Key Points
Question
What is the association between receipt of the Pfizer-BioNTech BNT162b2 vaccine and the incidence of symptomatic and asymptomatic SARS-CoV-2 infection among health care workers?
Findings
In this retrospective cohort study conducted in Tel Aviv, Israel, that included 6710 health care workers who underwent periodic testing for SARS-CoV-2 infection, vaccination with the BNT162b2 vaccine was associated with an adjusted incidence rate ratio of 0.03 for symptomatic infection and 0.14 for asymptomatic infection more than 7 days after the second dose. Both incidence rate ratios were statistically significant.
Meaning
Receipt of the BNT162b2 vaccine was significantly associated with lower incidence of symptomatic and asymptomatic SARS-CoV-2 infection among health care workers.
Abstract
Importance
Randomized clinical trials have provided estimates of the effectiveness of the BNT162b2 vaccine against symptomatic SARS-CoV-2 infection, but its effect on asymptomatic infections remains unclear.
Objective
To estimate the association of vaccination with the Pfizer-BioNTech BNT162b2 vaccine with symptomatic and asymptomatic SARS-CoV-2 infections among health care workers.
Design, Setting, and Participants
This was a single-center, retrospective cohort study conducted at a tertiary medical center in Tel Aviv, Israel. Data were collected on symptomatic and asymptomatic SARS-CoV-2 infections confirmed via polymerase chain reaction (PCR) tests in health care workers undergoing regular screening with nasopharyngeal swabs between December 20, 2020, and February 25, 2021. Logistic regression was used to calculate incidence rate ratios (IRRs) comparing the incidence of infection between fully vaccinated and unvaccinated participants, controlling for demographics and the number of PCR tests performed.
Exposures
Vaccination with the BNT162b2 vaccine vs unvaccinated status was ascertained from the employee health database. Full vaccination was defined as more than 7 days after receipt of the second vaccine dose.
Main Outcomes and Measures
The primary outcome was the regression-adjusted IRR for symptomatic and asymptomatic SARS-CoV-2 infection of fully vaccinated vs unvaccinated health care workers. The secondary outcomes included IRRs for partially vaccinated health care workers (days 7-28 after first dose) and for those considered as late fully vaccinated (>21 days after second dose).
Results
A total of 6710 health care workers (mean [SD] age, 44.3 [12.5] years; 4465 [66.5%] women) were followed up for a median period of 63 days; 5953 health care workers (88.7%) received at least 1 dose of the BNT162b2 vaccine, 5517 (82.2%) received 2 doses, and 757 (11.3%) were not vaccinated. Vaccination was associated with older age compared with those who were not vaccinated (mean age, 44.8 vs 40.7 years, respectively) and male sex (31.4% vs 17.7%). Symptomatic SARS-CoV-2 infection occurred in 8 fully vaccinated health care workers and 38 unvaccinated health care workers (incidence rate, 4.7 vs 149.8 per 100 000 person-days, respectively, adjusted IRR, 0.03 [95% CI, 0.01-0.06]). Asymptomatic SARS-CoV-2 infection occurred in 19 fully vaccinated health care workers and 17 unvaccinated health care workers (incidence rate, 11.3 vs 67.0 per 100 000 person-days, respectively, adjusted IRR, 0.14 [95% CI, 0.07-0.31]). The results were qualitatively unchanged by the propensity score sensitivity analysis.
Conclusions and Relevance
Among health care workers at a single center in Tel Aviv, Israel, receipt of the BNT162b2 vaccine compared with no vaccine was associated with a significantly lower incidence of symptomatic and asymptomatic SARS-CoV-2 infection more than 7 days after the second dose. Findings are limited by the observational design.
This cohort study estimates the association between Pfizer-BioNTech BNT162b2 vaccination and symptomatic and asymptomatic SARS-CoV-2 infections among health care workers more than 7 days after receipt of a second vaccine dose.
Introduction
The BNT162b2 COVID-19 vaccine manufactured by Pfizer and BioNTech demonstrated 95% efficacy in preventing symptomatic SARS-CoV-2 infection in a phase 3, placebo-controlled randomized clinical trial,1 and became the first COVID-19 vaccine to receive Emergency Use Authorization by the US Food and Drug Administration.2 On December 19, 2020, 8 days after the US Food and Drug Administration authorization, a large-scale campaign was launched in Israel to vaccinate its population.3 Health care workers were among the first eligible for vaccination, along with individuals at risk of COVID-19 complications and death. Concurrently with the vaccination campaign, the number of new COVID-19 cases surged in Israel, with up to 10 000 new cases per day (eFigure 1 in the Supplement),4,5 possibly due to the spread of the B.1.1.7 SARS-CoV-2 variant.6,7
Analysis of uncontrolled data from the Israeli vaccination campaign has been consistent with the results from the phase 3 randomized clinical trial. The risk ratio for symptomatic disease, estimated from a cohort of more than 500 000 vaccinated adults and matched unvaccinated controls, was 0.06.8 However, the association of BNT162b2 vaccination with asymptomatic infection and transmission remains unclear, with important implications for public health policy. Data on vaccine effectiveness for health care workers, who are frequently at risk of exposure to SARS-CoV-2, are also limited. Asymptomatically infected individuals are estimated to account for 40% to 45% of all SARS-CoV-2 infections and may silently spread the virus for extended periods.9 Asymptomatic infections have been proposed as a major barrier to controlling the spread of SARS-CoV-2 infection and are a possible explanation for the rapid evolution of the COVID-19 pandemic.9
This study was conducted to assess the association between vaccination with the BNT162b2 vaccine and SARS-CoV-2 infections among health care workers at a tertiary medical center in Tel Aviv, Israel. Periodic screening for SARS-CoV-2 infection, coupled with detailed investigation of each infection confirmed via polymerase chain reaction (PCR) tests, allowed reliable estimation of symptomatic and asymptomatic infection rates.
Methods
Study Design
This was a retrospective cohort study designed to estimate the association between vaccination with the BNT162b2 vaccine and SARS-CoV-2 infections among health care workers. The study was conducted at the Tel Aviv Sourasky Medical Center, a tertiary medical center that employs approximately 7500 health care workers and close to 4000 nonsalaried health care workers (eg, students, volunteers). The hospital has implemented since May 2020 a policy of routinely screening health care workers with potential exposure to SARS-CoV-2 using nasopharyngeal swabs and PCR-based virus detection. In addition, each case of SARS-CoV-2 infection in a health care worker triggered an epidemiological investigation that included the collection and recording of clinical and virological data.
All health care workers were eligible to receive the vaccine from the first day of the vaccination campaign and throughout the study period. Data were retrieved from the hospital information system database, which aggregates data from multiple sources including personnel files, vaccination reports, laboratory databases, digitized epidemiological questionnaires, and external reports from health authorities.
Ethics approval and review were performed according to the Declaration of Helsinki10 and were obtained from the institutional review board along with a waiver of written informed consent.
Group Assignment and Definition of the Follow-up Period
Health care workers who received at least 1 vaccine dose between December 20, 2020, and February 25, 2021, were assigned to the vaccinated group. The control group was composed of health care workers who did not receive any doses of the BNT162b2 vaccine during this period. This partition of the groups was used throughout the analysis unless explicitly mentioned otherwise.
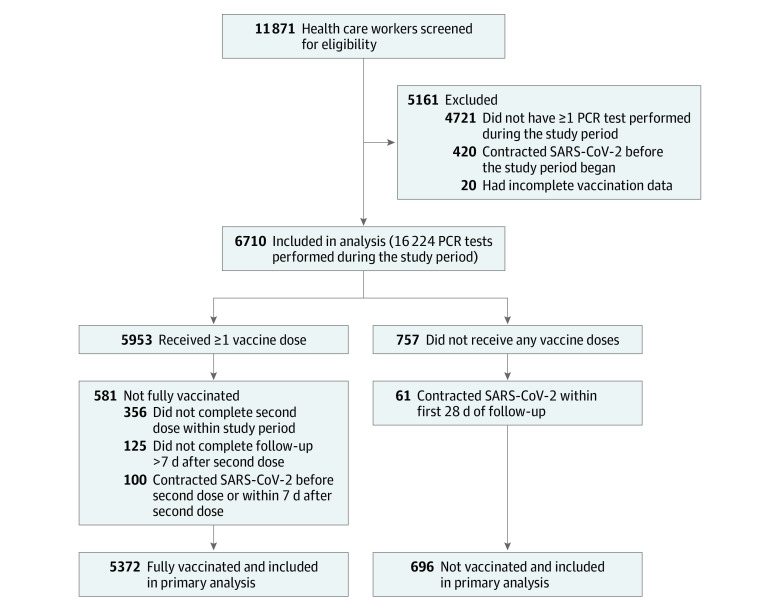
The PCR tests were performed according to the screening policy of the hospital. The screening policy changed during the course of the study period as follows: from December 20, 2020, to January 2, 2021 (period 1), health care workers were screened monthly or biweekly depending on their risk of SARS-CoV-2 exposure; from January 3 to 14, 2021 (period 2), hospital-wide screening was done including all health care workers regardless of vaccination status; and from January 15, 2021-onward (period 3), health care workers with medium to high exposure risk (defined below) and non–fully vaccinated health care workers were screened monthly to weekly in accordance with evolving hospital directives (Figure 1A and eMethods in the Supplement). All health care workers had open and free access to same-day PCR testing at their own discretion. Health care workers who did not undergo at least 1 PCR test during the study period, had incomplete data pertaining to vaccination dates, or contracted SARS-CoV-2 infection prior to the study period were excluded from the analysis (Figure 2).
Figure 1. Institutional Screening Policies, Vaccine Uptake in the Vaccinated Group, and the Daily Proportion of Vaccinated and Unvaccinated Participants Tested for SARS-CoV-2.
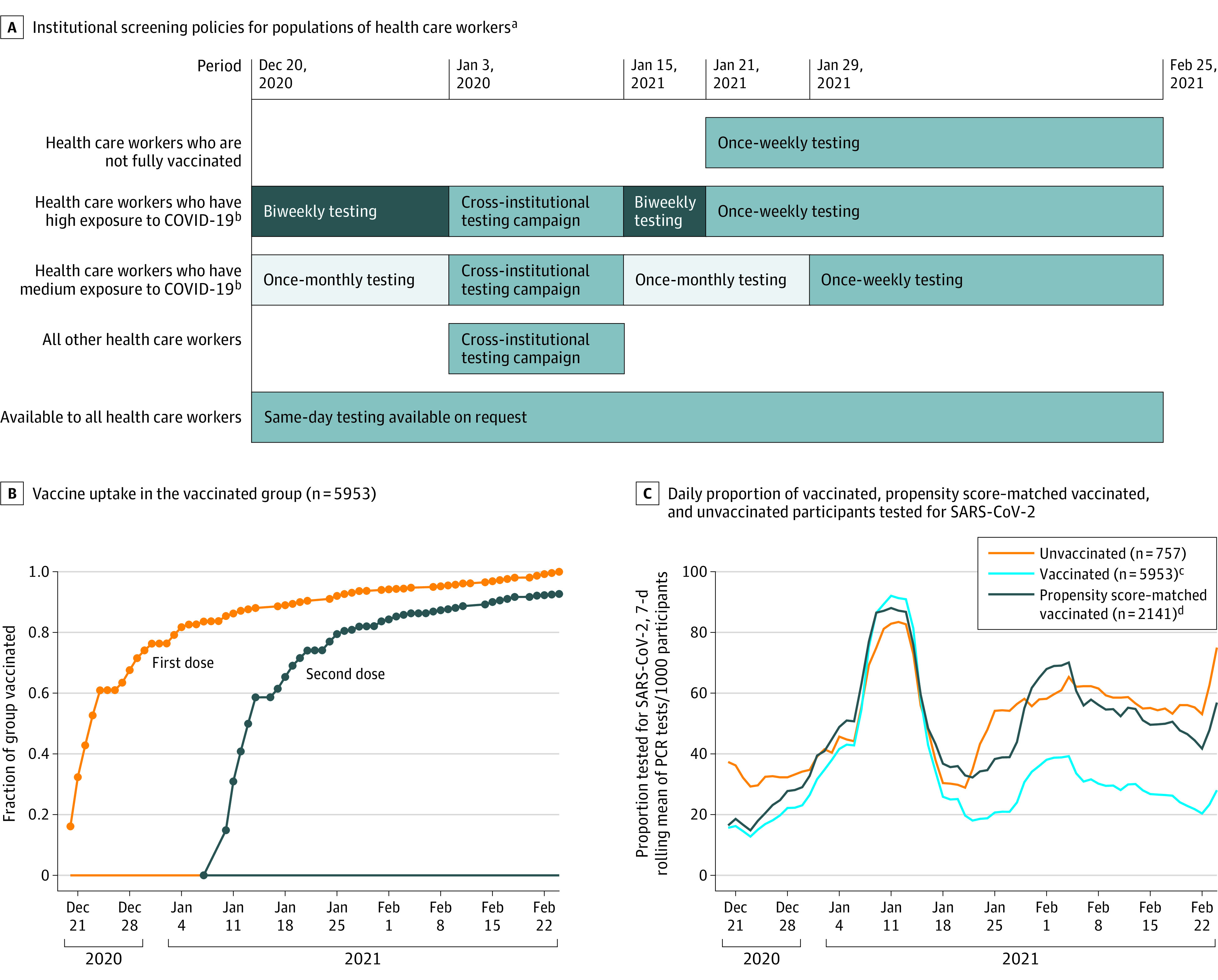
PCR indicates polymerase chain reaction.
aA detailed description of the study periods appears in the eMethods in the Supplement.
bRegardless of vaccination status.
cUnadjusted for propensity score.
dAdjusted for propensity score.
Figure 2. Study Cohort Derivation Process.
PCR indicates polymerase chain reaction.
The follow-up period for each health care worker was defined as starting either on the day of receiving the first vaccine dose for vaccinated health care workers or on December 20, 2020, for unvaccinated health care workers. Participants were censored at the first positive PCR test result or on February 25, 2021.
Definitions
The risk of exposure to a SARS-CoV-2–positive person was classified as high (for persons working in the emergency department or dedicated COVID-19 units), medium (for persons working in internal medicine departments or who performed high-risk procedures), or low (for other population subgroups). If several levels of exposure were listed for a given health care worker over the course of the study period (eg, worked in a dedicated COVID-19 unit for part of the study period), the highest risk level of exposure was used.
Participants infected with SARS-CoV-2 were defined as symptomatic if they had any of the following: temperature greater than 37.6 °C, headache, sore throat, cough, dyspnea, rhinorrhea, diarrhea, myalgia, malaise, or loss of sense of taste or smell. Symptoms were obtained from postinfection epidemiological interviews performed by the hospital’s infection prevention and control unit. Participants were considered fully vaccinated more than 7 days after the second dose of the vaccine, late fully vaccinated more than 21 days after the second dose of the vaccine, and partially vaccinated 7 to 28 days after the first dose of the vaccine.
Study Outcomes
The primary outcome was incidence of SARS-CoV-2 infection among fully vaccinated health care workers compared with unvaccinated health care workers who did not test positive for SARS-CoV-2 infection within the first 28 days of follow-up.
Secondary outcomes were the incidence of SARS-CoV-2 infection for partially vaccinated participants and for late fully vaccinated participants, as defined above, compared with unvaccinated participants. Outcomes were also analyzed for predefined subgroups according to age, sex, employment sector, and estimated level of exposure to SARS-CoV-2.
Statistical Analysis
Continuous variables are expressed as mean (SD) for normally distributed variables and as median (interquartile range) for non–normally distributed variables. Categorical variables are expressed as number (percentage) of health care workers within each group. The groups were compared using t tests for normally distributed continuous variables and χ2 tests for categorical variables. Missing values for categorical variables were considered as a separate category. All reported tests were 2-sided and a P value of less than .05 was considered significant.
Incidence Rate Ratio Analysis
The incidence rate ratio (IRR) was used as an estimate of vaccine effectiveness between vaccinated and unvaccinated health care workers and was calculated for each of the outcomes defined above.
The unadjusted IRR (uIRR) was computed directly from the incidence rates of the 2 groups as
 |
The incidence rates were defined for each of the 2 groups as the number of positive SARS-CoV-2 cases divided by the cumulative follow-up time of the health care workers in the group within the examined time frame. The 95% CI was calculated as (eln(IRR)–1.96 × SE, eln(IRR)+1.96 × SE). The standard error (SE) of the ln(IRR) was defined as
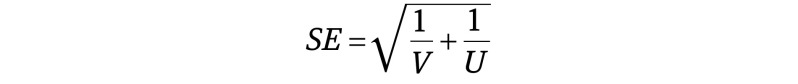 |
where V and U represent the number of SARS-CoV-2–positive cases confirmed via PCR tests in the vaccinated and unvaccinated groups, respectively.
The adjusted IRR was estimated using a multivariable Poisson regression model with confirmed cases as a response variable and group assignment (vaccinated vs unvaccinated), age, sex, employment sector, exposure risk, and number of PCR tests for each health care worker in the time frame under observation as explanatory variables. The IRRs were computed by exponentiating the group assignment coefficient from the final regression model and the 95% CIs and P values were estimated from the model. Because of the potential for type I error, the results from the secondary outcomes and the subgroup analyses should be interpreted as exploratory. Tests of interaction between subgroups were not conducted because of inadequate power.
Cumulative incidence curves for the vaccinated and unvaccinated groups were estimated using the Kaplan-Meier method and the 95% CIs for each curve were estimated using the percentile bootstrap method. Full details of estimation for the IRRs and the bootstrap method appear in the eMethods in the Supplement.
Sensitivity Analyses
To account for possible differences between the groups, including the possibility of a detection bias due to a difference in the number of tests carried out per group, a propensity score–adjusted sensitivity analysis11 was performed. The propensity score for each health care worker was computed using a multivariable logistic regression model with group assignment (vaccinated vs unvaccinated) as the response variable and age, sex, employment sector, exposure risk, and the number of PCR tests performed for each health care worker as explanatory variables. Each unvaccinated health care worker was matched with 2 to 3 vaccinated health care workers with similar propensities of being vaccinated. Full details of the propensity score computation and matching procedure appear in the eMethods in the Supplement.
To account for the possibility of bias introduced by the arbitrary definition of the index date for unvaccinated individuals, a sensitivity analysis was performed by offsetting the beginning of the follow-up period for unvaccinated participants to each of the 28 consecutive days between December 21, 2020, and January 18, 2021, and computing the IRR using the same procedure described above.
All statistical analyses were performed using R software version 4.0.3 (R Foundation for Statistical Computing).
Results
Participants
A total of 11 871 health care workers were screened for eligibility. After excluding the health care workers who contracted SARS-CoV-2 infection prior to the study period, those who had incomplete data regarding vaccination dates, and those who did not undergo any PCR testing throughout the study period, 6710 health care workers (mean [SD] age, 44.3 [12.5] years; 4465 [66.5%] women) remained and comprised the study cohort (Figure 2). Of this cohort, 5953 health care workers (88.7%) received at least 1 dose of the BNT162b2 vaccine, 5517 (82.2%) received 2 doses, and 757 (11.3%) were not vaccinated. Sixty-one percent of vaccinated health care workers received the first dose within 5 days of the initiation of the vaccination program and 75% within 12 days. Eighty percent of first dose recipients received the second dose within 21 days of receiving the first dose and 92% within 30 days (Figure 1B). The median overall follow-up period was 63 days.
The baseline characteristics of the participants appear in Table 1. Vaccinated health care workers were older compared with unvaccinated health care workers (mean age, 44.8 vs 40.7 years, respectively; P < .001) and were more frequently males (31.4% vs 17.7%; P < .001). The estimated work-related risk of exposure to SARS-CoV-2 was similar between the groups. Sex and employment sector data were not available for 242 (3.6%) and 170 (2.5%) participants, respectively.
Table 1. Baseline Characteristics of Study Participants.
| Unvaccinated | Vaccinated original cohort | SMD | Propensity score–matched vaccinated cohort | SMDa | |
|---|---|---|---|---|---|
| No. of participants | 757 | 5953 | 2141 | ||
| Received 2 doses of vaccine, No. (%) | 0 | 5517 (92.7) | 1984 (92.7) | ||
| Age, mean (SD), y | 40.7 (11.7) | 44.8 (12.5) | 0.337 | 41.0 (12.0) | 0.024 |
| Age group, No. (%) | |||||
| ≤39 y | 381 (50.3) | 2278 (38.3) | 0.289 | 1094 (51.1) | 0.043 |
| 40-59 y | 321 (42.4) | 2840 (47.7) | 872 (40.7) | ||
| ≥60 y | 55 (7.3) | 835 (14.0) | 175 (8.2) | ||
| Sex, No. (%) | |||||
| Female | 585 (77.3) | 3880 (65.2) | 0.325 | 1634 (76.3) | 0.062 |
| Male | 134 (17.7) | 1869 (31.4) | 419 (19.6) | ||
| Missing data | 38 (5.0) | 204 (3.4) | 88 (4.1) | ||
| Employment sector, No. (%) | |||||
| Administration | 324 (42.8) | 2421 (40.7) | 0.473 | 850 (39.7) | 0.160 |
| Nursing | 263 (34.7) | 1438 (24.1) | 732 (34.2) | ||
| Medicine | 44 (5.8) | 1231 (20.7) | 215 (10.0) | ||
| Other health professionsb | 98 (13.0) | 721 (12.1) | 271 (12.7) | ||
| Missing data | 28 (3.7) | 142 (2.4) | 73 (3.4) | ||
| Estimated exposure level to COVID-19, No. (%) | |||||
| Low | 661 (87.3) | 5070 (85.2) | 0.064 | 1791 (83.7) | 0.106 |
| Medium | 22 (2.9) | 188 (3.2) | 72 (3.4) | ||
| High | 74 (9.8) | 695 (11.7) | 278 (13.0) | ||
| Follow-up time, median (IQR), d | 66.0 (66.0-66.0) | 63.0 (52.0-65.0) | 0.322 | 62.0 (51.0-65.0) | 0.412 |
| No. of PCR tests per person, median (IQR) | 3.0 (1.0-5.0) | 2.0 (1.0-3.0) | 0.551 | 3.0 (1.0-5.0) | 0.090 |
| Test density per 100 d of follow-up, median (IQR) | 5.88 (2.99-8.96) | 3.03 (1.56-5.97) | 0.312 | 5.26 (2.99-8.96) | 0.076 |
Abbreviations: IQR, interquartile range; PCR, polymerase chain reaction; SMD, standardized mean difference.
Compares the propensity score–matched adjusted vaccinated cohort (n = 2141) with the unvaccinated cohort (n = 757).
Included physiotherapists, speech therapists, phlebotomists, respiratory technicians, and other paramedical staff.
A total of 16 224 PCR tests to detect SARS-CoV-2 infection were performed during the study period (13 615 in the vaccinated group and 2609 in the unvaccinated group). The daily rate of testing in each group changed over the course of the study period, with significantly more tests performed per participant in the unvaccinated cohort during period 3 (defined as January 15, 2021-onward; Figure 1A, C).
A total of 243 PCR-confirmed cases of SARS-CoV-2 infection were documented during the study period, which were categorized after epidemiological investigation as symptomatic (n = 149 [61.3%]) and asymptomatic (n = 94 [38.7%]; eTable 1 in the Supplement).
Association Between Vaccination Status and Incidence of Symptomatic SARS-CoV-2 Infection
Symptomatic SARS-CoV-2 infection was detected in 64 health care workers who received at least 1 dose of the vaccine (incidence rate, 19.4 per 100 000 person-days) and in 85 health care workers in the unvaccinated cohort (incidence rate, 186.1 per 100 000 person-days) (Figure 3 and eFigure 2 in the Supplement).
Figure 3. Cumulative Incidence of SARS-CoV-2 Infection Among Vaccinated, Propensity Score–Matched Vaccinated, and Unvaccinated Participants Screened for SARS-CoV-2 Infection.
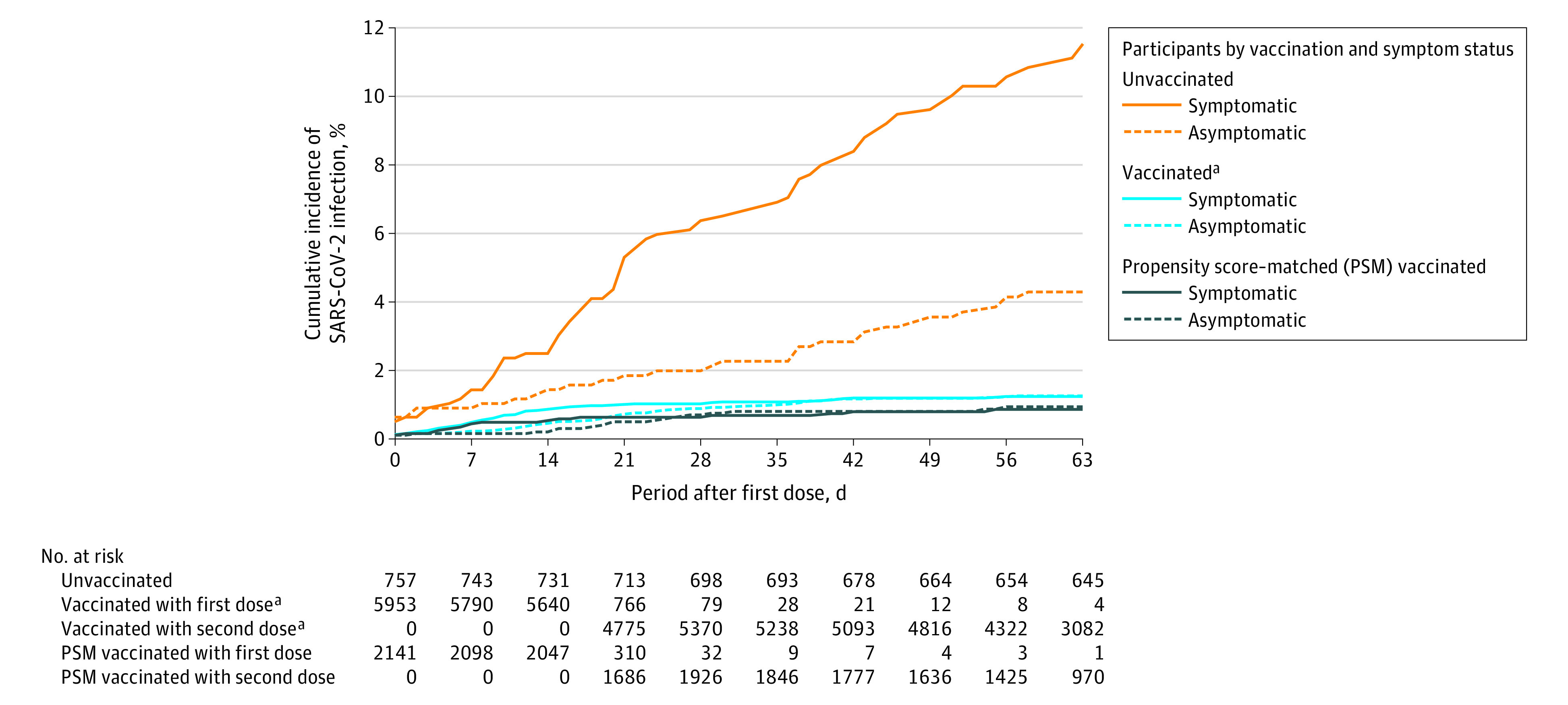
Data are the cumulative incidence of symptomatic and asymptomatic SARS-CoV-2 cases after the first dose of the vaccine in the unvaccinated cohort (n = 757), in the original vaccinated cohort (n = 5953), and in the propensity score–matched vaccinated cohort (n = 2141). Additional information appears in eFigure 2 in the Supplement.
aUnadjusted for propensity score.
For the primary outcome analysis, the incidence rate of symptomatic SARS-CoV-2 infection was 4.7 vs 149.8 per 100 000 person-days in the fully vaccinated and unvaccinated cohorts, respectively, corresponding with an adjusted IRR of 0.03 (95% CI, 0.01-0.06; Table 2). The corresponding adjusted IRRs for the late fully vaccinated and partially vaccinated cohorts were 0.02 (95% CI, 0-0.06) and 0.11 (95% CI, 0.06-0.17), respectively.
Table 2. Observed Incidence Rate Ratios of Symptomatic and Asymptomatic SARS-CoV-2 Infectiona.
| Vaccination status | Subgroup | Vaccinated | Unvaccinatedb | Incidence rate ratio (95% CI)c | P value | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of cases | No. | Surveillance time, person-days | Incidence rate per 100 000 person-daysc | No. of cases | No. | Surveillance time, person-days | Incidence rate per 100 000 person-daysc | Unadjustedd | Adjustede | |||
| Original cohort | ||||||||||||
| Fully vaccinatedf | Symptomatic | 8 | 5372 | 168 571 | 4.7 | 38 | 696 | 25 359 | 149.8 | 0.03 (0.01-0.07) | 0.03 (0.01-0.06) | <.001 |
| Asymptomatic | 19 | 11.3 | 17 | 67.0 | 0.17 (0.09-0.32) | 0.14 (0.07-0.31) | <.001 | |||||
| Late fully vaccinatedg | Symptomatic | 2 | 5036 | 95 689 | 2.1 | 23 | 675 | 15 726 | 146.3 | 0.01 (0-0.06) | 0.02 (0-0.06) | <.001 |
| Asymptomatic | 4 | 4.2 | 11 | 69.9 | 0.06 (0.02-0.19) | 0.06 (0.02-0.22) | <.001 | |||||
| Partially vaccinatedh | Symptomatic | 31 | 5761 | 117 389 | 26.4 | 37 | 741 | 15 091 | 245.2 | 0.11 (0.07-0.17) | 0.11 (0.06-0.17) | <.001 |
| Asymptomatic | 37 | 31.5 | 8 | 53.0 | 0.59 (0.28-1.28) | 0.64 (0.31-1.51) | .27 | |||||
| Propensity score–matched adjusted cohort | ||||||||||||
| Fully vaccinatedf | Symptomatic | 2 | 1916 | 57 274 | 3.5 | 38 | 696 | 25 359 | 149.8 | 0.02 (0.01-0.10) | 0.02 (0-0.07) | <.001 |
| Asymptomatic | 4 | 7.0 | 17 | 67.0 | 0.10 (0.04-0.31) | 0.09 (0.03-0.25) | <.001 | |||||
| Late fully vaccinatedg | Symptomatic | 1 | 1748 | 31 645 | 3.2 | 23 | 675 | 15 726 | 146.3 | 0.02 (0-0.16) | 0.02 (0-0.10) | <.001 |
| Asymptomatic | 2 | 6.3 | 11 | 69.6 | 0.09 (0.02-0.41) | 0.09 (0.01-0.35) | .002 | |||||
| Partially vaccinatedh | Symptomatic | 4 | 2085 | 42 414 | 9.4 | 37 | 741 | 15 091 | 245.2 | 0.04 (0.01-0.11) | 0.03 (0.01-0.09) | <.001 |
| Asymptomatic | 11 | 25.9 | 8 | 53.0 | 0.49 (0.20-1.22) | 0.48 (0.19-1.26) | .12 | |||||
Participants with fewer than 7 days of follow-up or who contracted SARS-CoV-2 less than 7 days after the first vaccine dose were not included in this analysis (Figure 2).
The surveillance period used corresponded to the period used for the vaccinated participants (ie, 28, 42, or 7-28 days after the beginning of follow-up, which was set as December 20, 2020).
When estimating incidence rates and incidence rate ratios of symptomatic infection, asymptomatic cases were censored on the day of case confirmation (and vice versa).
Indicates the ratio of incidence rates in each group.
Calculated using Poisson regression as detailed in the Methods section.
Included those with data for longer than 7 days after the second dose to the end of follow-up.
Included those with data for longer than 21 days after the second dose to the end of follow-up.
Included those with data for days 7 to 28 after first dose.
Association Between Vaccination Status and Incidence of Asymptomatic SARS-CoV-2 Infection
Asymptomatic SARS-CoV-2 infection was documented in 63 health care workers who received at least 1 dose of vaccine (incidence rate, 19.1 per 100 000 person-days) and in 31 health care workers in the unvaccinated cohort (incidence rate, 67.9 per 100 000 person-days; Figure 3 and eFigure 2 in the Supplement).
For the primary outcome analysis, the incidence rate of asymptomatic SARS-CoV-2 infection was 11.3 vs 67.0 per 100 000 person-days in the fully vaccinated and unvaccinated cohorts, respectively, corresponding with an adjusted IRR of 0.14 (95% CI, 0.07-0.31; Table 2). The corresponding adjusted IRRs for late fully vaccinated and partially vaccinated cohorts were 0.06 (95% CI, 0.02-0.22) and 0.64 (95% CI, 0.31-1.51), respectively.
Sensitivity and Subgroup Analyses
The vaccinated and unvaccinated groups in the propensity score–adjusted cohort were well-balanced with respect to the participant demographics and the number of PCR tests performed throughout the study period (Table 1 and Figure 1C). The primary outcome for symptomatic and asymptomatic SARS-CoV-2 infections remained significantly lower in the propensity score–matched cohort of fully vaccinated health care workers vs those unvaccinated, with IRRs similar to those from the primary analysis (Figure 3 and Table 2). The results were similar for the partially vaccinated and late fully vaccinated cohorts (Table 2).
The overall estimations of the adjusted IRR for symptomatic and asymptomatic SARS-CoV-2 infection in the subgroup analyses were consistent with the results of the primary analysis. However, the results were not statistically significant for certain health care worker populations (asymptomatic infections in males as well as all infections in persons aged >60 years and in those with medium or high SARS-CoV-2 exposure) (eTable 2 in the Supplement).
The use of offsetting in the sensitivity analysis for the beginning of the follow-up period for the unvaccinated cohort resulted in shorter follow-up periods but did not affect the observed adjusted IRR for symptomatic and asymptomatic infection (eFigure 3 in the Supplement).
Discussion
In this retrospective cohort study of regularly screened health care workers, vaccination with 2 doses of the BNT162b2 messenger RNA vaccine was associated with significantly lower incidence rates for both symptomatic and asymptomatic SARS-CoV-2 infection. The adjusted IRR for the primary outcome was 0.03 for symptomatic infection and 0.14 for asymptomatic infection, corresponding to an estimated vaccine effectiveness (1−IRR) of 97% and 86%, respectively.
The association between vaccination status and symptomatic SARS-CoV-2 infection is similar to the 95% efficacy reported in the phase 3 randomized clinical trial1 and the risk ratio of 0.06 observed in a study of the nationwide vaccination campaign in Israel.8 The risk ratio for the asymptomatic infection proxy in the nationwide vaccination study8 was somewhat lower than the results of this study (risk ratio of 0.10 vs IRR of 0.14), which may be due to a difference in study methods or may reflect more stringent classification of symptomatic cases in the current analysis. Separation of cumulative incidence curves occurred later for asymptomatic than for symptomatic SARS-CoV-2 infections, with the adjusted IRR for asymptomatic SARS-CoV-2 infections reaching 0.06 (estimated vaccine effectiveness of 94%) 21 days after the second vaccine dose.
Asymptomatic infections accounted for 38.7% of all cases of PCR-confirmed SARS-CoV-2 infection in this cohort, which is similar to the results reported in other cross-sectional studies.9 Asymptomatically infected individuals can harbor high titers of culturable virus in their upper airways, similar to those of symptomatic patients,12,13 and may have clinically silent pulmonary infiltrates on chest imaging,14 making them a potential source of transmissible SARS-CoV-2 infection in the community and within hospitals.13,15
Vaccine effectiveness for symptomatic and asymptomatic infections among health care workers is the subject of ongoing research. Health care workers were excluded from many studies including the Israeli nationwide mass vaccination study8 because of the high variability of exposure among health care workers. Amit et al16 reported a rate reduction of 85% for SARS-CoV-2 infection among vaccinated compared with unvaccinated health care workers within 15 to 28 days of the first vaccine dose, with a shorter mean follow-up period than the current study. Benenson et al17 found a lower incidence of COVID-19 among vaccinated health care workers compared with unvaccinated health care workers that was apparent 4 weeks after the first vaccine dose and remained lower throughout the follow-up time; however, vaccinated individuals were not regularly screened. Neither of these studies specifically assessed rates of asymptomatic SARS-CoV-2 infection.
The strengths of this data set include the availability of systematically collected PCR data and clinical symptoms on a well characterized cohort of health care workers, allowing relatively robust assessment of asymptomatic infections. The potential implications of the observed reduction in asymptomatic infections on SARS-CoV-2 transmission require further study. Given the potential role of asymptomatically infected persons in driving the COVID-19 pandemic,15,18 the effect of this reduction on the silent spread of SARS-CoV-2 infection likely has important public health consequences.
Limitations
This study has several limitations. First, the inherent characteristics of a single-center, retrospective cohort study might limit the generalizability of the findings.
Second, the vaccinated and unvaccinated cohorts differed in size and in some individual characteristics. Unvaccinated health care workers were younger and were more frequently females. However, the primary outcome was stable in the propensity score–matched cohort, which was well-balanced with regard to sex and age.
Third, vaccinated health care workers underwent fewer PCR tests for SARS-CoV-2 infection than unvaccinated health care workers after January 15, 2021, potentially biasing toward more observed infections in the unvaccinated cohort, although the analyses attempted to account for this possible imbalance.
Fourth, other confounders may be present that were unaccounted for in the regression analyses and in the adjustments for propensity score. For example, a healthy vaccinee bias, referring to an association between adherence to vaccination recommendations and general health status and behavior, cannot be excluded.
Conclusions
Among health care workers at a single center in Tel Aviv, Israel, receipt of the BNT162b2 vaccine compared with no vaccine was associated with a significantly lower incidence of symptomatic and asymptomatic SARS-CoV-2 infection more than 7 days after the second dose. Findings are limited by the observational design.
eFigure 1. COVID-19 in Israel during the study period
eFigure 2. Cumulative incidence of SARS-CoV-2 infection, including 95% confidence intervals
eFigure 3. Sensitivity analysis for index date in the unvaccinated group
eMethods
eTable 1. Characteristics of SARS-CoV-2 positive participants
eTable 2. Subgroup analyses for the primary endpoint
References
- 1.Polack FP, Thomas SJ, Kitchin N, et al. ; C4591001 Clinical Trial Group . Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020;383(27):2603-2615. doi: 10.1056/NEJMoa2034577 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.US Food and Drug Agency . Emergency Use Authorization for Pfizer-BioNTech COVID-19 vaccine. Accessed February 25, 2021. https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-covid-19/pfizer-biontech-covid-19-vaccine
- 3.Rosen B, Waitzberg R, Israeli A. Israel’s rapid rollout of vaccinations for COVID-19. Isr J Health Policy Res. 2021;10(1):6. doi: 10.1186/s13584-021-00440-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Israel Ministry of Health . Israeli COVID-19 dataset. Accessed March 22, 2021. https://data.gov.il/dataset/covid-19
- 5.Roser M, Ritchie H, Ortiz-Ospina E, Hasell J. Coronavirus pandemic (COVID-19): our world in data. Accessed February 25, 2021. https://ourworldindata.org/coronavirus
- 6.Mascola JR, Graham BS, Fauci AS. SARS-CoV-2 viral variants—tackling a moving target. JAMA. 2021;325(13):1261-1262. doi: 10.1001/jama.2021.2088 [DOI] [PubMed] [Google Scholar]
- 7.Munitz A, Yechezkel M, Dickstein Y, Yamin D, Gerlic M. BNT162b2 vaccination effectively prevents the rapid rise of SARS-CoV-2 variant B.1.1.7 in high risk populations in Israel. Cell Rep Med. Published online April 18, 2021. doi: 10.1016/j.xcrm.2021.100264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dagan N, Barda N, Kepten E, et al. BNT162b2 mRNA Covid-19 vaccine in a nationwide mass vaccination setting. N Engl J Med. 2021;384(15):1412-1423. doi: 10.1056/NEJMoa2101765 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Oran DP, Topol EJ. Prevalence of asymptomatic SARS-CoV-2 infection: a narrative review. Ann Intern Med. 2020;173(5):362-367. doi: 10.7326/M20-3012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.World Medical Association . World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310(20):2191-2194. doi: 10.1001/jama.2013.281053 [DOI] [PubMed] [Google Scholar]
- 11.Desai RJ, Franklin JM. Alternative approaches for confounding adjustment in observational studies using weighting based on the propensity score: a primer for practitioners. BMJ. 2019;367:l5657. doi: 10.1136/bmj.l5657 [DOI] [PubMed] [Google Scholar]
- 12.Kimball A, Hatfield KM, Arons M, et al. ; Public Health – Seattle & King County; CDC COVID-19 Investigation Team . Asymptomatic and presymptomatic SARS-CoV-2 infections in residents of a long-term care skilled nursing facility—King County, Washington, March 2020. MMWR Morb Mortal Wkly Rep. 2020;69(13):377-381. doi: 10.15585/mmwr.mm6913e1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Singanayagam A, Patel M, Charlett A, et al. Duration of infectiousness and correlation with RT-PCR cycle threshold values in cases of COVID-19, England, January to May 2020. Euro Surveill. 2020;25(32):1-5. doi: 10.2807/1560-7917.ES.2020.25.32.2001483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ooi EE, Low JG. Asymptomatic SARS-CoV-2 infection. Lancet Infect Dis. 2020;20(9):996-998. doi: 10.1016/S1473-3099(20)30460-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee S, Kim T, Lee E, et al. Clinical course and molecular viral shedding among asymptomatic and symptomatic patients with SARS-CoV-2 Infection in a community treatment center in the Republic of Korea. JAMA Intern Med. 2020;180(11):1447-1452. doi: 10.1001/jamainternmed.2020.3862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Amit S, Regev-Yochay G, Afek A, Kreiss Y, Leshem E. Early rate reductions of SARS-CoV-2 infection and COVID-19 in BNT162b2 vaccine recipients. Lancet. 2021;397(10277):875-877. doi: 10.1016/S0140-6736(21)00448-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Benenson S, Oster Y, Cohen MJ, Nir-Paz R. BNT162b2 mRNA Covid-19 vaccine effectiveness among health care workers. N Engl J Med. Published online March 23, 2021. doi: 10.1056/NEJMc2101951 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nogrady B. What the data say about asymptomatic COVID infections. Nature. 2020;587(7835):534-535. doi: 10.1038/d41586-020-03141-3 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eFigure 1. COVID-19 in Israel during the study period
eFigure 2. Cumulative incidence of SARS-CoV-2 infection, including 95% confidence intervals
eFigure 3. Sensitivity analysis for index date in the unvaccinated group
eMethods
eTable 1. Characteristics of SARS-CoV-2 positive participants
eTable 2. Subgroup analyses for the primary endpoint