Abstract
Nano drug-delivery systems (DDS) may significantly improve efficiency and reduce toxicity of loaded drugs, but a few nano-DDS are highly successful in clinical use. Unprotected nanoparticles in blood flow are often quickly cleared, which could limit their circulation time and drug delivery efficiency. Elongating their blood circulation time may improve their delivery efficiency or grant them new therapeutic possibilities. Erythrocytes are abundant endogenous cells in blood and are continuously renewed, with a long life span of 100–120 days. Hence, loading nanoparticles on the surface of erythrocytes to protect the nanoparticles could be highly effective for enhancing their in vivo circulation time. One of the key questions here is how to properly attach nanoparticles on erythrocytes for different purposes and different types of nanoparticles to achieve ideal results. In this review, we describe various methods to attach nanoparticles and drugs to the erythrocyte surface, and discuss the key factors that influence the stability and circulation properties of the erythrocytes-based delivery system in vivo. These data show that using erythrocytes as a host for nanoparticles possesses great potential for further development.
Keyword: drug delivery systems, nanoparticle, erythrocytes, prolonged circulation time
Introduction
In recent decades, nanomedicine research has made tremendous progress in applications such as imaging, diagnostics, and targeted drug delivery [1, 2]. When properly designed, nanomedicines have less severe adverse effects, better delivery efficiency, and better effects than free drugs. However, without proper design, rapid blood clearance and poor targeting may limit the clinical application of nanoparticle (NP)-based drug delivery systems [3, 4]. Various design strategies have been developed to overcome these shortcomings. Due to the large quantity of available data, it could be helpful to summarize these data and organize them from different perspectives.
Generally, using NPs for drug delivery requires optimized NP properties, such as material, size, shape, and surface modification, which need to be tailored to achieve the desired functions, such as the ability to circulate in the blood, target specific tissues, enter target cells, and release drugs under certain conditions [5–11]. For many medical applications, the retention time of NPs in blood circulation is of great importance. Almost all nanomedicines will interact with blood vessels and blood contents at some point during their life cycle in the human body, regardless of their final destination. A longer retention time provides more opportunities for the NPs to reach the target tissue, increases the period of drug release [12], increases their duration as functional imaging contrast agents [13], or allows them to interact with blood longer [14–17]. Therefore, various strategies have been investigated to elongate the blood circulation time of NPs [1, 18]. Among these strategies, erythrocyte-assisted NP retention has attracted great interest in recent years. Erythrocytes, or red blood cells (RBCs), are the most abundant endogenous cells in human blood and have very simple structures; thus, they are easy to access and manipulate [19]. They are continuously generated in the bone marrow and renewed throughout the lifetime, and they typically have a long life span between 100 and 120 days [5, 14, 20]. These characteristics make it possible for erythrocytes to help NPs escape clearance mechanisms in vivo by carrying and camouflaging the NPs in the bloodstream. The earliest attempts to use erythrocytes to carry cargo date back to the 1970s, and studies have continued to explore different applications and methods, with NPs becoming a new type of cargo much later. A study in 2004 showed that certain bacteria (e.g., M. haemominutum) could adhere to RBCs and use them as a disguise to stay in the circulation for several weeks [4]. Hence, there is potential to elongate the circulation time of NPs in a similar manner.
In this review, we present and compare traditional and novel engineering strategies in the development of RBC-based drug delivery systems (DDS), with a focus on attachment methods mainly used to achieve longer retention. We aim to provide comprehensive coverage of both natural and synthetic systems, illustrating the potential of RBC-based long-circulation delivery systems and their wide applications in the field of drug delivery. We further discuss the aspects of surface erythrocyte drug delivery that are most pertinent in the current context: factors related to long-term delivery regimens, and immunologic considerations of RBC-based DDS.
Using erythrocytes for drug delivery
As discussed above, the inherent biochemical and biophysical properties of erythrocytes, especially their long half-life in the circulatory system and their interactions with certain organs and tissues, make them good drug delivery platforms for specific applications. When properly designed, RBC-based systems could have significant advantages over alternative technologies in terms of half-life, stability, versatility, and safety [21]. However, how to efficiently exploit the characteristics of RBCs is a complicated problem. The first question here is whether to encapsulate the cargoes inside RBCs or to attach them to the outside of the cells.
Encapsulation inside or adherence outside RBCs
Many studies have explored the encapsulation of nanomedicines in RBCs or the membrane of RBCs [22, 23]. Here, we will first discuss the encapsulation strategy. As a functional cell with a biconcave shape (diameter of ~7.5 μm and thickness of ~2.5 μm) and low internal proteolytic activity, erythrocytes can house many cargoes—especially NPs—inside them, which also offers full protection for the cargoes [17, 24]. Because cargoes are basically isolated from the blood environment, they are well protected from extracellular clearance, possible inactivation by blood contact, and immune recognition [25–27].
Because erythrocytes are surrounded by the cell membrane, putting cargoes inside them without damaging the cell membrane is a key part of the preparation process. The most common way to do this is to use hypotonic dialysis [28]. For example, Baysal et al. used this method to encapsulate PEG-urease/PEG-aldehyde dehydrogenase into homologous sheep RBCs. These engineered RBCs could maintain enzyme activity for 6 days when injected into sheep, compared with 2 days after injecting free PEG-modified enzymes [27]. In engineered RBCs, the lack of lysosomal enzymes leads to the gradual accumulation of the substrates in lysosomes, which ultimately leads to lysosomal destruction, dysfunction and cell death. Another study used β-glucocerebrosidase, which is the first enzyme incorporated into RBCs for enzyme replacement therapy, and a fourfold increase in circulation time was observed [26]. Currently, a variety of free lysosomal enzymes are used to treat lysosomal storage diseases, but they have high immunogenicity and high costs. Loading the appropriate enzymes into RBCs can overcome these limitations and reduce the overall treatment cost. In addition to enzymes [29], other cargoes, including antigens (to modulate the immune response for malignant tumor treatment [30]) and reactive substances (for removing toxic compounds from the bloodstream [31]), are also encapsulated in RBCs. Multiple studies using a drug encapsulation approach have confirmed the feasibility and prospects of infusing drug-loaded RBCs [12].
However, the cargoes that can be encapsulated via hypotonic dialysis are mostly limited by size, and most NPs cannot be encapsulated in this way. In addition, internal loading often requires carefully controlled conditions or procedures and can easily result in membrane disruption that may compromise biocompatibility [32–34]. Although there are other encapsulation methods, such as chemical-assisted encapsulation and electroporation, these methods are often less manipulatable and may cause more damage to erythrocytes, and NP encapsulation remains difficult. Endocytosis is another way to encapsulate NPs into erythrocytes, but erythrocytes appear to have low endocytosis efficiency [35], which could lead to a low encapsulation rate, and the endocytosis pathway also involves many cellular mechanisms, which could make the results less controllable. Another disadvantage is that the encapsulated substances need to penetrate the cell membrane (and the endosomal membrane if endocytosed) to enter the blood and for changes in the blood to reach the cargoes, which severely limits the design and the type of cargo.
One compromised way to encapsulate NPs is to use only part of the erythrocytes. For instance, in a previous study by our group, we designed a glucose-sensitive insulin nanoplatform that has an inner polymeric NP “core” with an outer erythrocyte membrane cover as a “shell” [17]. The NPs could achieve repeatable insulin release in response to changes in environmental glucose concentrations, while the RBC membrane-derived coating elongated the circulation time. However, because only the membrane of erythrocytes was used, which limits the ability of this core-shell system to escape immune clearance, the elimination half-life reached only 36.3 h.
In light of these problems, some researchers turned their attention to the surface of the RBC membrane [16, 36], as the large specific surface area of RBCs is also advantageous for cell hitchhiking or surface engineering. Employing erythrocyte surface loading or modification strategies, maintaining a longer cycle time without disrupting the normal physiological functions of erythrocytes can be achieved if the degree of surface coupling and loading are well controlled. The useable cargo types are significantly expanded. In addition to biomacromolecules (e.g., enzymes, antibodies, and functional peptides), drugs, imaging probes, and antigens, other larger substances up to micron size, such as NPs and nanorobots, can be loaded onto the surface of RBCs (mostly in a “hitchhiking” fashion) for different purposes [36–38]. With this strategy, the cargoes are less protected and are partially exposed to the bloodstream. Therefore, the cargoes need to be able to stay stable through interactions with substances in the blood or on the surface of blood vessels. On the other hand, it provides more opportunities for cargo to respond to the blood environment and act more directly (such as releasing drugs) in the body. As this review is focused on detailed strategies and applications of erythrocyte surface loading to achieve long circulation time, we will present related advances in detail in the sections below.
Common medical applications
There are many vastly different medical applications for cargo-loaded or engineered erythrocytes. Here, a brief introduction of these applications will be presented to help the reader better understand why surface loading on erythrocytes could be useful. However, as this is not the main focus of this review, we recommend reading the excellent reviews from the Gu group and the Muzykantov group for additional information if these topics interest you [14, 18, 39, 40].
As mentioned, one of the most common goals is to elongate their retention time inside the body, especially in the blood. In some specific cases, the long retention time in blood is especially useful to control the formation and dissolution of blood clot proteins by binding active agents to the surface of carrier RBCs when cargoes are loaded on the erythrocyte surface [41, 42]. Moreover, engineered or natural RBCs as hybrid and/or biological carriers constitute a unique drug delivery system that is capable of extensively changing drug pharmacokinetics. Several studies using erythrocytes as vehicles for proteins showed that they can reversibly and specifically bind diffusible drugs, making new modalities possible and increasing drug absorption in circulating erythrocytes [29, 43], which suggests that organ-specific drug absorption and drug distribution may change as a result [36, 44, 45].
As discussed above, erythrocytes also have an innate targeting capability that can be exploited. For instance, they are able to transfer attached pathogens to immune cells and hence can be used to deliver loaded antigens to antigen-presenting cells (APCs) in the spleen and generate strong immune responses [46]. This method was named erythrocyte-driven immune targeting by the group who developed it and used it to successfully slow the growth of cancerous tumors in mice. The team also suggested that NP-carrying RBCs could be applied as biocompatible adjuvants, which would be an intriguing trend in the vaccine field [47].
The application of erythrocytes as containers or hosts for various cargoes was also shown to reduce the toxicity of therapies and decrease the risk of side effects and unwanted immune reactions [12], as the delivery of biologics by erythrocytes could protect the entrapped agent from rapid clearance and immune responses. Moreover, this camouflage could (under appropriate conditions) target biologics to selected cells and/or cell compartments without damaging healthy tissues [48, 49]. Sometimes, even RBCs themselves could be sheltered for reduced side effects. For instance, a widely used strategy is to covalently attach methoxy PEG (mPEG) to the surface of erythrocytes to block antigenic determinants, thereby minimizing the immune responses to blood transfusion [50, 51].
The development of these erythrocyte-based delivery systems demonstrated the great versatility and immense potential of these novel DDS in treating various pathological conditions. More work in this field could help accelerate the application of these types of DDS in the clinical setting, and the NP surface-loading strategy is an important topic.
Safety concerns—the influence of NPs on RBCs
While using RBCs to protect drug-loaded NPs has many advantages, it is essential to keep the RBCs physiologically normal. Indeed, RBC membrane disorders (such as hereditary spherocytosis) have demonstrated the significance of maintaining RBC integrity, deformability, membrane fluidity, shape, rheology, and resistance to phagocytosis and clearance [2, 18]. In addition, abnormal RBCs or the exogenous substances attached to RBCs may also activate immune reactions and/or certain stress-responsive signaling pathways and mechanisms, invoking complicated consequences, which could be highly unpredictable and lead to other adverse effects. Below, these safety-related issues will be discussed.
The interactions between RBCs and many objects—such as NPs, agents used for disguising RBC antigenic determinants, and other DDS—may adversely affect RBCs and their functions and even cause hemolysis. Therefore, it is necessary to evaluate the extent of functional abnormalities in erythrocytes after these interactions. To do so, a study used a set of high-throughput in vitro assays to examine the sensitivity of NP-carrying erythrocytes to biologically relevant injuries (especially osmotic, mechanical, oxidative and complement stress) and RBC agglutination, which are often frequently encountered in circulation [9, 52]. The results show that the noncovalent adsorption of NPs to mouse and human RBCs is not detrimental at NP:RBC ratios lower than NP/RBC 200:1. This pattern also applies for most other related particles. These tests could be used as prescreen formulations to exclude those that exhibit inappropriate effects on erythrocytes, thereby eliminating unnecessary or dangerous preclinical and clinical settings in vivo and in vitro.
Furthermore, when inside the body, almost all nanomedicines will interact with blood vessels at some point regardless of their intended destination [53]. Previous studies demonstrated that NPs induced hemolysis of RBCs and activated cell aggregation [54], and some groups have examined the influence of NPs on RBC adhesion to endothelial cells (ECs) [53, 54]. However, knowledge of the details of these interactions between ECs and RBCs in the presence of NPs is still insufficient, and further research is needed to determine the mechanism by which NPs penetrate the endothelium and enter the tissue of interest.
There are also a few studies that investigated the effects of particle-cell dynamics or capillary stresses on loaded drugs in the blood flow [53, 55, 56], which provided valuable insights into the biophysical behavior of nanomedicines that could help us evaluate their delivery and side effects.
To reduce immunogenicity and stress signaling, the basic principle is to use biocompatible materials. However, beyond this principle, little is known about the detailed interactions between materials and cells/tissues/organs, and the consequences that may occur are ambiguous in many cases. For instance, the modification of foreign molecules on the membrane of RBCs may induce alloimmunization, which is a potential risk in clinical use, especially after repeated administrations, but the exact mechanism is not clear [57]. In fact, these problems form an essential bottleneck that limits the use of most new nanomaterials in clinical applications. Due to the lack of data, we expect that in-depth investigations could be conducted in this field and improve our understanding of the long-term safety of these exogenous nanomaterials.
Nevertheless, our understanding of this topic is far from satisfactory. To increase the clinical use of erythrocyte-based nanomedicines, more works concerning the detailed effects of loaded cargoes on erythrocytes and the underlying mechanism are needed.
Characteristics and circulation time of erythrocyte-anchored NPs
When an erythrocyte-based drug delivery system is employed to achieve long circulation of NPs, if the erythrocytes are physiologically normal and have a normal life span, then the final NP circulation time may be determined by a few critical factors, including the stability of the NPs, the detachment rate of the NPs, and the internalization rate of the NPs. Because the stability of NPs is generally governed by the material used and purpose of the nanomedicine, we disregard this factor and assume that the NPs are highly stable. For the other two factors, the interaction between nanomaterials and the cell surface is essential, which largely governs whether the NPs will drop off the surface and whether the surface will engulf the NPs [58]. Both the physical properties and chemical properties (especially the surface chemical properties) of the NPs will significantly influence these interactions. Because these properties are the major tailorable properties of NPs, their impacts will be dissected in detail below. Because of the importance and diversity of NP-RBC binding mechanisms, which are often carefully selected and specifically designed, we will break the section into two parts. The first part introduces more general characteristics across different types of NPs, which are mostly physical properties, and the second part is centered on the different binding mechanisms that are often intrinsically different in different NPs.
General characteristics of NPs that could affect circulation time
For the design of NPs for RBC-centered long-term circulation strategies, it is vital to screen appropriate physical characteristics (e.g., size, charge, shape, and elasticity) and surface modifications (such as chemical or polymer binding, targeting or tissue‐penetrating peptides) and to consider their impacts on drug delivery. Some of these parameters of long-circulation cellular-hitchhiking-based DDS have been covered in several previous reviews [48, 59, 60]. In general, a combination of appropriate NP physical characteristics, surface modifications, a natural cell shape, and cell physiological properties is required to achieve long circulation and other goals [5]. More specifically, Anselmo et al. reviewed the pros and cons of different cell-particle attachment methods [59] (Table 1); Stephan et al. reviewed various bioengineering functionalizations to rationally modify the surface of treated cells and utilized the properties of these membranes based on the complicated structure of the plasma membrane [58]. Here, we will simply cover some basics in this research area.
Table 1.
Methods, advantages, and disadvantages of nanoparticle attachment techniques to cells. WIth permission from [59].
| Attachment method | Advantages | Disadvantages | Ideal cell type |
|---|---|---|---|
|
Adsorption (electrostatic and hydrophobic)
|
Requires no pre-binding cell preparation. Simple particle requirements: hydrophobic or positively charged. |
Attachment may not be strong and particles may detach unpredictably in vivo. Positively charged particles can damage cell membranes. |
Cells that persist in circulation: red blood cells and platelets. |
|
Ligand–receptor-mediated binding
|
Can potentially be used to attach particles to cells in vivo since attachment is mediated by specific cell receptors. The binding affinity could be quite strong. |
Ligand–receptor interaction may activate certain signaling pathway. |
Cells that express sufficient receptors which allow for multivalent binding: i.e., CD44–hyaluronan interaction allows for stable multivalent bonding to multiple cell types. |
|
Covalent surface coupling
|
Offers the strongest bond between particle and cell. May conjugate directly to amine or thiol groups present on all cells. |
Requires permanent modification to cell surface | Cells that migrate through endothelium: monocytes, macrophages and T cells since particle attachment must be strong. |
|
Encapsulation by endocytosis and other internalization mechanism
|
Leaves the cell membrane unmodified Can carry particles through endothelium |
Can lead to degradation of internalized biodegradable particles. May block sensing and releasing of cargoes from the environment. |
Cells capable of endocytosis, phagocytosis, etc. |
Size and charge
Size and charge are the two most basic parameters of NPs, and small differences in size and charge could have significant biological effects on cellular uptake and biodistribution [7, 61, 62]. Before moving into more specific RBC-related contents, it is beneficial to introduce the general influences of size and charge on NPs in vivo.
He et al. systematically synthesized a batch of chitosan derivative polymeric NPs [61]. These NP particles had sizes between 150 to 500 nm and had ζ-potentials from −40 mV to +35 mV. The study clarified that NPs with a higher surface charge and a larger particle size were more likely to be phagocytized by macrophages, while NPs that were approximately 150 nm in diameter with a slight negative charge tended to efficiently accumulate in tumors. This is consistent with the widely accepted understanding that highly positive NPs are more likely to attract a corona, be cleared, and display stronger acute toxicity. However, negatively charged NPs have been investigated less thoroughly and need further research. Nevertheless, these findings have served as guidelines in the design of drug nanocarriers. Another study pointed out that smaller NPs (<200 nm) could evade recognition by the MPS and thus prolong the circulation of NPs in the bloodstream [63]. Indeed, particles larger than 200 nm in size can activate the human complement system and then be cleared rapidly in the liver and spleen upon intravenous administration [7], which could occur because the higher curvature of smaller particles could prevent the efficient adhesion of opsonins to a certain extent [64, 65]. Thus, this appears to be an alternative way to reduce NP size for longer durations of action and higher activity. In the field of particle-delivered vaccine research, an increasing number of reports have shown that particle size may affect vaccine efficiency with different particle compositions. Benne et al. [66] demonstrated that smaller particles (<50 nm) can directly penetrate deeper into the lymph nodes, in contrast to larger particles, which are efficiently retained in the lymph nodes upon intradermal injection. Hence, it is worth selecting the optimal size to balance drainage and retention. Generally, for long-term circulation of NPs, the size of the particles should be either large enough to avoid renal filtration or small enough to minimize opsonization and MPS clearance. Based on the above studies, spherical particles with diameters between 10 and 200 nm appear to be a suitable range for long circulation times [5].
Focusing on RBC-centered systems, Zelepukin et al. demonstrated that using certain types of small 100 nm magnetic particles (50, 100 and 200 nm NPs, coated with polyacrylic acid (FluidMag-PAS, PAS); 100 nm NPs coated with glucuronic acid (FluidMag-ARA, ARA), chitosan (FluidMagChitosan, CTS), carboxymethyl-dextran (FluidMag-CMX, CMX), polyethylene glycol α-,ω-diphosphate (FluidMag-PEG/P, PEG/P), polystyrenesulfonate (FluidMag-PS); 100 nm uncoated anionic NPs (FluidMag-UC/A, UC/A); and 100 nm uncoated cationic NPs (FluidMag-UC/C, UC/C))—unlike larger conventional submicron NPs – to attach to RBCs can boost their lung accumulation and the accumulation of the drugs increased nearly 120-fold (up to 40% of the injected dose) [49]. In addition, sub-200 nm NPs of different sizes, coatings, and zeta potentials were examined to obtain the best RBC adsorption/desorption behavior. Data have proven that in the washing step, only the positively charged NPs are tightly bound to RBCs and hardly detach from the cell surface, which may be largely due to the negative charge of the RBC membrane. Therefore, for non-positively charged NPs, further binding mechanisms need to be employed to ensure satisfactory RBC adherence.
Shape
The role of shape in vascular dynamics has long been known, as it significantly affects the behavior of circulating cells such as RBCs and platelets [67, 68]. Unsurprisingly, the shape of NPs in the circulation has also attracted much interest because it could also have a significant impact on their cellular internalization, fluid dynamics, and interaction with vascular targets [69, 70]. However, there have been far fewer studies concerning the relationship between NP shape and RBCs. Hence, in this section, we will introduce some of the latest reports about NP shape from a more general perspective, which could provide clues about what may happen with RBCs and serve as the basis for more specific future research.
The interaction of spherical particles with cells has been investigated extensively, and many in vivo studies using animal models have been conducted. Recently, comparisons between spherical and nonspherical NPs showed some interesting results, bringing substantial attention to the effects of nonspherical NP shapes. To date, evidence has shown that the rate of internalization of NPs (such as filaments or rods) with a relatively large diameter is significantly reduced compared with that of spherical materials [71, 72].
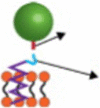
Kolhar et al. [68]. showed that the specificity of endothelial targeting by NPs with surface ligand coatings could be further enhanced by using rod shapes (Fig. 1). Compared with spherical NPs, these nonspherical NPs showed a higher distribution in the endothelium in diseased tissues under various experimental settings, including static cell cultures, microfluidics, and a mouse model. Moreover, a mathematical model of particle-surface interactions was employed to simulate the dynamic NP-EC contact process. The results showed that the higher affinity and specificity of nanorods are probably due to the balance of multivalent interactions, which is conducive to adhesion and entropic loss, as well as shear-induced separation that reduces the binding force.
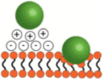
Fig. 1. The forces acting on nanoparticles under flow.
a Schematic of particles interacting with cells under flow. b SEM of polystyrene spheres and c elongated polystyrene particles (200 nm). Scale bar, 1 μm. d SMN of RBE4. With permission from [68].
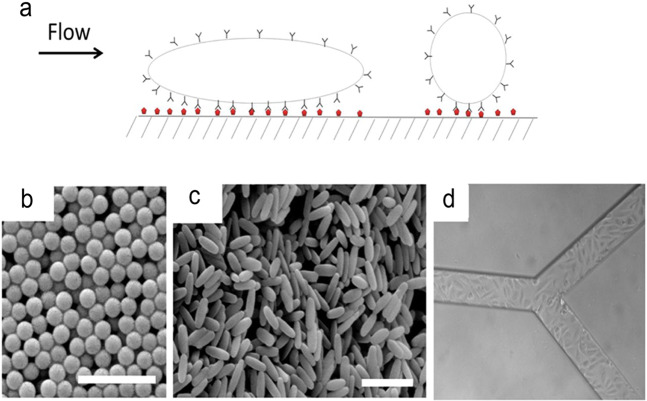
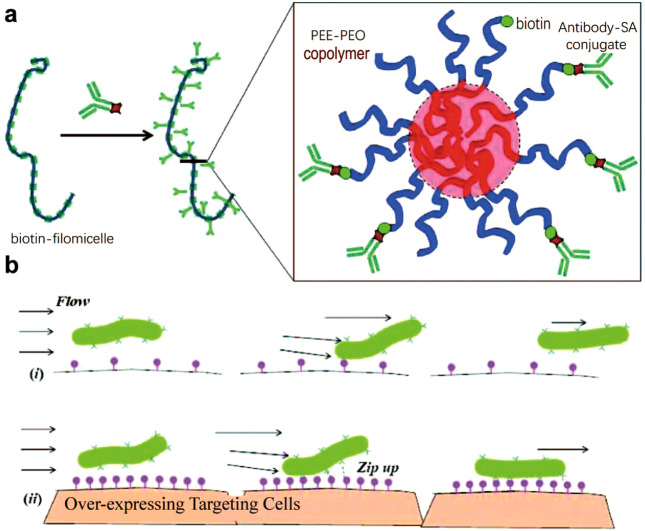
Similarly, to explore the role of carrier geometry in endothelial targeting, Shuvaev et al. designed and tested NPs with different shapes, notably disc-shaped and long flexible polymeric filomicelles that were self-assembled from polyethylene oxide diblock copolymers [71]. Figure 2 illustrates the design of these antibody-decorated filomicelles (Ab/filomicelles) and the proposed mechanism for anchoring them to target ECs in the bloodstream. Their results indicated that elongated carriers (mainly large disc-shaped and filamentous cells with a length from 3 to 7.5 μm) exhibit longer residence times in the bloodstream than spherical carriers (~150 nm). The author proposed that the decreased interactions with phagocytes and the blood vessel wall were largely attributed to the stretching effect of blood flow on nonspherical NPs. Hence, NPs with shorter cylindrical shapes are less affected by blood flow and have stronger interactions with phagocytes, resulting in more effective absorption and faster clearance from the circulation. These targeted Ab/filamentous cells combine high stealth, long circulation, high-target binding affinity and the ability to load cargoes; therefore, compared with spherical carriers, the new slender carriers have the potential to improve bioavailability and pharmacological effects [72].
Fig. 2. The design of antibody-modified fibroblasts (Ab/fibroblasts) and their anchoring mechanism to target endothelial cells in the bloodstream.
a The upper image shows the overall schematic diagram of biotinylated and antibody-coated filamentous cells (Ab/filomicelle). b Targeting long, soft filaments in the bloodstream with fewer (i) or more (ii) targeting sites. With permission from [71].
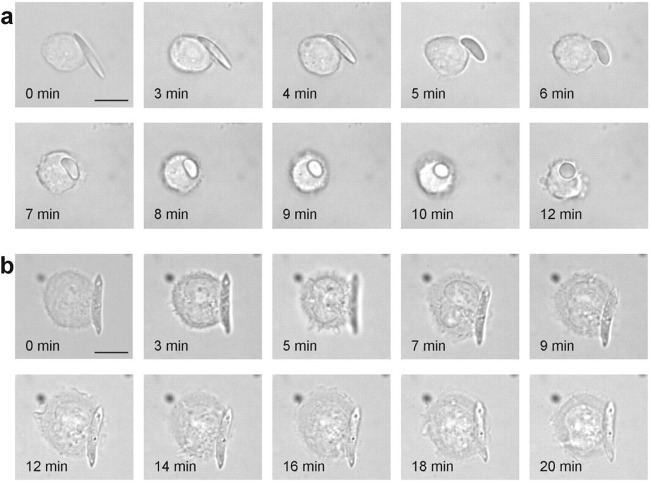
Another interesting topic is the interaction between shape-switchable particles and cells. Mitragotri et al. explored this field using polymeric particles with shape-changing ability, which is driven by a subtle balance between polymer viscosity and interfacial tension in a stimulus-responsive manner [73]. As demonstrated in Fig. 3, PLGA-ester elliptical disks (EDs) switched their shape to spheres after contacting the cell surface and were consequently internalized (Fig. 3a), while PLGA-acid EDs without shape-changing ability were not phagocytosed by macrophages (Fig. 3b).
Fig. 3. Time-lapse video microscopy clip of the shape-dependent phagocytosis of macrophages.
a The shape-switching PLGA ester ED initially attaches to macrophages without being engulfed. Once the shape changes to a nearly spherical shape, the macrophages rapidly internalize the particles. b Macrophages are scattered on PLGA acid ED, which does not change shape at pH 7.4 but cannot be engulfed completely (Scale bar: 10 μm). With permission from [73].
It has been suggested that the uptake of spherical particles is always advantageous, while the uptake of rod-shaped particles depends on the possibility that the particles will achieve a favorable contact angle, thereby negatively affecting the absorption of such particles [66, 74] (Fig. 4). This theory may explain the difference between the internalization of shape-changing EDs and non-shape-changing EDs.
Fig. 4. Flexible spheres, rigid spheres or rigid rods approaching the cell membrane at different angles, such as vertical or tangential angles, are absorbed by cells with different efficiencies.

Rigid spheres have greater uptake efficiency than flexible spheres, as flexible spheres deforms leading to slower uptake; while the angle at which the nanoparticles are in contact with the cell surface also makes a difference, with rigid rods that tend to be vertically close to the cell requiring much less energy to be taken up. With permission from [66].
To date, the investigation of nonspherical NPs is still in its early stage, and there are many unexplored yet promising areas in this field. There are even fewer data on using nonspherical NPs together with erythrocytes. Although it may be reasonably speculated that NPs with a large aspect ratio could avoid RBC internalization, there is no solid evidence on this topic to date. The influence of various shapes on NP-RBC binding, circulation time, membrane deformation, and RBC function remains to be explored. We expect more work to be carried out on this topic in the future.
Elasticity
Elasticity is a less explored field, but recent advances have suggested its importance in hemodynamic interactions. Again, due to the lack of RBC-specific studies, we will mainly present data on more general subjects.
Evidence suggests that elasticity could influence the margination and adhesion of RBCs to the vascular wall [75]. Some recent works reported the effect of elasticity (in the range of 0.5–63.8 kPa) on the circulation and biodistribution of RBC-carried particles [76, 77]. The results indicated that an eightfold reduction in the particle modulus brought about a more than 30-fold increase in the elimination half-life of the softer particles. Thus, it appears that softer RBC-shaped particles are less likely to be cleared. Anselmo et al. also proposed the potential benefits of using NPs with optimized elasticity for longer vascular-targeted circulation [76]. They fabricated PEG-based hydrogel NPs in a series of standard sizes (including 200 nm) with elastic moduli ranging from 0.255 to 3000 kPa and studied the pivotal role of particle elasticity in blood circulation time, biodistribution and endocytosis. The results demonstrated that softer NPs were more likely to be retained in the bloodstream in higher amounts at shorter time points and had increased blood retention times compared to those of harder NPs, which is likely caused by the tendency of softer particles to resist phagocytosis longer, as shown in in vitro macrophage phagocytosis experiments.
Guo et al. [78] synthesized a hybrid nanolipogel-hydrogel vehicle with different elastic moduli (from 45 ± 9 kPa to 19 ± 5 MPa). They found that lower elastic moduli were associated with greater accumulation in tumors, with up to a fivefold increase in in vitro cellular uptake and a 2.6-fold increase in in vivo tumor uptake. To understand how elasticity affects cell-NP interactions, Hui et al. [77] fabricated a series of silica nanocapsules (SNCs) (~150 nm) with elastic moduli ranging from 0.56 to 1.18 kPa and examined their performance in macrophage uptake and receptor-mediated cancer cell uptake. The results revealed that rigid SNCs remained spherical during cellular uptake, while soft SNCs were deformed due to specific ligand–receptor interactions and membrane encapsulation. The deformation appeared to reduce their cellular adhesion and endocytosis rate. This conclusion is also consistent with previously discussed studies.
Because softer particles seem to exhibit longer circulation time than hard particles, it is assumed that soft particles may be deformed under the phagocytic force of phagocytes, resulting in a decrease in the radius of curvature of the particles, so as the radius of curvature of the particles decreases, phagocytosis becomes more and more difficult [79, 80]. Another possibility is that the degree of phagocytosis may be reduced due to the increase in cell membrane tension caused by the actin structure, while the actin structure must continue to complete phagocytosis; in this case, phagocytosis is significantly reduced as the radius of curvature of spherical particles decrease [76, 81].
The synergistic effects of NP properties
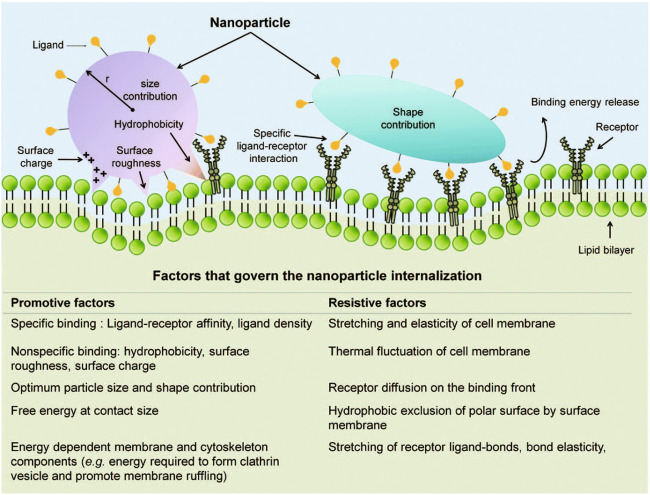
In the previous sections, the effects of NP size, charge, shape, and elasticity were separately reviewed, and most of the studies above illuminated the effects of either one or two physical properties on cellular interactions. However, in real-world application settings, different NP parameters and their interplay determine the final performance together, including the delivery efficiency, retention time, biodistribution, and toxicity. Indeed, many studies have systematically evaluated the combined effects of particle size, shape, and elasticity on particle margination to the vascular walls in cardiovascular diseases [10, 11]. Figure 5 presents an overview of the interplay among multiple properties in cellular interactions [8, 82]. In some specific application scenarios, such as organ-specific delivery, cancer imaging [10], cardiovascular diseases [11], and anticancer drug delivery [83], the synergistic effects of NP physical properties (e.g., size, shape and stiffness) have been clearly demonstrated. However, there is still a lack of data in many other settings, and hence, to date, there is no clear and widely recognized pattern governing particle behavior in general terms. Specific RBC-NP-related research on these combined effects is thus urgently needed for advancements in this field.
Fig. 5. Synergistic contributions to the NP internalization process.
Physical interactions, specific ligand–receptor interactions and the characteristics of NPs are required to overcome resistance to internalization. With permission from [53].
By manufacturing layer-by-layer adjustable particles with constant surface chemistry, the macrophage Fc receptor-mediated macrophage interaction can be studied. In the context of reduced internalization of spherical particles by macrophages, the increase in size plays an important role in reducing stiffness. The internalization of rod-shaped rather than spherical particles is highly dependent on stiffness [82]. These physical parameters affect the penetration of NPs across the blood–brain barrier (BBB) [67], and results have shown that 200 nm particles show higher BBB transmission than 100 and 500 nm spheres.
Rod-shaped particles showed higher BBB transport after normalization to endothelial binding, while soft particles showed transport comparable to that of hard particles, which proves that the physical parameters of NPs may enhance their ability to pass through the BBB.
Erythrocyte-binding methods
The methods used to attach NP carriers to the surface of RBCs vary widely and may be roughly categorized into a few groups: physical methods (mostly noncovalent interactions), covalent linking reactions with or without erythrocyte modification, and biological anchoring strategies, including cell surface receptor/molecule coupling and affinity conjugation (e.g., antibodies and antibody fragments). These binding methods will be introduced and discussed below.
Physical absorption
Noncovalent attachment of NPs to the surface of natural erythrocytes normally has low selectivity. In some cases, the surface of erythrocytes is modified for better binding strength or higher binding specificity. Either way, this type of surface loading is normally conducted ex vivo by incubation with intact or modified erythrocytes to reduce nonspecific binding or for easier manipulation.
Chambers et al. [4] physically attached NPs (200–450 nm) to the surface of erythrocytes to prolong their intravascular circulation, and the results showed that ~5% of RBCs loaded with NPs remained in the circulation for 12 h. Among the different NPs, 220 and 450 nm particles exhibited the longest circulation time. Scanning electron microscopy (SEM) was used to examine the morphology of erythrocyte-bound NPs of different sizes and revealed no significant shape change in NPs or host RBCs. The authors proposed that this physical adhesive interaction might be a result of van der Waals forces, electrostatic interactions, hydrogen bonding, and hydrophobic forces between PS NPs and RBCs.
Zelepukin et al. [49] studied the efficiency of RBC hitchhiking for eight types of small sub-200 nm magnetic NPs with different coatings (50, 100 and 200 nm NPs coated with polyacrylic acid (FluidMag-PAS, PAS); 100 nm NPs coated with glucuronic acid (FluidMag-ARA, ARA), chitosan (FluidMagChitosan, CTS), carboxymethyl-dextran (FluidMag-CMX, CMX), polyethylene glycol α-,ω-diphosphate (FluidMag-PEG/P, PEG/P), polystyrenesulfonate (FluidMag-PS); 100 nm uncoated anionic NPs (FluidMag-UC/A, UC/A); and 100 nm uncoated cationic NPs (FluidMag-UC/C, UC/C)). They found that only positively charged NPs displayed improved blood retention. The authors suggested that positively charged particles bind to the membrane surface more easily via electrostatic interactions since the surface of RBCs is negatively charged. They also discovered that hydrophobic NPs exhibited a higher binding rate than hydrophilic NPs, which may be attributed to the binding preferences of hydrophobic cell membranes. Importantly, they also found that two major factors—particle size and the particle/cell ratio—had a strong influence on adhesion efficiency. Oversized NPs displayed a much lower binding number. A high particle/cell ratio could increase the number of RBC-loaded NPs, but overloading of particles led to cell agglutination and deformation. Consistent with this result, other studies also found that at a lower particle/cell ratio (~3–4 NPs per RBC), 200 nm PS NPs did not cause changes in the circulation time of RBCs, while an inappropriately high particle/cell ratio induced strong local membrane deformation on RBCs [9, 84]. This deformation led to spiculation of RBCs, cellular uptake of the particles, and eventual hemolysis [85], or even increased RBC sensitivity to osmotic and mechanical stress [9, 52].
Normally, particle adhesion via physical interaction is reversible, as the affinity is not high enough to keep the NPs stably anchored (Fig. 6). Nevertheless, this adhesion could reduce NP uptake by MPS-related organs (liver and spleen) to some extent [48], and the pharmacokinetics of the RBC/NP complex were more favorable than those of free NPs in many clinical settings. Many studies have demonstrated that nontargeted particles delivered via physical interactions (RBC hitchhiking) could boost delivery to the lungs or aggressive and small-cell cancer types, as well as other lung diseases [48, 49, 59]. Although this type of nonpermanent binding may be suitable for some applications, in many other situations, higher cargo-RBC affinity is more desirable; hence, other strategies need to be employed.
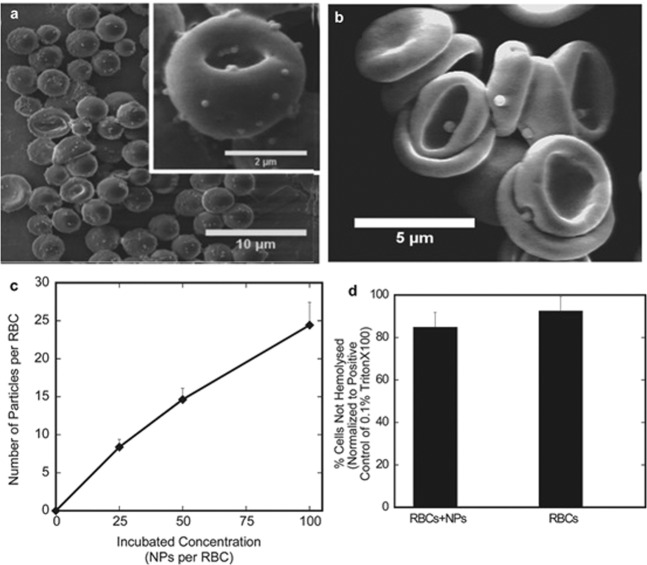
Fig. 6. Attachment of nanoparticles to erythrocytes.
a SEM of 200 nm polystyrene spheres (PS spheres) attached to RBCs. b SEM of 500 nm PS spheres attached to erythrocytes. c The average number of 200 nm nanoparticles attached to each RBC as the particle concentration increases, shown by 3H labeling. d RBC-NPs under high load (100:1 NP:RBC load ratio) have no obvious lysis effect compared to RBCs in the same buffer. With permission from [48].
Covalent binding
Cargos can also be loaded into erythrocytes by covalent coupling to the surface of RBC membranes, which normally offers higher binding affinity. Because this type of process normally requires a controlled reaction environment, they are normally performed ex vivo as well. In early studies, avidin-functionalized particles were often used to readily bind biotinylated RBCs via chemical conjugation, which results in stable adhesion between synthetic drug-carrier NPs and RBCs [18, 86]. Various covalent binding methods were subsequently explored, including conjugation via thiol groups [87] and lipids [88].
Chambers et al. [15] analyzed the behavior of RBC-attached NPs and improved adhesion by covalently binding C-mPEG (cyanuric chloride functionalized mPEG) to amine groups on the cell surface. By PEGylating erythrocytes, the interaction between erythrocytes can be reduced, thus diminishing the shedding rate of particles. In this study, the circulation half-life of RBC-bound particles was increased to over 24 h, and ~3% of particles remained in the circulation even after 1 week.
There is a substantial amount of free thiols on the surfaces of cells, which could be another option to anchor NPs. For instance, thiol-reactive maleimide-modified liposome-like synthetic NPs with a size of 100–300 nm were covalently linked to the surface of thiol-rich T cells via thiol-maleimide conjugation in a study performed by Stephan et al. [87]. This is achieved through a simple two-step process (Fig. 7b): NPs bind to targeted cells via maleimide-thiol coupling first, followed by in situ conjugation with thiol-terminated polyethylene glycol (PEGylation) to quench unreactive groups on the particles. As a result, the T cells maintained physical attachment of 83% (± 3%) of their original NP cargo without compromising key cellular functions after crossing the endothelial barrier. Though the authors did not use RBCs for conjugation in this study, similar methods could be used with RBCs, as they also have abundant free surface thiol groups.
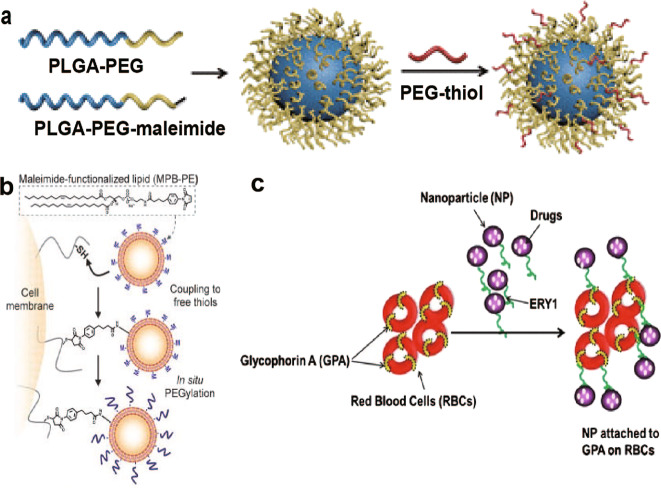
Fig. 7. Some modifications based on covalent binding and ligand-anchoring.
a Schematic illustration of the long blood circulation of PLGA-PEG-NPs with a dynamic topographical structure of the PEG shell (PLGA-TPEG-NPs). b Covalent coupling of NPs to the T-cell membrane via thiol-maleimide conjugation. c Schematic diagram of the targeting peptide ERY1 (green chain) conjugated to nanoparticles (purple circles) binding to glycophorin A (GPA) receptor (yellow bands) on the RBC membrane (red). Modified with permission from [16, 87, 89].
Another interesting mechanism to achieve long circulation is to engineer a hierarchical PEG structure on the NP surface [89]. This type of PEG cloak is theorized to have dual physical effects: the dense inner PEG layer would thermodynamically prevent proteins via steric repulsion, while an outer layer of PEG arranged in a mushroom-like conformation would yield a topographical structure that provides fluctuating conformational effects to interfere with protein-binding kinetics (Fig. 7a), leading to long circulation [89]. The authors explored the effect of topographical structure on NP blood circulation and used a series of PLGA-PEG-MAL percentages (PLGA-TPEG-NPs-10, 20, 40, 60, 80, and 100, in which the numbers indicate the percentages of PLGA-PEG-MAL used in NP fabrication). The results revealed that PLGA-TPEG-NPs-20 displayed the longest circulation half-life of 10.01 ± 0.52 h. This circulation time appears to be lower than that reported for the cellular-hitchhiking strategy in [48, 49], probably due to the absence of RBC camouflaging and the reduced inhibition of nonspecific protein adsorption. It is worth noting that recent studies have reported a phenomenon called “accelerated blood clearance (ABC)”, in which a second dose of PEGylated NPs is rapidly cleared when administered a few days after the first dose [90]. Although it is possible that ABC is related to the adaptive immune response, the mechanism underlying this phenomenon is still unclear, and further investigations are required. Nevertheless, combining this alternative NP surface modification strategy with RBC hitchhiking may be a promising way to further improve the performance of nano-DDS.
Ligand-anchoring
Conjugating NPs with antibodies could grant NPs specific and selective antigen-binding ability [88]. Compared with the typical ex vivo covalent binding manipulation of RBCs and reinfusion, specific targeting makes it possible to load NPs into RBCs in vivo, which may reduce the possibility of RBC damage and greatly simplify clinical procedures, making this approach feasible for clinical applications. Moreover, it is generally expected that noncovalent surface modification using affinity ligands will use milder conditions than covalent coupling schemes, which usually include multiple washes, hypotonic opening, and chemical modification. In addition to this straightforward one-step loading approach, a two-step approach is also possible. It is possible to load intermediate substances that specifically bind to RBC surface proteins, such as peptides, antibodies and antibody fragments, and then bind the drugs to these ligands. This method can avoid potential changes in surface molecules that regulate the survival of RBCs in vivo and achieve cargo attachment in specific locations at the same time [1]. Despite all these advantages, there are also disadvantages for this type of binding method. Because biomolecules with high molecular weights are often used, this type of binding mechanism may have a higher cost, and the full complex may contain a large ratio of binding molecules.
Nevertheless, despite the abundance of potential sites for coupling, these epitopes are rarely evaluated for their stability and suitability in drug delivery. Since the surface ligands selected for RBC coupling should have a sufficient quantity to maintain an appropriate binding reservoir and reduce interference with the original function, some target antigens that may meet this requirement were listed by Villa et al. [18].
In one study, a large random peptide library from bacteria was screened to identify unique peptide ligands in RBCs [91]. One of these substances, glycophorin A (GPA), which is a type I membrane-bound sialo glycoprotein with receptor function, accounts for ~2% of the total RBC membrane protein and can be used as a target site [18]. Sahoo et al. attached a synthetic 12-amino acid peptide (ERY1)—which specifically binds to GPA—to NPs loaded with BSA (as a model drug) to target GPA and improve the circulation time of NPs (Fig. 7c) [16]. The results showed that drug targeting with ERY1-NPs was efficiently achieved. Confocal and flow cytometry data indicate that ERY1-NPs could bind to RBCs efficiently and irreversibly, with ~70% of erythrocytes bound after 24 h in a physiologic flow loop model, compared to 10% binding of NPs without ERY1. Further analysis also indicates that this binding is relatively safe for RBCs.
Complement receptor type 1 (CR1) is another anchoring candidate. The majority of CR1 (~90%) is found on RBCs (~500–1500 copies per cell) [92]. Tissue plasminogen activator (tPA, which accelerates clot breakdown) was combined with a monoclonal antibody that recognizes CR1 and anchors tPA to circulating RBCs (Fig. 8) [41]. This anti-CR1/tPA conjugate shows stable CR1 binding and prolonged fibrinolytic activity in blood vessels, while reducing harmful tPA-induced hemostasis and thrombolysis.
Fig. 8. Coupling of tPA to circulating RBCs reduces rebleeding.
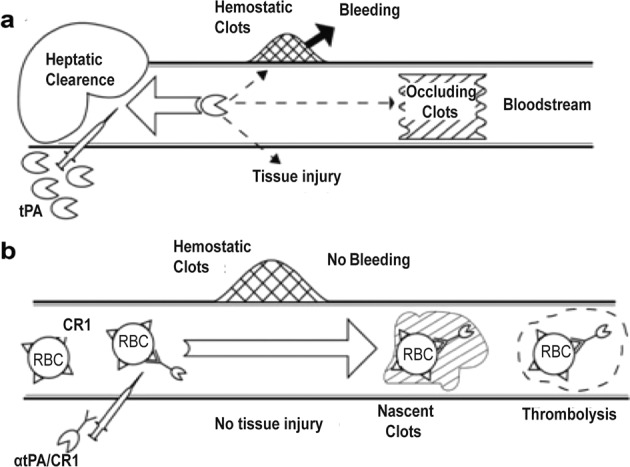
a Rapid clearance by the liver prohibits preventive use and necessitates large doses of tPA for injection, which can induce hemostasis and clots in tissues, causing bleeding and other side effects. b The injected anti-CR1/tPA is mainly combined with RBCs rather than being present in hemostatic clots and extravascular tissues; at the same time, new intravascular clots are incorporated and dissolved. Modified with permission from [41].
To solve the challenges related to the translation and application of chemically generated antibody/tPA conjugates and improve clinical applicability, the scFv fragment of Ter-119, a monoclonal antibody that recognizes mouse GPA, was used to anchor the injected PA to circulating RBCs [93]. As a result, at 48 h after intravenous (IV) injection in mice, ~95% of circulating scFv/uPA-T was still bound to RBCs, and scFv/uPA-T could effectively prevent arterial and venous thrombosis for up to 24 h. These developments reveal practical possibilities for noncovalent RBC anchoring, and there are still many candidates and strategies to explore.
Lipid modification
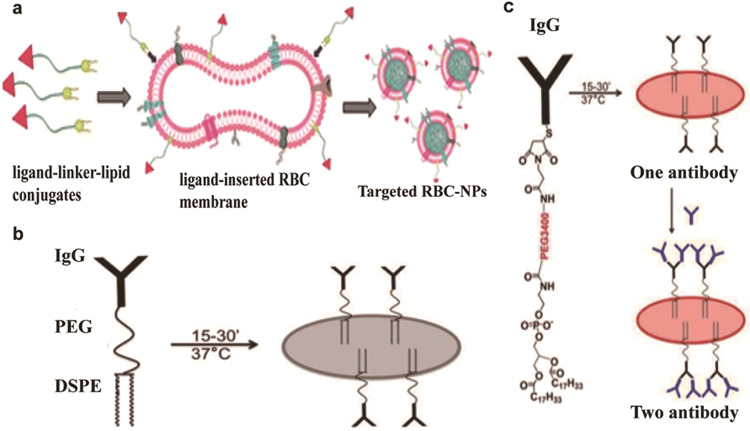
Fully functional membrane proteins are necessary for immune evasion and other physiological activities of RBCs; thus, preserving the function of these proteins is important during NP anchoring [94]. Because chemical conjugation technology widely uses carboxyl-, amine-, or thiol-based chemical methods to modify synthetic nanocarriers with targeted ligands, the presence of biological components on the cell surface requires noninterference functionalization strategies. To address the problem of chemical denaturation, Fang et al. [95] reported a method of physically inserting a ligand-linker-lipid conjugate into RBC membranes to produce functionalized RBC membranes without destroying existing surface proteins. As shown in Fig. 9a, the targeting moiety was bound to the RBC membrane via lipid tethering. In this study, both folic acid (FA) and the insertion of the nucleolar targeting aptamer AS1411 showed receptor-specific targeting for model cancer cell lines. It seems that this scheme can also be transferable to anchor NPs to RBCs.
Fig. 9. Methods based on lipid modification on the surface of red blood cells.
a Schematic diagram of the preparation of ligand-functionalized RBC membranes, which encapsulated polymeric cores as targeted RBC-NPs. b Schematic diagram of the DSPE-PEG-IgG construct incorporated into the RBC membrane. c Schematic diagram of the DSPE-PEG3400-maleimide construct modified with thiolated IgG incorporated into the RBC membrane. Modified with permission from [95–97].
In another study, extraneously added anchors made of dioleoyl phosphatidylethanolamine lipoprotein fragments were constructed to add functional molecules to the RBC surface (Fig. 9b) [96]. These molecules were then used to link the model ligand thiolated IgG (already coupled with DSPE-PEG3400-mal). The prepared IgG-RBC complex showed prolonged circulation in vivo (70% of the injected dose after 48 h) and efficient retention of IgG on the RBC membrane, with a half-life of 73 h. Based on a previously described lipophilic ligand painting strategy, the same group also used RBCs to selectively capture cancer cells or leukocytes in vivo and in vitro (Fig. 9c) [97]. In summary, lipophilic ligand-related modifications could be very useful to functionalize RBCs for selective targeting, tagging, and depletion of cargos (including cells) [98, 99].
Integrating different adherence methods and NP properties
The studies presented above mostly used the different anchoring methods separately but generally did not take the influence of NP properties into account in detail. However, using different methods together with more careful NP design is also possible. Although our understanding of these combined effects is still far from satisfactory, some groups have made interesting attempts and obtained valuable results, as discussed below. It was suggested that the combined use of different methods and NP design could improve overall efficiency and reduce adverse effects. These integrated designs may also make the resulting therapy more practical in clinical settings.
Anselmo’s group [60] conducted a research by applying a combination of RBC hitchhiking, a rod-like shape, and antibody attachment to investigate synergistic contributions based on a lung inflammation model (Fig. 10). In this model, ICAM is overexpressed on the lung endothelium; hence, an ICAM antibody was used to decorate the NP surface. To evaluate the ultimate ability of NPs to avoid immune system clearance and improve lung targeting, the authors compared NPs generated with the ICAM antibody (for lung targeting), a rod-like shape (for phagocytosis escape), and/or RBC hitchhiking (for long circulation), individually and in combination. The results showed that, compared with any single aspect and any combination of two aspects, the combination of these three aspects can effectively reduce the cumulative concentration of NPs in the main clearance organs by more than twofold. This combination also increased the accumulation of NPs in the lungs by 1.2- to 8.9-fold.
Fig. 10. Schematic diagram of the chemical, physical, and biological modifications investigated for RES avoidance and lung targeting.
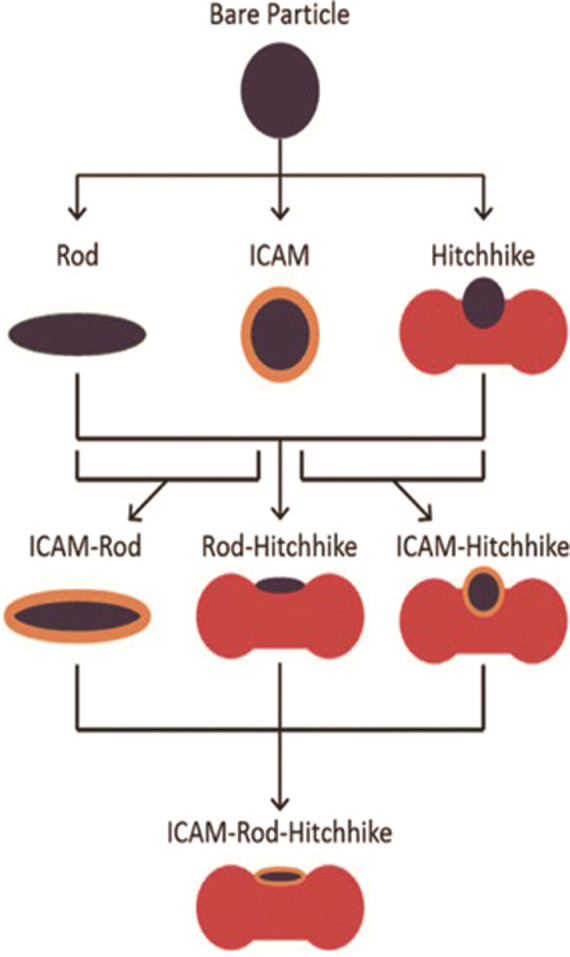
Combining the three factors of chemistry, physics and biology, ICAM-Rod-Hitchhike complex was synthesized. With permission from [60].
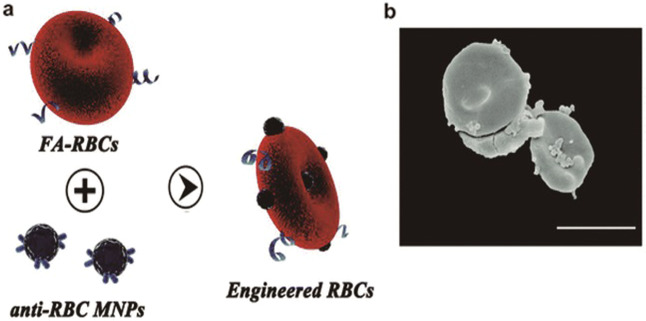
Another example is the use of NP anchoring to RBCs to capture circulating tumor cells (CTCs) in the blood flow for early cancer diagnosis, treatment monitoring, metastasis diagnosis, and prognostic evaluation [99]. In this study, the tumor-targeting molecule FA and magnetic nanoparticles were bound on the surface of RBCs by hydrophobic interactions and chemical conjugation, respectively (Fig. 11), leading to the isolation of the CTC–RBC complex in a magnetic field. This system showed an outstanding capture efficiency above 90%, and the purity of the obtained CTCs was higher than 75%, which is comparable to that of the magnetic-activated cell sorting method (80% capture efficiency and 20% purity) under the same conditions. This study implies that RBCs could also be used as a platform for cargos attached to RBCs to repeatedly interact with substances or objects in the blood flow.
Fig. 11. Preparation of engineered RBCs.
a Schematic of preparation of engineered RBCs. b SEM image of engineered RBCs. (Scale bar: 10 μm). Modified with permission from [99].
These studies provide good examples showing the potential of erythrocyte-based therapies when different techniques are used together appropriately. With more knowledge on how different methods and factors can influence these NP erythrocyte systems, it is possible to further improve efficiency and safety by appropriately coordinating these factors and methods. A deeper understanding of individual methods/factors and additional attempts to combine them are both important.
Using RBCs across individuals
One of the key issues for wider application of erythrocyte-based nanomedicines is how to safely use heterogeneous erythrocytes; this issue is primarily linked to their immunogenicity. Without properly addressing this issue, in the worst case, the source of erythrocytes is limited to the recipient him- or herself. These limitations could severely restrict the clinical use of this type of therapy. Although this issue is not the exact theme of this review, as surface modification (which is a major topic in this review) is an important process in engineering heterogeneous erythrocytes and could be of interest to some readers, some of the recent advances in this field will be presented below.
In clinical practice, RBCs remain antigenically challenged despite ABO/RhD matching. Thus, for subjects receiving multiple blood transfusions, the immune response to allogeneic erythrocytes represents a serious adverse impact of blood transfusion. Injecting covalently linked antigen peptides into RBCs can induce or inhibit antigen-specific immune responses, so this approach has been used as an immunotherapy based on RBC delivery for experimental cancers and autoimmune diseases in vivo [37]. RBCs have also been tested in innovative cancer immunotherapy, which can deliver tumor-associated antigens to APCs, thereby inducing antigen-specific immune responses in tumor animal models [100, 101]. For this reason, modified RBCs or an RBC-derived carrier system should match the blood type and Rh type of patients [102]; in these cases, whole blood should be collected to avoid harmful immunogenic reactions. Alternatively, RBCs could be membrane engineered, and PEG modification could effectively camouflage RBC-based NPs from being recognized by the immune system to a certain extent without affecting the functions of RBC membrane glycoproteins or RBC biocompatibility [103]. Additional studies using alternative PEG coupling techniques with different chemical groups produced promising results in concealing ABO and Rh(D) blood group antigens [104, 105] (e.g., mPEG surface modification; some information is given in Fig. 12 [51]) to reduce RBC antigen immunogenicity and reduce phagocytic degradation by heterologous phagocytes [38, 106]. These “universal” RBCs may reduce the risk of potentially life-threatening hemolytic transfusion reactions as well as shortages of matched blood type reserves. These findings also have important implications for certain types of RBC-assisted DDS, as it will significantly increase clinical usability when one type of ready-made formulation can be used on all patients without additional customization.
Fig. 12. Schematic diagram of a series of reactions for camouflaging erythrocytes with poly(ethylene glycol) (PEG) via reaction with erythrocytes surface amines.
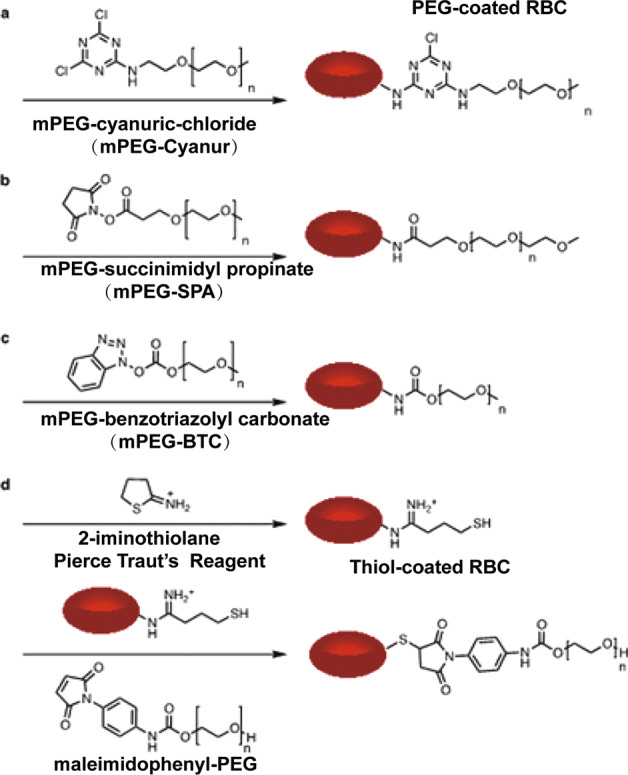
a Cyanuric chloride activated PEGylation, b N-hydroxysuccinimide (NHS) amide coupling, c benzotriazolyl carbonate amide coupling, and d Pierce Traut’s reagent thiol generation followed by a thiol-Michael reaction with maleimide. With permission from [51].
In fact, two companies in Europe are actively engaged in industrializing this process and may take this technology to clinical trials. They successfully developed a controlled drug release therapeutic solution(GR-ASPA) and a stable enzyme delivery system [107]. The complex interaction between donor and recipient biology after blood transfusion is still unclear in many respects. The addition of another variable parameter (drug factor) to the system represents a new challenge in the field of blood transfusion and transfusion medicine [100].
With an adequate understanding of the mechanisms of allogeneic transfusion, pharmacodynamic delivery, and RBC-based drug delivery, unnecessary complications can be avoided. RBC-based drug delivery also represents a new type of system that can address foreseeable indications in the future.
Conclusion
Long-circulating erythrocyte-based DDS are attractive not only because they can prolong the half-life of drugs but also because of their rich pharmaceutical functions, good biocompatibility, and abundant modification options. In this type of DDS, NP fabrication and RBC modification are two essential aspects. This review introduces and analyzes the effects of the properties of NPs and the characteristics of various surface-anchoring strategies in erythrocyte-NP DDS. Due to the insufficient research data on many specific topics in this field, we also presented some more general studies, which could provide valuable insights for further investigations. In addition, we briefly discussed the pros and cons of these strategies.
There are a growing number of studies in this field, but these delivery regimens currently remain in an early stage of development. Hence, there are still many challenges to overcome in the production and complete characterization of these delivery regimens. For instance, so far, the RBC-NP drug delivery method is mainly used in in vitro treatment, and there are relatively few studies on in vivo manipulation. Therefore, further animal and clinical experiments need to be performed to test the efficacy and safety of cell-drug binding in the future.
Nonetheless, despite all these challenges, RBC-NP-based delivery systems still offer a promising approach to exploit the most abundant endogenous cells in a multitude of medical applications.
Acknowledgements
This work was supported by National Natural Science Foundation of China (Nos. 81872824 and 51721091). Sichuan University provided necessary services for the writing of the manuscript.
Competing interests
The authors declare no competing interests.
Contributor Information
Zhi-rong Zhang, Email: zrzzl@vip.sina.com.
Zhen-mi Liu, Email: zhenmiliu@scu.edu.cn.
References
- 1.Hu CMJ, Fang RH, Zhang LF. Erythrocyte-inspired delivery systems. Adv Healthc Mater. 2012;1:537–47. doi: 10.1002/adhm.201200138. [DOI] [PubMed] [Google Scholar]
- 2.Mohandas N, Gallagher PG. Red cell membrane: past, present, and future. Blood. 2008;112:3939–48. doi: 10.1182/blood-2008-07-161166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sheng Y, Liu CS, Yuan Y, Tao XY, Yang F, Shan XQ, et al. Long-circulating polymeric nanoparticles bearing a combinatorial coating of PEG and water-soluble chitosan. Biomaterials. 2009;30:2340–8. doi: 10.1016/j.biomaterials.2008.12.070. [DOI] [PubMed] [Google Scholar]
- 4.Chambers E, Mitragotri S. Prolonged circulation of large polymeric nanoparticles by non-covalent adsorption on erythrocytes. J Control Release. 2004;100:111–9. doi: 10.1016/j.jconrel.2004.08.005. [DOI] [PubMed] [Google Scholar]
- 5.Yoo JW, Chambers E, Mitragotri S. Factors that control the circulation time of nanoparticles in blood: challenges, solutions and future prospects. Curr Pharm Des. 2010;16:2298–307. doi: 10.2174/138161210791920496. [DOI] [PubMed] [Google Scholar]
- 6.Schipper ML, Iyer G, Koh AL, Cheng Z, Ebenstein Y, Aharoni A, et al. Particle size, surface coating, and PEGylation influence the biodistribution of quantum dots in living mice. Small. 2009;5:126–34. doi: 10.1002/smll.200800003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hoshyar N, Gray S, Han HB, Bao G. The effect of nanoparticle size on in vivo pharmacokinetics and cellular interaction. Nanomedicine. 2016;11:673–92. doi: 10.2217/nnm.16.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Albanese A, Tang PS, Chan WC. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu Rev Biomed Eng. 2012;14:1–16. doi: 10.1146/annurev-bioeng-071811-150124. [DOI] [PubMed] [Google Scholar]
- 9.Pan D, Vargas-Morales O, Zern B, Anselmo AC, Gupta V, Zakrewsky M, et al. The effect of polymeric nanoparticles on biocompatibility of carrier red blood cells. PLoS One. 2016;11:e152074. doi: 10.1371/journal.pone.0152074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Toy R, Hayden E, Shoup C, Baskaran H, Karathanasis E. The effects of particle size, density and shape on margination of nanoparticles in microcirculation. Nanotechnology. 2011;22:115101. doi: 10.1088/0957-4484/22/11/115101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ta HT, Truong NP, Whittaker AK, Davis TP, Peter K. The effects of particle size, shape, density and flow characteristics on particle margination to vascular walls in cardiovascular diseases. Expert Opin Drug Deliv. 2018;15:33–45. doi: 10.1080/17425247.2017.1316262. [DOI] [PubMed] [Google Scholar]
- 12.Magnani M, Pierigè F, Rossi L. Erythrocytes as a novel delivery vehicle for biologics: from enzymes to nucleic acid-based therapeutics. Ther Deliv. 2012;3:405–14. doi: 10.4155/tde.12.6. [DOI] [PubMed] [Google Scholar]
- 13.Brahler M, Georgieva R, Buske N, Muller A, Muller S, Pinkernelle J, et al. Magnetite-loaded carrier erythrocytes as contrast agents for magnetic resonance imaging. Nano Lett. 2006;6:2505–9. doi: 10.1021/nl0618501. [DOI] [PubMed] [Google Scholar]
- 14.Muzykantov VR. Drug delivery by red blood cells: vascular carriers designed by mother nature. Expert Opin Drug Deliv. 2010;7:403–27. doi: 10.1517/17425241003610633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chambers E, Mitragotri S. Long circulating nanoparticles via adhesion on red blood cells: mechanism and extended circulation. Exp Biol Med. 2007;232:958. [PubMed] [Google Scholar]
- 16.Sahoo K, Koralege RSH, Flynn N, Koteeswaran S, Clark P, Hartson S, et al. Nanoparticle attachment to erythrocyte via the glycophorin A targeted ERY1 ligand enhances binding without impacting cellular function. Pharmacol Res. 2016;33:1191–203. doi: 10.1007/s11095-016-1864-x. [DOI] [PubMed] [Google Scholar]
- 17.Fu Y, Liu W, Wang LY, Zhu BY, Qu MK, Yang LQ, et al. Erythrocyte-membrane-camouflaged nanoplatform for intravenous glucose-responsive insulin delivery. Adv Funct Mater. 2018;28:1802250. [Google Scholar]
- 18.Villa CH, Anselmo AC, Mitragotri S, Muzykantov V. Red blood cells: supercarriers for drugs, biologicals, and nanoparticles and inspiration for advanced delivery systems. Adv Drug Deliv Rev. 2016;106:88–103. doi: 10.1016/j.addr.2016.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jensen FB. The dual roles of red blood cells in tissue oxygen delivery: oxygen carriers and regulators of local blood flow. J Exp Biol. 2009;212:3387–93. doi: 10.1242/jeb.023697. [DOI] [PubMed] [Google Scholar]
- 20.Hamidi M, Zarrin A, Foroozesh M, Mohammadi-Samani S. Applications of carrier erythrocytes in delivery of biopharmaceuticals. J Control Release. 2007;118:145–60. doi: 10.1016/j.jconrel.2006.06.032. [DOI] [PubMed] [Google Scholar]
- 21.Rossi L, Pierige F, Antonelli A, Bigini N, Gabucci C, Peiretti E, et al. Engineering erythrocytes for the modulation of drugs’ and contrasting agents’ pharmacokinetics and biodistribution. Adv Drug Deliv Rev. 2016;106:73–87. doi: 10.1016/j.addr.2016.05.008. [DOI] [PubMed] [Google Scholar]
- 22.He HN, Ye JX, Wang YS, Liu Q, Chung HS, Kwon YM, et al. Cell-penetrating peptides meditated encapsulation of protein therapeutics into intact red blood cells and its application. J Control Release. 2014;176:123–32. doi: 10.1016/j.jconrel.2013.12.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Villa CH, Pan DC, Zaitsev S, Cines DB, Siegel DL, Muzykantov VR. Delivery of drugs bound to erythrocytes: new avenues for an old intravascular carrier. Ther Deliv. 2015;6:795–826. doi: 10.4155/tde.15.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bird J, Best R, Lewis DA. The encapsulation of insulin in erythrocytes. J Pharm Pharmacol. 1983;35:246–7. doi: 10.1111/j.2042-7158.1983.tb02921.x. [DOI] [PubMed] [Google Scholar]
- 25.Harisa G, Ibrahim MF, Alanazi FK. Characterization of human erythrocytes as potential carrier for pravastatin: an in vitro study. Int J Med Sci. 2011;8:222–30. doi: 10.7150/ijms.8.222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Beutler E, Dale GL, Guinto E, Kuhl W. Enzyme replacement therapy in Gaucher’s disease preliminary clinical trial of a new enzyme preparation. Proc Natl Acad Sci USA. 1977;74:4620–3. doi: 10.1073/pnas.74.10.4620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Baysal SH, Uslan AH, Pala HH, Tuncoku O. Encapsulation of PEG-Urease/PEG-AlaDH within sheep erythrocytes and determination of the system’s activity in lowering blood levels of urea in animal models. Artif Cells Blood Substit Immobil Biotechnol. 2007;35:391–403. doi: 10.1080/10731190701460259. [DOI] [PubMed] [Google Scholar]
- 28.Millan CG, Castaneda AZ, Marinero MLS, Lanao JM. Factors associated with the performance of carrier erythrocytes obtained by hypotonic dialysis. Blood Cells Mol Dis. 2004;33:132–40. doi: 10.1016/j.bcmd.2004.06.004. [DOI] [PubMed] [Google Scholar]
- 29.Bax BE, Bain MD, Fairbanks LD, Webster ADB, Chalmers RA. In vitro and in vivo studies with human carrier erythrocytes loaded with polyethylene glycol-conjugated and native adenosine deaminase. Br J Haematol. 2000;109:549–54. doi: 10.1046/j.1365-2141.2000.02059.x. [DOI] [PubMed] [Google Scholar]
- 30.Banz A, Cremel M, Rembert A, Godfrin Y. In situ targeting of dendritic cells by antigen-loaded red blood cells: A novel approach to cancer immunotherapy. Vaccine. 2010;28:2965–72. doi: 10.1016/j.vaccine.2010.02.013. [DOI] [PubMed] [Google Scholar]
- 31.Wang GP, Guan YS, Jin XR, Jiang SS, Lu ZJ, Wu Y, et al. Development of novel 5-fluorouracil carrier erythrocyte with pharmacokinetics and potent antitumor activity in mice bearing malignant ascites. J Gastroenterol Hepatol. 2010;25:985–90. doi: 10.1111/j.1440-1746.2009.06155.x. [DOI] [PubMed] [Google Scholar]
- 32.Gallagher PG, Chang SH, Rettig MP, Neely JE, Hillery CA, Smith BD, et al. Altered erythrocyte endothelial adherence and membrane phospholipid asymmetry in hereditary hydrocytosis. Blood. 2003;101:4625–7. doi: 10.1182/blood-2001-12-0329. [DOI] [PubMed] [Google Scholar]
- 33.Podsiedlik M, Markowicz-Piasecka M, Sikora J. Erythrocytes as model cells for biocompatibility assessment, cytotoxicity screening of xenobiotics and drug delivery. Chem Biol Interact. 2020;332:109305. doi: 10.1016/j.cbi.2020.109305. [DOI] [PubMed] [Google Scholar]
- 34.Villa CH, Cines DB, Siegel DL, Muzykantov V. Erythrocytes as carriers for drug delivery in blood transfusion and beyond. Transfus Med Rev. 2017;31:26–35. doi: 10.1016/j.tmrv.2016.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gao XL, Yue TT, Tian FL, Liu ZP, Zhang XR. Erythrocyte membrane skeleton inhibits nanoparticle endocytosis. AIP Adv. 2017;7:65303. [Google Scholar]
- 36.Zhao ZM, Ukidve A, Gao YS, Kim J, Mitragotri S. Erythrocyte leveraged chemotherapy (ELeCt): nanoparticle assembly on erythrocyte surface to combat lung metastasis. Sci Adv. 2019;5:eaax9250. doi: 10.1126/sciadv.aax9250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pishesha N, Bilate AM, Wibowo MC, Huang NJ, Li ZY, Dhesycka R, et al. Engineered erythrocytes covalently linked to antigenic peptides can protect against autoimmune disease. Proc Natl Acad Sci USA. 2017;114:3157–62. doi: 10.1073/pnas.1701746114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Glassman PM, Villa CH, Ukidve A, Zhao ZM, Smith P, Mitragotri S, et al. Vascular drug delivery using carrier red blood cells: focus on RBC surface loading and pharmacokinetics. Pharmaceutics. 2020;12:440. doi: 10.3390/pharmaceutics12050440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Han X, Wang C, Liu Z. Red blood cells as smart delivery systems. Bioconjug Chem. 2018;29:852–60. doi: 10.1021/acs.bioconjchem.7b00758. [DOI] [PubMed] [Google Scholar]
- 40.Yan JJ, Yu JC, Wang C, Gu Z. Red blood cells for drug delivery. Small Methods. 2017;1:1700270. [Google Scholar]
- 41.Zaitsev S, Danielyan K, Murciano JC, Ganguly K, Krasik T, Taylor RP, et al. Human complement receptor type 1–directed loading of tissue plasminogen activator on circulating erythrocytes for prophylactic fibrinolysis. Blood. 2006;108:1895–902. doi: 10.1182/blood-2005-11-012336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zaitsev S, Spitzer D, Murciano JC, Ding BS, Tliba S, Kowalska MA, et al. Targeting of a mutant plasminogen activator to circulating red blood cells for prophylactic fibrinolysis. J Pharmacol Exp Ther. 2010;332:1022–31. doi: 10.1124/jpet.109.159194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Domenech C, Thomas X, Chabaud S, Baruchel A, Gueyffier F, Mazingue F, et al. l-asparaginase loaded red blood cells in refractory or relapsing acute lymphoblastic leukaemia in children and adults: results of the GRASPALL 2005-01 randomized trial. Br J Haematol. 2011;153:58–65. doi: 10.1111/j.1365-2141.2011.08588.x. [DOI] [PubMed] [Google Scholar]
- 44.Anselmo AC, Kumar S, Gupta V, Pearce AM, Ragusa A, Muzykantov V, et al. Exploiting shape, cellular-hitchhiking and antibodies to target nanoparticles to lung endothelium: Synergy between physical, chemical and biological approaches. Biomaterials. 2015;68:1–8. doi: 10.1016/j.biomaterials.2015.07.043. [DOI] [PubMed] [Google Scholar]
- 45.Brenner JS, Pan DC, Myerson JW, Marcos-Contreras OA, Villa CH, Patel P, et al. Red blood cell-hitchhiking boosts delivery of nanocarriers to chosen organs by orders of magnitude. Nat Commun. 2018;9:2684. doi: 10.1038/s41467-018-05079-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Red Blood Cells Harnessed as Nanoparticle Carriers for Vaccines. Genetic Engineering and Biotechnology News. 2020 July 14. https://www.genengnews.com/news/red-blood-cells-harnessed-as-nanoparticle-carriers-for-vaccines/
- 47.Ukidve A, Zhao ZM, Fehnel A, Krishnan V, Pan DIC, Gao YS, et al. Erythrocyte-driven immunization via biomimicry of their natural antigen-presenting function. Proc Natl Acad Sci USA. 2020;117:17727–36. doi: 10.1073/pnas.2002880117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Anselmo AC, Gupta V, Zern BJ, Pan D, Zakrewsky M, Muzykantov V, et al. Delivering nanoparticles to lungs while avoiding liver and spleen through adsorption on red blood cells. ACS Nano. 2013;7:11129–37. doi: 10.1021/nn404853z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zelepukin IV, Yaremenko AV, Shipunova VO, Babenyshev AV, Balalaeva IV, Nikitin PI, et al. Nanoparticle-based drug delivery via RBC-hitchhiking for the inhibition of lung metastases growth. Nanoscale. 2019;11:1636–46. doi: 10.1039/c8nr07730d. [DOI] [PubMed] [Google Scholar]
- 50.Scott MD, Murad KL, Koumpouras F, Talbot M, Eaton JW, et al. Chemical camouflage of antigenic determinants: Stealth erythrocytes. Proc Natl Acad Sci USA. 1997;14:7566–71. doi: 10.1073/pnas.94.14.7566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Park J, Andrade B, Seo Y, Kim MJ, Zimmerman SC, Kong H. Engineering the surface of therapeutic “living”cells. Chem Rev. 2018;118:1664–90. doi: 10.1021/acs.chemrev.7b00157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Pan DC, Myerson JW, Brenner JS, Patel PN, Anselmo AC, Mitragotri S, et al. Nanoparticle properties modulate their attachment and effect on carrier red blood cells. Sci Rep. 2018;8:1615. doi: 10.1038/s41598-018-19897-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Setyawati MI, Tay CY, Docter D, Stauber RH, Leong DT. Understanding and exploiting nanoparticles’ intimacy with the blood vessel and blood. Chem Soc Rev. 2015;44:8174–99. doi: 10.1039/c5cs00499c. [DOI] [PubMed] [Google Scholar]
- 54.Barshtein G, Livshits L, Shvartsman LD, Shlomai NO, Arbell D, Yedgar S. Polystyrene nanoparticles activate erythrocyte aggregation and adhesion to endothelial cells. Cell Biochem Biophys. 2016;74:19–27. doi: 10.1007/s12013-015-0705-6. [DOI] [PubMed] [Google Scholar]
- 55.Charoenphol P, Onyskiw PJ, Carrasco-Teja M, Eniola-Adefeso O. Particle-cell dynamics in human blood flow: Implications for vascular-targeted drug delivery. J Biomech. 2012;45:2822–8. doi: 10.1016/j.jbiomech.2012.08.035. [DOI] [PubMed] [Google Scholar]
- 56.Down LA, Papavassiliou DV, O’Rear EA. Significance of extensional stresses to red blood cell lysis in a shearing flow. Ann Biomed Eng. 2011;39:1632–42. doi: 10.1007/s10439-011-0262-0. [DOI] [PubMed] [Google Scholar]
- 57.Chinol M, Casalini P, Maggiolo M, Canevari S, Omodeo ES, Caliceti P, et al. Biochemical modifications of avidin improve pharmacokinetics and biodistribution, and reduce immunogenicity. Br J Cancer. 1998;78:189–97. doi: 10.1038/bjc.1998.463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Stephan MT, Irvine DJ. Enhancing cell therapies from the outside in: Cell surface engineering using synthetic nanomaterials. Nano Today. 2011;6:309–25. doi: 10.1016/j.nantod.2011.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Anselmo AC, Mitragotri S. Cell-mediated delivery of nanoparticles: Taking advantage of circulatory cells to target nanoparticles. J Control Release. 2014;190:531–41. doi: 10.1016/j.jconrel.2014.03.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Anselmo AC, Kumar S, Gupta V, Pearce AM, Ragusa A, Muzykantov V, et al. Exploiting shape, cellular-hitchhiking and antibodies to target nanoparticles to lung endothelium: synergy between physical, chemical and biological approaches. Biomaterials. 2015;68:1–8. doi: 10.1016/j.biomaterials.2015.07.043. [DOI] [PubMed] [Google Scholar]
- 61.He CB, Hu YP, Yin LC, Tang C, Yin CH. Effects of particle size and surface charge on cellular uptake and biodistribution of polymeric nanoparticles. Biomaterials. 2010;31:3657–66. doi: 10.1016/j.biomaterials.2010.01.065. [DOI] [PubMed] [Google Scholar]
- 62.Foged C, Brodin B, Frokjaer S, Sundblad A. Particle size and surface charge affect particle uptake by human dendritic cells in an in vitro model. Int J Pharm. 2005;298:315–22. doi: 10.1016/j.ijpharm.2005.03.035. [DOI] [PubMed] [Google Scholar]
- 63.Kulkarni SA, Feng SS. Effects of particle size and surface modification on cellular uptake and biodistribution of polymeric nanoparticles for drug delivery. Pharmacol Res. 2013;30:2512–22. doi: 10.1007/s11095-012-0958-3. [DOI] [PubMed] [Google Scholar]
- 64.Aggarwal P, Hall JB, McLeland CB, Dobrovolskaia MA, McNeil SE. Nanoparticle interaction with plasma proteins as it relates to particle biodistribution, biocompatibility and therapeutic efficacy. Adv Drug Deliv Rev. 2009;61:428–37. doi: 10.1016/j.addr.2009.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Xiao K, Li YP, Luo JT, Lee JS, Xiao WW, Gonik AM, et al. The effect of surface charge on in vivo biodistribution of PEG-oligocholic acid based micellar nanoparticles. Biomaterials. 2011;32:3435–46. doi: 10.1016/j.biomaterials.2011.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Benne N, van Duijn J, Kuiper J, Jiskoot W, Slutter B. Orchestrating immune responses: How size, shape and rigidity affect the immunogenicity of particulate vaccines. J Control Release. 2016;234:124–34. doi: 10.1016/j.jconrel.2016.05.033. [DOI] [PubMed] [Google Scholar]
- 67.Nowak M, Brown TD, Graham A, Helgeson ME, Mitragotri S. Size, shape, and flexibility influence nanoparticle transport across brain endothelium under flow. Bioeng Transl Med. 2020;5:e10153. doi: 10.1002/btm2.10153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kolhar P, Anselmo AC, Gupta V, Pant K, Prabhakarpandian B, Ruoslahti E, et al. Using shape effects to target antibody-coated nanoparticles to lung and brain endothelium. Proc Natl Acad Sci USA. 2013;110:10753–8. doi: 10.1073/pnas.1308345110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Doshi N, Prabhakarpandian B, Rea-Ramsey A, Pant K, Sundaram S, Mitragotri S. Flow and adhesion of drug carriers in blood vessels depend on their shape: a study using model synthetic microvascular networks. Release. J Control Release. 2010;146:196–200. doi: 10.1016/j.jconrel.2010.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lee S, Ferrari M, Decuzzi P. Shaping nano-/micro-particles for enhanced vascular interaction in laminar flows. Nanotechnology. 2009;20:495101. doi: 10.1088/0957-4484/20/49/495101. [DOI] [PubMed] [Google Scholar]
- 71.Shuvaev VV, Ilies MA, Simone E, Zaitsev S, Kim Y, Cai SS, et al. Endothelial targeting of antibody-decorated polymeric filomicelles. ACS Nano. 2011;5:6991–9. doi: 10.1021/nn2015453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Geng Y, Dalhaimer P, Cai SS, Tsai R, Tewari M, Minko T, et al. Shape effects of filaments versus spherical particles in flow and drug delivery. Nat Nanotechnol. 2007;2:249–55. doi: 10.1038/nnano.2007.70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Yoo J, Mitragotri S. Polymer particles that switch shape in response to a stimulus. Proc Natl Acad Sci USA. 2010;107:11205–10. doi: 10.1073/pnas.1000346107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Champion JA, Mitragotri S. Shape induced inhibition of phagocytosis of polymer particles. Pharmacol Res. 2009;26:244–9. doi: 10.1007/s11095-008-9626-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Gutierrez M, Ojeda LS, Eniola-Adefeso O. Vascular-targeted particle binding efficacy in the presence of rigid red blood cells: implications for performance in diseased blood. Biomicrofluidics. 2018;12:42217. doi: 10.1063/1.5027760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Anselmo AC, Zhang M, Kumar S, Vogus DR, Menegatti S, Helgeson ME, et al. Elasticity of nanoparticles influences their blood circulation, phagocytosis, endocytosis, and targeting. ACS Nano. 2015;9:3169–77. doi: 10.1021/acsnano.5b00147. [DOI] [PubMed] [Google Scholar]
- 77.Hui Y, Yi X, Wibowo D, Yang GZ, Middelberg APJ, Gao HJ, et al. Nanoparticle elasticity regulates phagocytosis and cancer cell uptake. Sci Adv. 2020;6:eaaz4316. doi: 10.1126/sciadv.aaz4316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Guo P, Liu D, Subramanyam K, Wang BR, Yang J, Huang J, et al. Nanoparticle elasticity directs tumor uptake. Nat Commun. 2018;9:130. doi: 10.1038/s41467-017-02588-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Champion JA, Mitragotri S. Role of target geometry in phagocytosis. Proc Natl Acad Sci USA. 2006;103:4930–4. doi: 10.1073/pnas.0600997103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Anselmo AC, Mitragotri S. Impact of particle elasticity on particle-based drug delivery systems. Adv Drug Deliv Rev. 2017;108:51–67. doi: 10.1016/j.addr.2016.01.007. [DOI] [PubMed] [Google Scholar]
- 81.Herant M. Mechanics of neutrophil phagocytosis: behavior of the cortical tension. J Cell Sci. 2005;118:1789–97. doi: 10.1242/jcs.02275. [DOI] [PubMed] [Google Scholar]
- 82.Garapaty A, Champion JA. Tunable particles alter macrophage uptake based on combinatorial effects of physical properties. Bioeng Transl Med. 2017;2:92–101. doi: 10.1002/btm2.10047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Carboni E, Tschudi K, Nam J, Lu XL, Ma AWK. Particle margination and its implications on intravenous anticancer drug delivery. AAPS PharmSciTech. 2014;15:762–71. doi: 10.1208/s12249-014-0099-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Gupta N, Kozlovskaya V, Dolmat M, Kharlampieva E. Shape recovery of spherical hydrogen-bonded multilayer capsules after osmotically induced deformation. Langmuir. 2019;35:10910–9. doi: 10.1021/acs.langmuir.9b01795. [DOI] [PubMed] [Google Scholar]
- 85.Zhao YN, Sun XX, Zhang GN, Trewyn BG, Slowing II, Lin VSY. Interaction of mesoporous silica nanoparticles with human red blood cell membranes: size and surface effects. ACS Nano. 2011;5:1366–75. doi: 10.1021/nn103077k. [DOI] [PubMed] [Google Scholar]
- 86.Muzykantov VR, Taylor RP. Attachment of biotinylated antibody to red blood cellsantigen-binding capacity of immunoerythrocytes and their susceptibility to lysis by complement. Anal Biochem. 1994;1:142–8. doi: 10.1006/abio.1994.1559. [DOI] [PubMed] [Google Scholar]
- 87.Stephan MT, Moon JJ, Um SH, Bershteyn A, Irvine DJ. Therapeutic cell engineering with surface-conjugated synthetic nanoparticles. Nat Med. 2010;16:1035–41. doi: 10.1038/nm.2198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Arruebo M, Valladares M, González-Fernández Á. Antibody-conjugated nanoparticles for biomedical applications. J Nanomater. 2009;2009:439389. [Google Scholar]
- 89.Zhou H, Fan Z, Li PY, Deng JJ, Arhontoulis DC, Li CY, et al. Dense and dynamic polyethylene glycol shells cloak nanoparticles from uptake by liver endothelial cells for long blood circulation. ACS Nano. 2018;12:10130–41. doi: 10.1021/acsnano.8b04947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Ishida T, Maeda R, Ichihara M, Irimura K, Kiwada H. Accelerated clearance of PEGylated liposomes in rats after repeated injections. J Control Release. 2003;88:35–42. doi: 10.1016/s0168-3659(02)00462-5. [DOI] [PubMed] [Google Scholar]
- 91.Hall SS, Mitragotri S, Daugherty PS. Identification of peptide ligands facilitating nanoparticle attachment to erythrocytes. Biotechnol Prog. 2007;23:749–54. doi: 10.1021/bp060333l. [DOI] [PubMed] [Google Scholar]
- 92.Lindorfe MA, Hahn CS, Foley PL, Taylor RP. Heteropolymer-mediated clearance of immune complexes via erythrocyte CR1 mechanisms and applications. Immunol Rev. 2001;183:10–24. doi: 10.1034/j.1600-065x.2001.1830102.x. [DOI] [PubMed] [Google Scholar]
- 93.Zaitsev S, Spitzer D, Murciano J, Ding BS, Tliba S, Kowalska MA, et al. Sustained thromboprophylaxis mediated by an RBC-targeted pro-urokinase zymogen activated at the site of clot formation. Blood. 2010;115:5241–8. doi: 10.1182/blood-2010-01-261610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Potempa M, Potempa J. Protease-dependent mechanisms of complement evasion by bacterial pathogens. Biol Chem. 2012;393:873–88. doi: 10.1515/hsz-2012-0174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Fang RH, Hu CJ, Chen KNH, Luk BT, Carpenter CW, Gao WW, et al. Lipid-insertion enables targeting functionalization of erythrocyte membrane-cloaked nanoparticles. Nanoscale. 2013;5:8884–8. doi: 10.1039/c3nr03064d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Shi G, Mukthavaram R, Kesari S, Simberg D. Distearoyl anchor-painted erythrocytes with prolonged ligand retention and circulation properties in vivo. Adv Healthc Mater. 2014;3:142–8. doi: 10.1002/adhm.201300084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Mukthavaram R, Shi G, Kesari S, Simberg D. Targeting and depletion of circulating leukocytes and cancer cells by lipophilic antibody-modified erythrocytes. J Control Release. 2014;183:146–53. doi: 10.1016/j.jconrel.2014.03.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Reist CJ, Combs MJ, Croft BY, Taylor RP. Antigens pre-bound to the primate erythrocyte complement receptor via cross-linked bispecific monoclonal antibody heteropolymers are rapidly cleared from the circulation. Eur J Immunol. 1993;23:3021–7. doi: 10.1002/eji.1830231144. [DOI] [PubMed] [Google Scholar]
- 99.Zhu DM, Wu L, Suo M, Gao S, Xie W, Zan MH, et al. Engineered red blood cells for capturing circulating tumor cells with high performance. Nanoscale. 2018;10:6014–23. doi: 10.1039/c7nr08032h. [DOI] [PubMed] [Google Scholar]
- 100.Tzounakas VL, Karadimas DG, Papassideri IS, Seghatchian J, Antonelou MH. Erythrocyte-based drug delivery in Transfusion Medicine: Wandering questions seeking answers. Transfus Apher Sci. 2017;56:626–34. doi: 10.1016/j.transci.2017.07.015. [DOI] [PubMed] [Google Scholar]
- 101.Banz A, Cremel M, Mouvant A, Guerin N, Horand F, Godfrin Y. Tumor growth control using red blood cells as the antigen delivery system and poly(I:C) J Immunother. 2012;35:409–17. doi: 10.1097/CJI.0b013e3182594352. [DOI] [PubMed] [Google Scholar]
- 102.Gao WW, Zhang LF. Engineering red‐blood‐cell‐membrane–coated nanoparticles for broad biomedical applications. AIChE J. 2015;61:738–46. [Google Scholar]
- 103.Murad KT, Mahany KL, Brugnara C, Kuypers FA, Eaton JW, Scott ML. Structural and functional consequences of antigenic modulation of red blood cells. Blood. 1999;93:2121–7. [PubMed] [Google Scholar]
- 104.Tan YX, Qiu Y, Xu H, Ji SP, Li SB, Gong F, et al. Decreased immunorejection in unmatched blood transfusions by attachment of methoxypolyethylene glycol on human red blood cells and the effect on D antigen. Transfusion. 2006;46:2122–7. doi: 10.1111/j.1537-2995.2006.01038.x. [DOI] [PubMed] [Google Scholar]
- 105.Nacharaju P, Boctor FN, Manjula BN, Acharya SA. Surface decoration of red blood cells with maleimidophenyl-polyethylene glycol facilitated by thiolation with iminothiolane: an approach to mask A, B, and D antigens to generate universal red blood cells. Transfusion. 2005;45:374–83. doi: 10.1111/j.1537-2995.2005.04290.x. [DOI] [PubMed] [Google Scholar]
- 106.Sarvi F, Najafabadi SH, Farahani EV, Shojaosadati SA. Surface coating of red blood cells with monomethoxy poly(ethylene glycol) activated with two different reagents. Iran J Chem Chem Eng-Int Engl Ed. 2008;27:1–9. [Google Scholar]
- 107.Bourgeaux V, Lanao JM, Bax BE, Godfrin YL. Drug-loaded erythrocytes: on the road toward marketing approval. Drug Des Devel Ther. 2016;10:665–76. doi: 10.2147/DDDT.S96470. [DOI] [PMC free article] [PubMed] [Google Scholar]