Highlights
-
•
High prolactin (PRL) levels are associated with weight gain and impaired metabolic profiles.
-
•
Long-term control of hyperprolactinemia can be attained by first-line surgery and medical therapy.
-
•
Normalization of PRL improves patients’ BMI and fasting glucose levels.
-
•
Marginal changes in patients’ metabolic profiles are noted regardless of the primary therapy.
-
•
Not dopamine agonists per se, but rather the control of hyperprolactinemia plays a role in metabolic profile alterations.
Keywords: BMI, Dopamine agonist, Metabolic profile, Prolactinoma, Surgery
Abstract
Objectives
High prolactin levels have been associated with weight gain and impaired metabolic profiles. While treatment with dopamine agonists (DAs) has been shown to improve these parameters, there is a lack of surgical series on its comparative effect in prolactinoma patients.
Methods
In this retrospective, comparative study, consecutive patients with a prolactinoma were enrolled if treated with first-line transsphenoidal surgery (TSS) or with DAs. Patients with prolactinomas of Knosp grade >2 and those with a follow-up <24 months were excluded, as were patients with missing laboratory metabolic parameters at baseline and over the long-term. Effects of either treatment on BMI and the metabolic profile were analyzed, and independent risk factors for long-term obesity were calculated.
Results
Primary treatment was TSS for 12 patients (40%) and DAs for 18 patients (60%). At diagnosis, no significant differences between the two cohorts were observed with regard to adenoma size, Knosp grading, baseline prolactin (PRL) levels, prevalence of hypogonadism, or laboratory metabolic parameters. Mean follow-up was 51.9 months (range, 24–158). Over the long-term, both TSS and DAs led to the control of hyperprolactinemia (92% vs. 72%) and hypogonadism (78% vs. 83%) in the majority of patients. While a significant decrease in patients’ BMI and fasting glucose were observed, changes in the lipid profile were marginal and independent of the treatment modality. At baseline, increased BMI—but not the primary treatment strategy—was an independent predictor of long-term obesity.
Conclusions
Over the long-term, patients’ BMI and FG improve, but changes in the metabolic profile are marginal and independent of the primary treatment. It is presumable that not DAs per se, but rather the control of hyperprolactinemia plays a role in patients’ metabolic profile alterations.
Introduction
Obesity represents a global epidemic with increased morbidity and mortality [1]. Endocrine disorders, in particular Cushing's syndrome, hyperprolactinaemia-induced hypogonadism, acromegaly and hypothyroidism have been associated with weight gain and exacerbated metabolic dysfunction [2], [3]. The relationship between the metabolic syndrome and hyperprolactinemia suggests that prolactin (PRL) may be per se a modulator of body weight [4]. Prolactinoma is a frequent source of hyperprolactinemia and represents the most common hormone-secreting pituitary tumour [5]. While dopamine agonists (DAs) are the first-line approach [6], [7], [8], transsphenoidal surgery (TSS) has increasingly emerged as an alternative treatment option [9], [10], [11]. Reasons include the need for ongoing DA therapy in up to four fifths of patients and potential adverse medical effects over the long-term [12], [13], [14], [15], [16].
Improvement of the metabolic profile in prolactinoma patients has been attributed to DA therapy per se [17], [18], [19]. Currently, there is a paucity of data on the comparative impact of first-line surgery on prolactinoma patients’ body mass index (BMI) and metabolic profile.
In this cross-sectional long-term follow-up study, we aimed at analyzing the effect of either treatment approach in a matched cohort of patients, based on their BMI, glucose and lipid profiles.
Methods
Study design
This is a retrospective comparative analysis of a prospectively maintained database. We reviewed records from prolactinoma patients consecutively treated by either TSS or DAs as first-line therapy between January 1997 and December 2015. Clinical and laboratory metabolic parameters were assessed at baseline and over the long-term (≥2years). All patients fulfilled the diagnostic criteria of having a PRL-secreting pituitary adenoma [20]. The Human Research Ethics Committee of Bern (Kantonale Ethikkommission KEK Bern, Bern, Switzerland) approved the study (KEK n° 10-10-2006 and 8-11-2006).
Inclusion and exclusion criteria
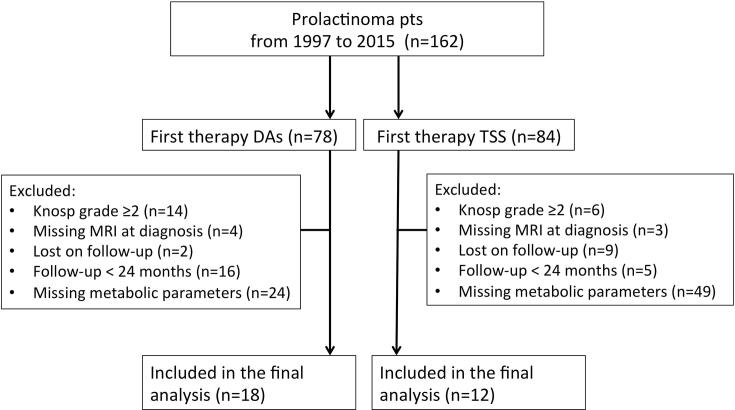
A flow chart of the patient selection process is depicted in Fig. 1. Patients with mixed-secreting adenomas, or those with secondary hyperprolactinemia due to antidepressants, opiates or neuroleptics, were excluded from the analysis. There were 84 patients treated by TSS and 78 patients treated with DAs. Patients with missing metabolic parameters at baseline and at last follow-up were excluded (n = 73). The same was true for patients with a follow-up <24 months (n = 21) or those with prolactinomas of a Knosp grade ≥2 (n = 22). The latter was done to prevent any selection bias towards medical therapy in prolactinomas infiltrating the cavernous sinus [21].
Fig. 1.
Flow chart of patient selection process Out of 162 patients with a prolactinoma, first therapy was TSS in 84 patients and DA in 78 patients. 30 patients met the final inclusion criteria.
Biochemical assessment
The following metabolic parameters were measured after an overnight fast: glucose (i.e., fasting glucose, FG), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), and triglyceride (TG) concentrations. Impaired fasting glucose (IFG) was defined as FG values between 6.1 and 6.9 mmol/l. PRL levels, including the immunoradiometric PRL assay with serum dilution in order to overcome the high-dose PRL hook effect [22], [23], and pituitary axis deficits were assessed. The upper limit of the PRL level was 25 µg/L for women and 20 µg/L for men [24]. Impaired secretion of one or more pituitary hormones was indicative of hypopituitarism. Secondary adrenal insufficiency was defined by the presence of low serum cortisol levels (<50 nmol/L) or normal cortisol but inadequate responses to the adrenocorticotropin (ACTH) stimulation test or insulin tolerance test. Secondary hypothyroidism was defined as low-normal thyroid-stimulating hormone (TSH) levels and a low free thyroxin (FT4) level. A gonadotropin deficiency or central hypogonadism was defined as low-normal levels of gonadotropins in parallel with low total estradiol/testosterone levels. Immunohistochemical analysis according to the WHO classification for neuroendocrine tumors was confirmed in the surgical cohort [25].
Assessment of BMI
Standard BMI was calculated for all patients [26]. A BMI of 21–25 kg/m2 was considered normal. BMI 26–30 kg/m2 was overweight, and BMI > 30 kg/m2) was obese.
Radiological assessment
A conventional 1.5-T or 3-Tesla pituitary MRI was conducted at diagnosis and at follow-up. The standard protocol included a proton density/T2-weighted whole-brain scan with 5 mm slice thickness and both unenhanced and contrast-enhanced overlapping 3 mm scans in the sagittal and coronal planes over the sellar region, as previously reported [21], [27]. An adenoma with a diameter of 1–10 mm was defined as a microadenoma and an adenoma ≥10 mm as a macroadenoma. Infiltration of the cavernous sinus was recorded according to the Knosp classification [28], [29].
Treatment modalities for prolactinomas
Treatment options were discussed at the weekly interdisciplinary pituitary board meeting. Surgery was considered for prolactinomas not extending beyond the medial carotid line (i.e., Knosp grade ≤1) [21], [30]. The indication for first-line surgery was further discussed with the patient. Based on his/her preferences, medical therapy was initiated or surgery was performed. Types of DA-agonists, maximal doses, and duration of treatment were noted (e.g., bromocriptine, and cabergoline). In Switzerland, both treatment options are covered by health insurance, so cost should not be a factor in the choice of treatment. In the surgical cohort, pituitary surgery was performed using a transseptal, transsphenoidal microsurgical approach (i.e., TSS) with sellar reconstruction to prevent cerebrospinal fistula, as previously described [9], [10], [31], [32], [33], [34].
Long-term assessment
If PRL levels had normalized and a tumor reduction of >50% was attained at the time of radiological follow-up, DAs were tapered 24 months after initiation of the medical therapy [35], [36]. Recurrence was defined as an increase in PRL levels above the normal range (>25 µg/L for women, >20 µg/L for men) during the last follow-up period after a previous remission, irrespective of radiological findings [37], [38].
Statistical analysis
Data were analyzed using IBM SPSS statistical software Version 24.0 (IBM Corp., New York, NY, USA) and GraphPad Prism (V7.04 software, San Diego, CA, USA). Continuous variables were examined for homogeneity of variance and are expressed as mean ± SD unless otherwise noted. Serum PRL levels are presented as median values and interquartile range (IQR). Categorical variables are given as numbers and percentages. For comparisons of means between groups, Student’s t-test was used for normally distributed data, and the Mann–Whitney test for nonparametric data (i.e., FG, HDL-C, and TG levels). The Wilcoxon signed-rank test was used to evaluate paired differences in PRL levels before and after treatment. Categorical variables were compared using Pearson’s chi-square test or Fisher’s exact test, as appropriate. Odds ratios (ORs) and 95% confidence intervals (CIs) of independent factors for obesity at last follow-up were analyzed by univariable and multivariable logistic regression. The variables tested were: age at diagnosis, sex, patient’s BMI (kg/m2), PRL levels at baseline, the primary treatment (i.e., DA), hypogonadism, and follow-up time. The multivariable logistic regression analysis included all dependent risk factors in the univariable regression with a p value ≤ 0.3. Baseline PRL values were log transformed before being imputed in the regression analysis, as data showed a positively skewed distribution. Significance level was set at <5%.
Results
Baseline characteristics
Among all patients treated for prolactinomas at our institution in the study period, 13 women and 17 men fulfilled the study inclusion criteria. Baseline characteristics of patients are detailed in Table 1. First therapy was TSS in 12 patients (40%) and DA in 18 (60%). No significant differences between the two cohorts were observed with regard to prevalence of a macroadenoma, the presence of a Knosp grade 1 adenoma, baseline PRL levels, affected pituitary axes including the prevalence of hypogonadism, or the metabolic profile (i.e., glucose and lipid concentrations).
Table 1.
Patient characteristics at baseline.
| Baseline characteristics | All patients | DA | TSS | p value |
|---|---|---|---|---|
| Number of patients, n (%) | 30 (100) | 18[60] | 12[40] | |
| Sex (women), n (%) | 13[43] | 8[44] | 5[42] | 1 |
| Age (yrs) | 48.0 ± 12.6 | 47.5 ± 11.5 | 48.8 ± 14.5) | 0.8 |
| Adenoma size (Macroadenoma), n (%) | 20[67] | 12[67] | 8[67] | 1 |
| Knosp grading (Knosp grade 1), n (%) | 16[53] | 10[56] | 6[50] | 1 |
| Affected pituitary axes, n (%) | ||||
| Gonadotropin deficiency, n (%) | 23 (77) | 14 (78) | 9 (75) | 1 |
| Secondary hypothyroidism, n (%) | 5[17] | 2[11] | 3[25] | 0.64 |
| Secondary adrenal insufficiency, n (%) | 3[10] | 1[6] | 2[17] | 0.55 |
| Prolactin (μg/L) | 856 (154–6473) | 856 (154–8117) | 928 (113–6473) | 0.48 |
| BMI (kg/m2) | 28.6 ± 6 | 29.5 ± 6.4 | 27.7 ± 5.5 | 0.68 |
| Triglycerides (mmol/L) | 1.6 ± 1.1 | 1.7 ± 1.3 | 1.3 ± 0.7 | 0.69 |
| Total cholesterol (mmol/L) | 5.5 ± 1.2 | 5.3 ± 1.2 | 5.5 ± 1.1 | 0.99 |
| HDL cholesterol (mmol/L) | 1.2 ± 0.4 | 1.1 ± 0.5 | 1.3 ± 0.4 | 0.58 |
| LDL cholesterol (mmol/L) | 3.6 ± 0.8 | 3.5 ± 0.9 | 3.6 ± 0.8 | 0.82 |
| Fasting glucose (mmol/L) | 5.4 ± 1.1 | 5.4 ± 0.3 | 5.4 ± 1.7 | 0.17 |
| Follow-up (months) | 51.9 ± 38.1 | 59.2 ± 38.9 | 41.0 ± 35.7 | 0.21 |
IQR, interquartile range; n, numbers; BMI, body mass index; SD, standard deviation.
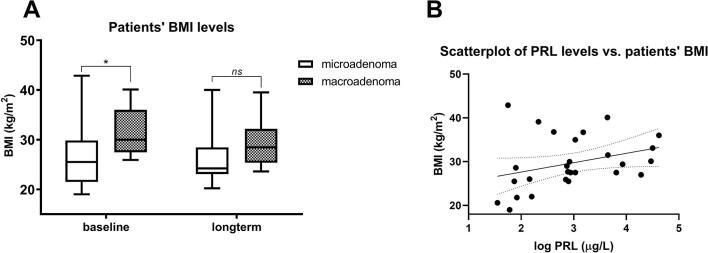
Patients with a macroprolactinoma vs. those with a microprolactinoma showed significant differences in their BMI (31.5 ± 4.4 vs. 26.8 ± 7.5, p = 0.04, Fig. 2A) and HDL-C levels (1.1 ± 0.2 vs. 1.4 ± 0.6; p = 0.02), while no significant differences were noted with regard to LDL-C (p = 0.35), TC (p = 0.98), TG (p = 0.09), or FG levels (p = 0.24). In addition, a significant positive correlation between all patients’ PRL and BMI values (r = 0.4, p = 0.03, Fig. 2B) was observed. Likewise, there was a significant correlation in all patients between the prevalence of hypogonadism and HDL-C levels (r = −0.48, p = 0.007), as well as TG levels (r = 0.52, p = 0.004).
Fig. 2.
Patients’ BMI as a function of their adenoma size and their relation with PRL levels (A) Patients with a macroprolactinoma vs. those with a microprolactinoma showed significant differences in their BMI at diagnosis, (31.5 ± 4.4 vs. 26.8 ± 7.5, p = 0.04), but not over the long-term (29.4 ± 5.1 vs. 26.5 ± 6.0, p = 0.18). (B) Scatterplot showing a significant positive correlation between all patients’ PRL and BMI values (r = 0.4, p = 0.03).
Characteristics at long-term follow-up
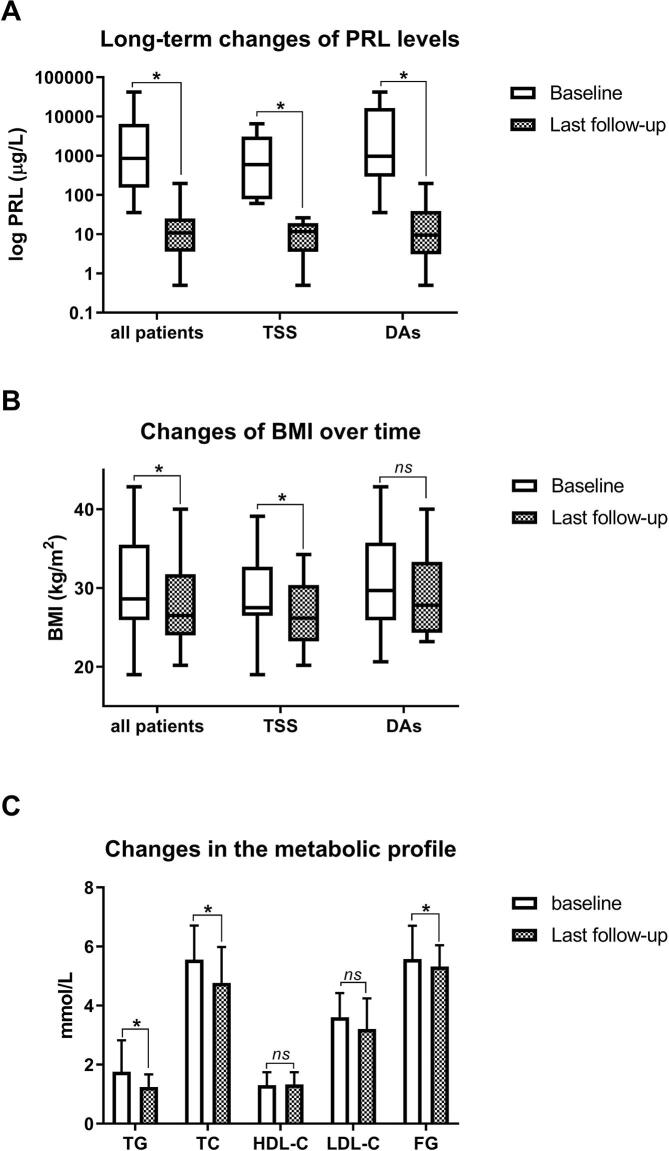
After a mean follow-up of 51.9 months [24], [25], [26], [27], [28], [29], [30], [31], [32], [33], [34], [35], [36], [37], [38], [39], [40], [41], [42], [43], [44], [45], [46], [47], [48], [49], [50], [51], [52], [53], [54], [55], [56], [57], [58], [59], [60], [61], [62], [63], [64], [65], [66], [67], long-term control of hyperprolactinemia (92% vs. 72%) and hypogandism (78% vs. 83%) was attained in the majority of patients, independent of the primary treatment (TSS vs. DAs, respectively). The prevalence of secondary hypothyroidism and secondary adrenal insufficiency was not significantly altered compared to baseline, independent of the primary treatment strategy (i.e. TSS vs. DAs, see Supplemental Tables 1 and 2). The use of gonadotropin replacement therapy was noted in 10 men (59%) and 2 women (15%) (p = 0.03). With regard to patients’ lipid profiles, no significant differences were detected between patients with replacement therapy and those without for HCL-C (p = 0.35), LCL-C (p = 0.73), TG (p = 0.63), and TC (p = 0.50). This was independent of the gender.
Long-term changes in patients’ BMI and metabolic parameters are detailed in Table 2. In particular, PRL levels decreased significantly over the long-term (p = 0.03), independent of the primary treatment; i.e., surgery (p = 0.05) or DA therapy (p = 0.01; Fig. 3A). Patients’ BMI decreased significantly (p = 0.05), a phenomenon more distinct in the surgical (p = 0.02) than in the medical cohort (p = 0.36; Fig. 3B). In the long-term, patients with a macroprolactinoma vs. those with a microprolactinoma showed no significant differences in their BMI (29.4 ± 5.1 vs. 26.5 ± 6.0, p = 0.18; Fig. 2B) While concentrations in FG significantly decreased in all patients (p = 0.01), significant improvements in the lipid profile were noted for levels of TG (p = 0.02) and TC (p = 0.01), but not for HDL-C (p = 0.18), or LDL-C (p = 0.07; Fig. 3C).
Table 2.
Long-term changes in metabolic parameters following treatment of hyperprolactinaemia.
| All patients | Baseline | Long-term FU | p value |
|---|---|---|---|
| Prolactin (μg/L) | 856 (154–6473) | 11[4–25] | 0.01 |
| Affected pituitary axes, n (%) | |||
| Gonadotropin deficiency, n (%) | 23 (77) | 6[20] | <0.001 |
| Secondary hypothyroidism, n (%) | 5[17] | 2[7] | 0.42 |
| Secondary adrenal insufficiency, n (%) | 3[10] | 4[13] | 1 |
| BMI (kg/m2) | 28.6 ± 6 (19–42.9) | 26.5 ± 6 (20.2–40) | 0.05 |
| Triglycerides (mmol/L) | 1.6 ± 1.1 | 1.1 ± 0.4 | 0.02 |
| Total cholesterol (mmol/L) | 5.5 ± 1.2 | 4.7 ± 1.2 | 0.01 |
| HDL-cholesterol (mmol/L) | 1.2 ± 0.4 | 1.3 ± 0.4 | 0.18 |
| LDL-cholesterol (mmol/L) | 3.6 ± 0.8 | 3.4 ± 1.0 | 0.07 |
| Fasting glucose (mmol/L) | 5.4 ± 1.1 | 5.2 ± 0.7 | 0.01 |
BMI, body mass index; SD, standard deviation.
Fig. 3.
Changes in PRL levels, metabolic parameters and patients’ BMI over the long-term (A) PRL levels significantly decreased at long-term follow-up (p = 0.03), independent of the primary treatment strategy; i.e. surgery (p = 0.05) and DA therapy (p = 0.01). (B) Over the long-term, a significant reduction in levels of TG (p = 0.02), TC (p = 0.01), and FG (p = 0.003), but not HDL-C (p = 0.16) or LDL-C (p = 0.07), was noted. (C) Patients’ BMI significantly decreased over the long-term (p = 0.05), and was significant in the surgical (p = 0.02) but not the medical (p = 0.34) cohort.
Risk factors for obesity (i.e., BMI ≥ 31 kg/m2) are detailed in Table 3. Increased BMI at baseline (OR 1.6, 95% CI 1.1–2.3; p = 0.01), but not the primary treatment strategy, was an independent predictor of long-term obesity. Baseline PRL levels were significantly higher in patients with persisting obesity at last follow-up: 1287 µg/L (IQR 264–38,862 µg/L) vs. 784 µg/L (IQR 99–4414 µg/L; p = 0.02).
Table 3.
Risk factors for obesity (i.e., BMI > 30 kg/m2) at last follow-up.
| Risk factors for obesity at last FU | Univariable analyses OR (95% CI) | p value | Multivariable analyses OR (95% CI) | p value |
|---|---|---|---|---|
| Age | 1.0 (0.9–1.1) | 0.94 | ||
| Sex (women) | 1.2 (0.3–6.3) | 0.81 | ||
| PRL levels at diagnosis | 1.8 (0.7–4.7) | 0.22 | 2.3 (0.4–14.8) | 0.38 |
| BMI at diagnosis | 1.6 (1.2–2.2) | 0.004 | 1.6 (1.1–2.3) | 0.01 |
| Hypogonadism at diagnosis | 3.0 (0.3–30.0) | 0.35 | ||
| Primary therapy (DAs) | 2.0 (0.3–12.5) | 0.46 | ||
| Follow-up time | 1.0 (1.0–1.0) | 0.33 |
PRL, prolactin; CI, confidence interval; DAs, dopamine agonists.
Metabolic impact of surgery or DAs as first-line treatment
The impact of TSS or DAs per se on patients’ metabolic profile is summarized in the supplementary tables (Tables S1 and S2). Surgery per se significantly decreased levels of TG (p = 0.02) and FG (p = 0.04), whereas DAs significantly decreased levels of TC (p = 0.05) and FG (p = 0.05). Net changes in patients’ BMI or PRL levels over the long-term were not significantly different between the two cohorts (i.e., TSS vs. DAs, Table S3).
With regard to the DA being used, cabergoline was more frequently prescribed than bromocriptine. Bromocriptine doses ranged from 2.5 to 10 mg/day, whereas cabergoline doses ranged from 0.5 to 2 mg/week.
In the medical cohort, DAs were required in 16 patients (53%) at last follow-up, with bromocriptine being noted in 5 (16%) and cabergoline in 11 (37%). Thereby, mean (±SD) doses at last follow-up were 3.9 ± 3.4 mg for bromocriptine (daily), and 1.3 ± 1.0 mg for cabergoline (weekly). Mean cumulative doses per patient at last follow-up were 971 mg (range 312–2340 mg) for bromocriptine, and 35 mg (range 3–80 mg) for cabergoline.
In the surgical cohort, cabergoline was required in 5 patients (17%) at last follow-up. While none of these patients were taking bromocriptine, mean (±SD) doses for cabergoline (weekly) were 1.1 ± 0.3 mg. Mean cumulative doses per patient were 28 mg (range 3–64 mg) for cabergoline.
At last follow-up, 6 patients (20%) were treated with statins: 3 (17%) in the medical cohort and 3 (25%) in the surgical cohort (p = 0.66). Levels of LDL-C (2.5 ± 1.1 vs. 3.4 ± 1.0; p = 0.07), TC (4.0 ± 1.2 vs. 5.0 ± 1.1; p = 0.09), and TG (1.3 ± 0.5 vs. 1.2 ± 0.4; p = 0.69) were lower in the statin cohort, though not significantly.
Morbidity and mortality
No mortality has been noted. In the surgical cohort, postoperative complications consisted of transient diabetes insipidus in one patient. In the medical group, prolonged nausea and vertigo were noted in one patient.
Discussion
The present analysis significantly adds to the existing literature on the comparative effect of first-line surgery and medical therapy on prolactinoma patients’ BMI and metabolic profile. Our long-term results indicate that i) hyperprolactinemia and associated hypogonadism can be controlled in the majority of patients regardless of the primary treatment, ii) normalization of PRL improves patients’ BMI and FG levels, iii) marginal changes in patients’ metabolic profiles are noted, and iv) high baseline BMI but not the first therapy is a risk for persistent long-term obesity.
Impact on patients’ BMI
Prolactinoma patients display higher BMI and increased prevalence of obesity compared to the general population [17], [39]. While we noted a significant correlation between BMI and PRL levels, corroborating recent results [39], the role of PRL in the pathophysiology of obesity remains unclear. Decreases in patients’ BMI levels are not an uncommon phenomenon following the control of hyperprolactinaemia [4], [40]. DAs have been found to reduce body weight, particularly in patients with high baseline PRL levels [19], [41], [42].Interestingly, we noted that normalization of PRL improves patients’ BMI, with a significant reduction in the TSS but not the DA cohort. Although PRL levels in both cohorts were not significantly different over the long-term, it is presumable that not DAs per se, but rather the control of hyperprolactinemia and hypogonadism might account for this effect. Namely, the association between BMI and PRL levels has recently been strengthened. Increased BMI in men with prolactinomas compared to the general population has been noted [39]. Similarly, our findings indicate a significantly higher BMI in patients with a macroprolactinoma compared to those with a microprolactinoma, corroborating previous results [43]. It is conceivable that longer exposure to increased PRL levels, as occurs in men, afford for the true effect. Namely, while amenorrhea in women is easily detected and investigated, men often do not report the non-specific symptoms of hypogonadism, such as loss of libido. Consequently, men suffer from hyperprolactinemia and hypogonadism over a much longer period, a reason that macroprolactinomas are more commonly seen in them [44], [45]. Whether hypogonadism and its associated lack of energy and physical exercise contributes to the increased BMI in females and males remains speculative. Increased BMI could also be related to an increase in adenoma-associated hypothalamic pressure rather than the effect of hyperprolactinemia itself, but this mechanism might only be valid in solid and large adenomas [46], [47]. Finally, regardless of how hyperprolactinemia influences patients’ BMI [43], control of hyperprolactinemia is key in overweight prolactinoma patients.
Impact on patients’ FG profile
It has been hypothesized that DAs directly decrease the level of FG in obese patients [48], [49]. Likewise, Pala et al. described a significant reduction in FG levels following treatment with Das [50], in keeping with our data showing significantly improved FG levels after DA therapy, but also after TSS. In contrast, Schwetz et al. did not confirm the previously described changes in the normalization of the glucose metabolism by Das [18]. This is of interest, given the postulated association between high PRL levels and an adverse glucose profile [51], [52]. Yet, FG may independently improve based on a decrease in PRL levels and/or changes in BMI [53]. This corroborates the hypothesis that DAs per se alter the glucose metabolism by fortifying the suppression of endogenous glucose product, or enhancing the splanchnic glucose uptake, or through direct hypothalamic alterations [54]. These mechanisms were demonstrated by the use of quick-release bromocriptin in obese type 2 diabetic patients [54], [55]. The same effect is associated with cabergoline therapy [17]. This would not explain why changes were also observed in our study cohort following TSS, but it affirms the importance of controlling hyperprolactinemia over the long-term.
Impact on patients’ lipid profiles
The proposed direct effect of DA therapy on the metabolic profile is debatable, and might rather be caused by the control of hyperprolactinemia and associated hypogonadism over the long-term. Namely, we observed that restoration of normoprolactinaemia and hypogonadism using either treatment led to a significant decrease in TG and TC levels, with no significant changes in LDL-C or HDL-C. Cross-sectional studies have shown an association between high PRL levels and adverse lipid profile [17], [51], [53]. Our results are in line with most published studies to date which found amelioration of the lipid profile following DA therapy, although changes have not been observed in all studied lipid parameters. In addition, it has been suggested that TC is one of the first metabolic parameters affected by a decrease in PRL levels following DA therapy [18]. Namely, levels of TC, but not TG, HDL-C or LDL-C, improved at 12 months, with the latter significantly decreasing at 24 months [56]. In particular, changes in TG and LDL-C levels have been attributed to the antilipogenic action in the liver tissue and antilipolytic action in adipose tissue afforded by Das [55]. In line with this finding, prolactinomas were associated with higher BMI and LDL-C [41]. In addition, while gonadotropin replacement didn’t alter patients’ lipid profile, it has been shown that low testosterone levels in men might be associated with decreased HDL-C and high LDL-C and TG levels [57], but this association can be confounded by obesity [58]. While changes in the lipid profile following gonadotropin replacement can be marginal or nonexistent in men [59], the administration of estrogen in women more distinctly decreases LCL-C levels, while increasing HDL-C and TG levels [57], [60], although we couldn’t confirm this result.
The reason we did not observe significant improvement in the levels of LDL-C or HDL-C following treatment with DAs or gonadotropin replacement therapy is unclear, as other studies noted a decrease in LDL-C levels with therapy [18]. Reasons may include differences in study characteristics, number of patients included, modalities or doses of gonadal replacement, as well as the risk of bias in uncontrolled studies. Namely, differences in the severity and duration of the diseases and intrinsic and extrinsic factors may influence lipid parameters.
Beside age-specific changes in BMI and metabolic parameters [61], [62], glucocorticoids, thyroxine and statin therapy likewise may affect the lipid profile [63], [64]. Namely, while levels of HDL-C are generally increased with glucocorticoids, changes in plasma TG and LDL-C vary considerably [65], [66]. Secondary hypothyroidism is known to increase TC, LDL-C, and TG [67]. In addition, statin therapy lowers LDL-C and TC [63]. In the present cohort, the prevalence of pituitary axes deficits—apart from hypogonadism—was not high at baseline or over the long-term. In addition, 6 patients were undergoing statin therapy at last follow-up, and their levels of LDL-C and TC were lower compared to the levels of the other subjects, although not significantly. Thus, it is conceivable that their effect on the metabolic profile may be less strong compared to the long-term control of hyperprolactinemia and associated hypogonadism. However, the small sample size of both subjects on statin therapy and those with pituitary axes deficits other than secondary hypogonadism may fail to reveal a true association.
Study strengths and limitations
The main limitations of our study are its retrospective design, the lack of randomization, and the single-center design. Despite the relatively small number of patients included, which may not reveal a true association between other factors influencing the metabolic profile (i.e., hormonal deficits, statin therapy), a strong point of our study is the collection of data over 20 years, as well as its homogeneity in terms of indications, treatment and follow-up. However, given the long-term follow-up, and there have been advances in the medical treatment and patients’ surveillance over time, which may confound outcomes. In addition, GH may vary the metabolic parameters, but was not systematically measured in this study.
Conclusion
Over the long-term, patients’ BMI and FG improve. However, changes in the metabolic profile are marginal and independent of the primary treatment. It is possible that it is not DAs per se, but rather the management of hyperprolactinemia and hypogonadism that plays a role in patients’ metabolic profile alterations. Considering the clinical significance of obesity and impaired metabolic profile in prolactinoma patients, our results underline the importance of long-term control of hyperprolactinemia.
Funding
No funding was received for this publication.
Declarations
This work is original and has not been published elsewhere nor is it currently under consideration for publication elsewhere.
Ethical standards and patient consent
The Human Research Ethics Committee of Bern (Kantonale Ethikkommission KEK Bern, Bern, Switzerland) approved the project (KEK n° 10-10-2006 and 8-11-2006). The study was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
Acknowledgments
The assistance of Ms. Jeannie Wurz in editing the manuscript is greatly appreciated.
Contributors
LA contributed to study conception and design, statistical analysis and interpretation, drafting of the manuscript, critical revision and final approval of the article. EC contributed to study conception and design, data interpretation, critical revision and final approval of the article. JF contributed to the acquisition of data, and final approval of the article. RHA, MML, JG, GAS JB, and LM contributed to critical revision and final approval of the article.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.jcte.2021.100258.
Appendix A. Supplementary data
The following are the Supplementary data to this article:
References
- 1.Flegal K.M., Kit B.K., Orpana H., Graubard B.I. Association of all-cause mortality with overweight and obesity using standard body mass index categories: a systematic review and meta-analysis. JAMA. 2013;309:71–82. doi: 10.1001/jama.2012.113905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wilding J.P.H. Endocrine testing in obesity. Eur J Endocrinol. 2020;182:C13–C15. doi: 10.1530/EJE-20-0099. [DOI] [PubMed] [Google Scholar]
- 3.Gorvin C.M. The prolactin receptor: Diverse and emerging roles in pathophysiology. J Clin Transl Endocrinol. 2015;2:85–91. doi: 10.1016/j.jcte.2015.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shibli-Rahhal A., Schlechte J. The effects of hyperprolactinemia on bone and fat. Pituitary. 2009;12:96–104. doi: 10.1007/s11102-008-0097-3. [DOI] [PubMed] [Google Scholar]
- 5.Raverot G., Jouanneau E., Trouillas J. Management of endocrine disease: clinicopathological classification and molecular markers of pituitary tumours for personalized therapeutic strategies. Eur J Endocrinol. 2014;170:R121–R132. doi: 10.1530/EJE-13-1031. [DOI] [PubMed] [Google Scholar]
- 6.Colao A., Di Sarno A., Guerra E. Predictors of remission of hyperprolactinaemia after long-term withdrawal of cabergoline therapy. Clin Endocrinol (Oxf) 2007;67:426–433. doi: 10.1111/j.1365-2265.2007.02905.x. [DOI] [PubMed] [Google Scholar]
- 7.Kars M., Souverein P.C., Herings R.M. Estimated age- and sex-specific incidence and prevalence of dopamine agonist-treated hyperprolactinemia. J Clin Endocrinol Metab. 2009;94:2729–2734. doi: 10.1210/jc.2009-0177. [DOI] [PubMed] [Google Scholar]
- 8.Levy A. Pituitary disease: presentation, diagnosis, and management. J Neurol Neurosurg Psychiatry. 2004;75(Suppl 3) doi: 10.1136/jnnp.2004.045740. iii47-52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Andereggen L., Frey J., Andres R.H. Long-term follow-up of primary medical versus surgical treatment of prolactinomas in men: effects on hyperprolactinemia, hypogonadism, and bone health. World Neurosurg. 2017;97:595–602. doi: 10.1016/j.wneu.2016.10.059. [DOI] [PubMed] [Google Scholar]
- 10.Andereggen L., Frey J., Andres R.H. 10-year follow-up study comparing primary medical vs. surgical therapy in women with prolactinomas. Endocrine. 2017;55:223–230. doi: 10.1007/s12020-016-1115-2. [DOI] [PubMed] [Google Scholar]
- 11.Donegan D., Atkinson J.L., Jentoft M. Surgical outcomes of prolactinomas in recent era: results of a heterogenous group. Endocr Pract. 2017;23:37–45. doi: 10.4158/EP161446.OR. [DOI] [PubMed] [Google Scholar]
- 12.Tampourlou M., Trifanescu R., Paluzzi A., Ahmed S.K., Karavitaki N. THERAPY OF ENDOCRINE DISEASE: surgery in microprolactinomas: effectiveness and risks based on contemporary literature. Eur J Endocrinol/Eur Federat Endocrine Soc. 2016;175:R89–R96. doi: 10.1530/EJE-16-0087. [DOI] [PubMed] [Google Scholar]
- 13.Demartini B., Ricciardi L., Ward A., Edwards M.J. Dopamine agonist withdrawal syndrome (DAWS) in a patient with a microprolactinoma. J Neurol Neurosurg Psychiatry. 2014;85:471. doi: 10.1136/jnnp-2013-306043. [DOI] [PubMed] [Google Scholar]
- 14.Moore T.J., Glenmullen J., Mattison D.R. Reports of pathological gambling, hypersexuality, and compulsive shopping associated with dopamine receptor agonist drugs. JAMA Intern Med. 2014;174:1930–1933. doi: 10.1001/jamainternmed.2014.5262. [DOI] [PubMed] [Google Scholar]
- 15.Honegger J., Nasi-Kordhishti I., Aboutaha N., Giese S. Surgery for prolactinomas: a better choice? Pituitary. 2020;23:45–51. doi: 10.1007/s11102-019-01016-z. [DOI] [PubMed] [Google Scholar]
- 16.Dekkers O.M., Lagro J., Burman P., Jorgensen J.O., Romijn J.A., Pereira A.M. Recurrence of hyperprolactinemia after withdrawal of dopamine agonists: systematic review and meta-analysis. J Clin Endocrinol Metab. 2010;95:43–51. doi: 10.1210/jc.2009-1238. [DOI] [PubMed] [Google Scholar]
- 17.dos Santos Silva C.M., Barbosa F.R., Lima G.A. BMI and metabolic profile in patients with prolactinoma before and after treatment with dopamine agonists. Obesity (Silver Spring) 2011;19:800–805. doi: 10.1038/oby.2010.150. [DOI] [PubMed] [Google Scholar]
- 18.Schwetz V., Librizzi R., Trummer C. Treatment of hyperprolactinaemia reduces total cholesterol and LDL in patients with prolactinomas. Metab Brain Dis. 2017;32:155–161. doi: 10.1007/s11011-016-9882-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Byberg S., Futtrup J., Andreassen M., Krogh J. Metabolic effects of dopamine agonists in patients with prolactinomas: a systematic review and meta-analysis. Endocr Connect. 2019;8:1395–1404. doi: 10.1530/EC-19-0286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Freda P.U., Wardlaw S.L. Clinical review 110: diagnosis and treatment of pituitary tumors. J Clin Endocrinol Metab. 1999;84:3859–3866. doi: 10.1210/jcem.84.11.6202. [DOI] [PubMed] [Google Scholar]
- 21.Andereggen L., Frey J., Andres R.H. First-line surgery in prolactinomas: lessons from a long-term follow-up study in a tertiary referral center. J Endocrinol Invest. 2021 doi: 10.1007/s40618-021-01569-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Karavitaki N., Thanabalasingham G., Shore H.C. Do the limits of serum prolactin in disconnection hyperprolactinaemia need re-definition? a study of 226 patients with histologically verified non-functioning pituitary macroadenoma. Clin Endocrinol. 2006;65:524–529. doi: 10.1111/j.1365-2265.2006.02627.x. [DOI] [PubMed] [Google Scholar]
- 23.Andereggen L., Frey J., Christ E. Long-term IGF-1 monitoring in prolactinoma patients treated with cabergoline might not be indicated. Endocrine. 2020 doi: 10.1007/s12020-020-02557-1. [DOI] [PubMed] [Google Scholar]
- 24.Melmed S., Casanueva F.F., Hoffman A.R. Diagnosis and treatment of hyperprolactinemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96:273–288. doi: 10.1210/jc.2010-1692. [DOI] [PubMed] [Google Scholar]
- 25.Saeger W., Ludecke D.K., Buchfelder M., Fahlbusch R., Quabbe H.J., Petersenn S. Pathohistological classification of pituitary tumors: 10 years of experience with the German Pituitary Tumor Registry. Eur J Endocrinol. 2007;156:203–216. doi: 10.1530/eje.1.02326. [DOI] [PubMed] [Google Scholar]
- 26.Mei Z., Grummer-Strawn L.M., Pietrobelli A., Goulding A., Goran M.I., Dietz W.H. Validity of body mass index compared with other body-composition screening indexes for the assessment of body fatness in children and adolescents. Am J Clin Nutr. 2002;75:978–985. doi: 10.1093/ajcn/75.6.978. [DOI] [PubMed] [Google Scholar]
- 27.Andereggen L., Mono M.L., Kellner-Weldon F., Christ E. Cluster headache and macroprolactinoma: Case report of a rare, but potential important causality. J Clin Neurosci. 2017;40:62–64. doi: 10.1016/j.jocn.2017.01.028. [DOI] [PubMed] [Google Scholar]
- 28.Knosp E, Steiner E, Kitz K, Matula C. Pituitary adenomas with invasion of the cavernous sinus space: a magnetic resonance imaging classification compared with surgical findings. Neurosurgery. 1993;33:610–7; discussion 617–618. [DOI] [PubMed]
- 29.Micko A.S., Wohrer A., Wolfsberger S., Knosp E. Invasion of the cavernous sinus space in pituitary adenomas: endoscopic verification and its correlation with an MRI-based classification. J Neurosurg. 2015;122:803–811. doi: 10.3171/2014.12.JNS141083. [DOI] [PubMed] [Google Scholar]
- 30.Andereggen L., Commentary C.E. Prolactinomas: prognostic factors of early remission after transsphenoidal surgery. Front Endocrinol (Lausanne) 2021 doi: 10.3389/fendo.2021.695498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Andereggen L., Schroth G., Gralla J. Selective inferior petrosal sinus sampling without venous outflow diversion in the detection of a pituitary adenoma in Cushing's syndrome. Neuroradiology. 2012;54:495–503. doi: 10.1007/s00234-011-0915-6. [DOI] [PubMed] [Google Scholar]
- 32.Andereggen L., Gralla J., Schroth G. Influence of inferior petrosal sinus drainage symmetry on detection of adenomas in Cushing's syndrome. J Neuroradiol. 2021;48:10–15. doi: 10.1016/j.neurad.2019.05.004. [DOI] [PubMed] [Google Scholar]
- 33.Andereggen L., Mariani L., Beck J. Lateral one-third gland resection in Cushing patients with failed adenoma identification leads to low remission rates: long-term observations from a small, single-center cohort. Acta Neurochir (Wien) 2021 doi: 10.1007/s00701-021-04830-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Andereggen L., Beck J., Gralla J., Luedi M.M., Christ E. Letter to the editor from Lukas Andereggen: “pitfalls in performing and interpreting inferior petrosal sinus sampling: personal experience and literature review”. J Clin Endocrinol Metab. 2021 doi: 10.1210/clinem/dgab329. [DOI] [PubMed] [Google Scholar]
- 35.Wass J.A. When to discontinue treatment of prolactinoma? Nat Clin Pract Endocrinol Metab. 2006;2:298–299. doi: 10.1038/ncpendmet0162. [DOI] [PubMed] [Google Scholar]
- 36.Colao A., Di Sarno A., Cappabianca P., Di Somma C., Pivonello R., Lombardi G. Withdrawal of long-term cabergoline therapy for tumoral and nontumoral hyperprolactinemia. N Engl J Med. 2003;349:2023–2033. doi: 10.1056/NEJMoa022657. [DOI] [PubMed] [Google Scholar]
- 37.Qu X., Wang M., Wang G. Surgical outcomes and prognostic factors of transsphenoidal surgery for prolactinoma in men: a single-center experience with 87 consecutive cases. Eur J Endocrinol/Eur Federat Endocrine Soc. 2011;164:499–504. doi: 10.1530/EJE-10-0961. [DOI] [PubMed] [Google Scholar]
- 38.Raverot G., Wierinckx A., Dantony E. Prognostic factors in prolactin pituitary tumors: clinical, histological, and molecular data from a series of 94 patients with a long postoperative follow-up. J Clin Endocrinol Metab. 2010;95:1708–1716. doi: 10.1210/jc.2009-1191. [DOI] [PubMed] [Google Scholar]
- 39.Al Sabie F., Tariq Z., Erickson D., Donegan D. Association between prolactinoma and body mass index. Endocr Pract. 2020 doi: 10.1016/j.eprac.2020.09.001. [DOI] [PubMed] [Google Scholar]
- 40.Doknic M., Pekic S., Zarkovic M. Dopaminergic tone and obesity: an insight from prolactinomas treated with bromocriptine. Eur J Endocrinol. 2002;147:77–84. doi: 10.1530/eje.0.1470077. [DOI] [PubMed] [Google Scholar]
- 41.Peric B., Kruljac I., Sundalic S. Obesity and hypercholesterolemia in patients with prolactinomas: Could DHEA-S and growth hormone be the missing link? Endocr Res. 2016;41:200–206. doi: 10.3109/07435800.2015.1135444. [DOI] [PubMed] [Google Scholar]
- 42.Korner J., Lo J., Freda P.U., Wardlaw S.L. Treatment with cabergoline is associated with weight loss in patients with hyperprolactinemia. Obes Res. 2003;11:311–312. doi: 10.1038/oby.2003.46. [DOI] [PubMed] [Google Scholar]
- 43.Schmid C., Goede D.L., Hauser R.S., Brandle M. Increased prevalence of high Body Mass Index in patients presenting with pituitary tumours: severe obesity in patients with macroprolactinoma. Swiss Med Wkly. 2006;136:254–258. doi: 10.4414/smw.2006.10955. [DOI] [PubMed] [Google Scholar]
- 44.Andereggen L., Frey J., Andres R.H. Persistent bone impairment despite long- term control of hyperprolactinemia and hypogonadism in men and women with prolactinomas. Sci Rep. 2021;11:5122. doi: 10.1038/s41598-021-84606-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tirosh A., Benbassat C., Shimon I. Short-term decline in prolactin concentrations can predict future prolactin normalization, tumor shrinkage, and time to remission in men with macroprolactinomas. Endocr Pract. 2015;21:1240–1247. doi: 10.4158/EP15804.OR. [DOI] [PubMed] [Google Scholar]
- 46.Andereggen L., Hess B., Andres R. A ten-year follow-up study of treatment outcome of craniopharyngiomas. Swiss Med Wkly. 2018;148 doi: 10.4414/smw.2018.14521. [DOI] [PubMed] [Google Scholar]
- 47.Hamidi O., Van Gompel J., Gruber L. Management and outcomes of giant prolactinoma: a series of 71 patients. Endocr Pract. 2019;25:340–352. doi: 10.4158/EP-2018-0392. [DOI] [PubMed] [Google Scholar]
- 48.Liang W., Gao L., Li N. Efficacy and safety of bromocriptine-QR in type 2 diabetes: a systematic review and meta-analysis. Horm Metab Res. 2015;47:805–812. doi: 10.1055/s-0035-1559684. [DOI] [PubMed] [Google Scholar]
- 49.Manning P.J., Grattan D., Merriman T., Manning T., Williams S., Sutherland W. Pharmaceutical interventions for weight-loss maintenance: no effect from cabergoline. Int J Obes (Lond) 2018;42:1871–1879. doi: 10.1038/s41366-018-0165-3. [DOI] [PubMed] [Google Scholar]
- 50.Pala N.A., Laway B.A., Misgar R.A., Dar R.A. Metabolic abnormalities in patients with prolactinoma: response to treatment with cabergoline. Diabetol Metab Syndr. 2015;7:99. doi: 10.1186/s13098-015-0094-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ciresi A., Amato M.C., Guarnotta V., Lo Castro F., Giordano C. Higher doses of cabergoline further improve metabolic parameters in patients with prolactinoma regardless of the degree of reduction in prolactin levels. Clin Endocrinol (Oxf) 2013;79:845–852. doi: 10.1111/cen.12204. [DOI] [PubMed] [Google Scholar]
- 52.Auriemma R.S., Granieri L., Galdiero M. Effect of cabergoline on metabolism in prolactinomas. Neuroendocrinology. 2013;98:299–310. doi: 10.1159/000357810. [DOI] [PubMed] [Google Scholar]
- 53.Inancli S.S., Usluogullari A., Ustu Y. Effect of cabergoline on insulin sensitivity, inflammation, and carotid intima media thickness in patients with prolactinoma. Endocrine. 2013;44:193–199. doi: 10.1007/s12020-012-9857-y. [DOI] [PubMed] [Google Scholar]
- 54.Pijl H., Ohashi S., Matsuda M. Bromocriptine: a novel approach to the treatment of type 2 diabetes. Diabetes Care. 2000;23:1154–1161. doi: 10.2337/diacare.23.8.1154. [DOI] [PubMed] [Google Scholar]
- 55.Scranton R., Cincotta A. Bromocriptine–unique formulation of a dopamine agonist for the treatment of type 2 diabetes. Expert Opin Pharmacother. 2010;11:269–279. doi: 10.1517/14656560903501544. [DOI] [PubMed] [Google Scholar]
- 56.Auriemma R.S., Galdiero M., Vitale P. Effect of chronic cabergoline treatment and testosterone replacement on metabolism in male patients with prolactinomas. Neuroendocrinology. 2015;101:66–81. doi: 10.1159/000371851. [DOI] [PubMed] [Google Scholar]
- 57.Feingold KR, Brinton EA, Grunfeld C. The Effect of Endocrine Disorders on Lipids and Lipoproteins. In: Feingold KR, Anawalt B, Boyce A, et al., eds. Endotext. South Dartmouth (MA)2000.
- 58.Pivonello R., Menafra D., Riccio E. Metabolic disorders and male hypogonadotropic hypogonadism. Front Endocrinol (Lausanne) 2019;10:345. doi: 10.3389/fendo.2019.00345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Bayram F., Elbuken G., Korkmaz C., Aydogdu A., Karaca Z., Cakir I. The effects of gonadotropin replacement therapy on metabolic parameters and body composition in men with idiopathic hypogonadotropic hypogonadism. Horm Metab Res. 2016;48:112–117. doi: 10.1055/s-0035-1564252. [DOI] [PubMed] [Google Scholar]
- 60.Ostberg J.E., Storry C., Donald A.E., Attar M.J., Halcox J.P., Conway G.S. A dose-response study of hormone replacement in young hypogonadal women: effects on intima media thickness and metabolism. Clin Endocrinol (Oxf) 2007;66:557–564. doi: 10.1111/j.1365-2265.2007.02772.x. [DOI] [PubMed] [Google Scholar]
- 61.Hayes A., Gearon E., Backholer K., Bauman A., Peeters A. Age-specific changes in BMI and BMI distribution among Australian adults using cross-sectional surveys from 1980 to 2008. Int J Obes (Lond) 2015;39:1209–1216. doi: 10.1038/ijo.2015.50. [DOI] [PubMed] [Google Scholar]
- 62.Feng L., Nian S., Tong Z. Age-related trends in lipid levels: a large-scale cross- sectional study of the general Chinese population. BMJ Open. 2020;10 doi: 10.1136/bmjopen-2019-034226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kofink D., Eppinga R.N., van Gilst W.H. Statin Effects on Metabolic Profiles: Data From the PREVEND IT (Prevention of Renal and Vascular End-stage Disease Intervention Trial) Circ Cardiovasc Genet. 2017;10 doi: 10.1161/CIRCGENETICS.117.001759. [DOI] [PubMed] [Google Scholar]
- 64.Benvenga S., Klose M., Vita R., Feldt-Rasmussen U. Less known aspects of central hypothyroidism: Part 1 - Acquired etiologies. J Clin Transl Endocrinol. 2018;14:25–33. doi: 10.1016/j.jcte.2018.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Taskinen M.R., Kuusi T., Yki-Jarvinen H., Nikkila E.A. Short-term effects of prednisone on serum lipids and high density lipoprotein subfractions in normolipidemic healthy men. J Clin Endocrinol Metab. 1988;67:291–299. doi: 10.1210/jcem-67-2-291. [DOI] [PubMed] [Google Scholar]
- 66.van Raalte D.H., Brands M., van der Zijl N.J. Low-dose glucocorticoid treatment affects multiple aspects of intermediary metabolism in healthy humans: a randomised controlled trial. Diabetologia. 2011;54:2103–2112. doi: 10.1007/s00125-011-2174-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Duntas L.H. Thyroid disease and lipids. Thyroid. 2002;12:287–293. doi: 10.1089/10507250252949405. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.