Abstract
Obesity is strongly and independently associated with an increased risk of severe illness and death from coronavirus disease 2019 (COVID-19). The pathophysiological changes that result from elevated body weight lead to metabolic dysfunction, chronic inflammation, impaired immunological responses, and multisystem disorders, which increase vulnerability to severe illness from COVID-19. While vaccination strategies are under way across the world, the second and third waves of the pandemic, along with the emergence of novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) strains, continue to threaten the stability of medical systems worldwide. Furthermore, evidence from previous pandemics suggests that vaccines are less effective in obese individuals than in their healthy-weight counterparts over the long term. Therefore, a consideration of lifestyle changes that can boost metabolic health and immunity is critical to reduce the risk of complications and severe illness from viral infection. In this review, we discuss the potential mechanisms linking excess body weight with COVID-19 morbidity. We also present evidence that intermittent fasting (IF), a dietary program that has gained popularity in recent years, may be an effective strategy to improve metabolic health and immunity and thus reduce the impact of obesity on COVID-19 morbidity and mortality.
Keywords: obesity, COVID-19, diabetes, intermittent fasting, immune response
Intersection of two pandemics
The COVID-19 pandemic has resulted in a major health crisis with devastating social and economic consequences. Studies from multiple clinical cohorts have shown that obesity is associated with increased complications, disease severity, and mortality on SARS-CoV-2 infection [1., 2., 3.]. A risk assessment analysis of over 900 000 COVID-19 hospitalizations across the USA found that almost two-thirds of these hospitalizations were attributable to cardiometabolic conditions; namely, obesity, diabetes mellitus (DM), hypertension, and heart failure, with obesity accounting for 30% of hospitalizations [4]. COVID-19 patients with elevated body mass index (BMI) were reported to have significantly increased need for invasive mechanical ventilation, a robust proxy for severity of disease [3], were more likely to develop severe pneumonia, exhibited more severe lung pathological changes and injury, and had increased risk of mortality compared with non-obese patients (BMI <25 kg/m2), independent of age, sex, diabetes, and hypertension [1,2,5,6].
Unlike the rapid spread of COVID-19, the obesity pandemic has been slowly gaining momentum for decades. The global prevalence of obesity tripled between 1975 and 2016, and according to the World Health Organization over 650 million adults aged 18 years and over worldwide were obese in 2016 (https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight). Excess adipose tissue is a major source of proinflammatory factors that contribute to chronic systemic inflammation and a host of metabolic disturbances including type 2 DM (T2DM) [7]. Infection with SARS-CoV-2 elicits a wide spectrum of clinical responses (Box 1 ). Severe complications of COVID-19 infection and cytokine storm are more pronounced in individuals with elevated BMI and diabetes [2,8], suggesting that obesity-associated inflammation and metabolic dysfunction exacerbate the severity of illness, thus culminating in the dangerous intersection of two global pandemics.
Box 1. Clinical characteristics of SARS-CoV-2 infection.
Infection with SARS-CoV-2 elicits a wide spectrum of clinical responses, ranging from asymptomatic infection to severe multisystem failure and respiratory distress. During the initial phase of infection, the entry of SARS-CoV-2 into the host cells activates innate immune signaling cascades, leading to increased secretion of proinflammatory cytokines, followed by functional adaptive immune responses that are critical for recovery and long-term immunity [93]. Approximately 1 week after the onset of COVID-19 symptoms, SARS-CoV-2-specific B and T cell responses can be detected in the blood [94]. These initial immune responses play a vital role in viral clearance through antibody production, the direct killing of virus-infected cells, and the production of cytokines that aid in immune cell recruitment [93,94]. While the majority of COVID-19 cases exhibit mild to moderate symptoms and eventually recover, approximately 15% exhibit dysfunctional immune responses characterized by uncontrolled proinflammatory cytokine release [95,96]. This state of hyperinflammation, termed a cytokine storm, is thought to be the main driver of tissue injury, resulting in acute respiratory distress syndrome (ARDS), multiorgan failure, and ultimately death [32]. Thus, compared with individuals who experience a normal recovery, patients with severe COVID-19 infection exhibit markedly increased serum levels of numerous proinflammatory cytokines [e.g., IL-6, IL-1β, IL-2, IL-8, IL-17, granulocyte colony-stimulating factor (G-CSF), granulocyte–macrophage CSF (GM-CSF), leptin, TNF-α] accompanied by immune cell impairment (e.g., higher percentages of pathogenic inflammatory monocytes, which propagate cytokine storms) [8,32,97]. Decreased lymphocyte counts and abnormal lymphocyte functionality, including T cell exhaustion, have also been associated with increased COVID-19 severity [32,93].
Alt-text: Box 1
Currently, a number of COVID-19 vaccine candidates have undergone regulatory approval and are being used for vaccination. However, there remain barriers to achieving herd immunity, which is estimated to require that 60–70% of the population achieve immunity [9]. Factors such as vaccine hesitancy, manufacturing delays, distribution disparities, and the emergence of new, more transmissible SARS-CoV-2 variants that may be resistant to vaccines increase the potential for continued outbreaks [9]. Furthermore, obese populations have a known history of poor response to vaccination [10,11]. An unhealthy metabolic profile, including elevated baseline levels of proinflammatory cytokines and hyperglycemia, is significantly correlated with a dysfunctional immune response to COVID-19 [12,13]. Therefore, now more than ever, the world could benefit from the implementation of lifestyle changes that boost metabolic and immune health to mitigate the impacts of COVID-19.
In this review, we present an overview of the relationship between obesity, adiposity, DM, and COVID-19 severity. We also propose a lifestyle regimen that incorporates IF (Box 2 ) as a promising strategy to improve metabolic health and immune function, potentially reducing the risk of severe illness from COVID-19. Finally, we offer our perspectives on some of the unique benefits and challenges of adopting IF during a global pandemic.
Box 2. Managing obesity through diet and exercise.
Restriction of energy intake has beneficial effects on human health, including extended longevity, disease protection, and delayed aging [74,98]. To address the difficulty of adhering to conventional, calorically restrictive weight-loss regimes, IF has gained popularity as an effective and practical alternative. Recently, IF has been shown to be an effective intervention for body weight regulation and glycemic control in many preclinical and clinical studies [59., 60., 61.,64,65]. Unlike CR, which is defined as a chronic 20–40% reduction of total caloric intake without any changes in meal frequency or timing [74], IF involves recurring periods of fasting, alternating with AL food consumption [56]. Subtypes of IF are described in Figure I.
Regular exercise of moderate to high intensity has been shown to reduce body weight and fat mass; however, evidence suggests that exercise alone generally leads to modest or clinically insignificant weight loss [99]. One trial assessed the effects of combining IF with endurance exercise on 3 days per week for 12 weeks in obese adults [100]. While body weight and BMI were reduced in both groups, the combination of both interventions resulted in a superior reduction in these parameters [100]. Interestingly, fat mass significantly decreased in the IF and combination group but not the exercise group, potentially highlighting the limitations of exercise alone as an antiobesity approach [100]. By contrast, one meta-analysis found that combining IF with resistance-training exercise had either small or inconsistent effects on decreasing body and fat mass compared with resistance training alone [101], indicating a need for further studies to validate the advantages of combining exercise with IF.
As recent evidence suggests that physical activity is protective against severe COVID-19 outcomes [102], a lifestyle that incorporates both physical activity and dietary intervention is likely to be an ideal approach to mitigating the impacts of COVID-19. However, during the COVID-19 pandemic, evidence suggests that social distancing has increased barriers to participation in physical activity [83]. One benefit of IF is that it is a zero-cost and -time approach to managing weight, whereas exercise generally requires time, resources, and access to facilities – barriers associated with inactivity [103]. Thus, while exercise and IF are both attractive options for managing weight, IF may be a more pragmatic approach for some, particularly during the global pandemic. See Figure I for a description and comparison of weight-loss approaches.
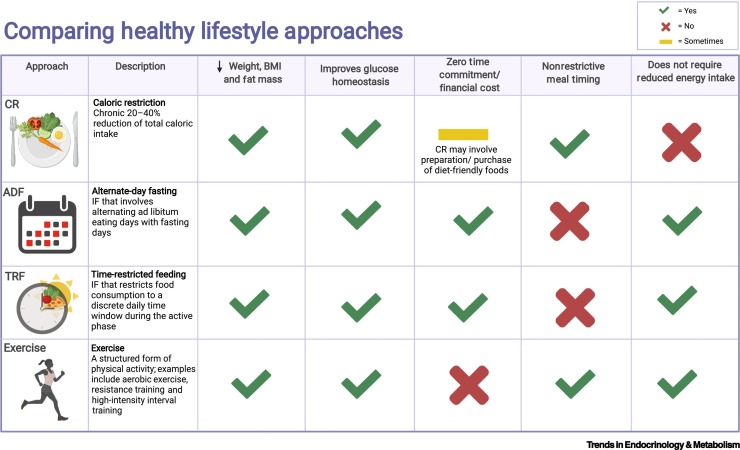
Figure I.
Comparing healthy lifestyle approaches.
Lifestyle interventions that support the management of obesity and promote metabolic health include caloric restriction (CR), alternate-day fasting (ADF), time-restricted feeding (TRF), and physical exercise. These interventions have all been shown to improve body composition and glucose homeostasis [56,63,98,99]. Unlike ADF and TRF [subtypes of intermittent fasting (IF)], exercise regimens require time and, often, financial resources to implement. Depending on the circumstances, this can be a burden associated with CR regimens as well. Moreover, energy intake is chronically restricted with CR, creating unique challenges for long-term adherence. A major benefit of ADF and TRF is that overall energy intake is not restricted, although this is often an unintended consequence of these regimens [59]. Meal timing is, however, restricted with IF, posing distinct adherence challenges. Figure created with BioRender.com. Abbreviation: BMI, body mass index.
Alt-text: Box 2
Hyperglycemia and COVID-19 pathogenesis
Diabetes, a common comorbidity of COVID-19, is associated with a higher risk of excessive uncontrolled inflammatory responses, higher computerized tomography (CT) imaging scores indicating SARS-CoV-2 pneumonia, an increased likelihood of mechanical ventilation, and a shorter duration of survival, even after adjusting for other comorbidities (e.g., age, sex, hypertension, cardiovascular disease) [14., 15., 16.]. Studies point to hyperglycemia, resulting from poor glycemic control, as a driver of COVID-19 progression and poor prognosis [17., 18., 19.]. In one large retrospective, multicenter study of over 7000 patients with COVID-19 in Wuhan, China, among a cohort with COVID-19 and T2DM, individuals with well-controlled blood glucose exhibited significant reductions in adverse outcomes, major organ injuries, all-cause mortality, and overall comorbidities compared with those with poor glycemic control [18]. In other studies, elevated fasting blood glucose at the time of admission was associated with longer hospitalization and higher levels of in-hospital complications and mortality [20., 21., 22.]. Thus, fasting blood glucose at the time of admission was reported to be the best predictor of radiographic imaging of SARS-CoV-2 [21] and was an independent predictor of mortality, regardless of past medical history of diabetes [20., 21., 22.].
Mechanisms linking impaired glucose metabolism with the severity of COVID-19
Increased cellular glucose metabolism may be a key link to the severity of COVID-19 (Figure 1 ). One study showed that increased hexosamine biosynthesis, a glucose metabolic pathway that is activated by influenza infection, led to the O-GlcNAcylation of interferon regulatory factor-5 (IRF-5), which is critical for the induction of cytokine storms during viral infections [23]. Similarly, patients with influenza had more O-GlcNAcylation of IRF-5 and higher blood glucose levels, which were tightly correlated with levels of circulating inflammatory cytokines [23]. Thus, interfering with glucose metabolism and/or glucose utilization pathways may prevent deadly cytokine storms. The expression of IRF-5 and its related proinflammatory cytokines was associated with the severity and prognosis of community-acquired pneumonia influenza virus infection [24]. Furthermore, IRF-5, a marker of metabolic inflammation, has been reported to be significantly elevated in the subcutaneous adipose tissues of diabetic obese patients compared with diabetic lean/overweight individuals [25] and is implicated in macrophage polarization toward a proinflammatory M1 phenotype [25,26] (Figure 1). Inhibition of IRF-5 signaling pathways has been proposed as a potential therapy for severe COVID-19 [27].
Figure 1.
Hyperglycemia-mediated coronavirus disease 2019 (COVID-19) pathogenesis.
Hyperglycemia and poor glycemic control are independently associated with the severity of COVID-19 and increase the risk of mortality [17., 18., 19.,21,22]. The pathophysiological mechanisms for this association may be related to increased activation of glucose metabolic pathways. Increased hexosamine biosynthesis by viral infection contributes to cytokine storms through upregulation of interferon regulatory factor-5 (IRF5) [23,24], a marker of metabolic inflammation that is elevated in adipose tissues of obese individuals [25,26]. Hyperglycemia was associated with increased severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) replication in monocytes from human obese diabetic patients, leading to sustained viral proliferation, induction of cytokine storm, and T cell dysfunction [8]. Furthermore, hyperglycemia may potentiate the glycosylation of the SARS-CoV-2 receptor angiotensin-converting enzyme 2 (ACE2) [28,29]. Figure created with BioRender.com.
Abbreviations: ARDS, acute respiratory distress syndrome; HIF-1a, hypoxia-inducible factor-1α; ROS, reactive oxygen species.
Increased glycolysis exacerbated by elevated glucose levels was shown to promote SARS-CoV-2 replication and proinflammatory cytokine secretion in monocytes [8]. Accordingly, monocytes isolated from obese diabetic patients who were infected with SARS-CoV-2 exhibited increased viral load compared with monocytes from healthy controls, providing further evidence that hyperglycemic individuals may be more susceptible to severe COVID-19 [8]. Furthermore, mitochondrial reactive oxygen species production, induced by SARS-CoV-2 infection, led to the stabilization of hypoxia-inducible factor-1α (HIF-1α), which upregulates glycolytic genes. Thus, uncontrolled hyperglycemia may create a permissive environment for SARS-CoV-2 by increasing glycolytic flux [8].
The relationship between expression of angiotensin-converting enzyme 2 (ACE2), the SARS-CoV-2 receptor, and COVID-19 pathogenesis is unclear (Box 3 ); however, hyperglycemia could lead to increased glycosylation of ACE2, which is thought to be important for its function and may facilitate viral entry [28,29]. Expression of glycosylated ACE2 rather than total ACE2 is associated with viral binding and fusion in the lung [30].
Box 3. Tissue ACE2 expression and relationship to COVID-19 pathogenesis.
SARS-CoV-2 gains entry into host cells through the binding of its envelope spike protein to the human cell receptor ACE2 [104]. The serine protease transmembrane serine protease 2 (TMPRSS2) is essential for ACE2-receptor-mediated binding and viral entry [104]. ACE2 is expressed ubiquitously, with its expression highest in the digestive system, specifically ileal epithelial cells, as well as in the lungs, kidneys, heart, bladder, and adipose tissue [48]. In the lungs, alveolar type II pneumocytes (AT2 cells) and transient bronchial secretory cell populations coexpress ACE2 and TMPRSS2 and are suspected to be the main targets for SARS-CoV-2 infection [105].
ACE2 is an important negative regulator of the renin–angiotensin system (RAS) [106]. Signaling of angiotensin II (AngII) through the AngII type 1 receptor promotes vasoconstrictive responses [106]. ACE2 catalyzes the cleavage of AngII, producing angiotensin 1-7, a vasodilator associated with beneficial regulation of blood pressure homeostasis [106].
Adipose tissue, where ACE2 is highly expressed and is even further upregulated in obesity, has been reported to be a reservoir for SARS-CoV-2 [107]. The relationship between adipocyte expression of ACE2 and the pathogenesis of COVID-19, however, is unknown. ACE2 expression was significantly upregulated in lung tissue from obese mice compared with lean mice and both ACE2 and TMPRSS2 were increased in human lung epithelial cells from obese subjects compared with non-obese individuals [108], which could enhance the entry of SARS-CoV-2 into the cell. However, SARS-CoV-2 infection downregulates the expression of ACE2, a process theorized to contribute to lung injury and increase disease severity [104,109]. Based on observations that conditions reported to be strongly associated with severe symptoms of COVID-19 infection show a degree of ACE2 deficiency, it has been suggested that the downregulation of ACE2 induced by the entry of SARS-CoV-2 into the host cell may be particularly detrimental in individuals with a baseline deficiency of ACE2, who may have a more marked imbalance of the RAS system [104]. Therapies that can increase baseline ACE2 expression may reduce the exaggerated imbalance of the RAS system on COVID-19 infection. Notably, in mice a 4-week IF treatment mitigated HFD-induced increases in systolic blood pressure and increased left ventricular expression of ACE2 associated with beneficial remodeling of cardiac tissue [110]. Furthermore, human studies have also reported positive benefits of IF on hypertension [58,63]. Evidently, the relationship between tissue ACE2 expression and susceptibility to SARS-CoV-2 infection as well as COVID-19 pathogenesis is complex and warrants further investigation.
Alt-text: Box 3
Obesity, immunity, and COVID-19
The accumulation of excess adipose tissue leads to a state of chronic inflammation characterized by a phenotypic shift from an anti-inflammatory to a proinflammatory metabolic milieu with increased secretion of proinflammatory cytokines and dysfunctional immune responses that could play a major role in predisposing individuals to severe COVID-19 [7,31] (Figure 2 ). At least one-third of circulating interleukin (IL)-6, a central driver of inflammation and a strong clinical predictor of disease severity and mortality resulting from COVID-19 infection, is estimated to come from adipose tissue [32,33]. Compared with patients with nonsevere COVID-19, patients with severe COVID-19 had significantly higher baseline circulating IL-6 levels on admission, which increased over the course of the disease, suggesting that baseline circulating IL-6 may be a prognostic marker of COVID-19 [13]. Similarly, high serum IL-6, IL-8, and tumor necrosis factor alpha (TNF-α) at baseline (i.e., at the time of hospitalization) are strong and independent predictors of patient survival [12].
Figure 2.
Impact of obesity on metabolic and immune function.
The accumulation of excess fat is characterized by a phenotypic shift from an anti-inflammatory to a proinflammatory tissue microenvironment, including an increased ratio of proinflammatory (M1-like) to anti-inflammatory (M2-like) macrophages, enhanced polarization toward T helper 1 (Th1), Th17, and CD8+ cytotoxic T cells, and reduced regulatory T cells [7]. These changes result in an increase in the baseline circulating levels of proinflammatory cytokines [e.g., interleukin-6 (IL-6), tumor necrosis factor alpha (TNFα), IL-1β, leptin] [7,33] and the frequency of monocytes. Increased proinflammatory cytokines may exacerbate the risk of virus-induced cytokine storms and drive tissue injury, resulting in acute respiratory distress syndrome (ARDS), multiorgan failure, and ultimately death [12,97]. Attenuated immune cell activation and impaired memory T cell responses in obese populations result in poor outcomes from viral infection and reduce the efficacy of vaccines [10]. Obesity results in impaired gut physiology, including increased barrier permeability and microbiome dysbiosis [51]. As the gastrointestinal system (GI) is thought to be a target for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) entry, poor gut health may be a predisposing factor for the development of severe coronavirus disease 2019 (COVID-19). Figure created with BioRender.com. Abbreviation: T2DM, type 2 diabetes mellitus.
Leptin in COVID-19 pathogenesis
Circulating leptin is well known to be elevated in obese individuals [7,31]. High levels of circulating leptin and selective leptin resistance have been implicated in impaired immune response to viral infection, diminished vaccine response, and chronic low-grade inflammation [34]. Leptin could be a key contributor to acute respiratory distress syndrome (ARDS) and cytokine storms during the advanced stages of COVID-19. Obese mice challenged with H1N1 infection developed more extensive lung inflammation and damage as well as enhanced proinflammatory cytokine expression compared with lean mice [35]. Importantly, leptin inhibition with an antileptin antibody improved the cytokine profiles and survival rates of these obese mice [35]. In one cross-sectional study of intensive care unit (ICU) patients in The Netherlands, patients with COVID-19 had significantly higher serum leptin levels compared with BMI-matched controls (who did not have COVID-19) [36]. Circulating leptin, together with IL-6, CXCL10, and TNF-α, has been reported to be significantly associated with severe COVID-19 illness and inflammatory monocyte activation [37].
Obesity impairs vaccine responses
In a recent study, obese mice inoculated with a human influenza virus showed increased disease severity and viral spread [38]. Moreover, viruses from obese hosts had increased replicative ability, genetic diversity, and virulence [38]. Memory T cell responses, which are critical for conferring immunological memory and serve as major components of vaccination strategies, are impaired in obese individuals [39,40]. Among adults who received influenza vaccines, those who were obese were twice as likely to develop influenza or influenza-like disease as healthy-weight counterparts, despite having similar antibody titers 1 month after influenza vaccination [10]. In addition, CD4+ and CD8+ T cell activation and cytotoxicity, which correlate with disease protection, were defective in cells taken from obese individuals compared with those taken from healthy individuals [10,11]. Obesity was also associated with attenuated influenza vaccine responses, reduced circulating switched memory B cells, and increased proinflammatory late/exhausted dysfunctional B cells [41]. Leptin resistance may be implicated in these responses. Downregulation of the leptin receptor (LEPR) and the upregulation of suppressors of cytokine signaling (e.g., SOCS1, SOCS3), which contribute to leptin resistance, led to decreased maintenance of influenza-specific CD8+ effector memory T cells in the lungs of a diet-induced obese mouse model 84 days post-influenza infection [39].
As COVID-19 vaccines continue to gain widespread regulatory approval globally, these data are critically relevant to the overall long-term success of COVID-19 vaccine strategies. According to published clinical trial results, Pfizer-BioNTech’s COVID-19 vaccine was 95% effective in preventing COVID-19 disease, including in people with obesity (BMI ≥30 kg/m2), who comprised one-third of participants in the clinical trial [42]. Similarly, in the Moderna vaccine trial, vaccine efficacy in participants with severe obesity (BMI ≥40 kg/m2) was comparable (>90%) with that in other participants at low risk [43]. Thus, the evidence suggests that this higher-risk group responds extremely well to the vaccine in the short-term. However, it remains unclear how well obese individuals maintain long-term SARS-CoV-2 vaccine-induced immunity (Figure 2). One recent study, published as a preprint, reported that the Pfizer vaccine was less effective in people with obesity, as healthcare workers with obesity had a significantly lower antibody titer in response to vaccination than those classified as normal weight [44]. Further, compared with lean COVID-19 patients, obese patients were reported to have significantly lower serum levels of SARS-CoV-2-specific IgG antibodies, which negatively correlated with BMI and serum levels of proinflammatory markers [45]. These data, while preliminary, indicate that the dysfunctional immune responses in obesity may extend to COVID-19.
Gut health and COVID-19
Aside from the lungs, the gastrointestinal (GI) system is likely to be another target of SARS-CoV-2, as clinical presentations of GI symptoms in COVID-19 patients have been well documented [46] and viral fecal RNA is known to be present in a significant proportion of COVID-19 patients [47]. Relative to other human tissues, ACE2 is highly expressed in small intestinal enterocytes [48]. In addition to the potential role of the gut as a direct entry point for SARS-CoV-2, poor gut health could have important implications for the severity and outcomes of COVID-19. Gut barrier dysfunction and an altered gut microbiome are hallmark changes associated with obesity [49]. Changes in the composition of the gut microbiome have been postulated to play a role in abnormal immune responses and the severity of COVID-19 [50,51], although substantial evidence for this is forthcoming. Constant interactions between the mammalian host and its commensal gut microbes are crucial for proper immune system development, maintenance, and function [52]. While gut dysbiosis has been widely observed in a number of chronic inflammatory conditions, including obesity, dramatic dysbiosis has also been observed in critically ill patients and is hypothesized to be involved in the pathogenesis of ARDS and sepsis [53]. Accordingly, patients positive for COVID-19 exhibited significant gut dysbiosis, with decreased microbiota diversity and increased opportunistic pathogens that persisted even after clearance of the virus [50,54]. Another study found that COVID-19 patients had significantly altered microbiomes compared with non-COVID-19 controls and that the microbiota composition was associated with disease severity [51]. Given that persistent dysbiosis is frequently observed in COVID-19 patients, improving the parameters of the gut microbiome could ameliorate long-term complications and accelerate recovery from COVID-19 [51].
Increased gut barrier permeability in individuals with obesity and T2DM is associated with leakage of bacteria and bacterial products into the circulation, which leads to metabolic endotoxemia, a major contributor to inflammation [49,55]. It is postulated that enhanced synergistic interactions of bacterial and viral pathogens can potentiate lung injury, suggesting a mechanism by which gut barrier dysfunction in obesity and T2DM may worsen the severity of COVID-19 [49].
IF improves metabolic health
Compelling evidence from human and animal studies indicates that IF regimens (Box 2) promote numerous metabolic benefits, including weight loss, reduced adiposity, and improved glucose homeostasis (Figure 3 ) [56]. One group used a mobile application to track the eating patterns of healthy adults and found that over half of the individuals exhibited erratic eating patterns, consuming food for an average of 15 h/day [57]. Shifting overweight individuals to a time-restricted feeding (TRF) regimen in which they consumed food within a daily 10–11-h window resulted in body weight reductions [57]. Furthermore, restricting daily food intake to an 8–10-h duration resulted in significant reductions in body weight and adiposity and improved glucose tolerance in overweight/obese adults [58,59]; this intervention also reduced abdominal adiposity in individuals who exhibited one or more components of the metabolic syndrome [60]. Similarly, 4-h and 6-h TRF regimens were found to correspond to reductions in body weight, fat mass, markers of oxidative stress, and fasting insulin as well as improved insulin sensitivity in a group of obese individuals [61]. A short, 4-day 6-h early TRF regimen decreased mean 24-h glucose levels in overweight adults [62]. One study reported positive metabolic effects of IF that were independent of weight reduction. In men at risk for T2DM, 5 weeks of 6-h early TRF (in which all calories are consumed early in the day) resulted in improvements in insulin sensitivity, reduced markers of oxidative stress, and reduced blood pressure even in the absence of weight reduction and without change in overall energy expenditure compared with individuals who consumed calories over a 12-h period [63]. Similarly, isocaloric IF improved glucose tolerance in genetically obese mice in the absence of weight or fat mass reduction [64].
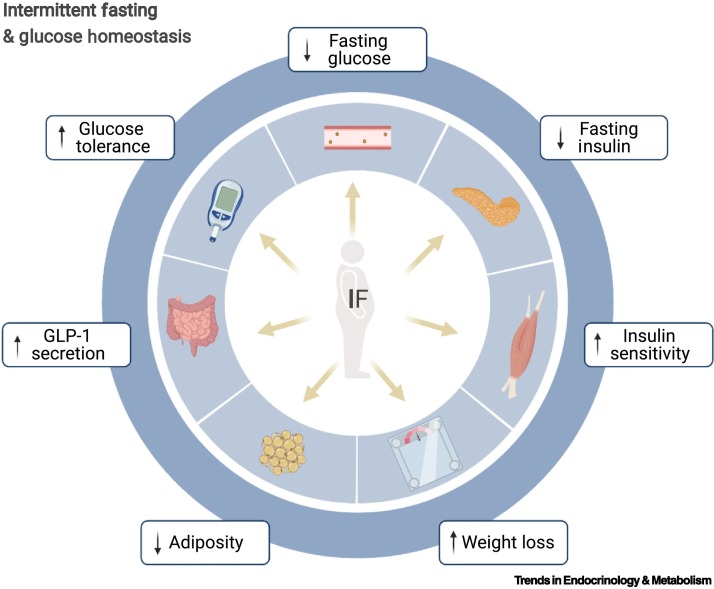
Figure 3.
Intermittent fasting (IF) and glucose homeostasis.
Evidence from both preclinical and clinical studies indicates that various IF regimens improve glucose homeostasis and insulin sensitivity, even in the absence of weight loss or no change in overall energy expenditure [63,65]. We propose that glycemic control by IF could contribute to lowering the risk of severe complications from coronavirus disease 2019 (COVID-19). Figure created with BioRender.com. Abbreviation: GLP-1, glucagon-like peptide 1.
Mice consuming an isocaloric 2:1 IF diet (2 days feeding, 1 day fasting) for 16 weeks were protected from diet-induced obesity and exhibited improved glucose homeostasis and increased insulin sensitivity compared with ad libitum (AL)-fed mice [65]. In mice consuming various obesogenic diets, limiting food access to 8–9 h during the active phase prevented body weight gain and fat accumulation, reversed pre-existing obesity and glucose intolerance, and reduced markers of adipose tissue inflammation in obese mice, regardless of diet and caloric intake [66].
Glucose-lowering dipeptidyl-peptidase-4 (DPP4) inhibitors, which are widely used in the treatment of T2DM, have been proposed as a strategy to offset the severity of COVID-19 infection [67,68]. However, evidence for this application is scant and more studies on this intervention are needed. Notably, DPP4 plays a role in glucose and insulin metabolism and is positively correlated with adipocyte inflammation and insulin resistance [69]. DPP4 inhibitors act as agonists of the gut hormone glucagon-like peptide 1 (GLP-1) receptor, leading to increased circulating GLP-1 levels, improved pancreatic islet responses, and enhanced glucose homeostasis [69] (Figure 3). In obese mice, IF increased the secretion of GLP-1 [64], which could improve insulin sensitivity by essentially mimicking the function of DPP4 inhibitors. Taken together, these findings suggest that through improvements in glucose homeostasis and adiposity, IF and TRF diet regimens could lower the risk of severe COVID-19 complications.
IF reduces inflammation
Evidence for the prognostic role of inflammatory factors and defective immune cells in weakened responses to SARS-CoV-2 infection raises the possibility that strategies to modulate these circulating factors or boost memory T cells could potentially improve the immune response to COVID-19. Concomitant with improvements in glucose homeostasis, some studies have also reported reductions in inflammatory cytokines with IF [70,71], although others report no change in these markers despite other metabolic improvements [58,61,63]. Ramadan fasting, observed by millions of Muslims globally, involves daily diurnal IF for a period of 12–19 h. Both during and outside the month of Ramadan, IF has been shown to decrease circulating TNF-α, IL-1β, and IL-6 levels compared with non-fasting baseline levels, accompanied by significant weight loss and improved glucose metabolism [71,72]. Similarly, intermittent caloric restriction (CR) in rodents significantly reduced circulating and adipose tissue levels of IL-6, TNF-α, insulin-like growth factor 1 (IGF-1), and leptin compared with AL-fed mice [70]. Fasting and IF strategies have been shown to lower leptin levels [66,70] and correct immune defects associated with aging and other conditions characterized by dysregulated leptin signaling, including obesity, metabolic syndrome, and autoimmunity [65,73,74].
In a mouse model of a chronic inflammatory disease, a 6-week alternate-day fasting (ADF) protocol prior to disease onset led to reduced proinflammatory monocyte numbers during disease development and downregulated the expression of the proinflammatory genes TNF-α, IL-1β, CXCL2, and CXCL10 in monocytes compared with an AL feeding group [75]. In addition, short-term fasting reduced monocyte numbers and the frequency of circulating and tissue-resident proinflammatory monocytes in humans and mice, respectively [75].
A CR diet in mice led to the homing of memory T cells to a bone marrow (BM) niche from which these cells were rapidly mobilized during secondary infection, providing enhanced protection against subsequent microbial challenge and prolonging host survival compared with AL-fed mice [76]. Given the similar responses to the CR and IF regimens, it is plausible that IF could also promote BM storage of memory T cells. Multiple cycles of a prolonged 48-h fast led to the renewal and regeneration of hematopoietic stem cells and reduced circulating IGF-1 levels, suggesting that cycles of fasting and refeeding can alter the frequency and composition of circulating immune cells infiltrating peripheral tissues and lead to a reset of the immune system [77]. These findings raise the possibility that IF could potentially reverse immune deficits in obese individuals, thus serving as a preventive, disease-mitigating approach to lower the risk of COVID-19 complications and potentially increase vaccine success (Figure 2).
IF effects on autophagy
As recently reviewed, IF may also boost the host immune system by activating pathways involved in autophagy [78]. While obesity, hypernutrition, and associated lipotoxicity have been associated with the attenuation of autophagic processes [79], CR regimens enhance autophagy through AMP-activated protein kinase negative regulation of mammalian target of rapamycin (mTOR) [78]. Autophagy improves the body’s ability to handle cellular stress and primes the host immune system by strengthening the survival and responses of T and B cells as well as antigen-presenting cells, which could prevent severe complications of SARS-CoV-2 infection [78]. TRF was reported to increase markers of autophagy in overweight humans [62] and decreased oxidative stress [61,63].
IF improves gut health
Much of what we know about the effects of IF on the gut comes from animal studies. Compared with AL-fed controls, a fasting mimicking diet (FMD) has been shown to increase the diversity of the gut microbiota and decrease the Firmicutes:Bacteroidetes ratio; the resulting phenotype is consistent with that of a healthy, lean gut [80]. At the genus level, the FMD group showed decreases in several genera that were positively correlated with fasting blood glucose as well as increases in genera that were negatively correlated with fasting glucose [80]. In a separate study in HFD-induced obese mice, compared with AL-fed controls, ADF-fed mice showed significant changes in gut microbiota composition and significant metabolic improvements that were abolished in microbiota-depleted mice [81]. Finally, IF has been shown to reduce widespread systemic inflammation in a gut-dependent manner [73]. Taken together, these studies suggest that IF-mediated benefits for gut health could improve metabolic and immune health, thus ameliorating outcomes for COVID-19 patients.
Concluding remarks and future perspectives
As we have reviewed, obesity and T2DM increase the incidence of severe complications, cytokine storms, and mortality from COVID-19. Disparities in vaccine distribution, vaccine hesitancy, and the emergence of SARS-CoV-2 variants are barriers to achieving population immunity, increasing the potential for continued outbreaks, hospitalizations, and mortality resulting from COVID-19 illness [9]. With obesity on the rise and potential for continued COVID-19 outbreaks globally, it is more important than ever to develop strategies that could reduce inflammation, improve glycemic control, and boost immune health. Naturally, this necessitates curbing overnutrition and maladaptive eating habits that lead to poor metabolic health. We have highlighted the numerous beneficial effects of IF on adiposity, metabolic health, glucose homeostasis, the antiviral immune response, long-term immunity, inflammatory markers, and the gut. One critical question remains surrounding the long-term efficacy of COVID-19 vaccination in obese individuals (see Outstanding questions), as memory T cell activation and responses to vaccination are blunted in this population [10,11,44]. In a recent study, an obesity-related microenvironment in mice promoted the emergence of more virulent pathogenic influenza virus strains that induced greater disease severity in lean hosts [38]. This raises the intriguing possibility that obese metabolic phenotypes could impact future generations of novel COVID-19 variants.
Outstanding questions.
Due to the relative speed at which COVID-19 vaccines were developed, tested, and approved, there are currently no long-term data on their efficacy in obese populations. Will long-term immunity from COVID-19 vaccination be hampered in obese individuals, as has been observed for other vaccines? We predict that obese individuals would experience quicker declines in vaccine-induced immunity than lean individuals, making this population at higher risk for infection and resulting in negative consequences for the overall success of COVID-19 vaccination programs worldwide.
Most clinical trials of IF have examined small samples of participants over relatively short periods of time. Evidence on whether adherence can be sustained in the long term is forthcoming. Furthermore, much of the evidence for the impact of IF and/or CR on adaptive immunity, memory T cells, immune system reset, and gut health, which are critical parameters of antiviral immunity, have come from preclinical studies. This raises the question of whether IF strategies can impact immunopathological mechanisms specific to SARS-CoV-2. There is a critical need for studies testing the role of dietary strategies, including IF, in improving antiviral immunity and reducing the severity of COVID-19.
In this review, we provide evidence that IF may help to reduce the risk of severe COVID-19 in obese, diabetic, and/or metabolically unhealthy individuals. However, it is less clear whether these putative benefits extend to lean, metabolically healthy individuals. Given that the risk of developing severe COVID-19 symptoms is greatly increased in older individuals as well as in those with weakened immune systems and pre-existing conditions, it is conceivable that IF could worsen disease conditions in these individuals who may already be frail and vulnerable to nutritional deficiencies. Ultimately, further investigation is needed to clarify whether the putative benefits and safety profile of IF are dependent on the host’s caloric intake and/or baseline fat mass and metabolic health.
Alt-text: Outstanding questions
IF in the time of COVID?
As a result of the COVID-19 pandemic, billions of individuals continue to practice various measures of social distancing and imposed lockdowns. This has resulted in increased sedentary lifestyles and prolonged periods of social isolation, which have been reported to be detrimental to physical and mental health [82,83]. Globally, eating habits have changed drastically during the pandemic [84., 85., 86.]. Generally, more time spent cooking and eating at home, less time spent exercising, boredom, and stress have all given rise to overeating and mindless snacking [85., 86., 87.]. Pandemic-induced lifestyle changes have also led some to increase their focus on healthy eating and seek out strategies to boost their immunity and health [84,87]. Given the current social climate, with limited opportunities for physical activity, dietary changes such as IF regimens may be a practical way to curb unhealthy eating habits and maximize metabolic health during the pandemic. In particular, TRF regimens have been associated with decreased feelings of hunger throughout the day, curbing non-hungry snacking and mindless eating [57,63]. Furthermore, IF is also associated with improvements in mood and emotional well-being [88,89] (Figure 4 , Key figure). We propose that TRF or IF may be more sustainable than traditional diets because it allows individuals to maintain their usual diets with shorter eating durations. In many studies, reducing the duration of eating each day led to unintentional decreases in caloric intake, leading to weight loss and associated metabolic benefits [57,59]. However, IF has been shown to have positive metabolic consequences, such as improved glucose homeostasis, even in the absence of weight reduction and/or no changes in overall energy expenditure [63., 64., 65.]. It must be cautioned that most of the evidence for these findings are from small controlled clinical trials, and an absence of significant effects of TRF on weight and metabolic parameters has also been reported [90]. Importantly, IF regimens have not been addressed in relation to COVID-19 illness (see Outstanding questions). Ultimately, as outlined in Box 2, a combination of nutritional lifestyles and physical activity are likely to be most effective.
Figure 4.
Key figure. Intermittent fasting (IF) in the time of coronavirus disease 2019 (COVID-19).
While the number of vaccine candidates for COVID-19 continues to increase, many hurdles exist surrounding global distribution, access, and vaccine hesitancy [9]. Thus, the feasibility of tackling this global pandemic and achieving population herd immunity remains unclear. In addition to these challenges, second and third waves of infection have resulted in stay-at-home orders and imposed lockdowns for billions of individuals worldwide. The toll of widespread social isolation includes a shift to more sedentary lifestyles, drastic changes in eating and lifestyle habits, and increased stress culminating in declining physical and mental health [82,84,86,87]. We propose that the incorporation of healthy eating strategies, such as IF, that are reported to boost immune health, restore metabolic function, and improve glucose homeostasis is especially important during this time, particularly for obese individuals. IF strategies are particularly suited to the unique social climate of stay-at-home orders, as they do not require major alterations in diet composition but instead restrict eating to discrete time windows. Figure created with BioRender.com.
Finally, there are some concerns about whether IF strategies are appropriate for everyone. It is possible that certain dietary regimens involving reduced caloric intake could harness immunosuppressive effects, thus weakening the anti-SARS-CoV-2 immune response [91]. There are also limited reports on the effects of IF in elderly populations. Although IF has been shown to have physical and mental benefits in an elderly population (>65 years of age) [92], caution must be exercised when recommending fasting strategies to improve health for the elderly and those with weakened immunity (see Outstanding questions).
In conclusion, while IF is an exciting and growing field of investigation, further research efforts are needed to establish the effects of IF on antiviral immunity, particularly in the context of COVID-19. Given that IF has shown profound promise in improving metabolic health, we argue that now is a better time than ever to adopt this healthy lifestyle strategy (Figure 4).
Acknowledgments
Acknowledgments
We thank Lauren Pickel, Christy Yeung, and Irisa Qianwen Shi for critical reading of the manuscript and helpful suggestions and Irisa Qianwen Shi for help creating figures. This research was funded by the Canadian Institutes of Health Research (CIHR, PJT-162083) of Canada, a Sun Life Financial New Investigator Award, and a Reuben and Helene Dennis Scholar Award of the Banting and Best Diabetic Centre (BBDC), University of Toronto.
Declaration of interests
None are declared.
Author contributions
K.N.E., J.P., and H-K.S. conceived and designed the research. All authors agreed to the final version of the manuscript.
References
- 1.Gao M., et al. Associations between body-mass index and COVID-19 severity in 6.9 million people in England: a prospective, community-based, cohort study. Lancet Diabetes Endocrinol. 2021;9:350–359. doi: 10.1016/S2213-8587(21)00089-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kang Z., et al. Obesity is a potential risk factor contributing to clinical manifestations of COVID-19. Int. J. Obes. (Lond.) 2020;44:2479–2485. doi: 10.1038/s41366-020-00677-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Simonnet A., et al. High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity (Silver Spring) 2020;28:1195–1199. doi: 10.1002/oby.22831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.O’Hearn M., et al. Coronavirus disease 2019 hospitalizations attributable to cardiometabolic conditions in the United States: a comparative risk assessment analysis. J. Am. Heart Assoc. 2021;10 doi: 10.1161/JAHA.120.019259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hendren N.S., et al. Association of body mass index and age with morbidity and mortality in patients hospitalized with COVID-19: results from the American Heart Association COVID-19 Cardiovascular Disease Registry. Circulation. 2021;143:135–144. doi: 10.1161/CIRCULATIONAHA.120.051936. [DOI] [PubMed] [Google Scholar]
- 6.Guerson-Gil A., et al. Sex-specific impact of severe obesity in the outcomes of hospitalized patients with COVID-19: a large retrospective study from the Bronx, New York. Eur. J. Clin. Microbiol. Infect. Dis. 2021 doi: 10.1007/s10096-021-04260-z. Published online May 6, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Alarcon P.C., et al. Adipocyte inflammation and pathogenesis of viral pneumonias: an overlooked contribution. Mucosal Immunol. 2021 doi: 10.1038/s41385-021-00404-8. Published online May 6, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Codo A.C., et al. Elevated glucose levels favor SARS-CoV-2 infection and monocyte response through a HIF-1α/glycolysis-dependent axis. Cell Metab. 2020;32:437–446.e435. doi: 10.1016/j.cmet.2020.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Aschwanden C. Five reasons why COVID herd immunity is probably impossible. Nature. 2021;591:520–522. doi: 10.1038/d41586-021-00728-2. [DOI] [PubMed] [Google Scholar]
- 10.Neidich S.D., et al. Increased risk of influenza among vaccinated adults who are obese. Int. J. Obes. (Lond.) 2017;41:1324–1330. doi: 10.1038/ijo.2017.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Paich H.A., et al. Overweight and obese adult humans have a defective cellular immune response to pandemic H1N1 influenza A virus. Obesity (Silver Spring) 2013;21:2377–2386. doi: 10.1002/oby.20383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Del Valle D.M., et al. An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat. Med. 2020;26:1636–1643. doi: 10.1038/s41591-020-1051-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Laguna-Goya R., et al. IL-6-based mortality risk model for hospitalized patients with COVID-19. J. Allergy Clin. Immunol. 2020;146:799–807.e799. doi: 10.1016/j.jaci.2020.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cariou B., et al. Phenotypic characteristics and prognosis of inpatients with COVID-19 and diabetes: the CORONADO study. Diabetologia. 2020;63:1500–1515. doi: 10.1007/s00125-020-05180-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Guo W., et al. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab. Res. Rev. 2020;36 doi: 10.1002/dmrr.3319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yan Y., et al. Clinical characteristics and outcomes of patients with severe Covid-19 with diabetes. BMJ Open Diabetes Res. Care. 2020;8 doi: 10.1136/bmjdrc-2020-001343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bode B., et al. Glycemic characteristics and clinical outcomes of COVID-19 patients hospitalized in the United States. J. Diabetes Sci. Technol. 2020;14:813–821. doi: 10.1177/1932296820924469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhu L., et al. Association of blood glucose control and outcomes in patients with COVID-19 and pre-existing type 2 diabetes. Cell Metab. 2020;31:1068–1077.e1063. doi: 10.1016/j.cmet.2020.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Smith S.M., et al. Impaired glucose metabolism in patients with diabetes, prediabetes, and obesity is associated with severe COVID-19. J. Med. Virol. 2021;93:409–415. doi: 10.1002/jmv.26227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Carrasco-Sanchez F.J., et al. Admission hyperglycaemia as a predictor of mortality in patients hospitalized with COVID-19 regardless of diabetes status: data from the Spanish SEMI-COVID-19 Registry. Ann. Med. 2021;53:103–116. doi: 10.1080/07853890.2020.1836566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Iacobellis G., et al. Admission hyperglycemia and radiological findings of SARS-CoV2 in patients with and without diabetes. Diabetes Res. Clin. Pract. 2020;164 doi: 10.1016/j.diabres.2020.108185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wang S., et al. Fasting blood glucose at admission is an independent predictor for 28-day mortality in patients with COVID-19 without previous diagnosis of diabetes: a multi-centre retrospective study. Diabetologia. 2020;63:2102–2111. doi: 10.1007/s00125-020-05209-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wang Q., et al. O-GlcNAc transferase promotes influenza A virus-induced cytokine storm by targeting interferon regulatory factor-5. Sci. Adv. 2020;6 doi: 10.1126/sciadv.aaz7086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang X., et al. Expression levels of interferon regulatory factor 5 (IRF5) and related inflammatory cytokines associated with severity, prognosis, and causative pathogen in patients with community-acquired pneumonia. Med. Sci. Monit. 2018;24:3620–3630. doi: 10.12659/MSM.910756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sindhu S., et al. Enhanced adipose expression of interferon regulatory factor (IRF)-5 associates with the signatures of metabolic inflammation in diabetic obese patients. Cells. 2020;9:730. doi: 10.3390/cells9030730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dalmas E., et al. Irf5 deficiency in macrophages promotes beneficial adipose tissue expansion and insulin sensitivity during obesity. Nat. Med. 2015;21:610–618. doi: 10.1038/nm.3829. [DOI] [PubMed] [Google Scholar]
- 27.Stoy N. Involvement of interleukin-1 receptor-associated kinase 4 and interferon regulatory factor 5 in the immunopathogenesis of SARS-CoV-2 infection: implications for the treatment of COVID-19. Front. Immunol. 2021;12 doi: 10.3389/fimmu.2021.638446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Walls A.C., et al. Structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein. Cell. 2020;181:281–292.e286. doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mehdipour A.R., Hummer G. Dual nature of human ACE2 glycosylation in binding to SARS-CoV-2 spike. Proc. Natl Acad. Sci. U. S. A. 2021;118 doi: 10.1073/pnas.2100425118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Brufsky A. Hyperglycemia, hydroxychloroquine, and the COVID-19 pandemic. J. Med. Virol. 2020;92:770–775. doi: 10.1002/jmv.25887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Porsche C.E., et al. Obesity results in adipose tissue T cell exhaustion. JCI Insight. 2021;6 doi: 10.1172/jci.insight.139793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen G., et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J. Clin. Invest. 2020;130:2620–2629. doi: 10.1172/JCI137244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mohamed-Ali V., et al. Subcutaneous adipose tissue releases interleukin-6, but not tumor necrosis factor-alpha, in vivo. J. Clin. Endocrinol. Metab. 1997;82:4196–4200. doi: 10.1210/jcem.82.12.4450. [DOI] [PubMed] [Google Scholar]
- 34.Alti D., et al. Emergence of leptin in infection and immunity: scope and challenges in vaccines formulation. Front. Cell. Infect. Microbiol. 2018;8:147. doi: 10.3389/fcimb.2018.00147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhang A.J., et al. Leptin mediates the pathogenesis of severe 2009 pandemic influenza A(H1N1) infection associated with cytokine dysregulation in mice with diet-induced obesity. J. Infect. Dis. 2013;207:1270–1280. doi: 10.1093/infdis/jit031. [DOI] [PubMed] [Google Scholar]
- 36.van der Voort P.H.J., et al. Leptin levels in SARS-CoV-2 infection related respiratory failure: a cross-sectional study and a pathophysiological framework on the role of fat tissue. Heliyon. 2020;6 doi: 10.1016/j.heliyon.2020.e04696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang J., et al. Leptin correlates with monocytes activation and severe condition in COVID-19 patients. J. Leukoc. Biol. 2021 doi: 10.1002/JLB.5HI1020-704R. Published online January 6, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Honce R., et al. Obesity-related microenvironment promotes emergence of virulent influenza virus strains. mBio. 2020;11 doi: 10.1128/mBio.03341-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Karlsson E.A., et al. Diet-induced obesity in mice reduces the maintenance of influenza-specific CD8+ memory T cells. J. Nutr. 2010;140:1691–1697. doi: 10.3945/jn.110.123653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Rebeles J., et al. Obesity-induced changes in T-cell metabolism are associated with impaired memory T-cell response to influenza and are not reversed with weight loss. J. Infect. Dis. 2019;219:1652–1661. doi: 10.1093/infdis/jiy700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Frasca D., et al. Obesity decreases B cell responses in young and elderly individuals. Obesity (Silver Spring) 2016;24:615–625. doi: 10.1002/oby.21383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Polack F.P., et al. Safety and efficacy of the BNT162b2 mRNA covid-19 vaccine. N. Engl. J. Med. 2020;383:2603–2615. doi: 10.1056/NEJMoa2034577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Baden L.R., et al. Efficacy and safety of the mRNA-1273 SARS-CoV-2 vaccine. N. Engl. J. Med. 2021;384:403–416. doi: 10.1056/NEJMoa2035389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pellini R., et al. Obesity may hamper SARS-CoV-2-specific vaccine. medRxiv. 2021 doi: 10.1101/2021.02.24.21251664. Published online February 26, 2021. [DOI] [Google Scholar]
- 45.Frasca D., et al. Influence of obesity on serum levels of SARS-CoV-2-specific antibodies in COVID-19 patients. PLoS One. 2021;16 doi: 10.1371/journal.pone.0245424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Mao R., et al. Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: a systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2020;5:667–678. doi: 10.1016/S2468-1253(20)30126-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wang W., et al. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020;323:1843–1844. doi: 10.1001/jama.2020.3786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Li M.Y., et al. Expression of the SARS-CoV-2 cell receptor gene ACE2 in a wide variety of human tissues. Infect. Dis. Poverty. 2020;9:45. doi: 10.1186/s40249-020-00662-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kruglikov I.L., et al. Obesity and diabetes as comorbidities for COVID-19: underlying mechanisms and the role of viral-bacterial interactions. Elife. 2020;9 doi: 10.7554/eLife.61330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gu S., et al. Alterations of the gut microbiota in patients with coronavirus disease 2019 or H1N1 influenza. Clin. Infect. Dis. 2020;71:2669–2678. doi: 10.1093/cid/ciaa709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Yeoh Y.K., et al. Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut. 2021;70:608–706. doi: 10.1136/gutjnl-2020-323020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Belkaid Y., Harrison O.J. Homeostatic immunity and the microbiota. Immunity. 2017;46:562–576. doi: 10.1016/j.immuni.2017.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Dickson R.P. The microbiome and critical illness. Lancet Respir. Med. 2016;4:59–72. doi: 10.1016/S2213-2600(15)00427-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zuo T., et al. Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology. 2020;159:944–955.e948. doi: 10.1053/j.gastro.2020.05.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mohammad S., Thiemermann C. Role of metabolic endotoxemia in systemic inflammation and potential interventions. Front. Immunol. 2020;11 doi: 10.3389/fimmu.2020.594150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lee J.H., et al. Intermittent fasting: physiological implications on outcomes in mice and men. Physiology (Bethesda) 2020;35:185–195. doi: 10.1152/physiol.00030.2019. [DOI] [PubMed] [Google Scholar]
- 57.Gill S., Panda S. A smartphone app reveals erratic diurnal eating patterns in humans that can be modulated for health benefits. Cell Metab. 2015;22:789–798. doi: 10.1016/j.cmet.2015.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wilkinson M.J., et al. Ten-hour time-restricted eating reduces weight, blood pressure, and atherogenic lipids in patients with metabolic syndrome. Cell Metab. 2020;31:92–104.e105. doi: 10.1016/j.cmet.2019.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Gabel K., et al. Effects of 8-hour time restricted feeding on body weight and metabolic disease risk factors in obese adults: a pilot study. Nutr. Healthy Aging. 2018;4:345–353. doi: 10.3233/NHA-170036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kesztyus D., et al. Adherence to time-restricted feeding and impact on abdominal obesity in primary care patients: results of a pilot study in a pre-post design. Nutrients. 2019;11:2854. doi: 10.3390/nu11122854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cienfuegos S., et al. Effects of 4- and 6-h time-restricted feeding on weight and cardiometabolic health: a randomized controlled trial in adults with obesity. Cell Metab. 2020;32:366–378.e363. doi: 10.1016/j.cmet.2020.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Jamshed H., et al. Early time-restricted feeding improves 24-hour glucose levels and affects markers of the circadian clock, aging, and autophagy in humans. Nutrients. 2019;11:1234. doi: 10.3390/nu11061234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Sutton E.F., et al. Early time-restricted feeding improves insulin sensitivity, blood pressure, and oxidative stress even without weight loss in men with prediabetes. Cell Metab. 2018;27:1212–1221.e1213. doi: 10.1016/j.cmet.2018.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kim Y.H., et al. Thermogenesis-independent metabolic benefits conferred by isocaloric intermittent fasting in ob/ob mice. Sci. Rep. 2019;9:2479. doi: 10.1038/s41598-019-39380-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kim K.H., et al. Intermittent fasting promotes adipose thermogenesis and metabolic homeostasis via VEGF-mediated alternative activation of macrophage. Cell Res. 2017;27:1309–1326. doi: 10.1038/cr.2017.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chaix A., et al. Time-restricted feeding is a preventative and therapeutic intervention against diverse nutritional challenges. Cell Metab. 2014;20:991–1005. doi: 10.1016/j.cmet.2014.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Solerte S.B., et al. Sitagliptin treatment at the time of hospitalization was associated with reduced mortality in patients with type 2 diabetes and COVID-19: a multicenter, case-control, retrospective, observational study. Diabetes Care. 2020;43:2999–3006. doi: 10.2337/dc20-1521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Mirani M., et al. Impact of comorbidities and glycemia at admission and dipeptidyl peptidase 4 inhibitors in patients with type 2 diabetes with COVID-19: a case series from an academic hospital in Lombardy, Italy. Diabetes Care. 2020;43:3042–3049. doi: 10.2337/dc20-1340. [DOI] [PubMed] [Google Scholar]
- 69.Deacon C.F. Physiology and pharmacology of DPP-4 in glucose homeostasis and the treatment of type 2 diabetes. Front. Endocrinol. (Lausanne) 2019;10:80. doi: 10.3389/fendo.2019.00080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Dogan S., et al. The influence of different calorie restriction protocols on serum pro-inflammatory cytokines, adipokines and IGF-I levels in female C57BL6 mice: short term and long term diet effects. Meta Gene. 2017;12:22–32. doi: 10.1016/j.mgene.2016.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Faris M.A., et al. Intermittent fasting during Ramadan attenuates proinflammatory cytokines and immune cells in healthy subjects. Nutr. Res. 2012;32:947–955. doi: 10.1016/j.nutres.2012.06.021. [DOI] [PubMed] [Google Scholar]
- 72.Almeneessier A.S., et al. The effects of diurnal intermittent fasting on proinflammatory cytokine levels while controlling for sleep/wake pattern, meal composition and energy expenditure. PLoS One. 2019;14 doi: 10.1371/journal.pone.0226034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Cignarella F., et al. Intermittent fasting confers protection in CNS autoimmunity by altering the gut microbiota. Cell Metab. 2018;27:1222–1235.e1226. doi: 10.1016/j.cmet.2018.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Longo V.D., Mattson M.P. Fasting: molecular mechanisms and clinical applications. Cell Metab. 2014;19:181–192. doi: 10.1016/j.cmet.2013.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Jordan S., et al. Dietary intake regulates the circulating inflammatory monocyte pool. Cell. 2019;178:1102–1114.e1117. doi: 10.1016/j.cell.2019.07.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Collins N., et al. The bone marrow protects and optimizes immunological memory during dietary restriction. Cell. 2019;178:1088–1101.e1015. doi: 10.1016/j.cell.2019.07.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Cheng C.W., et al. Prolonged fasting reduces IGF-1/PKA to promote hematopoietic-stem-cell-based regeneration and reverse immunosuppression. Cell Stem Cell. 2014;14:810–823. doi: 10.1016/j.stem.2014.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hannan M.A., et al. Intermittent fasting, a possible priming tool for host defense against SARS-CoV-2 infection: crosstalk among calorie restriction, autophagy and immune response. Immunol. Lett. 2020;226:38–45. doi: 10.1016/j.imlet.2020.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Namkoong S., et al. Autophagy dysregulation and obesity-associated pathologies. Mol. Cells. 2018;41:3–10. doi: 10.14348/molcells.2018.2213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Wei S., et al. Intermittent administration of a fasting-mimicking diet intervenes in diabetes progression, restores beta cells and reconstructs gut microbiota in mice. Nutr. Metab. (Lond.) 2018;15:80. doi: 10.1186/s12986-018-0318-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Li G., et al. Intermittent fasting promotes white adipose browning and decreases obesity by shaping the gut microbiota. Cell Metab. 2017;26:672–685.e674. doi: 10.1016/j.cmet.2017.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Pietrabissa G., Simpson S.G. Psychological consequences of social isolation during COVID-19 outbreak. Front. Psychol. 2020;11:2201. doi: 10.3389/fpsyg.2020.02201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Stockwell S., et al. Changes in physical activity and sedentary behaviours from before to during the COVID-19 pandemic lockdown: a systematic review. BMJ Open Sport Exerc. Med. 2021;7 doi: 10.1136/bmjsem-2020-000960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Di Renzo L., et al. Eating habits and lifestyle changes during COVID-19 lockdown: an Italian survey. J. Transl. Med. 2020;18:229. doi: 10.1186/s12967-020-02399-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Giacalone D., et al. Reported changes in dietary habits during the COVID-19 lockdown in the Danish population: the Danish COVIDiet Study. Front. Nutr. 2020;7 doi: 10.3389/fnut.2020.592112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Rolland B., et al. Global changes and factors of increase in caloric/salty food intake, screen use, and substance use during the early COVID-19 containment phase in the general population in France: survey study. JMIR Public Health Surveill. 2020;6:19630. doi: 10.2196/19630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ruiz-Roso M.B., et al. COVID-19 lockdown and changes of the dietary pattern and physical activity habits in a cohort of patients with type 2 diabetes mellitus. Nutrients. 2020;12:2327. doi: 10.3390/nu12082327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Hussin N.M., et al. Efficacy of fasting and calorie restriction (FCR) on mood and depression among ageing men. J. Nutr. Health Aging. 2013;17:674–680. doi: 10.1007/s12603-013-0344-9. [DOI] [PubMed] [Google Scholar]
- 89.Igwe O., et al. A review of effects of calorie restriction and fasting with potential relevance to depression. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2020 doi: 10.1016/j.pnpbp.2020.110206. Published online December 11, 2020. [DOI] [PubMed] [Google Scholar]
- 90.Lowe D.A., et al. Effects of time-restricted eating on weight loss and other metabolic parameters in women and men with overweight and obesity: the TREAT randomized clinical trial. JAMA Intern. Med. 2020;180:1491–1499. doi: 10.1001/jamainternmed.2020.4153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Duriancik D.M., et al. Age, calorie restriction, and age of calorie restriction onset reduce maturation of natural killer cells in C57Bl/6 mice. Nutr. Res. 2018;55:81–93. doi: 10.1016/j.nutres.2018.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Anton S.D., et al. The effects of time restricted feeding on overweight, older adults: a pilot study. Nutrients. 2019;11:1500. doi: 10.3390/nu11071500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Garcia L.F. Immune response, inflammation, and the clinical spectrum of COVID-19. Front. Immunol. 2020;11:1441. doi: 10.3389/fimmu.2020.01441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Suthar M.S., et al. Rapid generation of neutralizing antibody responses in COVID-19 patients. Cell Rep. Med. 2020;1 doi: 10.1016/j.xcrm.2020.100040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Tang Y., et al. Cytokine storm in COVID-19: the current evidence and treatment strategies. Front. Immunol. 2020;11:1708. doi: 10.3389/fimmu.2020.01708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Wu Z., McGoogan J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020;323:1239–1242. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 97.Zhang D., et al. Frontline science: COVID-19 infection induces readily detectable morphologic and inflammation-related phenotypic changes in peripheral blood monocytes. J. Leukoc. Biol. 2021;109:13–22. doi: 10.1002/JLB.4HI0720-470R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Golbidi S., et al. Health benefits of fasting and caloric restriction. Curr. Diab. Rep. 2017;17:123. doi: 10.1007/s11892-017-0951-7. [DOI] [PubMed] [Google Scholar]
- 99.Bellicha A., et al. Effect of exercise training on weight loss, body composition changes, and weight maintenance in adults with overweight or obesity: an overview of 12 systematic reviews and 149 studies. Obes. Rev. 2021 doi: 10.1111/obr.13256. Published online May 6, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Bhutani S., et al. Alternate day fasting and endurance exercise combine to reduce body weight and favorably alter plasma lipids in obese humans. Obesity (Silver Spring) 2013;21:1370–1379. doi: 10.1002/oby.20353. [DOI] [PubMed] [Google Scholar]
- 101.Ashtary-Larky D., et al. Effects of intermittent fasting combined with resistance training on body composition: a systematic review and meta-analysis. Physiol. Behav. 2021;237 doi: 10.1016/j.physbeh.2021.113453. [DOI] [PubMed] [Google Scholar]
- 102.Sallis R., et al. Physical inactivity is associated with a higher risk for severe COVID-19 outcomes: a study in 48 440 adult patients. Br. J. Sports Med. 2021 doi: 10.1136/bjsports-2021-104080. Published online April 13, 2021. [DOI] [PubMed] [Google Scholar]
- 103.Herazo-Beltran Y., et al. Predictors of perceived barriers to physical activity in the general adult population: a cross-sectional study. Braz. J. Phys. Ther. 2017;21:44–50. doi: 10.1016/j.bjpt.2016.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Verdecchia P., et al. The pivotal link between ACE2 deficiency and SARS-CoV-2 infection. Eur. J. Intern. Med. 2020;76:14–20. doi: 10.1016/j.ejim.2020.04.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Lukassen S., et al. SARS-CoV-2 receptor ACE2 and TMPRSS2 are primarily expressed in bronchial transient secretory cells. EMBO J. 2020;39 doi: 10.15252/embj.20105114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Donoghue M., et al. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1-9. Circ. Res. 2000;87:E1–E9. doi: 10.1161/01.res.87.5.e1. [DOI] [PubMed] [Google Scholar]
- 107.Kruglikov I.L., Scherer P.E. The role of adipocytes and adipocyte-like cells in the severity of COVID-19 infections. Obesity (Silver Spring) 2020;28:1187–1190. doi: 10.1002/oby.22856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Al Heialy S., et al. Regulation of angiotensin- converting enzyme 2 in obesity: implications for COVID-19. Front. Physiol. 2020;11 doi: 10.3389/fphys.2020.555039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Datta P.K., et al. SARS-CoV-2 pandemic and research gaps: understanding SARS-CoV-2 interaction with the ACE2 receptor and implications for therapy. Theranostics. 2020;10:7448–7464. doi: 10.7150/thno.48076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Camelo L., et al. Intermittent fasting exerts beneficial metabolic effects on blood pressure and cardiac structure by modulating local renin-angiotensin system in the heart of mice fed high-fat or high-fructose diets. Nutr. Res. 2019;63:51–62. doi: 10.1016/j.nutres.2018.12.005. [DOI] [PubMed] [Google Scholar]