Abstract
Objective
Cognitive–behavioural therapy (CBT) has been shown to be effective in the management of chronic widespread pain (CWP); we now test whether it can prevent onset among adults at high risk.
Methods
A population-based randomised controlled prevention trial, with recruitment through UK general practices. A mailed screening questionnaire identified adults at high risk of CWP. Participants received either usual care (UC) or a short course of telephone CBT (tCBT). The primary outcome was CWP onset at 12 months assessed by mailed questionnaire. There were seven secondary outcomes including quality of life (EuroQol Questionnaire-five dimensions-five levels/EQ-5D-5L) used as part of a health economic assessment.
Results
996 participants were randomised and included in the intention-to-treat analysis of which 825 provided primary outcome data. The median age of participants was 59 years; 59% were women. At 12 months there was no difference in the onset of CWP (tCBT: 18.0% vs UC: 17.5%; OR 1.05; 95% CI 0.75 to 1.48). Participants who received tCBT were more likely to report better quality of life (EQ-5D-5L utility score mean difference 0.024 (95% CI 0.009 to 0.040)); and had 0.023 (95% CI 0.007 to 0.039) more quality-adjusted life-years at an additional cost of £42.30 (95% CI −£451.19 to £597.90), yielding an incremental cost-effectiveness ratio of £1828. Most secondary outcomes showed significant benefit for the intervention.
Conclusions
A short course of tCBT did not prevent onset of CWP in adults at high risk, but improved quality of life and was cost-effective. A low-cost, short-duration intervention benefits persons at risk of CWP.
Trial registration number
ClinicalTrials.gov Registry (NCT02668003).
Keywords: fibromyalgia, economics, epidemiology
Key messages.
What is already known about this subject?
Cognitive–behavioural therapy (CBT) has demonstrated long-term effectiveness in managing chronic widespread pain (CWP), the characteristic symptom of fibromyalgia.
It improves patient global assessment of change and quality of life.
What does this study add?
A short course of telephone CBT in persons evaluated at high risk of developing CWP does not change onset of CWP but does result in a wide range of health benefits including improved quality of life.
How might this impact on clinical practice or future developments?
CBT derives benefit for a wider group of people with pain than previously established and in relation to this wider group is highly cost-effective.
Introduction
Chronic widespread pain (CWP) is common, with an estimated population prevalence of 10.6% (95% CI 8.6% to 12.9%)1 and is the key feature of fibromyalgia which is the second most common reason (after osteoarthritis) for referral to a rheumatologist.2 CWP and fibromyalgia result in a substantial impact on health-related quality of life3 even in comparison with other musculoskeletal disorders.4
The road to diagnosis is often tortuous and can take many years. Using general practitioner records in the UK, Hughes et al 5 noted that people diagnosed with fibromyalgia had higher rates of primary care visits (average 25 visits/year), prescriptions (11/year) and testing from at least 10 years prior to diagnosis, in comparison with matched persons without such a diagnosis (12 visits/year and 4.5 prescriptions/year). Current European guidelines emphasise the primary role of non-pharmacological therapies for fibromyalgia.6 Evidence in relation to musculoskeletal pain generally, is that the longer the duration of symptoms the less likely they are to improve, including with specific interventions.7
A Versus Arthritis ‘Research roadmap for pain’ produced by scientists, clinicians and patients identified preventing future musculoskeletal pain as one of four main priorities.8 Further recognising its importance, the International Association for the Study of Pain nominated 2020 as ‘The Global Year for the Prevention of Pain’. Despite this, we are not aware of any large-scale trials which have tested approaches to the future prevention of pain.
We have previously shown, in a randomised controlled trial, short-term and long-term effectiveness of a course of cognitive–behavioural therapy delivered by telephone (tCBT) for CWP, compared with usual care (UC).9 10 These results are consistent with a meta-analysis of 29 trials involving 2509 participants and comparing CBT (across all modes of delivery) with control interventions for the management of fibromyalgia, which found high-quality evidence for improving pain and reducing disability, negative mood and fatigue.11
We have developed, validated and refined a statistical model which identifies people at high risk for the future development of CWP.12 13 On the basis of reporting somatic symptoms, sleep problems and aspects of illness behaviour, those classified as ‘high risk’ have around one in four chance of reporting CWP 1 year later. Therefore, building on the evidence for the use of tCBT in the management of CWP and the ability to identify those with risk factors for its development, we undertook a trial to test whether tCBT can reduce CWP onset among those at high risk.
Methods
Study design
We conducted a randomised controlled parallel prevention trial, recruiting through a population-based sampling frame, in three health boards within the UK (National Health Service (NHS) Grampian, NHS Greater Glasgow and Clyde, and NHS Highland), the protocol for which has been previously published.14 Recruitment was through 16 general practices.
Participants
A short screening questionnaire, to determine eligibility for the trial, was mailed to persons aged 25 years and over registered at participating general practices in the study area. Respondents eligible for the trial were those assessed as at high risk of developing CWP, namely that they reported pain which did not satisfy the definition of CWP used in the 1990 American College of Rheumatology criteria for fibromyalgia (namely axial and contralateral body pain present for at least 3 months), and hereafter referred to as ‘ACR criteria’,15 and satisfied at least two of the following: (a) a score >4 on the Illness Behaviour Subscale of the Illness Attitudes Scale,16 (b) a score >2 on the Somatic Symptom Scale score (but excluding items on pain),17 (c) a score >4 on the Sleep Problem Scale.18 In order to ensure that in the event of the trial showing benefit there was a relevant clinical population to which the intervention could be applied, we added to the risk models we had developed the requirement that persons had consulted to primary care within the previous 6 months or reported consulting a doctor frequently. Respondents were not eligible to take part if they had a medical condition which would make the proposed intervention unsuitable (eg, lacked cognitive ability).
Randomisation
Potentially eligible participants were contacted by post with information about the study, and subsequently by a study researcher by telephone to confirm their willingness to take part and provide informed consent. Participants were allocated into groups using a computer randomisation program (1:1 allocation ratio), stratified in blocks by two factors (a) the number of non-pain ‘high-risk’ factors they reported (two or three) since this is related to the risk of CWP onset, and (b) the general practice at which they were registered.
Procedures
The tCBT intervention consisted of an initial assessment (45–60 min), six weekly sessions (each 30–45 min) over 6 weeks, and then booster sessions at 3 and 6 months. The intervention was delivered by therapists trained for the study and accredited by the British Association for Behaviour and Cognitive Psychotherapies. Participants were supported by a self-management manual. The therapist conducted an assessment for problem identification, and they developed with each participant a shared formulation of the current health problem. The sessions involved education about musculoskeletal pain, somatic symptoms and specific techniques such as pacing of activity, behavioural activation, diary keeping, identifying and challenging negative and unhelpful thinking patterns, and the development of a longer term management plan. Participants would record in the manuals agreed goals for the therapist and patient to work towards, and some activities to complete between sessions. Therapists delivering the intervention received a 2-day training programme conducted by the investigators. Therapists were supervised every 2 weeks (by investigators KL and PK) throughout the delivery of the intervention. The number of telephone consultations conducted was recorded, although the therapist and participant could jointly agree that no further sessions were required before all planned sessions had been completed.
The group allocated to UC received no additional intervention, reflecting the fact there is no specific intervention provided to patients currently for the prevention of CWP. There was no restriction on what this care could involve.
Follow-up questionnaires were mailed to participants at 3, 12 and 24 months after the treatment start date (for participants in the active treatment group) or dummy treatment start date (for those in UC). The dummy treatment start date for a participant randomised to UC was determined by the treatment start date of the last participant to be randomised to receive active treatment. At 3 and 12 months, participants who did not return their questionnaire were telephoned to ask them to complete and return it, while at 24 months the follow-up call also offered the option of completing a shortened version by telephone.
Outcomes
The principal outcome time was at 12-month follow-up and the primary outcome was ACR criteria for CWP. Secondary outcomes were: Global Impression of Change, Illness Behaviour Subscale of the Illness Attitudes Scale,16 the Somatic Symptom Scale (excluding items on pain),17 the Sleep Problem Scale,18 the presence of pain over the past month, Widespread Pain Index (WPI) and Symptom Severity Scale (SSS) of the 2010 (revised) criteria for fibromyalgia,19 psychological distress measured using the General Health Questionnaire (GHQ),20 Chalder Fatigue Scale,21 quality of life (EuroQol Questionnaire-five dimensions-five levels/EQ-5D-5L)22 and capability (ICEpop CAPability measure for Adults/ICECAP-A).23 Further details of secondary outcome (including coding) are given in the online supplemental file.
annrheumdis-2020-219091supp001.pdf (225.5KB, pdf)
Statistical analysis
All analyses were undertaken using Stata V.15. The a priori target sample size was 946 participants, which would provide 90% power to detect a group difference of 9% (21% vs 12%) in the percentage of participants with CWP at 12-month follow-up, assuming a 5% significance level and an 80% response rate.
Where there were missing data within a scale score, we followed standard procedures (where available) as to if and how the missing values could be imputed. The analysis of the primary outcome used a binary logistic regression model with results expressed as an OR with 95% CI. Secondary outcomes were analysed using linear, binary logistic, ordinal logistic or Poisson regression models for continuous, binary, ordinal and count variables, respectively. Model results were reported using mean differences, ORs or incidence rate ratios (IRRs) as appropriate. Except for EQ-5D-5L, mean differences less than 0 and ORs/IRRs less than 1 favour the treatment group. All models were adjusted (adj) for the number of non-pain risk factors on screening (two or three), age (years), gender, general practice (random effect) and baseline score of the outcome measure (where applicable). The primary analysis was by intention to treat—that is, participants were analysed according to randomised group regardless of the number of sessions received. Separate analyses were performed for each time point (3, 12 and 24 months). For the primary outcome, a p value less than 0.05 was regarded as statistically significant; for secondary outcomes p<0.01 was used. Additional sensitivity analyses were conducted for the primary outcome only and are detailed in the online supplemental file.
Health economic analysis
Health service resource used over 24 months was assessed using responses from self-reported questionnaires. Participants were asked to recall their usage for the previous 4-week period at each follow-up. Resource use was then valued using published UK sources—NHS Reference Cost and the Personal and Social Service Research Unit for NHS primary and secondary care, and published literature for care obtained from private providers.24 The unit costs used for the valuation of health service resource use are reported in online supplemental table S1. The intervention cost was based on the actual number and duration of telephone calls per participant (‘direct time’), plus time spent on training and supervision. An allowance for indirect time spent was also included and this was based on an assumed ratio of 1:1 between time spent on participant contact and other activities conducted by therapists. Training costs were estimated using the time spent in training by trainers and trainees (tCBT therapists). A fortnightly supervision cost was estimated by assuming 30 sessions per therapist (30 min per session) were provided. Costs were expressed in 2017/2018 prices. Health utility scores were assigned based on responses to the EQ-5D-5L at each follow-up, and these were converted using the ‘crosswalk’ procedure to EQ-5D-3L.25 There is currently no consensus on the preferred EQ-5D-5L tariff for use in economic evaluation, although the National Institute for Health and Care Excellence (NICE) recommends the use of the ‘crosswalk’ procedure (a validated mapping function) to derive health utility scores for the EQ-5D-5L from the EQ-5D-3L tariff (https://www.nice.org.uk/about/what-we-do/our-programmes/nice-guidance/technology-appraisal-guidance/eq-5d-5l accessed 20 November 2020). These utility scores were used to estimate quality-adjusted life-years (QALYs) over the 24 months using the area under the curve method.26 Costs and QALYs incurred beyond 12 months were discounted at the rate of 3.5% per annum.
annrheumdis-2020-219091supp002.pdf (248.5KB, pdf)
The within-trial economic analysis was conducted over 24 months from a UK NHS cost perspective. To estimate the differences in mean costs and QALYs between groups, generalised linear models with adjustment for minimisation factors, baseline cost and baseline utility score were performed. A γ family with log-link function and a Poisson family with power 0.5 link function were specified for the cost and QALY data, respectively. Missing data were addressed using multiple imputation by chained equations (MICE). Variance surrounding the incremental costs and QALYs was characterised using non-bootstrapping (500 iterations), with MICE (m=5) nested within the bootstrap loops.27 Cost-effectiveness acceptability curves were constructed, using 500 replications of each incremental cost-effectiveness ratio (ICER) and the net monetary benefit framework, to determine the probability of the alternative interventions being considered cost-effective at different willingness to pay per QALY (£20 000–£30 000 per QALY used are commonly applied ceiling ratios in the UK). Several sensitivity analyses were performed to explore the impact on the results of uncertainty in estimates made—(1) using complete cases of costs and QALYs, (2) including private care costs, (3) using alternative tCBT costing methodology (actual trial expenses incurred by therapists and the cost of a complete tCBT course) and (4) using ICECAP tariff as the measure of effectiveness.
The trial was evaluated by the Trial Steering Committee as not requiring a Data Monitoring Committee.
Results
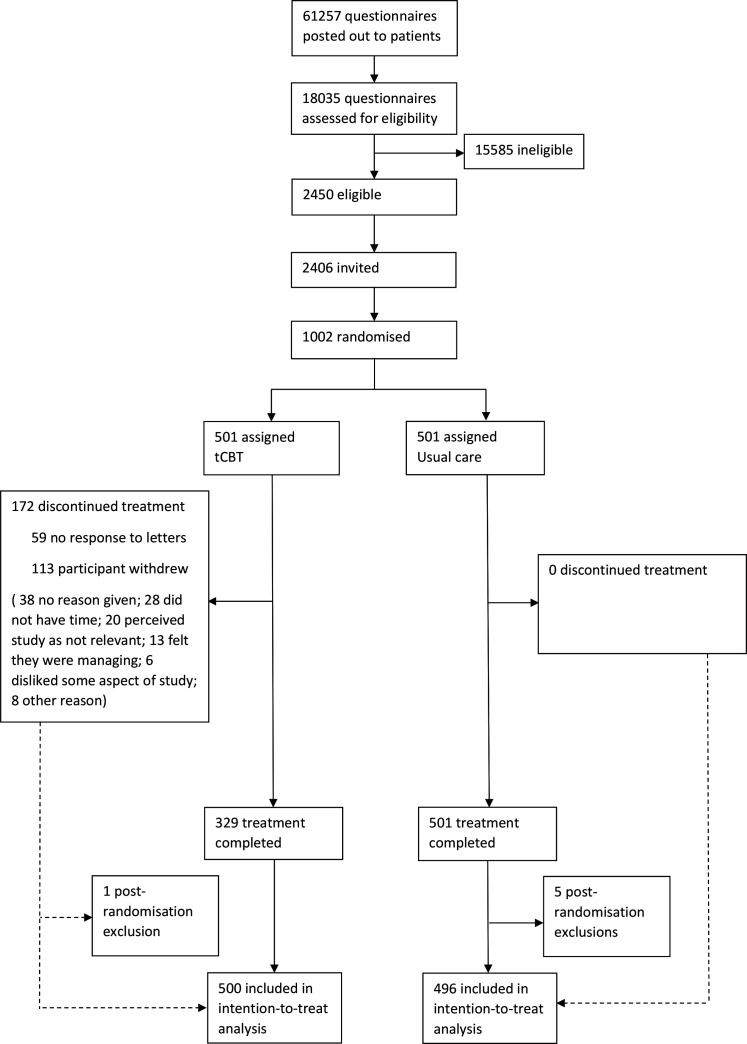
Of 61 257 screening questionnaires sent between 4 April 2016 and 4 November 2016 to patients registered at 16 general practices, 18 035 completed questionnaires were returned. From those returning a completed questionnaire, 2406 were identified as potentially eligible and sent invitations to take part in the trial. A total of 1002 participants were recruited to the trial and randomised, 501 to tCBT and 501 to UC, between May 2016 and March 2017. Six participants were subsequently determined to be ineligible for the trial and were excluded from analyses (see Trial profile: figure 1) leaving a final study size of 500 and 496 in the tCBT and UC arms, respectively. At the 3-month, 12-month and 24-month follow-up, there were 823, 825 and 853 respondents who provided primary outcome data, respectively. Most participants (51%) came from the lowest two quintiles of deprivation, while 18% came from the two most deprived quintiles.
Figure 1.
Trial profile. tCBT, telephone-delivered cognitive–behavioural therapy.
Participants at the time of recruitment had a median age of 59 years (IQR 48–69), 59% were women, and 52% were working full-time or part-time (table 1). The median EQ-5D utility score was 0.74 (IQR 0.65–0.80). The vast majority satisfied only two of the non-pain criteria for eligibility, nearly always on the basis of a high score on the illness behaviour subscale of the Illness Attitudes Scale and having sleep problems. Only 6% of the study sample satisfied the somatic symptoms criterion. The tCBT and UC groups were well matched in terms of the measured health-related factors.
Table 1.
Baseline characteristics by treatment arm in the ITT population
| Characteristic | Randomised groups | |
| tCBT (n=500) | Usual care (n=496) | |
| Median (IQR) | Median (IQR) | |
| Age (years) | 58.8 (47.7–68.7) | 59.5 (47.9–68.9) |
| N (%) | N (%) | |
| Gender | ||
| Male | 209 (41.8) | 204 (41.0) |
| Female | 291 (58.2) | 292 (58.9) |
| Employment status | ||
| Working (full or part-time) | 277 (55.4) | 244 (49.2) |
| Unable to work because of health | 18 (3.6) | 30 (6.0) |
| Retired | 168 (33.6) | 177 (35.7) |
| Other | 37 (7.4) | 45 (9.1) |
| CWP risk profile: | ||
| Illness behaviour score >4 | ||
| No | 1 (0.2) | 2 (0.5) |
| Yes | 498 (99.6) | 494 (99.5) |
| Not known* | 1 (0.2) | 0 (0.0) |
| Somatic Symptom Scale score >2 | ||
| No | 472 (94.4) | 462 (93.1) |
| Yes | 28 (5.6) | 34 (6.9) |
| Sleep problems score >4 | ||
| No | 1 (0.2) | 2 (0.4) |
| Yes | 499 (99.8) | 493 (99.4) |
| Not known* | 0 (0.0) | 1 (0.2) |
| CWP risk profile factors present (N) | ||
| 2 | 474 (94.8) | 466 (94.0) |
| 3 | 26 (5.2) | 30 (6.0) |
| Median (IQR) (n)† | Median (IQR) (n) | |
| Psychological distress (GHQ) | 1 (0–4) (499) | 1 (0–4) (494) |
| Quality of Life (EQ-5D-5L utility score) | 0.74 (0.65–0.80) (499) | 0.74 (0.64–0.80) (496) |
| ICECAP-A | 0.91 (0.81–0.95) (495) | 0.90 (0.79–0.95) (491) |
| Fibromyalgia research criteria | ||
| WPI | 3 (1–4) (499) | 2 (1–4) (492) |
| SSS | 4 (3–6) (497) | 4 (3–5) (494) |
*Where individuals completed half or fewer items, the score was classified as not known, but individuals could still be eligible for recruitment based on their responses to other items answered.
†The number of persons for whom a scale score could be calculated.
EQ-5D-5L, EuroQol Questionnaire-five dimensions-five levels; GHQ, General Health Questionnaire; ICECAP-A, ICEpop CAPability measure for Adults; ITT, intention to treat; SSS, Symptom Severity Scale; tCBT, telephone-delivered cognitive–behavioural therapy; WPI, Widespread Pain Index.
Results for all outcome measures at the primary time point (12 months) are shown in table 2. The corresponding results at 3 and 24 months are shown in online supplemental tables S2–S3. Table 3 provides a summary of all primary and secondary outcomes at all time points and shows adjusted and unadjusted effect sizes.
Table 2.
Outcomes by treatment arm at 12 months (intention-to-treat analysis)
| Characteristic | Randomised groups | |
| tCBT (n=500) | Usual care (n=496) | |
| Primary outcome | N (%) | N (%) |
| Chronic widespread pain | ||
| No | 315 (82.0) | 364 (82.5) |
| Yes | 69 (18.0) | 77 (17.5) |
| Secondary outcome | ||
| Global impression of change | ||
| Very much better | 24 (6.5) | 15 (3.5) |
| Much better | 88 (23.7) | 59 (13.8) |
| A little better | 90 (24.3) | 84 (19.6) |
| No change | 83 (22.4) | 126 (29.4) |
| A little worse | 65 (17.5) | 119 (27.7) |
| Much worse | 18 (4.9) | 23 (5.4) |
| Very much worse | 3 (0.8) | 3 (0.7) |
| Pain reported | ||
| No | 79 (20.6) | 68 (15.4) |
| Yes | 305 (79.4) | 373 (84.6) |
| CWP risk profile | ||
| Somatic symptoms score | ||
| 0 | 210 (56.5) | 228 (52.8) |
| 1 | 103 (27.7) | 123 (28.5) |
| 2–5 | 59 (15.9) | 81 (18.8) |
| Illness behaviour score, mean (SD) (n)* | 8.21 (4.04) (371) | 8.96 (4.19) (431) |
| Sleep problems score, mean (SD) (n) | 8.20 (4.89) (373) | 9.20 (5.16) (432) |
| Psychological distress (GHQ score) | ||
| 0 | 201 (54.5) | 202 (46.8) |
| 1 | 59 (16.0) | 54 (12.5) |
| 2–5 | 68 (18.4) | 113 (26.2) |
| 6–12 | 41 (11.1) | 63 (14.6) |
| Mean (SD) (n) | Mean (SD) (n) | |
| Chalder Fatigue Score | 12.6 (4.5) (370) | 13.6 (4.4) (433) |
| Median (IQR) (n) | Median (IQR) (n) | |
| Quality of Life (EQ-5D utility score) | 0.74 (0.66–0.84) (371) | 0.74 (0.65–0.82) (435) |
| ICECAP-A | 0.91 (0.82–0.97) (368) | 0.89 (0.78–0.95) (429) |
| Fibromyalgia research criteria | ||
| WPI | 2 (1–4) (366) | 2 (1–4) (427) |
| SSS | 3 (2–5) (369) | 4 (2–5) (431) |
*The number of persons for whom a scale score could be calculated.
EQ-5D, EuroQol Questionnaire-five dimensions; GHQ, General Health Questionnaire; ICECAP-A, ICEpop CAPability measure for Adults; SSS, Symptom Severity Scale; tCBT, telephone-delivered cognitive–behavioural therapy; WPI, Widespread Pain Index.
Table 3.
Summary of the primary and secondary outcomes across follow-up points*
| Outcome | Time point (months) | Analysis method (effect size) | Adjusted† effect size (95% CI) | P value | Unadjusted effect size (95% CI) | P value | |
| Primary outcome | |||||||
| CWP CWP (per protocol) CWP (with multiple imputation) |
3 | Logistic regression (OR) | 1.08 (0.74 to 1.58) | 0.691 | 1.07 (0.75 to 1.53) | 0.716 | |
| 1.15 (0.75 to 1.75) | 0.519 | 1.18 (0.77 to 1.66) | 0.522 | ||||
| 1.06 (0.74 to 1.54) | 0.749 | 1.05 (0.72 to 1.53) | 0.816 | ||||
| CWP‡
CWP (per protocol) CWP (with multiple imputation) |
12 | 1.05 (0.75 to 1.48) | 0.771 | 1.04 (0.72 to 1.48) | 0.849 | ||
| 1.11 (0.81 to 1.50) | 0.519 | 1.09 (0.74 to 1.60) | 0.673 | ||||
| 1.04 (0.75 to 1.45) | 0.982 | 1.03 (0.74 to 1.42) | 0.964 | ||||
| CWP CWP (per protocol) CWP (with multiple imputation) |
24 | 0.85 (0.68 to 1.07) | 0.163 | 0.84 (0.61 to 1.18) | 0.317 | ||
| 0.85 (0.64 to 1.12) | 0.241 | 0.84 (0.58 to 1.20) | 0.330 | ||||
| 0.85 (0.66 to 1.09) | 0.220 | 0.84 (0.65 to 1.09) | 0.196 | ||||
| CWP | 3, 12, 24 | GEE (OR) | 1.00 (0.96 to 1.04) | 0.923 | 1.00 (0.96 to 1.04) | 0.835 | |
| Secondary outcomes | |||||||
| Global impression of change§ | 3 | Ordinal logistic regression (OR) | 0.42 (0.32 to 0.55) | <0.001 | 0.43 (0.34 to 0.56) | <0.001 | |
| 12 | 0.51 (0.39 to 0.67) | <0.001 | 0.53 (0.41 to 0.68) | <0.001 | |||
| 24 | 0.55 (0.43 to 0.70) | <0.001 | 0.58 (0.45 to 0.73) | <0.001 | |||
| CWP risk profile | Somatic symptoms score | 3 | Ordinal logistic regression (OR) | 0.79 (0.60 to 1.03) | 0.084 | 0.83 (0.64 to 1.08) | 0.173 |
| 12 | 0.86 (0.71 to 1.04) | 0.112 | 0.85 (0.65 to 1.11) | 0.237 | |||
| 24 | 0.81 (0.59 to 1.12) | 0.206 | 0.90 (0.67 to 1.21) | 0.498 | |||
| Illness behaviour score | 3 | Linear regression (mean difference) | −0.17 (−0.58 to 0.24) | 0.385 | −0.25 (−0.79 to 0.29) | 0.360 | |
| 12 | −0.81 (−1.54 to −0.09) | 0.030 | −0.74 (−1.32 to −0.17) | 0.011 | |||
| 24 | −1.25 (−2.15 to −0.35) | 0.010 | −1.20 (−1.83 to −0.58) | <0.001 | |||
| Sleep problems score | 3 | Linear regression (mean difference) | −0.62 (−1.26 to 0.02) | 0.057 | −0.62 (−1.31 to 0.08) | 0.081 | |
| 12 | −0.95 (−1.48 to −0.42) | 0.002 | −1.00 (−1.70 to −0.30) | 0.005 | |||
| 24 | −0.51 (−1.25 to 0.23) | 0.161 | −0.52 (−1.39 to 0.16) | 0.117 | |||
| Psychological distress (GHQ) | 3 | Ordinal logistic regression (OR) | 0.55 (0.43 to 0.69) | <0.001 | 0.58 (0.45 to 0.76) | <0.001 | |
| 12 | 0.65 (0.50 to 0.86) | 0.002 | 0.70 (0.54 to 0.90) | 0.007 | |||
| 24 | 0.76 (0.60 to 0.96) | 0.024 | 0.74 (0.56 to 0.98) | 0.037 | |||
| Chalder Fatigue Score | 3 | Linear regression (mean difference) | −1.36 (−2.10 to −0.64) | 0.001 | −1.40 (−1.97 to −0.82) | <0.001 | |
| 12 | −1.02 (−1.63 to −0.42) | 0.003 | −1.03 (−1.64 to −0.42) | 0.001 | |||
| 24 | −0.93 (−1.62 to −0.23) | 0.012 | −0.93 (−1.58 to −0.27) | 0.006 | |||
| Quality of Life (EQ-5D-5L utility score) | 3 | Linear regression (mean difference) | 0.009 (−0.009 to 0.028) | 0.304 | 0.021 (−0.004 to 0.046) | 0.101 | |
| 12 | 0.024 (0.009 to 0.040) | 0.004 | 0.037 (0.010 to 0.064) | 0.007 | |||
| 24 | 0.030 (0.009 to 0.050) | 0.008 | 0.040 (0.011 to 0.069) | 0.007 | |||
| ICECAP-A tariff | 3 | Ordinal logistic regression (OR) | 1.14 (0.89 to 1.48) | 0.304 | 1.17 (0.86 to 1.59) | 0.323 | |
| 12 | 1.39 (0.94 to 2.04) | 0.096 | 1.39 (1.01 to 1.91) | 0.042 | |||
| 24 | 0.88 (0.67 to 1.15) | 0.338 | 0.99 (0.70 to 1.41) | 0.966 | |||
| Fibromyalgia criteria | Widespread Pain Index | 3 | Poisson regression (IRR) | 0.98 (0.90 to 1.07) | 0.698 | 1.01 (0.93 to 1.10) | 0.771 |
| 12 | 0.88 (0.80 to 0.98) | 0.018 | 0.92 (0.84 to 0.99) | 0.036 | |||
| 24 | 0.88 (0.78 to 0.98) | 0.022 | 0.92 (0.84 to 1.00) | 0.058 | |||
| Symptom Severity Scale | 3 | Linear regression (mean difference) | −0.28 (−0.52 to −0.04) | 0.026 | −0.25 (−0.57 to 0.65) | 0.118 | |
| 12 | −0.52 (−0.75 to −0.28) | <0.001 | −0.59 (−0.91 to −0.27) | <0.001 | |||
| 24 | −0.29 (−0.55 to −0.02) | 0.040 | −0.28 (−0.61 to 0.05) | 0.100 | |||
*Analyses shaded in grey favour tCBT over usual care at prespecified significance level for secondary outcomes (p<0.01). Except for EQ-5D-5L, mean differences less than 0 and ORs less than 1 favour the treatment group.
†Adjusted analyses control for the number of risk factors (two or three), age, gender, baseline score (if applicable) and centre (random effect). Analyses are intention to treat unless otherwise stated.
‡Primary outcome.
§OR of 1 point increase in global impression of change score (worsening of health).
CWP, chronic widespread pain; EQ5D-5D-5L, EuroQol Questionnaire-five dimensions-five levels; GEE, generalised estimating equations; GHQ, General Health Questionnaire; ICECAP-A, ICEpop CAPability measure for Adults; IRR, incidence rate ratio;tCBT, telephone-delivered cognitive–behavioural therapy.
Primary outcome
At the 12-month time point similar percentages in the tCBT and UC groups reported having CWP (tCBT: 69/384 (18.0%), UC: 77/441 (17.5%); adj OR 1.05; 95% CI: 0.75 to 1.48; difference in percentages: adj 0.73, 95% CI: −4.15 to 5.61) (tables 2 and 3)). Very similar results were obtained at 3 months (17.9% vs 16.9%; adj OR: 1.08; 95% CI: 0.74 to 1.58) and 24 months (19.6% vs 22.3%; adj OR: 0.85; 95% CI: 0.68 to 1.07) (online supplemental tables S2–S3, table 3). There was no difference in the interpretation when examining unadjusted results, per protocol results or the analyses using multiple imputation (table 3). The generalised estimating equations model, incorporating data from all three time points, also showed no evidence of a difference (adj OR: 1.00; 95% CI: 0.96 to 1.04; p=0.91).
Secondary outcomes
At 12 months, those randomised to tCBT were more likely to perceive their health to be improved (adj OR (ordinal logistic regression/OLR): 0.51, 95% CI: 0.39 to 0.67) and to report better quality of life (EQ-5D-5L utility scores) (adj mean difference (diff): 0.024, 95% CI: 0.009 to 0.040) (tables 2 and 3). While those who received tCBT had lower illness behaviour (adj mean diff: −0.81; 95% CI: −1.54 to −0.09) and sleep problem scores (adj mean diff: −0.95; 95% CI: −1.48 to −0.42), but there was no significant difference in relation to somatic symptoms (adj OR: 0.86; 95% CI: 0.71 to 1.04). Participants randomised to tCBT had improved distress (GHQ scores) (adj OR: 0.65, 95% CI: 0.50 to 0.86) and lower levels of fatigue (Chalder Scale scores) (adj mean diff: −1.02, 95% CI: −1.63 to −0.42). There was no evidence of a difference for ICECAP-A tariffs (adj OR (OLR): 1.39, 95% CI: 0.94 to 2.04; p=0.10). In relation to the components of criteria for fibromyalgia, they had lower scores on the WPI (adj IRR: 0.88; 95% CI: 0.80 to 0.98) and SSS (adj mean diff: −0.52, 95% CI: −0.75 to −0.28). Of these receiving tCBT, 3.8% met fibromyalgia research criteria at follow-up (in comparison to 6.0% among those receiving UC).
Outcomes across time points
Sensitivity analyses, unadjusted results and findings at 3-month and 24-month time points generally yielded similar observations as those for 12 months (table 3, online supplemental tables S1–S2). There was consistently no effect on the primary outcome. The strongest and most consistent effects were on patient global assessment of change—which showed large and consistent effects across all time points. There were also clear effects of the intervention (in comparison with UC) across all time points with respect to improvement in levels of fatigue and psychological distress. Quality of life was better in the intervention group from 12 months onwards. There was only one serious adverse event reported, it was in the intervention group but unrelated to the intervention.
Health economic analysis
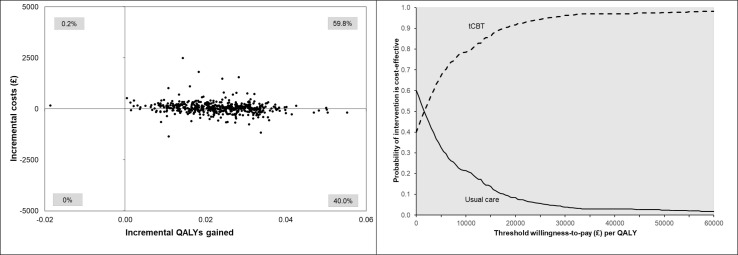
The unadjusted health service resource use and costs per participant are summarised in online supplemental table S4. Participants randomised to tCBT group had an average time of 139 min of direct contact with therapists over the 6-month t-CBT course, and the average tCBT cost was £270.19 per participant. Compared with the UC group, NHS primary and secondary care costs were lower among tCBT group, and private care costs higher. All cost-effectiveness analyses showed that tCBT was associated with an increase in health service costs and an increase in QALYs (table 4). The primary analysis generated a mean of 0.023 (95% CI 0.007 to 0.039) more QALYs per participant at an additional cost of £42.30 (95% CI −£451.19 to £597.90), yielding an ICER of £1828. Based on the results of the non-parametric bootstrap, tCBT was found to have a 91.6% chance of being the preferred strategy at a ceiling ratio of £20 000 per QALY gained (figure 2). Sensitivity analyses showed that this finding was robust to changes in study perspective, inclusion of complete cases only and different assumptions relating to delivery of the intervention in terms of tCBT staff time (online supplemental figure S1 a–d).
Table 4.
Adjusted* mean incremental costs, incremental QALYs and incremental cost-effectiveness ratio over 24 months between tCBT versus usual care
| Analysis | Mean costs, (95% CI) | Mean QALYs, (95% CI) | Incremental mean costs, £ (95% CI)† |
Incremental mean QALYs (95% CI) | ICER (£/QALY) | ||
| tCBT | Usual care | tCBT | Usual care | ||||
| Imputed dataset/ITT analysis (NHS perspective)‡ | 3094.68 (1775.65 to 9074.15) | 3052.38 (1735.77 to 8567.24) | 1.254 (1.238 to 1.270) | 1.231 (1.215 to 1.245) | 42.30 (−451.19 to 597.90) | 0.023 (0.007 to 0.039) | 1828 |
| SA: complete cases (NHS perspective)§ | 2684.53 (1817.69 to 5221.86) | 2454.67 (1645.66 to 4769.87) | 1.444 (1.415 to 1.471) | 1.420 (1.392 to 1.447) | 229.86 (−228.74 to 734.09) | 0.024 (−0.005 to 0.053) | 9608 |
| SA: imputed dataset (NHS+private care perspective) | 4239.22 (2135.82 to 15 332.80) | 4149.10 (2110.98 to 14 039.06) | 1.253 (1.238 to 1.270) | 1.231 (1.215 to 1.247) | 90.12 (−475.79 to 772.98) | 0.022 (0.007 to 0.039) | 4022 |
| SA: imputed dataset using actual trial expenses (NHS perspective)¶ | 3128.61 (1809.54 to 9164.04) | 3027.54 (1734.31 to 8587.83) | 1.254 (1.238 to 1.270) | 1.231 (1.215 to 1.245) | 101.07 (−373.14 to 641.98) | 0.023 (0.007 to 0.039) | 4367 |
| SA: imputed dataset using the cost of a complete tCBT course (NHS perspective)** | 3314.57 (1966.93 to 9059.99) | 2960.98 (1729.67 to 7781.19) | 1.254 (1.238 to 1.270) | 1.231 (1.215 to 1.245) | 353.59 (−80.46 to 1238.07) | 0.023 (0.007 to 0.039) | 15 280 |
| SA: imputed dataset using ICECAP (NHS perspective)†† | 4659.66 (1764.56 to 10 400.07) | 4787.56 (1815.05 to 10 632.20) | 1.288 (1.278 to 1.297) | 1.275 (1.266 to 1.284) | −127.90 (−603.19 to 545.33) | 0.013 (0.003 to 0.023)‡‡ | NA |
*Adjusted for baseline differences (age, gender, number of risk factors present, employment status, centre, baseline EQ-5D health utility score and baseline cost).
†Bootstrapped non-parametric 95% CI (2.5th/97.5th centile). Generalised linear model with γ distribution and log-link function to estimate incremental costs and generalised linear model with Poisson distribution and power 0.5 link function to estimate incremental QALYs/years of full capacity. Discounted at 3.5% per year.
‡Imputed dataset is the ITT analysis. Missing values were imputed to account for all participants included in the ITT analysis.
§593 complete cases were included (tCBT, n=297 and usual care, n=326). Complete cases are those with no missing data on cost and health utility at each time point.
¶Included the actual trial expenses per tCBT participant, £301. This was estimated using the lump-sum trial expenses incurred by therapists, including therapists’ training and tCBT delivery.
**Included the cost of a complete tCBT course per participant, £443. Time spent by therapist, training and supervision were included. The total time spent by the therapist was estimated by assuming that all tCBT participants attended a complete tCBT course consisting of nine sessions.
††Adjusted for baseline differences (age, gender, number of risk factors present, employment status, centre, baseline ICECAP value and baseline cost).
‡‡Incremental years of full capability.
EQ-5D, EuroQol Questionnaire-five dimensions; ICECAP, ICEpop CAPability measure for Adults; ICER, incremental cost-effectiveness ratio; ITT, intention to treat; NA, not applicable; NHS, National Health Service; QALYs, quality-adjusted life-years; SA, sensitivity analysis; tCBT, telephone-delivered cognitive–behavioural therapy.
Figure 2.
Cost-Effectiveness plane and cost-effectiveness acceptability curve between groups (primary analysis using imputed dataset, NHS perspective). Cost-effectiveness planes were based on 500 bootstrap cost-effect pairs (adjusted for age, gender, number of risk factors present, employment status, centre, baseline EQ-5D health utility score and baseline cost). EQ-5D, EuroQol Questionnaire-five dimensions; NHS, National Health Service; QALY, quality-adjusted life-year; tCBT, telephone-delivered cognitive–behavioural therapy.
annrheumdis-2020-219091supp003.pdf (1.4MB, pdf)
Discussion
A short course of tCBT among persons at high risk did not change the proportion of people developing CWP (compared with UC). Those receiving the active intervention were more likely to perceive their health as having improved and report better quality of life as well as lower levels of fatigue and psychological distress. The intervention was highly cost-effective in terms of incremental cost per QALY gained.
Undertaking a primary prevention study presents different challenges to undertaking a treatment study. Most people eligible for the trial probably would not have known what CWP is, nor that they were at high risk of its development. Thus, the intervention was described as ‘maintaining musculoskeletal health’ and introduced in the context of participants having reported pain and other symptoms. Although a set number of sessions for the intervention was planned, it was agreed that at any point the intervention could be stopped with mutual agreement between therapist and participant; with the intervention considered completed. Among participants, 329 (66%) were considered to be completers that is, had the assessment session and either had at least two completed treatment sessions (n=297) or had the assessment session and up to one treatment session with mutual agreement that the intervention was complete (n=32). Of those classed as ‘non-completers’, 97 had no assessment while 75 had an assessment and up to one treatment session.
Why did the trial clearly not change the likelihood of CWP onset while showing positive effects for a range of secondary outcomes (including quality of life)? First, it may be that CBT is not effective in relation to preventing CWP onset. We know that there is a large body of evidence that CBT (including tCBT) is effective in relation to managing CWP, and also for managing some of the symptoms which characterised people at high risk, but it may not be effective at improving the pain in CWP. Our previous trial using CBT in the management of CWP while showing large improvement in patient perception of their condition and in quality of life, did not demonstrate any benefit in terms of the Chronic Pain Grade.10 Second, our risk model may not be the causal model. A change in hypothesised risk factors would only effect a change in outcome if the relationship was causal. This suggests that it would be beneficial to explore, among those at risk, what is the underlying causal mechanism. Altered hypothalamic–pituitary–adrenal axis function is one possible underlying causal mechanism which has been investigated.28 Third, it is understood that there are life-course influences, specifically early life factors, on the development of CWP,29 so it could be that intervening across the adult age range is too late to be effecting a change by means of a short-term intervention. Fourth, it may be that CWP was a poor choice as the primary outcome. There is evidence that people with CWP can move in and out of meeting criteria30 and indeed it may be that we have identified people who commonly experience CWP but recruited them at a time when they did not meet criteria—and the interpretation would be that the intervention did not move participants off that trajectory. Recent data from a longitudinal study in Norway have shown that the transition, among people with pain, to CWP did not represent a clinically significant change in state.31
It is already known that CBT is effective in the management of fibromyalgia11 and this study provides evidence that a wider range of patients may benefit in terms of quality of life. In total 54.5% of the intervention group considered their health had improved (between a little and very much) compared with 36.9% of the UC group, as well as improvements in fatigue, distress and changes in response to symptoms. The incremental cost per QALY gained of £1828 (which was robust to different assumptions modelled in various sensitivity analyses) means that this intervention is highly likely to be cost-effective at the limit, which NICE in the UK, is willing to pay. In terms of delivering behavioural therapies, it has long been recognised that there is a shortage of clinical psychologists in the UK. It is not necessary to have such persons delivering behavioural therapy to all such patients even where CBT is identified as appropriate. In this study, the intervention was delivered by therapists accredited by the British Association for Behaviour and Cognitive Psychotherapies. At a minimum this requires a Bachelor of Science degree and a 2-year course leading to a postgraduate diploma in cognitive–behaviour psychotherapies. Further there has been a considerable amount of research in terms of internet-based therapies. The potential advantage of such a self-directed approach is that it requires less input by the therapist (usually somewhere between 1 and 15 mins/week). Further, a meta-analysis of 20 studies involving 1460 participants showed that internet-delivered CBT was effective in the treatment of insomnia,32 while a meta-analysis of 20 studies involving 1418 participants comparing face-to-face and internet-delivered CBT for psychiatric and somatic symptoms found that ‘there was no evidence to conclude that they were not equivalent’.33 Studies have also examined training members of the care team (usually nurses) to deliver behavioural therapy in terms of making any service for chronic pain sustainable, and these have been shown to be effective.34 Thus, we need to consider different professionals and ways of delivering CBT, particularly if we widen the group eligible to receive it, and there is no doubt that the large changes to how health services are delivered, caused by COVID-19, will only accelerate moves to the greater use of remote delivery of care.
In summary, this trial has shown that a short course of tCBT does not prevent the onset of CWP in adults assessed as being at high risk. It did however positively change most other health indicators measured, including quality of life, and was highly cost-effective. It demonstrates that a low-cost, short-duration intervention benefits a wider range of people with musculoskeletal symptoms than previously considered.
Acknowledgments
We acknowledge the contribution of the Trial Steering Committee to the successful conduct of the study. The members were Professors Ernest Choy (Cardiff University), Tamar Pincus (Royal Holloway, University of London) and Gordon Taylor (Bath University). We thank Brian Taylor and Mark Forrest from the Centre for Healthcare Randomised Trials (CHaRT) at the University of Aberdeen for their technical assistance and Professor Graeme MacLennan, Director of CHaRT, for methodological input. Professor John Norrie (originally University of Aberdeen now University of Edinburgh) and Dr Majid Artus (originally Keele University, now the Osmaston surgery, Derbyshire) were study investigators at the time of grant award but subsequently left the study. We thank Kathy Longley (a representative of Fibromyalgia Action UK) for her input to the grant application and the project, as well as from members of the public on the University of Aberdeen College of Life Sciences and Medicine Research Interest Group. The prioritisation of 'Prevention of chronic pain' arose from a 2012 meeting of the Arthritis Research UK Clinical Study Group in Pain to which patients contributed.
Footnotes
Handling editor: Josef S Smolen
Twitter: @UAberdeenEpi, @mahkusjaybee, @hteraG_senoJ
Contributors: GJM was CI and MB study coordinator. KL and PK were responsible for designing and overseeing the delivery of the intervention. GP was the trial statistician and designed the analysis plan, and this role was latterly taken over by NS who conducted the analysis. PM was responsible for designing the health economic analysis which was undertaken by HC. GJM drafted the manuscript with input from MB, HC, PM and NS. All authors contributed important intellectual content to trial design and execution, and commented on drafts of the manuscript.
Funding: The study was funded by Arthritis Research UK (now Versus Arthritis) grant number: 20748. Costs for delivery of the intervention were provided by NHS Grampian, NHS Greater Glasgow and Clyde, and NHS Highland.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request. There is an application process by which researchers may request to access data in this manuscript. In principle we are willing to share de-identified data.
Ethics statements
Patient consent for publication
Not required.
Ethics approval
Ethical approval was obtained from Cornwall and Plymouth Research Ethics Committee reference 16/SW/0019.
References
- 1. Mansfield KE, Sim J, Jordan JL, et al. A systematic review and meta-analysis of the prevalence of chronic widespread pain in the general population. Pain 2016;157:55–64. 10.1097/j.pain.0000000000000314 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Jones GT, Atzeni F, Beasley M, et al. The prevalence of fibromyalgia in the general population: a comparison of the American College of rheumatology 1990, 2010, and modified 2010 classification criteria. Arthritis Rheumatol 2015;67:568–75. 10.1002/art.38905 [DOI] [PubMed] [Google Scholar]
- 3. Burckhardt CS, Clark SR, Bennett RM. Fibromyalgia and quality of life: a comparative analysis. J Rheumatol 1993;20:475–9. [PubMed] [Google Scholar]
- 4. Picavet HSJ, Hoeymans N. Health related quality of life in multiple musculoskeletal diseases: SF-36 and EQ-5D in the DMC3 study. Ann Rheum Dis 2004;63:723–9. 10.1136/ard.2003.010769 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Hughes G, Martinez C, Myon E, et al. The impact of a diagnosis of fibromyalgia on health care resource use by primary care patients in the UK: an observational study based on clinical practice. Arthritis Rheum 2006;54:177–83. 10.1002/art.21545 [DOI] [PubMed] [Google Scholar]
- 6. Macfarlane GJ, Kronisch C, Dean LE, et al. EULAR revised recommendations for the management of fibromyalgia. Ann Rheum Dis 2017;76:318–28. 10.1136/annrheumdis-2016-209724 [DOI] [PubMed] [Google Scholar]
- 7. Von Korff M, Dunn KM. Chronic pain reconsidered. Pain 2008;138:267–76. 10.1016/j.pain.2007.12.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Versus arthritis research roadmap for pain. Available: https://www.versusarthritis.org/media/1672/research-roadmap-pain.pdf
- 9. McBeth J, Prescott G, Scotland G, et al. Cognitive behavior therapy, exercise, or both for treating chronic widespread pain. Arch Intern Med 2012;172:48–57. 10.1001/archinternmed.2011.555 [DOI] [PubMed] [Google Scholar]
- 10. Beasley M, Prescott GJ, Scotland G, et al. Patient-reported improvements in health are maintained 2 years after completing a short course of cognitive behaviour therapy, exercise or both treatments for chronic widespread pain: long-term results from the MUSICIAN randomised controlled trial. RMD Open 2015;1:e000026. 10.1136/rmdopen-2014-000026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Bernardy K, Klose P, Welsch P, et al. Efficacy, acceptability and safety of cognitive behavioural therapies in fibromyalgia syndrome - A systematic review and meta-analysis of randomized controlled trials. Eur J Pain 2018;22:242–60. 10.1002/ejp.1121 [DOI] [PubMed] [Google Scholar]
- 12. McBeth J, Macfarlane GJ, Benjamin S, et al. Features of somatization predict the onset of chronic widespread pain: results of a large population-based study. Arthritis Rheum 2001;44:940–6. [DOI] [PubMed] [Google Scholar]
- 13. Gupta A, Silman AJ, Ray D, et al. The role of psychosocial factors in predicting the onset of chronic widespread pain: results from a prospective population-based study. Rheumatology 2007;46:666–71. 10.1093/rheumatology/kel363 [DOI] [PubMed] [Google Scholar]
- 14. Macfarlane GJ, Beasley M, Prescott G, et al. The maintaining musculoskeletal health (mammoth) study: protocol for a randomised trial of cognitive behavioural therapy versus usual care for the prevention of chronic widespread pain. BMC Musculoskelet Disord 2016;17:179. 10.1186/s12891-016-1037-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Wolfe F, Smythe HA, Yunus MB, et al. The American College of rheumatology 1990 criteria for the classification of fibromyalgia. Report of the multicenter criteria Committee. Arthritis Rheum 1990;33:160–72. 10.1002/art.1780330203 [DOI] [PubMed] [Google Scholar]
- 16. Kellner R. Abridged manual of the illness attitudes scale. University of New Mexico, 1983. [Google Scholar]
- 17. Othmer E, DeSouza C. A screening test for somatization disorder (hysteria). Am J Psychiatry 1985;142:1146–9. 10.1176/ajp.142.10.1146 [DOI] [PubMed] [Google Scholar]
- 18. Jenkins CD, Stanton BA, Niemcryk SJ, et al. A scale for the estimation of sleep problems in clinical research. J Clin Epidemiol 1988;41:313–21. 10.1016/0895-4356(88)90138-2 [DOI] [PubMed] [Google Scholar]
- 19. Wolfe F, Clauw DJ, Fitzcharles M-A, et al. Fibromyalgia criteria and severity scales for clinical and epidemiological studies: a modification of the ACR preliminary diagnostic criteria for fibromyalgia. J Rheumatol 2011;38:1113–22. 10.3899/jrheum.100594 [DOI] [PubMed] [Google Scholar]
- 20. Goldberg DP, Williams P. A user’s guide to the General Health Questionnaire. Windsor, ON, Canada: NFER-NELSON, 1988. [Google Scholar]
- 21. Chalder T, Berelowitz G, Pawlikowska T, et al. Development of a fatigue scale. J Psychosom Res 1993;37:147–53. 10.1016/0022-3999(93)90081-P [DOI] [PubMed] [Google Scholar]
- 22. Herdman M, Gudex C, Lloyd A, et al. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual Life Res 2011;20:1727–36. 10.1007/s11136-011-9903-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Coast J, Peters TJ, Natarajan L, et al. An assessment of the construct validity of the descriptive system for the icecap capability measure for older people. Qual Life Res 2008;17:967–76. 10.1007/s11136-008-9372-z [DOI] [PubMed] [Google Scholar]
- 24. Curtis L. Unit costs of health and social care (2014). Kent, UK: Personal Social Services Research Unit, University of Kent, 2014. https://www.gov.uk/government/publications/nhs-reference-costs-2013-to-2014 [Google Scholar]
- 25. van Hout B, Janssen MF, Feng Y-S, et al. Interim scoring for the EQ-5D-5L: mapping the EQ-5D-5L to EQ-5D-3L value sets. Value Health 2012;15:708–15. 10.1016/j.jval.2012.02.008 [DOI] [PubMed] [Google Scholar]
- 26. Manca A, Hawkins N, Sculpher MJ. Estimating mean QALYs in trial-based cost-effectiveness analysis: the importance of controlling for baseline utility. Health Econ 2005;14:487–96. 10.1002/hec.944 [DOI] [PubMed] [Google Scholar]
- 27. Brand J, van Buuren S, le Cessie S, et al. Combining multiple imputation and bootstrap in the analysis of cost-effectiveness trial data. Stat Med 2019;38:210–20. 10.1002/sim.7956 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Blackburn-Munro G. Hypothalamo-Pituitary-Adrenal axis dysfunction as a contributory factor to chronic pain and depression. Curr Pain Headache Rep 2004;8:116–24. 10.1007/s11916-004-0025-9 [DOI] [PubMed] [Google Scholar]
- 29. Jones GT, Power C, Macfarlane GJ. Adverse events in childhood and chronic widespread pain in adult life: results from the 1958 British birth cohort study. Pain 2009;143:92–6. 10.1016/j.pain.2009.02.003 [DOI] [PubMed] [Google Scholar]
- 30. Glette M, Stiles TC, Borchgrevink PC, et al. The natural course of chronic pain in a general population: stability and change in an Eight-Wave longitudinal study over four years (the HUNT pain study). J Pain 2019;S1526-5900(19)30845-4. [DOI] [PubMed] [Google Scholar]
- 31. Landmark T, Romundstad P, Butler S, et al. Development and course of chronic widespread pain: the role of time and pain characteristics (the HUNT pain study). Pain 2019;160:1976–81. 10.1097/j.pain.0000000000001585 [DOI] [PubMed] [Google Scholar]
- 32. Zachariae R, Lyby MS, Ritterband LM, et al. Efficacy of internet-delivered cognitive-behavioral therapy for insomnia - A systematic review and meta-analysis of randomized controlled trials. Sleep Med Rev 2016;30:1–10. 10.1016/j.smrv.2015.10.004 [DOI] [PubMed] [Google Scholar]
- 33. Carlbring P, Andersson G, Cuijpers P, et al. Internet-Based vs. face-to-face cognitive behavior therapy for psychiatric and somatic disorders: an updated systematic review and meta-analysis. Cogn Behav Ther 2018;47:1–18. 10.1080/16506073.2017.1401115 [DOI] [PubMed] [Google Scholar]
- 34. Rutledge T, Atkinson JH, Holloway R, et al. Randomized controlled trial of nurse-delivered cognitive-behavioral therapy versus supportive psychotherapy telehealth interventions for chronic back pain. J Pain 2018;19:1033–9. 10.1016/j.jpain.2018.03.017 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
annrheumdis-2020-219091supp001.pdf (225.5KB, pdf)
annrheumdis-2020-219091supp002.pdf (248.5KB, pdf)
annrheumdis-2020-219091supp003.pdf (1.4MB, pdf)
Data Availability Statement
Data are available upon reasonable request. There is an application process by which researchers may request to access data in this manuscript. In principle we are willing to share de-identified data.