Abstract
Introduction
Pluripotent stem cells (PSCs) are promising tools for modern regenerative medicine applications because of their stemness properties, which include unlimited self-renewal and the ability to differentiate into all cell types in the body. Evidence suggests that a rare population of cells within a tumor, termed cancer stem cells (CSCs), exhibit stemness and phenotypic plasticity properties that are primarily responsible for resistance to chemotherapy, radiotherapy, metastasis, cancer development, and tumor relapse. Different therapeutic approaches that target CSCs have been developed for tumor eradication.
Results and Discussion
In this review, we first provide an overview of different viewpoints about the origin of CSCs. Particular attention has been paid to views believe that CSCs are probably appeared through dysregulation of very small embryonic-like stem cells (VSELs) which reside in various tissues as the main candidate for tissue-specific stem cells. The expression of pluripotency markers in these two types of cells can strengthen the validity of this theory. In this regard, we discuss the common properties of CSCs and PSCs, and highlight the potential of PSCs in cancer studies, therapeutic applications, as well as educating the immune system against CSCs.
Conclusion
In conclusion, the resemblance of CSCs to PSCs can provide an appropriate source of CSC-specific antigens through cultivation of PSCs which brings to light promising ideas for prophylactic and therapeutic cancer vaccine development.
Graphical Abstract
Keywords: Cancer stem cells, Embryonic stem cells, Pluripotency, Cancer vaccine
Introduction
Cancer is a major worldwide health issue and leading cause of death. It is estimated that the worldwide cancer rate may surpass 29 million cases annually with approximately 1 out of every 6 deaths in 2040 [1]. Despite outstanding progress in many therapeutic approaches, various factors such as the failure of current treatments, drug resistance [2], delayed diagnosis [3] and absence of a strong immune system in affected patients [4] have led to the lack of a definitive cancer treatment. Most importantly, metastasis post-treatment and recurrence of tumors after resection are the main causes of cancer death [5–7]. Therefore, therapeutic strategies, which can treat or prevent tumors from metastasis and recurrence could be an outstanding achievement in cancer therapy.
Currently, the role of cancer stem cells (CSCs) in the propagation of malignant tumors and resistance to therapy has become more accentuated. CSCs comprise a group of cells that can form transplantable tumors with the same cellular heterogeneity that exists in their tumor of origin [8]. Beside the ability both to self-renew and to differentiate into several heterogeneous cells with less potency, major disorders related to malignancy such as plasticity, drug resistance, epithelial to mesenchymal transition (EMT) and metastasis, and tumor recurrence are attributed to the existence of CSCs. Many attempts have been made to isolate and characterize CSCs to enable further diagnosis and treatment; however, the lack of specific markers for cellular recognition is a serious challenge in dealing with CSCs [9]. There are several questions that should be answered about CSCs and understanding the origin of these cells may help define a useful model for further studies.
Pluripotent stem cells (PSCs), with an indefinite self-renewal capacity and the potential to generate all cell types of the body, have many similarities with CSCs. PSCs are usually derived via direct cultivation of early embryos (embryonic stem cells [ESCs]) or from reprogramming of somatic cells (induced PSCs [iPSCs]) [10, 11]. These cells have been used as promising sources for cell based therapies in clinical trials of spinal cord injuries, diabetes and age-related macular degeneration (AMD) treatment (reviewed in [12]). However, PSC-based cancer therapy studies are mainly restricted to derivation of functional cancer fighting cells that include PSC-derived dendritic cells (DCs) [13], lymphocytes [14], and transplantable cells like hematopoietic stem cells (HSCs) [15]. The results of some studies have shown that exposure of different types of cancer cells to the PSC microenvironment could inhibit Nodal expression and suppress the malignant characteristics of these cells [16]. The importance of these PSC-based research priorities signifies a novel vaccination strategy against CSCs that possibly could induce long-term memory against a broad-spectrum of tumors [17].
In this review, we discuss the existing theories that concern the origins of cancer or, more specifically, those of CSCs, their resemblance to PSCs and application of PSCs against tumors as a novel cancer therapy approach.
Cancer Stem Cells (CSCs) as Stem Cell Malignancies: Origin of CSCs
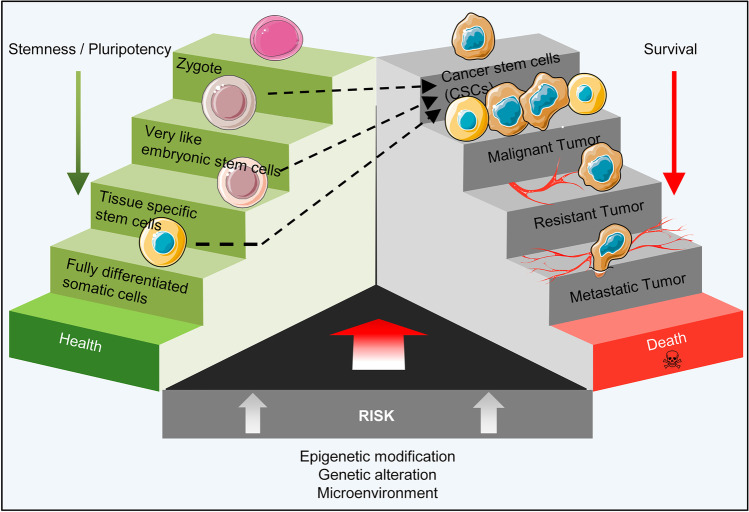
Growing evidence support the idea of representing cancer as a stem cell malignancy and the introduction of CSCs as being responsible for metastasis and tumor recurrence [18, 19]. Cell–cell fusion, horizontal gene transfer, genetic instability and the cell microenvironment are proposed mechanisms for these malignancies [20]. Accordingly, there are various assumptions about the origin of CSCs that include disrupted tissue-resident stem cells such as specific adult stem cells and their precursors, dedifferentiated somatic cells, and improperly activated residual embryonic cells in adult tissues.
Residual Embryonic Cells
The presence of cells with a semi-embryonic morphology in cancerous biopsies was initially reported in the nineteenth century. This finding ultimately led to the formation of a hypothesis entitled "embryonic rest of cancer origin" developed by Rudolf Virchow and his student Julius Cohnheim, which suggests that the remnants of embryonic cells persist in adult tissues and remain silent until adulthood [21]. In certain conditions, however, these cells can be activated by the influence of internal and external factors and they play a role in cancer development [22]. The close relation between embryonic and cancer cells was reported in cancers such as nephroblastomas that originate from germ cell tumors [23]. Researchers also identified some cells with embryonic morphology nominated as “very small embryonic-like stem cells” (VSELs). Possibly, during early embryogenesis when primordial germ cells (PGCs) originate from the proximal epiblast, a group of PGCs seats in other tissues where they remain silent. Therefore, it was hypothesized that migrating PGCs from the primary epiblast may be the origin of VSELs [24]. There is a hypothesis that places these cells at the top of the stem cell hierarchy in bone marrow, which results in the production of HSCs, mesenchymal stem cells (MSCs), endothelial progenitor cells, and production of tissue-committed stem cells (TCSCs) in other tissues (Fig. 1) [25]. Furthermore, it is suggested that VSELs are a type of migratory stem cells that are released from bone marrow into the bloodstream during stressful conditions and participate to regenerate damaged tissues [26]. These cells could be found in bone marrow, brain, pancreas, thymus and intestinal epithelium, and ovarian and testis surface epithelium in human and rodents [25, 27]. Improperly, fusion of normal cell with VSEL as a circulating stem cell provides the possibility of formation cancer initiating cell or CSCs in solid tumors [26]. VSELs are also isolated from human ESCs’ culture using stem-cell-related markers (CD133, SSEA-4) and germinal lineage markers (DDX4/VASA, PRDM14) [28]. These cells possess many characteristics of pluripotent cells; for example, a prototype of undifferentiated morphology with high chromatin content in the nucleus and high nucleus to cytoplasm ratio; presence of bivalent domains in chromatin; pluripotency marker expressions such as Oct4, NANOG, and SSEA1 (murine) or SSEA4 (humans); and the ability to form spheres and in vitro differentiation into derivatives of the three germ layers [29, 30]. Moreover, like ESCs, these cells have elevated telomerase activity and lack MHC class I expression on their surfaces [29, 31–33]. These also express germ cell markers such as Stella and Blimp1, as well as epiblast markers Gbx2, Fgf5 and Nodal [24].
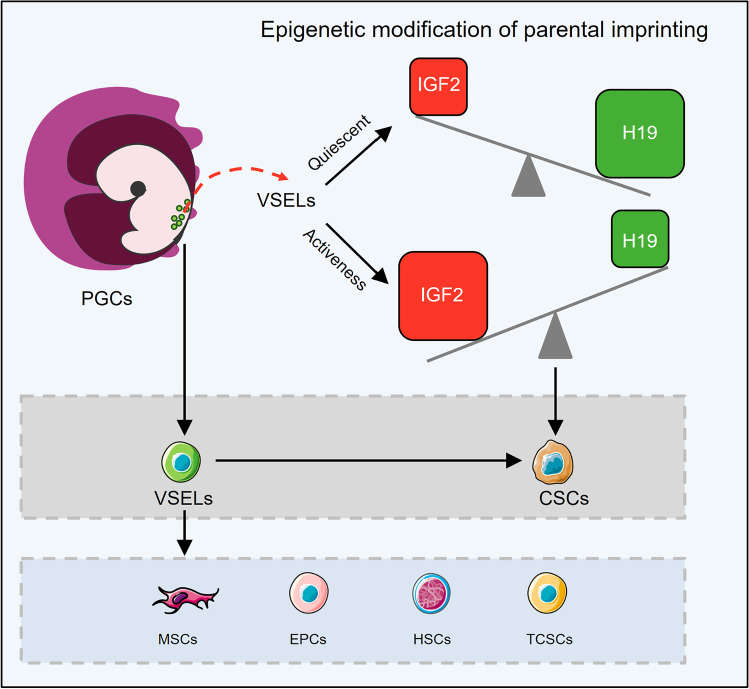
Fig. 1.
Very small embryonic-like stem cells (VSELs) support the embryonic rest theory. Primordial germ cells (PGCs) and VSELs are connected by a similar molecular signature. VSELs are present in variety of tissues and are thought to be a source of mesenchymal stem cells (MSCs), hematopoietic stem cell (HSCs), and endothelial progenitor cells (EPCs) in bone marrow as well as tissue-specific stem/progenitor cells in other tissues. Microenvironment condition, mutations, and epigenetic modifications can change the fate of VSELs into cancer initiating cells or cancer stem cells (CSCs). In the quiescent state, VSELs highly express growth-repressive imprinted gene transcripts such as H19 and show the downregulation of growth-promoting ones like IGF2. However, in activated VSELs, there is higher expression of IGF2 compared to H19, which ultimately leads to uncontrolled cell proliferation and can promote cancer formation. The cartoon in all figures of this article was created using Servier Medical Art templates (https://smart.servier.com) licensed under a Creative Commons License (https://creativecommons.org/licenses/by/3.0/)
To have a historical look at the development of VSELs, it should be referred to the mechanism of PGCs migration in the gastrulating embryo. At the onset of PGC migration, the complete erasure of imprinting marks from DNA of these cells results in downregulation of insulin-like growth factor 2 (IGF2) and upregulation of the tumor suppressor gene H19, which promote and inhibit growth, respectively [34]. Indeed, parental allele-specific expression of Igf2/H19 genes is controlled by differentially methylated regions (DMRs) and imprinting control regions (ICRs) which its regulation relies on DNA methylation. Normally, growth factor gene Igf2 is expressed from the paternal allele and tumor suppressor gene H19 is expressed from the maternal allele. These genes are neighboring genes, and both respond to the same enhancers located at H19's distal end. Briefly, the monoallelic expression of Igf2 is mediated by allele-specific DNA methylation at CTCF-binding sites in ICR, located immediately upstream of the H19 gene. Actually, CTCF (known as CCCTC-binding factor) mediates the insulation at the H19-Igf2 imprinted domain [35]. The methylated paternal ICR inhibits CTCF protein binding, which allows the Igf2 promoter to interact with the enhancers and be transcribed. Unmethylated maternal ICR binds to CTCF and prevents the Igf2 promoter from interacting with the enhancers. As a result, only H19 is transcribed on the maternal chromosome. However, in the late migratory PGCs, and similarly in developing VSELs, the complete erasure of imprinting marks from DNA results in down-regulation of Igf2 and up-regulation of H19 which led to the prevention of uncontrolled proliferation in these cells and explain the quiescent state of VSELs in adult tissues (Fig. 1) [34]. Improperly, if VSELs in adult testes affected by endocrine disruption and alteration of epigenetic modifications in imprinted genes, they may initiate self-renewal and promote testicular cancer [36]. Furthermore reduced sperm count and infertility can occur as a result of increased VSEL proliferation and impaired differentiation [37]. According to Ratajczak and his colleagues, VSELs can convert to CSCs and promote the formation of teratomas and teratocarcinomas, germinal tumors, or pediatric sarcomas if they achieve relevant mutations [26]. The presence of VSELs in ovarian teratoma and testicular cancer suggests these cells as tumor-initiating stem cells. In these tumors, VSELs express both pluripotency and germinal lineage markers and suggests to play important role in tumor invasion through the activation of EMT [38]. On the other hand, like VSELs, the expression of cancer testis antigens (CTAs; for example: MageB3, Ssbx2, and BORIS) and epiblast/germline-related cells are seen in some tumors [26, 38]. It's worth noting that identifying these small putative NANOG-, SOX2-, and SSEA-4-positive cells that are responsible for tumor formation in ovarian cancer is crucial from a clinical point of view as the potential therapeutic targets [39].
Tissue-specific Stem/Progenitor Cells
Tissue-specific stem/progenitor cells, also known as adult stem cells, are a rare cell population that have self-renewal and multi-lineage differentiation abilities. These cells are present in various tissues such as the intestines [40], breasts [41], lungs [42], ovaries [43] and prostate [44] and they function to repair or maintain these tissues. In a “classical model”, it has been assumed that the asymmetric mitotic division invariably creates one new stem cell and one daughter cell in each niche space of the adult tissues. However, in a “neutral competition model” it is supposed that each of the daughter cells, and even fully differentiated cells, can re-enter the niche and dedifferentiate to replace lost stem cells. The evidence to support this claim is that, with some rare exceptions such as muscle satellite cells or hair-follicle stem cells, the finding of such tissue-specific stem cells in most tissues of the body is almost impossible [45].
On the other hand, tissue-specific stem/progenitor cells are also considered as a primary suspect of cancer onset. However, definitions of these cells described in some studies, for example the assuming of a quiescent state of these cells during the lifetime of their host [46] and their activation under adverse conditions such as tissue damage that may lead to the conversion of these cells to CSCs is more in line with the definition that applies to the VSELs.
It is generally believed that the proliferation of tissue-specific stem/progenitor cells is precisely under control while they also are prompt enough to take action in confronting any awakening signal [47]. Furthermore, these cells can maintain their homeostasis and survival during differentiation by controlling the metabolic process of autophagy [48]. By receiving different signals from their surrounding cells and the microenvironment, tissue-specific stem/progenitor cells can quit quiescence and regain self-renewal, proliferation, and the ability to differentiate into different cell types, which enables them to maintain an effective role in regeneration [49]. However, it cannot be ignored the accumulation of genetic and epigenetic mutations that specifically be caused by aging may exert adverse effects on the accuracy of self-renewal and cell survival pathways of these cells [50–52]. Moreover, it is also possible that issue-specific stem/progenitor cells may turn into cancer initiating cells by fusion with abnormal cells or by absorbing damaging cellular material from cells trapped by phagocytosis and cause alterations in their normal mechanisms [53]. It is likely that processes such as autophagy can apply changes to the stem cell cellular microenvironment, homeostasis, and maintenance, and affect adult stem cell survival by inducing apoptosis or a cancerous state (Fig. 2) [54–57].
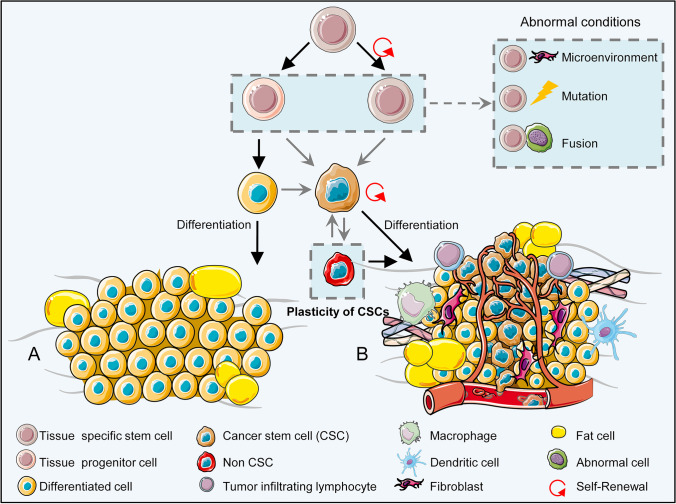
Fig. 2.
Schematic illustration of transformation of specific tissue stem cells into cancer stem cells (CSCs). A) Tissue-specific stem cells normally program into precursor cells that can differentiate into specified tissue cells. B) Cancer-derived external (i.e., microenvironment) or internal (genetic or epigenetic) factors or fusion with an abnormal cell can cause tissue specific stem cells or their precursor derivatives to revert into a cancerous state. Immune cells are suppressed by cancer cells in the affected area. Other normal resident cells, such as fibroblasts, may change in favor of the tumor and create a new microenvironment
On the other hand, CSCs can generate progenitors that have self-renewal ability and less stemness capability, which is similar to multipotent tissue-specific stem/progenitor cells [46, 58]. In addition, signaling networks, like the Wnt and Notch pathways, are common between CSCs and multipotent tissue-specific stem/progenitor cells [59, 60], and signal transduction occurs through some of the same surface receptors such as CD24 [61], CD44 [62], CD133 [63, 64], and CD49f [65, 66]. This data suggests a close relationship between these two cell types [42, 44, 67].
Stem/Progenitor Cells Produced by Reprogramming
In 2006, Takahashi and Yamanaka established a major breakthrough with the generation of iPSCs by reprogramming differentiated somatic cells into the pluripotent state by induction of four transcription factors (TFs)—Oct4 (Pou5f1), Sox2, c-Myc, and KLF4 [11, 68]. Since most of these TFs are oncogenes [69], genetic and epigenetic alterations in differentiated somatic cells have been hypothesized to activate dedifferentiation processes in adult cells to convert them to CSCs [70]. In this scenario, the expression of differentiation-specific genes in somatic cells is gradually downregulated and upregulation of stemness-related genes occurs, which leads to the establishment of a pluripotency-like state. This transformation has been observed in basal-like human mammary epithelial cells [71] and differentiated airway epithelial cells [72]. Similarly, in intestinal tumors, activation of NF-κB in response to inflammatory pathways leads to activation of Wnt signaling and consequently results in reprogramming of non-stem cell intestinal epithelial cells (IEC) into a pluripotent state [73]. Proto-oncogene activating mutations such as KRas and its downstream target c-Myc are among the other dynamic ways that transform cells into cancer stem-like cells. Interestingly, KRas activating mutations have been detected in over 90% of pancreatic cancers and is one of the key mutations in various cancers [74–76].
The tumor microenvironment is another important factor that may contribute to transformation of differentiated cells into stem-like cells. In colorectal tumors, cancer-associated myofibroblasts can induce dedifferentiation pathways by secreting hepatocyte growth factor (HGF) that consequently activates Wnt signaling, which enhances stemness properties in differentiated cells [77]. T-cells and macrophages can direct dedifferentiation of differentiated cells through the secretion of tumor necrosis factor alpha (TNF-α) in the induced inflammatory microenvironment in melanoma. On the other hand, it has been assumed that these microenvironments induce plasticity in tumor cells that lead to changes in certain surface markers and the resistance of the tumor cells to an anti-cancer immune response [78]. In general, the tumor microenvironment increases cellular plasticity, which induces various cell types at various differentiation levels to transform into CSCs (Fig. 3).
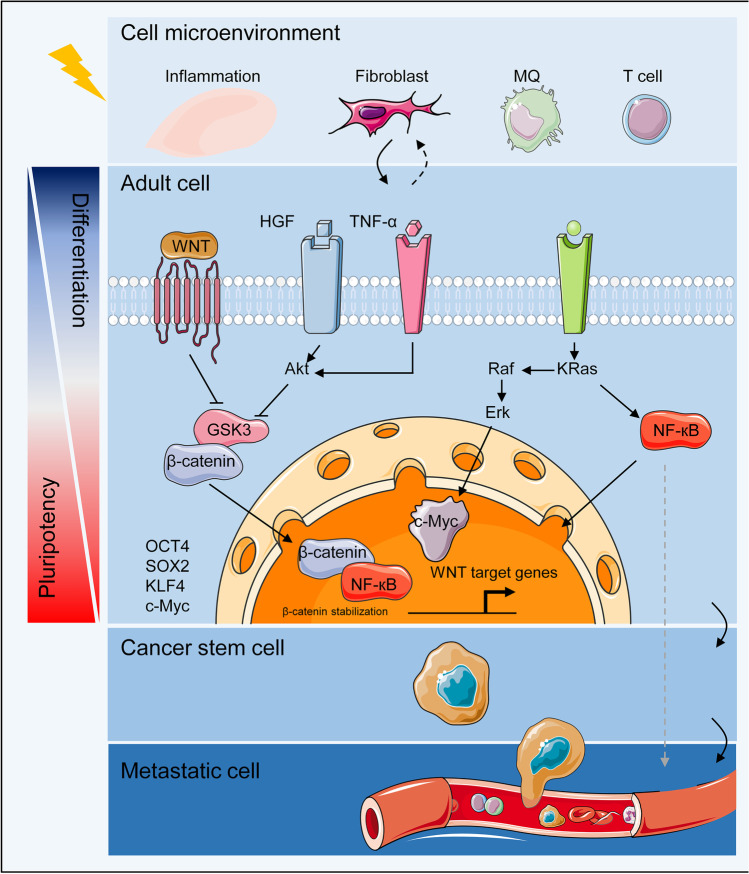
Fig. 3.
Adult stem cell reprogramming. Tissue adult stem cells are usually unipotent; however, under microenvironment-induced transformation, the activation of pluripotency-related signaling pathways like Wnt and transcription factors (TFs), such as Oct4, lead to their reprogramming into a pluripotent state The newly formed cells go through genetic and epigenetic alterations that favor cancer stem cell (CSC) formation
Comparisons between Pluripotent Stem Cells (PSCs) and Cancer Stem Cells (CSCs): Key Factors
Regardless of the cell of origin, the stemness characteristics and the resemblance of CSCs to early developmental cells is significant in the stem cell theory of cancer (Fig. 4). In many ways cancer cells imitate the early developmental pathways at the biological and molecular levels. The activation of initial developmental signaling pathways [79] and the TF expressions associated with pluripotency in both CSCs and PSCs [80] (Table 1). Moreover, the similarities in protein content [79] and metabolism [81] can show the proximity of CSCs and PSCs.
Fig. 4.
Yin-yang of pluripotency. While stemness and pluripotency in the early embryo are crucial factors in creating a new being, the activation of pluripotency in some cells, such as very small embryonic-like stem cells (VSELs) and tissue-specific stem cells in adult tissues leads to formation of cancerous cells and cancer stem cells (CSCs)
Table 1.
Pluripotency-related TF in PSCs and CSCs
| Pluripotency marker | Role in cancer development | Type of Cancer | References |
|---|---|---|---|
| OCT4 | Stemness, EMT, Metastasis and drug resistance | Breast cancer | [168–170] |
| Pancreatic cancer | [171] | ||
| Liver cancer | [172] | ||
| SOX2 | Stemness, EMT, Metastasis and drug resistance | Squamous cell carcinoma | [90, 173] |
| Small cell lung carcinoma | [91] | ||
| Non- Small cell lung carcinoma | [174] | ||
| Glioblastoma | [175] | ||
| Colorectal cancer | [176] | ||
| Bladder Urothelial carcinoma | [93] | ||
| Osteosarcoma | [94] | ||
| Tumor suppressor | Colorectal cancer | [95] | |
| Neuroblastoma, | [96] | ||
| Nasopharyngeal sarcoma | [97, 177] | ||
| NANOG | Stemness, EMT, Metastasis and drug resistance | Glioblastoma | [86] |
| Liver | [87, 178] | ||
| Colorectal cancer | [88] | ||
| Prostate cancer | [89, 179] | ||
| Gastric cancer | [180] |
Key Transcription Factors (TFs)
The induction of the main pluripotency-associated TFs Oct4, Sox2, KLF4 and NANOG results in induction of pluripotency in terminally differentiated cells into iPSCs. These TFs are considered to be key controllers of stemness in CSCs [82]. Oct4 plays an important role in germ cell tumor formation and is a diagnostic factor in these tumors. Post-translational modifications of Oct4 increase its stability in breast CSCs [83]. Oct4 is considered to be a renal CSC marker along with high expression of NANOG, and it is responsible for maintenance of CSCs, which negatively affects survival [84]. Oct4 expression increases stemness, drug resistance and migration of pancreatic CSCs (PCSCs) [85]. Similar to Oct4, NANOG plays an important role in maintaining malignant stem cell populations and CSCs of glioblastoma, liver, colon and prostate cancers [86–89]. Sox2 expression can be seen in squamous cell carcinoma of the esophagus [90] and small cell lung carcinoma [91], and its expression is necessary in maintenance of the CSC microenvironment in glioblastoma and breast cancer [92].
On the other hand, KLF4, is a double-edged sword in cancer development. Although this TF promotes malignancies in bladder urothelial carcinoma and osteosarcoma [93, 94], its inhibitory role in colorectal cancer [95], neuroblastoma [96], and nasopharyngeal sarcoma [97] has been reported. KLF4 interaction with c-Myc, a proto-oncogene, and its dissuasive effect on p53, a tumor suppressor, results in an increased proliferation and enhanced reprogramming that can be seen in p53 deficiency. Lack of efficient p53 expression can result in tumor formation in the adult stem cell populations, and diminish their differentiation and trigger malignant features in iPSCs [98]. KLF4 also triggers induction of telomerization activity in cancer cells, which is similar to ESCs [99]. C-Myc augments cell proliferation in many human malignancies and plays a role in reprogramming of fibroblasts to iPSCs [100]. This factor also affects the enzymes involved in transcription and can increase total transcription levels [101]. Together, these factors form a network of interactions that have two sides—one promotes malignancy and the other is responsible for the pluripotent state of cells. The balance between these two defines the normal cell state.
Signaling Pathways
Self-renewal and proliferation are two characteristics of both CSCs and PSCs; however, unlike PSCs, these mechanisms are not strictly controlled by CSCs. Both cells adjust the self-renewal process through the same signaling pathways (Table 2). However, irregular expressions of key factors in CSCs disrupt their balanced state and make them prone to tumor formation [102].
Table 2.
Common regulatory signaling cascades in PSCs and CSCs
| Signaling pathway | Targets | Role in cancer development | Cancer type | References |
|---|---|---|---|---|
| Notch | AKT, NF-kB, p21, p27 and CyclinD1 | Pluripotency maintenance, cell fate determination and proliferation and apoptosis regulation | Pancreatic Ductal Adenocarcinoma (PDAC) | [117, 181, 182] |
| Melanomas | [107] | |||
| Medulloblastoma | [183] | |||
| Ovarian cancer | [109] | |||
| Breast cancer | [110] | |||
| Prostate cancer | [184] | |||
| Hepatocellular Carcinoma | [185] | |||
| Non-Small Cell Lung Cancer (NSCLC) | [186] | |||
| Cyclin A, Cyclin D1, Cyclin E, CDK2, Retinoblastoma protein, HES-1, P21, P63 and P53 | Tumor suppressor | Cutaneous Squamous Cell Carcinoma (CSCC) | [187–189] | |
| head and neck squamous cell carcinoma (HNSCC) | [190] | |||
| Small Cell Lung Cancer (SCLC) | [191, 192] | |||
| WNT | β-catenin and STAT3 | Pluripotency maintenance,Stem cell self-renewal and differentiation and EMT | Colorectal Carcinoma | [118] |
| Leukemia | [114] | |||
| Melanoma | [119] | |||
| Liver cancer | [120] | |||
| Prostate cancer | [121] | |||
| Breast cancer | [122] | |||
| Hedgehog (HH) | GLI families | Pluripotency maintenance and Stem cell self-renewal | Basal Cell Carcinoma | [193, 194] |
| Medulloblastoma | [123, 125, 193, 195] | |||
| Colon cancer | [196] | |||
| Breast cancer | [197] | |||
| Chronic Myelogenous Leukemia (CML) | [198] | |||
| T- cell Acute Lymphoblastic Leukemia (T-ALL) | [199] | |||
| pancreatic Ductal Adenocarcinoma (PDA) | [200] |
Notch:
During development, the Notch signaling pathway is one of the central pathway that makes contact between adjacent cells; it is necessary for differentiation and determination of the cell fate and homeostasis of adult tissues [59]. The Notch signaling pathway reportedly functions in human ESCs (hESCs); however, it appears to be unnecessary for maintenance of pluripotency in these cells. Conversely, Notch signaling plays an important role in maintenance of the differentiating cell types in hESC cultures [103] such as neural differentiation of hESCs [104], as well as migration and proliferation of retinal pigment epithelial cells [105]. Upon activation by direct cell contact, cleaved Notch intracellular domain (NICD) in conjunction with a complex of proteins affects downstream targets that include AKT, NF-kB, p21, p27 and CyclinD1 [106]. Changes in these targets may result in changes in the cell fate, differentiation and uncontrolled proliferation, and may result in melanomas, medulloblastoma, ovarian and breast cancers [102, 107–110]. As a cell context dependent pathway, Notch signaling may also act as a tumor suppressor [111]. Several therapeutic approaches target components of this pathway. Delta like (DLL-1, DLL2, DLL4) or Jagged (JAG1, JAG2) ligand, Notch receptors (Notch1-4) and ɣ-secretase complex have been studied. However, in CSCs, several pathways are interconnected and suggested therapies should cover all features of signaling [60].
Wnt:
Wnt is one of the most conserved signaling pathways that is active in a wide variety of organisms and is essential for maintenance and self-renewal of adult stem cells (i.e., HSCs) and PSCs [112–114]. Its first connection to cancer was discovered when Wnt overexpression resulted in tumor formation in mice [115]. Nearly a decade later, mutations in the adenomatous polyposis coli (APC) gene were found to be responsible for hereditary colon cancer syndrome [116]. Normally, in the absence of the Wnt ligand, APC along with Axin and GSK3β form a regulatory complex which degrades β-catenin. Mutated APC activates Wnt signaling by stabilizing β-catenin, which results in T-cell factor (TCF) 4 transcriptional activity [117]. Non-canonical Wnt signaling, which is β-catenin independent, may play a role in cancer malignancy by activating planar cell polarity (PCP) and calcium signaling, and result in cell migration and possible metastasis. However, canonical Wnt signaling has been observed more frequently [116]. Telomerase subunit TERT expression after Wnt activation helps to keep telomerase activity, which also links Wnt to CSCs [99]. Other than colorectal carcinoma (CRC) [118], the role of Wnt signaling in leukemia [114], melanoma [119] liver cancer [120], prostate cancer [121] and triple negative breast cancer [122] has been studied and several drugs against this pathway have entered clinical trials [60].
Hedgehog (Hh):
Hh signaling is responsible for cell fate determination, neuro-ectodermal lineage differentiation, and polarity and pattern formation during organogenesis. Hh ligand binding to the PTCH1/2 receptor and Smoothened activation enables GLI families of TFs to target the expressions of several genes [123, 124]. Although this pathway is not a key regulator of hESC self-renewal and maintenance [123], disruptions in the normal activity of downstream targets such as the PTCH1 and SMO genes have been discovered in patients with basal cell carcinoma (BCC), medulloblastoma, colon cancer, breast cancer, chronic myelogenous leukemia (CML), acute myelogenous leukemia (AML), and acute lymphoblastic leukemia (ALL) [123, 125]. Interestingly non-canonical Hh signaling (Shh-Ptch-SMO independent) seems to be important in pancreatic ductal adenocarcinoma (PDAC). Several different therapies that use SMO antagonists and PTCH1 inhibitors have shown that cancer cells are resistant to some of the SMO inhibitors [125]. Li et al. recently reported that some SMO inhibitor substitutes could be used successfully against this resistance and present a novel opportunity for treatment of medulloblastoma [126].
Surface Antigens
Tumor and fetal cells have many surface antigens in common; hence, these markers are called oncofetal antigens and they include carcinoembryonic antigen (CEA), prostate-specific antigen (PSA), cancer-testis antigen (CTA), human placental lactogen (hPL), alpha-fetoprotein (AFP), beta-human chorionic gonadotropin (β-hCG) and placental alkaline phosphatase (PLAP) [127–131]. Exposure of mature animals to embryonic materials has been shown to increase tumor resistance and tumor transplant rejection [127]. The similarity between the serum antibodies of a cancer patient to a pregnant woman can be attributed to the similarities between cancer and embryonic antigens [132, 133]. Thus, cancer diagnosis during pregnancy may be difficult because both conditions have elevations in the cancer antigen 15–3 (CA 15–3), squamous cell carcinoma antigen (SCC), CA 125, and AFP markers [134]. Many differentiated tissue antigens lose their expressions after tumor formation, which is called “antigenic simplification” [127]. In return, the tumor cells begin to re-express the embryonic genes/antigens. More importantly, the immunity against tumors created by embryonic material is both humoral and cellular [17, 135]. Based on these findings, one therapeutic approach against cancer could be a vaccine based on embryonic/fetal materials.
Developmental Biology: A New Approach to Cancer Treatment
The aforementioned similarities between fetal and cancer cells, especially malignant ones, show that early developmental programs that are active in ESCs and are responsible for fetal development play a role in cancer formation. As these developmental processes are under strict regulation in an embryo, mimicking those regulations may be effective in finding new therapeutic agents against cancer.
Cancer and Cancer Stem Cell (CSC) Modeling Using Pluripotent Stem Cells (PSCs)
The discovery of the ability to reprogramming of somatic cells into PSCs has developed the view of almost all aspects of biomedical research, including cancer. Recently, similar to iPSC derivation technology, the reprogramming protocols that utilize key pluripotency-associated TFs have been used to induce pluripotency in cancer cell lines. In these studies, melanoma-R545, CML-KBM7, osteosarcoma cell lines-(SAOS2, HOS, MG63), non-small cell lung cancer lines-(H358, H460, IMR90), and gastrointestinal cancer cell lines (GCC) have been placed through dedifferentiation that resulted in the generation of iPSCs. However, some of these cells have karyotype abnormalities and are resistance to anti-cancer drugs [136, 137]. On the other hand, in order to observe the cancer formation process, iPSCs were derived from somatic cells that contained germline mutations and subsequently differentiated into lineages that had several early signs of malignancy. This method was used as a model in Li-Fraumeni syndrome [138], myelodysplastic syndrome [139], PDAC [140], and colorectal cancer [141] to determine the role of genetic mutations responsible for these malignancies.
In the CSC modeling related studies, it was shown that exposure of normal iPSCs to the mouse Lewis lung carcinoma (LLC) cell line microenvironment and extracellular vesicles could turn them into cells with CSC phenotypes that had high tumorigenic and metastatic potentials once transplanted into nude mice [142, 143]. Besides self-renewal, these CSCs could differentiate into endothelial cells that resulted in tumor angiogenesis both in vitro and in vivo [144]. Additionally, drug testing on prostate CSC-like cells derived from iPSCs by the same protocol indicated that these cells are resistant to chemotherapeutic drugs and sensitive to agents that target their stemness [145]. These findings from cancer and CSC modeling with iPSC technology suggest that these cells can help to determine the signaling pathways and mechanisms of tumorigenesis, metastasis, and angiogenesis, and also act as platforms to study and test drugs against stemness and CSC-like characteristics of different types of cancer.
It is important to note that despite the increasing advances in iPSC production and further lineage differentiation with improved protocols, cancer modeling with iPSCs has not yet made much progress. Technically, this limitation can be attributed to two factors. First, some studies have shown that cancer cells are more resistance to reprogramming than normal cells. This resistance may be due to various cancer-related mutations in cancer cells as the starting material for reprogramming. The second factor is that the process of creating iPSCs from a cancer cell and further lineage differentiation does not work in the cell culture in exactly the same that it does in cancer. As mentioned before, most likely, the cancer starts from VSELs or tissue-specific stem/progenitor cells. Therefore, a proper modeling of cancer with iPSCs that was close to reality must first be able to create progenitor cells in this simulation instead of directly producing the final differentiated cells. Moreover, most differentiated cells from iPSCs are similar to cells in early development rather than phenotypes in adults [146].
Another challenge that limits the modelling of cancer is that iPSCs inherit their starting cells’ genetic memory. Therefore, the variable genetic instabilities of the primary cancer cells are captured and amplified in their iPSCs. It may be resolved by isolation of single cells and clonal propagation in order to establishment of a stable original genome. There are also other challenges related to iPSCs’ production per se, such as epigenetic instability, incomplete reprogramming, and line-to line variation, that are true for modeling of cancer with iPSCs, too [146].
Differentiation Therapy by Induction of the Embryonic Microenvironment
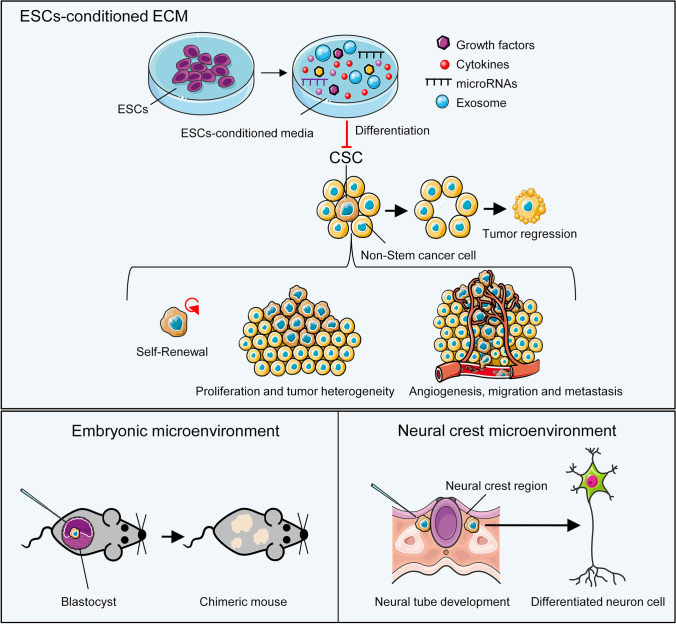
There is an overlap between signaling pathways that control developmental processes and tumor progression. Thus, the question arises whether or not cancer cells can respond to embryonic messages. Several studies have been designed to answer this question. By using matrix conditioned with hESCs, Postovit and colleagues showed an increase in melan-A (MLANA) expression in human melanoma cells and a reduction in their invasiveness [147]. The inhibitory nature of the conditioned medium of ESCs and secretory factors released by these cells increased the number of CSCs in the G1 phase and with the cell cycle arrest without inducing apoptosis [148]. Investigation of intercellular interactions between breast cancer cells and ESCs in a 3D-engineered tumor model showed that low molecular weight factors released by pluripotent cells suppressed Stat3 signaling and inhibited cancer cell proliferation and migration [149]. Examination of this microenvironment showed that exposed exosomes were the main factors that affected tumor cells and reduced their invasiveness [150]. Notch signaling is also responsible for the antitumor activity of this specific environment against colorectal cancer [151]. Another important factor in the embryonic microenvironment that regulates cancer cell proliferation and differentiation is Gremlin, a BMP4 antagonist. Inhibition of BMP4 activates senescence in melanoma cells [152]. Various miRNAs have been shown to play roles as tumor inhibitors or as oncogenes in cancer. Melanoma cells exposed to hESCs had increased miR-302 expression. An augmented level of miR302 by Notch4 inhibition causes Nodal reduction. On the other hand, expression of miR-27b, which is responsible for angiogenesis and progression of melanoma in the early stages, is reduced after exposure to the hESCs microenvironment [153]. These studies show the modulation effects of ESCs and their microenvironment on cancer cells, and indicate the potential impact of this approach on cancer treatment. However, the mechanism of these effects is still not clear.
Furthermore, the effects of the embryonic microenvironment on tumor cell progression and metastasis has been investigated in animal models. In a study by Illmensee and Mintz, injection of terato-carcinoma cells into mouse blastocysts not only inhibited the tumor phenotype of the cells, but the terato-carcinoma cells could participate in the formation of normal tissues. In a chick model, the tumor-suppressing activity of the embryonic microenvironment also inhibited sarcoma formation after injection of Rous sarcoma virus [154]. It has been shown that tumorigenicity of the mouse-melanoma cells decreased when the cells were exposed to factors secreted by neural crest region and the cells gained specific markers of differentiated cells such as neural marker TUJ1 [155]. Therefore, embryonic microenvironments appear to provide regulatory factors that are lost in cancer cells during transformation and these factors can be used to restructure embryonic signaling in cancer cells and their microenvironment (Fig. 5).
Fig. 5.
Differentiation therapy. Communication between the cells and their microenvironment is essential for the maintenance of normal tissues or formation of cancers and their metastases. In cancer cells, there is disrupted regulation of cell differentiation, which leads to the exhibition of a tumor promoting phenotype. The embryonic niche or embryonic stem cell (ESC) conditioned-media can promote undifferentiated cancer stem cells (CSCs) to become differentiated, which ultimately inhibits tumor growth. After injection into an early embryo, aggressive CSCs learn to “speak the same language” of the embryonic cells and participate in embryo formation instead of tumor growth. The specific niche of the embryo can change the CSC phenotype toward specific cell lineages related to that niche
Using Pluripotent Stem Cells (PSCs) as a Cancer Vaccine
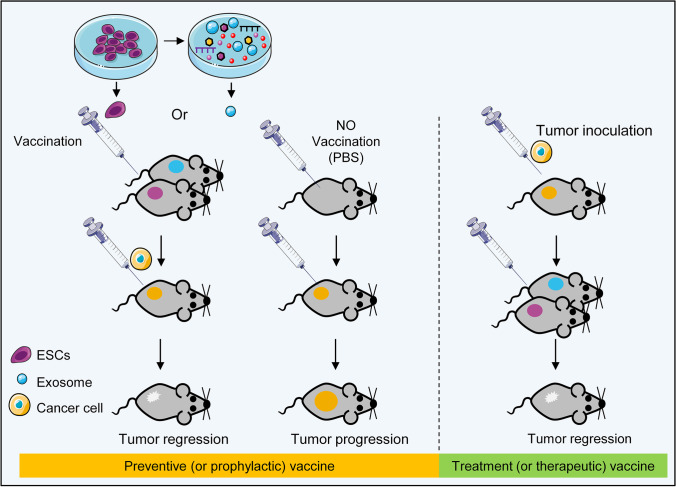
One of the new potential application of PSCs in cancer research that has been raised in recent years, is the use of PSCs as a global prophylactic cancer vaccine. This idea may seem strange at first; whereas both PSCs and CSCs have common stemness factors, and just as CSCs, PSCs can pose the same threat for body. But, in fact, the possibility of large scale cultivation of genetically stable PSCs in vitro, makes available the high amounts of CSCs-specific antigens. In one hand, the conditioned medium of PSCs per se can provide an environment similar to the embryonic environment that promote cancer treatment through differentiation therapy (Fig. 5), and on the other hand, PSCs can be considered as a valuable source to have large amounts of CSCs-specific antigens. Thus far, inactivated PSCs have been used to make prophylactic vaccines against colon [156], lung [17] and ovarian cancer [157] in animal models.
While in the immunosuppressive environment of tumors, cancer cells can suppress the activation of immune cells through various mechanisms, including the recruitment of immunomodulatory MSCs, myeloid-derived suppressor cells (MDSCs), and regulatory T cells (Tregs), the attenuation of MHC class I expression, and the use of the PD-1/PD-L1 axis, cancer vaccines based on cancer–specific antigens can educate the immune system against cancer cells, so that it can encounters and eliminates them. The basic idea behind a cancer vaccine is to find the optimal antigen and vaccine delivery system that is capable of stimulates immune cells. As a result of cancer vaccines, immune responses could be guided selectively toward patients’ cancerous cells while preventing their normal cells from immune attack, potentially avoiding side effects. For example, DCs as a professional APC can induce tumor-specific cytotoxicity by processing and presenting tumor antigens to T lymphocytes. DCs can load with different type of antigens including peptide, RNA and also with whole tumor antigen via tumor lysate or fusion with tumor cells and present numerous antigens to immune effector CD8+- and CD4+-T cells [158, 159]. In addition, induction of immunological memory by DCs overcomes cancer cell plasticity and tumor recurrence [160]. However, DC vaccine therapies require an effective and adequate number of DCs to produce the desired results while in most patients, functional DC is difficult to achieve due to chemotherapy or the tumor’s immunosuppressive state [161].
Based on this idea, it is suggested that the exposure to conditioned medium, lysate, or mitotically inactivated CSCs or PSCs can activate the immune system and makes immunity against both embryonic and cancer cells [162]. This ability of PSCs can be used by introduction of embryonic materials to cancer cells. The studies show significant reductions in tumor growth and activation of both the humoral and cellular immune systems in response to PSCs. These immunities prevent the growth and proliferation of cancer cells, and an increase in APCs and activation of lymphocytes has been also reported [163]. Interestingly, in these studies, hESCs also produced immune responses in rats, and it can be concluded that the immune function of these cells is species-independent. These vaccines did not show any side effects such as autoimmune reactions [164]. A PSC-derived based vaccine for cancer combined with a histone deacetylase inhibitor (HDACi) could modify the immune suppression microenvironment of the tumor by increasing CD4+ or CD8+ tumor infiltrating lymphocytes and reducing T regulatory cells. This combined regimen targets CSCs; hence, it could decrease the incidence of metastasis and improve survival rates [165]. Furthermore, vaccination with ESC exosomes, a derivative of these cells in the presence of GM-CSF, resulted in reduced tumor size and immune response [166]. The enhanced targeting ability of ESC exosome carriers combined with chemotherapeutic agents can act highly reactive against tumors [167]. These studies show how ESCs and their secretome act as tumor suppressors and this can be a potential therapeutic approach. The use of exosomes instead of inactivated cells can be a tremendous step towards a cell-free vaccination against cancers (Fig. 6). Overall, these studies showed that with the antigenic similarity to CSCs, PSCs or PSCs-derived secretome therapeutic cancer vaccines can be highly immunogenic against CSCs. As cancer treatment has always been a challenging issue due to the high rate of tumor recurrence and metastasis, PSC-based therapeutic cancer vaccines can theoretically help to overcome the effects of immunosuppression in the tumor microenvironment.
Fig. 6.
Pluripotent stem cell (PSC)-derived vaccine. Left: Inactivated PSCs or their extracellular vesicles can induce antitumor immunity that suppresses cancer cell proliferation and prevents tumor development. Right: PSC-based vaccines can educate the immune system against the cancer and overcome low or no immunogenicity that exist in the tumor microenvironment
Conclusion and Future Outlook
Despite multiple therapy attempts, cancer has not been defeated. Tumor cell heterogeneity that causes differences in their biology, metabolism and signaling pathways as well as the effects of the microenvironment on normal cells are challenges that complicate the choice of treatment. Fortunately, recognition of processes such as autophagy has contributed greatly to better identification of cancer cells, and allows for a more effective treatment approach. In order to achieve successful and effective treatment, cancer cells have been targeted based on their recurrence and metastasis. As noted above, there is stemness, pluripotency and the expression of early developmental markers in cancer cells, especially in malignant cancers with poor prognosis. Apart from the assumptions related to the origin of CSCs, the existence of morphological and molecular similarities between cancer cells and early-stage stem cells suggests that early developmental programs are inappropriately activated in cancer; however, the mechanism of this phenomenon is not completely understood. It is assumed that the inappropriate expressions of pluripotency factors and reprogramming of somatic or adult stem cells are the main sources of tumor-like stem cells. There is a defect in developmental biology in these scenarios despite the fact that most developmental biology is observed in embryonic development. Therefore, it seems that a study of embryonic development and its differences and similarities with the developmental processes in cancer can help us to find new therapeutic approaches. Targeting markers and stemness and pluripotency pathways, the use of ESCs as a vaccine, and consideration of embryonic microenvironments in order to regulate developmental phenomena have been investigated to improve cancer treatments.
Acknowledgements
Manuscript was written during a doctoral program was approved and funded by Royan Institute for Stem Cell Biology and Technology. This project is also co-funded by Cancer Research Center of Cancer Institute of Iran (Shams cancer charity, Grant No: 37309-202-01- 97), and Royan Stem Cell Technology Company.
Author Contributions
MB, MA, and SN–H designed the concept of the manuscript. BM, MA, NS, NH, and AG wrote manuscript. MB designed and prepared the figs. HB, ME, and SN–H revised manuscript.
Declarations
Ethical Approval
The content of the manuscript is original and has not been published or accepted somewhere, either in whole or in part. Not part of the manuscript is now under evaluation for publication elsewhere.
Conflict of Interest
The authors have no competing interests to declare.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Mojgan Barati and Maryam Akhondi authors contributed equally to this work.
Contributor Information
Marzieh Ebrahimi, Email: m.ebrahimi@royan-rc.ac.ir.
Seyedeh-Nafiseh Hassani, Email: snafiseh.hassani@royaninstitute.org, Email: sn.hassani@royan-rc.ac.ir.
References
- 1.WHO. (2020). WHO report on cancer: setting priorities, investing wisely and providing care for all.
- 2.Vasan N, Baselga J, Hyman DM. A view on drug resistance in cancer. Nature. 2019;575(7782):299–309. doi: 10.1038/s41586-019-1730-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brand NR, et al. Delays and Barriers to Cancer Care in Low- and Middle-Income Countries: A Systematic Review. The Oncologist. 2019;24(12):e1371–e1380. doi: 10.1634/theoncologist.2019-0057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Otto-Meyer S, et al. The interplay among psychological distress, the immune system, and brain tumor patient outcomes. Current Opinion in Behavioral Sciences. 2019;28:44–50. doi: 10.1016/j.cobeha.2019.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chapuis PH, et al. Recurrence and cancer-specific death after adjuvant chemotherapy for Stage III colon cancer. Colorectal Disease. 2019;21(2):164–173. doi: 10.1111/codi.14434. [DOI] [PubMed] [Google Scholar]
- 6.Pan H, et al. 20-Year Risks of Breast-Cancer Recurrence after Stopping Endocrine Therapy at 5 Years. New England Journal of Medicine. 2017;377(19):1836–1846. doi: 10.1056/NEJMoa1701830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Reardon, D. A., et al. (2020). Effect of Nivolumab vs Bevacizumab in Patients With Recurrent Glioblastoma: The CheckMate 143 Phase 3 Randomized Clinical Trial. JAMA Oncolology. [DOI] [PMC free article] [PubMed]
- 8.Rycaj K, Tang DG. Cell-of-Origin of Cancer versus Cancer Stem Cells: Assays and Interpretations. Cancer Research. 2015;75(19):4003–4011. doi: 10.1158/0008-5472.CAN-15-0798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Akbarzadeh, M., et al. (2019). Current approaches in identification and isolation of cancer stem cells. Journal of Cell Physiology. [DOI] [PubMed]
- 10.Ilic D, Ogilvie C. Concise Review: Human Embryonic Stem Cells-What Have We Done? What Are We Doing? Where Are We Going? Stem Cells. 2017;35(1):17–25. doi: 10.1002/stem.2450. [DOI] [PubMed] [Google Scholar]
- 11.Takahashi K, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131(5):861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 12.Trounson A, DeWitt ND. Pluripotent stem cells progressing to the clinic. Nature Reviews Molecular Cell Biology. 2016;17(3):194–200. doi: 10.1038/nrm.2016.10. [DOI] [PubMed] [Google Scholar]
- 13.Yanagimachi, M. D., et al. (2013). Robust and highly-efficient differentiation of functional monocytic cells from human pluripotent stem cells under serum- and feeder cell-free conditions. PLoS One, 8(4), e59243. [DOI] [PMC free article] [PubMed]
- 14.Zhu H, et al. Concise Review: Human Pluripotent Stem Cells to Produce Cell-Based Cancer Immunotherapy. Stem Cells. 2018;36(2):134–145. doi: 10.1002/stem.2754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Uenishi GI, et al. NOTCH signaling specifies arterial-type definitive hemogenic endothelium from human pluripotent stem cells. Nature Communications. 2018;9(1):1828. doi: 10.1038/s41467-018-04134-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Postovit L-M, et al. Human embryonic stem cell microenvironment suppresses the tumorigenic phenotype of aggressive cancer cells. Proceedings of the National Academy of Sciences of the United States of America. 2008;105(11):4329–4334. doi: 10.1073/pnas.0800467105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yaddanapudi, K., et al. (2012). Vaccination with Embryonic Stem Cells Protects against Lung Cancer: Is a Broad-Spectrum Prophylactic Vaccine against Cancer Possible? PLoS One. 7(7), e42289. [DOI] [PMC free article] [PubMed]
- 18.Aponte PM, Caicedo A. Stemness in Cancer: Stem Cells, Cancer Stem Cells, and Their Microenvironment. Stem Cells Int. 2017;2017:5619472. doi: 10.1155/2017/5619472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yadav AK, Desai NS. Cancer Stem Cells: Acquisition, Characteristics, Therapeutic Implications, Targeting Strategies and Future Prospects. Stem Cell Rev Rep. 2019;15(3):331–355. doi: 10.1007/s12015-019-09887-2. [DOI] [PubMed] [Google Scholar]
- 20.Bu Y, Cao D. The origin of cancer stem cells. Frontiers in Bioscience (Scholar Edition) 2012;4(3):819–830. doi: 10.2741/s302. [DOI] [PubMed] [Google Scholar]
- 21.Cooper M. Regenerative pathologies: Stem cells, teratomas and theories of cancer. Medicine Studies. 2009;1(1):55. doi: 10.1007/s12376-008-0002-4. [DOI] [Google Scholar]
- 22.Ratajczak, M. Z., et al. (2020). The Embryonic Rest Hypothesis of Cancer Development: 150 Years Later, in Trends in Stem Cell Proliferation and Cancer Research, 51–63.
- 23.Hohenstein P, Pritchard-Jones K, Charlton J. The yin and yang of kidney development and Wilms' tumors. Genes & Development. 2015;29(5):467–482. doi: 10.1101/gad.256396.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shin DM, et al. Molecular signature of adult bone marrow-purified very small embryonic-like stem cells supports their developmental epiblast/germ line origin. Leukemia. 2010;24(8):1450–1461. doi: 10.1038/leu.2010.121. [DOI] [PubMed] [Google Scholar]
- 25.Ratajczak MZ, Ratajczak J, Kucia M. Very Small Embryonic-Like Stem Cells (VSELs) Circulation Research. 2019;124(2):208–210. doi: 10.1161/CIRCRESAHA.118.314287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ratajczak MZ, et al. Cancer from the perspective of stem cells and misappropriated tissue regeneration mechanisms. Leukemia. 2018;32(12):2519–2526. doi: 10.1038/s41375-018-0294-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zuba-Surma EK, et al. Very small embryonic-like stem cells in adult tissues-potential implications for aging. Mechanisms of Ageing and Development. 2009;130(1–2):58–66. doi: 10.1016/j.mad.2008.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Virant-Klun I, et al. Similar population of CD133+ and DDX4+ VSEL-like stem cells sorted from human embryonic stem cell, ovarian, and ovarian Cancer ascites cell cultures: The real embryonic stem cells? Cells. 2019;8(7):706. doi: 10.3390/cells8070706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ratajczak MZ, et al. Identification of very small embryonic/epiblast-like stem cells (VSELs) circulating in peripheral blood during organ/tissue injuries. Methods in Cell Biology. 2011;103:31–54. doi: 10.1016/B978-0-12-385493-3.00003-6. [DOI] [PubMed] [Google Scholar]
- 30.Bhartiya D. Pluripotent Stem Cells in Adult Tissues: Struggling To Be Acknowledged Over Two Decades. Stem Cell Rev Rep. 2017;13(6):713–724. doi: 10.1007/s12015-017-9756-y. [DOI] [PubMed] [Google Scholar]
- 31.Ratajczak MZ, et al. Very small embryonic-like stem cells (VSELs) represent a real challenge in stem cell biology: Recent pros and cons in the midst of a lively debate. Leukemia. 2014;28(3):473–484. doi: 10.1038/leu.2013.255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ratajczak MZ, et al. RasGrf1: Genomic imprinting, VSELs, and aging. Aging (Albany NY) 2011;3(7):692. doi: 10.18632/aging.100354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shin DM, et al. Novel epigenetic mechanisms that control pluripotency and quiescence of adult bone marrow-derived Oct4(+) very small embryonic-like stem cells. Leukemia. 2009;23(11):2042–2051. doi: 10.1038/leu.2009.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Peters J. The role of genomic imprinting in biology and disease: An expanding view. Nature Reviews Genetics. 2014;15(8):517–530. doi: 10.1038/nrg3766. [DOI] [PubMed] [Google Scholar]
- 35.Singh P, Lee D-H, Szabó PE. More than insulator: Multiple roles of CTCF at the H19-Igf2 imprinted domain. Frontiers in genetics. 2012;3:214. doi: 10.3389/fgene.2012.00214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kaushik A, Anand S, Bhartiya D. Altered biology of testicular VSELs and SSCs by neonatal endocrine disruption results in defective spermatogenesis, reduced fertility and tumor initiation in adult mice. Stem Cell Reviews and Reports. 2020;16(5):893–908. doi: 10.1007/s12015-020-09996-3. [DOI] [PubMed] [Google Scholar]
- 37.Kaushik A, Bhartiya D. Pluripotent very small embryonic-like stem cells in adult testes–an alternate premise to explain testicular germ cell tumors. Stem cell reviews and reports. 2018;14(6):793–800. doi: 10.1007/s12015-018-9848-3. [DOI] [PubMed] [Google Scholar]
- 38.Virant-Klun I, Stimpfel M. Novel population of small tumour-initiating stem cells in the ovaries of women with borderline ovarian cancer. Scientific reports. 2016;6(1):1–23. doi: 10.1038/srep34730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Virant-Klun I, Kenda-Suster N, Smrkolj S. Small putative NANOG, SOX2, and SSEA-4-positive stem cells resembling very small embryonic-like stem cells in sections of ovarian tissue in patients with ovarian cancer. Journal of ovarian research. 2016;9(1):1–15. doi: 10.1186/s13048-016-0221-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Beumer J, Clevers H. Regulation and plasticity of intestinal stem cells during homeostasis and regeneration. Development. 2016;143(20):3639–3649. doi: 10.1242/dev.133132. [DOI] [PubMed] [Google Scholar]
- 41.Colacino JA, et al. Heterogeneity of Human Breast Stem and Progenitor Cells as Revealed by Transcriptional Profiling. Stem Cell Reports. 2018;10(5):1596–1609. doi: 10.1016/j.stemcr.2018.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kim CF, et al. Identification of bronchioalveolar stem cells in normal lung and lung cancer. Cell. 2005;121(6):823–835. doi: 10.1016/j.cell.2005.03.032. [DOI] [PubMed] [Google Scholar]
- 43.Ng A, Barker N. Ovary and fimbrial stem cells: Biology, niche and cancer origins. Nature Reviews Molecular Cell Biology. 2015;16(10):625–638. doi: 10.1038/nrm4056. [DOI] [PubMed] [Google Scholar]
- 44.Wang X, et al. A luminal epithelial stem cell that is a cell of origin for prostate cancer. Nature. 2009;461(7263):495–500. doi: 10.1038/nature08361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Batlle E, Clevers H. Cancer stem cells revisited. Nature medicine. 2017;23(10):1124. doi: 10.1038/nm.4409. [DOI] [PubMed] [Google Scholar]
- 46.Visvader JE, Clevers H. Tissue-specific designs of stem cell hierarchies. Nature Cell Biology. 2016;18(4):349–355. doi: 10.1038/ncb3332. [DOI] [PubMed] [Google Scholar]
- 47.Rodgers JT, et al. mTORC1 controls the adaptive transition of quiescent stem cells from G0 to G(Alert) Nature. 2014;510(7505):393–396. doi: 10.1038/nature13255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chen X, He Y, Lu F. Autophagy in Stem Cell Biology: A Perspective on Stem Cell Self-Renewal and Differentiation. Stem Cells Int. 2018;2018:9131397. doi: 10.1155/2018/9131397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Montagnani S, et al. Adult Stem Cells in Tissue Maintenance and Regeneration. Stem Cells Int. 2016;2016:7362879. doi: 10.1155/2016/7362879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Blokzijl F, et al. Tissue-specific mutation accumulation in human adult stem cells during life. Nature. 2016;538(7624):260–264. doi: 10.1038/nature19768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Covey MV, et al. REST regulates the pool size of the different neural lineages by restricting the generation of neurons and oligodendrocytes from neural stem/progenitor cells. Development. 2012;139(16):2878–2890. doi: 10.1242/dev.074765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Adams PD, Jasper H, Rudolph KL. Aging-Induced Stem Cell Mutations as Drivers for Disease and Cancer. Cell Stem Cell. 2015;16(6):601–612. doi: 10.1016/j.stem.2015.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bjerkvig R, et al. The origin of the cancer stem cell: Current controversies and new insights. Nature Reviews Cancer. 2005;5(11):899–904. doi: 10.1038/nrc1740. [DOI] [PubMed] [Google Scholar]
- 54.Biswas S, et al. Microenvironmental control of stem cell fate in intestinal homeostasis and disease. The Journal of Pathology. 2015;237(2):135–145. doi: 10.1002/path.4563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Redondo PA, et al. Elements of the niche for adult stem cell expansion. J Tissue Eng. 2017;8:2041731417725464. doi: 10.1177/2041731417725464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sharif T, et al. Autophagic homeostasis is required for the pluripotency of cancer stem cells. Autophagy. 2017;13(2):264–284. doi: 10.1080/15548627.2016.1260808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Nazio F, et al. Autophagy and cancer stem cells: Molecular mechanisms and therapeutic applications. Cell Death and Differentiation. 2019;26(4):690–702. doi: 10.1038/s41418-019-0292-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cole AJ, et al. An evolving paradigm of cancer stem cell hierarchies: Therapeutic implications. Theranostics. 2020;10(7):3083–3098. doi: 10.7150/thno.41647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Bigas A, Espinosa L. Notch Signaling in Cell-Cell Communication Pathways. Current Stem Cell Reports. 2016;2(4):349–355. doi: 10.1007/s40778-016-0065-1. [DOI] [Google Scholar]
- 60.Takebe N, et al. Targeting Notch, Hedgehog, and Wnt pathways in cancer stem cells: Clinical update. Nature Reviews. Clinical Oncology. 2015;12(8):445–464. doi: 10.1038/nrclinonc.2015.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Uchida N, et al. Direct isolation of human central nervous system stem cells. Proceedings of the national academy of sciences. 2000;97(26):14720–14725. doi: 10.1073/pnas.97.26.14720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Williams K, et al. CD44 integrates signaling in normal stem cell, cancer stem cell and (pre)metastatic niches. Experimental Biology and Medicine (Maywood, N.J.) 2013;238(3):324–338. doi: 10.1177/1535370213480714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Barzegar Behrooz, A., Syahir, A., Ahmad S. (2019). CD133: beyond a cancer stem cell biomarker. Journal of Drug Targeting, 27(3), 257–269. [DOI] [PubMed]
- 64.Prager BC, et al. Cancer Stem Cells: The Architects of the Tumor Ecosystem. Cell Stem Cell. 2019;24(1):41–53. doi: 10.1016/j.stem.2018.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Krebsbach PH, Villa-Diaz LG. The Role of Integrin alpha6 (CD49f) in Stem Cells: More than a Conserved Biomarker. Stem Cells Dev. 2017;26(15):1090–1099. doi: 10.1089/scd.2016.0319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Martin TA, Jiang WG. Evaluation of the expression of stem cell markers in human breast cancer reveals a correlation with clinical progression and metastatic disease in ductal carcinoma. Oncology Reports. 2014;31(1):262–272. doi: 10.3892/or.2013.2813. [DOI] [PubMed] [Google Scholar]
- 67.Zhu L, et al. Prominin 1 marks intestinal stem cells that are susceptible to neoplastic transformation. Nature. 2009;457(7229):603–607. doi: 10.1038/nature07589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 69.Villodre ES, et al. Silencing of the transcription factors Oct4, Sox2, Klf4, c-Myc or Nanog has different effect on teratoma growth. Biochemical and Biophysical Research Communications. 2019;517(2):324–329. doi: 10.1016/j.bbrc.2019.07.064. [DOI] [PubMed] [Google Scholar]
- 70.Friedmann-Morvinski D, Verma IM. Dedifferentiation and reprogramming: Origins of cancer stem cells. EMBO Reports. 2014;15(3):244–253. doi: 10.1002/embr.201338254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Chaffer CL, et al. Normal and neoplastic nonstem cells can spontaneously convert to a stem-like state. Proc Natl Acad Sci U S A. 2011;108(19):7950–7955. doi: 10.1073/pnas.1102454108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Tata PR, et al. Dedifferentiation of committed epithelial cells into stem cells in vivo. Nature. 2013;503(7475):218–223. doi: 10.1038/nature12777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Schwitalla S, et al. Intestinal tumorigenesis initiated by dedifferentiation and acquisition of stem-cell-like properties. Cell. 2013;152(1–2):25–38. doi: 10.1016/j.cell.2012.12.012. [DOI] [PubMed] [Google Scholar]
- 74.Guerra C, et al. Chronic pancreatitis is essential for induction of pancreatic ductal adenocarcinoma by K-Ras oncogenes in adult mice. Cancer Cell. 2007;11(3):291–302. doi: 10.1016/j.ccr.2007.01.012. [DOI] [PubMed] [Google Scholar]
- 75.Gidekel Friedlander SY, et al. Context-dependent transformation of adult pancreatic cells by oncogenic K-Ras. Cancer Cell. 2009;16(5):379–389. doi: 10.1016/j.ccr.2009.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Hwang JH, et al. A mutant KRAS-induced factor REG4 promotes cancer stem cell properties via Wnt/beta-catenin signaling. International Journal of Cancer. 2020;146(10):2877–2890. doi: 10.1002/ijc.32728. [DOI] [PubMed] [Google Scholar]
- 77.Vermeulen L, et al. Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nature Cell Biology. 2010;12(5):468–476. doi: 10.1038/ncb2048. [DOI] [PubMed] [Google Scholar]
- 78.Landsberg J, et al. Melanomas resist T-cell therapy through inflammation-induced reversible dedifferentiation. Nature. 2012;490(7420):412–416. doi: 10.1038/nature11538. [DOI] [PubMed] [Google Scholar]
- 79.Hadjimichael C, et al. Common stemness regulators of embryonic and cancer stem cells. World J Stem Cells. 2015;7(9):1150–1184. doi: 10.4252/wjsc.v7.i9.1150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kim J, Orkin SH. Embryonic stem cell-specific signatures in cancer: Insights into genomic regulatory networks and implications for medicine. Genome medicine. 2011;3(11):1–8. doi: 10.1186/gm291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Jang H, et al. Metabolism in embryonic and cancer stemness. Archives of Pharmacal Research. 2015;38(3):381–388. doi: 10.1007/s12272-015-0558-y. [DOI] [PubMed] [Google Scholar]
- 82.Muller M, et al. The role of pluripotency factors to drive stemness in gastrointestinal cancer. Stem Cell Res. 2016;16(2):349–357. doi: 10.1016/j.scr.2016.02.005. [DOI] [PubMed] [Google Scholar]
- 83.Cho Y, et al. Post-translational modification of OCT4 in breast cancer tumorigenesis. Cell Death and Differentiation. 2018;25(10):1781–1795. doi: 10.1038/s41418-018-0079-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Rasti A, et al. Co-expression of Cancer Stem Cell Markers OCT4 and NANOG Predicts Poor Prognosis in Renal Cell Carcinomas. Science and Reports. 2018;8(1):11739. doi: 10.1038/s41598-018-30168-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Lu Y, et al. Knockdown of Oct4 and Nanog expression inhibits the stemness of pancreatic cancer cells. Cancer Letters. 2013;340(1):113–123. doi: 10.1016/j.canlet.2013.07.009. [DOI] [PubMed] [Google Scholar]
- 86.Zbinden M, et al. NANOG regulates glioma stem cells and is essential in vivo acting in a cross-functional network with GLI1 and p53. EMBO Journal. 2010;29(15):2659–2674. doi: 10.1038/emboj.2010.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Lee TK, et al. CD24(+) liver tumor-initiating cells drive self-renewal and tumor initiation through STAT3-mediated NANOG regulation. Cell Stem Cell. 2011;9(1):50–63. doi: 10.1016/j.stem.2011.06.005. [DOI] [PubMed] [Google Scholar]
- 88.Ibrahim EE, et al. Embryonic NANOG activity defines colorectal cancer stem cells and modulates through AP1- and TCF-dependent mechanisms. Stem Cells. 2012;30(10):2076–2087. doi: 10.1002/stem.1182. [DOI] [PubMed] [Google Scholar]
- 89.Wang, X., et al. (2019). AMPK Promotes SPOP-Mediated NANOG Degradation to Regulate Prostate Cancer Cell Stemness. Developmental Cell, 48(3), 345–360 e7. [DOI] [PMC free article] [PubMed]
- 90.Wang Z, et al. AKT drives SOX2 overexpression and cancer cell stemness in esophageal cancer by protecting SOX2 from UBR5-mediated degradation. Oncogene. 2019;38(26):5250–5264. doi: 10.1038/s41388-019-0790-x. [DOI] [PubMed] [Google Scholar]
- 91.Bora-Singhal N, et al. Novel HDAC11 inhibitors suppress lung adenocarcinoma stem cell self-renewal and overcome drug resistance by suppressing Sox2. Science and Reports. 2020;10(1):4722. doi: 10.1038/s41598-020-61295-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Yin Y, et al. The FBXW2-MSX2-SOX2 axis regulates stem cell property and drug resistance of cancer cells. Proc Natl Acad Sci U S A. 2019;116(41):20528–20538. doi: 10.1073/pnas.1905973116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Tseng, W. C., et al. (2016). Kruppel-like factor 4 is a novel prognostic predictor for urothelial carcinoma of bladder and it regulates TWIST1-mediated epithelial-mesenchymal transition. Urologic Oncology, 34(11), 485 e15–485 e24. [DOI] [PubMed]
- 94.Qi XT, et al. KLF4 functions as an oncogene in promoting cancer stem cell-like characteristics in osteosarcoma cells. Acta Pharmacologica Sinica. 2019;40(4):546–555. doi: 10.1038/s41401-018-0050-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ma Y, et al. KLF4 inhibits colorectal cancer cell proliferation dependent on NDRG2 signaling. Oncology Reports. 2017;38(2):975–984. doi: 10.3892/or.2017.5736. [DOI] [PubMed] [Google Scholar]
- 96.Shum CK, et al. Kruppel-like factor 4 (KLF4) suppresses neuroblastoma cell growth and determines non-tumorigenic lineage differentiation. Oncogene. 2013;32(35):4086–4099. doi: 10.1038/onc.2012.437. [DOI] [PubMed] [Google Scholar]
- 97.Li X, et al. Klf4 reduces stemness phenotype, triggers mesenchymal-epithelial transition (MET)-like molecular changes, and prevents tumor progression in nasopharygeal carcinoma. Oncotarget. 2017;8(55):93924–93941. doi: 10.18632/oncotarget.21370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Tapia N, Scholer HR. p53 connects tumorigenesis and reprogramming to pluripotency. Journal of Experimental Medicine. 2010;207(10):2045–2048. doi: 10.1084/jem.20101866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Hoffmeyer K, et al. Wnt/beta-catenin signaling regulates telomerase in stem cells and cancer cells. Science. 2012;336(6088):1549–1554. doi: 10.1126/science.1218370. [DOI] [PubMed] [Google Scholar]
- 100.Dang CV. MYC on the path to cancer. Cell. 2012;149(1):22–35. doi: 10.1016/j.cell.2012.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Lin CY, et al. Transcriptional amplification in tumor cells with elevated c-Myc. Cell. 2012;151(1):56–67. doi: 10.1016/j.cell.2012.08.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Dreesen O, Brivanlou AH. Signaling pathways in cancer and embryonic stem cells. Stem Cell Rev. 2007;3(1):7–17. doi: 10.1007/s12015-007-0004-8. [DOI] [PubMed] [Google Scholar]
- 103.Noggle SA, Weiler D, Condie BG. Notch signaling is inactive but inducible in human embryonic stem cells. Stem Cells. 2006;24(7):1646–1653. doi: 10.1634/stemcells.2005-0314. [DOI] [PubMed] [Google Scholar]
- 104.Lowell, S., et al. (2006). Notch promotes neural lineage entry by pluripotent embryonic stem cells. PLoS Biology, 4(5), e121. [DOI] [PMC free article] [PubMed]
- 105.Liu, W., et al. (2013). Blockage of Notch signaling inhibits the migration and proliferation of retinal pigment epithelial cells. Scientific World Journal, 178708. [DOI] [PMC free article] [PubMed]
- 106.Vujovic F, Hunter N, Farahani RM. Notch pathway: A bistable inducer of biological noise? Cell Communication and Signaling: CCS. 2019;17(1):133. doi: 10.1186/s12964-019-0453-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Asnaghi L, et al. Notch signaling promotes growth and invasion in uveal melanoma. Clinical Cancer Research. 2012;18(3):654–665. doi: 10.1158/1078-0432.CCR-11-1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Hallahan AR, et al. The SmoA1 mouse model reveals that notch signaling is critical for the growth and survival of sonic hedgehog-induced medulloblastomas. Cancer research. 2004;64(21):7794–7800. doi: 10.1158/0008-5472.CAN-04-1813. [DOI] [PubMed] [Google Scholar]
- 109.McAuliffe SM, et al. Targeting Notch, a key pathway for ovarian cancer stem cells, sensitizes tumors to platinum therapy. Proc Natl Acad Sci U S A. 2012;109(43):E2939–E2948. doi: 10.1073/pnas.1206400109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Stylianou S, Clarke RB, Brennan K. Aberrant activation of notch signaling in human breast cancer. Cancer Research. 2006;66(3):1517–1525. doi: 10.1158/0008-5472.CAN-05-3054. [DOI] [PubMed] [Google Scholar]
- 111.Nowell CS, Radtke F. Notch as a tumour suppressor. Nature Reviews Cancer. 2017;17(3):145–159. doi: 10.1038/nrc.2016.145. [DOI] [PubMed] [Google Scholar]
- 112.Loh KM, van Amerongen R, Nusse R. Generating Cellular Diversity and Spatial Form: Wnt Signaling and the Evolution of Multicellular Animals. Developmental Cell. 2016;38(6):643–655. doi: 10.1016/j.devcel.2016.08.011. [DOI] [PubMed] [Google Scholar]
- 113.Xu Z, et al. Wnt/beta-catenin signaling promotes self-renewal and inhibits the primed state transition in naive human embryonic stem cells. Proc Natl Acad Sci U S A. 2016;113(42):E6382–E6390. doi: 10.1073/pnas.1613849113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Duchartre Y, Kim YM, Kahn M. The Wnt signaling pathway in cancer. Critical Reviews in Oncology Hematology. 2016;99:141–149. doi: 10.1016/j.critrevonc.2015.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Polakis, P. (2012). Wnt signaling in cancer. Cold Spring Harbor Perspectives Biology, 4(5). [DOI] [PMC free article] [PubMed]
- 116.Zhan T, Rindtorff N, Boutros M. Wnt signaling in cancer. Oncogene. 2017;36(11):1461–1473. doi: 10.1038/onc.2016.304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Borah, A., et al. (2015). Targeting self-renewal pathways in cancer stem cells: clinical implications for cancer therapy. Oncogenesis, 4, e177. [DOI] [PMC free article] [PubMed]
- 118.Krausova M, Korinek V. Wnt signaling in adult intestinal stem cells and cancer. Cellular Signalling. 2014;26(3):570–579. doi: 10.1016/j.cellsig.2013.11.032. [DOI] [PubMed] [Google Scholar]
- 119.Xue G, et al. Wnt/beta-catenin signaling in melanoma: Preclinical rationale and novel therapeutic insights. Cancer Treatment Reviews. 2016;49:1–12. doi: 10.1016/j.ctrv.2016.06.009. [DOI] [PubMed] [Google Scholar]
- 120.Sun J, et al. Low-level shear stress induces differentiation of liver cancer stem cells via the Wnt/beta-catenin signalling pathway. Experimental Cell Research. 2019;375(1):90–96. doi: 10.1016/j.yexcr.2018.12.023. [DOI] [PubMed] [Google Scholar]
- 121.Zhang K, et al. WNT/beta-Catenin Directs Self-Renewal Symmetric Cell Division of hTERT(high) Prostate Cancer Stem Cells. Cancer Research. 2017;77(9):2534–2547. doi: 10.1158/0008-5472.CAN-16-1887. [DOI] [PubMed] [Google Scholar]
- 122.Pohl, S. G., et al. (2017). Wnt signaling in triple-negative breast cancer. Oncogenesis, 6(4), e310. [DOI] [PMC free article] [PubMed]
- 123.Oren O, Smith BD. Eliminating Cancer Stem Cells by Targeting Embryonic Signaling Pathways. Stem Cell Rev Rep. 2017;13(1):17–23. doi: 10.1007/s12015-016-9691-3. [DOI] [PubMed] [Google Scholar]
- 124.Katoh M. Network of WNT and other regulatory signaling cascades in pluripotent stem cells and cancer stem cells. Current pharmaceutical biotechnology. 2011;12(2):160–170. doi: 10.2174/138920111794295710. [DOI] [PubMed] [Google Scholar]
- 125.Sari, I. N., et al. (2018). Hedgehog Signaling in Cancer: A Prospective Therapeutic Target for Eradicating Cancer Stem Cells. Cells, 7(11). [DOI] [PMC free article] [PubMed]
- 126.Li QR, et al. Novel-smoothened inhibitors for therapeutic targeting of naive and drug-resistant hedgehog pathway-driven cancers. Acta Pharmacologica Sinica. 2019;40(2):257–267. doi: 10.1038/s41401-018-0019-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Brewer BG, et al. Embryonic vaccines against cancer: An early history. Experimental and Molecular Pathology. 2009;86(3):192–197. doi: 10.1016/j.yexmp.2008.12.002. [DOI] [PubMed] [Google Scholar]
- 128.Jiang XP, et al. Vaccination with a mixed vaccine of autogenous and allogeneic breast cancer cells and tumor associated antigens CA15-3, CEA and CA125-results in immune and clinical responses in breast cancer patients. Cancer biotherapy & radiopharmaceuticals. 2000;15(5):495–505. doi: 10.1089/cbr.2000.15.495. [DOI] [PubMed] [Google Scholar]
- 129.Malatesta M, et al. Prostate-specific antigen synthesis and secretion by human placenta: A physiological kallikrein source during pregnancy. The Journal of Clinical Endocrinology & Metabolism. 2000;85(1):317–321. doi: 10.1210/jcem.85.1.6302. [DOI] [PubMed] [Google Scholar]
- 130.Gjerstorff MF, Andersen MH, Ditzel HJ. Oncogenic cancer/testis antigens: Prime candidates for immunotherapy. Oncotarget. 2015;6(18):15772. doi: 10.18632/oncotarget.4694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Skinner J, Whitehead R. Tumor-associated antigens in polyps and carcinoma of the human large bowel. Cancer. 1981;47(6):1241–1245. doi: 10.1002/1097-0142(19810315)47:6<1241::AID-CNCR2820470602>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 132.Han SN, et al. Physiologic variations of serum tumor markers in gynecological malignancies during pregnancy: A systematic review. BMC medicine. 2012;10(1):86. doi: 10.1186/1741-7015-10-86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Beaman KD, et al. Pregnancy is a model for tumors, not transplantation. American Journal of Reproductive Immunology. 2016;76(1):3–7. doi: 10.1111/aji.12524. [DOI] [PubMed] [Google Scholar]
- 134.Hepner A, et al. Cancer During Pregnancy: The Oncologist Overview. World J Oncol. 2019;10(1):28–34. doi: 10.14740/wjon1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Ouyang X, Telli ML, Wu JC. Induced Pluripotent Stem Cell-Based Cancer Vaccines. Frontiers in Immunology. 2019;10:1510. doi: 10.3389/fimmu.2019.01510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Camara DA, et al. Advances and Challenges on Cancer Cells Reprogramming Using Induced Pluripotent Stem Cells Technologies. Journal of Cancer. 2016;7(15):2296–2303. doi: 10.7150/jca.16629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Bindhya S, et al. Induced pluripotent stem cells: A new strategy to model human cancer. International Journal of Biochemistry & Cell Biology. 2019;107:62–68. doi: 10.1016/j.biocel.2018.12.008. [DOI] [PubMed] [Google Scholar]
- 138.Lee DF, et al. Modeling familial cancer with induced pluripotent stem cells. Cell. 2015;161(2):240–254. doi: 10.1016/j.cell.2015.02.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Kotini AG, et al. Functional analysis of a chromosomal deletion associated with myelodysplastic syndromes using isogenic human induced pluripotent stem cells. Nature Biotechnology. 2015;33(6):646–655. doi: 10.1038/nbt.3178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Kim J, et al. An iPSC line from human pancreatic ductal adenocarcinoma undergoes early to invasive stages of pancreatic cancer progression. Cell Reports. 2013;3(6):2088–2099. doi: 10.1016/j.celrep.2013.05.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Sommer, C. A., et al. (2018). Modeling APC mutagenesis and familial adenomatous polyposis using human iPS cells. PLoS One, 13(7), e0200657. [DOI] [PMC free article] [PubMed]
- 142.Chen, L., et al. (2012). A model of cancer stem cells derived from mouse induced pluripotent stem cells. PLoS One, 7(4), e33544. [DOI] [PMC free article] [PubMed]
- 143.Yan T, et al. Characterization of cancer stem-like cells derived from mouse induced pluripotent stem cells transformed by tumor-derived extracellular vesicles. Journal of Cancer. 2014;5(7):572–584. doi: 10.7150/jca.8865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Prieto-Vila M, et al. iPSC-derived cancer stem cells provide a model of tumor vasculature. American Journal of Cancer Research. 2016;6(9):1906–1921. [PMC free article] [PubMed] [Google Scholar]
- 145.Xu N, et al. Induction of cells with prostate cancer stem-like properties from mouse induced pluripotent stem cells via conditioned medium. American journal of cancer research. 2018;8(8):1624. [PMC free article] [PubMed] [Google Scholar]
- 146.Papapetrou EP. Patient-derived induced pluripotent stem cells in cancer research and precision oncology. Nature medicine. 2016;22(12):1392–1401. doi: 10.1038/nm.4238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Postovit LM, et al. A three-dimensional model to study the epigenetic effects induced by the microenvironment of human embryonic stem cells. Stem Cells. 2006;24(3):501–505. doi: 10.1634/stemcells.2005-0459. [DOI] [PubMed] [Google Scholar]
- 148.Giuffrida D, et al. Human embryonic stem cells secrete soluble factors that inhibit cancer cell growth. Cell Proliferation. 2009;42(6):788–798. doi: 10.1111/j.1365-2184.2009.00640.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Raof NA, et al. Bioengineering embryonic stem cell microenvironments for exploring inhibitory effects on metastatic breast cancer cells. Biomaterials. 2011;32(17):4130–4139. doi: 10.1016/j.biomaterials.2011.02.035. [DOI] [PubMed] [Google Scholar]
- 150.Zhou, S., et al. (2017). Reprogramming Malignant Cancer Cells toward a Benign Phenotype following Exposure to Human Embryonic Stem Cell Microenvironment. PLoS One, 12(1), e0169899. [DOI] [PMC free article] [PubMed]
- 151.Lan G, et al. Notch pathway is involved in the suppression of colorectal cancer by embryonic stem cell microenvironment. Oncotargets and Therapy. 2019;12:2869–2878. doi: 10.2147/OTT.S199046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Raof NA, Mooney BM, Xie Y. Bioengineering embryonic stem cell microenvironments for the study of breast cancer. International Journal of Molecular Sciences. 2011;12(11):7662–7691. doi: 10.3390/ijms12117662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Costa FF, et al. Epigenetically reprogramming metastatic tumor cells with an embryonic microenvironment. Epigenomics. 2009;1(2):387–398. doi: 10.2217/epi.09.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Kulesa PM, et al. Reprogramming metastatic melanoma cells to assume a neural crest cell-like phenotype in an embryonic microenvironment. Proceedings of the National Academy of Sciences. 2006;103(10):3752–3757. doi: 10.1073/pnas.0506977103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Illmensee K, Mintz B. Totipotency and normal differentiation of single teratocarcinoma cells cloned by injection into blastocysts. Proceedings of the National Academy of Sciences. 1976;73(2):549–553. doi: 10.1073/pnas.73.2.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Li Y, et al. Vaccination with human pluripotent stem cells generates a broad spectrum of immunological and clinical responses against colon cancer. Stem Cells. 2009;27(12):3103–3111. doi: 10.1002/stem.234. [DOI] [PubMed] [Google Scholar]
- 157.Zhang Z, et al. Vaccination with embryonic stem cells generates effective antitumor immunity against ovarian cancer. International Journal of Molecular Medicine. 2013;31(1):147–153. doi: 10.3892/ijmm.2012.1195. [DOI] [PubMed] [Google Scholar]
- 158.Wang, Q. T., et al. (2020). Tumor-associated antigen-based personalized dendritic cell vaccine in solid tumor patients. Cancer Immunology, Immunotherapy, 1–13. [DOI] [PMC free article] [PubMed]
- 159.Avigan D, Rosenblatt J. Vaccine therapy in hematologic malignancies. Blood. 2018;131(24):2640–2650. doi: 10.1182/blood-2017-11-785873. [DOI] [PubMed] [Google Scholar]
- 160.Wang Y, et al. Dendritic cell biology and its role in tumor immunotherapy. Journal of Hematology & Oncology. 2020;13(1):1–18. doi: 10.1186/s13045-020-00939-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Rami, F., H. (2016). Mollainezhad, and M. Salehi, Induced pluripotent stem cell as a new source for cancer immunotherapy. Genetics Research International. [DOI] [PMC free article] [PubMed]
- 162.Dhodapkar MV. Immunity to stemness genes in human cancer. Current Opinion in Immunology. 2010;22(2):245–250. doi: 10.1016/j.coi.2010.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Kooreman, N. G., et al. (2020). Autologous iPSC-Based Vaccines Elicit Anti-tumor Responses In Vivo.Cell Stem Cell, 22(4), 501–513 e7. [DOI] [PMC free article] [PubMed]
- 164.Zhang ZJ, et al. Human embryonic stem cells–a potential vaccine for ovarian cancer. Asian Pacific Journal of Cancer Prevention. 2012;13(9):4295–4300. doi: 10.7314/APJCP.2012.13.9.4295. [DOI] [PubMed] [Google Scholar]
- 165.Heront-Kishi, M., et al. (2020). Pharmacologically modified pluripotent stem cell-based cancer vaccines with anti-metastatic potential, bioRxiv.
- 166.Yaddanapudi K, et al. Exosomes from GM-CSF expressing embryonic stem cells are an effective prophylactic vaccine for cancer prevention. Oncoimmunology. 2019;8(3):1561119. doi: 10.1080/2162402X.2018.1561119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Zhu Q, et al. Embryonic Stem Cells-Derived Exosomes Endowed with Targeting Properties as Chemotherapeutics Delivery Vehicles for Glioblastoma Therapy. Adv Sci (Weinh) 2019;6(6):1801899. doi: 10.1002/advs.201801899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Gwak JM, et al. Expression of embryonal stem cell transcription factors in breast cancer: Oct4 as an indicator for poor clinical outcome and tamoxifen resistance. Oncotarget. 2017;8(22):36305. doi: 10.18632/oncotarget.16750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Cheng C-C, et al. Stat3/Oct-4/c-Myc signal circuit for regulating stemness-mediated doxorubicin resistance of triple-negative breast cancer cells and inhibitory effects of WP1066. International journal of oncology. 2018;53(1):339–348. doi: 10.3892/ijo.2018.4399. [DOI] [PubMed] [Google Scholar]
- 170.Ghanei Z, et al. Isolation and characterization of breast cancer stem cell-like phenotype by Oct4 promoter-mediated activity. Journal of cellular physiology. 2020;235(11):7840–7848. doi: 10.1002/jcp.29437. [DOI] [PubMed] [Google Scholar]
- 171.Shankar, S., et al. (2011) Resveratrol inhibits pancreatic cancer stem cell characteristics in human and Kras G12D transgenic mice by inhibiting pluripotency maintaining factors and epithelial-mesenchymal transition. PloS one, 6(1), e16530. [DOI] [PMC free article] [PubMed]
- 172.Jia Q, et al. Positive correlation of Oct4 and ABCG2 to chemotherapeutic resistance in CD90+ CD133+ liver cancer stem cells. Cellular reprogramming. 2013;15(2):143–150. doi: 10.1089/cell.2012.0048. [DOI] [PubMed] [Google Scholar]
- 173.Boumahdi S, et al. SOX2 controls tumour initiation and cancer stem-cell functions in squamous-cell carcinoma. Nature. 2014;511(7508):246–250. doi: 10.1038/nature13305. [DOI] [PubMed] [Google Scholar]
- 174.Xiang R, et al. Downregulation of transcription factor SOX2 in cancer stem cells suppresses growth and metastasis of lung cancer. British journal of cancer. 2011;104(9):1410–1417. doi: 10.1038/bjc.2011.94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Gangemi RMR, et al. SOX2 silencing in glioblastoma tumor-initiating cells causes stop of proliferation and loss of tumorigenicity. Stem cells. 2009;27(1):40–48. doi: 10.1634/stemcells.2008-0493. [DOI] [PubMed] [Google Scholar]
- 176.Lundberg IV, et al. SOX2 expression is associated with a cancer stem cell state and down-regulation of CDX2 in colorectal cancer. BMC Cancer. 2016;16(1):1–11. doi: 10.1186/s12885-016-2509-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Zhang P, et al. MicroRNA-10b regulates epithelial-mesenchymal transition by modulating KLF4/Notch1/E-cadherin in cisplatin-resistant nasopharyngeal carcinoma cells. American journal of cancer research. 2016;6(2):141. [PMC free article] [PubMed] [Google Scholar]
- 178.Shan J, et al. Nanog regulates self-renewal of cancer stem cells through the insulin-like growth factor pathway in human hepatocellular carcinoma. Hepatology. 2012;56(3):1004–1014. doi: 10.1002/hep.25745. [DOI] [PubMed] [Google Scholar]
- 179.Jeter CR, et al. NANOG promotes cancer stem cell characteristics and prostate cancer resistance to androgen deprivation. Oncogene. 2011;30(36):3833–3845. doi: 10.1038/onc.2011.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Yong X, et al. Helicobacter pylori upregulates Nanog and Oct4 via Wnt/β-catenin signaling pathway to promote cancer stem cell-like properties in human gastric cancer. Cancer letters. 2016;374(2):292–303. doi: 10.1016/j.canlet.2016.02.032. [DOI] [PubMed] [Google Scholar]
- 181.Avila JL, Kissil JL. Notch signaling in pancreatic cancer: Oncogene or tumor suppressor? Trends in molecular medicine. 2013;19(5):320–327. doi: 10.1016/j.molmed.2013.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Wang Y, et al. A side population of cells from a human pancreatic carcinoma cell line harbors cancer stem cell characteristics. Neoplasma. 2009;56(5):371. doi: 10.4149/neo_2009_05_371. [DOI] [PubMed] [Google Scholar]
- 183.Andrew, R., Hallahan, et al. (2004). The SmoA1 Mouse Model Reveals That Notch Signaling Is Critical for the Growth and Survival of Sonic Hedgehog-Induced Medulloblastomas. Cancer Research, 64,7794-7800. [DOI] [PubMed]
- 184.Wang L, et al. Inhibition of Notch pathway enhances the anti-tumor effect of docetaxel in prostate cancer stem-like cells. Stem Cell Research & Therapy. 2020;11(1):1–8. doi: 10.1186/s13287-020-01773-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Huang Q, et al. The carcinogenic role of the notch signaling pathway in the development of hepatocellular carcinoma. Journal of Cancer. 2019;10(6):1570. doi: 10.7150/jca.26847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Westhoff B, et al. Alterations of the Notch pathway in lung cancer. Proceedings of the National Academy of Sciences. 2009;106(52):22293–22298. doi: 10.1073/pnas.0907781106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Nguyen B-C, et al. Cross-regulation between Notch and p63 in keratinocyte commitment to differentiation. Genes & development. 2006;20(8):1028–1042. doi: 10.1101/gad.1406006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Koch U, Radtke F. Notch and cancer: A double-edged sword. Cellular and molecular life sciences. 2007;64(21):2746–2762. doi: 10.1007/s00018-007-7164-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Zhang M, et al. Does Notch play a tumor suppressor role across diverse squamous cell carcinomas? Cancer medicine. 2016;5(8):2048–2060. doi: 10.1002/cam4.731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Shah PA, et al. NOTCH1 Signaling in Head and Neck Squamous Cell Carcinoma. Cells. 2020;9(12):2677. doi: 10.3390/cells9122677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Sriuranpong V, et al. Notch signaling induces cell cycle arrest in small cell lung cancer cells. Cancer research. 2001;61(7):3200–3205. [PubMed] [Google Scholar]
- 192.Augert, A., et al. (2019). Targeting NOTCH activation in small cell lung cancer through LSD1 inhibition. Science signaling, 12(567). [DOI] [PMC free article] [PubMed]
- 193.Teglund, S., Toftgård, R. (2010). Hedgehog beyond medulloblastoma and basal cell carcinoma. Biochimica et Biophysica Acta (BBA)-Reviews on Cancer, 1805(2), 181–208. [DOI] [PubMed]
- 194.Bonifas JM, et al. Activation of expression of hedgehog target genes in basal cell carcinomas. Journal of investigative dermatology. 2001;116(5):739–742. doi: 10.1046/j.1523-1747.2001.01315.x. [DOI] [PubMed] [Google Scholar]
- 195.Berman DM, et al. Medulloblastoma growth inhibition by hedgehog pathway blockade. Science. 2002;297(5586):1559–1561. doi: 10.1126/science.1073733. [DOI] [PubMed] [Google Scholar]
- 196.Varnat F, et al. Human colon cancer epithelial cells harbour active HEDGEHOG-GLI signalling that is essential for tumour growth, recurrence, metastasis and stem cell survival and expansion. EMBO molecular medicine. 2009;1(6–7):338–351. doi: 10.1002/emmm.200900039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Liu S, et al. Hedgehog signaling and Bmi-1 regulate self-renewal of normal and malignant human mammary stem cells. Cancer research. 2006;66(12):6063–6071. doi: 10.1158/0008-5472.CAN-06-0054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Zhao C, et al. Hedgehog signalling is essential for maintenance of cancer stem cells in myeloid leukaemia. Nature. 2009;458(7239):776–779. doi: 10.1038/nature07737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Burns MA, et al. Hedgehog pathway mutations drive oncogenic transformation in high-risk T-cell acute lymphoblastic leukemia. Leukemia. 2018;32(10):2126–2137. doi: 10.1038/s41375-018-0097-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.di Magliano MP, et al. Hedgehog/Ras interactions regulate early stages of pancreatic cancer. Genes & development. 2006;20(22):3161–3173. doi: 10.1101/gad.1470806. [DOI] [PMC free article] [PubMed] [Google Scholar]