Abstract
Objectives
This study aims to estimate the prevalence of coronavirus disease 2019 (COVID-19) in the general population of Iran.
Methods
The target population was all Iranian people aged 6 years and older in the country. A stratified random sampling design was used to select 28 314 people from among the individuals registered in the electronic health record systems used in primary health care in Iran. Venous blood was taken from each participant and tested for the IgG antibody against COVID-19. The prevalence of COVID-19 was estimated at provincial and national levels after adjusting for the measurement error of the laboratory test, non-response bias and sampling design.
Results
Of the 28 314 Iranians selected, 11 256 (39.75%) participated in the study. Of these, 5406 (48.0%) were male and 6851 (60.9%) lived in urban areas. The mean (standard deviation) participant age was 35.89 (18.61) years. The adjusted prevalence of COVID-19 until 20 August 2020 was estimated as 14.2% (95% uncertainty interval 13.3%–15.2%), which was equal to 11 958 346 (95% CI 11 211 011–12 746 776) individuals. The adjusted prevalences of infection were 14.6%, 13.8%, 16.6%, 11.7% and 19.4% among men, women, urban population, rural population and individuals aged 60 years or more, respectively. Ardabil, Golestan and Khuzestan provinces had the highest prevalence and Alborz, Hormozgan and Kerman provinces had the lowest.
Conclusions
Based on the study results, a large proportion of the Iranian population had not yet been infected by COVID-19. The observance of hygienic principles and social restrictions should therefore continue until the majority of the population has been vaccinated.
Keywords: Iran, Nationwide, Population-based, Seroprevalence, Severe acute respiratory syndrome coronavirus 2
Introduction
Although daily reports of the number of PCR-confirmed cases of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) are available, the number of diagnosed cases is a function of the number of tests performed per day. For example, in Iran, because of the limited number of laboratory kits available at the beginning of the pandemic, the number of daily tests was very limited and PCR tests were performed only for hospitalized patients in a serious physical condition with suspected coronavirus disease 2019 (COVID-19) [1]. Furthermore, the PCR test has a relatively high false-negative rate, which is affected by the sampling method and the time interval from the onset of the disease [2]. On the other hand, a significant proportion (17%) of COVID-19 patients remain asymptomatic [3,4]. Given the above limitations, it is impossible to rely solely on daily reports of the number of definitively diagnosed cases for pandemic management and policy-making.
Serological tests are used to measure the response of antibodies to the virus and are also able to detect a history of infection in asymptomatic individuals. Seroepidemiological surveys can therefore provide reliable information about the prevalence of the infection and the history of population immunity [[5], [6], [7]].
To date, several seroprevalence studies have been performed in cities and some provinces in Iran and on high-risk populations [8,9]. However, the surveys have had some limitations in terms of the sampling designs and analysis methods, which has raised debate on the validity and generalizability of the findings [[10], [11], [12]]. This study from the Iranian COVID-19 Serological Surveillance programme, which is supported by the Ministry of Health and Medical Education (MOHME) of Iran [13], is the first report of the series of nationwide, population-based serological surveys for COVID-19 that are conducted at regular intervals.
Materials and methods
Study design, population and sampling
The survey was conducted from August to October 2020 in all provinces of Iran to estimate the prevalence of COVID-19 in the country in total and by province, urban/rural area of residence, sex and age group. The target population of the survey was all Iranians aged 6 years and older living in Iran. People with a unique Iranian national identification number registered in the primary health-care electronic health record systems (PHC-EHRS) (SIB, SINA and NAB), who were 6 years of age or older and had sufficient physical ability to attend blood sampling centers were included. Individuals who had contraindications for venous blood sampling were excluded.
A stratified random sampling scheme was used in the survey; each province was considered a stratum. In each province, sampling was conducted through a simple random sampling method using the list of eligible individuals registered in the PHC-EHRS as the sampling frame.
A national sample of 28 314 individuals was recruited. The sample included 858 individuals for each of the provinces in Iran except for Tehran province. For Tehran province, because of the socio-economic status heterogeneity of the population covered by its three universities of medical sciences, three times as many participants were recruited compared with the other provinces. The provincial sample size was calculated based on the estimated COVID-19 prevalence of 33% [14], a relative estimation error of 10%, a confidence interval of 95% and considering a non-response rate of 10%.
Procedures
To invite the participants selected for the survey, the relevant lists and personal profiles in the PHC-EHRS were made available to urban and rural community health workers. These community health workers called the selected people and invited them to each district's specified blood sampling centers for blood sampling.
The qualified blood sampling centers in each district were selected in such a way that the participants would face minimal risk of being infected by SARS-CoV-2 and the centers were easily accessible to the participants. Blood sampling was performed in full compliance with the health protocols and after obtaining written informed consent from the participants. A volume of 10 mL of intravenous blood was taken from each person. The serum samples were transferred to the selected laboratories of the medical universities (not more than 2 hours driving distance) in a three-layer package at a temperature of 4°C–8°C (by icepacks) up to 24 hours after sampling (see Supplementary material for details, Appendix S1).
Measurements and other variables
To determine the IgG antibody against COVID-19 of each sample, serological testing was undertaken using the Iran's Food and Drug Organization-approved SARS-CoV-2 ELISA kit (Pishtaz Teb, Tehran, Iran; catalogue number PT-SARS-COV-2.IgG-96) according to the relevant protocol [13] by the trained staff in the selected qualified laboratories. Participants with a cut-off index of more than 1 were considered positive [13]. The test result was then recorded by the laboratory staff in the PHC-EHRS. Other required variables such as age, gender, province/district of residence and urban/rural area of residence were extracted from the participant's profile in the PHC-EHRS. Fig. S1 in the Supplementary material (Appendix S1) shows the study implementation process.
Ethical considerations
The study protocol was approved by the ethics committee of the National Institute of Health Research of the Islamic Republic of Iran (ethics code: IR.TUMS.NIHR.REC.1399.019). Written informed consent was obtained from all the participants in the study (see Supplementary material for details, Appendix S1).
Statistical analysis
We estimated the prevalence of COVID-19 and its 95% uncertainty interval (UI) at a national and provincial level by urban/rural area of residence, sex and age (6–17, 18–39, 40–59 and ≥ 60 years). A participant was considered ‘positive’ in the presence of anti-SARS-CoV-2 IgG. After correcting the false negatives and false positives of the IgG test results and using post-stratification, inverse probability of response and sampling design weights, a minimum bias estimate of the prevalence was obtained.
We followed three stages in the statistical analysis: (a) correction of the crude (unadjusted) prevalence resulting from the measurement error of the laboratory kit based on the sensitivity and specificity of the kit, (b) conversion of the corrected prevalence of the previous stage into individual data and (c) weighing the individual data of the second stage using post-stratification, response rates and sampling design weights. All of these stages were performed for 16 strata made up of a combination of four age groups, two genders and two urban/rural categories in each province separately (496 categories at a national level) (see Supplementary material for details, Appendix S2). Microsoft Excel, STATA [15] and R [16] software were used in the statistical analysis.
Appendix S3 (see Supplementary material) describes the education, monitoring and supervision of the study. In addition to the reports of the diagnostic accuracy of the Pishtaz Teb kit by the manufacturer (sensitivity 94.1% (32/34) and specificity 98.3% (109/111)), we re-evaluated its accuracy in a separate study (see Supplementary material Appendix S4). The full details of the survey methods are also provided in a protocol article, which has been published elsewhere [13].
Results
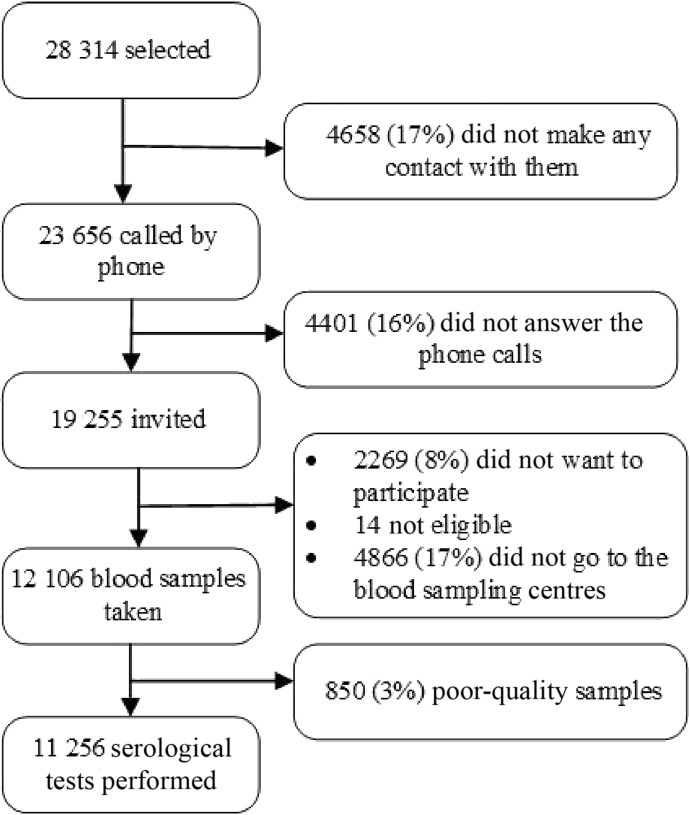
Among the 28 314 Iranians selected to participate in the study from all the provinces, 11 256 (39.75%) participated in the study (Fig. 1 ). The highest rate of non-response was reported from Tehran, Qom and Hamadan provinces (90.4%, 76.9% and 76.5%, respectively), and the provinces of Razavi Khorasan, Sistan and Baluchestan, and Mazandaran had the lowest non-response rates (14.7%, 24.2% and 33.7%, respectively; see Supplementary material, Appendix S5). The multiple logistic regression analysis used to investigate the factors affecting participation in the study revealed a statistically significant association between study participation and province of residence (odds ratio (OR) 1.074, 95% CI 1.071–1.077), 1-year aging (OR 1.007, 95% CI 1.005–1.008), being female (OR 1.19, 95% CI 1.13–1.30) and having a rural residence (OR 2.04, 95% CI 1.93–2.15). To include the province of residence in this model, the provinces of the country were first ranked from large to small based on their non-response rates, then they were given a rank from 1 to 31. Thereafter, the rank of each province based on the non-response rate was entered into the model. The OR of the province of residence indicated the rise in the chance of responding with each increase in the rank of the non-response rate of the province.
Fig. 1.
Profile of participation in the study.
The blood sampling of the study participants started on 3 August 2020, in Tehran province and continued until 31 October 2020 in Kohgiluyeh and Boyer-Ahmad province. Half the participants were sampled before 10 September 2020 and the other half after that date. To increase the response rate, we extended the data collection period by 1 month. The mean (standard deviation) of the age of the study participants in the country was 35.89 (18.61) years (age range 6–109 years). Table 1 and Table S3 (Supplementary material, Appendix S5) show the distribution of the age, sex and area of residence of the study participants across the country and by province, respectively.
Table 1.
Demographic characteristics of the sampled and participant individuals
| Sampled individuals, n (%) | Participants, n (%) | |
|---|---|---|
| Total | 28 314 (100) | 11 256 (100) |
| Age (years) | ||
| 6–17 | 6157 (21.74) | 2302 (20.45) |
| 18–39 | 11 885 (41.97) | 4496 (39.94) |
| 40–59 | 7261 (25.64) | 3100 (27.54) |
| ≥60 | 3011 (10.63) | 1358 (12.06) |
| Gender | ||
| Male | 14 209 (50.18) | 5406 (48.03) |
| Female | 14 105 (49.81) | 5850 (51.97) |
| Area | ||
| Urban | 20 281 (71.63) | 6851 (60.86) |
| Rural | 8033 (28.37) | 4405 (39.13) |
We estimated the sensitivity and specificity of the IgG test of the laboratory kit as 0.74 (95% CI 0.67–0.80) and 0.98 (95% CI 0.96–0.99), respectively (see Supplementary material for details, Appendix S4).
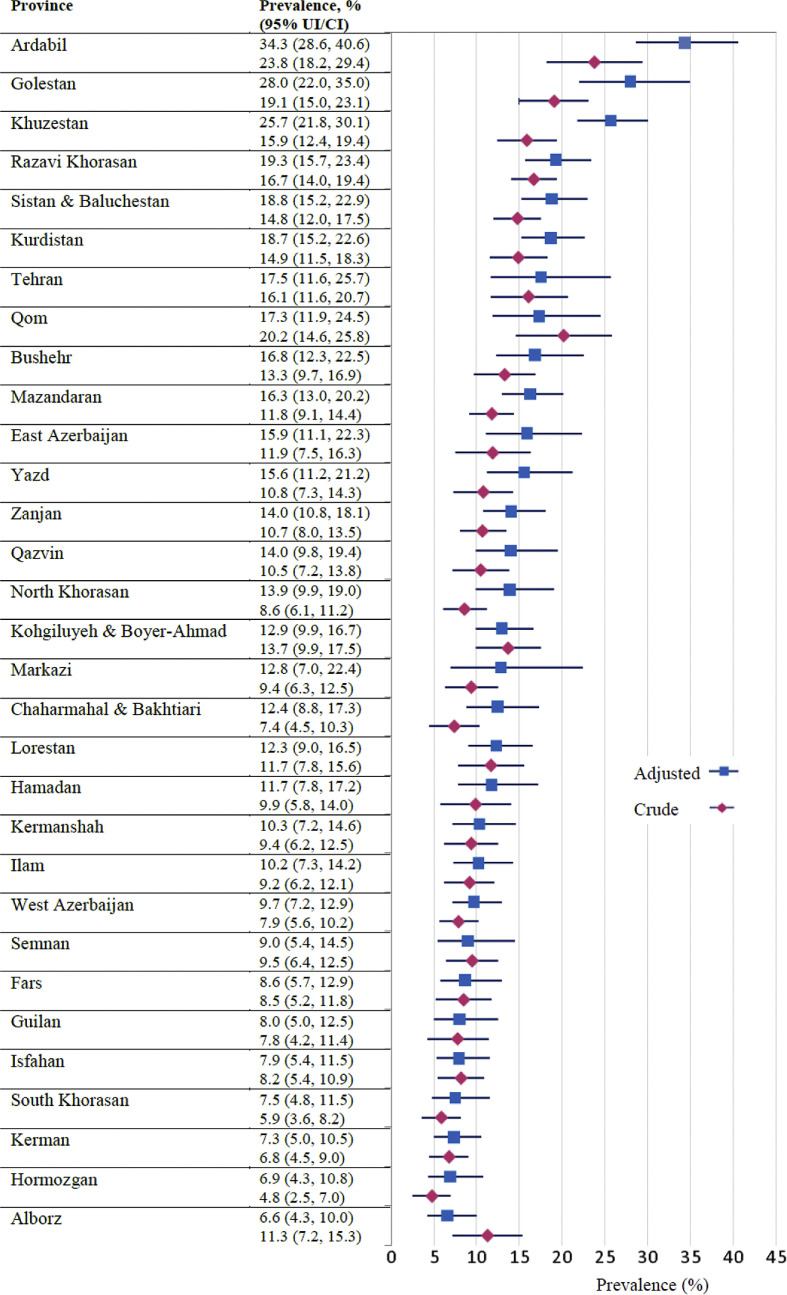
Of the 11 256 participants in the study nationally, the IgG serological test was positive in 1303 (11.6%, 95% CI 11.0%–12.0%). The crude prevalence of COVID-19 based on the IgG serological testing is presented in total in Fig. 2 and separately by age, sex and urban/rural subgroups in Table 2 and Table S4 (see Supplementary material, Appendix S6) for the country and the provinces, respectively. We also provided the national estimates of COVID-19 prevalence after weighting data by post-stratification, inverse probability of response and sampling design weights and without adjusting for the laboratory kit measurement error in Table 2.
Fig. 2.
The crude (unadjusted) prevalence (95% CI) and test measurement error adjusted and weighted prevalence (95% UI) of coronavirus disease 2019 in Iran (by province).
Table 2.
Crude prevalence, weighted prevalence and test measurement error adjusted and weighted prevalence of coronavirus disease 2019 in Iran by age groups, gender and urban/rural area of residence
| Crude prevalence (95% CI) | Weighted prevalence (95% CI) | Test measurement error adjusted and weighted prevalence (95% uncertainty intervals) | |
|---|---|---|---|
| Total | 11.6 (11.0–12.0) | 11.8 (10.9–12.7) | 14.2 (13.3–15.2) |
| Age (years) | |||
| 6–17 | 10.3 (9.1–11.5) | 10.0 (8.5–11.8) | 11.5 (9.9–13.4) |
| 18–39 | 10.3 (9.5–11.2) | 9.6 (8.6–10.7) | 11.6 (10.5–12.8) |
| 40–59 | 13.0 (11.8–14.2) | 11.9 (10.4–13.5) | 14.3 (12.8–16.0) |
| ≥60 | 14.7 (12.8–16.5) | 15.5 (13.2–18.2) | 19.4 (17.0–22.1) |
| Gender | |||
| Male | 11.3 (10.5–12.2) | 12.0 (10.8–13.3) | 14.6 (13.4–15.9) |
| Female | 11.8 (11.0–12.6) | 11.6 (10.3–12.9) | 13.8 (12.5–15.2) |
| Area | |||
| Urban | 12.4 (11.6–13.2) | 13.6 (12.3–14.9) | 16.6 (15.3–18.0) |
| Rural | 10.3 (9.4–11.2) | 9.9 (8.8–11.2) | 11.7 (10.6–13.0) |
The prevalence of COVID-19 in Iran after correcting for the laboratory kit measurement error and weighting data by post-stratification, inverse probability of response and sampling design weights was estimated as 14.2% (95% UI 13.3%–15.2%) at 20 August 2020. It was therefore estimated that from the beginning of the pandemic to this date, more than 11 958 346 (95% CI 11 211 011–12 746 776) of the population over 6 years of age had been infected with COVID-19 in Iran. Nationwide, the infection prevalence was higher among men than women, urban than rural populations and the age groups ≥60 and 40–59 years than other age groups (Table 2). The corrected prevalence estimates by age, gender and urban/rural area of residence for each province are also presented in Table S4 (see Supplementary material, Appendix S6). Ardabil, Golestan, Khuzestan, Razavi Khorasan, and Sistan and Baluchestan provinces had the highest prevalence, and Alborz, Hormozgan, Kerman, South Khorasan and Isfahan provinces had the lowest (see Fig. 2, and see Supplementary material, Fig. S2 in Appendix S7).
Discussion
We estimated the prevalence of COVID-19 in the population aged 6 years and older in Iran from the beginning of the pandemic until 20 August 2020 as 14.2%.
In a study conducted in Iran on the general population in 18 of the most densely populated cities across 17 provinces in the country, the prevalence of COVID-19 from the start of the pandemic until the end of April 2020 was estimated as 17.1% [8,[10], [11], [12]]. The estimated prevalence of the disease would probably have been higher than that in our study because the sampling population of the study only included cities with high population densities—not small and sparsely populated cities and villages—and the study was conducted only in provinces with the highest reported number of COVID-19 cases based on MOHME reports [10]. Although the laboratory kit used in the study was the same as ours, the sensitivity of the IgG test was estimated in different situations (within 2–4 weeks of symptom onset versus 3–16 weeks in our study) and was found to be lower than that in our study (61% versus 74%, respectively). In terms of COVID-19 infection, the IgG titre increased after week 3 of symptom onset, but the samples were used within 2–4 weeks in their sensitivity estimation study. These factors, as well as some of the study's methodological and statistical issues [12], may have contributed to the differences between the results of that study and ours.
In another seroprevalence study in Guilan province, the prevalence of COVID-19 from the beginning of the pandemic until April 2020 was estimated at 22.2% [9], which is significantly higher than our estimate for Guilan province (8.0%). There could be several reasons for this discrepancy, including household sampling compared with the simple random sampling undertaken in our study (because of the high risk of infection of all household members if one of the members is infected), the inclusion of only a number of districts with high and low incidences (based on their hospitalization rate) compared with the inclusion of all districts in the present study, and possibly greater participation of infected individuals in that study compared with ours because of participants' fear of becoming infected in the comprehensive health centers (because of this, 17% of the participants did not participate in that study). More importantly, as Guilan province was among the first provinces hit by the COVID-19 pandemic, it is quite possible that the proportion of infected people with negative serological test results would have been higher than that of many other provinces with delayed peaks. This may have led to a higher underestimation of the infection in Guilan province in our study. In terms of statistical methods, in the multiple bias correction, the true sequence should be the inverse of the bias occurrence sequence [17,18]. Accordingly, in that study, the measurement error had to be corrected first and then the non-response bias; however, the opposite was done. This could also have affected the results.
The first limitation of our study was the use of populations registered in the PHC-EHRS as a sampling framework. Despite the coverage of over 90% of the population of many provinces of the country by these systems, in some provinces, such as Tehran province, the coverage of this system was about 80%. When calculating the prevalence estimates by weighting, the distribution of age, sex, urban/rural residence and the province of residence of the survey sample was matched with the distribution of the provinces and the country population (based on the projection of the population in 2020 by the Statistics Center of Iran). Accordingly, the problem of the incomplete coverage of the sampling framework was partially solved, except for the influential variables not included in our weightings, such as the distribution of socio-economic status. Second, there was a high rate of non-responses in our study. This reduced the precision of the estimates and increased the risk of selection bias. To counter the possibility of selection bias, the elements of such bias relating to gender, age, urban/rural location and the province of residence because of non-responses were corrected during the statistical analysis by inverse probability of response weighting. It is expected that the weighting controlled most of the non-response bias of the present survey. Third, in the laboratory kit sensitivity study, the included patients were individuals who presented at 3–16 weeks following disease symptom onset. This combination of patients did not include those individuals who presented after 16 weeks from symptom onset. Such participants may have provided false-negative results because of a drop in the antibody titres following the extended period since the onset of the symptoms. We may therefore have estimated the sensitivity of the study kit as being slightly higher than the true value. Accordingly, the true prevalence from the start of the pandemic to 20 August 2020 is expected to be slightly higher than our estimate, especially for the provinces with earlier peaks in the pandemic (see Supplementary material, Appendix S8, for further discussion).
Conclusion
According to the study results, a large proportion of the population had not yet been infected. The observance of the principles of hygiene and social restrictions should therefore continue until the majority of the population has been vaccinated.
Authors' contributions
All authors contributed to the study's conception and design. Material preparation, data collection and analysis were performed by AO, KK, SG, DK, MM, SM, SA, MMR, SMH, KT, HN, KA, JT and KM. The first draft of the manuscript was written by KK, AO, FH, SN and AR, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Transparency declaration
SN and AR are the Minister of Health and Medical Education and Deputy Minister of Public Health of MOHME in Iran, respectively. SMS, SMH, SA, MMR, KT, HNT, JST and AO also hold positions in MOHME. The rest of the authors declare no conflict of interest, real or perceived.
Funding
This work was supported by the Deputy of Public Health of the Iranian MOHME and Iran National Institute for Health Research (grant number 990027).
Acknowledgements
This study, the first survey of the Iranian COVID-19 Serological Surveillance programme, was implemented through the active participation of the Network Management Center, the National Comprehensive Health Laboratory, and the Centers for Communicable and Non-Communicable Diseases Control of the MOHME, as well as the departments of health laboratories, communicable diseases and network management at medical universities, and district primary health networks across the country. We are grateful for the cooperation of the staff of the comprehensive health centers, rural health houses, urban health posts and health laboratories throughout the country.
Editor: L. Leibovici
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.cmi.2021.06.002.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Christoph H. WHO Iran Country Office; Tehran: 2020. Coronavirus update: Islamic Repulic of Iran, coronavirus disease 2019 (COVID-19), 01 december 2020. [Google Scholar]
- 2.Arevalo-Rodriguez I., Buitrago-Garcia D., Simancas-Racines D., Zambrano-Achig P., Del Campo R., Ciapponi A., et al. False-negative results of initial RT-PCR assays for COVID-19: a systematic review. PloS One. 2020;15 doi: 10.1371/journal.pone.0242958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Byambasuren O., Cardona M., Bell K., Clark J., McLaws M.-L., Glasziou P. Estimating the extent of asymptomatic COVID-19 and its potential for community transmission: systematic review and meta-analysis. J Assoc Med Microbiol Infect Dis Can. 2020;5:223–234. doi: 10.3138/jammi-2020-0030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.He J., Guo Y., Mao R., Zhang J. Proportion of asymptomatic coronavirus disease 2019: a systematic review and meta-analysis. J Med Virol. 2021;93:820–830. doi: 10.1002/jmv.26326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bryant J.E., Azman A.S., Ferrari M.J., Arnold B.F., Boni M.F., Boum Y., et al. Serology for SARS-CoV-2: apprehensions, opportunities, and the path forward. Sci Immunol. 2020;5 doi: 10.1126/sciimmunol.abc6347. [DOI] [PubMed] [Google Scholar]
- 6.Pollán M., Pérez-Gómez B., Pastor-Barriuso R., Oteo J., Hernán M.A., Pérez-Olmeda M., et al. Prevalence of SARS-CoV-2 in Spain (ENE-COVID): a nationwide, population-based seroepidemiological study. Lancet. 2020;396:535–544. doi: 10.1016/S0140-6736(20)31483-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Theel E.S., Slev P., Wheeler S., Couturier M.R., Wong S.J., Kadkhoda K. The role of antibody testing for SARS-CoV-2: is there one? J Clin Microbiol. 2020;58 doi: 10.1128/JCM.00797-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Poustchi H., Darvishian M., Mohammadi Z., Shayanrad A., Delavari A., Bahadorimonfared A., et al. SARS-CoV-2 antibody seroprevalence in the general population and high-risk occupational groups across 18 cities in Iran: a population-based cross-sectional study. Lancet Infect Dis. 2020 doi: 10.1016/S1473-3099(20)30858-6. epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shakiba M., Nazemipour M., Salari A., Mehrabian F., Nazari S.S., Rezvani S.M., et al. Seroprevalence of SARS-CoV-2 in guilan province, Iran, April 2020. Emerg Infect Dis. 2020;27:636–638. doi: 10.3201/eid2702.201960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Darvishian M., Sharafkhah M., Poustchi H., Malekzadeh R. Estimates of anti-SARS-CoV-2 antibody seroprevalence in Iran – authors' reply. Lancet Infect Dis. 2021 doi: 10.1016/S1473-3099(21)00058-X. epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ghafari M., Kadivar A., Katzourakis A. Estimates of anti-SARS-CoV-2 antibody seroprevalence in Iran. Lancet Infect Dis. 2021 doi: 10.1016/S1473-3099(21)00053-0. epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nazemipour M., Shakiba M., Mansournia M.A. Estimates of anti-SARS-CoV-2 antibody seroprevalence in Iran. Lancet Infect Dis. 2021 doi: 10.1016/S1473-3099(21)00044-X. epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Khalagi K., Gharibzadeh S., Mirab Samiee S., Hashemi S.M., Khalili D., Aghamohamadi S., et al. Nationwide population-based surveys of Iranian COVID-19 Serological Surveillance (ICS) program: the surveys protocol. Med J Islam Repub Iran. 2021;35 doi: 10.47176/mjiri.35.61. epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shakiba M., Nazari S.S.H., Mehrabian F., Rezvani S.M., Ghasempour Z., Heidarzadeh A. Seroprevalence of COVID-19 virus infection in Guilan province, Iran. medRxiv. 2020 doi: 10.3201/eid2702.201960. epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.StataCorp L . StataCorp; College Station, TX: 2017. Stata statistical software: release 15. [Google Scholar]
- 16.R Core Team: a language and environment for statistical computing. R Foundation; Vienna, Austria: 2020. R Foundation for Statistical Computing. [Google Scholar]
- 17.Lash T.L., Fox M.P., Fink A.K. Springer Science & Business Media; Heidelberg: 2011. Applying quantitative bias analysis to epidemiologic data. [Google Scholar]
- 18.Rothman K.J., Greenland S., Lash T.L. Lippincott Williams & Wilkins; Philadelphia: 2008. Modern epidemiology. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.