Abstract
Electrophysiological studies show that nicotine enhances neural responses to characteristic frequency stimuli. Previous behavioral studies partially corroborate these findings in young adults, showing that nicotine selectively enhances auditory processing in difficult listening conditions. The present work extended previous work to include both young and older adults and assessed the nicotine effect on sound frequency and intensity discrimination. Hypotheses were that nicotine improves auditory performance and that the degree of improvement is inversely proportional to baseline performance. Young (19–23 years old) normal-hearing nonsmokers and elderly (61–80) nonsmokers with normal hearing between 500 and 2000 Hz received nicotine gum (6 mg) or placebo gum in a single-blind, randomized crossover design. Participants performed three experiments (frequency discrimination, frequency modulation identification, and intensity discrimination) before and after treatment. The perceptual differences were analyzed between pre- and post-treatment, as well as between post-treatment nicotine and placebo conditions as a function of pre-treatment baseline performance. Compared to pre-treatment performance, nicotine significantly improved frequency discrimination. Compared to placebo, nicotine significantly improved performance for intensity discrimination, and the improvement was more pronounced in the elderly with lower baseline performance. Nicotine had no effect on frequency modulation identification. Nicotine effects are task-dependent, reflecting possible interplays of subjects, tasks and neural mechanisms.
Subject terms: Neuroscience, Physiology, Psychology, Medical research
Introduction
Nicotine, an exogenous agonist for nicotinic acetylcholine receptors, has been shown to improve cognitive functions such as attention, learning, and memory for healthy young adults1,2, healthy older adults3, and those with dementia4. In the mouse auditory cortex, systemic nicotine sharpens receptive fields at characteristic frequencies while suppressing spectrally distant, non-characteristic frequency responses5–7. Conversely, mice lacking β2-containing nicotinic acetylcholine receptors, the predominant receptor subtype in forebrain, exhibit impaired auditory discrimination8. These results suggest that nicotine aids with auditory gating in order to facilitate processing of relevant sounds while filtering out irrelevant sounds9. Human electrophysiological studies did not find consistent evidence for the stimulus-filter model in auditory processing10,11. Human behavioral studies in younger adults found that nicotinic effects depend upon task difficulty, with a greater benefit in more difficult conditions, such as tone-in-noise detection and auditory selective attention tasks11,12.
It is possible that older individuals may derive a greater benefit from nicotine treatment compared to their younger counterparts because older adults often have difficulty with speech understanding even with little to no hearing loss13,14. This difficulty, which cannot be overcome by traditional hearing aids or cochlear implants, is related to suprathreshold impairments in both peripheral and central processing15–18. To the extent that these behavioral impairments reflect the age-related changes in nicotinic acetylcholine receptor signaling19–21, nicotine treatment may improve auditory performance in the elderly.
The present study selected two frequency tasks that rely on information processing over a relatively broad frequency region22 and one intensity task that relies on information processing at the characteristic frequency23. Both young and elderly participants, all healthy nonsmokers, were evaluated for these three tasks before and after treatment with either nicotine or placebo. The primary hypothesis was that, relative to placebo, nicotine treatment would improve auditory performance. The secondary hypothesis was that individuals with lower pre-treatment baseline performance would obtain a greater benefit from nicotine treatment24–26.
Materials and methods
Subjects
Twenty individuals participated in the study: 10 young adults (age range = 19–23, M ± SD = 21 ± 4 years, 5 females) and 10 elderly adults (age range = 61–80, M = 69 ± 6, 5 females). Participants gave written informed consent approved by the University of California Irvine’s Institutional Review Board and were monetarily compensated. The methods were in accordance with principles set forth in the Belmont Report and Declaration of Helsinki. Participants were screened prior to the start of the study to ensure no known severe hearing dysfunction, medical, or mental health illnesses. Participants were also given hearing tests to screen for hearing loss. Young adult participants had normal hearing with thresholds ≤ 20 dB HL (Hearing Level) at octave frequencies between 0.125 and 8 kHz. Except for one who had mild hearing of 30–35 dB HL at 125 and 250 Hz, all elderly participants had normal hearing (≤ 20 dB HL) at octave frequencies between 0.125 and 2 kHz. On average, the elderly participants had mild-to-moderate hearing loss at 4 kHz (33 ± 21 dB HL) and 8 kHz (45 ± 24 dB HL). To minimize the influence of hearing loss, all three tasks had stimulus frequencies below 2 kHz (see “Experimental protocol” below).
To ensure little to no nicotine dependence from use or exposure, participants were required to have a score of 0–2 out of 10 on the Fagerstrӧm index of smoking dependency27,28. Eighteen participants were non- or social-smokers, defined as smoking no more than 100 cigarettes in their lifetime and none in the past year24. Two subjects reported quitting smoking more than 20 years ago. To avoid chemical interactions, participants were instructed to abstain from alcohol consumption for 24 h and food consumption for ≥ 1 h prior to testing. To avoid caffeine withdrawal in regular caffeine consumers, ½ cup of a caffeine-containing beverage was permitted ≥ 1 h prior to testing29.
Experimental protocol
Three tasks were selected for their lower performance in older than younger adults, including frequency and intensity discrimination17 and frequency modulation identification18. The previously described experimental protocols were closely followed in the present study. Briefly, the frequency discrimination experiment measured the just-noticeable-difference in frequency for a 400-ms, 500-Hz pure tone presented at 55 dB SPL. The intensity discrimination experiment measured the just-noticeable-difference in loudness for a 400-ms, 500-Hz pure tone presented at 55 dB SPL. The just-noticeable-difference was obtained by a three-interval, two-alternative, forced-choice adaptive procedure using a two-down, one-up rule to yield 71% correct performance30. The just-noticeable-difference in Hz was logarithmically transformed to conform to a normal distribution.
The frequency modulation identification experiment measured the signal-to-noise ratio required to identify an up-down or down-up frequency modulation “signal” in the presence of a ‘noise”18. The 400-ms signal consisted of harmonics with a fundamental frequency of 189 Hz and a single spectral peak or formant at 1000 Hz. The formant frequency was dynamically modulated by a 5-Hz triangular wave with a 55% modulation swing. The 800-ms noise also consisted of similarly-modulated harmonics but with a different fundamental frequency of 107 Hz. The signal was presented at the temporal center of the noise. The signal level was constant at 55 dB SPL, while the noise was adaptively varied. The participant had to report whether the modulation was up-down or down-up in a single-interval, two-alternative, forced-choice task. A three-down, one-up rule estimated the signal-to-noise ratio, at which the participant correctly identified 79% of the frequency modulated signal31.
All experiments took place in a double-walled, sound-attenuated booth. The 55-dB SPL was chosen because previous studies17 found that the performance difference between young and elderly listeners is greater at lower than higher levels. Given that the present subjects had a threshold of 10–15 dB HL (= 25–30 dB SPL) at 500 and 1000 Hz, 55 dB SPL was a reasonable choice, presenting the stimuli at 25–30 dB SL, which sounded relatively soft but clearly audible. Stimuli were presented binaurally via a sound card (Creative Labs E-MU 0404 USB digital audio system, Creative Technology Ltd., Singapore, 16-bit, 44.1 kHz), through circumaural headphones (Sennheiser HAD-200, Wedemark, Germany). The reported result for each participant was the arithmetic mean of the estimate obtained in three to five runs. In all three tasks, lower values reflected better performance.
Study design
All procedures leading up to experiments, including method of drug delivery and time between sessions to allow for drug clearance followed those specified by Pham et al.12. Briefly, six mg of nicotine was delivered in the form of two pieces of mint-flavored polacrilex gum (4 mg and 2 mg; Nicorette, Johnson & Johnson, Inc). Two mint-flavored gum (Eclipse), resembling the nicotine gum in size, shape, color, and texture, served as the placebo. Furthermore, subjects wore a blindfold during both administrations to mask any potential visual differences between placebo and nicotine gums; a drop of Tabasco sauce was added to each gum piece to disguise taste bias32. Test occurred between 8:00 am and 6:00 pm and took place at a consistent time across sessions to avoid confounding arousal and attention effects. All three experiments were completed in two sessions, in which a treatment (nicotine or placebo) was given while pre- and post-treatment data were collected. Audiograms were measured prior to pre-treatment testing in the first session. Afterwards, participants received either nicotine or placebo gum in a randomized design. The protocol was repeated with the alternate treatment (nicotine or placebo) in the second session adhering to a single-blind intra-subject design. Treatment sessions were separated by ≥ 48 h to allow for treatment clearance. Timing was carefully controlled to assure that nicotine plasma concentration at this dosage reached and maintained peak levels for the duration of the three experiments12,33.
Pulse oximetry, mood changes, and side effects
Neither nicotine nor placebo treatment significantly changed blood oxygen saturation in either group (mean ± SD: young, pre-nicotine 97.9 ± 0.3%; post-nicotine 98.2 ± 0.6; pre-placebo 97.9 ± 0.9; post-placebo 98.1 ± 1.1; elderly, pre-nicotine 96.0 ± 1.8; post-nicotine 96.8 ± 1.5; pre-placebo 97.0 ± 1.8; post-placebo 97.0 ± 1.0). Nicotine treatment did not significantly change pulse rate in either group (young, pre-nicotine 71.0 ± 4.5/min; post-nicotine 73.9 ± 8.1; elderly, pre-nicotine 71.0 ± 8.2; post-nicotine 70.6 ± 6.4). Placebo treatment did not significantly alter pulse rate for the young (pre-placebo 70.5 ± 8.2; post-placebo 68.8 ± 8.5) but significantly decreased pulse rate for the elderly (pre-placebo 72.2 ± 8.2; post-placebo 66.6 ± 8.0, p < 0.01). The reason for the decreased pulse rate was not clear.
Participants also provided subjective pre- and post-treatment ratings using a 9-category mood profile, where responses for each category were binary (e.g., tense/relaxed) and using a 5-point side effects scale, where 1 corresponded to no symptoms and 5 corresponded to severe symptoms such as jittery, headache, nausea, or vomiting10. Ratings were averaged across the three experiments. No significant pre- versus post-treatment change in mood was found for nicotine or placebo treatment in either group (both p > 0.05). No post-treatment difference between placebo and nicotine was observed in either group for ratings of side effects (both p > 0.05).
Data analysis
Mixed 2 × 2 × 2 ANOVA was used to examine the effects of age (young vs. elderly), treatment (nicotine vs. placebo), and time (pre vs post), with age being a between-subjects factor, and treatment and time being within-subjects factors. For any significant factor or interactions, effect sizes were reported as eta squared or η2 (small effect: ~ 0.01, medium effect: ~ 0.06, large effect: ~ 0.14). To test the primary hypothesis that nicotine improves auditory performance, two-tailed, two-sample t-tests were used to test if combined (i.e., young and elderly) post-nicotine minus placebo scores were significantly different from zero, and effect sizes were determined with Cohen’s d (small effect: ~ 0.2, medium effect: ~ 0.5, large effect: ~ 0.8). To test the secondary hypothesis that participants with lower baseline performance would benefit more from nicotine treatment, a linear regression was conducted between the nicotine-placebo post-treatment difference and the baseline performance for each of the three auditory tasks. Baseline performance was the average of the two sets of pre-treatment data from nicotine and placebo conditions. Justification for combining pre-treatment data was performed via a Kolmogorov–Smirnov test, revealing that the combined data follow a normal distribution for each experiment (frequency discrimination, p = 0.34; frequency modulation identification, p = 0.83; intensity discrimination, p = 0.83).
Results
Task-dependent nicotine effects
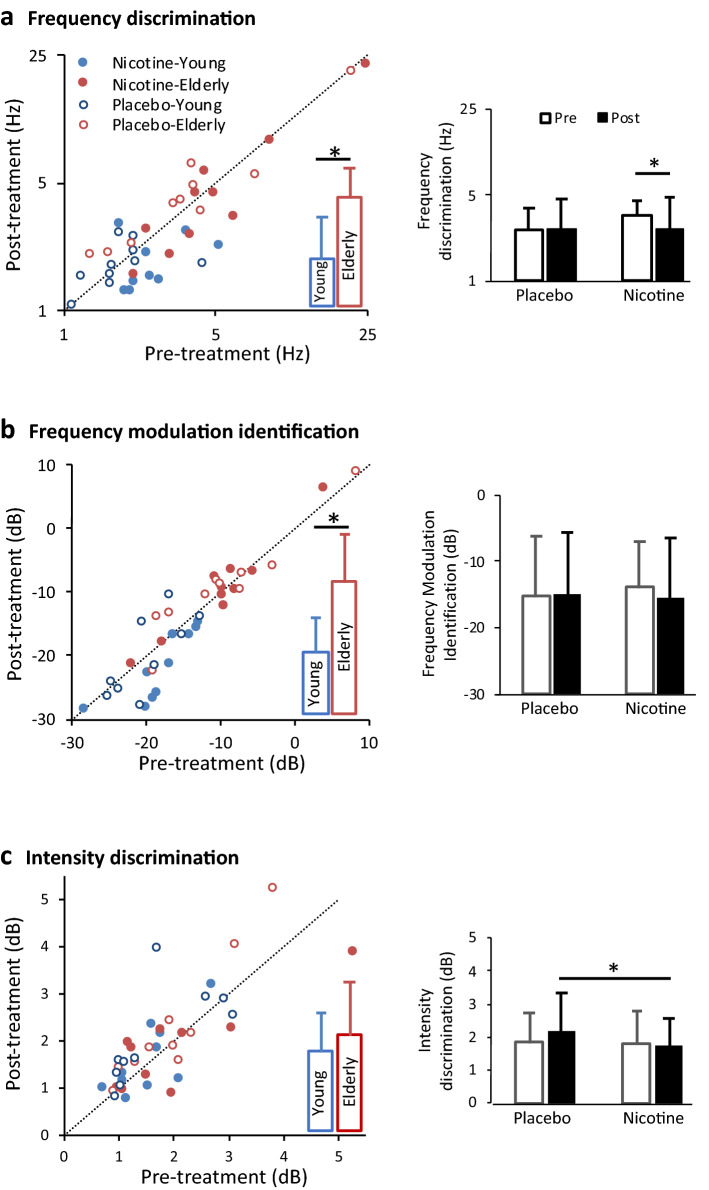
Figure 1 contrasts pre- versus post-treatment results from the three tasks. Figure 1a (left panel) shows frequency discrimination results from both nicotine (filled circles) and placebo (open circles) conditions for young (blue) and elderly adults (red). Data points below the diagonal line indicate improved post-treatment performance, whereas those above indicate worsened performance. Overall, frequency discrimination was significantly worse (i.e., greater) for the elderly (red bar) than younger (blue bar) individuals (see insert in the left panel: 4.2 ± 2.0 Hz vs. 1.9 ± 1.5 Hz; F(1,18) = 10.66, p = 0.004, η2 = 0.37). This age-related difference replicates the previous result17. Within-subjects comparisons (right panel) found a borderline significant effect for both treatment [F(1,18) = 4.12, p = 0.057] and time [F(1,18) = 4.18, p = 0.056], but a significant interaction between the two factors [F(1,18) = 13.22, p = 0.002, η2 = 0.78]. The significant interaction was that nicotine significantly improved frequency discrimination by 24% over the pre-treatment baseline (3.4 ± 2.0 Hz vs. 2.6 ± 2.1 Hz, open bar vs. filled bar above “Nicotine”). There was no significant three-way interaction.
Figure 1.
Post- versus pre-treatment performance for three auditory tasks. (a) Frequency discrimination thresholds at 500 Hz. The left panel shows individual results from both nicotine (filled circles) and placebo (open circles) conditions for young (blue) and elderly adults (red). Data points below the diagonal line indicate improved post-treatment performance, whereas points above denote worsened performance. The insert in the left panel shows between-subjects comparisons between young (blue bar) and elderly (red bar) adults. The right panel shows 2 × 2 within-subjects comparisons between the treatment (nicotine vs. placebo on the x-axis) and time (pre- vs. post-treatment = open vs. filled bars) factors. Error bars represent one standard deviation. The asterisk above the line indicates a significant difference between the two conditions. (b) Frequency modulation identification results are displayed following the same convention as (a). (c) Intensity discrimination results are displayed following the same convention as (a).
Figure 1b uses the same format to display frequency modulation identification results. Similar to the previous result18, younger individuals outperformed elderly individuals by about 10 dB (− 19.5 ± 5.1 dB vs. − 9.7 ± 7.4 dB; F(1,18) = 15.77, p = 0.001, η2 = 0.96). Within-subjects comparisons found no significant effect for either treatment or time, nor was there any significant interaction between the two factors [F(1,18) < 3.29, p > 0.09]. The three-way interaction was not significant.
Figure 1c displays intensity discrimination results. Different from the previous significant result17, the present study found no difference between the elderly and younger individuals [F(1,18) = 0.71, p = 0.41]. Within-subjects comparisons found a significant treatment effect [F(1,18) = 5.37, p = 0.03, η2 = 0.23] but no time effect [F(1,18) = 1.69, p = 0.21] nor any significant interaction [F(1,18) = 2.60, p = 0.12]. Different from the frequency discrimination result, the significant nicotine effect here was due to the 29% worsened post-treatment performance in the placebo condition (2.2 ± 1.2 dB vs. 1.7 ± 0.8 dB, the two filled bars in the right panel). There was no significant three-way interaction.
Post-treatment performance as a function of baseline
Figure 2 shows the individual differences between nicotine and placebo post-treatment performance as a function of pre-treatment baseline performance (blue circles for young individuals; red circles for elderly individuals). For frequency discrimination (Fig. 2a), there was no significant difference between nicotine and placebo post-treatment performance (the horizontal dashed line represents the mean difference: M = − 0.01 ± 0.02 logHz, t(19) = 0.06, p = 0.95). There was no correlation between post-treatment difference and baseline performance (r2 = 0.13, p = 0.11).
Figure 2.
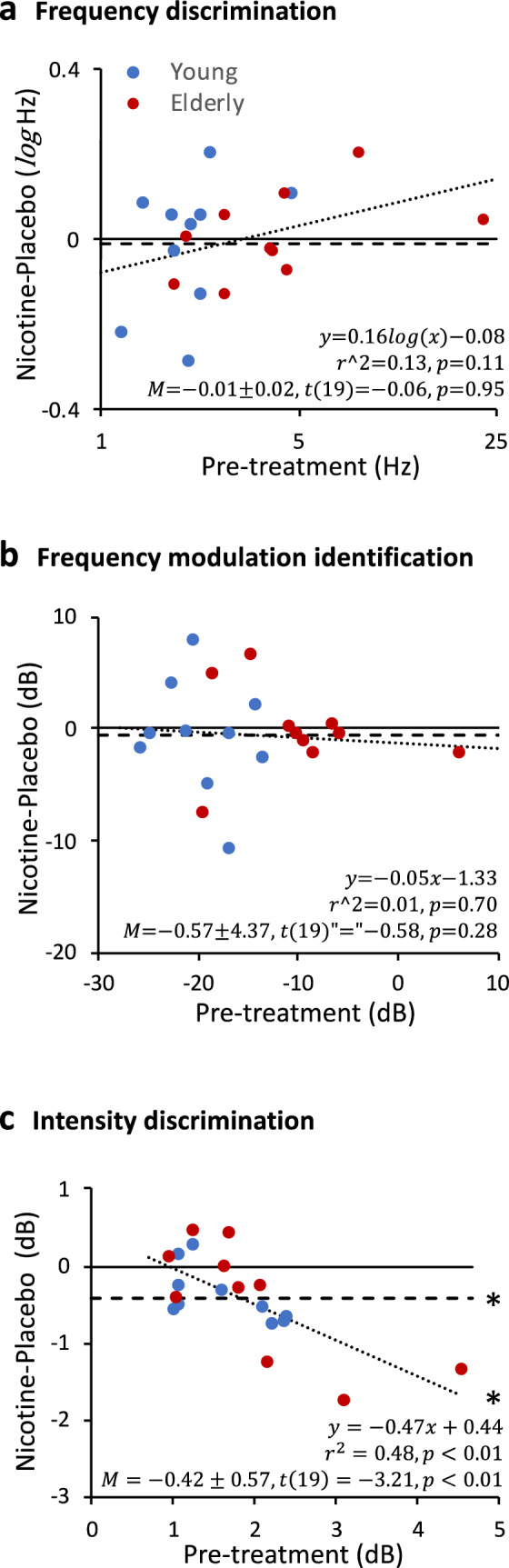
Post-treatment nicotine and placebo differences as a function of pre-treatment baseline performance in three experiments. (a) Frequency discrimination for young (blue circles) and elderly (red circles) adults. The baseline-dependent regression line is represented by the dotted line, with the linear regression equation being shown by text in the bottom (top line), r2 and p-value (second from top). The nicotine effect in terms of the mean difference between post-nicotine and post-placebo performance is represented by the dashed horizontal line, with the mean difference, standard deviation and the two-sample t-test result being shown by text in the bottom. The solid horizontal line crossing zero on the y-axis indicates no difference between post-nicotine and post-placebo performance. (b) Frequency modulation identification results are displayed following the same convention as (a). (c) Intensity discrimination results are displayed following the same convention as (a). The asterisk next to the dashed line indicates a significant difference from the baseline (solid line) and that next to the dotted indicates significant regression.
Figure 2b shows the frequency modulation identification result. Similar to frequency discrimination, there was no significant difference between nicotine and placebo post-treatment performance [M = − 0.57 ± 4.37 dB, t(19) = − 0.58, p 0.05], nor any significant correlation between the post-treatment difference and the pre-treatment baseline (r2 = 0.01, p = 0.70).
In contrast to the results from the two frequency tasks, Fig. 2c shows that intensity discrimination produced a significant nicotine effect. First, nicotine improved intensity discrimination performance over placebo (the horizontal dashed line: M = − 0.42 ± 0.57 dB, t(19) = − 3.21, p < 0.01). Second, greater improvement with nicotine treatment was correlated with worse baseline performance (the downward dotted line represents the linear regression: r2 = 0.48, p < 0.01). Note that the three subjects whose performance was improved the most were all from the elderly group (the three red bottom circles).
Discussion
The present study tested two hypotheses in three auditory tasks: (1) nicotine improves auditory processing, and (2) the amount of improvement is greater in those with poorer baseline performance. The results were task-dependent. First, frequency and intensity discrimination results were consistent with the first hypothesis, but for different reasons. The nicotine effect in frequency discrimination came from improved post-nicotine performance over the pre-nicotine performance (Fig. 1a, right panel), whereas that in intensity discrimination came from improved post-nicotine performance over the post-placebo performance (Fig. 1c, right panel and Fig. 2c). Second, only the intensity discrimination result supported the second hypothesis, with the greatest improvement from those with poorer baseline performance, especially in some elderly individuals (Fig. 2c). Third, the frequency modulation identification result did not support either of these two hypotheses. We note, however, the data were trending towards the right direction, with placebo producing worse post-treatment performance and nicotine producing better post-treatment performance (Figs. 1b, 2b). The present study was underpowered to reach the level of statistical significance.
Nicotine effects on human auditory performance
Different from relatively large and consistent nicotine effects in animal studies7,34, human studies, due to inherent limitations in sample selection and experimental control, often produce small and inconsistent results. For example, Harkrider and Hedrick10 found a significant nicotine effect on speech perception, whereas Knott et al.11 did not. Another example is the inconsistent effect of baseline performance on nicotine-induced improvement. Knott et al.24,25 found that nicotine’s effects were baseline-dependent, but Pham et al.12 did not observe such a dependence. Moreover, the nicotine effect may depend on the task. Pham et al.12 found that nicotine improved performance for auditory selective attention and tone-in-noise detection but not for easier temporal or spectral resolution tasks. Similar to the previous studies, the present study found a significant nicotine effect on intensity and frequency discrimination but not on frequency modulation identification (see “Limitations and Future Directions” below).
Physiological mechanisms
While age-related impairments have been observed in nicotinic acetylcholine receptor signaling along the auditory pathway19–21, it is not clear how these impairments are related to the observed nicotine effects on auditory processing35–38. A particularly difficult issue to untangle is the interaction between aging and hearing loss, which may differentially influence the present auditory tasks. Frequency discrimination, even at 500 or 1000 Hz as in the present study, still relies on information from high frequencies22. The presence of high-frequency hearing loss (30–45 dB HL, see the Subjects section) in the elderly participants may not only produce poorer performance compared to younger participants in the two frequency tasks, but also potentially confound any age-related nicotine effect. On the other hand, intensity discrimination relies on changes in neural activities at the local frequency channel23. The presence of high-frequency hearing loss in the elderly participants had no effect on intensity discrimination at 500 Hz, thereby revealing a significant nicotine effect. Interestingly, similar results for frequency and intensity discrimination were also observed in individuals with tinnitus39, further suggesting the importance of considering both subject and task variables in investigating the effects of nicotine on auditory processing.
Limitations and future directions
One limitation of the present study involves testing only a small number of participants, limiting its statistical power for uncovering potential effects of age and sex11,40. For example, the small sample size was likely responsible for a lack of a significant difference in intensity discrimination between the young and elderly adults in the present study. A second limitation is that a single dose of orally-administered nicotine may not be sufficient to effectively change perception, reflecting variability of absorption rate across participants and their different pharmacodynamic thresholds33. A third limitation is the choice of tasks, which were all relatively simple auditory processing tasks. Future work could improve upon these present limitations by increasing sample size, by using alternative administration routes (e.g., transdermal patch), multiple dosing levels and monitoring individual plasma level data41, and by targeting central auditory processing involving both temporal and spectral variance cues42.
Conclusions
The present study evaluated acute effects of oral nicotine treatment on three auditory tasks in young adult and elderly, healthy, non-smoking individuals. All had normal hearing within the frequency range of the stimuli presented for the three tasks. Compared to pre-treatment performance, nicotine improved frequency discrimination. Compared to placebo, nicotine produced no overall effects on the two frequency related tasks, but significantly improved intensity discrimination, with more improvement obtained for those who had lower baseline performance. The present results support the hypothesis that nicotine enhances auditory processing, but this enhancement is task-dependent.
Acknowledgements
The authors thank Sahara George and Phillip Tran for technical assistance. This research was partially supported by the National Institutes of Health (1R01-AG067073 and 2T32-DC010775).
Author contributions
S.S., M.K. and F.G.Z. wrote the main manuscript text. All authors designed the experiments and reviewed the manuscript.
Competing interests
FGZ owns stock in Axonics, DiaNavi, Nurotron, Syntiant, Velox and Xense. The other authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Levin ED, et al. Transdermal nicotine effects on attention. Psychopharmacology. 1998;140:135–141. doi: 10.1007/s002130050750. [DOI] [PubMed] [Google Scholar]
- 2.Levin ED, McClernon FJ, Rezvani AH. Nicotinic effects on cognitive function: Behavioral characterization, pharmacological specification, and anatomic localization. Psychopharmacology. 2006;184:523–539. doi: 10.1007/s00213-005-0164-7. [DOI] [PubMed] [Google Scholar]
- 3.Min S, Moon IW, Ko R, Shin H. Effects of transdermal nicotine on attention and memory in healthy elderly non-smokers. Psychopharmacology. 2001;159:83–88. doi: 10.1007/s002130100899. [DOI] [PubMed] [Google Scholar]
- 4.White HK, Levin ED. Four-week nicotine skin patch treatment effects on cognitive performance in Alzheimer’s disease. Psychopharmacology. 1999;143:158–165. doi: 10.1007/s002130050931. [DOI] [PubMed] [Google Scholar]
- 5.Kawai HD, Kang H-A, Metherate R. Heightened nicotinic regulation of auditory cortex during adolescence. J. Neurosci. 2011;31:14367–14377. doi: 10.1523/JNEUROSCI.1705-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Intskirveli I, Metherate R. Nicotinic neuromodulation in auditory cortex requires MAPK activation in thalamocortical and intracortical circuits. J. Neurophysiol. 2012;107:2782–2793. doi: 10.1152/jn.01129.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Askew, C., Intskirveli, I. & Metherate, R. Systemic nicotine increases gain and narrows receptive fields in A1 via integrated cortical and subcortical actions. Eneuro4, ENEURO.0192–17.2017 (2017). [DOI] [PMC free article] [PubMed]
- 8.Horst NK, et al. Impaired auditory discrimination learning following perinatal nicotine exposure or β2 nicotinic acetylcholine receptor subunit deletion. Behav. Brain Res. 2012;23:170–180. doi: 10.1016/j.bbr.2012.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kassel JD. Smoking and attention: A review and reformulation of the stimulus-filter hypothesis. Clin. Psychol. Rev. 1997;17:451–478. doi: 10.1016/S0272-7358(97)00032-9. [DOI] [PubMed] [Google Scholar]
- 10.Harkrider AW, Hedrick MS. Acute effect of nicotine on auditory gating in smokers and non-smokers. Hear. Res. 2005;202:114–128. doi: 10.1016/j.heares.2004.11.009. [DOI] [PubMed] [Google Scholar]
- 11.Knott VJ, et al. Effects of acute nicotine on event-related potential and performance indices of auditory distraction in nonsmokers. Nicotine Tob. Res. 2009;11:519–530. doi: 10.1093/ntr/ntp044. [DOI] [PubMed] [Google Scholar]
- 12.Pham CQ, Kapolowicz MR, Metherate R, Zeng FG. Nicotine enhances auditory processing in healthy and normal-hearing young adult nonsmokers. Psychopharmacology. 2020;237:833–840. doi: 10.1007/s00213-019-05421-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dubno JR, Lee F, Matthews LJ, Mills JH. Age-related and gender-related changes in monaural speech recognition. J. Speech Lang. Hear. Res. 1997;40:444–452. doi: 10.1044/jslhr.4002.444. [DOI] [PubMed] [Google Scholar]
- 14.Helfer KS, Freyman RL. Aging and speech-on-speech masking. Ear Hear. 2008;29:87–98. doi: 10.1097/AUD.0b013e31815d638b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Humes LE, et al. Central presbycusis: A review and evaluation of the evidence. J. Acad. Audiol. 2012;23:635–666. doi: 10.3766/jaaa.23.8.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Schoof T, Rosen S. The role of auditory and cognitive factors in understanding speech in noise by normal-hearing older listeners. Front. Aging Neurosci. 2014;6:1–14. doi: 10.3389/fnagi.2014.00307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.He N, Dubno JR, Mills JH. Frequency and intensity discrimination measured in a maximum-likelihood procedure from young and aged normal-hearing subjects. J. Acoust. Soc. Am. 1998;103:553–565. doi: 10.1121/1.421127. [DOI] [PubMed] [Google Scholar]
- 18.Divenyi P. Decreased ability in the segregation of dynamically changing vowel-analog streams: A factor in the age-related cocktail-party deficit? Front. Neurosci. 2014;8:1–15. doi: 10.3389/fnins.2014.00144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tang X, et al. Age-related hearing loss: GABA, nicotinic acetylcholine and NMDA receptor expression changes in spiral ganglion neurons of the mouse. Neuroscience. 2014;14:184–193. doi: 10.1016/j.neuroscience.2013.11.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sottile SY, Ling L, Cox BC, Caspary DM. Impact of ageing on postsynaptic neuronal nicotinic neurotransmission in auditory thalamus. J. Physiol. 2017;595:5375–5385. doi: 10.1113/JP274467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ghimire M, Cai R, Ling L, Hackett TA, Caspary DM. Nicotinic receptor subunit distribution in auditory cortex: Impact of aging on receptor number and function. J. Neurosci. 2020;40:5724–5739. doi: 10.1523/JNEUROSCI.0093-20.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zurek PM, Formby C. Frequency-discrimination ability of hearing-impaired listeners. J. Speech Lang. Hear. Res. 1981;24:108–112. doi: 10.1044/jshr.2401.108. [DOI] [PubMed] [Google Scholar]
- 23.Viemeister NF. Auditory intensity discrimination at high frequencies in the presence of noise. Science (80-) 1983;221:1206–1208. doi: 10.1126/science.6612337. [DOI] [PubMed] [Google Scholar]
- 24.Knott V, et al. Baseline-dependent modulating effects of nicotine on voluntary and involuntary attention measured with brain event-related P3 potentials. Pharmacol. Biochem. Behav. 2014;122:107–117. doi: 10.1016/j.pbb.2014.03.020. [DOI] [PubMed] [Google Scholar]
- 25.Knott V, et al. Modulation of auditory deviance detection by acute nicotine is baseline and deviant in healthy nonsmokers: a mismatch negativity study. Hum. Psychopharmacol. 2014;29:446–458. doi: 10.1002/hup.2418. [DOI] [PubMed] [Google Scholar]
- 26.Newhouse PA, Potter A, Singh A. Effects of nicotinic stimulation on cognitive performance. Curr. Opin. Pharmacol. 2004;4:36–46. doi: 10.1016/j.coph.2003.11.001. [DOI] [PubMed] [Google Scholar]
- 27.Bramer SL, Kallungal BA. Clinical considerations in study designs that use cotinine as a biomarker. Biomarkers. 2003;8:187–203. doi: 10.1080/13547500310012545. [DOI] [PubMed] [Google Scholar]
- 28.Heatherton TF, Kozlowski LT, Frecker RC, Fagerström KO. The Fagerström test for nicotine dependence: A revision of the Fagerström tolerance questionnaire. Br. J. Addict. 1991;86:1119–1127. doi: 10.1111/j.1360-0443.1991.tb01879.x. [DOI] [PubMed] [Google Scholar]
- 29.Lawrence NS, Ross TJ, Stein EA. Cognitive mechanisms of nicotine on visual attention. Neuron. 2002;36:539–548. doi: 10.1016/S0896-6273(02)01004-8. [DOI] [PubMed] [Google Scholar]
- 30.Levitt H. Transformed up-down methods in psychoacoustics. J. Acoust. Soc. Am. 1971;49:467–477. doi: 10.1121/1.1912375. [DOI] [PubMed] [Google Scholar]
- 31.Levitt H. Transformed up-down methods in psychoacoustics. J. Acoust. Soc. Am. 2005;49:467–477. doi: 10.1121/1.1912375. [DOI] [PubMed] [Google Scholar]
- 32.Thiel CM, Fink GR. Visual and auditory alertness: Modality-specific and supramodal neural mechanisms and their modulation by nicotine. J. Neurophysiol. 2007;97:2758–2768. doi: 10.1152/jn.00017.2007. [DOI] [PubMed] [Google Scholar]
- 33.Hukkanen J, Jacob P, III, Benowitz NL. Metabolism and disposition kinetics of nicotine. Pharmacol. Rev. 2005;57:79–115. doi: 10.1124/pr.57.1.3. [DOI] [PubMed] [Google Scholar]
- 34.Metherate R, Intskirveli I, Kawai HD. Nicotinic filtering of sensory processing in auditory cortex. Front. Behav. Neurosci. 2012;6:1–8. doi: 10.3389/fnbeh.2012.00044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sarter M, Givens B, Bruno JP. The cognitive neuroscience of sustained attention: Where top-down meets bottom-up. Brain Res. Rev. 2001;35:146–160. doi: 10.1016/S0165-0173(01)00044-3. [DOI] [PubMed] [Google Scholar]
- 36.Sarter M, Gehring WJ, Kozak R. More attention must be paid: The neurobiology of attentional effort. Brain Res. Rev. 2006;51:145–160. doi: 10.1016/j.brainresrev.2005.11.002. [DOI] [PubMed] [Google Scholar]
- 37.Schofield, B. R. & Hurley, L. Circuits for modulation of auditory function. in The mammalian auditory pathways: Synaptic organization and microcircuits (eds. Oliver, D. L., Cant, N. B., Fay, R. R. & Popper, A. N.) 235–267 (Springer International Publishing, berlin, 2018). 10.1007/978-3-319-71798-2_9
- 38.Gil SM, Metherate R. Enhanced sensory-cognitive processing by activation of nicotinic acetylcholine receptors. Nicotine Tob. Res. 2019;21:377–382. doi: 10.1093/ntr/nty134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zeng FG, Richardson M, Turner K. Tinnitus does not interfere with auditory and speech perception. J. Neurosci. 2020;40:6007–6017. doi: 10.1523/JNEUROSCI.0396-20.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Benowitz NL, Lessov-Schlaggar CN, Swan GE, Jacob P. Female sex and oral contraceptive use accelerate nicotine metabolism. Clin. Pharmacol. Ther. 2006;79:480–488. doi: 10.1016/j.clpt.2006.01.008. [DOI] [PubMed] [Google Scholar]
- 41.Gourlay SG, Benowitz NL. Arteriovenous differences in plasma concentration of nicotine and catecholamines and related cardiovascular effects after smoking, nicotine nasal spray, and intravenous nicotine. Clin. Pharmacol. Ther. 1997;62:453–463. doi: 10.1016/S0009-9236(97)90124-7. [DOI] [PubMed] [Google Scholar]
- 42.Pham CQ, et al. Central auditory processing of temporal and spectral-variance cues in cochlear implant listeners. PLoS ONE. 2015;10:1–21. doi: 10.1371/journal.pone.0132423. [DOI] [PMC free article] [PubMed] [Google Scholar]