Abstract
In March 2019, the global COVID-19 pandemic caused by the novel SARS-CoV-2 coronavirus began. The first cases of SARS-CoV-2 infection occurred in November 19 in Wuhan, China. Preventive measures taken have not prevented the rapid spread of the virus to countries around the world. To date, there are approximately 3 million deaths, and a massive worldwide vaccination campaign has recently begun. SARS-CoV-2 uses the ACE-2 protein as an intracellular carrier. ACE-2 is a key component of the renin-angiotensin system (RAS), a key regulator of cardiovascular function. Considering the key role of ACE-2 in COVID-19 infection, both as an entry receptor and as a protective role, especially for the respiratory tract, and considering the variations of ACE-2 during the phases of viral infection, it is clear the important role that pharmacological regulation of RAS and ACE-2 may take. In this article, we describe the importance of ACE-2 in COVID-19 infection, the pharmacological aspects of a modulation with RAS-modifying agents, new therapeutic strategies, trying to provide a deep understanding and explanation of the complex mechanisms underlying the relationship between the virus and ACE-2, providing opinions and personal hypotheses on the best strategies of therapeutic intervention.
Keywords: RAS, ACE-2, COVID-19, SARS-CoV-2
Introduction
The global pandemic COVID-19
The 2019 global coronavirus pandemic (COVID-19) is caused by a new coronavirus, SARS-CoV-2. To date, COVID-19 has caused approximately 3 Mln deaths (World Health Organization 2021). The COVID-19 pandemic represents a health, social, and economic challenge for all countries. A massive vaccination campaign has recently begun worldwide (Vitiello et al. 2021e). To date, drug treatments are mostly experimental; some antivirals such as remdesivir have shown good efficacy in reducing mortality and healing time (Ferrara et al. 2020b; Vitiello and Ferrara 2020a; Vitiello et al. 2021a). COVID-19 infection in most cases is asymptomatic or slightly symptomatic; however in a small percentage of cases, especially in elderly people and those with pre-existing conditions, the infection can be severe and in some cases fatal (Ruan et al. 2020; Yang et al. 2020). Although respiratory symptoms are predominant (Ferrara et al. 2020a), in the most severe stages of infection, multi-organ dysfunction can occur (Vitiello et al. 2021c; Ferrara and Vitiello 2021a) due to an abnormal and generalized inflammatory response (Vitiello et al. 2021b; Vitiello and Ferrara 2021a, b), causing injury to vital organs such as the lungs, heart, liver, and CNS (Shi et al. 2020; Vitiello et al. 2020a). Most patients with COVID-19 have a good prognosis, and rapid healing times; however, patients with diseases, such as diabetes, hypertension, and heart disease, are at greater risk of serious complications; this suggests that during the COVID-19, infection treatment of the underlying diseases should not be interrupted (Ferrara 2020; Guo et al. 2020). SARS-CoV-2 has high structural homology with SARS-CoV, demonstrating that it also shares the same cell entry receptor, the angiotensin 2 conversion enzyme (ACE-2) (Walls et al. 2020). ACE-2 is a protein with a key role in the renin-angiotensin system (RAS). RAS is a key regulator of the cardiovascular system.
RAS and COVID-19
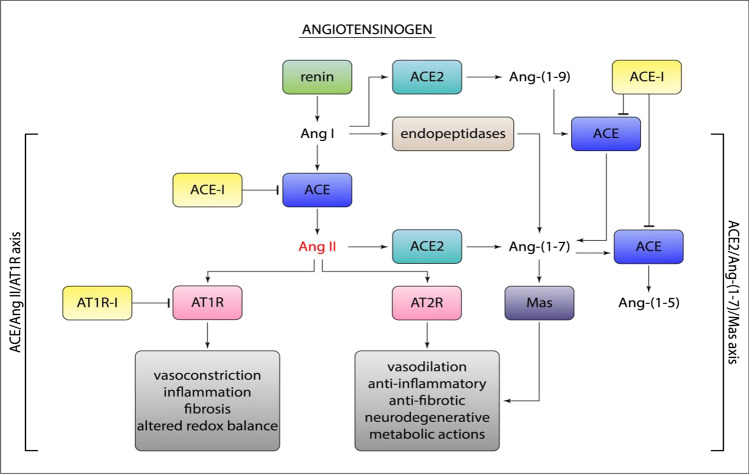
The renin-angiotensin system (RAS) is a physiological mechanism with a key regulatory role in various functions of the cardiovascular system. RAS is an enzymatic cascade consisting of a “classical way” and a “non-classical way”. In the main (classical) enzymatic pathway, renin converts angiotensinogen to angiotensin I (Ang I). Ang I is then converted to angiotensin II (Ang II) by the angiotensin conversion enzyme (ACE). In the non-classical enzyme pathway, ACE-2 converts Ang II to angiotensin 1–7 (Ang 1–7) and Ang I to angiotensin 1–9 (Ang 1–9). ACE-2 is expressed in renal, cardiovascular, pulmonary, and gastrointestinal tissues. The biological effects of Ang-II on AT-1r receptors are vasoconstriction and stimulant of aldosterone release, myocardial hypertrophy, interstitial fibrosis, endothelial dysfunction, increased inflammatory state, increased oxidative stress, and increased coagulation (Fig. 1).
Fig. 1.
The renin-angiotensin system (RAS) and the biological effects after activation of AT-1r, AT-2r, and MASr
Ang II can also cause increased inflammation through the production of IL-6, TNF (tumor necrosis factor), and other inflammatory cytokines (Yamamoto et al. 2011; Recinos et al. 2007). Ang 1–7 and Ang 1–9 have opposite biological effects to Ang II through stimulation of the Mas receptor (MASr) and angiotensin type II receptors (AT-2r). MASr are expressed on the surface of the bronchial muscle and alveolar epithelium of the lungs (Pai et al. 2017; Magalhães et al. 2015). Ang 1–7 and Ang1-9 have anti-inflammatory, anti-fibrous, vasodilation effects. Evidence shows that the RAS system varies in conditions of pathologies such as COPD or viral infections, suggesting a possible involvement in homeostasis and function of the respiratory system and other ongoing systems of certain diseases. Some data demonstrate the variation of RAS and ACE-2 during COVID-19 infection, in particular an increase of ACE-2 in the early stages of infection and a rapid decrease in the more severe stages. It may be possible that ACE-2 has a protective effect, and when it decreases, there is a worsening of the inflammatory pulmonary state (Vitiello et al. 2020b; Vitiello and Ferrara 2021c).
ACE-2
ACE-2 is expressed both on the cell surface (in bound form) and in plasma and urine (soluble form) (Warner et al. 2005) Evidence shows that membrane ACE-2, during SARS-CoV infection, is internalized into the cytoplasm at the time of virus binding. ACE-2 is expressed in almost all organs (Donoghue et al. 2000), including the oral and nasal mucosa, nasopharynx, lung, liver, kidney, and brain. Some data show that the greatest expression of ACE-2 is in the lungs, heart, and liver, suggesting a possible correlation between the tissue distribution of ACE-2, and the organs may be more vulnerable in the more severe stages of COVID-19 infection. Some proteases, in particular TMPRSS2, are also involved in viral infection. TMPRSS2 may promote membrane fusion between the virus and the host cell as a result of ACE2 involvement and activation of S-protein SARS-CoV-2 (Hoffmann et al. 2020). Evidence shows that ACE-2 decreases occur in severe COVID-19 patients. This variation could probably be one of the causes of the damage caused by COVID-19. As described above, the decrease in ACE-2 expression leads to a decrease in the ACE2-Ang (1–7)-MasR axis and an increase in Ang-II. The imbalance between Ang 1–7 and Ang II leads to the prevalence of the biological effects of Ang II such as profibrotics, proinflammatory vasoconstriction, and indirect procoagulation (Vitiello and Ferrara 2020b). Biological effects may be responsible for pulmonary and cardiac lesions that may occur in severe COVID-19 patients (Vitiello et al. 2020c).
ACE inhibitor(ACE-i) and angiotensin-receptor blocker (ARB)
Recent studies have shown that the use of ACE-i or ARB can affect the expression of ACE-2 (Li et al. 2020a). Since the beginning of the COVID-19 pandemic, a scientific debate is ongoing on whether the use of ACE-i and ARB may represent a COVID-19 risk factor. To date, epidemiological evidence shows that they do not represent a risk factor, and it is not recommended to stop the therapeutic treatment (Reynolds et al. 2020). Moreover, according to the different mechanism of action, it is assumed that the use of ACE-i increases the expression of ACE-2, and the use of ARB causes a counter-regulation of the RAS system with an increase of ACE and ACE-2. Currently, it is not completely clear whether ACE-I and ARB increase ACE-2 expression in all tissues, whether under certain conditions and at what times and doses. For example, perindopril (ACE-i) has been able to increase ACE-2 expression in the liver under conditions of liver fibrosis (Huang et al. 2010). Ramipril (ACE-i) decreased the expression of the ACE-2 protein after myocardial infarction (Burchill et al. 2012). Losartan (ARB) may not only increase cardiac mRNA ACE2 expression but also significantly increase ACE-2 activity in the lung, suggesting that losartan may have protective effects on cardiac and lung lesions caused by COVID-19. In addition, in vivo experiments have shown that lozartan may protect against lung injury caused by SARS-CoV coronavirus by decreasing the production of pro-inflammatory cytokines (Ferrario et al. 2005; Kuba et al. 2005).
Pharmacological target of SARS-CoV-2
A causa della gravità della pandemia, sono urgentemente necessarie terapie efficaci contro la SARS-CoV-2. Come già descritto, l'ACE-2 è il recettore di ingresso cellulare della SARS-CoV-2, ed è anche un componente chiave del RAS. Pertanto, una piena comprensione della correlazione tra ACE-2 e SARS-CoV-2 sarebbe di fondamentale importanza per agire con strategie di intervento farmacologico mirate (Ferrara and Vitiello 2020; Vitiello and Ferrara 2021d; Ferrara and Vitiello 2021b; Muslim et al. 2020). Tuttavia, sebbene l'ACE-2 sia stato identificato come recettore della SARS-CoV-2, potrebbero esserci altri co-recettori della SARS-CoV-2 ancora da scoprire (Muslim et al. 2020). Ciò solleva ulteriori importanti implicazioni per i bersagli terapeutici della SARS-CoV-2 (Marovich et al. 2020; Vitiello et al. 2021d; Singh et al. 2021). One of the most important strategies to control viral infections is to block the initial binding of the virus to its functional receptors. Several candidate drugs have been developed to block binding of S protein and ACE-2, including drugs based on S protein and ACE-2. Monoclonal antibodies to COVID-19 act by neutralizing the spike protein of SARS-CoV-2, which blocks the binding of the spike protein to human ACE-2 receptors, thereby preventing subsequent viral entry into human cells and virus replication (Marovich et al. 2020; Tian et al. 2020). Treatment with a soluble recombinant human form of ACE2 (rhACE2) could prove useful as a trap effect for circulating SARS-CoV2 and decrease viral load and hinder infection (Monteil et al. 2020). Administration of recombinant soluble human ACE2 has shown good efficacy in subjects with acute respiratory distress syndrome (ARDS) (Muslim et al. 2020). From a molecular pharmacological point of view, administration of rhACE2 activates the Ang 1–7 and Ang 1–9 synthesis pathway of the RAS system (non-classical pathway) by decreasing Ang II levels with a tendency to lower the concentration of proinflammatory cytokines (Gaddam et al. 2014). Some clinical trials show excellent results when administered in combination rhACE2 and remdesivir (Monteil et al. 2021). Theoretically, administration of soluble ACE2 protein, in sufficient quantities, binding to the spike protein of SARS-CoV-2, could reduce the attachment to ACE-2 at the plasma membrane. This could be used therapeutically as a way to reduce infectivity in patients treated with COVID-19. Studies in healthy volunteers have demonstrated a reduction in Ang II after administration of soluble ACE-2 (Imai et al. 2005). In contrast, direct administration of Ang (1–7) failed to demonstrate significant effects in humans (Oudit and Penninger 2011). In addition, preclinical studies have shown that administration of soluble ACE-2 can decrease lung injury (Haschke et al. 2013). In patients with acute respiratory distress syndrome (ARDS), hrACE2 (GSK2586881) was well-tolerated (Khan et al. 2017). Human rACE2 caused a decrease in circulating Ang II levels (73), whereas angiotensin (1–5) and angiotensin (1–7) levels increased and remained elevated for 48 h (Khan et al. 2017). Some in vitro and in silico assays revealed two compounds (xanthenone and resorcinolnaphthalein) that increase ACE2 activity in a dose-dependent manner (Li et al. 2020b). They may be useful in increasing Ang II turnover in Ang (1–7). Finally, vitamin D is known for its anti-inflammatory effects. The hormonal form of vitamin D, calcitriol (1,25(OH)2D3), is a negative endocrine regulator of the RAS and inhibits renin biosynthesis, and it blocks the expression of ACE and Ang II and elevates ACE2 levels in LPS-induced acute lung injury (Xu et al. 2015). Thus, it could potentially play a role in combating SARS-CoV-2 infection and related complications.
Conclusions
The COVID-19 pandemic has caused about 1.34 million deaths and is currently ongoing. Due to this severity, effective therapies against SARS-CoV-2 are urgently needed. RAS and ACE-2 have an important correlation with SARS-CoV-2. A modulation of the system could represent a protective effect against SARS-CoV-2 infection. Interesting drug strategies are being developed. The blocking of SARS-CoV-2 cell entry is potentially the best pharmacological strategy to follow. Well-structured clinical trials are urgent in this direction to generate the necessary EBM.
Author contribution
The authors certify that the manuscript is original, never submitted to other journal for publication before. All authors contributed equally to the manuscript and had the opportunity to revise and approve the final text.
Declarations
Conflict of interest
The authors declare no completing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
11/5/2021
A Correction to this paper has been published: 10.1007/s00210-021-02165-4
Contributor Information
Francesco Ferrara, Email: francesco.ferrara@uslumbria1.it.
Antonio Vitiello, Email: antonio.vitiello2@uslumbria1.it.
References
- Burchill LJ, Velkoska E, Dean RG, Griggs K, Patel SK, Burrell LM. Combination renin-angiotensin system blockade and angiotensin-converting enzyme 2 in experimental myocardial infarction: implications for future therapeutic directions. Clin Sci (Lond) 2012;123(11):649–658. doi: 10.1042/CS20120162. [DOI] [PubMed] [Google Scholar]
- Donoghue M, Hsieh F, Baronas E, Godbout K, Gosselin M, Stagliano N, Donovan M, Woolf B, Robison K, Jeyaseelan R, Breitbart RE, Acton S. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1–9. Circ Res. 2000;87(5):E1–9. doi: 10.1161/01.res.87.5.e1. [DOI] [PubMed] [Google Scholar]
- Ferrara F. Antirheumatic in SARS-cov-2: benefit or risk? Italian J Med. 2020;14(2):114–115. doi: 10.4081/itjm.2020.1290. [DOI] [Google Scholar]
- Ferrara F, Vitiello A (2020) Potential pharmacological approach in the regulation of ACE-2 and DPP-IV in diabetic COVID-19 patient. Italian J Med (AOP). 10.4081/itjm.2020.1435
- Ferrara F, Granata G, Pelliccia C, La Porta R, Vitiello A. The added value of pirfenidone to fight inflammation and fibrotic state induced by SARS-CoV-2: anti-inflammatory and anti-fibrotic therapy could solve the lung complications of the infection? Eur J Clin Pharmacol. 2020;76(11):1615–1618. doi: 10.1007/s00228-020-02947-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrara F, Porta R, D’Aiuto V, Vitiello A (2020b) Remdesivir and COVID-19. Ir J Med Sci 17:1–2. 10.1007/s11845-020-02401-5 [DOI] [PMC free article] [PubMed]
- Ferrara F, Vitiello A. Efficacy of synthetic glucocorticoids in COVID-19 endothelites Naunyn Schmiedebergs. Arch Pharmacol. 2021;394(5):1003–1007. doi: 10.1007/s00210-021-02049-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrara F, Vitiello A. Scientific hypothesis for treatment of COVID-19’s lung lesions by adjusting ACE/ACE2 imbalance. Cardiovasc Toxicol. 2021;9:1–6. doi: 10.1007/s12012-021-09649-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrario CM, Jessup J, Chappell MC, Averill DB, Brosnihan KB, Tallant EA, Diz DI, Gallagher PE. Effect of angiotensin-converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2. Circulation. 2005;111(20):2605–2610. doi: 10.1161/CIRCULATIONAHA.104.510461. [DOI] [PubMed] [Google Scholar]
- Gaddam RR, Chambers S, Bhatia M. ACE and ACE2 in inflammation: a tale of two enzymes. Inflamm Allergy Drug Targets. 2014;13(4):224–234. doi: 10.2174/1871528113666140713164506. [DOI] [PubMed] [Google Scholar]
- Guo T, Fan Y, Chen M, Wu X, Zhang L, He T, Wang H, Wan J, Wang X, Lu Z. Cardiovascular implications of fatal outcomes of patients with coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5(7):811–818. doi: 10.1001/jamacardio.2020.1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haschke M, Schuster M, Poglitsch M. Pharmacokinetics and pharmacodynamics of recombinant human angiotensin- converting enzyme 2 in healthy human subjects. Clin Pharmacokinet. 2013;52(9):783–792. doi: 10.1007/s40262-013-0072-7. [DOI] [PubMed] [Google Scholar]
- Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche A, Müller MA, Drosten C, Pöhlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271–280.e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang ML, Li X, Meng Y, Xiao B, Ma Q, Ying SS, Wu PS, Zhang ZS. Upregulation of angiotensin-converting enzyme (ACE) 2 in hepatic fibrosis by ACE inhibitors. Clin Exp Pharmacol Physiol. 2010;37(1):e1–6. doi: 10.1111/j.1440-1681.2009.05302.x. [DOI] [PubMed] [Google Scholar]
- Imai Y, Kuba K, Rao S. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436(7047):112–116. doi: 10.1038/nature03712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan A, Benthin C, Zeno B, et al. A pilot clinical trial of recombinant human angiotensin-converting enzyme 2 in acute respiratory distress syndrome. Crit Care. 2017;21(1):234. doi: 10.1186/s13054-017-1823-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuba K, Imai Y, Rao S, Gao H, Guo F, Guan B, Huan Y, Yang P, Zhang Y, Deng W, Bao L, Zhang B, Liu G, Wang Z, Chappell M, Liu Y, Zheng D, Leibbrandt A, Wada T, Slutsky AS, Liu D, Qin C, Jiang C, Penninger JM. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med. 2005;11(8):875–879. doi: 10.1038/nm1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li G, He X, Zhang L, Ran Q, Wang J, Xiong A, Wu D, Chen F, Sun J, Chang C. Assessing ACE2 expression patterns in lung tissues in the pathogenesis of COVID-19. J Autoimmun. 2020;112:102463. doi: 10.1016/j.jaut.2020.102463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y, Zhou W, Yang L, You R. Physiological and pathological regulation of ACE2, the SARS-CoV-2 receptor. Pharmacol Res. 2020;157:104833. doi: 10.1016/j.phrs.2020.104833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magalhães GS, Rodrigues-Machado MG, Motta-Santos D, Silva AR, Caliari MV, Prata LO, Abreu SC, Rocco PR, Barcelos LS, Santos RA, Campagnole-Santos MJ. Angiotensin-(1–7) attenuates airway remodelling and hyperresponsiveness in a model of chronic allergic lung inflammation. Br J Pharmacol. 2015;172(9):2330–2342. doi: 10.1111/bph.13057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marovich M, Mascola JR, Cohen MS. Monoclonal antibodies for prevention and treatment of COVID-19. JAMA. 2020;324(2):131–132. doi: 10.1001/jama.2020.10245. [DOI] [PubMed] [Google Scholar]
- Monteil V, Kwon H, Prado P, Hagelkrüys A, Wimmer RA, Stahl M, Leopoldi A, Garreta E, Hurtado Del Pozo C, Prosper F, Romero JP, Wirnsberger G, Zhang H, Slutsky AS, Conder R, Montserrat N, Mirazimi A, Penninger JM. Inhibition of SARS-CoV-2 infections in engineered human tissues using clinical-grade soluble human ACE2. Cell. 2020;181(4):905–913.e7. doi: 10.1016/j.cell.2020.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monteil V, Dyczynski M, Lauschke VM, Kwon H, Wirnsberger G, Youhanna S, Zhang H, Slutsky AS, Hurtado Del Pozo C, Horn M, Montserrat N, Penninger JM, Mirazimi A. Human soluble ACE2 improves the effect of remdesivir in SARS-CoV-2 infection. EMBO Mol Med. 2021;13(1):e13426. doi: 10.15252/emmm.202013426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muslim S, Nasrin N, Alotaibi FO, Prasad G, Singh SK, Alam I, Mustafa G. Treatment options available for COVID-19 and an analysis on possible role of combination of rhACE2, angiotensin (1–7) and angiotensin (1–9) as effective therapeutic measure. SN Compr Clin Med. 2020;22:1–6. doi: 10.1007/s42399-020-00407-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oudit GY, Penninger JM. Recombinant human angiotensin- converting enzyme 2 as a new renin-angiotensin system peptidase for heart failure therapy. Curr Heart Fail Rep. 2011;8(3):176–183. doi: 10.1007/s11897-011-0063-7. [DOI] [PubMed] [Google Scholar]
- Pai WY, Lo WY, Hsu T, Peng CT, Wang HJ. Angiotensin-(1–7) inhibits thrombin-induced endothelial phenotypic changes and reactive oxygen species production via NADPH oxidase 5 downregulation. Front Physiol. 2017;8(8):994. doi: 10.3389/fphys.2017.00994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Recinos A, 3rd, LeJeune WS, Sun H, Lee CY, Tieu BC, Lu M, Hou T, Boldogh I, Tilton RG, Brasier AR. Angiotensin II induces IL-6 expression and the Jak-STAT3 pathway in aortic adventitia of LDL receptor-deficient mice. Atherosclerosis. 2007;194(1):125–133. doi: 10.1016/j.atherosclerosis.2006.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reynolds HR, Adhikari S, Pulgarin C, Troxel AB, Iturrate E, Johnson SB, Hausvater A, Newman JD, Berger JS, Bangalore S, Katz SD, Fishman GI, Kunichoff D, Chen Y, Ogedegbe G, Hochman JS. Renin-angiotensin-aldosterone system inhibitors and risk of Covid-19. N Engl J Med. 2020;382(25):2441–2448. doi: 10.1056/NEJMoa2008975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruan Q, Yang K, Wang W, Jiang L, Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan China. Intensive Care Med. 2020;46(5):846–848. doi: 10.1007/s00134-020-05991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi S, Qin M, Shen B, Cai Y, Liu T, Yang F, Gong W, Liu X, Liang J, Zhao Q, Huang H, Yang B, Huang C. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan China. JAMA Cardiol. 2020;5(7):802–810. doi: 10.1001/jamacardio.2020.0950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh SP, Pritam M, Pandey B, Yadav TP. Microstructure, pathophysiology, and potential therapeutics of COVID-19: a comprehensive review. J Med Virol. 2021;93(1):275–299. doi: 10.1002/jmv.26254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tian X, Li C, Huang A, Xia S, Lu S, Shi Z, Lu L, Jiang S, Yang Z, Wu Y, Ying T. Potent binding of 2019 novel coronavirus spike protein by a SARS coronavirus-specific human monoclonal antibody. Emerg Microbes Infect. 2020;9(1):382–385. doi: 10.1080/22221751.2020.1729069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitiello A, Ferrara F. Remdesivir versus ritonavir/lopinavir in COVID-19 patients. Ir J Med Sci. 2020;18:1–2. doi: 10.1007/s11845-020-02440-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitiello A, Ferrara F. Pharmacological agents to therapeutic treatment of cardiac injury caused by Covid-19. Life Sci. 2020;1(262):118510. doi: 10.1016/j.lfs.2020.118510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitiello A, Ferrara F, Pelliccia C, Granata G, La Porta R. Cytokine storm and colchicine potential role in fighting SARS-CoV-2 pneumonia. Italian J Med. 2020;14(2):88–94. doi: 10.4081/itjm.2020.1284. [DOI] [Google Scholar]
- Vitiello A, La Porta R, Ferrara F (2020b) Correlation between the use of statins and COVID-19: what do we know? BMJ Evid Based Med. 10.1136/bmjebm-2020-111589 [DOI] [PubMed]
- Vitiello A, La Porta R, Ferrara F (2020c) Sacubitril, valsartan and SARS-CoV-2. BMJ Evid Based Med. 10.1136/bmjebm-2020-111497 [DOI] [PubMed]
- Vitiello A, Ferrara F. Anti-fibrotic therapy for the treatment of pulmonary sequelae in patients healed by COVID-19. Lung India. 2021;38(Supplement):S129–S130. doi: 10.4103/lungindia.lungindia_803_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitiello A, Ferrara F. Colchicine and SARS-CoV-2: Management of the hyperinflammatory state. Respir Med. 2021;178:106322. doi: 10.1016/j.rmed.2021.106322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitiello A, Ferrara F. Therapeutic strategies for SARS-CoV-2 acting on ACE-2. Eur J Pharm Sci. 2021;1(156):105579. doi: 10.1016/j.ejps.2020.105579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitiello A, Ferrara F. Pharmacological agents modifying the renin angiotensin and natriuretic peptide systems in COVID-19 patients. Wien Klin Wochenschr. 2021 doi: 10.1007/s00508-021-01855-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitiello A, Ferrara F, Porta R. Remdesivir and COVID-19 infection, therapeutic benefits or unnecessary risks? Ir J Med Sci. 2021;12:1–2. doi: 10.1007/s11845-020-02482-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitiello A, La Porta R, D’Aiuto V, Ferrara F (2021b) Pharmacological approach for the reduction of inflammatory and prothrombotic hyperactive state in COVID-19 positive patients by acting on complement cascade. Hum Immunol 82(4):264–269. 10.1016/j.humimm.2021.01.007 [DOI] [PMC free article] [PubMed]
- Vitiello A, La Porta R, Ferrara F. Scientific hypothesis and rational pharmacological for the use of sacubitril/valsartan in cardiac damage caused by COVID-19. Med Hypotheses. 2021;147:110486. doi: 10.1016/j.mehy.2021.110486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitiello A, Pelliccia C, Ferrara F (2021d) Drugs acting on the renin-angiotensin system and SARS-CoV-2. Drug Discov Today S1359–6446(21):00037–00044. 10.1016/j.drudis.2021.01.010 [DOI] [PMC free article] [PubMed]
- Vitiello A, Porta R, Pianesi L, Ferrara F. COVID-19 pandemic: vaccine and new monoclonal antibodies, point of view. Ir J Med Sci. 2021 doi: 10.1007/s11845-021-02584-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walls AC, Park YJ, Tortorici MA, Wall A, McGuire AT, Veesler D. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020;181:894–904. doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warner FJ, Lew RA, Smith AI, Lambert DW, Hooper NM, Turner AJ. Angiotensin-converting enzyme 2 (ACE2), but not ACE, is preferentially localized to the apical surface of polarized kidney cells. J Biol Chem. 2005;280(47):39353–39362. doi: 10.1074/jbc.M508914200. [DOI] [PubMed] [Google Scholar]
- World Health Organization (WHO). https://www.who.int/emergencies/diseases/novel-coronavirus2019/situation-reports. Accessed Apr 2021
- Yamamoto S, Yancey PG, Zuo Y, Ma LJ, Kaseda R, Fogo AB, Ichikawa I, Linton MF, Fazio S, Kon V. Macrophage polarization by angiotensin II-type 1 receptor aggravates renal injury-acceleration of atherosclerosis. Arterioscler Thromb Vasc Biol. 2011;31(12):2856–2864. doi: 10.1161/ATVBAHA.111.237198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J, Zheng Y, Gou X, Pu K, Chen Z, Guo Q, Ji R, Wang H, Wang Y, Zhou Y. Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: a systematic review and meta-analysis. Int J Infect Dis. 2020;94:91–95. doi: 10.1016/j.ijid.2020.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu S, Chen YH, Tan ZX, Xie DD, Zhang C, Xia MZ. Vitamin D3 pretreatment alleviates renal oxidative stress in lipopolysaccharide-induced acute kidney injury. J Steroid Biochem Mol Biol. 2015;152:133–141. doi: 10.1016/j.jsbmb.2015.05.009. [DOI] [PubMed] [Google Scholar]