Abstract
Esophageal cancer (EC) is relatively common; at the time of diagnosis, 50% of cases present with distant metastases, and most patients are men. This study aimed to examine and compare the clinicopathological characteristics and metastatic patterns of male EC (MEC) and female EC (FEC). In addition, risk factors associated with MEC prognosis were evaluated.
The present study population was extracted from the Surveillance Epidemiology and End Results database. MEC characteristics and factors associated with prognosis were evaluated using descriptive analysis, the Kaplan–Meier method, and the Cox regression model.
A total of 12,558 MEC cases were included; among them, 3454 cases had distant organ metastases. Overall, 27.5% of the entire cohort were patients with distant organ metastases. Compared with patients with non-metastatic MEC, patients with metastatic MEC were more likely to be aged ≤60 years, of Black and White race, have a primary lesion in the overlapping esophagus segments, and have a diagnosis of adenocarcinoma of poorly differentiated and undifferentiated grade that was treated with radiotherapy and chemotherapy rather than surgery; moreover, they were also more likely to be married and insured. In addition, patients with MEC were more likely to be aged ≤60 years, White race, and diagnosed with a primary lesion in the lower third of the esophagus and overlapping esophagus segments, and treated without chemotherapy, compared with those with FEC. Patients in the former group were also more likely than those in the latter group to be unmarried and have bone metastasis only and lung metastasis only. Liver, lung, and bone metastases separately, and simultaneous liver and lung metastases were associated with poor survival in MEC patients.
Metastatic MEC is associated with clinicopathological characteristics and metastatic patterns different from those associated with non-metastatic MEC and metastatic FEC. Metastatic MEC and FEC patients may have similar prognoses. Distant organ metastasis may be associated with poor prognosis in patients with MEC and FEC.
Keywords: esophageal cancer, male, metastasis, prognosis, SEER
1. Introduction
Esophageal cancer (EC) is the seventh most common cancer and the sixth leading cause of mortality worldwide.[1] In 2019, 17,650 new EC cases and 16,080 associated deaths were reported in the United States,[2] including 13,750 new diagnoses and 13,020 deaths among men. Men account for two-thirds of EC patients.[1,3–5] Approximately, 50% of EC patients presented with distant site metastases at the time of diagnosis.[6,7]
EC survival has improved with progress in treatment. Some population-based studies have reported that the overall 5-year survival rates of this patient group increased from <5% in the 1960s to >20% in the past decade.[8] However, in some European countries, the United States, and China, the prognosis of male EC (MEC) patients remains poor, in particular, in cases of distant organ metastasis.[9] Previous studies have reported on distant metastasis of EC; however, a systematic examination of MEC with distant metastasis has not been undertaken to date; thus, the clinical characteristics, metastatic patterns, and factors associated with prognosis in this patient group remain unclear.
The present study was based on a large population of patients with EC and metastasis. The data were extracted from the Surveillance Epidemiology and End Results (SEER) database (2010–2015). This study aimed to examine the clinical and epidemiological characteristics of MEC. Moreover, EC characteristics were compared between male and female EC (FEC) patients. Factors affecting MEC prognosis were examined.
2. Methods
2.1. Populations and characteristics
SEER∗stat software was used to extract data from the SEER-18 database, which is maintained by the National Cancer Institute and accounts for approximately 28% of the United States’ population. SEER∗stat provided information up to 2016; we included patients aged ≥18 years diagnosed with EC between January 1, 2010 and December 31, 2015 (N = 24,082). We excluded patients with diagnoses of other primary malignant tumors, diagnoses made at autopsy or via a death certificate, diagnosis not pathologically confirmed, and those who lacked information about distant organ metastasis status (Fig. 1).
Figure 1.
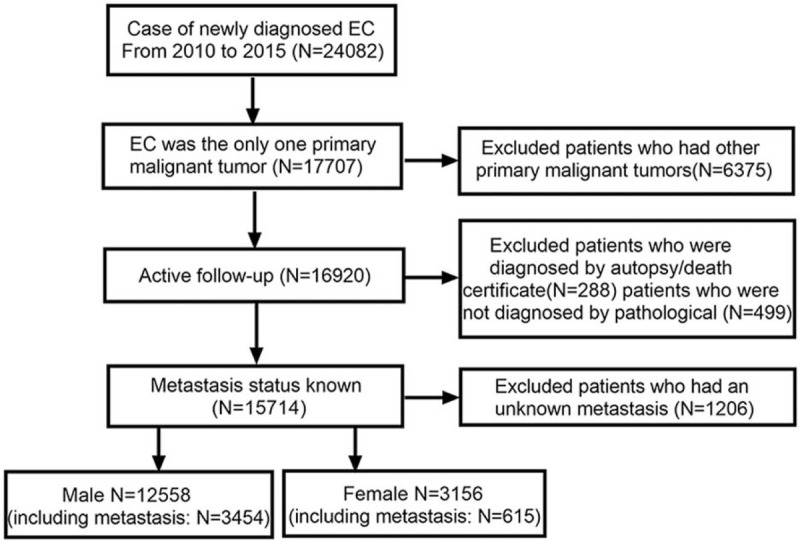
Flowchart of selection of patients with metastatic esophageal cancer used the SEER database. SEER = surveillance epidemiology and end results.
Characteristics of interest were age at diagnosis (≤60 years old and >60 years old); race (White, Black, and other race); primary lesion site (esophagus upper1/3, esophagus middle1/3, esophagus lower1/3, and spanning 2 or more esophageal segments means overlapping lesion); histopathology type (squamous cell carcinoma, adenocarcinoma, and other types); pathology grade (I well-differentiated, II moderately, III poorly differentiated, and IV undifferentiated); surgery, radiotherapy, chemotherapy, marital, and insurance status; and residence type.
Distant metastases were observed in the bone, brain, liver, and lungs. Metastasis patterns were divided into 15 groups: single-organ metastases (bone, brain, liver, and lung), 2-organ metastases (bone and brain, bone and liver, bone and lung, brain and liver, brain and lung, and liver and lung), 3-organ metastases (bone, brain, and liver; bone, brain, and lung; bone, liver, and lung; and brain, liver, and lung), and4-organ metastases (bone, brain, liver, and lung).
2.2. Statistical analysis
In this cohort study, descriptive analysis was used to calculate the proportion and an absolute number of cases per characteristic. The chi-squared test or Fisher exact test was used for between-group comparisons of characteristics. The metastatic proportion was defined as the percentage of patients with MEC with or without distant organ metastasis to the total number of MEC cases, and the number of FEC with distant metastasis to the total number of FEC cases. Survival estimates were obtained with the Kaplan–Meier method and compared using the log-rank test in MEC and FEC patients with different metastasis organs; univariable Cox regression was used to determine factors associated with all-cause mortality. Factors statistically significant (P < .05) in univariable analysis were entered into the multivariable Cox regression model.
The Kaplan–Meier curves were generated using GraphPad Prism software (version 8.0; La Jolla, CA, USA); other statistical analyses were performed with SPSS (version 25.0; IBM Corp., USA). Statistical significance was set as two-sided P-values of <.05. The SEER dataset used was publicly available and thus exempt from an ethics board review and the informed consent requirement. This study complied with the 1964 Helsinki Declaration and its subsequent amendments and other relevant ethical standards.
3. Results
3.1. Patients’ criteria
Among 12,558 MEC cases, 3454 patients had metastases. Among 3156 FEC cases, 615 patients had metastases. All patients were diagnosed during 2010–2015; the age range was 18 to 105 years. MEC and FEC patients with metastasis were aged 63.68 ± 11.135 and 66.30 ± 12.525 years, respectively. Metastatic MEC patients were more likely than their counterparts to be aged <60 years, of Black or White race, married, and without insurance, and to have an overlapping lesion primary site, and the diagnosis of poorly differentiated (grade III) or undifferentiated (grade IV) adenocarcinoma that was treated with radiotherapy and chemotherapy rather than surgery. The place of residence was similar in both groups. The patients’ clinical and demographic characteristics are presented in Table 1.
Table 1.
Clinical characteristics of male and female patients with esophageal cancer.
| MEC without metastasis | MEC with metastasis | FEC without metastasis | FEC with metastasis | |||||||
| N 9104 | % 72.5 | N 3454 | % 27.5 | N 2541 | % 80.5 | N 615 | % 19.5 | P value† | P value‡ | |
| Age at diagnosis(year) | .000∗∗ | .046∗ | ||||||||
| ≤60 | 2858 | 31.4 | 1343 | 38.9 | 647 | 25.5 | 213 | 34.6 | ||
| >60 | 6246 | 68.6 | 2111 | 61.1 | 1894 | 74.5 | 402 | 65.4 | ||
| Race | .001∗ | .000∗∗ | ||||||||
| Black | 758 | 8.3 | 322 | 9.3 | 389 | 15.3 | 99 | 16.1 | ||
| White | 7792 | 85.6 | 2973 | 86.1 | 1991 | 78.4 | 486 | 79.0 | ||
| Others | 512 | 5.6 | 154 | 4.5 | 149 | 5.9 | 29 | 4.7 | ||
| Unknown | 42 | 0.5 | 5 | 0.1 | 12 | 0.5 | 1 | 0.2 | ||
| Primary site | .000∗∗ | .000∗∗ | ||||||||
| Upper1/3 | 518 | 5.7 | 143 | 4.1 | 283 | 11.1 | 40 | 6.5 | ||
| Middle1/3 | 1460 | 16.0 | 455 | 13.2 | 772 | 30.4 | 144 | 23.4 | ||
| Lower1/3 | 6102 | 67.0 | 2266 | 65.6 | 1186 | 46.7 | 325 | 52.8 | ||
| Overlapping lesion | 364 | 4.0 | 215 | 6.2 | 107 | 4.2 | 25 | 4.1 | ||
| Unknown | 660 | 7.2 | 375 | 10.9 | 193 | 7.6 | 81 | 13.2 | ||
| Histopathology type | .000∗∗ | .000∗∗ | ||||||||
| Squamous cell carcinoma | 2363 | 26.0 | 709 | 20.5 | 1381 | 54.3 | 230 | 37.4 | ||
| Adenocarcinoma | 5938 | 65.2 | 2443 | 70.7 | 979 | 38.5 | 328 | 53.3 | ||
| Others | 803 | 8.8 | 302 | 8.7 | 181 | 7.1 | 57 | 9.3 | ||
| Pathology grade | .000∗∗ | .565 | ||||||||
| I well-differentiated | 540 | 5.9 | 86 | 2.5 | 145 | 5.7 | 14 | 2.3 | ||
| II moderately | 3103 | 34.1 | 943 | 27.3 | 965 | 38.0 | 168 | 27.3 | ||
| III poorly differentiated | 3682 | 40.4 | 1700 | 49.2 | 859 | 33.8 | 289 | 47.0 | ||
| IV undifferentiated | 111 | 1.2 | 56 | 1.6 | 32 | 1.3 | 8 | 1.3 | ||
| Unknown | 1668 | 18.3 | 669 | 19.4 | 540 | 21.3 | 136 | 22.1 | ||
| Surgery | .000∗∗ | .111 | ||||||||
| No | 5578 | 61.3 | 3376 | 97.7 | 1843 | 72.5 | 602 | 97.9 | ||
| Yes | 3362 | 36.9 | 67 | 1.9 | 653 | 25.7 | 8 | 1.3 | ||
| Unknown | 164 | 1.8 | 11 | 0.3 | 45 | 1.8 | 5 | 0.8 | ||
| Radiotherapy | .000∗∗ | .562 | ||||||||
| No | 5949 | 65.3 | 1340 | 38.8 | 1013 | 39.9 | 231 | 37.6 | ||
| Yes | 3155 | 34.7 | 2114 | 61.2 | 1528 | 60.1 | 384 | 62.4 | ||
| Chemotherapy | .000∗∗ | .034∗ | ||||||||
| No | 6229 | 68.4 | 2089 | 60.5 | 1049 | 41.3 | 344 | 55.9 | ||
| Yes | 2875 | 31.6 | 1365 | 39.5 | 1492 | 58.7 | 271 | 44.1 | ||
| Marital status | .000∗∗ | .000∗∗ | ||||||||
| Married | 3249 | 35.7 | 1372 | 39.7 | 907 | 35.7 | 366 | 59.5 | ||
| Unmarried | 5323 | 58.5 | 1925 | 55.7 | 1482 | 58.3 | 215 | 35.0 | ||
| Unknown | 532 | 5.8 | 157 | 4.5 | 152 | 6.0 | 34 | 5.5 | ||
| Residence type | .270 | .378 | ||||||||
| Rural | 173 | 1.9 | 53 | 1.5 | 33 | 1.3 | 5 | 0.8 | ||
| Urban | 8911 | 97.9 | 3396 | 98.3 | 2498 | 98.3 | 609 | 99.0 | ||
| Unknown | 20 | 0.2 | 5 | 0.1 | 10 | 0.4 | 1 | 0.2 | ||
| Insurance situation | .000∗∗ | .114 | ||||||||
| Insurance | 8581 | 94.3 | 3221 | 93.3 | 2399 | 94.4 | 562 | 91.4 | ||
| No insurance | 310 | 3.4 | 168 | 4.9 | 77 | 3.0 | 34 | 5.5 | ||
| Unknown | 213 | 2.3 | 65 | 1.9 | 65 | 2.6 | 19 | 3.1 | ||
FEC = female esophageal cancer, MEC = male esophageal cancer
Comparison between male esophageal cancer without metastasis and male esophageal cancer with metastasis.
Comparison between male esophageal cancer with metastasis and female esophageal cancer with metastasis.
P < .05.
P < .001.
MEC patients with distant organ metastasis were more likely than their female counterparts to be aged <60 years, unmarried, of White race, diagnosed with adenocarcinoma in the lower third of the esophagus and overlapping lesions; concurrently, the former group was less likely than the latter group to receive chemotherapy. Pathology grade, surgery and radiotherapy status, residence type, and insurance status were similar in both groups.
3.2. Metastasis patterns
There were 3454 patients in this cohort. The most common distant single-, 2-, and 3-organ metastasis sites were the liver (N = 1238, 35.8%), liver and lung (N = 466, 13.5%), and bone, liver, and lung (N = 140, 4.1%), respectively. Overall, the most common metastasis patterns were liver metastasis only (N = 1238, 35.8%), lung metastasis only (N = 483, 14.0%), bone metastasis only (N = 482, 14.0%), and concurrent liver and lung metastasis (N = 466, 13.5%).
MEC patients with distant organ metastasis were more and less likely than their FEC counterparts to have bone metastasis only (14.0% vs 10.9%), and lung metastasis only (14.0% vs 24.6%), respectively. The remaining metastatic patterns were observed at a similar frequency in both groups (Table 2).
Table 2.
Compare organ metastasis patterns between male and female patients with esophageal cancer.
| Male | Female | ||||
| N = 3454 | N = 615 | P value | |||
| Variable | n | % | n | % | |
| Bone metastasis only | 482 | 14.0 | 67 | 10.9 | .041∗ |
| Brain metastasis only | 95 | 2.8 | 10 | 1.6 | .105∗ |
| Liver metastasis only | 1238 | 35.8 | 204 | 33.2 | .202∗ |
| Lung metastasis only | 483 | 14.0 | 151 | 24.6 | <.001∗ |
| Bone and brain | 29 | 0.8 | 4 | 0.7 | .809∗∗ |
| Bone and liver | 268 | 7.8 | 40 | 6.5 | .278∗ |
| Bone and lung | 117 | 3.4 | 20 | 3.3 | .864∗ |
| Brain and liver | 39 | 1.1 | 3 | 0.5 | .147∗ |
| Brain and lung | 20 | 0.6 | 6 | 1.0 | .268∗∗ |
| Liver and lung | 466 | 13.5 | 74 | 12.0 | .326∗ |
| Bone, brain, and liver | 14 | 0.4 | 5 | 0.8 | .192∗∗ |
| Bone, brain, and lung | 11 | 0.3 | 5 | 0.8 | .081∗∗ |
| Bone, liver, and lung | 140 | 4.1 | 22 | 3.6 | .578∗ |
| Brain, liver, and lung | 27 | 0.8 | 2 | 0.3 | .300∗∗ |
| Bone, brain, liver, and lung | 25 | 0.7 | 2 | 0.3 | .416∗∗ |
| One site metastasis | 2298 | 66.5 | 432 | 70.2 | .071∗ |
| Two sites metastasis | 939 | 27.2 | 147 | 23.9 | .090∗ |
| Three sites metastasis | 192 | 5.6 | 34 | 5.5 | .976∗ |
| Four sites metastasis | 25 | 0.7 | 2 | 0.3 | .416∗∗ |
Pearson chi-squared test.
Fisher exact test.
3.3. Survival
The patients with the above-mentioned metastasis patterns accounted for over 80% of the sample and were included in survival analysis.
There was no difference in survival rates between metastatic MEC and FEC patients (Fig. 2). However, overall survival rates were different between metastatic and non-metastatic MEC patients (Fig. 3). Among patients with metastatic MEC, survival rates decreased with an increase in the number of metastatic sites (Fig. 4).
Figure 2.

OS rate of MEC and FEC patients at different metastasis sites. (A) OS of bone alone metastasis between MEC and FEC patients; (B) OS of liver alone metastasis between MEC and FEC patients; (C) OS of lung alone metastasis between MEC and FEC patients; and (D) OS of both liver and lung metastasis between MEC and FEC patients. FEC = female esophageal cancer, MEC = male esophageal cancer, OS = overall survival.
Figure 3.

The survival difference among the different metastasis sites in MEC patients. MEC = male esophageal cancer.
Figure 4.

The survival difference among the different number of metastasis sites in MEC patients. MEC = male esophageal cancer
Univariate Cox regression revealed 11 factors associated with all-cause mortality in metastatic MEC patients (Table 3). Multivariable Cox regression revealed that the following factors were associated with poor prognosis: age at diagnosis of >60 (vs ≤60) years, primary lesion site in the middle or lower third of the esophagus, and overlapping lesions (vs lesions in the upper third), pathology grade II (moderately differentiated), III (poorly differentiated), and IV (undifferentiated) (vs grade I, well-differentiated), no insurance (vs insurance), and distant organ metastatic (vs non-metastatic). Concurrently, factors associated with good prognosis in this patient group were being of non-Black race, diagnosis of adenocarcinoma (vs squamous cell carcinoma), treatment with surgery (vs without surgery) and chemotherapy (vs without chemotherapy), and being married (vs unmarried). Radiotherapy did not affect outcomes in the present study.
Table 3.
Univariate and multivariate survival analysis of male esophageal cancer patients with bone alone, liver alone, lung alone, and simultaneous liver and lung metastasis.
| Univariate analysis | Multivariate analysis | |||
| Characteristics | P | Hazard ratio | 95%CI | P value |
| Age at diagnosis (year) | <.001 | .011 | ||
| ≤60 | Reference | |||
| >60 | 1.062 | 1.014–1.113 | .011 | |
| Race | <.001 | <.001 | ||
| Black | Reference | |||
| White | 0.921 | 0.852–0.995 | .038 | |
| Others | 0.867 | 0.775–0.971 | .014 | |
| Unknown | 0.401 | 0.247–0.652 | <.001 | |
| Primary site | .010 | <.001 | ||
| Upper1/3 | Reference | |||
| Middle1/3 | 1.178 | 1.062–1.306 | .002 | |
| Lower1/3 | 1.217 | 1.101–1.346 | <.001 | |
| Overlapping lesion | 1.460 | 1.280–1.665 | <.001 | |
| Unknown | 1.272 | 1.129–1.432 | <.001 | |
| Histopathology type | <.001 | <.001 | ||
| Squamous cell carcinoma | Reference | |||
| Adenocarcinoma | 0.844 | 0.794–0.898 | <.001 | |
| Others | 0.964 | 0.882–1.053 | .414 | |
| Pathology grade | <.001 | <.001 | ||
| I well-differentiated | Reference | |||
| II moderately | 1.430 | 1.275–1.605 | <.001 | |
| III poorly differentiated | 1.872 | 1.670–2.098 | <.001 | |
| IV undifferentiated | 1.932 | 1.564–2.386 | <.001 | |
| Unknown | 1.295 | 1.149–1.461 | <.001 | |
| Surgery | <.001 | <.001 | ||
| No | Reference | |||
| Yes | 0.277 | 0.261–0.295 | <.001 | |
| Unknown | 0.652 | 0.543–0.782 | <.001 | |
| Radiotherapy | <.001 | .526 | ||
| No | Reference | |||
| Yes | 1.017 | 0.966–1.070 | .526 | |
| Chemotherapy | <.001 | <.001 | ||
| No | Reference | |||
| Yes | 0.534 | 0.507–0.562 | <.001 | |
| Marital status | <.001 | <.001 | ||
| Unmarried | Reference | |||
| Married | 0.846 | 0.809–0.885 | <.001 | |
| Unknown | 0.802 | 0.725–0.886 | <.001 | |
| Residence type | .882 | NA | ||
| Rural | ||||
| Urban | ||||
| Unknown | ||||
| Insurance situation | <.001 | <.001 | ||
| Insurance | Reference | |||
| No insurance | 1.262 | 1.132–1.406 | <.001 | |
| Unknown | 0.850 | 0.730–0.989 | .035 | |
| Metastasis | <.001 | <.001 | ||
| None | Reference | |||
| Bone only | 2.105 | 1.910–2.320 | <.001 | |
| Liver only | 1.836 | 1.712–1.968 | <.001 | |
| Lung only | 1.609 | 1.458–1.776 | <.001 | |
| Liver and lung | 2.372 | 2.146–2.623 | <.001 | |
CI = confidence intervals.
4. Discussion
In the present study, we examined the clinical characteristics, metastatic patterns, and factors affecting prognosis in patients with metastatic MEC, registered in the SEER database. Outcomes were compared among metastatic and non-metastatic MEC patients and metastatic FEC patients. In the present study, 27.5% of MEC patients were metastatic cases; this rate was higher than that of the FEC group. The clinicopathological characteristics were different between the cohorts. To the best of our knowledge, this is the first large study on EC metastasis in men.
Some previous studies have shown similar findings in patients with metastatic EC,[7,9] including higher incidence among men than among women; in the present study, the rate of EC was 1.5-fold higher among men than among women. Male sex hormones may promote EC cell proliferation and metastasis,[10,11] while men are more likely than women to drink alcohol and smoke cigarettes. These hormonal and behavioral differences between men and women may account for the differences in EC rates between the sexes.
Younger patients with EC are more likely than their older counterparts to have metastasis[12]; men are particularly vulnerable to metastatic EC, as seen in the present study. In the populations of the United States and Europe, adenocarcinoma is the main histopathological type of EC,[1,13] particularly common among men with metastatic EC. Poorly differentiated malignant tumors are associated with poor prognosis and increased incidence of distant organ metastasis.[14,15] Advanced-stage cancers tend to be treated with chemotherapy and radiation rather than surgery, which might also be the reason why patients with metastatic MEC rarely receive surgery. Metastatic MEC patients were treated with surgery and radiotherapy at a rate similar to that observed in metastatic FEC patients, and with marginally reduced rates of chemotherapy. Metastatic MEC patients were more and less likely than non-metastatic MEC and FEC patients to be married, respectively. Understanding whether malignant tumor metastasis is associated with marital status requires further research. The rate of insurance holders was lower among patients with metastatic MEC than among those with non-metastatic MEC; this rate was similar to that among metastatic FEC patients. Uninsured patients have reduced access to medical care, which may increase their risk of progressing to advanced-stage cancer.
We compared distant organ metastasis patterns between MEC and FEC patients; overall, the most and least common metastatic sites among EC patients were the liver and brain, respectively; this finding is consistent with that of previous studies.[16–19] The incidence of bone-only metastasis among metastatic MEC patients was higher than that among metastatic FEC patients.[20] Ma et al reported that female sex was associated with a lower risk of bone metastasis in digestive system cancers.[21] Sex hormone levels differ between men and women, and musculoskeletal health may be the primary cause of these differences.[22] Differences in lifestyle factors may also account for these outcomes; for example, men are 1.5-times more likely than women to smoke.[23] Smoking is a risk factor for breast cancer metastasis, including bone and lung metastases.[24,25]
However, metastatic MEC patients had a lower incidence of lung only metastasis than did metastatic FEC patients; this finding is consistent with that of previous studies.[7,26] The lung-only metastatic pattern in MEC patients was different from that observed in patients with gastric cancer and hepatocellular carcinoma. [27,28] The reason behind this phenomenon is unknown, which might indicate that many more MEC patients with lung metastasis had simultaneously other distant organ metastasis, and further studies are needed to explain this. The rate of simultaneous bone and lung metastases was similar in both MEC and FEC patients. Single-site metastasis was the most common metastatic pattern in both MEC and FEC patients, followed by 2- and 3-site metastasis; 4-site metastasis was the least common type.
In the present study, there were differences in clinicopathological characteristics and metastatic patterns between metastatic MEC and FEC patients. However, the overall survival rates were similar in both groups and among patients with the most common metastatic sites (the liver, bone, lung, liver, and lung). [9]
Similar findings were reported in other malignant tumors such as breast and colorectal cancers with distant metastases, among others.[29,30] Compared to non-metastatic MEC patients, metastatic MEC patients had poor overall survival (P < .001). Multivariable Cox regression revealed that distant organ metastasis was associated with poor prognosis in MEC patients (Table 3). In addition, among all metastatic patterns, simultaneous liver and lung metastases were associated with the poorest prognosis. Overall, the prognosis associated with metastatic sites was poor. In the present study, radiotherapy did not seem to affect outcomes; this finding is in contrast to that of previous studies. However, this study did not account for radiotherapy dose or target due to the lack of data. However, the role of both palliative and definitive radiotherapy in the treatment of metastatic EC is controversial.[31–35] Clinical trials are required to validate the present findings.
4.1. Limitations
This study has some limitations. First, this study was based on the SEER database, which is a retrospective database, and we could only obtain information on liver, lung, bone, and brain metastases; data on other metastatic sites were not available. Second, only cases of synchronous metastases were recorded; data on asynchronous metastases were lacking. Third, we could not establish what factors were associated with the differences in metastatic patterns between men and women; further research is required to elucidate the mechanisms of these differences. Fourth, the present study sample was extracted from the United States population; the present findings may not generalize to other populations.
5. Conclusion
The present study is the first large-scale report on MEC characteristics and metastasis patterns in EC. The present findings are relevant to clinicians managing patients with EC. Although metastatic patterns may differ between MEC and FEC patients, the prognosis may be similar for both patient groups. Distant organ metastasis in EC patients may be a risk factor for poor outcomes.
Acknowledgments
We would like to thank the staff members of the National Cancer Institute and the researchers who have been involved with the SEER Program.
Author contributions
Shengqiang Zhang performed data collection, data analysis, and manuscript writing. Jida Guo, Hongyan Zhang, and Huawei Li performed data collection and data analysis. Mohamed Osman Omar Hassan took part in manuscript writing. Linyou Zhang performed project development. All authors contributed to the article and approved the submitted version.
Conceptualization: Shengqiang Zhang.
Data curation: Shengqiang Zhang, Jida Guo, Hongyan Zhang, Huawei Li.
Formal analysis: Jida Guo, Hongyan Zhang, Huawei Li.
Investigation: Shengqiang Zhang, Hongyan Zhang.
Project administration: Shengqiang Zhang, Linyou Zhang.
Supervision: Linyou Zhang.
Writing – original draft: Shengqiang Zhang.
Writing – review & editing: Mohamed Osman Omar Osman Omar Hassan.
Footnotes
Abbreviations: CI = confidence intervals, EC = esophageal cancer, FEC = female esophageal cancer, MEC = male esophageal cancer, OS = overall survival, SEER = surveillance epidemiology and end results.
How to cite this article: Zhang S, Guo J, Zhang H, Li H, Hassan MO, Zhang L. Metastasis pattern and prognosis in men with esophageal cancer patients: a SEER-based study. Medicine. 2021;100:25(e26496).
The authors have no funding and conflicts of interest to disclose.
The datasets generated during and/or analyzed during the present study are available from the corresponding author on reasonable request.
References
- [1].Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer 2018;68:394–424. [DOI] [PubMed] [Google Scholar]
- [2].Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer 2019;69:07–34. [DOI] [PubMed] [Google Scholar]
- [3].Thrumurthy SG, Chaudry MA, Thrumurthy SSD, Mughal M. Oesophageal cancer: risks, prevention, and diagnosis. BMJ 2019;366:l4373. [DOI] [PubMed] [Google Scholar]
- [4].Botterweck AA, Schouten LJ, Volovics A, Dorant E, van Den Brandt PA. Trends in incidence of adenocarcinoma of the oesophagus and gastric cardia in ten European countries. Int J Epidemiol 2000;29:645–54. [DOI] [PubMed] [Google Scholar]
- [5].Devesa SS, Blot WJ, Fraumevni JF. Changing patterns in the incidence of esophageal and gastric carcinoma in the United States. Cancer 1998;83:2049–53. [PubMed] [Google Scholar]
- [6].Enzinger PC, Mayer RJ. Esophageal cancer. N Engl J 2003;349:2241–52. [DOI] [PubMed] [Google Scholar]
- [7].Wu SG, Zhang WW, He ZY, Sun JY, Chen YX, Guo L. Sites of metastasis and overall survival in esophageal cancer: a population-based study. Cancer Manag Res 2017;9:781–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Forde PM, Kelly RJ. Chemotherapeutic and targeted strategies for locally advanced and metastatic esophageal cancer. J Thorac Oncol 2013;8:673–84. [DOI] [PubMed] [Google Scholar]
- [9].Ai D, Zhu H, Ren W, et al. Patterns of distant organ metastases in esophageal cancer: a population-based study. J Thorac Dis 2017;9:3023–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Sukocheva OA, Li B, Due SL, Hussey DJ, Watson DI. Androgens and esophageal cancer: what do we know? World J Gastroenterol 2015;21:6146–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Busby J, Karasneh R, Murchie P, et al. The role of 5α-reductase inhibitors in gastro-oesophageal cancer risk: a nested case-control study. Pharmacoepidemiol Drug Saf 2020;29:48–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Barz H, Barz D. [Age dependence of metastases. A study of more than 5000 cases of death from cancer]. Arch Geschwulstforsch 1984;54:77–83. [PubMed] [Google Scholar]
- [13].Edgren G, Adami HO, Weiderpass E, Nyrén O. A global assessment of the oesophageal adenocarcinoma epidemic. Gut 2013;62:1406–14. [DOI] [PubMed] [Google Scholar]
- [14].Rice TW, Rusch VW, Apperson-Hansen C, et al. Worldwide esophageal cancer collaboration. Dis Esophagus 2009;22:01–8. [DOI] [PubMed] [Google Scholar]
- [15].Dickson GH, Singh KK, Escofet X, Kelley K. Validation of a modified GTNM classification in peri-junctional oesophago-gastric carcinoma and its use as a prognostic indicator. Eur J Surg Oncol 2001;27:641–4. [DOI] [PubMed] [Google Scholar]
- [16].Bosch A, Frias Z, Caldwell WL, Jaeschke WH. Autopsy findings in carcinoma of the esophagus. Acta Radiol Oncol Radiat Phys Biol 1979;18:103–12. [DOI] [PubMed] [Google Scholar]
- [17].Chen MQ, Xu BH, Zhang YY. Analysis of prognostic factors for esophageal squamous cell carcinoma with distant organ metastasis at initial diagnosis. J Chin Med Assoc 2014;77:562–6. [DOI] [PubMed] [Google Scholar]
- [18].Tustumi F, Kimura CMS, Takeda FR, Sallum RA, Ribeiro-Junior U, Cecconello I. Evaluation of lymphatic spread, visceral metastasis and tumoral local invasion in esophageal carcinomas. Arq Bras Cir Dig 2016;29:215–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Mariette C, Balon JM, Piessen G, Fabre S, Van Seuningen I, Triboule JP. Pattern of recurrence following complete resection of esophageal carcinoma and factors predictive of recurrent disease. Cancer 2003;97:1616–23. [DOI] [PubMed] [Google Scholar]
- [20].Zhang J, Ma W, Wu H, et al. Analysis of homogeneous and heterogeneous factors for bone metastasis in esophageal cancer. Med Sci Monit 2019;25:9416–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Ma W, Peltzer K, Qi L, et al. Female sex is associated with a lower risk of bone metastases and favourable prognosis in non-sex-specific cancers. BMC Cancer 2019;19:1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Wiren KM, Zhang XW, Olson DA, Turner RT, Iwaniec UT. Androgen prevents hypogonadal bone loss via inhibition of resorption mediated by mature osteoblasts/osteocytes. Bone 2012;51:835–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Szklo AS, de Souza MC, Szklo M, de Almeida LM. Smokers in Brazil: who are they? Tob Control 2016;25:564–70. [DOI] [PubMed] [Google Scholar]
- [24].Yao S, Zhang Y, Tang L, et al. Bone remodeling and regulating biomarkers in women at the time of breast cancer diagnosis. Breast Cancer Res Treat 2017;161:501–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Scanlon EF, Suh O, Murthy SM, Mettlin C, Reid SE, Cummings KM. Influence of smoking on the development of lung metastases from breast cancer. Cancer 1995;75:2693–9. [DOI] [PubMed] [Google Scholar]
- [26].Ai D, Chen Y, Liu Q, Deng J, Zao K. The effect of tumor locations of esophageal cancer on the metastasis to liver or lung. J Thorac Dis 2019;11:4205–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Qiu MZ, Shi SM, Chen ZH, et al. Frequency and clinicopathological features of metastasis to liver, lung, bone, and brain from gastric cancer: a SEER-based study. Cancer Med 2018;7:3662–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Wu C, Ren X, Zhang Q. Incidence, risk factors, and prognosis in patients with primary hepatocellular carcinoma and lung metastasis: a population-based study. Cancer Manag Res 2019;11:2759–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Xie J, Ying YY, Xu B, Li Y, Zhang X, Li C. Metastasis pattern and prognosis of male breast cancer patients in US: a population-based study from SEER database. Ther Adv Med Oncol 2019;11:1758835919889003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Shi T, Huang M, Han D, et al. Chemotherapy is associated with increased survival from colorectal signet ring cell carcinoma with distant metastasis: A Surveillance, Epidemiology, and End Results database analysis. Cancer Med 2019;8:1930–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Hingorani M, Dixit S, Johnson M, et al. Palliative radiotherapy in the presence of well-controlled metastatic disease after initial chemotherapy may prolong survival in patients with metastatic esophageal and gastric cancer. Cancer Res Treat 2015;47:706–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Lyu J, Li T, Wang Q, et al. Outcomes of concurrent chemoradiotherapy versus chemotherapy alone for stage IV esophageal squamous cell carcinoma: a retrospective controlled study. Radiat Oncol 2018;13:233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Xu J, Lu D, Zhang L, Li J. Palliative resection or radiation of primary tumor prolonged survival for metastatic esophageal cancer. Cancer Med 2019;8:7253–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Huang S, Li Y, Ma H, et al. Investigating the survival benefit of combining radiotherapy for surgery treated locally advanced esophageal squamous cell carcinoma patients aged 65 and older. J Gastrointest Surg 2019;23:2111–8. [DOI] [PubMed] [Google Scholar]
- [35].Zhang R, Jia M, Li P, et al. Radiotherapy improves the survival of patients with metastatic esophageal squamous cell carcinoma: a propensity score matched analysis of Surveillance, Epidemiology, and End Results database. Dis Esophagus 2019;32: undefined. [DOI] [PubMed] [Google Scholar]


