Abstract
Background
In the coronavirus disease 2019 (COVID-19) global pandemic, patients with cardiovascular disease represent a vulnerable population with higher risk for contracting COVID-19 and worse prognosis with higher case fatality rates. However, the relationship between COVID-19 and heart failure (HF) is unclear, specifically whether HF is an independent risk factor for severe infection or if other accompanying comorbidities are responsible for the increased risk.
Methods
This is a retrospective analysis of 1331 adult patients diagnosed with COVID-19 infection between March and June 2020 admitted at Rush University System for Health (RUSH) in metropolitan Chicago, Illinois, USA. Patients with history of HF were identified by International Classification of Disease, Tenth Revision (ICD-10) code assignments extracted from the electronic medical record. Propensity score matching was utilized to control for the numerous confounders, and univariable logistic regression was performed to assess the relationship between HF and 60-day morbidity and mortality outcomes.
Results
The propensity score matched cohort consisted of 188 patients in both the HF and no HF groups. HF patients did not have lower 60-day mortality (OR 0.81; p = 0.43) compared to patients without HF. However, those with HF were more likely to require readmission within 60 days (OR 2.88; p < 0.001) and sustain myocardial injury defined as troponin elevation within 60 days (OR 3.14; p < 0.05).
Conclusions
This study highlights the complex network of confounders present between HF and COVID-19. When balanced for these numerous factors, those with HF appear to be at no higher risk of 60-day mortality from COVID-19 but are at increased risk for morbidity.
Keywords: Heart failure, Mortality/survival, Complications, COVID-19
1. Introduction
Coronavirus disease-2019 (COVID-19) and has rapidly evolved into a global pandemic responsible for 2.5 million deaths worldwide as of March 2021 [1,2]. Patients with cardiovascular comorbidities are at increased risk for complications and severe COVID-19 infection though it remains unclear whether pre-existing heart failure (HF) is by itself an independent risk factor for severe infection [1,3,4].
Studies that have specifically explored the impact of pre-existing HF on COVID-19 morbidity and mortality, each has found that those with HF have a higher in-hospital mortality rate [3,[5], [6], [7], [8]]. Some of these studies also found higher rates of other adverse events including acute kidney injury, acute HF, mechanical ventilation, and myocardial injury [3,5,6]. However, isolating the effect of HF on COVID-19 outcomes is difficult due to the complex network of confounding variables present. For example, in one study, though HF patients had higher in-hospital mortality rates, after adjustment for comorbidities, HF itself was not an independent risk factor of mortality [6]. Conversely, others have found that HF is an independent risk factor after adjusting for these comorbidities [3,8,9].
Isolating and fully understanding the effects of pre-existing HF on COVID-19 infection is crucial as healthcare for patients with HF has been restructured during the pandemic. Therefore, identifying risk factors and high-risk patients is important for appropriate resource allocation and triage [10].
.
This study of 1331 hospitalized COVID-19 patients sought to compare outcomes between those with and without a previous HF diagnosis. We investigated additional measures of in-hospital morbidity such as need for renal replacement therapy, intubation, and inotropic agents, as well as rates of readmission, and mortality at 60 days follow-up. We utilized propensity score matching to reduce the effects of any cohort differences other than existing HF to investigate whether HF is an independent risk factor for severe COVID-19 infection or if other accompanying comorbidities are responsible for the increased risk.
2. Methods
This retrospective cohort study consisted of patients ≥18 years old diagnosed with COVID-19 admitted to Rush University System for Health (RUSH) between March to June 2020. Data was collected through a combination of automatic and manual data extraction, and patients were followed through medical record review for a minimum of 60 days after the date of initial COVID-19 admission.
HF data was automatically extracted using International Statistical Classification of Diseases and Related Health Problems, 10th revision (ICD-10) codes I50.20, I50.21, I50.22, I50.23, I50.30, I50.31, I50.33, I50.32, I50.40, I50.41, I50.42, and I50.43 (Supplemental Table 1).
Baseline characteristics included age, sex, body mass index (BMI), race, and a variety of comorbidities that were included as covariables in propensity score matching (Table 1). Covariables were chosen based on clinical relevance with emphasis on including factors with a known association with both HF and COVID-19 in the baseline characteristics.
Table 1.
Patient characteristics pre- and post-matching by heart failure status.
| Before matching |
After matching |
|||||||
|---|---|---|---|---|---|---|---|---|
| No HF | HF | p-Value | SMD | No HF | HF | p-Value | SMD | |
| n | 1075 | 256 | 188 | 188 | ||||
| Age (mean (SD)) | 55.22 (16.31) | 65.08 (15.51) | <0.001 | 0.620 | 64.11 (15.70) | 63.54 (16.03) | 0.728 | 0.036 |
| Male (%) | 607 (56.5) | 131 (51.2) | 0.144 | 0.106 | 107 (56.9) | 104 (55.3) | 0.835 | 0.032 |
| BMI (mean (SD)) | 28.62 (7.34) | 30.24 (8.61) | 0.002 | 0.203 | 29.73 (8.32) | 29.96 (8.43) | 0.791 | 0.027 |
| Race (%) | <0.001 | 0.497 | 0.796 | 0.070 | ||||
| White | 312 (29.0) | 62 (24.2) | 47 (25.0) | 51 (27.1) | ||||
| Other | 405 (37.7) | 51 (19.9) | 46 (24.5) | 41 (21.8) | ||||
| Black or African American | 358 (33.3) | 143 (55.9) | 95 (50.5) | 96 (51.1) | ||||
| Comorbidities | ||||||||
| Current smoker (%) | 59 (5.5) | 9 (3.5) | 0.258 | 0.095 | 9 (4.8) | 6 (3.2) | 0.598 | 0.082 |
| Atrial fibrillation (%) | 95 (8.8) | 88 (34.4) | <0.001 | 0.653 | 52 (27.7) | 49 (26.1) | 0.816 | 0.036 |
| Coronary artery disease (%) | 176 (16.4) | 152 (59.4) | <0.001 | 0.989 | 93 (49.5) | 93 (49.5) | 1.000 | <0.001 |
| Hypertension (%) | 597 (55.5) | 226 (88.3) | <0.001 | 0.782 | 163 (86.7) | 160 (85.1) | 0.767 | 0.046 |
| Chronic kidney disease (%) | 175 (16.3) | 152 (59.4) | <0.001 | 0.992 | 92 (48.9) | 96 (51.1) | 0.757 | 0.043 |
| COPD (%) | 38 (3.5) | 51 (19.9) | <0.001 | 0.527 | 19 (10.1) | 25 (13.3) | 0.422 | 0.099 |
| Diabetes mellitus (%) | 441 (41.0) | 163 (63.7) | <0.001 | 0.466 | 111 (59.0) | 110 (58.5) | 1.000 | 0.011 |
| Asthma (%) | 128 (11.9) | 43 (16.8) | 0.046 | 0.140 | 25 (13.3) | 29 (15.4) | 0.659 | 0.061 |
| Cancer (%) | 97 (9.0) | 42 (16.4) | 0.001 | 0.223 | 31 (16.5) | 27 (14.4) | 0.668 | 0.059 |
| Ventricular arrhythmia (%) | 19 (1.8) | 32 (12.5) | <0.001 | 0.426 | 16 (8.5) | 18 (9.6) | 0.857 | 0.037 |
| Stroke (%) | 104 (9.7) | 74 (28.9) | <0.001 | 0.503 | 43 (22.9) | 43 (22.9) | 1.000 | <0.001 |
| Acute myocardial infarction (%) | 94 (8.7) | 89 (34.8) | <0.001 | 0.665 | 52 (27.7) | 48 (25.5) | 0.726 | 0.048 |
| DVT or pulmonary embolism (%) | 107 (10.0) | 62 (24.2) | <0.001 | 0.386 | 39 (20.7) | 36 (19.1) | 0.796 | 0.040 |
HF = heart failure; SMD = standardized mean difference; SD = standard deviation; BMI = body mass index; COPD = chronic obstructive pulmonary disorder; DVT = deep vein thrombosis; PE = pulmonary embolism.
Imputation was performed on race (7.6% missing), smoking status (9.2% missing), BMI (1.4% missing) through modeling with multinomial logistic regression, linear regression, and logistic regression, respectively.
To balance baseline characteristics, we utilized propensity score matching. Whereas prior studies concluded that those with HF have higher mortality rates using multivariable regression to adjust for confounding variables, proponents of propensity score matching argue it is better able to generate a similar distribution of baseline characteristics and comorbidities between the groups, which more closely approximates a quasi-randomized controlled trial [3,6,8,9,11,12]. Due to the large number of intertwined comorbidities that exist with HF patients, creating similar groups by optimally balancing covariates is critical. In a simulation study comparing the two statistical methods, propensity score methods were superior at estimating effect sizes than multivariable regression modeling [11].
Propensity scores were calculated using a logistic regression model based on pre-defined covariates. Matching was performed using a 1:1 nearest-neighbor technique with a caliper width of 0.10, an even more stringent caliper than usually recommended for observational studies [13].
The primary outcome was 60-day mortality. Secondary outcomes included need for intubation, need for tracheostomy, need for intensive care unit, vasopressor requirement, and inotrope requirement.
Patient records were also reviewed for the occurrence of major adverse cardiovascular events (MACE) up to 60 days from hospital admission, which included myocardial injury (defined as cardiac troponin I > 0.09 ng/mL), life-threatening arrhythmia, deep venous thrombosis, symptoms of acute HF, acute renal failure requiring renal replacement therapy, or pulmonary embolism. Data was also collected on readmission and mortality. If there was no documentation of mortality, the patient was assumed to alive.
Baseline characteristics and the incidence of key outcomes were also compared between the matched and non-matched HF patients to better understand why they went unmatched and to determine whether they had significant differences in the frequency of outcomes.
2.1. Statistical analysis
All data analysis, including statistical analyses, was performed using RStudio version 1.3 (Boston, Massachusetts). Propensity score analysis and matching were performed using the MatchIt package. Kaplan-Meier survival estimates were generated and plotted with the survival and survminer packages respectively.
Continuous variables are reported with mean and standard deviation for normally distributed variables and with median and interquartile range for variables not normally distributed. Categorical variables are reported as counts and proportions. Continuous variables were compared with t-tests, and categorical variables with the Pearson chi-square test.
Univariable logistic regression was performed with presence of HF as the predictor variable for the various outcome variables, both pre- and post-matching. Odds ratios (OR) with 95% confidence intervals (CI) are reported. The threshold for statistical significance was set to a p-value <0.05.
3. Results
Among 1331 patients, 256 (19.2%) had a history of HF. Patients with HF were more likely to be older (p < 0.001), higher BMI (p < 0.01), and African American (p < 0.001) (Table 1). Those with HF were also more likely to have a variety of comorbidities including a history of atrial fibrillation, coronary artery disease, hypertension, chronic kidney disease, chronic obstructive pulmonary disorder, diabetes mellitus, asthma, cancer, ventricular arrhythmias, stroke, acute myocardial infarction, and DVT or PE (Table 1).
Propensity score matching using all the pre-defined variables in Table 1 allowed for the matching of 188 in both the heart failure and no heart failure groups. Post-matching, all variables in Table 1 had a standardized mean difference (SMD) less than 0.1, indicating a well-balanced match.
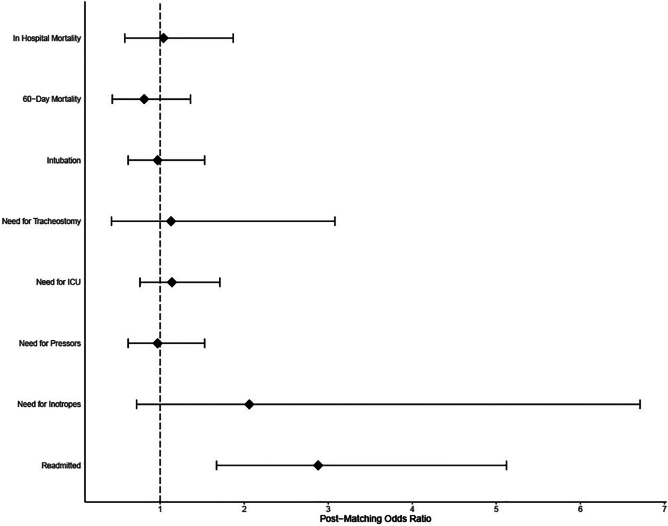
After propensity score matching, patients with HF did not have an increased risk of 60-day mortality versus those without HF (14.4% vs 13.8%; OR 1.04 [CI: 0.58–1.87]; p = 0.88) (Fig. 1, Table 2). Those with HF also had similar rates of intubation, tracheostomy, need for intensive care unit, and vasopressor or inotropic requirement as those without HF. HF patients were however more likely to require readmission within 60 days (26.6% vs 11.2%; OR 2.88 [CI: 1.67–5.12]; p < 0.001).
Fig. 1.
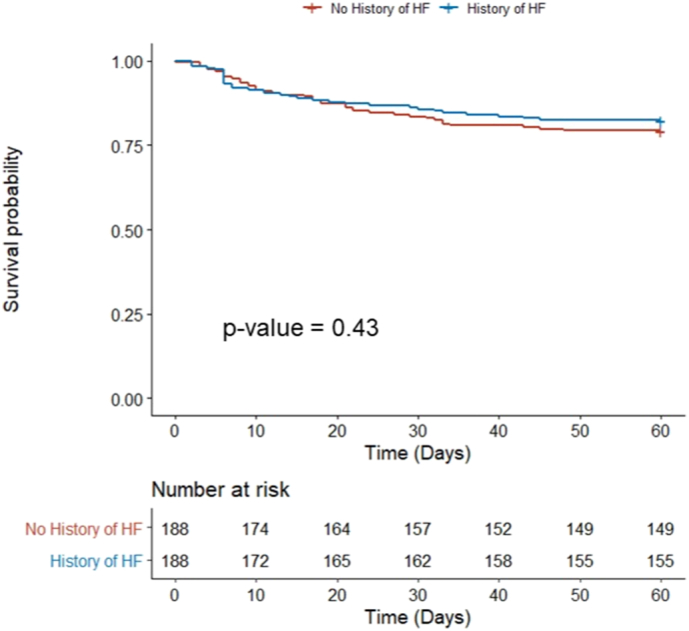
Kaplan-Meier 60-day survival estimates in COVID-19 patients with and without pre-existing heart failure.
Table 2.
Outcomes by heart failure status, both pre- and post-matching.
| Before matching |
After matching |
|||||
|---|---|---|---|---|---|---|
| No HF | HF | p-Value | No HF | HF | p-Value | |
| In-hospital mortality | ||||||
| Incidence (%) | 7.9 | 14.5 | – | 13.8 | 14.4 | |
| Univariable OR (95% CI) | Ref | 1.97 (1.29–2.95) | <0.01 | Ref | 1.04 (0.58–1.87) | 0.88 |
| 60-day mortality | ||||||
| Incidence (%) | 10 | 18.8 | – | 20.7 | 17.6 | |
| Univariable OR (95% CI) | Ref | 2.07 (1.42–2.98) | <0.001 | Ref | 0.81 (0.48–1.36) | 0.43 |
| Intubation | ||||||
| Incidence (%) | 17.6 | 30.1 | – | 28.7 | 28.2 | |
| Univariable OR (95% CI) | Ref | 2.02 (1.47–2.74) | <0.001 | Ref | 0.97 (0.62–1.53) | 0.91 |
| Need for tracheostomy | ||||||
| Incidence (%) | 1.9 | 6.3 | – | 4.3 | 4.8 | |
| Univariable OR (95% CI) | Ref | 3.52 (1.77–6.87) | <0.001 | Ref | 1.13 (0.42–3.08) | 0.80 |
| Need for ICU | ||||||
| Incidence (%) | 32.7 | 48.8 | – | 43.6 | 46.8 | |
| Univariable OR (95% CI) | Ref | 1.97 (1.49–2.60) | <0.001 | Ref | 1.14 (0.76–1.71) | 0.53 |
| Need for pressors | ||||||
| Incidence (%) | 16.7 | 28.9 | – | 28.2 | 27.7 | |
| Univariable OR (95% CI) | Ref | 2.02 (1.47–2.76) | <0.001 | Ref | 0.97 (0.62–1.53) | 0.91 |
| Need for inotropes | ||||||
| Incidence (%) | 0.9 | 6.6 | – | 2.7 | 5.3 | |
| Univariable OR (95% CI) | Ref | 7.58 (3.48–17.4) | <0.001 | Ref | 2.06 (0.72–6.71) | 0.18 |
| Readmitted | ||||||
| Incidence (%) | 8.3 | 27.3 | – | 11.2 | 26.6 | |
| Univariable OR (95% CI) | Ref | 4.17 (2.93–5.91) | <0.001 | Ref | 2.88 (1.67–5.12) | <0.001 |
HF = heart failure; OR = odds ratio; CI = confidence interval; Ref = reference; ICU = intensive care unit.
From day 1 of their hospitalization to a minimum of day 60, those with HF did not have an increased risk of MACE (27.7% vs 22.3%; OR 1.33 [CI: 0.83–2.13]; p = 0.23) (Table 3). However, when looking at the individual MACE, those with HF were more likely to have myocardial injury (6.4% vs 2.1%; OR 3.14 [CI: 1.07–11.38]; p < 0.05) and have an acute HF exacerbation (11.2% vs 0.5%; OR 23.51 [CI: 4.84–423.82]; p < 0.01) (Fig. 2). Those with and without HF were similarly likely to have deep venous thrombosis, life-threatening arrhythmia, need for renal replacement therapy, and pulmonary embolism.
Table 3.
Major adverse cardiovascular events (MACE) by heart failure status, both pre- and post-matching.
| Major adverse cardiovascular events | Before matching |
After matching |
||||
|---|---|---|---|---|---|---|
| No HF | HF | p-Value | No HF | HF | p-Value | |
| Myocardial injury | ||||||
| Incidence (%) | 0.8 | 7.4 | – | 2.1 | 6.4 | |
| Univariable OR (95% CI) | Ref | 9.50 (4.36–22.3) | <0.001 | Ref | 3.14 (1.07–11.38) | <0.05 |
| Life-threatening arrhythmia | ||||||
| Incidence (%) | 4.7 | 12.1 | – | 8.5 | 12.2 | |
| Univariable OR (95% CI) | Ref | 2.82 (1.75–4.50) | <0.001 | Ref | 1.50 (0.77–2.98) | 0.24 |
| Deep venous thrombosis | ||||||
| Incidence (%) | 2 | 5.1 | – | 2.7 | 3.7 | |
| Univariable OR (95% CI) | Ref | 2.69 (1.29–5.38) | <0.01 | Ref | 1.42 (0.44–4.86) | 0.56 |
| HF exacerbation | ||||||
| Incidence (%) | 0.1 | 10.9 | – | 0.5 | 11.2 | |
| Univariable OR (95% CI) | Ref | 65.9 (19.6–409.9) | <0.001 | Ref | 23.51 (4.84–423.82) | <0.01 |
| Renal replacement therapy | ||||||
| Incidence (%) | 4.2 | 12.1 | – | 10.1 | 10.6 | |
| Univariable OR (95% CI) | Ref | 3.15 (1.94–5.07) | <0.001 | Ref | 1.06 (0.54–2.07) | 0.87 |
| Pulmonary embolism | ||||||
| Incidence (%) | 2.8 | 3.9 | – | 4.8 | 4.8 | |
| Univariable OR (95% CI) | Ref | 1.42 (0.65–2.84) | 0.36 | Ref | 1.00 (0.38–2.62) | 1.00 |
| Total | ||||||
| Incidence (%) | 12.2 | 29.7 | – | 22.3 | 27.7 | |
| Univariable OR (95% CI) | Ref | 3.04 (2.19–4.20) | <0.001 | Ref | 1.33 (0.83–2.13) | 0.23 |
HF = heart failure; OR = odds ratio; CI = confidence interval; Ref = reference.
Fig. 2.
Odds ratios with 95% confidence intervals for various COVID-19 outcomes if pre-existing heart failure is present.
When analyzing only those with HF, the 68 non-matched patients tended to be older (p < 0.01) and were more likely to be female (p < 0.05) than their 188 matched counterparts (Supplemental Table 2). In addition, non-matched patients were more likely to have a variety of comorbidities including atrial fibrillation (p < 0.001), coronary artery disease (p < 0.001), hypertension (p < 0.01), chronic kidney disease (p < 0.001), COPD (p < 0.001), diabetes mellitus (p < 0.01), ventricular arrhythmia (p < 0.05), and acute myocardial infarction (p < 0.01). Non-matched HF patients did not have increased risk of the key study outcomes including 60-day mortality, requiring ICU, and overall MACE.
4. Discussion
4.1. HF readmissions and outcomes of HF in COVID-19 infection
Compared to patients without HF, pre-existing HF did not appear to be an independent risk factor for increased 60-day mortality in our cohort. While prior studies have observed higher mortality in HF patients with COVID-19, our study design utilized propensity score matching to reduce bias and create well balanced groups. This reduction in bias is particularly important in isolating the effect of HF on COVID-19 as numerous comorbidities have independent and potentially confounding relationships between these two diseases including diabetes mellitus, chronic kidney disease, chronic respiratory disease, tobacco use, and more [3,14]. Our findings further echo this importance as prior to matching, an approximately 2-fold higher risk of mortality was present, which approximates that reported in prior studies (Table 2) [3,8]. However, after balance was achieved through matching, this mirage of an effect was no longer observable. Of note, Rey et al. similarly demonstrated a higher mortality rate in HF patients, but when adjusted for comorbidities, they found that HF was not an independent risk factor for mortality [6].
Our findings suggest that the variables controlled for during our propensity score matching are the drivers of the higher mortality in HF patients with COVID-19 rather than the HF itself. In examining prior studies asking this same research question, each has adjusted for different comorbidities, which could in part be responsible for differences in results (Supplemental Table 3). For example, the largest study of COVID-19 patients with pre-existing HF did not control for coronary artery disease, which is associated with HF and worse COVID-19 outcomes [9]. To our knowledge, our study provides the most comprehensive attempt, to date, at controlling for confounders between HF and COVID-19.
Importantly, pre-existing HF was a significant predictor for readmission and the occurrence of an acute HF exacerbation within 60 days. Prior studies assessing the association between HF and increased readmissions have shown mixed results, but many of these studies have been limited by smaller sample sizes and shorter follow-up durations [[15], [16], [17]]. In our study cohort, 159 total patients (pre-matching) were readmitted during 60-days follow-up, which provided a robust sample for analysis. With the continued spread of COVID-19, high rates of readmission in patients with COVID-19 and history of HF requires more structured remote care for HF patients [10].
4.2. HF and myocardial injury in COVID-19
While myocardial injury is known to occur in up to 60% of hospitalized COVID-19 patients, our data suggest that those with pre-existing HF are at increased risk for this complication. Several mechanisms of COVID-19 induced acute myocardial injury have been proposed including myocyte damage from hypoxia, inflammation, downregulation of angiotensin converting enzyme 2 (ACE2), hypercoagulability leading to development of coronary microvascular thrombi, and supply-demand mismatch leading to plaque rupture and myocardial infarction [18,19]. It is unclear why HF patients are at increased risk, but troponin elevation is prevalent in patients with chronic HF and present in nearly all patients presenting with acute decompensated HF [20]. Therefore, this suggests that the increased risk for myocardial injury could be due to a combination of both COVID-19 effects on myocardium and a generalized increased risk for troponin elevation in HF patients.
4.3. Outcome duration of follow up
While many COVID-19 studies focus on in-hospital outcomes, our follow up of 60 days provides critical information surrounding both in-hospital and post-discharge events. While COVID-19 has significantly changed health care delivery across virtually all specialties, special consideration must be given to HF patients due to their baseline increased risk for complications, readmission, and overall mortality. In fact, the most common cause of readmission in Medicare patients is chronic heart failure [21]. Therefore, our data provide a longitudinal view of adverse outcomes from COVID-19 in patient population known to be at risk for medical complications.
4.4. Propensity matching effects
We also analyzed the non-matched HF patients in comparison to matched HF patients to provide insight into why these patients went unmatched and whether they may represent a sicker subpopulation of HF patients that could limit generalizability of our study. At baseline, the non-matched patients were older and had increased frequency of a variety of comorbidities. However, there was no difference in the frequency of key outcomes between the matched and non-matched HF patients, indicating that this is not a limitation of this study.
4.5. External validity of results
In our cohort, approximately 1 of every 5 patients admitted with COVID-19 had a pre-existing diagnosis of HF, which is considerably higher than prior HF studies [3,6,8,9]. Our mortality rate for both the entire cohort (9.2%) and heart failure patients (18.8%) was also lower than these prior HF studies. This difference is, in part, attributable to differences in city-specific disease burdens at the time of the study. For example, 3 of the major HF studies took place in New York City from late February to June 2020, Italy from March to April 2020, and Madrid, Spain from March to April 2020. Each of these areas was profoundly strained by disease burden during the time of the study, which therefore limits each's external validity. In contrast, Chicago was experiencing a more manageable disease burden at this time. As of April 13, 2020, New York City had the most reported cases of any city in the United States at 106,764, which included 7154 deaths [22]. Chicago on the other hand, while fifth overall, had only 15,474 reported cases and 543 deaths [22]. Internationally, the United States' 7-day rolling average of COVID-19 confirmed deaths per million people on April 1, 2020 was 2.29 compared to 13.55 and 17.54 in Italy and Spain, respectively [23]. Because Chicago as a whole was less strained at the time these data were collected, our data are likely more applicable to other populations.
4.6. Study limitations
The use of electronic health records has inherent sources for error. In this case, identification of diagnosis of HF was done with the use of ICD-10 codes, which have been shown to be specific (96.8%) but not sensitive (75.3%) for identifying a prior HF diagnosis [24]. Another potential source of error is in the method of COVID-19 diagnosis. Confirmed COVID-19 patients were included based on either positive polymerase chain reaction (PCR) testing or point-of-care rapid testing, with each test subject to their own sensitivity and specificities. Based on data using multiple cohort studies, the overall sensitivity and specificity of COVID-19 testing has been estimated to be 71% and 100%, respectively [25]. Lastly, readmission rates were only captured for patients who had a subsequent hospitalization to a health care system in or around Chicago that also use the EPIC electronic medical record.
5. Conclusions
In a propensity-matched population of hospitalized COVID-19 patients in metropolitan Chicago, a pre-existing HF history was not associated with increased risk of 60-day mortality. However, those with HF were found to be at higher risk for a complicated course including myocardial injury, as well as need for structured hospital follow up to avoid hospital readmission.
Funding
The authors received no financial support for the research, authorship, or publication of this manuscript.
Declaration of competing interest
None.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ahjo.2021.100022.
Appendix A. Supplementary data
Supplementary tables
References
- 1.Yang J., Zheng Y., Gou X., Pu K., Chen Z., Guo Q., Ji R., Wang H., Wang Y., Zhou Y. Prevalence of comorbidities and its effects in coronavirus disease 2019 patients: a systematic review and meta-analysis. Int. J. Infect. Dis. 2020;94:91–95. doi: 10.1016/j.ijid.2020.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Johns Hopkins coronavirus resource center. https://coronavirus.jhu.edu/ Available at.
- 3.Alvarez-Garcia J., Lee S., Gupta A., Cagliostro M., Joshi A.A., Rivas-Lasarte M., Contreras J., Mitter S.S., LaRocca G., Tlachi P., Brunjes D., Glicksberg B.S., Levin M.A., Nadkarni G., Fayad Z., Fuster V., Mancini D., Lala A. Prognostic impact of prior heart failure in patients hospitalized with COVID-19. J. Am. Coll. Cardiol. 2020;76:2334–2348. doi: 10.1016/j.jacc.2020.09.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zheng Y.Y., Ma Y.T., Zhang J.Y., Xie X. COVID-19 and the cardiovascular system. Nat. Rev. Cardiol. 2020:17. doi: 10.1038/s41569-020-0360-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chatrath N., Kaza N., Pabari P.A., Fox K., Mayet J., Barton C., Cole G.D., Plymen C.M. The effect of concomitant COVID-19 infection on outcomes in patients hospitalized with heart failure. ESC Heart Fail. 2020 doi: 10.1002/ehf2.13059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rey J.R., Caro-Codón J., Rosillo S.O., Iniesta Á.M., Castrejón-Castrejón S., Marco-Clement I., Martín-Polo L., Merino-Argos C., Rodríguez-Sotelo L., García-Veas J.M., Martínez-Marín L.A., Martínez-Cossiani M., Buño A., Gonzalez-Valle L., Herrero A., López-Sendón J.L., Merino J.L. Heart failure in COVID-19 patients: prevalence, incidence and prognostic implications. Eur. J. Heart Fail. 2020 doi: 10.1002/ejhf.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yonas E., Alwi I., Pranata R., Huang I., Lim M.A., Gutierrez E.J., Yamin M., Siswanto B.B., Virani S.S. Effect of heart failure on the outcome of COVID-19 - a meta analysis and systematic review. Am. J. Emerg. Med. 2020 doi: 10.1016/j.ajem.2020.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tomasoni D., Inciardi R.M., Lombardi C.M., Tedino C., Agostoni P., Ameri P., Barbieri L., Bellasi A., Camporotondo R., Canale C., Carubelli V., Carugo S., Catagnano F., Dalla Vecchia L.A., Danzi G.B., di Pasquale M., Gaudenzi M., Giovinazzo S., Gnecchi M., Iorio A., la Rovere M.T., Leonardi S., Maccagni G., Mapelli M., Margonato D., Merlo M., Monzo L., Mortara A., Nuzzi V., Piepoli M., Porto I., Pozzi A., Sarullo F., Sinagra G., Volterrani M., Zaccone G., Guazzi M., Senni M., Metra M. Impact of heart failure on the clinical course and outcomes of patients hospitalized for COVID-19. Results of the cardio-COVID-Italy multicentre study. Eur. J. Heart Fail. 2020;22:2238–2247. doi: 10.1002/ejhf.2052. [DOI] [PubMed] [Google Scholar]
- 9.Bhatt A.S., Jering K.S., Vaduganathan M., Claggett B.L., Cunningham J.W., Rosenthal N., Signorovitch J., Thune J.J., Vardeny O., Solomon S.D. Clinical outcomes in patients with heart failure hospitalized with COVID-19. JACC Heart Fail. 2021;9:65–73. doi: 10.1016/j.jchf.2020.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.DeFilippis E.M., Reza N., Donald E., Givertz M.M., Lindenfeld J., Jessup M. Considerations for heart failure care during the COVID-19 pandemic. JACC Heart Fail. 2020;8:681–691. doi: 10.1016/j.jchf.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Martens E.P., Pestman W.R., de Boer A., Belitser S. v, Klungel O.H. Systematic differences in treatment effect estimates between propensity score methods and logistic regression. Int. J. Epidemiol. 2008;37:1142–1147. doi: 10.1093/ije/dyn079. [DOI] [PubMed] [Google Scholar]
- 12.Amoah J., Stuart E.A., Cosgrove S.E., Harris A.D., Han J.H., Lautenbach E., Tamma P.D. Comparing propensity score methods versus traditional regression analysis for the evaluation of observational data: a case study evaluating the treatment of gram-negative bloodstream infections. Clin. Infect. Dis. 2020;71:e497–e505. doi: 10.1093/cid/ciaa169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Austin P.C. Optimal caliper widths for propensity-score matching when estimating differences in means and differences in proportions in observational studies. Pharm. Stat. 2011;10:150–161. doi: 10.1002/pst.433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Williamson E.J., Walker A.J., Bhaskaran K., Bacon S., Bates C., Morton C.E., Curtis H.J., Mehrkar A., Evans D., Inglesby P., Cockburn J., McDonald H.I., MacKenna B., Tomlinson L., Douglas I.J., Rentsch C.T., Mathur R., Wong A.Y.S., Grieve R., Harrison D., Forbes H., Schultze A., Croker R., Parry J., Hester F., Harper S., Perera R., Evans S.J.W., Smeeth L., Goldacre B. Factors associated with COVID-19-related death using OpenSAFELY. Nature. 2020;584:430–436. doi: 10.1038/s41586-020-2521-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lavery A.M., Preston L.E., Ko J.Y., Chevinsky J.R., DeSisto C.L., Pennington A.F., Kompaniyets L., Datta S.D., Click E.S., Golden T., Goodman A.B., mac Kenzie W.R., Boehmer T.K., Gundlapalli A. v. Characteristics of hospitalized COVID-19 patients discharged and experiencing same-hospital readmission - United States, March-August 2020. MMWR Morb. Mortal. Wkly Rep. 2020;69:1695–1699. doi: 10.15585/mmwr.mm6945e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jeon W.-H., Seon J.Y., Park S.-Y., Oh I.-H. Analysis of risk factors on readmission cases of COVID-19 in the Republic of Korea: using nationwide health claims data. Int. J. Environ. Res. Public Health. 2020:17. doi: 10.3390/ijerph17165844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Somani S., Richter F., Fuster V., de Freitas J., Naik N., Sigel K., Boettinger E.P., Levin M.A., Fayad Z., Just A.C., Charney A., Zhao S., Glicksberg B.S., Lala A., Nadkarni G. Characterization of patients who return to hospital following discharge from hospitalization for COVID-19. medRxiv: The Preprint Server for Health Sciences. 2020 doi: 10.1101/2020.05.17.20104604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lang J.P., Wang X., Moura F.A., Siddiqi H.K., Morrow D.A., Bohula E.A. A current review of COVID-19 for the cardiovascular specialist. Am. Heart J. 2020;226:29–44. doi: 10.1016/j.ahj.2020.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bavishi C., Bonow R.O., Trivedi V., Abbott J.D., Messerli F.H., Bhatt D.L. Special article - acute myocardial injury in patients hospitalized with COVID-19 infection: a review. Prog. Cardiovasc. Dis. 2020;63:682–689. doi: 10.1016/j.pcad.2020.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Felker G.M., Mentz R.J., Teerlink J.R., Voors A.A., Pang P.S., Ponikowski P., Greenberg B.H., Filippatos G., Davison B.A., Cotter G., Prescott M.F., Hua T.A., Lopez-Pintado S., Severin T., Metra M. Serial high sensitivity cardiac troponin T measurement in acute heart failure: insights from the RELAX-AHF study. Eur. J. Heart Fail. 2015;17:1262–1270. doi: 10.1002/ejhf.341. [DOI] [PubMed] [Google Scholar]
- 21.Chamberlain R.S., Sond J., Mahendraraj K., Lau C.S., Siracuse B.L. Determining 30-day readmission risk for heart failure patients: the Readmission After Heart Failure scale. Int. J. Gen. Med. 2018;11:127–141. doi: 10.2147/IJGM.S150676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Maroko A.R., Nash D., Pavilonis B.T. COVID-19 and inequity: a comparative spatial analysis of New York City and Chicago hot spots. J. Urban Health: Bull. New York Acad. Med. 2020;97:461–470. doi: 10.1007/s11524-020-00468-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Our world in data, data explorer. https://ourworldindata.org/ Available at.
- 24.McCormick N., Lacaille D., Bhole V., Avina-Zubieta J.A. Validity of heart failure diagnoses in administrative databases: a systematic review and meta-analysis. PLoS One. 2014;9 doi: 10.1371/journal.pone.0104519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hanson K.E., Caliendo A.M., Arias C.A., Hayden M.K., Englund J.A., Lee M.J., Loeb M., Patel R., el Alayli A., Altayar O., Patel P., Falck-Ytter Y., Lavergne V., Morgan R.L., Murad M.H., Sultan S., Bhimraj A., Mustafa R.A. The Infectious Diseases Society of America guidelines on the diagnosis of COVID-19: molecular diagnostic testing. Clin. Infect. Dis.: Off. Publ. Infect. Dis. Soc. Am. 2021 doi: 10.1093/cid/ciab048. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary tables