupta GP, Giri DD, Doubrovin M, Ponomarev V, Gerald WL, Blasberg R, Massague J. Distinct organ-specific metastatic potential of individual breast cancer cells and primary tumors. J Clin Invest. 2005;115:44–55. doi: 10.1172/JCI22320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Guleng B, Tateishi K, Ohta M, Kanai F, Jazag A, Ijichi H, Tanaka Y, Washida M, Morikane K, Fukushima Y, Yamori T, Tsuruo T, Kawabe T, Miyagishi M, Taira K, Sata M, Omata M. Blockade of the stromal cell-derived factor-1/CXCR4 axis attenuates in vivo tumor growth by inhibiting angiogenesis in a vascular endothelial growth factor-independent manner. Cancer Res. 2005;65:5864–5871. doi: 10.1158/0008-5472.CAN-04-3833. [DOI] [PubMed] [Google Scholar]
- 28.Orimo A, Gupta PB, Sgroi DC, Arenzana-Seisdedos F, Delaunay T, Naeem R, Carey VJ, Richardson AL, Weinberg RA. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell. 2005;121:335–348. doi: 10.1016/j.cell.2005.02.034. [DOI] [PubMed] [Google Scholar]
- 29.Liang Z, Brooks J, Willard M, Liang K, Yoon Y, Kang S, Shim H. CXCR4/CXCL12 axis promotes VEGF-mediated tumor angiogenesis through Akt signaling pathway. Biochem Biophys Res Commun. 2007;359:716–722. doi: 10.1016/j.bbrc.2007.05.182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Li T, Lu H, Mukherjee D, Lahiri SK, Shen C, Yu L, Zhao J. Identification of epidermal growth factor receptor and its inhibitory microRNA141 as novel targets of Kruppel-like factor 8 in breast cancer. Oncotarget. 2015;6:21428–42. doi: 10.18632/oncotarget.4077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lu H, Wang X, Urvalek AM, Li T, Xie H, Yu L, Zhao J. Transformation of human ovarian surface epithelial cells by Kruppel-like factor 8. Oncogene. 2014;33:10–18. doi: 10.1038/onc.2012.545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang X, Lu H, Urvalek AM, Li T, Yu L, Lamar J, DiPersio CM, Feustel PJ, Zhao J. KLF8 promotes human breast cancer cell invasion and metastasis by transcriptional activation of MMP9. Oncogene. 2011;30:1901–1911. doi: 10.1038/onc.2010.563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang X, Zhao J. KLF8 transcription factor participates in oncogenic transformation. Oncogene. 2007;26:456–461. doi: 10.1038/sj.onc.1209796. [DOI] [PubMed] [Google Scholar]
- 34.Lahiri SK, Lu H, Mukherjee D, Yu L, Zhao J. ERK2 phosphorylates Kruppel-like factor 8 protein at serine 48 to maintain its stability. Am J Cancer Res. 2016;6:910–923. [PMC free article] [PubMed] [Google Scholar]
- 35.Li T, Lu H, Shen C, Lahiri SK, Wason MS, Mukherjee D, Yu L, Zhao J. Identification of epithelial stromal interaction 1 as a novel effector downstream of Kruppel-like factor 8 in breast cancer invasion and metastasis. Oncogene. 2014;33:4746–4755. doi: 10.1038/onc.2013.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gyorffy B, Lanczky A, Eklund AC, Denkert C, Budczies J, Li Q, Szallasi Z. An online survival analysis tool to rapidly assess the effect of 22,277 genes on breast cancer prognosis using microarray data of 1,809 patients. Breast Cancer Res Treat. 2010;123:725–731. doi: 10.1007/s10549-009-0674-9. [DOI] [PubMed] [Google Scholar]
- 37.Cojoc M, Peitzsch C, Trautmann F, Polishchuk L, Telegeev GD, Dubrovska A. Emerging targets in cancer management: role of the CXCL12/CXCR4 axis. Onco Targets Ther. 2013;6:1347–1361. doi: 10.2147/OTT.S36109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Caulfield J, Fernandez M, Snetkov V, Lee T, Hawrylowicz C. CXCR4 expression on monocytes is up-regulated by dexamethasone and is modulated by autologous CD3+ T cells. Immunology. 2002;105:155–162. doi: 10.1046/j.0019-2805.2001.01359.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Joly M, Pinto JM. CXCR4 and CCR5 regulation and expression patterns on T- and monocyte-macrophage cell lineages: implications for susceptibility to infection by HIV-1. Math Biosci. 2005;195:92–126. doi: 10.1016/j.mbs.2005.01.002. [DOI] [PubMed] [Google Scholar]
- 40.Capoccia BJ, Shepherd RM, Link DC. G-CSF and AMD3100 mobilize monocytes into the blood that stimulate angiogenesis in vivo through a paracrine mechanism. Blood. 2006;108:2438–2445. doi: 10.1182/blood-2006-04-013755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Epstein RJ. The CXCL12-CXCR4 chemotactic pathway as a target of adjuvant breast cancer therapies. Nat Rev Cancer. 2004;4:901–909. doi: 10.1038/nrc1473. [DOI] [PubMed] [Google Scholar]
- 42.Muller A, Homey B, Soto H, Ge N, Catron D, Buchanan ME, McClanahan T, Murphy E, Yuan W, Wagner SN, Barrera JL, Mohar A, Verastegui E, Zlotnik A. Involvement of chemokine receptors in breast cancer metastasis. Nature. 2001;410:50–56. doi: 10.1038/35065016. [DOI] [PubMed] [Google Scholar]
- 43.Zlotnik A, Burkhardt AM, Homey B. Homeostatic chemokine receptors and organ-specific metastasis. Nat Rev Immunol. 2011;11:597–606. doi: 10.1038/nri3049. [DOI] [PubMed] [Google Scholar]
- 44.Chatterjee S, Behnam Azad B, Nimmagadda S. The intricate role of CXCR4 in cancer. Adv Cancer Res. 2014;124:31–82. doi: 10.1016/B978-0-12-411638-2.00002-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Balkwill F. Cancer and the chemokine network. Nat Rev Cancer. 2004;4:540–550. doi: 10.1038/nrc1388. [DOI] [PubMed] [Google Scholar]
- 46.Pusic I, DiPersio JF. Update on clinical experience with AMD3100, an SDF-1/CXCL12-CXCR4 inhibitor, in mobilization of hematopoietic stem and progenitor cells. Curr Opin Hematol. 2010;17:319–326. doi: 10.1097/MOH.0b013e328338b7d5. [DOI] [PubMed] [Google Scholar]
- 47.Calandra G, Bridger G, Fricker S. CXCR4 in clinical hematology. Curr Top Microbiol Immunol. 2010;341:173–191. doi: 10.1007/82_2010_26. [DOI] [PubMed] [Google Scholar]
- 48.Valastyan S, Weinberg RA. Tumor metastasis: molecular insights and evolving paradigms. Cell. 2011;147:275–292. doi: 10.1016/j.cell.2011.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Williams SA, Harata-Lee Y, Comerford I, Anderson RL, Smyth MJ, McColl SR. Multiple functions of CXCL12 in a syngeneic model of breast cancer. Mol Cancer. 2010;9:250. doi: 10.1186/1476-4598-9-250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Liang Z, Yoon Y, Votaw J, Goodman MM, Williams L, Shim H. Silencing of CXCR4 blocks breast cancer metastasis. Cancer Res. 2005;65:967–971. [PMC free article] [PubMed] [Google Scholar]
- 51.Huang EH, Singh B, Cristofanilli M, Gelovani J, Wei C, Vincent L, Cook KR, Lucci A. A CXCR4 antagonist CTCE-9908 inhibits primary tumor growth and metastasis of breast cancer. J Surg Res. 2009;155:231–236. doi: 10.1016/j.jss.2008.06.044. [DOI] [PubMed] [Google Scholar]
- 52.Salvucci O, Bouchard A, Baccarelli A, Deschenes J, Sauter G, Simon R, Bianchi R, Basik M. The role of CXCR4 receptor expression in breast cancer: a large tissue microarray study. Breast Cancer Res Treat. 2006;97:275–283. doi: 10.1007/s10549-005-9121-8. [DOI] [PubMed] [Google Scholar]
- 53.Zhang Z, Ni C, Chen W, Wu P, Wang Z, Yin J, Huang J, Qiu F. Expression of CXCR4 and breast cancer prognosis: a systematic review and meta-analysis. BMC Cancer. 2014;14:49. doi: 10.1186/1471-2407-14-49. [DOI] [PMC free article] [PubMed] [Google Scholar]