Abstract
Pembrolizumab (mAb to PD-1) has been recently approved for the therapy of pretreated urothelial cancer. Despite the efficacy, it is often accompanied by unpredictable and sometime severe immune-related (ir) adverse events (AEs). Here, we report the clinical and immune–biological characterization of a patient with a metastatic bladder cancer who developed myositis signs (M) and a myasthenia-like syndrome (MLS) during treatment with pembrolizumab. The patient presented an autoimmunity-associated HLA haplotype (HLA-A*02/HLA-B*08/HLA-C*07/HLA-DRB1*03) and experienced an increase in activated CD8 T-cells along the treatment. The symptomatology regressed after pembrolizumab discontinuation and a pyridostigmine and steroids-based therapy. This is the first report of concurrent M and MLS appearance in cancer patients receiving pembrolizumab. More efforts are needed to define early the risk and the clinical meaning of irAEs in this setting.
Keywords: urothelial cancer, PD1-checkpoint inhibitors, class-I/II HLA, myasthenia
1. Introduction
Tumor-infiltrating T-cell rescue by using PD-1/PDL1 immune checkpoint-blocking mAbs represents an innovative therapeutic strategy for a number of different malignancies, including NSCLC, malignant melanoma and urothelial, as well as head and neck cancer [1,2,3]. Although very active in a number of patients in terms of clinical benefit and survival, this treatment could be complicated by severe immune-related (ir) adverse events (AEs) (irAEs) [4]. Interestingly, these immunological events have been occasionally reported to correlate with a good outcome in different malignancies including malignant melanoma, colorectal cancer and mNSCLC [5,6,7,8]. However, less is known for patients with urological malignancies, where the study of potential correlations between outcome and irAEs under immune-checkpoint inhibitors treatment is still in its infancy (it has been recently demonstrated that patients responding to anti-PD-1/L1 treatment are more likely to develop irAEs) [9].
It has been hypothesized that these adverse events are strictly related to the immunological nature of the treatment that, other than impairing tolerance against cancer (private) antigens, even breaks endogenous tolerance to multiple and different (physiological) self-antigens [4,10,11]. At present, however, no clear classification of tumor-specific irAEs has been proposed, thus the report of unexpected and infrequent irAEs is eagerly welcome.
2. Case Report
VG is a seventy-two year-old male with a good performance status, with a moderate smoking habit, no professional or familiar risk of bladder malignancy or autoimmunity and hypertension in medical treatment, who was diagnosed with a locally advanced bladder cancer for which he underwent radical cystectomy and locoregional lymphadenectomy on October 2018. Histological examination detected an undifferentiated urothelial carcinoma infiltrating the muscularis mucosae with no nodal involvement. A pre-operatory radiological study excluded the presence of disseminated disease. He was therefore staged as T3N0Mo and no adjuvant chemotherapy was recommended. On February 2019 he become symptomatic with a rise in sacral and pelvic pain associated with weight loss and ankle swelling. A CT/PET scan detected the presence of soft tissue relapse within the left ileo-psoas muscle as well as bone (costs, pelvis, ischiopubic bones) and iliac, lombo-aortic and inguinal node metastasis. This patient was therefore addressed to palliative chemotherapy with cisplatin and gemcitabine that was ineffective in controlling the disease and was suspended just after three cycles upon instrumental demonstration of disease progression (from 20 March to 10 May 2019). Subsequently, he received radiation therapy (8Gy) and then metronomic chemotherapy with oral vinorelbine (20 mg three times a week) from 1 September to 10 December 2019, before new disease progression was demonstrated (Figure 1A). He therefore was addressed to immunotherapy, receiving pembrolizumab at a dose of 200 mg every three weeks, starting on 17 December 2019. He showed a fast benefit in terms of quality of life and pain decrease and swelling after the first administration. The treatment was well tolerated and he could receive three treatment cycles with no alterations in blood tests or symptoms suggestive for irAEs, with the exception of a mild arthromyalgia (g1), stiffness and moderate asthenia. A few days after the third treatment course he developed left eyelid ptosis, which progressively worsened until becoming bilateral within a few days. In parallel, diplopia with limitation of eye movements, proximal weakness to the limbs and dysphonia also occurred. A biochemical blood test showed no change in inflammatory markers and blood cell counts, a sudden rise in cell lysis enzymes and increased levels of anti-nuclear antibodies (ANA) (Table 1).
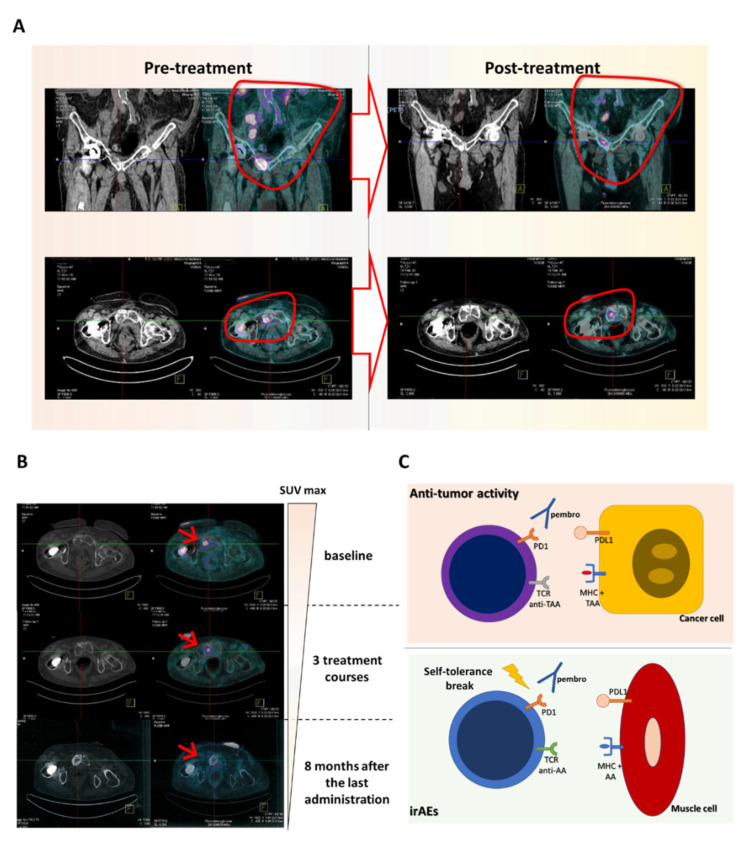
Figure 1.
(A) CT/PET scan performed prior to and after three treatment cycles of pembrolizumab showed a good disease control with volume reduction in all the involved sites. (B) CT/PET scan performed during the follow-up (after 8 months from the last pembrolizumab administration) demonstrated a further reduction in cancer metabolism coupled with a sclerotic reaction. Arrows indicate the point where SUV reduction is more evident. (C) Cartoon representing the possible mechanism responsible for (muscle) immune-related adverse events (irAEs). Pembro: pembrolizumab; AA: auto-antigens; TAA: tumor-associated antigens.
Table 1.
Immune and inflammatory parameters modulation along pembrolizumab treatment.
| Blood Tests | Baseline | Post-Treatment | Follow-Up |
|---|---|---|---|
| Inflammatory markers | |||
| CRP (mg/L) | 40.7 (#) | 15.5 (#) | 6.59 (#) |
| ESR (mm/h) | 104 (#) | 50 (#) | 24 (#) |
| Cell lysis enzymes | |||
| AST (U/L) | 35 | 445 (#) | 19 |
| ALT (U/L) | 15 | 109 (#) | 7 |
| LDH (U/L) | 154 | 4403 (#) | 177 |
| CK (U/L) | ND | 4403 (#) | 100 |
| CK MB(U/L) | ND | 189 (#) | 9 |
| Troponin I (ng/mL) | ND | 5.42 (#) | <0.012 |
| Auto-antibodies | |||
| ASMA | ND | negative | negative |
| ENA | ND | negative | negative |
| ANA | ND | 1/1280 (#) | 1/160 (#) |
| Anti-peroxidase (U/l) | ND | 94.64 (#) | 125.80 (#) |
| Anti-ChR-abs (U/mL) | ND | 2.8 (#) | 2.68 (#) |
| Anti-MuSK (U/mL) | ND | <0.4 | <0.4 |
| CD4+/CD8+ T cell ratio | 1.50 | 1.1 | ND |
| Activated T cells % (HLA-DR+) | 21 | 27 | ND |
| Activated T cells % (HLA-DR+) |
# out of normality range.
The study of his peripheral blood mononuclear cells performed by flow cytometry revealed a decline in CD4+/CD8+ T-cell ratio and a rise in activated T cells (Table 1).
As long as these symptoms were rising, this patient was hospitalized. Contrast-enhanced brain MRI, hearth ultrasonography, single-fiber electromyography and repetitive nerve stimulation test, lumbar puncture and subsequent cerebrospinal fluid analysis were carried out (Table 2) in order to exclude possible metastases, traumas or infectious diseases. We then performed laboratory research of M- and MSL-specific immunoglobulins, detecting the presence of AAbs to the muscle acetylcholine receptors (AChR-abs) (while AAbs to muscle-specific kinase (MuSK) remained within the normal ranges) commonly associated with myasthenia. A HLA genotype study of this patient finally confirmed the expression of a high-risk autoimmunity haplotype (Table 2). Additionally, this finding was accompanied by a reduction in the CD4/CD8 ratio and an increase in the presence of activated HLADR+ cells.
Table 2.
Instrumental evaluation and HLA determination at adverse event onset.
| Patient’s Characteristics | |
|---|---|
| Contrast-enhanced brain MRI | No CNS metastases |
| Single-fiber EMG and repetitive nerve stimulation test | Within normal ranges |
| Cerebrospinal fluid | Within normal ranges |
| hearth ultrasonography | Within normal ranges |
| HLA haplotype | HLA-A*02/*29 HLA-B*08/*35 HLA-C*04/*07 DRB1*03/*04 |
The patient discontinued immunotherapy and received a proper treatment, including intravenous dexamethasone (16 mg a day) associated with oral pyridostigmine bromide (60 mg three times a day), that led to full recovery of the patient within fifteen days and hospital discharge in a good clinical shape. The patient was re-evaluated by a CT/PET scan, showing a partial antitumor response (Figure 1A). He remained free of symptoms for a further six months, showing no blood cell, instrumental or biochemical alterations (Table 1). He continued the treatment with oral prednisone (10 mg a day) and pyridostigmine bromide until July 2020 when a distal urethral soft tissue relapse required palliative radiation therapy (30Gy in ten fraction). Our patient is currently in a good clinical shape, free from systemic progression with no further neurologic impairment or other irAEs or signs of other common adverse events, sixteen months after the last pembrolizumab dose on February 2020 (Figure 1B).
3. Discussion
PD1/PDL1 immune-checkpoint blockade is often complicated by irAEs whose occurrence is unpredictable and heterogenous in term of physiopathology, symptoms and severity. The majority of them usually present mild (g1–2 irAEs) articular, endocrine, mucosal and cutaneous symptoms, while 10–14% develop more severe irAEs (g3–4), including pneumonia, hypophysitis and neurological impairments [4,11,12,13]. It is reasonable to believe that irAEs present substantial differences correlated with gender, class I/II HLA-typing, malignant histology and metastatic sites, as well as with the different PD-1/PDL1 mAb (used alone or in a combination strategy) [14,15,16,17]. All these factors may affect the release and processing of different tissue-specific auto-antigens and relative immune-inflammatory response, sufficient to ignite an autoimmune response. Among them, idiopathic inflammatory myopathies represent rare autoimmune diseases defined by muscle weakness and heterogeneous systemic organic involvement. Interestingly, a irAEs involving muscles have been reported to occur in cancer patients receiving immune-checkpoint blockade in a variable range of 1–43% [4,18,19,20,21,22,23,24]. Additionally, several fatal events (especially if associated with heart muscle damage) have been reported [25].
Tumor microenvironment is characterized by an unstable equilibrium where cells belonging to different arms of the immune system (including lymphocytes and myeloid-derived cells) create a tolerogenic niche where malignant cells could freely proliferate [26,27,28,29,30,31,32,33,34]. This aspect closely resembles pro-inflammatory/tolerogenic phenomena happening around an injured tissue, a mechanism which physiologically protects us from the development of autoimmune diseases. Accordingly, the development of irAEs after PD1/PDL1 inhibition could indeed rely (from a pathogenetic point of view) on mechanisms of “epitope-sharing” between normal and cancer cells or “epitope-spreading” (release in the microenvironment of self-antigens as a consequence of tumor cell death) in the cross-activation of T cells against health tissues [24], which finally cause an inappropriate immune-mediated inflammatory response to muscle cells (Figure 1C). This point, in particular, warrants further investigation; in the view of a broader context of “private” tumor-associated antigens which could trigger irAEs, the advent of whole exome sequencing methodologies and computational docking strategies, able to derive the final 3D structure of the HLA–epitope association, will clarify these aspects in the future [35].
Additionally, the specific rise in AABs and specific genetic risk partially related to class I and II HLA should been taken carefully into account [15,36,37]. Indeed, the expression of HLA-B*08 as well as DRB1*03, *07 or *11 are all correlated with the development of myositis in not-neoplastic patients [38]. Specifically, the risk of early onset myasthenia has been correlated with the presence of HLA-A*01 and *02 together with B*08, C*05 and *07 and DRB1*03, *07 and *13 [37].
On the other hand, acquired myasthenia gravis is another rare antibody-mediated autoimmune disease caused by impaired neuromuscular transmission that leads to abnormal muscle fatigability, affecting in some cases only the eye muscles (ocular MG), but in most cases several muscle groups. It often occurs as para-neoplastic syndromes associated with thymoma, endocrine neoplasms and malignant melanoma [39,40]. The etiology seems to be very complex and includes both environmental and genetic risk factors mainly involving (again) Class I and II HLA complexes. Indeed, the risk of early onset myasthenia has been correlated with the germinal expression of HLA-A*01 and *02 together with B*08, C*05 and *07 and DRB1*03, *07 and *13 [37]. The occurrence of symptoms in these patients is strictly correlated with the presence of AChR-abs (80–85% of the cases) and/or to MuSK AAbs (10–50%) [41,42]. The prognosis of the disease depends on the possibility to treat the “triggering” malignancy as well as on the response to therapeutics such as steroids, immunoglobulins or plasmapheresis. However, a recent pooled analysis suggested an overall dismal outcome for these patients [12,43,44]. The occurrence of myasthenia with myositis signs has been sporadically observed in patients with malignant melanoma, NSCLC and kidney cancer receiving PD-1/PDL1 blockade but, so far, it has never been recorded in patients with urothelial cancer.
In our patient, the hypothesis that both myositis- and myasthenia-like syndrome are consequent to pembrolizumab treatment is sustained by clinical as well as biological/biochemical considerations. In the first place, the symptoms occurred a few months after the beginning of the treatment in concomitance with a significant anti-tumor response, progressive rise in muscular cell lysis markers such as ALT, AST, CK, CK, MB and LDH, a significant serum conversion for ANA, anti-thyroglobulin Abs and finally, AChR-abs that confirmed the diagnosis of myasthenia. Additionally, all symptoms disappeared upon corticosteroid treatment and pembrolizumab discontinuation excluding the hypothesis of a paraneoplastic condition. Our analysis of his HLA genotype revealed the presence of an autoimmune haplotype presenting class I HLA alleles associated with an increased risk of both myositis (A*02, B*08, C*07, DRB1*03) and myasthenia gravis (B*08 and DRB1*03) [36,37].
4. Conclusions
The fact that irAEs symptoms occurred in parallel with an increased serum level of AAbs and clinical response to pembrolizumab suggests that the equilibrium between antitumor activity and tolerance to auto-antigens was affected by the immunological treatment. On the basis of these considerations, we believe that the research of autoimmune-associated HLA haplotypes prior starting PD-1/PDL1 immune-checkpoint mAb administration could be a safe modality to warn physicians of an increased risk of life-threatening irAEs.
Author Contributions
C.B. and P.C. composed the manuscript and performed the literature review; V.D., V.C. and N.D.C. performed the acquisition and analysis of laboratory data for the work; R.M.A., M.C. and P.C. critically revised and interpreted the data; R.M.A., G.B., A.M., R.M., E.I., G.O., D.M., A.C.F., R.E.S., R.G., G.R. and P.C. took care of the patient from the clinical point of view. U.A., C.B. and P.C. fully revised and improved the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by a grant of the ARCO (Associazione Riuniti Calabria Oncologia) non-profit patients’ association and by “the rare tumor network project 2017”; subproject: “Prevention and monitoring of immune-related adverse events in cancer patients receiving immunological treatments” financed by the Calabria Region.
Informed Consent Statement
Written informed consent was obtained from the individual for the publication of any potentially identifiable images or data included in this article.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Pardoll D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer. 2012;12:252–264. doi: 10.1038/nrc3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Xia L., Liu Y., Wang Y. PD-1/PD-L1 Blockade Therapy in Advanced Non-Small-Cell Lung Cancer: Current Status and Future Directions. Oncologist. 2019;24:S31–S41. doi: 10.1634/theoncologist.2019-IO-S1-s05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhang F., Liu Z., Liang J., Zhang F., Wu K., Zhou C., Lu Y., Wang X. The efficacy and safety of immunotherapy targeting the PD-1 pathway for advanced urothelial carcinoma: A meta-analysis of published clinical trials. Clin. Transl. Oncol. :2020. doi: 10.1007/s12094-020-02316-8. [DOI] [PubMed] [Google Scholar]
- 4.Day D., Hansen A.R. Immune-Related Adverse Events Associated with Immune Checkpoint Inhibitors. BioDrugs. 2016;30:571–584. doi: 10.1007/s40259-016-0204-3. [DOI] [PubMed] [Google Scholar]
- 5.Correale P., Tagliaferri P., Fioravanti A., Del Vecchio M.T., Remondo C., Montagnani F., Rotundo M.S., Ginanneschi C., Martellucci I., Francini E., et al. Immunity feedback and clinical outcome in colon cancer patients undergoing chemoimmunotherapy with gemcitabine + FOLFOX followed by subcutaneous granulocyte macrophage colony-stimulating factor and aldesleukin (GOLFIG-1 Trial) Clin. Cancer Res. 2008;14:4192–4199. doi: 10.1158/1078-0432.CCR-07-5278. [DOI] [PubMed] [Google Scholar]
- 6.Giannicola R., D’Arrigo G., Botta C., Agostino R., Del Medico P., Falzea A.C., Barbieri V., Staropoli N., Del Giudice T., Pastina P., et al. Early blood rise in auto-antibodies to nuclear and smooth muscle antigens is predictive of prolonged survival and autoimmunity in metastatic-non-small cell lung cancer patients treated with PD-1 immune-check point blockade by nivolumab. Mol. Clin. Oncol. 2019;11:81–90. doi: 10.3892/mco.2019.1859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Leonardi G.C., Candido S., Falzone L., Spandidos D.A., Libra M. Cutaneous melanoma and the immunotherapy revolution (Review) Int. J. Oncol. 2020;57:609–618. doi: 10.3892/ijo.2020.5088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rodriguez-Cerdeira C., Carnero Gregorio M., Lopez-Barcenas A., Sanchez-Blanco E., Sanchez-Blanco B., Fabbrocini G., Bardhi B., Sinani A., Guzman R.A. Advances in Immunotherapy for Melanoma: A Comprehensive Review. Mediat. Inflamm. 2017;2017:3264217. doi: 10.1155/2017/3264217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Maher V.E., Fernandes L.L., Weinstock C., Tang S., Agarwal S., Brave M., Ning Y.M., Singh H., Suzman D., Xu J., et al. Analysis of the Association Between Adverse Events and Outcome in Patients Receiving a Programmed Death Protein 1 or Programmed Death Ligand 1 Antibody. J. Clin. Oncol. 2019;37:2730–2737. doi: 10.1200/JCO.19.00318. [DOI] [PubMed] [Google Scholar]
- 10.Mohn N., Beutel G., Gutzmer R., Ivanyi P., Satzger I., Skripuletz T. Neurological Immune Related Adverse Events Associated with Nivolumab, Ipilimumab, and Pembrolizumab Therapy-Review of the Literature and Future Outlook. J. Clin. Med. 2019;8:1777. doi: 10.3390/jcm8111777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mohn N., Suhs K.W., Gingele S., Angela Y., Stangel M., Gutzmer R., Satzger I., Skripuletz T. Acute progressive neuropathy-myositis-myasthenia-like syndrome associated with immune-checkpoint inhibitor therapy in patients with metastatic melanoma. Melanoma Res. 2019;29:435–440. doi: 10.1097/CMR.0000000000000598. [DOI] [PubMed] [Google Scholar]
- 12.Cuzzubbo S., Javeri F., Tissier M., Roumi A., Barlog C., Doridam J., Lebbe C., Belin C., Ursu R., Carpentier A.F. Neurological adverse events associated with immune checkpoint inhibitors: Review of the literature. Eur. J. Cancer. 2017;73:1–8. doi: 10.1016/j.ejca.2016.12.001. [DOI] [PubMed] [Google Scholar]
- 13.Wang D.Y., Salem J.E., Cohen J.V., Chandra S., Menzer C., Ye F., Zhao S., Das S., Beckermann K.E., Ha L., et al. Fatal Toxic Effects Associated With Immune Checkpoint Inhibitors: A Systematic Review and Meta-analysis. JAMA Oncol. 2018;4:1721–1728. doi: 10.1001/jamaoncol.2018.3923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Correale P., Botta C., Martino E.C., Ulivieri C., Battaglia G., Carfagno T., Rossetti M.G., Fioravanti A., Guidelli G.M., Cheleschi S., et al. Phase Ib study of poly-epitope peptide vaccination to thymidylate synthase (TSPP) and GOLFIG chemo-immunotherapy for treatment of metastatic colorectal cancer patients. Oncoimmunology. 2016;5:e1101205. doi: 10.1080/2162402X.2015.1101205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Correale P., Saladino R.E., Giannarelli D., Sergi A., Mazzei M.A., Bianco G., Giannicola R., Iuliano E., Forte I.M., Calandruccio N.D., et al. HLA Expression Correlates to the Risk of Immune Checkpoint Inhibitor-Induced Pneumonitis. Cells. 2020;9:1964. doi: 10.3390/cells9091964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cusi M.G., Botta C., Pastina P., Rossetti M.G., Dreassi E., Guidelli G.M., Fioravanti A., Martino E.C., Gandolfo C., Pagliuchi M., et al. Phase I trial of thymidylate synthase poly-epitope peptide (TSPP) vaccine in advanced cancer patients. Cancer Immunol. Immunother. 2015;64:1159–1173. doi: 10.1007/s00262-015-1711-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nardone V., Pastina P., Giannicola R., Agostino R., Croci S., Tini P., Pirtoli L., Giordano A., Tagliaferri P., Correale P. How to Increase the Efficacy of Immunotherapy in NSCLC and HNSCC: Role of Radiation Therapy, Chemotherapy, and Other Strategies. Front. Immunol. 2018;9:2941. doi: 10.3389/fimmu.2018.02941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cappelli L.C., Gutierrez A.K., Bingham C.O., 3rd, Shah A.A. Rheumatic and Musculoskeletal Immune-Related Adverse Events Due to Immune Checkpoint Inhibitors: A Systematic Review of the Literature. Arthritis Care Res. (Hoboken) 2017;69:1751–1763. doi: 10.1002/acr.23177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kennedy L.B., Salama A.K.S. A Review of Immune-Mediated Adverse Events in Melanoma. Oncol. Ther. 2019;7:101–120. doi: 10.1007/s40487-019-0096-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu Y.H., Zang X.Y., Wang J.C., Huang S.S., Xu J., Zhang P. Diagnosis and Management of Immune Related Adverse Events (irAEs) in Cancer Immunotherapy. Biomed. Pharmacother. 2019;120:109437. doi: 10.1016/j.biopha.2019.109437. [DOI] [PubMed] [Google Scholar]
- 21.Simeone E., Grimaldi A.M., Festino L., Trojaniello C., Vitale M.G., Vanella V., Palla M., Ascierto P.A. Immunotherapy in metastatic melanoma: A novel scenario of new toxicities and their management. Melanoma Manag. 2019;6:MMT30. doi: 10.2217/mmt-2019-0005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Xiao Y., Zeng L., Shen Q., Zhou Z., Mao Z., Wang Q., Zhang X., Li Y., Yao W. Diagnosis and Treatment of Rheumatic Adverse Events Related to Immune Checkpoint Inhibitors. J. Immunol. Res. 2020;2020:2640273. doi: 10.1155/2020/2640273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhong H., Zhou J., Xu D., Zeng X. Rheumatic immune-related adverse events induced by immune checkpoint inhibitors. Asia Pac. J. Clin. Oncol. :2020. doi: 10.1111/ajco.13346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Solimando A.G., Crudele L., Leone P., Argentiero A., Guarascio M., Silvestris N., Vacca A., Racanelli V. Immune Checkpoint Inhibitor-Related Myositis: From Biology to Bedside. Int. J. Mol. Sci. 2020;21:3054. doi: 10.3390/ijms21093054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Martinez-Calle N., Rodriguez-Otero P., Villar S., Mejias L., Melero I., Prosper F., Marinello P., Paiva B., Idoate M., San-Miguel J. Anti-PD1 associated fulminant myocarditis after a single pembrolizumab dose: The role of occult pre-existing autoimmunity. Haematologica. 2018;103:e318–e321. doi: 10.3324/haematol.2017.185777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cuce M., Gallo Cantafio M.E., Siciliano M.A., Riillo C., Caracciolo D., Scionti F., Staropoli N., Zuccala V., Maltese L., Di Vito A., et al. Trabectedin triggers direct and NK-mediated cytotoxicity in multiple myeloma. J. Hematol. Oncol. 2019;12:32. doi: 10.1186/s13045-019-0714-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Botta C., Misso G., Martino E.C., Pirtoli L., Cusi M.G., Tassone P., Tagliaferri P., Caraglia M., Correale P. The route to solve the interplay between inflammation, angiogenesis and anti-cancer immune response. Cell Death Dis. 2016;7:e2299. doi: 10.1038/cddis.2016.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Botta C., Di Martino M.T., Ciliberto D., Cuce M., Correale P., Rossi M., Tagliaferri P., Tassone P. A gene expression inflammatory signature specifically predicts multiple myeloma evolution and patients survival. Blood Cancer J. 2016;6:e511. doi: 10.1038/bcj.2016.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Botta C., Cuce M., Caracciolo D., Fiorillo L., Tagliaferri P., Tassone P. Immunomodulatory Activity of MicroRNAs: Potential Implications for Multiple Myeloma Treatment. Curr. Cancer Drug Targets. 2017;17:819–838. doi: 10.2174/1568009617666170330154756. [DOI] [PubMed] [Google Scholar]
- 30.Botta C., Bestoso E., Apollinari S., Cusi M.G., Pastina P., Abbruzzese A., Sperlongano P., Misso G., Caraglia M., Tassone P., et al. Immune-modulating effects of the newest cetuximab-based chemoimmunotherapy regimen in advanced colorectal cancer patients. J. Immunother. 2012;35:440–447. doi: 10.1097/CJI.0b013e31825943aa. [DOI] [PubMed] [Google Scholar]
- 31.Botta C., Barbieri V., Ciliberto D., Rossi A., Rocco D., Addeo R., Staropoli N., Pastina P., Marvaso G., Martellucci I., et al. Systemic inflammatory status at baseline predicts bevacizumab benefit in advanced non-small cell lung cancer patients. Cancer Biol. Ther. 2013;14:469–475. doi: 10.4161/cbt.24425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Botta C., Gulla A., Correale P., Tagliaferri P., Tassone P. Myeloid-derived suppressor cells in multiple myeloma: Pre-clinical research and translational opportunities. Front. Oncol. 2014;4:348. doi: 10.3389/fonc.2014.00348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Perez C., Botta C., Zabaleta A., Puig N., Cedena M.T., Goicoechea I., Alameda D., San Jose-Eneriz E., Merino J., Rodriguez-Otero P., et al. Immunogenomic identification and characterization of granulocytic myeloid-derived suppressor cells in multiple myeloma. Blood. 2020;136:199–209. doi: 10.1182/blood.2019004537. [DOI] [PubMed] [Google Scholar]
- 34.Sacco A., Battaglia A.M., Botta C., Aversa I., Mancuso S., Costanzo F., Biamonte F. Iron Metabolism in the Tumor Microenvironment-Implications for Anti-Cancer Immune Response. Cells. 2021;10:303. doi: 10.3390/cells10020303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Nerli S., Sgourakis N.G. Structure-based modeling of SARS-CoV-2 peptide/HLA-A02 antigens. bioRxiv. :2020. doi: 10.3389/fmedt.2020.553478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chinoy H., Payne D., Poulton K.V., Fertig N., Betteridge Z., Gunawardena H., Davidson J.E., Oddis C.V., McHugh N.J., Wedderburn L.R., et al. HLA-DPB1 associations differ between DRB1*03 positive anti-Jo-1 and anti-PM-Scl antibody positive idiopathic inflammatory myopathy. Rheumatology. 2009;48:1213–1217. doi: 10.1093/rheumatology/kep248. [DOI] [PubMed] [Google Scholar]
- 37.Maniaol A.H., Elsais A., Lorentzen A.R., Owe J.F., Viken M.K., Saether H., Flam S.T., Brathen G., Kampman M.T., Midgard R., et al. Late onset myasthenia gravis is associated with HLA DRB1*15:01 in the Norwegian population. PLoS ONE. 2012;7:e36603. doi: 10.1371/journal.pone.0036603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rothwell S., Chinoy H., Lamb J.A., Miller F.W., Rider L.G., Wedderburn L.R., McHugh N.J., Mammen A.L., Betteridge Z.E., Tansley S.L., et al. Focused HLA analysis in Caucasians with myositis identifies significant associations with autoantibody subgroups. Ann. Rheum. Dis. 2019;78:996–1002. doi: 10.1136/annrheumdis-2019-215046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Carr A.S., Cardwell C.R., McCarron P.O., McConville J. A systematic review of population based epidemiological studies in Myasthenia Gravis. BMC Neurol. 2010;10:46. doi: 10.1186/1471-2377-10-46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Osserman K.E., Genkins G. Studies in myasthenia gravis: Review of a twenty-year experience in over 1200 patients. Mt. Sinai J. Med. 1971;38:497–537. [PubMed] [Google Scholar]
- 41.Hoch W., McConville J., Helms S., Newsom-Davis J., Melms A., Vincent A. Auto-antibodies to the receptor tyrosine kinase MuSK in patients with myasthenia gravis without acetylcholine receptor antibodies. Nat. Med. 2001;7:365–368. doi: 10.1038/85520. [DOI] [PubMed] [Google Scholar]
- 42.Lindstrom J.M., Seybold M.E., Lennon V.A., Whittingham S., Duane D.D. Antibody to acetylcholine receptor in myasthenia gravis. Prevalence, clinical correlates, and diagnostic value. Neurology. 1976;26:1054–1059. doi: 10.1212/WNL.26.11.1054. [DOI] [PubMed] [Google Scholar]
- 43.Larkin J., Chmielowski B., Lao C.D., Hodi F.S., Sharfman W., Weber J., Suijkerbuijk K.P.M., Azevedo S., Li H., Reshef D., et al. Neurologic Serious Adverse Events Associated with Nivolumab Plus Ipilimumab or Nivolumab Alone in Advanced Melanoma, Including a Case Series of Encephalitis. Oncologist. 2017;22:709–718. doi: 10.1634/theoncologist.2016-0487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Safa H., Johnson D.H., Trinh V.A., Rodgers T.E., Lin H., Suarez-Almazor M.E., Fa’ak F., Saberian C., Yee C., Davies M.A., et al. Immune checkpoint inhibitor related myasthenia gravis: Single center experience and systematic review of the literature. J. Immunother. Cancer. 2019;7:319. doi: 10.1186/s40425-019-0774-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.