Abstract
To report outcomes and risk factors of ultrahypofractionated (UHF) radiotherapy for Japanese prostate cancer patients. This multi-institutional retrospective analysis comprised 259 patients with localized prostate cancer from 6 hospitals. A total dose of 35–36 Gy in 4–5 fractions was prescribed for sequential or alternate-day administration. Biochemical failure was defined according to the Phoenix ASTRO consensus. Toxicities were assessed using National Cancer Institute Common Toxicity Criteria version 4. Tumor control and toxicity rates were analyzed by competing risk frames. Median follow-up duration was 32 months (range 22–97 months). 2- and 3-year biochemical control rates were 97.7% and 96.4%, respectively. Initial prostate-specific antigen (p < 0.01) and neoadjuvant androgen deprivation therapy (p < 0.05) were identified as risk factors for biochemical recurrence. 2- and 3-year cumulative ≥ Grade 2 late genitourinary (GU) toxicities were 5.8% and 7.4%, respectively. Corresponding rates of gastrointestinal (GI) toxicities were 3.9% and 4.5%, respectively. Grade 3 rates were lower than 1% for both GU and GI toxicities. No grade 4 or higher toxicities were encountered. Biologically effective dose was identified as a risk factor for ≥ Grade 2 late GU and GI toxicities (p < 0.05). UHF radiotherapy offered effective, safe treatment for Japanese prostate cancer with short-term follow-up. Our result suggest higher prescribed doses are related to higher toxicity rates.
Subject terms: Cancer, Urological cancer
Introduction
Ultrahypofractionated (UHF) radiotherapy is defined as > 5 Gy per fraction and has gradually been recognized as a standard treatment for localized prostate cancer. The biological features of prostate cancer with a low α/β ratio have encouraged widespread adoption of UHF radiotherapy around the world. In addition to these biological advantages, UHF radiotherapy offers benefits in terms of cost effectiveness and patient convenience. In addition, patients treated with UHF radiotherapy reported significantly “less regret” and “less toxicity” than expected compared to patients treated with other radiotherapy techniques1.
The current guidelines conditionally recommend UHF radiotherapy only for low- or intermediate-risk patients2. However, as the potential advantages of UHF radiotherapy over other treatment techniques are gradually confirmed, candidates for this treatment are expected to expand to not only low- and intermediate-risk patients, but also high-risk patients.
Meanwhile, the current state of UHF radiotherapy in Japan is unclear, although several Japanese institutions are likely to have already started UHF radiotherapy. In addition, treatment results for Japanese patients have not yet been reported except in a few papers3–5. We therefore conducted a survey of the current status of UHF radiotherapy in Japan and undertook a multi-institutional retrospective analysis of UHF radiotherapy for Japanese prostate cancer patients. We report herein the outcomes and risk factors of UHF radiotherapy in Japanese prostate cancer patients.
Materials and methods
Patients and treatments
We sent a questionnaire survey to around 160 Japanese institutions that were participants in the Japanese Radiation Oncology Study Group (JROSG) or that were equipped with CyberKnife systems between December 2019 and February 2020. The results showed that at least 10 institutions in Japan currently apply UHF radiotherapy for localized prostate cancer, and more than 1300 patients have already received treatment with UHF radiotherapy. Detailed results of the questionnaire survey are shown in the Supplementary Table S1.
Six of the 10 institutions agreed to participate in further retrospective analysis of patients treated with UHF radiotherapy. The Kitasato University Hospital institutional review board, Shonan Fujisawa Tokusyukai Hospital institutional review board, Toyama CyberKnife Center institutional review board, Saiseikai Yokohamashi Tobu Hospital institutional review board, Tobata Kyoritsu Hospital institutional review board, and National Cancer Center Hospital institutional review board approved the study protocol. Informed consent was obtained in the form of opt-out on the web-site. Those who rejected were excluded. This study was conducted in accordance with the Declaration of Helsinki.
Table 1 shows the background characteristics of patients. More than half of the patients (61%) were categorized as intermediate risk based on the National Comprehensive Cancer Network criteria. More than half of the patients (57%) had received neoadjuvant androgen deprivation therapy (ADT), and 32% had received adjuvant ADT. The mean (± standard deviation) duration of therapy was 6.5 (± 6.3) months for neoadjuvant ADT, and 12.5 (± 12.6) months for adjuvant ADT. The total dose was 35–36 Gy in 4–5 fractions, prescribed as sequential or alternate-day doses. The mean biologically effective dose (BED) based on α/β = 1.5 was 222.8 Gy. Most patients in our study population had no hydrogel spacer. Table 2 shows treatment protocols in the 6 participating institutions. Four of 6 institutions used CyberKnife. Five of 6 institutions used fiducial markers implanted in the prostate.
Table 1.
Patient characteristics.
| Age, years | 72 | (6.8) |
| T | ||
| 1a | 2 | |
| 1b | 1 | |
| 1c | 96 | |
| 2a | 83 | |
| 2b | 22 | |
| 2c | 30 | |
| 3a | 14 | |
| 3b | 11 | |
| Gleason score | ||
| 5 + 5 | 7 | |
| 5 + 4 | 3 | |
| 4 + 5 | 19 | |
| 4 + 4 | 29 | |
| 4 + 3 | 51 | |
| 3 + 4 | 94 | |
| 3 + 3 | 56 | |
| iPSA | 12.1 | (16.9) |
| Risk criteria | ||
| Low | 33 | |
| Intermediate | 158 | |
| High | 68 | |
| Neo ADT | ||
| Yes | 147 | |
| No | 112 | |
| Adj ADT | ||
| Yes | 83 | |
| No | 176 | |
| Total dose | 35.6 | (0.9) |
| Number of fractions | 4.6 | (0.5) |
| BED | 222.8 | (22.5) |
| Hydrogel spacer | ||
| Yes | 31 | |
| No | 228 | |
ADT androgen deprivation therapy; iPSA initial prostate cancer-specific antigen; GS Gleason score; BED biologically effective dose. Values are number or mean (standard deviation).
Table 2.
Treatment protocols.
| Institution | Dose per fraction | Number of fractions | Prescription | Schedule | Target | Treatment system | Fiducial marker | Urethral catheter |
|---|---|---|---|---|---|---|---|---|
| 1 | 7.25 Gy | 5 | D95 | Sequential | CTV = prostate + (SV 1 cm + 3 mm (posterior 1 mm)); PTV = CTV + 2 mm | Cyberknife | Yes | No |
| 2 | 7.25 Gy | 5 | D95 | Sequential |
Low risk: CTV = GTV + 3 mm; PTV = CTV + 2 mm Intermediate risk: CTV = GTV + 3 mm; PTV = CTV + 2 mm |
Cyberknife M6 | Yes | No |
| 3 | 7.25 Gy | 5 | D83 | Alternate-day |
Low risk: CTV = prostate; PTV = CTV + 5 mm (posterior 3 mm) Intermediate risk: CTV = prostate + SV 1 cm; PTV = CTV + 5 mm (posterior 3 mm) High risk: CTV = prostate + SV 1–2 cm; PTV = CTV + 5 mm (posterior 3 mm) |
Cyberknife | Yes | No |
| 4 | 8–9 Gy | 4 | D95 | Sequential (2-day break) | CTV = prostate + SV 1 cm; PTV = CTV + 5 mm (posterior 3 mm) | Tomotherapy | Yes | Yes |
| 5 | 7 Gy | 5 | D98-99 |
Sequential Alternate-day Twice-weekly |
Low risk: CTV = prostate; PTV = CTV + 3 mm Intermediate risk: CTV = prostate + proximal SV; PTV = CTV + 3 mm high Risk: CTV = prostate + SV; PTV = CTV + 3 mm |
Liniac | No | No |
| 6 | 7.25 Gy | 5 | D95 | Alternate-day |
low Risk: CTV = prostate; PTV = CTV + 5 mm (posterior 4 mm) Intermediate risk: CTV = prostate + half of SV; PTV = CTV + 5 mm (posterior 4 mm) |
Cyberknife Liniac |
Yes | No |
CTV clinical target volume, SV seminal vesicle, PTV planning target volume.
Statistical analysis
Statistical analyses were performed using R version 3.5.1 software (R Project for Statistical Computing, Vienna, Austria). Overall survival was calculated using the Kaplan–Meier method. Biochemical failure was defined according to the Phoenix ASTRO consensus (Nadir + 2)6. Genitourinary (GU) and gastrointestinal (GI) toxicities were assessed using the National Cancer Institute Common Toxicity Criteria version 4.
A competing risk analysis (Gray’s test and Fine and Gray’s regression) was used for biochemical control, local recurrence, pelvic lymph-node recurrence, distant metastasis, castration-resistant prostate cancer (CRPC) and cumulative GU and GI toxicity rates. Crude rates of GU and GI toxicities are also reported for comparison with other reports. Age, T stage, Gleason score, initial prostate-specific antigen (PSA), BED, neoadjuvant ADT, and adjuvant ADT were included as variates in univariate analyses. Multivariate models for each endpoint were constructed by including all factors with values of p < 0.20 from univariate analyses. Values of p < 0.05 were considered statistically significant.
Ethics approval and consent to participate
This study was approved by the local institutional review boards.
Consent for publication
Informed consent was obtained in the form of opt-out on the web-site. Those who rejected were excluded.
Results
The median duration of follow-up was 32 months (range 22–97 months). Two- and 3-year overall survival rates were 99.6% (95% CI 0.99–100%) and 99.1% (95% CI 97.9–100%), respectively. No prostate cancer deaths were reported. Two- and 3-year biochemical control rates were 97.7% (95% CI 95.2–99.0%) and 96.4% (95% CI 93.3–98.4), respectively. Corresponding rates of each risk category were as follows: low risk, 100% and 100% (95% CI na); intermediate risk, 97.5% (95% CI 94.0–99.2%) and 96.6% (95% CI 92.7–98.7%); and high risk, 97.1% (95% CI 90.8–99.5%) and 93.7% (95% CI 83.2–98.6%), respectively. Initial PSA and neoadjuvant ADT were detected as risk factors for biochemical recurrence by multivariate analysis (Table 3, Fig. 1).
Table 3.
Uni- and multivariate analyses of biochemical relapse-free rate.
| Risk factor | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p value | HR | 95% CI | p value | |
| Age | 0.96 | (0.88–1.05) | 0.78 | – | ||
| iPSA | ||||||
| < 10 | Ref | 0.00 | Ref | 0.00 | ||
| ≧10 | 10.1 | (2.30–44.40) | 10.70 | (2.69–42.89) | ||
| GS | 1.45 | (0.75–2.81) | 0.51 | – | ||
| T stage | ||||||
| < 3a | Ref | 0.72 | – | |||
| ≧3a | 1.45 | (0.18–11.70) | ||||
| Neoadjuvant ADT | ||||||
| No | Ref | 0.14 | Ref | 0.03 | ||
| Yes | 0.37 | (0.01–1.43) | 0.21 | (0.05–0.85) | ||
| Adjuvant ADT | ||||||
| No | Ref | 0.57 | – | |||
| Yes | 0.63 | (0.13–3.04) | ||||
| BED | 1.02 | (1.00–1.05) | 0.10 | 1.02 | (1.00–1.05) | 0.15 |
iPSA initial prostate cancer-specific antigen, GS Gleason score; BED biologically effective dose; ADT androgen deprivation therapy.
Figure 1.
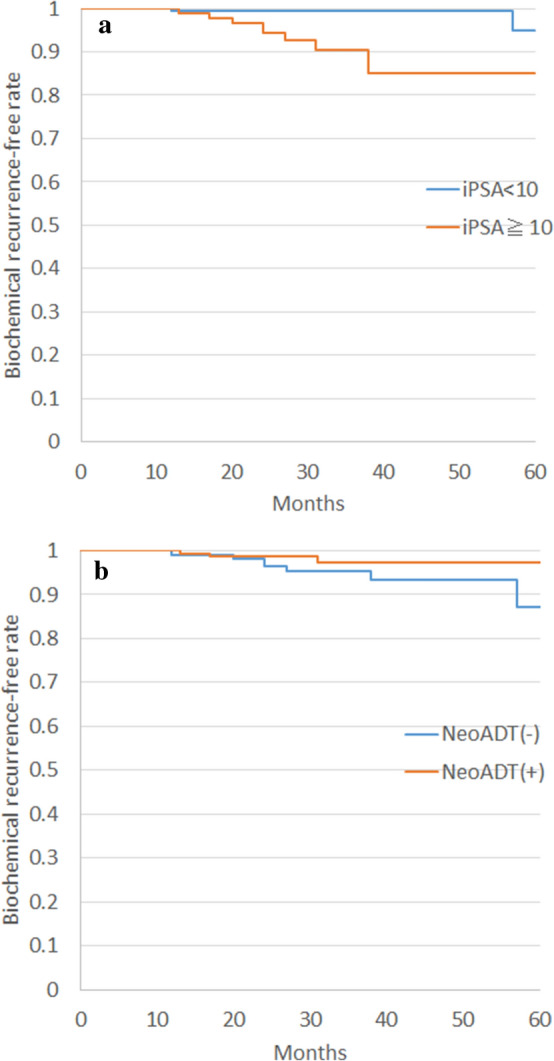
Differences in biochemical recurrence-free rates depend on initial PSA value (a) and neoadjuvant androgen deprivation therapy (b).
Two-year local recurrence, pelvic lymph-node recurrence, and distant metastasis rates were 0.4%, 0.4%, and 1.2%, respectively. Corresponding rates at 3 years were 1.3%, 0.4%, and 1.8%, respectively. Two- and 3-year CRPC rates were 0.4% and 1%, respectively.
Table 4 shows crude rates of acute and late toxicities. Grade 3 toxicity rate was lower than 1%. No grade 4 or higher toxicity was seen during follow-up. The most common acute ≥ Grade 2 GU toxicities were frequency (17/37, 46%), retention (13/37, 35%), and pain (3/37, 4%). Corresponding GI toxicities were diarrhea (7/13, 54%), proctitis (3/13, 23%), and bleeding (3/13, 23%). The most common late > Grade 2 GU toxicities were frequency (8/18, 44%), retention (2/18, 11%), and pain (2/18, 11%). The corresponding GI toxicity was bleeding (10/12, 83%).
Table 4.
Toxicity rates.
| Acute GU | Acute GI | Late GU | Late GI | |||||
|---|---|---|---|---|---|---|---|---|
| Grade 1 | 95 | (36.7%) | 55 | (21.2%) | 71 | (27.4%) | 36 | (13.9%) |
| Grade 2 | 36 | (13.9%) | 13 | (5.0%) | 16 | (6.2%) | 10 | (3.9%) |
| Grade 3 | 1 | (0.4%) | 0 | (0.0%) | 2 | (0.8%) | 2 | (0.8%) |
GU genitourinary. GI gastrointestinal.
2- and 3-year cumulative ≥ Grade 2 late GU toxicity rates were 5.8% and 7.4%, respectively. Corresponding rates of GI toxicities were 3.9% and 4.5%, respectively. BED was detected as a risk factor for ≥ Grade 2 late GU and GI toxicities (Table 5, Fig. 2). No other variables was detected with values of p < 0.20 in univariate analyses.
Table 5.
Univariate analyses of > Grade 2 late toxicity.
| Risk factor | Late GU toxicity | Late GI toxicity | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p value | HR | 95% CI | p value | |
| Age | 0.96 | (0.89–1.03) | 0.24 | 1.02 | (0.93–1.12) | 0.66 |
| iPSA | ||||||
| < 10 | Ref | 0.31 | Ref | 0.55 | ||
| ≧10 | 1.65 | (0.63–4.30) | 1.41 | (0.45–4.40) | ||
| GS | 1.20 | (0.77–1.89) | 0.42 | 1.07 | (0.57–2.00) | 0.84 |
| T stage | ||||||
| < 3a | Ref | 0.65 | Ref | 0.96 | ||
| ≧3a | 0.63 | (0.08–4.72) | 0.95 | (0.13–7.25) | ||
| Neoadjuvant ADT | ||||||
| No | Ref | 0.57 | Ref | 0.71 | ||
| Yes | 1.31 | (0.51–3.39) | 0.80 | (0.25–2.60) | ||
| Adjuvant ADT | ||||||
| No | Ref | 0.82 | Ref | |||
| Yes | 0.89 | (0.32–2.48) | 0.44 | (0.10–2.01) | 0.29 | |
| BED | 1.02 | (1.00–1.04) | 0.03 | 1.03 | (1.00–1.07) | 0.03 |
GU genitourinary, GI gastrointestinal, iPSA initial prostate cancer-specific antigen, GS Gleason score, BED biologically effective dose, ADT androgen deprivation therapy.
Figure 2.
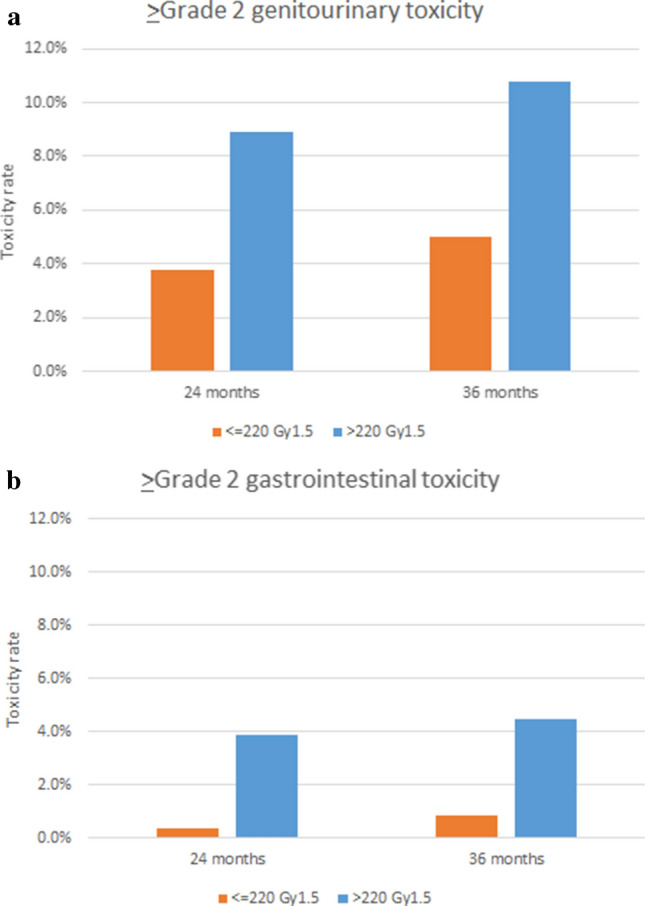
Crude toxicity rates for Grade ≥ 2 genitourinary (a) and gastrointestinal, (b) toxicities depend on biologically equivalent doses (> 220 or ≤ 220 Gy1.5).
Discussion
Our retrospective analysis showed that the UHF radiotherapy is safe and effective for Japanese patients, at least in a short-term follow-up. Considering recently reported long-term7 and large cohort8 outcomes from Western countries, this treatment is also valuable as a treatment option for Japanese prostate cancer patients. Although a cautious approach is warranted until long-term results become available, there are no obstacles to applying UHF radiotherapy for Japanese patients.
Our analysis revealed that prescription doses were significantly related to toxicity rates. The American Society for Radiation Oncology (ASTRO), American Society of Clinical Oncology (ASCO), and American Urological Association (AUA) guidelines recommend prescription doses between 35 Gy and 36.25 Gy in 5 fractions, and doses above 36.25 Gy are not suggested outside the setting of clinical trials due to the risk of late toxicities2. Table 6 shows the results of previous reports of relatively high incidences of ≥ Grade 2 toxicities7,9–16. Although it was not always high-dose prescriptions that led to high toxicity rates in previous studies, prescribed doses seem related to more severe toxicity, as our study suggested. Because our study included patients who participated in dose-escalation trials testing relatively high doses, these patients suffered relatively severe late toxicities compared to patients treated with current standard doses following the guidelines4.
Table 6.
Selected series reported > 5% Grade 2 toxicities.
| Author | Year | Schedule | BED α/β = 1.5 | Median follow-up | Late GU | Late GI |
|---|---|---|---|---|---|---|
| Aluwini | 2013 | 38 Gy/4 fr | 278.7 Gy | 28.35 months |
G2: 15% G3: 5% |
G2: 3% |
| Chen | 2013 | 35–36.5 Gy/5 fr | 198–211.5 Gy | 27.6 months | G2: 31% (2-year) | G2: 1% (2-year) |
| Meier | 2018 | 40 Gy/5 fr | 253.3 Gy | 61 months |
G2: 12% G3: 1.3% |
G2: 2% |
| Katz | 2017 | 35–36.5 Gy/5 fr | 198–211.5 Gy | 108 months |
G2: 9% G3: 3% |
G2: 4% |
| Zimmermann | 2016 | 45 Gy/9 fr | 315 Gy | 83 months |
G2: 27.5% G3: 2.5% G4: 1.3% |
G2: 17.5% G3: 12.5% |
| Kim | 2014 | 45–50 Gy/5 fr | 315–383.3 Gy | 24.5 months | NA |
G4: 2.2% G3: 3.3% G2: 23.1% |
| Bernetich | 2014 | 35–37.5 Gy/5 fr | 198–225 Gy | 38 months |
G2: 14% G3: 2% |
G2: 3% |
| Zhang | 2017 | 38 Gy/4 fr | 278.7 Gy | 35.5 months |
G2: 19.2% G3: 2.6% |
NA |
| Helou | 2017 | 35–40 Gy/5 fr | 198–253.3 Gy | 38 months |
G3: 1.9% G2: 32.6% |
G2: 12.0% G3: 0.8% G4: 1.1% |
BED biologically effective dose, GU genitourinary, GI gastrointestinal.
Koontz et al. published a review analyzing data from pioneering institutions and reported that over 100 Gy-equivalents in 2-Gy fractions might cause higher rates of > Grade 2 toxicities17. Because the schedule of 36 Gy/4 fractions included in our study was equal to 108 Gy-equivalents in 2-Gy fractions and a BED of 226 Gy, our results appear consistent with their suggestion.
From the perspective of toxicity, a lower prescribed dose might be suitable, especially for low- and favorable intermediate-risk patients. However, a lower dose might cause lower tumor control, and the importance of dose escalation is well known in conventional fractionation18,19. In the field of UHF radiotherapy, Zelefsky et al. suggested the importance of dose escalation. Although long-term control rates were not determined, they reported positive biopsy rates of 47.6%, 19.2%, 16.7% and 7.7% after 32.5 Gy, 35 Gy, 37.5 Gy and 40 Gy in 5 fractions, respectively20. These results suggest that unfavorable tumor control rates might be seen in lower-dose groups.
Helou et al. reported that the 3-year PSA level correlated with the prescribed dose in a comparison between 40 Gy (0.27 ng/ml) and 35 Gy (0.64 ng/ml) in 5 fractions. The higher dose of 40 Gy was an independent predictor of a lower 3-year PSA level in their multivariate analysis16. The 3-year PSA value was previously found to offer an early predictor of biochemical failure after high-dose-rate brachytherapy21. The concept of a well-balanced, optimal dose during UHF radiotherapy thus remains contentious and should be explored in future trials.
Meanwhile, periprostatic hydrogel spacers have been approved for use with transperineal injection in Japan since 2017. Current practices in UHF radiotherapy are thus expected to usually be combined with spacer injection, and may decrease severe toxicities even using the same dose levels22.
Regarding risk factors for biochemical recurrence, our results were not surprising from the point of view of experience with conventional external beam radiotherapy (EBRT). The nomogram established by Kattan et al. more than 20 years ago revealed that the initial PSA level was the most powerful indicator of biochemical recurrence23. ADT is well known as another powerful indicator of biochemical recurrence24. Our results thus suggested that UHF radiotherapy shows similar trends to conventional EBRT, although our short follow-up makes the value of these risk factors difficult to confirm.
Because our study was a retrospective analysis, several limitations should be considered. First, as collected items were limited, other factors might correlate with tumor control and toxicity rates. Second, our analysis of toxicities might have been biased due to the lack of information on dose-volume histograms. Third, because various treatment schedules were used at each hospital, results reported in this paper would have varied depending on treatment schedules. Fourth, because no follow-up schedule was defined, the timings of follow-up visits and examinations among hospitals were highly heterogeneous. Tumor control and toxicity rates might thus be relatively ambiguous.
Conclusion
This multi-institutional analysis showed that UHF radiotherapy is effective for Japanese prostate cancer with limited severe toxicity. However, the optimal dose during UHF radiotherapy should be continuously explored, as our results also suggested that higher prescribed doses were related to higher toxicity rates.
Supplementary Information
Acknowledgements
This study was supported in part by the Japanese Radiation Oncology Study Group.
Abbreviations
- UHF
Ultrahypofractionated
- GU
Genitourinary
- GI
Gastrointestinal
- ASTRO
American Society for Radiation Oncology
- JROSG
Japanese Radiation Oncology Study Group
- ADT
Androgen deprivation therapy
- CRPC
Castration-resistant prostate cancer
- PSA
Prostate-specific antigen
- BED
Biologically effective dose
- ASCO
American Society of Clinical Oncology
- AUA
American Urological Association
- EBRT
External beam radiotherapy
Author contributions
H.I., H.T. and H.K. drafted the manuscript. H.I. and K.N. participated in the design of the study. H.I. performed the statistical analysis. K.N. supervised the study. H.I., H.N., M.W., E.M., M.T., H.K., T.E., H.I. and K.I. collected medical information about this study. All authors read and approved the final manuscript.
Funding
This study was supported in part by the Japanese Radiation Oncology Study Group.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing interests
Dr. Ishiyama reports grants from Nihon Medi-physics, Co,Ltd., outside the submitted work. Dr. Inaba reports grants from Elekta K.K., outside the submitted work.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-021-92307-8.
References
- 1.Shaverdian N, Verruttipong D, Wang PC, Kishan AU, Demanes DJ, McCloskey S, et al. Exploring value from the patient’s perspective between modern radiation therapy modalities for localized prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 2017;97(3):516–525. doi: 10.1016/j.ijrobp.2016.11.007. [DOI] [PubMed] [Google Scholar]
- 2.Morgan SC, Hoffman K, Loblaw DA, Buyyounouski MK, Patton C, Barocas D, et al. Hypofractionated radiation therapy for localized prostate cancer: An ASTRO, ASCO, and AUA evidence-based guideline. J. Clin. Oncol. 2018 doi: 10.1016/j.juro.2018.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nakamura R, Sugawara J, Yamaguchi S, Kakuhara H, Kikuchi K, Ariga H. Stereotactic body radiotherapy with a single isocentre for multiple pulmonary metastases. BJR Case Rep. 2020;6(4):20190121. doi: 10.1259/bjrcr.20190121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kainuma T, Kawakami S, Tsumura H, Satoh T, Tabata KI, Iwamura M, et al. A phase I dose-escalation trial of stereotactic body radiotherapy using 4 fractions for patients with localized prostate cancer. Radiat. Oncol. 2019;14(1):158. doi: 10.1186/s13014-019-1369-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kobayashi H, Eriguchi T, Tanaka T, Ogata T, Ishida M, Nakajima Y, et al. Distribution analysis of hydrogel spacer and evaluation of rectal dose reduction in Japanese prostate cancer patients undergoing stereotactic body radiation therapy. Int. J. Clin. Oncol. 2021;26:736. doi: 10.1007/s10147-020-01855-y. [DOI] [PubMed] [Google Scholar]
- 6.Roach M, Hanks G, Thames H, Jr, Schellhammer P, Shipley WU, Sokol GH, et al. Defining biochemical failure following radiotherapy with or without hormonal therapy in men with clinically localized prostate cancer: Recommendations of the RTOG-ASTRO Phoenix Consensus Conference. Int. J. Radiat. Oncol. Biol. Phys. 2006;65(4):965–974. doi: 10.1016/j.ijrobp.2006.04.029. [DOI] [PubMed] [Google Scholar]
- 7.Katz A. Stereotactic body radiotherapy for low-risk prostate cancer: A ten-year analysis. Cureus. 2017;9(9):e1668. doi: 10.7759/cureus.1668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kishan AU, Dang A, Katz AJ, Mantz CA, Collins SP, Aghdam N, et al. Long-term outcomes of stereotactic body radiotherapy for low-risk and intermediate-risk prostate cancer. JAMA Netw. Open. 2019;2(2):e188006. doi: 10.1001/jamanetworkopen.2018.8006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Aluwini S, Beltramo G, Van Rooij P, Boormans J, Kirkels W, Kolkman-Deurloo I-K. P068 stereotactic body radiotherapy with four fractions for low-and intermediate-risk prostate cancer: Acute and late toxicity. Eur. Urol. Suppl. 2013;12(6):156. doi: 10.1016/S1569-9056(13)62393-3. [DOI] [Google Scholar]
- 10.Chen LN, Suy S, Uhm S, Oermann EK, Ju AW, Chen V, et al. Stereotactic body radiation therapy (SBRT) for clinically localized prostate cancer: The Georgetown University experience. Radiat. Oncol. 2013;8:58. doi: 10.1186/1748-717X-8-58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Meier RM, Bloch DA, Cotrutz C, Beckman AC, Henning GT, Woodhouse SA, et al. Multicenter trial of stereotactic body radiation therapy for low-and intermediate-risk prostate cancer: Survival and toxicity endpoints. Int. J. Radiat. Oncol. Biol. Phys. 2018;102(2):296–303. doi: 10.1016/j.ijrobp.2018.05.040. [DOI] [PubMed] [Google Scholar]
- 12.Zimmermann M, Taussky D, Menkarios C, Vigneault É, Beauchemin M-C, Bahary J-P, et al. Prospective phase II trial of once-weekly hypofractionated radiation therapy for low-risk adenocarcinoma of the prostate: Late toxicities and outcomes. Clin. Oncol. 2016;28(6):386–392. doi: 10.1016/j.clon.2015.12.024. [DOI] [PubMed] [Google Scholar]
- 13.Kim DW, Cho LC, Straka C, Christie A, Lotan Y, Pistenmaa D, et al. Predictors of rectal tolerance observed in a dose-escalated phase 1–2 trial of stereotactic body radiation therapy for prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 2014;89(3):509–517. doi: 10.1016/j.ijrobp.2014.03.012. [DOI] [PubMed] [Google Scholar]
- 14.Bernetich M, Oliai C, Lanciano R, Hanlon A, Lamond J, Arrigo S, et al. SBRT for the primary treatment of localized prostate cancer: The effect of gleason score, dose and heterogeneity of intermediate risk on outcome utilizing 2.2014 NCCN risk stratification guidelines. Front. Oncol. 2014;4:312. doi: 10.3389/fonc.2014.00312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhang L, Johnson J, Gottschalk AR, Chang AJ, Hsu IC, Roach M, 3rd, et al. Receiver operating curves and dose-volume analysis of late toxicity with stereotactic body radiation therapy for prostate cancer. Pract. Radiat. Oncol. 2017;7(2):e109–e116. doi: 10.1016/j.prro.2016.07.004. [DOI] [PubMed] [Google Scholar]
- 16.Helou J, D'Alimonte L, Quon H, Deabreu A, Commisso K, Cheung P, et al. Stereotactic ablative radiotherapy in the treatment of low and intermediate risk prostate cancer: Is there an optimal dose? Radiother. Oncol. 2017;123(3):478–482. doi: 10.1016/j.radonc.2017.03.006. [DOI] [PubMed] [Google Scholar]
- 17.Koontz BF, Bossi A, Cozzarini C, Wiegel T, D’Amico A. A systematic review of hypofractionation for primary management of prostate cancer. Eur. Urol. 2015;68(4):683–691. doi: 10.1016/j.eururo.2014.08.009. [DOI] [PubMed] [Google Scholar]
- 18.Viani GA, Stefano EJ, Afonso SL. Higher-than-conventional radiation doses in localized prostate cancer treatment: A meta-analysis of randomized, controlled trials. Int. J. Radiat. Oncol. Biol. Phys. 2009;74(5):1405–1418. doi: 10.1016/j.ijrobp.2008.10.091. [DOI] [PubMed] [Google Scholar]
- 19.Zelefsky MJ, Yamada Y, Fuks Z, Zhang Z, Hunt M, Cahlon O, et al. Long-term results of conformal radiotherapy for prostate cancer: Impact of dose escalation on biochemical tumor control and distant metastases-free survival outcomes. Int. J. Radiat. Oncol. Biol. Phys. 2008;71(4):1028–1033. doi: 10.1016/j.ijrobp.2007.11.066. [DOI] [PubMed] [Google Scholar]
- 20.Zelefsky MJ, Kollmeier M, McBride S, Varghese M, Mychalczak B, Gewanter R, et al. 5-year outcomes of a phase I dose escalation study using stereotactic body radiosurgery for patients with low and intermediate risk prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 2019;104:42. doi: 10.1016/j.ijrobp.2018.12.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Helou J, D'Alimonte L, Loblaw A, Chung H, Cheung P, Szumacher E, et al. High dose-rate brachytherapy boost for intermediate risk prostate cancer: Long-term outcomes of two different treatment schedules and early biochemical predictors of success. Radiother. Oncol. 2015;115(1):84–89. doi: 10.1016/j.radonc.2015.02.023. [DOI] [PubMed] [Google Scholar]
- 22.Karsh LI, Gross ET, Pieczonka CM, Aliotta PJ, Skomra CJ, Ponsky LE, et al. Absorbable hydrogel spacer use in prostate radiotherapy: A comprehensive review of phase 3 clinical trial published data. Urology. 2018;115:39–44. doi: 10.1016/j.urology.2017.11.016. [DOI] [PubMed] [Google Scholar]
- 23.Kattan MW, Zelefsky MJ, Kupelian PA, Scardino PT, Fuks Z, Leibel SA. Pretreatment nomogram for predicting the outcome of three-dimensional conformal radiotherapy in prostate cancer. J. Clin. Oncol. 2000;18(19):3352–3359. doi: 10.1200/JCO.2000.18.19.3352. [DOI] [PubMed] [Google Scholar]
- 24.Bolla M, Collette L, Blank L, Warde P, Dubois JB, Mirimanoff RO, et al. Long-term results with immediate androgen suppression and external irradiation in patients with locally advanced prostate cancer (an EORTC study): A phase III randomised trial. Lancet. 2002;360(9327):103–106. doi: 10.1016/S0140-6736(02)09408-4. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.


