Abstract
Background.
Genetics hold promise of predicting long-term post-traumatic stress disorder (PTSD) outcomes following trauma. The aim of the current study was to test whether six hypothesized polygenic risk scores (PRSs) developed to capture genetic vulnerability to psychiatric conditions prospectively predict PTSD onset, severity, and 18-year course after trauma exposure.
Methods.
Participants were 1490 responders to the World Trade Center (WTC) disaster (mean age at 9/11 = 38.81 years, S.D. = 8.20; 93.5% male; 23.8% lifetime WTC-related PTSD diagnosis). Prospective longitudinal data on WTC-related PTSD symptoms were obtained from electronic medical records and modelled as PTSD trajectories using growth mixture model analysis. Independent regression models tested whether six hypothesized psychiatric PRSs (PTSD-PRS, Re-experiencing-PRS, Generalized Anxiety-PRS, Schizophrenia-PRS, Depression-PRS, and Neuroticism-PRS) are predictive of WTC-PTSD outcomes: lifetime diagnoses, average symptom severity, and 18-year symptom trajectory. All analyses were adjusted for population stratification, 9/11 exposure severity, and multiple testing.
Results.
Depression-PRS predicted PTSD diagnostic status (OR 1.37, CI 1.17–1.61, adjusted p = 0.001). All PRSs, except PTSD-PRS, significantly predicted average PTSD symptoms (β = 0.06–0.10, adjusted p < 0.05). Re-experiencing-PRS, Generalized Anxiety-PRS and Schizophrenia-PRS predicted the high severity PTSD trajectory class (ORs 1.21–1.28, adjusted p < 0.05). Finally, PRSs prediction was independent of 9/11 exposure severity and jointly accounted for 3.7 times more variance in PTSD symptoms than the exposure severity.
Conclusions.
Psychiatric PRSs prospectively predicted WTC-related PTSD lifetime diagnosis, average symptom severity, and 18-year trajectory in responders to 9/11 disaster. Jointly, PRSs were more predictive of subsequent PTSD than the exposure severity. In the future, PRSs may help identify at-risk responders who might benefit from targeted prevention approaches.
Keywords: Polygenic risk score, posttraumatic stress disorder, trauma, orld trade center
Introduction
Post-traumatic stress disorder (PTSD) is a persistent and debilitating psychiatric condition (Kessler et al., 2017; Koenen et al., 2017). Symptoms of PTSD include re-experiencing of the trauma, avoidance of situations that bring back memories of the event, emotional numbing, and hyperarousal. Genetics hold promise of predicting who will develop PTSD following trauma, and how PTSD will unfold in terms of severity and persistence. The aim of the current study was to test whether polygenic biomarkers can be used to prospectively predict PTSD diagnosis, symptom severity, and 18-year course in responders to 9/11 World Trade Center (WTC) attacks.
PTSD has a complex multifactorial etiology, is moderately heritable, and genome-wide association studies (GWASs) indicate that multiple genetic variants contribute to PTSD (Duncan, Cooper, & Shen, 2018). Although individual variants associated with PTSD in discovery GWASs have small effects, cumulatively they explain a substantial proportion of genetic variation and can be collapsed into a polygenic risk score (PRS) to improve prediction of PTSD (Duncan et al., 2017; Gelernter et al., 2019; Nievergelt et al., 2019). Specifically, for each individual, the number of risk alleles carried at each variant (0, 1, or 2) can be summed and weighted by its effect size, resulting in a single score of each individual’s genetic vulnerability to a disorder. PRSs are usually computed with variants up to a certain effect size (i.e. p value threshold). The largest PTSD GWAS to date employed a sample of over 30 000 PTSD cases and 170 000 controls from 60 cohorts (Nievergelt et al., 2019). Moreover, an independent GWAS of PTSD re-experiencing symptoms was conducted by the US Million Veteran Program (MVP) in over 166 000 individuals (Gelernter et al., 2019). Both studies provide an unprecedented opportunity for deriving polygenic risk metrics for PTSD and examining the predictive utility of PTSD-related PRSs in clinical settings.
PRSs can be used across diagnostic boundaries, as PRSs developed for one disorder can significantly predict many other conditions (Docherty et al., 2018; Jones et al., 2018). This is in line with a well-documented genetic overlap among psychiatric disorders, indicating transdiagnostic genetic vulnerability to mental illness and a widespread pleiotropy (Lee et al., 2019; Smoller et al., 2018; Waszczuk et al., 2020). In particular, the Schizophrenia-PRS (Ripke et al., 2014) is among the most frequently used PRSs in psychiatry (Bogdan, Baranger, & Agrawal, 2018) and it was previously found to predict PTSD (R2 = 0.009) (Sumner, Duncan, Ratanatharathorn, Roberts, & Koenen, 2016). PTSD is also highly comorbid and shares significant molecular genetic covariance with depression and the personality trait neuroticism (rg = 0.62–0.80 and rg = 0.59–0.66, respectively), as well as with schizophrenia (rg = 0.34) (Nievergelt et al., 2019). Furthermore, generalized anxiety, depression, and neuroticism constitute major components of internalizing psychopathology alongside PTSD (Hettema, Neale, Myers, Prescott, & Kendler, 2006; Waszczuk, Kotov, Ruggero, Gamez, & Watson, 2017), and PRSs for these conditions are by far the most robust among internalizing problems due to the large GWAS discovery sample sizes (Ns > 200 000 participants) (Howard et al., 2019; Levey et al., 2020; Nagel et al., 2018). Therefore, PRSs for schizophrenia and internalizing psychopathology constitute plausible transdiagnostic markers of genetic vulnerability to PTSD.
Psychiatric PRSs derived in cross-sectional discovery GWASs can predict longitudinal trajectories of psychopathology (Rice et al., 2018). However, it remains unknown whether psychiatric PRSs can predict the course of PTSD. Furthermore, although genetic risk can encompass a wide range of environmental effects due to gene–environment correlations and interactions (Assary, Vincent, Keers, & Pluess, 2018; Avinun, 2019), PRSs have been found to predict depression independently of trauma (Lehto et al., 2019; Peyrot et al., 2018). Nonetheless, given that PTSD is contingent on trauma exposure, it remains unknown whether polygenetic risk contributes to the prediction of PTSD over and above exposures. Twin studies find that a significant proportion of genetic influences is common to trauma exposure and subsequent risk for developing PTSD (Sartor et al., 2011; Stein, Jang, Taylor, Vernon, & Livesley, 2002). Thus, PRSs could capture this broad genetic vulnerability, as well as influences specific to PTSD. Finally, psychiatric PRSs were derived from heterogeneous samples, and PTSD-PRS and Re-experiencing-PRS were additionally obtained in participants with a wide variety of trauma exposures. Thus, to gain an in-depth understanding of how PRSs relate to PTSD outcomes, it is imperative to relate markers of genetic risk to longitudinally-tracked PTSD, using cohorts with a common type of trauma exposure.
One such population are WTC responders, who were exposed to a major disaster and subsequently monitored for over 18 years (Dasaro et al., 2015). The rate of WTC-related PTSD is very high in this population – about 20%, and is associated with significant functional impairment (Bromet et al., 2016; Wisnivesky et al., 2011). The current study aims to test whether psychiatric PRSs predict long-term PTSD outcomes in 9/11 responders. We hypothesized that PRSs for PTSD and re-experiencing will predict a clinical diagnosis of WTC-related PTSD, greater PTSD symptom severity across all clusters: re-experiencing, avoidance, numbing, and hyperarousal (King, Leskin, King, & Weathers, 1998), and poorer symptom course over the 18-year post-exposure follow-up. In addition, given transdiagnostic genetic vulnerability to mental illness, we anticipated that PRSs for schizophrenia and the three internalizing phenotypes: depression, generalized anxiety, and neuroticism, will also predict PTSD outcomes. Finally, we hypothesized that all six PRSs will predict PTSD even after accounting for the severity of trauma exposure.
Methods
Participants
Participants were 1490 WTC responders enrolled in the Stony Brook University WTC Health Program (Dasaro et al., 2015). The Stony Brook program monitors more than 10 000 WTC responders primarily from Long Island, NY, USA. The mean age on 11 September 2001 was 38.81 years (S.D. = 8.20, range = 19–70 years); 93.5% of the sample was male, and 67.1% was employed in law enforcement on 9/11. All participants were of European Ancestry, with ancestry evaluated using genetic data. This is because PRSs are sensitive to ancestry (Martin et al., 2019), thus analyses were limited to individuals with ancestry matching the discovery GWASs. Participants were selected randomly into the project and do not differ from the total clinical population on other demographic characteristics and symptom levels.
Study timeline
Due to Centers for Disease Control and Prevention mandated rolling admission to the WTC Health Program, the first visit took place on average 7.06 years (S.D. = 3.98, range = 0.88–15.20) after 9/11. WTC exposure severity and the first available PTSD symptom score were obtained at the participant’s first monitoring visit. We note here that the associations of the year of first monitoring visit with exposure severity and first symptom score were non-significant. Across the follow-up period, participants attended on average 7.75 (S.D. = 2.92, range = 1–14) monitoring visits, at an average interval of 1.49 years (S.D. = 0.62, range = 0.95–12.61). PTSD symptom severity was assessed at each visit. As expected, the number of visits and duration of the intervals between visits were not associated with PTSD outcomes, as participants are invited to annual monitoring visits irrespective of their health status. Blood samples were drawn as a routine part of the monitoring examination at the WTC Health Program. The study was approved by the Institutional Reviewer Board of Stony Brook University, and all participants provided written informed consent.
Clinical measures
PTSD symptoms were assessed with the PTSD Checklist (PCL) – Specific Version (Weathers, Litz, Herman, Huska, & Keane, 1993). PCL-17 is a 17-item self-report questionnaire assessing, on a five-point scale (1 = not at all to 5 = extremely), the severity of WTC-related DSM-IV PTSD symptoms in the past month. The scale encompasses four PTSD clusters: re-experiencing, hyperarousal, avoidance, and numbing (King et al., 1998). PCL-17 demonstrates sound psychometric properties (Wilkins, Lang, & Norman, 2011) and had excellent internal consistency in the current sample (α = 0.96). For cross-sectional analyses, PTSD symptoms were defined as participants’ average scores across all available visits, and these data were available for N = 1474.
PTSD diagnosis was obtained by master’s level clinical assessors trained to administer selected modules of the Structured Clinical Interview for DSM-IV (SCID) (First, Spitzer, Gibbon, & Williams, 1998). The SCID was modified to assess PTSD symptoms related to traumatic WTC exposures (Criterion A). Participants reported on their worst episode since 9/11. Inter-rater agreement for 55 independently rated audio-tapes was very good (kappa ⩾ 0.82). The SCID was administered once, to a subset of participants (N = 891, 59.8% of the total sample, resulting in 212 lifetime WTC-related PTSD cases). The diagnostic interview was conducted on average 12.20 years (S.D. = 0.96, range = 10.33–15.07) after 9/11, as part of another study (Bromet et al., 2016). There were no significant differences in demographic characteristics between responders by SCID administration. However, individuals who were administered the SCID had slightly higher PCL score than those were not (mean = 27.5 v. 29.5, t(1405.8) = −3.04, p = 0.002, d = 0.16).
The 9/11 exposure severity was assessed by a clinician-administered interview at the first monitoring visit, and operationalized dimensionally using an established index composed of the total time spent working at the WTC site, exposure to the cloud of debris from the collapse of the WTC buildings, and work on the pile of debris (Wisnivesky et al., 2011). The highest trauma exposure group (2.9%) worked more than 90 days on the WTC site and at least some time on the pile, and was exposed to the dust cloud. The high exposure group (18.2%) was in the dust could, but either worked less than 90 days or did not work on the debris pile. The majority of the sample (61.5%) experienced an intermediate level of trauma exposure – no dust cloud exposure and either worked between 40 and 90 days or did not work on the pile. Finally, 17.3% of participants were in the low exposure group – worked less than 40 days at the WTC site and had neither dust cloud nor debris pile exposure.
Polygenic risk scores
Genotyping of blood samples was performed in a single batch at the Genomics Shared Resource at Roswell Park Cancer Institute, using the Infinium Global Screening Array (Illumina, San Diego, CA, USA), according to protocols of the manufacturer. Genotypes were imputed on the Michigan Imputation Server pipeline v1.2.4, using the Haplotype Reference Consortium reference panel (McCarthy et al., 2016). Before imputation, the genotypes were filtered for ambiguous strand orientation, missingness rate>5% (by marker exclusion, then by an individual), Hardy-Weinberg equilibrium violation (p < 10−6), sex mismatch, and non-European ancestry. After imputation, the SNPs were excluded for imputation R2 < 0.5 and average call rate below 90%. Genotype imputation was performed on 552 230 SNPs, resulting in 25 514 638 SNPs after QC which were used for the final polygenic risk scoring.
PRSs were created using PRSice 2.0 (Euesden, Lewis, & O’Reilly, 2015). Specifically, PTSD-PRS was calculated using summary statistics from the most recent Psychiatric Genetics Consortium (PGC) GWAS in 23 212 PTSD cases and 151 447 controls of European Ancestry (Nievergelt et al., 2019). The Re-experiencing-PRS (REXP-PRS) was based on the MVP GWAS conducted in over 166 000 individuals with continuous PCL re-experiencing symptom score (Gelernter et al., 2019). Likewise, the Generalized anxiety-PRS (GAD-PRS) comes from a GWAS in the MVP cohort of close to 200 000 veterans (Levey et al., 2020). The MVP sample is predominantly of European Ancestry. The Schizophrenia-PRS (SZ-PRS) was created using summary statistics from the PGC GWAS in 36 989 schizophrenia cases and 113 075 controls, predominantly of European Ancestry (Ripke et al., 2014). The Depression-PRS (DEP-PRS) was calculated using summary statistics from a GWAS meta-analysis on 246 363 cases and 561 190 controls of European Ancestry (Howard et al., 2019). Finally, the Neuroticism-PRS (N-PRS) was created using summary statistics from a GWAS meta-analysis in 449 484 individuals of European Ancestry with a neuroticism score (Nagel et al., 2018). None of these GWASs included genetic data from participants from the WTC cohort.
PRSs were created by aggregating genetic variants up to varying thresholds of significance from a GWAS discovery sample, weighted by the associations in the discovery sample. Our primary analysis used a full list of SNPs after clumping and their corresponding weights from GWAS discovery samples (p value threshold = 1), to incorporate more of the genome (Wray et al., 2014). See online Supplementary Table S1 for sensitivity analyses at other thresholds. All analyses were adjusted for the first 10 genetic ancestry principle components (PCs). See online Supplementary Table S2 for repeated analyses examining up to 100 PCs.
Analytic approach
Cross-sectional PRS predictions
All variables were standardized prior to analyses. Cross-sectional associations of each PRS with PTSD symptoms and diagnoses were tested using linear and logistic regressions, respectively. PTSD was a dependent variable, the first 10 principal components of the population structure were included in the first block as covariates, and a single PRS was an independent variable. In cross-sectional analyses, PTSD symptoms were analyzed both as a total scale, and separately as four symptom clusters. For illustrative purposes, we also computed effect sizes for PTSD per PRS quartiles relative to the first quartile. A false detection rate (FDR) of 5% was used to control for multiple comparisons. For sensitivity testing, all analyses were repeated with 9/11 exposure severity included as a covariate. Furthermore, regressions were conducted using employment in law enforcement, and the 9/11 exposure severity, as dependent variables, in order to test if these key covariates are predicted by psychiatric PRSs.
Incremental prediction by PRS and 9/11 exposure
In order to compare PTSD symptoms prediction by PRSs and 9/11 exposure severity, two hierarchical linear regression models were conducted. In each regression, 10 ancestry PCs were included in the first block. Next, in the first model, the 9/11 exposure severity was added in the second block, and the 6 psychiatric PRSs together in the third block, with R2 change test used to evaluate an incremental prediction by PRSs over and above the 9/11 exposures. In the second model, the order of the second and third blocks was reversed, to test an incremental prediction by the 9/11 exposures when accounting for six PRSs.
PRS prediction of PTSD trajectories
PTSD trajectories were characterized using growth mixture model analysis in Mplus, version 8 (Muthén & Muthén, 2007). This analysis was used to identify types of course in the longitudinal PTSD data, i.e. latent classes, and assign each participant to a latent class. In other words, participants were assigned to a longitudinal trajectory class depending on the pattern of all available PTSD symptom scores across the monitoring period. Models were run with an increasing number of classes specified, up to the point where model fit worsened or the model extracted a class too small for further analyses (set to N < 200), (see online Supplementary Table S3 for fit statistics and N per class). The model with N = 2 classes was selected because each class in this model was large enough for further analysis (N > 200) and readily interpretable, with the solution showing acceptable fit statistics. Full information maximum likelihood estimation was used to handle missing data. Next, we used logistic regression to estimate the associations between continuous PRSs and trajectory class, adjusting for the first 10 ancestry principal components of the population structure and 9/11 exposure severity.
Results
Descriptive statistics
Descriptive statistics for the analytic sample are reported in Table 1. Average PTSD symptom severity was significantly higher in participants with lifetime WTC-related PTSD diagnosis (mean = 24.43 v. 46.00, t(251.54) = −21.40, p = 1.49 × 10−58, d = 2.01). Accordingly, 77% of responders with an average PCL score indicative of probable PTSD also had a SCID diagnosis of WTC-PTSD.
Table 1.
Descriptive statistics
| Total PCL sample (N = 1474) | Healthy trajectory (N = 1219) | High PTSD trajectory (N = 255) | |||||
|---|---|---|---|---|---|---|---|
| Mean | S.D. | Range | Mean | S.D. | Mean | S.D. | |
| PTSD Total | 28.67 | 12.61 | 17–79 | 24.18 | 7.06 | 50.13 | 11.13 |
| PTSD Re-experiencing | 8.06 | 3.51 | 5–23.50 | 6.90 | 1.99 | 13.60 | 3.88 |
| PTSD Avoidance | 3.51 | 1.86 | 2–10 | 2.92 | 1.16 | 6.38 | 1.89 |
| PTSD Numbing | 7.78 | 3.78 | 5–24 | 6.50 | 2.13 | 13.92 | 3.89 |
| PTSD Hyperarousal | 9.28 | 4.30 | 5–25 | 7.84 | 2.76 | 16.13 | 3.55 |
| 9/11 Exposure severity | 1.06 | 0.68 | 0–3 | 1.05 | 0.68 | 1.17 | 0.69 |
| N | Valid % | N | Valid % | N | Valid % | ||
| WTC-PTSD diagnosis | 205 | 23.3% | 87 | 12.3% | 118 | 69.0% | |
| Law enforcement occupation | 989 | 67.1% | 844 | 69.2% | 145 | 56.9% | |
Note: The lifetime WTC-PTSD diagnosis is reported for the total N = 880 for whom both PCL and SCID were available. The total analytic sample for SCID data was N = 891 (212 cases and 679controls).
Cross-sectional PRS predictions
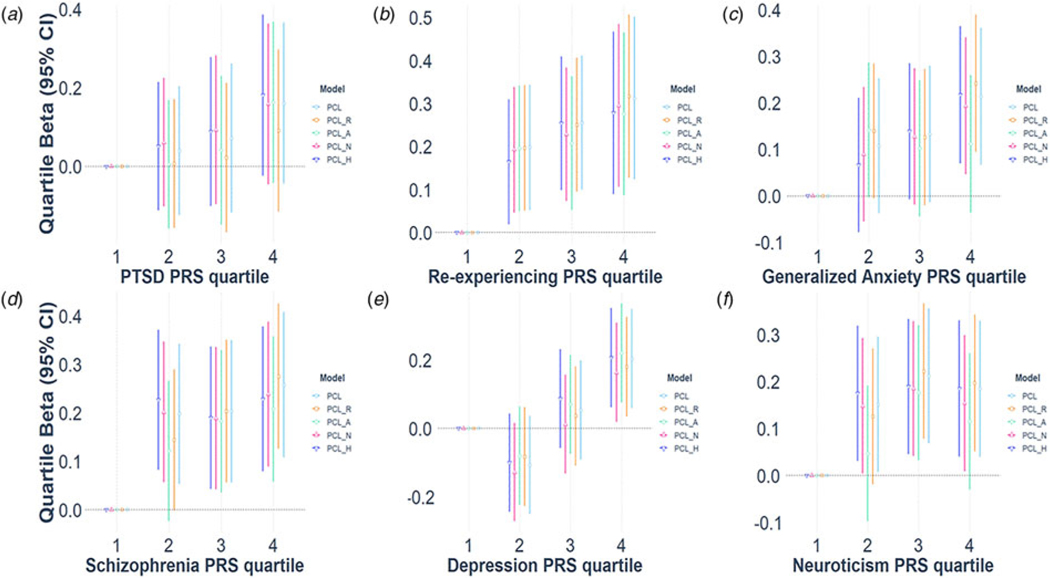
After multiple testing correction, PTSD-PRS did not significantly predict SCID diagnosis or any of the PTSD symptom dimensions (Table 2). In contrast, Re-experiencing-PRS significantly predicted total and subscale PTSD symptoms (β = 0.08–0.10), but not the diagnostic status. Moreover, PRSs for Schizophrenia, Generalized Anxiety, Depression, and Neuroticism significantly predicted PTSD symptoms, with Neuroticism-PRS effect sizes ranging β = 0.03–0.07 and Schizophrenia, Generalized Anxiety, and Depression PRSs effect sizes ranging β = 0.08–0.11. Notably, Neuroticism-PRS was not associated with the avoidance and numbing clusters, with avoidance also not associated with the Generalized Anxiety-PRS. Finally, Depression-PRS was the only genetic instrument predictive of PTSD diagnostic status (OR 1.37, CI1.17–1.61, adjusted p = 0.001, see Table 2).
Table 2.
PRS prediction of WTC-related PTSD symptoms and diagnostic status
| Symptoms: | PTSD-PRS | REXP-PRS | GAD-PRS | SZ-PRS | DEP-PRS | N-PRS | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| β | p | β | p | β | p | β | p | β | p | β | p | |
| PTSD Total | 0.064 | 0.126 | 0.096 | 0.011 | 0.094 | 0.002 | 0.103 | 0.001 | 0.099 | 0.001 | 0.059 | 0.040 |
| PTSD Re-experiencing | 0.046 | 0.253 | 0.094 | 0.012 | 0.100 | 0.001 | 0.110 | 0.001 | 0.088 | 0.003 | 0.067 | 0.021 |
| PTSD Avoidance | 0.071 | 0.116 | 0.079 | 0.035 | 0.051 | 0.090 | 0.082 | 0.008 | 0.094 | 0.002 | 0.035 | 0.216 |
| PTSD Numbing | 0.053 | 0.203 | 0.092 | 0.015 | 0.087 | 0.003 | 0.095 | 0.003 | 0.087 | 0.003 | 0.045 | 0.119 |
| PTSD Hyperarousal | 0.072 | 0.090 | 0.089 | 0.018 | 0.098 | 0.002 | 0.093 | 0.003 | 0.101 | 0.001 | 0.060 | 0.037 |
| OR (95% CI) | p | OR (95% CI) | p | OR (95% CI) | p | OR (95% CI) | p | OR (95% CI) | p | OR (95% CI) | p | |
| WTC-PTSD Diagnosis | 1.119 (0.892–1.405) | 0.356 | 1.010 (0.822–1.232) | 0.954 | 0.988 (0.846–1.154) | 0.902 | 1.148 (0.976–1.350) | 0.126 | 1.368 (1.165–1.607) | 0.001 | 1.109 (0.948–1.296) | 0.221 |
REXP, Re-experiencing symptoms of PTSD; GAD, Generalized Anxiety; SZ, Schizophrenia; DEP, Depression, N, Neuroticism.
Notes:
Linear regressions were conducted for symptoms (N = 1474), logistic regressions for diagnoses (N = 891).
All significant results at 5% FDR multiple testing correction are in bold.
Adjusting for 9/11 exposure severity did not affect significant findings.
Analyses were repeated for the PCL scores at the first available monitoring visit, see online Supplementary Table S4.
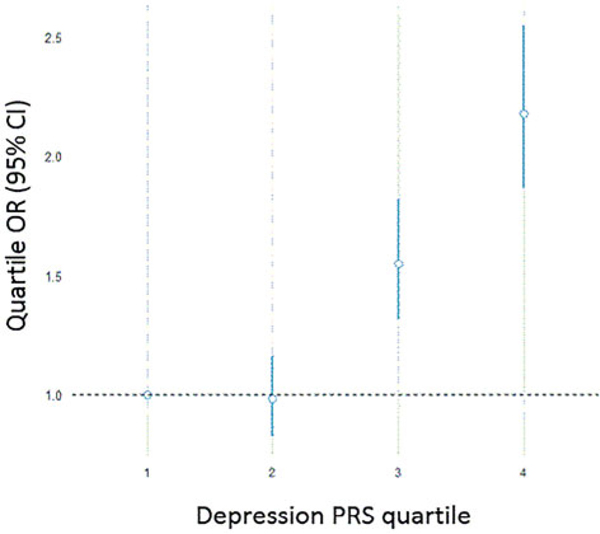
The pattern of associations was generally consistent at other PRS p value thresholds (online Supplementary Table S1) and when adjusting for a much larger number of ancestry PCs (online Supplementary Table S2). The analyses were also repeated for PTSD symptoms at the first available monitoring visit (online Supplementary Table S4). Furthermore, all associations remained significant after controlling for the 9/11 exposure severity. Figures 1 and 2 illustrate the effect sizes across PRS quartile, as compared with the lowest risk quartile. For example, responders in the 4th quartile of genetic risk for depression had over twice the odds of receiving a PTSD diagnosis than responders in the 1st quartile (Fig. 3e). Nonetheless, effect sizes were small and their increase across PRS quartiles was not monotonic in some cases. Finally, neither employment in law enforcement nor 9/11 exposure severity were predicted by any of the six PRSs.
Fig. 1.
Polygenic prediction of WTC-related PTSD symptoms.
Notes: R, Re-experiencing; A, Avoidance; N-Numbing; H, Hyperarousal. PRS predictions of PTSD symptoms are reported as standardized regression betas per PRS quartile, relative to the first quartile. For example, participants in the 4th quartile of genetic risk for depression had significantly higher PTSD symptoms than subjects in the 1st quartile.
Fig. 2.
Depression Polygenic Risk Score prediction of WTC-related PTSD diagnosis.
Notes: Depression-PRS prediction of PTSD diagnosis is illustrated as odds ratios per PRS quartile, relative to the first quartile. For example, responders in the highest 4th quartile of genetic risk for depression had over twice the odds of receiving a PTSD diagnosis than responders in the lowest 1st quartile of genetic risk.
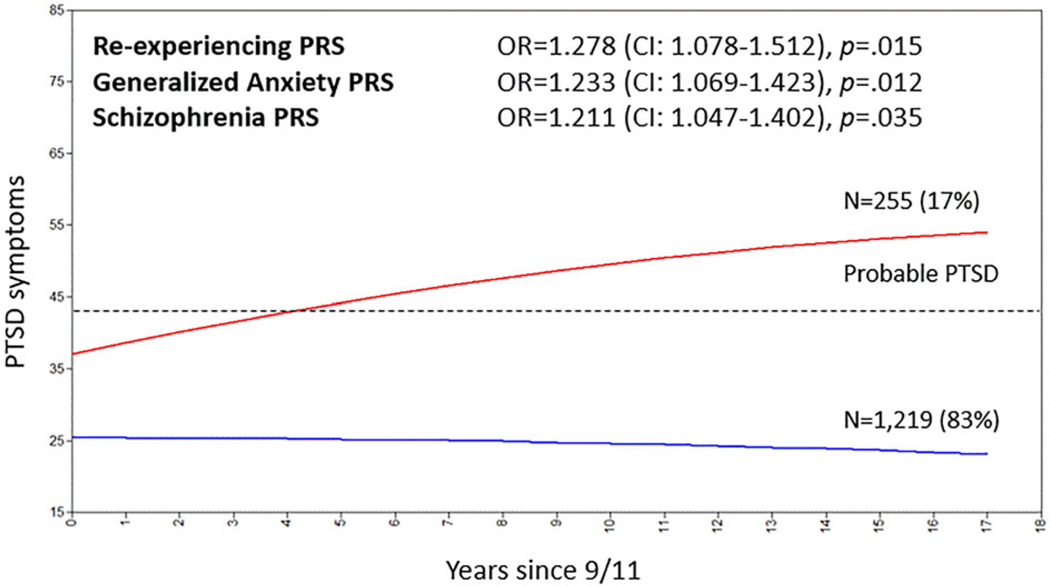
Fig. 3.
PTSD trajectory classes since 9/11, and prediction of class membership by Polygenic Risk Scores.
Notes: Analyses were repeated controlling for the 9/11 exposure severity. The three PRSs remained significant predictors of trajectory class membership, while exposure severity contributed independent prediction of comparable magnitude. These results are reported in online Supplementary Table S5. Results for PTSD-PRS, DEP-PRS, and N-PRS were non-significant (all adjusted p > 0.050) and are not illustrated. PTSD symptoms are reported as PCL score on a scale from 17 to 85. A score of 43 or above is indicative of probable PTSD (Freedy et al., 2010), represented with the dashed line.
Incremental prediction by PRS and 9/11 exposure
PRSs and 9/11 exposure severity constituted independent, significant predictors of PTSD severity, jointly accounting for 2.8% of variance in PTSD symptoms after 10 PCs were controlled (Table 3). However, PRSs predicted about four times more variance in PTSD than the 9/11 exposure severity (ΔR2 = 0.022 v. ΔR2 = 0.006).
Table 3.
Exposure severity and PRS incremental prediction of total WTC-related PTSD symptoms
| Predictors of PTSD | R2 | Δ R2 | p | |
|---|---|---|---|---|
| 10 PCs | 0.015 | |||
| Model 1 | 10 PCs + Exposure | 0.021 | 0.006 | 0.002 |
| 10 PCs + Exposure + 6 PRSs | 0.043 | 0.022 | 1.70 × 10−5 | |
| Model 2 | 10 PCs + 6 PRSs | 0.037 | 0.022 | 1.00 × 10−5 |
| 10 PCs + 6 PRSs + Exposure | 0.043 | 0.006 | 0.004 | |
Notes: In each hierarchical regression model, 10 ancestry PCs were included first to estimate R2. Then, each predictor was added in consecutive blocks, to estimate the change in R2 and thus determine whether each added predictor provides novel information about PTSD symptoms, over and above the predictors already included in the model. The predictors were entered in two orders (Model 1: exposure followed by PRSs, Model 2: PRSs followed by exposure).
In 6 PRSs block, PTSD PRS, Re-experiencing-PRS, GAD-PRS, Schizophrenia-PRS, Depression-PRS, and Neuroticism-PRS were entered simultaneously.
p value indicates the significance of ΔR2, testing whether including an additional predictor to the model significantly increased the prediction of PTSD symptoms.
PRS prediction of PTSD trajectories
Growth mixture modeling resulted in a two classes solution (online Supplementary Table S3). As expected, a majority of responders (83%) were resilient since 9/11 (Fig. 3). However, the remaining 17% showed a severe symptom trajectory. The severe symptom trajectory was generally within the threshold indicative of probable PTSD and responders in this group had a much higher probability of receiving a PTSD diagnosis (OR 15.92, CI10.73–23.61, p = 4.38 ×10−43), 69.0% of all individuals in the symptomatic class trajectory had a lifetime PTSD diagnosis, v. only 12.3% participants in the non-symptomatic trajectory receiving the diagnosis. Descriptive statistics per trajectory class are reported in Table 1.
Re-experiencing, Generalized Anxiety, and Schizophrenia PRSs predicted trajectory class membership (ORs 1.21–1.28, adjusted p < 0.05). The prediction was independent of 9/11 exposure severity (online Supplementary Table S5). However, PTSD, Depression, and Neuroticism PRSs were not associated with PTSD trajectory.
Discussion
The current study found that selected psychiatric polygenic biomarkers prospectively predicted WTC-related PTSD diagnosis, symptom severity, and long-term course in responders to 9/11 disaster. Clinical data collected over 18-years of monitoring provided an opportunity to take PTSD genetics beyond the case-control comparisons to longitudinal models, investigating how genetics related to the trajectory of PTSD after a traumatic event. The results indicated that Re-experiencing, Generalized Anxiety, and Schizophrenia PRSs were predictive of a severe PTSD symptom trajectory after 9/11. Moreover, these three PRSs, as well as Depression and Neuroticism PRSs, predicted WTC-related PTSD symptom severity. Depression-PRS additionally predicted PTSD diagnostic status. Finally, genetic vulnerability was not correlated with the severity of 9/11 trauma exposure, and instead contributed an independent and larger prediction of PTSD than the exposure severity. Overall, the current study is the first to demonstrate that existing polygenetic profiles can predict long-term PTSD outcomes in traumatized populations, although the effect sizes are small.
The current study is the first to use PRSs to predict long-term course of PTSD symptoms. The results indicate that Re-experiencing, Generalized Anxiety, and Schizophrenia PRSs significantly predicted a severe PTSD symptom trajectory since 9/11. The first two PRSs were based on the GWASs from the MVP project, indicating that the genetic discoveries from the US veteran population capture genetic vulnerability to PTSD in 9/11 responders population. There are notable demographic similarities between the two cohorts (e.g. predominantly male sex, occupation in uniformed services) that could contribute to this performance (Gaziano et al., 2016). Our results are also in line with one previous study that reported a cross-sectional association between Schizophrenia-PRS and PTSD symptoms (Sumner et al., 2016), extending this finding to the long-term course of PTSD.
Moreover, Re-experiencing, Generalized Anxiety, and Schizophrenia PRSs, alongside Depression and Neuroticism PRSs, significantly predicted average PTSD symptoms during the monitoring period. In particular, Depression-PRS emerged as a consistent cross-sectional predictor of PTSD, and in addition uniquely predicted PTSD diagnostic status obtained via diagnostic interview. The associations were the most prominent when contrasting the lowest and highest genetic risk groups, in line with the view that psychiatric PRSs may be particularly informative at the very high genetic susceptibility scores (Lewis & Vassos, 2017). This finding is in line with the known genetic overlap between PTSD and depression (Sartor et al., 2012), as well as with the evidence for common genetic vulnerability to internalizing psychopathology more broadly (Hettema et al., 2006).
Notably, the PRS derived for PTSD by the PGC (Nievergelt et al., 2019) was not predictive of this condition in the current sample, although the association was in the expected direction. This might be due to small effect sizes reported in this study, which may only reach significance when the PTSD-PRS is tested in very large samples (Gelernter et al., 2019). The null findings might also reflect sex differences in genetic influences on PTSD, such as higher heritability of PTSD in women (Duncan et al., 2018), and notably better performance of the PGC-PTSD-PRS in female samples (Nievergelt et al., 2019). The sex differences in the genetic architecture of PTSD could also affect associations with other PRSs. Finally, cross-sectional associations with PRSs were comparable across all PTSD symptom clusters, although only three PRSs significantly predicted avoidance symptoms. Overall, the current results show little phenotype-specificity in PRS prediction and instead support transdiagnostic applications of PRSs in translational studies. This is consistent with the notion of shared genetic vulnerability across diagnostic categories (Smoller et al., 2018; Waszczuk et al., 2020). Emerging cross-disorder analyses suggest that pleiotropic genes are uniquely involved in neurodevelopmental processes, such as neurogenesis and neuron differentiation (Lee et al., 2019).
The six selected PRSs and 9/11 trauma exposure severity constituted independent, significant predictors of PTSD. Notably, PRSs accounted for approximately four times more variance in PTSD than exposure severity. This novel finding suggests that at the point of trauma exposure, genetic vulnerability to psychopathology may be a better indicator of long-term PTSD outcomes than the severity of trauma. Nonetheless, the trauma exposure is by definition necessary for PTSD onset and an inclusion of both sources of information maximizes prediction. The weak association between trauma exposure severity and WTC-related PTSD outcome has been reported previously in this population and reflects a multifactorial etiology of enduring PTSD (Luft et al., 2012). Finally, the lack of a significant association between exposure severity and any of the PRSs argues against the gene–environment correlation, i.e. genetic vulnerability to psychopathology contributing to higher levels of trauma exposures. This is in line with some (Peterson et al., 2018), but not all (Dunn et al., 2016; Peyrot et al., 2018) molecular studies of the gene–environment correlation in depression. Nonetheless, a gene–environment correlation could still occur for selection into responder occupations or for experiencing the trauma exposure itself.
There was no association between genetic vulnerability to psychiatric conditions and employment in law enforcement occupations. Yet in the WTC cohort, police officers show more resilient outcomes than non-traditional responders (Bromet et al., 2016; Perrin et al., 2007; Wisnivesky et al., 2011), in line with findings from other law enforcement samples (Renck, Weisaeth, & Skarbö, 2002). The current results suggest that these differences are not due to lower genetic vulnerability to PTSD or other psychiatric traits in police officers. Instead, this occupational group might report fewer PTSD symptoms due to resilience training and preparedness for disasters, ‘selective survival’ of resilient police officers remaining in the law enforcement, or underreporting due to stigma (Perrin et al., 2007).
An improved understanding of PTSD genetics holds promise for reducing the burden of this disorder in first responders. While currently the PRS effect sizes are too small to be clinically informative, larger discovery sample sizes will result in more predictive PRSs that may be useful for risk stratification at the extreme end of the distribution., Moreover, integrating genotype data with patient’s electronic medical records, and combining polygenic information with exposure severity and other established clinical predictors, may enhance screening algorithms to better identify high-risk individuals vulnerable to PTSD (Schultebraucks et al., 2020). Together, these approaches might in the future help identify those who would benefit from more frequent screenings and interventions such as resilience training (Bogdan et al., 2018; Lewis & Vassos, 2020; Torkamani, Wineinger, & Topol, 2018). However, they also bring about ethical considerations, including the potential for unnecessary stress due to deterministic interpretations of genetic influences or false-positive results of screening algorithms, as well as a risk for genetic discrimination.
Limitations
The current study has several strengths, including a prospective design with repeated clinical assessments spanning 18 years following common trauma exposure, in a large cohort. Nonetheless, some limitations are notable. First, the proportion of individuals with ancestry admixture in the WTC cohort is too small to allow genetic analyses in non-Northern European ancestries. Thus, the current findings and potential clinical implications arising from this study and other work building on it may not generalize to individuals of other ancestries. This limitation occurs in the context of psychiatric PRSs having been primarily developed in samples of Northern European ancestry and performing more poorly in individuals from other ancestries (Martin et al., 2019). The low generalizability of genetic tools across diverse populations may exacerbate health disparities. One solution would be additional discovery GWAS including ancestry admixture, as with new methods that have recently been demonstrated to improve prediction in schizophrenia (Bigdeli et al., 2019). In turn, these new PRSs should be tested in longitudinal epidemiological samples composed of individuals of corresponding ancestries, including diverse trauma-exposed populations.
Second, the WTC responder population is mostly male, and the number of females was insufficient to explore sex differences. While there are differences in PTSD heritability between males and females that could have affected the observed associations (Duncan et al., 2018), on the whole, psychiatric PRSs have been demonstrated to perform well in both sexes. Third, we did not investigate the interaction between genetic vulnerability and trauma exposure severity (gene–environment interaction). While of interest to the field, this research question has led to notable non-replications (Assary et al., 2018; Lehto et al., 2019), including no meta-analytic evidence for the interaction between Depression-PRS and childhood trauma (Peyrot et al., 2018). We were also underpowered to detect very small effect sizes.
Fourth, we did not have data on other trauma exposures or potential PTSD treatment. These unmeasured sources of heterogeneity might have affected PTSD symptoms and trajectories, as well as associations with PRSs. Finally, studies focused on delineating PTSD course in WTC cohorts report more than two trajectory classes (Bromet et al., 2016; Pietrzak et al., 2014). However, subdividing the current sample into additional, more nuanced PTSD trajectories resulted in classes with less than 200 participants, which is underpowered for genetic analyses. Nonetheless, the 2-class conceptualization is in line with previous studies broadly differentiating between a healthy majority, and a minority of responders with severe PTSD courses across the post 9/11 follow-up period.
Conclusion
Five psychiatric PRSs – Re-experiencing, Generalized Anxiety, Schizophrenia, Depression, and Neuroticism PRSs – prospectively predicted PTSD outcomes across the 18-years of post-trauma monitoring in responders to the 9/11 disaster. In this cohort, the Re-experiencing, Generalized Anxiety, and Schizophrenia PRSs in particular emerged as the best predictors of long-term PTSD course following trauma exposure. The current effect sizes are small, but with additional research, PRSs may eventually be translated to first responder cohorts and help to identify individuals who might benefit from a more thorough medical monitoring.
Supplementary Material
Acknowledgements.
The study was funded by the NIOSH/CDC awards: U01OH011864 to Monika Waszczuk and Roman Kotov, 200–2017-93432, U01OH010718, U01OH010416 to Benjamin Luft, and 200–2011-39410 to Evelyn Bromet, Roman Kotov, and Benjamin Luft, NIMH K01MH093731 to Anna Docherty, and a Brain & Behavior Research Foundation grant to Andrey Shabalin. We thank participants for their commitment to the project. We are grateful to Drs Stein, Gelernter, and Levey for kindly sharing summary statistics from the Million Veteran Program. The findings and conclusions in this article are those of the authors and do not represent the official position of NIOSH, CDC, NIMH, or Brain & Behavior Research Foundation.
Footnotes
Ethical standards. The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
Supplementary material. The supplementary material for this article can be found at https://doi.org/10.1017/S0033291720003839
Conflict of interest. The authors declare no conflicts of interest.
References
- Assary E, Vincent JP, Keers R, & Pluess M. (2018). Gene-environment interaction and psychiatric disorders: Review and future directions. Seminars in cell & Developmental Biology, 77, 133–143. [DOI] [PubMed] [Google Scholar]
- Avinun R. (2019). The E Is in the G: Gene–environment–trait correlations and findings from genome-wide association studies. Perspectives on Psychological Science, 15(1), 81–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bigdeli TB, Genovese G, Georgakopoulos P, Meyers JL, Peterson RE, Iyegbe CO, … Pato CN (2019). Contributions of common genetic variants to risk of schizophrenia among individuals of African and Latino ancestry. Molecular Psychiatry, 25(10), 2455–2467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bogdan R, Baranger DA, & Agrawal A. (2018). Polygenic risk scores in clinical psychology: Bridging genomic risk to individual differences. Annual Review of Clinical Psychology, 14, 119–157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bromet E, Hobbs M, Clouston S, Gonzalez A, Kotov R, & Luft B. (2016). DSM-IV post-traumatic stress disorder among World Trade Center responders 11–13 years after the disaster of 11 September 2001 (9/11). Psychological Medicine, 46(04), 771–783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dasaro CR, Holden WL, Berman KD, Crane MA, Kaplan JR, Lucchini RG, … Todd AC (2015). Cohort profile: World trade center health program general responder cohort. International Journal of Epidemiology, 46(2), e9–e9. doi: 10.1093/ije/dyv099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Docherty AR, Moscati A, Dick D, Savage JE, Salvatore JE, Cooke M, … Riley BP (2018). Polygenic prediction of the phenome, across ancestry, in emerging adulthood. Psychological Medicine, 48(11), 1814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan L, Cooper BN, & Shen H. (2018). Robust findings from 25 years of PSTD genetics research. Current Psychiatry Reports, 20(12), 115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan L, Ratanatharathorn A, Aiello AE, Almli LM, Amstadter AB, Ashley-Koch AE, … Koenen KC (2017). Largest GWAS of PTSD (N = 20 070) yields genetic overlap with schizophrenia and sex differences in heritability. Molecular Psychiatry, 23(3), 666–673. doi: 10.1038/mp.2017.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dunn EC, Wiste A, Radmanesh F, Almli LM, Gogarten SM, Sofer T, … Weir DR (2016). Genome-wide association study (GWAS) and genome-wide by environment interaction study (GWEIS) of depressive symptoms in African American and Hispanic/Latina women. Depression and Anxiety, 33(4), 265–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Euesden J, Lewis CM, & O’Reilly PF (2015). PRSice: Polygenic risk score software. Bioinformatics (Oxford, England), 31(9), 1466–1468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- First MB, Spitzer RL, Gibbon M, & Williams JB (1998). Structured Clinical Interview for DSM-IV Axis I Disorders: Patient Edition (February 1996 Final), SCID-I/P: Biometrics Research Department, New York: State Psychiatric Institute. [Google Scholar]
- Freedy JR, Steenkamp MM, Magruder KM, Yeager DE, Zoller JS, Hueston WJ, & Carek PJ (2010). Post-traumatic stress disorder screening test performance in civilian primary care. Family Practice, 27(6), 615–624. [DOI] [PubMed] [Google Scholar]
- Gaziano JM, Concato J, Brophy M, Fiore L, Pyarajan S, Breeling J, … Humphries D. (2016). Million Veteran Program: A mega-biobank to study genetic influences on health and disease. Journal of Clinical Epidemiology, 70, 214–223. [DOI] [PubMed] [Google Scholar]
- Gelernter J, Sun N, Polimanti R, Pietrzak R, Levey DF, Bryois J, … Radhakrishnan K. (2019). Genome-wide association study of post-traumatic stress disorder reexperiencing symptoms in> 165 000 US veterans. Nature Neuroscience, 22(9), 1394–1401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hettema JM, Neale MC, Myers JM, Prescott CA, & Kendler KS (2006). A population-based twin study of the relationship between neuroticism and internalizing disorders. American Journal of Psychiatry, 163(5), 857–864. [DOI] [PubMed] [Google Scholar]
- Howard DM, Adams MJ, Clarke T-K, Hafferty JD, Gibson J, Shirali M, … Wigmore EM (2019). Genome-wide meta-analysis of depression identifies 102 independent variants and highlights the importance of the prefrontal brain regions. Nature Neuroscience, 22(3), 343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones HJ, Heron J, Hammerton G, Stochl J, Jones PB, Cannon M, … Linden DE (2018). Investigating the genetic architecture of general and specific psychopathology in adolescence. Translational Psychiatry, 8(1), 145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kessler RC, Aguilar-Gaxiola S, Alonso J, Benjet C, Bromet EJ, Cardoso G, … Ferry F. (2017). Trauma and PTSD in the WHO world mental health surveys. European Journal of Psychotraumatology, 8(Suppl 5), 1353383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King DW, Leskin GA, King LA, & Weathers FW (1998). Confirmatory factor analysis of the clinician-administered PTSD Scale: Evidence for the dimensionality of posttraumatic stress disorder. Psychological Assessment, 10(2), 90. [Google Scholar]
- Koenen K, Ratanatharathorn A, Ng L, McLaughlin K, Bromet E, Stein D, … Scott K. (2017). Posttraumatic stress disorder in the world mental health surveys. Psychological Medicine, 47(13), 2260–2274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee PH, Anttila V, Won H, Feng Y-CA, Rosenthal J, Zhu Z, … Posthuma D. (2019). Genomic relationships, novel loci, and pleiotropic mechanisms across eight psychiatric disorders. Cell, 179(7), 1469–1482, e1411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehto K, Hägg S, Lu D, Karlsson R, Pedersen NL, & Mosing MA (2019). Childhood adoption and mental health in adulthood: The role of gene-environment correlations and interactions in the UK Biobank. Biological Psychiatry, 87(8), 708–716. [DOI] [PubMed] [Google Scholar]
- Levey DF, Gelernter J, Polimanti R, Zhou H, Cheng Z, Aslan M, … Bryois J. (2020). Reproducible genetic risk loci for anxiety: Results from∼ 200000 participants in the million veteran program. American Journal of Psychiatry, 177(3), 223–232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis CM, & Vassos E. (2017). Prospects for using risk scores in polygenic medicine. Genome Medicine, 9(1), 96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis CM, & Vassos E. (2020). Polygenic risk scores: From research tools to clinical instruments. Genome Medicine, 12, 1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luft B, Schechter C, Kotov R, Broihier J, Reissman D, Guerrera K, … Friedman-Jimenez G. (2012). Exposure, probable PTSD and lower respiratory illness among World Trade Center rescue, recovery and clean-up workers. Psychological Medicine, 42(05), 1069–1079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin AR, Kanai M, Kamatani Y, Okada Y, Neale BM, & Daly MJ (2019). Clinical use of current polygenic risk scores may exacerbate health disparities. Nature Genetics, 51(4), 584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCarthy S, Das S, Kretzschmar W, Delaneau O, Wood AR, Teumer A, … Sharp K. (2016). A reference panel of 64 976 haplotypes for genotype imputation. Nature Genetics, 48(10), 1279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muthén L, & Muthén B. (2007). Mplus. Statistical analysis with latent variables. Version, 3. [Google Scholar]
- Nagel M, Jansen PR, Stringer S, Watanabe K, de Leeuw CA, Bryois J, … Muñoz-Manchado AB (2018). Meta-analysis of genome-wide association studies for neuroticism in 449484 individuals identifies novel genetic loci and pathways. Nature Genetics, 50(7), 920. [DOI] [PubMed] [Google Scholar]
- Nievergelt CM, Maihofer AX, Klengel T, Atkinson EG, Chen C-Y, Choi KW, … Gelernter J. (2019). International meta-analysis of PTSD genome-wide association studies identifies sex-and ancestry-specific genetic risk loci. Nature Communications, 10(1), 1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perrin MA, DiGrande L, Wheeler K, Thorpe L, Farfel M, & Brackbill R. (2007). Differences in PTSD prevalence and associated risk factors among World Trade Center disaster rescue and recovery workers. American Journal of Psychiatry, 164(9), 1385–1394. [DOI] [PubMed] [Google Scholar]
- Peterson RE, Cai N, Dahl AW, Bigdeli TB, Edwards AC, Webb BT, … Kendler KS (2018). Molecular genetic analysis subdivided by adversity exposure suggests etiologic heterogeneity in major depression. American Journal of Psychiatry, 175(6), 545–554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peyrot WJ, Van der Auwera S, Milaneschi Y, Dolan CV, Madden PA, Sullivan PF, … Nivard MG (2018). Does childhood trauma moderate polygenic risk for depression? A meta-analysis of 5765 subjects from the psychiatric genomics consortium. Biological Psychiatry, 84(2), 138–147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pietrzak R, Feder A, Singh R, Schechter C, Bromet EJ, Katz C, … Crane M. (2014). Trajectories of PTSD risk and resilience in World Trade Center responders: An 8-year prospective cohort study. Psychological Medicine, 44(1), 205–219. [DOI] [PubMed] [Google Scholar]
- Renck B, Weisaeth L, & Skarbö S. (2002). Stress reactions in police officers after a disaster rescue operation. Nordic Journal of Psychiatry, 56(1), 7–14. [DOI] [PubMed] [Google Scholar]
- Rice F, Riglin L, Thapar AK, Heron J, Anney R, O’donovan MC, … Thapar A. (2018). Characterizing developmental trajectories and the role of neuropsychiatric genetic risk variants in early-onset depression. JAMA Psychiatry, 76(3), 306–313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ripke S, Neale BM, Corvin A, Walters JT, Farh K-H, Holmans PA, … Huang H. (2014). Biological insights from 108 schizophrenia-associated genetic loci. Nature, 511(7510), 421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sartor CE, Grant JD, Lynskey MT, McCutcheon VV, Waldron M, Statham DJ, … Martin NG (2012). Common heritable contributions to low-risk trauma, high-risk trauma, posttraumatic stress disorder, and major depression. Archives of General Psychiatry, 69(3), 293–299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sartor CE, McCutcheon V, Pommer N, Nelson E, Grant J, Duncan AE, … Heath A. (2011). Common genetic and environmental contributions to post-traumatic stress disorder and alcohol dependence in young women. Psychological Medicine, 41(7), 1497–1505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schultebraucks K, Shalev AY, Michopoulos V, Grudzen CR, Shin S-M, Stevens JS, … Galatzer-Levy IR (2020). A validated predictive algorithm of post-traumatic stress course following emergency department admission after a traumatic stressor. Nature Medicine, 26(7), 1084–1088. doi: 10.1038/s41591-020-0951-z. [DOI] [PubMed] [Google Scholar]
- Smoller JW, Andreassen OA, Edenberg HJ, Faraone SV, Glatt SJ, & Kendler KS (2018). Psychiatric genetics and the structure of psychopathology. Molecular Psychiatry, 24(3), 409–420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein MB, Jang KL, Taylor S, Vernon PA, & Livesley WJ (2002). Genetic and environmental influences on trauma exposure and post-traumatic stress disorder symptoms: A twin study. American Journal of Psychiatry, 159(10), 1675–1681. [DOI] [PubMed] [Google Scholar]
- Sumner JA, Duncan L, Ratanatharathorn A, Roberts AL, & Koenen KC (2016). PTSD has shared polygenic contributions with bipolar disorder and schizophrenia in women. Psychological Medicine, 46(3), 669–671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torkamani A, Wineinger NE, & Topol EJ (2018). The personal and clinical utility of polygenic risk scores. Nature Reviews Genetics, 19(9), 581–590. [DOI] [PubMed] [Google Scholar]
- Waszczuk MA, Eaton NR, Krueger RF, Shackman AJ, Waldman ID, Zald DH, … Kotov R. (2020). Redefining phenotypes to advance psychiatric genetics: Implications from hierarchical taxonomy of psychopathology. Journal of Abnormal Psychology, 129(2), 143–161. doi: 10.31234/osf.io/sf46g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waszczuk MA, Kotov R, Ruggero CJ, Gamez W, & Watson D. (2017). Hierarchical structure of internalizing psychopathology: From individual symptoms to the spectrum. Journal of Abnormal Psychology, 126(5), 613–634. [DOI] [PubMed] [Google Scholar]
- Weathers FW, Litz BT, Herman DS, Huska JA, & Keane TM (1993). The PTSD Checklist (PCL): Reliability, validity, and diagnostic utility. Paper presented at the Annual Convention of the International Society for Traumatic Stress Studies. [Google Scholar]
- Wilkins KC, Lang AJ, & Norman SB (2011). Synthesis of the psychometric properties of the PTSD checklist (PCL) military, civilian, and specific versions. Depression and Anxiety, 28(7), 596–606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wisnivesky JP, Teitelbaum SL, Todd AC, Boffetta P, Crane M, Crowley L, … Herbert R. (2011). Persistence of multiple illnesses in World Trade Center rescue and recovery workers: A cohort study. The Lancet, 378(9794), 888–897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wray NR, Lee SH, Mehta D, Vinkhuyzen AA, Dudbridge F, & Middeldorp CM (2014). Research review: Polygenic methods and their application to psychiatric traits. Journal of Child Psychology and Psychiatry, 55(10), 1068–1087. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.