Abstract
Dendrobium mixture (DMix) is a Traditional Chinese Medicine widely used for preventing and treating diabetic nephropathy (DN). Autophagy contributes to DN development and progression. The present study aimed to investigate the mechanism underlying the protective effects of DMix on the kidneys of rats with DN and to determine whether this involves autophagy. Herein, a high-sugar and high-fat diet, combined with the intra-abdominal injection of low-dose streptozocin, was used to induce DN in 40 Sprague-Dawley male rats. In total, 10 additional rats were used as controls. The rats with DN were then randomly divided into three groups and treated with DMix, gliquidone or saline via gastric administration for 8 weeks. Body weight, kidney weight, kidney index, fasting blood glucose (FBG), blood lipid, hemoglobin A1c (HbA1c), insulin, blood urea nitrogen and serum creatinine levels, as well as the 24-h urinary albumin excretion rate (UAER) were measured. H&E, Periodic Acid-Schiff and Masson staining were used to examine the renal pathology. The mRNA and protein expression levels of LC3 and Beclin-1 in renal tissues were measured using reverse transcription-quantitative PCR and immunohistochemistry, respectively. Western blotting was conducted to measure the protein expression levels of PI3K, phosphorylated (p)-PI3K, Akt, p-Akt, mTOR, p-mTOR, LC3 and Beclin-1 in renal tissues. It was found that DMix significantly reduced the FBG, blood lipids, HbA1c and insulin levels, kidney weight, kidney index and UAER in rats with DN, as well as improved renal function. Rats with DN showed notable glomerular hypertrophy, an increase in mesangial matrix content and renal interstitial fibrosis. Moreover, DMix notably reduced kidney damage. The results demonstrated that DMix inhibited the phosphorylation of PI3K, Akt and mTOR in the kidney tissues of rats with DN, and increased the protein and mRNA expression levels of LC3 and Beclin-1. Therefore, it was suggested that DMix has protective effects on the kidney of rats with DN, which may be associated with the inhibition of the PI3K/Akt/mTOR signaling pathway and activation of renal autophagy by this traditional medicine.
Keywords: DMix, DN, autophagy, PI3K/Akt/mTOR signaling pathway, Traditional Chinese Medicine
Introduction
Diabetic nephropathy (DN) is a common microvascular complication of diabetes mellitus and is a major cause of chronic kidney failure and end-stage renal disease (ESRD) (1,2). The increasing incidence of DN worldwide affects the quality of life of patients and has a heavy burden on society and the economy. According to a US Renal Data System report, 20–40% of diabetic patients in the United States display different degrees of renal injury, and DN is the first secondary factor leading to ESRD (3). The main clinical features of DN are proteinuria, progressive renal impairment and hypertension (4,5), and its prognosis and outcome are associated with a variety of mechanisms, such as hemodynamic changes, oxidative stress and inflammation. Therefore, research should be conducted to further understand the pathogenesis of DN, in order to develop novel therapeutic agents.
Recent studies have reported that autophagy was closely associated with the occurrence and development of DN (6,7). The process of autophagy is an effective method to remove damaged proteins and senescent organelles in cells to maintain the stability of the intracellular environment (8,9). The PI3K/Akt/mTOR signaling pathway is the most typical signaling pathway regulating autophagy, and mTOR is a key target of autophagy regulation and it negatively regulates autophagy (10,11). mTOR is a downstream target of P13K/Akt, and its activity is dependent on regulation of the PI3K/Akt signaling pathway. Under normal physiological circumstances, a tyrosine kinase receptor activates PI3K, which then converts its substrate phosphatidylinositol diphosphate into inositol triphosphate (PIP3). Then, PIP3 and phosphoinositide-dependent kinase-1 synergistically activate Akt, which then transmits the signals for mTOR activation (12). Activation of mTOR and its downstream factors promotes protein synthesis and cell proliferation, accelerates cell metabolism and inhibits cell autophagy (13,14).
Beclin-1 and LC3 are important autophagy markers. Beclin-1, as one of the related genes regulating autophagy formation, is an important component of the autophagy core complex, and it serves a key role in maintaining autophagy activity (15). The LC3 gene also belongs to the autophagy system, and it is involved in the encoding of proteins associated with autophagy. The encoded proteins are mainly expressed on the surface of the pre-autophagosome and autophagosome membranes, and serve a major role in the construction of autophagosome (16). Moreover, the proportion of LC3II/LC3I can reflect the degree of autophagy (17,18). Therefore, the LC3II/LC3I ratio and Beclin-1 expression are usually used as references to evaluate the level of autophagy (19,20).
Dendrobium mixture (DMix) is a preparation used at The Second Affiliated Hospital of Fujian Traditional Chinese Medical University (batch. no. Min Q/YZ-2012-315; patent. no. ZL201110408411.0), which was developed by Professor Hong Shi for the long-term clinical treatment of diabetes and its complications (21,22). DMix is composed of Dendrobium, Astragalus, Schisandra, Radix puerariae, Salvia miltiorrhiza, Rehmanniae and Rhizoma anemarrhenae. In clinical applications, it can lower glucose and lipid levels, and alleviate insulin resistance (22); however, the molecular mechanisms underlying its action remain unknown.
The PI3K/Akt signaling pathway is a signal transduction pathway that promotes cell survival and proliferation in response to extracellular signals (23). It has been reported that the PI3K/Akt/mTOR signaling pathway serves an important role in the pathogenesis of diabetes mellitus; therefore, the regulation of this signaling pathway may be a potential therapeutic target (24). In the present study, a DN rat model was established using a high-sugar and high-fat feed diet, combined with a low-dose streptozocin (STZ) peritoneal injection. Based on the regulatory effect of the PI3K/Akt/mTOR signaling pathway on autophagy, the mechanism of DMix in renal protection in rats with DN was further examined to provide an experimental basis for clinical treatment of DN.
Materials and methods
Drugs
The DMix decoction consisting of the seven herbs listed in Table I (15 g Dendrobium, 20 g Astragalus, 8 g Schisandra, 15 g Radix puerariae, 20 g Salvia miltiorrhiza, 18 g Rehmanniae and 12 g Rhizoma anemarrhenae) was purchased from the Guoyitang Clinic of Fujian University of Traditional Chinese Medicine. The aforementioned herbs were added to distilled water and soaked for 30 min, boiled for 1 h and filtered. The filtered liquid was concentrated to 1.6 g/ml by boiling and stored at this concentration for use. Gliquidone tablets (batch. no. 1140573) were purchased from Beijing Wanhui Shuanghe Pharmaceutical Co., Ltd.
Table I.
Composition of Dendrobium mixture.
| Ingredient, plant name | Plant organ | Weight, g |
|---|---|---|
| Dendrobium | Stem | 15 |
| Astragalus | Radix | 20 |
| Schisandra | Fruit | 8 |
| Radix puerariae | Radix | 15 |
| Salvia miltiorrhiza | Radix and Rhizoma | 20 |
| Rehmanniae | Root tuber | 18 |
| Rhizoma anemarrhenae | Rhizoma | 12 |
Animals
A total of 50 specific-pathogen free healthy male Sprague-Dawley rats (age, 6 weeks; weight, 180–220 g) were provided by Shanghai Slyke Experimental Animals Co., Ltd. [license. no. SCXK (HU) 2017-0005]. Rats were kept in a specific-pathogen free environment (temperature, 22–25°C; humidity, 50–60%; 12-h light/dark cycles) at the Experimental Animal Center of Fujian University of Traditional Chinese Medicine, with free access to a standard diet and water. All animal experiments were conducted in accordance with internationally recognized animal welfare guidelines (25) and were approved by the Medical Ethics Committee of Fujian University of Traditional Chinese Medicine (approval. no. 2019-031). Rats were rapidly euthanized via cervical dislocation under deep anesthesia using 20% urethane (1,000 mg/kg; intraperitoneal).
Experimental design
All rats were fed an adaptive routine diet for 1 week. In total, 10 rats were randomly selected as the normal control group and were fed a routine diet. The remaining 40 rats were given a high-sugar and high-fat diet (main ingredients: 60.7% basic feed, 10% lard, 15% sucrose, 10% egg yolk powder, 4% cholesterol and 0.3% cholate). After 6 weeks of feeding, the rats were fasted for 12 h and given an intraperitoneal injection of 25 mg/kg STZ (Sigma-Aldrich; Merck KGaA) dissolved in 0.1 mol/l citrate buffer (pH 4.2). The control group was given an intraperitoneal injection of equal volume of citrate buffer. After 72 h, a second intraperitoneal injection of the same dose was given using the same method. Then, 3 days later, tail vein blood sampling was performed for detecting random blood glucose levels of ≥16.7 mmol/l, and the unqualified rats (blood glucose level, <16.7 mmol/l) were excluded from the experiment. Rat urine was collected from a metabolic cage for 24 h, and the urinary albumin excretion rate (UAER) was measured. Blood glucose level at ≥16.7 mmol/l and an UAER of ≥30 mg/24 h were used as the establishment criteria for DN (26). In total, 30 rats with DN successfully met the DN model criteria and were randomly allocated into three groups: DN group (n=10), DMix group (n=10) and gliquidone group (n=10). Gliquidone (cat. no. 1140573; Beijing Wanhui Double Crane Pharmaceutical Co., Ltd.) is an anti-DN drug and was used as a positive control. At 9:00 a.m every day, all animals were treated via intragastric administration at the clinical equivalent dose for adults with body weight of 60 kg. The rats in the DMix group were administered 8 g/(kg·d) DMix, and those in the gliquidone group were given 3 mg/(kg·d) gliquidone via gastric gavage. The control rats and rats with DN were administered the same amount of normal saline daily via gastric gavage for 8 weeks.
Measurement of fasting blood glucose (FBG), body weight, kidney weight, kidney index, blood lipid level, insulin and renal function
During the treatment period, the rats were weighed, and FBG was measured every 2 weeks. The rats were fasted (with free access to water) for 12 h, and the tails of the rats were pierced with disposable sterile blood collection needles. After collecting 0.1 ml tail blood, the FBG of the rats was measured using a Roche glucose meter (Roche Diagnostics). At the end of 0, 4 and 8 weeks, rat urine was collected from a metabolic cage for 24 h; then, the urine volume was measured using a volumetric canister and the urine protein concentration was measured using a urine protein quantitative kit (cat. no. C035-2, Nanjing Jiancheng Bioengineering Institute), according to the manufacturer's protocol. Values of the total urine volume and protein concentration were used to calculate the UAER.
After 8 weeks of treatment, all rats were fasted for 12 h, with water available, and then anesthetized using intraperitoneal 20% urethane (1,000 mg/kg). Blood was collected from the abdominal aorta and the serum was separated via centrifugation at 4°C for 15 min at 1,200 × g. Serum total cholesterol (TC; cat. no. A111-2), triglyceride (TG; cat. no. A110-2), low-density lipoprotein cholesterol (LDL-C, cat. no. A113-1), high-density lipoprotein cholesterol (HDL-C; cat. no. A112-1), hemoglobin A1c (HbA1c; cat. no. A056-2), blood urea nitrogen (BUN; cat. no. C013-2) and serum creatinine (Scr; cat. no. C011-2) levels were measured using biochemical analysis kits according to the manufacturer's protocols. All biochemical analysis kits were purchased from the Nanjing Jiancheng Bioengineering Institute. Serum insulin levels were measured with an ELISA kit (cat. no. F15960; Shanghai Xitang Biotechnology Co., Ltd.) according to the manufacturer's protocol. After the rats were sacrificed, both kidneys were removed, washed with normal saline and weighed. Kidney index was calculated as follows: Kidney index=weight (mg)/body weight (g).
Renal histology
Kidney tissue was fixed in 4% paraformaldehyde solution at room temperature for 24 h, embedded in paraffin and cut into 4-µm-thick sections. H&E, Periodic Acid-Schiff (PAS) and Masson's staining were used to evaluate the pathological changes in the kidney tissue. All staining steps were performed at room temperature. The stained kidney sections were examined under a light microscope (Nikon Corporation) at a magnification of ×400.
H&E staining
The dried kidney tissue sections were dewaxed using xylene, graded alcohol (100, 95, 90, 80 and 70%) and distilled water. The sections were stained with hematoxylin for 10 min, differentiated with 1% hydrochloric acid alcohol for 5 sec and then immersed in eosin for 3 min. Dehydration and transparent sealing were then performed for examination under a light microscope.
PAS staining
The dried kidney tissue sections were dewaxed using xylene, graded alcohol and distilled water. Sections were immersed in iodic acid oxidation solution for 5 min, followed by immersion in Schiff reagent for 15 min. After counterstaining with hematoxylin for 1 min, 1% hydrochloric acid alcohol differentiation for 3 sec, dehydration and transparent sealing, examination was performed under a light microscope.
Masson staining
The dried kidney tissue sections were dewaxed using xylene, gradient alcohol and distilled water. The sections were fixed for 1 h in the Bouin's fixative solution (15:5:1 of picric acid saturated liquid, formaldehyde and glacial acetic acid), immersed in Masson composite dyeing solution for 10 min and differentiated in 1% phosphomolybdate for 10 min. The collagen fiber showed a reddish color and was immersed in 2% aniline blue solution for 5 min. Dehydration and transparent sealing were then performed for examination under a light microscope.
Reverse transcription-quantitative PCR (RT-qPCR)
Total RNA was extracted from rat kidney tissue using the RNAiso Plus reagent (cat. no. 9108; Takara Bio, Inc.), and the concentration was determined. Then, cDNA was synthesized using a PrimeScript RT Reagent kit (cat. no. RR047A; Takara Bio, Inc.) at 37°C for 15 min and 85°C for 5 sec. PCR was performed using a PCR kit (cat. no. RR420A; Takara Bio, Inc.) under the following reaction conditions: Initial denaturation at 95°C for 30 sec; followed by 40 cycles of denaturation at 95°C for 5 sec, annealing at 55°C for 30 sec and extension at 72°C for 30 sec. SDS2.4 software (Thermo Fisher Scientific, Inc.) was used to analyze the Cq values of the samples detected during the PCR process, using GAPDH as the internal reference and the ΔΔCq method for relative quantitative analysis, with 2−∆∆Cq as a quantity for the relative expression of the target RNA. PCR primers (Table II) were designed and provided by Fuzhou Shangya Biotechnology Co., Ltd.
Table II.
Primers used for reverse transcription-quantitative PCR.
| Gene | Primer sequence | Product length, bp |
|---|---|---|
| LC3 | Forward: 5′-GCGAGTTGGTCAAGATCATCC-3′ | 138 |
| Reverse: 5′-CGTCTTCATCCTTCTCCTGTTC-3′ | ||
| Beclin-1 | Forward: 5′-AATCTAAGGAGTTGCCGTTGT-3′ | 191 |
| Reverse: 5′-GCCTCCAGTGTCTTCAATCTT-3′ | ||
| GAPDH | Forward: 5′-ACGGCAAGTTCAACGGCACAG-3′ | 149 |
| Reverse: 5′-GAAGACGCCAGTAGACTCCACGAC-3′ |
Immunohistochemistry
The kidney tissue was fixed in 4% paraformaldehyde solution at room temperature for 24 h, embedded in paraffin and cut into 4-µm-thick sections. The paraffin-embedded kidney tissue was cut into 4-µm-thick slices, adhered to microscope slides, baked at 58°C for 2 h, dewaxed using xylene twice, hydrated with gradient alcohol (100, 95, 90, 80 and 70%), placed into boiled sodium citrate solution for antigen repair and naturally cooled to room temperature (18–30°C). The sections were rinsed with PBS three times, co-incubated with an endogenous peroxidase blocker at room temperature for 10 min, rinsed with PBS three times and co-incubated with non-immunized animal serum (10%; Fuzhou Maixin Biotech Co., Ltd.) at room temperature for 10 min. After removing the serum, the following primary antibodies were added in a dropwise manner: Rabbit anti-LC3 polyclonal antibody (1:200; cat. no. ab48394; Abcam) and rabbit anti-Beclin-1 polyclonal antibody (1:500; cat. no. ab62557; Abcam). Slides were then incubated overnight at 4°C and then rinsed with PBS three times. Biotin-labeled sheep anti-rabbit IgG (ready to use; cat. no. KIT-9710; Fuzhou Maixin Biotech Co., Ltd.) was added and slides were incubated at room temperature for 10 min, after which they were rinsed with PBS thrice, incubated with streptavidin-peroxidase (Fuzhou Maixin Biotech Co., Ltd.) at room temperature for 10 min and rinsed with PBS three times. Then, the sections were treated with DAB (Wuhan Boster Biological Technology Co., Ltd.) for color development at room temperature for 3 min, rinsed with distilled water, dyed with hematoxylin at room temperature for 1 min and rinsed using tap water for blueness. Gradient alcohol was used for dehydration, sections were dried and made transparent using xylene, following which neutral gum was used for sealing. Brown staining indicated positive expression as observed under an optical microscope (magnification, ×400). Image-pro Plus 6.0 software (Media Cybernetics, Inc.) was used for analysis, and the relative protein expression was represented in terms of mean density.
Western blotting
The kidney tissues stored in liquid nitrogen were lysed in RIPA buffer (cat. no. P0013B; Beyotime Institute of Biotechnology) and fully ground to create tissue homogenate. After centrifugation (4°C; 1,200 × g; 15 min), the supernatant was absorbed to obtain the total protein of kidney tissue, and the protein concentration was determined using a BCA assay. Then, 30 µg each sample was used for 10% SDS-PAGE, after which the samples were transferred to a PVDF membrane, and the membrane was blocked with 5% skim milk at room temperature for 1 h. Next, the membrane was incubated with primary antibodies overnight at 4°C. After rinsing with TBS-0.1% Tween 20 (TBST), the membrane was incubated with a secondary antibody at room temperature for 1 h. After rinsing with TBST, the membrane was stained using ECL (cat. no. P0018S; Beyotime Institute of Biotechnology) reagent and viewed with a gel imaging system (Bio-Rad Laboratories, Inc.). The antibodies and dilutions used were as follows: β-actin (1:1,000; cat. no. ab8226; Abcam), PI3K (1:1,000; cat. no. ab191606; Abcam), phosphorylated (p)-PI3K (1:500; cat. no. ab182651; Abcam), Akt (1:500; cat. no. ab8805; Abcam), p-Akt (1:500; cat. no. ab222489; Abcam), mTOR (1:10,000; cat. no. ab134903; Abcam), p-mTOR (1:1,000; cat. no. ab137133; Abcam), LC3 (1:2,000; cat. no. ab48394; Abcam), Beclin-1 (1:1,000; cat. no. ab62557; Abcam), goat anti-mouse IgG secondary antibody (1:2,000; cat. no. A0216; Biyuntian Biotechnology Research Institute) and goat anti-rabbit IgG secondary antibody (1:1,000; cat. no. A0208; Biyuntian Biotechnology Research Institute). Image Lab software (version 5.2.1; Bio-Rad Laboratories, Inc.) was used to analyze and semi-quantify the images.
Statistical analysis
All experiments were repeated three times. SPSS 22.0 statistical software (IBM Corp.) was used to analyze the data, which are presented as the mean ± SD if they followed a normal distribution. If not, the data are expressed as median and interquartile range. If the data conformed to the normal distribution pattern, differences among multiple groups were analyzed using one-way and mixed two-way ANOVA. Kruskal-Wallis test was used for analyzing data that did not follow a normal distribution. The Bonferroni method was used for pairwise comparison between groups when the variances were homogeneous, and Dunnett's T3 comparison was used when the variances were heterogeneous. P<0.05 was considered to indicate a statistically significant difference.
Results
General conditions of rats during the treatment period
The rats in the control group displayed a quick reaction, good mental state and glossy hair. The rats in the DN group showed slow reaction and weight loss, and increased diet, water intake and urine output, compared with the control group. In comparison with the DN group, these characteristics were improved to varying degrees in the DMix and gliquidone groups.
DMix lowers FBG levels in rats with DN
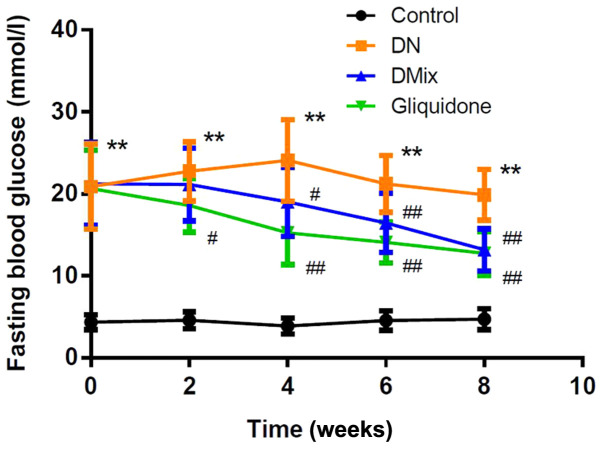
As shown in Fig. 1, the FBG levels of rats in the DN group were significantly increased compared with those in the control group (P<0.01). After treatment with DMix, FBG levels were gradually decreased with treatment time, and they were significantly different from those in the DN group from the 4th week onwards (week 4, P=0.034; week 6, P=0.009; and week 8, P<0.01); the difference was not statistically significant compared with the gliquidone group (P=0.767).
Figure 1.
DMix lowers fasting blood glucose levels in rats with DN. Fasting blood glucose levels at 0–8 weeks after DMix treatment in the control, DN, DMix and gliquidone groups. Data are presented as the mean ± SD. **P<0.01 vs. control; #P<0.05, ##P<0.01 vs. DN. DMix, Dendrobium mixture; DN, diabetic nephropathy.
Effect of DMix on serum HbA1c and insulin levels of rats with DN
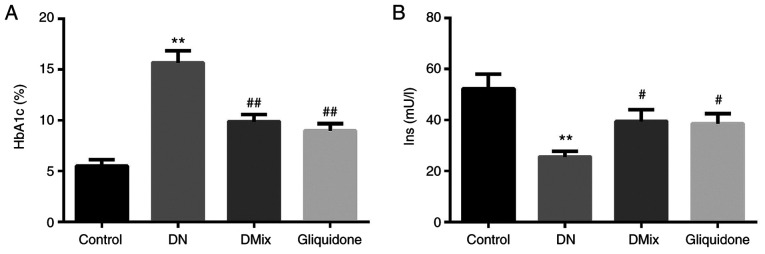
It was found that compared with the control group, HbA1c levels of rats in the DN group were increased significantly (P=0.000; Fig. 2A), and insulin levels were decreased significantly (P<0.01; Fig. 2B). Compared with the DN group, HbA1c and insulin levels in the DMix group were significantly decreased and increased, respectively (HbA1c, P<0.01; insulin, P=0.033).
Figure 2.
Comparison of serum HbA1c and Ins levels in each group. Effect of DMix on serum (A) HbA1c and (B) Ins levels in rats with DN. **P<0.01 vs. control; #P<0.05, ##P<0.01 vs. DN. DMix, Dendrobium mixture; HbA1c, hemoglobin A1c; Ins, insulin; DN, diabetic nephropathy.
Comparison of body weight, kidney weight and kidney index in each group
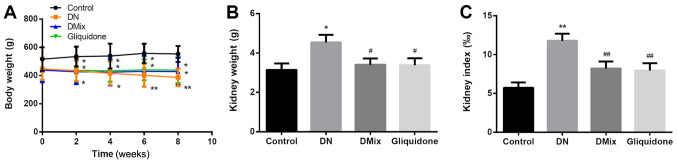
Compared with the control group, the DN group continued to lose weight over time (week 2, P=0.043; week 4, P=0.022; week 6, P=0.004; and week 8, P=0.002). However, after treatment with DMix, further weight loss of the rats was inhibited (P=0.006; Fig. 3A). The kidney weight and index of the DN group were significantly higher compared with those of the normal group (kidney weight, P=0.011; kidney index, P<0.01; Fig. 3B and C), suggesting that the renal tissue was damaged. After 8 weeks of treatment with DMix, the kidney weight and index of the rats were significantly decreased (kidney weight, P=0.034; kidney index, P=0.008; Fig. 3B and C), which showed alleviated renal tissue injury.
Figure 3.
Comparison of body weight, kidney weight and kidney index in each group. Changes in (A) body weight, (B) kidney weight and (C) kidney index after DMix treatment. *P<0.05, **P<0.01 vs. control; #P<0.05, ##P<0.01 vs. DN. DMix, Dendrobium mixture; DN, diabetic nephropathy.
Effects of DMix on serum TC, TG, LDL-C and HDL-C levels in rats with DN
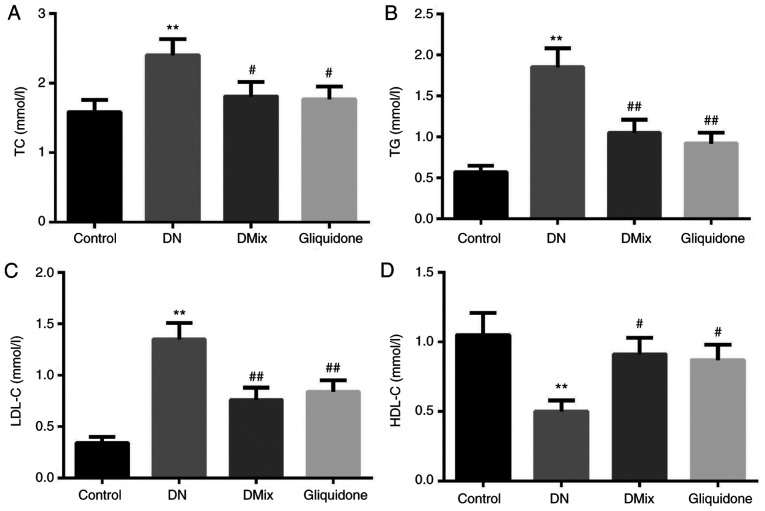
As shown in Fig. 4, the serum TC, TG and LDL-C levels of rats in the DN group were significantly higher compared with those in the control group (TC, P=0.009; TG, P<0.01; LDL-C, P<0.01), while HDL-C levels were significantly lower (P=0.005). Moreover, compared with the DN group, TC, TG and LDL-C levels were significantly lower in the DMix group (TC, P=0.049; TG, P=0.048; LDL-C, P=0.003), while HDL-C levels were significantly higher (P=0.029). There was no statistically significant difference between the DMix and gliquidone groups (TC, P=0.894; TG, P=0.579; LDL-C, P=0.664; HDL-C, P=0.848).
Figure 4.
Comparison of blood lipid levels in each group. Effects of DMix on serum (A) TC, (B) TG, (C) LDL-C and (D) HDL-C levels in rats with DN. **P<0.01 vs. control; #P<0.05, ##P<0.01 vs. DN. DMix, Dendrobium mixture; TC, total cholesterol; TG, triglyceride; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; DN, diabetic nephropathy.
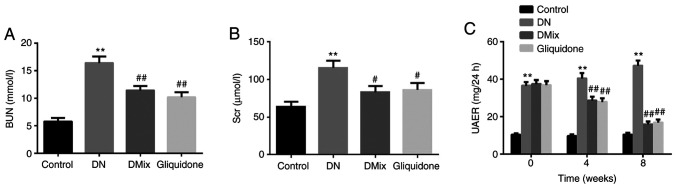
DMix improves renal function in rats with DN
Indices of renal function, including BUN, Scr and UAER, were detected in each group, and compared with those of the control group. All indices in the DN group were found to be significantly increased (BUN, P<0.01; Scr, P<0.01; UAER, P<0.01; Fig. 5A-C). Renal insufficiency was observed in rats with DN. Compared with the DN group, BUN and Scr levels in the DMix group were significantly reduced (BUN, P=0.001; Scr, P=0.013; Fig. 5A and B), but the difference was not statistically significant compared with that of the gliquidone group (BUN, P=0.347; Scr, P=0.792). UAER was measured every 4 weeks. It was identified that the UAER in the DN group was gradually increased (week 4, P=0.293; week 8, P=0.011), and it was significantly decreased after treatment with DMix (week 4, P=0.001; week 8, P<0.01; Fig. 5C). These data indicated that DMix protected renal function in rats with DN.
Figure 5.
DMix improves renal function in rats with DN. DMix improved (A) BUN, (B) Scr and (C) 24-h UAER in rats with DN. **P<0.01 vs. control; #P<0.05, ##P<0.01 vs. DN. DMix, Dendrobium mixture; BUN, blood urea nitrogen; Scr, serum creatinine; UAER, urinary albumin excretion rate; DN, diabetic nephropathy.
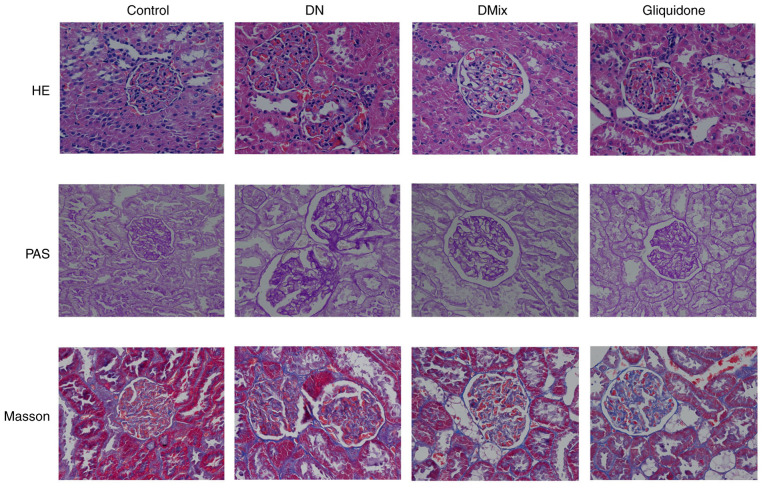
Effect of DMix on pathological renal morphology of rats with DN
H&E, PAS and Masson staining revealed that the kidney structure of the control rats was clear and complete, the size and shape of the glomeruli were normal, the number of matrix and mesangial cells was not increased and the renal tubular lumen was not obvious. Moreover, the epithelial cells were intact, there was no glycogen deposition, no signs of fibrous tissue hyperplasia were observed and the basement membrane was not thickened (Fig. 6). In the DN group, the glomerular volume was higher, the number of mesangial cells and extracellular matrix deposition were increased, the mesangial area was wider and there were signs of vacuolar degeneration of the renal tubular epithelial cells. In addition, the number of renal interstitial cells was increased, the amount of red-stained glycogen deposits was increased and there was obvious collagen fiber accumulation (Fig. 6). Compared with the DN group, both the DMix and gliquidone groups had significantly improved observable morphology, as the proliferation of glomerular mesangial cells was significantly reduced, the deposition of extracellular matrix was decreased and the basement membrane was thinner than that of the DN group. Furthermore, the structure of renal tubules was essentially restored to normal, a small amount of glycogen was observed and the deposition of collagen fibers was decreased (Fig. 6).
Figure 6.
Photomicrographs of H&E, PAS and Masson staining of rat kidneys from each group, as observed under a light microscope (magnification, ×400). The kidney specimen of the DN group showed markedly severe destruction in glomerular and tubulointerstitial lesions, such as glomerular hypertrophy, increased mesangial cell number and extracellular matrix deposition, interstitial cell infiltration and accumulation of glycogen and collagen fibres. After treatment with DMix, the overall morphology of glomerular and tubulointerstitial lesions improved significantly. DMix, Dendrobium mixture; DN, diabetic nephropathy; PAS, Periodic Acid-Schiff.
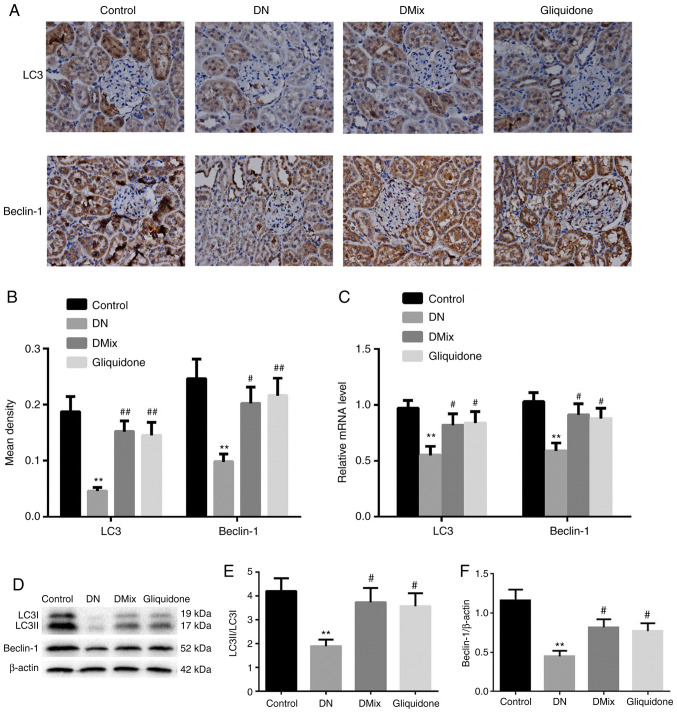
DMix increases the protein and mRNA expression levels of LC3 and Beclin-1 in the kidneys of rats with DN
Immunohistochemical staining and western blot analysis demonstrated that the protein expression levels of LC3 and Beclin-1, and the ratio of LC3II to LC3I in rat kidney tissues of the DN group were significantly lower compared with those in the control group (immunohistochemical: LC3, P<0.01; Beclin-1, P=0.002; western blotting: LC3II/LC3I, P=0.004; Beclin-1, P<0.01; Fig. 7A, B, D-F). However, the protein expression levels of LC3 and Beclin-1 and the ratio of LC3II to LC3I were significantly increased in the DMix and gliquidone groups (immunohistochemical, DMix: LC3, P=0.001; Beclin-1, P=0.019; immunohistochemical, gliquidone: LC3, P=0.002; Beclin-1, P=0.008; western blotting, DMix: LC3II/LC3I, P=0.018; Beclin-1, P=0.023; western blotting, gliquidone: LC3II/LC3I, P=0.029; Beclin-1, P=0.049; Fig. 7A, B, D-F). Additionally, the mRNA expression levels of LC3 and Beclin-1 in rat kidney tissues of the DN group were significantly lower (LC3, P=0.003; Beclin-1, P=0.002; Fig. 7C). DMix upregulated the mRNA expression levels of LC3 and Beclin-1 in rats with DN (LC3, P=0.048; Beclin-1, P=0.017; Fig. 7C). These results indicated that DMix may promote the expression levels of autophagy-related proteins in the kidney tissues of rats with DN to protect renal function.
Figure 7.
DMix increases the protein and mRNA expression levels of LC3 and Beclin-1 in the kidneys of rats with DN. (A) Immunohistochemical staining of LC3 and Beclin-1 (magnification, ×400). (B) Average optical density values of LC3 and Beclin-1. (C) Reverse transcription-quantitative PCR analysis of LC3 and Beclin-1. (D) Western blot analysis of (E) LC3 and (F) Beclin-1 protein expression levels. **P<0.01 vs. control; #P<0.05, ##P<0.01 vs. DN. DMix, Dendrobium mixture; DN, diabetic nephropathy.
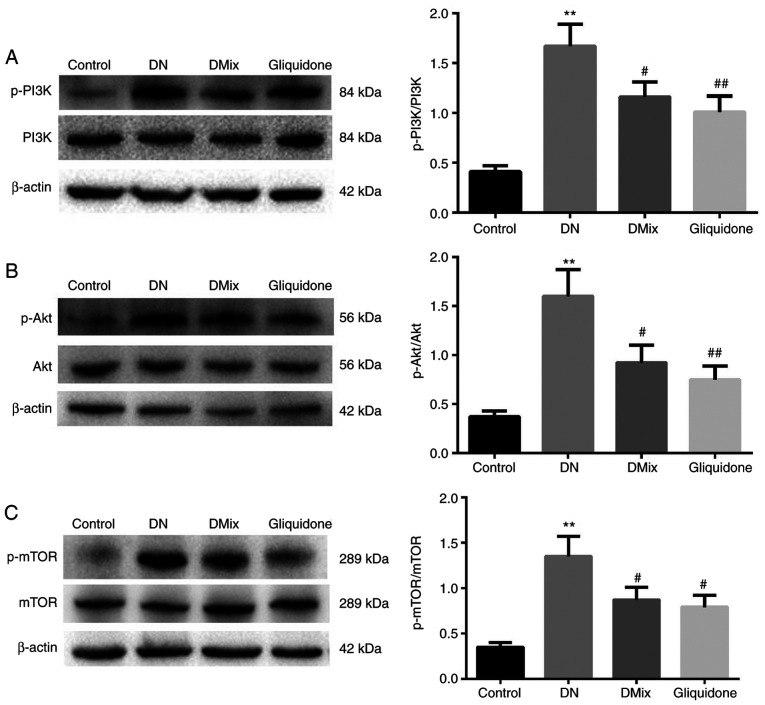
DMix inhibits the PI3K/Akt/mTOR signal transduction pathway in the renal tissues of rats with DN
It is well known that the PI3K/Akt/mTOR signaling pathway is one of the main pathways regulating autophagy (27). In order to understand the molecular mechanism underlying the effect of DMix on renal autophagy in DN, the effect of DMix on the PI3K/Akt/mTOR signaling pathway was examined (Fig. 8). The results demonstrated that the protein expression levels of p-PI3K, p-Akt and p-mTOR in the DN group were significantly higher compared with those in the control group (p-PI3K, P<0.01; p-Akt, P<0.01; p-mTOR, P<0.01), indicating that the PI3K/Akt/mTOR signaling pathway was more active in the kidney tissues of rats with DN. After 8 weeks of DMix treatment, the protein expression levels of p-PI3K, p-Akt and p-mTOR were significantly decreased (p-PI3K, P=0.036; p-Akt, P=0.014; p-mTOR, P=0.022), but there were no significant changes in the expression levels of PI3K, Akt and mTOR proteins. Furthermore, there was no statistically significant difference between the DMix and gliquidone groups (p-PI3K, P=0.716; p-Akt, P=0.531; p-mTOR, P=0.522). This indicated that DMix inhibited the PI3K/Akt/mTOR signaling pathway in the kidney tissues of rats with DN.
Figure 8.
DMix inhibits the PI3K/Akt/mTOR signal transduction pathway in the renal tissues of rats with DN. The protein expression levels of (A) PI3K, p-PI3K, (B) Akt, p-Akt, (C) mTOR and p-mTOR were detected using western blotting. β-actin, PI3K, Akt and mTOR were used as the internal control. The relative ratios of (A) p-PI3K/PI3K, (B) p-Akt/Akt and (C) p-mTOR/mTOR were used to analyze the gray value. **P<0.01 vs. control; #P<0.05, ##P<0.01 vs. DN. DMix, Dendrobium mixture; DN, diabetic nephropathy; p-, phosphorylated.
Discussion
In the present study, it was identified that DMix inhibited the PI3K/Akt/mTOR signaling pathway, which promoted renal autophagy and improved renal function. Moreover, DMix inhibited the phosphorylation of PI3K, Akt and mTOR, increased the expression levels of LC3 and Beclin-1, and alleviated renal damage based on the morphological analysis of the kidneys, thereby delaying the progression of DN. The basic pathological changes in DN are glomerular mesangial cell proliferation, increase in the extracellular matrix contents, glomerular basement membrane thickening and glomerular sclerosis (28–30). Its pathogenesis is complex, and there are a lack of effective treatments for this condition.
Recently, Traditional Chinese Medicine (TCM) has achieved good efficacy in the treatment of DN. Numerous TCMs have been used in clinical practice and have shown promising results in the treatment of diabetes and its complications (31,32). Unlike a single compound that has only one active chemical component, TCM is usually a complex combination of chemicals that work synergistically. Moreover, any active ingredient of TCM alone cannot reflect the final therapeutic effect of the whole medicine (33,34). In preliminary experimental studies and clinical practice, DMix has been proved to have a good therapeutic effect against diabetes and its complications (21,22).
The present study used a high-sugar and high-fat feed combined classic model of STZ-induced DN to successfully reproduce the typical pathological changes of DN in rats. The biochemical indexes of these rats were abnormal to varying degrees, and these were significantly improved after DMix treatment, which was consistent with the findings of our previous studies (21,22). Furthermore, the current study demonstrated the therapeutic effects of DMix in protecting renal function and preventing renal fibrosis. Gliquidone is a sulfonylurea-based hypoglycemic agent that can bind to the specific receptors on the pancreatic β cell membrane to promote insulin secretion; it has a certain protective effect on the kidney (35). Gliquidone is a commonly used drug in the clinical treatment of DN and was used as a positive control in the present study. The current results demonstrated that the clinical equivalent dose of DMix had effects similar to those of gliquidone.
Autophagy is considered an important potential mechanism underlying the development of DN; thus, activation of autophagy may be a potential therapeutic target for the treatment of DN (36–38). Our previous studies confirmed that Beclin-1 and LC3 levels in the kidneys of rats with DN were downregulated, suggesting that autophagy was a self-repairing mechanism of the body to fight against the damage caused by stress (39,40). The results of the present study also indicated that the autophagy activity in the renal tissue of DN was reduced, and that DMix enhanced autophagy and improved renal function.
Previous studies have reported that in DN, the renal PI3K/Akt/mTOR signaling pathway was abnormally activated and autophagy was inhibited, leading to kidney injury, glomerular hypertrophy and accelerated renal fibrosis (41–43). p-PI3K, p-Akt and p-mTOR are key participants in the PI3K/Akt/mTOR signaling pathway that activate this signaling pathway (44,45). In the current study, the intervention of DMix significantly inhibited phosphorylation of PI3K, Akt and mTOR. These results suggest that DMix may restore renal autophagy activity and serve a protective role in DN kidney tissue by inhibiting the PI3K/Akt/mTOR signaling pathway.
Although the results support the current hypothesis, this study has some limitations. For example, the duration of drug therapy was relatively short; the present findings are similar to studies of multiple other drug mixtures (46,47); and other mechanisms that have not been investigated here cannot be excluded. Therefore, further studies in larger cohorts over a longer duration, as well as clinical trials, are required to verify the present findings.
In conclusion, the present study demonstrated that DMix has a protective effect on the kidneys of rats with DN, which may be due to the negative regulation of the PI3K/Akt/mTOR signaling pathway, promotion of renal autophagy and inhibition of renal fibrosis, thereby delaying the progression of DN. Therefore, DMix could be used as a potentially beneficial TCM for the prevention and treatment of DN.
Acknowledgements
Not applicable.
Glossary
Abbreviations
- DMix
Dendrobium mixture
- DN
diabetic nephropathy
- STZ
streptozocin
- FBG
fasting blood glucose
- UAER
urinary albumin excretion rate
- BUN
blood urea nitrogen
- Scr
serum creatinine
- HbA1c
hemoglobin A1c
Funding Statement
The present study was supported by the Fujian University of Traditional Chinese Medicine's research platform management project (grant. no. X2019001-platform), the Natural Science Foundation of Fujian Province (grant. no. 2018J01873) and the National Natural Science Foundation of China (grant. no. 81973827).
Funding
The present study was supported by the Fujian University of Traditional Chinese Medicine's research platform management project (grant. no. X2019001-platform), the Natural Science Foundation of Fujian Province (grant. no. 2018J01873) and the National Natural Science Foundation of China (grant. no. 81973827).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
YC, YFZ, XHL, JPZ, FL and HS participated in the study design. YC, YFZ and XHL performed the experiments. YC, YFZ, XHL, JPZ, FL and HS performed the data analysis. YC and HS confirm the authenticity of all the raw data. YC drafted the manuscript. All authors have read and approved the final manuscript.
Ethics approval and consent to participate
The present study was carried out in strict accordance with the requirements in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The experiments were carried out strictly in line with the requirements of the International Association for the Study of Pain. The present study was approved by the Medical Ethics Committee of Fujian University of Traditional Chinese Medicine (Fujian, China; approval. no. 2019-031).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Li X, Wang L, Ma H. Betaine alleviates high glucose-induced mesangial cell proliferation by inhibiting cell proliferation and extracellular matrix deposition via the AKT/ERK1/2/p38 MAPK pathway. Mol Med Rep. 2019;20:1754–1760. doi: 10.3892/mmr.2019.10391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hsieh AR, Huang YC, Yang YF, Lin HJ, Lin JM, Chang YW, Wu CM, Liao WL, Tsai FJ. Lack of association of genetic variants for diabetic retinopathy in Taiwanese patients with diabetic nephropathy. BMJ Open Diabetes Res Care. 2020;8:e000727. doi: 10.1136/bmjdrc-2019-000727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Perkovic V, Jardine MJ, Neal B, Bompoint S, Heerspink HJL, Charytan DM, Edwards R, Agarwal R, Bakris G, Bull S, et al. Canagliflozin and renal outcomes in type 2 diabetes and nephropathy. N Engl J Med. 2019;380:2295–2306. doi: 10.1056/NEJMoa1811744. [DOI] [PubMed] [Google Scholar]
- 4.Zhang Q, Yang M, Xiao Y, Han Y, Yang S, Sun L. Towards better drug repositioning: Targeted immunoinflammatory therapy for diabetic nephropathy. Curr Med Chem. 2021;8:1003–1024. doi: 10.2174/0929867326666191108160643. [DOI] [PubMed] [Google Scholar]
- 5.Oraby MA, El-Yamany MF, Safar MM, Assaf N, Ghoneim HA. Amelioration of early markers of diabetic nephropathy by linagliptin in fructose-streptozotocin-induced type 2 diabetic rats. Nephron. 2019;141:273–286. doi: 10.1159/000495517. [DOI] [PubMed] [Google Scholar]
- 6.Li X, Zhu Q, Zheng R, Yan J, Wei M, Fan Y, Deng Y, Zhong Y. Puerarin attenuates diabetic nephropathy by promoting autophagy in podocytes. Front Physiol. 2020;11:73. doi: 10.3389/fphys.2020.00073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yang C, Chen XC, Li ZH, Wu HL, Jing KP, Huang XR, Ye L, Wei B, Lan HY, Liu HF. SMAD3 promotes autophagy dysregulation by triggering lysosome depletion in tubular epithelial cells in diabetic nephropathy. Autophagy. 2020;10:1–20. doi: 10.1080/15548627.2020.1824694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kim JH, Kim KM, Jeong JU, Shin JH, Shin JM, Bang KT. Nrf2-Heme oxygenase-1 modulates autophagy and inhibits apoptosis triggered by elevated glucose levels in renal tubule cells. Kidney Res Clin Pract. 2019;38:318–325. doi: 10.23876/j.krcp.18.0152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cao T, Xu R, Xu Y, Liu Y, Qi D, Wan Q. The protective effect of cordycepin on diabetic nephropathy through autophagy induction in vivo and in vitro. Int Urol Nephrol. 2019;51:1883–1892. doi: 10.1007/s11255-019-02241-y. [DOI] [PubMed] [Google Scholar]
- 10.Dai H, Liu Q, Liu B. Research progress on mechanism of podocyte depletion in diabetic nephropathy. J Diabetes Res. 2017;2017:2615286. doi: 10.1155/2017/2615286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chen Y, Liu Q, Shan Z, Mi W, Zhao Y, Li M, Wang B, Zheng X, Feng W. Catalpol ameliorates podocyte injury by stabilizing cytoskeleton and enhancing autophagy in diabetic nephropathy. Front Pharmacol. 2019;10:1477. doi: 10.3389/fphar.2019.01477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhang MZ, Jang H, Nussinov R. The structural basis for ras activation of PI3Kα lipid kinase. Phys Chem Chem Phys. 2019;21:12021–12028. doi: 10.1039/C9CP00101H. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang X, Wang D, Liu B, Jin X, Wang X, Pan J, Tu W, Shao Y. IMP3 accelerates the progression of prostate cancer through inhibiting PTEN expression in a SMURF1-dependent way. J Exp Clin Cancer Res. 2020;39:190. doi: 10.1186/s13046-020-01657-0. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 14.Clement E, Inuzuka H, Nihira NT, Wei W, Toker A. Skp2-dependent reactivation of AKT drives resistance to PI3K inhibitors. Sci Signal. 2018;11:eaao3810. doi: 10.1126/scisignal.aao3810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chen XB, Sun Y, Wang BR, Wang HH. Prognostic significance of autophagy-related genes beclin1 and LC3 in ovarian cancer: A meta-analysis. J Int Med Res. 2020;48:300060520968299. doi: 10.1177/0300060520968299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yu H, Shao J, Huang R, Guan Y, Li G, Chen S, Zhou F, Yao Q, Shen J. Targeting PTEN to regulate autophagy and promote the repair of injured neurons. Brain Res Bull. 2020;165:161–168. doi: 10.1016/j.brainresbull.2020.10.008. [DOI] [PubMed] [Google Scholar]
- 17.Jung G, Roh J, Lee H, Gil M, Yoon DH, Suh C, Jang S, Park CJ, Huh J, Park CS. Autophagic markers beclin1 and LC3 are associated with prognosis of multiple myeloma. Acta Haematol. 2015;134:17–24. doi: 10.1159/000368848. [DOI] [PubMed] [Google Scholar]
- 18.Ebrahim N, Ahmed IA, Hussien NI, Dessouky AA, Farid AS, Elshazly AM, Mostafa O, Gazzar WBE, Sorour SM, Seleem Y, et al. Mesenchymal stem cell-derived exosomes ameliorated diabetic nephropathy by autophagy induction through the mTOR signaling pathway. Cells. 2018;7:226. doi: 10.3390/cells7120226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fu L, Wu W, Sun X, Zhang P. Glucocorticoids enhanced osteoclast autophagy through the PI3K/Akt/mTOR signaling pathway. Calcif Tissue Int. 2020;107:60–71. doi: 10.1007/s00223-020-00687-2. [DOI] [PubMed] [Google Scholar]
- 20.Vurusaner B, Gargiulo S, Testa G, Gamba P, Leonarduzzi G, Poli G, Basaga H. The role of autophagy in survival response induced by 27-hydroxycholesterol in human promonocytic cells. Redox Biol. 2018;17:400–410. doi: 10.1016/j.redox.2018.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xu Q, Liu Y, Cong YB, Zheng YY, Zhang JP, Shi H. Gene expression and microarray investigation of dendrobium mixture as progressive therapy for the treatment of type 2 diabetes mellitus. Trop J Pharmaceutical Res. 2013;12:195–201. [Google Scholar]
- 22.Lin X, Shi H, Cui Y, Wang X, Zhang J, Yu W, Wei M. Dendrobium mixture regulates hepatic gluconeogenesis in diabetic rats via the phosphoinositide-3-kinase/protein kinase B signaling pathway. Exp Ther Med. 2018;16:204–212. doi: 10.3892/etm.2018.6194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nitulescu GM, Margina D, Juzenas P, Peng Q, Olaru OT, Saloustros E, Fenga C, Spandidos DA, Libra M, Tsatsakis AM. Akt inhibitors in cancer treatment: The long journey from drug discovery to clinical use. Int J Oncol. 2016;48:869–885. doi: 10.3892/ijo.2015.3306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nitulescu GM, Van De Venter M, Nitulescu G, Ungurianu A, Juzenas P, Peng Q, Olaru OT, Grădinaru D, Tsatsakis A, Tsoukalas D, et al. The Akt pathway in oncology therapy and beyond. Int J Oncol. 2018;53:2319–2331. doi: 10.3892/ijo.2018.4597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Brown MJ, Symonowicz C, Medina LV, Bratcher NA, Buckmaster CA, Klein H, Anderson LC. Management of Animal Care and Use Programs in Research, Education, and Testing. 2nd edition. Boca Raton (FL): CRC Press/Taylor & Francis; 2018. Culture of care: Organizational responsibilities. [PubMed] [Google Scholar]
- 26.Tesch GH, Allen TJ. Rodent models of streptozotocin-induced diabetic nephropathy. Nephrology (Carlton) 2007;12:261–266. doi: 10.1111/j.1440-1797.2007.00796.x. [DOI] [PubMed] [Google Scholar]
- 27.Liu K, Yang Y, Zhou F, Xiao Y, Shi L. Inhibition of PI3K/AKT/mTOR signaling pathway promotes autophagy and relieves hyperalgesia in diabetic rats. Neuroreport. 2020;31:644–649. doi: 10.1097/WNR.0000000000001461. [DOI] [PubMed] [Google Scholar]
- 28.Zeng LF, Xiao Y, Sun L. A glimpse of the mechanisms related to renal fibrosis in diabetic nephropathy. Adv Exp Med Biol. 2019;1165:49–79. doi: 10.1007/978-981-13-8871-2_4. [DOI] [PubMed] [Google Scholar]
- 29.Qi C, Mao X, Zhang Z, Wu H. Classification and differential diagnosis of diabetic nephropathy. J Diabetes Res. 2017;2017:8637138. doi: 10.1155/2017/8637138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dong Z, Sun Y, Wei G, Li S, Zhao Z. Ergosterol ameliorates diabetic nephropathy by attenuating mesangial cell proliferation and extracellular matrix deposition via the TGF-β1/smad2 signaling pathway. Nutrients. 2019;11:483. doi: 10.3390/nu11020483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Park CH, Hiratani K, Natazuka T, Yokozawa T. Therapeutic effect of Chinese prescription Kangen-Karyu in patients with diabetic nephropathy. Drug Discov Ther. 2020;14:84–88. doi: 10.5582/ddt.2020.03077. [DOI] [PubMed] [Google Scholar]
- 32.Wang J, Ma Q, Li Y, Li P, Wang M, Wang T, Wang C, Wang T, Zhao B. Research progress on Traditional Chinese Medicine syndromes of diabetes mellitus. Biomed Pharmacother. 2020;121:109565. doi: 10.1016/j.biopha.2019.109565. [DOI] [PubMed] [Google Scholar]
- 33.Cheng T, Ye J, Li H, Dong H, Xie N, Mi N, Zhang Z, Zou J, Jin H, Zhang W. Hybrid multidimensional data acquisition and data processing strategy for comprehensive characterization of known, unknown and isomeric compounds from the compound dan zhi tablet by UPLC-TWIMS-QTOFMS. Rsc Advances. 2019;9:8714–8727. doi: 10.1039/C8RA10100K. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Shen H, Qu Z, Harata-Lee Y, Aung TN, Cui J, Wang W, Kortschak RD, Adelson DL. Understanding the mechanistic contribution of herbal extracts in compound kushen injection with transcriptome analysis. Front Oncol. 2019;9:632. doi: 10.3389/fonc.2019.00632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tian HY, Yang JB, Xie ZC, Liu JL. Gliquidone alleviates diabetic nephropathy by inhibiting notch/snail signaling pathway. Cell Physiol Biochem. 2018;51:2085–2097. doi: 10.1159/000495827. [DOI] [PubMed] [Google Scholar]
- 36.Kim H, Dusabimana T, Kim SR, Je J, Jeong K, Kang MC, Cho KM, Kim HJ, Park SW. Supplementation of abelmoschus manihot ameliorates diabetic nephropathy and hepatic steatosis by activating autophagy in mice. Nutrients. 2018;10:1703. doi: 10.3390/nu10111703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Liu WJ, Huang WF, Ye L, Chen RH, Yang C, Wu HL, Pan QJ, Liu HF. The activity and role of autophagy in the pathogenesis of diabetic nephropathy. Eur Rev Med Pharmacol Sci. 2018;22:3182–3189. doi: 10.26355/eurrev_201805_15079. [DOI] [PubMed] [Google Scholar]
- 38.Tu Q, Li Y, Jin J, Jiang X, Ren Y, He Q. Curcumin alleviates diabetic nephropathy via inhibiting podocyte mesenchymal transdifferentiation and inducing autophagy in rats and MPC5 cells. Pharm Biol. 2019;57:778–786. doi: 10.1080/13880209.2019.1688843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jiang Y, Zhao Y, Zhu X, Liu Y, Wu B, Guo Y, Liu B, Zhang X. Effects of autophagy on macrophage adhesion and migration in diabetic nephropathy. Ren Fail. 2019;41:682–690. doi: 10.1080/0886022X.2019.1632209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wu W, Zhang M, Liu Q, Xue L, Li Y, Ou S. Piwil 2 gene transfection changes the autophagy status in a rat model of diabetic nephropathy. Int J Clin Exp Pathol. 2015;8:10734–10742. [PMC free article] [PubMed] [Google Scholar]
- 41.Ribback S, Cigliano A, Kroeger N, Pilo MG, Terracciano L, Burchardt M, Bannasch P, Calvisi DF, Dombrowski F. PI3K/AKT/mTOR pathway plays a major pathogenetic role in glycogen accumulation and tumor development in renal distal tubules of rats and men. Oncotarget. 2015;6:13036–13048. doi: 10.18632/oncotarget.3675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Huang C, Lin MZ, Cheng D, Braet F, Pollock CA, Chen XM. KCa3.1 mediates dysfunction of tubular autophagy in diabetic kidneys via PI3k/Akt/mTOR signaling pathways. Sci Rep. 2016;6:23884. doi: 10.1038/srep23884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Yang F, Qu Q, Zhao C, Liu X, Yang P, Li Z, Han L, Shi X. Paecilomyces cicadae-fermented Radix astragali activates podocyte autophagy by attenuating PI3K/AKT/mTOR pathways to protect against diabetic nephropathy in mice. Biomed Pharmacother. 2020;129:110479. doi: 10.1016/j.biopha.2020.110479. [DOI] [PubMed] [Google Scholar]
- 44.Zhang Q, Wang X, Cao S, Sun Y, He X, Jiang B, Yu Y, Duan J, Qiu F, Kang N. Berberine represses human gastric cancer cell growth in vitro and in vivo by inducing cytostatic autophagy via inhibition of MAPK/mTOR/p70S6K and akt signaling pathways. Biomed Pharmacother. 2020;128:110245. doi: 10.1016/j.biopha.2020.110245. [DOI] [PubMed] [Google Scholar]
- 45.Li D, Lu Z, Xu Z, Ji J, Zheng Z, Lin S, Yan T. Spironolactone promotes autophagy via inhibiting PI3K/AKT/mTOR signalling pathway and reduce adhesive capacity damage in podocytes under mechanical stress. Biosci Rep. 2016;36:e00355. doi: 10.1042/BSR20160086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yang P, Tian YM, Deng WX, Cai X, Liu WH, Li L, Huang HY. Sijunzi decoction may decrease apoptosis via stabilization of the extracellular matrix following cerebral ischaemia-reperfusion in rats. Exp Ther Med. 2019;18:2805–2812. doi: 10.3892/etm.2019.7878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lee D, Lee SH, Lee M, Lee SH, Shin YJ, Lee JY, Kim H, Kim YS, Song J. Effects of Siwu decoction on chondrocyte proliferation of growth plate in adolescent rats. J Ethnopharmacol. 2019;236:108–113. doi: 10.1016/j.jep.2019.01.048. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.