Abstract
Introduction: Following vaccination of Israeli population with Pfizer-BioNTech COVID-19 Vaccine, an unusual increase in axillary-lymphadenopathy was noted. This study assesses the rate and magnitude of this trend from breast-imaging standpoint.
Materials and Methods: Participants undergoing breast-imaging, in whom isolated axillary-lymphadenopathy was detected were questioned regarding SARS-CoV-2 vaccine to the ipsilateral arm. Patients’ and imaging characteristics were statistically compared. In order to perform a very short-term follow-up, twelve healthy vaccinated medical staff-members, underwent axillary-ultrasound shortly after the second dose, and follow-up.
Results: Axillary-lymphadenopathy attributed to vaccination was found in 163 women undergoing breast-imaging, including BRCA-carriers. During the study, number of detected lymphadenopathies increased by 394% (p = 0.00001) in comparison with previous 2 consecutive years. Mean cortical-thickness of abnormal lymph-nodes after second dose vaccination was 5 ± 2 mm. Longer lymph-node diameter after second vaccination was noted (from 15 ± 5 mm, to 18 ± 6 mm, p = 0.005). In the subgroup of medical staff members, following trends were observed: in patients with positive antibodies, lymph-node cortical-thickness was larger than patients with negative serology (p = 0.03); lymph-node cortical-thickness decreased in 4-5 weeks follow-up (p = 0.007). Lymphadenopathy was evident on mammography in only 49% of cases.
Discussion: Vaccine-associated lymphadenopathy is an important phenomenon with great impact on breast-imaging clinic workload. Results suggest the appearance of cortical thickening shortly after both doses. Positive serology is associated with increased lymph-node cortical-thickness. In asymptomatic vaccinated women with ipsilateral axillary-lymphadenopathy as the only abnormal finding, radiological follow-up is probably not indicated. BRCA-carriers, although at higher risk for breast-cancer, should probably receive the same management as average-risk patients.
Key Words: COVID-19 vaccine, SARS-Cov-2, axillary lymphadenopathy, breast imaging, BRCA-carriers
INTRODUCTION
In December 2020, following the approval of the BNT162b2 COVID-19 vaccine (Pfizer-BioNTech COVID-19 Vaccine), a massive vaccination program of the Israeli population began. Within a few weeks, vaccination was offered to all Israelis over 16 years of age excluding those with prior SARS-Cov-2 infection and pregnant women. At the time of reporting, 54.8% of the total population have been fully vaccinated, and 9% tested positive for the virus (Israel`s Ministry of Health https://govextra.gov.il/ministry-of-health/corona/corona-virus/).
Our breast imaging center performs over 15,000 mammography exams and 3500 breast MRI's annually, the latter offered primarily to young women at high risk for developing breast cancer (e.g., BRCA1 BRCA2 mutation carriers).
In early January 2021, an unusual increase in the number of radiologically detected and clinically palpable unilateral axillary lymphadenopathy in women undergoing breast imaging at our breast imaging center was noted. This increase encompassed women who underwent breast imaging as part of the average-risk population screening, for surveillance after diagnosis of breast cancer, and for high-risk screening in BRCA1 BRCA2 mutation carriers. Most of the cases with lymphadenopathies were detected by ultrasound and MRI, with a part of them shown also by mammography.
Ipsilateral axillary lymphadenopathy has been previously reported shortly after smallpox, Bacille Calmette-Guerin (BCG), human papillomavirus HPV and H1N1 influenza A virus vaccines (1, 2, 3). Yet these vaccination campaigns have never been so massive and deployed on a nation-wide basis as the current SARS -CoV-2 vaccine campaign in Israel. Recently, several case reports on the occurrence of post-COVID 19 vaccination axillary lymphadenopathy were published (4, 5, 6, 7, 8). In February 2021, a scientific expert panel published recommendations regarding post vaccination lymphadenopathy for patients undergoing imaging that includes the axillae (9,10).
Herein, we performed an observational study to assess the rate and magnitude of post SARS-CoV-2 vaccination axillary lymphadenopathy on ultrasound and MRI, and the change in appearance and size of lymph nodes over time. Since our breast imaging unit serves the largest Israeli BRCA carrier surveillance clinic, information on lymphadenopathy in that high risk group was also collected. Furthermore, data on antibody levels against SARS CoV- 2 and comparison to the magnitude of the lymph node cortical thickening was performed.
MATERIAL AND METHODS
Patients
A retrospective observational study was carried out: the study population included all women who underwent breast imaging during the study period (between January 11, 2021 and February 04, 2021) at our breast imaging center, who presented newly detected unilateral axillary lymphadenopathy on imaging, and had a history of recent COVID-19 vaccination to the ipsilateral arm. In the regular anamnesis, if the radiologist observed axillary lymphadenopathy, the patients were asked if they were vaccinated for COVID-19 in the ipsilateral arm, in order to decide if the lymphadenopathy would be attributable to the vaccine or if further investigation was necessary. This was noted in the report, which was afterwards retrieved from the computerized hospital information system using the OpisoftCare software. The study population was composed of women undergoing routine average-risk screening for breast cancer (n = 67), women with past history of breast cancer undergoing annual imaging surveillance (n = 28), high-risk population undergoing screening for breast cancer due to family history of breast cancer (n = 5), women with newly-diagnosed breast cancer (n = 3), women with a clinically palpable mass in the axilla and lymphadenopathy detected on breast mammography or ultrasound (n = 4). Additionally, women who are carriers of pathogenic sequence variants in the BRCA1 or BRCA2 genes that underwent regular breast imaging surveillance between January 11, 2021 and March 31, 2021 were also included (n = 44). For each participant the data collected included age, relevant clinical data on patient's personal history of breast cancer (if diagnosed), vaccinated arm side, dates of first and second vaccination, postvaccination symptoms such as pain, fever, were recorded. In addition, serology dates and results and biopsy results were also collected, when these were performed and available on the patients’ medical files. Furthermore, in order to perform a very short term follow-up on the imaging course of the axillary lymphadenopathy, twelve healthy medical staff members who were vaccinated, underwent baseline axillary US shortly after the second dose (range: 1–7 days) and a follow-up axillary US (average 4–5 weeks, range: 3–6 weeks post vaccination).
Finally, in order to evaluate the vaccination-associated excessive incidence of cases of ipsilateral lymphadenopathy, a comparison was performed with the rate of cases showing unilateral lymphadenopathy detected during the same time frame in 2020 and 2019. Using the OpisoftCare software, we searched our institutional radiological information system on a prospectively maintained database for women who underwent US examination during the same period last year. Case-by-case confirmation was then performed to identify cases with documented unilateral lymphadenopathy.
Imaging Technique
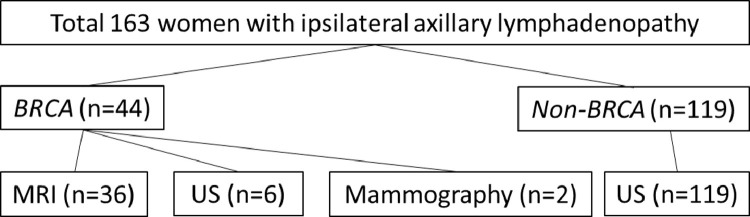
For the non-BRCA cohort, which accounts for the majority of participants 119/163 (73%), ultrasonography (US) was the imaging modality mostly applied to detect, qualify, and perform analysis of the axillary lymphadenopathy. For BRCA carriers’ cohort, MRI was the main imaging modality. The flowchart with the exact distribution of subgroups and imaging modalities is shown in Figure 1 .
Figure 1.
Flowchart showing patients subgroups distribution and imaging modalities.
Ultrasound: Whole breast and bilateral axillae US examinations were performed by certified fellowship-trained breast radiologists, using Siemens Acuson S2000 ultrasound system with a linear transducer 18-6Mhz, (Siemens Medical Solutions USA, Inc., Mountain View, CA, USA). Image parameters were as follows: frame rate, 60 frames/second; gain, 0 dB; dynamic range, 65 Db.
MRI: Scan was performed on 1.5-T MRI (Signa Excite HDX, GE Healthcare) using a dedicated double breast coil equipped with 8 channels. A standard dynamic contrast enhanced MRI protocol was via axial vibrant multiphase 3D DCE T1-weighted sequence with the following parameters: echo time (TE)/ repetition time (TR) of 2.6/5.4 ms, flip angle 15°, bandwidth 83.3 kHz, matrix 512 × 364; field of view (FOV) 340 mm and slice thickness of 2 mm. DCE was acquired prior to and 4 times after an automated injection of contrast agent bolus (0.1 ml/kg at 2 ml/sec Dotarem, (gadoterate meglumine, Guerbet)) followed by a 20-ml saline flush. Post-contrast images were acquired, with the first acquisition centered at 1:25 minutes after injection and the delayed acquisition centered at 7:35 minutes after injection, as was previously described (11).
In our breast imaging center, we screen average risk patients with mammography, and we add handheld radiologist performed ultrasound when the breasts are heterogeneously dense or extremely dense (BI-RADS Category C or D by BI-RADS 5th ed), or if there is a clinical or mammographic finding. High-risk patients (BRCA carriers or patients with patients with ≥20% lifetime risk of having breast cancer) will undergo MRI and mammography every year.
Image Analysis
Ultrasound imaging features of the axillary lymphadenopathy included: maximal dimension on long axis, and cortex width of the largest lymph node on the vaccinated arm side. In this regard, it should be mentioned though, that a lack of consensus exists for axillary node size assessment on ultrasound (12), while focal or diffuse thickening greater than 3 mm cortical thickness are considered proper parameters (13, 14, 15). Measurements were obtained by a single reader (RF, certified fellowship-trained breast radiologist, with 8 years of experience in breast imaging). The same measurements were also performed on MRI, and were obtained on the second post contrast T1-weighted image with fat-suppression.
For non-BRCA women, 81 mammograms and 107 screening ultrasounds were performed. For BRCA carriers, 44 MRIs were performed.
Serology
In 9 participants, serology results for SARS-CoV-2 antibodies were available, as part of a service offered by the hospital to the staff. Serology was measured in units, when results equal or higher than 1 unit were considered positive. Results equal or under 0.8 unit were considered negative. Results between 0.81 and 0.99 unit were considered borderline. This results were correlated with axillary lymphadenopathy.
Statistical Analysis
Comparison of the rate of unilateral lymphadenopathy detected during the same periods of the study with those reported in 2019 and 2020 was carried out using chi-square test. In addition, imaging measurements were compared between cases that were examined after the first and second dose (Student's paired 2-tailed t-test). Finally, intra-individual comparison between the staff members with lymphadenopathy that were examined twice, at baseline and follow-up US was performed (Student's paired 2-tailed t-test). All statistical tests were calculated using SPSS V27. P values were considered significant at p = 0.05.
Our institutional review board approved this retrospective study and waived the requirement for informed consent.
RESULTS
Overall, the study encompassed 107 participants (non-BRCA carriers) undergoing breast and axillary ultrasound, 12 medical staff members, and 44 BRCA carriers who underwent breast ultrasound, mammography or breast MRI, for a total of 163 participants. In all participants, post vaccination lymphadenopathy was not present on previous breast imaging of the same participant and was limited to the ipsilateral axilla of the vaccinated arm.
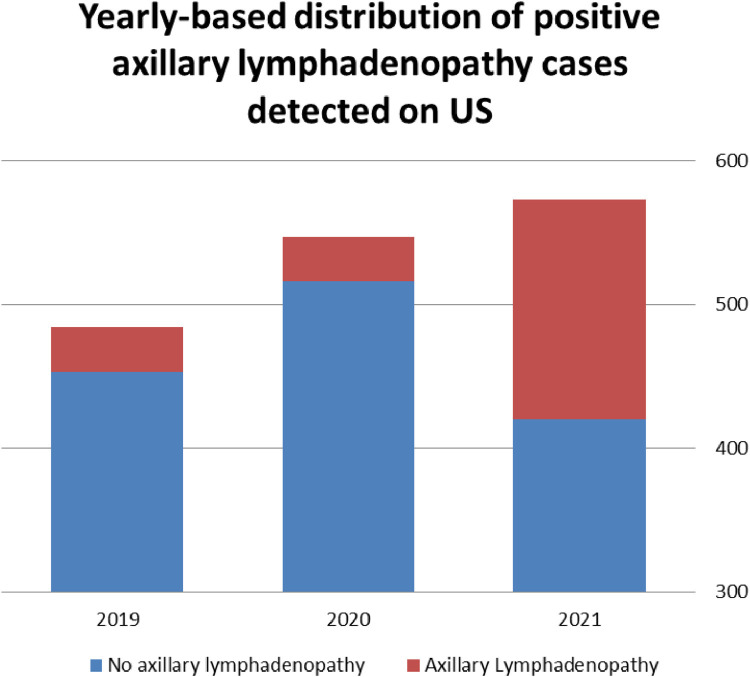
Comparing the rate of newly diagnosed ipsilateral lymphadenopathies of all causes detected by US in the current year, with those detected in the previous 2 consecutive years, during the same period of January 11, 2020 until February 04, 2020, and January 11, 2019 until February 04, 2019 the following results were noted: 2019 – 31/484 (6.4%); 2020 – 31/547 (5.7%) and the current study 153/573 (26.7%), an increase of 394% (p = 0.00001) (Fig 2 ). Vaccination associated lymphadenopathy was attributed to 77.8% of lymphadenopathy cases of all causes in the current period (119 lymphadenopaties were attributed to the vaccine, while 34 were due to other causes, including metastatic cancer and reactive lymphadenopaties due to other causes). Illustration of the rise in the lymphadenopathy cases as compared to previous years is presented in Figure 2.
Figure 2.
Rise in lymphadenopathy cases as compared to previous years. (Color version of figure is available online.)
Non-BRCA Carriers
Thirty participants had ultrasound after the first vaccine dose and 77 patients after the second vaccine dose, and 12 staff members after the second dose. The mean time in days of the ultrasound after the first vaccine dose (mean ± SD) was 9 ± 6 days (range 1–20 days), and after the second vaccine dose imaging occurred after 9 ± 8 days (range 1–38 days). Mean cortical thickening of the abnormal lymph nodes after the first and second dose of vaccination, in the group of non-BRCA carriers was (mean ± SD), respectively: 5 ± 1.5 mm, range 3-9 mm (after the first dose) and 5 ± 2 mm, range of 3-15 mm (after the second dose) (p = 0.70). Mean ± SD length of abnormal lymph nodes after the first vaccination dose was 15 ± 5 mm (range 7–30 mm), and increased to 18 ± 6 mm (range 9–48 mm) after the second vaccination dose (p = 0.005) Table 1.
Table 1.
Patients and Lymph Node Imaging Data as Demonstrated on Ultrasound (Excluding Medical Staff-Members)
| First Dose | Second Dose | ||
|---|---|---|---|
| Patients that had cortical thickening (n) | 30 | 77 | |
| Mean ± SD lymph node cortical thickness | 5 ± 1.5 mm | 5 ± 2 mm | p = 0.70 |
| Mean ± SD lymph node length | 15 ± 6 mm | 18 ± 6 mm | 0.005 |
| Previous history of breast cancer | 9 patients | 25 patients | |
In the subgroup of patients having breast imaging after the first dose, 17 underwent mammography, and ipsilateral lymphadenopathy was shown in 9 of them 9/17 (52%). In the subgroup of patients having breast imaging after the second dose, 64 underwent mammography, and ipsilateral lymphadenopathy was shown in 31 of them 31/64 (48%). Altogether, lymphadenopathy was seen in only 40/81 (49%) of ultrasound positive cases.
In the non-BRCA participant group, 33/107 (30%) had a personal history of breast cancer, of whom 8 were vaccinated in the ipsilateral arm to the side that breast cancer was diagnosed. In 5/8 of these cases, the imaging score was BI-RADS 2 with no recommendation for a repeat interval breast imaging, and mild lymphadenopathy was attributed to the vaccination (the assessment was BI-RADS 2 since breast imaging was normal and there was a known clinical event which explained ipsilateral mild lymphadenopathy – the vaccination); 2 cases (1 with a palpable axillary mass) were instructed to undergo a short term (2 month) repeated US, and another one was recommended to undergo biopsy since the cortex was very thickened up to 15 mm, with a benign pathological result. Both follow up ultrasounds demonstrated normal sonographic appearing lymph node with cortex width up to 0.2 mm.
BRCA Carriers
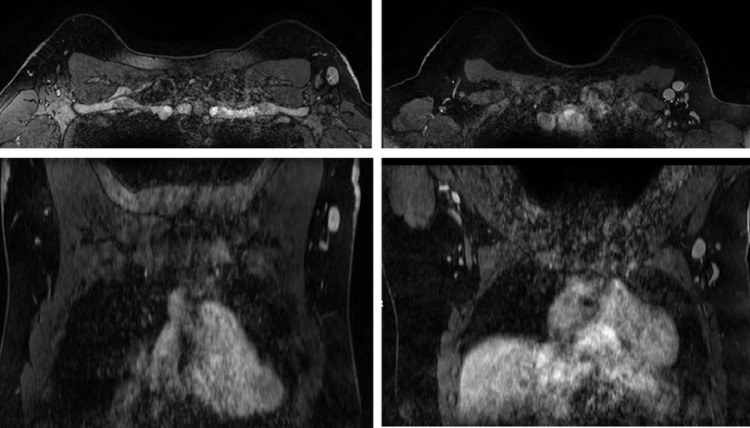
44 BRCA carriers with lymphadenopathy detected by breast imaging were included: 27 BRCA1, 16 BRCA2 carriers, and 1 patient that is carrier of both a BRCA1 and BRCA2 mutation. Six participants had breast ultrasound (1 due to palpable lymphadenopathy and 5 as part of the routine protocol surveillance scheme), 2 had a mammogram and 36 had a breast MRI. Figure 3 shows 2 examples of axillary lymphadenopathy on MRI. Mean ± SD cortical thickening was 6 ± 2 mm (range 3-13 mm) and mean lymph node length was 14 ± 4 mm (range 8-24 mm). All mild lymphadenopathy were also considered secondary to vaccination and these women also received a BI-RADS 2 categorization. Fourteen BRCA carriers had past history of breast cancer (32%), 3 of them bilateral, 6 in the contralateral vaccinated arm and 2 in the ipsilateral vaccinated arm. One BRCA carrier with past history of bilateral breast cancer had an ultrasound guided biopsy, in which pathology showed reactive lymph node. Both patients vaccinated in the ipsilateral arm to the previous breast cancer received a BI-RADS 3 categorization with recommendation of short term ultrasound follow up. One of the women with a personal history of left breast cancer that was vaccinated in the right arm had a new focus in the right breast – she received recommendation of biopsy of the focus, which was not seen in the day of the biopsy - this patient received a recommendation of sonographic short term follow up, that was performed 11 weeks after the second dose – which showed complete resolution of the lymphadenopathy, with a normal cortical thickness of 1 mm. Participants vaccinated in the contralateral arm and without history of breast cancer received a BI-RADS 2 with recommendation of continued routine protocol approved breast imaging surveillance).
Figure 3.
Examples of left axillary lymphadenopathy on breast MRI.
Biopsy of Ipsilateral Axillary Lymphadenopathy
In 3 patients with a newly diagnosed breast cancer and ipsilateral axillary lymphadenopathy, ultrasound guided core needle biopsy was performed (an example is shown in Fig 4). Additionally, 3 cancer free women with clinically suspicious lymphadenopathy (1 with family history, 1 with previous ipsilateral breast cancer and 1 with no history of breast cancer), and an additional BRCA carrier with past history of bilateral breast cancer also had an ultrasound guided biopsy. In all 7 participants pathology results showed benign reactive lymph node. A summary of outcomes for both groups is shown in Table 2 .
Figure 4.
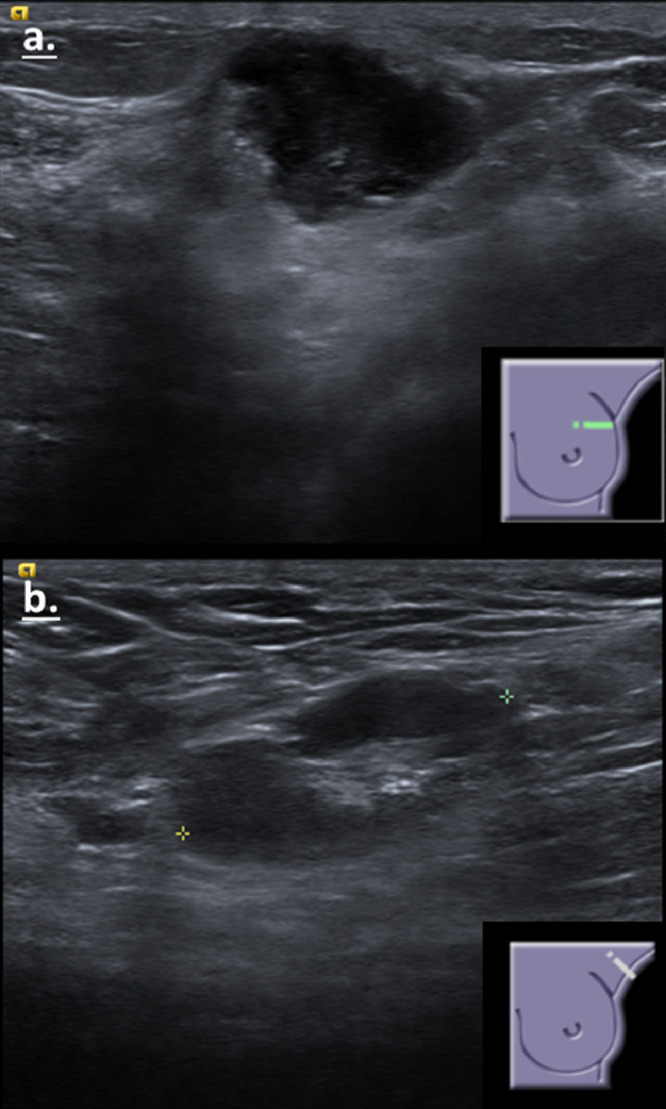
A 27 years old patient that had pain in the left arm and left breast after the first dose of the vaccine (in the ipsilateral arm), and felt a mass in the left breast. Ultrasound showed a suspicious mass in the upper outer quadrant of the left breast (A) and a pathological axillary lymph node (B). A biopsy was performed of both the mass and the axillary lymph node – on pathology, the mass was a triple negative invasive ductal breast cancer and the lymph node was reactive (considered to be reactive to the vaccine). (Color version of figure is available online.)
Table 2.
Summary of Outcomes in 107 Non-BRCA (excluding staff- members) and 44 BRCA Women
| Non-BRCA | BRCA | |
|---|---|---|
| BIRADS 2 | 99 | 40 |
| BI-RADS 3 | 2 | 3 |
| BIRADS 4 | 6 | 1 |
| Pathology | Reactive lymph node | Reactive lymph node |
| Outcome of BI-RADS 3 | Cortex up to 2 mm after 2-8 weeks | Cortex up to 2 mm after 6-8 weeks |
| Number of breast cancers detected | 3 | - |
Palpable Versus Subclinical Lymphadenopathy
Overall, in 159/163 (97.5%) participants lymphadenopathy was detected only by imaging, whereas in 4 cases a palpable lymphadenopathy was noted (all of them in the non-BRCA carriers’ group, 1 of them with a personal history of breast cancer in the contralateral breast). A short-term ultrasound follow-up after 6-12 weeks was recommended to all the patients with palpable lymphadenopathy. One already performed repeat US after 6 weeks and adenopathy had resolved.
Serology
In 9 participants, serology results for SARS-CoV-2 antibodies were available. In 7 participants, serology levels were positive (higher than 1 unit). All participants with positive serology presented with enlarged lymph nodes (mean ± SD lymph node length of 18 ± 5.2 mm, range 9-25 mm; mean ± SD cortical thickness 5 ± 2.5 mm, range 4–10 mm). Two additional participants who performed serology testing had negative serology (0.03 serology units), both had only slight cortical thickness of 3 mm which was statistically significant smaller compared to the size of the 7 serology positive participants (p = 0.03).
Short-Term Follow-Up
Short term ultrasonographic follow up in the medical staff cohort demonstrated that the lymphadenopathy started to recede within 4 weeks from the second dose, with a mean cortical thickness of 3 mm. Subsequent US follow up in 4 of these medical staff after 6 weeks showed continued reduction of the cortical thickness, being less than 3 mm (p = 0.007). Summary of the staff-members lymph node data is provided in table 3 .
Table 3.
Summary of Ultrasound Lymph Node Data and Follow Up in Medical Staff-Members
| After 2nd dose | Follow up 4-5 weeks | ||
|---|---|---|---|
| Mean ± SD cortical thickening | 5 ± 1.9 mm | 3 ± 1.2 mm | p = 0.007 |
| Mean ± SD lymph node length | 17 ± 5 mm | 16 ± 5 mm | p = 0.83 |
DISCUSSION
In the current study we show an almost 4-fold increase in axillary lymphadenopathy detected on breast imaging in 2021 compared to 2019 and 2020 in the same medical center, an increase attributed to the massive COVID-19 vaccination campaign in Israel. Vaccination with the Pfizer-BioNTech vaccine was associated with a mostly radiologically detected, clinically subtle, ipsilateral lymphadenopathy in women who underwent breast imaging for a variety of clinical reasons: average and high risk women and in cancer free and breast cancer affected women.
Moderna reported on higher rates of axillary lymphadenopathy compared to prior vaccines (16,17); In the Moderna clinical trials, axillary swelling or tenderness was reported in 11.6% of patients (5.0% placebo) after the first dose and 16.0% (4.3% placebo) after the second, while per Pfizer trials, reports of ipsilateral axillary and supraclavicular lymphadenopathy in the first month after the second dose were imbalanced, with more cases in the vaccine group versus the placebo group, on clinical examination (18, 19, 20). In our study, most of the adenopathy 159/163 (97.5%) was subclinical and mostly detected by imaging only. Previously vaccine associated lymphadenopathy was anecdotally reported. Mehta et al described 3 cases of patients coming for routine screening mammography and ultrasound, and 1 patient presented due to palpable lump in the ipsilateral vaccinated arm (4). Eifer and Eshet reported a patient who underwent lumpectomy for breast cancer in whom contralateral lymphadenopathy and FDG uptake were attributed to reactive post vaccination lymphadenopathy (5). Özütemiz reported 5 cases of oncology patients with axillary lymphadenopathy that mimicked metastasis (7).
The size of enlarged lymph nodes was roughly but statistically significant correlated with vaccination schedule: 15 mm after the first dose and 18 mm after second vaccination dose (p = 0.005), and mean cortical thickness was abnormal and unchanged over time, measuring 5 mm. The reassuring feature of this radiological phenomenon is that it is self-regressing as shown by the limited follow up in the present study.
Despite the limited number of cases where serology data were available, it seems that minimal cortical thickening is associated with negative serology for SARS-CoV-2 antibodies. It seems plausible that the absence of sizable adenopathy in individuals with a limited measurable antibody levels could be due to a low local immune response and could be related to the absence of vaccine induced antibody formation. This possibility needs to be tested in larger future studies.
The study limitations include a limited number of cases in a single medical center in Israel, and the selection bias that may have resulted. Furthermore since not all vaccinated women underwent axillary sonography, the precise rate of this subclinical phenomenon cannot be accurately determined. The strength of this study is the fact that it was done in a country, where approximately 90% of the adult population have been vaccinated (Israel Ministry of Health).
The clinical impaction of this study may be to defer breast imaging screening in asymptomatic individuals to 6 weeks after vaccination. In addition, it would be helpful to query all women undergoing breast imaging, if a vaccine were given, when and in which arm. In asymptomatic vaccinated women, subclinical lymphadenopathy as the only abnormal finding is in all likelihood secondary to vaccination, and radiological follow up is probably not indicated. Similarly, Keshavarz et al. have recently recommended in their review to rely on patient's clinical context and updated resources to prevent potential disease upstaging and change in therapy, regarding post vaccination axillary lymphadenopathy (21). Patients with current and past breast cancer should be vaccinated in the contralateral arm, as recommended by Becker et al (22). Patients with newly diagnosed breast cancer and ipsilateral lymphadenopathy should have a biopsy performed to exclude malignancy, even if they have a history of vaccination. BRCA carriers, although at a higher risk for breast cancer, should probably receive the same management as patients in average risk.
In conclusion, imaging detected axillary lymphadenopathy appears shortly after vaccination, with a mean cortical thickening of 5 mm, which starts to decrease after 4-5 weeks. In asymptomatic low risk women with normal breast exam, isolated axillary lymphadenopathy and a history of recent COVID-19 vaccination to the ipsilateral arm, a further investigation or follow up is most probably not warranted, though additional studies are required in order to confirm these preliminary observations.
Declarations of interest
None.
References
- 1.Shirone N, Shinkai T, Yamane T. Axillary lymph node accumulation on FDG-PET/CT after influenza vaccination. Ann Nucl Med. 2012 doi: 10.1007/s12149-011-0568-x. [DOI] [PubMed] [Google Scholar]
- 2.Studdiford J, Lamb K, Horvath K. Development of unilateral cervical and supraclavicular lymphadenopathy after human papilloma virus vaccination. Pharmacotherapy. 2008 doi: 10.1592/phco.28.9.1194. [DOI] [PubMed] [Google Scholar]
- 3.Barouni AS, Augusto C, Queiroz MVNP. BCG lymphadenopathy detected in a BCG-vaccinated infant. Brazilian J Med Biol Res. 2004 doi: 10.1590/S0100-879X2004000500011. [DOI] [PubMed] [Google Scholar]
- 4.Mehta N, Sales RM, Babagbemi K. Unilateral axillary Adenopathy in the setting of COVID-19 vaccine. Clin Imaging. 2021 doi: 10.1016/j.clinimag.2021.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Eifer M, Eshet Y. Imaging of COVID-19 Vaccination at FDG PET/CT. Radiology. 2021 doi: 10.1148/radiol.2020210030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bernstine H, Priss M, Anati T. Axillary lymph nodes hypermetabolism after BNT162b2 mRNA COVID-19 vaccination in cancer patients undergoing 18F-FDG PET/CT: A cohort study. Clin Nucl Med. 2021;00:1–6. doi: 10.1097/RLU.0000000000003648. [DOI] [PubMed] [Google Scholar]
- 7.Özütemiz C, Krystosek LA, Church AL. Lymphadenopathy in COVID-19 vaccine recipients: diagnostic dilemma in oncology patients. Radiology. 2021 doi: 10.1148/radiol.2021210275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hiller N, Goldberg SN, Cohen-Cymberknoh M. Lymphadenopathy associated with the COVID-19 vaccine. Cureus. 2021 doi: 10.7759/cureus.13524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lehman CD, D'Alessandro HA, Mendoza DP. Unilateral Lymphadenopathy After COVID-19 vaccination: a practical management plan for radiologists across specialties. J Am Coll Radiol. 2021 doi: 10.1016/j.jacr.2021.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Edmonds CE, Zuckerman SP, Conant EF. Management of unilateral axillary lymphadenopathy detected on breast mri in the era of coronavirus disease (COVID-19) vaccination. Am J Roentgenol. 2021 doi: 10.2214/ajr.21.25604. [DOI] [PubMed] [Google Scholar]
- 11.Nissan N, Allweis T, Menes T. Breast MRI during lactation: effects on tumor conspicuity using dynamic contrast-enhanced (DCE) in comparison with diffusion tensor imaging (DTI) parametric maps. Eur Radiol. 2020 doi: 10.1007/s00330-019-06435-x. [DOI] [PubMed] [Google Scholar]
- 12.Marino MA, Avendano D, Zapata P. Lymph node imaging in patients with primary breast cancer: concurrent diagnostic tools. Oncologist. 2020 doi: 10.1634/theoncologist.2019-0427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Choi YJ, Ko EY, Han BK. High-resolution ultrasonographic features of axillary lymph node metastasis in patients with breast cancer. Breast. 2009 doi: 10.1016/j.breast.2009.02.004. [DOI] [PubMed] [Google Scholar]
- 14.Abe H, Schmidt RA, Kulkarni K. Axillary lymph nodes suspicious for breast cancer metastasis: Sampling with US-guided 14-gauge core-needle biopsy - Clinical experience in 100 patients. Radiology. 2009 doi: 10.1148/radiol.2493071483. [DOI] [PubMed] [Google Scholar]
- 15.Bedi DG, Krishnamurthy R, Krishnamurthy S. Cortical morphologic features of axillary lymph nodes as a predictor of metastasis in breast cancer: In vitro sonographic study. Am J Roentgenol. 2008 doi: 10.2214/AJR.07.2460. [DOI] [PubMed] [Google Scholar]
- 16.Tu W, Gierada DS, Joe BN. COVID-19 Vaccination-related lymphadenopathy: what to be aware of. Radiol Imaging Cancer. 2021 doi: 10.1148/rycan.2021210038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Local Reactions Systemic reactions, adverse events, and serious adverse events: moderna covid-19 vaccine. Centers for Disease Control and Prevention. 2021 https://www.cdc.gov/vaccines/covid-19/info-by-product/moderna/reactogenicity.html January 16,Available from. [Google Scholar]
- 18.Mortazavi S. Coronavirus disease (COVID-19) vaccination associated axillary adenopathy: imaging findings and follow-up recommendations in 23 women. Am J Roentgenol. 2021 doi: 10.2214/ajr.21.25651. [DOI] [PubMed] [Google Scholar]
- 19.Local Reactions Systemic reactions, adverse events, and serious adverse events: Pfizer-BioNTech COVID-19 vaccine. Centers for Disease Control and Prevention. 2021 https://www.cdc.gov/vaccines/covid-19/info-by-product/pfizer/reactogenicity.html January 16Available from: [Google Scholar]
- 20.Polack FP, Thomas SJ, Kitchin N. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020 doi: 10.1056/nejmoa2034577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Keshavarz P, Yazdanpanah Fereshteh, Rafiee Faranak. Lymphadenopathy following COVID-19 vaccination : imaging findings review running title : lymphadenopathy following COVID-19 vaccination. Acad Radiol. 2021 doi: 10.1016/j.acra.2021.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Becker AS, Perez-Johnston R, Chikarmane SA. Multidisciplinary recommendations regarding post-vaccine adenopathy and radiologic imaging: radiology scientific expert panel. Radiology. 2021 doi: 10.1148/radiol.2021210436. [DOI] [PMC free article] [PubMed] [Google Scholar]