Drosophila is a useful model system to study the regulation of energy metabolism and its diseases like obesity and diabetes.
Abstract
The organs and metabolic pathways involved in energy metabolism, and the process of ATP production from nutrients, are comparable between humans and Drosophila melanogaster. This level of conservation, together with the power of Drosophila genetics, makes the fly a very useful model system to study energy homeostasis. Here, we discuss the major organs involved in energy metabolism in Drosophila and how they metabolize different dietary nutrients to generate adenosine triphosphate. Energy metabolism in these organs is controlled by cell-intrinsic, paracrine, and endocrine signals that are similar between Drosophila and mammals. We describe how these signaling pathways are regulated by several physiological and environmental cues to accommodate tissue-, age-, and environment-specific differences in energy demand. Last, we discuss several genetic and diet-induced fly models of obesity and diabetes that can be leveraged to better understand the molecular basis of these metabolic diseases and thereby promote the development of novel therapies.
ENERGY METABOLISM IN DROSOPHILA
Energy metabolism is the process of using nutrients to produce adenosine triphosphate (ATP), the energy currency of cells. The high-energy bond in ATP is hydrolyzed to generate adenosine diphosphate (ADP) and inorganic phosphate and to release energy, which is used in cellular processes such as active transmembrane transport of solutes, DNA and RNA synthesis, signal transduction, and assembly of cytoskeletal elements, as well as for voluntary skeletal muscle contraction (1).
The machinery
There are many similarities among organs involved in energy metabolism in Drosophila and mammals. In both, for example, nutrients are absorbed by the intestine and skeletal muscles are responsible for voluntary energy expenditure. The Drosophila fat body (FB) plays a dual role: It stores excess energy in the form of triglycerides, similar to what is seen in the white adipose tissue in mammals, and, similar to the mammalian liver, is involved in metabolism, glycogen storage, and nutrient sensing (2, 3). Moreover, analogous to the insulin/glucagon endocrine system in mammals, Drosophila insulin-like peptides (dILPs) and glucagon-like adipokinetic hormone (AKH) play central roles in glucose and lipid metabolism in Drosophila (4, 5). However, unlike the mammalian system where the blood flowing through the cardiovascular system delivers both the nutrients and the oxygen for oxidative metabolism to different tissues, nutrients and oxygen are delivered separately in Drosophila. Different organs in Drosophila, including muscles and FB, derive nutrients from an open circulatory system through which hemolymph is circulated by a linear heart. A separate open tubular network called the trachea directly provides the oxygen necessary for metabolism to different organs (6).
Role of FB and oenocytes in energy metabolism
The FB is the principal site of metabolism in Drosophila. In rapidly growing larvae, the FB stores nutrients in large quantities in the form of glycogen and triglycerides and synchronizes systemic growth through insulin and TOR signaling (4, 6). Carbohydrates in the food are enzymatically broken down to monosaccharides and absorbed in the intestine and then released into the hemolymph. They are then taken up by the FB, where they are either converted and stored as the multibranched polysaccharide glycogen or condensed into the nonreducing disaccharide trehalose, which is released back into the hemolymph to be metabolized in other tissues (6). Lipids in the food are digested and absorbed in the intestine and then transported in the hemolymph as lipoprotein particles. These are taken up by the FB using lipophorin receptors and are stored as triglycerides (aka triacylglycerol or TAG) in lipid droplets, mostly in the FB and in the gut in smaller quantities (7, 8). TAGs in the FB are broken down by lipases and metabolized in response to starvation (4). In addition, the FB coordinates nutrient availability with energy storage and expenditure and thus plays a central role in energy metabolism (3, 9). For instance, Unpaired 2 (Upd2), a cytokine secreted from the FB in response to feeding, regulates dILP secretion and thereby alters the systemic metabolic state (10). Likewise, a high-sugar diet (HSD) induces secretion of Neural Lazarillo (NLaz) from the FB and leads to insulin resistance (11).
In addition to the FB, oenocytes, a group of secretory cells located dorsally in the adult fly abdomen, uptake mobilized lipids during starvation and thereby function similarly to mammalian hepatocytes. Genetic ablation studies have shown that oenocytes are required for larval growth and pupal development as well as for starvation resistance in adults (12, 13). Chatterjee et al. (13) showed that dILP6 secreted from the FB promotes lipid uptake in oenocytes in response to starvation and thereby coordinates lipid turnover and the starvation response in these two different tissues. The similarities between oenocytes and the mammalian liver are further highlighted by the results of a recent RiboTag translatomic profiling study by Huang et al. (14). They found that genes regulating pathways such as long-chain fatty acid metabolism, peroxisomal β-oxidation, ketogenesis, reactive oxygen species (ROS) metabolism, and xenobiotic metabolism are enriched in the oenocytes, as is true for the mammalian liver. There was little overlap between oenocyte-enriched and FB-enriched genes, indicating that the two tissues have distinct functions in Drosophila metabolism.
Role of skeletal muscles in energy metabolism
Like mammals, Drosophila has both glycolytic and oxidative muscles (in Drosophila, these are the leg and flight muscles, respectively) (5). In insects, flight muscles constitute a large proportion of body weight and are of two types: direct flight muscles (DFMs) and indirect flight muscles (IFMs). Insect IFMs are one of the most energy-consuming muscles found in animals and must maintain a high rate of oxidative metabolism to meet this energy demand (15). Thus, the high levels of trehalose normally found in the hemolymph are essential to meet the energy demands of flight muscles (16). In addition, Drosophila also stores glycogen in the skeletal muscles as an energy reserve (2). Intriguingly, some glycolytic enzymes have been reported to colocalize with the Z line in the sarcomere of Drosophila flight muscles, indicating that high rates of glycolysis are localized in these areas (17, 18). The importance of skeletal muscle on metabolic homeostasis and associated life-span regulation in Drosophila has been underscored in a study by Demontis and Perrimon. They showed that the age-related decline of skeletal muscle function can be prevented by overexpression of the transcription factor dFOXO in muscles. The Drosophila forkhead box O (dFOXO)/eukaryotic translation initiation factor 4E (eIF4E)-binding protein (4E-BP) signaling maintains proteostasis in the aging muscle as well as in distal organs, decreases feeding, and reduces insulin secretion, thereby altering systemic metabolism and extending life span (19).
In addition to energy expenditure, cytokines and growth factors secreted from the skeletal muscles (myokines) also regulate the metabolic state in Drosophila. Zhao and Karpac recently showed that dFOXO activity in Drosophila muscle regulates the expression of the cytokine Unpaired 2 (Upd2), which, in turn, modulates the secretion of the glucagon-like peptide AKH. AKH is an important regulator of lipid metabolism and storage in the FB and the intestine. Upd2 myokine-mediated cross-talk between an energy-expending tissue, i.e., the skeletal muscle, and energy-storing tissues, i.e., the FB and the gut, plays an important role in diurnal lipid homeostasis in Drosophila (20).
Role of the digestive tract in energy metabolism
Like mammals, the digestive tract is the principal site of digestion and absorption of nutrients in Drosophila. Notably, the genes involved in carbohydrate digestion are enriched in the anterior adult midgut, whereas those for protein digestion are enriched in the posterior parts (21). The digestion and absorption of different nutrients have been reviewed previously (21). The structure and the function of the digestive tract are closely coordinated with the energy status of the organism. On one hand, starvation induces the expression of carbohydrate digestive enzymes such as amylases (13, 22). On the other hand, nutritious sugar feeding stimulates the secretion of the transforming growth factor–β (TGFβ) ligand Dawdle (Daw) from the adult FB that, in turn, represses multiple carbohydrases and lipases (23). Similarly, low dietary cholesterol induces the expression of the nuclear receptor DHR96, which, in turn, induces the expression of cholesterol and lipid metabolism genes including the gastric lipase magro. Consequently, mutation of DHR96 prevents gastric lipid uptake and leads to depletion of lipid stores in the FB (24, 25). In addition, dietary sugar can also suppress magro expression (26). Intestinal stem cells (ISCs), which give rise to enterocytes (ECs) and enteroendocrine cells (EEs), are critical for the structural and functional integrity of the Drosophila digestive tract. Prolonged nutrient deprivation reversibly reduces ISC proliferation. Conversely, insulin signaling promotes ISC proliferation (21). This is important for gut growth in response to refeeding after extended starvation.
In addition to responding to distant signals that modulate the functions of the Drosophila digestive tract, signals emanating from it also influence metabolic processes in different organs. Tachykinins (TKs), which are produced in adult midgut EEs in response to starvation, reduce lipogenesis in ECs in a protein kinase A (PKA)–dependent manner (27). Furthermore, a loss of midgut TK leads to a systemic increase in lipid levels and lipid accumulation in FB. In addition, chronic HSD leads to Activin-β (Actβ) secretion from larval midgut EEs, which, in turn, stimulates AKH signaling in FB by increasing AKH receptor (AKHR) expression, eventually leading to hyperglycemia. Long-term activation of activin signaling also promotes glucagon response and concomitant increases in glucose production in mouse primary hepatocytes, indicating a conserved role of TGFβ/Activin signaling in the maintenance of blood sugar levels (28).
Role of the nervous system in energy metabolism
The Drosophila nervous system plays a critical role in sensing the metabolic state, in coordinating metabolic processes of different organs, and in regulating energy metabolism accordingly. Similar to mammals, insulin signaling plays a central role in this regulation. However, unlike mammals, four dILPs (dILP1, dILP2, dILP3, and dILP5) are mainly secreted from insulin-producing cells (IPCs), a group of median neurosecretory cells (MNCs) in the Drosophila brain (29, 30). Like mammalian insulin-producing pancreatic β cells, IPCs in adult flies can directly sense an increase in sugar levels in the hemolymph and secrete dILPs in response (31–33). dILP secretion from IPCs, in turn, is regulated by multiple neuronal inputs. For example, short neuropeptide F (sNPF) released from sugar-sensing dorsolateral CN neurons [corazonin (Crz)+ and sNPF+ neurons] promotes dILP2 secretion from IPCs (34). On the other hand, dILP secretion from IPCs is inhibited by gamma-aminobutyric acid–releasing (GABAergic) neurons. This inhibition is relieved by Upd2 released from FB upon feeding (10). Drosophila AKH, the counterpart of mammalian glucagon that plays a pivotal role in energy homeostasis, is secreted from the corpora cardiaca (CC) neuroendocrine cells, which are located in the ring gland in larvae but attached to the esophagus after metamorphosis (35, 36). In larvae, AKH secreted from CC in response to sugar triggers dILP3 secretion from larval IPCs (37). sNPF released from sugar-sensing dorsolateral CN neurons inhibit AKH secretion from adult CCs (34). The nervous system also affects digestive tract functions. For instance, nutritive sugar-induced secretion of Diuretic hormone 44, the ortholog of the mammalian corticotropin-releasing hormone, from six neurosecretory cells in the Drosophila brain promotes intestinal ingestion and digestion (38). At the same time, signals from the gut affect dILP secretion from the brain. For example, nutrient-dependent CCHamide-2 (CCHa2) secretion from the larval midgut controls dILP secretion from IPCs (39). Conversely, Limostatin secretion from gut-associated endocrine CC cells in response to nutrient deprivation suppresses dILP secretion (40).
The fuel
Carbohydrates as an energy source
Carbohydrates such as glucose and trehalose are circulating energy sources in Drosophila hemolymph (35). Glucose is metabolized through the glycolytic pathway, tricarboxylic acid (TCA) cycle, and oxidative phosphorylation to produce ATP (41). Drosophila mitochondrial pyruvate carrier (Mpc1), which transfers pyruvate generated in glycolysis into mitochondria for pyruvate catabolism in the TCA cycle, is modestly induced by sugar feeding (42). Trehalose, a disaccharide that can be tolerated at high concentrations in the hemolymph due to its nonreducing nature, is the primary sugar in the hemolymph of Drosophila (16, 43). Trehalose is synthesized in the FB by condensation of two glucose molecules in a process catabolized by the enzyme trehalose-6-phosphate synthase (Tps1). To be used in ATP production, trehalose must be broken down into glucose, a process performed by the enzyme trehalase (44). As noted above, high circulating levels of trehalose are necessary to supply energy to flight muscles (16). Trehalose is also required in the Drosophila brain. Glycolytic metabolism of trehalose in the glial cells that form the blood-brain-barrier produces lactate and alanine, which are secreted to provide energy to neurons. Notably, mutants deficient in tps1 can survive up to the late pupal stage, suggesting that glucose can partially compensate for a lack of trehalose during development (41). Increased availability of dietary sugars, as occurs in HSD conditions, elevates the levels of both glucose and trehalose (45). There are few notable differences between Drosophila and mammalian carbohydrate energy metabolism. Unlike mammals, where glucose is the principal circulating sugar, trehalose is the most abundant sugar in Drosophila. Because of the nonreducing nature of trehalose, nonspecific glycation of proteins and associated complications that are seen in diabetic mammals with high circulating levels of glucose are absent in Drosophila. Therefore, the homeostatic regulation of circulating sugar levels might not be as necessary in Drosophila as in mammals. As a consequence, the levels of circulating sugars in insects increase with an increase in the sugar content of the food (46). The readily available energy from high trehalose concentration also allows Drosophila to meet the rapid increase in energy demand during flight. It has recently been shown that water supplementation can rescue the negative effect of HSD on Drosophila life span although it does not affect HSD-induced metabolic changes such as obesity and insulin resistance, indicating that the metabolic effects of HSD do not have a long-term effect on Drosophila survival (47).
As in many other animals, in Drosophila, excess energy from food is stored in the form of glycogen (48). HSD induces the expression of glycogen synthase (glyS) and causes developmental delay in larvae lacking GlyS (a key enzyme in glycogen synthesis) in the FB (49). In larvae, glycogen is mainly found in the body wall muscle and the FB, whereas in adults, it is stored in the FB, flight muscle, and oocytes (41). Glycogen is mobilized from the larval FB upon starvation to maintain the circulating sugar levels (50). Deficiency in glycogen metabolism causes semi-lethality in larvae and affects the fitness of adults upon starvation (51). Intriguingly, glycogen mobilization from larval FB upon fasting is prevented after the “pupal commitment” stage and it is conserved to be used during the metamorphosis (52). In addition to glycogenolysis, which breaks down glycogen into glucose, glycogen autophagy is also involved in glycogen catabolism in larval muscles in Drosophila. Either glycogenolysis or glycogen autophagy is necessary for efficient glycogen catabolism in larval muscles during starvation. Simultaneous suppression of both pathways completely impairs this starvation-induced glycogen breakdown (53). However, glycogen phosphorylase (GlyP), the rate-limiting enzyme of glycogenolysis, is essential in the Drosophila adult flight muscle, which exhibits a marked reduction in beat frequency when glyP is mutated (54).
Lipids as an energy source
Excess energy from food is largely stored in cytoplasmic lipid droplets in the form of calorie-dense and hydrophobic TAG. In addition to being stored in the FB, TAG is also stored in other Drosophila tissues such as the gut, oenocytes, ovaries, and brain (8, 12). Stored TAG is lipolyzed under conditions of nutrient deprivation and/or high energy demand (55). Brummer lipase (Bmm), an ortholog of mammalian adipocyte triglyceride lipase, is the principal lipase in Drosophila. It is expressed in multiple tissues including the FB, gut, oenocytes, and heart. Bmm hydrolyzes TAG to produce free fatty acid (FFA) and diacylglycerol (DAG), both of which can be transported in the hemolymph (56). In addition, putative lipases encoded by CG1882, CG5966, and dob are also expressed in the FB (57). Drosophila hormone-sensitive lipase (Hsl), which is an enzymatically uncharacterized protein orthologous to mammalian DAG lipase Hsl, has been proposed to further hydrolyze DAG to release FFA (56).
FFA molecules then undergo β-oxidation in the mitochondria and peroxisomes to produce ATP (55). A loss of bubblegum (bgm), which encodes an acyl–coenzyme A (CoA) synthetase (ACS) enzyme that activates long-chain FFAs for β-oxidation by coupling them with CoA, leads to the accumulation of lipids in gastric caeca in larvae (58). Similarly, mutation of another ACS gene, pudgy (pdgy), also results in excess lipid storage and impaired TAG mobilization (59). Mutations in the α or β subunits of the mitochondrial trifunctional protein, which catalyzes the last three steps in long-chain fatty acid β-oxidation, cause significant lipid accumulation as well (60).
Dietary proteins in energy metabolism
In addition to carbohydrates and lipids, proteins can also be catabolized to yield energy. However, proteins principally serve as the source for amino acids, which are the building blocks of the body. They are neither the readily available form of energy such as glucose nor the preferred form of energy storage such as fat with few exceptions like hexamerins in insects. Hexamerins, which are generally made of six polypeptide subunits, are insect storage proteins that are synthesized in the larval FB and secreted in large amounts into the hemolymph. They provide amino acids to build new tissue during development and mostly disappear by the adult stage. The comparable proteins in Drosophila are the larval storage proteins (61). In extreme energy-deficient conditions, such as prolonged starvation, proteins serve as an important source of energy. During these conditions, amino acids are converted into pyruvate, acetyl-CoA, or TCA cycle intermediates and then are further metabolized to generate ATP. The relative contribution of proteins to the energy budget of Drosophila is not well studied. However, Marron and colleagues studied the effect of starvation and desiccation on the energy metabolism in Drosophila and estimated that proteins contribute about 10% to overall metabolism (62).
Amino acids are also important modulators of metabolism. Mammalian mechanistic target of rapamycin (mTOR), which regulates growth and metabolism by phosphorylating proteins such as 4E-BP and ribosomal protein S6 kinase (S6K), monitors the nutritional status by sensing the levels of amino acids (63). Similarly, dietary amino acids are sensed by target of rapamycin (TOR)/regulatory associated protein of TOR (RAPTOR) in the larval FB in Drosophila. This, in turn, remotely regulates dILP secretion from the brain and thereby regulates systemic metabolism (64). Tiebe and colleagues showed that REPTOR and REPTOR-BP transcription factors are the principal downstream effectors of TOR signaling in Drosophila. They influence energy metabolism by regulating multiple genes including those involved in lipid biosynthesis and energy expenditure (65). Moreover, Colombani and colleagues have shown that slimfast, an amino acid transporter, regulates TOR signaling in the FB and thereby remotely regulates phosphatidyl-inositol 3-kinase (PI3K) signaling in peripheral tissues to modulate growth (9). PI3K/AKT signaling also inhibits the function of 4E-BP, which represses protein translation, by inhibiting dFOXO that promotes 4E-BP expression (66). IPCs in larval brains can directly sense the extracellular concentration of the amino acid leucine through minidiscs, a leucine transporter. This, in turn, regulates dILP2 and dILP5 secretion to regulate growth and hemolymph sugar levels (67). Thus, amino acids can serve both as a source of energy and as regulators of energy homeostasis in Drosophila. However, we will focus on carbohydrate and lipid energy metabolism in this review because they are the major sources of energy under most conditions.
REGULATION OF ENERGY METABOLISM IN DROSOPHILA
The cell-intrinsic regulation of energy metabolism in Drosophila is largely determined by the cell type–specific expression of a variety of metabolic genes. Tissue-specific gene expression, combined with environmental and physiological signals that modify gene expression and activity, generates a complex spatiotemporal pattern of metabolic regulation that meets the energy demands of the organism. The importance of tissue-specific regulation of energy metabolism is highlighted by Placais et al. (68), who recently showed that up-regulation of energy flux in neurons of the mushroom body is important for long-term memory (LTM) formation in Drosophila and is accompanied by an increase in sucrose intake during the early stages of LTM formation. This elevation of energy flux is triggered by dopamine signaling coming from a pair of afferent MP1 neurons. This signal exerts its effect through a D5-like dopamine receptor on cells in the mushroom body (DAMB), a memory center in the Drosophila brain.
Below, we discuss some of the major regulators of energy metabolism in Drosophila, which themselves are regulated by nutrient availability and the metabolic state of the organism. As a result, multiple interconnected feedback loops are formed that maintain energy homeostasis in Drosophila. Like mammals, insulin signaling responds to nutrient availability and plays a central role in energy metabolism in Drosophila. Other major regulators of Drosophila energy homeostasis, such as AKH, TGFβ signaling, and the Mondo/Mlx complex, which themselves are regulated by nutrient availability, influence the systemic metabolic state in Drosophila partially by modulating insulin signaling (Fig. 1).
Fig. 1. Sugar-induced dILP secretion in Drosophila larvae and adults.
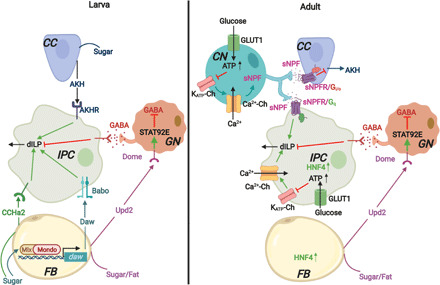
Adult Drosophila IPCs can sense sugar in the hemolymph but larval IPCs cannot. Thus, in larvae, sugar-induced AKH secretion from CC acts through AKHR and indirectly stimulates dILP3 secretion from larval IPCs. Daw, which is transcriptionally induced in the FB by sugar-activated Mondo-Mlx complex, binds the type I (Babo)/type II TGFβ receptor complex on larval IPCs, promoting dILP2 and dILP5 secretion. Likewise, CCHamide-2 (CCHa2), a peptide hormone secreted from the larval FB and midgut (not shown) in response to nutrients including sugar also promotes the expression and secretion of dILPs from IPCs. In contrast, IPCs in adult Drosophila respond to hemolymph glucose directly, through a mechanism analogous to the one described for mammalian pancreatic β cells. GLUT1-mediated glucose uptake in adult IPCs increases glucose catabolism and accompanying mitochondrial ATP production. This leads to the closure of ATP-sensitive potassium channels (KATP-Ch), membrane depolarization, the opening of voltage-dependent Ca2+ channels (Ca2+-Ch), an influx of Ca2+, and subsequent dILP secretion through exocytosis. HNF4, which is highly expressed in adult IPCs, is required for glucose-induced dILP secretion. The CN neuron, which projects to both adult IPCs and AKH-secreting CC with bifurcated axons, responds to hemolymph glucose in a similar fashion in adults. However, Ca2+ influx in these neurons triggers the release of sNPF that binds to sNPF receptors (sNPFR) on IPCs and promotes dILP2 secretion through a Gq-coupled signaling mechanism. This is accompanied by a suppression of AKH secretion from adult CC through an sNPFR/Gi/o-induced signaling cascade. Upd2 secreted from the FB in response to nutrients, including sugar, stimulates Janus kinase/signal transducers and activators of transcription (JAK/STAT) signaling in GABAergic neurons (GN) by binding to its receptor Dome and inhibits their firing in a STAT92E-dependent manner. This relieves their inhibitory effect on dILP secretion from both larval and adult IPCs.
Insulin/insulin-like growth factor signaling
Effect of IIS on carbohydrate energy metabolism
There are eight dILPs (dILP1 to dILP8) (5). Most dILPs act through an insulin-like receptor (InR) encoded by a single gene. They stimulate a well-conserved insulin receptor substrate (IRS)/PI3K/protein kinase B (PKB, also known as AKT) signaling pathway. Different from dILP1 to dILP7, dILP8 acts through Leucine-rich repeat-containing G protein–coupled receptor 3 (41). In mammals, the same IRS/PI3K/AKT pathway can be activated by insulin-like growth factor 1 (IGF-1) through its receptor IGF-1R. In contrast, InR is the only insulin receptor in Drosophila and the sole input for activation of IRS/PI3K/AKT signaling cascade (69).
In Drosophila, insulin/insulin-like growth factor signaling (IIS) is required for both energy metabolism and growth of larval tissues (29, 30, 70). dILP1, dILP2, dILP3, and dILP5 are mainly expressed from a group of MNCs in the Drosophila brain. This group of cells is also called IPCs. Genetic ablation of IPCs in larvae severely affects larval growth. It also increases glucose, trehalose, glycogen, and lipid levels that persist even in adulthood (29, 30). Similarly, larvae lacking dILP2, dILP3, and dILP5 have high hemolymph glucose concentrations, indicating that these dILPs play a major role in the regulation of glucose metabolism. This is accompanied by an elevation in trehalose, glycogen, and lipid levels (30, 70, 71). Ectopic expression of dILP2 can reverse the IPC-ablation phenotypes, highlighting the partial functional redundancy of dILPs in this context (29). However, it should be noted that different dILP deficiencies have different effects on circulating sugar levels, energy storage, and feeding preferences, indicating a divergence in dILPs function. The evolutionary history of dILPs, the phenotypes of individual dILP mutations, and the compensatory interaction among different dILPs have been studied by Gronke et al. (70). In addition, Semaniuk and colleagues studied the effect of particular dILP mutations on hemolymph sugar levels, energy storage, and feeding preferences (72). As for mammalian insulin signaling, Drosophila IIS facilitates glucose clearance by promoting glycogen synthesis and storage (50). However, a loss of systemic insulin signaling due to either IPC ablation or dILP2-3,5 triple deficiency also induces glycogen storage, indicating the presence of an additional IIS-independent regulatory mechanism that promotes glycogen synthesis (30, 48, 70).
AKT-mediated activation of TOR signaling, a master regulator of growth, plays a crucial role in the growth-promoting effects of IIS in Drosophila larvae and thereby increases utilization of nutrients in anabolic processes (41). In addition, inhibition of dFOXO by AKT-mediated phosphorylation promotes growth through its effects on 4E-BP and Myc (41, 66). Suppression of dFOXO by IIS also reduces glucose levels by preventing dFOXO-mediated activation of PEPCK, the rate-limiting enzyme of gluconeogenesis (29, 30, 73).
Effect of IIS on lipid energy metabolism
Like mammalian insulin signaling, IIS in Drosophila promotes fat storage in the adipose tissue (48). FB-specific overexpression of a constitutively active InR elevates fat storage in the FB, whereas overexpression of dominant-negative InR depletes the lipid reserves (74). However, perturbations of FB-specific and systemic insulin signaling show opposite effects on TAG levels. Mutations in both InR and the IRS protein Chico increase TAG levels (75, 76). Similarly, genetic ablation of dILP-producing IPCs, as well as triple mutation of dILP2-3,5, results in elevated whole-body TAG levels (30, 70). Moreover, the results of studies using dILP-binding proteins that impair dILP function also indicate that systemic insulin signaling suppresses lipid storage in Drosophila (77–79). It has been proposed that the systemic effect of IIS on lipid metabolism masks its FB-specific effect (55).
IIS regulates lipolysis by regulating the Bmm lipase. IIS suppresses dFOXO through AKT-mediated phosphorylation. dFOXO, in turn, induces bmm expression when IIS signaling is low (80). In addition, IIS also activates salt-induced kinase 3 (SIK3), an adenosine monophosphate (AMP)–activated protein kinase (AMPK)–related protein, by AKT-mediated phosphorylation (81). SIK3 phosphorylates and inhibits HDAC4, a histone deacetylase that activates dFOXO (80, 81). Therefore, loss of IIS signaling promotes bmm expression by removing the suppressive effect of SIK3 on the dFOXO-activator HDAC4 (Fig. 2). This is supported by the observation that loss of SIK3 leads to enhanced bmm expression and depletion of stored fat (80, 81). It should be noted that the Bmm lipase is also regulated by TOR signaling, which itself is activated by IIS. Animals carrying hypomorphic alleles of TOR show induction of bmm expression (82). In addition to its effects on lipolysis, IIS also modulates β-oxidation of FFA generated through lipolysis by regulating the ACS Pdgy. Xu et al. (59) showed that dFOXO directly regulates pdgy expression and is required for starvation-induced expression of pdgy mRNA. In addition to dILPs secreted from IPCs, dILP6 secreted from the FB also plays a role in lipid mobilization during starvation. Starvation induces dILP6 mRNA expression in the larval FB in a dFOXO-dependent fashion (83). Chatterjee et al. (13) have shown that dILP6 secreted from the adult FB is important for lipid uptake and processing in the oenocytes during starvation.
Fig. 2. Convergence of nutrient-sensitive IIS, AKH signaling, and Mondo/Mlx activity on the SIK3/HDAC4/dFOXO axis.
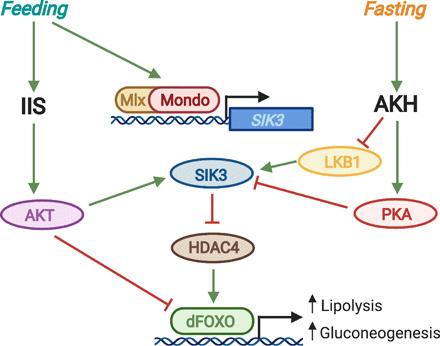
Feeding increases levels of sugar, which binds to and activates the Mondo/Mlx transcription factor complex. The Mondo/Mlx complex then binds to the carbohydrate response element (ChoRE) at the SIK3 promoter and induces SIK3 expression. Feeding also stimulates insulin/insulin-like growth factor signaling (IIS), which leads to AKT activation downstream of the InR/Chico/PI3K cascade. AKT phosphorylates and activates SIK3, which, in turn, phosphorylates and inhibits HDAC4. Active HDAC4 removes inhibitory acetylation on dFOXO and thereby activates it. Thus, SIK3-mediated inhibition of HDAC4 in response to enhanced IIS inhibits dFOXO. In addition, AKT can directly phosphorylate dFOXO to inactivate it and reduce its nuclear localization. dFOXO-induced expression of bmm lipase and PEPCK leads to an increase in lipolysis and gluconeogenesis that eventually elevates sugar levels. Consequently, feeding-mediated IIS activation reduces lipolysis and gluconeogenesis by inhibiting dFOXO. In contrast, fasting stimulates glucagon-like AKH signaling, which leads to PKA activation and inhibition by PKA of SIK3. Moreover, AKH inhibits LKB1 and thereby prevents LKB1-mediated activation of SIK3. As a result, fasting-mediated activation of AKH signaling ultimately leads to dFOXO activation and a subsequent increase in lipolysis and gluconeogenesis.
Regulation of dILP secretion
To coordinate the regulation of insulin-mediated carbohydrate metabolism with changes in blood glucose levels, mammalian pancreatic insulin secretion is controlled by glucose. How IPCs in Drosophila respond to sugars differs in larvae and adults. In Drosophila larvae, IPCs cannot directly sense sugar and dILP secretion by larval IPCs is indirectly regulated by AKH secretion (37). In contrast, in Drosophila adults, IPCs respond to hemolymph sugars directly through a mechanism that is similar to the one seen in pancreatic β cells. GLUT1-mediated glucose uptake in IPCs increases glucose catabolism and subsequent mitochondrial ATP production and thereby shuts down ATP-sensitive potassium channels (KATP-ch) (Fig. 1). This leads to membrane depolarization, the opening of voltage-dependent calcium channels, an influx of Ca2+, and eventual exocytosis of dILP-containing granules (31–33). Hepatocyte nuclear factor 4 (HNF4), which is highly expressed in adult IPCs, is required for glucose-induced dILP secretion (84). Recently, Oh et al. showed that a pair of glucose-sensing Crz+ and sNPF+ dorsolateral neurons (CN neurons) project to both IPCs and AKH-secreting CC with bifurcated axons. An increase in the hemolymph sugar level triggers these neurons to release sNPF, which binds to sNPF receptors on IPCs and promotes dILP2 secretion in adult flies through a Gq-coupled signaling mechanism. This is accompanied by a suppression of AKH secretion. Thus, CN neurons balance the opposing effects of dILPs and AKH on sugar homeostasis in Drosophila, and as a consequence, silencing of these neurons leads to hyperglycemia (34). It will be interesting to explore the role of CN neurons in dILP regulation in larvae.
Apart from direct glucose sensing, dILP release from IPCs responds to multiple additional signals secreted in response to nutrient sensing. Upd2, secreted from the FB in response to food intake, stimulates Janus kinase/signal transducers and activators of transcription signaling in GABAergic neurons and relieves their inhibitory effect on the dILP secretion from IPCs (Fig. 1) (10). Thus, Upd2, like mammalian Leptin, plays an important role in metabolic regulation in response to nutrient availability (85). Notably, FB-specific expression of human leptin can rescue the upd2 mutant phenotype of small body size and increased hemolymph glucose levels (10). The FB can also specifically sense dietary amino acids, leading to TOR-dependent release of Stunted (Sun) and growth-blocking peptides (GBPs), GBP1 and GBP2. Sun acts on its receptor, Methuselah, on IPCs to trigger the secretion of dILPs and thereby promote organ growth (86). Similar to Upd2, GBPs suppress an inhibitory neuronal relay to IPCs by binding to EGFR on inhibitory neurons and thus promote dILP release (87, 88). An opposing activity is provided by Eiger, the ortholog of mammalian tumor necrosis factor–α that is secreted from the FB in response to nutrient deprivation, which binds to its receptor, Grindelwald, on IPCs to prevent dILP production in a c-Jun N-terminal kinase (JNK)–dependent manner (89). Daw, a TGFβ ligand that is regulated by dietary sugar through Mondo-Mlx complex, promotes secretion of dILP2 and dILP5 from larval IPCs and thereby regulates sugar homeostasis (Fig. 1) (42, 90). Similarly, CCHa2, a peptide hormone that is secreted from larval FB and midgut in a nutrient-dependent manner, also regulates the expression and secretion of dILPs from IPCs (39).
Regulation of dILP sensitivity
In addition to the regulation of dILP expression and secretion, the effect of IIS on carbohydrate energy metabolism is modulated by a group of secreted factors that perturb the sensitivity of target tissues to dILPs. ImpL2, an ortholog of mammalian insulin growth factor binding proteins (IGFBPs) in Drosophila, is expressed in the FB, CC, and IPCs. ImpL2 binds to and blunts the effects of dILP2 and dILP5 (77, 79). ImpL2 has been shown to drive tumor-induced insulin resistance and organ wasting in Drosophila (91, 92). Drosophila acid-labile subunit (dALS), secreted from FB and IPCs, acts in a complex of dILP2/ImpL2/dALS to impede dILP2 function (78). In contrast, the glia-derived secreted decoy of InR (SDR), which resembles the extracellular domain of InR, binds to multiple dILPs independently of ImpL2, with the highest affinity for dILP3, and impairs their functions (93). In addition, the lipocalin-like protein NLaz mediates HSD-induced insulin resistance in Drosophila. It has been proposed that high hemolymph sugar levels activate stress-responsive JNK signaling in the FB, leading to NLaz secretion (11, 94). In addition to these secreted factors, a mutation in the microRNA miR-278 can also cause insulin resistance in the FB. Consequently, lipid stores in the FB are depleted, an effect accompanied by hyperglycemia and increased dILP secretion (95). Thus, IIS signaling integrates various metabolic signals and thereby plays a principal role in the regulation of systemic metabolism in Drosophila.
Adipokinetic hormone
Effect of AKH on carbohydrate energy metabolism
Like mammalian glucagon, AKH maintains sugar levels in the Drosophila hemolymph (96). AKH is secreted from CC neuroendocrine cells, which are located in the ring gland in larvae but attached to the esophagus after metamorphosis (35, 36). AKH acts through the AKHR, a G protein–coupled receptor, and elevates intracellular Ca2+ and cyclic AMP levels (48). Recently, Song et al. (28) showed that AKH, like mammalian glucagon, triggers the protein kinase A (PKA)/inositol-requiring enzyme (IRE)/cyclic-AMP response element binding protein 2 (CREB2) pathway downstream of AKHR in the larval FB. A loss of AKH signaling due to ablation of AKH-releasing neurons leads to a sharp decrease in hemolymph trehalose levels (97). Consistent with this, the ectopic expression of AKH in the FB elevates trehalose levels (36). The CC senses dietary sugars in the hemolymph and promotes dILP3 secretion from larval IPCs in an AKH-regulated fashion (Fig. 1) (37). AKHR mutant flies show a moderate increase in glycogen levels accompanied by high triglyceride stores and are resistant to starvation (98).
Effect of AKH on lipid energy metabolism
Analogous to mammalian glucagon, AKH promotes lipid catabolism in Drosophila. Mutations in either AKH or AKHR cause obesity in adult Drosophila, whereas AKH or AKHR overexpression leads to a lean phenotype (36, 48, 96, 99). Loss of AKH does not increase TAG levels in larvae (36, 96). Like IIS, AKH signaling can also impinge on SIK3/HDAC4/dFOXO signaling to regulate Bmm lipase and thereby regulate lipolysis (Fig. 2). AKH signaling inhibits LKB1, a kinase that directly activates SIK3 (81). The consequent lack of SIK3-mediated HDAC4 inhibition activates dFOXO, which, in turn, elevates bmm expression. However, it should be noted that obesity caused by mutation of AKHR can be exacerbated by an accompanying bmm mutation, indicating that an additional mechanism is involved in the fat depletion caused by AKH signaling (55).
Regulation of AKH signaling
AKH secretion from the larval CC is regulated by ATP-sensitive K+ channels composed of a sulfonylurea receptor (Sur) and a potassium channel (Ir) that respond to changes in the intracellular AMP/ATP ratio (35). In addition, AMPK controls Ca2+ influx into CC cells and ensuing AKH release into the hemolymph (100). Thus, energy metabolism regulates AKH secretion through a feedback loop.
How AKH secretion is regulated by hemolymph sugar levels is not fully understood. Similar to hypoglycemia-induced glucagon secretion in mammals, a steep reduction in hemolymph trehalose level leads to Ca2+ influx into larval CC cells and subsequent AKH secretion (35, 101). Opposite this, hyperglycemia triggers sNPF release from CN neurons and sNPF acts on sNPF receptors in CC to suppress AKH secretion in adult flies through a Gi/o-coupled signaling mechanism (34). Unexpectedly, high trehalose levels are also reported to promote AKH secretion in larvae (37). Furthermore, chronic HSD leads to an increase in Actβ production by the midgut that induces AKHR expression in the FB and subsequently to hyperglycemia (28). Notably, the biphasic nature of the effect of blood sugar levels on glucagon secretion is also seen in humans. High glucagon secretion despite high blood glucose levels is an important component of diabetic hyperglycemia (101).
Several secreted proteins also have been reported to regulate AKH secretion. dILP2 induces AKH expression in CC in a dILP1-dependent fashion in adult Drosophila (102). Similarly, Upd2 secreted from skeletal muscle promotes AKH secretion from CC in adults (20). On the other hand, Buriscon α (Bursα), which is secreted from the intestinal EEs in response to nutrients and binds to neuronal DLgr2 receptor, triggers a neuronal relay that prevents AKH production in CC and thereby reduces AKH signaling in the FB (103). A reduction in Bursα/DLgr2 signaling heightens the oxidative metabolism of glucose and depletes lipid stores and, as a result, renders the animals more sensitive to starvation. Apart from nutrient sensing, water-sensitive pickpocket 28 (ppk28), which is expressed in water-sensing gustatory neurons, also regulates AKH (104, 105). Lipid mobilization is increased in ppk28 mutant Drosophila due to up-regulation of AKH signaling. Thus, the secretion of AKH, a prominent regulator of energy metabolism in Drosophila, is regulated by intracellular ATP levels as well as by nutrient-sensing signals coming from distal tissues.
Mondo/Mlx in Drosophila energy metabolism
Effect of Mondo/Mlx on carbohydrate energy metabolism
Mondo [aka Mio, carbohydrate-response element-binding protein (ChREBP)] is a basic helix-loop-helix-leucine zipper (bHLH-LZ) transcription factor, which, in combination with its interacting partner Max-like X (Mlx aka Bigmax), another bHLH-LZ transcription factor, binds to carbohydrate response elements (ChoREs) in the DNA and regulates the expression of multiple genes involved in carbohydrate and lipid metabolism (41). Glycolysis plays an important role in the clearance of excess glucose from the hemolymph. Sugar feeding causes a sharp induction of glycolytic genes in a Mondo/Mlx-dependent manner (42). Consistent with this, Drosophila larvae with impaired Mondo/Mlx function display reduced survival in a sugar-rich diet (106). This recapitulates the low survival of ChREBP mutant mice in a carbohydrate-rich diet (107).
In addition to direct transcriptional regulation of target genes, Mondo/Mlx also indirectly regulates metabolic genes through the transcription factors Cabut and Sugarbabe (42, 108). Both cabut and sugarbabe mutant larvae also show sugar intolerance, indicating that they are also involved in sugar metabolism (42, 106, 108). Cabut binds to the promoter of the PEPCK gene, suppressing PEPCK expression, and thereby inhibits glucose generation through gluconeogenesis (108). In addition to these transcription factors, Mondo/Mlx also regulates other genes, including the TGFβ ligand daw and the kinase SIK3 (42, 80, 81, 109). As discussed below, Daw has a profound effect on carbohydrate metabolism. SIK3 mutants are also sugar-intolerant (80, 110).
Effect of Mondo/Mlx on lipid energy metabolism
In addition to regulating genes relevant to carbohydrate metabolism, Mondo/Mlx also regulates genes involved in lipid metabolism upon sugar feeding. Genes involved in fatty acid synthesis from TCA cycle–derived citrate, such as ATP citrate lyase, acetyl-CoA carboxylase (ACC), and Fatty acid synthase 1 (FASN1), are induced upon sugar feeding (41). It has been shown that, like mammalian ChREBP, Mondo/Mlx regulates the sugar-induced expression of ACC and FASN1 (42). In addition, the Mondo/Mlx target gene sugarbabe also positively regulates sugar-mediated induction of lipogenic genes such as stearoyl-CoA desaturase (desat1) (42). Notably, Mondo/Mlx also regulates several pentose phosphate pathway (PPP) genes and thereby controls the production of PPP-generated NADPH [reduced form of nicotinamide adenine dinucleotide (NAD+) phosphate], which is required for fatty acid synthesis (41, 42). Mondo/Mlx-induced SIK3 activates PPP by phosphorylation of glucose-6-phosphate dehydrogenase (G-6-PD) (110). Moreover, as discussed above, SIK3 suppresses the expression of the bmm lipase gene by inhibiting HDAC4/dFOXO and thereby suppresses lipolysis upon sugar feeding (Fig. 2).
Regulation of Mondo/Mlx
Mondo and Mlx, which are mainly expressed in the FB, gut, and Malpighian tubules, are induced upon sugar feeding (22, 42). The glucose sensing module including the glucose-response activation conserved element, which is present in mammalian ChREBP and MondA proteins and is required for glucose sensing, is conserved between Drosophila and mammals (111, 112). As discussed above, SIK3/HDAC4/dFOXO, in addition to sugar-induced Mondo/Mlx, is also regulated by IIS and AKH signaling and therefore acts as a point of convergence for these key determinants of energy homeostasis in Drosophila (Fig. 2). Sugarbabe, on the other hand, is regulated by nutrient-sensitive microRNA miR-14 (113). Thus, sugar levels directly regulate the Mondo/Mlx complex, whereas some of the downstream effectors of Mondo/Mlx are also modulated by other nutrient-sensing signals.
TGFβ/BMP signaling
Effect of TGFβ/BMP signaling on carbohydrate energy metabolism
The core components of TGFβ/bone morphogenetic protein (BMP) signaling are conserved between Drosophila and mammals. The TGFβ/BMP signaling pathway in Drosophila can be broadly divided into two branches: TGFβ/Activin and BMP signaling. The ligands for the TGFβ/Activin branch are Daw, Actβ, and Myo. The binding of these ligands to a complex of type I (Babo) and type II (Punt or Wit) receptors leads to phosphorylation of R-Smad (Smox), which then, in combination with a co-Smad (Med), binds to target DNA and regulates gene expression. Similarly, BMP ligands Gbb, Dpp, Scw, and Mav can bind to a complex of type I (Tkv or Sax) and type II (Punt or Wit) receptors, and their effects are mediated through a different R-Smad (Mad)/co-Smad (Med) complex (114).
The TGFβ ligand Daw affects carbohydrate metabolism in multiple ways. Daw stimulates dILP2 and dILP5 secretion from IPCs in larvae and thereby modulates sugar homeostasis (90). Moreover, Daw inhibits the expression of digestive amylases and induces the expression of sugarbabe, a sugar-sensitive lipogenic transcription factor (23, 42). A mutation in the daw gene leads to high levels of glucose and trehalose in the hemolymph, which is accompanied by elevated glycogen storage. It also leads to enhanced expression of multiple mitochondrial genes and accumulation of TCA cycle intermediates in the FB. In addition, daw mutant larvae suffer from sugar intolerance, and as a consequence, larvae grown in a sugar-rich diet show delayed development and a high frequency of death (90). Drosophila sechellia, which is defective in carbohydrate-induced, TGFβ signaling-mediated gene regulation, is similarly sensitive to a sugar-rich diet (115). Apart from Daw, Actβ also modulates sugar metabolism, but unlike daw mutants, actβ null mutants are hypoglycemic. Conversely, Actβ secreted from the midgut induces AKHR expression in the FB by acting through the Babo-A receptor (A isoform of Babo), which leads to hyperglycemia (28).
Effect of TGFβ/BMP signaling on lipid energy metabolism
The increased circulating and stored carbohydrate levels in daw mutant larvae are accompanied by elevated TAG levels (90). An increase in Actβ secretion from larval skeletal muscle due to mitochondrial electron transport chain (ETC) complex-I perturbation and the resultant increase in Actβ/Babo-A signaling in the FB also lead to increased TAG levels (116). This is accompanied by a decrease in ADP-induced oxygen consumption and ATP production in mitochondria isolated from the FB of these larvae. In addition to TGFβ/Activin ligands, the BMP ligand Gbb also affects lipid metabolism. The name of the gene glass bottom boat (gbb) is based on the glassy appearance of the gbb mutant larvae. The FBs of these larvae show significantly reduced TAG storage with a concomitant decrease in total TAG, glucose, and trehalose levels. These recapitulate metabolic changes seen during starvation. Unexpectedly, gbb mutant larvae show increased nutrient uptake, suggesting that an unidentified mechanism is involved in the TAG depletion in gbb mutant larvae (114, 117).
Regulation of TGFβ/BMP signaling
daw is strongly expressed in the FB and skeletal muscles. In response to HSD, Mondo-Mlx binds to the daw promoter and drives its expression, most prominently in the larval FB (42, 118). Moreover, dFOXO, which is inhibited by sugar-sensitive IIS signaling, suppresses daw expression (118). The mechanism involved in Actβ induction in the EEs of the midgut in response to chronic HSD is not completely understood. However, the accompanying increase in the number of EEs is at least partially responsible for Actβ induction (28). An opposite activity is exerted by perturbation of the ETC complex-I, which increases the ROS load and activates Relish, which, in turn, induces Actβ expression in the Drosophila muscle (116, 119).
HNF4 in Drosophila energy metabolism
Effect of HNF4 on carbohydrate energy metabolism
HNF4, like Mondo/Mlx, plays an important role in both sugar sensing and sugar tolerance in Drosophila. As discussed earlier, adult IPCs, unlike larval IPCs, can directly sense glucose in the hemolymph and modulate dILP secretion in a glucose-dependent manner. IPC-specific knockdown of HNF4 renders adult IPCs unresponsive to glucose, indicating that HNF4 is essential for glucose-stimulated insulin secretion (GSIS) (84). HNF4-mediated expression of glycolytic enzyme hexokinase C and oxidative phosphorylationa genes is required for the transition to the adult metabolic state, which is characterized by high oxidative phosphorylation (84). HNF4 mutant larvae develop normally but cannot eclose when fed regular laboratory food. However, a fivefold reduction in dietary sugar rescues the phenotype indicating sugar intolerance in these larvae. This sugar intolerance is also seen in HNF4 mutant adults. The metabolic state of HNF4 mutant adult flies, characterized by hyperglycemia, sugar intolerance, and the absence of GSIS, is reminiscent of maturity onset diabetes of the young 1 (MODY1), which is caused by HNF4A mutations in mammals (84).
Effect of HNF4 on lipid energy metabolism
In addition to carbohydrate metabolism, HNF4 also plays a role in lipid catabolism. Accordingly, HNF4 mutant larvae are sensitive to starvation. They show impaired TAG depletion in the midgut and FB upon starvation and also have elevated levels of long-chain fatty acids. Consistent with this observation, the results of microarray studies have shown that HNF4 regulates multiple lipolysis genes and β-oxidation genes such as lip3, bgm, and CPT1 (120).
Regulation of HNF4
The increase in HNF4 expression, which is seen in the IPCs and the FB upon the transition to adulthood, is responsible for the switch to high oxidative phosphorylation in adults (84). HNF4 is also activated by external cues such as starvation and exogenous fatty acids (120). The starvation-mediated HNF4 induction indicates a feedback loop triggered by nutrient depletion. In contrast, fatty acid–induced HNF4 activation points toward a feedforward loop in which excess fatty acid increases energy production by inducing HNF4-mediated expression of enzymes involved in β-oxidation. In addition, HNF4 is also regulated by NAD+-dependent deacetylase sirtuin 2 (Sir2). Sir2-mediated deacetylation of HNF4 enhances its activity and stability (121). Sir2 was previously identified as a regulator of lipid storage in a genetic screen in Drosophila (122). Reis et al. showed that knockdown of Sir2 in the larval FB leads to increased lipid levels and Sir2 prevents lipid accumulation in a cell-autonomous manner. Moreover, sir2 knockdown in adult FB affects fat metabolism and reduces dILP5 expression in the IPCs, thereby affecting systemic IIS (123). A study by Palu and Thummel showed that loss of Sir2 causes the age-progressive onset of obesity, hyperglycemia, glucose intolerance, and insulin resistance (121). Moreover, there is a substantial overlap between genes regulated by Sir2 and HNF4. Congruent with this observation, HNF4 mutations recapitulate the diabetic phenotypes seen in sir2 mutants. Notably, enhanced HNF4 expression rescues the IIS-associated phenotypes in sir2 mutants, suggesting that HNF4 is a key effector of the metabolic functions of Sir2 in Drosophila (121). Thus, HNF4 is regulated by nutrient availability as well as by the metabolic sensor Sir2.
DROSOPHILA AS A MODEL SYSTEM TO STUDY OBESITY AND DIABETES
Obesity, caused by an imbalance in energy metabolism, is a modern-day epidemic. It increases the risk of several diseases, including diabetes, cardiovascular diseases, and cancer. The term “diabesity” is used to describe the frequent coexistence of obesity and type 2 diabetes (T2D). A better understanding of the molecular etiology of obesity and diabetes is of utmost importance to improve treatment. Drosophila is an attractive model for the study of obesity and diabetes because, as discussed above, the organs involved in energy metabolism in Drosophila are mostly analogous to those in humans and many of the molecular regulators of energy metabolism are common between Drosophila and humans. A large majority of disease-associated genes, including those involved in metabolic diseases, are conserved between Drosophila and humans (124). Like humans, excess dietary calories lead to obesity in Drosophila, and this diet-induced obesity is accompanied by the hallmarks of T2D (45, 125). Last, the availability of a wide variety of genetic tools and the feasibility of large-scale screens make Drosophila a particularly useful model system for the study of obesity and diabetes. We have already discussed how sugar-rich diet and genetic perturbation of different regulators of Drosophila energy metabolism affect lipid accumulation and impair insulin signaling, leading to obesity and diabetes. In this section, we describe some of these in more detail and discuss additional widely used models of obesity and diabetes in Drosophila.
High-fat diet–induced obesity model
Similar to mammals, when fed a high-fat diet (HFD), Drosophila develops obesity characterized by increased total TAG content (125). Moreover, flies on HFD also develop obesity in a dose-dependent fashion and deposit fat in nonadipose tissues such as the gut and the heart. The ensuing cardiomyopathy in Drosophila, characterized by reduced cardiac contractility, conduction block, and structural pathologies, can be prevented, along with whole-body TAG accumulation, by systemic inhibition of TOR signaling, which is activated by HFD (125). Inhibition of TOR signaling or its downstream target fatty acid synthase, specifically in the heart or FB, can preserve heart function in flies on HFD. Similarly, heart-specific or FB-specific overexpression of dFOXO or its target bmm lipase can also ameliorate HFD-induced cardiomyopathy (125). It should be noted that HFD also leads to life-span reduction in Drosophila (126).
Comparable to mammalian diabesity, HFD causes hyperglycemia, hyperinsulinemia (elevated dILP2 secretion), and a progressive decline in insulin signaling characterized by a reduction in phosphorylated AKT levels (125, 126). Recently, Hong et al. (127) showed that HFD induces expression of the BMP ligand gbb, particularly in the FB. Intriguingly, FB-specific gbb overexpression is sufficient to cause obesity and insulin resistance in Drosophila. Consistent with this observation, FB-specific Gbb inhibition mitigates HFD-mediated metabolic changes (127). As discussed earlier, gbb mutation also leads to depletion of lipids in the larval FB. spargel (srl) is the Drosophila ortholog of the transcriptional coactivator PGC1α that encodes a master regulator of genes involved in oxidative phosphorylation and mitochondrial biogenesis in mammals (128). srl also regulates mitochondrial activity and biogenesis (129–131). In addition, Srl regulates the expression of multiple OXPHOS and TCA cycle genes in adipose tissues and is necessary for insulin signaling-mediated induction of these genes in the larvae (132). Diop et al. (133) found that srl mutation mimics and exacerbates the effect of HFD on obesity and cardiac dysfunction. srl overexpression reduces TAG levels in Drosophila. Diop and colleagues reported that HFD-mediated activation of TOR signaling suppresses the expression of srl. Diop et al. (133) also found that the Bmm lipase acted downstream of TOR and upstream of Srl in HFD-mediated cardiac lipotoxicity.
Drosophila genetic models of obesity
The usefulness of Drosophila genetics in uncovering previously unidentified regulators of human obesity is highlighted by multiple examples, the adipose mutation being one of the earliest and most prominent among them. A mutation in the adipose gene in Drosophila promotes obesity, which is accompanied by low carbohydrate storage (134). Notably, a mutation in the adipose ortholog WD and tetratricopeptide repeats 1 (WDTC1) gene, which encodes a substrate receptor for a Cullin4-RING E3 ligase, DDB 1-CUL 4-ROC 1 (CRL 4), leads to obesity in both mice and humans (135). Groh and colleagues proposed that WDTC1 causes epigenetic silencing of adipogenic genes via histone H2A monoubiquitylation. Intriguingly, Baumbach and colleagues previously identified Cullin 4 as an anti-obesity gene in a targeted in vivo RNA interference (RNAi) screen in Drosophila, highlighting the similarities between Drosophila and mammalian obesity regulation (136). In the same RNAi screen, it was also found that knockdown of the stromal interaction molecule (stim) gene, a core component of store-operated calcium entry (SOCE) in fat storage tissues (i.e., the FB and the midgut), leads to hyperphagia and obesity. The authors also showed that the effect of stim knockdown on fat storage is mediated via an increase in sNPF expression in the brain. Neuropeptide Y, which is a functional homolog of sNPF in mammals, is a strong orexigenic peptide that promotes diet-induced obesity in mammals (136). The expression of bmm lipase in FB is reduced in stim knockdown flies. Incidentally, a mutation in bmm itself almost doubles fat storage and can serve as a model for obesity in Drosophila (137). Similarly, AKH mutation, which also affects bmm expression (Fig. 2), significantly elevates fat content in adults (36, 96).
The genome-wide RNAi screen conducted by Pospisilik and colleagues provides another prime example of how large-scale screens in Drosophila can successfully illuminate conserved mechanisms of adiposity regulation in mammals. More than 60% of candidate genes identified in this screen were conserved between Drosophila and humans, and notably, a large number were previously uncharacterized (138). Hedgehog (hh) signaling was the top-scoring FB-specific pathway in Drosophila. Moreover, hh was one of the candidate genes and was subsequently shown to play an important role in white versus brown adipocyte cell fate determination in mice. Activation of hh signaling prevents the differentiation of white adipocytes by its direct action on early adipogenic genes (138). In a different approach, Lee and colleagues used Drosophila genetics to further investigate how the cardiac muscle-specific expression of MED13, which encodes a subunit of the Mediator complex, regulates systemic fat storage in mice. It was previously reported that a decrease in MED13 expression in the cardiac tissue promotes obesity in mice whereas cardiac-specific MED13 overexpression results in a lean phenotype. Consistent with that observation, Lee et al. (139) showed that muscle-specific knockdown of MED13 ortholog skuld (skd) promotes obesity in Drosophila. Moreover, through a large-scale screen, they found that wingless (wg), which encodes a ligand of the Wnt/Wg signaling pathway, acts as a downstream effector of MED13 in the regulation of obesity in Drosophila.
Regulation of sedentary energy expenditure affects metabolism and obesity in mammals. Sarcolipin increases energy expenditure in muscle by interacting with Sarco/ER Ca2+ adenosine triphosphatase (SERCA) and promoting ATP hydrolysis. SERCA uses the energy produced by ATP hydrolysis to pump Ca2+ into the endoplasmic reticulum (ER) lumen. Sarcolipin blocks Ca2+ accumulation in the ER lumen without affecting ATP hydrolysis and thereby uncouples ATP hydrolysis from Ca2+ transport. Sarcolipin thus promotes metabolic heat generation without any muscle activity (nonshivering thermogenesis). It also improves glucose tolerance and prevents obesity (140). Changes in energy expenditure control obesity in Drosophila as well (141, 142). Moraru et al. recently reported that thyroid adenoma associated (THADA) uncouples ATP hydrolysis from the Ca2+ transport conducted by SERCA. This leads to reduced Ca2+ transport into the ER, and as a consequence, the energy from ATP hydrolysis is dissipated in the form of heat (141). Consistent with this observation, THADA-deficient Drosophila has reduced energy production, is hyperphagic and obese, and is sensitive to cold. Notably, THADA had been associated with diabetes and cold adaptation in humans, but the molecular basis of its function was unknown (141). This illustrates how Drosophila can be used to uncover the mechanism of action of known regulators of energy homeostasis in humans.
Like Sarcolipin, mitochondrial uncoupling proteins (UCPs) can affect energy metabolism by increasing sedentary energy expenditure. Mitochondrial ATP synthase produces ATP by using the proton gradient generated across the inner mitochondrial membrane due to electron transfer through the mitochondrial ETC. Mitochondrial UCPs, a family of transmembrane proton carriers, can cause proton leakage across the inner mitochondrial membrane and dissipate the electrochemical energy in the form of heat. This dissociates mitochondrial respiration from ATP synthesis, hence the name “uncoupled respiration.” The mammalian uncoupling protein UCP1, which is induced in response to cold exposure, promotes nonshivering thermogenesis in brown adipose tissue through this mechanism. Like Sarcolipin, UCP1 overexpression increases whole-body energy expenditure, promotes weight loss, and improves glucose homeostasis (143). There are four mitochondrial UCPs in Drosophila (dUCP4a, dUCP4b, dUCP4c, and dUCP5). Da-Re and colleagues found that dUCP4c promotes mitochondrial uncoupling and is required for the development of larvae when raised at cold temperatures (144). Recently, Ulgherait et al. showed that the expression of dUCP4b and dUCP4c is regulated by the circadian rhythm master-regulator gene period (per). Their periodic expression reduces the ROS load by reducing the mitochondrial “proton motive force.” Moreover, their function is important in Drosophila for the regulation of proliferative homeostasis in ISCs and life span (145).
HSD-induced T2D model
The HSD approach has been widely used to cause caloric overload and to perturb the carbohydrate homeostasis in Drosophila. For example, Musselman and colleagues showed that like human diabetics, larvae fed HSD develop hyperglycemia, characterized by an increase in hemolymph glucose levels (45). This is accompanied by an increase in the levels of trehalose, which, as discussed above, is the primary carbohydrate molecule in the Drosophila hemolymph. Larvae fed an HSD diet also develop insulin resistance in peripheral tissues, a hallmark of T2D (45). Analogous to compensatory hyperinsulinemia seen in T2D patients, HSD feeding of larvae leads to enhanced expression of dILP2, dILP3, and dILP5, although they cannot activate IIS (45, 94). Notably, HSD also leads to obesity, characterized by TAG accumulation and increased lipid droplet size (45). As discussed above, lipocalin-like protein NLaz is implicated in HSD-induced insulin resistance in the peripheral tissues of Drosophila. It has been argued that high hemolymph sugar levels activate stress-responsive JNK signaling in the FB leading to NLaz secretion (11, 94). Overexpression of Apolipoprotein D (ApoD), the mammalian ortholog of NLaz, leads to glucose intolerance, insulin resistance, and hepatic steatosis in mice (146). Thus, Drosophila can be used to identify additional factors that mediate the metabolic effects of HSD.
Adult Drosophila, when fed HSD, shows similar metabolic effects such as hyperglycemia, insulin resistance, and obesity (147). In addition, they develop cardiomyopathy manifested by fat and collagen accumulation in the cardiac tissue, and arrhythmias that gradually progress to fibrillations and asystolic periods. Previously, an increase in hexosamine flux has been implicated in hyperglycemia and T2D-initiated cardiac dysfunction in mammals (147). Consistent with that observation, the hexosamine pathway was activated in adult Drosophila fed HSD. Knockdown of hexosamine pathway components alleviates HSD-induced cardiac dysfunction in adult Drosophila. HSD also reduces life span in adult flies (147). However, it should be noted that flies on HSD do not have access to additional water to compensate for the elevated osmolarity of the diet and Rovenko et al. (148) have shown that flies on HSD have reduced body water content. In agreement with this observation, van Dam and colleagues have recently demonstrated that the life span of these animals can be rescued by water supplementation (47). However, it does not rescue metabolic effects like obesity and insulin resistance. Water supplementation thereby uncouples the metabolic effects of HSD from its life-span effect in adult Drosophila. van Dam et al. also found that HSD-mediated dehydration leads to the accumulation of uric acid, an end product of purine metabolism, as well as the deposition of renal stones. Inhibition of uric acid production partially rescued the life-span effects of HSD in Drosophila (47). Consistent with Drosophila studies, the results of a large human cohort analysis showed that dietary sugar intake is a strong predictor of circulating purine levels and renal function (47).
Drosophila genetic models of diabetes
Drosophila genetic models of T2D
The inability to respond to insulin due to impaired insulin signaling (i.e., insulin resistance) is a hallmark of T2D. Impaired activity of InR or any of the downstream components of IIS can lead to insulin resistance. For instance, a loss of InR activity due to a heterozygous InR mutant combination (InR05545/InR+) leads to insulin resistance in Drosophila. Similar to diabetic hyperinsulinemia, these flies exhibit an increase in dILP secretion (33, 76). However, there is no significant change either in the circulating carbohydrate levels or in adiposity. This is unlike the HSD-induced T2D model where increased lipid accumulation is accompanied by elevated levels of circulating sugars (149). It will be interesting to study the link between lipid accumulation and circulating carbohydrate levels in Drosophila T2D models. FB-specific knockdown of InR is sufficient to phenocopy InR05545/InR+ mutation, indicating that IIS in the FB is a major determinant of systemic metabolism in Drosophila (33). In addition to this mechanism, the aforementioned secreted peptides that bind to dILPs and impede their function can also lead to insulin resistance. For example, Drosophila ImpL2 can bind to dILP2 and dILP5 and, like mammalian IGFBPs, cause insulin resistance (77, 79). As discussed above, ImpL2 has been implicated in tumor-induced insulin resistance and organ wasting in Drosophila (91, 92). dALS and SDR might also cause insulin resistance through a similar mechanism (78, 93).
Drosophila genetic models of type 1 diabetes
Type 1 diabetes (T1D) is caused by the lack of insulin secretion. This can be modeled in Drosophila by removing either the dILPs or the dILP-producing IPCs. Genetic ablation of IPCs severely impedes larval growth. This is accompanied by an increase in circulating glucose and trehalose levels that persists even in adulthood (29, 30). Likewise, larvae with dILP2-3,5 triple mutation show high glucose and trehalose concentrations in the hemolymph, suggesting that these dILPs play a prominent role in glucose metabolism (30, 70, 71). Intriguingly, a loss of systemic insulin signaling due to either IPC ablation or dILP2-3,5 triple deficiency elevates glycogen storage, indicating the involvement of an alternative mechanism of regulation of glycogen synthesis (30, 70). Similarly, lipid levels are also elevated by both larval IPC ablation and dILP2-3,5 triple mutations (29, 30, 70, 71). Haselton and colleagues performed an oral glucose tolerance test and showed that the glucose levels were higher and clearance was slower in adults in which IPCs were ablated (150). Moreover, this phenotype was rescued by an injection of bovine insulin, indicating that this Drosophila model closely resembles mammalian T1D.
CONCLUSIONS AND OUTLOOK
The Drosophila FB, like the mammalian liver, serves as a principal metabolic organ that coordinates nutrient availability with energy demand. Its metabolic function is complemented by oenocytes, which, like mammalian hepatocytes, facilitate lipid mobilization in response to starvation. In addition, the FB also serves as the primary storage organ, similar to the mammalian WAT. Skeletal muscles are the main site of energy expenditure, store glycogen, and affect metabolism through secreted myokines. Drosophila, like mammals, depends on circulating glucose as a readily available source of energy and uses stored glycogen and fat during nutrient scarcity. Unlike in mammals, in Drosophila, trehalose is found in high concentration in the hemolymph and is particularly suited to meet the high energy demand of the flight muscles. Metabolic activities in different tissues are regulated by endocrine signals such as dILPs and AKH from neuroendocrine cells, and paracrine signals such as Actβ and Upd2 from other organs. In addition, metabolic processes are also influenced by the developmental stage and external signals such as nutrient availability. These factors are integrated to appropriately modulate ATP production according to tissue type, developmental stage, and environmental conditions.
In addition to endocrine and paracrine signals, intestinal microbiota plays an important role in metabolic regulation in mammals (151). Recent studies have shown that commensal gut bacteria also modulate metabolism in Drosophila. For example, acetate, a metabolic by-product of Drosophila intestinal bacteria, enhances insulin signaling by suppressing ImpL2 (152, 153). In addition, HSD promotes the growth of resident uracil-secreting gut bacteria, which, in turn, promotes larval growth and protects against HSD-induced damages (154). Whereas humans have a wide variety of commensal bacteria in their intestines, the intestinal microbiota of Drosophila is composed of a limited variety of bacteria (155). This, combined with the ease of generating axenic flies and the genetic tractability of the host, makes Drosophila a good model system to study the effect of microbiota on metabolism and how dietary changes cross-talk with gut bacteria to influence energy metabolism.
Drosophila can also be used to study the “Warburg effect” seen in cancer cells characterized by a metabolic reprogramming from an energy-efficient metabolism driven by the TCA cycle and oxidative phosphorylation-driven to a growth-promoting glycolytic metabolism. Drosophila undergoes metabolic reprogramming in the reverse direction, i.e., from a glycolytic to an OXPHOS-dependent metabolism during the transition from larvae to adults. As discussed earlier, HNF4 expression plays a crucial role in this transition. In addition, Drosophila estrogen-related receptor (dERR), another nuclear receptor, promotes larval growth by inducing glycolytic and pentose phosphate pathway genes (156). Consequently, dERR mutant larvae display reduced expression of multiple glycolytic genes, elevated levels of circulating sugars, and low levels of lactate and TAG, and die during larval stages. Both human HNF4A and ERRs have been implicated in cancer (157, 158). Additional studies in Drosophila will shed light on the regulation of these genes and elucidate the mechanistic details of their role in metabolic reprogramming.
The role of UCPs in Drosophila energy metabolism is not well understood. As discussed above, recent studies have shown that dUCPs, like mammalian UCPs, can protect against cold and oxidative stress, but their regulation is poorly studied. Mammalian UCP1 is regulated by environmental signals such as cold and diet and by signaling pathways like β-adrenergic signaling and TGfβ/BMP signaling (143, 159, 160). It will be interesting to study how different environmental and physiological signals regulate UCPs in Drosophila. In addition, the signaling pathways that coordinate the expression of dUCPs with these regulatory factors should be studied to better understand how they influence energy metabolism in Drosophila. For example, exploration of the molecular mechanism of the regulation of dUCP4b and dUCP4c expression by circadian rhythm-controlling period gene will establish a mechanistic link between the activity/rest cycle and energy metabolism. Drosophila recapitulates salient features of human obesity and diabetes, two highly prevalent and frequently coexisting disorders of energy metabolism. Moreover, key determinants of these disorders, such as hedgehog signaling in obesity and hexosamine flux in T2D-induced cardiomyopathy, are conserved from Drosophila to humans. These parallels allow Drosophila genetics to be used to further dissect the molecular etiology of these diseases and uncover druggable targets for new therapies.
Acknowledgments
We thank A, Ghosh, P. Jouandin, S. Entwisle, I. Droujinine, J. Li, and S. Mohr for comments on the manuscript. Figures are created with BioRender.com. Funding: Work in the Perrimon laboratory is supported by 5P01CA120964, R01AR057352, and HHMI. N.P. is an investigator of the Howard Hughes Medical Institute. Author contributions: N.C. and N.P. conceptualized and wrote the manuscript. Both authors reviewed the completed manuscript. Competing interests: The authors declare that they have no competing interests. Data and materials availability: Not applicable.
REFERENCES AND NOTES
- 1.J. Dunn, M. H. Grider, Physiology, adenosine triphosphate (ATP), in StatPearls (Treasure Island, 2020). [PubMed] [Google Scholar]
- 2.Wigglesworth V. B., The utilization of reserve substances in Drosophila during flight. J. Exp. Biol. 26, 150–163 (1949). [DOI] [PubMed] [Google Scholar]
- 3.Rajan A., Perrimon N., Of flies and men: Insights on organismal metabolism from fruit flies. BMC Biol. 11, 38 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baker K. D., Thummel C. S., Diabetic larvae and obese flies-emerging studies of metabolism in Drosophila. Cell Metab. 6, 257–266 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Owusu-Ansah E., Perrimon N., Modeling metabolic homeostasis and nutrient sensing in Drosophila: Implications for aging and metabolic diseases. Dis. Model. Mech. 7, 343–350 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Padmanabha D., Baker K. D., Drosophila gains traction as a repurposed tool to investigate metabolism. Trends Endocrinol. Metab. 25, 518–527 (2014). [DOI] [PubMed] [Google Scholar]
- 7.Canavoso L. E., Jouni Z. E., Karnas K. J., Pennington J. E., Wells M. A., Fat metabolism in insects. Annu. Rev. Nutr. 21, 23–46 (2001). [DOI] [PubMed] [Google Scholar]
- 8.Palm W., Sampaio J. L., Brankatschk M., Carvalho M., Mahmoud A., Shevchenko A., Eaton S., Lipoproteins in Drosophila melanogaster—Assembly, function, and influence on tissue lipid composition. PLOS Genet. 8, e1002828 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Colombani J., Raisin S., Pantalacci S., Radimerski T., Montagne J., Leopold P., A nutrient sensor mechanism controls Drosophila growth. Cell 114, 739–749 (2003). [DOI] [PubMed] [Google Scholar]
- 10.Rajan A., Perrimon N., Drosophila cytokine unpaired 2 regulates physiological homeostasis by remotely controlling insulin secretion. Cell 151, 123–137 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hull-Thompson J., Muffat J., Sanchez D., Walker D. W., Benzer S., Ganfornina M. D., Jasper H., Control of metabolic homeostasis by stress signaling is mediated by the lipocalin NLaz. PLOS Genet. 5, e1000460 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gutierrez E., Wiggins D., Fielding B., Gould A. P., Specialized hepatocyte-like cells regulate Drosophila lipid metabolism. Nature 445, 275–280 (2007). [DOI] [PubMed] [Google Scholar]
- 13.Chatterjee D., Katewa S. D., Qi Y., Jackson S. A., Kapahi P., Jasper H., Control of metabolic adaptation to fasting by dILP6-induced insulin signaling in Drosophila oenocytes. Proc. Natl. Acad. Sci. U.S.A. 111, 17959–17964 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Huang K., Chen W., Zhu F., Li P. W.-L., Kapahi P., Bai H., RiboTag translatomic profiling of Drosophila oenocytes under aging and induced oxidative stress. BMC Genomics 20, 50 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wegener G., Flying insects: Model systems in exercise physiology. Experientia 52, 404–412 (1996). [DOI] [PubMed] [Google Scholar]
- 16.Becker A., Schloder P., Steele J. E., Wegener G., The regulation of trehalose metabolism in insects. Experientia 52, 433–439 (1996). [DOI] [PubMed] [Google Scholar]
- 17.Wojtas K., Slepecky N., von Kalm L., Sullivan D., Flight muscle function in Drosophila requires colocalization of glycolytic enzymes. Mol. Biol. Cell 8, 1665–1675 (1997). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sullivan D. T., MacIntyre R., Fuda N., Fiori J., Barrilla J., Ramizel L., Analysis of glycolytic enzyme co-localization in Drosophila flight muscle. J. Exp. Biol. 206, 2031–2038 (2003). [DOI] [PubMed] [Google Scholar]
- 19.Demontis F., Perrimon N., FOXO/4E-BP signaling in Drosophila muscles regulates organism-wide proteostasis during aging. Cell 143, 813–825 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhao X., Karpac J., Muscle directs diurnal energy homeostasis through a myokine-dependent hormone module in Drosophila. Curr. Biol. 27, 1941–1955.e6 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Miguel-Aliaga I., Jasper H., Lemaitre B., Anatomy and physiology of the digestive tract of Drosophila melanogaster. Genetics 210, 357–396 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zinke I., Schutz C. S., Katzenberger J. D., Bauer M., Pankratz M. J., Nutrient control of gene expression in Drosophila: Microarray analysis of starvation and sugar-dependent response. EMBO J. 21, 6162–6173 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chng W. A., Sleiman M. S. B., Schupfer F., Lemaitre B., Transforming growth factor β/Activin signaling functions as a sugar-sensing feedback loop to regulate digestive enzyme expression. Cell Rep. 9, 336–348 (2014). [DOI] [PubMed] [Google Scholar]
- 24.Bujold M., Gopalakrishnan A., Nally E., King-Jones K., Nuclear receptor DHR96 acts as a sentinel for low cholesterol concentrations in Drosophila melanogaster. Mol. Cell. Biol. 30, 793–805 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sieber M. H., Thummel C. S., Coordination of triacylglycerol and cholesterol homeostasis by DHR96 and the Drosophila LipA homolog magro. Cell Metab. 15, 122–127 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Karpac J., Biteau B., Jasper H., Misregulation of an adaptive metabolic response contributes to the age-related disruption of lipid homeostasis in Drosophila. Cell Rep. 4, 1250–1261 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Song W., Veenstra J. A., Perrimon N., Control of lipid metabolism by tachykinin in Drosophila. Cell Rep. 9, 40–47 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Song W., Cheng D., Hong S., Sappe B., Hu Y., Wei N., Zhu C., O’Connor M. B., Pissios P., Perrimon N., Midgut-derived activin regulates glucagon-like action in the fat body and glycemic control. Cell Metab. 25, 386–399 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rulifson E. J., Kim S. K., Nusse R., Ablation of insulin-producing neurons in flies: Growth and diabetic phenotypes. Science 296, 1118–1120 (2002). [DOI] [PubMed] [Google Scholar]
- 30.Broughton S. J., Piper M. D., Ikeya T., Bass T. M., Jacobson J., Driege Y., Martinez P., Hafen E., Withers D. J., Leevers S. J., Partridge L., Longer lifespan, altered metabolism, and stress resistance in Drosophila from ablation of cells making insulin-like ligands. Proc. Natl. Acad. Sci. U.S.A. 102, 3105–3110 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fridell Y.-W., Hoh M., Kréneisz O., Hosier S., Chang C., Scantling D., Mulkey D. K., Helfand S. L., Increased uncoupling protein (UCP) activity in Drosophila insulin-producing neurons attenuates insulin signaling and extends lifespan. Aging 1, 699–713 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kréneisz O., Chen X., Fridell Y.-W., Mulkey D. K., Glucose increases activity and Ca2+ in insulin-producing cells of adult Drosophila. Neuroreport 21, 1116–1120 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Park S., Alfa R. W., Topper S. M., Kim G. E., Kockel L., Kim S. K., A genetic strategy to measure circulating Drosophila insulin reveals genes regulating insulin production and secretion. PLOS Genet. 10, e1004555 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Oh Y., Lai J. S.-Y., Mills H. J., Erdjument-Bromage H., Giammarinaro B., Saadipour K., Wang J. G., Abu F., Neubert T. A., Suh G. S. B., A glucose-sensing neuron pair regulates insulin and glucagon in Drosophila. Nature 574, 559–564 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kim S. K., Rulifson E. J., Conserved mechanisms of glucose sensing and regulation by Drosophila corpora cardiaca cells. Nature 431, 316–320 (2004). [DOI] [PubMed] [Google Scholar]
- 36.Lee G., Park J. H., Hemolymph sugar homeostasis and starvation-induced hyperactivity affected by genetic manipulations of the adipokinetic hormone-encoding gene in Drosophila melanogaster. Genetics 167, 311–323 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kim J., Neufeld T. P., Dietary sugar promotes systemic TOR activation in Drosophila through AKH-dependent selective secretion of Dilp3. Nat. Commun. 6, 6846 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Dus M., Lai J. S.-Y., Gunapala K. M., Min S., Tayler T. D., Hergarden A. C., Geraud E., Joseph C. M., Suh G. S., Nutrient sensor in the brain directs the action of the brain-gut axis in Drosophila. Neuron 87, 139–151 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sano H., Nakamura A., Texada M. J., Truman J. W., Ishimoto H., Kamikouchi A., Nibu Y., Kume K., Ida T., Kojima M., The Nutrient-Responsive Hormone CCHamide-2 controls growth by regulating insulin-like peptides in the brain of Drosophila melanogaster. PLOS Genet. 11, e1005209 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Alfa R. W., Park S., Skelly K. R., Poffenberger G., Jain N., Gu X., Kockel L., Wang J., Liu Y., Powers A. C., Kim S. K., Suppression of insulin production and secretion by a decretin hormone. Cell Metab. 21, 323–334 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mattila J., Hietakangas V., Regulation of carbohydrate energy metabolism in Drosophila melanogaster. Genetics 207, 1231–1253 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mattila J., Havula E., Suominen E., Teesalu M., Surakka I., Hynynen R., Kilpinen H., Vaananen J., Hovatta I., Kakela R., Ripatti S., Sandmann T., Hietakangas V., Mondo-Mlx mediates organismal sugar sensing through the Gli-similar transcription factor sugarbabe. Cell Rep. 13, 350–364 (2015). [DOI] [PubMed] [Google Scholar]
- 43.Ugrankar R., Berglund E., Akdemir F., Tran C., Kim M. S., Noh J., Schneider R., Ebert B., Graff J. M., Drosophila glucome screening identifies Ck1alpha as a regulator of mammalian glucose metabolism. Nat. Commun. 6, 7102 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Shukla E., Thorat L. J., Nath B. B., Gaikwad S. M., Insect trehalase: Physiological significance and potential applications. Glycobiology 25, 357–367 (2015). [DOI] [PubMed] [Google Scholar]
- 45.Musselman L. P., Fink J. L., Narzinski K., Ramachandran P. V., Hathiramani S. S., Cagan R. L., Baranski T. J., A high-sugar diet produces obesity and insulin resistance in wild-type Drosophila. Dis. Model. Mech. 4, 842–849 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Graham P., Pick L., Drosophila as a model for diabetes and diseases of insulin resistance. Curr. Top. Dev. Biol. 121, 397–419 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.van Dam E., van Leeuwen L. A. G., Dos Santos E., James J., Best L., Lennicke C., Vincent A. J., Marinos G., Foley A., Buricova M., Mokochinski J. B., Kramer H. B., Lieb W., Laudes M., Franke A., Kaleta C., Cochemé H. M., Sugar-induced obesity and insulin resistance are uncoupled from shortened survival in Drosophila. Cell Metab. 31, 710–725.e7 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Galikova M., Klepsatel P., Obesity and aging in the Drosophila model. Int. J. Mol. Sci. 19, 1896 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Garrido D., Rubin T., Poidevin M., Maroni B., Le Rouzic A., Parvy J. P., Montagne J., Fatty acid synthase cooperates with glyoxalase 1 to protect against sugar toxicity. PLOS Genet. 11, e1004995 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Yamada T., Habara O., Kubo H., Nishimura T., Fat body glycogen serves as a metabolic safeguard for the maintenance of sugar levels in Drosophila. Development 145, dev158865 (2018). [DOI] [PubMed] [Google Scholar]
- 51.Yamada T., Habara O., Yoshii Y., Matsushita R., Kubo H., Nojima Y., Nishimura T., The role of glycogen in development and adult fitness in Drosophila. Development 146, dev176149 (2019). [DOI] [PubMed] [Google Scholar]
- 52.Yamada T., Hironaka K. I., Habara O., Morishita Y., Nishimura T., A developmental checkpoint directs metabolic remodelling as a strategy against starvation in Drosophila. Nat. Metab. 2, 1096–1112 (2020). [DOI] [PubMed] [Google Scholar]
- 53.Zirin J., Nieuwenhuis J., Perrimon N., Role of autophagy in glycogen breakdown and its relevance to chloroquine myopathy. PLoS Biol. 11, e1001708 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Eanes W. F., Merritt T. J., Flowers J. M., Kumagai S., Sezgin E., Zhu C.-T., Flux control and excess capacity in the enzymes of glycolysis and their relationship to flight metabolism in Drosophila melanogaster. Proc. Natl. Acad. Sci. U.S.A. 103, 19413–19418 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lehmann M., Endocrine and physiological regulation of neutral fat storage in Drosophila. Mol. Cell. Endocrinol. 461, 165–177 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Heier C., Kuhnlein R. P., Triacylglycerol metabolism in Drosophila melanogaster. Genetics 210, 1163–1184 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Horne I., Haritos V. S., Oakeshott J. G., Comparative and functional genomics of lipases in holometabolous insects. Insect Biochem. Mol. Biol. 39, 547–567 (2009). [DOI] [PubMed] [Google Scholar]
- 58.Sivachenko A., Gordon H. B., Kimball S. S., Gavin E. J., Bonkowsky J. L., Letsou A., Neurodegeneration in a Drosophila model of adrenoleukodystrophy: The roles of the Bubblegum and Double bubble acyl-CoA synthetases. Dis. Model. Mech. 9, 377–387 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Xu X., Gopalacharyulu P., Seppanen-Laakso T., Ruskeepaa A. L., Aye C. C., Carson B. P., Mora S., Oresic M., Teleman A. A., Insulin signaling regulates fatty acid catabolism at the level of CoA activation. PLOS Genet. 8, e1002478 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kishita Y., Tsuda M., Aigaki T., Impaired fatty acid oxidation in a Drosophila model of mitochondrial trifunctional protein (MTP) deficiency. Biochem. Biophys. Res. Commun. 419, 344–349 (2012). [DOI] [PubMed] [Google Scholar]
- 61.Martins J. R., Bitondi M. M., The HEX 110 hexamerin is a cytoplasmic and nucleolar protein in the ovaries of Apis mellifera. PLOS ONE 11, e0151035 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Marron M. T., Markow T. A., Kain K. J., Gibbs A. G., Effects of starvation and desiccation on energy metabolism in desert and mesic Drosophila. J. Insect Physiol. 49, 261–270 (2003). [DOI] [PubMed] [Google Scholar]
- 63.Saxton R. A., Sabatini D. M., mTOR signaling in growth, metabolism, and disease. Cell 169, 361–371 (2017). [DOI] [PubMed] [Google Scholar]
- 64.Geminard C., Rulifson E. J., Leopold P., Remote control of insulin secretion by fat cells in Drosophila. Cell Metab. 10, 199–207 (2009). [DOI] [PubMed] [Google Scholar]
- 65.Tiebe M., Lutz M., De La Garza A., Buechling T., Boutros M., Teleman A. A., REPTOR and REPTOR-BP regulate organismal metabolism and transcription downstream of TORC1. Dev. Cell 33, 272–284 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Puig O., Marr M. T., Ruhf M. L., Tjian R., Control of cell number by Drosophila FOXO: Downstream and feedback regulation of the insulin receptor pathway. Genes Dev. 17, 2006–2020 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Maniere G., Ziegler A. B., Geillon F., Featherstone D. E., Grosjean Y., Direct sensing of nutrients via a LAT1-like transporter in Drosophila insulin-producing cells. Cell Rep. 17, 137–148 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Placais P. Y., de Tredern E., Scheunemann L., Trannoy S., Goguel V., Han K. A., Isabel G., Preat T., Upregulated energy metabolism in the Drosophila mushroom body is the trigger for long-term memory. Nat. Commun. 8, 15510 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Brogiolo W., Stocker H., Ikeya T., Rintelen F., Fernandez R., Hafen E., An evolutionarily conserved function of the Drosophila insulin receptor and insulin-like peptides in growth control. Curr. Biol. 11, 213–221 (2001). [DOI] [PubMed] [Google Scholar]
- 70.Gronke S., Clarke D. F., Broughton S., Andrews T. D., Partridge L., Molecular evolution and functional characterization of Drosophila insulin-like peptides. PLOS Genet. 6, e1000857 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ikeya T., Galic M., Belawat P., Nairz K., Hafen E., Nutrient-dependent expression of insulin-like peptides from neuroendocrine cells in the CNS contributes to growth regulation in Drosophila. Curr. Biol. 12, 1293–1300 (2002). [DOI] [PubMed] [Google Scholar]
- 72.Semaniuk U. V., Gospodaryov D. V., Feden’ko K. M., Yurkevych I. S., Vaiserman A. M., Storey K. B., Simpson S. J., Lushchak O., Insulin-like peptides regulate feeding preference and metabolism in Drosophila. Front. Physiol. 9, 1083 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Harvey K. F., Mattila J., Sofer A., Bennett F. C., Ramsey M. R., Ellisen L. W., Puig O., Hariharan I. K., FOXO-regulated transcription restricts overgrowth of Tsc mutant organs. J. Cell Biol. 180, 691–696 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.DiAngelo J. R., Birnbaum M. J., Regulation of fat cell mass by insulin in Drosophila melanogaster. Mol. Cell. Biol. 29, 6341–6352 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Bohni R., Riesgo-Escovar J., Oldham S., Brogiolo W., Stocker H., Andruss B. F., Beckingham K., Hafen E., Autonomous control of cell and organ size by CHICO, a Drosophila homolog of vertebrate IRS1-4. Cell 97, 865–875 (1999). [DOI] [PubMed] [Google Scholar]
- 76.Tatar M., Kopelman A., Epstein D., Tu M. P., Yin C. M., Garofalo R. S., A mutant Drosophila insulin receptor homolog that extends life-span and impairs neuroendocrine function. Science 292, 107–110 (2001). [DOI] [PubMed] [Google Scholar]
- 77.Honegger B., Galic M., Kohler K., Wittwer F., Brogiolo W., Hafen E., Stocker H., Imp-L2, a putative homolog of vertebrate IGF-binding protein 7, counteracts insulin signaling in Drosophila and is essential for starvation resistance. J. Biol. 7, 10 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Arquier N., Geminard C., Bourouis M., Jarretou G., Honegger B., Paix A., Leopold P., Drosophila ALS regulates growth and metabolism through functional interaction with insulin-like peptides. Cell Metab. 7, 333–338 (2008). [DOI] [PubMed] [Google Scholar]
- 79.Alic N., Hoddinott M. P., Vinti G., Partridge L., Lifespan extension by increased expression of the Drosophila homologue of the IGFBP7 tumour suppressor. Aging Cell 10, 137–147 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Wang B., Moya N., Niessen S., Hoover H., Mihaylova M. M., Shaw R. J., Yates J. R. III, Fischer W. H., Thomas J. B., Montminy M., A hormone-dependent module regulating energy balance. Cell 145, 596–606 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Choi S., Lim D. S., Chung J., Feeding and fasting signals converge on the LKB1-SIK3 pathway to regulate lipid metabolism in Drosophila. PLOS Genet. 11, e1005263 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Luong N., Davies C. R., Wessells R. J., Graham S. M., King M. T., Veech R., Bodmer R., Oldham S. M., Activated FOXO-mediated insulin resistance is blocked by reduction of TOR activity. Cell Metab. 4, 133–142 (2006). [DOI] [PubMed] [Google Scholar]
- 83.Slaidina M., Delanoue R., Gronke S., Partridge L., Leopold P., A Drosophila insulin-like peptide promotes growth during nonfeeding states. Dev. Cell 17, 874–884 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Barry W. E., Thummel C. S., The Drosophila HNF4 nuclear receptor promotes glucose-stimulated insulin secretion and mitochondrial function in adults. eLife 5, e11183 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Stern J. H., Rutkowski J. M., Scherer P. E., Adiponectin, leptin, and fatty acids in the maintenance of metabolic homeostasis through adipose tissue crosstalk. Cell Metab. 23, 770–784 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Delanoue R., Meschi E., Agrawal N., Mauri A., Tsatskis Y., McNeill H., Leopold P., Drosophila insulin release is triggered by adipose Stunted ligand to brain Methuselah receptor. Science 353, 1553–1556 (2016). [DOI] [PubMed] [Google Scholar]
- 87.Koyama T., Mirth C. K., Growth-blocking peptides as nutrition-sensitive signals for insulin secretion and body size regulation. PLOS Biol. 14, e1002392 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Meschi E., Léopold P., Delanoue R., An EGF-responsive neural circuit couples insulin secretion with nutrition in Drosophila. Dev. Cell 48, 76–86.e5 (2019). [DOI] [PubMed] [Google Scholar]
- 89.Agrawal N., Delanoue R., Mauri A., Basco D., Pasco M., Thorens B., Leopold P., The Drosophila TNF eiger is an adipokine that acts on insulin-producing cells to mediate nutrient response. Cell Metab. 23, 675–684 (2016). [DOI] [PubMed] [Google Scholar]
- 90.Ghosh A. C., O’Connor M. B., Systemic activin signaling independently regulates sugar homeostasis, cellular metabolism, and pH balance in Drosophila melanogaster. Proc. Natl. Acad. Sci. U.S.A. 111, 5729–5734 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Kwon Y., Song W., Droujinine I. A., Hu Y., Asara J. M., Perrimon N., Systemic organ wasting induced by localized expression of the secreted insulin/IGF antagonist ImpL2. Dev. Cell 33, 36–46 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Figueroa-Clarevega A., Bilder D., Malignant Drosophila tumors interrupt insulin signaling to induce cachexia-like wasting. Dev. Cell 33, 47–55 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Okamoto N., Nakamori R., Murai T., Yamauchi Y., Masuda A., Nishimura T., A secreted decoy of InR antagonizes insulin/IGF signaling to restrict body growth in Drosophila. Genes Dev. 27, 87–97 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Pasco M. Y., Leopold P., High sugar-induced insulin resistance in Drosophila relies on the lipocalin Neural Lazarillo. PLOS ONE 7, e36583 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Teleman A. A., Maitra S., Cohen S. M., Drosophila lacking microRNA miR-278 are defective in energy homeostasis. Genes Dev. 20, 417–422 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Galikova M., Diesner M., Klepsatel P., Hehlert P., Xu Y., Bickmeyer I., Predel R., Kuhnlein R. P., Energy homeostasis control in Drosophila adipokinetic hormone mutants. Genetics 201, 665–683 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Isabel G., Martin J. R., Chidami S., Veenstra J. A., Rosay P., AKH-producing neuroendocrine cell ablation decreases trehalose and induces behavioral changes in Drosophila. Am. J. Physiol. Regul. Integr. Comp. Physiol. 288, R531–R538 (2005). [DOI] [PubMed] [Google Scholar]
- 98.Bharucha K. N., Tarr P., Zipursky S. L., A glucagon-like endocrine pathway in Drosophila modulates both lipid and carbohydrate homeostasis. J. Exp. Biol. 211, 3103–3110 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Gronke S., Muller G., Hirsch J., Fellert S., Andreou A., Haase T., Jackle H., Kuhnlein R. P., Dual lipolytic control of body fat storage and mobilization in Drosophila. PLOS Biol. 5, e137 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Braco J. T., Gillespie E. L., Alberto G. E., Brenman J. E., Johnson E. C., Energy-dependent modulation of glucagon-like signaling in Drosophila via the AMP-activated protein kinase. Genetics 192, 457–466 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Gylfe E., Glucose control of glucagon secretion-‘There’s a brand-new gimmick every year’. Ups. J. Med. Sci. 121, 120–132 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Post S., Liao S., Yamamoto R., Veenstra J. A., Nassel D. R., Tatar M., Drosophila insulin-like peptide dilp1 increases lifespan and glucagon-like Akh expression epistatic to dilp2. Aging Cell 18, e12863 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Scopelliti A., Bauer C., Yu Y., Zhang T., Kruspig B., Murphy D. J., Vidal M., Maddocks O. D. K., Cordero J. B., A neuronal relay mediates a nutrient responsive gut/fat body axis regulating energy homeostasis in adult Drosophila. Cell Metab. 29, 269–284.e10 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Waterson M. J., Chung B. Y., Harvanek Z. M., Ostojic I., Alcedo J., Pletcher S. D., Water sensor ppk28 modulates Drosophila lifespan and physiology through AKH signaling. Proc. Natl. Acad. Sci. U.S.A. 111, 8137–8142 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Jourjine N., Mullaney B. C., Mann K., Scott K., Coupled sensing of hunger and thirst signals balances sugar and water consumption. Cell 166, 855–866 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Havula E., Teesalu M., Hyotylainen T., Seppala H., Hasygar K., Auvinen P., Oresic M., Sandmann T., Hietakangas V., Mondo/ChREBP-Mlx-regulated transcriptional network is essential for dietary sugar tolerance in Drosophila. PLOS Genet. 9, e1003438 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Iizuka K., Bruick R. K., Liang G., Horton J. D., Uyeda K., Deficiency of carbohydrate response element-binding protein (ChREBP) reduces lipogenesis as well as glycolysis. Proc. Natl. Acad. Sci. U.S.A. 101, 7281–7286 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Bartok O., Teesalu M., Ashwall-Fluss R., Pandey V., Hanan M., Rovenko B. M., Poukkula M., Havula E., Moussaieff A., Vodala S., Nahmias Y., Kadener S., Hietakangas V., The transcription factor Cabut coordinates energy metabolism and the circadian clock in response to sugar sensing. EMBO J. 34, 1538–1553 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Hirabayashi S., Cagan R. L., Salt-inducible kinases mediate nutrient-sensing to link dietary sugar and tumorigenesis in Drosophila. eLife 4, e08501 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Teesalu M., Rovenko B. M., Hietakangas V., Salt-inducible kinase 3 provides sugar tolerance by regulating NADPH/NADP+ redox balance. Curr. Biol. 27, 458–464 (2017). [DOI] [PubMed] [Google Scholar]
- 111.Li M. V., Chang B., Imamura M., Poungvarin N., Chan L., Glucose-dependent transcriptional regulation by an evolutionarily conserved glucose-sensing module. Diabetes 55, 1179–1189 (2006). [DOI] [PubMed] [Google Scholar]
- 112.Havula E., Hietakangas V., Glucose sensing by ChREBP/MondoA-Mlx transcription factors. Semin. Cell Dev. Biol. 23, 640–647 (2012). [DOI] [PubMed] [Google Scholar]
- 113.Varghese J., Lim S. F., Cohen S. M., Drosophila miR-14 regulates insulin production and metabolism through its target, sugarbabe. Genes Dev. 24, 2748–2753 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Upadhyay A., Moss-Taylor L., Kim M. J., Ghosh A. C., O’Connor M. B., Erratum: TGF-β family signaling in Drosophila. Cold Spring Harb. Perspect. Biol. 9, a031963 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Watanabe K., Kanaoka Y., Mizutani S., Uchiyama H., Yajima S., Watada M., Uemura T., Hattori Y., Interspecies comparative analyses reveal distinct carbohydrate-responsive systems among Drosophila species. Cell Rep. 28, 2594–2607.e7 (2019). [DOI] [PubMed] [Google Scholar]
- 116.Song W., Owusu-Ansah E., Hu Y., Cheng D., Ni X., Zirin J., Perrimon N., Activin signaling mediates muscle-to-adipose communication in a mitochondria dysfunction-associated obesity model. Proc. Natl. Acad. Sci. U.S.A. 114, 8596–8601 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Ballard S. L., Jarolimova J., Wharton K. A., Gbb/BMP signaling is required to maintain energy homeostasis in Drosophila. Dev. Biol. 337, 375–385 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Bai H., Kang P., Hernandez A. M., Tatar M., Activin signaling targeted by insulin/dFOXO regulates aging and muscle proteostasis in Drosophila. PLOS Genet. 9, e1003941 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Song W., Ghosh A. C., Cheng D., Perrimon N., Endocrine regulation of energy balance by Drosophila TGF-β/activins. Bioessays 40, e1800044 (2018). [DOI] [PubMed] [Google Scholar]
- 120.Palanker L., Tennessen J. M., Lam G., Thummel C. S., Drosophila HNF4 regulates lipid mobilization and beta-oxidation. Cell Metab. 9, 228–239 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Palu R. A., Thummel C. S., Sir2 acts through hepatocyte nuclear factor 4 to maintain insulin signaling and metabolic homeostasis in Drosophila. PLOS Genet. 12, e1005978 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Reis T., Van Gilst M. R., Hariharan I. K., A buoyancy-based screen of Drosophila larvae for fat-storage mutants reveals a role for Sir2 in coupling fat storage to nutrient availability. PLOS Genet. 6, e1001206 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Banerjee K. K., Ayyub C., Sengupta S., Kolthur-Seetharam U., dSir2 deficiency in the fatbody, but not muscles, affects systemic insulin signaling, fat mobilization and starvation survival in flies. Aging 4, 206–223 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Reiter L. T., Potocki L., Chien S., Gribskov M., Bier E., A systematic analysis of human disease-associated gene sequences in Drosophila melanogaster. Genome Res. 11, 1114–1125 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Birse R. T., Choi J., Reardon K., Rodriguez J., Graham S., Diop S., Ocorr K., Bodmer R., Oldham S., High-fat-diet-induced obesity and heart dysfunction are regulated by the TOR pathway in Drosophila. Cell Metab. 12, 533–544 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Woodcock K. J., Kierdorf K., Pouchelon C. A., Vivancos V., Dionne M. S., Geissmann F., Macrophage-derived upd3 cytokine causes impaired glucose homeostasis and reduced lifespan in Drosophila fed a lipid-rich diet. Immunity 42, 133–144 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Hong S. H., Kang M., Lee K.-S., Yu K., High fat diet-induced TGF-β/Gbb signaling provokes insulin resistance through the tribbles expression. Sci. Rep. 6, 30265 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Puigserver P., Spiegelman B. M., Peroxisome proliferator-activated receptor-γ coactivator 1α (PGC-1α): Transcriptional coactivator and metabolic regulator. Endocr. Rev. 24, 78–90 (2003). [DOI] [PubMed] [Google Scholar]
- 129.Rera M., Bahadorani S., Cho J., Koehler C. L., Ulgherait M., Hur J. H., Ansari W. S., Lo T. Jr., Jones D. L., Walker D. W., Modulation of longevity and tissue homeostasis by the Drosophila PGC-1 homolog. Cell Metab. 14, 623–634 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Tinkerhess M. J., Healy L., Morgan M., Sujkowski A., Matthys E., Zheng L., Wessells R. J., The Drosophila PGC-1α homolog spargel modulates the physiological effects of endurance exercise. PLOS ONE 7, e31633 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Dumesic P. A., Egan D. F., Gut P., Tran M. T., Parisi A., Chatterjee N., Jedrychowski M., Paschini M., Kazak L., Wilensky S. E., Dou F., Bogoslavski D., Cartier J. A., Perrimon N., Kajimura S., Parikh S. M., Spiegelman B. M., An evolutionarily conserved uORF regulates PGC1α and oxidative metabolism in mice, flies, and bluefin tuna. Cell Metab. 30, 190–200.e6 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Tiefenböck S. K., Baltzer C., Egli N. A., Frei C., The Drosophila PGC-1 homologue Spargel coordinates mitochondrial activity to insulin signalling. EMBO J. 29, 171–183 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Diop S. B., Bisharat-Kernizan J., Birse R. T., Oldham S., Ocorr K., Bodmer R., PGC-1/spargel counteracts high-fat-diet-induced obesity and cardiac lipotoxicity downstream of TOR and brummer ATGL lipase. Cell Rep. 10, 1572–1584 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Hader T., Muller S., Aguilera M., Eulenberg K. G., Steuernagel A., Ciossek T., Kuhnlein R. P., Lemaire L., Fritsch R., Dohrmann C., Vetter I. R., Jackle H., Doane W. W., Bronner G., Control of triglyceride storage by a WD40/TPR-domain protein. EMBO Rep. 4, 511–516 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Groh B. S., Yan F., Smith M. D., Yu Y., Chen X., Xiong Y., The antiobesity factor WDTC1 suppresses adipogenesis via the CRL4WDTC1 E3 ligase. EMBO Rep. 17, 638–647 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Baumbach J., Hummel P., Bickmeyer I., Kowalczyk K. M., Frank M., Knorr K., Hildebrandt A., Riedel D., Jackle H., Kuhnlein R. P., A Drosophila in vivo screen identifies store-operated calcium entry as a key regulator of adiposity. Cell Metab. 19, 331–343 (2014). [DOI] [PubMed] [Google Scholar]
- 137.Gronke S., Mildner A., Fellert S., Tennagels N., Petry S., Muller G., Jackle H., Kuhnlein R. P., Brummer lipase is an evolutionary conserved fat storage regulator in Drosophila. Cell Metab. 1, 323–330 (2005). [DOI] [PubMed] [Google Scholar]
- 138.Pospisilik J. A., Schramek D., Schnidar H., Cronin S. J., Nehme N. T., Zhang X., Knauf C., Cani P. D., Aumayr K., Todoric J., Bayer M., Haschemi A., Puviindran V., Tar K., Orthofer M., Neely G. G., Dietzl G., Manoukian A., Funovics M., Prager G., Wagner O., Ferrandon D., Aberger F., Hui C.-c., Esterbauer H., Penninger J. M., Drosophila genome-wide obesity screen reveals hedgehog as a determinant of brown versus white adipose cell fate. Cell 140, 148–160 (2010). [DOI] [PubMed] [Google Scholar]
- 139.Lee J. H., Bassel-Duby R., Olson E. N., Heart- and muscle-derived signaling system dependent on MED13 and Wingless controls obesity in Drosophila. Proc. Natl. Acad. Sci. U.S.A. 111, 9491–9496 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Bal N. C., Maurya S. K., Sopariwala D. H., Sahoo S. K., Gupta S. C., Shaikh S. A., Pant M., Rowland L. A., Bombardier E., Goonasekera S. A., Tupling A. R., Molkentin J. D., Periasamy M., Sarcolipin is a newly identified regulator of muscle-based thermogenesis in mammals. Nat. Med. 18, 1575–1579 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Moraru A., Cakan-Akdogan G., Strassburger K., Males M., Mueller S., Jabs M., Muelleder M., Frejno M., Braeckman B. P., Ralser M., Teleman A. A., THADA regulates the organismal balance between energy storage and heat production. Dev. Cell 41, 72–81.e6 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Chatterjee N., Perrimon N., Thermogenesis by THADA. Dev. Cell 41, 1–2 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Lowell B. B., Spiegelman B. M., Towards a molecular understanding of adaptive thermogenesis. Nature 404, 652–660 (2000). [DOI] [PubMed] [Google Scholar]
- 144.Da-Re C., De Pitta C., Zordan M. A., Teza G., Nestola F., Zeviani M., Costa R., Bernardi P., UCP4C mediates uncoupled respiration in larvae of Drosophila melanogaster. EMBO Rep. 15, 586–591 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Ulgherait M., Chen A., McAllister S. F., Kim H. X., Delventhal R., Wayne C. R., Garcia C. J., Recinos Y., Oliva M., Canman J. C., Picard M., Owusu-Ansah E., Shirasu-Hiza M., Circadian regulation of mitochondrial uncoupling and lifespan. Nat. Commun. 11, 1927 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Do Carmo S., Fournier D., Mounier C., Rassart E., Human apolipoprotein D overexpression in transgenic mice induces insulin resistance and alters lipid metabolism. Am. J. Physiol. Endocrinol. Metab. 296, E802–E811 (2009). [DOI] [PubMed] [Google Scholar]
- 147.Na J., Musselman L. P., Pendse J., Baranski T. J., Bodmer R., Ocorr K., Cagan R., A Drosophila model of high sugar diet-induced cardiomyopathy. PLOS Genet. 9, e1003175 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Rovenko B. M., Perkhulyn N. V., Gospodaryov D. V., Sanz A., Lushchak O. V., Lushchak V. I., High consumption of fructose rather than glucose promotes a diet-induced obese phenotype in Drosophila melanogaster. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 180, 75–85 (2015). [DOI] [PubMed] [Google Scholar]
- 149.Alfa R. W., Kim S. K., Using Drosophila to discover mechanisms underlying type 2 diabetes. Dis. Model. Mech. 9, 365–376 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Haselton A., Sharmin E., Schrader J., Sah M., Poon P., Fridell Y. W., Partial ablation of adult Drosophila insulin-producing neurons modulates glucose homeostasis and extends life span without insulin resistance. Cell Cycle 9, 3063–3071 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Wong A. C., Vanhove A. S., Watnick P. I., The interplay between intestinal bacteria and host metabolism in health and disease: Lessons from Drosophila melanogaster. Dis. Model. Mech. 9, 271–281 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Shin S. C., Kim S. H., You H., Kim B., Kim A. C., Lee K. A., Yoon J. H., Ryu J. H., Lee W. J., Drosophila microbiome modulates host developmental and metabolic homeostasis via insulin signaling. Science 334, 670–674 (2011). [DOI] [PubMed] [Google Scholar]
- 153.Hang S., Purdy A. E., Robins W. P., Wang Z., Mandal M., Chang S., Mekalanos J. J., Watnick P. I., The acetate switch of an intestinal pathogen disrupts host insulin signaling and lipid metabolism. Cell Host Microbe 16, 592–604 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Whon T. W., Shin N. R., Jung M. J., Hyun D. W., Kim H. S., Kim P. S., Bae J. W., Conditionally pathogenic gut microbes promote larval growth by increasing redox-dependent fat storage in high-sugar diet-fed Drosophila. Antioxid. Redox Signal. 27, 1361–1380 (2017). [DOI] [PubMed] [Google Scholar]
- 155.Wong C. N., Ng P., Douglas A. E., Low-diversity bacterial community in the gut of the fruitfly Drosophila melanogaster. Environ. Microbiol. 13, 1889–1900 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Beebe K., Robins M. M., Hernandez E. J., Lam G., Horner M. A., Thummel C. S., Drosophila estrogen-related receptor directs a transcriptional switch that supports adult glycolysis and lipogenesis. Genes Dev. 34, 701–714 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Deblois G., Giguere V., Oestrogen-related receptors in breast cancer: Control of cellular metabolism and beyond. Nat. Rev. Cancer 13, 27–36 (2013). [DOI] [PubMed] [Google Scholar]
- 158.Walesky C., Apte U., Role of hepatocyte nuclear factor 4α (HNF4α) in cell proliferation and cancer. Gene Expr. 16, 101–108 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Yadav H., Quijano C., Kamaraju A. K., Gavrilova O., Malek R., Chen W., Zerfas P., Zhigang D., Wright E. C., Stuelten C., Sun P., Lonning S., Skarulis M., Sumner A. E., Finkel T., Rane S. G., Protection from obesity and diabetes by blockade of TGF-β/Smad3 signaling. Cell Metab. 14, 67–79 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Modica S., Straub L. G., Balaz M., Sun W., Varga L., Stefanicka P., Profant M., Simon E., Neubauer H., Ukropcova B., Ukropec J., Wolfrum C., Bmp4 promotes a brown to white-like adipocyte shift. Cell Rep. 16, 2243–2258 (2016). [DOI] [PubMed] [Google Scholar]


