Abstract
Background
Despite high psychosocial needs that negatively affect the quality of life of adults living with cancer and their family caregivers, there is a lack of interventions that are culturally sensitive to low-income countries. This protocol tests the feasibility of a randomised controlled trial on the efficacy of a socio-spiritual intervention to improve the quality of life of adult Nigerians living with cancer and their family caregivers.
Methods/design
This two-arm trial will recruit 152 adults with cancer and their family caregivers (76 dyads). Participants will be recruited from a clinical facility in Zaria, Kaduna State, Nigeria. Eligible participants will be randomly assigned to either the intervention or control group at a 1:1 ratio. The intervention consists of four weekly face-to-face sessions with a focus on spirituality, social support, and information needs. Control participants will receive usual care. Outcome measures include feasibility, spiritual need, social need, information, cancer health literacy, and quality of life collected at baseline and immediate post-intervention.
Discussion
Nigeria has the highest rate of extreme poverty globally with high rates of cancer mortality. Testing the feasibility of social-spiritual interventions in resource poor settings is important to establish preliminary efficacy and sustainability. Family-centred interventions for adults living with cancer and their family caregivers can strengthen their coping capabilities. If this intervention is feasible and effective, it could be implemented both in clinical practice and communities in other low and middle income countries.
Keywords: Adult patient, Cancer, Family caregiver, Health literacy, Quality of life, Socio-spiritual intervention
1. Introduction
Cancer mortality is on the rise in sub-Saharan Africa, where there is limited health care infrastructure [1]. According to cancer incidence estimates, about 238,000 new cancer cases in this region will occur annually, with an almost triple rise in the number of new cancer cases expected by 2050 [2]. Several sub-Saharan African countries are under-resourced to provide treatment for adults with cancer despite significant progress in cancer management globally [3]. Lack of resources place many adults with cancer in the care of close relatives who may have little or no capability [4]. While adults with cancer struggle with symptoms and uncertainty, family caregivers may also suffer significant long-term psychosocial burden [5,6]. Psychosocial burden may be associated with a range of needs related to information, practical, psychological, physical, social, and spiritual support [7]. Unmet psychosocial needs may adversely affect quality of life (QoL) of individuals with cancer and family caregivers [8].
A study of psychosocial needs and health-related QoL among 600 colorectal cancer survivors in the United Kingdom (U.K) found that participants with one or more unmet needs had decreased QoL, as psychosocial needs increased QoL decreased [9]. Similarly, the prevalence and impact of psychosocial needs were predictive of QoL among 52 South Korean women with recurrent breast cancer [10].
Information needs are critical for individuals living with cancer and their family caregivers in sub-Saharan Africa where low health literacy about the disease and socio-cultural issues influence help-seeking and care behaviours [11]. Socio-cultural beliefs about cancer in sub-Saharan Africa contribute to late presentation, failure to adhere to treatment and discontinuation of treatment [12]. Although the provision of relevant education about the disease to both adults with cancer and family caregivers may contribute to better decision-making, socio-cultural beliefs must also be considered. In traditional African culture, diseases such as cancer are conceptualised as a lack of connection with the supernatural/ancestral forces. This understanding influences how the disease is perceived and steps taken towards diagnosis and management [13].
Social support is one of the foremost contributors of QoL and plays an important role in cancer care. It encompasses practical help, personal care for patients, preservation or enhancement of social networks, and emotional support [14]. Social support has a positive effect on a variety of outcomes, including physical, psychosocial, social, and spiritual wellbeing [15]. The ability of the family to maintain cohesion and adequate support for each other is an important consideration in cancer care. In Nigeria, family bonds are considered essential in an individual's life and are the main source of social support.
Socio-spiritual needs are amenable to intervention. This is consistent with systematic reviews and meta-analyses on the positive effects of psychosocial interventions on QoL in cancer patients [16,17]. Similarly, various family-based interventions have been used to support people living with cancer and their families manage the condition and caregiving burden and improve QoL [18,19]. However, these studies were mainly conducted in high-income countries with no published studies emerging from Africa, or Nigeria in particular [20]. Interventions tested in high-income countries may not apply in LMIC as content may lack certain components that are important to African people, such as spirituality, concept of familism, and contextualised information about cancer.
A recent survey of 240 adult Nigerians living with cancer and their families found a decline in QoL. Adults with cancer had the greatest need for information and spiritual support, while family caregivers had the greatest need for family/social support and spiritual needs [21].
1.1. Theoretical framework
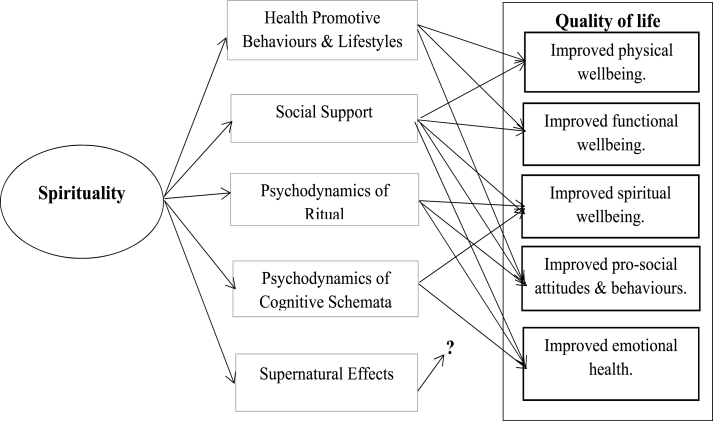
The proposed study is guided by a spiritual framework adapted from Hodge [22]. The framework identifies seven discrete explanatory mechanisms, or pathways through which spirituality produces beneficial outcomes: health promoting behaviours and lifestyles, social sychodynamics of ritual, psychodynamics of cognitive schemata, ego challenges, quantum effects, and supernatural effects. For this study, five specific issues were identified within the pathways (health promoting behaviours and lifestyles, social support, psychodynamics of ritual, psychodynamics of cognitive schemata, and supernatural effects) to explain how spirituality influences QoL (see Fig. 1). This framework provides a theoretical approach to assessing change in QoL.
Fig. 1.
The relationship between discrete pathways and mediating outcomes.
1.2. Rationale and previous work
This study includes four work package for adult Nigerians with cancer and their family caregivers. Work Package 1 included a systematic review [20], while Work Package 2 included a quantitative needs survey of 240 adult Nigerians living with cancer and their family caregivers [21]. Based on the data obtained in work packages one and two, the Behaviour Change Wheel (BCW) [23] was used to develop the intervention (work package three). Target behaviours were described as (1) involvement in socio-spiritual behaviours to increase QoL across the study population, and (2) appropriate knowledge of the disease process and caregiving among adults with cancer and family caregivers. Thereafter, through application of the BCW, seven intervention functions were selected: education, persuasion, incentivisation, environmental restructuring, training, modelling, and enablement.
The outcomes of previous work packages and the development of the intervention are described in more detail elsewhere [20,21].
1.3. Aims
The proposed study aims to (1) test the feasibility of a socio-spiritual intervention suitable for the clinical realities of LMICs (primary outcome); and (2) evaluate the effectiveness of the socio-spiritual intervention on spiritual, family/social, and information needs, health literacy, and QoL (secondary outcome).
1.4. Objectives
The socio-spiritual intervention will be assessed according to the Donabedian Framework [24] of structure, process, and outcome as described below.
Structure: to evaluate rates of participant eligibility for inclusion, acceptance, and retention.
Process:
-
a.
conduct the socio-spiritual intervention using an RCT design with adults with cancer and their family caregivers.
-
b.
monitor intervention fidelity.
-
c.
compare data collection rates for those allocated to the intervention and control groups.
Outcome: determine self-reported spiritual needs, family/social needs, information needs, cancer health literacy, and perceived QoL of adults with cancer and their family caregivers receiving the intervention (pre-post, intra and inter group comparison) compared to the control group.
1.5. Hypotheses
-
•
Adults with cancer and their family caregivers receiving the intervention will have significantly higher overall family/social support, lower spiritual needs, and information needs scores compared to the control group.
-
•
Adults with cancer and their family caregivers receiving the intervention will have significantly higher QoL and health literacy compared to the control group.
2. Methods
2.1. Study design
A single-centre, two-arm RCT will test the feasibility and evaluates efficacy of the socio-spiritual intervention for adults living with cancer and their family caregivers. Eligible participants will be randomly assigned to either the intervention or control group at a 1:1 ratio.
This study protocol was developed in accordance with the Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) statement guidelines for designing and reporting RCTs [25]; the SPIRIT checklist (additional file 2) and the schedule of procedures can be seen in Table 1.
Table 1.
SPIRIT protocol schedule of procedures.
This trial was registered at the Pan African Clinical Trial Registry (www.pactr.org) (PACTR202007829295775) on 15 June 2020.
2.2. Setting
The study will be conducted at the Oncology Unit of a large, publicly funded tertiary referral hospital in Nigeria. Patients are referred to the hospital from all over the Northern region of Nigeria.
2.3. Eligibility criteria
Adults living with cancer and their family caregivers will be recruited. Eligibility criteria are adults (aged 18 or older) living with cancer (or adult family caregiver), receiving their first diagnosis within the last three months, literate to at least junior secondary school level (Grade 9) to complete self-administered questionnaires, and willingness to participate in a 2-hour program each week for four weeks. Exclusion criteria include patients with advanced cancer, or family caregivers who at the time of the study were also receiving treatment for cancer, or a condition that increased their physical and/or psychological vulnerability; unable to understand and speak English, and not able to complete self-reported surveys.
2.4. Sample size
A power analysis was performed to determine the sample size required to evaluate the efficacy of the intervention. Based on a previous related intervention study [26], a sample of 76 participants (38 dyads) dyads in each arm will provide 85% power for repeated-measures analysis of variance (time 1 vs time 2) with a moderate effect size and probability of 0.05. A final sample of 152 participants (adults with cancer and family caregivers) will be needed.
2.5. Recruitment and consent
Participants will be recruited at the oncology and outpatient clinics. All staff working in the oncology unit will be informed about the study by the research team. The recruitment is a three-step process involving the following:
Initially identifying potential participants against inclusion and exclusion criteria: Nurses will identify and refer all potential participants who meet the inclusion criteria to research assistant who will be present at the clinic.
Approaching and informing potential participants of the study: The research assistant will provide written and verbal information to adults living with cancer and family caregivers about the purpose and processes of the study. Patients who attend the clinic without a family caregiver but are interested in participating will be encouraged to discuss the study with their family, decide on a nominee, and notify the research assistant.
Consent: Those interested in the study will be given research information sheet, and then sign an informed consent form, which will be collected by the research assistant.
2.6. Randomisation and blinding
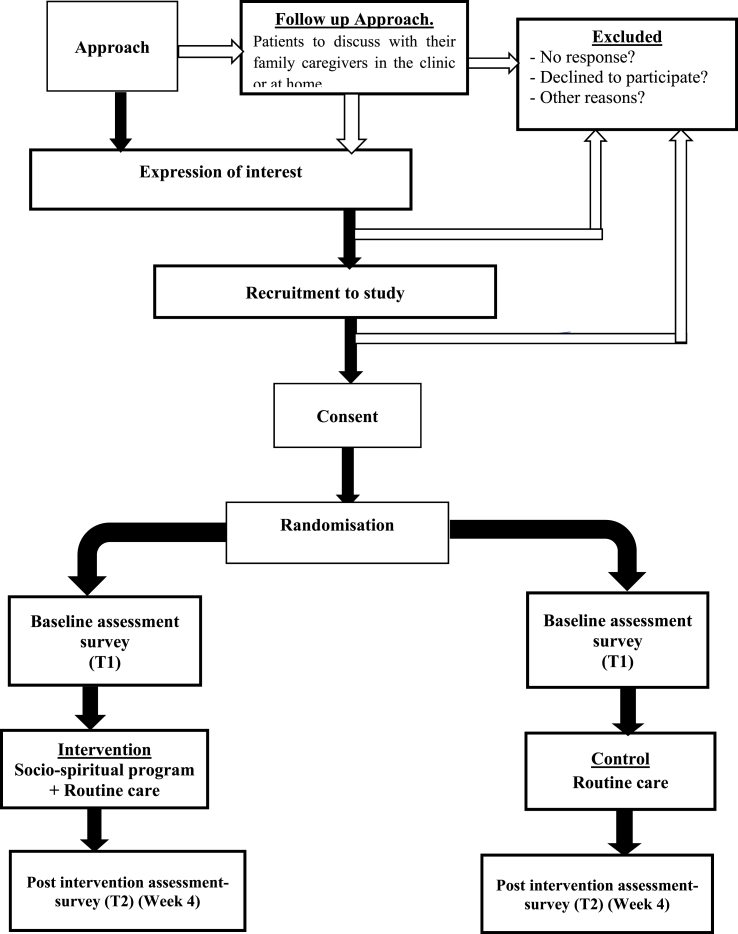
Participants who meet the study criteria and give informed consent to participate in the study will be randomly assigned either to the intervention or control group (Fig. 2). Computer-generated block randomisation will be performed to ensure equal (1:1) allocation. An independent researcher who is not involved in the study will carry out the allocation procedure. As a result, the interventionists who will facilitate the group and the participants will be blind to the participants allocation (allocation concealment). The allocation outcome will be conveyed via phone to interventionists, who will inform participants of their allocation and subsequent study procedure.
Fig. 2.
Participant flow chart.
2.7. Routine care versus intervention
All dyads in the control and intervention groups will receive routine medical and nursing care from health care professionals, which may include cancer treatment, medication, counselling, and adherence to a nutritional plan. Over a four-week period, dyads assigned to the intervention group will receive face-to-face socio-spiritual intervention aimed at strengthening social support, reducing spiritual and information needs, and improving QoL.
Although adult with cancer-caregiver dyads in the control group will not participate in the intervention programme, they will engage with independent researcher/interventionalists during recruitment, baseline, and post intervention data collection. For ethical reasons, and if desired; participants assigned to the control group will be offered a printed copy of intervention resources and a one-day workshop after data collection is complete. All participants in the study will receive a nominal amount ($10) for attendance which can be used to purchase a meal or pay for public transport.
2.8. Zaman Lafiya intervention
The Behaviour Change Wheel framework was used to develop an evidence-based socio-spiritual intervention (referred to here as the “Zaman Lafiya Programme,” a local vernacular name that means “Living Well”). Guided by the findings of a local needs survey, the intervention was designed to meet the needs of adult Nigerians living with cancer and their family caregivers to improve family/social support, spiritual needs, and information/health literacy [21]. The spiritual framework informed program content and processes to be congruent with the Nigerian culture. It assumed that lifestyles, social support, psychodynamics of ritual, psychodynamics of cognitive schemata, and supernatural effects influence QoL (see Fig. 1).
The intervention consists of four, 2-h sessions addressing various behavioural and social aspects affecting QoL. This includes (i) understanding cancer journey – need for family/social support and spirituality, (ii) sharing the journey – communication and building trusting relationships, (iii) finding the right support – family/social support and spiritual support and (iv) staying strong to overcome adversity – understanding strengths and resources of the family by building spiritual support. The intervention programme is detailed in Table 2.
Table 2.
Socio-spiritual intervention content.
| Theme | Typical adult with cancer/caregivers' concerns | The spiritual Framework's discrete pathways | Intervention |
|---|---|---|---|
| Understanding cancer journey – need for family/social support and spirituality. | - Assess baseline health literacy of the disease. Discuss results amongst the group. | - Health promotive behaviours and lifestyles. | - Assess baseline health literacy of the disease and caregiving. Discuss results amongst the group. |
| - Educate about disease and treatments according to identified knowledge gaps and misconceptions. | - Social support. | - Educate about illness and treatments and caregiving according to identified knowledge gaps and misconceptions. | |
| - Discuss communication strategies and how to be assertive to obtain additional information | - Psychodynamics of ritual. | - Discuss communication strategies and how to be assertive to obtain additional information. | |
| - Clearly and concisely communicate and demonstrate a range of possible therapies such as relaxation and massage. | - Psychodynamics of cognitive schemata, | - Clearly and concisely communicate and demonstrate a range of possible therapies such as relaxation, massage. | |
| Sharing your journey – communication and building trusting relationships | Concerns about how much support they will receive during illness and treatment trajectories (e.g., will people continue to visit, or will they forget about me, will caregivers receive support from other family members) | - Health promotive behaviours and lifestyles. | - Ask participants to write down their concerns and fears. Discuss results amongst the group. |
| - Discuss strategies to promote open communication. | |||
| - Social support. | - Brainstorm strategies to enhance mutual support and teamwork. | ||
| - Highlight the value of living “here and now” and promote approaches to helping dyad work within the limits of their new limitations. | |||
| Finding the right support – family/social support and spiritual support. | Loneliness and reduced social support. | - Social support. | - Identify family strengths using a short survey. Discuss results amongst the group. |
| - Psychodynamics of ritual. | |||
| - Psychodynamics of cognitive schemata. | - Social connections; link between the past and present. | ||
| - Provide reality-oriented approaches and pragmatic social support – maintain regular and brief interventions. | |||
| - Work to address cognitive distortions and devastating behaviours. | |||
| Stay strong overcoming adversity – understanding strengths and resources of the family building spiritual support. | Coming to terms with “who I am now.” Creating meaning, finding purpose; achieve a sense of wisdom through life's experience and lessons. | - Health promotive behaviours and lifestyles. | - Use reflective writing to deepen insights, reflect on life changes and what is essential in life. |
| - Social support. | - Discuss the use of spiritual coping with health challenges, especially dealing with those situations that are not in our control. | ||
| - Psychodynamics of ritual. | - Consider what provides a sense of inner peace for the individual. | ||
| - Psychodynamics of cognitive schemata. | - Explore the views of each other and come to a new shared understanding. | ||
| - Supernatural effects. |
2.9. Delivery of intervention
The intervention is a face-to-face, nurse-led programme that incorporates activities and resources (video and paper) to assist in the delivery of the intervention. A programme booklet will be developed with detailed information about the themes discussed in each session, and each session will have a 10-min video clip featuring cancer survivors and their families sharing their experiences. Group sessions will provide opportunities to express feelings and discuss issues of shared concern. For example, discussions will enable participants to gain an understanding of their experiences and how these may have changed their view of life, guiding them to adopt an optimistic cognitive framework for coping with stress and finding benefits in their situation.
Two interventionists will conduct two concurrent group sessions on various days of the week in the hospital hall. Each group will meet weekly for four weeks and will consist of five to seven adults with cancer and caregiver dyads. All participants will receive the programme within four months. Research assistants will contact participants by telephone in advance as a reminder about the forthcoming session and encourage attendance as much as possible. This approach aims to be supportive and identify any difficulties participants may be experiencing (such as transport; appointment clashes).
2.10. Interventionist training and background
Interventionists are certified nurse educators with an extensive background in oncology nursing and research methodology. Five days of training have been provided to interventionists and a research assistant on the study aims, procedures, surveys, content of four sessions, group processes, use of registration forms, and data collection, during which they will become acquainted with the programme and their position within it. Additionally, the significance of rigour will be emphasised. As part of their orientation and training for the study, interventionists were provided with a digital version of the intervention protocol and facilitator guide.
2.11. Withdrawal criteria
No one is obligated to participate in this trial. As a result, they are also totally free to withdraw from the study at any moment and without penalty, as well as to decline to answer certain questions. Participation in the study has no impact on the care participants get at the Hospital. Furthermore, because the study is pair-based, if one of the dyads attends intervention sessions, they will be able to continue, but their data will be excluded from the study, and no re-training chances will be available for those who missed any of the programme's sessions.
2.12. Quality assurance
We are evaluating sessions using a systematic adherence checklist form to ensure the consistency of intervention content and delivery for quality assurance purposes. Participants answer a brief questionnaire at baseline and again at the 4-week follow-up to evaluate retention of key points learned and information covered throughout intervention sessions. Although all instruments have been validated and confirmed to be reliable, the survey will be piloted using a Nigerian sample of eight adults with cancer and eight family caregivers. Weekly staff meetings, process evaluations, and monitoring reports will ensure that interventionists receive ongoing quality assurance training and supervision. Additionally, all sessions are audio recorded and reviewed in weekly supervision meetings with the researchers.
To ensure intervention fidelity, efforts would be taken to ensure that each participant received the same dose, and that the intervention period was set for a fixed number of groups. Each session, interventionists will be asked to complete an intervention diary to keep note of any protocol deviations. Data will be collected at baseline and post intervention by an independent researcher who is blind to participant allocation and does not participate in group facilitation. All analyses will be performed based on intention to treat.
2.13. Outcomes
The primary and secondary outcomes are presented in Table 3. The study will follow the guidelines used by Leon., Davis [27]. to demonstrate feasibility of a study according to rates of eligibility (per cent screened for participants who were eligible), acceptance (proportion of those who screen eligible who enrol), retention (attrition and number of sessions completed by study arm), assessment process (proportion of planned assessments that are completed by participants), and intervention fidelity (percentage and similarity of session content covered across sessions by interventionalists).
Table 3.
Overview of study outcomes.
| Outcome | Domain (Aim) | Name | Source | Time point |
|||
|---|---|---|---|---|---|---|---|
| W1 (T1) | W2 | W3 | W4 (T2) | ||||
| Primary | Feasibility (1) | Recruitment rate | Study records | X | |||
| Primary | Feasibility (1) | Acceptance | Study records | X | |||
| Primary | Feasibility (1) | Retention rate | Study records | X | |||
| Primary | Feasibility (1) | Assessment process | Study records | X | X | X | X |
| Primary | Feasibility (1) | Fidelity | Fidelity rating form – done at every session. | X | X | X | X |
| Secondary | Preliminary efficacy (2) | Spiritual needs | - The Spiritual Needs Assessment for Patients (SNAP) (Likert scale 23 items) | X | X | ||
| - Modified version of the Spiritual Needs Assessment for Patient (SNAP) (Likert scale 23 items) | |||||||
| Secondary | Preliminary efficacy (2) | Social needs | - Multidimensional Scale of Perceived Social Support (MSPSS) (Survey: Likert scale 12 items) | X | X | ||
| Secondary | Preliminary efficacy (2) | Information need | - Information Scale of the Comprehensive Needs Assessment Tool for Cancer (Likert scale 10 items) and Cancer health literacy (CHLT-6) | X | X | ||
| - Information Scale of the Comprehensive Needs Assessment Tool for Cancer- Caregivers (Likert scale 8 items) and Cancer health literacy (CHLT-6) | |||||||
| Secondary | Preliminary efficacy (2) | Quality of life | - The Functional Assessment of Chronic Illness Therapy-Spirituality (FACIT-Sp) (Likert scale 39 items) | X | X | ||
| - The Functional Assessment of Chronic Illness Therapy-Spirituality (FACIT-Sp) (Likert scale 39 items) (Modified) | |||||||
| Descriptive (2) | Demographic and clinical information | Socio-demographic and health-related information (10 items for adults with cancer and 9 items for family caregivers). | X | ||||
Key: W: Week; T1: Time 1; T2: Time 2.
Primary outcome measure – To test the feasibility (rates of eligibility, acceptance, retention, assessment process, and fidelity) of the intervention, interventionists will score.
-
○
Recruitment and acceptance rates at baseline (T1).
-
○
Assessment process and fidelity will be directly observed throughout the trial.
-
○
Retention rate at T2.
Fidelity will be rated using a structured adherence checklist form to evaluate sessions in the study arm for a subset (50%) session. The intervention fidelity scores range from 0 to 100, depending on the number of topics discussed during the session.
Feasibility of the study will be demonstrated if 80% of participants complete all sessions and assessments. A 60% recruitment rate will be considered acceptable. The retention and assessment process benchmarks are 80% and covering 80–100% of topics would be regarded as high fidelity [28].
Secondary outcome measures – Efficacy of the intervention will include measures of the outcome variables (spiritual needs, family/social needs, information needs, health literacy, and QoL), repeated measurements will be conducted pre and post intervention. The surveys will be completed by participants in both the intervention and control groups.
2.14. Participant's timeline
For everyone in the intervention group, surveys will be completed at two-time points: baseline (T1), and immediately following completion of socio-spiritual intervention (four weeks, T2) (Fig. 2) to determine whether the intervention has an immediate impact on the study outcomes. Participants in the control group will complete surveys within the same timeframe (baseline; + 4 weeks).
2.15. Data collection
For all participants, socio-demographic data (age, gender, ethnicity, education, marital status, religion, economic status, relationship to the patient, duration of caregiving), clinical information (including cancer type, and stage of cancer), primary outcome (recruitment rate and acceptance, retention rate, and assessment process), and secondary outcomes (social/family needs, spiritual needs, information needs, health literacy, and QoL) will be measured. Well-validated measures will be used (see Table 3).
Independent variables
Social/family needs – The Multidimensional Scale of Perceived Social Support (MSPSS) is a brief instrument designed to measure perceptions of support from 3 sources: Family, Friends, and Significant Other. The scale is comprised of 12 items, with 4 items for each subscale [29]. Each item is scored on a 7-point Likert scale from 1 = very strongly disagree to 7 = very strongly agree. The MSPSS was initially validated in western, high-income countries but has since been validated in a number of non-western settings and LMICs with good reliability ranging from 0.86 to 0.90 [30,31]. In this study, MSPSS will be used to measure both adults living with cancer and family caregivers’ perception of the adequacy of the social support.
Spiritual need – The Spiritual Needs Assessment for Patients (SNAP) comprises 23 items in three domains: psychosocial (5 items), spiritual (13 items), and religious (5 items) [32]. Each question is scored on a 4-point Likert scale ranging from 1 = very much to 4 = not at all. The total score can vary from 23 to 92, and higher values reflect more spiritual needs [33]. Good reliability ranging from 0.74 to 0.95 has been reported [32].
A modified version of the SNAP will be used to measure the spiritual needs of the family caregiver. Three items will be modified (“getting in touch with other patients with similar illnesses” to “getting in touch with other family caregivers”, “finding meaning in your experience of illness” to “finding meaning in your experience of caregiving”, and “making decisions about your medical treatment that are in keeping with your spiritual or religious beliefs” to “making decisions about your loved one's medical treatment that are in keeping with their spiritual or religious beliefs”) to reflect the family caregivers needs.
Information needs – The ten-item Information subscale of the Comprehensive Needs Assessment Tool for Cancer (CNAT) will be used to measure adults with cancer information needs [34]. The eight-item Information subscale of the Comprehensive Needs Assessment Tool for Cancer- Caregivers (CNAT-C) [35] will measure family caregivers' information needs. Items are scored on a 4-point Likert scale ranging from 0 = no need to 3 = high need. The CNAT and CNAT-C have Cronbach's alpha internal consistency reliability ranging from of 0.79–0.97 [34,35].
Cancer health literacy – Cancer health literacy (CHLT-6) [36] is a cancer-specific health literacy tool which differentiates between patients with adequate and limited cancer health literacy with a high degree of precision. Responses to the six items are scored as correct [1] or incorrect (0) and summed. Participants are considered to either have adequate cancer health literacy (total score 4 to 6) or limited cancer literacy (total score less than 4) [37]. The CHLT-6 has a Cronbach's alpha internal consistency reliability of 0.96–0.99 [36]. It has been tested and used in Nigeria with good reliability of 0.74 [21]. In this study, CHLT-6 will be used to measure both adults living with cancer and family caregivers' health literacy.
Dependent variable
Quality of life – The Functional Assessment of Chronic Illness Therapy-Spirituality (FACIT-Sp) (version 4) combines the 27-item FACT-G and the 12-item FACIT-Sp [38,39]. FACIT-Sp will be used to assess the QoL of participants and a modified version will be administered to family caregivers. Items three, five, eleven, twelve, sixteen, seventeen, nineteen, twenty, twenty-four, thirty-eight, and thirty-nine were rephrased for family caregivers.
The survey consists of 39 items with five domains, assessing physical well-being (seven items), social/family well-being (seven items), emotional well-being (six items), functional well-being (seven items), and spiritual well-being (twelve items). Items are rated on a Likert-scale from 0 (not at all) to 4 (very much) [40]. Higher scores indicate a more favourable perception of QoL [41]. It has good internal consistency reliability ranging from 0.72 to 0.85 [42].
2.16. Data management and privacy procedures
All data will be password protected. All identifying information of participants will be coded and de-identified. Signed informed consent forms and returned coded/anonymised surveys will be stored in a secured project file, and access will be restricted to research team members, who will all sign a privacy statement. Data will be entered into the Statistical Package for the Social Sciences (SPSS) version 27 [43] database by a researcher not involved with the intervention and blind to participants' allocation. Data integrity will be assured by random consistency checks/re-entry of data. Archived electronic data will be kept for a maximum of seven years. Collected data will be processed anonymously in publications and reports, preventing identification of individual participants.
2.17. Approach to analysis
Data will be entered, cleaned, and analysed using SPSS. All statistical analyses will be performed based on intention-to-treat.
Descriptive statistics – Continuous demographic and clinical variables will be summarised by means and standard deviations, while categorical data will be described using frequencies and percentages and 95% confidence intervals. Normality assumptions of continuous outcome variables will be evaluated.
Inferential statistics – Parametric or non-parametric inferential statistical tests will be performed to assess for differences between adults with cancer and family caregivers' demographic/medical variables (e.g., age, gender, economic status, cancer type, and dyadic relationship) in intervention and control groups at baseline. If differences are detected, these variables will be controlled in subsequent analyses. To assess intervention efficacy, repeated measures Multivariate Analysis of Variance (MANOVA) will be used to analyse changes in study variables from time 1 to 2, with time and role (patient and caregiver) as within-subject variables to control for the correlated nature of the data.
2.18. Ethical considerations
The Declaration of Helsinki's ethical research principles will guide the study [44]. The Ethics Committee of Ahmadu Bello University Teaching Hospital (ABUTH) Zaria, Nigeria (ABUTHZ/HREC/W38/2020) and the Human Research Ethics Committee of Griffith University, Queensland, Australia (GU Ref No 2020/554) approved this study in May and July 2020, respectively. Written consent will be obtained from the participants.
Participation will be voluntary without coercion, manipulation, or undue influence. Participants will be duly informed that their consent is fluid and that they are free to withdraw at any point during the study process. The principle of justice and fairness will be adhered to by ensuring that all potential participants are treated equally without favour to individual on the grounds of religion, gender, age, or cadre.
2.19. Dissemination policy
After the study is completed, the findings will be disseminated across a variety of media, including open-access peer-reviewed journals and national and international conferences. The demonstrated efficacy would improve the intervention's implementation in clinical practice and communities in low and middle-income countries. Furthermore, the findings will inform a larger longitudinal RCT study.
3. Discussion
This protocol describes an RCT to test the feasibility and efficacy of a nurse-delivered socio-spiritual intervention for adults living with cancer and their family caregivers in a LMICs. The primary focus will be on feasibility (recruitment rate, retention rate, and fidelity). Secondary outcomes evaluate the efficacy of this intervention in addressing spirituality, family/social needs, information needs and QoL among participants. The intervention is theory-driven and in response to a recent needs survey conducted in Nigeria for adults with cancer and their family caregivers [21]. The emphasis on spirituality is novel and highly relevant to low-income countries such as Nigeria.
Research shows a dramatic rise in cancer incidence in many parts of Africa [45]. Although incidence is lower than high-income countries, mortality rates in Africa are the highest in the world [46]. The needs of patients with cancer and their family caregivers in low-income countries are greater than those in high-income countries, suggesting that African populations may share a higher burden of cancer. It is therefore imperative to develop and test brief, inexpensive, family-based interventions to assist coping and well-being.
Most intervention studies in psycho-oncology research have been conducted in high-income countries [20]. To the best of our knowledge, there has been no intervention research targeting socio-spiritual and information needs of the patient/caregiver dyad in Africa. With the rising prevalence of cancer, there is a great need for research on supportive therapy for individuals with cancer and family caregivers in Africa and Nigeria in particular.
In Nigeria, cancer is still seen as a spiritual issue with a superstitious belief in orthodox treatments, in addition to a family-centred model of decision-making that may influence how individuals access and respond to treatment. These are critical factors to be considered in resource-limited settings. The proposed intervention aims to address an often-neglected area of need. Results are expected to guide the further development and improvement of the intervention for future more extensive trials and application to other low and middle-income countries.
3.1. Limitations
This proposed study has some limitations, including being conducted in a single tertiary centre. The heterogeneity of adults living with cancer with respect to types and stages of cancer would inevitably affect the psychosocial well-being of participants and influence various outcomes. Thus, variations in reported outcomes cannot be attributed solely to the proposed intervention. If confounding differences arise from demographic and medical variables, this will be controlled in the data analysis. Another limitation is that the intervention is limited to only those who speak English. This excludes non-English - speaking adults with cancer and family caregivers who may have different needs. Generalizability of findings may therefore be limited to the English-speaking population.
3.2. Conclusion
This study describes a socio-spiritual intervention and proposed feasibility and evaluation for adults living with cancer and their family caregivers. The intervention was developed based on current literature, theoretical framework, and views of adults with cancer/family caregivers in Nigeria to maximise the efficacy and relevance of the target population. The intervention is important as there is currently an insufficient/poor provision of services for adults with cancer and their family caregivers in Nigeria. There is currently no provision in the form of interventions explicitly designed to meet the needs of this population in sub-Saharan Africa. Findings are expected to help assess and evaluate the likely success and efficacy of rolling out this intervention to more people in a range of settings.
Funding
The first author is supported by a Griffith University International Postgraduate Research Scholarship.
Author declaration
1) We wish to confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome.2) We confirm that the manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed. We further confirm that the order of authors listed in the manuscript has been approved by all of us.3) We confirm that neither the entire paper nor any of its content has been submitted, published, or accepted by another journal. The paper will not be submitted elsewhere if accepted for publication in the Journal.4) We confirm that we have given due consideration to the protection of intellectual property associated with this work and that there are no impediments to publication, including the timing of publication, with respect to intellectual property. In so doing we confirm that we have followed the regulations of our institutions concerning intellectual property.5) We confirm that any aspect of the work covered in this manuscript that has involved either experimental animals or human patients has been conducted with the ethical approval of all relevant bodies and that such approvals are acknowledged within the manuscript. 6) We understand that the Corresponding Author is the sole contact for the Editorial process (including Editorial Manager and direct communications with the office). He/she is responsible for communicating with the other authors about progress, submissions of revisions and final approval of proofs.
An authorship statement
All authors listed meet the authorship criteria according to the latest guidelines of the International Committee of Medical Journal Editors and all authors are in agreement with the manuscript.
Declaration of competing interest
No conflict of interest has been declared by the authors.
Acknowledgments
We appreciate the support of the Ahmadu Bello University Teaching Hospital in Zaria, Nigeria, and Griffith University in Queensland, Australia, in developing the procedures for these RCTs.
Contributor Information
Israel Gabriel, Email: israel.gabriel@griffithuni.edu.au.
Debra Creedy, Email: d.creedy@griffith.edu.au.
Elisabeth Coyne, Email: e.coyne@griffith.edu.au.
References
- 1.Lekoane K.M.B., Kuupiel D., Mashamba-Thompson T.P., Ginindza T.G. Evidence on the prevalence, incidence, mortality and trends of human papilloma virus-associated cancers in sub-Saharan Africa: systematic scoping review. BMC Canc. 2019;19(1) doi: 10.1186/s12885-019-5781-3. 563-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pilleron S., Soerjomataram I., Charvat H., Chokunonga E., Somdyala N.I.M., Wabinga H. Cancer incidence in older adults in selected regions of sub‐Saharan Africa, 2008–2012. Int. J. Canc. 2019;144(8):1824–1833. doi: 10.1002/ijc.31880. [DOI] [PubMed] [Google Scholar]
- 3.Boyle P., Ngoma T., Sullivan R., Brawley O. Cancer in Africa: the way forward. ecancermedicalscience. 2019;13 doi: 10.3332/ecancer.2019.953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shah Kayamba, Peek Heimburger. Cancer control in low- and middle-income countries: is it time to consider screening? Journal of global oncology. 2019;5(5):1–8. doi: 10.1200/JGO.18.00200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ferrell B.R., Kravitz K., Borneman T., Friedmann E.T. Family caregivers: a qualitative study to better understand the quality-of-life concerns and needs of this population. Clin. J. Oncol. Nurs. 2018;22(3):286–294. doi: 10.1188/18.CJON.286-294. [DOI] [PubMed] [Google Scholar]
- 6.Gabriel I.O., Mayers P.M. Effects of a psychosocial intervention on the quality of life of primary caregivers of women with breast cancer. European journal of oncology nursing. the official journal of European Oncology Nursing Society. 2019;38:85–91. doi: 10.1016/j.ejon.2018.12.003. [DOI] [PubMed] [Google Scholar]
- 7.Chae B.J., Lee J., Lee S.K., Shin H.-J., Jung S.-Y., Lee J.W. Unmet needs and related factors of Korean breast cancer survivors: a multicenter, cross-sectional study. BMC Canc. 2019;19(1) doi: 10.1186/s12885-019-6064-8. 839-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ullrich A., Ascherfeld L., Marx G., Bokemeyer C., Bergelt C., Oechsle K. Quality of life, psychological burden, needs, and satisfaction during specialized inpatient palliative care in family caregivers of advanced cancer patients. BMC Palliat. Care. 2017;16(1) doi: 10.1186/s12904-017-0206-z. 31-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Santin O., Murray L., Prue G., Gavin A., Gormley G., Donnelly M. Self-reported psychosocial needs and health-related quality of life of colorectal cancer survivors. Eur. J. Oncol. Nurs. 2015;19(4):336–342. doi: 10.1016/j.ejon.2015.01.009. [DOI] [PubMed] [Google Scholar]
- 10.Park B.-W., Hwang S.Y. Unmet needs and their relationship with quality of life among women with recurrent breast cancer. Journal of Breast Cancer. 2012;15(4):454–461. doi: 10.4048/jbc.2012.15.4.454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Akuoko C.P., Armah E., Sarpong T., Quansah D.Y., Amankwaa I., Boateng D. Barriers to early presentation and diagnosis of breast cancer among African women living in sub-Saharan Africa. PloS One. 2017;12(2) doi: 10.1371/journal.pone.0171024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tetteh D., Faulkner S. Sociocultural factors and breast cancer in sub-Saharan Africa: implications for diagnosis and management. Women's Health. 2016;12(1):147–156. doi: 10.2217/whe.15.76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sarki A., Roni B. This disease is “not for hospital”: myths and misconceptions about cancers in Northern Nigeria. Health Rep. 2019;3 [Google Scholar]
- 14.Shin D.W., Park H.S., Lee S.H., Jeon S.H., Cho S., Kang S.H. Health-related quality of life, perceived social support, and depression in disease-free survivors who underwent curative surgery only for prostate, kidney and bladder cancer: comparison among survivors and with the general population. Cancer research and treatment. official journal of Korean Cancer Association. 2019;51(1):289. doi: 10.4143/crt.2018.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Goudarzian Sharifnia. The relationship between social support and quality of life in Iranian clients with Cancer. Iranian Journal of Cancer Nursing (ijcn) 2019;1(2):9–15. [Google Scholar]
- 16.Son H., Son Y.-J., Kim H., Lee Y. Effect of psychosocial interventions on the quality of life of patients with colorectal cancer: a systematic review and meta-analysis. Health Qual. Life Outcome. 2018;16(1):119. doi: 10.1186/s12955-018-0943-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Teo I., Krishnan A., Lee G.L. Psychosocial interventions for advanced cancer patients: a systematic review. Psycho Oncol. 2019;28(7):1394–1407. doi: 10.1002/pon.5103. [DOI] [PubMed] [Google Scholar]
- 18.Badger Segrin C., Hepworth J.T., Pasvogel A., Weihs K., Lopez A.M. Telephone‐delivered health education and interpersonal counseling improve quality of life for Latinas with breast cancer and their supportive partners. Psycho Oncol. 2013;22(5):1035–1042. doi: 10.1002/pon.3101. [DOI] [PubMed] [Google Scholar]
- 19.Badr H., Carmack C.L., Diefenbach M.A. Psychosocial interventions for patients and caregivers in the age of new communication technologies: opportunities and challenges in cancer care. J. Health Commun. 2015;20(3):328–342. doi: 10.1080/10810730.2014.965369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gabriel I., Creedy D., Coyne E. A systematic review of psychosocial interventions to improve quality of life of people with cancer and their family caregivers. Nursing open. 2020 doi: 10.1002/nop2.543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gabriel I., Creedy D., Coyne E. Quality of life and associated factors among adults living with cancer and their family caregivers. Nurs. Health Sci. 2021 doi: 10.1111/nhs.12823. [DOI] [PubMed] [Google Scholar]
- 22.Hodge D.R. Spirituality: towards a theoretical framework. Soc. Thought. 2000;19(4):1–20. [Google Scholar]
- 23.Michie S., Van Stralen M.M., West R. The behaviour change wheel: a new method for characterising and designing behaviour change interventions. Implement. Sci. 2011;6(1):1–12. doi: 10.1186/1748-5908-6-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Donabedian The quality of medical care. Methods for assessing and monitoring the quality of care for research and for quality assurance programs. Science. 1978;200(4344):856–864. doi: 10.1126/science.417400. [DOI] [PubMed] [Google Scholar]
- 25.Chan A.-W., Tetzlaff J.M., Altman D.G., Laupacis A., Gøtzsche P.C., Krleža-Jerić K. SPIRIT 2013 statement: defining standard protocol items for clinical trials. Ann. Intern. Med. 2013;158(3):200–207. doi: 10.7326/0003-4819-158-3-201302050-00583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Northouse L., Schafenacker A., Barr K.L.C., Katapodi M., Yoon H., Brittain K. A tailored web-based psychoeducational intervention for cancer patients and their family caregivers. Canc. Nurs. 2014;37(5):321–330. doi: 10.1097/NCC.0000000000000159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Leon, Davis, Kraemer The role and interpretation of pilot studies in clinical research. J. Psychiatr. Res. 2011;45(5):626–629. doi: 10.1016/j.jpsychires.2010.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Borrelli B. The assessment, monitoring, and enhancement of treatment fidelity in public health clinical trials. J. Publ. Health Dent. 2011;71:S52–S63. doi: 10.1111/j.1752-7325.2011.00233.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zimet G.D., Dahlem N.W., Zimet S.G., Farley G.K. The multidimensional scale of perceived social support. J. Pers. Assess. 1988;52(1):30–41. doi: 10.1080/00223891.1990.9674095. [DOI] [PubMed] [Google Scholar]
- 30.Bruwer B., Emsley R., Kidd M., Lochner C., Seedat S. Psychometric properties of the multidimensional scale of perceived social support in youth. Compr. Psychiatr. 2008;49(2):195–201. doi: 10.1016/j.comppsych.2007.09.002. [DOI] [PubMed] [Google Scholar]
- 31.Qadir F., Khalid A., Haqqani S., Medhin G. The association of marital relationship and perceived social support with mental health of women in Pakistan. BMC Publ. Health. 2013;13(1):1–13. doi: 10.1186/1471-2458-13-1150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sharma RKMDMHS. Astrow A.B.M.D., Texeira K.P., Sulmasy D.P.M.D.P. The spiritual needs assessment for patients (SNAP): development and validation of a comprehensive instrument to assess unmet spiritual needs. J. Pain Symptom Manag. 2012;44(1):44–51. doi: 10.1016/j.jpainsymman.2011.07.008. [DOI] [PubMed] [Google Scholar]
- 33.De Araujo Toloi D., Uema D., Matsushita F., Da Silva Andrade P.A., Branco T.P., De Carvalho Chino F.T.B. Validation of questionnaire on the spiritual needs assessment for patients (SNAP) questionnaire in Brazilian Portuguese. Ecancermedicalscience. 2016;10:694. doi: 10.3332/ecancer.2016.694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Shim E.-J., Lee k-S, Park J.-H., Park J.-H. Comprehensive needs assessment tool in cancer (CNAT): the development and validation. Support. Care Canc. 2011;19(12):1957–1968. doi: 10.1007/s00520-010-1037-0. [DOI] [PubMed] [Google Scholar]
- 35.Shin D.W., Park J.H., Shim E.J., Park J.H., Choi J.Y., Kim S.G. The development of a comprehensive needs assessment tool for cancer‐caregivers in patient–caregiver dyads. Psycho Oncol. 2011;20(12):1342–1352. doi: 10.1002/pon.1857. [DOI] [PubMed] [Google Scholar]
- 36.Dumenci L., Matsuyama R., Riddle D.L., Cartwright L.A., Perera R.A., Chung H. Measurement of cancer health literacy and identification of patients with limited cancer health literacy. J. Health Commun. 2014;19(sup2):205–224. doi: 10.1080/10810730.2014.943377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kanu C.N. 2019. The Relationship between Health Literacy, Patient Activation, and Health Outcomes in Breast Cancer Patients. [Google Scholar]
- 38.Canada A.L., Murphy P.E., Fitchett G., Peterman A.H., Schover L.R. A 3‐factor model for the FACIT‐Sp. Psycho Oncol. 2008;17(9):908–916. doi: 10.1002/pon.1307. [DOI] [PubMed] [Google Scholar]
- 39.Cella D.F., Tulsky D.S., Gray G., Sarafian B., Linn E., Bonomi A. The Functional Assessment of Cancer Therapy scale: development and validation of the general measure. J. Clin. Oncol. 1993;11(3):570–579. doi: 10.1200/JCO.1993.11.3.570. [DOI] [PubMed] [Google Scholar]
- 40.Schubart J.R.P.D., Wise J.D.M., Deshaies I.M.D., Kimchi E.T.M.D., Staveley-O'Carroll KFMDPD. Gusani N.J.M.D. Quality of life assessment in postoperative patients with upper GI malignancies. J. Surg. Res. 2010;163(1):40–46. doi: 10.1016/j.jss.2010.04.057. [DOI] [PubMed] [Google Scholar]
- 41.Weisbord S.D., Carmody S.S., Bruns F.J., Rotondi A.J., Cohen L.M., Zeidel M.L. Symptom burden, quality of life, advance care planning and the potential value of palliative care in severely ill haemodialysis patients. Nephrol. Dial. Transplant. 2003;18(7):1345–1352. doi: 10.1093/ndt/gfg105. [DOI] [PubMed] [Google Scholar]
- 42.Peterman A.H., Fitchett G., Brady M.J., Hernandez L., Cella D. Measuring spiritual well-being in people with cancer: the functional assessment of chronic illness therapy—spiritual Well-being Scale (FACIT-Sp) Ann. Behav. Med. 2002;24(1):49–58. doi: 10.1207/S15324796ABM2401_06. [DOI] [PubMed] [Google Scholar]
- 43.George D., Mallery P. Routledge; 2019. IBM SPSS Statistics 26 Step by Step: A Simple Guide and Reference. [Google Scholar]
- 44.World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. Jama. 2013;310(20):2191–2194. doi: 10.1001/jama.2013.281053. [DOI] [PubMed] [Google Scholar]
- 45.Ferlay J., Soerjomataram I., Dikshit R., Eser S., Mathers C., Rebelo M. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int. J. Canc. 2015;136(5):E359–E386. doi: 10.1002/ijc.29210. [DOI] [PubMed] [Google Scholar]
- 46.Azubuike S.O., Muirhead C., Hayes L., McNally R. Rising global burden of breast cancer: the case of sub-Saharan Africa (with emphasis on Nigeria) and implications for regional development: a review. World J. Surg. Oncol. 2018;16(1) doi: 10.1186/s12957-018-1345-2. 63-13. [DOI] [PMC free article] [PubMed] [Google Scholar]