Abstract
Non-small cell lung cancer (NSCLC) is the most common type of lung cancer. Chemotherapy-induced adverse effects and resistance of NSCLC to conventional drugs reduce the efficacy of current therapies. Tumors contain a small population of cancer stem cells (CSCs) that play a critical role in tumor initiation, maintenance, and drug resistance that finally lead to cancer recurrence. Therefore, CSC-targeting therapies can offer the best hope for developing curative cancer therapies. Vitamins have a high potential for cancer prevention and treatment. Vitamins also ameliorate the side effects which occur in chemo-radio therapy. Menadione (2-methyl-1,4-naphthoquinone/vitamin-K3) is a synthetic form of vitamin K that indicated antitumor activities. The purpose of this study was to evaluate the anti-CSCs effect of menadione and combination of cisplatin and gemcitabine as a first-line treatment in patients with NSCLC on the NSCLC cell line A549. MTT results displayed decreased cell survival after treatment with cisplatin/gemcitabine for 48 h treatment (IC50 values 0.25 µM for cisplatin and 5 µM for gemcitabine). Menadione also inhibited the cell growth in A549 cells (IC50: 16 µM). Quantitative RT-PCR showed significant downregulation of CSC markers (Oct4, Nanog, Sox2, Aldh1, Abcb1, CD44, and CD133) and Snail, epithelial-mesenchymal transition marker, after treatment with menadione and cisplatin/gemcitabine. Flow cytometry showed CD44-positive cells that constitute a high percentage (70%) of A549 cells reduced significantly after treatment with cisplatin/gemcitabine or menadione. However, A549 cells did not show a significant population positive for CD133 and ABCB1 (less than 0.05%), and these fractions did not change after treatment with two agents.
Key Words: Non-small cell lung cancer, Menadione, Gemcitabine, Cisplatin, Cancer stem cells, Cancer stem cell markers
Introduction
Lung cancer is one of the most commonly occurring cancers and the leading cause of cancer-associated death worldwide (1). Non-small cell lung cancer (NSCLC) is the most common type of lung cancer that is resistant to chemotherapy. Most NSCLC patients respond poorly to conventional chemotherapy (2). Several studies have identified cancer stem cells (CSCs) in NSCLC, responsible for tumor maintenance, therapeutic resistance, and malignant features of this type of lung cancer (3-8). CSCs are a subpopulation of cancer cells within the tumor which are characterized by the ability to self-renew as well as differentiation into multilineage cancer cell types (9). Therefore CSCs express elevated levels of pluripotent stem cell markers, OCT4, NANOG, SOX2 (10, 11). This subpopulation of cancer cells has been found to hold intrinsic resistance to radio-chemotherapy through high aldehyde dehydrogenase (ALDHs) enzyme activity that catalyzes the detoxification of a wide spectrum of drugs (12, 13), enhanced ability for DNA repair (14-17), overexpression of ATP-binding cassette (ABC) transporters which mediate the efflux of multiple chemotherapeutic drugs (18-21) and escaping the effects of the drug via quiescence (22-28). Considering the role of CSCs in cancer therapy failure, therapeutic strategies that target CSCs may bring new hopes to cancer therapy.
Vitamins are compounds required for normal physiological function and growth of the body. Vitamins are reported to have an apoptotic and inhibitory effect against various cancers. Therefore, the role of vitamins in cancer prevention and treatment has emerged in the past few decades. Furthermore, it is reported that vitamins involve in the amelioration of side effects that occur in chemotherapy and radiation therapy (29-31). As a result, the combination of vitamins with traditional chemotherapeutic drugs can potentiate anticancer efficacy and reduce the side-effects of chemotherapy. Vitamin K is a dietary nutrient that is required for blood clotting. Menadione (2-methyl-1,4-naphthoquinone/vitamin-K3), a synthetic form of vitamin K, is used as a component in multivitamin drugs. It is shown that menadione has antitumor activity against colon, cervix, liver, breast, stomach, nasopharynx, lung, leukemia, and lymphoma cancer cell lines (32-37). Moreover, anti-migratory effects of menadione by modulating the expression of epithelial to mesenchymal transition (EMT) markers was demonstrated in human oral cancer cells (38).
EMT is a process by which immotile epithelial cells lose cell-cell adhesion and obtain migratory and invasive properties, which have been shown to occur during metastasis in cancer progression (39, 40). Signaling pathways that are critical for CSCs self-renewal and maintenance, such as Wnt, Hedgehog, and Notch, are also activated during EMT. Therefore, according to some evidence, cells undergoing EMT enter the CSC state, acquire drug resistance phenotype as a consequence of elevated expression of anti-apoptotic proteins and increased levels of ABC transporters and express stem cell markers (26, 41-45). Hence, due to the inhibitory effect of menadione on EMT, we hypothesized that the menadione might also target the CSCs population. Therefore, in the present study, we compared the effect of menadione with gemcitabine plus cisplatin as first-line therapy in NSCLC on CSC population and expression of CSC markers in NSCLC cell lines A549
Experimental
Materials
We prepared the RPMI-1640 medium from Gibco, Grand Island, USA, and fetal bovine serum (FBS) and penicillin-streptomycin from Biowest, France. 3-(4,5-dimethylthiazol-2- yl)-2, 5-diphenyltetrazolium bromide (MTT) was from Atocel, Austria. For cell treatment, cisplatin (Platinol) and gemcitabine (Gemzar) were purchased from Vianex S.A, Greece and Sobhan, Iran, respectively, and menadione (Cat. No. M5625) was from Sigma-Aldrich. FITC anti-human CD44 antibody (Cat. No. 560977; BD Biosciences, San Jose, CA, U.S.A), PE anti- Human CD133/2 (clone: 293C3; Miltenyi Biotec, Bergisch Gladbach, Germany), PE anti-Human CD243 (ABCB1) antibody (Cat. No 919405; Biolegend, San Diego, C.A, U.S.A), Propidium iodide (PI) (Cat. No. P4170; Sigma-Aldrich) and 7-amino-actinomycin D (7-AAD) (Cat. No. 559763; B.D Bioscience) were used for flow cytometry. Total RNA isolation kit was purchased from DENAzist Asia, Mashhad, Iran. RNase-free DNase I (Cat. No. EN0521) and M-MuLV reverse transcriptase (Cat. No. EP0441) was from Thermo Scientific, Wilmington, USA. The real-time PCR was performed using Real QPCR 2x SYBR Green master mix (Cat. No. 5000850-1250; Amplicon, UK). Oligo(dT)18 primer and dNTP were obtained from Yekta Tajhiz Azma, Tehran, Iran.
Cell culture and treatment
Human lung cancer cell line A549 was obtained from the National Cell Bank of Iran (Pasteur Institute of Iran, Tehran). They were cultured in RPMI-1640 medium supplemented with 10% FBS, 100 U/mL penicillin, and 100 mg/mL streptomycin and maintained at 37 °C in humidified air with 5% CO2. For treatment experiments, A549 cells were treated for 7 days by combination of cisplatin (0.25 µM)/gemcitabine (5 µM) and 16 µM menadione separately. RNA isolation, quantitative reverse transcription PCR (qRT-PCR) and flow cytometry analysis was carried out on untreated and treated cells to evaluate the gene expression level.
Measurement of cell viability
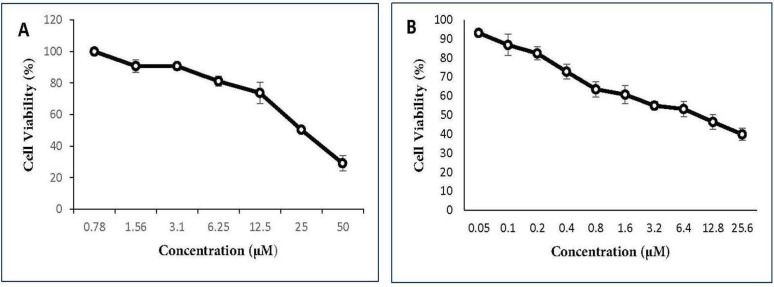
MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide) assay was conducted to evaluate in-vitro cytotoxicity of gemcitabine and cisplatin as single agents and their combinations as well as menadione on the A549 cell line. The cells were seeded in 96-plates at a density of 5 × 103 cells/well and allowed to adhere overnight. Cells were exposed to different concentrations of each drug as shown in Figures 1 and 2. After 48 h incubation, 20 µL MTT stock solution (5 mg/mL) was added into each well and cells were incubated for 3 h at 37 °C. Thereafter, 100 μL dimethyl sulfoxide (DMSO) was added to each well to dissolve the formazan crystals. The absorbance was measured at a wavelength of 540 nm by spectrofluorometry (BioTekELx800, USA). The percentage of cell viability calculated as: [(OD 490 treated cells)/OD 490 control cells] × 100. The IC50 values of agents were calculated using Prism 6.0 (GraphPad Software, Inc., San Diego, California, USA).
Figure 1.
The ani-proliferative effect of cisplatin and gemcitabine on A549 cell line. A549 cell line was cultured and treated with various concentrations of (A) cisplatin (0.78-50 µM) and (B) gemcitabine (0.05-25.6 µM) for 48 h and cell viability percentage was measured. All data are reported as mean ± SD of at least three separate experiments
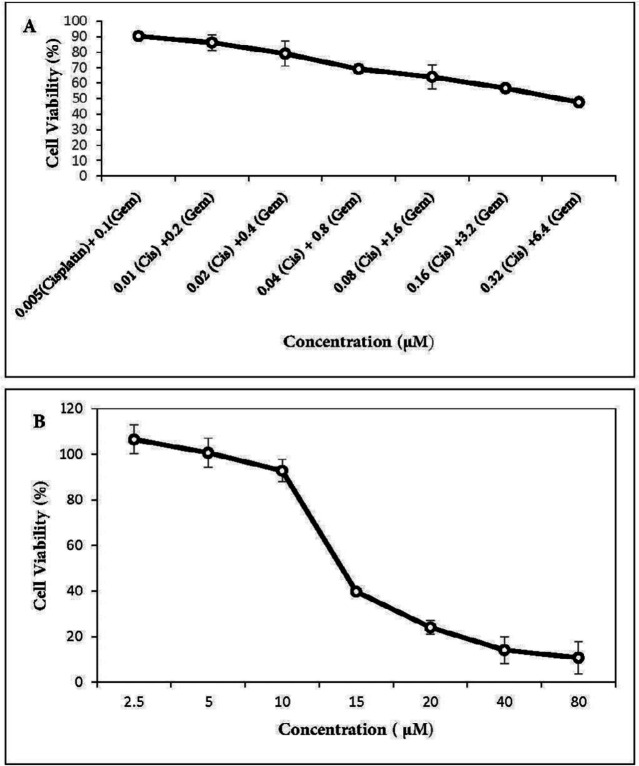
Figure 2.
Effect of cisplatin plus gemcitabine and menadione on the growth of A549 lung cancer cells. A549 cells were treated with increasing concentrations of (A) cisplatin plus gemcitabine as well as (B) menadione for 48 h and cell viability was determined by MTT assay. All the points represent results from three independent experiments performed in triplicate. Data are expressed as mean ± SD
Quantitative reverse-transcription PCR (qRT-PCR)
Total RNA was extracted using an RNA isolation kit according to the manufacturer’s instructions and was reverse-transcribed by using M-MuLV reverse transcriptase as described in the protocol.
Real-time PCR was carried out on Thermocycler (Analytik Jena, Jena, Germany). Each reaction consists of 1X SYBR Green Real-time PCR Master Mix, 1 µL cDNA template, and each primer at 250 nM in a 20 µL reaction volume. Gene-specific primers were designed using Oligo7 Primer Analysis Software. The sequence of primers and product length are described in Table 1. Amplification conditions for Oct4, Nanog, Aldh1a1, Abcb1, CD133, CD44, Gapdh and Snail were: 95 ºC for 15 min, followed by 40 cycles of 95 ºC for 20 sec, 64 ºC for 30 sec, and 72 ºC for 15 sec. The same program was used for Sox2, except that the annealing temperature was 66 ºC. At the end of the PCR runs to derive melting curves, the temperature was increased in steps of 1 ºC for 5 sec from 60 ºC to 95 ºC. Analysis of melting curves clearly indicated that each of the primer pairs amplified a single expected product with a distinct Tm. The accuracy of the amplification reaction was validated by gel electrophoresis. Gapdh was used as an endogenous control to normalize each sample. Relative quantification of mRNA within the samples was examined using the comparative Ct method (ΔCt treated cells – ΔCt control cells = ΔΔC; relative quantity = 2 -ΔΔct).
Flow cytometry
The following protocol was employed to examine the expression of cell surface markers CD44, CD133, and ABCB1 by flow cytometry. Cells were dissociated with trypsin-EDTA and after washing, one million cells were suspended in PBS/2% FBS. The antibodies were added to cells at a 1:5 dilution for FITC-CD44 antibody and 1:10 dilution for PE-CD133 and PE-ABCB1 antibodies for 30 min in the dark. Finally, after washing, cells were suspended in 0.5 mL of PBS/2% FBS and then analyzed by flow cytometry. To detect dead cells, PI was used with FITC-CD44 antibody and 7-AAD was used with PE-CD133 and PE-ABCB1 antibodies. Flow cytometry was done using a FACSCalibur (BD Biosciences, San Jose, CA, USA). Data were analyzed by Flow Jo software, which is provided with the system.
Statistical analysis
All data were expressed as Mean±SD and analyzed using SPSS 16.0 software (SPSS Inc, Chicago, IL, USA). Data analysis was performed using Student’s t-test, and P < 0.05 was considered to indicate a statistically significant difference. All the experiments were repeated for 3 times.
Results
Dose-dependent antitumor activity of chemotherapy drugs and menadione against NSCLC cell lines A549
The cytotoxic activities of gemcitabine and cisplatin were first determined individually on A549 cells. As expected, there was a dose-dependent cell-killing effect. IC50 values were 22.5 µM for cisplatin (Figure 1A) and 9 µM for gemcitabine (Figure 1B). Co-treatment with two drugs in gemcitabine/cisplatin ratio of 20:1 potentiated antitumor activity. Analysis of cell survival by MTT assay showed that IC50 values after 48 h treatment with two drugs were 0.25 µM for cisplatin and 5 µM for gemcitabine (Figure 2A). Menadione also inhibited the growth of the A549 cells with IC50: 16 µM (Figure 2B).
Downregulation of CSC-related genes following cisplatin/gemcitabine and menadione treatment in A549 cells
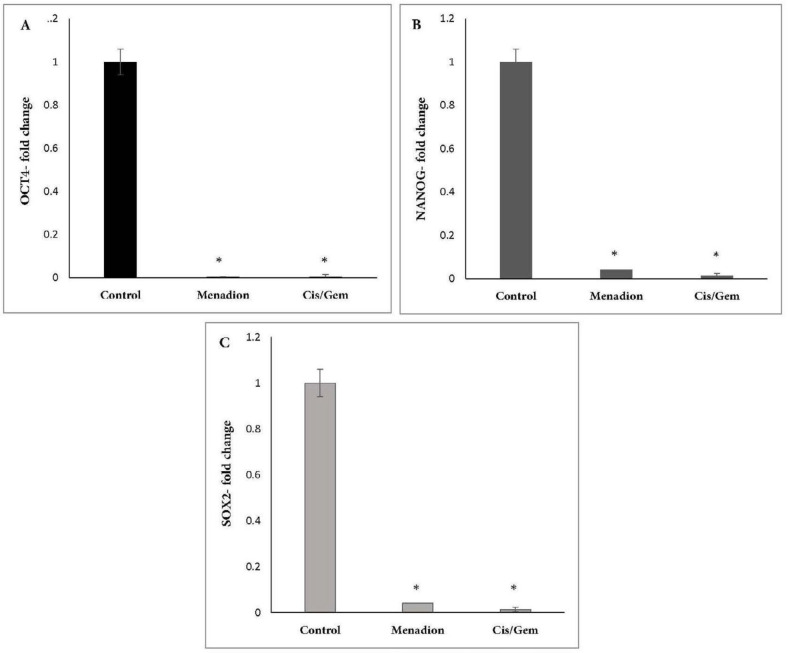
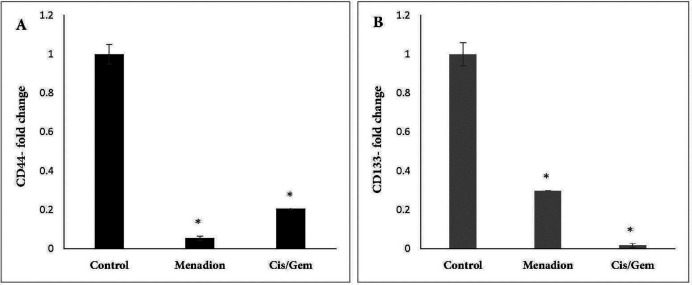
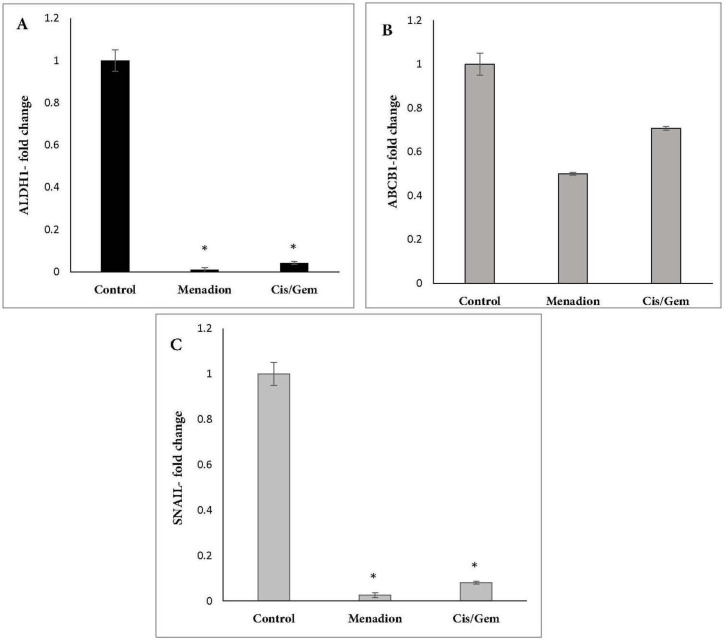
Gene expression analysis of A549 cancer cells treated with cisplatin/gemcitabine and menadione revealed several CSC-related genes and transcription factors that were significantly downregulated after treatment. In detail, stemness-related genes such as Oct4, Nanog, and Sox2 that control the pluripotency and self-renewal of stem cells showed more than 95% decrease after treatment by cisplatin/gemcitabine or menadione (Figures 3A-3C). Moreover, our data revealed a significant downregulation of CD44 and CD133 as well-known CSCs surface markers in response to treatment with both agents (Figure 4). The RNA expression level of Aldh1a1 as detoxifying enzyme after treatment with cisplatin/gemcitabine and menadione showed 91% and 97% reduction, whereas treatment with both components did not show significant alteration on the expression of drug efflux transporter Abcb1 (Figures 5A and 5B). Snail is a family of transcription factors that promote the repression of the adhesion molecule E-cadherin to regulate EMT. We demonstrated that the treatment with chemotherapy drugs cisplatin/gemcitabine and menadione decreased the expression of Snails by more than 90% (Figure 5C) (46, 47).
Figure 3.
Downregulation of pluripotency transcription factors Oct4, Nanog and Sox2 after A549 treatment with cisplatin/gemcitabine and menadione. A549 cells were treated for 7 days by a combination of cisplatin (0.25 µM)/gemcitabine (5 µM) and 16 µM menadione separately and expression of (A) Oct4, (B) Nanog and (C) Sox2 were analyzed. Y-axis represents the fold-change in transcript levels compared with untreated A549 cells (designated as 1.0). The graph represents the mean data ± SD of at least three independent experiments. The asterisk indicates a significant (p < 0.05) difference in mRNA expression in comparison with untreated cells
Figure 4.
Effect of cisplatin/gemcitabine and menadione treatment on the mRNA expression level of CSC surface markers CD44 and CD133 in A549 cell line. mRNA expression levels of (A) CD44 and (B) CD133 in untreated control 549 cells were compared to cells treated by cisplatin/gemcitabine (0.25 µM/5 µM) and menadione (16 µM) for 7 days
Figure 5.
mRNA expression analysis of CSC markers Aldh1a1, Abcb1 and Snail following cisplatin/gemcitabine and menadione treatment of A549. A549 cells were treated with cisplatin/gemcitabine and menadione and transcript expression levels of (A) Aldh1a1, (B) Abcb1 and (C) Snail were measured relative to untreated control cells by real-time PCR
FACS analysis of CSC surface markers following cisplatin/gemcitabine and menadione treatment in A549 cells
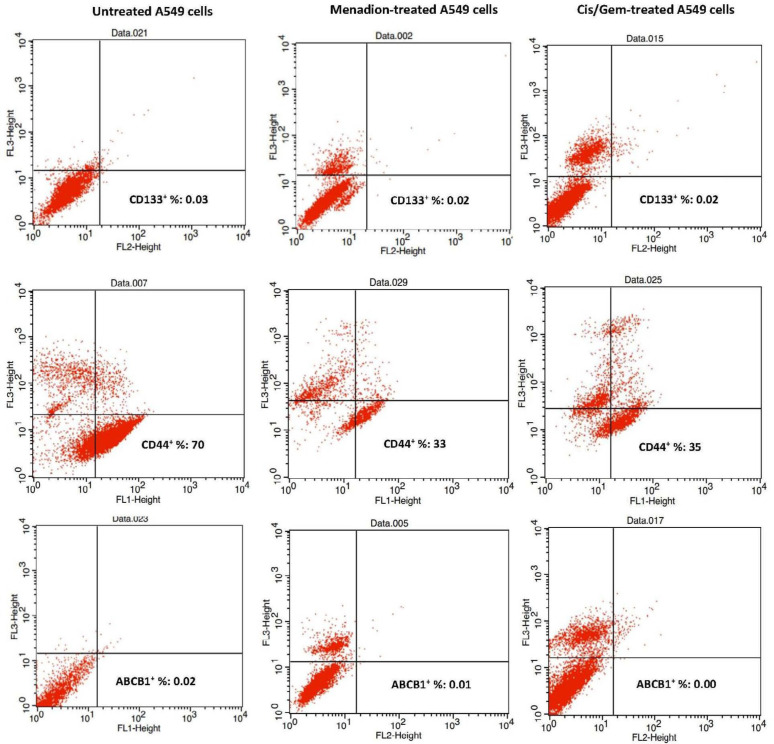
CD133 and CD44 are recognized as robust markers for CSCs in solid tumors (48-50). Therefore, in this study, the expression profile of CD133 and CD44 in cultures A549 cells before and after treatment with cisplatin/gemcitabine and menadione were analyzed by flow cytometry. Data showed that a high percentage of A549 cells expressed CD44 (70%) and this ratio dropped to half after treatment with menadione or cisplatin/gemcitabine. However, A549 cells did not show a significant population positive for CD133 and ABCB1 (less than 0.05%) and these fractions did not alter after treatment with two agents (Figure 6).
Figure 6.
Evaluation of CD133, CD44, and ABCB1 positive cells following A549 treatment with menadione and cisplatin/gemcitabine. A549 cells were treated with a combination of cisplatin (0.25 µM)/gemcitabine (5 µM) as well as 16 µM menadione for 7 days. Cells were analyzed after incubation with PE anti-Human CD133 FITC anti-Human CD44 and PE anti-Human ABCB1 antibodies with flow cytometry. The cell population represented in the right low quadrants are considered positive live cells for each marker
Discussion
Platinum-based doublet chemotherapy is the gold standard therapy for NSCLC patients where cisplatin or carboplatin are used in combination with drugs such as paclitaxel, gemcitabine, or vinorelbine (51). Despite advances in cancer therapy, a low survival rate in NSCLC patients displayed resistance of tumor cells to chemotherapy drugs (52). In various cancer types such as NSCLC, a subpopulation of cancer cells known as CSCs show resistance to therapeutic agents and lead to cancer relapse after therapeutic course (26, 53). On the other hand, traditional anticancer drugs are cytotoxic and inhibit normal cell division. Therefore, fast-dividing body cells such as blood cells and the cells lining the mouth, stomach, and intestines are sensitive to chemotherapy drugs. This results in a range of side-effect and chemotherapy-related toxicities after administration (54). Therefore, low effectiveness of common anticancer drugs against CSCs and their adverse side effects persuaded researcher to develop more effective treatments that could target CSCs and improve the clinical outcome of cancer patients.
In this study, we showed that menadione (Vitamin K3) reduced the expression of CSCs markers in NSCLC cell line A549. OCT4, NANOG, and SOX2 are three essential transcription factors contributing to pluripotency and self-renewal of normal and CSCs (10, 11). Menadione-induced downregulation of pluripotency markers Oct4, Nanog, and Sox2 show that this vitamin can inhibit self-renewal ability and induce differentiation of CSCs in the A549 cell line. The combination of cisplatin and gemcitabine which is used as a first-line regimen for patients with advanced NSCLC, also downregulated the expression of pluripotent markers. In other studies, downregulation of stem cell-associated genes was indicated after treatment with some chemotherapy drugs such as FOLFOX (5-fluorouracil plus oxaliplatin), docetaxel, and gemcitabine as well as phytochemicals agents such as berberine, apigenin, cinnamic acid, and kaempferol (55-58).
Moreover, CSCs are identified and isolated from various tumors based on overexpression of specific surface markers (59). CD44 and CD133 are two important surface membrane glycoproteins that have been identified as CSC markers in solid tumors and associate with growth, migration and invasion characteristic, drug resistance, and stem-like properties of CSCs (48, 60-62). In order to assess the effect of cancer cytotoxic agents on CSCs, expression of specific cell surface CSC markers was analyzed in various studies. For example, curcumin and its analog (Diflourinated Curcumin) either alone or in combination with chemotherapy drugs reduced CD44 and CD166 expression in colon and esophageal squamous carcinoma cell lines (63, 64). Here we showed that menadione as well as cisplatin/gemcitabine, meaningfully decreased CD44 and CD133 mRNA expression. As a result, it seems that these components can reduce proliferation, tumorigenesis, and stem cell-like properties in A549 cancer cells. Expression of cell surface markers CD44 was detectable on around 70% of the A549 cell line and this ratio decreased significantly after treatment with menadione and cisplatin/gemcitabine that is consistent with qRT-PCR results. However, using our antibody, we were not able to detect CD133 expression on a significant proportion of A549 cells as well as menadione or cisplatin/gemcitabine-treated cells.
Stem cell populations in many types of tumors are defined by the high activity of ALDHs. ALDHs are a superfamily of enzymes responsible for oxidizing endogenous and exogenous aldehydes to carboxylic acids. Therefore, ALDHs activity confers CSC resistance to antitumor drugs, so components that can reduce the expression of this marker or inhibit its activity play a key role in sensitizing resistant cells to cytotoxic agents (65-69). The current study also confirmed that cisplatin/gemcitabine, a standard drug for lung cancer treatment, and menadione decreased the expression of Aldh1a1. Overexpression of the ABC gene family is one of the principal mechanisms for protecting CSCs against many cytotoxic drugs that contribute to cancer resistance to chemotherapy (70). Drug-mediated inhibition in the expression of ABC transporters has the potential to re-sensitize CSCs to chemotherapy agents. Expression of ALDH1 and ABC transports was analyzed in many types of research to determine the effect of anticancer agents on CSCs. For example, expression of ALDH1A1 in the human esophageal squamous cell line was diminished after treatment with curcumin (63). In another research, berberine-treated MCF-7 and baicalein-treated myeloma cells were associated with a decrease in expression of ABCG2 relative to untreated cells (71, 72). Our results did not indicate significant downregulation of Abcb1 in cisplatin/gemcitabine and menadione-treated cells. Moreover, flow cytometry did not show detectable expression of ABCB1 on A549 cells, and this proportion did not change after treatment with two agents.
As a key transcriptional repressor of E-cadherin, Snail is recognized as a prominent inducer of EMT and tumor metastatic (73, 74). Considering that migratory cells with EMT phenotype have CSC characteristics, reduced expression of E-cadherin and increased expression of Snail can be considered as markers for detection of CSC population cells. (42, 43, 75). In research on human oral cancer cells, it was demonstrated that menadione reduced metastatic potential by expression induction of E-cadherin and down-regulation of EMT markers such as vimentin and fibronectin (38). Our results also revealed that Snail was downregulated significantly by menadione treatment. Reduced expression of EMT markers shows that menadione can suppress stem-like properties of cancer cells. Consequently, menadione, a synthetic version of vitamin K, exhibited anti-CSCs activities against lung cancer cells.
The promising role of vitamins, mostly vitamin A and D, in cancer prevention and treatment has emerged from the past few decades (29). Furthermore, vitamin A and D have also been shown to target CSCs in several studies. RA (used simplified here for all-trans-retinoic acid) is a metabolite of vitamin A1 that mediates the functions of vitamin A1 required for growth and development. RA- induced differentiation of CSCs in acute promyelocytic leukemia (APML) and breast cancer show the potential of vitamin A to eliminate CSCs (76, 77). In addition, RA decreased the expression of CSC markers (CD44 and ALDH) and stemness genes (KLF4 and SOX2) and inhibited CSC properties such as tumorspheres formation in gastric cancer (78, 79). β-carotene, a provitamin A carotenoid, inhibited self-renewal characteristics of CSCs and decreased expression of several stem cell markers (80). These findings suggest that RA may be regarded as a chemotherapeutic agent to target the CSC population. Inhibitory effects of vitamin D and its analogs on CSCs in various human cancer cells have been demonstrated in several studies. It was proved that vitamin D could induce differentiation of various neoplastic cells into a more mature phenotype (81-83). Signaling pathways, such as Notch, Hedgehog, Wnt and TGF-β are important signaling pathways in the maintenance of CSCs in human tumors. Therefore, these pathways have been considered as novel targets against CSCs (84, 85). Many studies have reported the inhibitory effect of vitamin D on Notch signaling, Hedgehog signaling and Wnt signaling pathways, indicating vitamin D can be considered as a promising therapeutic/preventive agent against CSCs (83, 86-91).
In conclusion, tumorigenic ability and resistance of CSCs to conventional chemo- and radio-therapy make them a potential target for anticancer drugs to reduce drug resistance and attenuate the likelihood of relapse. Here we introduced menadione as an anticancer agent that can down-regulate the expression of important CSC markers. Although treatment with cisplatin and gemcitabine showed approximately the same effect as menadione on downregulation of CSC-associated genes, combination therapy with menadione and chemotherapy drugs may not only reduce adverse side effects but improve the effectiveness of chemotherapy drugs and finally achieve prolonged survival of cancer patients. In-vivo experiment is needed to achieve more reliable results on the anti-CSCs effect of menadione and further validate the applicability of our results.
Acknowledgments
The authors thank “Vice Chancellor for Research and Technology”, Shahid Bahonar University of Kerman, Kerman, Iran for financial support.
Funding/Support
This work was supported by a grant from the Vice Chancellor for Research and Technology, Shahid Bahonar University of Kerman, Kerman, Iran.
References
- 1.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. . 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J. Clin. . 2016;66:7–30. doi: 10.3322/caac.21332. [DOI] [PubMed] [Google Scholar]
- 3.Hu B, Ma Y, Yang Y, Zhang L, Han H, Chen J. CD44 promotes cell proliferation in non-small cell lung cancer. Oncol. Lett. . 2018;15:5627–33. doi: 10.3892/ol.2018.8051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bertolini G, Roz L, Perego P, Tortoreto M, Fontanella E, Gatti L, Pratesi G, Fabbri A, Andriani F, Tinelli S. Highly tumorigenic lung cancer CD133+ cells display stem-like features and are spared by cisplatin treatment. Proc. Natl. Acad. Sci. . 2009;106:16281–6. doi: 10.1073/pnas.0905653106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Eramo A, Lotti F, Sette G, Pilozzi E, Biffoni M, Di Virgilio A, Conticello C, Ruco L, Peschle C, De Maria R. Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ. . 2008;15:504–14. doi: 10.1038/sj.cdd.4402283. [DOI] [PubMed] [Google Scholar]
- 6.Zheng Y, Cecile C, Sayles LC, Alleyne-Chin C, Vaka D, Knaak TD, Bigos M, Xu Y, Hoang CD, Shrager JB. A rare population of CD24+ ITGB4+ Notchhi cells drives tumor propagation in NSCLC and requires Notch3 for self-renewal. Cancer Cell . 2013;24:59–74. doi: 10.1016/j.ccr.2013.05.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lundholm L, Hååg P, Zong D, Juntti T, Mörk B, Lewensohn R, Viktorsson K. Resistance to DNA-damaging treatment in non-small cell lung cancer tumor-initiating cells involves reduced DNA-PK/ATM activation and diminished cell cycle arrest. Cell Death Dis. . 2013;4:e478. doi: 10.1038/cddis.2012.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liang S, Marti T, Dorn P, Froment L, Hall S, Berezowska S, Kocher G, Schmid R, Peng R. Blocking the epithelial-to-mesenchymal transition pathway abrogates resistance to anti-folate chemotherapy in lung cancer. Cell Death Dis. . 2015;6:e1824. doi: 10.1038/cddis.2015.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Soltanian S, Matin MM. Cancer stem cells and cancer therapy. Tumor Biol. . 2011;32:425–40. doi: 10.1007/s13277-011-0155-8. [DOI] [PubMed] [Google Scholar]
- 10.Liu A, Yu X, Liu S. Pluripotency transcription factors and cancer stem cells: small genes make a big difference. Chin. J. Cancer . 2013;32:483–7. doi: 10.5732/cjc.012.10282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Boyer LA, Lee TI, Cole MF, Johnstone SE, Levine SS, Zucker JP, Guenther MG, Kumar RM, Murray HL, Jenner RG. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell . 2005;122:947–56. doi: 10.1016/j.cell.2005.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tomita H, Tanaka K, Tanaka T, Hara A. Aldehyde dehydrogenase 1A1 in stem cells and cancer. Oncotarget. . 2016;7:11018–32. doi: 10.18632/oncotarget.6920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tanei T, Morimoto K, Shimazu K, Kim SJ, Tanji Y, Taguchi T, Tamaki Y, Noguchi S. Association of breast cancer stem cells identified by aldehyde dehydrogenase 1 expression with resistance to sequential Paclitaxel and epirubicin-based chemotherapy for breast cancers. Clin. Cancer Res. . 2009;15:4234–41. doi: 10.1158/1078-0432.CCR-08-1479. [DOI] [PubMed] [Google Scholar]
- 14.Abdullah LN, Chow EKH. Mechanisms of chemoresistance in cancer stem cells. Clin. Transl. Med. . 2013;2 doi: 10.1186/2001-1326-2-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bao S, Wu Q, McLendon RE, Hao Y, Shi Q, Hjelmeland AB, Dewhirst MW, Bigner DD, Rich JN. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature . 2006;444:756–60. doi: 10.1038/nature05236. [DOI] [PubMed] [Google Scholar]
- 16.Mathews LA, Cabarcas SM, Farrar WL. DNA repair: the culprit for tumor-initiating cell survival? Cancer Metast. Rev. . 2011;30:185–97. doi: 10.1007/s10555-011-9277-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mathews LA, Cabarcas SM, Hurt EM, Zhang X, Jaffee EM, Farrar WL. Increased expression of DNA repair genes in invasive human pancreatic cancer cells. Pancreas . 2011;40:730–9. doi: 10.1097/MPA.0b013e31821ae25b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wright MH, Calcagno AM, Salcido CD, Carlson MD, Ambudkar SV, Varticovski L. Brca1 breast tumors contain distinct CD44+/CD24-and CD133+ cells with cancer stem cell characteristics. Breast Cancer Res. . 2008;10:R10. doi: 10.1186/bcr1855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pan ST, Li ZL, He ZX, Qiu JX, Zhou SF. Molecular mechanisms for tumour resistance to chemotherapy. Clin. Exp. Pharmacol. Physiol. . 2016;43:723–37. doi: 10.1111/1440-1681.12581. [DOI] [PubMed] [Google Scholar]
- 20.Doyle LA, Ross DD. Multidrug resistance mediated by the breast cancer resistance Doyle LA, Ross DD. Multdrug resistance mediated by the breast cancer resistance protein BCRP Oncogene . 2003;22:7340. doi: 10.1038/sj.onc.1206938. [DOI] [PubMed] [Google Scholar]
- 21.Gottesman MM, Fojo T, Bates SE. Multidrug resistance in cancer: role of ATP–dependent transporters. Nat. Rev. Cancer . 2002;2:48–58. doi: 10.1038/nrc706. [DOI] [PubMed] [Google Scholar]
- 22.Viale A, De Franco F, Orleth A, Cambiaghi V, Giuliani V, Bossi D, Ronchini C, Ronzoni S, Muradore I, Monestiroli S. Cell-cycle restriction limits DNA damage and maintains self-renewal of leukaemia stem cells. Nature . 2009;457:51–6. doi: 10.1038/nature07618. [DOI] [PubMed] [Google Scholar]
- 23.Pece S, Tosoni D, Confalonieri S, Mazzarol G, Vecchi M, Ronzoni S, Bernard L, Viale G, Pelicci PG, Di Fiore PP. Biological and molecular heterogeneity of breast cancers correlates with their cancer stem cell content. Cell . 2010;140:62–73. doi: 10.1016/j.cell.2009.12.007. [DOI] [PubMed] [Google Scholar]
- 24.Valent P, Bonnet D, De Maria R, Lapidot T, Copland M, Melo JV, Chomienne C, Ishikawa F, Schuringa JJ, Stassi G. Cancer stem cell definitions and terminology: the devil is in the details. Nat. Rev. Cancer . 2012;12:767–75. doi: 10.1038/nrc3368. [DOI] [PubMed] [Google Scholar]
- 25.Prieto-Vila M, Takahashi RU, Usuba W, Kohama I, Ochiya T. Drug resistance driven by cancer stem cells and their niche. Int. J. Mol. Sci. . 2017;18:2574. doi: 10.3390/ijms18122574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Phi LTH, Sari IN, Yang YG, Lee SH, Jun N, Kim KS, Lee YK, Kwon HY. Cancer stem cells (CSCs) in drug resistance and their therapeutic implications in cancer treatment. Stem Cells Int. . 2018:2018. doi: 10.1155/2018/5416923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Soltanian S, Dehghani H. BORIS: a key regulator of cancer stemness. Cancer Cell Int. . 2018;18:154–67. doi: 10.1186/s12935-018-0650-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Soltanian S, Sheikhbahaei M, Mohamadi N. cytotoxicity evaluation of methanol extracts of some medicinal plants on P19 embryonal carcinoma cells. J. Appl. Pharm. Sci. . 2017;7:142–9. [Google Scholar]
- 29.Jain A, Tiwari A, Verma A, Jain S. Vitamins for cancer prevention and treatment: an insight. Curr. Mol. Med. . 2017;17:321–40. doi: 10.2174/1566524018666171205113329. [DOI] [PubMed] [Google Scholar]
- 30.Lockwood K, Moesgaard S, Yamamoto T, Folkers K. Progress on therapy of breast cancer with vitamin Q10 and the regression of metastases. Biochem. Biophys. Res. Commun. . 1995;212:172–7. doi: 10.1006/bbrc.1995.1952. [DOI] [PubMed] [Google Scholar]
- 31.Fortmann SP, Burda BU, Senger CA, Lin JS, Whitlock EP. Vitamin and mineral supplements in the primary prevention of cardiovascular disease and cancer: an updated systematic evidence review for the US Preventive Services Task Force. Ann. Intern. Med. . 2013;159:824–34. doi: 10.7326/0003-4819-159-12-201312170-00729. [DOI] [PubMed] [Google Scholar]
- 32.Nutter LM, Ann-Lii C, Hsiao-Ling H, Ruey-Kun H, Ngo EO, Tsang-Wu L. Menadione: spectrum of anticancer activity and effects on nucleotide metabolism in human neoplastic cell lines. Biochem. Pharmacol. . 1991;41:1283–92. doi: 10.1016/0006-2952(91)90099-q. [DOI] [PubMed] [Google Scholar]
- 33.Juan CC, Wu FY. Vitamin K3 inhibits growth of human hepatoma HepG2 cells by decreasing activities of both p34cdc2 kinase and phosphatase. Biochem. Bioph. Res. Co. . 1993;190:907–13. doi: 10.1006/bbrc.1993.1135. [DOI] [PubMed] [Google Scholar]
- 34.Matzno S, Yamaguchi Y, Akiyoshi T, Nakabayashi T, Matsuyama K. An attempt to evaluate the effect of vitamin K3 using as an enhancer of anticancer agents. Biol. Pharm. Bull. . 2008;31:1270–3. doi: 10.1248/bpb.31.1270. [DOI] [PubMed] [Google Scholar]
- 35.Akman SA, Dietrich M, Chlebowski R, Limberg P, Block JB. Modulation of cytotoxicity of menadione sodium bisulfite versus leukemia L1210 by the acid-soluble thiol pool. Cancer Res. . 1985;45:5257–62. [PubMed] [Google Scholar]
- 36.Akman SA, Doroshow JH, Dietrich MF, Chlebowski RT, Block JS. Synergistic cytotoxicity between menadione and dicumarol vs murine leukemia L1210. J. Pharmacol. Exp. Ther. . 1987;240:486–91. [PubMed] [Google Scholar]
- 37.Jamison JM, Gilloteaux J, Taper HS, Summers JL. Evaluation of the in-vitro and in-vivo antitumor activities of vitamin C and K-3 combinations against human prostate cancer. J. Nutr. . 2001;131:158S–60S. doi: 10.1093/jn/131.1.158S. [DOI] [PubMed] [Google Scholar]
- 38.Suresh S, Raghu D, Karunagaran D. Menadione (Vitamin K3) induces apoptosis of human oral cancer cells and reduces their metastatic potential by modulating the expression of epithelial to mesenchymal transition markers and inhibiting migration. Asian Pac. J. Cancer Prev. . 2013;14:5461–5. doi: 10.7314/apjcp.2013.14.9.5461. [DOI] [PubMed] [Google Scholar]
- 39.Tarin D. The fallacy of epithelial mesenchymal transition in neoplasia. Cancer Res. . 2005;65:5996–6001. doi: 10.1158/0008-5472.CAN-05-0699. [DOI] [PubMed] [Google Scholar]
- 40.Kalluri R, Weinberg RA. The basics of epithelial-mesenchymal transition. J. Clin. Invest. . 2009;119:1420–8. doi: 10.1172/JCI39104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Shibue T, Weinberg RA. EMT, CSCs, and drug resistance: the mechanistic link and clinical implications. Nat. Rev. Clin. Oncol. . 2017;14 doi: 10.1038/nrclinonc.2017.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Singh A, Settleman J. EMT, cancer stem cells and drug resistance: an emerging axis of evil in the war on cancer. Oncogene . 2010;29:4741–51. doi: 10.1038/onc.2010.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mani SA, Guo W, Liao MJ, Eaton EN, Ayyanan A, Zhou AY, Brooks M, Reinhard F, Zhang CC, Shipitsin M. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell . 2008;133:704–15. doi: 10.1016/j.cell.2008.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Du B, Shim J. Targeting epithelial–mesenchymal transition (EMT) to overcome drug resistance in cancer. Molecules . 2016;21:965. doi: 10.3390/molecules21070965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Moustakas A, de Herreros AG. Epithelial–mesenchymal transition in cancer. Mol. Oncol. . 2017;11:715–7. doi: 10.1002/1878-0261.12094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Villarejo A, Cortés-Cabrera Á, Molina-Ortíz P, Portillo F, Cano A. Differential role of Snail1 and Snail2 zinc fingers in E-cadherin repression and epithelial to mesenchymal transition. J. Biol. Chem. . 2014;289:930–41. doi: 10.1074/jbc.M113.528026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Batlle E, Sancho E, Francí C, Domínguez D, Monfar M, Baulida J, De Herreros AG. The transcription factor snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nat. Cell Biol. . 2000;2:84–9. doi: 10.1038/35000034. [DOI] [PubMed] [Google Scholar]
- 48.Lee WJ. The prospects of vitamin C in cancer therapy. Immune. Netw. . 2009;9:147–52. doi: 10.4110/in.2009.9.5.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Woodward WA, Sulman EP. Cancer stem cells: markers or biomarkers? Cancer Metast. Rev. . 2008;27:459–70. doi: 10.1007/s10555-008-9130-2. [DOI] [PubMed] [Google Scholar]
- 50.Marhaba R, Klingbeil P, Nuebel T, Nazarenko I, Buechler MW, Zoeller M. CD44 and EpCAM: cancer-initiating cell markers. Curr. Mol. Med. . 2008;8:784–804. doi: 10.2174/156652408786733667. [DOI] [PubMed] [Google Scholar]
- 51.Cosaert J, Quoix E. Platinum drugs in the treatment of non-small-cell lung cancer. Brit. J. cancer. . 2002;87:825–33. doi: 10.1038/sj.bjc.6600540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Leon G, MacDonagh L, Finn SP, Cuffe S, Barr MP. Cancer stem cells in drug resistant lung cancer: Targeting cell surface markers and signaling pathways. Pharmacol. Therapeut. . 2016;158:71–90. doi: 10.1016/j.pharmthera.2015.12.001. [DOI] [PubMed] [Google Scholar]
- 53.Chen LS, Wang AX, Dong B, Pu KF, Yuan LH, Zhu YM. A new prospect in cancer therapy: targeting cancer stem cells to eradicate cancer. Chin. J. Cancer. . 2012;31:564–72. doi: 10.5732/cjc.011.10444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Jiang Q. Natural forms of vitamin E as effective agents for cancer prevention and therapy. Adv. Nutr. . 2017;8:850–67. doi: 10.3945/an.117.016329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Park S, Sung J, Chung N. Berberine diminishes side population and down-regulates stem cell-associated genes in the pancreatic cancer cell lines PANC-1 and MIA PaCa-2. Mol Cell Biochem. . 2014;394:209–15. doi: 10.1007/s11010-014-2096-1. [DOI] [PubMed] [Google Scholar]
- 56.Soltanian S, Riahirad H, Pabarja A, Karimzadeh MR, Saeidi K, Perez-Tejada E, Morales-Corona J, Gomez-Quiroz L, Gutierrez-Ruiz M, Olayo R. Kaempferol and docetaxel diminish side population and down-regulate some cancer stem cell markers in breast cancer cell line MCF-7. Biocell. . 2017;41:33–40. [Google Scholar]
- 57.Soltanian S, Riahirad H, Pabarja A, Jafari E, Khandani BK. Effect of cinnamic acid and FOLFOX in diminishing side population and downregulating cancer stem cell markers in colon cancer cell line HT-29. DARU . 2018;26:19–29. doi: 10.1007/s40199-018-0210-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kim B, Jung N, Lee S, Sohng JK, Jung HJ. Apigenin inhibits cancer stem cell-like phenotypes in human glioblastoma cells via suppression of c-Met signaling. Phytother. Res. . 2016;30:1833–40. doi: 10.1002/ptr.5689. [DOI] [PubMed] [Google Scholar]
- 59.Ailles LE, Weissman IL. Cancer stem cells in solid tumors. Curr. Opin. Biotech. . 2007;18:460–6. doi: 10.1016/j.copbio.2007.10.007. [DOI] [PubMed] [Google Scholar]
- 60.Glumac PM, LeBeau AM. The role of CD133 in cancer: a concise review. Clin. Transl. Med. . 2018;7:18. doi: 10.1186/s40169-018-0198-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Taylor WF, Jabbarzadeh E. The use of natural products to target cancer stem cells. Am. J. Cancer Res. . 2017;7:1588–605. [PMC free article] [PubMed] [Google Scholar]
- 62.Wang C, Xie J, Guo J, Manning HC, Gore JC, Guo N. Evaluation of CD44 and CD133 as cancer stem cell markers for colorectal cancer. Oncol. Rep. . 2012;28:1301–8. doi: 10.3892/or.2012.1951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Almanaa TN, Geusz ME, Jamasbi RJ. Effects of curcumin on stem-like cells in human esophageal squamous carcinoma cell lines. BMC Ccmplem. Altern. M. . 2012;12:195. doi: 10.1186/1472-6882-12-195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kanwar SS, Yu Y, Nautiyal J, Patel BB, Padhye S, Sarkar FH, Majumdar AP. Difluorinated-curcumin (CDF): a novel curcumin analog is a potent inhibitor of colon cancer stem-like cells. Pharm. Res. . 2011;28:827–38. doi: 10.1007/s11095-010-0336-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Nakahata K, Uehara S, Nishikawa S, Kawatsu M, Zenitani M, Oue T, Okuyama H. Aldehyde dehydrogenase 1 (ALDH1) is a potential marker for cancer stem cells in embryonal rhabdomyosarcoma. PloS One . 2015;10:e0125454. doi: 10.1371/journal.pone.0125454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Han L, Shi S, Gong T, Zhang Z, Sun X. Cancer stem cells: therapeutic implications and perspectives in cancer therapy. Acta Pharm. Sin B . . 2013;3:65–75. [Google Scholar]
- 67.Vassalli G. Aldehyde dehydrogenases: Not just markers, but functional regulators of stem cells. Stem Cells Int. . 2019;2019:3904645. doi: 10.1155/2019/3904645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kohn FR, Landkamer GJ, Manthey CL, Ramsay NK, Sladek NE. Effect of aldehyde dehydrogenase inhibitors on the ex vivo sensitivity of human multipotent and committed hematopoietic progenitor cells and malignant blood cells to oxazaphosphorines. Cancer Res. . 1987;47:3180–5. [PubMed] [Google Scholar]
- 69.Landen CN, Goodman B, Katre AA, Steg AD, Nick AM, Stone RL, Miller LD, Mejia PV, Jennings NB, Gershenson DM. Targeting aldehyde dehydrogenase cancer stem cells in ovarian cancer. Mol. Cancer Ther. . 2010;9:3186–99. doi: 10.1158/1535-7163.MCT-10-0563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Dean M. ABC transporters, drug resistance, and cancer stem cells. J. Mammary Gland Biol. Neoplasia. . 2009;14:3–9. doi: 10.1007/s10911-009-9109-9. [DOI] [PubMed] [Google Scholar]
- 71.Kim JB, Ko E, Han W, Shin I, Park SY, Noh DY. Berberine diminishes the side population and ABCG2 transporter expression in MCF-7 breast cancer cells. Planta Medica . 2008;74:1693–700. doi: 10.1055/s-0028-1088313. [DOI] [PubMed] [Google Scholar]
- 72.Gu YY, Liu LP, Qin J, Zhang M, Chen Y, Wang D, Li Z, Tang JZ, Mo SL. Baicalein decreases side population proportion via inhibition of ABCG2 in multiple myeloma cell line RPMI 8226 in-vitro. Fitoterapia . 2014;94:21–8. doi: 10.1016/j.fitote.2014.01.019. [DOI] [PubMed] [Google Scholar]
- 73.Barrallo-Gimeno A, Nieto MA. The Snail genes as inducers of cell movement and survival: implications in development and cancer. Development . 2005;132:3151–61. doi: 10.1242/dev.01907. [DOI] [PubMed] [Google Scholar]
- 74.Wang Y, Shi J, Chai K, Ying X, P Zhou B. The role of Snail in EMT and tumorigenesis. Curr. Cancer Drug Targets . 2013;13:963–72. doi: 10.2174/15680096113136660102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kong D, Li Y, Wang Z, Sarkar F. Cancer stem cells and epithelial-to-mesenchymal transition (EMT)-phenotypic cells: are they cousins or twins? Cancers . 2011;3:716–29. doi: 10.3390/cancers30100716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Tallman MS, Andersen JW, Schiffer CA, Appelbaum FR, Feusner JH, Ogden A, Shepherd L, Willman C, Bloomfield CD, Rowe JM. All-trans-retinoic acid in acute promyelocytic leukemia. New. Engl. J. Med. . 1997;337:1021–8. doi: 10.1056/NEJM199710093371501. [DOI] [PubMed] [Google Scholar]
- 77.Ginestier C, Wicinski J, Cervera N, Monville F, Finetti P, Bertucci F, Wicha MS, Birnbaum D, Charafe-Jauffret E. Retinoid signaling regulates breast cancer stem cell differentiation. Cell Cycle. . 2009;8:3297–302. doi: 10.4161/cc.8.20.9761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Nguyen PH, Giraud J, Staedel C, Chambonnier L, Dubus P, Chevret E, Bœuf H, Gauthereau X, Rousseau B, Fevre M. All-trans retinoic acid targets gastric cancer stem cells and inhibits patient-derived gastric carcinoma tumor growth. Oncogene . 2016;35:5619–28. doi: 10.1038/onc.2016.87. [DOI] [PubMed] [Google Scholar]
- 79.Bouriez D, Giraud J, Gronnier C, Varon C. Efficiency of all-trans retinoic acid on gastric cancer: a narrative literature review. Int. J. Mol. Sci. . 2018;19:3388. doi: 10.3390/ijms19113388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lee HA, Park S, Kim Y. Effect of β-carotene on cancer cell stemness and differentiation in SK-N-BE (2) C neuroblastoma cells. Oncol. Rep. . 2013;30:1869–77. doi: 10.3892/or.2013.2643. [DOI] [PubMed] [Google Scholar]
- 81.Gocek E, Studzinski GP. Vitamin D and differentiation in cancer. Crit. Rev. Clin. Lab. Sci. . 2009;46:190–209. doi: 10.1080/10408360902982128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Pálmer HG, González-Sancho JM, Espada J, Berciano MT, Puig I, Baulida J, Quintanilla M, Cano A, de Herreros AG, Lafarga M. Vitamin D3 promotes the differentiation of colon carcinoma cells by the induction of E-cadherin and the inhibition of β-catenin signaling. J. Cell Biol. . 2001;154:369–88. doi: 10.1083/jcb.200102028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.So JY, Lee HJ, Smolarek AK, Paul S, Wang CX, Maehr H, Uskokovic M, Zheng X, Conney AH, Cai L. A novel oemini vitamin D analog represses the expression of a stem cell marker CD44 in breast cancer. Mol. Pharmacol. . 2011;79:360–7. doi: 10.1124/mol.110.068403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Takebe N, Harris PJ, Warren RQ, Ivy SP. Targeting cancer stem cells by inhibiting Wnt, Notch, and Hedgehog pathways. Nat. Rev. Clin. Oncol. . 2011;8:97. doi: 10.1038/nrclinonc.2010.196. [DOI] [PubMed] [Google Scholar]
- 85.Karamboulas C, Ailles L. Developmental signaling pathways in cancer stem cells of solid tumors. Biochim. Biophys. Acta . 2013;1830:2481–95. doi: 10.1016/j.bbagen.2012.11.008. [DOI] [PubMed] [Google Scholar]
- 86.Kovalenko PL, Zhang Z, Cui M, Clinton SK, Fleet JC. 1, 25 dihydroxyvitamin D-mediated orchestration of anticancer, transcript-level effects in the immortalized, non-transformed prostate epithelial cell line, RWPE1. BMC Genomics . 2010;11:26. doi: 10.1186/1471-2164-11-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Bijlsma MF, Spek CA, Zivkovic D, van de Water S, Rezaee F, Peppelenbosch MP. Repression of smoothened by patched-dependent (pro-) vitamin D3 secretion. PLoS Biol. . 2006;4:e232. doi: 10.1371/journal.pbio.0040232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Tang JY, Xiao TZ, Oda Y, Chang KS, Shpall E, Wu A, So PL, Hebert J, Bikle D, Epstein EH. Vitamin D3 inhibits hedgehog signaling and proliferation in murine Basal cell carcinomas. Cancer Prev. Res. . 2011;4:744–51. doi: 10.1158/1940-6207.CAPR-10-0285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Larriba M, González-Sancho J, Barbáchano A, Niell N, Ferrer-Mayorga G, Muñoz A. Vitamin D is a multilevel repressor of Wnt/b-catenin signaling in cancer cells. Cancers . 2013;5:1242–60. doi: 10.3390/cancers5041242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.So JY, Suh N. Targeting cancer stem cells in solid tumors by vitamin D. J. Steroid Biochem. Mol. Biol. . 2015;148:79–85. doi: 10.1016/j.jsbmb.2014.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Li MX, Li LF, Zhang L, Xiao ZG, Shen J, Hu W, Zeng Q, Cho CH. Vitamin D and cancer stem cells in the gastrointestinal tract. Curr. Med. Chem. . 2017;24:918–27. doi: 10.2174/0929867324666170214110633. [DOI] [PubMed] [Google Scholar]