Highlights
Comprehensive summary of crystalline structures and morphologies of carbon nitride-based materials (CNBMs).
Density functional theory computation for the design of functional CNBMs for rechargeable battery applications.
The experimental synthesis strategies of CNBMs for rechargeable battery application.
Keywords: Carbon nitrides, Metal-ion batteries, Density functional theory, g-C3N4, Anode
Abstract
Carbon nitrides (including CN, C2N, C3N, C3N4, C4N, and C5N) are a unique family of nitrogen-rich carbon materials with multiple beneficial properties in crystalline structures, morphologies, and electronic configurations. In this review, we provide a comprehensive review on these materials properties, theoretical advantages, the synthesis and modification strategies of different carbon nitride-based materials (CNBMs) and their application in existing and emerging rechargeable battery systems, such as lithium-ion batteries, sodium and potassium-ion batteries, lithium sulfur batteries, lithium oxygen batteries, lithium metal batteries, zinc-ion batteries, and solid-state batteries. The central theme of this review is to apply the theoretical and computational design to guide the experimental synthesis of CNBMs for energy storage, i.e., facilitate the application of first-principle studies and density functional theory for electrode material design, synthesis, and characterization of different CNBMs for the aforementioned rechargeable batteries. At last, we conclude with the challenges, and prospects of CNBMs, and propose future perspectives and strategies for further advancement of CNBMs for rechargeable batteries.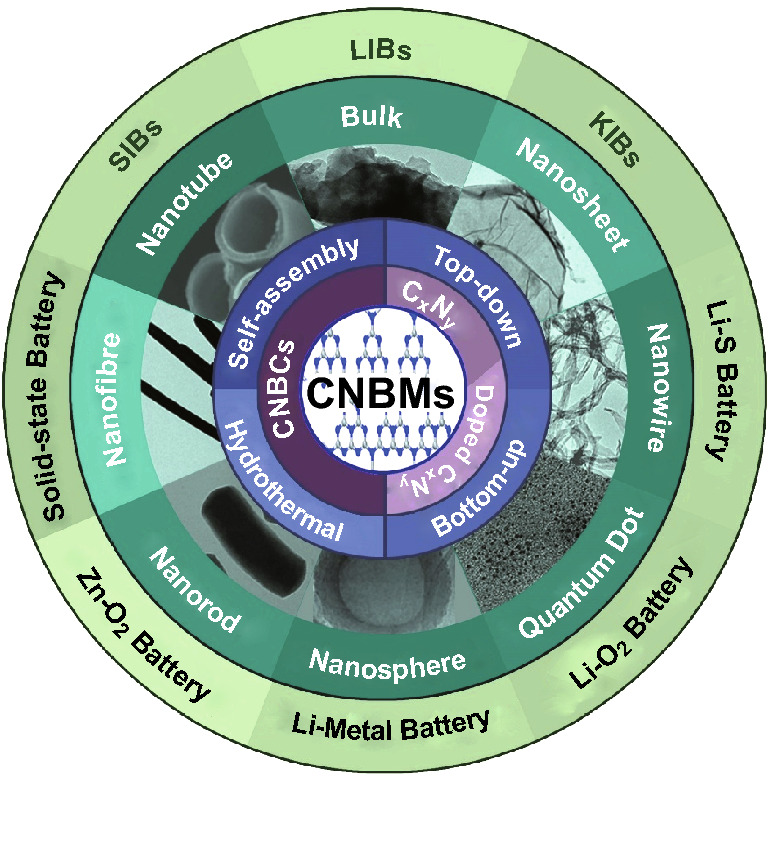
Introduction
Rechargeable metal ion batteries (MIBs) are one of the most reliable portable energy storage devices today because of their high power density, exceptional energy capacity, high cycling stability, and low self-discharge [1, 2]. Lithium-ion batteries (LIBs) remain the most developed and commercially viable alternative among all rechargeable batteries, and graphite is widely accepted as a preferred negative electrode (anode) material for LIBs [3]. Graphite is affordable, and it does not suffer significant volumetric changes (compared to other metal-based electrodes); it operates at a very low voltage close to that of lithium metal (~ 0.1 V) and exhibits moderately stable cycle life with a theoretical capacity of 372 mAh g (LiC6). Despite these positive characteristics, the performance of graphite is still very limited, and one of the effective ways to modulate its properties and electrochemical performance is through nitrogen doping [4]. Precisely, the work of Liu et al. showed that the N-doping of graphene results in improved conductivity and charge transfer, which boosts its performance and cyclability [5].
Carbon nitrides are a family of nitrogen-rich graphite analogues which contain a high nitrogen content and porous defect sites for effective charge transfer in energy storage devices [6, 7]. However, carbon nitrides are limited by poor electrical conductivity, chemical inertness, and ineffective intercalation/deintercalation process [8]. Due to these issues, several research studies have focused on the design of unique carbon-nitride-based materials (CNBMs), including pure carbon nitrides, doped carbon nitrides (DCNs) as well as carbon-nitride-based composites (CNBCs) [9]. Most of these reports have focused on g-C3N4 because it is easy to synthesize, low cost, environmentally safe, and it has a theoretical capacity of 524 mAh g−1 (Li2C3N4). In contrast, though other carbon nitride structures (such as CN, C2N, C3N, C4N, and C5N) which have been studied through density functional theory (DFT) calculations exhibit exceptional structural and electronic properties which demonstrates their capabilities as promising MIB electrode materials, they have attracted less attention.
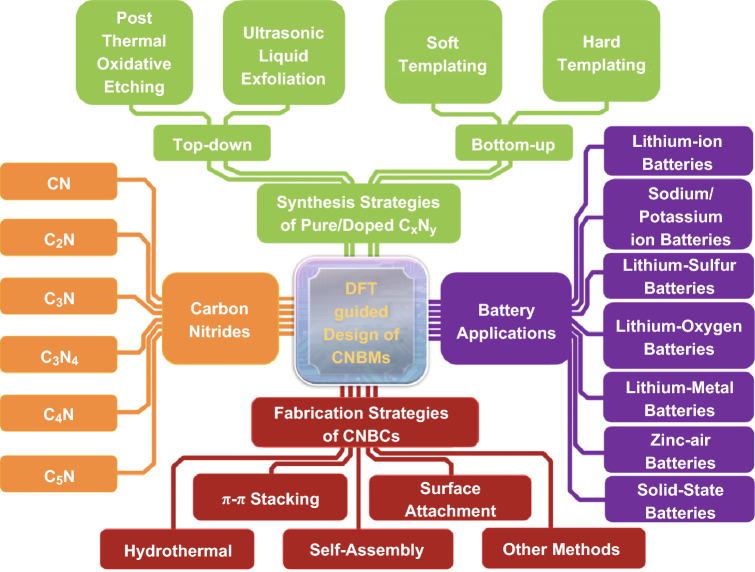
On the other hand, DFT has been adopted more and more in studying CNBMs and their electronic properties [10]. In fact, DFT could provide effective guidance for the synthesis of electrode materials and/or interpretation of the structure–performance relationship in energy storage devices, including LIBs, sodium- and potassium-ion batteries (SIBs and PIBs), lithium-sulfur batteries (Li–S), lithium-oxygen (Li-O2) batteries, lithium metal batteries (LMBs), zinc air batteries (ZABs), and solid-state batteries (SSBs). This review aims to comprehensively discuss the relationships between the structural and electronic properties and the MIB performance of pure carbon nitrides, doped carbon nitrides, and CNBCs, summaries the theoretical computation for the design of functional CNBMs for different rechargeable MIB applications, and generalizes the synthesis strategies of pure carbon nitrides and CNBCs for rechargeable metal ion battery application (Fig. 1). At the end of this work, we also offer a perspective on the existing challenges of carbon nitrides for energy storage devices and relevant resolving strategies.
Fig. 1.
Overview of the main topics of this work, including DFT-guided design (symbolized by the “CPU” at the center), the molecular configuration of carbon nitrides (yellow block), the synthesis strategies of pure/doped carbon nitrides (green block), the fabrication strategies of CNBCs (red block) and the battery applications of CNBMs (purple block). (Color figure online)
CNBMs
To study the carbon nitrides, we must begin by exploring their crystalline structure, structural and electronic properties, and functionality. Carbon nitrides are a family of nitrogen-rich carbon materials, and they have different crystalline structures and molecular configurations. Generally, there are seven types of nitrogen species (Fig. 2), with at least two most common species of nitrogen (the graphitic-N and pyridinic-N). The nature of the nitrogen and its percentage concentration has been proven to impact the electronic configuration and characteristic of the carbon nitride for different applications. The classification of carbon nitrides is often based on the carbon to nitrogen content (i.e., the C/N ratio) which is often associated with the degree of surface defects in their structure [11]. The carbon nitrides discussed in this review are categorized based on the dominant N species in their crystalline structure.
Fig. 2.
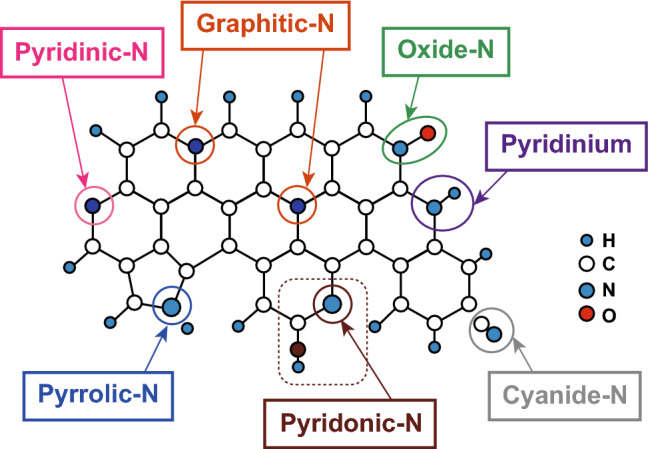
Common forms of nitrogen species in nitrogen-doped carbon materials. Reproduced with permission from Ref. [12]. Copyright permissions from Wiley–VCH. (Color figure online)
Pure Carbon Nitrides
Based on their generic family molecular formula of CxNy, the carbon nitride family includes CN (C2N2, C3N3, C4N4), C2N, C3N, C3N2, C3N4, C3N5, C3N6, C3N7, C4N, C4N3, C5N, C6N6, C6N8, C9N4, C9N7, C10N3, C10N9, and C14N12 [11, 13]. All these carbon nitride materials possess some similarity with graphene in that they are all two-dimensional (2D) materials with sp2/sp3 hybridized conjugated C atoms [14], but they all exhibit different structural frameworks and C/N ratio. In this section of the review, we summarize the crystalline structure, surface functionalities, and electronic properties of different carbon nitrides, including pyridinic nitrogen-based carbon nitrides (CN, C2N, and C3N4) and graphitic nitrogen-based carbon nitrides (C3N, C4N, and C5N) (Fig. 3).
Fig. 3.
Geometric structures of the pyridinic and graphitic-N-based carbon nitrides reported for rechargeable batteries and their surface functionalities. The brown spheres represent carbon atoms, and the light blue one represents nitrogen atoms in the 2D carbon nitrides structures. (Color figure online)
Pyridinic Nitrogen-Based Carbon Nitrides (CN, C2N, C3N4)
Pyridinic-N is one of the edge site nitrogen species in an N-doped carbon material or carbon nitride. It is bonded to two carbon atoms and supplies one p electron to the π–π conjugated system of carbon [12]. Due to the co-ordination of pyridinic-N to 2 carbon atoms, it donates 4 electrons to the sp2 of each carbon atom along with a lone pair. Because of its superior electronegativity, it is able to pull π-electrons from the conjugated system of carbon, thereby making it negatively charged N-atom. When the π-conjugated system of carbon is broken (the aromaticity), a non-bonding pz orbital is formed, and this orbital can also be observed around defected carbon materials. Carbon materials that contain pyridinic-N can function as Lewis base because the pyridinic-N exhibits a negative charge on surrounding carbon atom, which makes them positively charged with a lone pair of electrons as a result of the non-bonding pz orbital [12]. The pyridinic-N-based carbon nitrides in this category include CN, C2N, and C3N4.
CN
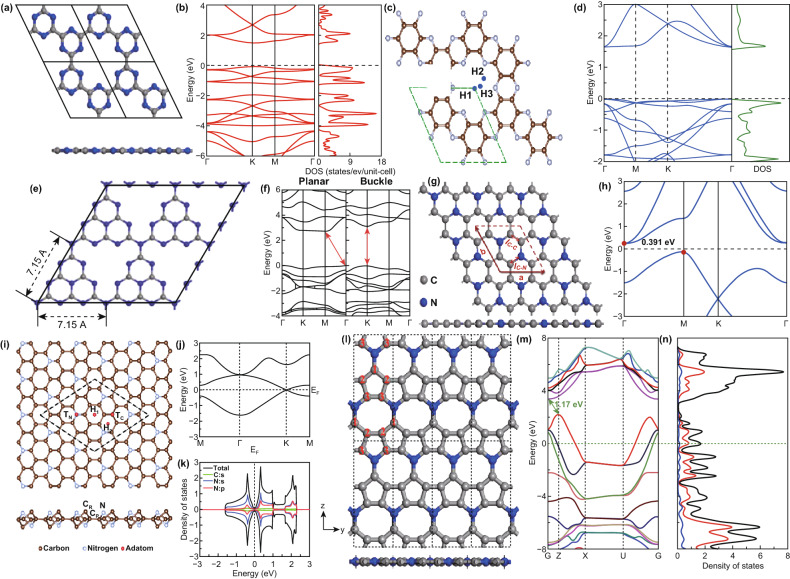
CN exists in different compositions such as C2N2, C3N3, and C4N4, and the structure of these carbon nitride materials possesses round and uniform pores, six pyridinic nitrogens in each unit cell and a bandgap of ~ 1.5–1.6 eV (Fig. 4a, b) [15]. Most of the carbon nitrides contain both graphitic and pyridinic nitrogen except for CN. Due to its rich pyridinic nitrogen structure and absence of graphitic nitrogen, CN exhibits high structural stability, better conductivity and does not experience loss of crystallinity when it interacts with alkali metal ions like Li+.
Fig. 4.
a Top view and side view of the supercell (2 × 2) g-C3N3. b Band structure and total density of state for 1 × 1 g-C3N3. Reproduced with permission from Ref. [15]. c Relaxed structure of 2 × 2 C2N monolayer, H1, H2, and H3 are possible binding sites for transition metal atom doping on the C2N. d Band structure and density of states of C2N monolayer. Reproduced with permission from Ref. [28]. e Schematic structure of monolayer g-C3N4. Reproduced with permission from Ref. [18]. f Calculated band structures of monolayer g-C3N4 with planar or buckled topology. Reproduced with permission from Ref. [20]. g Optimized structure and h band structure of C3N monolayer. The unit cell is shown by the red dashed line. Reproduced with permission from Ref. [29]. i Top and side view of the atomic structure of monolayer C4N. The black dashed lines show the 3 × 3 × 1 supercell of monolayer C4N. Four adsorption sites were considered (H1, H2, TC, TN). j Electronic band structures and k PDOS of the unit cell of a pristine C4N monolayer. Reproduced with permission from Ref. [30]. l Top (upper) and side (lower) view of the atomic structure of C5N monolayer. The gray and blue balls represent C atoms and N atoms, respectively. m Band structure and n density of states (DOS) of C5N monolayer obtained from HSE06 calculations. The black, red, and blue lines denote the total DOS of C5N, the partial DOS of C atoms, and the partial DOS of N atoms, respectively. Reproduced with permission from Ref. [27]. Copyright permissions from Elsevier, Royal Society of Chemistry and Wiley–VCH. (Color figure online)
C2N
The structure of C2N exhibits sp2 hybridization, and it is filled with uniformly sized holes and one large hole at the center, which is due to the six-member nitrogen-containing ring (Fig. 4c). Like the structure of graphene, it possesses several benzene rings that are bridged by pyrazine rings, which have two nitrogen atoms opposite to each other [16]. Also, unlike graphene, which has a fully conjugated π–electron structure, the presence of nitrogen in the structure of C2N makes the π–electronic structure of its benzene rings isolated. Therefore, it has a flat band and exists as a semiconductor with a bandgap of 1.96 eV (Fig. 4d). Due to its wider structure and the abundance of several benzene and pyrazine rings, it is expected that it will possess several active sites for alkali metal ion storage. Moreover, the defect sites and higher nitrogen population of C2N suggest that it will exhibit superior conductivity to boron nitride (BN) [17].
C3N4
C3N4 carbon nitride is the most reported member of the carbon nitride family. It is composed of consistently repeated tri-s-triazine units having planar sp2-hybridized conjugation structures held together by van de Waals forces. It often occurs as bulk C3N4, which has a relatively low indirect bandgap of ~ 2.7 eV and an interplanar distance of 0.324 nm and can be functionalized to obtain other architectures such as the monolayer 2D sheet (Fig. 4e) [18]. This indirect bandgap of C3N4 highly has spurred massive interest in its application for several applications such as photocatalysis, oxygen evolution, reduction reactions, sensing devices, etc. However, the semiconductor property of bulk C3N4 limits the electronic conductivity for application in electronic devices [19]. Under buckling, the indirect bandgap of bulk C3N4 can be tuned into a direct bandgap material with superior electronic conductivity (Fig. 4f) [20]. This ability to easily modify the electronic and structural properties of C3N4 makes it one of the most preferred and widely applied carbon nitride structures [21].
Graphitic Nitrogen-Based Carbon Nitrides (C3N, C4N, and C5N)
Graphitic nitrogen is one of the most widely known forms of nitrogen. It has a nitrogen bonded to three carbon atoms, and it is the nitrogen-doped into the basal graphitic carbon plane of the material. Due to its superior negativity in comparison with carbon, it is possible for graphitic nitrogen to induce a positive charge on adjacent carbon, thereby making it readily active site for attraction to negatively charged species [22]. This makes it a commonly desired form of nitrogen doping, just like pyridinic nitrogen. The graphitic-N-based carbon nitrides, which we shall discuss in this review, include C3N, C4N, and C5N.
C3N
The structure of C3N contains well-defined homogenous rings with no large holes. It is a 2D honeycomb lattice configuration that displays a D6h-symmetry that facilitates high structural stability and superior thermal conductivity (Fig. 4g). It is a semiconductor which displays a unique molecular orbital with a low unoccupied molecular orbital gap (~ 2.7 eV) and an indirect bandgap of 0.39 eV (Fig. 4h) [23].
C4N
Compared to the atomic structure of another carbon nitride, the C4N structure is filled with sp3 hybridized species and due to the presence of 4 pz atomic orbitals in the projected electron density of states (PDOS), it exhibits a Dirac cone shape in its band structure like that of graphene (Fig. 4i, j). It is often called the dumbbell (DB) C4N and depending on the positions of the raised C/N atoms in its structure, two structural configurations exist, namely the DB C4N-I and the DB C4N-II. The former refers to a C4N monolayer structure in which the raised C/N atoms are located on the same side, while the latter is a C4N monolayer structure where C/N atoms are on opposite sides [24, 25]. These two configurations of C4N are semiconductors in nature and display a narrow and zero bandgap (Fig. 4k); however, this property can be tuned easily [24, 26].
C5N
C5N is the most recently reported graphitic-N-based carbon nitrides; like C4N, the atomic structure of C5N is filled with sp3 hybridized carbon atoms; however, it contains two cyclic rings of different sizes (Fig. 4l). A 5-membered carbon ring and an eight-membered carbon ring with two nitrogen atoms are opposite to each other, and these rings are side-by-side to each other (see the atomic structure of C5N in Fig. 4k). The C5N structure has some interesting features such as high chemical, mechanical and thermodynamic stability. Also, analysis of the band structure shows that the Fermi level is located below the valence-band maximum, which suggests it will be metallic and display superior conductivity than other carbon nitrides (Fig. 4m, n) [27].
Doped Carbon Nitrides
It has been proven that the metal ion storage capacity of an electrode material such as carbon nitride is significantly dependent on the electronic property. Most carbon nitrides are semiconductors, the electronic density of states (DOS) of the majority of the carbon nitrides possess a lone pair orbital of nitrogen at their Fermi level, and this leads to a large effective mass for hole/electron and results in poor electronic conductivity [19]. Therefore, doping is important for carbon nitride structures. Heteroatoms of elements at the top end of the periodic table such as sulfur, nitrogen, boron, and phosphorus, display narrow bandgap and lower high occupied molecular orbital, which signifies superior electronic conductivity. Therefore, replacing some of the carbon atoms in the crystalline structure of carbon nitrides with metallic or non-metallic atoms can boost their electronic conductivity and electron mobility. Doping also creates surface defects on the carbon nitride structure, and this is beneficial for effective charge transport.
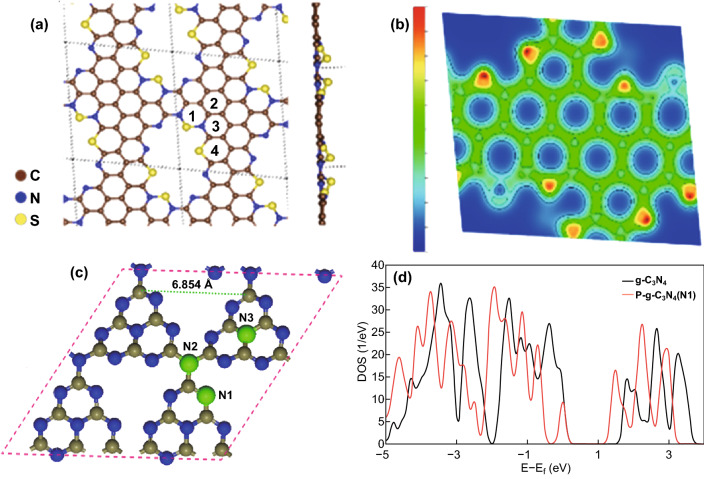
Three heteroatoms are commonly used to dope carbon nitrides. These are carbon, nitrogen, sulfur, and phosphorus. C doping in carbon nitrides can facilitate the formation of delocalized π-bonds, which can result in n − π* electronic transition due to interaction with inherent nitrogen, and this will boost the electronic conductivity and charge transfer. N and S doping have proven to enlarge the interlayer distance of carbon nitrides; this facilitates the adsorption of metal ions and improves the conductivity. Cha et al. reported S-doped mesoporous CN carbon nitride (Fig. 5a shows the optimized structure), and from the electron density profile presented in Fig. 5b, it is obvious that effective charge distribution can be achieved at S-doped sites [31]. Boron (B) and phosphorus (P) doping can decrease the indirect bandgap of semiconductor carbon nitrides and make them metallic. Molaei et al. studied the effect of doping C3N4 with phosphorus by replacing some of the N atoms with P atoms (Fig. 5c), and the DOS result shown in Fig. 5d confirms a shift of the Fermi level toward the conduction band. This suggests a narrowed band gap and enhanced electronic conductivity compared to pure C3N4 [32]. In summary, heteroatom doping of carbon nitrides is beneficial for boosting their electrical conductivity, improving charge transfer, enlarging their interlayer distance, and boosting metal ion storage.
Fig. 5.
a Optimized atomic structure of S-doped mesoporous CN (S-MCN). b Charge density profile of S-MCN, blue color—lowest electron density (0%), and red color—highest electron density (100%). Reproduced with permission from Ref. [31]. c Optimized structure g-C3N4 monolayer, showing possible P-doping sites. d DOS plots of P-doped g-C3N4 for P substitution at N1 and N2 sites. Reproduced with permission from Ref. [32]. Copyright permissions from American Chemical Society and Springer Nature. (Color figure online)
CNBCs
Although doping is an effective way of modulating the electronic conductivity of carbon nitrides, they are still limited by poor reversible capacity due to ineffective metal ion storage mechanisms and inadequate surface area. Most carbon nitrides are not able to achieve the desired performance in battery testing because the adsorption energy may be too strong for effective adsorption and desorption of metal ions or the diffusion barrier may be too high for easy metal ion transport. Also, apart from poor electronic conductivity, pure carbon nitrides are characterized by smaller surface area, which provides inadequate coverage for metal ion adsorption. CNBCs can solve these problems because combining pure carbon nitride with highly conductive materials like reduced graphene oxide (rGO) will produce a material with improved electronic conductivity and larger surface area. Moreover, the formation of CNBCs with high theoretical capacity metal oxides such as Fe2O3 will provide an anode material with an improved reversible capacity [33]. CNBCs can also be formed by combining pure carbon nitride with metal chalcogenides, perovskites, etc., for different metal-ion batteries. Hence, we summarize that an effective way to resolve the problems of poor reversible capacity and limited surface area is through CNBCs.
DFT-Guided Studies of CNBMs for Energy Storage Devices
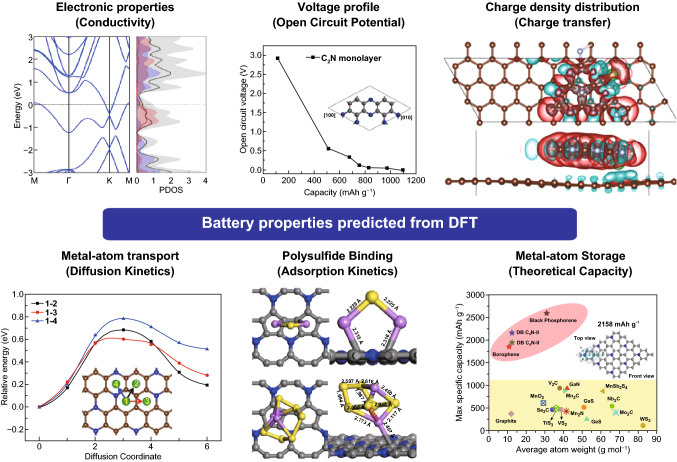
After the first discovery/synthesis of tri-s-triazine-based carbon nitride by Berzelius in 1834, the prediction and synthesis of beta-C3N4 in 1993, the discovery of other carbon nitride structures have largely depended on theoretical first-principle simulations. First-principle calculations allow researchers to calculate or predict the structures and properties of material before its synthesis. It can also be used to resolve the discrepancy between experimental measurements and theoretical calculations by considering various possible alkali-metal atom diffusion kinetics, including diffusions in the bulk structure, on the surface, in the defect rich sites, etc. [1, 34]. This prevents random experimental tests or synthesis, thereby facilitating materials discovery. Furthermore, modification strategies for improving the properties of prospective materials can be easily explored through first-principle calculations. DFT is one of the most common computational simulation methods. It is a very powerful tool used to predict different electrochemical parameters of electrode materials. DFT calculations can be used to determine the alkali-metal atom insertion voltage of electrode materials, calculate the migration energy barriers of the alkali-metal atom, and directly visualize the transport pathways and dynamics [35, 36]. In addition, the theoretical capacity of a potential electrode material can be predicted from DFT calculations by investigating the maximum metal atom loading on its structure. The adsorption energy of important molecules on the structure of electrode materials can also be calculated; for example, the adsorption of polysulfides on Li–S cathode materials. Crystal and surface defects are important properties of an electrode material because they impact the alkali-metal atom transport mechanism and pathways, and their effect can also be studied through first-principle calculations. Moreover, information on the intrinsic electronic conductivity and bandgap can be directly inferred from the calculated electronic density of states by observing the Fermi level, conduction, and valence bands. The important role of DFT calculations in CNBMs for energy storage devices includes the DFT-guided synthesis and the DFT-predicted electrochemical properties. In terms of DFT-guided synthesis, DFT studies can determine important synthesis parameters such as the formation energy, exfoliation energy, and cohesive energy of a CNBM. These are criteria that indicate the ease/feasibility of synthesizing a CNBM experimentally. DFT calculations can also provide an atomistic understanding of the interaction between the C/N and the adsorbed metal atom, which will reveal the structural limitations of the CNBMs and guide the synthesis of superior material. DFT studies also play a significant role in predicting fundamental electrochemical properties such as the bandgap, metal atom adsorption energy, open-circuit potential, charge transport kinetics, and theoretical capacity of the CNBMs. These parameters forecast the prospect of CNBMs for energy storage devices and reveal potential limitations that must be considered and improved upon. The electrochemical properties that can be predicted from DFT are summarized in Fig. 6.
Fig. 6.
Electrochemical properties of carbon nitrides can be predicted from DFT calculations. Reproduced with permission from [27, 37–40]. Copyright permissions from Elsevier, American Chemical Society, Wiley–VCH, and IOP Publishing. (Color figure online)
Theoretical Calculations on CNBMs for Energy Storage Devices
Density functional theory calculations are one of the most useful tools for discovery and electrochemical investigation of different carbon nitrides for rechargeable batteries [41]. In this section, we discuss different carbon nitrides that have been studied through DFT.
Pure Carbon Nitrides
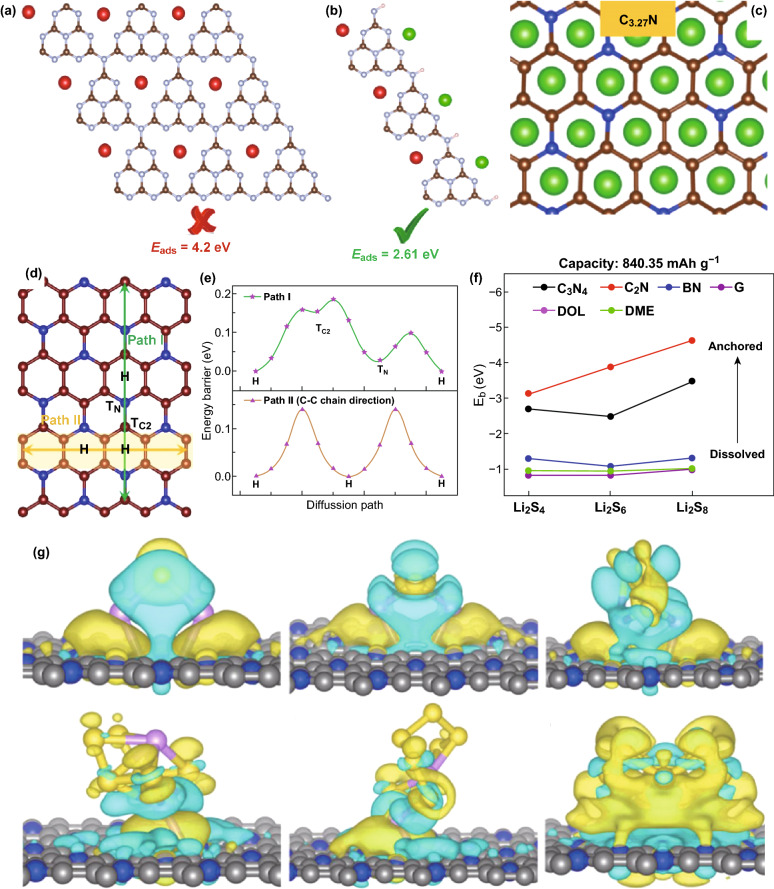
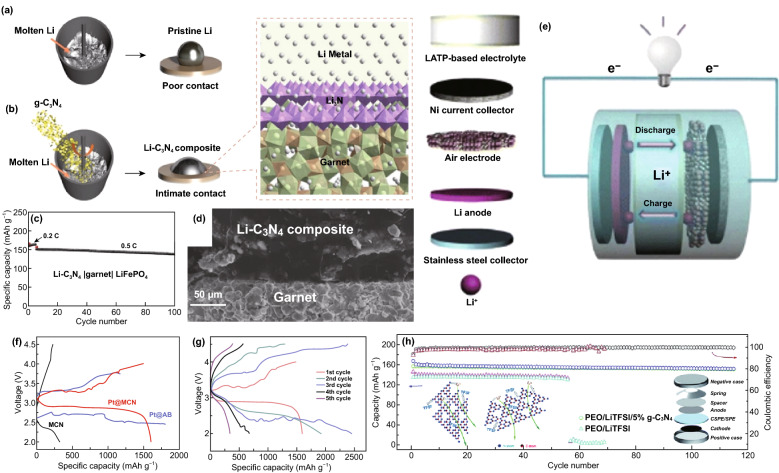
Through DFT calculations, it was predicted that C3N4 carbon nitride could deliver a theoretical capacity of up to 524 mAh g−1 (Li2C3N4) [42], and this capacity has not been achieved in battery testing. Mao et al. [43], Veith et al. [44], and Hankel et al. [45] showed that this poor performance was due to the high content of graphitic nitrogen in its structure which resulted in ineffective intercalation. We reported the first C3N4 with reduced graphitic-N and increased pyridinic-N for LIBs, and our DFT calculations showed that unlike bulk/sheet C3N4 which adsorbs Li atom in the triangular pore at high energy of ~ 4 eV, a 1D-C3N4 fiber possesses lower Li binding energy (2.61 eV) at its edges. Hence, it can effectively adsorb/desorb Li atoms (Fig. 7a, b) [46]. Hui Pan reported a first-principle study on C3N4 carbon nitride nanotubes for LIBs and predicted that it could store Li atoms internally and externally due to its porous structure, which might make it better than other dimensions of carbon nitride [47].
Fig. 7.
Reversible Li active sites in a 2D-C3N4 sheet, and b 1D-C3N4 fiber. Green means reversible Li atoms and red means non-reversible Li atoms. c Top and side view of stable intercalation structures of n Li ions into C3N. Reproduced with permission from Ref. [40]. d Diffusion paths and e corresponding energy barriers of Li migration in C3N-S2. Reproduced with permission from Ref. [51]. f Binding energy of Li2S4/Li2S6/Li2S8 interacting with G, BN, C2N, C3N4 and DOL/DME solvent, respectively. Reproduced with permission from Ref. [17]. g Isosurfaces of charge density difference of Li2S, Li2S2, Li2S4, Li2S6, Li2S8, and S8 adsorbed on the surface of C5N with the isovalue of 0.003 A−3. Blue wireframes denote loss of electrons and yellow wireframes denote gain of electrons. Reproduced with permission from Ref. [27]. Copyright permissions from American Chemical Society, Elsevier and Wiley–VCH. (Color figure online)
Other carbon nitride structures have also been studied for LIBs through DFT. For example, Hankel et al. showed that although g-CN can deliver a high capacity of 454 mAh g−1. Its high Li binding energy of > 3 eV leads to ineffective intercalation/deintercalation and limits its prospects for LIBs [48]. The study of Hussain et al. on C2N-h2D showed that despite the pyridinic nitrogen, which facilitates Li storage, the high initial Li adsorption energy would result in poor battery performance [16]. By using a “pair of particle (metals) model,” Wu et al. [49] carried out a DFT study on C2N for LIBs and SIBs to show that C2N monolayer can achieve a capacity of 2939 and 2469 mAh g−1 for LIBs and SIBs with low diffusion barrier and an OCV of 0.45 V [16]. Liu et al. carried out a DFT study on different C3N compositions (C3N, C2.67N, and C3.33N) and showed that the drop in capacity experience by Xu et al. [50] was due to ineffective intercalation of Li. They concluded that C3.33N is the preferred C3N composition, and it delivered a reversible capacity of 840.35 mAh g−1 (Fig. 7c), operated at a low open-circuit potential of 0.12 V, and displayed superior electronic conductivity [40]. Guo et al. proposed other C3N allotropes (C3N-S1, C3N-S2, and C3N-S3) with unique electronic properties, and the C3N-S2 structure displayed the most feasible Li diffusion pathway with lower diffusion barrier (0.14 eV) for LIBs (Fig. 7d, e) [51]. Yang et al. also reported the first-principle study of two structural configurations of DB C4N as LIBs anode for LIBs, and they predicted that DB C4N-I and DB C4N-II could deliver high theoretical capacities of 1942 mAh g−1 (DB C4N-I) and 2158 mAh g−1 (DB C4N-II) [52].
DFT studies of pure carbon nitrides for large-sized metal ion batteries have also been reported. For instance, Weng et al. showed that pure C3N4 nanosheet displays high adsorption energy for Na-atom, but it shows an exceedingly high diffusion barrier, which makes it fail as a SIBs anode [53]. Moreover, through DFT we showed that thanks to the high pyridinic-N of 1D-C3N4 fiber it exhibits a high affinity for potassium ions, but it suffers from a rather high K-diffusion barrier which limits effective ion transport [54]. Bhuriyal et al. also reported the first-principle study of C3N monolayer as a promising anode material for SIBs and PIBs, and they showed that multilayer adsorption of the metal atoms generated a high capacity of 1072 mAh g−1 and low diffusion barrier of 0.03 and 0.07 eV for Na and K-ion [55]. Xu et al. showed that metal ion (Li, Na, and K) adsorption on C4N is an effective way to open the zero bandgap and modulate the electronic property because the adsorbed metal ion transfers charges to the surface of DB C4N [30].
Thanks to the nitrogen-rich structure, carbon nitrides have also been studied for Li–S and other battery systems. Li et al. reported that due to the accumulation of charges at the N–N bond of the C4N4 structure, it demonstrates superior lithium polysulfide species (LIPSs) anchorage than commercial electrolyte solvent molecules (DOL and DME) [56]. Zheng and co-workers compared the LIPSs anchoring property of graphene, boron nitride (BN), C2N, and C3N4 with commercial solvents and showed that C2N and C3N4 were the most effective due to their stronger binding energy (Fig. 7f) surface interaction with LiPSs via the Li-N/C-S bonds formed during their interaction [17]. Liang et al. also showed that interaction between LIPSs and polymeric C3N4 (p-C3N4) exhibits strong ionic bonding, electrostatic, and vdW interactions which are beneficial for altering the bonding and spatial configuration of LIPSs which then modifies their redox kinetics [57]. Meng et al. also reported that the abundant nitrogen species on the surface of C3N4 nanosheet is able to facilitate LiPSs anchoring by a surface chemical adsorption mechanism due to the formation of a Li-N bond [58]. Wang et al. reported a first-principle study on the application of C5N as a LIPSs host. The isosurfaces of charge density difference presented in Fig. 7g showed that it exhibits an effective physical/chemical adsorption property, which enables LIPSs anchoring and charge transfer to its surface [27]. Carbon nitrides have also been explored for metal-air batteries because of their unique properties. For example, Shinde et al. reported the prospect of C2N for zinc-air (Zn-Air) batteries and showed that the open holey structure of C2N enabled reversible oxygen reactions and improved the electronic conductivity [59]. Je et al. also carried out first-principle calculations on heptazine and triazine sourced C3N4 for non-aqueous Li-O2 battery, and the results showed that triazine was better because it delivered a higher overpotential. Interaction of C3N4 with LiO2 resulted in the formation of a Li-N bonding, which is dependent on the ratio of N in the material and greatly influences their overpotential [60].
Doped Carbon Nitrides
Theoretical study on doped carbon nitrides of different atomic compositions has been reported. For example, Nong et al. considered the effect of non-metal doping (boron, oxygen, and sulfur) and the impact of strain on the Li storage capacity and Li absorptivity of C3N. They reported that oxygen-doped C3N (ON–C3N) was the preferred dopant, it operated at 0.02 V and delivered a higher theoretical capacity of 534.42 mAh g−1 although it exhibited a high Li diffusion barrier of 0.78 eV, and this could be lowered by applying strain to the C3N structure [61]. Tian et al. also studied the effect of boron doping on the performance of C3N for alkali metal ion battery (Li, Na, and K) and showed that B4-doped C3N displays superior capacity, cycle, and thermal stability than pristine C3N [29]. Cha et al. showed that CN possesses a high Na adsorption energy, which can foster attraction of Na atoms into its structure for SIBs batteries [31]. Although Weng et al. [62] proved that C3N4 displays a very high Na-adsorption energy which limits its application for SIBs, the report of Molaei et al. on P-doped C3N4 showed that phosphorus doping is an effective way to decrease the high Na adsorption energy and diffusion barrier in C3N4 (Fig. 8a-d). The more P-atoms introduced into the structure of C3N4, the lower these energies [32]. Moreover, the work of Cha et al. [31] on Si-doped CN suggests that S-doping of carbon nitrides can successfully enlarge their interlayer distance for such large-sized metal-ion battery applications.
Fig. 8.
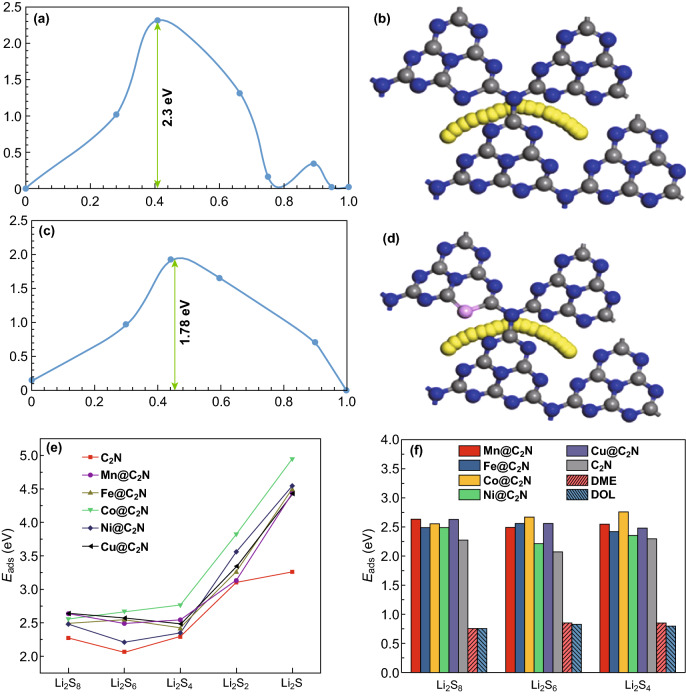
a Energy barrier for Na diffusion through g-C3N4, b corresponding Na diffusion path. c Energy barriers for Na diffusion through P-g-C3N4, d corresponding Na diffusion path. Reproduced with permission from Ref. [32]. e Adsorption energies of LiPSs on transition metal embedded C2N monolayers. f Adsorption energies of long-chain LiPSs with transition metal embedded C2N, 1,2-dimethoxyethane (DME) and 1,3-dioxolane (DOL). Reproduced with permission from Ref. [28]. Copyright permissions from Springer Nature and Elsevier. (Color figure online)
Doped carbon nitrides have also been studied for Li–S battery, Lin et al. studied the effect of transition metal doping (Mn, Fe, Co, Ni, and Cu) in C2N for improved immobilization of polysulfides in Li–S batteries, their results showed that a Lewis acid–base interaction which boosted LiPSs anchoring was established, and a significant charge was transferred from the transition metal dopant to the C2N monolayer with Co@C2N transferring the most in line with its superior adsorption energy (Fig. 8e, f) [28]. Zhao and co-workers reported confined single-atom Pt in holey g-C3N4, and DFT calculations showed that incorporation of single-atom Pt in holey g-C3N4 leads to improved electrical conductivity and a more stable structure with efficient electron and ionic transfer [63].
CNBCs
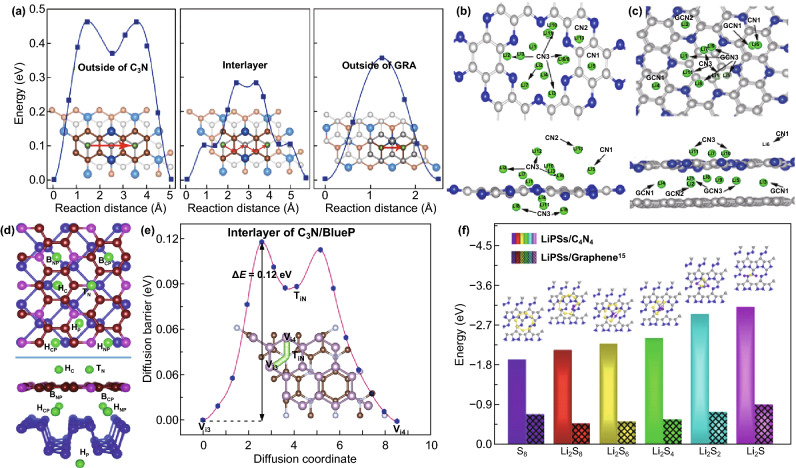
CNBCs with superior electronic conductivity and higher capacity have also been studied through DFT. For example, Wang et al. reported a DFT study of C3N/graphene heterostructure for LIBs and proved that the electrical conductivity and structural stability can be significantly improved. The heterostructure recorded a high theoretical capacity of 1079 mAh g−1, a low Li diffusion barrier of 0.28 eV at the interlayer (Fig. 9a), and operated at a low open-circuit voltage of 0.13 V [37]. Ding et al. reported the first-principle study of C2N/graphene bilayer for LIBs, and by using molecular dynamic (MD) simulations, they predicted that Li storage follows a two-step process, i.e., migration through the z-direction via the large hole in the center of the C2N structure and on to the surface of the C2N membrane. The structures of different compositions of the bilayer heterostructures are presented in Fig. 9b, c. [38]. Guo et al. [64] and Lin et al. [65] also reported a DFT study on C3N/phosphorene heterostructure for LIBs, while Bao et al. explored its potential for Li/Na battery. The three reports concluded that the design of such C3N/phosphorene heterostructure would not only address the issues of phosphorene but also alleviate the problems of C3N for Li/Na storage. Storage of Li-ions occurred at the outer surface and interlayer of the heterostructure (Fig. 9d, e) [66].
Fig. 9.
a Energy profiles for Li atom diffusion on C3N/GRA, along with the corresponding pathways denoted as red arrows. Carbon atoms from graphene—gray balls, carbon atoms from C3N—orange balls, nitrogen atoms—blue and lithium atoms—green. Reproduced with permission from Ref. [37]. b Top and side views of Li13–C2N structure; c top and side views of the Li11–C2N/graphene bilayer. Different adsorption sites are indicated as CN1, CN2, CN3, GCN1, GCN2, and GCN3 for both structures. Reproduced with permission from Ref. [38]. d Top and side views of the Li adsorption site on the C3N/P heterostructure (Li/C3N/P, C3N/Li/P, and C3N/P/Li). HC and TN sites are on the outer surface of C3N, and HP site is on the outer surface of phosphorene, HCP, HNP, BCP, and BNP sites are in the interlayer of the C3N/P heterostructure. Reproduced with permission from Ref. [64]. e Lithium migration pathway and corresponding energy profile through Path II of the interlayer of C3N/Blue P heterostructure. Reproduced with permission from Ref. [65]. f Adsorption energy for S8 cluster and LiPSs on C4N4 monolayer (bars without patterns) and graphene (bars with patterns), respectively. The insets on the pillars show S8 or Li2Sn/C4N4 structures generated by the principle of minimum energy. Reproduced with permission from Ref. [56]. Copyright permissions from Elsevier, American Chemical Society and Royal Society of Chemistry. (Color figure online)
DFT reports on CNBCs for Li–S batteries have also been widely reported. For instance, Liao and co-workers showed that the anchoring ability of C3N4 could be exploited in CNBCs [67]. Fan et al. also reported a carbon black blended g-C3N4 (g-C-coated) for Li–S battery and showed that two bonds (C-S and N-Li) are formed as a result of its interaction with LIPSs. These bonds inhibit the migration of LiPSs because it effectively binds to the LiPSs [68]. The work of Li et al. on porous C4N4 monolayer and C4N4/graphene composite for Li–S battery reported that LIPSs adsorption occurred by a chemisorption process and addition of graphene enhanced this process (Fig. 9f) [56]. Chen et al. studied the effect of transition metal (Fe, Ni, Cu, and Co)-doped g-C3N4/C composite for Li–S battery and showed that this would increase the electron mobility, while g-C3N4 will prevent polysulfide dissolution by anchoring LiPSs species [69]. In summary, DFT has been very key in the synthesis and electrochemical study of carbon nitrides for metal-ion batteries. Table 1 compares the DFT predicted parameters of carbon nitrides with commercial graphite anode for energy storage devices. From this table, it is obvious that most of the carbon nitrides can function as anode materials with superior theoretical capacity compared to graphite. These electrochemical properties have inspired researchers into the synthesis and fabrication of pure and doped carbon nitrides as well as CNBCs for energy storage devices.
Table 1.
Comparison of the DFT predicted parameters of pure carbon nitrides with commercial graphite for energy storage devices
| Materials | Conductivity | Capacity (mAh g−1) | Adsorption energy (eV) | Diffusion barrier (eV) | Operating voltage (V) |
|---|---|---|---|---|---|
| Graphite | Metallic |
372 (LIBs) 31 (SIBs) [70] 279 (PIBs) [71] |
1.1–1.29 (LIBs) [72] − 0.67 (SIBs) [73] − 0.44 to 2.0 (PIBs) [74] |
0.22 (LIBs) [75] 0.09–0.35 (SIBs) [76] 0.039 (PIBs) [77] |
0–0.5 (LIBs) [78] 0.3 (SIBs) [79] ~ 0.24 (PIBs) [80] |
| CN | Semi-conductor | 454 (LIBs) [48] | – 5.03 (LIBs) [48] | ~ 3 (LIBs) [45] | ~ 0.6 (LIBs) [81] |
| C2N | Semi-conductor |
2939 (LIBs) [49] 2469 (SIBs) [49] |
– 3.437 (LIBs) [49] – 2.868 (SIBs) [49] |
0.409 (LIBs) [49] 0.116 (SIBs) [49] |
0.452 (LIBs) [49] 0.458 (SIBs) [49] |
| C3N | Semi-conductor |
837.06 (LIBs) [40] 1072 (SIBs) [55] 1072 (PIBs) [55] |
− 0.01 (LIBs) [40] − 1.806 (SIBs) [55] – 2.230 (PIBs) [55] |
0.8 (LIBs) [40] 0.03 (SIBs) [55] 0.07 (PIBs) [55] |
0.15 (LIBs) [40] 0.13 (SIBs) [55] 0.26 (PIBs) [55] |
| C3N4 | Semi-conductor | 524 (LIBs) [44] | – 4.56 (LIBs) [45] | ~1.8 (LIBs) [47] | 0.8 (LIBs) [44] |
| C4N | Semi-conductor |
I—1942 (LIBs) [52] II -2158 (LIBs) [52] |
I—1.3 (LIBs) II—0.93 (LIBs) [52] |
I—0.26 [52] II—0.21 [52] |
I—0.60 [52] II—0.68 [52] |
| C5N | Metallic | N/A | N/A | N/A | N/A |
Synthesis Strategies of Pure and Doped Carbon Nitrides
The conclusions obtained from DFT studies showed that the poor performance of most CNBMs can be traced to high metal atom adsorption energy due to excess graphitic-N, which leads to irreversible intercalation/deintercalation, poor conductivity, and low charge transfer mobility, as well as inferior structural stability after metal atom adsorption. DFT calculations showed that these problems could be overcome by regulating the ratio of C/N in the structure and prioritizing pyridinic-N, heteroatom doping, and CNBCs design with other superior electrode materials. Inspired by these DFT conclusions, several researchers have modified the general experimental synthesis strategies to obtain CNMBs that exhibit the desired structural/electronic properties. Such modified experimental synthesis strategies that are motivated by the findings in DFT studies can be considered DFT-guided synthetic protocols. In this section, we summarize some significant DFT-guided synthesis strategies for CNBMs, including pure/doped carbon nitrides as well as CNBCs.
Top-Down Strategy
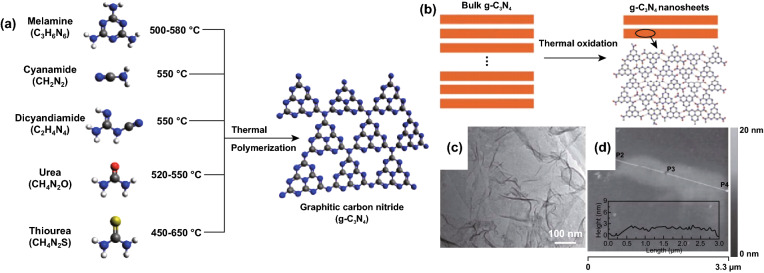
Carbon nitrides generally occur in the bulk state, and this form of carbon nitride is obtained via thermal decomposition of N-rich amine compounds such as melamine [82] (Fig. 10a). However, bulk carbon nitride suffers from the poor conductivity, limited surface area, and sluggish reaction kinetics [83, 84]. Bulk carbon nitride also contains excess graphitic-N which is proven to limit the performance of carbon nitrides in rechargeable batteries according to DFT conclusions. One way to regulate the C/N ratio and decrease the graphitic-N is by converting bulk carbon nitride to nanosheets and other morphologies. This can be achieved through the top-down strategy which is grouped into post-thermal oxidative etching and ultrasonic liquid exfoliation.
Fig. 10.
a Schematic illustration of one of the top-down synthesis approach (thermal polymerization) for g-C3N4 using different precursors. Black balls—carbon (C), blue balls—nitrogen (N), white balls—hydrogen (H), red balls—oxygen (O), and yellow balls—sulfur (S), respectively. Reproduced with permission from Ref. [84]. b Schematic of the synthesis of g-C3N4 nanosheets from bulk g-C3N4. In the atomic model, carbon atoms—gray balls, nitrogen atoms—blue balls and hydrogen atoms—red. c TEM image of g-C3N4 nanosheets. d Tapping-mode AFM image of a single g-C3N4 nanosheet deposited on the silicon wafer substrate. The inset is the height curve determined along the line between P1 and P4. Reproduced with permission from Ref. [88]. Copyright permissions from American Chemical Society and Wiley–VCH. (Color figure online)
Thermal Oxidation
As the name implies, this method involves the heat treatment of bulk carbon nitride at high temperature and it is an effective way to break the van der Waals forces which hold the layer of carbon nitride together. This process produces a sheet-like carbon nitride with large surface area, surface defects and adjusted layer thickness [22, 85]. As depicted in Fig. 10b, bulk g-C3N4 can be converted to g-C3N4 nanosheets via thermal oxidation and Fig. 10c, d shows that the layer thickness can also be regulated. Guo et al. reported the synthesis of graphite/sheet-like carbon nitride (CN) as well as nanotube, nanoribbon, and microsphere morphologies via thermal treatment [86, 87]. 2D-C3N carbon nitride with a needle-like morphology was also synthesized by via a direct solid-state reaction [23].
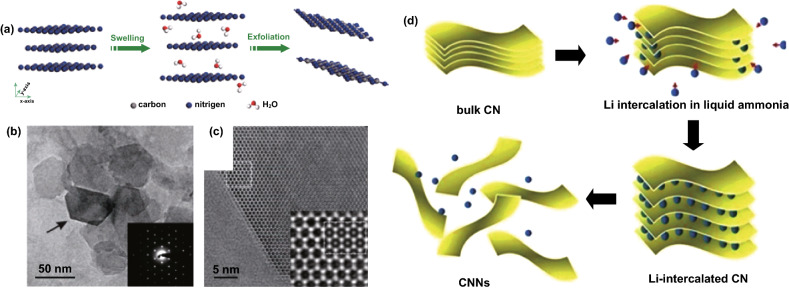
Ultrasonic Liquid Exfoliation
Another effective strategy to convert bulk multilayered carbon nitrides to few/single layer carbon nitrides is through the intercalation of solvent molecules through the bulk structure, thereby resulting in delamination (Fig. 11a). This liquid exfoliation process is effective for synthesizing nanosheets or flakes of carbon nitrides (Fig. 11b, c). Mahmood et al. reported the synthesis of two-dimensional C2N carbon nitride by a top-down (facile wet-chemical reaction) approach [89]. This process can also be achieved through lithiation exfoliation which uses Li ions as exfoliant to penetrate the layers of bulk carbon nitride and separate them to individual nanosheets (Fig. 11d).
Fig. 11.
a Schematic illustration of liquid-exfoliation process from bulk g-C3N4 to ultrathin nanosheets. Reproduced with permission from Ref. [90]. b TEM image of exfoliated ultrathin nanosheets, c higher magnification of a carbon nitride nanosheet edge viewed along [001]. Reproduced with permission from Ref. [91] d Schematic diagram of the lithiation and exfoliation of g-C3N4 nanosheets from bulk g-C3N4. Reproduced with permission from Ref. [92]. Copyright permissions from American Chemical Society. (Color figure online)
Bottom-Up Strategy
The bottom-up strategy or template-aided synthesis is divided into soft and hard templating method.
Hard Templating
Templates are often used as morphology directing agents during the synthesis of unique carbon nitride morphologies. Hard templates such as silica [93–95], anodic alumina oxide [96], and carbon [97] are often reported for synthesizing CNBMs. For instance, DFT studies showed that by increasing the N content in C3N4 to C3N5 the electrochemical properties can be improved. Recently, Kim et al. reported the synthesis of a graphene-like mesoporous carbon nitride C3N5 with superior electronic properties by using KIT-6 as a hard template [98]. Moreover, their DFT/electrochemical study showed that such composite can deliver excellent performance for energy storage devices [34]. The C3N5 sample showed outstanding interesting electrochemical properties, and it was combined with graphene oxide to form a composite (MCN-11). Figure 12a shows a scheme of the synthesis procedure of the C3N5 and the hybrid.
Fig. 12.
a Schematic representation of the synthesis protocol for synthesizing the mesoporous C3N5 and its graphene hybrid. Reproduced with permission from Ref. [98]. b Schematic illustration of the synthesis of ordered porous g-C3N4 by using close-packed silica nanospheres (SNSs) as the primary template. c–e FE-SEM images of porous g-C3N4 at different resolutions. Reproduced with permission from Ref. [93]. Copyright permissions from Wiley–VCH and Royal Society of Chemistry. (Color figure online)
Soft Templating
Templating materials which demonstrate self-assembling properties have also been employed for synthesizing carbon nitrides by using ionic liquids [99, 100], surfactants [101–103], and amphiphilic block polymers [102, 104]. Yong et al. reported the synthesis of a nanoporous carbon nitride by using ionic liquid as soft templates (Fig. 12b-e), and such carbon nitride structure demonstrated superior conductivity to bulk material [101]. In summary, by using the conclusions of DFT studies as a guide, the general experimental synthesis approach of carbon nitrides can be optimized to achieve carbon nitrides with regulated C/N ratio, increased pyridinic-N, superior structural stability, and conductivity for improved performance.
Fabrication Strategies of Doped Carbon Nitrides
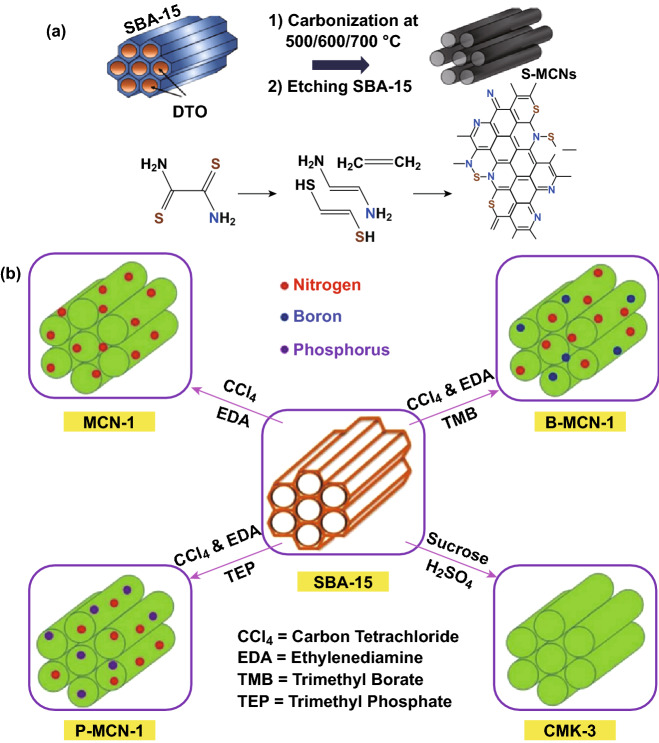
The DFT study of Molaei et al. [32] proved that heteroatom doping of carbon nitride will boost the electronic conductivity and improve metal atom storage, and the DFT conclusion of this work motivated the Vinu group to report the synthesis of rod-like sulfur-doped mesoporous CN (S-CN) by a templating method (Fig. 13a) [31]. Phosphorus-doped mesoporous carbon nitride (P-MCN) was also synthesized via a simple template approach [105]. A schematic of the template synthesis method for heteroatom-doped mesoporous carbon nitrides is presented in Fig. 13b. To summarize, doped carbon nitrides can be synthesized through a combination of some top-down and bottom-up approaches which often involves the use of a salt of the heteroatom or a surfactant.
Fig. 13.
a Schematic of the synthesis procedure of S-MCN [31]. b Schematic of the template synthesis method for heteroatom-doped mesoporous carbon nitrides. Reproduced with permission from Ref. [105]. Copyright permission from American Chemical Society. (Color figure online)
Fabrication Strategies for CNBCs
DFT studies have proved that CNBCs design is another effective way to resolve the challenges of carbon nitrides because such CNBCs will demonstrate superior conductivity, better structural stability, and enhanced charge transfer. Therefore, in this section, we highlight some recent CNBCs that have been synthesized through commonly reported experimental synthesis strategies but inspired by the conclusions of DFT studies.
Hydrothermal Method
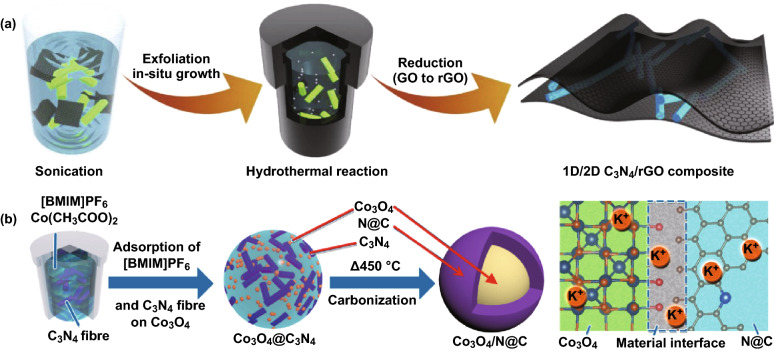
Hydrothermal method involves the controlled synthesis of a composite in a tightly sealed vessel under high temperature and pressure [106, 107]. Although 2D-C3N4 is widely reported for designing CNBCs, our DFT studies showed that 1D-C3N4 exhibits superior structural stability and metal storage capability than 2D-C3N4 [40]. Moreover, our comparative DFT study between 1D/2D C3N4/graphene and 1D/2D C3N4/graphene showed that 1D/2D will perform better [38]. These DFT conclusions motivated us to combine 1D-C3N4 with 2D-rGO for energy storage devices. Therefore, we reported the synthesis of a 1D/2D C3N4/rGO composite via a freeze-drying-assisted hydrothermal approach by deploying the π–π interaction between C3N4 and graphene [54]. Figure 14a shows the synthesis scheme of the composite. Guided by the result of our DFT calculations, we also reported the design of a composite of Co3O4@N–C derived from 1D-C3N4 via an ionic liquid-assisted solvothermal method [108]. Scheme illustration of the synthesis strategy is depicted in Fig. 14b.
Fig. 14.
a Schematic illustration of the synthesis of 1D/2D C3N4/rGO composite via a hydrothermal/freeze-drying method. Reproduced with permission from Ref. [54]. b Scheme illustrating suggested potassiation and depotassiation mechanism of the Co3O4@N–C electrode. Reproduced with permission from Ref. [108]. Copyright permission from Elsevier and American Chemical Society. (Color figure online)
Self-Assembly Method
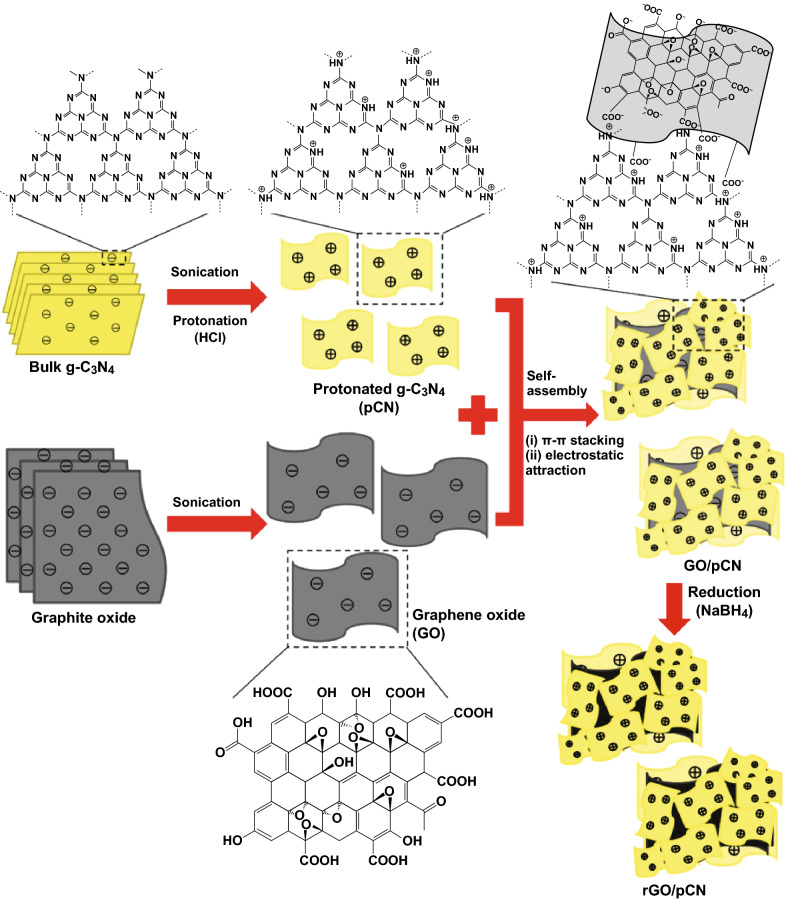
Due to their amphoteric nature, carbon nitrides possess tunable surface functional groups and surface charges which enable surface attachment with other functional materials such as graphene and other functional 2D materials [130]. Such structural interaction has been identified through the molecular dynamic simulation and DFT studies of Ding et al. and Wang et al. The heterostructure will display superior electronic conductivity and structural stability than the pure carbon nitride. This concept is employed in self-assembly synthesis of C3N4 and other CNBMs. Figure 15 shows a detailed schematic of the self-assembly approach for a surface modified C3N4 (pCN) with graphene oxide and reduced graphene oxide to achieve a GO/pCN and rGO/pCN composite, respectively [109].
Fig. 15.
Schematic diagram for the synthesis process of rGO/pCN samples via a combined ultrasonic dispersion and electrostatic self-assembly strategy followed by a NaBH4-reduction process. Reproduced with permission from Ref. [109]. Copyright permission from Elsevier. (Color figure online)
Other Composite Fabrication Strategies
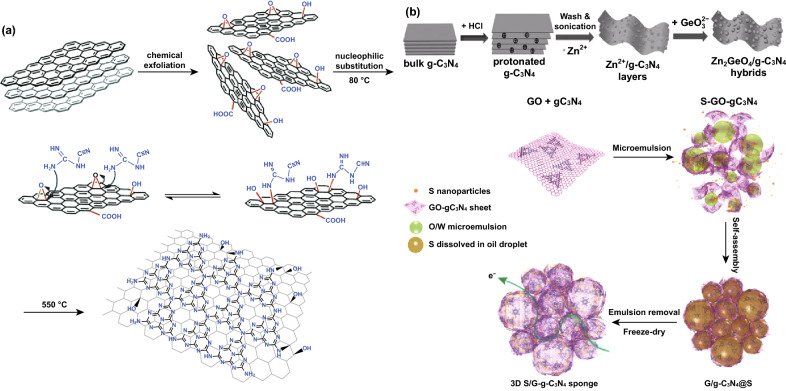
CNBCs can also be designed by some other unique methods, and Fu et al. designed a 2D-2D g-C3N4-rGO hybrid via an in situ chemical method by initiating a nucleophilic reaction between epoxy groups on the surface of GO and amine/amide groups on dicyanamide-C2H4N4. The synthesis method is depicted in Fig. 16a, and through this approach, the problem of poor electrical conductivity, aggregation, and restacking was resolved [110]. Similarly, Li et al. reported the design of a Zn2GeO4/g-C3N4 composite by growing Zn2GeO4 NPs in-between the layers of g-C3N4 via a solution approach to inhibit agglomeration of Zn2GeO4 NPs and restacking of g-C3N4 (Fig. 16b). Zn2GeO4 also functioned as a spacer to enlarge the interlayer distance of g-C3N4 sheet enabling metal ion adsorption and improved conductivity [111]. Wang et al. reported the synthesis of a GO/g-C3N4 microsphere by an ethanol-assisted spray drying approach [112]. Zhang et al. also reported the design of a sponge-like free-standing 3D S/graphene@g-C3N4 hybrid using a microemulsion-assisted assembly method (Fig. 16c) [113].
Fig. 16.
a Illustration of the formation process of g-C3N4–rGO. Reproduced with permission from Ref. [110]. b Schematic of the synthesis process of the Zn2GeO4/g-C3N4 hybrids. Reproduced with permission from Ref. [111]. c Schematic illustration of the procedure for preparing S/GCN hybrid sponge. Reproduced with permission from Ref. [113]. Copyright permissions from Royal Society of Chemistry and Wiley–VCH. (Color figure online)
In summary, the advancement in experimental synthesis strategies of pure/doped carbon nitrides and CNBCs has been propelled by the conclusions of DFT studies on CNBMs. Precisely, DFT studies proposed that adjusting the C/N ratio, increasing the concentration of pyridinic-N, heteroatom doping and CNBCs design are effective ways to inhibit the problem of irreversible intercalation/deintercalation, poor conductivity, and instability experienced by pure layered CNBMs in rechargeable batteries. These conclusions from DFT studies have guided the experimental synthesis of CNBMs to fabricate functional CNBMs which exhibit the desired requirements and deliver superior battery performance.
Electrochemical Studies of CNBMs for Energy Storage Devices
The structural/electronic properties and surface functionalities of CNBMs qualify them as promising electrode materials for energy storage devices. In this section, we give an overview of experimental works on carbon nitrides for energy storage devices including LIBs, SIBs and PIBs, Li–S, LABs, LMBs, ZABs, and SSBs.
Lithium-Ion Batteries (LIBs)
LIBs offer benefits such as lightweight, superior energy density, and long cycle life [3, 114, 115]; CNBMs (pure/doped carbon nitrides and CNBCs) have been reported as electrodes.
Pure Carbon Nitrides for LIBs
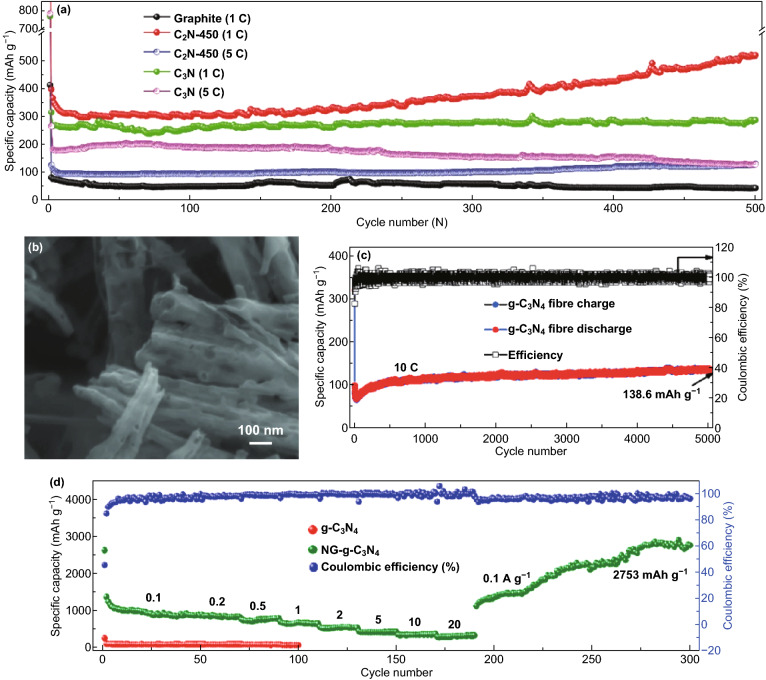
Due to the structural and electronic properties of carbon nitrides, different atomic composition of carbon nitrides has been studied for LIBs. Yin and co-workers reported C3N3 for LIBs but only a low reversible capacity of 197.8 mAh g−1 was obtained at 100 mA g−1 after 300 cycles and a ICE of 34.3% [81]. Xu et al. tested C2N (C2N-450) and C3N for LIBs and at 1C, C3N only retained 285.1 mAh g−1 after 500 cycles, while C2N‐450 maintained a reversible capacity of 516.1 mAh g−1 although it displayed large activation (Fig. 17a) [50]. Other carbon nitride compositions such as C4N and C5N have not been experimentally tested for any metal ion batteries.
Fig. 17.
a Long cycle life of C2N-450, C3N. Reproduced with permission from Ref. [50]. b SEM image of the 1D-g-C3N4 fiber. c Cycling performance of 1D-g-C3N4 fiber structure at a high current density of 10 C. Reproduced with permission from Ref. [46]. d Rate performance for ND-g-C3N4 electrode at current density of 0.1, 0.2, 0.5, 1, 2, 5, 10, and 20 A g−1 and galvanostatic discharge property for g-C3N4 electrode. Reproduced with permission from Ref. [8]. Copyright permissions from Wiley–VCH, and American Chemical Society. (Color figure online)
We reported the synthesis of a porous 1D-C3N4 fiber with large surface area and multiple active sites (see SEM image—Fig. 17b) for LIBs; a reversible capacity of 181.7 mAh g−1 was achieved at 0.5 C and 138.6 mAh g−1 at 10 C (Fig. 17c) [46]. Chen et al. also reported the application of C3N4 (ND-g-C3N4) for LIBs and obtained a capacity of 2753 mAh g−1 after 300 cycles) although significant activation occurred (Fig. 17d). To provide a brief summary, pure carbon nitrides of different crystalline structures have been reported for LIBs, and commendable performances have been recorded (Table 2). However, the issues of ineffective intercalation/deintercalation due to extremely high Li adsorption energy, high Li diffusion barrier, structural deformation, and loss of crystallinity has severely limited the exploitation of their full potential. Therefore, DCNs and CNBCs are necessary to achieve improved performance.
Table 2.
Pure and doped carbon nitrides for LIBs and SIBs
| Battery type | Materials | Synthesis | Morphology | Initial capacity (mAh g−1) | ICEa | Reversible capacity (mAh g−1) | References |
|---|---|---|---|---|---|---|---|
| LIBs | ND-g-C3N4 | Magnesiothermic denitriding method | Porous nanosheet | 2627 @ 100 mA/g | 45.7% | 2753 @ 300 cycles | [8] |
| CN-480–600 | Solid-state Wurtz reaction | Macroporous graphite-like | 575.7 @ 100 mA/g | 19.9% | 197.8 @ 300 cycles | [144] | |
| C2N-450 | Bottom-up wet-chemical reaction. | Stacked sheet | 1629.6 @ 0.1 C | 57.2% | 516.1 @ 500 cycles | [146] | |
| C3N | Bottom-up wet-chemical reaction. | Rod-like | 787.3 @ 0.1 C | 48.7% | 1C, 285.1 @ 500 cycles | [126] | |
| Li-C3N4 | Electrochemical and solid-state reactions | Sheet-like | 188 | – | 38 @ 6 cycles | [121] | |
| C6N8 (C3N4) | Thermal oxidation | – | 250 @ 30 mA/g | – | 50 @ 50 cycles | [122] | |
| 1D-C3N4 fiber | Polyaddition/polycondensation reaction | Porous layered fiber | 419.7 @ 0.5 C | 84.1% | 181.7 @ 200 cycles | [46] | |
| NGC | Carbonization | Graphene-like | 2749 @ 50 mA/g | 50.3% | 1143 @ 200 cycles | [145] | |
| P-MCN-1 | Template synthesis | Spherical particles | 2850 @ 1 A/g | 44.2% | 963 @ 1000 cycles | [105] | |
| SIBs | C6N8 (C3N4) | Thermal oxidation | – | 250 @ 30 mA/g | – | 10 @ 50 cycles | [122] |
| SIBs | S-MCN | Hard template approach | Rod-like morphology | ~850 @ 100 mA/g | 47% | 304.2 @ 100 cycles | [31] |
aICE Initial coulombic efficiency
CNBCs for LIBs
CNBCs can deliver superior structural/electronic properties, enhanced structural stability and extremely large surface area which will boost the overall LIBs performance. In this section, we highlight some significant CNBCs including binary CNBCs and ternary CNBCs.
Binary CNBCs for LIBs
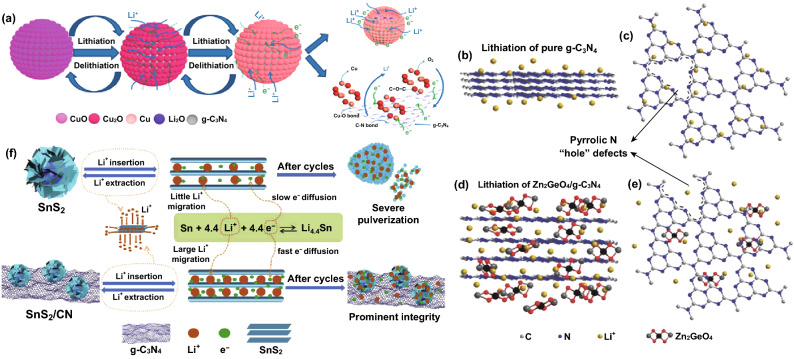
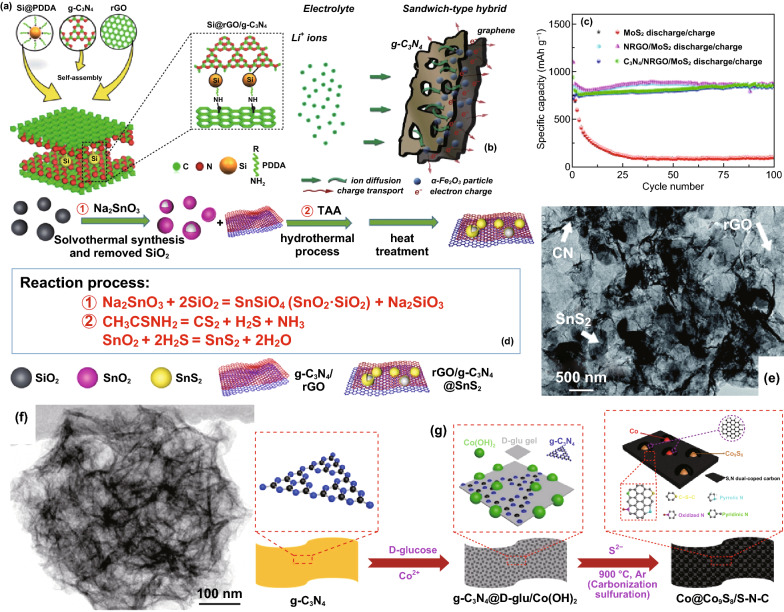
Binary CNBCs involving carbonaceous materials such as graphene oxide are commonly reported because of the inherent π–π stacking, extremely large surface area, surface functional groups, and electronic conductivity. For instance, Fu et al. [110] reported the application of a 2D-2D stacked g-C3N4-rGO hybrid for LIBs. The 2D-2D stacked g-C3N4-rGO hybrid achieved a reversible capacity of 1525 mAh g−1 at 100 mA g−1 and up to 943 mAh g−1 at 1000 mA g−1 [110]. Mohamed et al. designed a CuO/O-doped g-C3N4 composite which delivered a reversible capacity of 738 mAh g−1 after 100 cycles and 503 mAh g−1 after 500 cycles when tested at 100 and 1000 mA g−1, respectively. A depiction of the Li+ storage mechanism is shown in Fig. 18a [116]. Li et al. also reported the application of metal oxide-based composite—Zn2GeO4/g-C3N4 composite for LIBs. The lithium storage mechanism for g-C3N4 and the composite is depicted by the scheme provided in Fig. 18b-e. The composite exhibited a superior reversible capacity of 1370 mAh g−1 at 200 mA g−1 after 140 cycles [111]. Yin et al. designed a composite comprising of SnS2 anchored on g-C3N4 nanosheet, and the SnS2/CN composite followed the conversion and alloying/dealloying lithium storage mechanism (). Schematic of this storage mechanism is provided in Fig. 18f. A high reversible capacity of 444.7 mAh g−1 was delivered at 100 mA g−1 after 100 cycles with structural stability and no pulverization [117].
Fig. 18.
a Schematic description explaining the reaction mechanism of the CuO/O-doped g-C3N4 anode during the charge/discharge process. Reproduced with permission from Ref. [116]. Illustrations of pure (b, c) g-C3N4 and (d, e) Zn2GeO4/g-C3N4 hybrids for Li-insertion viewed from the (b, c) edge and (d, e) basal plan directions. Reproduced with permission from Ref. [111]. f Schematic illustration of the Li-ions diffusion and electronic transport in the SnS2 and SnS2/CN composite electrode during the charge/discharge processes. Reproduced with permission from Ref. [117]. Copyright permissions from Elsevier, Royal Society of Chemistry, American Chemical Society and Wiley–VCH. (Color figure online)
Ternary CNBCs for LIBs
Ternary composites which can provide additional benefits than binary composites have also been reported. Wang et al. reported the application of a Si@rGO/g-C3N4 composite for LIBs. By taking advantage of the interfacial chemical bonding on functionalized rGO/g-C3N4, Si nanoparticles (NPS) were effectively anchored on 2D-composite of rGO/g-C3N4 (Fig. 19a), and such composite design resulted in a reversible capacity of 1354.8 and 799.6 mAh g−1 when cycled at 0.1 and 0.5 C, respectively [118]. Kong et al. also reported a red phosphorus/rGO-C3N4 composite which delivered a capacity of 1032.6 mA g−1 after 600 cycles when tested at 200 mA g−1 [119]. Shi et al. reported the synthesis of a Fe2O3/C3N4-graphene composite for LIBs. The Li storage mechanism of the Fe2O3/CN-G composite is depicted in Fig. 19b, a high reversible capacity of 980 mAh g−1 at 50 mA g−1 after 50 cycles along with a stable rate performance exceeding that of Fe2O3/G [120] (Table 3).
Fig. 19.
a Schematic reaction scheme of the Si@rGO/g-C3N4 hybrid and illustration of Si NPs anchored on rGO/g-C3N4 via strong covalent and hydrogen bonding formed during the pyrolysis process. Reproduced with permission from Ref. [118]. b Schematic diagram of ionic diffusion and charge transport in the porous Fe2O3/CN–G anode with a 2D sandwich-like nanosheet architecture. Reproduced with permission from Ref. [124]. c Cycle performance over the voltage range of 0.01–3.0 V vs. Li/Li+ at the same current density of 100 mA/g for MoS2, NRGO/MoS2, and C3N4/NRGO/MoS2. Reproduced with permission from Ref. [121]. d Schematic synthesis process of rGO/g-C3N4@SnS2. Reproduced with permission from Ref. [123]. e TEM micrograph of porous ternary composite architectures of reduced graphene oxide, SnS2, and CN (GSC6). Reproduced with permission from Ref. [122]. Copyright permissions from Royal Society of Chemistry, Elsevier, and Springer Nature. (Color figure online)
Table 3.
CNBCs for metal-ion batteries LIBs, SIBs and PIBs
| Battery type | Materials | Synthesis | Morphology | Initial capacity (mAh g−1) | ICEa | Reversible capacity (mAh g−1) | References |
|---|---|---|---|---|---|---|---|
| LIBs | huCP/g-C3N4 | Hydrothermal treatment | Carbon fibers with large pores and hollow structure | 1199 @ 1 A/g | ~ 93% | 1030 @ 1000 cycles | [147] |
| g-C3N4–rGO | In-situ chemical synthetic approach | 2D sheets | 3002 @ 100 mA/g | 57% | 1525 @ 50 cycles | [108] | |
| CN-rGO | Hydrothermal synthesis | 3D Sandwich architecture | 1632 @ 50 mA/g | 41.3% | 970 @ 300 cycles | [130] | |
| g-C3N4@rGO | Hydrothermal reaction | Spongy, porous, and tangled ultrathin sheets | 2731 @ 0.1 C | ~89% | 901 @ 50 cycles | [131] | |
| SN/CN | Hydrothermal | Cauliflower-like morphology | 733 @ 0.1 C | 55.4% | ~420 @ 60 cycles | [132] | |
| SnO2@C3N4 | Scalable solid-state reaction | Porous and wrinkled material | 1200 @ 0.1 C | ~50% | 550 @ 100 cycles | [149] | |
| TiO2@CNS (TCNS) | Self-assembly approach/hydrothermal | Spherical Core–shell particles | 359 @ 0.1 C | ~90% | 303 @ 125 cycles | [100] | |
| Zn2GeO4/g-C3N4 | Facile solution approach | ultrathin nanosheet. | 1068 @ 200 mA/g | 58.6% | 1370 @ 140 cycles | [109] | |
| LIBs | NiCo2O4/g-C3N4 | Facile ultrasonic treatment | Nanosheet structure | 1367 @ 100 mA/g | 84.5% | 1252 @ 100 cycles | [150] |
| Li4Ti5O12/g-C3N4 | Solvothermal method | Nanoparticles | 173.7 @ 0.5 C | – | 150.8 @ 502 cycles | [133] | |
| Nitrogen-doped LTO/C (NCLTO) | Thermal decomposition | Irregular sized nanoparticles | 189 @ 1 C | ~92% | 122 @ 500 cycles | [109] | |
| CuO/O-doped g-C3N4 | Hydrothermal | Nanospheres | 980 @ 100 mA/g | 94.7% | 738 @ 100 cycles | [101] | |
| 3D N-rich C3N4@MoS2 | Hydrothermal | Nanosphere | 2390 @ 0.1 C | ~74% | 857 @ 50 cycles | [154] | |
| MoS3/g-C3N4–H+/GO | Sonication/thermal treatment | 2D sheet-like structure | 1728 @ 0.1 mA/g | – | 1450 @ 200 cycles | [103] | |
| MoS2/g-C3N4 | Thermal treatment (calcination) | Spherical particles | 2467 @ 0.05 C | ~41% | 1204 @ 200 cycles | [175] | |
| WS2/g-C3N4 | Solid-state reaction | Nano-sized petal-like sheets | 1933.6 @ 100 mA/g | 63.6% | 622.7 @ 400 cycles | [156] | |
| SnS2/CN | Microwave hydrothermal method | Nanoflower | 1465.9 @ 100 mA/g | 47% | 383.8 | [153] | |
| Co1−xS@g-C3N4 | Solvothermal method | Spherical-like | ~1250 @ 0.1 A/g | ~55% | 789.59 @ 210 cycles | [155] | |
| C3N5/MoS2 | Soft- and hard templating methods | Highly ordered mesoporous nanosheet | 271 @ 100 mA/g | 60% | 193 @ 100 cycles | [112] | |
| SIBs | huCP/g-C3N4 | Hydrothermal treatment | Carbon fibers with large pores and hollow structure | 222 @ 0.1 A/g | – | 345 @ 380 cycles | [147] |
| N-doped MoC | Pyrolysis | Hollow microspheres | 1040 @0.16 A/g | – | 410@ 200 cycles | [172] | |
| Amorphous carbon nitride (ACN) | Copolymerization/direct carbonization | Uniform rhombic dodecahedral shape | 640 @ 83 mA/g | 67.2% | 175 @ 2000 cycles | [173] | |
| N-FLG-800 | Pyrolysis | 2D lamellar structure of with plenty of wrinkles | 256.7 @ 0.5 A/g | 83.5% | 211.3 @ 2000 cycles | [175] | |
| C/g-C3N4 | One-pot calcination | 2D-sheet-like structure | ~200 @ 0.4 A/g | 99 % | ~ 160 @ 400 cycles | [128] | |
| g-C3N4 film | Chemical vapor deposition (CVD) | 2D thin film | – | 98 % | ~6 Ah/g @ 500 cycles | [174] | |
| CN/MoS2-600 | Nanotemplating approach | Corrugated cardboard-like morphology | – | 70% | 605 | [98] | |
| C3N5/MoS2 | Combined soft- and hard templating methods | Ordered mesoporous nanosheet | 126 @ 100 mA/g | 46% | 54 @ 100 cycles | [112] | |
| PIBs | 1D/2D C3N4/rGO | Hydrothermal/freeze drying method | 1D-C3N4 rod infused in-between the 2D-sheet | 682.7 @ 0.5 A/g | 56.8% | 557.4 @ 50 cycles | [54] |
| PIBs | Co3O4@N–C | Ionic liquid-assisted solvothermal method | Spherical | 1229.2 @ 50 mA/g | 48.2% | 448.7 @ 40 cycles | [176] |
| LIBs | Si@rGO/g-C3N4 | Template-free self-assembly and pyrolysis process | Multilayered 3D framework | 1354.8 @ 0.5 C | 70.9% | 799.6 @ 1000 cycles | [159] |
| P/rGO-C3N4 | Ball milling | Nubby structure (40–200 nm) | 2423.4 @ 200 mA/g | 99% | 1032.6 @ 600 cycles | [160] | |
| rGO/g-C3N4/SnS2 (GSC6) | Hydrothermal route | 3D porous structure | 1073.1 @ 100 mA/g | 61.1% | 1118.6 @ 100 cycles | [122] | |
| rGO/g-C3N4@SnS2 | Solvothermal synthesis | Nano-spheres anchored on hybrid sheet | ~600 @ 800 mA/g | —— | 864.9 @ 1000 cycles | [164] | |
| MnO/C3N4/C | Sonication/calcination | 3D Porous sphere | 918.9 @ 0.5 C | 66% | 781.9 @ 86 cycles | [165] | |
| SnO2-TiO2-C3N4 | Self-assembly deposition | Microspherical shaped NPs | 853 @ 0.1 C | —— | 114.1 @ 20 cycles | [188] | |
| Co@Co9S8/S–N-C | Hydrothermal reaction and pyrolysis method | Porous structure with smooth-faced surfaces | 1033.25 @ 0.2 A/g | 60% | 652.1 @ 610 cycles | [166] |
aICE Initial coulombic efficiency
Ternary CNBCs involving metal chalcogenides such as MoS2 and SnS2 have also been studied. Hou et al. reported a C3N4/NRGO/MoS2 composite which exhibited a reversible capacity of 855 mAh g−1 after 100 cycles when tested at 100 mA g−1 (Fig. 19c) [121]. Shah et al. reported the design of a SnS2, rGO and g-C3N4 composite with intimate 2D-2D contact for LIBs. A stepwise detail of the synthesis method and the reaction processes involved is depicted in Fig. 19d. A reversible capacity of 1248.4 mAh g−1 was retained after 276 cycles at 100 mAg−1 [122]. Shi et al. also prepared a rGO/g-C3N4@SnS2 composite with effective surface contact of all constituents which can be seen from the TEM image (Fig. 19e). After 1000 cycles at 800 mA g−1, a reversible capacity of 864.9 mAh g−1 was retained [123].
Sodium and Potassium-Ion Batteries (SIBs and PIBs)
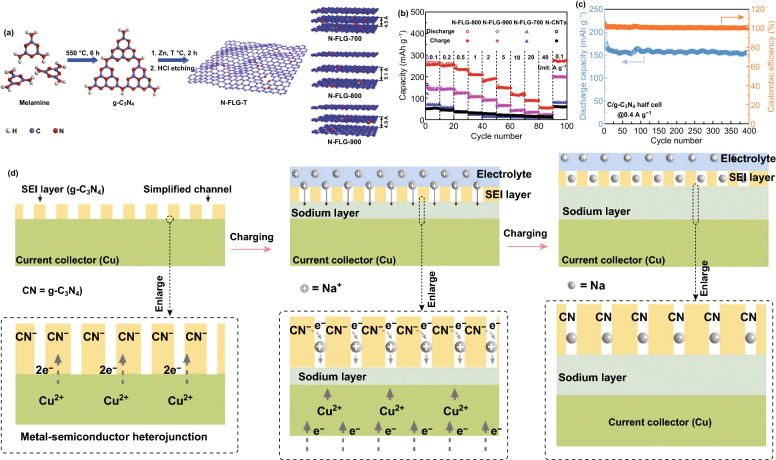
Due to their large-sized ions, one of the major focus for sodium and potassium ion battery electrode materials is the enlargement of their interlayer distance. The low interlayer/d-spacing of graphite (0.334 nm) and its low theoretical capacity (279 mAh g−1) has limited its application in either of these two battery systems [125–127]. g-C3N4 can be a source of N-doped carbon with large interlayer distance; this approach was reported by Qiao et al. who successfully fabricated a series of N-doped graphene 2D-sheet with different interlayer distances (Fig. 20c). The optimized N-doped graphene sheet (N-FLG-800) delivered a superior rate capability of 56.6 mAh g−1 at a current density of 40 A g−1 (Fig. 20e) and outstanding long-cycle stability of 211.3 mAh g−1 after 2000 cycles at 0.5 A g−1 [128]. Weng et al. reported the synthesis of a C/g-C3N4 composite via a simple one-pot synthesis approach, and the composite delivered a capacity of 254 mAh g−1 at 0.1 A g−1 and 160 mAh g−1 at 0.4 A g−1 (see Fig. 20g) [53]. Recently, Chen et al. showed that coating the surface of copper metal current collector with a thin film of 2D-C3N4 can improve Na storage by inhibiting unwanted surface interaction with the liquid electrolyte. The Na+ storage mechanism is illustrated by the scheme in Fig. 20h. At an areal current density of 0.013 mA cm−2, a high areal capacity of 0.036 mAh cm−2 was achieved [129].
Fig. 20.
a Schematic illustration of the synthetic routes for the fabrication of N-FLG-T. b Rate capability of N–CNTs and N-FLG-T at various current densities. Reproduced with permission from Ref. [128]. c Cycling performance of C/g-C3N4 Na half cells at 0.4 A g−1. Reproduced with permission from Ref. [53]. d Illustration of the mechanism of the sodium storage in g-C3N4 film activated Cu foil. Reproduced with permission from Ref. [129]. Copyright permission from Wiley–VCH. (Color figure online)
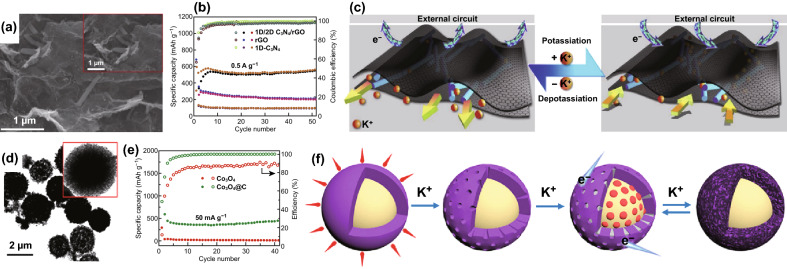
In the case of potassium-ion batteries (PIBs), we showed that the high K+ affinity of 1D-C3N4 fiber facilitates high initial PIBs capacity, but it suffers from poor cycle life and DFT study showed that this was due to the high potassium diffusion barrier and poor conductivity. Guided by this theoretical discovery, we combined 1D-C3N4 fiber with rGO and fabricated a 1D/2D C3N4/rGO composite (Fig. 21a—SEM) which exhibited a larger surface area, superior K+ diffusivity, and improved conductivity. The composite delivered a superior reversible capacity 557.4 mAh g−1 after 50 cycles with impressive cycle stability (Fig. 21b). The K-ion storage mechanism is depicted in Fig. 21c and shows the bi-directional transport of ions through the interlayer of the composite [54]. In our recent work, we resolved the inadequate interlayer spacing of Co3O4 by coating it with N-doped carbon sourced from 1D-C3N4 fiber. The Co3O4@N–C possessed a core–shell morphology (Fig. 21d) in which N–C at the surface of Co3O4 ensured effective transfer of attracted K+ to the Co3O4 core. The composite delivered a superior reversible capacity of 448.7 mAh g−1 unlike pure Co3O4 spheres which only recorded ~ 10 mAh g−1 after 40 cycles (Fig. 21e). The potassium storage mechanism of the composite is depicted in Fig. 21f.
Fig. 21.
a SEM image of 1D/2D C3N4/rGO. b Cycling performances and coulombic efficiency of 1D-C3N4, rGO and 1D/2D C3N4/rGO at of 0.5 A g−1. c Possible mechanism of potassium storage in the 1D/2D C3N4/rGO composite. Reproduced with permission from Ref. [54]. d TEM and e comparison of the cycle life of Co3O4 and Co3O4@N–C at 50 mA g−1. f Scheme illustrating suggested potassiation and depotassiation mechanism of the Co3O4@N-C electrode. Reproduced with permission from Ref. [108]. Copyright permissions from Elsevier and American Chemical Society. (Color figure online)
Lithium-Sulfur Batteries
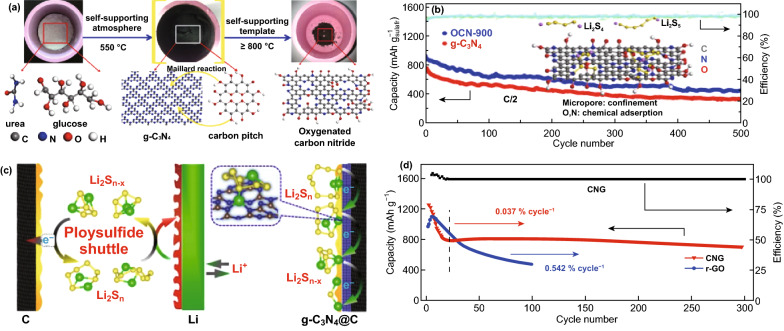
Carbon nitrides have proven to be effective at anchoring LiPSs in Li–S batteries, thereby inhibiting their dissolution in the electrolyte and boosting overall battery performance [134, 135]. Experimental battery studies on CNBMs as sulfur hosts for Li–S battery have confirmed the DFT predictions discussed earlier. For example, Liu et al. reported the application of a 2D graphene-like oxygenated C3N4 via a scalable one-step self-supporting solid-state pyrolysis process (Fig. 22a). The oxygen-rich functional groups possess strong chemical adsorption toward sulfur atoms; hence, they can inhibit the dissolution of polysulfides in Li–S electrolyte. As a cathode material, the resultant electrode delivered an initial columbic efficiency of 98% (S utilization of 84%) with a reversible capacity of 1407.6 mAh g−1 at C/20 (Fig. 22b) thanks to the surface functional groups [136].
Fig. 22.
a Schematic illustration of graphene-like oxygenated carbon nitride (OCN) prepared by one-step self-supporting solid-state pyrolysis. b Cycling performances of the OCN sample prepared at different conditions and g-C3N4 at C/2 after initial activation process to allow complete access of the electrolyte to the active material. Inset: OCN with adsorbed LiPSs molecules. Reproduced with permission from Ref. [136]. c Schematic of the mechanism for polysulfide adsorption by the g-C3N4@CFM cathode. Reproduced with permission from Ref. [67]. d Cycling performance of LiPSs on CNG and r-GO. Reproduced with permission from Ref. [57]. Copyright permissions from American Chemical Society and Wiley–VCH. (Color figure online)
Carbon coating for anchoring soluble polysulfides is an effective strategy to improve the conductivity of electrode materials in Li–S batteries [135]. Based on the conclusion of DFT calculations, Liao and co-workers showed that coating g-C3N4 on carbon-fiber mesh can serve as an effective anchoring strategy for LiPSs, and the g-C3N4@CFM electrode delivered 905 mAh g−1 at 0.1 C. Figure 22c shows the polysulfide adsorption mechanism of the composite electrode [67]. Liang et al. deployed polymeric C3N4 (p-C3N4) as a suitable cathode material to effectively attract LiPSs in common electrolyte solvents (1,3-dioxolane (DOL) and 1,2-dimethoxyethane (DME)). DFT calculations proved that interaction between p-C3N4 and LiPSs can alter the bonding and spatial configuration of the lithium polysulfides, which in turn tunes their redox kinetics. The results of Li–S battery testing in Fig. 22d showed that the electrode consisting of p-C3N4 and rGO (CNG) performed better than its individual constituents, improved stabilization, and kinetics of LiPSs and restrained shuttling effect [57].
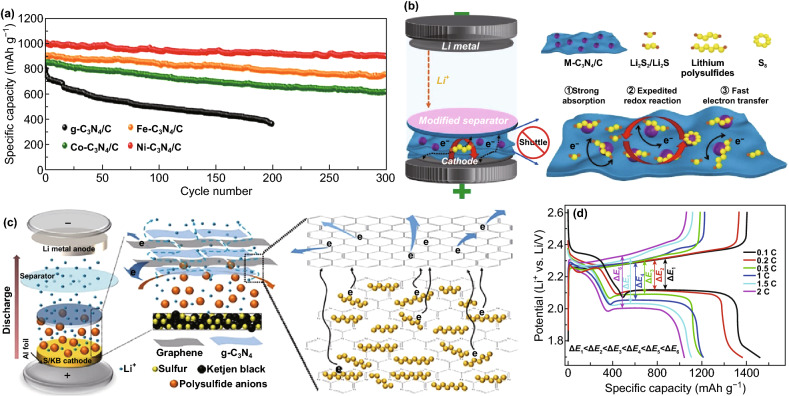
Coating of the commercial polypropylene separator with g-C3N4 is another strategy to facilitate LiPSs adsorption and effectively suppress polysulfide dissolution [134, 135]. Fan et al. reported the blending of commercial separator with carbon black (g-C-coated) and coating it on commercial glass fiber separator for LIPSs adsorption. The g-C3N4-coated separator showed better capacity retention in that after 400 cycles at 0.2 C, it could still deliver 773.2 mAh g−1, while the carbon-coated separator only had 611 mAh g−1 [68]. Chen et al. showed that coating of a transition metal (Fe, Ni, Cu, and Co) coordinated g-C3N4/C commercial separator improved LiPSs adsorption and performance. Experimental Li–S battery testing and DFT calculations concluded that Ni-modified C3N4 facilitated the LiPSs adsorption the best; hence, it outperformed the other metal modified C3N4/C-coated separators in terms of battery performance (Fig. 23a). A schematic of the polysulfide adsorption mechanism of the metal modified electrode is depicted in Fig. 23b. In terms of CNBCs, Qu et al. reported the use of graphene as a conductive material combined with C3N4 to form a g-C3N4/GS interlayer for a sulfur filled kejten black cathode for Li–S battery. Due to the electrostatic forces present as a result of π–π bonding between both materials, graphene and g-C3N4 heterogeneously intercalated with each in a closely packed fashion to form a laminated channel which inhibited diffusion of LiPSs toward the anode (Fig. 23c). The cathode material delivered a high reversible capacity (> 1200 mAh g−1) when cycled at 1 C and retained up to 50% of its capacity after 100 cycles. Moreover, from Fig. 23d a stable and consistent charge/discharge profile was maintained even when the electrode was cycled at various current densities [137]. To summarize the discussion on Li–S, CNBMs have been used for several functions in Li–S batteries and they have shown great promise as advanced electrode materials (Table 4). However, the percentage of S loading needs to be increased from an average of 50% to an average of ~ 80% for all CNBCs reports. Also, the binding mechanism and chemical interactions between the CNBMs and the binder need to be further investigated.
Fig. 23.
a Cycling performance of the heteroatom-doped-C3N4/C-modified separators at 0.5 A g−1. b Schematic illustration of the M-C3N4/C-modified separator to suppress the shuttle of polysulfides and expedite conversion reaction kinetics of polysulfides. Reproduced with permission from Ref. [69]. c Schematic of cell configuration with a laminated structure g-C3N4/GS cathode interlayer. d Charge/discharge profiles for the S/KB@C3N4/GS cathode at various scan rates. Reproduced with permission from Ref. [137]. Copyright permissions from Elsevier and Wiley–VCH. (Color figure online)
Table 4.
CNBMs (pure, doped, and CNBCs) for different functions in Li–S batteries
| Battery function | Materials | Synthesis | Morphology | Initial capacity (mAh g−1) | Sulfur loading | Reversible capacity (mAh g−1) | References |
|---|---|---|---|---|---|---|---|
| Cathode | OCN | One-step self-supporting solid-state pyrolysis (OSSP) | Free-standing 2D sheets | 1407.6 @ C/20 | 56 wt% | 447.3 @ 500 cycles | [177] |
| GCN | Chemical deposition method | Sheet-like morphology | 1250.1 @ C/0.05 | 70.4 wt% | 578.0 @ 750 cycles | [132] | |
| C-NC/GN/g-C3N4 | Low-temperature dissolution and high-temperature carbonization | 3D interconnected network ultrathin sheets | ~ 1270 @ 0.5 C | ~ 78.1 wt% | ~ 1130 @ 500 cycles | [178] | |
| CN@NSHPC | Double-solvent approach with controllable pyrolysis | Solid spheroidal nanoparticle | 1099 @ 0.1 C | 73% (1.7–2 mg/cm2) | 445 @ 500 cycles | [179] | |
| CN/MoS2 | Hydrothermal method | Laminar nanosheet | 1252 @ 0.5 C | 4 mg/cm2 | 680 @ 200 cycles | [184] | |
| rGO/g-C3N4/CNT | Ethanol-assisted spray-drying method | Microsphere-like structure | 1030 @ 0.2 C | 1.5 mg/cm2 | 820 @ 200 cycles | [182] | |
| 3D PCN@rGO | Hydrothermal and freeze-drying | Interconnected sheets stacked by several layers | 1240 @ 0.2 C | ~ 1.5 mg/cm2 | 1005 @ 170 cycles | [181] | |
| Sulfur host | C3N4@CFM | In situ coating method | Carbon fiber mesh | 905 @ 0.1 C | 0.6 mg/cm2 | 801 @ 100 cycles | [140] |
| C3N4/rGO aerogel (CG12) | Hydrothermal, freeze-drying method | Inter-connected thin nanosheets | 1024 @ 0.5 C | 80 wt% (~ 1 mg/cm2) | 770 @ 100 cycles | [138] | |
| MoS2/g-C3N4 | Melt-diffusion method | Multi-layered sheet like structure | 943 @ 1 C | ~ 1.5 mg/cm2 (59.1%) | 569 @ 400 cycles | [102] | |
| Facilitate LiPS redox | CNG (Polymeric g-C3N4 and rGO) | Simple cyro-drying and annealing process | Interconnected thin sheets | ~1250 @ ~ 330 mA/g | ~ 0.6 mg of sulfur on 1.2 mg of CNG or r-GO | over 700 @ 280 cycles | [131] |
| Separator coating for Li–S battery | |||||||
|---|---|---|---|---|---|---|---|
| Battery type | Materials | Synthesis | Morphology | Initial capacity (mAh g−1) | Sulfur loading | Reversible capacity (mAh g−1) | References |
| LiPS-trapping separator | C3N4/CNT@PP | Thermal condensation and oxidization etching with simple filtration process | Long-range continuous framework | ~1350 @ 0.1 C | 7.2 mg/cm2 | 870.5 @ 100 cycles | [180] |
| g-C3N4 | Thermal condensation and oxidization etching | Ultrathin nanosheet | ~1000 | − 4 mg/cm2 | 829 @ 200 cycles | [183] | |
| g-C3N4-C | Polymerization of melamine | Uniform and porous sheet layer | ~1500 @ 0.2 C | 1.7 mg/cm2 | 773.2 @ 400 cycles | [141] | |
| Ni-C3N4/C | Template assisted thermal polymerization | Sheet-like morphology | 999 @ 0.5 A/g | 0.5 mg/cm2 | 893 @ 300 cycles | [142] | |
| Interlayer for LIPs | 2D g-C3N4/Graphene | Ultrasonic treatment | 2D micro sized shaped flakes | ~1600 @ 0.2 C | 50.9 wt% | 710.4 @ 100 cycles | [137] |
| Fe3O4/C3N4 | Ultrasonication | Nanospheres coated tubular structure | 1255 @ 0.2 C | 1.0–6.4 mg/cm2 | 1048 @ 100 cycles | [180] | |
Lithium-Oxygen Batteries
As discussed earlier, DFT predictions show that carbon nitrides are promising for Li-O2 battery because they possess a superior overpotential than most commercial electrodes because molecular level interaction with LiO2 indicates that they form a Li-N bonding which is dependent on the ratio of N in the material and greatly influences their overpotential [60].
Experimental study of pure and CNBCs in Li-O2 batteries has been reported in the literature. For instance, Liu et al. showed that g-C3N4 due to the lower conduction band (CB) potential of g-C3N4 and its low bandgap of ~ 2.7 eV the use of g-C3N4 together with an I− ion redox mediator and non-aqueous electrolyte resulted in a drastic decrease in charging voltage [139].
Heteroatom doping (especially single atom doping) is a well-known method of modulating the electronic properties [140, 141]. Zhao et al. reported the application of a Pt-doped g-C3N4 (CNHS) for Li-O2 battery, and the performance and cycle stability of Pt-g-C3N4 (Pt-CNHS) exceeded those of pure holey g-C3N4. A schematic illustration of the Li-O2 reaction process is presented in Fig. 24a. Also, Li-O2 battery analysis (charge/discharge voltage plateau) shows that Pt-CNHS possess a lower overpotential when compared to CNHS with a round trip efficiency of 69% compared to 65% for CNHS [63]. Gao et al. also reported the design of a tungsten carbide-modified N-doped defective carbon (W2C@NC), and the composite exhibited a significant synergistic interaction (Fig. 24b) and delivered an initial reversible capacity of 10,976 mAh g−1 at a current density of 100 mAh g−1 with a low overpotential and long cycle life [142].
Fig. 24.
a Schematic illustration for the reaction process during cycling. Reproduced with permission from Ref. [63]. b Schematic diagram of the synergistic effect of N-doped carbon layer and W2C nanoparticles as Li-O2 battery catalyst. Reproduced with permission from Ref. [142]. c Comparison of the cycling performances of graphene and G@CN free-standing macroporous electrode. Reproduced with permission from Ref. [143]. d Cycle performance of RuO2@m-BCN in Li-O2 batteries with a current density of 0.3 mA cm−2. Reproduced with permission from Ref. [144]. e First discharge curves of Co3O4 and Ag/g-C3N4/Co3O4 as catalysts at a current density of 100 mA g−1 f First charge/discharge curves of the Ag/g-C3N4/Co3O4 as a catalyst at a current density of 500 mA g−1. Reproduced with permission from Ref. [145]. Copyright permissions from Springer Nature, Elsevier, Wiley–VCH, and Royal Society of Chemistry. (Color figure online)
Luo et al. reported the fabrication of a 3D free-standing graphene@g-C3N4 (G@CN) composite which delivered superior performance (~ 17,300 mAh g−1 at a discharge voltage plateau of 2.6 V), energy efficiency (71%), and stable cycle life compared to the graphene electrode when applied in Li-O2 battery (Fig. 24c) [143]. Lou et al. also reported the application of mesoporous boron-doped carbon nitride (m-BCN) as a support for even distribution of ruthenium oxide (RuO2 nanoparticles). As a Li-O2 cathode, the RuO2@m-BCN composite displayed a superior discharge capacity (2.57 mAh cm−2 = 512 mAh g−1 based on the mass of the composite), smaller overpotential (0.18—discharge and 0.54—recharge), and excellent cycle stability even after 126 cycles. The charge/discharge curve for the composite when cycled at 0.3 mA cm−2 is presented in Fig. 24d [144]. Guo and co-workers reported the synthesis of a ternary composite of Co3O4-modified Ag/g-C3N4 for Li-O2. The synergistic interaction between these three constituents produced a composite with superior battery performance and catalytic activity with stable cycle life, high reversible capacity, and round-trip efficiency (Fig. 24e, f) [145].
Lithium-Metal Batteries
Despite the benefits of lithium metal as a preferred anode for high energy batteries, its large-scale application is hampered by two major issues: performance and safety [1, 146, 147]. These two problems are related in that they are both caused by Li dendrite growth. Moreover, other issues such as formation of SEI film [148–150] and non-uniform Li+ deposition are still of concern. These issues have been tackled in different ways, and because of the surface charges and functional groups of g-C3N4, it has been considered a very useful material for these various solutions. For instance, Luan and co-workers applied O- and N-rich graphene-like g-C3N4 as an effective artificial protective layer for Li-metal anode in half and full LIB and Li–S cells. Pristine g-C3N4 (P-G) and acid-treated g-C3N4 (A-G) were tested for Li-metal, and the A-G-Li exhibited the best wettability, structural stability, and least contact angle of almost zero indicating uniform Li+ distribution (Fig. 25a). From Fig. 25b, c, A-G-Li delivered the highest reversible capacity and superior capacity retention (80%) in both battery tests (LIB and Li–S, respectively) [151].
Fig. 25.
a Schematic illustration and SEM images of the cross-sectional view of Li deposition on pristine Cu and A-G-Cu electrodes before cycling and after depositing 5 mAh cm−2 of Li. b Cycle performance of the Li//LFP full cells with Li, P-G-Li, and A-G-Li. c Rate capability and of the Li//S full cells with Li, P-G-Li, and A-G-Li. Reproduced with permission from Ref. [151]. d Scheme of Li dendrite growth and inhibition depending on Li symmetric cells with g-C3N4 or without addition. e Galvanostatic charge/discharge curves of Li/FeS2 cell based on LiTFSI-DGM-C3N4 electrolyte of at 0.1C in a voltage range of 1—3 V. f Cycling performance of LiTFSI-DGM-C3N4-based Li/FeS2 cell (red circles) and its comparison with Li/LiTFSI-DGM/FeS2 cell. Reproduced with permission from Ref. [152]. g Schematic of the Li nucleation and plating process on Ni foam and g-C3N4@Ni foam. h Discharge capacity and CE of Li@g-C3N4@Ni foam|LiCoO2 and Li@Ni foam|LiCoO2 cells at 1.0 C. i Discharge capacity and CE of Li@g-C3N4@Ni foam|S and Li@Ni foam|S cells at 1.0 C. Reproduced with permission from Ref. [153]. Copyright permissions from American Chemical Society and Wiley–VCH. (Color figure online)
Considering the lithophilic nature of g-C3N4, its incredible mechanical strength, and unique morphology, Hu et al. reported the use of porous g-C3N4 microspheres as a polymeric electrolyte filler for lithium metal anode. Li dendrite was successfully suppressed in the g-C3N4 filler (Fig. 25d). It delivered superior performance and stability to the commercial electrolyte (Fig. 25e, f) [152]. Luo et al. reported the uniform coating of g-C3N4 on a 3D Ni foam (Fig. 25g) to design a g-C3N4@Ni foam which was applied as a current collector for Li-metal anode. The g-C3N4@Ni foam anode delivered superior capacity (Fig. 25h, i) to the pure Ni foam with excellent coulombic efficiency (98%), cycle stability, and capacity retention (72.9% at 1 C after 200 cycles) [153].
Zinc-Air Batteries
The affordability, safety, and high specific energy density of ZABs are some of the reasons for the intensive research into this type of metal-air battery technology. However, the high cost and stability of metal-based catalysts for ZABs application [154, 155] have motivated researchers into designing metal-doped carbon materials [156, 157]. The high nitrogen content of C3N4 makes it a viable material for composite design or as a reliable precursor for N-doped carbon synthesis. For instance, Shinde et al. developed a 3D carbon NP sandwiched in phosphorus and sulfur co-doped g-C3N4 as a metal-free hybrid cathode material for ZAB. Electrochemical battery testing shows that the hybrid air–cathode operated at a voltage of approximately 1.51 V (Fig. 26a), delivered a high reversible capacity of 830 mAh g−1 along with a high energy density of 970 Wh kg−1, 198 mW cm−2 power density with excellent stability even after been recharged more than 210 h [158]. The composite was also tested in a tri-electrode ZABs system, and a schematic of the tri-electrode ZABs is shown in Fig. 26b.
Fig. 26.
a Three-electrode ZABs charge and discharge polarization plots of commercial Pt/C, P-CNS, S-CNS, and P, S-CNS catalyst as air electrodes. b Schematic representation for the tri-electrode ZABs. Reproduced with permission from Ref. [158]. c Charge and discharge polarization curves for three-electrode ZABs with P-CNF, S-CNF, PS-CNF, and Pt/C catalysts as both air electrodes. d Schematic representation of the all-solid-state rechargeable Zn–air battery performance with N-GCNT/FeCo-3 acting as the air cathode. Reproduced with permission from Ref. [160]. e Galvanostatic discharge curves of the N–C or Fe–N–C cathode at the specific discharge current density of 10 mA cm−2. f Photograph of blue-light LED powered by two primary Zn-air batteries connected in series with N–C or Fe–N–C as the cathode catalyst. Reproduced with permission from Ref. [161]. Copyright permissions from Wiley–VCH, American Chemical Society, Elsevier, and Royal Society of Chemistry. (Color figure online)
Shinde et al. also synthesized 3D phosphorus, sulfur co-doped carbon nitride nanofibers. As a cathode material in a primary zinc-air battery, the hybrid nanofiber architecture operated at a 1.49 circuit voltage (consider Fig. 26c) and delivered a reversible capacity of 698 mAh g−1, power and energy density of 231 mW/cm2 and 785 Wh/kg, respectively, with great stability even after 240 h of operation [159, 160]. Ma et al. designed a flexible ZAB with bimetallic iron and cobalt (FeCo) sandwiched in N-doped carbon nanotubes (N-CNT). As an air–cathode in ZAB (Fig. 26d), the FeCo/N-CNT composite operated at an open-circuit voltage of 1.25 V, delivered a power density of 97.8 mW cm−2, and showed excellent stability at 100 mA cm−2 even after 144 cycles [160]. Zhang et al. reported the synthesis of a zinc air electrode using g-C3N4 as a template for N-doped carbon. The Fe–N-C air cathode operated at an open-circuit potential of 1.51 V, delivered a power density of 100 mW cm−2 and was able to power a 3 V blue LED lamp. As seen in Fig. 26e, the air–cathode operated with excellent stability at a current density of 10 mA cm−2 even after 6 h outshining the N–C electrode. The cathode materials were able to light up LED lamps; photographs of the LED lights powered by the cathode materials are presented in Fig. 26f [161].
Solid-State Batteries
Unlike liquid organic electrolyte systems, the possibility of maximizing electron transfer in a solid-state electrolyte and lithium metal battery system is rather arduous because of the reducing property of Li metal when it interacts with most solid-state electrolytes [162]. This reductive process occurs because of the high reduction potential of solid-state electrolytes against that of Li, for example Li10GeP2S12 (1.71 V), Li1.3Ti1.7Al0.3(PO4)3 (2.17 V), and Li0.33La0.56TiO3 (1.75) [163, 164]. While the reductive potential of Li7La3Zr5O12 (0.05 V) is very far from that of Li, it possesses a lithiophobic surface which means that it will experience poor contact and great surface resistance with lithium metal anode. However, this problem can be solved by meticulously coating its surface with a lithophilic material, thereby making changing its lithiophobicity and making Li7La3Zr5O12 lithophilic. Huang et al. used this approach when they reported the in situ formation of a Li3N layer between Li and Li7La3Zr5O12 such that Li+ can be effectively conducted and Li dendrite formation suppressed. This Li+ conducting and electron-insulating Li3N layer was created because of the reaction between molten Li and g-C3N4 to form a Li-C3N4 composite such that the role of C3N4 is for interface formation between Li and Li7La3Zr5O12 (Fig. 27a, b). The composite delivered a high capacity and stable cycle performance when tested in a solid-state battery (Fig. 27c) and maintained a strong/intimate contact with the Garnet (Fig. 27d) [165]. Lu et al. also reported the application of mesoporous carbon nitride (MCN) and acetylene black (AB) as a support for in situ growth of Pt nanoparticles for an all-solid-state lithium air battery system. A schematic of the assembled MCN-based electrode solid-state lithium-air battery is presented in Fig. 27e. As a cathode material in all-solid-state Li-air battery, the Pt@MCN electrode synthesized by Lu et al. recorded a significant high round trip efficiency (87%) at a high discharge voltage of ~ 2.87 V along with a low charge voltage of ~ 3.3 V (Fig. 27f, g) [166]. Sun et al. reported the application of g-C3N4 as a polymeric filler to improve the coupling effect between the amorphous specie and coordination unit of a solid polymer electrolyte in solid-state batteries. When applied as an electrolyte with LiFePO4 in an all-solid-state battery, 60 °C an initial reversible capacity of 161 mAh g−1 was delivered, and after 100 cycles, 155 mAh g−1 was still retained (Fig. 27h) [167].
Fig. 27.
a Schematic illustration of the synthesis process of Li-C3N4 composite and the interfacial contact comparison of Li/garnet and Li-C3N4/garnet. a Garnet SSE presents a lithiophobic surface that forms pristine Li droplets. b Li-C3N4 composite shows an intimate contact with garnet and a Li3N layer is in situ formed at the interface. c Cycling performance of Li-C3N4|garnet|LiFePO4 full cell at a current rate of 0.5 C. d SEM images of the cross sections of Li-C3N4/garnet interfaces. Reproduced with permission from Ref. [165]. e Scheme of the configuration of the MCN-based electrode solid-state lithium-air battery. f Charge and discharge curves of the first cycle of the prepared air electrodes at a current density of 0.02 mA cm−2. g Cycling performance of Pt@MCN electrode at a current density of 0.02 mA cm−2. Reproduced with permission from Ref. [166]. h Capacity and coulombic efficiency versus cycle number for solid-state batteries using SPE and 5% g-C3N4 CSPE at 0.2 C rate. Inset: Schematics of enhanced lithium-ion migration in g-C3N4 CSPE. Reproduced with permission from Ref. [167]. Copyright permissions from Wiley–VCH, Springer Nature and Royal Society of Chemistry. (Color figure online)
Conclusions and Future Perspectives
A comprehensive overview of structure-guided synthesis/fabrication and DFT-guided application of CNBMs for energy storage devices has been discussed in this review. CNBMs, including pure, doped, and CNBCs, exhibit high mechanical strength, excellent structural stability, abundant nitrogen-rich active sites, and surface functionalities, which are important for energy storage devices. Through the DFT-guided approach, CNBMs with superior structural and electronic properties are designed and their electrochemical properties can be modulated for improved performance. Moreover, the structure-guided approach facilitates the synthesis and fabrication of pure/doped CxNy materials and CNBC’s, respectively. The massive attention into CNBMs for electrochemical energy storage and conversion can be attributed to the combination of these two approaches, and they are paramount to the advancement in this field [140, 168–172].
Despite the significant progress made so far in the study of CNBMs for energy storage devices, there are still unresolved issues; therefore, we provide some perspectives to resolve them. Firstly, the important preconditions must be considered when carrying out a DFT-guided synthesis/design of CNBMs. These prerequisites include the crystal phase and lattice parameter of the material, the interlayer distance (if it is a multi-layered material) and the exposed surface facet if it is a single-layered material, the ratio of C/N or other elements and type of nitrogen (pyridinic or graphitic-N) dominant in the structure of the material. These preconditions are critical when designing a material via a DFT-guided approach. Secondly, a clear and concise investigation of the metal ion storage mechanism of CNBMs is essential. This will provide a fundamental/atomic-scale understanding of the electrochemical processes occurring at the surface and carbon matrix of the CNBMs. It will also show which surface functionalities of CNBMs impact their metal ion storage mechanism and how the interfacial interactions can be optimized. Thirdly, in situ characterization of CNBMs is required for a deeper understanding of structural changes, surface functional group transformation, and metal ion storage. For example, through in situ Fourier-transform infrared spectroscopy (FTIR), functional groups such as hydroxyl, amine, and carboxylic groups on the surface of CNBMs can be observed. Phase and oxidation state changes can also be identified through in situ X-ray photoelectron spectroscopy (XPS) analysis. These advanced characterization techniques can provide crucial information that will enlighten researchers on the relationship between the electronic properties and the CNBMs. Lastly, theoretical/computational studies of CNBMs have proven to be fundamental in understanding their electronic properties for energy storage devices; hence, it must remain a crucial part of its future research. However, DFT is limited in studying CNBMs for energy storage device because it involves the use of metal atoms instead of ions, and it cannot effectively account for the effect of electrolyte, SEI layer formation, pulverization of electrode, the surface charge of materials and interfacial interactions. Therefore, improvement of existing DFT tools/functionals or the development of new ones that can account for these critical parameters is essential to achieve a closer alignment between DFT studies and experimental testing.
On a general note, the study of CNBMs is still at the early stage. Therefore, there is much room for improvement, and resolving these issues will surely propel the research to advance at a rapid pace.
Acknowledgements
The authors gratefully acknowledge financial support from the Australia Research Council Discovery Projects (DP160102627 and DP1701048343) of Australia and Shenzhen Peacock Plan of China (KQTD2016112915051055), and the 111 Project (D20015) of China Three Gorges University.
References
- 1.Su Z, Liu J, Li M, Zhu Y, Qian S, et al. Defect engineering in titanium-based oxides for electrochemical energy storage devices. Electrochem. Energ. Rev. 2020;3:286–343. doi: 10.1007/s41918-020-00064-5. [DOI] [Google Scholar]
- 2.Zhang Y, Shi G, Qin J, Lowe SE, Zhang S, Zhao H, Zhong YL. Recent progress of direct ink writing of electronic components for advanced wearable devices. ACS Appl. Electron. Mater. 2019;1(9):1718–1734. doi: 10.1021/acsaelm.9b00428. [DOI] [Google Scholar]
- 3.Wu Z, Xie J, Xu ZJ, Zhang S, Zhang Q. Recent progress in metal–organic polymers as promising electrodes for lithium/sodium rechargeable batteries. J. Mater. Chem. A. 2019;7(9):4259–4290. doi: 10.1039/C8TA11994E. [DOI] [Google Scholar]
- 4.Wu C, Tong X, Ai Y, Liu D-S, Yu P, Wu J, Wang ZM. A review: enhanced anodes of Li/Na-ion batteries based on yolk–shell structured nanomaterials. Nano Micro Lett. 2018;10(3):40. doi: 10.1007/s40820-018-0194-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Liu C, Liu X, Tan J, Wang Q, Wen H, Zhang C. Nitrogen-doped graphene by all-solid-state ball-milling graphite with urea as a high-power lithium ion battery anode. J. Power Sources. 2017;342:157–164. doi: 10.1016/j.jpowsour.2016.11.110. [DOI] [Google Scholar]
- 6.He B, Wang Y, Zhai Q, Qiu P, Dong G, Liu X, Chen Y, Li Z. From polymeric carbon nitride to carbon materials: extended application to electrochemical energy conversion and storage. Nanoscale. 2020;12(16):8636–8646. doi: 10.1039/D0NR01612H. [DOI] [PubMed] [Google Scholar]
- 7.Hao Q, Jia G, Wei W, Vinu A, Wang Y, Arandiyan H, Ni B-J. Graphitic carbon nitride with different dimensionalities for energy and environmental applications. Nano Res. 2020;13(1):18–37. doi: 10.1007/s12274-019-2589-z. [DOI] [Google Scholar]
- 8.Chen J, Mao Z, Zhang L, Wang D, Xu R, Bie L, Fahlman BD. Nitrogen-deficient graphitic carbon nitride with enhanced performance for lithium ion battery anodes. ACS Nano. 2017;11(12):12650–12657. doi: 10.1021/acsnano.7b07116. [DOI] [PubMed] [Google Scholar]
- 9.Pang H, Luo Y, Yan Y, Zheng S, Xue H. Graphitic carbon nitride based materials for electrochemical energy storage. J. Mater. Chem. A. 2018;7(3):901–924. doi: 10.1039/C8TA08464E. [DOI] [Google Scholar]
- 10.Zhu B, Cheng B, Zhang L, Yu J. Review on dft calculation of s-triazine-based carbon nitride. Carbon Energy. 2019;1(1):32–56. doi: 10.1002/cey2.1. [DOI] [Google Scholar]
- 11.Bafekry A, Shayesteh SF, Peeters FM. Two-dimensional carbon nitride (2DCN) nanosheets: tuning of novel electronic and magnetic properties by hydrogenation, atom substitution and defect engineering. J. Appl. Phys. 2019;126(21):215104. doi: 10.1063/1.5120525. [DOI] [Google Scholar]
- 12.Shibuya R, Kondo T, Nakamura J. Active sites in nitrogen-doped carbon materials for oxygen reduction reaction. Carbon-Based Metal-Free Catalysts. 2018;1:227–249. doi: 10.1002/9783527811458.vol1-ch8. [DOI] [Google Scholar]
- 13.Lakhi KS, Park D-H, Al-Bahily K, Cha W, Viswanathan B, Choy J-H, Vinu A. Mesoporous carbon nitrides: synthesis, functionalization, and applications. Chem. Soc. Rev. 2017;46(1):72–101. doi: 10.1039/C6CS00532B. [DOI] [PubMed] [Google Scholar]
- 14.Xia H, Xu Q, Zhang J. Recent progress on two-dimensional nanoflake ensembles for energy storage applications. Nano-Micro Lett. 2018;10(4):66. doi: 10.1007/s40820-018-0219-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Qiu H, Wang Z, Sheng X. First-principles prediction of an intrinsic half-metallic graphitic hydrogenated carbon nitride. Phys. Lett. A. 2013;377(3–4):347–350. doi: 10.1016/j.physleta.2012.11.050. [DOI] [Google Scholar]
- 16.Hussain T, Searles DJ, Hankel M. Insights into the trapping mechanism of light metals on C2N-h2d: utilisation as an anode material for metal ion batteries. Carbon. 2020;160:125–132. doi: 10.1016/j.carbon.2019.12.063. [DOI] [Google Scholar]
- 17.Zheng Y, Li H, Yuan H, Fan H, Li W, Zhang J. Understanding the anchoring effect of graphene, BN, C2N and C3N4 monolayers for lithium–polysulfides in Li–S batteries. Appl. Surf. Sci. 2018;434:596–603. doi: 10.1016/j.apsusc.2017.10.230. [DOI] [Google Scholar]
- 18.Zhu B, Zhang L, Cheng B, Yu J. First-principle calculation study of tri-s-triazine-based g-C3N4: a review. Appl. Cat. B Environ. 2018;224:983–999. doi: 10.1016/j.apcatb.2017.11.025. [DOI] [Google Scholar]
- 19.Weng Q, Li G, Feng X, Nielsch K, Golberg D, Schmidt OG. Electronic and optical properties of 2D materials constructed from light atoms. Adv. Mater. 2018;30(46):1801600. doi: 10.1002/adma.201801600. [DOI] [PubMed] [Google Scholar]
- 20.Wu H-Z, Liu L-M, Zhao S-J. The effect of water on the structural, electronic and photocatalytic properties of graphitic carbon nitride. Phys. Chem. Chem. Phys. 2014;16(7):3299–3304. doi: 10.1039/C3CP54333A. [DOI] [PubMed] [Google Scholar]
- 21.Wang A, Wang C, Fu L, Wong-Ng W, Lan Y. Recent advances of graphitic carbon nitride-based structures and applications in catalyst, sensing, imaging, and leds. Nano-Micro Lett. 2017;9(4):47. doi: 10.1007/s40820-017-0148-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang J, Chen Y, Wang X. Two-dimensional covalent carbon nitride nanosheets: synthesis, functionalization, and applications. Energy Environ. Sci. 2015;8(11):3092–3108. doi: 10.1039/C5EE01895A. [DOI] [Google Scholar]
- 23.Mahmood J, Lee EK, Jung M, Shin D, Choi H-J, et al. Two-dimensional polyaniline (C3N) from carbonized organic single crystals in solid state. Proc. Natl. Acad. Sci. 2016;113(27):7414–7419. doi: 10.1073/pnas.1605318113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Li L, Kong X, Leenaerts O, Chen X, Sanyal B, Peeters FM. Carbon-rich carbon nitride monolayers with dirac cones: dumbbell C4N. Carbon. 2017;118:285–290. doi: 10.1016/j.carbon.2017.03.045. [DOI] [Google Scholar]
- 25.Peng B, Mortazavi B, Zhang H, Shao H, Xu K, et al. Tuning thermal transport in C3N monolayers by adding and removing carbon atoms. Phys. Rev. Appl. 2018;10(3):034046. doi: 10.1103/PhysRevApplied.10.034046. [DOI] [Google Scholar]
- 26.Pu C, Zhou D, Li Y, Liu H, Chen Z, Wang Y, Ma Y. Two-dimensional C4N global minima: unique structural topologies and nanoelectronic properties. J. Phys. Chem. C. 2017;121(5):2669–2674. doi: 10.1021/acs.jpcc.6b09960. [DOI] [Google Scholar]
- 27.Wang D, Li H, Zhang L, Sun Z, Han D, Niu L, Zhao J. 2D nitrogen-containing carbon material C5N as potential host material for lithium polysulfides: a first-principles study. Adv. Theory Simul. 2019;2(2):1800165. doi: 10.1002/adts.201800165. [DOI] [Google Scholar]
- 28.Lin H, Jin R, Wang A, Zhu S, Li H. Transition metal embedded C2N with efficient polysulfide immobilization and catalytic oxidation for advanced lithium-sulfur batteries: a first principles study. Ceram. Int. 2019;45(14):17996–18002. doi: 10.1016/j.ceramint.2019.06.018. [DOI] [Google Scholar]
- 29.Tian B, Huang T, Guo J, Shu H, Wang Y, Dai J. Performance effects of doping engineering on graphene-like C3N as an anode material for alkali metal ion batteries. Mater. Sci. Semicond. Proc. 2020;109:104946. doi: 10.1016/j.mssp.2020.104946. [DOI] [Google Scholar]
- 30.Xu M, Wang H, Sun S, Li H, Li X, Chen Y, Ni Y. First-principles study of metal atoms adsorption on 2D dumbbell C4N. Phys. Status Solidi B. 2020;257(1):1900205. doi: 10.1002/pssb.201900205. [DOI] [Google Scholar]
- 31.Cha W, Kim IY, Lee JM, Kim S, Ramadass K, Gopalakrishnan K, Premkumar S, Umapathy S, Vinu A. Sulfur-doped mesoporous carbon nitride with an ordered porous structure for sodium-ion batteries. ACS Appl. Mater. Interfaces. 2019;11(30):27192–27199. doi: 10.1021/acsami.9b07657. [DOI] [PubMed] [Google Scholar]
- 32.Molaei M, Mousavi-Khoshdel SM, Ghiasi M. Exploring the effect of phosphorus doping on the utility of g-C3N4 as an electrode material in Na-ion batteries using DFT method. J. Mol. Model. 2019;25(8):256. doi: 10.1007/s00894-019-4109-1. [DOI] [PubMed] [Google Scholar]
- 33.Balogun M-S, Wu Z, Luo Y, Qiu W, Fan X, Long B, Huang M, Liu P, Tong Y. High power density nitridated hematite (α-Fe2O3) nanorods as anode for high-performance flexible lithium ion batteries. J. Power Sources. 2016;308:7–17. doi: 10.1016/j.jpowsour.2016.01.043. [DOI] [Google Scholar]
- 34.Kim S, Hankel M, Cha W, Singh G, Lee JM, Kim IY, Vinu A. Theoretical and experimental investigations of mesoporous C3N5/MoS2 hybrid for lithium and sodium ion batteries. Nano Energy. 2020;72:104702. doi: 10.1016/j.nanoen.2020.104702. [DOI] [Google Scholar]
- 35.Wu X, Kang F, Duan W, Li J. Density functional theory calculations: a powerful tool to simulate and design high-performance energy storage and conversion materials. Prog. Nat. Sci. 2019;29(3):247–255. doi: 10.1016/j.pnsc.2019.04.003. [DOI] [Google Scholar]
- 36.Li M, Gould T, Su Z, Li S, Pan F, Zhang S. Electrochromic properties of Li4Ti5O12: from visible to infrared spectrum. Appl. Phys. Lett. 2019;115(7):073902. doi: 10.1063/1.5099330. [DOI] [Google Scholar]
- 37.Wang Y, Jiao Z, Ma S, Guo Y. Probing C3N/graphene heterostructures as anode materials for Li-ion batteries. J. Power Sources. 2019;413:117–124. doi: 10.1016/j.jpowsour.2018.12.031. [DOI] [Google Scholar]
- 38.Ding Y, Xiao B, Li J, Deng Q, Xu Y, Wang H, Rao D. Improved transport properties and novel Li diffusion dynamics in van der waals C2N/graphene heterostructure as anode materials for lithium-ion batteries: a first-principles investigation. J. Phys. Chem. C. 2019;123(6):3353–3367. doi: 10.1021/acs.jpcc.8b11044. [DOI] [Google Scholar]
- 39.Adekoya D, Zhang S, Hankel M. 1D/2D C3N4/graphene composite as a preferred anode material for lithium ion batteries: importance of heterostructure design via DFT computation. ACS Appl. Mater. Interfaces. 2020;12(23):25875–25883. doi: 10.1021/acsami.0c04900. [DOI] [PubMed] [Google Scholar]
- 40.Liu Q, Xiao B, Cheng J, Li Y-C, Li Q, et al. Carbon excess C3N: a potential candidate as Li-ion battery material. ACS Appl. Mater. Interfaces. 2018;10(43):37135–37141. doi: 10.1021/acsami.8b14183. [DOI] [PubMed] [Google Scholar]
- 41.Jain A, Shin Y, Persson KA. Computational predictions of energy materials using density functional theory. Nat. Rev. Mater. 2016;1(1):15004. doi: 10.1038/natrevmats.2015.4. [DOI] [Google Scholar]
- 42.Wu M, Wang Q, Sun Q, Jena P. Functionalized graphitic carbon nitride for efficient energy storage. J. Phys. Chem. C. 2013;117(12):6055–6059. doi: 10.1021/jp311972f. [DOI] [Google Scholar]
- 43.Mao Y, Duan H, Xu B, Zhang L, Hu Y, et al. Lithium storage in nitrogen-rich mesoporous carbon materials. Energy Environ. Sci. 2012;5(7):7950–7955. doi: 10.1039/c2ee21817h. [DOI] [Google Scholar]
- 44.Veith GM, Baggetto L, Adamczyk LA, Guo B, Brown SS, et al. Electrochemical and solid-state lithiation of graphitic C3N4. Chem. Mater. 2013;25(3):503–508. doi: 10.1021/cm303870x. [DOI] [Google Scholar]
- 45.Hankel M, Ye D, Wang L, Searles DJ. Lithium and sodium storage on graphitic carbon nitride. J. Phys. Chem. C. 2015;119(38):21921–21927. doi: 10.1021/acs.jpcc.5b07572. [DOI] [Google Scholar]
- 46.Adekoya D, Gu X, Rudge M, Wen W, Lai C, Hankel M, Zhang S. Carbon nitride nanofibres with exceptional lithium storage capacity: from theoretical prediction to experimental implementation. Adv. Funct. Mater. 2018;28(50):1803972. doi: 10.1002/adfm.201803972. [DOI] [Google Scholar]
- 47.Pan H. Graphitic carbon nitride nanotubes as Li-ion battery materials: a first-principles study. J. Phys. Chem. C. 2014;118(18):9318–9323. doi: 10.1021/jp4122722. [DOI] [Google Scholar]
- 48.Hankel M, Searles DJ. Lithium storage on carbon nitride, graphenylene and inorganic graphenylene. J. Phys. Chem. C. 2016;18(21):14205–14215. doi: 10.1039/C5CP07356A. [DOI] [PubMed] [Google Scholar]
- 49.Wu D, Yang B, Chen H, Ruckenstein E. Nitrogenated holey graphene C2N monolayer anodes for lithium- and sodium-ion batteries with high performance. Energy Storage Mater. 2019;16:574–580. doi: 10.1016/j.ensm.2018.09.001. [DOI] [Google Scholar]
- 50.Xu J, Mahmood J, Dou Y, Dou S, Li F, Dai L, Baek JB. 2D frameworks of C2N and C3N as new anode materials for lithium-ion batteries. Adv. Mater. 2017;29(34):1702007. doi: 10.1002/adma.201702007. [DOI] [PubMed] [Google Scholar]
- 51.Guo G, Wang R, Luo S, Ming B, Wang C, et al. Metallic two-dimensional C3N allotropes with electron and ion channels for high-performance Li-ion battery anode materials. Appl. Surf. Sci. 2020;518:146254. doi: 10.1016/j.apsusc.2020.146254. [DOI] [Google Scholar]
- 52.Yang C, Zhang X, Ma J, Shi B, Zhang H, et al. Ultrahigh capacity of monolayer dumbbell C4N as a promising anode material for lithium-ion battery. J. Electrochem. Soc. 2020;167(2):020538. doi: 10.1149/1945-7111/ab6bbd. [DOI] [Google Scholar]
- 53.Weng GM, Xie Y, Wang H, Karpovich C, Lipton J, et al. A promising carbon/g-C3N4 composite negative electrode for a long-life sodium-ion battery. Angew. Chem. Int. Ed. 2019;131(39):13865–13871. doi: 10.1002/ange.201905803. [DOI] [PubMed] [Google Scholar]
- 54.Adekoya D, Li M, Hankel M, Lai C, Balogun M-S, Tong Y, Zhang S. Design of a 1D/2D C3N4/rGO composite as an anode material for stable and effective potassium storage. Energy Storage Mater. 2019;25:495–501. doi: 10.1016/j.ensm.2019.09.033. [DOI] [Google Scholar]
- 55.Bhauriyal P, Mahata A, Pathak B. Graphene-like carbon–nitride monolayer: a potential anode material for Na-and K-ion batteries. J. Phys. Chem. C. 2018;122(5):2481–2489. doi: 10.1021/acs.jpcc.7b09433. [DOI] [Google Scholar]
- 56.Li T, He C, Zhang W. A novel porous C4N4 monolayer as a potential anchoring material for lithium–sulfur battery design. J. Mater. Chem. A. 2019;7(8):4134–4144. doi: 10.1039/C8TA10933H. [DOI] [Google Scholar]
- 57.Liang J, Yin L, Tang X, Yang H, Yan W, et al. Kinetically enhanced electrochemical redox of polysulfides on polymeric carbon nitrides for improved lithium–sulfur batteries. ACS Appl. Mater. Interfaces. 2016;8(38):25193–25201. doi: 10.1021/acsami.6b05647. [DOI] [PubMed] [Google Scholar]
- 58.Meng Z, Xie Y, Cai T, Sun Z, Jiang K, Han W-Q. Graphene-like g-C3N4 nanosheets/sulfur as cathode for lithium–sulfur battery. Electrochim. Acta. 2016;210:829–836. doi: 10.1016/j.electacta.2016.06.032. [DOI] [Google Scholar]
- 59.Shinde SS, Lee CH, Yu J-Y, Kim D-H, Lee SU, Lee J-H. Hierarchically designed 3D holey C2N aerogels as bifunctional oxygen electrodes for flexible and rechargeable Zn-air batteries. ACS Nano. 2017;12(1):596–608. doi: 10.1021/acsnano.7b07473. [DOI] [PubMed] [Google Scholar]
- 60.Je M, Chung Y-C. Investigation of the initial reactions of lithium oxides on the graphitic carbon nitrides (g-C3N4) for catalyst in non-aqueous lithium—air batteries: a first-principles calculations. Thin Solid Films. 2018;660:186–190. doi: 10.1016/j.tsf.2018.06.017. [DOI] [Google Scholar]
- 61.Nong W, Li Y, Wang C. C3N monolayer with substitutional doping and strain modulation serving as anode material of lithium-ion batteries. Appl. Surf. Sci. 2020;510:145324. doi: 10.1016/j.apsusc.2020.145324. [DOI] [Google Scholar]
- 62.Weng G-M, Xie Y, Wang H, Karpovich C, Lipton J, et al. A promising carbon/g-C3N4 composite negative electrode for a long-life sodium-ion battery. Angew. Chem. Int. Ed. 2019;58(39):13727–13733. doi: 10.1002/anie.201905803. [DOI] [PubMed] [Google Scholar]
- 63.Zhao W, Wang J, Yin R, Li B, Huang X, Zhao L, Qian L. Single-atom pt supported on holey ultrathin g-C3N4 nanosheets as efficient catalyst for Li-O2 batteries. J. Colloid Inter. Sci. 2020;564:28–36. doi: 10.1016/j.jcis.2019.12.102. [DOI] [PubMed] [Google Scholar]
- 64.Guo G-C, Wang R-Z, Ming B-M, Wang C, Luo S-W, Zhang M, Yan H. C3N/phosphorene heterostructure: a promising anode material in lithium-ion batteries. J. Mater. Chem. A. 2019;7(5):2106–2113. doi: 10.1039/C8TA10972A. [DOI] [Google Scholar]
- 65.Lin H, Jin R, Zhu S, Huang Y. C3N/blue phosphorene heterostructure as a high rate-capacity and stable anode material for lithium ion batteries: insight from first principles calculations. Appl. Surf. Sci. 2020;505:144518. doi: 10.1016/j.apsusc.2019.144518. [DOI] [Google Scholar]
- 66.Bao J, Li H, Duan Q, Jiang D, Liu W, et al. Graphene-like C3N/blue phosphorene heterostructure as a potential anode material for li/na-ion batteries: a first principles study. Solid State Ionics. 2020;345:115160. doi: 10.1016/j.ssi.2019.115160. [DOI] [Google Scholar]
- 67.Liao K, Mao P, Li N, Han M, Yi J, He P, Sun Y, Zhou H. Stabilization of polysulfides via lithium bonds for Li–S batteries. J. Mater. Chem. A. 2016;4(15):5406–5409. doi: 10.1039/C6TA00054A. [DOI] [Google Scholar]
- 68.Feng N, Liu F, Huang M, Zheng A, Wang Q, et al. Unravelling the efficient photocatalytic activity of boron-induced Ti 3+ species in the surface layer of TiO2. Sci. Rep. 2016;6:34765. doi: 10.1038/srep34765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Chen M, Zhao X, Li Y, Zeng P, Liu H, et al. Kinetically elevated redox conversion of polysulfides of lithium-sulfur battery using a separator modified with transition metals coordinated g-C3N4 with carbon-conjugated. Chem. Eng. J. 2020;385:123905. doi: 10.1016/j.cej.2019.123905. [DOI] [Google Scholar]
- 70.Xiao B, Rojo T, Li X. Hard carbon as sodium-ion battery anodes: progress and challenges. Chemsuschem. 2019;12(1):133–144. doi: 10.1002/cssc.201801879. [DOI] [PubMed] [Google Scholar]
- 71.Gabaudan V, Stievano L, Monconduit L, Berthelot R. Snapshot on negative electrode materials for potassium-ion batteries. Front. Energy Res. 2019;7:46. doi: 10.3389/fenrg.2019.00046. [DOI] [Google Scholar]
- 72.Garay-Tapia AM, Romero AH, Barone V. Lithium adsorption on graphene: from isolated adatoms to metallic sheets. J. Chem. Theory Comput. 2012;8(3):1064–1071. doi: 10.1021/ct300042p. [DOI] [PubMed] [Google Scholar]
- 73.Ullah S, Denis PA, Sato F. Unusual enhancement of the adsorption energies of sodium and potassium in sulfur − nitrogen and silicon − boron codoped graphene. ACS Omega. 2018;3(11):15821–15828. doi: 10.1021/acsomega.8b02500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Lugo-Solis A, Vasiliev I. Ab initio study of K adsorption on graphene and carbon nanotubes: role of long-range ionic forces. Phys. Rev. B. 2007;76(23):235431. doi: 10.1103/PhysRevB.76.235431. [DOI] [Google Scholar]
- 75.Liu T, Jin Z, Liu D-X, Du C, Wang L, Lin H, Li Y. A density functional theory study of high-performance pre-lithiated MS2 (M = Mo, W, V) monolayers as the anode material of lithium ion batteries. Sci. Rep. 2020;10(1):6897. doi: 10.1038/s41598-020-63743-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Sun X, Wang Z, Fu YQ. Adsorption and diffusion of sodium on graphene with grain boundaries. Carbon. 2017;116:415–421. doi: 10.1016/j.carbon.2017.01.024. [DOI] [Google Scholar]
- 77.Yang J, Yuan Y, Chen G. First–principles study of potassium adsorption and diffusion on graphene. Mol. Phys. 2019;118(1):1–7. doi: 10.1080/00268976.2019.1581291. [DOI] [Google Scholar]
- 78.Eftekhari A. Low voltage anode materials for lithium-ion batteries. Energy Storage Mater. 2017;7:157–180. doi: 10.1016/j.ensm.2017.01.009. [DOI] [Google Scholar]
- 79.Kim J-H, Kim DK. Conversion-alloying anode materials for na-ion batteries: recent progress, challenges, and perspective for the future. J. Korean Ceram. Soc. 2018;55(4):307–324. doi: 10.4191/kcers.2018.55.4.07. [DOI] [Google Scholar]
- 80.Jian Z, Luo W, Ji X. Carbon electrodes for K-ion batteries. J. Am. Chem. Soc. 2015;137(36):11566–11569. doi: 10.1021/jacs.5b06809. [DOI] [PubMed] [Google Scholar]
- 81.Yin H, Guo Q, He D, Li J, Sun S. Structural characterization and electrochemical performance of macroporous graphite-like C3N3 prepared by the wurtz reaction and heat treatment. RSC Adv. 2017;7(69):44001–44008. doi: 10.1039/C7RA07707F. [DOI] [Google Scholar]
- 82.Zhu J, Xiao P, Li H, Carabineiro SAC. Graphitic carbon nitride: synthesis, properties, and applications in catalysis. ACS Appl. Mater. Interfaces. 2014;6(19):16449–16465. doi: 10.1021/am502925j. [DOI] [PubMed] [Google Scholar]
- 83.Gong Y, Li M, Wang Y. Carbon nitride in energy conversion and storage: recent advances and future prospects. Chemsuschem. 2015;8(6):931–946. doi: 10.1002/cssc.201403287. [DOI] [PubMed] [Google Scholar]
- 84.Ong W-J, Tan L-L, Ng YH, Yong S-T, Chai S-P. Graphitic carbon nitride (g-C3N4)-based photocatalysts for artificial photosynthesis and environmental remediation: are we a step closer to achieving sustainability? Chem. Rev. 2016;116(12):7159–7329. doi: 10.1021/acs.chemrev.6b00075. [DOI] [PubMed] [Google Scholar]
- 85.Dong X, Cheng F. Recent development in exfoliated two-dimensional g-C3N4 nanosheets for photocatalytic applications. J. Mater. Chem. A. 2015;3(47):23642–23652. doi: 10.1039/C5TA07374J. [DOI] [Google Scholar]
- 86.Guo Q, Yang Q, Yi C, Zhu L, Xie Y. Synthesis of carbon nitrides with graphite-like or onion-like lamellar structures via a solvent-free route at low temperatures. Carbon. 2005;43(7):1386–1391. doi: 10.1016/j.carbon.2005.01.005. [DOI] [Google Scholar]
- 87.Li J, Cao C, Hao J, Qiu H, Xu Y, Zhu H. Self-assembled one-dimensional carbon nitride architectures. Diam. Relat. Mater. 2006;15(10):1593–1600. doi: 10.1016/j.diamond.2006.01.013. [DOI] [Google Scholar]
- 88.Niu P, Zhang L, Liu G, Cheng HM. Graphene like carbon nitride nanosheets for improved photocatalytic activities. Adv. Funct. Mater. 2012;22(22):4763–4770. doi: 10.1002/adfm.201200922. [DOI] [Google Scholar]
- 89.Mahmood J, Lee EK, Jung M, Shin D, Jeon I-Y, et al. Nitrogenated holey two-dimensional structures. Nat. Commun. 2015;6(1):6486. doi: 10.1038/ncomms7486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Zhang X, Xie X, Wang H, Zhang J, Pan B, Xie Y. Enhanced photoresponsive ultrathin graphitic-phase C3N4 nanosheets for bioimaging. J. Am. Chem. Soc. 2013;135(1):18–21. doi: 10.1021/ja308249k. [DOI] [PubMed] [Google Scholar]
- 91.Schwinghammer K, Mesch MB, Duppel V, Ziegler C, Senker J, Lotsch BV. Crystalline carbon nitride nanosheets for improved visible-light hydrogen evolution. J. Am. Chem. Soc. 2014;136(5):1730–1733. doi: 10.1021/ja411321s. [DOI] [PubMed] [Google Scholar]
- 92.Yin Y, Han J, Zhang X, Zhang Y, Zhou J, et al. Facile synthesis of few-layer-thick carbon nitride nanosheets by liquid ammonia-assisted lithiation method and their photocatalytic redox properties. RSC Adv. 2014;4(62):32690–32697. doi: 10.1039/C4RA06036A. [DOI] [Google Scholar]
- 93.Fukasawa Y, Takanabe K, Shimojima A, Antonietti M, Domen K, Okubo T. Synthesis of ordered porous graphitic-C3N4 and regularly arranged Ta3N5 nanoparticles by using self-assembled silica nanospheres as a primary template. Chem. Asian J. 2011;6(1):103–109. doi: 10.1002/asia.201000523. [DOI] [PubMed] [Google Scholar]
- 94.Jun Y-S, Hong WH, Antonietti M, Thomas A. Mesoporous, 2D hexagonal carbon nitride and titanium nitride/carbon composites. Adv. Mater. 2009;21(42):4270–4274. doi: 10.1002/adma.200803500. [DOI] [Google Scholar]
- 95.Park SS, Chu S-W, Xue C, Zhao D, Ha C-S. Facile synthesis of mesoporous carbon nitrides using the incipient wetness method and the application as hydrogen adsorbent. J. Mater. Chem. 2011;21(29):10801–10807. doi: 10.1039/C1JM10849B. [DOI] [Google Scholar]
- 96.Li X-H, Zhang J, Chen X, Fischer A, Thomas A, Antonietti M, Wang X. Condensed graphitic carbon nitride nanorods by nanoconfinement: promotion of crystallinity on photocatalytic conversion. Chem. Mater. 2011;23(19):4344–4348. doi: 10.1021/cm201688v. [DOI] [Google Scholar]
- 97.Liu J, Liu Y, Liu N, Han Y, Zhang X, et al. Metal-free efficient photocatalyst for stable visible water splitting via a two-electron pathway. Science. 2015;347(6225):970–974. doi: 10.1126/science.aaa3145. [DOI] [PubMed] [Google Scholar]
- 98.Kim S, Cha W, Ramadass K, Singh G, Kim IY, Vinu A. Single-step synthesis of mesoporous carbon nitride/molybdenum sulfide nanohybrids for high-performance sodium-ion batteries. Chem. Asian J. 2020;15(12):1863–1868. doi: 10.1002/asia.202000349. [DOI] [PubMed] [Google Scholar]
- 99.Paraknowitsch JP, Zhang J, Su D, Thomas A, Antonietti M. Ionic liquids as precursors for nitrogen-doped graphitic carbon. Adv. Mater. 2010;22(1):87–92. doi: 10.1002/adma.200900965. [DOI] [PubMed] [Google Scholar]
- 100.Lin Z, Wang X. Ionic liquid promoted synthesis of conjugated carbon nitride photocatalysts from urea. Chemsuschem. 2014;7(6):1547–1550. doi: 10.1002/cssc.201400016. [DOI] [PubMed] [Google Scholar]
- 101.Wang Y, Wang X, Antonietti M, Zhang Y. Facile one-pot synthesis of nanoporous carbon nitride solids by using soft templates. Chemsuschem. 2010;3(4):435–439. doi: 10.1002/cssc.200900284. [DOI] [PubMed] [Google Scholar]
- 102.Yan H. Soft-templating synthesis of mesoporous graphitic carbon nitride with enhanced photocatalytic H2 evolution under visible light. Chem. Commun. 2012;48(28):3430–3432. doi: 10.1039/C2CC00001F. [DOI] [PubMed] [Google Scholar]
- 103.Wang Y, Zhang J, Wang X, Antonietti M, Li H. Boron- and fluorine-containing mesoporous carbon nitride polymers: metal-free catalysts for cyclohexane oxidation. Angew. Chem. Int. Ed. 2010;49(19):3356–3359. doi: 10.1002/anie.201000120. [DOI] [PubMed] [Google Scholar]
- 104.Yang Z, Zhang Y, Schnepp Z. Soft and hard templating of graphitic carbon nitride. J. Mater. Chem. A. 2015;3(27):14081–14092. doi: 10.1039/C5TA02156A. [DOI] [Google Scholar]
- 105.Kesavan T, Partheeban T, Vivekanantha M, Prabu N, Kundu M, et al. Design of P-doped mesoporous carbon nitrides as high-performance anode materials for Li-ion battery. ACS Appl. Mater. Interfaces. 2020;12(21):24007–24018. doi: 10.1021/acsami.0c05123. [DOI] [PubMed] [Google Scholar]
- 106.Dou Y, Zhang L, Xu X, Sun Z, Liao T, Dou SX. Atomically thin non-layered nanomaterials for energy storage and conversion. Chem. Soc. Rev. 2017;46(23):7338–7373. doi: 10.1039/C7CS00418D. [DOI] [PubMed] [Google Scholar]
- 107.Wang Z, Gao H, Zhang Q, Liu Y, Chen J, Guo Z. Recent advances in 3D graphene architectures and their composites for energy storage applications. Small. 2019;15(3):1803858. doi: 10.1002/smll.201803858. [DOI] [PubMed] [Google Scholar]
- 108.Adekoya D, Chen H, Hoh HY, Gould T, Balogun M-SJT, Lai C, Zhao H, Zhang S. Hierarchical Co3O4@ N-doped carbon composite as an advanced anode material for ultrastable potassium storage. ACS Nano. 2020;14(4):5027–5035. doi: 10.1021/acsnano.0c01395. [DOI] [PubMed] [Google Scholar]
- 109.Ong W-J, Tan L-L, Chai S-P, Yong S-T, Mohamed AR. Surface charge modification via protonation of graphitic carbon nitride (g-C3N4) for electrostatic self-assembly construction of 2D/2D reduced graphene oxide (rGO)/g-C3N4 nanostructures toward enhanced photocatalytic reduction of carbon dioxide to methane. Nano Energy. 2015;13:757–770. doi: 10.1016/j.nanoen.2015.03.014. [DOI] [Google Scholar]
- 110.Fu Y, Zhu J, Hu C, Wu X, Wang X. Covalently coupled hybrid of graphitic carbon nitride with reduced graphene oxide as a superior performance lithium-ion battery anode. Nanoscale. 2014;6(21):12555–12564. doi: 10.1039/C4NR03145H. [DOI] [PubMed] [Google Scholar]
- 111.Li X, Feng Y, Li M, Li W, Wei H, Song D. Smart hybrids of Zn2GeO4 nanoparticles and ultrathin g-C3N4 layers: synergistic lithium storage and excellent electrochemical performance. Adv. Funct. Mater. 2015;25(44):6858–6866. doi: 10.1002/adfm.201502938. [DOI] [Google Scholar]
- 112.Wang J, Meng Z, Yang W, Yan X, Guo R, Han W-Q. Facile synthesis of rgo/g-C3N4/CNT microspheres via ethanol-assisted spray drying method for high performance lithium-sulfur batteries. ACS Appl. Mater. Interfaces. 2018;11(1):819–827. doi: 10.1021/acsami.8b17590. [DOI] [PubMed] [Google Scholar]
- 113.Zhang J, Li J-Y, Wang W-P, Zhang X-H, Tan X-H, Chu W-G, Guo Y-G. Microemulsion assisted assembly of 3D porous S/graphene@g-C3N4 hybrid sponge as free-standing cathodes for high energy density Li–S batteries. Adv. Energy Mater. 2018;8(14):1702839. doi: 10.1002/aenm.201702839. [DOI] [Google Scholar]
- 114.Wang C, Zhang L, Al-Mamun M, Dou Y, Liu P, et al. A hollow-shell structured V2O5 electrode-based symmetric full Li-ion battery with highest capacity. Adv. Energy Mater. 2019;9(31):1900909. doi: 10.1002/aenm.201900909. [DOI] [Google Scholar]
- 115.Balogun M-S, Qiu W, Lyu F, Luo Y, Meng H, et al. All-flexible lithium ion battery based on thermally-etched porous carbon cloth anode and cathode. Nano Energy. 2016;26:446–455. doi: 10.1016/j.nanoen.2016.05.017. [DOI] [Google Scholar]
- 116.Mohamed HSH, Wu L, Li C-F, Hu Z-Y, Liu J, et al. In-situ growing mesoporous Cuo/O-doped g-C3N4 nanospheres for highly enhanced lithium storage. ACS Appl. Mater. Interfaces. 2019;11(36):32957–32968. doi: 10.1021/acsami.9b10171. [DOI] [PubMed] [Google Scholar]
- 117.Yin L, Cheng R, Song Q, Yang J, Kong X, et al. Construction of nanoflower SnS2 anchored on g-C3N4 nanosheets composite as highly efficient anode for lithium ion batteries. Electrochim. Acta. 2018;293:408–418. doi: 10.1016/j.electacta.2018.10.020. [DOI] [Google Scholar]
- 118.Wang G, Wen Z, Yang Y-E, Yin J, Kong W, et al. Ultra-long life Si@rGO/g-C3N4 with a multiply synergetic effect as an anode material for lithium-ion batteries. J. Mater. Chem. A. 2018;6(17):7557–7565. doi: 10.1039/C8TA00539G. [DOI] [Google Scholar]
- 119.Kong W, Yu J, Shi X, Yin J, Yang H, Wen Z. Encapsulated red phosphorus in rgo-C3N4 architecture as extending-life anode materials for lithium-ion batteries. J. Electrochem. Soc. 2020;167(6):060518. doi: 10.1149/1945-7111/ab8406. [DOI] [Google Scholar]
- 120.Wang Y, Han J, Gu X, Dimitrijev S, Hou Y, Zhang S. Ultrathin Fe2O3 nanoflakes using smart chemical stripping for high performance lithium storage. J. Mater. Chem. A. 2017;5(35):18737–18743. doi: 10.1039/C7TA05798A. [DOI] [Google Scholar]
- 121.Hou Y, Li J, Wen Z, Cui S, Yuan C, Chen J. N-doped graphene/porous gC3N4 nanosheets supported layered-MoS2 hybrid as robust anode materials for lithium-ion batteries. Nano Energy. 2014;8:157–164. doi: 10.1016/j.nanoen.2014.06.003. [DOI] [Google Scholar]
- 122.Shah MSAS, Park AR, Rauf A, Hong SH, Choi Y, et al. Highly interdigitated and porous architected ternary composite of SnS2, gC3N4, and reduced graphene oxide (RGO) as high performance lithium ion battery anodes. RSC Adv. 2017;7(6):3125–3135. doi: 10.1039/C6RA25886G. [DOI] [Google Scholar]
- 123.Shi X, Zhou Z, Yin J, Li S, Ji S, Sun J, Wen Z. Fabrication of rGO/g-C3N4@SnS2 and its rate-performance enhancement. Chem. Phys. Lett. 2020;746:137296. doi: 10.1016/j.cplett.2020.137296. [DOI] [Google Scholar]
- 124.Shi M, Wu T, Song X, Liu J, Zhao L, Zhang P, Gao L. Active Fe2O3 nanoparticles encapsulated in porous gC3N4/graphene sandwich-type nanosheets as a superior anode for high-performance lithium-ion batteries. J. Mater. Chem. A. 2016;4(27):10666–10672. doi: 10.1039/C6TA03533G. [DOI] [Google Scholar]
- 125.Hwang JY, Myung ST, Sun YK. Recent progress in rechargeable potassium batteries. Adv. Funct. Mater. 2018;28(43):1802938. doi: 10.1002/adfm.201802938. [DOI] [Google Scholar]
- 126.Song K, Liu C, Mi L, Chou S, Chen W, Shen C. Recent progress on the alloy-based anode for sodium-ion batteries and potassium-ion batteries. Small. 2019;1:1903194. doi: 10.1002/smll.201903194. [DOI] [PubMed] [Google Scholar]
- 127.Ali Z, Zhang T, Asif M, Zhao L, Yu Y, Hou Y. Transition metal chalcogenide anodes for sodium storage. Mater. Today. 2020;3(35):131–167. doi: 10.1016/j.mattod.2019.11.008. [DOI] [Google Scholar]
- 128.Liu J, Zhang Y, Zhang L, Xie F, Vasileff A, Qiao S-Z. Graphitic carbon nitride (g-C3N4)-derived n-rich graphene with tuneable interlayer distance as a high-rate anode for sodium-ion batteries. Adv. Mater. 2019;31(24):1901261. doi: 10.1002/adma.201901261. [DOI] [PubMed] [Google Scholar]
- 129.Chen L, Yan R, Oschatz M, Jiang L, Antonietti M, Xiao K. Ultrathin 2D graphitic carbon nitride on metal films: underpotential sodium deposition in adlayers for sodium-ion batteries. Angew. Chem. Int. Ed. 2020;59(23):9067. doi: 10.1002/anie.202000314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Subramaniyam CM, Deshmukh KA, Tai Z, Mahmood N, Deshmukh AD, et al. 2D layered graphitic carbon nitride sandwiched with reduced graphene oxide as nanoarchitectured anode for highly stable lithium-ion battery. Electrochim. Acta. 2017;237(20):69–77. doi: 10.1016/j.electacta.2017.03.194. [DOI] [Google Scholar]
- 131.Senthil C, Kesavan T, Bhaumik A, Sasidharan M. N-rich graphitic carbon nitride functionalized graphene oxide nanosheet hybrid as anode for high performance lithium-ion batteries. Mater. Res. Express. 2018;5(1):016307. doi: 10.1088/2053-1591/aaa6b9. [DOI] [Google Scholar]
- 132.Vo V, Nguyen Thi XD, Jin Y-S, Ly Thi G, Nguyen TT, Duong TQ, Kim S-J. SnO2 nanosheets/g-C3N4 composite with improved lithium storage capabilities. Chem. Phys. Lett. 2017;674:42–47. doi: 10.1016/j.cplett.2017.02.057. [DOI] [Google Scholar]
- 133.Liu K, Man J, Cui J, Zhang H, Li T, Yang J, Wen Z, Sun J. Li4Ti5O12/g-C3N4 composite with an improved lithium storage capability. Mater. Lett. 2019;234:117–120. doi: 10.1016/j.matlet.2018.09.083. [DOI] [Google Scholar]
- 134.Wang Y, Huang X, Zhang S, Hou Y. Sulfur hosts against the shuttle effect. Small Methods. 2018;2(6):1700345. doi: 10.1002/smtd.201700345. [DOI] [Google Scholar]
- 135.Hencz L, Chen H, Ling HY, Wang Y, Lai C, Zhao H, Zhang S. Housing sulfur in polymer composite frameworks for Li–S batteries. Nano-Micro Lett. 2019;11(1):17. doi: 10.1007/s40820-019-0249-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Liu J, Li W, Duan L, Li X, Ji L, et al. A graphene-like oxygenated carbon nitride material for improved cycle-life lithium/sulfur batteries. Nano Lett. 2015;15(8):5137–5142. doi: 10.1021/acs.nanolett.5b01919. [DOI] [PubMed] [Google Scholar]
- 137.Qu L, Liu P, Yi Y, Wang T, Yang P, et al. Enhanced cycling performance for lithium–sulfur batteries by a laminated 2D g-C3N4/graphene cathode interlayer. Chemsuschem. 2019;12(1):213–223. doi: 10.1002/cssc.201802449. [DOI] [PubMed] [Google Scholar]
- 138.Song J, Feng S, Zhu C, Lee J-I, Fu S, Dong P, Song M-K, Lin Y. Tuning the structure and composition of graphite-phase polymeric carbon nitride/reduced graphene oxide composites towards enhanced lithium-sulfur batteries performance. Electrochim. Acta. 2017;248:541–546. doi: 10.1016/j.electacta.2017.07.149. [DOI] [Google Scholar]
- 139.Liu Y, Li N, Wu S, Liao K, Zhu K, Yi J, Zhou H. Reducing the charging voltage of a Li–O2 battery to 1.9 V by incorporating a photocatalyst. Energy Environ. Sci. 2015;8(9):2664–2667. doi: 10.1039/C5EE01958C. [DOI] [Google Scholar]
- 140.Zhao Z, Sun Y, Dong F. Graphitic carbon nitride based nanocomposites: a review. Nanoscale. 2015;7(1):15–37. doi: 10.1039/c4nr03008g. [DOI] [PubMed] [Google Scholar]
- 141.Pomerantseva E, Gogotsi Y. Two-dimensional heterostructures for energy storage. Nat. Energy. 2017;2(7):17089. doi: 10.1038/nenergy.2017.89. [DOI] [Google Scholar]
- 142.Gao R, Zhou Y, Liu X, Wang J. N-doped defective carbon layer encapsulated W2C as a multifunctional cathode catalyst for high performance Li-O2 battery. Electrochim. Acta. 2017;245:430–437. doi: 10.1016/j.electacta.2017.05.177. [DOI] [Google Scholar]
- 143.Luo WB, Chou SL, Wang JZ, Zhai YC, Liu HK. A metal-free, free-standing, macroporous graphene@ g-C3N4 composite air electrode for high-energy lithium oxygen batteries. Small. 2015;11(23):2817–2824. doi: 10.1002/smll.201403535. [DOI] [PubMed] [Google Scholar]
- 144.Lou P, Cui Z, Guo X. Achieving highly stable Li–O2 battery operation by designing a carbon nitride-based cathode towards a stable reaction interface. J. Mater. Chem. A. 2017;5(34):18207–18213. doi: 10.1039/C7TA05009G. [DOI] [Google Scholar]
- 145.Guo Q, Zhang C, Zhang C, Xin S, Zhang P, et al. Co3O4 modified Ag/g-C3N4 composite as a bifunctional cathode for lithium-oxygen battery. J. Energy Chem. 2020;41:185–193. doi: 10.1016/j.jechem.2019.05.018. [DOI] [Google Scholar]
- 146.Cha H, Kim J, Lee Y, Cho J, Park M. Issues and challenges facing flexible lithium-ion batteries for practical application. Small. 2018;14(43):1702989. doi: 10.1002/smll.201702989. [DOI] [PubMed] [Google Scholar]
- 147.Xiong T, Su H, Yang F, Tan Q, Appadurai PBS, et al. Harmonizing self-supportive VN/MoS2 pseudocapacitance core-shell electrodes for boosting the areal capacity of lithium storage. Mater. Today Energy. 2020;17:100461. doi: 10.1016/j.mtener.2020.100461. [DOI] [Google Scholar]
- 148.Yue X-Y, Li X-L, Wang W-W, Chen D, Qiu Q-Q, et al. Wettable carbon felt framework for high loading Li-metal composite anode. Nano Energy. 2019;60:257–266. doi: 10.1016/j.nanoen.2019.03.057. [DOI] [Google Scholar]
- 149.Shen K, Wang Z, Bi X, Ying Y, Zhang D, et al. Magnetic field–suppressed lithium dendrite growth for stable lithium-metal batteries. Adv. Energy Mater. 2019;9(20):1900260. doi: 10.1002/aenm.201900260. [DOI] [Google Scholar]
- 150.Yue X-Y, Wang W-W, Wang Q-C, Meng J-K, Wang X-X, et al. Cuprite-coated cu foam skeleton host enabling lateral growth of lithium dendrites for advanced Li metal batteries. Energy Storage Mater. 2019;21:180–189. doi: 10.1016/j.ensm.2018.12.007. [DOI] [Google Scholar]
- 151.Luan X, Wang C, Wang C, Gu X, Yang J, Qian Y. Stable lithium deposition enabled by an acid-treated g-C3N4 interface layer for a lithium metal anode. ACS Appl. Mater. Interfaces. 2020;12(9):11265–11272. doi: 10.1021/acsami.9b23520. [DOI] [PubMed] [Google Scholar]
- 152.Hu J, Tian J, Li C. Nanostructured carbon nitride polymer-reinforced electrolyte to enable dendrite-suppressed lithium metal batteries. ACS Appl. Mater. Interfaces. 2017;9(13):11615–11625. doi: 10.1021/acsami.7b00478. [DOI] [PubMed] [Google Scholar]
- 153.Lu Z, Liang Q, Wang B, Tao Y, Zhao Y, et al. Graphitic carbon nitride induced micro-electric field for dendrite-free lithium metal anodes. Adv. Energy Mater. 2019;9(7):1803186. doi: 10.1002/aenm.201803186. [DOI] [Google Scholar]
- 154.Jiao D, Ma Z, Li J, Han Y, Mao J, Ling T, Qiao S. Test factors affecting the performance of zinc–air battery. J. Energy Chem. 2020;44:1–7. doi: 10.1016/j.jechem.2019.09.008. [DOI] [Google Scholar]
- 155.Ran F, Chen S. Advanced Nanomaterials for Electrochemical-based Energy Conversion and Storage. Amsterdam: Elsevier; 2019. p. 416. [Google Scholar]
- 156.Li Y-J, Cui L, Da P-F, Qiu K-W, Qin W-J, et al. Multiscale structural engineering of ni-doped coo nanosheets for zinc–air batteries with high power density. Adv. Mater. 2018;30(46):1804653. doi: 10.1002/adma.201804653. [DOI] [PubMed] [Google Scholar]
- 157.Lu H-S, Zhang H, Zhang X, Sun N, Zhu X, Zhao H, Wang G. Transformation of carbon-encapsulated metallic Co into ultrafine Co/CoO nanoparticles exposed on N-doped graphitic carbon for high-performance rechargeable zinc-air battery. Appl. Surf. Sci. 2018;448:369–379. doi: 10.1016/j.apsusc.2018.04.146. [DOI] [Google Scholar]
- 158.Shinde SS, Lee C-H, Sami A, Kim D-H, Lee S-U, Lee J-H. Scalable 3D carbon nitride sponge as an efficient metal-free bifunctional oxygen electrocatalyst for rechargeable Zn–air batteries. ACS Nano. 2017;11(1):347–357. doi: 10.1021/acsnano.6b05914. [DOI] [PubMed] [Google Scholar]
- 159.Shinde SS, Yu J-Y, Song J-W, Nam Y-H, Kim D-H, Lee J-H. Highly active and durable carbon nitride fibers as metal-free bifunctional oxygen electrodes for flexible Zn–air batteries. Nanoscale Horiz. 2017;2(6):333–341. doi: 10.1039/C7NH00058H. [DOI] [PubMed] [Google Scholar]
- 160.Su C-Y, Cheng H, Li W, Liu Z-Q, Li N, et al. Atomic modulation of FeCo–nitrogen–carbon bifunctional oxygen electrodes for rechargeable and flexible all-solid-state zinc–air battery. Adv. Energy Mater. 2017;7(13):1602420. doi: 10.1002/aenm.201602420. [DOI] [Google Scholar]
- 161.Zhang L, Xiong J, Qin Y-H, Wang C-W. Porous N-C catalyst synthesized by pyrolyzing g-C3N4 embedded in carbon as highly efficient oxygen reduction electrocatalysts for primary Zn-air battery. Carbon. 2019;150:475–484. doi: 10.1016/j.carbon.2019.05.044. [DOI] [Google Scholar]
- 162.Wang S, Xiong P, Zhang J, Wang G. Recent progress on flexible lithium metal batteries: composite lithium metal anodes and solid-state electrolytes. Energy Storage Mater. 2020;29:310–331. doi: 10.1016/j.ensm.2020.04.032. [DOI] [Google Scholar]
- 163.Zhu Y, He X, Mo Y. Origin of outstanding stability in the lithium solid electrolyte materials: insights from thermodynamic analyses based on first-principles calculations. ACS Appl. Mater. Interfaces. 2015;7(42):23685–23693. doi: 10.1021/acsami.5b07517. [DOI] [PubMed] [Google Scholar]
- 164.Zhu Y, He X, Mo Y. First principles study on electrochemical and chemical stability of solid electrolyte–electrode interfaces in all-solid-state Li-ion batteries. J. Mater. Chem. A. 2016;4(9):3253–3266. doi: 10.1039/C5TA08574H. [DOI] [Google Scholar]
- 165.Huang Y, Chen B, Duan J, Yang F, Wang T, et al. Graphitic carbon nitride (g-C3N4): an interface enabler for solid-state lithium metal batteries. Angew. Chem. Int. Ed. 2020;59(9):3699–3704. doi: 10.1002/anie.201914417. [DOI] [PubMed] [Google Scholar]
- 166.Lu Y, Wen Z, Jin J, Cui Y, Wu M, Sun S. Mesoporous carbon nitride loaded with Pt nanoparticles as a bifunctional air electrode for rechargeable lithium-air battery. J. Solid State Electrochem. 2012;16(5):1863–1868. doi: 10.1007/s10008-012-1640-8. [DOI] [Google Scholar]
- 167.Sun Z, Li Y, Zhang S, Shi L, Wu H, Bu H, Ding S. G-C3N4 nanosheets enhanced solid polymer electrolytes with excellent electrochemical performance, mechanical properties, and thermal stability. J. Mater. Chem. A. 2019;7(18):11069–11076. doi: 10.1039/C9TA00634F. [DOI] [Google Scholar]
- 168.Liu T, Zhang Y, Chen C, Lin Z, Zhang S, Lu J. Sustainability-inspired cell design for a fully recyclable sodium ion battery. Nat. Commun. 2019;10(1):1965. doi: 10.1038/s41467-019-09933-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Su Z, Ling HY, Li M, Qian S, Chen H, Lai C, Zhang S. Honeycomb-like carbon materials derived from coffee extract via a “salty” thermal treatment for high-performance Li-I2 batteries. Carbon Energy. 2020;2(2):265–275. doi: 10.1002/cey2.40. [DOI] [Google Scholar]
- 170.Sha M, Liu L, Zhao H, Lei Y. Anode materials for potassium-ion batteries: current status and prospects. Carbon Energy. 2020 doi: 10.1002/cey2.57. [DOI] [Google Scholar]
- 171.Shi Y, Liu G, Jin R, Xu H, Wang Q, Gao S. Carbon materials from melamine sponges for supercapacitors and lithium battery electrode materials: a review. Carbon Energy. 2019;1(2):253–275. doi: 10.1002/cey2.19. [DOI] [Google Scholar]
- 172.Chen J, Park N-G. Materials and methods for interface engineering towards stable and efficient perovskite solar cells. ACS Energy Lett. 2020;5:180–185. doi: 10.1021/acsenergylett.0c01240. [DOI] [Google Scholar]
- 173.J.-M. Fan, J.-J. Chen, Q. Zhang, B.-B. Chen, J. Zang, M.-S. Zheng, Q.-F. Dong. An amorphous carbon nitride composite derived from ZIF-8 as anode material for sodium-ion batteries. ChemSusChem. 8(11), 1856–1861 (2015). 10.1002/cssc.201500192 [DOI] [PubMed]
- 174.Chen L, Yan R, Oschatz M, Jiang L, Antonietti M, Xiao K. Ultrathin 2D graphitic carbon nitride on metal films: Underpotential sodium deposition in adlayers for sodium-ion batteries. Angew. Chem. Int. 2020;59(23):9067. doi: 10.1002/anie.202000314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Liu J, Zhang Y, Zhang L, Xie F, Vasileff A, Qiao S-Z. Graphitic carbon nitride (g-C3N4)-derived N-rich graphene with tuneable interlayer distance as a high-rate anode for sodium-ion batteries. Adv Mater. 2019;31(24):1901261. doi: 10.1002/adma.201901261. [DOI] [PubMed] [Google Scholar]
- 176.Adekoya D, Chen H, Hoh HY, Gould T, Balogun M-SJT, Lai C, Zhao H, Zhang S. Hierarchical Co3o4@N-doped carbon composite as an advanced anode material for ultrastable potassium storage. ACS Nano. 2020;14(4):5027–5035. doi: 10.1021/acsnano.0c01395. [DOI] [PubMed] [Google Scholar]
- 177.Liu J, Li W, Duan L, Li X, Ji L, Geng Z, Huang K, Lu L, Zhou L, Liu Z. A graphene-like oxygenated carbon nitride material for improved cyclelife lithium/sulfur batteries. Nano Lett. 2015;15(8):5137–5142. doi: 10.1021/acs.nanolett.5b01919. [DOI] [PubMed] [Google Scholar]
- 178.Wu X, Li S, Wang B, Liu J, Yu M. Free-standing 3D network-like cathode based on biomass-derived N-doped carbon/graphene/g-C3N4 hybrid ultrathin sheets as sulfur host for high-rate Li-S battery. Renew. Energy. 2020;158:509–519. doi: 10.1016/j.renene.2020.05.098. [DOI] [Google Scholar]
- 179.Zhang H, Zhao Z, Hou Y-N, Tang Y, Dong Y, Wang S, Hu X, Zhang Z, Wang X, Qiu J. Nanopore-confined g-C3N4 nanodots in N, S Codoped hollow porous carbon with boosted capacity for lithium–sulfur batteries. J. Mater. Chem. A. 2018;6(16):7133–7141. doi: 10.1039/C8TA00529J. [DOI] [Google Scholar]
- 180.Wang X, Li G, Li M, Liu R, Li H, Li T, Sun M, Deng Y, Feng M, Chen Z. Reinforced polysulfide barrier by g-C3N4/CNT composite towards superior lithium-sulfur batteries. J. Energy Chem. 2021;53:234–240. doi: 10.1016/j.jechem.2020.05.036. [DOI] [Google Scholar]
- 181.Gong Y, Fu C, Zhang G, Zhou H, Kuang Y. Three-dimensional porous C3N4 nanosheets@reduced graphene oxide network as sulfur hosts for high performance lithium-sulfur batteries. Electrochim. Acta. 2017;256:1–9. doi: 10.1016/j.electacta.2017.10.032. [DOI] [Google Scholar]
- 182.Wang J, Meng Z, Yang W, Yan X, Guo R, Han W-Q. Facile synthesis of rGO/g-C3N4/CNT microspheres via ethanol-assisted spray drying method for high performance lithium-sulfur batteries. ACS Appl. Mater. Interfaces. 2018;11(1):819–827. doi: 10.1021/acsami.8b17590. [DOI] [PubMed] [Google Scholar]
- 183.Huangfu Y, Zheng T, Zhang K, She X, Xu H, Fang Z, Xie K. Facile fabrication of permselective g-C3N4 separator for improved lithium-sulfur batteries. Electrochim. Acta. 2018;272:60–67. doi: 10.1016/j.electacta.2018.03.149. [DOI] [Google Scholar]
- 184.Angamuthu G, Babu DB, Ramesha K, Rangarajan V. MoS2 anchored carbon nitride based mesoporous material as a polysulfide barrier for high capacity lithium-sulfur battery. J. Electroanal. Chem. 2019;843:37–46. doi: 10.1016/j.jelechem.2019.05.006. [DOI] [Google Scholar]