Abstract
A 68-year-old man was referred to the general surgeons on account of his abdominal pain of unknown cause. He had contracted COVID-19, 9 days prior. CT chest abdomen and pelvis revealed an extensive thrombus extending from the portal vein to the superior mesenteric vein. Further investigation ruled out haematological causes, and COVID-19 was determined to be the cause. He was treated with an extended course of therapeutic dose low molecular weight heparin under the guidance of the haematology team. He was discharged once he was clinically stable and pain-free, with a plan to be followed up by both the surgeons and haematologists. This case highlights the different ways in which COVID-19 presents, and the need for clearer guidance on the treatment and prevention of thromboembolism in COVID-19.
Keywords: COVID-19, haematology (incl blood transfusion), radiology, general surgery, portal vein
Background
COVID-19 is a respiratory viral infection caused by the novel coronavirus SARS-CoV-2, and its rapid global spread has led to more than three million deaths in 12 months.1 The vast majority of these deaths are due to the virus’ effects on the respiratory system, causing widespread patchy pneumonia and resultantly respiratory insufficiency, sepsis and organ failure.2 However, significant morbidity and mortality is beginning to be reported from the non-respiratory effects of infection with COVID-19. In particular, the virus has the ability to produce a procoagulant state that predisposes to venous thromboembolism (VTE) and arterial thromboembolism, commonly in the form of pulmonary embolism (PE), though there are emerging case reports of thromboembolism in other locations.3 4 In this case report, we describe a 68-year-old man who presented with abdominal pain, and was found to have superior mesenteric vein and portal vein thrombosis after contracting COVID-19.
Case presentation
A 68-year-old man presented to our Accident & Emergency department with a 1-day history of abdominal pain and distension. The pain was of sudden onset, sharp in character and localised to the central abdomen with no radiation. He denied nausea, diarrhoea and vomiting. He had experienced constipation for the past week, but had passed flatus. On examination, his abdomen was soft with generalised tenderness. There was no guarding or peritonism. A soft, reducible, 2 cm umbilical hernia was noted. Bowel sounds were present on auscultation. There was no lower extremity oedema or sign of venous congestion. Digital rectal examination revealed soft stools in the rectum. He also had a mild dry cough. On auscultation of his chest, he had mild bi-basal crepitations. He had tested positive for COVID-19, 9 days prior on nasopharyngeal swab. The rest of his medical history included chronic obstructive pulmonary disease, mild Alzheimer’s dementia and recurrent urinary tract infection. There was no family history of thrombotic disease. His only regular medications were salbutamol and tiotropium bromide inhalers and lubricating eye drops taken as required.
He was clinically stable with observations showing respiratory rate 18/min, oxygen saturation 94% on air, blood pressure 116/65 mm Hg, heart rate 83/min and temperature 36.7°C. Laboratory findings showed raised inflammatory markers and D-dimer (table 1). Lactate was 1.2 mmol/L. The primary diagnosis was referred pain due to COVID-19 pneumonia. He was given analgesia, intravenous fluids and phosphate enemas.
Table 1.
Laboratory findings on admission and on discharge
| Test | Result on admission | Result on discharge | Units | Normal range |
| Haemoglobin | 150 | 119 | g/L | 130–170 |
| Lymphocytes | 0.95 | 1.28 | *109/L | 1.0–3.0 |
| Neutrophils | 10.52 | 3.19 | *109/L | 2.0–7.0 |
| Platelet count | 318 | 352 | *109/L | 150–410 |
| WBC | 12.44 | 5.48 | *109/L | 4.0–10.0 |
| Albumin | 29 | 25 | g/L | 35–50 |
| ALP | 78 | 88 | U/L | 30–130 |
| ALT | 41 | 24 | U/L | <41 |
| Bilirubin | 13 | 6 | µmol/L | <21 |
| Creatinine | 87 | 91 | µmol/L | 62–106 |
| CRP | 307 | 117 | mg/L | <5 |
| Potassium | 4.5 | 4.1 | mmol/L | 3.5–5.3 |
| Sodium | 137 | 137 | mmol/L | 133–146 |
| D-dimer | 894 | ng/mL | <243 |
ALP, Alkaline phosphatase; ALT, Alanine transaminase; CRP, C-reactive protein; WBC, White blood cells.
Investigations
CT chest, abdomen and pelvis showed lung changes consistent with viral pneumonia (figure 1). There was no evidence of major PE. There was, however, a central venous filling defect within the portal vein extending to the superior mesenteric vein, including some terminal branches. There was associated engorgement of small bowel mesenteric vessels. There was no evidence of bowel wall changes, and no signs of obstruction or dilation. For the section of the portal vein system in the liver hilum, there were no changes. There was no free air in the abdomen (figure 2). These changes were concurrent with the diagnosis of portal and superior mesenteric vein thrombosis.
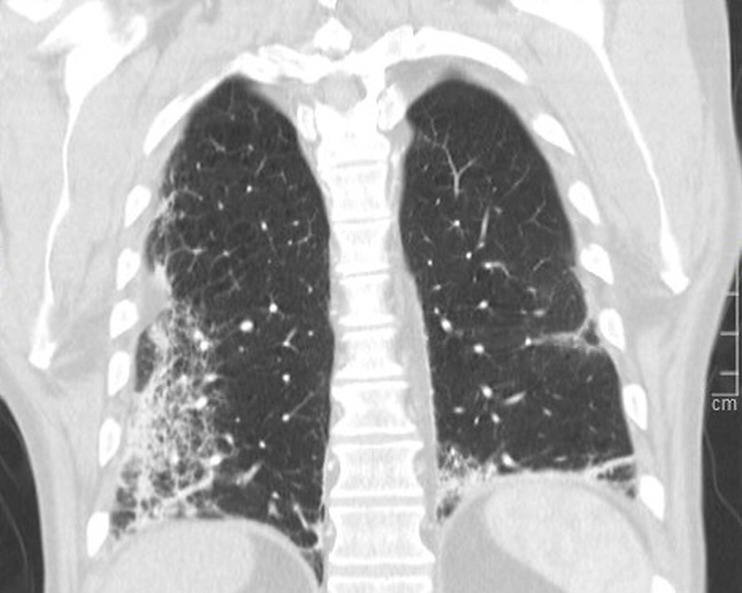
Figure 1.
CT coronal cross-section of the thorax showing patchy infiltrates in the right lower lobe and posterior basal segment of the left lower lobe, consistent with COVID-19 pneumonia.
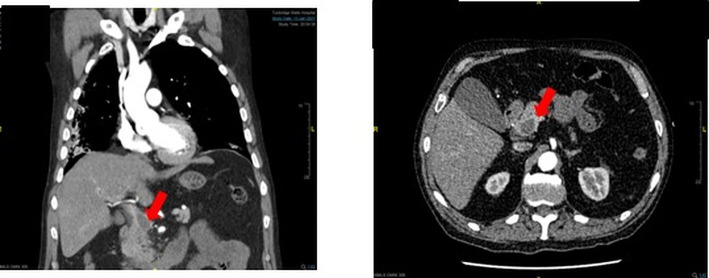
Figure 2.
CT coronal (left) and transverse (right) cross-sections of the abdomen and pelvis showing portal vein thrombus, annotated.
Treatment
Due to the location of the VTE, the case was discussed with the haematology team who advised therapeutic dose low molecular weight heparin (LMWH) for at least 3 months, as well as investigatory blood tests for prothrombotic disease which were unremarkable (table 2).
Table 2.
Haematology findings
| Test | Result | Units | Normal range |
| Anticardiolipin IgM | 8.26 | MPL U/mL | 0.00–6.99 |
| Anticardiolipin IgG | 7.0 | GPL | 0.0–10.0 |
| Beta-2-glycoprotein | 2.2 | U/mL | 0.0–5.0 |
| Calcium | 2.4 | mmol/L | 2.2–2.6 |
| Erythrocyte sedimentation rate (ESR) | 8 | mm/hour | 0–10 |
| Lactate dehydrogenase (LDH) | 532 | IU/L | 240–480 |
| Serum total protein | 55 | g/L | 60–80 |
| BCR-ABL | No translocations detected | ||
| Antinuclear antibody (ANA) | Negative | ||
| Antineutrophil cytoplasmic antibody (ANCA) | Negative | ||
| Myeloproliferative neoplasms (MPN) panel | No calreticulin, JAK2 or MPL mutations detected | ||
| CD55 and CD59 | No paroxysmal nocturnal haemoglobinuria (PNH) clones detected | ||
| Serum protein electrophoresis | Physiological gamma globulins reduced. Moderate acute phase reaction. No paraprotein present | ||
| Paraprotein typing | Oligoclonal response due to acute/chronic inflammatory reaction | ||
BCR-ABL, Translocation seen on the Philadelphia chromosome of CML patients.
Outcome and follow-up
Apart from a brief drop in oxygen saturation and temperature spike (39.3°C) that resolved after 24 hours, there were no signs of systemic illness, and his abdominal pain improved over the course of admission.
CT abdomen and pelvis 4 days after starting dalteparin showed little change in the extent of the thrombus, and there was a new additional finding of a small filling defect within the right lower lobe pulmonary arterial branches, consistent with a finding of acute PE (figure 3). On discussion with the haematology team, they suggested that repeat imaging not be routinely performed so early because residual thrombosis will be present in the majority of cases despite anticoagulation, and therefore imaging is unlikely to change the patient’s management. Having said this, the new finding of PE made them more certain that at least 3 months of anticoagulation was appropriate.
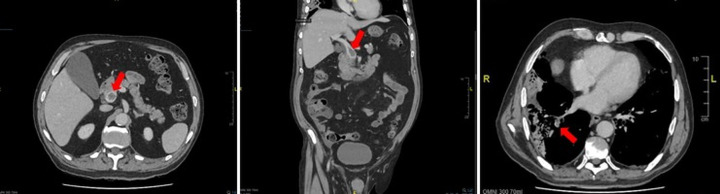
Figure 3.
CT transverse (left) and coronal (middle) cross-sections of the abdomen and pelvis showing unchanged portal vein thrombus after 4 days, annotated. CT transverse (right) cross-section of the thorax showing right-sided filling defect consistent with PE, annotated. PE, pulmonary embolism.
Eight days after admission, he was clinically stable and pain-free with improved inflammatory markers (table 1), and was subsequently discharged with a plan to be followed up in our surgical assessment unit in 2 weeks, by haematology in 10 weeks, and in thrombosis clinic in 12 weeks where his dalteparin dose would be reviewed.
Discussion
The pathophysiology of COVID-19 leading to a procoagulant state is being increasingly researched and seems to be multifactorial.5 Evidence suggests that the initiating event occurs in the lungs, with alveolar endothelial activation and proinflammatory cytokine release in COVID-19 pneumonia leading to a local thrombotic state in the pulmonary vasculature. This is then followed by a more generalised hypercoagulable state resulting in microvasculature and macrovasculature thrombosis.6 In this case study, we first ruled out any underlying haematological causes for the superior mesenteric and portal vein thrombosis, before concluding that this rare case was caused by the procoagulant state induced by COVID-19. This included investigating for myeloproliferative neoplasms, antiphospholipid syndrome, paroxysmal nocturnal haemoglobinuria, leukaemias such as chronic myeloid leukaemia, vasculitidies, myeloma and other causes of acquired thrombophilia (table 2). Furthermore, there was no evidence of overt neoplasm on CT scanning. Given that the patient denied any family history of prothrombotic disease, he was not tested for congenital causes of thrombophilia. However, an association has recently been noted between elevated factor V activity and COVID-19 coagulopathy.7 Clinicians may therefore wish to consider measurement of factor V activity for VTE risk stratification.
In the absence of research into the optimal management of COVID-19-associated VTE, a number of bodies including the National Institute of Health and the International Society on Thrombosis and Haemostasis have advised conforming to non-COVID VTE management guidelines.8 Our haematology team therefore directed us to anticoagulate the patient for at least 3 months, in line with other provoked VTEs such as PEs. They advised LMWH over oral anticoagulation because of the stronger general evidence base in relation to its anticoagulant effect in VTE. However, some suggest these guidelines are not adequate, and that a lower threshold for thrombolysis should be considered in acute PE with COVID-19 pneumonia where lung inflammation and arterial hypoperfusion both contribute to hypoxaemia.9
What is more hotly contested is thromboprophylaxis. There is currently much conflicting research, and no clear central guidance on regular inpatient VTE prophylaxis, with many trusts opting to produce and follow their own guidelines. Early studies suggested that VTEs were developing in patients with COVID-19 already on prophylactic LMWH,4 10 and in light of the finding that the risk of VTE is particularly high in patients with COVID-19 admitted to the intensive care unit (ICU), a number of trials sought to reduce thrombotic events in ICU patients by adopting ‘high-intensity’ thromboprophylaxis regimes, with mixed results.8 More recently, the REMAP-CAP trial has reported that therapeutic dose anticoagulation does not improve clinical outcomes for patients with severe COVID-19 in the ICU setting, whereas it does for those hospitalised patients with moderate COVID-19 not requiring organ support.11 12 Other ongoing trials aim to assess the efficacy and safety of extended postdischarge thromboprophylaxis.13
This case highlights the need for healthcare professionals to be aware that COVID-19 can present in ways other than respiratory distress—in particular, it can lead to significant and rare thrombotic complications. Clinicians should therefore have a lower threshold for suspecting thromboses other than PEs and deep vein thromboses (DVTs) in patients with COVID-19. These complications are potentially avoidable with early identification of high-risk groups and optimal thromboprophylaxis, and it will be imperative for clinicians and trusts to keep up to date with ongoing clinical trials and the guidelines that will soon follow. However, research into the prevention and management of VTE in COVID-19 will likely focus on PEs and DVTs due to their frequency, and therefore trusts should be prepared to develop local guidelines for rarer VTEs where felt appropriate.
Learning points.
COVID-19 has multimodal effects on the body, and therefore results in a number of non-respiratory presentations.
The procoagulant state in COVID-19 can result in thromboses in uncommon locations.
There is general agreement that COVID-19-associated venous thromboembolism (VTE) should be managed in accordance with non-COVID VTE guidelines, whereas recommendations on VTE prophylaxis in COVID-19 have been far more disparate.
There is a paucity of research and guidelines on the treatment and prevention of thrombosis in COVID-19 infection, but this is changing. Regardless, trusts may feel it necessary to develop their own guidelines for rarer thromboses.
Acknowledgments
We thank all authors and contributors for their work.
Footnotes
Contributors: MJ led the project and wrote the main bulk of the case report, including the introduction, case report, results and discussion. RH acquired the data, processed it and designed the tables. He also made significant contributions to the discussion. MR provided useful input on the language of the paper, chose the appropriate images and helped with the data interpretation. YA oversaw the planning of the case report and directed its overall theme, and made edits in accordance to this theme. He also provided direct access to the patient and his imaging and lab results.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Ethics statements
Patient consent for publication
Obtained.
References
- 1.WHO coronavirus (COVID-19) dashboard. Available: https://covid19.who.int/ [Accessed 17 Feb 2021].
- 2.Vincent J-L, Taccone FS. Understanding pathways to death in patients with COVID-19. Lancet Respir Med 2020;8:430–2. 10.1016/S2213-2600(20)30165-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Connors JM, Levy JH. COVID-19 and its implications for thrombosis and anticoagulation. Blood 2020;135:2033–40. 10.1182/blood.2020006000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Klok FA, Kruip MJHA, van der Meer NJM, et al. Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: an updated analysis. Thromb Res 2020;191:148–50. 10.1016/j.thromres.2020.04.041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Moschonas I, Tselepis A. SARS-CoV-2 infection and thrombotic complications: a narrative review. J Thromb Thrombolysis 2021:1–13. 10.1007/s11239-020-02374-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Abou-Ismail MY, Diamond A, Kapoor S, et al. The hypercoagulable state in COVID-19: incidence, pathophysiology, and management. Thromb Res 2020;194:101–15. 10.1016/j.thromres.2020.06.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stefely JA, Christensen BB, Gogakos T, et al. Marked factor V activity elevation in severe COVID-19 is associated with venous thromboembolism. Am J Hematol 2020;95:1522–30. 10.1002/ajh.25979 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kaptein FHJ, Stals MAM, Huisman MV. Prophylaxis and treatment of COVID-19 related venous thromboembolism. Postgrad Med 2021;4:1–9. 10.1080/00325481.2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Qanadli SD, Gudmundsson L, Rotzinger DC. Catheter-directed thrombolysis in COVID-19 pneumonia with acute PE: thinking beyond the guidelines. Thromb Res 2020;192:9–11. 10.1016/j.thromres.2020.05.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Santoliquido A, Porfidia A, Nesci A, et al. Incidence of deep vein thrombosis among non-ICU patients hospitalized for COVID-19 despite pharmacological thromboprophylaxis. J Thromb Haemost 2020;18:2358–63. 10.1111/jth.14992 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.The REMAP-CAP, ACTIV-4a, ATTACC Investigators . Therapeutic anticoagulation in critically ill patients with Covid-19 – preliminary report. medRxiv 2021. [Google Scholar]
- 12.ATTACC, ACTIV-4a and REMAP-CAP trials - results of interim analysis (Release date: 28/01/2021. Available: https://static1.squarespace.com/static/5cde3c7d9a69340001d79ffe/t/6013892709de942b53f6e3da/1611893037749/mpRCT+interim+presentation_v21-slides+22+and+23+corrected.pdf [Accessed 26 Mar 2021].
- 13.Talasaz AH, Sadeghipour P, Kakavand H, et al. Recent randomized trials of antithrombotic therapy for patients with COVID-19: JACC state-of-the-art review. J Am Coll Cardiol 2021;77:1903–21. 10.1016/j.jacc.2021.02.035 [DOI] [PMC free article] [PubMed] [Google Scholar]