Highlights
-
•
Simple, yet flexible, model of human movement patterns.
-
•
Analytic formalism which can be used to derive important spatial scales.
-
•
Introduces a novel drift–diffusion approximation for stochastic reservoirs.
-
•
A new critical spatial scale predicted for helminth reservoirs of infection.
-
•
The necessary data needed to test these predictions is outlined in detail.
Keywords: Mathematical models, Spatial infection model, Control policies, Monitoring and evaluation
Abstract
The life cycle of parasitic organisms that are the cause of much morbidity in humans often depend on reservoirs of infection for transmission into their hosts. Understanding the daily, monthly and yearly movement patterns of individuals between reservoirs is therefore of great importance to implementers of control policies seeking to eliminate various parasitic diseases as a public health problem. This is due to the fact that the underlying spatial extent of the reservoir of infection, which drives transmission, can be strongly affected by inputs from external sources, i.e., individuals who are not spatially attributed to the region defined by the reservoir itself can still migrate and contribute to it. In order to study the importance of these effects, we build and examine a novel theoretical model of human movement between spatially-distributed focal points for infection clustered into regions defined as ‘reservoirs of infection’. Using our model, we vary the spatial scale of human moment defined around focal points and explicitly calculate how varying this definition can influence the temporal stability of the effective transmission dynamics – an effect which should strongly influence how control measures, e.g., mass drug administration (MDA), define evaluation units (EUs). Considering the helminth parasites as our main example, by varying the spatial scale of human movement, we demonstrate that a critical scale exists around infectious focal points at which the migration rate into their associated reservoir can be neglected for practical purposes. This scale varies by species and geographic region, but is generalisable as a concept to infectious reservoirs of varying spatial extents and shapes. Our model is designed to be applicable to a very general pattern of infectious disease transmission modified by the migration of infected individuals between clustered communities. In particular, it may be readily used to study the spatial structure of hosts for macroparasites with temporally stationary distributions of infectious focal point locations over the timescales of interest, which is viable for the soil-transmitted helminths and schistosomes. Additional developments will be necessary to consider diseases with moving reservoirs, such as vector-born filarial worm diseases.
1. Introduction and background
Defining the spatial scales over which transmission should be considered is a relatively recent research area in the context of neglected tropical diseases (NTDs), where much of the effort has been focused on the development of geostatistical methods for each disease in turn (Pullan et al., 2011, Schur et al., 2013, Pullan et al., 2014, Moyes et al., 2014, Karagiannis-Voules et al., 2015, Moraga et al., 2015, Hamm et al., 2015, OHanlon et al., 2016, Ciddio et al., 2017, Mari et al., 2017, Flueckiger et al., 2019). In particular, mathematical models of helminth transmission have only recently begun to incorporate the dynamical effects of human movement between reservoirs of infection (Ciddio et al., 2017, Vegvari et al., 2019, Hardwick et al., 2019, Hardwick et al., 2021).
Infection with parasitic worms, also known as helminths, is the causative agent of a number of neglected tropical diseases including soil transmitted helminthiasis, schistosomiasis and lymphatic filariasis. Following World Health Organisation (WHO) guidelines, drugs have been donated by pharmaceutical companies since 2007 (Brooker et al., 2004, Crompton, 2006, Ending the neglect and reaching, 2020; Progress reports of the london declaration; World Health Organization, 2012) to countries significantly affected by helminth-associated morbidity. Initially the aim of mass treatment was morbidity reduction, but over time this has evolved into reducing transmission (World Health Organisation, 2020). Control initiatives have been developed specifically to investigate the prospects for elimination as a public health problem in the long term (Halliday et al., 2019, Ásbjörnsdóttir et al., 2018).
Transmission of human helminth infections generally involves an environmental stage, rather than person-to-person transmission (see Fig. 1). In the case of STHs, these infectious focal points consistute eggs, or larval stages in the soil, that are either ingested or enter the body via skin penetration. Similarly, in the case of schistosomiasis transmission, these focal points exist in water sources where larvae, that are released by freshwater snails (intermediate hosts), penetrate the skin to infect human hosts.
Fig. 1.
Illustration of the transmission dynamics of human helminth infections via a defined focal point.
One possible means for controlling, e.g., STH or schistosomiasis transmission is the administration of antihelminthic drugs in a structured programme with repeated rounds of treatment – we shall refer to this means for control as ‘mass drug administration’ (MDA). Note that MDA does not require each individual to be tested prior to treatment itself. For this paper, let us define an evaluation unit (EU) as a specified population of people (often associated to a particular spatial region) which is used to evaluate the impact of a MDA control programme. Let us also define an implementation unit (IU) similarly, but for the purpose of implementing MDA. Note that for some NTDs, IUs and EUs differ.
It may be possible to minimise the effect human movement has on EUs by specifying a critical spatial scale around the focal points within a reservoir (and corresponding to a human community) over which one may sample and treat infections. At this scale (or larger), the number of migrants contributing to the reservoir per unit time is potentially low enough such that their effect may essentially be neglected. The present work demonstrates the existence of this critical scale, which is expected to vary between helminths and regions, by developing a spatial model of human movement between defined locations which is consistent with the current literature. Current frameworks for choosing an appropriate spatial scale at which programme managers can evaluate the impact of NTD programmes focus primarily on empirical correlations, with a limited reference to the more fundamental disease models which can generate them (Fronterre et al., 2020). In this paper, we shall hence look at the spatial scale of EUs through a more model-focused lens.
The distances travelled by human movement patterns exhibit certain known characteristics similar to those of known random walks, e.g., the same heavy-tailed distributions as those of Lévy flights (Raichlen et al., 2014, Rhee et al., 2011). Furthermore, in a variety of countries and levels of urbanisation, Kung et al. (2014) uses mobile phone data in many different countries to conclude that there is evidence for some universality in the distribution shape of daily work-home commute distances, which appear to fit a broken power-law. Note that the distributions are similar to, e.g., those observed for radial distance in Lenormand et al., 2015, Zhao et al., 2015, where these works make use of geolocated tweets and GPS data, respectively.
The findings in all cases above, and other examples of broken power-laws for human mobility in the NTD disease modelling literature (see, e.g., Cosner et al., 2009, Citron et al., 2020), motivates us to use a similar description to develop a simple model of human movement from households to focal points of infection. We also note that Ciddio et al., 2017, Mari et al., 2017 exhibit spatial NTD frameworks which utilise deterministic disease models. The fundamentals-based modelling approach we will take in this paper is distinct from these references because it is constructed in a fully individual-based stochastic manner.
In Section 2, we derive a broken power-law model with one jump per individual, which both replicates the distribution behaviour and also suggests a possible mechanism for its origin. The model we introduce, however, will remain flexible (by varying parameters) to other possible power-law descriptions for human mobility and so we will not be wholly reliant on one possible description. In Section 3 we briefly investigate a computationally-efficient extension to this model which uses an approximation to describe multiple jumps per individual for movement patterns with longer timescales – both of which might be important for an accurate representation of the relevant human behaviour.
Having developed our model and illustrated its possible extensions, in Section 4 we apply it to obtain a critical spatial scale at which the migration of individuals to focal points clustered as infectious reservoirs declines to a negligible level. This result is shown to vary with helminth species, human movement patterns and regional geometry of settlement patterns. The analysis also provides insight into the most important pieces of information which are necessary to build an accurate model of human migration patterns between reservoirs of infection. Collecting such information will be key to the success of future helminth control programmes in reaching their targets, e.g., achieving STH elimination as a public health problem (Helminthiases, 2012). Lastly, in Section 5, we discuss how the spatial extent of reservoirs of infection considered here should influence the scale of EUs for control programmes and conclude with a summary of our findings and prospects for future work.
2. A one-jump model
2.1. Locations of infectious focal points and households
There are important ethical implications to the public availability of data on both the spatial locations and movement patterns of individuals over various scales in time, especially in low- and middle-income countries (Taylor, 2016). Among these are data privacy and the potential identifiability of individuals through the use of ‘big data’ by governments, companies and universities, and the potential use of this information for non-research purposes. Therefore, access to such high-quality data for study is scarce. This represents a challenge for those concerned with the various consequences of such movements and their methods of model validation. In this paper, we present mathematical models that can be used in the absence of access to high-quality movement data. Model dynamics are implemented on the spatial patterns of, e.g., building locations, obtained from real-world datasets, e.g., the high resolution settlement layer dataset generated by the Facebook Connectivity Lab (High Resolution Settlement Layer (HRSL), 2016). Our methodology should allow researchers to draw useful conclusions on the impact of human spatial movement on NTD programmes from readily available geospatial data (coordinates of households, settlements and environmental features, such as water bodies). Other data sources, e.g., call data records (from mobile phones) and data from migration questionnaires in control studies, may be available under certain circumstances and can easily be integrated in our modelling framework. The models we introduce here should also be able to take this specific case-study information into account by appropriate parameter inference.
Let us first consider the embedding of spatial locations of households as points in a 2-dimensional flat space. In addition to these household points there will also exist focal points of infectious contact that depend on the helminth species, e.g., near buildings, water sources, parts of farmland with large concentrations of infectious material and households themselves. Choosing a particular focal point, we will use the observed spatial distributions of buildings in the high resolution settlement layer dataset (High Resolution Settlement Layer (HRSL), 2016) as a proxy for the spatial distribution of household locations around it. Therefore, the expectation of having nearby households or buildings within a radial distance of r from the focal point, i.e., , scales according to the following radially-dependent power law
| (1) |
It is important to note that, since at first we consider only a single focal point from which household distances are measured, the distribution of focal points themselves is not relevant to the calculation. The spatial distribution of focal points will, however, become relevant later on when we cluster them into reservoirs of infection and investigate their spatial extent. Note, however, that households themselves may be a reasonable tracer of the underlying distribution for infectious focal points of STH (Steinbaum et al., 2016, Steinbaum et al., 2019), though our calculations throughout will not depend on this possibility.
In the cases of STH and schistosomiasis transmission, although it is clear that the location and importance of their infectious focal points can vary over time – e.g., a chance decline in snail population for a particular location at the edge of a fresh water source leads to a drop in infectious spread – throughout this work, we shall assume that their spatial distribution is effectively temporally stationary over the timescales of interest. Note that for some NTDs, the infectious contact event locations can also be mobile, e.g., in the case of lymphatic filariasis transmission, where the larval stage of microfilariae enter the body by bites from infected mosquitoes – hence, the contact event locations will migrate with the human and mosquito populations.
By averaging over the observed point spread patterns in the high resolution settlement layer dataset, we obtain the following cumulative neighbouring point number distributions as a function of radial distance. This can be compared to the radial dependence of the power-law index we have quoted in Eq. (1). In order to give an indication of the effect of spatial heterogeneity in the distances between buildings for this averaged quantity, we have plotted some example lines (left column of plots) and the sample mean lines with root-mean-square deviation (RMSD) shaded regions of (right column of plots) from individual randomly sampled initial building locations for different countries in Fig. 2. In Central Malawi and Northern Benin, up to some substantial variance, below a transitioning radial scale , the index seems to have a mean of and above this scale the mean of the index begins a transition to . In the Central Ivory Coast, we see that the mean trend rapidly varies between and below the transition scale , and then at radial distances much greater than this scale most of the power-laws appear to begin a transition to . The substantial variation below the transition scale in the Ivory Coast is likely due to a greater degree of clustering on particularly small scales (note the drop towards before the transition scale indicates this as very few new points are added to the cumulative total for this range).
Fig. 2.
Left column: The cumulative number of buildings (vertical axis) below a given radial distance (horizontal axis) from sample locations in Malawi, Benin and the Ivory Coast obtained from the high resolution settlement layer dataset generated by the Facebook Connectivity Lab (High Resolution Settlement Layer (HRSL), 2016). Lines for different constant and values are provided only as rough indicators for comparison. Right column: The sample mean lines and root-mean-square deviation (RMSD) shaded regions of the power law index plotted against radial distance in each case. The RMSD was calculated by computing discrete derivatives between log-frequency bins as a function of log-radial distance. In each country, the collection of sample locations was randomly drawn from: the central region of Malawi, including Lilongwe; the wide northern region of Benin, spanning between Kandi and Djougou; and a wide central region of the Ivory Coast, including Yamoussoukro.
For all countries and initial locations in Fig. 2, we can confirm that the majority of the power-laws begin to converge to a mean of above a particular transition scale which varies substantially between location. This convergence reflects the emergence of statistical spatial homogeneity only on very large spatial scales, but this is clearly not present below in all cases. The power-law signature of statistical homogeneity being should become clear by noting that another way to generate it is to draw a Poisson point process with an intensity (the flat Euclidean measure in 2 dimensions). Note that statistical isotropy is also implicitly confirmed by choosing different locations for each line and observing the same behaviour up to some noise that is captured by the RMSD in the right column of plots. Deviations from this trend towards (particularly in the Ivory Coast) are also present in many other locations in the same countries, due to the presence of substantial global heterogeneity, e.g., through proximity to large voids in the distribution of points, which exist for geographic reasons.
2.2. Obtaining a model using maximum entropy arguments
In this section we provide a simple argument to generate the broken power-law frequency distribution of work-home commute distances (as observed and described in, e.g., Kung et al., 2014, Zhao et al., 2015) using maximum entropy arguments. The distributions of movement distances we derive will be used to model the migration of individuals from their household locations to a focal point of infection. In the absence of access high-quality site-specific data that we discussed in the previous section, we are using the work-home commuter distance distributions as a guide for the likely patterns of movement. Work-home commuter distances should be a reasonable proxy for the overall movement of individuals in many areas. However, the model that we develop in this section is adaptive and flexible to modification, even in circumstances where this proxy is inappropriate, we will be able to vary distribution parameters to quantify their impact on our conclusions.
For greater analytic insight in the expressions we derive in this section, and throughout most of the subsequent arguments made in this article, we shall now assume that Eq. (1) has a constant index . When considering the heterogeneity in the real power-law behaviour below in Fig. 2, this assumption will not be appropriate in all locations/situations. Note, however, the results we obtain in all cases may be easily numerically generalised to take into account complex transitions between different power-law behaviours, as are observed in Fig. 2. In addition, choosing several key values for constant (which fit within the range of observed power law behaviours) in what follows will aid with quantifying its impact on our conclusions.
Assuming that each individual jumps to a single new location and then returns back to their starting position over the course of each day, we may follow a simple argument that incidentally generates a distribution over travel distances which is consistent with those observed and described in, e.g., Kung et al., 2014, Zhao et al., 2015. Let us take the distribution of neighbouring points to be isotropic. Defining as the probability density function (PDF) of an individual jumping to a given location as a function of its radial distance, one may then transform the flat radial probability measure by a Jacobian like so
| (2) |
which accounts for the power-law density of points that may depart from linear scaling according to Eq. (1). Another way to view this rescaling is to consider the expectation of the probability measure for a radially-generated, statistically isotropic Poisson point process.
Note that if we additionally assume that the PDF of the jump distribution for each individual has a mean then the maximum entropy PDF is that of an exponential distribution , then by using Eq. (2) we can trivially demonstrate that the resulting normalised PDF is a Gamma distribution
| (3) |
One possible interpretation for here is a number, with physical units of distance, which quantifies the access of that individual to varying degrees of transportation methods that are themselves capable of travelling varying distances. To clarify, without access to any motorised transportation, an individual may receive a low value for , and in contrast, if an indvidiual has access to a vehicle then they may receive a larger value. Despite this simple suggested explanation, the presence of variable scales for human travel distance distributions is not contingent on access to vehicular transportation at all. Indeed, human walking patterns themselves are known to exhibit multi-distance-scale characteristics in many contexts – see, e.g., Reynolds et al. (2018).
Assuming that values may vary across the population of individuals with a distribution of known mean and expected scaling , the maximum entropy distribution for values of is another gamma distribution. Choosing specific parameter values, we therefore have
| (4) |
where the resulting marginal jump PDF is obtained by integration in the following way
| (5) |
where denotes the generalised beta prime distribution (or beta distribution of the second kind), which takes a form similar to that of a beta distribution. Note that, to avoid a divergent integral, one must specify that .
The main motivations for using maximum entropy probability distributions in the case above (and in general) include the beneficial properties that such distributions have when limited a priori information is available. In the present instance, these properties translate into distributions which account for the maximum amount of potential variability in outcomes while still satisfying the basic pre-defined classifying properties, e.g., the gamma distribution we have used above is maximally entropic under the condition that the mean and expected scaling of the distribution are known. Furthermore, it is often reasonable to expect that the steady states of many systems observed across the sciences are well-described by maximum entropy configurations.
We introduced another radial spatial scale (distinct from in the previous section) in Eq. (4) whose effect, in conjunction with and , we shall explore. In Fig. 3 we have plotted the marginal jump PDF given by Eq. (5) for a range of parameter values and . By inspection of Eq. (5), in the limit where the distribution scales as , whereas in the opposite limit the distribution exhibits a scaling . The scale of therefore plays the role of separating two regimes in the distribution of jump lengths for a given individual. For radial distances below , the geometric distribution of available spatial locations to jump to (which is encoded in the parameter) dominates the behaviour, whereas, for radial distances above , the predisposition of individuals to travel a given distance combines with the geometry of points to give the distribution behaviour and this is encoded in the parameter of the second gamma distribution in Eq. (4).
Fig. 3.
Plots of the marginal probability density as a function of radial distance (in units of km) generated using Eq. (5) for a range of and values for comparison.
So far we have not assumed that the parameter in the distribution over inverse-jump scales given by Eq. (4) is related to any purely geometrically-driven break in the power laws of Fig. 2. By comparing our observed power laws for the Ivory Coast with the observed work-home commuter distance distributions in Kung et al. (2014), for instance, we see that in general. This is perhaps unsurprising since the scale may arise due to the availability of easy access to vehicles that allow for longer-distance travel for either work or school, or water sources near a given settlement. Despite this fact, it is intriguing to note that the radial scale associated to the peak in clustering ( well below ) for the Ivory Coast dataset in Fig. 2 occurs at the same rough scale that is observed for in Kung et al. (2014). We leave the investigation into the potential link between these two scales for future work.
Despite statistical isotropy and homogeneity on large spatial scales, we have observed that the variation between the power-laws for cumulative building numbers observed in Fig. 2, and hence the index of Eq. (1), exhibits a large degree of location dependence between regions. In Fig. 4, we see the effect that this small-scale heterogeneity in the distances between buildings has on the probability density for our one-jump model. We plot a comparison between the binned frequency of individuals (with exponential jump distributions each with a scale drawn from Eq. (4)) simulated to travel (solid lines) between two buildings on real-world map data. The data for building locations corresponds to the same regions of each country as in Fig. 2, and the marginalised jump probability densities derived from Eq. (5) for choices of . In all cases we have fixed to a single value for easier comparison of different lines; this value is approximately the same as the tails observed in the distance distributions of Kung et al. (2014).
Fig. 4.
The binned frequency of individuals simulated to travel a between two buildings (solid lines) whose locations correspond to data from the same regions of each country as in Fig. 2 (where some globally-applied uniform random point density thinning has been used to reduce computational load of exploring the distribution tails). This corresponds to the same marginalised jump probability density as Eq. (5) but instead of using a geometric power-law given by Eq. (1) we have implemented our single jumps on real-world map data (with exponential jump distributions each with a scale drawn from Eq. (4)). For comparison, we have also plotted some jump probabilities calculated using Eq. (5) with (dashed lines) and (dotted lines). In all cases we have fixed .
The agreement between the curves and the distances generated from single simulated jumps between buildings on the real-world map data appears to be variable between countries. Overall, it is unsurprising that the shapes of the curves all seem to agree for scales , because the asymptotic behaviour dominates in all cases, which is consistent with our model. In the opposing limit, however, the small-scale variability in power laws for each country dominates and the departure from our constant- models appears more pronounced – in favour of more transitory distance-dependent behaviour. Note also that the range in variability that the region between our and lines can account for is quite substantial. Such a range in behaviour motivates the use of both and in quoting the most important results that follow so as to correctly assess the impact of small-scale region-specific building distributions.
3. Multi-jump processes
In the previous section, a single jump model from a household to an infectious focal point location was developed in order to describe the movement of individuals between locations before contributing infectious material. An important caveat to this treatment of movement patterns is that one must also consider the possibility that multiple movements are performed either: between buildings before making a contribution to a focal point, or between infectious focal points themselves, supplying each some portion of infectious material in turn. The latter of these will be very difficult to quantify without some form of data, but for the former, we can explore some potential modifications to our model.
In this section, we will briefly explore an extension to the basic model of human movement presented in Section 2 to include successive jumps by an individual to multiple buildings before reaching an infectious focal point. The type of human movement we aim to capture is not just that of daily commuting, but also potentially longer distance travel across multiple days/weeks. The latter form of movement can contribute to the variability in migrant seasonal labour or family visits which can change the population numbers in a given region on a yearly timescale.
3.1. Homogeneous randomly-directed jumps
Following along similar lines to Section 2.2, let us now consider an individual who completes multiple successive jumps with exponentially-distributed lengths (where we have indexed each successive jump event with an i) with a fixed mean scale within a day. For a fixed jump rate in time, consider the following compound Poisson process for a two-dimensional vector which encodes the 2-dimensional Euclidean coordinate position of an individual over time
| (6) |
| (7) |
| (8) |
where is the uniform distribution PDF (assuming statistical isotropy) over the change in angle and is a Heaviside function. In Eq. (6) above, note that we are using an indicator function which takes value unity if , else zero.
The process specified by Eq. (6) assumes isotropy of both the geometric distribution of the points and a uniform-random direction choice of the individual. The distribution is not known as a closed-form expression, however, it clearly has both a vanishing first moment
| (9) |
and a second moment which scales according to
| (10) |
Furthermore, with a scale distribution over jump lengths applied – as in Eq. (5) – the second moment given in Eq. (10) becomes
| (11) |
In the left panel of Fig. 5 we have plotted the binned frequency of total daily distances travelled by a population of individuals following the process defined by Eq. (6) and drawing each individual’s inverse-jump scale from Eq. (4). We have also fixed per day (in red and black lines, respectively), and as indicated by the increasing opacity within each triplet of lines. Contrasting these distributions, it is immediately clear that by increasing the jump rate , the effect on the distribution of daily distances travelled is similar to increasing the value of – see the middle panel of Fig. 3 for comparison.
Fig. 5.
Numerical plots of the distance distributions generated by the multi-jump processes introduced in Section 3.1 with randomly-directed (left panel) and in Section 3.2 with unidirectional jumps (right panel). In the left panel, a population of individuals following the process defined by Eq. (6) have been drawn (drawing from Eq. (4) for their jump scale predispositions) and the binned frequency of the total distance evaluated at the end of the day with jump rates of per day (in red and black lines, respectively), and as indicated by the increasing opacity within each triplet of lines. In the right panel, the marginal probability density given by Eq. (15) is depicted with jump rates of per day (in red and black lines, respectively), and as indicated by the increasing opacity within each triplet of lines.
3.2. Homogeneous unidirectional jumps
Human movement patterns are not truly random, and instead, one might anticipate a strong directional dependence, e.g., to long distance travel of individuals over the course of multiple days to an intended destination. Due to this fact, let us try to quantify the effect of this behaviour by assuming the logical extreme, i.e., that the angular direction of an individual’s long-distance travel is fixed to a particular value. Following multiple successive jumps while assuming no variation in angle, Eq. (6) then becomes a compound Poisson process for the total jump distance in a single dimension. This process has a known characteristic function
| (12) |
where denotes the characteristic function of the stationary increments given by Eq. (3). Hence, we may write
| (13) |
which has no closed-form inverse transformation, but is useful as an expression to calculate the moments. Alternatively, in order to find the distribution over , we may marginalise over the number of jumps performed (which is Poisson-distributed by construction), which are themselves drawn from the jump distance distribution in the following way
| (14) |
Therefore, if , Eq. (14) yields
| (15) |
where is the modified Bessel function of the first kind.
In the right panel of Fig. 5 we have plotted the marginal probability density given by integrating Eqs. (15), (4) over for direct comparison with the one-jump model of Eq. (5). We have used values of per day (in red and black lines, respectively), and as indicated by the increasing opacity within each triplet of lines. Comparing this distribution with the one observed for randomly-directed multiple jumps (see Section 3.1), a similar, but even more pronounced, effect by varying is observed – since only a value of is required to achieve roughly the same change. This is to be expected, as the unidirectional jumpers will always travel at least the same (or more often) a greater distance in total in comparison with the randomly-directed ones, which will likely induce a more severe deformation of the overall distribution when using the former.
The parameter degeneracy between of the one-jump model and in the multi-jump processes described above suggests a test can be performed, to either validate a model, or discard it in favour of a modified version for a given situation with real data. By combining collected data on the total distance travelled per day and the local geometry of distances between buildings travelled to in a given region, the jump rate for each individual (if the same) could be statistically inferred. Comparing this inferred value to real data would provide a test of the movement models we have suggested in this work or potentially provide insight into where they may be improved to better reflect real human daily movement in a given setting.
The simple multi-jump models we have considered in this section have assumed that the distribution for the cumulative number of nearby buildings is homogeneous for each new jump. This approximation is not likely to work well on real-world map data as we have shown there is substantial small-scale heterogeneity in the distances between buildings exhibited in, e.g., Fig. 2. Furthermore, if the multi-jump movement model is purely diffusive over the building locations themselves (itself an assumption which must be investigated further), then there will be correlations in the location distributions between successive jumps that can lead to the distribution of jump distances exhibiting localising effects akin to those exhibited by power-law random banded matrices (Mirlin et al., 1996). In other words, when an individual jumps from a cluster of points to, e.g., a point on the edge of their cluster, then they may experience a stronger ‘pull’ back towards the centre of this cluster on the next jump due to the anisotropy in their apparent local point density. This effect should be investigated in future work to evaluate its relevance to movement patterns.
A more sophisticated method to modify the model and deal with (uncorrelated) small-scale heterogeneity could be to sample from the numerically-obtained power laws from Fig. 2 to emulate the spatial heterogeneity in building distances on the real-world map data directly in the processes of Section 3. This method would have significant advantages, in terms of computational complexity, to direct simulation methods over the real world map data.
4. The effect on reservoirs of infection
4.1. A coarse-grained stochastic reservoir network
Consider a group of focal points that are clustered into a ‘reservoir of infection’. Such a cluster is a source of infectious material from which disease-free individuals are exposed to new infections and, for those already with infections, to potentially increase them in intensity. With each of these clusters, let us now also associate a population of individuals with household locations that are sufficiently close such that the majority of their infectious inputs must be into one of their associated reservoir’s focal points.
Let us now denote the set of indicies which indentify all individuals who live in households that are less than a radial distance away from a focal point that is included in the reservoir cluster as . Additionally, to make a reservoir cluster, let us use as a radial separation distance threshold below which any two focal points are clustered together to be part of the same infectious reservoir. For reservoirs of infection of either schistosomiasis or STH, assuming that the associated human population number (the number of elements in ) does not change, the force of infection (FOI) at time t is updated according to
| (16) |
where is the death rate of the infectious material, is the worm death rate, d is the human death rate,1 is the basic reproduction number and is a compound Poisson process associated to the time-dependent infectious material input of the i-th individual. Since the helminths we study here are dioecious, the definition for differs from its standard interpretation in standard microparasite diseases. Here, it is the average number of female parasites produced by a female worm that themselves infect hosts and survive to reproductive maturity in a susceptible host population in the absence of density-dependent processes (Anderson and May, 1992, Truscott et al., 2016).
By clustering focal points into a reservoir of infection by using a radial distance threshold, one might reasonably question the scaling in the spatial extent of the infectious focal points themselves. As was discussed in Section 3, the distribution of focal points of infection is likely to be both helminth species-dependent and variable according to local geographic considerations. Due to this variability, the spatial scales associated with clusters of focal points themselves will likely vary across a map. However, this fact will not directly affect the conclusions of the present work since we will consider distances between each individual focal point and its neighbouring households in turn. For specific case studies, the implementation of our algorithm for binding focal points together should be well-defined in most cases for STH and schistosomiasis transmission settings, as long as the distribution of focal points can be inferred. Recall also our previously stated point that, for STH, our household distribution itself could potentially be a tracer for many of the focal points of infection (Steinbaum et al., 2016, Steinbaum et al., 2019).
A more important effect, which is indirectly related to the variability in spatial scale of focal point clusters, is how variable cluster sizes might induce internal variability in the exposure of each individual to new infections. In models of helminth transmission (Anderson and May, 1982), it is common to account for the variability of worms within hosts through assigning each individual a predisposition factor which is drawn from a gamma distribution, , where k is the ‘aggregation parameter’ which modifies the variance-to-mean ratio of the distribution of worms within hosts. Due to the epidemiological processes that it aims to capture – and assuming that the basic reproduction number , which accounts for the transmission intensity, remains unchanged – this aggregation parameter will likely vary to capture a change in exposure according to the spatial scale chosen for the reservoir of infection. One simple model to capture this variability in exposure might be to consider the value of k itself to be drawn from a gamma distribution, i.e., from which samples are drawn across the map for the individuals closest to each focal point of infection. When binding such focal points together to get the global behaviour of the reservoir of infection, one would then have a mixture model of separate behaviours to consider in the distribution of worms within hosts, affecting the summation term of Eq. (16). Since variability of exposure with spatial scaling is not the main focus of the present work, we shall not investigate this any further here. However, due to its obvious importance to the dynamical behaviour of the reservoir by association, it will be an important component to be cognisent of in future work.
In Hardwick et al. (2021) it was shown that, to good approximation for moderate-to-large population numbers, the worm burden distributions of individuals evolve according to the following reservoir birth–death process master equation
| (17) |
The solution to this equation is a Poisson distribution with time-dependent intensity
| (18) |
in which we have inserted the solution to Eq. (16). Summing over an ensemble of these individuals to get an overall distribution of worms within hosts, many epidemiogical variables, e.g., the prevalence of infection or mean parasite burden of hosts associated to the reservoir can be calculated (up to specifying the additional gamma-distributed predisposition to infection for each individual) while maintaining the finite population variance neglected by deterministic disease models. Therefore, in order to understand how these variables which are associated with each reservoir of infection are affected by spatial movements of human hosts, it is essential to correctly assess how is modified by inward migration.
Due to the movement patterns we have discussed, there may be individuals who contribute to one of the focal points of the reservoir but come from a region further away such that they are not counted in . There may be individuals who are counted in but instead contribute to a focal point outside of their own associated cluster, e.g., on a particular day. Eq. (16) will hence be perturbed by these movements in the following way
| (19) |
where are also compound Poisson processes which sum over the amount of infectious material generated by these migrating individuals that is either net entering (+) or leaving (−) the reservoir focal points.
Eq. (19) implicitly describes a network of spatially coarse-grained reservoirs of infection, each of which varying in spatial extent according to a combination of the distribution of their infectious foci, as well as the spatial coarse-graining scale choice . This network of reservoir ‘nodes’ is connected by migratory ‘pulses’, continually affecting each reservoir’s temporal stability (which we shall discuss later on) as well as potentially non-negligible statistical cross-correlations in local epidemiological indicators (e.g., prevalence or intensity of infection) with respect to other reservoirs. Note also that the choice of can therefore control the strength of these migratory interactions between reservoirs in the network. In Fig. 6 we have ilustrated this point explicitly on a digram with real map data based on buildings in central Malawi. The black (red) arrows on the map and zoomed illustrations show migratory movements of a radial distance below (above or equal to) , which has been arbitrarily chosen for clear visual effect.
Fig. 6.
A diagram of real map data (based on buildings in central Malawi) and a zoomed illustration which indicate the coarse-graining procedure of Eq. (21) – in which the black arrows are ‘removed’. Arrows depict individual movements from households (grey hollow dots on the right hand side zoomed illustration) to focal points of infection (black hollow dots on the right hand side zoomed illustration) where black arrows on both the map and its zoomed counterpart correspond to jumps over a radial distance (where is the spatial coarse-graining scale) and red arrows correspond to jumps over a radial distance . On the left hand side zoomed illustration we see the emergence of the spatially coarse-grained reservoir network ‘nodes’ (nodes are neither drawn to scale nor have geometrically-accurate reservoir spatial shapes) connected by red migratory ‘links’ in time.
Let us now define as the average rate of individuals travelling greater than a distance of to a focal point of the reservoir of infection and contributing material to it. Following Hardwick et al. (2021), this translates into the following decomposition of the positive migratory process in Eq. (19)
| (20) |
where is the contribution to the infectious material quantity of a focal point by an individual: a random variable with a distribution which varies between disease, but likely follows a negative binomial character in some cases. This distribution may be specifically derived by transforming the travelling individual’s worm burden (e.g., drawn from a Poisson distribution and Eq. (18) or an individual-based simulation) into an expected count of fertilised eggs or larvae using a helminth-specific mating function (Anderson and May, 1992). Note that this compound Poisson process is exact since it tracks the contribution entering the reservoir directly. If one wishes to temporally coarse-grain over the timescale and see the effect on the mean worm burden deterministic ODE model (as is done in Hardwick et al., 2019), an additional non-Markovian component arises to account for the reservoir pulse decay.
When an individual contributes to an infectious focal point which is further than away from their household, they do not just make an additional contribution to the latter’s reservoir of infection, but they also remove their contribution from one of the focal points which is within of their household (their local reservoir). In this sense, each random pulse within the process of one reservoir is exactly correlated to a pulse within the process of another’s. So, for model completeness, it is sufficient to define only the process (attributing an intended location of travel) for each reservoir of infection defined over a map. This is tantamount to defining ‘links’ between the coarse-grained nodes of the network illustrated by Fig. 6.
Let us now consider the influence that the coarse-graining procedure has on Eq. (20). By analogy to physical theories in nature, note that the fundamental dynamical theory should not change – e.g., individual contributions to each reservoir should not ‘disappear’ – simply because of an arbitrary choice for , and so one must define a procedure whereby the effective dynamics in Eq. (20) are always describing the same fundamental phenomenon, for any choice of . An obvious way to do this is to consider the variation in probability mass associated to individual movement rates as is varied. Based on the arguments of Section 2, we hence point out that the following scaling should exist for which accounts for this difference in probability mass arising from a rescaling of distances
| (21) |
where we have defined as the average rate of individuals travelling greater than a distance of to a focal point of the reservoir of infection and contributing material to it.
In order to derive Eq. (21) we have assumed that all individuals jump once per day, so that the marginalised jump PDF of Section 2 may be used directly. By integration of Eq. (21), one generally obtains
| (22) |
where is the (Gauss) hypergeometric function. In Fig. 7 we use the solution given by Eq. (22) to compute the quantity as a function of for a range of parameter choices.
Fig. 7.
The spatially coarse-grained average daily pulse rate into a focal point of infection as a fraction of its value from individuals arriving a distance of or greater away. This value is plotted as a function of the radial coarse-graining scale ratio used. The relationship is given by Eq. (22) for a range of and power-law parameters.
The variation of as a function of provides a mapping between the average pulse rate for Eq. (20) that is associated to each focal point within the reservoir of infection (whose pulse amplitudes can be easily summed over to get a total contribution to and from the reservoir) and the distribution of households at different spatial coarse-graining scales. The value of may hence be fixed to identify movement of individuals between structures at different scales, e.g., buildings, villages, towns, cities, etc. We may visualise this coarse-graining of movement distances by referring back to Fig. 6 – there is an emergence of black clusters of connected locations which are connected by red arrows at longer distances. By then clustering the focal points on this map (note this is a mock illustration and these are not real focal points) according to whether or not they are within a radial distance of one another, we can then build coarse-grained reservoirs of infection associated to the scale .
It is important to note that infectious reservoir clusters may themselves have a significant spatial extent due to the particular geometry of the nearby focal points, and so should be considered to apply to each and every focal point (or small clusters of focal points) within the reservoir individually. Hence, for reservoirs of infection which correspond to clusters of focal points which, when bound together, have a radial spatial extent that is much smaller than then the results of this section may be used directly for the entire reservoir. In situations where this is not the case – e.g., long and thin geometric relationships binding focal points around the edge of a lake – then our results may still be applied to each focal point (or small cluster of focal points) in turn. The application of our more general formalism is then subject to change depending on the specific relationships between focal points and hence, indirectly, on the species of helminth.
4.2. Reservoir Wiener process approximation
Reconsidering Eq. (16), note that the following Gaussian sample mean approximation applies for the terms summing over all (non-migratory) infectious material input into the reservoir
| (23) |
where is a Wiener process in time, is a time-dependent mean and is a time-dependent variance for the reservoir inputs. Such an approximation is motivated by the central limit theorem and will be most accurate in the limit of large population number . By inserting Eq. (23) into Eq. (19), one finds a drift–diffusion which satisfies the following Gaussian distribution for
| (24) |
Calculating an exact analytic form for in the case of each helminth species is still an open research question since this is very likely to be quite complex to model. For some analytic insight, however, in Hardwick et al. (2019) it was shown that, with a negative binomial distribution of worms within hosts, the distribution over typical egg outputs from hosts for hookworm is well-approximated by another negative binomial (with different mean and variance). Under these conditions, one may infer, from the sum of negative binomial variances, that
| (25) |
where is the aggregation parameter for the reservoir input negative binomial, which can be time-dependent before the system relaxes to the endemic steady state configuration.
Eq. (24) provides a significant improvement in computational efficiency over a full stochastic simulation. In particular, for endemic steady-state regions where the constant values of and can be computed quickly for each parameter configuration, it could be used to generate a simulation likelihood for statistical inference over an entire map affected by helminth transmission and migrating infected individuals.
Due to migratory inputs from (and outputs to) other reservoirs, applying Eq. (24) consistently to each node of our reservoir network will require the inclusion of spatial covariances (or higher-order statistics) between samples. The strength of these spatial covariances can, in principle, be computed by including the migratory terms of Eq. (19) into the approximation above. We can see this by recalling that the rate of these migratory compound Poisson processes (see Eq. (20)) is fundamentally connected to the spatial distance scales via Eq. (22). This ‘fundamentals-based’ method of spatial epidemiological inference for helminth transmission would be distinct from other methods since it would not only include important effects which are inherent to a simulation, such as finite population variance of the underlying stochastic process, but also a theoretical understanding of how the effective dynamical description itself changes with spatial coarse-graining into separate EUs. Note that the ‘scale-invariance’ of the theoretical model we have suggested here can therefore, in principle, be inferred with data collected at one EU scale and then interpolated/extrapolated for model predictions at another.
4.3. The critical spatial scale
In order to evaluate the success of control measures through the calculation of epidemiological observables, such as the prevalence or intensity of infection, it is common for an EU to correspond to a particular spatial scale for the reservoir of infection – see, e.g., Moyes et al., 2014, Criscione et al., 2010, Steinbaum et al., 2017. Therefore, an evaluation unit (or the spatial extent of MDA) needs to be large enough to include individuals migrating into the region and contributing infectious material to the reservoir.
It was shown in both Hardwick et al., 2019, Hardwick et al., 2021 that if the average rate at which reservoir pulses occur exceeds the average death rate of the infectious material in the reservoir, , then the effect of infected human migration on the transmission dynamics becomes particularly strong. In particular, the authors of Hardwick et al. (2021) demonstrate that, for a range of finite population sizes, migration rates at or above this critical rate into a reservoir of infection can restart transmission in regions where a mass treatment programme has already achieved transmission elimination. In addition, it was shown that this rate of migration can stabilise the stronger ‘fade-out’ effects exhibited near transmission breakpoints. By combining the concept of a critical rate with our proposed spatial model for migration between reservoirs, one may derive a new and important critical spatial scale around the focal points of infectious reservoirs which depends on: the type of helminth, the small-scale geometry of locations and the specific patterns of human movement. This spatial scale can be used to define the geographical size of EUs.
If the spatial scale of an EU is chosen which corresponds to a region smaller than the critical scale around each of its focal points of infection, the average daily rate of pulses will always be larger in magnitude than the value , calculated using the critical scale. This is because the curves shown in Fig. 7 are always decreasing with increasing radial scale. This implies that when regions around focal points are defined at this critical scale or smaller for an EU, pulses of the form given by Eq. (19) cannot be safely neglected and should become important to take into account when modelling the transmission dynamics to correctly assess reservoir stability and possible observation biases. This means that if EUs are at this critical scale, or smaller, NTD programmes need to have measures in place to mitigate the impact of human movement on programme targets. For example, individuals who regularly move beyond the boundaries of EUs should be specifically targeted for treatment. When defining an EU in the opposite case, however, effects from migration between reservoirs of infection may be safely neglected.
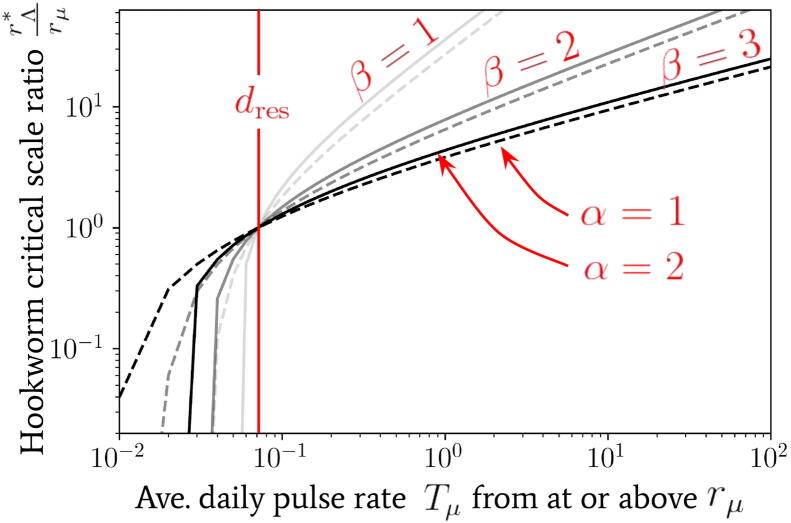
Using Eq. (22) the critical spatial scale at which becomes equal to the death rate of the infectious material in the reservoir per day, i.e., , has been plotted for hookworm (which has Truscott et al., 2016) in Fig. 8 as a function of the average daily pulse rate from individuals travelling distances at or above . In this plot we note that nearly all parameter combinations indicate a sharp decline in the critical scale ratio if is found to be below – where, in particular, for values of the scale ratio appears to fall extremely sharply and hence one can no longer find a finite critical scale below which migration becomes important.
Fig. 8.
The critical spatial scale as a ratio at which , for hookworm – where (Truscott et al., 2016) – plotted as a ratio of . The value of this scale is shown against the value of the average daily pulse rate at or above . We have used Eq. (22) to generate this relationship with a (bisection) root-finding algorithm.
In contrast, for values of increasing orders of magnitude, one infers from Fig. 8 that the value of is nearly irrelevant (which follows from the construction of ) and, particularly for , the increase in the critical scale ratio is very gradual. Such a relationship is also consistent with our expectation as the decline in the average pulse rate from people travelling from distances is particularly sharp for – see, e.g., Fig. 7 – and so one requires a significant increase in the amplitude of to achieve a significant change in the critical scale.
5. Discussion and conclusions
In this work we have developed and studied spatial models of human movement to infectious focal points – the important drivers of new infections for many parasitic organisms in humans. Our movement models have been motivated by the observed patterns of human mobility in, e.g., Kung et al., 2014, Zhao et al., 2015, but remain robust to setting-specific parametric variations should these observations prove unsuitable for all settings.
At its most basic, our model of human mobility assigns a single daily journey (or ‘jump’) for each individual, however, we have discussed many extensions to this description, which include: multiple Poisson-distributed jumps, each of which may either be in random directions or unidirectional; and substantial local heterogeneity in the distribution of distances between available travel locations. By combining these extensions into a generalised framework, one can generate a wide variety of possible daily human movement behaviours to/between focal points of infection under a unifying description which offers a model-focused explanation for the observed patterns that is also computationally efficient. In future work, it will be interesting to extend our approach to include time dependence in the location of infectious contact events. Such a model will be necessary for diseases such as lymphatic filariasis or onchocerciasis, where bites from mosquito or black fly vectors infected by filarial larvae can vary spatially over time, and may not be Markovian (i.e. memoryless). An investigation into the longer-term temporal changes in population number, e.g., due to seasonal migrant labour patterns or population displacement due to conflict or climate change, may also be interesting.
We note here that the human movement models we have studied are comparable to those developed in Cosner et al., 2009, Citron et al., 2020 for vector-bourne diseases, which label the single daily journey models as so-called ‘Lagrangian’ mobility models and the multiple successive movement models as ‘Eulerian’. We consider our stochastic approach to generating frequency-distance distributions useful and informative in understanding the underlying probability distributions for these works, especially in relation to demonstrating the regimes in radial spatial scales which are relevant to location-specific geometric effects and human movement distance predispositions.
By clustering focal points into reservoirs of infection, our human movement models have also highlighted the most important information necessary to efficiently (by which we mean low numbers of parameters) describe the effect of migration patterns on these reservoirs. Our model defines a stochastic network of dynamical reservoirs linked by migratory ‘pulses’ (see Eq. (19)) which accounts for these migratory movements at any defined spatial evaluation scale around each focal point using the movement models that we have developed. The necessary data to infer such a ‘reservoir network’ can, in principle, be collected by well-designed field studies. Based on our work here, the information to obtain for a particular study may include:
-
1.
Obtaining the appropriate (likely radially-dependent) power-law for the expected cumulative number of locations available to travel to by an individual – see, e.g., Fig. 2. These data can be obtained from geographic maps.
-
2.
Evaluating the amplitude of the effect of small-scale heterogeneity (and perhaps anisotropy) on the power-law cumulative distribution of locations available to travel to by an individual. One can obtain this information from high resolution maps of households.
-
3.
Obtaining the average number of journeys performed within a single day per individual. In this case some metric for having ‘completed’ a journey associated to the time spent at each location will likely need to be determined. In principle, this information can be obtained from questionnaires on human movement that can be integrated into a regular field survey.
-
4.
Evaluating the average number of people entering or leaving the relevant focal points for a given reservoir of infection at some particular spatial scale, e.g., this can be or evaluated at some other spatial scale since the reasoning of Eq. (21) can be easily adapted. Note that the true spatial scale of the reservoir does not need to be known for collecting these data. This information might be obtained through questionnaires.
-
5.
Potentially obtaining an appropriate scaling value for the distribution of predispositions to jump a certain distance (see Eq. (4)). This may either be consistent with the observed work-home commute patterns in Kung et al. (2014), or deviate to a different power-law relationship entirely, depending on local access to vehicles and other forms of transportation. This information might also be obtained through questionnaires.
-
6.
Obtaining the distribution of focal points themselves. This may be obtained through spatial surveys of the region of study.
While developing our stochastic reservoir network model, we also discussed a computationally-efficient approximate description for its dynamical behaviour near or at endemic steady states. In particular, a Gaussian sample mean approximation was made for the sum of infectious material in each reservoir over a coarse-grained temporal scale, rendering its dynamical behaviour the same as a drift–diffusion process with migratory ‘jumps’ – see Eq. (24). The drastic improvement in computational efficiency that such an approximation offers makes it an attractive option to explore when statistically inferring fully-stochastic infectious reservoirs from diagnostic data. We leave the implementation of this approach, and the analysis of its robustness for reservoir network states far from endemicity, to future work.
Although it is beyond the scope of this paper, in future work, it will be of interest to compare the results obtained from other NTD spatial epidemiological models – which typically employ deterministic models a particular spatial scale, e.g., Ciddio et al., 2017, Mari et al., 2017 – with the spatial correlations induced in the diagnostic output from our suggested approach. Such a comparison would help quantify the importance of finite population variance and EU choice on reservoir temporal stability. Other NTD spatial modelling approaches one might consider a comparison to are spatial GRF-based models (Moraga et al., 2015) – these are to be constrasted with the temporally Gaussian, but spatially complex, power-law-like, reservoir network we have proposed in this section. In addition, it would be interesting to explore how the approximate temporal Gaussianity of the model we have proposed in Eq. (24) changes when considering: dynamics further from endemic steady states; configurations closer to transmission breakpoints; or systems with significantly smaller population sizes (Hardwick et al., 2021).
The last, and perhaps most significant of our findings in this work concerns the spatial scales associated to EUs. An EU is commonly used to define epidemiological observables such as prevalence or intensity of infection for a given region, often in the context of assessing the perfomance of mass drug administration programmes (Ásbjörnsdóttir et al., 2018, Halliday et al., 2019). By combining our stochastic reservoir network model with a known critical rate for migration into/out of reservoirs of infection, we have been able to demonstrate that a critical radial spatial scale exists around the focal points of infection inside a defined EU. When an EU is spatially defined such that the spatial region of consideration around each of its included focal points is larger than this critical scale, the effects of migration may be safely neglected and our reservoir network may effectively remove its ‘links’. In the opposing limit at scales smaller than , however, we have described how: the temporal stability of the dynamics of infectious reservoirs defined at this scale may be affected through migratory jumps; statistical inference biases may occur through the use of models without migratory effects; and mass drug administration applied only to the population within an EU without considering individuals moving between EUs may experience strong bounce-back effects.
We acknowledge that there are important caveats to the relationship we have found for the critical scale (and the one shown in Fig. 7) which arise from the assumptions made in obtaining Eq. (21), i.e., the statistical homogeneity and isotropy of people living in the surrounding households who all jump only once per movement event/day directly to an infectious focal point. By relaxing the assumptions made in obtaining Eq. (21), the rate of pulses will vary between different reservoirs of infection. In addition to these modifying effects, the inclusion of multiple unidirectional jumps with different initial preferred directions may no longer be isotropically configured when viewed collectively over the course of the day – which would be the case when multiple individuals all converge to a globally preferred location such as a place of work or a school. We propose to consider such modifications in future work on specific case studies.
Perhaps an even more important model extension to consider was noted in Section 4.1, where the potential for the aggregation parameter k (for the distribution of worms within hosts) was hypothesised to also vary with spatial scale due to differences in infectious exposure. Although including this potential effect will not directly influence our conclusions with regard to migration, it is possible that another critical spatial scale exists which may be found by considering the value of k required to make endemic steady state stability possible. We leave the investigation into the possibility of this additional critical scale to future work.
The critical spatial scale we have found in this work is of biological and practical importance to many helminth disease transmission processes. We have also discussed how this model may need further modifications when the relative infectious exposure of individuals is varied according to spatial scale (through the worms-within–hosts aggregation parameter k) in addition to many other extensions we have proposed throughout. We therefore consider the model we have presented here to be a useful starting point for many interesting directions for future research.
CRediT authorship contribution statement
Robert J. Hardwick: Conceptualization, Methodology, Software, Formal analysis, Writing - original draft, Writing - review & editing. Carolin Vegvari: Conceptualization, Validation, Formal analysis, Writing - review & editing. Benjamin Collyer: Conceptualization, Formal analysis, Writing - review & editing. James E. Truscott: Conceptualization, Writing - review & editing. Roy M. Anderson: Conceptualization, Writing - review & editing, Funding acquisition.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
RJH, JET and RMA gratefully thank the Bill and Melinda Gates Foundation for research grant support via the DeWorm3 (OPP1129535) award to the Natural History Museum in London ( http://www.gatesfoundation.org/). CV gratefully acknowledges funding from the NTD Modelling Consortium (OPP1184344) by the Bill and Melinda Gates Foundation in partnership with the Task Force for Global Health ( http://www.taskforce.org/). The views, opinions, assumptions or any other information set out in this article are solely those of the authors. All authors acknowledge joint Centre funding from the UK Medical Research Council and Department for International Development.
Footnotes
Note that this human death rate must match the birth rate for our static population assumption to be consistent.
References
- Anderson R.M., May R.M. Population dynamics of human helminth infections: control by chemotherapy. Nature. 1982;297(5867):557. doi: 10.1038/297557a0. [DOI] [PubMed] [Google Scholar]
- Anderson, R., and May, R. 1992. Infectious Diseases of Humans: Dynamics and Control, Dynamics and Control, OUP Oxford, 1992. URL https://books.google.co.uk/books?id=HT0-xXBguQC.
- Ásbjörnsdóttir, K. H., Ajjampur, S. S. R., Anderson, R. M., Bailey, R., Gardiner, I., Halliday, K. E., Ibikounle, M., Kalua, K., Kang, G., Littlewood, D. T. J., Luty, A. J. F., Means, A. R., Oswald, W., Pullan, R. L., Sarkar, R., Schr, F., Szpiro, A., Truscott, J. E., Werkman, M., Yard, E., Walson, J. L., Team, T. D. T., 2018. Assessing the feasibility of interrupting the transmission of soil-transmitted helminths through mass drug administration: The deworm3 cluster randomized trial protocol, PLOS Neglected Trop. Diseases 12;1:1–16. https://doi.org/10.1371/journal.pntd.0006166. [DOI] [PMC free article] [PubMed]
- Brooker, S., Bethony, J., Hotez, P. J. 2004. Human hookworm infection in the 21st century, Vol. 58 of Advances in Parasitology, Academic Press, 2004, pp. 197–288. https://doi.org/10.1016/S0065-308X(04)58004-1. http://www.sciencedirect.com/science/article/pii/S0065308X04580041. [DOI] [PMC free article] [PubMed]
- Ciddio M., Mari L., Sokolow S.H., De Leo G.A., Casagrandi R., Gatto M. The spatial spread of schistosomiasis: A multidimensional network model applied to saint-louis region, senegal. Adv. Water Resourc. 2017;108:406–415. doi: 10.1016/j.advwatres.2016.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Citron, D. T., Smith, D. L., Wu, S. L., Sanchez Castellanos, H. M., Dolgert, A. J., Henry, J. M., Guerra, C. A., 2020. Comparing metapopulation dynamics of infectious diseases under different models of human movement, medRxiv (2020). arXiv: https://www.medrxiv.org/content/early/2020/04/07/2020.04.05.20054304.full.pdf, https://doi.org/10.1101/2020.04.05.20054304. [DOI] [PMC free article] [PubMed]
- Cosner C., Beier J.C., Cantrell R.S., Impoinvil D., Kapitanski L., Potts M.D., Troyo A., Ruan S. The effects of human movement on the persistence of vector-borne diseases. J. Theor. Biol. 2009;258(4):550–560. doi: 10.1016/j.jtbi.2009.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Criscione C.D., Anderson J.D., Sudimack D., Subedi J., Upadhayay R.P., Jha B., Williams K.D., Williams-Blangero S., Anderson T.J. Landscape genetics reveals focal transmission of a human macroparasite. PLoS Neglect. Trop. Diseas. 2010;4(4) doi: 10.1371/journal.pntd.0000665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crompton D.W.T. World Health Organization; 2006. Preventive chemotherapy in human helminthiasis: coordinated use of anthelminthic drugs in control interventions: a manual for health professionals and programme managers. http://www.who.int/iris/handle/10665/43545. [Google Scholar]
- Ending the neglect and reaching 2020 goals, https://unitingtocombatntds.org/london-declaration-neglected-tropical-diseases/.
- Flueckiger R.M., Giorgi E., Cano J., Abdala M., Amiel O.N., Baayenda G., Bakhtiari A., Batcho W., Bennawi K.H., Dejene M. Understanding the spatial distribution of trichiasis and its association with trachomatous inflammationfollicular. BMC Infect. Diseas. 2019;19(1):364. doi: 10.1186/s12879-019-3935-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fronterre, C., Amoah, B., Giorgi, E., Stanton, M. C., Diggle, P. J., 2020. Design and analysis of elimination surveys for neglected tropical diseases, J. Infect. Diseases 221;Supplement_5:S554–S560. [DOI] [PMC free article] [PubMed]
- Halliday K.E., Oswald W.E., Mcharo C., Beaumont E., Gichuki P.M., Kepha S., Witek-McManus S.S., Matendechero S.H., El-Busaidy H., Muendo R. Community-level epidemiology of soil-transmitted helminths in the context of school-based deworming: Baseline results of a cluster randomised trial on the coast of kenya. PLoS Neglect. Trop. Diseas. 2019;13(8) doi: 10.1371/journal.pntd.0007427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamm, N.A., Soares Magalhães, R.J., Clements, A.C., 2015. Earth observation, spatial data quality, and neglected tropical diseases, PLoS neglected tropical diseases 9;12:e0004164. [DOI] [PMC free article] [PubMed]
- Hardwick R.J., Vegvari C., Truscott J.E., Anderson R.M. The breakpointof soil-transmitted helminths with infected human migration. J. Theor. Biol. 2019;110076 doi: 10.1016/j.jtbi.2019.110076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hardwick R.J., Werkman M., Truscott J.E., Anderson R.M. Stochastic challenges to interrupting helminth transmission. Epidemics. 2021;34 doi: 10.1016/j.epidem.2021.100435. [DOI] [PubMed] [Google Scholar]
- Helminthiases W.S.-T. World Health Organization; France: 2012. Eliminating soil-transmitted helminthiases as a public health problem in children: progress report 2001–2010 and strategic plan 2011–2020; pp. 19–29. [Google Scholar]
- High Resolution Settlement Layer (HRSL), Facebook Connectivity Lab and Center for International Earth Science Information Network - CIESIN - Columbia University, 2016, Source imagery for HRSL 2016 DigitalGlobe, Accessed (31/10/2019).
- Karagiannis-Voules D.-A., Biedermann P., Ekpo U.F., Garba A., Langer E., Mathieu E., Midzi N., Mwinzi P., Polderman A.M., Raso G. Spatial and temporal distribution of soil-transmitted helminth infection in sub-saharan africa: a systematic review and geostatistical meta-analysis. Lancet Infectious Diseas. 2015;15(1):74–84. doi: 10.1016/S1473-3099(14)71004-7. [DOI] [PubMed] [Google Scholar]
- Kung K.S., Greco K., Sobolevsky S., Ratti C. Exploring universal patterns in human home-work commuting from mobile phone data. PloS One. 2014;9(6) doi: 10.1371/journal.pone.0096180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lenormand M., Gonçalves B., Tugores A., Ramasco J.J. Human diffusion and city influence. J. R. Soc. Interface. 2015;12(109):20150473. doi: 10.1098/rsif.2015.0473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mari L., Gatto M., Ciddio M., Dia E.D., Sokolow S.H., De Leo G.A., Casagrandi R. Big-data-driven modeling unveils country-wide drivers of endemic schistosomiasis. Sci. Rep. 2017;7(1):489. doi: 10.1038/s41598-017-00493-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirlin A.D., Fyodorov Y.V., Dittes F.-M., Quezada J., Seligman T.H. Transition from localized to extended eigenstates in the ensemble of power-law random banded matrices. Phys. Rev. E. 1996;54(4):3221. doi: 10.1103/physreve.54.3221. [DOI] [PubMed] [Google Scholar]
- Moraga P., Cano J., Baggaley R.F., Gyapong J.O., Njenga S.M., Nikolay B., Davies E., Rebollo M.P., Pullan R.L., Bockarie M.J. Modelling the distribution and transmission intensity of lymphatic filariasis in sub-saharan africa prior to scaling up interventions: integrated use of geostatistical and mathematical modelling. Parasites Vectors. 2015;8(1):560. doi: 10.1186/s13071-015-1166-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moyes C.L., Henry A.J., Golding N., Huang Z., Singh B., Baird J.K., Newton P.N., Huffman M., Duda K.A., Drakeley C.J. Defining the geographical range of the plasmodium knowlesi reservoir. PLoS Neglect. Trop. Diseas. 2014;8(3) doi: 10.1371/journal.pntd.0002780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OHanlon, S.J., Slater, H.C., Cheke, R.A., Boatin, B.A., Coffeng, L.E., Pion, S.D.S., Boussinesq, M., Zour, H.G.M., Stolk, W.A., Basez, M.-G., 2016. Model-based geostatistical mapping of the prevalence of onchocerca volvulus in west africa, PLOS Neglec. Trop. Diseases 10;1:1–36. https://doi.org/10.1371/journal.pntd.0004328. [DOI] [PMC free article] [PubMed]
- Progress reports of the london declaration, https://unitingtocombatntds.org/reports/.
- Pullan R.L., Gething P.W., Smith J.L., Mwandawiro C.S., Sturrock H.J., Gitonga C.W., Hay S.I., Brooker S. Spatial modelling of soil-transmitted helminth infections in kenya: a disease control planning tool. PLoS Neglect. Trop. Diseas. 2011;5(2) doi: 10.1371/journal.pntd.0000958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pullan R.L., Smith J.L., Jasrasaria R., Brooker S.J. Global numbers of infection and disease burden of soil transmitted helminth infections in 2010. Parasites Vectors. 2014;7(1):37. doi: 10.1186/1756-3305-7-37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raichlen D.A., Wood B.M., Gordon A.D., Mabulla A.Z., Marlowe F.W., Pontzer H. Evidence of lévy walk foraging patterns in human hunter–gatherers. Proc. Natl. Acad. Sci. 2014;111(2):728–733. doi: 10.1073/pnas.1318616111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reynolds, A., Ceccon, E., Baldauf, C., Karina Medeiros, T., Miramontes, O. 2018. Lévy foraging patterns of rural humans, PlOS one 13;6:e0199099. [DOI] [PMC free article] [PubMed]
- Rhee I., Shin M., Hong S., Lee K., Kim S.J., Chong S. On the levy-walk nature of human mobility. IEEE/ACM Trans. Network. (TON) 2011;19(3):630–643. [Google Scholar]
- Schur N., Hürlimann E., Stensgaard A.-S., Chimfwembe K., Mushinge G., Simoonga C., Kabatereine N.B., Kristensen T.K., Utzinger J., Vounatsou P. Spatially explicit schistosoma infection risk in eastern africa using bayesian geostatistical modelling. Acta Tropica. 2013;128(2):365–377. doi: 10.1016/j.actatropica.2011.10.006. [DOI] [PubMed] [Google Scholar]
- Steinbaum L., Njenga S.M., Kihara J., Boehm A.B., Davis J., Null C., Pickering A.J. Soil-transmitted helminth eggs are present in soil at multiple locations within households in rural kenya. PLoS One. 2016;11(6) doi: 10.1371/journal.pone.0157780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinbaum L., Kwong L.H., Ercumen A., Negash M.S., Lovely A.J., Njenga S.M., Boehm A.B., Pickering A.J., Nelson K.L. Detecting and enumerating soil-transmitted helminth eggs in soil: New method development and results from field testing in kenya and bangladesh. PLoS Neglect. Trop. Diseas. 2017;11(4) doi: 10.1371/journal.pntd.0005522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinbaum L., Mboya J., Mahoney R., Njenga S.M., Null C., Pickering A.J. Effect of a sanitation intervention on soil-transmitted helminth prevalence and concentration in household soil: A cluster-randomized controlled trial and risk factor analysis. PLoS Neglect. Trop. Diseas. 2019;13(2) doi: 10.1371/journal.pntd.0007180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor L. No place to hide? the ethics and analytics of tracking mobility using mobile phone data. Environ. Plann. D: Soc. Space. 2016;34(2):319–336. [Google Scholar]
- Truscott, J., Turner, H., Farrell, S., Anderson, R. 2016. Chapter three - soil-transmitted helminths: Mathematical models of transmission, the impact of mass drug administration and transmission elimination criteria, in: M. G. Basez, R. M. Anderson (Eds.), Mathematical Models for Neglected Tropical Diseases, Vol. 94 of Advances in Parasitology, Academic Press, 2016, pp. 133–198. https://doi.org/10.1016/bs.apar.2016.08.002. http://www.sciencedirect.com/science/article/pii/S0065308X1630077X. [DOI] [PubMed]
- Vegvari C., Truscott J.E., Kura K., Anderson R.M. Human population movement can impede the elimination of soil-transmitted helminth transmission in regions with heterogeneity in mass drug administration coverage and transmission potential between villages: a metapopulation analysis. Parasites Vectors. 2019;12(1):438. doi: 10.1186/s13071-019-3612-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Health Organisation, Ending the neglect to attain the Sustainable Development Goals – A road map for neglected tropical diseases 2021–2030 (2020).
- Accelerating work to overcome the global impact of neglected tropical diseases: a roadmap for implementation: executive summary, World Health Organization, 2012. URL http://www.who.int/iris/handle/10665/70809.
- Zhao K., Musolesi M., Hui P., Rao W., Tarkoma S. Explaining the power-law distribution of human mobility through transportationmodality decomposition. Sci. Rep. 2015;5(1):1–7. doi: 10.1038/srep09136. [DOI] [PMC free article] [PubMed] [Google Scholar]