Abstract
Abstract
Time and again, yeast has proven to be a vital model system to understand various crucial basic biology questions. Studies related to viruses are no exception to this. This simple eukaryotic organism is an invaluable model for studying fundamental cellular processes altered in the host cell due to viral infection or expression of viral proteins. Mechanisms of infection of several RNA and relatively few DNA viruses have been studied in yeast to date. Yeast is used for studying several aspects related to the replication of a virus, such as localization of viral proteins, interaction with host proteins, cellular effects on the host, etc. The development of novel techniques based on high-throughput analysis of libraries, availability of toolboxes for genetic manipulation, and a compact genome makes yeast a good choice for such studies. In this review, we provide an overview of the studies that have used yeast as a model system and have advanced our understanding of several important viruses.
Key points
• Yeast, a simple eukaryote, is an important model organism for studies related to viruses.
• Several aspects of both DNA and RNA viruses of plants and animals are investigated using the yeast model.
• Apart from the insights obtained on virus biology, yeast is also extensively used for antiviral development.
Keywords: Yeast, Virus, Replication, Saccharomyces cerevisiae, Schizosaccharomyces pombe
Introduction
Viruses are notorious as obligate intracellular parasites dependent on host cells for their replication and effect cells across kingdoms. The role of host cells in viral replication and the host cellular pathways affected by viral infections are widely studied. Extensive host cellular reorganization has been observed in various organisms upon viral infection. Understanding the molecular details of this reorganization will help to unravel the infection pathways. A role for the sub-cellular organelles for multiple facets of the viral replication cycle is highlighted in recent studies (Glingston et al. 2019). Host protein spatio-temporal localization and functions, their interaction with the viral proteins and localization of the viral proteins are few crucial aspects studied extensively (Nagy et al. 2014). Elucidating the virus lifecycle in the host organism of its infection may be the apt way, albeit it may not be possible in some cases due to technical, safety, and ethical hitches. Alternative suitable hosts are hence used as several cellular processes are conserved across kingdoms. Coronavirus pandemic of 2019 caused by severe acute respiratory syndrome coronovirus-2 (SARS-CoV-2) is an excellent recent example where studies of similar corona viruses in yeast and other model systems not only enabled researchers to rapidly understand the biology of the virus but also helped in developing antivirals/vaccines (Pollet et al. 2020). Virus research has undoubtedly benefitted using alternative hosts such as Caenorhabditis elegans, Drosophila, Zebra fish, mammalian cell lines, mouse models, and yeast with each one having its own pros and cons (Chua et al. 2019).
Yeast: apt but with caution
Yeast is a single-celled organism that belongs to the kingdom fungi. Several essential cellular processes/functions are conserved from yeast to higher eukaryotes. Interestingly, viruses target these conserved processes for successful infection and hence studies in yeast hold great significance. The ease of culturing, short doubling time, availability of a wealth of experimental tools for genetic manipulation, and possibility of heterologous expression of proteins of higher eukaryotes are some other advantages that make yeast an important model system for viral studies. Two commonly used yeast models for such studies are Saccharomyces cerevisiae (budding yeast) and Schizosaccharomyces pombe (fission yeast) (Zhao 2017; Li and Zhao 2018). Additionally, other yeast models such as Pichia pastoris and Hansenula polymorpha are also extensively used to express viral proteins for the production of vaccines (Vogl et al. 2013). Yeast studies have not only contributed toward understanding viral replication but also aided in the elucidation of the functions of individual proteins of various viruses. Expression of viral proteins in yeast cells can be achieved either by using centromere-containing (CEN; copy number 1–3) or 2 micron (copy number 40–60) plasmids or targeted homologous recombination to introduce a single copy of a gene into the yeast genome. The small genome of yeast with fewer genome duplications also provides an added advantage to study host-virus interactions. On the other hand, extensive gene duplication in physiological host organisms such as plants or human cells may hamper such studies. Identified interacting host factors of yeast have further been validated in physiological hosts highlighting the aptness of yeast for such studies. On the other hand, several studies have also used yeast for validating identified host (plant or animal) factors by orthologous expression of these proteins in yeast (Nagy et al. 2014). However, it cannot be ignored that the identified host factors in yeast may function differently in physiological hosts. Cellular effects in multicellular organisms may also be attributed to inter-cellular interactions and yeast is not capable of addressing such pehnotypes. In line with this, the physiological host may also have tissue-specific pathways/factors that aid specific interaction with virus and such studies may not be fruitful using yeast.
Several manuscripts have been published highlighting the importance of yeast in virology (Galao et al. 2007; Zhao 2017). This review aims to provide an updated overview of the insights obtained in virus research using yeast as a model system by extending the repertoire of viruses, both DNA and RNA studied in yeast recently and discussing the various aspects of the virus biology benefitted by yeast studies (Table 1). For ease of understanding, we have divided all the subsections into RNA and DNA viruses. Yeast studies have undoubtedly augmented our knowledge of viral genome replication, virus-host interaction, viral protein localization, and effect on intracellular structures, regulation of host cell cycle, and cell death (Fig. 1). Further, the utility of genome-wide studies in yeast and the potential of yeast for antiviral and vaccine development are also discussed.
Table 1.
An overview of the viruses listed in this paper and studied in yeast related to various aspects of the viral life cycle and virus-host interaction
| S. No | Virus | Viral genome replication | Virus host interaction | Localization and effects | Cell cycle regulation | Cell death and apoptosis |
|---|---|---|---|---|---|---|
| RNA viruses | ||||||
| 1 | Brome mosaic virus | ✓ | ✓ | ✓ | ||
| 2 | Carnation Italian ringspot virus | ✓ | ✓ | ✓ | ✓ | |
| 3 | Cucumber necrosis virus | ✓ | ✓ | |||
| 4 | Cymbidium ringspot virus | ✓ | ✓ | |||
| 5 | Flock house virus | ✓ | ✓ | ✓ | ||
| 6 | Hepatitis C virus | ✓ | ||||
| 7 | Human immunodeficiency virus | ✓ | ✓ | ✓ | ✓ | ✓ |
| 8 | Human T-lymphotropic type 1 virus | ✓ | ||||
| 9 | Influenza virus | ✓ | ✓ | |||
| 10 | Japanese encephalitis virus | ✓ | ||||
| 11 | Nodamura virus | ✓ | ||||
| 12 | Poliovirus | ✓ | ✓ | |||
| 13 | Rubellavirus | ✓ | ||||
| 14 | Sonchus yellow net nucleorhabdovirus | ✓ | ||||
| 15 | Tetravirus Helicoverpa armigera stunt virus | ✓ | ||||
| 16 | Tomato bushy stunt virus | ✓ | ✓ | ✓ | ||
| 17 | Zika virus | ✓ | ✓ | ✓ | ||
| DNA viruses | ||||||
| 1 | Abutilon mosaic virus | ✓ | ||||
| 2 | Adeno associated virus | ✓ | ||||
| 3 | Adenovirus | ✓ | ✓ | ✓ | ||
| 4 | African cassava mosaic virus | ✓ | ✓ | |||
| 5 | African swine fever virus | ✓ | ||||
| 6 | Bovine papilloma virus | ✓ | ✓ | |||
| 7 | Deerpox virus | ✓ | ||||
| 8 | Ebstein-Barr virus | ✓ | ||||
| 9 | Hepatitis B virus | ✓ | ||||
| 10 | Human papilloma virus | ✓ | ✓ | ✓ | ✓ | |
| 11 | Indian mungbean yellow mosaic virus | ✓ | ||||
| 12 | Maize streak virus | ✓ | ||||
| 13 | Mimivirus | ✓ | ||||
| 14 | Polyomavirus | ✓ | ||||
| 15 | Simian virus 40 | ✓ | ||||
| 16 | Vaccinia virus | ✓ | ||||
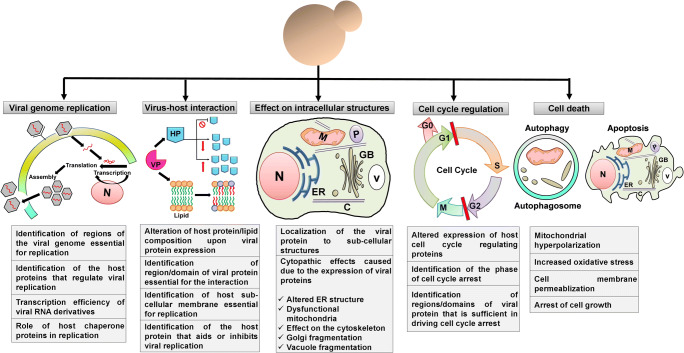
Fig. 1.
Various aspects of virus research benefitted by studies in yeast. The figure summarizes various aspects of virus research studied using yeast (both budding and fission) as a model system. Viral genome replication, virus-host interactions, effect of expression of the viral proteins on host intracellular structures, cell cycle regulation, and cell death of both RNA and DNA viruses are some important aspects extensively studied in these model organsims. Important contributions from studies in yeast in each of these topics are listed in the text boxes. HP host protein, VP viral protein, M mitochondria, N nucleus, V vacuole, ER endoplasmic reticulum, GB golgi body, C cytoskeleton.  -inhibition,
-inhibition,  -reduced expression, and
-reduced expression, and  -increased expression
-increased expression
Viral genome replication
Over the years, many DNA and RNA viruses have been reported to replicate in yeast cells, some of which are pathologically very important for humans (Alves-Rodrigues et al. 2006; Zhao 2017) (Table 1). Several positive strand RNA viruses are reported to utilize host sub-cellular membranes for their replication (Nagy 2008). Brome mosaic virus (BMV) was the first virus reported to replicate its genome in yeast cells (Janda and Ahlquist 1993). Transcription efficiency of viral RNA derivatives, regions of the viral genome essential for replication, identification of host proteins that regulate viral replication, role of host chaperone proteins in replication, and the mechanism of replication are some of the important topics addressed using yeast which are further validated in the physiological host (Fig. 1).
RNA viruses
Interestingly, most of the RNA viruses described to replicate in yeast are of positive polarity and include both plant and animal viruses. Plant viruses reported to replicate in yeast mainly belong to the family Bromoviridae (e.g., BMV) (Janda and Ahlquist 1993) and Tombusviridae (Carnation Italian ringspot virus (CIRV), Tomato bushy stunt virus (TBSV), Cymbidium ringspot virus (CymRSV)) (Nawaz-ul-Rehman et al. 2013; Pantaleo et al. 2003; Rubino et al. 2007). Animal RNA viruses like Flock House virus (FHV) and Nodamura virus (NoV) are also reported to replicate in yeast (Alves-Rodrigues et al. 2006).
Upon expression of BMV replication proteins, 1a and 2a in yeast, RNA-dependent replication and transcription of viral RNA3 derivatives was reported (Ishikawa et al. 1997). The expression of a reporter gene that replaced the coat protein in RNA3 derivatives was used as a screening marker for viral replication (Ishikawa et al. 1997; Kushner et al. 2003). An important advantage of using S. cerevisiae for viral studies is the presence of the yeast knock-out library that provides a systematic analysis of each gene deletion. Ninety-six non-essential host genes involved in several cellular pathways that either promote or inhibit TBSV replication were identified using this library (Panavas et al. 2005b). Yeast Tet promoter Hughes collection (yTHC) and temperature-sensitive (ts) essential gene library were used to identify additional host factors that regulate the accumulation of TBSV replicon in yeast cells. Several host factors involved in RNA transcription, protein transport, metabolism, or replication were identified in this study (Jiang et al. 2006; Nawaz-ul-Rehman et al. 2013). Panavas and colleagues reported that the expression of the viral replicase proteins in yeast was sufficient for the defective interfering RNA (DI RNA) replication of the TBSV virus (Panavas et al. 2005b). A role for enhanced phospholipid biosynthesis that facilitates TBSV replication in yeast cells was also proposed. p33 protein was reported to interact with phospholipid sensor proteins of the host cell leading to the upregulation of transcription of phospholipid biosynthesis genes facilitating TBSV replication (Barajas et al. 2014).
FHV and NoV are insect viruses that belong to the family Nodaviridae. Both FHV and NoV are reported to replicate their entire genome in budding yeast (Price et al. 2005; Price et al. 1996). Successful RNA replication, production of subgenomic RNA, and assembly of FHV viruses upon transfection of the isolated viral RNA in yeast cells was reported (Price et al. 2005). The co-chaperone of the heat shock protein Hsp70, Ydj1 was reported to play a crucial role in the assembly of the functional FHV replication complexes (which facilitates viral genome replication) in yeast cells (Weeks and Miller 2008).
S. cerevisiae has also contributed to understanding the mechanisms essential for the replication of the viral genome. One such common mechanism seen in RNA viruses is ribosomal frameshifting. This is used to increase the coding capacity of the viral genome and also regulates the stoichiometry of viral proteins (Advani and Dinman 2016). For instance, the human immunodeficiency virus-1 (HIV-1) utilizes this mechanism (− 1 ribosomal frameshifting) to produce Gag-Pol polyprotein from the Gag-coding mRNA. The − 1 ribosomal frameshifting signal comprises of a heptameric (U UUU UUA) slippery sequence and a stimulatory element P3 (Watts et al. 2009). A direct correlation between the frameshift efficiency and the presence of stimulatory elements has been reported (Bidou et al. 2010; Watts et al. 2009; Wilson et al. 1988). Interestingly, the molecular mechanism of this process is conserved from yeast to human cells and is reported to be crucial for viral replication (Bidou et al. 2010; Penno et al. 2017). Further, an important role of the Gag-Pol ribosomal frameshift signal in HIV-1 RNA packaging was also reported (Chamanian et al. 2013). The HIV-1 frameshift-stimulating RNA that plays a critical role in the replication of the virus can be a target for anti-HIV drug development. Triazole-containing compounds exhibit high affinity and selectivity toward this regulatory RNA and hence can be a target to inhibit viral replication (Hilimire et al. 2017). Positive-strand RNA viruses are a large group of viruses that includes many important human pathogens such as Hepatitis C virus (HCV), SARS, SARS-CoV-2, HIV, etc. and share a high degree of similarity in their replication cycles. Therefore, understanding strategies for replication may go beyond the specific virus studied.
Influenza virus belongs to the Orthomyxoviridae family, which are negative-sense single-stranded RNA viruses that cause respiratory diseases. Yeast has been used to study the transcription and replication of the viral genome, polyadenylation, translational control, and mechanism of viral budding out of the cell (Chua et al. 2019). The transcriptional elongation factor, Tat stimulatory factor 1 (Tat-SF1) homolog of yeast CUS2, was identified as an essential factor for synthesizing the influenza viral genome in yeast (Naito et al. 2007). Further, the authors reported that Tat-SF1 interacted with free nucleoprotein (NP) during viral genome synthesis and was suggested to serve as a molecular chaperone for NP aiding in the formation of NP-RNA complexes in mammalian cells. A system comprising the essential components for the efficient replication and transcription of the influenza virus in yeast cells that includes viral RNAs, viral RNA polymerases, and NP was developed (Naito et al. 2007). Such a system enables the identification of host factors interacting with virus components.
DNA viruses
Replication studies of DNA viruses in yeast are limited to only a handful of viruses, mainly belonging to the family Papillomaviridae, Geminiviridae, and Parvoviridae (Zhao 2017). Geminiviridae is a family of viruses that infects plants and causes a high rate of crop damage. Their genome consists of single-stranded DNA and includes viruses like African cassava mosaic virus (ACMV), maize streak virus (MSV), and Indian mungbean yellow mosaic virus (IMYMV) (Zhao 2017). These viruses have either one or two circular ssDNA molecules (two DNA molecules: DNA-A and DNA-B in ACMV), which encodes for proteins required for viral DNA replication and transcription regulation. Replication-associated protein (Rep) is one such quintessential protein encoded by DNA-B molecule and plays an essential role in rolling circle replication of the ACMV genome (Hanley-Bowdoin et al. 2004; Kittelmann et al. 2009). Along with Rep’s characteristic DNA cleavage activity, the unique potential of Rep in helping the cell to re-enter the cell cycle and induce genomic replication was studied in S. pombe (Kittelmann et al. 2009). Rep reactivates the S-phase of the host cell by binding to the heterologously expressed plant homolog (pRBR) of retinoblastoma protein to promote G1-S transition in yeast (Kittelmann et al. 2009). Rep expressing fission yeast cells exhibited aberrant cellular morphology with a single enlarged and less compact nucleus. These cells also showed ongoing replication with no interference in the mitotic division, confirming that Rep is necessary and sufficient to induce replication in fission yeast (Kittelmann et al. 2009).
Human papillomavirus (HPV) and Bovine papillomavirus (BPV) are double-stranded DNA viruses that have been widely studied in yeast. HPV type16 full-length genome, when linked in cis to a selectable yeast marker, was reported to stably replicate in S. cerevisiae as an episome (Kim et al. 2005). Other viruses like BPV type1 and HPV6b, -11, -16, -18, and -31 were also reported to replicate in budding yeast; however, only for short-term (for about 20 cell doublings) (Angeletti et al. 2002). A single element rep for ARS (autonomous replicating sequence) and multiple elements mtc for CEN were reported in HPV16 genome. These sequences help in a replication similar to that of extrachromosomal plasmid in yeast. This replication was further reported to be independent of the DNA helicase, E1. The rep and mtc elements constitute about 1000-bp-long region and support replication of a bacterial-yeast shuttle plasmid lacking both ARS and CEN elements (Kim et al. 2005). Similarly, the ARS region in the EBV genome was also identified using S. cerevisiae (Lista et al. 2015).
Adeno-associated virus (AAV, family Parvoviridae) is a non-pathogenic double-stranded DNA virus used as a potential gene delivery vector for gene therapy studies (Vasileva and Jessberger 2005). Production of AAV vector can be hugely benefited by understanding AAV biology and yeast has been a useful tool to understand AAV and host interactions. Single-stranded recombinant AAV (rAAV) was first produced by co-transformation of the double stranded rAAV genome containing inverted terminal repeats (ITRs) and the plasmid expressing Rep68 in S. cerevisiae (Cervelli et al. 2011). Expression of AAV proteins like VP1, VP2, VP3, AAP, Rep78, Rep52, and an ITR-flanked DNA was found to be crucial for encapsidation of virion particles, viral genome replication, and production of infectious particles in S. cerevisiae (Barajas et al. 2017). Further studies in yeast also reported that the production of rAAV could be increased upon overexpression of TOP2 and HEM4, two host genes that code for topoisomerase and uroporphyrinogen synthase, respectively (Aponte-Ubillus et al. 2019).
Virus-host interaction
Viral protein and host protein interactions may either suppress or facilitate replication of the virus (Xu et al. 2014; Zhang et al. 2016b). Such interactions also assist in the localization of viral proteins to their specific site of action in the host cell (Varadarajan et al. 2005). If the expression of viral proteins leads to distinct phenotypes in yeast, a testable hypothesis regarding its function and role in pathogenesis can be formulated. Further, this can be tested both in yeast as well as the physiological host. Numerous studies have used yeast to identify host factors that interact with various viral proteins and elucidate their role in the viral replication cycle (Diaz et al. 2010; Sasvari et al. 2013; Weeks and Miller 2008) (Table 1). Interesting aspects of virus-host interactions studied using the yeast model are alterations in host protein and lipid composition upon expression of viral proteins, identification of regions/domain of viral proteins essential for interaction with host proteins, host sub-cellular membranes essential for viral replication, and host factors that aid and inhibit viral replication (Fig. 1).
RNA viruses
Several important aspects related to BMV replication have been uncovered using the yeast model. As mentioned above, BMV replication proteins 1a and 2a are essential for RNA replication and were reported to colocalize and accumulate at the yeast perinuclear endoplasmic reticulum (ER), the site of nascent viral RNA synthesis (Restrepo-Hartwig and Ahlquist 1999). Lee and colleagues reported that 1a expression in yeast resulted in 25–33% increase of total membrane lipids and in the induction of spherules that are enveloped by the host ER membrane (Lee and Ahlquist 2003). The role of the Lsm1p-Lsm7p/pat1p deadenylation-dependent mRNA-decapping factors in the translation process of BMV genomic RNA in S. cerevisiae was also reported (Noueiry et al. 2003). Mutational studies further reported that the helicase motif of 1a was essential for the viral RNA replication, spherule formation, and RNA recruitment to the replication sites (Wang et al. 2005). Wang and colleagues reported the role of the multivesicular body (MVB) proteins Doa4 and Bro1 in BMV RNA replication. Doa4 and Bro1 are required for recycling ubiquitin from MVB cargos, which regulates the expression of fatty acid desaturase Ole1 essential for BMV RNA replication (Wang et al. 2011). The role of RNA capping and helicase domains of BMV1a in RNA replication was studied in S. cerevisiae. BMV 1a RNA capping and helicase domains were expressed individually or together or in combination with full length 1a in yeast. 1a capping domain alone was reported to be sufficient for the perinuclear ER membrane targeting of the protein and inducing the formation of lattice arrayed hexagonal tubes. Interestingly, it was reported that the presence and interaction of both capping and helicase domains were necessary for the formation of spherical replication compartment and viral RNA replication (Diaz et al. 2012).
Zhang and colleagues analyzed the role of host phosphatidylcholine (PC) in BMV replication in S. cerevisiae. Expression of BMV1a protein and its interaction and recruitment of host Cho2 (choline recruiting 2) resulted in the accumulation of PC at the perinuclear ER membrane. In the Cho2 deletion strain, BMV replication was significantly inhibited and larger spherule formation was reported (Zhang et al. 2016b). Host factors responsible for BMV1a localization were identified by high content screening. Interaction of BMV1a with ER-vesicle protein Erv14, a cargo receptor of core protein complex 2, was reported in this study. Deletion of ERV14 led to the mislocalization of BMV1a and reduced the number of BMV spherules formed and led to inhibition of BMV replication (Li et al. 2016).
The localization of TBSV p33 protein to peroxisomes was identified using yeast cells. Interestingly, when TBSV p33 was expressed in cells lacking peroxisomes (pex3 and pex19), it was found to relocalize to the ER membrane, supporting the concept that viruses can utilize alternate intracellular membranes for their replication (Jonczyk et al. 2007). Yeast Pex19 was reported to bind and interact with p33 as shown by pull-down assay and co-purification experiments. Upon re-targeting Pex19 to mitochondria, a significant fraction of p33 was also found to localize to mitochondria, which resulted in reduced replicase activity leading to decreased TBSV RNA accumulation (Pathak et al. 2008). Expression of TBSV p33 and p92 in the double mutant ssa1ssa2 (Hsp70 family) resulted in cytosolic localization. Hsp70 was also reported to play a vital role in the assembly of viral replicase and insertion of the replication proteins in the peroxisomal membrane (Wang et al. 2009). A role for the ER resident protein Sec39 in the localization of TBSV p33 to peroxisomes was reported (Sasvari et al. 2013). In the absence of Sec39, p33 was mislocalized to the ER membrane or cytosol instead of peroxisomes and further led to inhibition of TBSV RNA accumulation at the replication compartments (Sasvari et al. 2013). A role for the yeast phosphatidic acid phosphohydrolase (PAH1) in TBSV replication was also reported. In PAH1 deleted yeast cells, increased RNA replication due to the presence of expanded ER membrane was reported (Chuang et al. 2014). Inhibition of TBSV-mediated spherule formation was reported in cells lacking the endosomal sorting complex required for transport (ESCRT) proteins Vps23 and Bro1 (Kovalev et al. 2016). Mutational studies identified the presence of the cholesterol recognition motifs CRAC and CARC in the transmembrane domains of p33 required for binding to sterol (Xu and Nagy 2017). Mutation in CRAC or CARC sequences in p33 resulted in inhibition of TBSV RNA synthesis, emphasizing that cellular sterols act as vital proviral lipids essential for TBSV replication (Xu and Nagy 2017). Rsp5, a member of the Nedd4 family of E3 ubiquitin ligases, was identified as a potential inhibitor of TBSV replication (Barajas et al. 2009). Rsp5 interacts with TBSV replication proteins p33 and p92 with its WW domain. Further analysis showed that the interaction of Rsp5 with p92 resulted in reduced p92 stability followed by degradation and resulted in the inhibition of TBSV replicase activity. In addition, increased TBSV RNA replication was reported in cells with downregulated Rsp5 (Barajas et al. 2009). Cns1, a co-chaperone for Hsp70 and Hsp90 that inhibits TBSV replication, was identified using a conserved domain search (Lin and Nagy 2013). Cns1 interacts with the TBSV replication proteins, making them unavailable for the assembly of TBSV replicase complex, thus leading to the inhibition of TBSV RNA synthesis (Lin and Nagy 2013). Recruitment of the host glycolytic pyruvate kinase into the TBSV viral replication compartment resulted in ATP depletion in the host cytosol (Chuang et al. 2017). Two host factors, Vps34 and phosphatidylinositol 3-kinase (PI3k), were reported to be recruited by TBSV into its viral replication compartment (Feng et al. 2019). A recent study highlights the role of TBSV p33 protein in the recruitment of Vps34 to the replication compartment (Feng et al. 2019).
Localization of GFP-tagged replicase proteins of CIRV to mitochondria was reported in yeast (Weber-Lotfi et al. 2002). The tetratricopeptide repeat (TPR) domain-containing Hop-like-stress-inducible-protein 1 (Hop/Sti1p) co-chaperone interacts with the RNA-binding domain of CIRV replication proteins and prevents CIRV RNA accumulation in the viral replication compartment (VRC). The deletion of STI1 in baker’s yeast resulted in a significant increase in CIRV replication (Xu et al. 2014). CIRV was also reported to use the ER membrane for replication, albeit not as efficiently as the mitochondrial membrane (Xu et al. 2014). FHV RNA replication was studied in S. cerevisiae cells lacking Hsp70 and Hsp90 chaperones. Lack of Hsp90 did not affect RNA replication, while deletion of YDJ1 the cochaperone of Hsp70 resulted in decreased RNA replication. This decreased RNA replication in cells lacking Ydj1 was not observed when the viral RNA polymerase and replication complexes were retargeted to the ER from mitochondria (Weeks and Miller 2008).
Interaction of HIV-1 Vpr with yeast nuclear transport factors, importin α subunit, and nucleoporins was reported (Vodicka et al. 1998). Studies in fission yeast reported that HIV Vpr interacts with nucleoporin Nup124, which aids in its nuclear localization and subsequently results in several cellular effects (Varadarajan et al. 2005). The import mechanism of HIV integrase (IN) protein into the host nucleus was studied in S. cerevisiae. Colocalization of GFP-tagged IN with host microtubule-associated proteins, Stu2 and Dyn2, was reported. These host factors are required for the transportation of IN to the spindle pole body (SPB), a perinuclear microtubule organizing center in yeast. A role of the C-terminal of IN in this localization was also reported (Desfarges et al. 2009). Interaction with the host importin α, and the presence of nuclear localization signal (NLS) on IN aid in its nuclear localization. Inhibition of nuclear import of IN was reported in temperature-sensitive importin α mutants (Levin et al. 2009).
DNA viruses
HPV type16, BPV type 1, and BPV 4 encode E5 protein that localizes to the vacuolar membrane in yeast and binds to the Vma3 (V-ATPase) subunit C. The activity of yeast V-ATPase remained unaltered irrespective of the binding of the viral protein (Ashby et al. 2001). Expression of the polyomavirus major structural protein VP1 in S. cerevisiae resulted in the production of VP1 pseudocapsids that contain fragments of host DNA and accumulate in yeast nuclei. Further analysis reported that VP1 strongly interacts with host tubulin fibers of the mitotic spindle and resulted in growth inhibition (Palkova et al. 2000).
Localization of viral proteins and effect on intracellular structures
Deciphering the localization of the viral protein in a cell can be instrumental in designing methodologies to target viral infection. Yeast as an alternative eukaryotic model to carry out localization studies for both plant and animal viruses has been reported (Zhao 2017) (Table 1). Viral proteins fused to an epitope tag such as GFP can be expressed in yeast to study their localization. Important insights and hints into the molecular mechanisms of the viral proteins can be obtained by their localization studies in yeast. This may further aid in understanding pathogenesis strategies of these viruses. Strikingly, several studies also report the altered structure of organelles upon expression of viral proteins (Glingston et al. 2019). Change in the morphology of yeast organelles can also provide clues for the function of the viral protein (Fig. 1).
RNA viruses
Localization of the BMV RNA replicating proteins 1a and 2a to the ER in both protoplasts of barley leaves and yeast was reported (Restrepo-Hartwig and Ahlquist 1996; Restrepo-Hartwig and Ahlquist 1999). 1a protein was reported to localize independently to perinuclear ER while 2a protein depends on 1a for its localization and interaction with Kar2, an ER resident protein (Restrepo-Hartwig and Ahlquist 1999). Deletion analysis indicated that 120 residues present in the N-terminal of 2a were required for its 1a dependent ER localization (Chen and Ahlquist 2000). Amino acid sequence 240–280 of 1a was identified to be sufficient for its localization to the ER (den Boon et al. 2001). Later mutational studies of 1a by Johan and colleagues identified the sequence in the protein responsible for its targeting to the ER in yeast and further emphasized the use of the yeast model for viral protein localization studies (den Boon et al. 2001). Cytopathic effects such as the increased formation of small spherules or large multilayer stacks of the double membrane layers of the outer ER membrane were reported as a result of the expression of 1a and 2a in yeast (Schwartz et al. 2004).
The nucleocapsid (N) and phosphoprotein (P) of Sonchus Yellow Net Nucleorhabdovirus, when expressed independently, were reported to be imported into the nucleus in both plants and yeast cells (Goodin et al. 2001). However, upon co-expression, subnuclear localization of both the proteins was observed (Goodin et al. 2001). Nuclear localization of the heterologously expressed influenza virus proteins NS1 and NS2 in yeast was also reported (Chua et al. 2019; Ward et al. 1995). The CymRSV protein p33 fused to GFP was expressed in yeast and was analyzed for its localization. Two transmembrane domains and a short hydrophilic region present in p33 were reported to play a critical role in its association with peroxisomes (Navarro et al. 2004). Similarly, the overlapping replication proteins p33 and p92 of the cucumber necrosis tombus virus (CNV) were also reported to be localized to the peroxisomal membrane (Panavas et al. 2005a). A predominant role for p33 in recruiting viral RNA and p92 to the replication site (the peroxisomal membrane) was proposed in this study. The peroxisomal localization of p33 and not p92 was dependent on the N-proximal transmembrane domain of the protein (Panavas et al. 2005a). However, the p33:p33/p92 interaction domain was reported to be important for the efficient localization of both p33 and p92 to the peroxisomal membrane (Panavas et al. 2005a). S. cerevisiae strain, which does not grow on oleate medium due to the absence of mature peroxisomes, was used as a model to study the localization of CymRSV proteins p33 and p92. In the absence of peroxisomes, p33 and p92 localized to the nuclear membrane and ER. A further analysis reported that this altered localization of p33 and p92 did not affect the viral RNA replication (Rubino et al. 2007).
The tombus viruses, CNV and TBSV, were reported to replicate inside spherules formed along the membrane contact sites of the ER membrane. For the construction of these replication sites, the virus was reported to hijack host oxysterol-binding and ER-resident Vap protein and subsequently translocate it to the membrane contact sites. This facilitated the channeling of lipids to the replication sites. Accumulation of sterol at the membrane contact sites, in turn, resulted in increased replication of the tombusvirus (Barajas et al. 2014). During TBSV replication in yeast, the host metabolic enzyme Tdh2 (homolog of plant glyceraldehyde 3 phosphate dehydrogenase) was found to relocalize from the cytosol to the peroxisomal membrane, which also served as a site for viral RNA synthesis (Wang and Nagy 2008). Upon expression of TBSV p33 in yeast, interaction with cellular cofilin (Cof1), an actin depolymerization factor, was reported. This interaction prevented Cof1 binding to actin filaments and affected the transport of cellular components (Nawaz-ul-Rehman et al. 2016). The integral membrane movement proteins of potexvirus, TGBp2 and TGBp3, were expressed individually in yeast and analyzed for localization (Lee et al. 2010). Localization of TGBp2 to perinuclear ER and TGBp3 majorly to the yeast cell periphery and perinuclear ER was reported. This localization of potexvirus proteins in yeast cells was similar to that in plant cells and hence, yeast was used to study intracellular trafficking of these proteins. On simultaneous expression, TGBp2 and TGBp3 formed a ~ 1:1 stoichiometric protein complex, which polymerized gradually, and resulted in the formation of punctate structures at the perinuclear ER. These traffic along the ER network and finally settle at the cortical ER or peripheral ER (Lee et al. 2010).
Upon expression of GFP-tagged 36K protein of CIRV in yeast cells, mitochondrial localization was reported. Altered mitochondrial morphology and number were reported in these cells. Mitochondria with poor or undeveloped cristae with strands of membranous material attached to them were observed (Rubino et al. 2000). Similarly, mitochondrial localization of the rubella virus capsid protein was reported in yeast. Further analysis reported that the capsid protein inhibits the translocation of precursor proteins into mitochondria and prevents the processing of precursor proteins (Ilkow et al. 2010). Miller and colleagues reported mitochondrial localization of FHV RNA-dependent RNA polymerase in S. cerevisiae (Miller and Ahlquist 2002).
Zhao and colleagues reported that the expression of HIV Vpr protein in fission yeast resulted in various morphological changes in cell organelles (Zhao et al. 1998). After 24 h of Vpr expression, cells showed aberrant nuclear architecture as visualized by DAPI staining. Yeast cells expressing Vpr reported nuclei with several ring-like structures and fragmented chromatin, which resulted in cell death. An effect on the cytoskeletal system of yeast was also reported in this study (Zhao et al. 1998). Vpr expression also resulted in delocalization of two spindle pole body proteins causing impairment of actin ring formation and cytokinesis in yeast cells (Chang et al. 2004). Externalization of phosphatidylserine, hyperpolarization of mitochondria, and alteration of mitochondrial membrane potential were also observed in these cells (Huard et al. 2008a). Nkeze and co-workers performed a detailed study on the subcellular localization of all HIV-1 proteins in fission yeast. HIV-1 proteins P24, P7, P66, P51, IN, Gp120, Gp41, Vpr, Vif, Rev, and Tat were predominantly localized to the nuclei of the fission yeast (Nkeze et al. 2015). P17 and Vpu were primarily localized to the cytoplasm. HIV-1 Gag precursor protein was reported to aggregate in cytoplasm while PR, P6, and Nef protein were found to be distributed throughout the cell (Nkeze et al. 2015). The effect of the expression of individual HIV-1 viral protein on growth or cytotoxicity of yeast cells was studied. Expression of Vpr, PR, and Rev in fission yeast cells resulted in inhibitory effect on growth (Nkeze et al. 2015). HIV-1 Rev contains both NLS and nuclear export signal (NES) required for nuclear localization and for transportation of partially spliced and unspliced viral transcripts between the nucleus and the cytoplasm (Nkeze et al. 2015). Additionally, HIV-1 Rev expression triggered transient reactive oxygen species (ROS) production in fission yeast cells, leading to oxidative stress (Nkeze et al. 2015).
Upon expression, Hepatitis C core protein p23 was processed to p21 and localized to both cytoplasm and the nucleus (Isoyama et al. 2002). Interaction of the HCV core protein with yeast nuclear import receptors was also identified in this study (Isoyama et al. 2002). Expression of HCV1 core proteins 1b and 2a in yeast cells inhibited cell growth and degradation of Lro1 (phospholipid: diacylglycerol acyltransferase) required for lipid droplet formation (Iwasa et al. 2016; Kubota et al. 2012). Altered localization of Lro1 to the site adjacent to lipid droplets was also reported in this study (Iwasa et al. 2016). HCV immature core 191 (aa 1-191) inhibited both ER-associated protein degradation (ERAD)-L and ERAD-M required for degrading misfolded/unfolded proteins in the ER lumen and membrane, respectively. On the other hand, HCV mature core 177 (aa 1-177) inhibited only ERAD-M and not ERAD-L. Hence, Takashi and colleagues suggested that HCV core 191 may play a role in the induction of UPR in yeast by inhibiting ERAD-L (Takahashi et al. 2017).
Li and colleagues performed an extensive analysis of Zika virus (ZIKV) proteins using fission yeast. The genome corresponding to each viral protein was expressed individually in fission yeast and the intracellular localization and cytopathic effects on host cells were analyzed in this study (Li et al. 2017). Various sub-cellular localizations such as nuclear membrane (anaC and C proteins), ER (M, NS2B, NS4A, and NS4B proteins), ER and cytoplasmic membranes (prM protein), ER and cytoplasm (E protein), Golgi and ER (NS2A), evenly in the cell (Pr, NS5, 2K, NS3 proteins), punctate structures in the cytoplasm (NS1), were reported. These findings have great potential to act as a reference for further studies in mammalian cells and for designing strategies for ZIKV treatment (Li et al. 2017). Recent studies also reported localization of Newcastle disease virus (NDV) M protein to the nucleus and NP to the vacuole upon expression in yeast. Furthermore, fragmentation of vacuoles upon expression of the viral proteins was reported in this study (Glingston et al. 2021). In cell lines, a role for the lysosome (equivalent of vacuoles) mediated autophagy process in NDV replication has also been reported (Cheng et al. 2016). Similar results were also observed upon expression of Hepatitis A virus 3C protease in S. cerevisiae. The authors report distortion and fragmentation of vacuole (Shubin et al. 2021).
DNA viruses
The Geminivirus Abutilon mosaic virus (Abmv) nuclear shuttle protein (NSP) and movement protein (MP) were expressed in S. pombe to understand their role in host interaction. Upon expression, MP was targeted to the protoplasmic face of the plasma membrane while NSP accumulated within the host nuclei. However, when these proteins were co-expressed, NSP was observed to be localized to the plasma membrane indicating the role of MP in the movement of NSP from the nucleus to the cell periphery (Frischmuth et al. 2007). ACMV encodes two variants of the AC4 protein, and both the variants were fused to either GFP or glutathione-S-transferase and expressed in fission yeast to analyze their localization (Hipp et al. 2016). Plasma membrane localization of the shorter AC4 variant was reported while the longer AC4 variant was localized as discrete spots in the cytoplasm (Hipp et al. 2016). Similar localization upon expression of both AC4 variants in plants was also reported (Hipp et al. 2016).
Biemans and colleagues used yeast as a model system to express major (S), middle (M), and large (L) envelope proteins of hepatitis B virus (HBV) (Ralph et al. 1991). Modifications such as glycosylation and amino-terminal myristoylation in the L protein were reported. Electron microscopy analysis of yeast cells expressing L protein revealed accumulation of membranous structures in the ER (Ralph et al. 1991). Adenoviral proteins E1B-55KDa and E4-34KDa were expressed in S. cerevisiae to study their effect on cellular metabolism. Overexpression of E1B-55KDa affected the host mRNA transport and resulted in growth inhibition. Additionally, E4-34KDa was required for the association of E1B-55KDa with the host nuclear inclusion bodies (Liang et al. 1995). HPV type 16 E7 phosphoprotein was reported to localize to the nucleus in S. pombe, similarly as in infected human keratinocytes (Tommasino et al. 1990).
The African swine fever virus (ASFV) protein p10 was expressed in S. cerevisiae and was reported to be actively imported into yeast nucleus via the amino acid stretch 71-77 in the protein (Nunes-Correia et al. 2008). Recently, S. cerevisiae was used as a model to express the mimivirus nucleotide translocator protein Vmc1 (Zara et al. 2018). It was reported that the newly synthesized Vmc1 binds to the yeast mitochondrial outer membrane protein Tom70. Subsequently, Vmc1 translocated to the mitochondrial inner membrane through the β-barrel protein Tom40 constituted the import channel. Vmc1 plays a major role in the replication of the large DNA genome of the virus by aiding translocation of mitochondrial deoxyribonucleotides from the mitochondrial matrix into the cytosol (Zara et al. 2018).
Although sometimes the expression of viral proteins may result in different responses when introduced into yeast and their physiological host cells, analogous molecular activities upon expression are also reported in many studies.
Cell cycle regulation
Viruses tend to interfere with the host cell cycle to establish an environment favoring their replication and assembly (Mui et al. 2010). They induce cell cycle arrest that decreases the competition for cellular resources and takes control of the host cell cycle (Elder et al. 2001). Almost all the major cell cycle regulators are conserved across eukaryotic organisms, making them obvious candidates to study with respect to cell cycle defects during viral replication (Wixon 2002). Cell cycle regulation by viral proteins of various viruses has been studied using yeast as a model organism (Gutierrez 2000; Jin et al. 1998; Li et al. 2017; Zhao et al. 1996) (Table 1). The phase of cell cycle arrest, the domains or region of viral proteins that are sufficient in driving cell cycle arrest, altered expression of host proteins that drive changes in cell cycle are some of the important topics addressed using yeast (Fig. 1).
RNA viruses
As mentioned in the above sections, HIV-1 Vpr is one of the most extensively studied viral proteins using yeast as a model. Several functions of Vpr that aid replication of the retrovirus have been reported (Zhao et al. 2011). These functions are highly conserved in fission yeast and human cells (Elder et al. 2001). Cellular alterations such as abnormal cell enlargement and growth arrest were reported upon expression of HIV Vpr in S. cerevisiae. The end of the third alpha helix of the Vpr protein, particularly the sequence HFRIGCRHSRIG as a causal factor for this, was reported (Janette M. Berglez et al. 1999; Macreadie et al. 1995). Expression of Vpr resulted in host cell cycle arrest in the G2 phase with decreased p34/Cdc2 kinase activity, leading to the growth arrest of fission yeast cells (Zhang et al. 1997; Zhao et al. 1996). This cell cycle arrest phenotype, which was observed in yeast, was identical to that of mammalian cells (Huard et al. 2008a). Additionally, HIV-1 Vpr was found to be more cytotoxic compared to other HIV proteins (Zhang et al. 1997). The C-terminal of the protein was sufficient to cause G2 arrest of the yeast cell cycle, whereas the N-terminal α-helix was required for its nuclear localization (Chen et al. 1999). Further, the host cellular factors wee1, ppa2, and rad24 that contribute to Vpr-induced G2 arrest were identified in fission yeast (Matsuda et al. 2006). Hyperphosphorylation of Tyr15 on Cdc2 and reduced kinase activity as a mechanism of Vpr-induced cell cycle G2 arrest in fission yeast cells was reported (Elder et al. 2000). Vpr induces the upregulation of protein-phosphatase 2A (PP2A) holoenzyme that interacts with Wee1 kinase and Cdc25 phosphatase. This interaction leads to inhibition of Cdc25 phosphatase and activation of Wee1, which causes Tyr15 hyperphosphorylation on Cdc2. Additionally, overexpression of Rad25 leads to inhibition of Cdc25 phosphatase and enhances Vpr induced G2 cell cycle arrest (Elder et al. 2001). A role for the C-terminal region of Vpr in growth arrest was also identified by mutation and deletion studies (Nakazawa et al. 2005). Mutational studies also reported that Vpr-induced G2 cell cycle arrest by inducing the Rad24/14-3-3 dependent overexpression and phosphorylation of Wee1 (Matsuda et al. 2006). Suppressors of Vpr-induced cell cycle arrest such as Hsp16 and Hsp70 were identified using genome wide studies (Benko et al. 2004; Iordanskiy et al. 2004). A role for the heat shock factor (Hsf1), which regulates Hsp16 and inhibits Vpr-induced cell cycle arrest, was reported. Interestingly, it was reported that Vpr counteracts enhanced expression of Hsp16 by regulating its transcription (Benko et al. 2007). Yeast kinase-mediated relocalization of Cdc25 from nucleus to the cytoplasm upon expression of Vpr was reported. Vpr interacts and activates Srk1 kinase, which phosphorylates Cdc25 at the serine/threonine sites. This, in turn, leads to the binding of Rad24/14-3-3 to Cdc25, which results in the export of Cdc25 from the nucleus to the cytoplasm. Subsequent inhibition of Cdc25 mediated dephosphorylation of Cdc2 results in hyperphosphorylation of Cdc2 and further results in G2 cell cycle arrest (Huard et al. 2008b).
Poliovirus is an RNA virus that encodes for eleven mature viral proteins. The non-structural protein 2BC of the virus induced host cell growth arrest in the G2 phase of the cell cycle. However, the expression of mature proteins 2B and 2C either individually or in combination did not affect yeast cells. The expression also resulted in the formation of small membranous vesicles in the yeast cell cytoplasm (Barco and Carrasco 1995a). These morphological changes are suggested to be similar to the cytopathic vacuoles observed in human cells upon poliovirus infection (Bienz et al. 1983; Dales et al. 1965).
Expression of human T lymphotropic type 1 Tax protein in S. cerevisiae resulted in cell cycle G2/M arrest or cell death (Liu et al. 2003). A strong reduction in the levels of host proteins Pds1 and Clb2 due to the expression of Tax was identified in this study. This resulted in mitotic defects leading to chromosomal instability followed by DNA aneuploidy and was reported in yeast, rodent, and human cells (Liu et al. 2003). Yeast cells expressing two integrated copies of human protein kinase R (PKR) under galactose-inducible promoter were further transformed with a plasmid containing Japanese encephalitis virus (JEV) NS2A cDNA. PKR expression was reported to arrest translation initiation and cell growth. On the expression of JEV NS2A in the above, PKR-mediated growth suppression of yeast cells in galactose medium was prevented (Tu et al. 2012).
The effect of expression of ZIKV proteins on fission yeast was analyzed by Li and colleagues (Li et al. 2017). Expression of seven ZIKV proteins, namely anaC, C, prM, M, E, NS2B, and NS4A, either suppressed or inhibited the formation of yeast colonies. Yeast cells expressing anaC, C, M, E, and NS2A exhibited decreased growth rate while prM and NS2B expressing cells displayed complete growth inhibition. prM expression resulted in G1 phase arrest while anaC, M, E, and NS4A expression resulted in the accumulation of cells at G2/M phase of the cell cycle. Additionally, NS4A was reported to influence the target of rapamycin (TOR) cellular stress pathway, which involves Tor1 and type 2A phosphatase activator Tip41 that arrests growth and induces cellular hypertrophy (Li et al. 2017). Similar cellular effects caused by the expression of NS4A were also reported in human cells (Liang et al. 2016).
DNA viruses
Upon expression of HPV-16 E2 in yeast, delayed activation of Cdc2 kinase resulting in delayed G2-M transition was reported. This phenotype was similar to that observed in higher eukaryotic cells (Fournier et al. 1999). The epstein-barr virus (EBV) Ser/Thr kinase BGLF4 was expressed in S. cerevisiae to analyze its function in cell cycle regulation. Cells expressing BGLF4 were not able to proliferate at non-permissive temperature, reinforcing that BGLF4 lacks Cdc28 activity required for inducing cell cycle progression in S. cerevisiae. The authors also reported a Cdk1-like activity for BGLF4 in yeast (Chang et al. 2012). Elongated cell morphology and cell cycle arrest in the G2/M phase were reported upon expression of Adenovirus E4orf4 protein in S. cerevisiae. A role for the PP2A regulatory B subunit CD55 in regulating E4orf4 activity was reported. However, an alternative Cd55 independent pathway utilized by E4orf4 for growth suppression was also reported (Kornitzer et al. 2001; Roopchand et al. 2001). Further studies suggested that E4orf4 interacts with the Cdc55 subunit of PP2A and uncouples PP2ACdc55 resulting in abnormal activation of the Cdc20 form of the anaphase-promoting complex (APC) in budding yeast. This leads to the degradation of specific APC substrates and defective mitotic exit (Mui et al. 2010).
Upon expression of Simian virus 40 tumor (T) antigen in S. cerevisiae, interaction with proteins that regulate cell cycle G1 to S transition was reported. This induced morphological alterations and resulted in growth arrest. Further evidence for this interaction was obtained when T antigen co-immunoprecipitated with p34CDC28in yeast cells (Nacht et al. 1995). The carboxy-terminal 150 aa of SV40 T antigen was reported to induce morphological changes and cell cycle arrest (Fewell et al. 2002).
Cell death (apoptosis and autophagy)
Some viruses are reported to inhibit apoptosis to prolong the survival of the host cells and facilitate viral replication or maintain persistent infection (Orzalli and Kagan 2017; Upton and Chan 2014). On the other hand, some are involved in inducing cell death (Zhao and Elder 2005). Yeast serves as an excellent model to decipher the molecular details of such mechanisms utilized by viruses.
Yeast phenotypes associated with apoptosis and autophagy such as cell growth inhibition, mitochondrial depolarization and abnormal structure, increased cellular ROS, etc. are used to study the role of viruses in cell death (Fig. 1).
RNA viruses
The replication complex of the positive-strand RNA plant virus CIRV is targeted to the outer mitochondrial membrane in both plant and yeast cells. This targeting of the replication complex is facilitated by the CIRV ORF1 that encodes for p36 protein. Expression of p36 in yeast cells does not lead to cell death but reduces the growth rate. However, in the presence of acetic acid, abnormal mitochondria and necrotic cell death instead of apoptosis were reported in these cells (Rubino et al. 2017). Poliovirus encoded proteases 2Apro and 3Cpro are required for the processing of viral polyprotein and also cleave a number of host proteins and lead to high degree of toxicity in yeast cells. Growth arrest was reported in S. cerevisiae upon expression of these proteases. Electron-dense granules and autophagosomic bodies were observed in Apro-expressing yeast cells. Inhibition of host transcription and not translation by 2Apro due to protease toxicity was also reported (Barco and Carrasco 1995b).
Huard and colleagues developed and validated a fission yeast model system to study Vpr-mediated apoptosis. Vpr expression caused mitochondrial hyperpolarization resulting in mitochondrial membrane potential alteration, ROS production, and phosphatidylserine externalization. These morphological changes were similar to that of apoptosis in mammalian cells. Additionally, all three Vpr suppressors, EF2, Hsp16, and Skp1, inhibited Vpr-mediated cell death in mammalian cells, restored mitochondrial morphology, and prevented Vpr-mediated cell death in fission yeast (Huard et al. 2008a). To study Vpr-induced oxidative stress response in fission yeast at a molecular level, a mutant of Vpr (F34IVpr) capable of inducing cell cycle arrest but not cell death was analyzed. Enhanced concentration of superoxide, peroxide and downregulation of glutathione, hydroxyl radical concentrations were reported. Expression of F34IVpr also resulted in reduced activity of antioxidant enzymes in S. pombe (Stromajer-Racz et al. 2010).
Expression of HIV-1 Pr resulted in cell growth arrest, destruction of the cell wall, disruption of the actin cytoskeleton, and loss of plasma membrane integrity leading to cell lysis in S. cerevisiae, (Blanco et al. 2003). Similar cytopathic effects were also observed in mammalian cells (Blanco et al. 2003). Expression of Pr also induced ROS production and caused alterations in mitochondrial morphology leading to apoptosis in fission yeast (Benko et al. 2016). Genome-wide screens identified a serine/threonine kinase Hhp2 that downregulates Pr activity and prevents cell death and protease cleavage in fission yeast (Benko et al. 2016). Expression of HIV integrase resulted in a lethal phenotype in S. cerevisiae (Caumont et al. 1996). The catalytic domain of HIV-1 IN binds to the host chromatin and results in the induction of lethal phenotype in S. cerevisiae (Xu et al. 2008). Myristoylated N-terminal peptides of HIV Nef were found to cause cytotoxic effects in human lymphocytes and red blood cells (Curtain et al. 1997). This effect was also reported in yeast cells where expression of Nef caused cell membrane permeabilization leading to cell death (Macreadie et al. 1997).
Structural biology studies revealed that tetravirus procapsids undergo acid-dependent maturation in vitro in insect cell lines (Canady et al. 2000). To elucidate the significance of this acid-dependent maturation process, tetravirus Helicoverpa armigera stunt virus’s capsid coding sequence was expressed in S. cerevisiae. The maturation of procapsids in cells with accumulated hydrogen peroxide or acetic acid was reported. This cytosolic acidification induced by the virus may further lead to programmed cell death in S. cerevisiae (Tomasicchio et al. 2007). A systematic study where individual ZIKV proteins were expressed and studied for their effects identified seven ZIKV proteins in fission yeast that caused restricted cellular growth, triggered cellular autophagy, induced hypertrophy, and cellular oxidative stress associated with cell death (Li et al. 2017). Expression of five structural proteins (anaC, C, prM, M, and E), and two non-structural proteins (NS2B and NS4A) resulted in cytopathic effects that subsequently lead to cell death in fission yeast (Li et al. 2017).
DNA viruses
Adenovirus E4orf4 is known to induce PP2A-dependent apoptosis in mammalian cells and irreversible growth arrest in yeast (Afifi et al. 2001; Kornitzer et al. 2001). Using yeast deletion library as a tool, the host factor involved in Adenovirus E4orf4-mediated apoptosis was identified. YND1 that encodes yeast Golgi apyrase interacts with Cd55, and this interaction was reported to be disrupted in the presence of E40rf4. E4orf4 interacts with the cytosolic tail of Ynd1 and results in apoptosis (Maoz et al. 2005; Mittelman et al. 2010). On the other hand, E40rf4-induced cell toxicity was identified to be independent of the enzymatic activity of Ynd1 (Maoz et al. 2005). S. cerevisiae was used to identify non-apoptotic E4orf4 mutants that have a limited capacity to bind to PP2A (Afifi et al. 2001).
Poxviruses have double-stranded DNA as their genetic material and encode numerous proteins known to inhibit apoptosis (Banadyga et al. 2011). Yeast S. cerevisiae was used to construct a caspase inhibition assay to analyze the role of the viral protein in preventing Apaf-1 activated apoptosis. Expression of Apaf1, along with caspase-9 and caspase-3, leads to cell death in this assay unless a caspase inhibitor is overexpressed (Hawkins et al. 2001). Using the above assay, it was reported that vaccinia virus (belonging to poxvirus family) F1L does not prevent cell death in yeast, indicating its inability to prevent the Apaf-1 activated apoptosis pathway (Caria et al. 2016). Similar assays were used to validate the antiapoptotic property of the vaccinia virus M1 protein. M1 prevents Apaf1/procapsase9/procapsase3 expression mediated cell death in S. cerevisiae. Interestingly upon overexpression of active caspase 9 and procaspase 3, M1 inhibited cell death in Apaf1-independent apoptotic pathway. The authors suggest that M1 inhibited cell death at the apoptosome level, where it interacts with active capsase9, and prevents lethality in yeast cells (Ryerson et al. 2017). Deerpox virus genome codes for the protein DPV022. This protein shares regions of amino acid identity with poxvirus apoptotic inhibitors M11L and F1L (Afonso et al. 2005). Upon expression in S. cerevisiae, DPV022 interacts with Bak and Bax and prevents their conformational activation resulting in inhibition of apoptosis (Banadyga et al. 2011).
Genome-wide studies
Several higher eukaryotic model systems may not be very sutiable for high-throughput screening of mutants, inhibitors, and interacting proteins. Yeast (S. cerevisiae and S. pombe), on the other hand, is an ideal model for such studies given its relatively small genome and very well-developed functional genomic tools. Several studies have utilized this feature and identified pathways and processes regulated by specific genes/proteins. This can further provide insights into the phenotype's etiology. Genome-wide screens and proteomic approaches have been instrumental in identifying host proteins involved in viral replication (Fig. 2). For instance, both replication inhibitory and stimulatory proteins of a wide range of RNA viruses like BMV, TBSV, etc. have been identified using high throughput screening techniques in yeast (Kushner et al. 2003; Serva and Nagy 2006; Shah Nawaz-ul-Rehman et al. 2012).
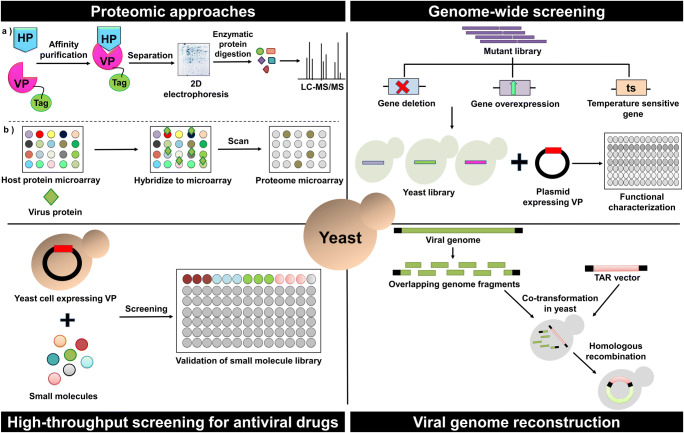
Fig. 2.
Yeast as a tool for high-throughput and rapid viral studies. An important advantage of using yeast for virus studies is the possibility of high-throughput studies. Expression of the viral protein with a tag in yeast cells and fishing out interacting partners using affinity-based approaches and subsequent identification of the host proteins using 2D gel electrophoresis and mass spectrometry is a commonly used proteomic approach. Host protein microarrays are used to identify the interacting partners that hybridize with the viral protein in another approach. Virus-host protein interactions can also be studied with the help of yeast genome libraries such as deletion library, over-expression library and ts library. Upon transformation of virus protein expressing plasmid into individual yeast strain of the library, phenotypic effects such as growth can be evaluated. Further functional characterization using microscopy, cell survival, etc. can also be performed. Screening of small molecule libraries with antiviral properties that can target virus protein expressing yeast cells is an efficient way of identifying antiviral molecules. Yeast can also be used as a tool for the reconstruction of whole viral genomes that can be valuable in rapid studies of the entire viral genome. Overlapping homologous fragments of the viral genome together with a vector are transformed into yeast and recombination within the cell leads to the expression of the entire viral genome. VP viral protein, HP host protein, ts temperature sensitive, TAR targeted amplified region
The yeast deletion library and luciferase reporter system were used to identify host factors involved in BMV RNA replication. Each strain of the deletion library was transformed to express the BMV RNA template and the BMV replicase. In order to measure the altered levels of viral replication, the BMV capsid gene in the RNA replication template was replaced with the luciferase reporter. Since the expression of luciferase reporter was dependent on the viral RNA replication and RNA-dependent mRNA synthesis, measurement of luciferase expression was utilized for quantitative analysis of viral RNA replication. This functional genomic approach was used to screen 4500 yeast deletion strains and more than 100 deletion strains showed significant alteration in the replication of BMV RNA (Kushner et al. 2003). The mutational analysis also led to the identification of several host proteins like Ded1, Ydj1 required for BMV replication (Noueiry et al. 2000; Tomita et al. 2003). Zhu and colleagues used S. cerevisiae protein arrays of approximately 5000 proteins to identify host proteins that bind specifically to a small BMV RNA hairpin. The small RNA hairpin was found to contain a motif called clamped adenine motif necessary for BMV replication (Zhu et al. 2007). An interesting study by Diaz and colleagues identified the role of reticulon homology proteins (RHPs) in BMV RNA replication. In yeast cells lacking RHPs, the localization of BMV1a to perinuclear ER and recruitment of BMV2a were not altered, but the viral replicase compartment formation was inhibited. Further, the authors reported that the interaction between RHPs and BMV1a protein was essential for the formation of the replication compartment (Diaz et al. 2010).
Host proteins that interact with CNV replication proteins were identified using a proteomics approach. Flag/six-His-tagged CNV p33 and p92 replication proteins along with DI-72 RNA replicon were expressed in S. cerevisiae. The CNV replicase complex formed in these yeast cells was isolated using two-step affinity purification. The obtained proteins were further subjected to two-dimensional electrophoresis, followed by mass spectrometry analysis (Serva and Nagy 2006). Host proteins such as the molecular chaperones Ssa1/Ssa2, Tdh2/3, and Pdc1 were identified in this study. Further analysis using double mutant strain ssa1ssa2 resulted in severe downregulation of CNV replicase activity, leading to reduced viral RNA accumulation. On the other hand, an approximately 3-fold increase in the viral replication was observed in yeast strains overexpressing either Ssa1 or Ssa2 (Serva and Nagy 2006). Li and colleagues used a yeast proteome microarray of 4080 yeast proteins to identify host proteins that interact with the viral RNA of TBSV and BMV. Proteins that selectively interact with TBSV RNA and BMV RNA, respectively and with both the viral RNA were identified in this study. Further analysis of interacting proteins revealed that 14 of these exhibited inhibitory effect while 4 enhanced TBSV replication (Li et al. 2009). The yeast ORF overexpression library was used to identify the host genes that play a role in TBSV replication. Host proteins that increased (40 host proteins) or decreased (101 host proteins) accumulation of TBSV replicon RNA were identified. Protein kinase C was identified as an inhibitor responsible for the downregulation of TBSV replication in this study (Shah Nawaz-ul-Rehman et al. 2012). Several other host factors such as Cpr1p, Ess1p, Wwm1, and Prp40 were also identified to act as potential inhibitors of TBSV replication (Mendu et al. 2010; Qin et al. 2012). In further studies, a temperature-sensitive yeast mutant library of essential genes was used to identify 118 host proteins that influence the replication of TBSV. Out of the 118, 72 proteins were found to facilitate TBSV replication and 46 inhibited it (Nawaz-ul-Rehman et al. 2013).
Sir2 as a suppressor of Middle East respiratory syndrome corona virus (MERS-CoV) ORF4a was identified using a suppressor screen and slow growth phenotype. Homolog of Sir2, Sirt1 in mammalian cells was also identified as a proviral factor for the replication of MERS-CoV. Upon inhibition of SIRT1 reduced MERS-CoV replication, was reported (Weston et al. 2019). Recently, a synthetic genomic platform was constructed for studying SARS-CoV-2 (Thao et al. 2020). Transformation-associated recombination (TAR) cloning technique for large RNA viruses was developed in S. cerevisiae in this study. This method aids in the rapid construction of the genome of the virus and is versatile and can be used to study the molecular biology and pathogenesis of novel RNA viruses (Fig. 2).
Yeast: a tool for vaccine development
The use of yeast as a system to produce viral vaccine dates back to 1982 when surface antigen of HBV was synthesized in S. cerevisiae and the protein secreted by yeast was reported to have properties similar to 22 nm particles produced by human cells (Valenzuela et al. 1982). The vaccine was first approved in 1986 and was the first licensed vaccine of any kind to be produced by recombinant technology. Several other vaccines were produced later using this methodology (Hilleman 1987). Since then, many yeast strains have been engineered, which serve as expression and screening systems to produce safe and effective recombinant antiviral drugs (Table 2) (Roldao et al. 2010).
Table 2.
An overview of vaccines produced using various yeast models
| S. No | Yeast model | Virus | Vaccine | Reference |
|---|---|---|---|---|
| 1 | Saccharomyces cerevisiae | Coxsackie virus |
VLP P1 and 3CD |
Zhao et al. 2013 |
|
VLP ChiEV-A71 |
Zhao et al. 2015 | |||
| Dengue virus |
PP Envelope domain III (scEDIII) |
Nguyen et al. 2013 | ||
| Enterovirus 71 |
VLP EV71 structural protein |
Li et al. 2013 | ||
|
VLP ChiEV-A71 |
Zhao et al. 2015 | |||
| Hepatitis B virus |
PP Hepatitis B surface antigen |
Jacobs et al. 1989 | ||
|
VLP HBsAg |
Pleckaityte et al. 2015 | |||
| Hepatitis C virus |
WRY NS3-Core fusion protein |
Haller et al. 2007 | ||
| HIV-1 |
YSD Envelope (Env) glycoprotein |
Wang et al. 2018 | ||
| Japanese encephalitis virus |
YSD Envelope protein (mannoprotein) |
Upadhyaya and Manjunath 2009 | ||
| New castle disease virus |
PP Hemagglutinin-neuraminidase |
Khulape et al. 2015 | ||
| Papillomavirus |
PP L1 protein |
Park et al. 2008 | ||
|
VLP Capsid proteins L1 or L1 plus L2 |
Lowe et al. 1997 | |||
|
PP Recombinant capsid protein |
Kim et al. 2014 | |||
| Parvovirus B19 |
VLP VP1 and VP2 |
Penkert et al. 2017 | ||
| Polyoma virus |
YCP VP1, a major capsid protein |
Tipper and Szomolanyi-Tsuda 2016 | ||
| Porcine circovirus type 2 |
VLP Capsid protein |
Chen et al. 2018 | ||
|
Red-spotted grouper NNV (RGNNV)/Nervous necrosis |
WRY Capsid protein |
Nguyet et al. 2013 | ||
| 2 | Pichia pastoris | Avian influenza virus |
PP Influenza A/H5N1 Neuraminidase |
Yongkiettrakul et al. 2009 |
| Avian leucosis virus |
PP ALV-J gp85 protein |
Jing et al. 2018 | ||
| Bursal disease virus (IBDV) |
PP VP2 gene of the Edgar strain of IBDV, hypervariable region of the VP2 gene (hvVP2) |
Villegas et al. 2008 | ||
| Chikungunia virus |
VLP Structural polyprotein |
Saraswat et al. 2016 | ||
| Classical swine fever virus |
PP Glycoprotein E2 |
Cheng et al. 2014 | ||
| Coxsackie virus A |
VLP P1 and 3CD proteins of CA6 |
Zhou et al. 2016 | ||
|
VLP P1 and 3CD proteins of CA16 |
Zhang et al. 2016a | |||
| Dengue virus |
VLP Envelope Glycoprotein |
Khetarpal et al. 2017 | ||
|
VLP Envelope protein domain III (EDIII) |
Ramasamy et al. 2018 | |||
|
VLP Envelope protein (E1-E4) |
Rajpoot et al. 2018 | |||
| Hepatitis B virus |
PP Recombinant hepatitis B surface antigen |
Hardy et al. 2000 | ||
| Hepatitis C virus |
PP Core E1E2 protein |
Fazlalipour et al. 2015 | ||
|
VLP Core protein (HCcAg) |
Acosta-Rivero et al. 2001 | |||
| Human papillomavirus |
VLP L1 proteins (16L1 and 18L1) |
Gupta et al. 2017 | ||
|
PP HPV type 16 L1-L2 Chimeric SAF protein |
Bredell et al. 2018 | |||
| Influenza virus |
PP Hemagglutinin (HA) protein |
Wang et al. 2019 | ||
|
YSD Alpha agglutinin |
Wasilenko et al. 2010 | |||
|
PP H5 haemagglutinin |
Pietrzak et al. 2016 | |||
| Norovirus |
VLP recombinant capsid protein |
Xia et al. 2007 | ||
| SARS-CoV |
PP Recombinant receptor binding domain of Spike protein |
Chen et al. 2020 | ||
| SARS-CoV-2 |
PP Recombinant receptor binding protein |
Pollet et al. 2020 | ||
| 3 | Hansenula polymorpha | Bovine viral diarrhoea virus |
VLP Chimera of membrane integral small surface protein (dS) of the duck hepatitis B virus fused with E2-BVDV |
Wetzel et al. 2018 |
| Classical swine fever virus |
VLP Chimera of membrane integral small surface protein (dS) of the duck hepatitis B virus fused with E2-CFSV |
Wetzel et al. 2018 | ||
| Feline leukemia virus |
VLP Chimera of membrane integral small surface protein (dS) of the duck hepatitis B virus fused with FeLVenv |
Wetzel et al. 2018 | ||
| West nile virus |
VLP Chimera of membrane integral small surface protein (dS) of the duck hepatitis B virus fused with WNVE |
Wetzel et al. 2018 | ||
| Hepatitis B virus |
PP VrHB-IB |
Caetano et al. 2017 | ||
|
PP Recombinant HBsAg |
Caetano et al. 2017 | |||
| Human Papillomavirus |
VLP Type 52 L1 Protein |
Liu et al. 2015 | ||
| Porcine circovirus type 2b |
VLP NLS-deleted capsid protein (ΔCP) |
Xiao et al. 2018 | ||
| 4 | Schizosaccharomyces pombe | Human papillomavirus type 6 & 16 |
VLP Capsid protein |
Sasagawa et al. 1995 |
| 5 | Kluyveromyces marxianus | Porcine circovirus type 2 |
VLP Capsid protein |
Duan et al. 2019 |
| 6 | Yarrowia lipolytica | Red spotted grouper Nervous necrosis virus |
VLP Capsid protein |
Luu et al. 2017 |
The table lists various yeast models such as S. cerevisiae, P. pastoris, H. polymorpha, K. lactis, S. pombe extensively used for the expression and production of vaccines. PP purified protein, VLP virus-like particle, WRY whole recombinant yeast, YSD yeast surface display
Pichia pastoris
Methylotrophic yeast P. pastoris is one of the most widely used expression systems for antiviral drug production in laboratory research and industrial production. (Vogl et al. 2013). The last year has witnessed one of the most severe pandemics caused by SARS-CoV2, and a need for the production of an effective vaccine was an emergency. The receptor-binding domain (RBD) protein of SARS-CoV-2 was expressed at high levels in P. pastoris and was also proposed to be a suitable vaccine candidate against COVID-19. To reduce the hyperglycosylation of the protein as a result of expression in yeast, modifications were made in the wild-type RBD gene that further improved the stability of the expressed protein. (Pollet et al. 2020). Similarly, vaccine candidates for SARS-CoV have also been developed using P. pastoris (Chen et al. 2014; Chen et al. 2020). Receptor-binding domain (RBD219-N1) of the SARS-CoV spike (S) protein adjuvanted with aluminum hydroxide (Alhydrogel®) was shown to be a promising candidate in this study (Chen et al. 2020). A universal influenza vaccine reported to combat the high mutation rate of the virus was developed using P. pastoris. To achieve this, a highly conserved stem region of hemagglutinin (HA) was used and immunogenic HA-stem-based protein was produced in P. pastoris. This yeast model was also used to achieve large scale production of recombinant influenza HA protein with a yield of 100 mg/L in a fermenter process (Wang et al. 2019). Large-scale production of recombinant HBV surface antigen, consequently producing a safe and effective vaccine against HBV infection (HEBERBIOVAC HB, Heberbiotec S. A, Cuba) was also reported (Hardy et al. 2000). For the production of virus-like particle (VLP)-based vaccine against a global threat, chikungunya virus (CHIKV), expression systems such as baculovirus and mammalian cells were screened. These systems, however, had disadvantages like contamination of baculovirus particles and high cost of production. P. pastoris was used as the expression system to overcome these disadvantages and to produce contamination free and highly specific vaccine at cheaper operating costs (Saraswat et al. 2016). Other VLP-based vaccines derived from P. pastoris include viruses like HPV (Gupta et al. 2017), norovirus (Xia et al. 2007), and dengue virus (Rajpoot et al. 2018) (Table 2).
Saccharomyces cerevisiae
S. cerevisiae is the most well-understood eukaryote at the molecular level. As mentioned above, the first antiviral vaccine against hepatitis B was made in the host S. cerevisiae. Since then, it has been used extensively in virus research for the production of antiviral drugs. S. cerevisiae has been used for the production of VLPs against viruses like coxsackievirus (Zhao et al. 2013) and enterovirus 71 both of which are leading causes of hand-mouth-foot disease (Li et al. 2013). It has also been used in the production of vaccine against Porcine circovirus type 2 (PCV2), which poses a big threat to pig industry. Recently, YeastFab assembly was used to produce VLP from PCV2 cap protein (Chen et al. 2018). YeastFab assembly is a term used for protocols demonstrated for efficient synthesis of standardized biological parts (genetic elements) of S. cerevisiae, which can be assembled into transcriptional units in one tube reaction (Guo et al. 2015). Apart from being used as an expression system, yeast has also been exploited as an agent for drug delivery. Figueiredo and colleagues demonstrated that yeast cell wall particles (YCWP) are promising nature-inspired microcarriers. Yeast cells have β-1,3-D-glucan (βG) on their outer surface, which is readily taken up by various phenotypes of APCs due to the dectin-1 receptor present on their membrane (Figueiredo et al. 2011). Upon removal of the layer of mannoprotein using various methods, the underlying layer of βG is exposed. This approach was used to produce YCWP vaccine against polyomavirus (Tipper and Szomolanyi-Tsuda 2016).
S. cerevisiae is also used as a tool to screen specific small molecules that can be used as antivirals. The slow growth of yeast as a response to viral protein expression has been utilized in such screens. Small molecules that can specifically target proteins of influenza virus and SARS-CoV have been identified using the above strategy (Basu et al. 2009; Frieman et al. 2011).
Other yeasts
The methylotrophic yeast H. polymorpha is a widely used expression system for producing hormones, antigens and enzymes such as Hep-B VLP-based vaccine, insulin, hirudin, and phytase (Celik and Calik 2012; Wetzel et al. 2018). It is very well suited for large-scale production because of the high yields and tolerance to very high cell density. It can also efficiently secrete proteins of upto 150 kDa, which makes it suitable for the production of a wide range of biopharmaceuticals (Celik and Calik 2012). Recently, it was used for the production of VLPs to be used as a platform for antigen presentation. In this study, membrane integral small surface protein (dS) of duck HBV was taken as scaffold VLP. The antigens to be presented were fused with dS and recombinant production strains of H. polymorpha were isolated. Eight different antigen molecules were chosen from four different viruses and chimeric VLPs co-producing dS and foreign antigen were successfully obtained. This yeast-based system was reported to be cost effective, can be scaled up, and used for presenting a variety of antigens with large molecular weight (Wetzel et al. 2018).
Fission yeast S. pombe is extensively used to study the viral replication cycle, as mentioned in the above sections (Takegawa et al. 2009). Its utility in the production of HPV type 16 vaccine and as a platform for high throughput screening of drugs has been reported (Sasagawa et al. 1995). Kluyveromyces marxianus is a non-conventional yeast recently reported to be a low-cost expression system for VLP formation against PCV2. Under certain fermenter conditions, the yield was reported to be more than other expression systems, making it a potential host system for the development of vaccines in the future (Duan et al. 2019). Yarrowia lipolytica is another non-conventional yeast where the glycosylation pattern of the expressed proteins is closer to the mammalian high-mannose type of glycosylation (Oh et al. 2016; Song et al. 2007). It was recently used for the expression of viral capsid proteins of nervous necrosis virus of fish as VLP based recombinant vaccines (Luu et al. 2017).
Concluding remarks
Yeast is one of the most widely used eukaryotic model organism to investigate several important research questions. Cell cycle, metabolism, apoptosis, regulation of gene expression, and signal transduction are a few cellular processes studied using yeast (Karathia et al. 2011). Such conserved eukaryotic cellular processes are often observed to be the target of viruses and hence validate the use of yeast for virus studies. In recent years, several important discoveries related to the life cycle of viruses have been made using yeast as study models. Viruses with both RNA and DNA genomes have been studied in yeast models and these have helped in understanding various aspects related to viral replication cycle and host-virus interactions.
Additionally, tremendous recent developments in the tools to analyze whole genomes and single cells make yeast an ideal choice for several basic biology research questions. However, the differences between the native virus host and yeast cell needs to be understood and results validated. Gaining insights into the functions of individual viral proteins, their structural and functional characterization, interactions and effects on the host cell, applications in biotechnology, including development of new antiviral methods are few aspects where contributions of yeast model are extremely valuable. Recently, an alternative yeast-based reverse genetics platform to genetically reconstruct SARS-CoV-2 was developed. Chemically synthesized clones of SARS-CoV-2 were generated using this method in a very short time, and this method will prove advantageous to tackle such outbreaks in future (Thao et al. 2020). Once described as an odd couple, the amount of research carried out using these two organisms may not make them the best couple but surely suitable for each other when the right questions are asked. Yeast will certainly play a key role in deciphering some of the viral mysteries yet to be unfolded.
Acknowledgements
Our sincere apologies to all our colleagues for any publication not being cited which is due to space limitation. This work was supported by Department of Biotechnology (DBT), Government of India [BT/PR25097/NER/95/1013/2017] and Top-up of start up grant from IITG.
Author contribution
SGR, JY, JR, and NJ wrote the manuscript. SN designed the structure, edited, and revised the manuscript. All authors read and approved the manuscript.
Declarations
Ethics approval and consent to participate
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no conflict of interest.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Acosta-Rivero N, Aguilar JC, Musacchio A, Falcón V, Vina A, de la Rosa MC, Morales J (2001) Characterization of the HCV core virus-like particles produced in the methylotrophic yeast Pichia pastoris. Biochem Biophys Res Commun 287(1):122–125. 10.1006/bbrc.2001.5561 [DOI] [PubMed]
- Advani VM, Dinman JD. Reprogramming the genetic code: The emerging role of ribosomal frameshifting in regulating cellular gene expression. Bioessays. 2016;38(1):21–26. doi: 10.1002/bies.201500131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Afifi R, Sharf R, Shtrichman R, Kleinberger T (2001) Selection of apoptosis-deficient adenovirus E4orf4 mutants in Saccharomyces cerevisiae. J Virol 75(9):4444–4447. 10.1128/JVI.75.9.4444-4447.2001 [DOI] [PMC free article] [PubMed]
- Afonso CL, Delhon G, Tulman ER, Lu Z, Zsak A, Becerra VM, Zsak L, Kutish GF, Rock DL. Genome of deerpox virus. J Virol. 2005;79(2):966–977. doi: 10.1128/JVI.79.2.966-977.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alves-Rodrigues I, Galao RP, Meyerhans A, Diez J (2006) Saccharomyces cerevisiae: a useful model host to study fundamental biology of viral replication. Virus Res 120(1-2):49–56. 10.1016/j.virusres.2005.11.018 [DOI] [PMC free article] [PubMed]
- Angeletti PC, Kim K, Fernandes FJ, Lambert PF (2002) Stable replication of papillomavirus genomes in Saccharomyces cerevisiae. J Virol 76(7):3350–3358. 10.1128/jvi.76.7.3350-3358.2002 [DOI] [PMC free article] [PubMed]
- Aponte-Ubillus JJ, Barajas D, Peltier J, Bardliving C, Shamlou P, Gold D. A rAAV2-producing yeast screening model to identify host proteins enhancing rAAV DNA replication and vector yield. Biotechnol Prog. 2019;35(1):e2725. doi: 10.1002/btpr.2725. [DOI] [PubMed] [Google Scholar]
- Ashby AD, Meagher L, Campo MS, Finbow ME. E5 transforming proteins of papillomaviruses do not disturb the activity of the vacuolar H(+)-ATPase. J Gen Virol. 2001;82(10):2353–2362. doi: 10.1099/0022-1317-82-10-2353. [DOI] [PubMed] [Google Scholar]
- Banadyga L, Lam SC, Okamoto T, Kvansakul M, Huang DC, Barry M. Deerpox virus encodes an inhibitor of apoptosis that regulates Bak and Bax. J Virol. 2011;85(5):1922–1934. doi: 10.1128/JVI.01959-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barajas D, Li Z, Nagy PD. The Nedd4-type Rsp5p ubiquitin ligase inhibits tombusvirus replication by regulating degradation of the p92 replication protein and decreasing the activity of the tombusvirus replicase. J Virol. 2009;83(22):11751–11764. doi: 10.1128/JVI.00789-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barajas D, Xu K, de Castro Martin IF, Sasvari Z, Brandizzi F, Risco C, Nagy PD. Co-opted oxysterol-binding ORP and VAP proteins channel sterols to RNA virus replication sites via membrane contact sites. PLoS Pathog. 2014;10(10):e1004388. doi: 10.1371/journal.ppat.1004388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barajas D, Aponte-Ubillus JJ, Akeefe H, Cinek T, Peltier J, Gold D (2017) Generation of infectious recombinant Adeno-associated virus in Saccharomyces cerevisiae. PLoS One 12(3):e0173010. 10.1371/journal.pone.0173010 [DOI] [PMC free article] [PubMed]
- Barco A, Carrasco L. A human virus protein, poliovirus protein 2BC, induces membrane proliferation and blocks the exocytic pathway in the yeast Saccharomyces cerevisiae. EMBO J. 1995;14(14):3349–3364. doi: 10.1002/j.1460-2075.1995.tb07341.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barco A, Carrasco L. Poliovirus 2Apro expression inhibits growth of yeast cells. FEBS Lett. 1995;371(1):4–8. doi: 10.1016/0014-5793(95)00841-v. [DOI] [PubMed] [Google Scholar]
- Basu D, Walkiewicz MP, Frieman M, Baric RS, Auble DT, Engel DA. Novel influenza virus NS1 antagonists block replication and restore innate immune function. J Virol. 2009;83(4):1881–1891. doi: 10.1128/JVI.01805-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benko Z, Liang D, Agbottah E, Hou J, Chiu K, Yu M, Innis S, Reed P, Kabat W, Elder RT, Di Marzio P, Taricani L, Ratner L, Young PG, Bukrinsky M, Zhao RY. Anti-Vpr activity of a yeast chaperone protein. J Virol. 2004;78(20):11016–11029. doi: 10.1128/JVI.78.20.11016-11029.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benko Z, Liang D, Agbottah E, Hou J, Taricani L, Young PG, Bukrinsky M, Zhao RY (2007) Antagonistic interaction of HIV-1 Vpr with Hsf-mediated cellular heat shock response and Hsp16 in fission yeast (Schizosaccharomyces pombe). Retrovirology 4:16. 10.1186/1742-4690-4-16 [DOI] [PMC free article] [PubMed]
- Benko Z, Elder RT, Li G, Liang D, Zhao RY (2016) HIV-1 Protease in the Fission Yeast Schizosaccharomyces pombe. PLoS One 11(3):e0151286. 10.1371/journal.pone.0151286 [DOI] [PMC free article] [PubMed]
- Bidou L, Rousset J-P, Namy O. Translational errors: from yeast to new therapeutic targets. FEMS Yeast Res. 2010;10(8):1070–1082. doi: 10.1111/j.1567-1364.2010.00684.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bienz K, Egger D, Rasser Y, Bossart W. Intracellular distribution of poliovirus proteins and the induction of virus-specific cytoplasmic structures. Virology. 1983;131(1):39–48. doi: 10.1016/0042-6822(83)90531-7. [DOI] [PubMed] [Google Scholar]
- Blanco R, Carrasco L, Ventoso I. Cell killing by HIV-1 protease. J Biol Chem. 2003;278(2):1086–1093. doi: 10.1074/jbc.M205636200. [DOI] [PubMed] [Google Scholar]
- Bredell H, Smith JJ, Görgens JF, van Zyl WH (2018) Expression of unique chimeric human papilloma virus type 16 (HPV-16) L1–L2 proteins in Pichia pastoris and Hansenula polymorpha. Yeast 35(9):519–529. 10.1002/yea.3318 [DOI] [PubMed]
- Caetano KAA, Del-Rios NHA, Pinheiro RS, Bergamaschi FPR, dos Santos Carneiro MA, Teles SA (2017) Low immunogenicity of recombinant Hepatitis B vaccine derived from Hansenula polymorpha in adults aged over 40 years. Am J Trop Med Hyg 96(1):118–121. 10.4269/ajtmh.16-0475 [DOI] [PMC free article] [PubMed]
- Canady MA, Tihova M, Hanzlik TN, Johnson JE, Yeager M. Large conformational changes in the maturation of a simple RNA virus, nudaurelia capensis omega virus (NomegaV) J Mol Biol. 2000;299(3):573–584. doi: 10.1006/jmbi.2000.3723. [DOI] [PubMed] [Google Scholar]
- Caria S, Marshall B, Burton RL, Campbell S, Pantaki-Eimany D, Hawkins CJ, Barry M, Kvansakul M. The N terminus of the vaccinia virus protein F1L is an intrinsically unstructured region that is not involved in apoptosis regulation. J Biol Chem. 2016;291(28):14600–14608. doi: 10.1074/jbc.M116.726851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caumont AB, Jamieson GA, Pichuantes S, Nguyen AT, Litvak S, Dupont C (1996) Expression of functional HIV-1 integrase in the yeast Saccharomyces cerevisiae leads to the emergence of a lethal phenotype: potential use for inhibitor screening. Curr Genet 29(6):503–510. 10.1007/bf02426953 [DOI] [PubMed]
- Celik E, Calik P. Production of recombinant proteins by yeast cells. Biotechnol Adv. 2012;30(5):1108–1118. doi: 10.1016/j.biotechadv.2011.09.011. [DOI] [PubMed] [Google Scholar]
- Cervelli T, Backovic A, Galli A (2011) Formation of AAV single stranded DNA genome from a circular plasmid in Saccharomyces cerevisiae. PLoS One 6(8):e23474. 10.1371/journal.pone.0023474 [DOI] [PMC free article] [PubMed]
- Chamanian M, Purzycka KJ, Wille PT, Ha JS, McDonald D, Gao Y, Le Grice SF, Arts EJ. A cis-acting element in retroviral genomic RNA links Gag-Pol ribosomal frameshifting to selective viral RNA encapsidation. Cell Host Microbe. 2013;13(2):181–192. doi: 10.1016/j.chom.2013.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang F, Re F, Sebastian S, Sazer S, Luban J (2004) HIV-1 Vpr induces defects in mitosis, cytokinesis, nuclear structure, and centrosomes. Mol Biol Cell 15(4):1793–1801. 10.1091/mbc.e03-09-0691 [DOI] [PMC free article] [PubMed]
- Chang YH, Lee CP, Su MT, Wang JT, Chen JY, Lin SF, Tsai CH, Hsieh MJ, Takada K, Chen MR. Epstein-Barr virus BGLF4 kinase retards cellular S-phase progression and induces chromosomal abnormality. PLoS One. 2012;7(6):e39217. doi: 10.1371/journal.pone.0039217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen J, Ahlquist P. Brome Mosaic Virus Polymerase-Like Protein 2a Is Directed to the Endoplasmic Reticulum by Helicase-Like Viral Protein 1a. J Virol. 2000;74(9):4310–4318. doi: 10.1128/JVI.74.9.4310-4318.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen M, Elder RT, Yu M, O'Gorman MG, Selig L, Benarous R, Yamamoto A, Zhao Y. Mutational analysis of Vpr-induced G2 arrest, nuclear localization, and cell death in fission yeast. J Virol. 1999;73(4):3236–3245. doi: 10.1128/JVI.73.4.3236-3245.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen W-H, Du L, Chag SM, Ma C, Tricoche N, Tao X, Seid CA, Hudspeth EM, Lustigman S, Tseng C-TK. Yeast-expressed recombinant protein of the receptor-binding domain in SARS-CoV spike protein with deglycosylated forms as a SARS vaccine candidate. Hum Vaccines Immunother. 2014;10(3):648–658. doi: 10.4161/hv.27464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen P, Zhang L, Chang N, Shi P, Gao T, Zhang L, Huang J. Preparation of virus-like particles for porcine circovirus type 2 by YeastFab Assembly. Virus Genes. 2018;54(2):246–255. doi: 10.1007/s11262-018-1537-4. [DOI] [PubMed] [Google Scholar]
- Chen W-H, Strych U, Hotez PJ, Bottazzi ME. The SARS-CoV-2 vaccine pipeline: an overview. Curr Trop Med Rep. 2020;7(2):61–64. doi: 10.1007/s40475-020-00201-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng CY, Wu CW, Lin GJ, Lee WC, Chien MS, Huang C. Enhancing expression of the classical swine fever virus glycoprotein E2 in yeast and its application to a blocking ELISA. J Biotechnol. 2014;174:1–6. doi: 10.1016/j.jbiotec.2014.01.007. [DOI] [PubMed] [Google Scholar]
- Cheng J-H, Sun Y-J, Zhang F-Q, Zhang X-R, Qiu X-S, Yu L-P, Wu Y-T, Ding C. Newcastle disease virus NP and P proteins induce autophagy via the endoplasmic reticulum stress-related unfolded protein response. Sci Rep. 2016;6(1):1–10. doi: 10.1038/s41598-016-0001-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chua SC, Tan HQ, Engelberg D, Lim LH. Alternative experimental models for studying influenza proteins, host–virus interactions and anti-influenza drugs. Pharmaceuticals. 2019;12(4):147. doi: 10.3390/ph12040147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chuang C, Barajas D, Qin J, Nagy PD. Inactivation of the host lipin gene accelerates RNA virus replication through viral exploitation of the expanded endoplasmic reticulum membrane. PLoS Pathog. 2014;10(2):e1003944. doi: 10.1371/journal.ppat.1003944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chuang C, Prasanth KR, Nagy PD. The glycolytic pyruvate kinase is recruited directly into the viral replicase complex to generate ATP for RNA Synthesis. Cell Host Microbe. 2017;22(5):639–652. doi: 10.1016/j.chom.2017.10.004. [DOI] [PubMed] [Google Scholar]
- Curtain CC, Lowe MG, Arunagiri CK, Mobley PW, Macreadie IG, Azad AA. Cytotoxic activity of the amino-terminal region of HIV type 1 Nef protein. AIDS Res Hum Retrovir. 1997;13(14):1213–1220. doi: 10.1089/aid.1997.13.1213. [DOI] [PubMed] [Google Scholar]
- Dales S, Eggers HJ, Tamm I, Palade GE. Electron microscopic study of the formation of poliovirus. Virology. 1965;26:379–389. doi: 10.1016/0042-6822(65)90001-2. [DOI] [PubMed] [Google Scholar]
- den Boon JA, Chen J, Ahlquist P. Identification of sequences in Brome mosaic virus replicase protein 1a that mediate association with endoplasmic reticulum membranes. J Virol. 2001;75(24):12370–12381. doi: 10.1128/JVI.75.24.12370-12381.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desfarges S, Salin B, Calmels C, Andreola ML, Parissi V, Fournier M (2009) HIV-1 integrase trafficking in S. cerevisiae: a useful model to dissect the microtubule network involvement of viral protein nuclear import. Yeast 26(1):39–54. 10.1002/yea.1651 [DOI] [PubMed]
- Diaz A, Wang X, Ahlquist P. Membrane-shaping host reticulon proteins play crucial roles in viral RNA replication compartment formation and function. Proc Natl Acad Sci U S A. 2010;107(37):16291–16296. doi: 10.1073/pnas.1011105107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diaz A, Gallei A, Ahlquist P. Bromovirus RNA replication compartment formation requires concerted action of 1a's self-interacting RNA capping and helicase domains. J Virol. 2012;86(2):821–834. doi: 10.1128/JVI.05684-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duan J, Yang D, Chen L, Yu Y, Zhou J, Lu H (2019) Efficient production of porcine circovirus virus-like particles using the nonconventional yeast Kluyveromyces marxianus. Appl Microbiol Biotechnol 103(2):833–842. 10.1007/s00253-018-9487-2 [DOI] [PubMed]
- Elder RT, Yu M, Chen M, Edelson S, Zhao Y (2000) Cell cycle G2 arrest induced by HIV-1 Vpr in fission yeast (Schizosaccharomyces pombe) is independent of cell death and early genes in the DNA damage checkpoint. Virus Res 68(2):161–173. 10.1016/s0168-1702(00)00167-2 [DOI] [PubMed]
- Elder RT, Yu M, Chen M, Zhu X, Yanagida M, Zhao Y (2001) HIV-1 Vpr induces cell cycle G2 arrest in fission yeast (Schizosaccharomyces pombe) through a pathway involving regulatory and catalytic subunits of PP2A and acting on both Wee1 and Cdc25. Virology 287(2):359–370. 10.1006/viro.2001.1007 [DOI] [PubMed]
- Fazlalipour M, Keyvani H, Monavari SHR, Mollaie HR (2015) Expression, purification and immunogenic description of a hepatitis C virus recombinant CoreE1E2 protein expressed by yeast Pichia pastoris. Jundishapur J Microbiol 8(4). https://doi.org/10.5812/jjm.8(4)2015.17157 [DOI] [PMC free article] [PubMed]
- Feng Z, Xu K, Kovalev N, Nagy PD. Recruitment of Vps34 PI3K and enrichment of PI3P phosphoinositide in the viral replication compartment is crucial for replication of a positive-strand RNA virus. PLoS Pathog. 2019;15(1):ss. doi: 10.1371/journal.ppat.1007530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fewell SW, Markle DM, Brodsky JL. The carboxy terminus of simian virus 40 large T antigen is required to disrupt the yeast cell cycle. J Virol. 2002;76(9):4621–4624. doi: 10.1128/jvi.76.9.4621-4624.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Figueiredo S, Moreira JN, Geraldes CF, Rizzitelli S, Aime S, Terreno E. Yeast cell wall particles: a promising class of nature-inspired microcarriers for multimodal imaging. Chem Commun. 2011;47(38):10635–10637. doi: 10.1039/c1cc14019a. [DOI] [PubMed] [Google Scholar]
- Fournier N, Raj K, Saudan P, Utzig S, Sahli R, Simanis V, Beard P (1999) Expression of human papillomavirus 16 E2 protein in Schizosaccharomyces pombe delays the initiation of mitosis. Oncogene 18(27):4015–4021. 10.1038/sj.onc.1202775 [DOI] [PubMed]
- Frieman M, Basu D, Matthews K, Taylor J, Jones G, Pickles R, Baric R, Engel DA. Yeast based small molecule screen for inhibitors of SARS-CoV. PLoS One. 2011;6(12):e28479. doi: 10.1371/journal.pone.0028479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frischmuth S, Wege C, Hulser D, Jeske H. The movement protein BC1 promotes redirection of the nuclear shuttle protein BV1 of Abutilon mosaic geminivirus to the plasma membrane in fission yeast. Protoplasma. 2007;230(1-2):117–123. doi: 10.1007/s00709-006-0223-x. [DOI] [PubMed] [Google Scholar]
- Galao RP, Scheller N, Alves-Rodrigues I, Breinig T, Meyerhans A, Díez J (2007) Saccharomyces cerevisiae: a versatile eukaryotic system in virology. Microbial Cell Fact 6(1):1–9. 10.1186/1475-2859-6-32 [DOI] [PMC free article] [PubMed]
- Glingston RS, Deb R, Kumar S, Nagotu S. Organelle dynamics and viral infections: at cross roads. Microbes Infect. 2019;21(1):20–32. doi: 10.1016/j.micinf.2018.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glingston S, Rajpoot J, Deori NM, Deb R, Kumar S, Nagotu S. Characterization of nucleocapsid and matrix proteins of Newcastle disease virus in yeast. 3. Biotech. 2021;11(2):1–13. doi: 10.1007/s13205-020-02624-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodin MM, Austin J, Tobias R, Fujita M, Morales C, Jackson AO. Interactions and nuclear import of the N and P proteins of sonchus yellow net virus, a plant nucleorhabdovirus. J Virol. 2001;75(19):9393–9406. doi: 10.1128/JVI.75.19.9393-9406.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo Y, Dong J, Zhou T, Auxillos J, Li T, Zhang W, Wang L, Shen Y, Luo Y, Zheng Y, Lin J, Chen GQ, Wu Q, Cai Y, Dai J (2015) YeastFab: the design and construction of standard biological parts for metabolic engineering in Saccharomyces cerevisiae. Nucleic Acids Res 43(13):e88. 10.1093/nar/gkv464 [DOI] [PMC free article] [PubMed]
- Gupta G, Glueck R, Rishi N (2017) Physicochemical characterization and immunological properties of Pichia pastoris based HPV16L1 and 18L1 virus like particles. Biologicals 46:11–22. 10.1016/j.biologicals.2016.12.002 [DOI] [PubMed]
- Gutierrez C. DNA replication and cell cycle in plants: learning from geminiviruses. EMBO J. 2000;19(5):792–799. doi: 10.1093/emboj/19.5.792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haller AA, Lauer GM, King TH, Kemmler C, Fiolkoski V, Lu Y, Bellgrau D, Rodell TC, Apelian D, Franzusoff A. Whole recombinant yeast-based immunotherapy induces potent T cell responses targeting HCV NS3 and Core proteins. Vaccine. 2007;25(8):1452–1463. doi: 10.1016/j.vaccine.2006.10.035. [DOI] [PubMed] [Google Scholar]
- Hanley-Bowdoin L, Settlage SB, Robertson D. Reprogramming plant gene expression: a prerequisite to geminivirus DNA replication. Mol Plant Pathol. 2004;5(2):149–156. doi: 10.1111/j.1364-3703.2004.00214.x. [DOI] [PubMed] [Google Scholar]
- Hardy E, Martínez E, Diago D, Díaz R, González D, Herrera L (2000) Large-scale production of recombinant hepatitis B surface antigen from Pichia pastoris. J Biotechnol 77(2):157–167. 10.1016/S0168-1656(99)00201-1 [DOI] [PubMed]
- Hawkins CJ, Silke J, Verhagen AM, Foster R, Ekert PG, Ashley DM. Analysis of candidate antagonists of IAP-mediated caspase inhibition using yeast reconstituted with the mammalian Apaf-1-activated apoptosis mechanism. Apoptosis. 2001;6(5):331–338. doi: 10.1023/A:1011329917895. [DOI] [PubMed] [Google Scholar]
- Hilimire TA, Chamberlain JM, Anokhina V, Bennett RP, Swart O, Myers JR, Ashton JM, Stewart RA, Featherston AL, Gates K, Helms ED, Smith HC, Dewhurst S, Miller BL. HIV-1 Frameshift RNA-targeted triazoles inhibit propagation of replication-competent and multi-drug-resistant HIV in human cells. ACS Chem Biol. 2017;12(6):1674–1682. doi: 10.1021/acschembio.7b00052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilleman MR. Yeast recombinant hepatitis B vaccine. Infection. 1987;15(1):3–7. doi: 10.1007/bf01646107. [DOI] [PubMed] [Google Scholar]
- Hipp K, Rau P, Schafer B, Pfannstiel J, Jeske H. Translation, modification and cellular distribution of two AC4 variants of African cassava mosaic virus in yeast and their pathogenic potential in plants. Virology. 2016;498:136–148. doi: 10.1016/j.virol.2016.07.011. [DOI] [PubMed] [Google Scholar]
- Huard S, Chen M, Burdette KE, Fenyvuesvolgyi C, Yu M, Elder RT, Zhao RY (2008a) HIV-1 Vpr-induced cell death in Schizosaccharomyces pombe is reminiscent of apoptosis. Cell Res 18(9):961–973. 10.1038/cr.2008.272 [DOI] [PubMed]
- Huard S, Elder RT, Liang D, Li G, Zhao RY. Human immunodeficiency virus type 1 Vpr induces cell cycle G2 arrest through Srk1/MK2-mediated phosphorylation of Cdc25. J Virol. 2008;82(6):2904–2917. doi: 10.1128/JVI.01098-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ilkow CS, Weckbecker D, Cho WJ, Meier S, Beatch MD, Goping IS, Herrmann JM, Hobman TC. The rubella virus capsid protein inhibits mitochondrial import. J Virol. 2010;84(1):119–130. doi: 10.1128/JVI.01348-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iordanskiy S, Zhao Y, Dubrovsky L, Iordanskaya T, Chen M, Liang D, Bukrinsky M. Heat shock protein 70 protects cells from cell cycle arrest and apoptosis induced by human immunodeficiency virus type 1 viral protein R. J Virol. 2004;78(18):9697–9704. doi: 10.1128/JVI.78.18.9697-9704.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishikawa M, Diez J, Restrepo-Hartwig M, Ahlquist P. Yeast mutations in multiple complementation groups inhibit brome mosaic virus RNA replication and transcription and perturb regulated expression of the viral polymerase-like gene. Proc Natl Acad Sci U S A. 1997;94(25):13810–13815. doi: 10.1073/pnas.94.25.13810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isoyama T, Kuge S, Nomoto A. The core protein of hepatitis C virus is imported into the nucleus by transport receptor Kap123p but inhibits Kap121p-dependent nuclear import of yeast AP1-like transcription factor in yeast cells. J Biol Chem. 2002;277(42):39634–39641. doi: 10.1074/jbc.M203939200. [DOI] [PubMed] [Google Scholar]
- Iwasa S, Sato N, Wang CW, Cheng YH, Irokawa H, Hwang GW, Naganuma A, Kuge S. The phospholipid:diacylglycerol acyltransferase Lro1 is responsible for hepatitis C virus core-induced lipid droplet formation in a yeast model system. PLoS One. 2016;11(7):e0159324. doi: 10.1371/journal.pone.0159324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobs E, Rutgers T, Voet P, Dewerchin M, Cabezon T, De Wilde M (1989) Simultaneous synthesis and assembly of various hepatitis B surface proteins in Saccharomyces cerevisiae. Gene 80(2):279–291. 10.1016/0378-1119(89)90292-8 [DOI] [PubMed]
- Janda M, Ahlquist P (1993) RNA-dependent replication, transcription, and persistence of brome mosaic virus RNA replicons in S. cerevisiae. Cell 72(6):961–970. 10.1016/0092-8674(93)90584-d [DOI] [PubMed]
- Janette M. Berglez, Laura A. Castelli, Sonia A. Sankovich, Stuart C. Smith, Cyril C. Curtain, Macreadie aIG (1999) Residues within the HFRIGC Sequence of HIV-1 Vpr Involved in Growth Arrest Activities. Biochem Biophys Res 264 287–290 [DOI] [PubMed]
- Jiang Y, Serviene E, Gal J, Panavas T, Nagy PD. Identification of essential host factors affecting tombusvirus RNA replication based on the yeast Tet promoters Hughes Collection. J Virol. 2006;80(15):7394–7404. doi: 10.1128/JVI.02686-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin DY, Spencer F, Jeang KT. Human T cell leukemia virus type 1 oncoprotein Tax targets the human mitotic checkpoint protein MAD1. Cell. 1998;93(1):81–91. doi: 10.1016/s0092-8674(00)81148-4. [DOI] [PubMed] [Google Scholar]
- Jing W, Zhou J, Wang C, Qiu J, Guo H, Li H. Preparation of the secretory recombinant ALV-J gp85 protein using Pichia pastoris and its Immunoprotection as vaccine antigen combining with CpG-ODN adjuvant. Viral Immunol. 2018;31(6):407–416. doi: 10.1089/vim.2017.0170. [DOI] [PubMed] [Google Scholar]
- Jonczyk M, Pathak KB, Sharma M, Nagy PD. Exploiting alternative subcellular location for replication: tombusvirus replication switches to the endoplasmic reticulum in the absence of peroxisomes. Virology. 2007;362(2):320–330. doi: 10.1016/j.virol.2007.01.004. [DOI] [PubMed] [Google Scholar]
- Karathia H, Vilaprinyo E, Sorribas A, Alves R (2011) Saccharomyces cerevisiae as a model organism: a comparative study. PloS one 6(2) [DOI] [PMC free article] [PubMed]
- Khetarpal N, Shukla R, Rajpoot RK, Poddar A, Pal M, Swaminathan S, Arora U, Khanna N (2017) Recombinant dengue virus 4 envelope glycoprotein virus-like particles derived from Pichia pastoris are capable of eliciting homotypic domain III-directed neutralizing antibodies. Am J Trop Med Hyg 96(1):126–134. 10.4269/ajtmh.16-0503 [DOI] [PMC free article] [PubMed]
- Khulape S, Maity H, Pathak D, Mohan CM, Dey S (2015) Antigenic validation of recombinant hemagglutinin-neuraminidase protein of Newcastle disease virus expressed in Saccharomyces cerevisiae. Acta Virol 59(3):240–246. 10.4149/av_2015_03_240 [DOI] [PubMed]
- Kim H, Lee J, Kang H, Lee Y, Park EJ, Kim HJ. Oral immunization with whole yeast producing viral capsid antigen provokes a stronger humoral immune response than purified viral capsid antigen. Lett Appl Microbiol. 2014;58(3):285–291. doi: 10.1111/lam.12188. [DOI] [PubMed] [Google Scholar]
- Kim K, Angeletti PC, Hassebroek EC, Lambert PF (2005) Identification of cis-acting elements that mediate the replication and maintenance of human papillomavirus type 16 genomes in Saccharomyces cerevisiae. J Virol 79(10):5933–5942. 10.1128/JVI.79.10.5933-5942.2005 [DOI] [PMC free article] [PubMed]
- Kittelmann K, Rau P, Gronenborn B, Jeske H. Plant geminivirus rep protein induces rereplication in fission yeast. J Virol. 2009;83(13):6769–6778. doi: 10.1128/JVI.02491-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornitzer D, Sharf R, Kleinberger T (2001) Adenovirus E4orf4 protein induces PP2A-dependent growth arrest in Saccharomyces cerevisiae and interacts with the anaphase-promoting complex/cyclosome. J Cell Biol 154(2):331–344. 10.1083/jcb.200104104 [DOI] [PMC free article] [PubMed]
- Kovalev N, de Castro Martin IF, Pogany J, Barajas D, Pathak K, Risco C, Nagy PD. Role of viral RNA and co-opted cellular ESCRT-I and ESCRT-III factors in formation of tombusvirus spherules harboring the tombusvirus replicase. J Virol. 2016;90(7):3611–3626. doi: 10.1128/JVI.02775-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubota N, Inayoshi Y, Satoh N, Fukuda T, Iwai K, Tomoda H, Kohara M, Kataoka K, Shimamoto A, Furuichi Y, Nomoto A, Naganuma A, Kuge S. HSC90 is required for nascent hepatitis C virus core protein stability in yeast cells. FEBS Lett. 2012;586(16):2318–2325. doi: 10.1016/j.febslet.2012.05.023. [DOI] [PubMed] [Google Scholar]
- Kushner DB, Lindenbach BD, Grdzelishvili VZ, Noueiry AO, Paul SM, Ahlquist P. Systematic, genome-wide identification of host genes affecting replication of a positive-strand RNA virus. Proc Natl Acad Sci U S A. 2003;100(26):15764–15769. doi: 10.1073/pnas.2536857100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee WM, Ahlquist P. Membrane synthesis, specific lipid requirements, and localized lipid composition changes associated with a positive-strand RNA virus RNA replication protein. J Virol. 2003;77(23):12819–12828. doi: 10.1128/jvi.77.23.12819-12828.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee SC, Wu CH, Wang CW. Traffic of a viral movement protein complex to the highly curved tubules of the cortical endoplasmic reticulum. Traffic. 2010;11(7):912–930. doi: 10.1111/j.1600-0854.2010.01064.x. [DOI] [PubMed] [Google Scholar]
- Levin A, Armon-Omer A, Rosenbluh J, Melamed-Book N, Graessmann A, Waigmann E, Loyter A. Inhibition of HIV-1 integrase nuclear import and replication by a peptide bearing integrase putative nuclear localization signal. Retrovirology. 2009;6:112. doi: 10.1186/1742-4690-6-112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li G, Zhao RY. Molecular cloning and characterization of small viral genome in fission yeast. Methods Mol Biol. 2018;1721:47–61. doi: 10.1007/978-1-4939-7546-4_5. [DOI] [PubMed] [Google Scholar]
- Li Z, Pogany J, Panavas T, Xu K, Esposito AM, Kinzy TG, Nagy PD. Translation elongation factor 1A is a component of the tombusvirus replicase complex and affects the stability of the p33 replication co-factor. Virology. 2009;385(1):245–260. doi: 10.1016/j.virol.2008.11.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li HY, Han JF, Qin CF, Chen R (2013) Virus-like particles for enterovirus 71 produced from Saccharomyces cerevisiae potently elicits protective immune responses in mice. Vaccine 31(32):3281–3287. 10.1016/j.vaccine.2013.05.019 [DOI] [PubMed]
- Li J, Fuchs S, Zhang J, Wellford S, Schuldiner M, Wang X. An unrecognized function for COPII components in recruiting the viral replication protein BMV 1a to the perinuclear ER. J Cell Sci. 2016;129(19):3597–3608. doi: 10.1242/jcs.190082. [DOI] [PubMed] [Google Scholar]
- Li G, Poulsen M, Fenyvuesvolgyi C, Yashiroda Y, Yoshida M, Simard JM, Gallo RC, Zhao RY. Characterization of cytopathic factors through genome-wide analysis of the Zika viral proteins in fission yeast. Proc Natl Acad Sci U S A. 2017;114(3):E376–e385. doi: 10.1073/pnas.1619735114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang S, Hitomi M, Tartakoff AM. Adenoviral ElB-55kDa protein inhibits yeast mRNA export and perturbs nuclear structure. Cell biology. 1995;92:7372–7375. doi: 10.1073/pnas.92.16.7372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang Q, Luo Z, Zeng J, Chen W, Foo SS, Lee SA, Ge J, Wang S, Goldman SA, Zlokovic BV, Zhao Z, Jung JU. Zika virus NS4A and NS4B proteins deregulate Akt-mTOR signaling in human fetal neural stem cells to inhibit neurogenesis and induce autophagy. Cell Stem Cell. 2016;19(5):663–671. doi: 10.1016/j.stem.2016.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin JY, Nagy PD. Identification of novel host factors via conserved domain search: Cns1 cochaperone is a novel restriction factor of tombusvirus replication in yeast. J Virol. 2013;87(23):12600–12610. doi: 10.1128/JVI.00196-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lista MJ, Voisset C, Contesse MA, Friocourt G, Daskalogianni C, Bihel F, Fåhraeus R, Blondel M (2015) The long-lasting love affair between the budding yeast Saccharomyces cerevisiae and the Epstein-Barr virus. Biotechnol J 10(11):1670–1681. 10.1002/biot.201500161 [DOI] [PubMed]
- Liu B, Liang MH, Kuo YL, Liao W, Boros I, Kleinberger T, Blancato J, Giam CZ. Human T-lymphotropic virus type 1 oncoprotein tax promotes unscheduled degradation of Pds1p/securin and Clb2p/cyclin B1 and causes chromosomal instability. Mol Cell Biol. 2003;23(15):5269–5281. doi: 10.1128/mcb.23.15.5269-5281.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu C, Yao Y, Yang X, Bai H, Huang W, Xia Y, Ma Y (2015) Production of recombinant human papillomavirus type 52 L1 protein in Hansenula polymorpha formed virus-like particles. J Microbiol Biotechnol 25:936–940. 10.4014/jmb.1412.12027 [DOI] [PubMed]
- Lowe RS, Brown DR, Bryan JT, Cook JC, George HA, Hofmann KJ, Hurni WM, Joyce JG, Lehman ED, Markus HZ. Human papillomavirus type 11 (HPV-11) neutralizing antibodies in the serum and genital mucosal secretions of African green monkeys immunized with HPV-11 virus-like particles expressed in yeast. J Infect Dis. 1997;176(5):1141–1145. doi: 10.1086/514105. [DOI] [PubMed] [Google Scholar]
- Luu VT, Moon HY, Hwang JY, Kang BK, Kang HA (2017) Development of recombinant Yarrowia lipolytica producing virus-like particles of a fish nervous necrosis virus. J Microbiol 55(8):655–664. 10.1007/s12275-017-7218-5 [DOI] [PubMed]
- Macreadie IG, Castelli LA, Hewish DR, Kirkpatrick A, Ward AC, Azad AA. A domain of human immunodeficiency virus type 1 Vpr containing repeated H(S/F)RIG amino acid motifs causes cell growth arrest and structural defects. Proc Natl Acad Sci U S A. 1995;92(7):2770–2774. doi: 10.1073/pnas.92.7.2770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Macreadie IG, Lowe MG, Curtain CC, Hewish D, Azad AA. Cytotoxicity resulting from addition of HIV-1 Nef N-terminal peptides to yeast and bacterial cells. Biochem Biophys Res Commun. 1997;232(3):707–711. doi: 10.1006/bbrc.1997.6349. [DOI] [PubMed] [Google Scholar]
- Maoz T, Koren R, Ben-Ari I, Kleinberger T. YND1 interacts with CDC55 and is a novel mediator of E4orf4-induced toxicity. J Biol Chem. 2005;280(50):41270–41277. doi: 10.1074/jbc.M507281200. [DOI] [PubMed] [Google Scholar]
- Matsuda N, Tanaka H, Yamazaki S, Suzuki J, Tanaka K, Yamada T, Masuda M. HIV-1 Vpr induces G2 cell cycle arrest in fission yeast associated with Rad24/14-3-3-dependent, Chk1/Cds1-independent Wee1 upregulation. Microbes Infect. 2006;8(12-13):2736–2744. doi: 10.1016/j.micinf.2006.08.003. [DOI] [PubMed] [Google Scholar]
- Mendu V, Chiu M, Barajas D, Li Z, Nagy PD. Cpr1 cyclophilin and Ess1 parvulin prolyl isomerases interact with the tombusvirus replication protein and inhibit viral replication in yeast model host. Virology. 2010;406(2):342–351. doi: 10.1016/j.virol.2010.07.022. [DOI] [PubMed] [Google Scholar]
- Miller DJ, Ahlquist P. Flock house virus RNA polymerase is a transmembrane protein with amino-terminal sequences sufficient for mitochondrial localization and membrane insertion. J Virol. 2002;76(19):9856–9867. doi: 10.1128/jvi.76.19.9856-9867.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mittelman K, Ziv K, Maoz T, Kleinberger T (2010) The cytosolic tail of the Golgi apyrase Ynd1 mediates E4orf4-induced toxicity in Saccharomyces cerevisiae. PLoS One 5(11):e15539. 10.1371/journal.pone.0015539 [DOI] [PMC free article] [PubMed]
- Mui MZ, Roopchand DE, Gentry MS, Hallberg RL, Vogel J, Branton PE (2010) Adenovirus protein E4orf4 induces premature APCCdc20 activation in Saccharomyces cerevisiae by a protein phosphatase 2A-dependent mechanism. J Virol 84(9):4798–4809. 10.1128/JVI.02434-09 [DOI] [PMC free article] [PubMed]
- Nacht M, Reed SI, Alwine JC (1995) Simian virus 40 large T antigen affects the Saccharomyces cerevisiae cell cycle and interacts with p34CDC28. J Virol 69(2):756–763 [DOI] [PMC free article] [PubMed]
- Nagy PD. Yeast as a model host to explore plant virus-host interactions. Annu Rev Phytopathol. 2008;46:217–242. doi: 10.1146/annurev.phyto.121407.093958. [DOI] [PubMed] [Google Scholar]
- Nagy PD, Pogany J, Lin JY. How yeast can be used as a genetic platform to explore virus-host interactions: from 'omics' to functional studies. Trends Microbiol. 2014;22(6):309–316. doi: 10.1016/j.tim.2014.02.003. [DOI] [PubMed] [Google Scholar]
- Naito T, Kiyasu Y, Sugiyama K, Kimura A, Nakano R, Matsukage A, Nagata K. An influenza virus replicon system in yeast identified Tat-SF1 as a stimulatory host factor for viral RNA synthesis. Proc Natl Acad Sci U S A. 2007;104(46):18235–18240. doi: 10.1073/pnas.0705856104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakazawa J, Watanabe N, Imoto M, Osada H (2005) Mutational analysis of growth arrest and cellular localization of human immunodeficiency virus type 1 Vpr in the budding yeast, Saccharomyces cerevisiae. J Gen Appl Microbiol 51(4):245–256 [DOI] [PubMed]
- Navarro B, Rubino L, Russo M (2004) Expression of the Cymbidium ringspot virus 33-kilodalton protein in Saccharomyces cerevisiae and molecular dissection of the peroxisomal targeting signal. J Virol 78(9):4744–4752. 10.1128/jvi.78.9.4744-4752.2004 [DOI] [PMC free article] [PubMed]
- Nawaz-ul-Rehman MS, Reddisiva Prasanth K, Baker J, Nagy PD. Yeast screens for host factors in positive-strand RNA virus replication based on a library of temperature-sensitive mutants. Methods. 2013;59(2):207–216. doi: 10.1016/j.ymeth.2012.11.001. [DOI] [PubMed] [Google Scholar]
- Nawaz-ul-Rehman MS, Prasanth KR, Xu K, Sasvari Z, Kovalev N, de Castro Martin IF, Barajas D, Risco C, Nagy PD. Viral replication protein inhibits cellular cofilin actin depolymerization factor to regulate the actin network and promote viral replicase assembly. PLoS Pathog. 2016;12(2):e1005440. doi: 10.1371/journal.ppat.1005440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen NL, Kim JM, Park JA, Park SM, Jang YS, Yang MS, Kim DH (2013) Expression and purification of an immunogenic dengue virus epitope using a synthetic consensus sequence of envelope domain III and Saccharomyces cerevisiae. Protein Expr Purif 88(2):235–242. 10.1016/j.pep.2013.01.009 [DOI] [PubMed]
- Nkeze J, Li L, Benko Z, Li G, Zhao RY (2015) Molecular characterization of HIV-1 genome in fission yeast Schizosaccharomyces pombe. Cell Biosci 5:47. 10.1186/s13578-015-0037-7 [DOI] [PMC free article] [PubMed]
- Noueiry AO, Chen J, Ahlquist P. A mutant allele of essential, general translation initiation factor DED1 selectively inhibits translation of a viral mRNA. Proc Natl Acad Sci U S A. 2000;97(24):12985–12990. doi: 10.1073/pnas.240460897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noueiry AO, Diez J, Falk SP, Chen J, Ahlquist P. Yeast Lsm1p-7p/Pat1p deadenylation-dependent mRNA-decapping factors are required for brome mosaic virus genomic RNA translation. Mol Cell Biol. 2003;23(12):4094–4106. doi: 10.1128/mcb.23.12.4094-4106.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nunes-Correia I, Rodriguez JM, Eulalio A, Carvalho AL, Citovsky V, Simoes S, Faro C, Salas ML, Pedroso de Lima MC. African swine fever virus p10 protein exhibits nuclear import capacity and accumulates in the nucleus during viral infection. Vet Microbiol. 2008;130(1-2):47–59. doi: 10.1016/j.vetmic.2007.12.010. [DOI] [PubMed] [Google Scholar]
- Oh HJ, Moon HY, Cheon SA, Hahn Y, Kang HA (2016) Functional analysis of recombinant human and Yarrowia lipolytica O-GlcNAc transferases expressed in Saccharomyces cerevisiae. J Microbiol 54(10):667–674. 10.1007/s12275-016-6401-4 [DOI] [PubMed]
- Orzalli MH, Kagan JC. Apoptosis and necroptosis as host defense strategies to prevent viral infection. Trends Cell Biol. 2017;27(11):800–809. doi: 10.1016/j.tcb.2017.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palkova Z, Adamec T, Liebl D, Stokrova J, Forstova J. Production of polyomavirus structural protein VP1 in yeast cells and its interaction with cell structures. FEBS Lett. 2000;478(3):281–289. doi: 10.1016/s0014-5793(00)01787-7. [DOI] [PubMed] [Google Scholar]
- Panavas T, Hawkins CM, Panaviene Z, Nagy PD. The role of the p33:p33/p92 interaction domain in RNA replication and intracellular localization of p33 and p92 proteins of Cucumber necrosis tombusvirus. Virology. 2005;338(1):81–95. doi: 10.1016/j.virol.2005.04.025. [DOI] [PubMed] [Google Scholar]
- Panavas T, Serviene E, Brasher J, Nagy PD. Yeast genome-wide screen reveals dissimilar sets of host genes affecting replication of RNA viruses. Proc Natl Acad Sci U S A. 2005;102(20):7326–7331. doi: 10.1073/pnas.0502604102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pantaleo V, Rubino L, Russo M (2003) Replication of carnation Italian ringspot virus defective interfering RNA in Saccharomyces cerevisiae. J Virol 77(3):2116–2123. 10.1128/jvi.77.3.2116-2123.2003 [DOI] [PMC free article] [PubMed]
- Park MA, Kim HJ, Kim HJ (2008) Optimum conditions for production and purification of human papillomavirus type 16 L1 protein from Saccharomyces cerevisiae. Protein Expr Purif 59(1):175–181. 10.1016/j.pep.2008.01.021 [DOI] [PubMed]
- Pathak KB, Sasvari Z, Nagy PD. The host Pex19p plays a role in peroxisomal localization of tombusvirus replication proteins. Virology. 2008;379(2):294–305. doi: 10.1016/j.virol.2008.06.044. [DOI] [PubMed] [Google Scholar]
- Penkert RR, Young NS, Surman SL, Sealy RE, Rosch J, Dormitzer PR, Settembre EC, Chandramouli S, Wong S, Hankins JS (2017) Saccharomyces cerevisiae-derived virus-like particle parvovirus B19 vaccine elicits binding and neutralizing antibodies in a mouse model for sickle cell disease. Vaccine 35(29):3615–3620. 10.1016/j.vaccine.2017.05.022 [DOI] [PMC free article] [PubMed]
- Penno C, Kumari R, Baranov PV, van Sinderen D, Atkins JF. Specific reverse transcriptase slippage at the HIV ribosomal frameshift sequence: potential implications for modulation of GagPol synthesis. Nucleic Acids Res. 2017;45(17):10156–10167. doi: 10.1093/nar/gkx690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pietrzak M, Macioła A, Zdanowski K, Protas-Klukowska AM, Olszewska M, Śmietanka K, Minta Z, Szewczyk B, Kopera E. An avian influenza H5N1 virus vaccine candidate based on the extracellular domain produced in yeast system as subviral particles protects chickens from lethal challenge. Antivir Res. 2016;133:242–249. doi: 10.1016/j.antiviral.2016.08.001. [DOI] [PubMed] [Google Scholar]
- Pleckaityte M, Bremer CM, Gedvilaite A, Kucinskaite-Kodze I, Glebe D, Zvirbliene A. Construction of polyomavirus-derived pseudotype virus-like particles displaying a functionally active neutralizing antibody against hepatitis B virus surface antigen. BMC Biotechnol. 2015;15(1):1–9. doi: 10.1186/s12896-015-0203-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollet J, Chen W-H, Versteeg L, Keegan B, Zhan B, Wei J, Liu Z, Lee J, Kundu R, Adhikari R (2020) SARS-CoV-2 RBD219-N1C1: a yeast-expressed SARS-CoV-2 recombinant receptor-binding domain candidate vaccine stimulates virus neutralizing antibodies and T-cell immunity in mice. BioRxiv. 10.1101/2020.11.04.367359 [DOI] [PMC free article] [PubMed]
- Price BD, Rueckert RR, Ahlquist P (1996) Complete replication of an animal virus and maintenance of expression vectors derived from it in Saccharomyces cerevisiae. Proc Natl Acad Sci U S A 93(18):9465–9470. 10.1073/pnas.93.18.9465 [DOI] [PMC free article] [PubMed]
- Price BD, Eckerle LD, Ball LA, Johnson KL (2005) Nodamura virus RNA replication in Saccharomyces cerevisiae: heterologous gene expression allows replication-dependent colony formation. J Virol 79(1):495–502. 10.1128/JVI.79.1.495-502.2005 [DOI] [PMC free article] [PubMed]
- Qin J, Barajas D, Nagy PD. An inhibitory function of WW domain-containing host proteins in RNA virus replication. Virology. 2012;426(2):106–119. doi: 10.1016/j.virol.2012.01.020. [DOI] [PubMed] [Google Scholar]
- Rajpoot RK, Shukla R, Arora U, Swaminathan S, Khanna N (2018) Dengue envelope-based 'four-in-one' virus-like particles produced using Pichia pastoris induce enhancement-lacking, domain III-directed tetravalent neutralising antibodies in mice. Sci Rep 8(1):8643. 10.1038/s41598-018-26904-5 [DOI] [PMC free article] [PubMed]
- Ralph B, Denise T, Tineke R, Michel DW, Teresa C. The large surface protein of Hepatitis B virus is retained in the yeast endoplasmic reticulum and provokes its unique enlargement. DNA Cell Biol. 1991;10:191–200. doi: 10.1089/dna.1991.10.191. [DOI] [PubMed] [Google Scholar]
- Ramasamy V, Arora U, Shukla R, Poddar A, Shanmugam RK, White LJ, Mattocks MM, Raut R, Perween A, Tyagi P (2018) A tetravalent virus-like particle vaccine designed to display domain III of dengue envelope proteins induces multi-serotype neutralizing antibodies in mice and macaques which confer protection against antibody dependent enhancement in AG129 mice. PLoS Negl Trop Dis 12(1) [DOI] [PMC free article] [PubMed]
- Restrepo-Hartwig MA, Ahlquist P. Brome mosaic virus helicase- and polymerase-like Proteins colocalize on the endoplasmic reticulum at sites of viral RNA synthesis. J Virol. 1996;70:8908–8916. doi: 10.1128/jvi.70.12.8908-8916.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Restrepo-Hartwig M, Ahlquist P. Brome mosaic virus RNA replication proteins 1a and 2a colocalize and 1a independently localizes on the yeast endoplasmic reticulum. J Virol. 1999;73(12):10303–10309. doi: 10.1128/JVI.73.12.10303-10309.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roldao A, Mellado MC, Castilho LR, Carrondo MJ, Alves PM. Virus-like particles in vaccine development. Expert Rev Vaccines. 2010;9(10):1149–1176. doi: 10.1586/erv.10.115. [DOI] [PubMed] [Google Scholar]
- Roopchand DE, Lee JM, Shahinian S, Paquette D, Bussey H, Branton PE (2001) Toxicity of human adenovirus E4orf4 protein in Saccharomyces cerevisiae results from interactions with the Cdc55 regulatory B subunit of PP2A. Oncogene 20(38):5279–5290. 10.1038/sj.onc.1204693 [DOI] [PubMed]
- Rubino L, Di Franco A, Russo M (2000) Expression of a plant virus non-structural protein in Saccharomyces cerevisiae causes membrane proliferation and altered mitochondrial morphology. J Gen Virol 81(Pt 1):279–286. 10.1099/0022-1317-81-1-279 [DOI] [PubMed]
- Rubino L, Navarro B, Russo M. Cymbidium ringspot virus defective interfering RNA replication in yeast cells occurs on endoplasmic reticulum-derived membranes in the absence of peroxisomes. J Gen Virol. 2007;88(Pt 5):1634–1642. doi: 10.1099/vir.0.82729-0. [DOI] [PubMed] [Google Scholar]
- Rubino L, Guaragnella N, Giannattasio S (2017) Heterologous expression of carnation Italian ringspot virus p36 protein enhances necrotic cell death in response to acetic acid in Saccharomyces cerevisiae. Mech Ageing Dev 161:255–261. 10.1016/j.mad.2016.09.004 [DOI] [PubMed]
- Ryerson MR, Richards MM, Kvansakul M, Hawkins CJ, Shisler JL. Vaccinia virus encodes a novel inhibitor of apoptosis that associates with the apoptosome. J Virol. 2017;91(23):e01385–e01317. doi: 10.1128/JVI.01385-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saraswat S, Athmaram TN, Parida M, Agarwal A, Saha A, Dash PK. Expression and characterization of yeast derived chikungunya virus like particles (CHIK-VLPs) and its evaluation as a potential vaccine candidate. PLoS Negl Trop Dis. 2016;10(7):e0004782. doi: 10.1371/journal.pntd.0004782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasagawa T, Pushko P, Steers G, Gschmeissner SE, Hajibagheri MA, Finch J, Crawford L, Tommasino M (1995) Synthesis and assembly of virus-like particles of human papillomaviruses type 6 and type 16 in fission yeast Schizosaccharomyces pombe. Virology 206(1):126–135. 10.1016/s0042-6822(95)80027-1 [DOI] [PubMed]
- Sasvari Z, Gonzalez PA, Rachubinski RA, Nagy PD. Tombusvirus replication depends on Sec39p endoplasmic reticulum-associated transport protein. Virology. 2013;447(1-2):21–31. doi: 10.1016/j.virol.2013.07.039. [DOI] [PubMed] [Google Scholar]
- Schwartz M, Chen J, Lee WM, Janda M, Ahlquist P. Alternate, virus-induced membrane rearrangements support positive-strand RNA virus genome replication. Proc Natl Acad Sci U S A. 2004;101(31):11263–11268. doi: 10.1073/pnas.0404157101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serva S, Nagy PD. Proteomics analysis of the tombusvirus replicase: Hsp70 molecular chaperone is associated with the replicase and enhances viral RNA replication. J Virol. 2006;80(5):2162–2169. doi: 10.1128/JVI.80.5.2162-2169.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah Nawaz-ul-Rehman M, Martinez-Ochoa N, Pascal H, Sasvari Z, Herbst C, Xu K, Baker J, Sharma M, Herbst A, Nagy PD. Proteome-wide overexpression of host proteins for identification of factors affecting tombusvirus RNA replication: an inhibitory role of protein kinase C. J Virol. 2012;86(17):9384–9395. doi: 10.1128/JVI.00019-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shubin AV, Komissarov AA, Karaseva MA, Padman BS, Kostrov SV, Demidyuk IV (2021) Human hepatitis A virus 3C protease exerts a cytostatic effect on Saccharomyces cerevisiae and affects the vacuolar compartment. Biologia 76(1):321–327. 10.2478/s11756-020-00569-w
- Song Y, Choi MH, Park JN, Kim MW, Kim EJ, Kang HA, Kim JY (2007) Engineering of the yeast Yarrowia lipolytica for the production of glycoproteins lacking the outer-chain mannose residues of N-glycans. Appl Environ Microbiol 73(14):4446–4454. 10.1128/AEM.02058-06 [DOI] [PMC free article] [PubMed]
- Stromajer-Racz T, Gazdag Z, Belagyi J, Vagvolgyi C, Zhao RY, Pesti M (2010) Oxidative stress induced by HIV-1 F34IVpr in Schizosaccharomyces pombe is one of its multiple functions. Exp Mol Pathol 88(1):38–44. 10.1016/j.yexmp.2009.10.002 [DOI] [PubMed]
- Takahashi S, Sato N, Kikuchi J, Kakinuma H, Okawa J, Masuyama Y, Iwasa S, Irokawa H, Hwang GW, Naganuma A, Kohara M, Kuge S. Immature Core protein of hepatitis C virus induces an unfolded protein response through inhibition of ERAD-L in a yeast model system. Genes Cells. 2017;22(2):160–173. doi: 10.1111/gtc.12464. [DOI] [PubMed] [Google Scholar]
- Takegawa K, Tohda H, Sasaki M, Idiris A, Ohashi T, Mukaiyama H, Giga-Hama Y, Kumagai H. Production of heterologous proteins using the fission-yeast (Schizosaccharomyces pombe) expression system. Biotechnol Appl Biochem. 2009;53(4):227–235. doi: 10.1042/BA20090048. [DOI] [PubMed] [Google Scholar]
- Thao TTN, Labroussaa F, Ebert N, V’kovski P, Stalder H, Portmann J, Kelly J, Steiner S, Holwerda M, Kratzel A. Rapid reconstruction of SARS-CoV-2 using a synthetic genomics platform. Nature. 2020;582(7813):561–565. doi: 10.1038/s41586-020-2294-9. [DOI] [PubMed] [Google Scholar]
- Tipper DJ, Szomolanyi-Tsuda E. Scaffolded antigens in yeast cell particle vaccines provide protection against systemic polyoma virus infection. J Immunol Res. 2016;2016:2743292. doi: 10.1155/2016/2743292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomasicchio M, Venter PA, Gordon KH, Hanzlik TN, Dorrington RA (2007) Induction of apoptosis in Saccharomyces cerevisiae results in the spontaneous maturation of tetravirus procapsids in vivo. J Gen Virol 88(Pt 5):1576–1582. 10.1099/vir.0.82250-0 [DOI] [PubMed]
- Tomita Y, Mizuno T, Diez J, Naito S, Ahlquist P, Ishikawa M. Mutation of host DnaJ homolog inhibits brome mosaic virus negative-strand RNA synthesis. J Virol. 2003;77(5):2990–2997. doi: 10.1128/jvi.77.5.2990-2997.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tommasino M, Contorni M, Searlato V, Buguoli M, b KM, Cavalieri aF (1990) Synthesis, phosphorylation, and nuclear IocaHzafian of human papillomaviros E7 protein in Schizosaccharomyces pombe. Gene 93(2):265–270 [DOI] [PubMed]
- Tu YC, Yu CY, Liang JJ, Lin E, Liao CL, Lin YL. Blocking double-stranded RNA-activated protein kinase PKR by Japanese encephalitis virus nonstructural protein 2A. J Virol. 2012;86(19):10347–10358. doi: 10.1128/JVI.00525-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Upadhyaya B, Manjunath R (2009) Baker’s yeast expressing the Japanese encephalitis virus envelope protein on its cell surface: induction of an antigen-specific but non-neutralizing antibody response. Yeast 26(7):383–397. 10.1002/yea.1676 [DOI] [PubMed]
- Upton JW, Chan FK. Staying alive: cell death in antiviral immunity. Mol Cell. 2014;54(2):273–280. doi: 10.1016/j.molcel.2014.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valenzuela P, Medina A, Rutter WJ, Ammerer G, Hall BD. Synthesis and assembly of hepatitis B virus surface antigen particles in yeast. Nature. 1982;298(5872):347–350. doi: 10.1038/298347a0. [DOI] [PubMed] [Google Scholar]
- Varadarajan P, Mahalingam S, Liu P, Ng SB, Gandotra S, Dorairajoo DS, Balasundaram D. The functionally conserved nucleoporins Nup124p from fission yeast and the human Nup153 mediate nuclear import and activity of the Tf1 retrotransposon and HIV-1 Vpr. Mol Biol Cell. 2005;16(4):1823–1838. doi: 10.1091/mbc.e04-07-0583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vasileva A, Jessberger R. Precise hit: adeno-associated virus in gene targeting. Nat Rev Microbiol. 2005;3(11):837–847. doi: 10.1038/nrmicro1266. [DOI] [PubMed] [Google Scholar]
- Villegas P, Hamoud M, Purvis L, Perozo F. Infectious bursal disease subunit vaccination. Avian Dis. 2008;52(4):670–674. doi: 10.1637/8289-032008-ResNote.1. [DOI] [PubMed] [Google Scholar]
- Vodicka MA, Koepp DM, Silver PA, Emerman M. HIV-1 Vpr interacts with the nuclear transport pathway to promote macrophage infection. Genes Dev. 1998;12(2):175–185. doi: 10.1101/gad.12.2.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogl T, Hartner FS, Glieder A (2013) New opportunities by synthetic biology for biopharmaceutical production in Pichia pastoris. Curr Opin Biotechnol 24(6):1094–1101. 10.1016/j.copbio.2013.02.024 [DOI] [PMC free article] [PubMed]
- Wang H, Chen X, Wang D, Yao C, Wang Q, Xie J, Shi X, Xiang Y, Liu W, Zhang L. Epitope-focused immunogens against the CD4-binding site of HIV-1 envelope protein induce neutralizing antibodies against auto-and heterologous viruses. J Biol Chem. 2018;293(3):830–846. doi: 10.1074/jbc.M117.816447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang RY, Nagy PD. Tomato bushy stunt virus co-opts the RNA-binding function of a host metabolic enzyme for viral genomic RNA synthesis. Cell Host Microbe. 2008;3(3):178–187. doi: 10.1016/j.chom.2008.02.005. [DOI] [PubMed] [Google Scholar]
- Wang X, Lee WM, Watanabe T, Schwartz M, Janda M, Ahlquist P. Brome mosaic virus 1a nucleoside triphosphatase/helicase domain plays crucial roles in recruiting RNA replication templates. J Virol. 2005;79(21):13747–13758. doi: 10.1128/JVI.79.21.13747-13758.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang RY, Stork J, Nagy PD. A key role for heat shock protein 70 in the localization and insertion of tombusvirus replication proteins to intracellular membranes. J Virol. 2009;83(7):3276–3287. doi: 10.1128/JVI.02313-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Diaz A, Hao L, Gancarz B, den Boon JA, Ahlquist P. Intersection of the multivesicular body pathway and lipid homeostasis in RNA replication by a positive-strand RNA virus. J Virol. 2011;85(11):5494–5503. doi: 10.1128/JVI.02031-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang SC, Liao HY, Zhang JY, Cheng TR, Wong CH (2019) Development of a universal influenza vaccine using hemagglutinin stem protein produced from Pichia pastoris. Virology 526:125–137. 10.1016/j.virol.2018.10.005 [DOI] [PubMed]
- Ward AC, Castelli LA, Lucantoni AC, White JF, Azad AA, Macreadie IG. Expression and analysis of the NS 2 protein of influenza A virus. Arch Virol. 1995;140(11):2067–2073. doi: 10.1007/BF01322693. [DOI] [PubMed] [Google Scholar]
- Wasilenko JL, Sarmento L, Spatz S, Pantin-Jackwood M (2010) Cell surface display of highly pathogenic avian influenza virus hemagglutinin on the surface of Pichia pastoris cells using α-agglutinin for production of oral vaccines. Biotechnol Prog 26(2):542–547. 10.1002/btpr.343 [DOI] [PubMed]
- Watts JM, Dang KK, Gorelick RJ, Leonard CW, Bess JW, Jr, Swanstrom R, Burch CL, Weeks KM. Architecture and secondary structure of an entire HIV-1 RNA genome. Nature. 2009;460(7256):711–716. doi: 10.1038/nature08237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber-Lotfi F, Dietrich A, Russo M, Rubino L. Mitochondrial targeting and membrane anchoring of a viral replicase in plant and yeast cells. J Virol. 2002;76(20):10485–10496. doi: 10.1128/jvi.76.20.10485-10496.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weeks SA, Miller DJ (2008) The heat shock protein 70 cochaperone YDJ1 is required for efficient membrane-specific flock house virus RNA replication complex assembly and function in Saccharomyces cerevisiae. J Virol 82(4):2004–2012. 10.1128/JVI.02017-07 [DOI] [PMC free article] [PubMed]
- Weston S, Matthews KL, Lent R, Vlk A, Haupt R, Kingsbury T, Frieman MB (2019) A yeast suppressor screen used to identify mammalian SIRT1 as a proviral factor for Middle East respiratory syndrome coronavirus replication. J Virol 93(16) [DOI] [PMC free article] [PubMed]
- Wetzel D, Rolf T, Suckow M, Kranz A, Barbian A, Chan JA, Leitsch J, Weniger M, Jenzelewski V, Kouskousis B, Palmer C, Beeson JG, Schembecker G, Merz J, Piontek M. Establishment of a yeast-based VLP platform for antigen presentation. Microb Cell Factories. 2018;17(1):17. doi: 10.1186/s12934-018-0868-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson W, Braddock M, Adams SE, Rathjen PD, Kingsman SM, Kingsman AJ. HIV expression strategies: ribosomal frameshifting is directed by a short sequence in both mammalian and yeast systems. Cell. 1988;55(6):1159–1169. doi: 10.1016/0092-8674(88)90260-7. [DOI] [PubMed] [Google Scholar]
- Wixon J (2002) Featured organism: Schizosaccharomyces pombe, the fission yeast. Comparative and functional genomics 3(2):194–204. 10.1002/cfg.92 [DOI] [PMC free article] [PubMed]
- Xia M, Farkas T, Jiang X. Norovirus capsid protein expressed in yeast forms virus-like particles and stimulates systemic and mucosal immunity in mice following an oral administration of raw yeast extracts. J Med Virol. 2007;79(1):74–83. doi: 10.1002/jmv.20762. [DOI] [PubMed] [Google Scholar]
- Xiao Y, Zhao P, Du J, Li X, Lu W, Hao X, Dong B, Yu Y, Wang L (2018) High-level expression and immunogenicity of porcine circovirus type 2b capsid protein without nuclear localization signal expressed in Hansenula polymorpha. Biologicals 51:18–24. 10.1016/j.biologicals.2017.11.003 [DOI] [PubMed]
- Xu K, Nagy PD (2017) Sterol Binding by the Tombusviral Replication Proteins Is Essential for Replication in Yeast and Plants. J Virol 91(7). 10.1128/jvi.01984-16 [DOI] [PMC free article] [PubMed]
- Xu Z, Zheng Y, Ao Z, Clement M, Mouland AJ, Kalpana GV, Belhumeur P, Cohen EA, Yao X. Contribution of the C-terminal region within the catalytic core domain of HIV-1 integrase to yeast lethality, chromatin binding and viral replication. Retrovirology. 2008;5:102. doi: 10.1186/1742-4690-5-102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu K, Lin JY, Nagy PD. The hop-like stress-induced protein 1 cochaperone is a novel cell-intrinsic restriction factor for mitochondrial tombusvirus replication. J Virol. 2014;88(16):9361–9378. doi: 10.1128/JVI.00561-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yongkiettrakul S, Boonyapakron K, Jongkaewwattana A, Wanitchang A, Leartsakulpanich U, Chitnumsub P, Eurwilaichitr L, Yuthavong Y. Avian influenza A/H5N1 neuraminidase expressed in yeast with a functional head domain. J Virol Methods. 2009;156(1–2):44–51. doi: 10.1016/j.jviromet.2008.10.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zara V, Ferramosca A, Gunnewig K, Kreimendahl S, Schwichtenberg J, Strater D, Cakar M, Emmrich K, Guidato P, Palmieri F, Rassow J. Mimivirus-Encoded Nucleotide Translocator VMC1 Targets the Mitochondrial Inner Membrane. J Mol Biol. 2018;430(24):5233–5245. doi: 10.1016/j.jmb.2018.09.012. [DOI] [PubMed] [Google Scholar]
- Zhang C, Rasmussen C, Chang LJ (1997) Cell cycle inhibitory effects of HIV and SIV Vpr and Vpx in the yeast Schizosaccharomyces pombe. Virology 230(1):103–112. 10.1006/viro.1997.8459 [DOI] [PubMed]
- Zhang C, Liu Q, Ku Z, Hu Y, Ye X, Zhang Y, Huang Z (2016a) Coxsackievirus A16-like particles produced in Pichia pastoris elicit high-titer neutralizing antibodies and confer protection against lethal viral challenge in mice. Antivir Res 129:47–51. 10.1016/j.antiviral.2016.02.011 [DOI] [PubMed]
- Zhang J, Zhang Z, Chukkapalli V, Nchoutmboube JA, Li J, Randall G, Belov GA, Wang X. Positive-strand RNA viruses stimulate host phosphatidylcholine synthesis at viral replication sites. Proc Natl Acad Sci U S A. 2016;113(8):E1064–E1073. doi: 10.1073/pnas.1519730113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao H, Li HY, Han JF, Deng YQ, Zhu SY, Li XF, Yang HQ, Li YX, Zhang Y, Qin ED. Novel recombinant chimeric virus-like particle is immunogenic and protective against both enterovirus 71 and coxsackievirus A16 in mice. Sci Rep. 2015;5(1):1–8. doi: 10.1038/srep07878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao RY. Yeast for virus research. Microb Cell. 2017;4(10):311–330. doi: 10.15698/mic2017.10.592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao RY, Elder RT. Viral infections and cell cycle G2/M regulation. Cell Res. 2005;15(3):143–149. doi: 10.1038/sj.cr.7290279. [DOI] [PubMed] [Google Scholar]
- Zhao Y, Cao J, O'Gorman MR, Yu M, Yogev R. Effect of human immunodeficiency virus type 1 protein R (vpr) gene expression on basic cellular function of fission yeast Schizosaccharomyces pombe. J Virol. 1996;70(9):5821–5826. doi: 10.1128/jvi.70.9.5821-5826.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Y, Yu M, Chen M, Elder RT, Yamamoto A, Cao J. Pleiotropic effects of HIV-1 protein R (Vpr) on morphogenesis and cell survival in fission yeast and antagonism by pentoxifylline. Virology. 1998;246(2):266–276. doi: 10.1006/viro.1998.9208. [DOI] [PubMed] [Google Scholar]
- Zhao RY, Li G, Bukrinsky MI. Vpr-host interactions during HIV-1 viral life cycle. J NeuroImmune Pharmacol. 2011;6(2):216–229. doi: 10.1007/s11481-011-9261-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao H, Li HY, Han JF, Deng YQ, Li YX, Zhu SY, He YL, Qin ED, Chen R, Qin CF (2013) Virus-like particles produced in Saccharomyces cerevisiae elicit protective immunity against Coxsackievirus A16 in mice. Appl Microbiol Biotechnol 97(24):10445–10452. 10.1007/s00253-013-5257-3 [DOI] [PubMed]
- Zhou Y, Shen C, Zhang C, Zhang W, Wang L, Lan K, Liu Q, Huang Z. Yeast-produced recombinant virus-like particles of coxsackievirus A6 elicited protective antibodies in mice. Antivir Res. 2016;132:165–169. doi: 10.1016/j.antiviral.2016.06.004. [DOI] [PubMed] [Google Scholar]
- Zhu J, Gopinath K, Murali A, Yi G, Hayward SD, Zhu H, Kao C. RNA-binding proteins that inhibit RNA virus infection. Proc Natl Acad Sci U S A. 2007;104(9):3129–3134. doi: 10.1073/pnas.0611617104. [DOI] [PMC free article] [PubMed] [Google Scholar]