Abstract
Wilson's disease (WD) is a rare condition caused by copper accumulation primarily in the liver and secondly in other organs, such as the central nervous system. It is a hereditary autosomal recessive disease caused by a deficiency in the ATP7B transporter. This protein facilitates the incorporation of copper into ceruloplasmin. More than 800 mutations associated with WD have been described. The onset of the disease frequently includes manifestations related to the liver (as chronic liver disease or acute liver failure) and neurological symptoms, although it can sometimes be asymptomatic. Despite it being more frequent in young people, WD has been described in all life stages. Due to its fatal prognosis, WD should be suspected in all patients with unexplained biochemical liver abnormalities or neurological or psychiatric symptoms. The diagnosis is established with a combination of clinical signs and tests, including the measurement of ceruloplasmin, urinary copper excretion, copper quantification in liver biopsy, or genetic assessment. The pharmacological therapies include chelating drugs, such as D-penicillamine or trientine, and zinc salts, which are able to change the natural history of the disease, increasing the survival of these patients. In some cases of end-stage liver disease or acute liver failure, liver transplantation must be an option to increase survival. In this narrative review, we offer an overview of WD, focusing on the importance of clinical suspicion, the correct diagnosis, and treatment.
Keywords: Wilson´s disease, Copper, ATP7B, Ceruloplasmin, Chelator, Liver disease
Core Tip: Wilson’s disease (WD) is a rare metabolic disorder caused by the deposition of copper in organs, particularly in the liver and the brain. As the symptoms and clinical presentation can be highly variable, WD is not always suspected. A detailed but practical review is presented to assist clinicians in the diagnosis and management of WD.
INTRODUCTION
Wilson's disease (WD) is an autosomal-recessive monogenic disorder characterized by an excessive accumulation of copper, firstly described in 1912 by Kinnear Wilson. The World Health Organization estimates the global prevalence of WD to be between 1/10000 and 1/30000[1]. It is caused by mutations in the ATP7B gene, which encodes a transporter protein with ATPase activity. This transporter is involved in incorporating copper into apoceruloplasmin, which is finally eliminated in bile. When a mutation affects the ATP7B transporter, free copper is released into the bloodstream and is removed by urine instead of feces[2]. Therefore, ATP7B is essential for copper biliary excretion[3].
In this review, we aimed to revise the clinical aspects of WD, including diagnosis, clinical manifestations, and the therapeutic approach, and discuss the future treatment of the disease.
GENETICS
The ATP7B gene is located on chromosome 13q14.3 and comprises 20 introns and 21 exons, encoding a protein of 165 amino acids[4,5], whose function is the incorporation of copper into ceruloplasmin. Currently, more than 800 mutations have been discovered in the gene[6], of which 380 have confirmed involvement in the pathogenesis of the disease[7,8]. Although mutations have been reported in almost all exons[5], they mainly affect the central regions of the gene (both 8 and 14 exons are the most frequently affected). The most common mutations are H1069Q and R778L in European and Asian populations, respectively[2,4]. Approximately 90%-98% of WD subjects are heterozygous, showing different mutations in each of the alleles encoding the ATP7B. On the other hand, the phenotype and the penetrance of WD can be extremely variable. Even patients carrying two disease-causing mutations do not necessarily have a demonstrable alteration of copper metabolism[9]. Some of the proposed reasons are differences in copper intake, individual antioxidant capacity or susceptibility to liver fibrosis, and hormonal influences[10].
The potential role that epigenetics could have in the gene expression of the disease should be highlighted. Some experimental models have shown changes in DNA methylation through breast milk enriched with methyl groups that could be related to the clinical manifestation of WD[11].
Considering the probability of late-onset, the fact of having asymptomatic cases, and the phenotypic variability, it seems vital to evaluate the previous and next generation of the index case[12]. Both the European Association for the Study of the Liver (EASL) and the American Association for the Study of Liver Diseases (AASLD) recommend an appropriate study of the index case taking into account the family history of liver- and brain-related disease[7,13]. These guidelines propose to assess the patient's siblings since the risk of WD is 25% (by presenting two mutations in both alleles). Subsequently, other first-degree family members should be evaluated, although the risk decreases to 0.5%[12].
CLINICAL MANIFESTATIONS
There is a wide variety of symptoms involved in WD, which predominantly affect the liver and brain (Table 1). Although WD may be present at any age, it is more common between the ages of 5 and 35. However, it should be investigated in patients with liver failure due to an unknown cause and those with liver disease and neuropsychiatric symptomatology[13]. Asymptomatic patients are commonly diagnosed during the family screening process[7].
Table 1.
Clinical manifestations
| Wilson's Disease Clinical Manifestations | |
| Liver | Hepatomegaly, jaundice, pain in right hypochondria, asthenia, elevation of transaminases, acute liver injury, acute liver failure, cirrhosis (compensated and decompensated), ACLF, steatosis |
| Neurological | Dystonia, tremor, dysarthria, dysphagia, Parkinson, chorea |
| Psychiatric | Behavioral changes, depression, anxiety, psychosis, school performance deficit, sexual disinhibition |
| Eye | Kayser-Fleischer Ring, Cataract |
| Hematologic | Hemolytic anemia, coagulopathy, thrombopenia |
| Renal | Acute renal failure, nephrolithiasis, urolithiasis, renal tubular acidosis |
| Musculoskeletal | Arthropathy, muscle weakness |
| Other | Heart disease, pancreatitis, hypoparathyroidism |
ACLF: Acute-on-Chronic Liver Failure.
Liver symptoms
Liver symptoms of WD occur mainly during childhood and adolescence[10]. In these cases, liver involvement appears up to 10 years before neurological manifestations[7]. The clinical spectrum ranges from asymptomatic patients, with mild analytical alterations, to subjects with fulminant liver failure. In this scenario, there are forms of acute (from acute hepatitis to fulminant liver failure) and chronic presentation (from steatosis to compensated and decompensated cirrhosis)[14].
Asymptomatic forms usually have only hepatomegaly, discretely elevated transaminases, or are identified during the screening of an index case[15].
Acute presentation: WD should be suspected in a patient with acute hepatitis, in which viral hepatitis is ruled out. Symptoms are similar to acute viral hepatitis, with jaundice and abdominal pain[14]. This situation, including acute liver injury (manifested by coagulopathy) or acute liver failure (with hepatic encephalopathy), occurs predominantly in women[16]. Beyond these signs and symptoms, the elevation of hemoglobin, cholinesterase, and low alkaline phosphatase are characteristic of acute WD. Sometimes, hemolytic anemia with a negative Coombs test is presented, one of the diagnostic criteria of WD[7]. WD causes 2%-5% of acute liver failure events, showing a fatal prognosis in the absence of liver transplantation (LT) [14].
Chronic hepatitis and cirrhosis: Typically, it starts as a slight transaminase elevation that progresses slowly to fibrosis and, finally, cirrhosis. When it manifests itself as cirrhosis, there is an increased risk of mortality[8]. Sometimes, patients may show splenomegaly uniquely as a sign of portal hypertension. In particular, young patients over three years of age showing cirrhosis should be evaluated for WD[15]. On the other hand, WD can initially be confused with autoimmune hepatitis, as they occur at a similar age and are manifested by jaundice and increased transaminases and gammaglobulins[14]. Also, WD has been described as causing hepatic steatosis, which is identified in up to 15% of biopsies[17].
Neurological symptoms
Neurological involvement typically appears after liver manifestations. WD affects the central nervous system mainly through extrapyramidal system dysfunction and bulbar involvement. The most common symptom is dysarthria, particularly in the early stages of the disease[8]. The neurological presentation can also be manifested by tremors, parkinsonism, or involuntary movements, even by cerebellar dysfunction, chorea, or hyperreflexia[14]. Furthermore, dystonia affecting the face and jaw is characteristic, producing a typical sign (Wilson’s face)[15]. Also, a postural tremor is common in WD patients[7].
Psychiatric symptoms
Psychiatric symptoms must be considered in WD. In fact, patients showing these symptoms often suffer a delayed diagnosis[18]. In fact, approximately one third of patients develop psychiatric symptoms as the initial manifestation[7]. Typical symptoms are depression and anxiety[14], although changes in behavior or personality or impulsivity can occur[19]. In addition, affective disorders are more common than psychotic spectrum disorders.
Ocular manifestations
Kayser-Fleischer´s (KF) ring represents a frequent manifestation of WD, which affects the Descemet membrane of the cornea. The slit-lamp examination shows a brown-gold colored ring on the periphery of the cornea[20]. It is present in more than 90% of patients with WD showing neurological involvement but only in half of cases with liver disease. Notably, the KF ring does not affect vision, and its disappearance has been seen in patients undergoing effective treatment and LT[15]. Although it is one of the most typical features of WD, this ring has been described in cholestatic syndromes and other diseases[21].
Other symptomatology
As copper can be accumulated in different organs and systems, WD has been associated with arthropathy[22], recurrent muscle weakness due to hypokalaemia[23], cardiomyopathy[24], symptomatic urolithiasis[25], pancreatitis[26], cases of hypoparathyroidism[27], and infertility[28,29].
DIAGNOSIS
There are no specific diagnostic tests for WD (Table 2). Instead, a combination of clinical signs and symptoms and some tests are required to achieve the final diagnosis.
Table 2.
Diagnosis tests for Wilson’s disease
|
Test
|
Normal values
|
Wilson disease
|
False negative
|
False positive
|
| Ceruloplasmin | 0.2-0.4 g/L | < 0.2 g/L | Increased levels: | Low levels: |
| Hepatic inflammation | Malabsorption | |||
| Malnutrition | ||||
| Estrogen | Aceruloplasminemia | |||
| Pregnancy | Menkes’ disease | |||
| Infection | Terminal liver disease | |||
| Children | Nephropathy with renal protein loss | |||
| Overestimation by immunological assay | Excess zinc ingestion | |||
| Healthy heterozygotes WD | ||||
| Non ceruloplasmin bound copper | < 0.3 μg/dL | > 10 μg/dL | Overestimation of ceruloplasmin by immunological assay | Increased levels: |
| Cholestatic syndromes | ||||
| Acute liver failure | ||||
| Copper intoxication | ||||
| Urinary copper excretion | < 0.6 μmol/24 h; < 40 μg/24 h | > 1.6 μmol/24 h; > 100 μg/24 h | Incomplete collection; Children | Increased levels: |
| Cholestatic syndromes | ||||
| Autoimmune hepatitis | ||||
| Chronic active liver disease or hepatocellular necrosis | ||||
| Healthy heterozygotes WD | ||||
| Liver biopsy | < 50 μg/g; < 0.8 μmol/g | > 250 μg/g; > 4 μmol/g | Uneven copper distribution | Increased levels: |
| Cholestatic syndromes | ||||
| Idiopathic copper toxicosis disorders | ||||
| Kayser Fleischer rings | Absence | Present: Neurological WD | Primary biliary cholangitis | |
| Absence: | ||||
| 50% hepatic WD | ||||
| Asymptomatic WD |
WD: Wilson’s disease.
Ceruloplasmin
Ceruloplasmin is the leading copper transporter protein, carrying 90% of serum circulating copper. It is synthesized in the liver and excreted into the circulation from hepatocytes, mostly as holoceruloplasmin (containing six copper atoms) and the remainder as apoceruloplasmin (not joined to copper)[30]. Ceruloplasmin levels may be determined enzymatically by its copper-dependent oxidase activity or by immunological assays. The immunological assay measures the total ceruloplasmin level but not the ceruloplasmin oxidase activity. Therefore, normal levels of ceruloplasmin do not rule out low oxidase activity and WD. For this reason, the use of enzymatic assays is more appropriate[31]. Blood ceruloplasmin levels are typically low (< 0.2 g/L) in patients with WD and neurological involvement. However, they may be higher in up to half of patients with WD[32]. On the other hand, ceruloplasmin levels are not decreased only in WD, but can be reduced in other conditions such as renal or enteric protein loss, malabsorption, end-stage liver disease, or aceruloplasminemia[33]. In addition, up to 20% of healthy heterozygous carriers have low non-pathological levels of ceruloplasmin. Ceruloplasmin is also an acute-phase reactant and may be elevated in inflammation or infections, resulting in false negatives in WD patients with both characteristics[7].
Serum copper
Serum copper decreases proportionally with ceruloplasmin levels. WD should be considered when normal or elevated serum copper levels along with decreased ceruloplasmin are identified, as this indicates an increase in the concentration of non-ceruloplasmin-bound copper[34]. However, in patients with deficient ceruloplasmin levels, low total serum copper levels can be found even though free copper (albumin-bound copper or non-ceruloplasmin bound copper) may be increased. For this reason, only the determination of free copper is important as total serum copper mostly reflects ceruloplasmin-bound copper. To calculate free copper, serum copper must be subtracted from the value of ceruloplasmin and multiplied by 3 (each ceruloplasmin molecule provides 3 mg of copper). Patients with WD have free copper levels between 10-20 mg/dL and symptomatic individuals have levels > 20 mg/dL[35]. Free copper levels may also be increased in cholestatic syndromes and copper intoxication[36] and strikingly elevated in acute WD liver failure due to the sudden release of copper from the liver.
Determining free copper is challenging due to the inadequacy of ceruloplasmin determination methods. It is preferable to use enzymatically determined ceruloplasmin levels when calculating free copper, but they do not detect apoceruloplasmin and overestimate ceruloplasmin. For this reason, the determination of ceruloplasmin non-bound copper is not commonly used as a diagnostic method[37]. In 2009, a new method called exchangeable copper (CuEXC) was proposed for the direct determination of labile copper. It can be performed routinely and allows a direct and accurate measurement of copper overload, representing an extrahepatic biomarker[38]. For instance, values greater than 2.08 mmol/L suggest a high risk of severe neurological disease[39]. Additionally, CuEXC facilitates calculation of the relative exchangeable copper. When its threshold is higher than 18.5%, this biomarker reaches a sensitivity and specificity close to 100% in WD diagnosis, without the presence of false negatives[40,41]. Therefore, it could differentiate WD from other liver diseases and healthy heterozygous subjects, representing a promising family screening marker[2,42].
Urinary copper excretion
Urinary copper excretion in 24 h reflects the amount of circulating non-ceruloplasmin copper and, therefore, represents the excess copper excreted in the urine. In children, a value greater than 0.64 mmol/24 h or 40 g/24 h is suggestive of WD, while the cut-off for adults is 1.6 mmol/24 h (100 g/24 h)[16]. However, in up to 16%-23%, especially in asymptomatic children and siblings, urinary copper excretion may be lower than the values set[34,43]. After D penicillamine (DPA) administration (1.000 mg administered in two doses), urinary copper excretion consists of measuring urinary copper excretion within 24 h on the same day. It has been proven that urinary copper excretion values > 160 μg/24 h is compatible with WD in children[44]. However, this test is not standardized in adults, so it is not currently recommended in that population.
The determination of urinary copper excretion is challenging in some scenarios, such as the presence of renal failure and an incomplete or inadequate collection of urine. In addition, patients with autoimmune hepatitis, cholestatic diseases, acute liver failure, or asymptomatic heterozygous patients can show elevated urinary copper excretion[45].
Liver biopsy
Liver biopsy is a non-risk-free invasive technique; thus, it is not easy to perform in asymptomatic patients. Its use is limited to patients with compatible clinical or biochemical findings but without a definite diagnosis.
WD has no specific histological changes, although there are suggestive changes. Mild steatosis may be observed in patients without risk factors (alcohol, overweight, diabetes mellitus, or dyslipidemia) who are often mistaken to have non-alcoholic fatty liver disease. Furthermore, staining of metallothionein (protein-bound to intrahepatocyte copper) by orcein or lysosomal copper complexes, using rodain or rubenic acid, show liver copper deposits[35]. The sensitivity of these stains increases when the sample is deposited in xylol for 24 h[46]. Despite this, the hepatic accumulation of copper cannot be ruled out with histochemistry as staining only reveals copper deposits in less than 10% of patients. Thus, intrahepatic copper quantification is essential for the diagnosis of WD after a hepatic biopsy. For the determination of copper in dry weight, it is necessary to obtain a significant sample (at least 1 cm) and its placement in a copper-free and dry container. Values greater than 250 μg/g (4 mmol/g) are diagnostic, while values less than 50 μg/g (0.8 μmol/g) make the diagnosis highly unlikely. The major problem of the intrahepatic quantification of copper is the heterogeneity of distribution of liver copper deposits (which could be unrepresentative), as well as the elevation of intrahepatic copper deposits in cholestatic diseases[47].
Neurological and psychiatric assessment
Patients with WD, even if they have predominantly hepatic involvement, should be evaluated neurologically. The neurological symptoms in WD are varied, and include Parkinsonian motor alterations and psychiatric symptoms[18]. Magnetic resonance imaging (MRI) shows structural abnormalities with a hyperintensity in the T2 sequence in the basal ganglia, tectum, spinal bulb, thalamus, and brainstem. Also, there is a decreased intensity in the T1 sequence in the basal ganglia[48]. During MRI, the "giant panda face" sign, found in 14% of patients, is characterized by hyperintensity of the tegmentum of the midbrain, especially around the red nucleus, which maintains its normal hypointensity on T2-weighted imaging axial sections of the brain. This sign, along with the tectal and center-protuberance plaque's hyperintensity and the simultaneous involvement of the basal ganglia, thalamus, and brainstem, are practically pathognomonic of WD[49].
Genetic testing
Direct sequencing of the ATP7B gene provides the greatest efficiency in clinical molecular diagnosis. The most common mutation (H1069Q) is present in 40%-50% of patients in Western countries; however, 17% of patients with a diagnosis established by the Leipzig criteria do not have any identifiable ATP7B gene mutation[50]. This may be explained by the inability of genetic testing to distinguish disease-specific mutations from polymorphisms of the gene and the absence of analyzing the non-coding regions of the gene, which can also affect gene expression. However, next-generation sequencing is becoming a very useful, reliable, time-saving, and cost-effective tool for diagnostic testing in the future.
How is the diagnosis established?
As previously described, a single test does not allow a definite diagnosis of WD. For this reason, a scoring system that combines clinical parameters with biochemical and imaging tests, known as the Leipzig criteria[7,13], is needed for patients (Table 3)[51]. More than 4 points are required to establish the diagnosis of WD according to these criteria, while an alternative diagnosis should be considered in individuals showing less than 4 points. Therefore, liver biopsy and the genetic assessment may not be needed if other test results add up to at least 4 points. However, the Leipzig criteria show some weaknesses that have to be taken into account, such as the lack of definition of the upper limit of normality of urinary copper excretion or the importance attributed to urinary copper excretion in 24 h after stimulation with DPA[52,53].
Table 3.
Leipzig scoring for Wilson’s disease
|
Typical clinical signs and symptoms
| |
| Kayser-Fleischer ring | |
| Present | 2 |
| Absent | 0 |
| Neurologic symptoms or typical abnormalities on MRI | |
| Severe | 2 |
| Mild | 1 |
| Absent | 0 |
| Serum ceruloplasmin | |
| Normal (> 0.2 g/L) | 0 |
| 0.1-0-2 g/L | 1 |
| < 0.1 g/L | 2 |
| Coombs negative hemolytic anemia | |
| Present | 1 |
| Absent | 0 |
| Other tests | |
| Liver copper1 | |
| > 4 μmol/g | 2 |
| 0.8-4 μmol/g | 1 |
| < 0.8 μmol/g | -1 |
| Rhodamine positive granules2 | 1 |
| Urinary copper excretion3 | |
| Normal | 0 |
| 1-2 times ULN | 1 |
| > 2 times ULN | 2 |
| 5 times ULN after penicillamine | 2 |
| Mutation analysis detected | |
| Both chromosomes | 4 |
| One chromosome | 1 |
| No mutations | 0 |
| Total Leipzig score | |
| Score | Evaluation |
| ≥ 4 | Diagnosis established |
| 3 | Diagnosis possible |
| ≤ 2 | Diagnosis very unlikely |
In the absence of cholestasis.
If no quantitative liver copper available.
In the absence of acute hepatitis. MRI: Magnetic resonance imaging; ULN: Upper limit of normal.
TREATMENT
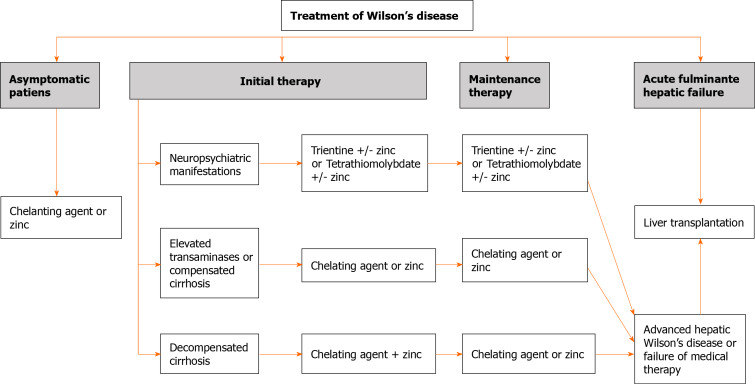
Lifelong treatment is necessary even in asymptomatic patients. There are several treatments for WD, including DPA, trientine, and zinc salts. Figure 1 summarizes the therapeutic approach for patients with WD. Once treatment is indicated for WD, it should be monitored in terms of efficacy (including adherence to treatment) and side effects. Briefly, urinary copper excretion should be assessed every two weeks within the first 4-6 wk and every 2-3 mo during the next 6-12 mo[10,54]. The objectives of copper excretion, according to the drug, are described in Table 4. Similarly, side effects of treatment should also be monitored using blood tests and the liver profile, as well as copper and serum ceruloplasmin[13].
Figure 1.
Therapeutic approach for Wilson’s disease.
Table 4.
Monitoring urinary copper excretion in the treatment of Wilson’s disease
|
Treatment
|
Initial treatment
|
Maintenance treatment
|
Undertreatment or non-compliance
|
Overtreatment or non-compliance
|
| D penicillamine | > 500 μg/24 h | 200-500 μg/24 h | > 500 μg/24 h | < 200 μg/24 h |
| Trientine | > 500 μg/24 h | 200-500 μg/24 h | > 500 μg/24 h | < 100 μg/24 h |
| Zinc | > 100-500 μg/24 h | < 75 μg/24 h | > 15 μg/24 h | < 5 μg/24 h |
DPA
DPA is the first-line drug for WD, and its mechanism involves chelation of circulating copper which will subsequently be excreted in the urine. DPA reduces copper's affinity for proteins by facilitating the removal of copper from tissues, and it induces the synthesis of metallothionein in the liver, a cysteine-rich protein with a high affinity for metal ions. It is metabolized in the liver and is mostly excreted in the urine.
DPA is administered orally, and its absorption is 40%-70% of the administered dose. The dose in adults is 750-1500 mg, and in children is 20 mg/kg/d, given in 2 or 3 divided doses in both cases. DPA should not be taken with food, antacids, or iron supplements because they decrease its absorption. Notably, pyridoxine supplementation should be recommended during treatment with DPA[7].
Up to 90% of patients under DPA therapy have hepatic improvements. However, the efficacy of DPA in neurologic WD is less satisfactory, with an improvement rate of 55%[55]. On the other hand, DPA has numerous adverse reactions; many of them can be severe (Table 5). In those situations, DPA should be discontinued and replaced with another drug. One of the most concerning scenarios is the severe and irreversible neurological worsening at the start of treatment, which can occur in 10%-50% of patients with previous neurological symptoms[56].
Table 5.
Adverse effects of medical therapy used in the treatment of Wilson’s disease
|
Medication
|
Side effects
|
| D penicillamine | Early (1-3 wk): |
| Fever, cutaneous eruptions, myelosuppression, lymphadenopathy, proteinuria | |
| Late: (> 3 wk-yr) | |
| Renal: Nephrotoxicity, nephrotic syndrome | |
| Lungs: Goodpasture syndrome | |
| Bone marrow: Aplasia | |
| Eye: Optic neuritis, retinitis | |
| Skin: Pemphigus, pemphigoid lesions, aphthous stomatitis, hair loss | |
| Autoimmunity: Lupus erythematosus, myasthenia gravis, polymyositis, immunoglobulin A depression | |
| Dose-dependent: | |
| Pyridoxine deficiency | |
| Mammary hypertrophy | |
| Skin: Elastosis serpiginosa, lichen planus, progeria-like skin changes | |
| Neurological deterioration (10%-50%) | |
| Trientine | Few side effects: |
| Bone marrow depression | |
| Sideroblastic anemia | |
| Hemorrhagic gastritis, loss of taste, and skin rash | |
| Neurological deterioration is less common | |
| Zinc | Very few side effects: |
| Gastric irritation | |
| Elevation of serum amylase and lipase | |
| Bone marrow depression | |
| Neurological deterioration is very uncommon | |
| Tetrathiomolybdate | Few side effects: |
| Bone marrow suppression | |
| Increased serum aminotransferase levels | |
| Anemia | |
| No neurological deterioration |
Although neurological worsening typically occurs with DPA treatment, it has also been demonstrated with trientine and to a lesser extent with zinc salts[16,57]. Free copper induces oxidative stress which damages brain tissue. Consequently, the chelating agent should be started at a low dose (125 mg/d) and should be increased every 3-4 d.
Trientine
Trientine or triethylenetetramine dihydrochloride is a chelating agent with a similar mechanism of action to DPA. The efficacy of trientine is similar to DPA. It forms a complex with four nitrogen atoms and copper to be excreted in the urine. It is administered orally, and is poorly absorbed from the gastrointestinal tract. The usual dose is 900 to 2700 mg/d for the initial chelation phase and 750 to 1500 mg/d for the maintenance phase in adults, while 20 mg/kg/d is recommended in children (always divided into two or three doses a day). Similar to DPA, trientine should also be administered separately from food and other drugs. Recent studies propose administering a single daily dose of 15 mg/kg, which would significantly improve adherence to treatment[58]. A particular challenge in trientine treatment is its instability as it must be kept cold (2ºC-8ºC). On the other hand, trientine is a well-tolerated chelating agent that decreases the discontinuation rate up to 4 times compared to DPA, but higher rates of neurological deterioration have been observed than with PDA therapy[55] (Table 5).
Zinc salts
Zinc induces metallothionein synthesis in enterocytes, binding to copper and preventing its absorption into the portal circulation. It is then excreted in feces due to the natural flaking of enterocytes. Zinc also induces metallothionein synthesis in hepatocytes by neutralizing copper in the liver[59,60]. The recommended dose is 150 mg/d, divided into three doses, while 75 mg is adequate for children lower than 50 kg, at least 30 min before meals. In combination with some chelating agents, zinc should be administered separately to avoid neutralization of salts. Evidence shows that zinc salts have few side effects, with gastric irritation being the most common side effect. Zinc salts are not recommended as the initial treatment, particularly in acute liver failure. Therefore, it should be used as first-line therapy only in asymptomatic patients or as maintenance treatment after initiation with chelating agents[61,62].
New treatment options
Trientine tetrahydrochloride is a new drug that is being studied in clinical trials. Compared with conventional trientine, it is stable at normal temperature. On the other hand, tetrathiomolybdate ammonium (TTM), a potent decoppering drug, reduces intestinal absorption of copper and forms a tripartite complex with proteins and copper that is subsequently excreted in bile. In contrast to chelating agents, TTM is not associated with neurological deterioration; thus, it can be used in the neurological phenotype of WD[63]. However, it has been associated with other side effects such as myelosuppression, anemia, and elevation of transaminases. Notably, the ammonium salt of TTM is unstable, although a new complex (Bis-choline TTM) is being developed to solve this issue[64]. Finally, methanobactins are a novel approach that is being investigated with positive results in WD treatment. They can remove copper from the mitochondria, avoiding cell toxicity and acute liver failure[65].
Is dietary copper restriction necessary?
As excessive accumulation of copper causes WD, it has been proposed that copper should be restricted in the diet. Significantly, foods to avoid are chocolate, fruits, nuts, mushrooms, liver, and seafood. Both AASLD and EASL guidelines recommend avoiding the intake of high-concentration copper foods or water, particularly within the first year of diagnosis[7,13]. Nevertheless, copper absorption depends on the content of copper in the diet, showing a self-regulatory mechanism. In fact, diets with a high copper concentration result in lower absorption by enterocytes and a higher copper excretion[66]. Thus, copper-rich foods should be consumed to generate excessive copper intake.
LT
LT has a particularly good survival rate in the WD setting[67]. It is indicated mainly in two situations: acute liver failure and end-stage liver disease. WD has a particular score (King's score) that should be used to decide on LT in the setting of acute liver failure, as an index greater than 11 is associated with a high risk of death without LT[68,69]. LT provides functionality for hepatic ATP7B, resulting in normalization of copper metabolism and removal; consequently, chelation therapy may be discontinued after LT. Although LT is controversial as a treatment for the neurological phenotype of WD, an improvement in neurological involvement has been documented[70,71].
Treatment in special situations: Pregnancy
Treatment should not be discontinued in pregnant patients as the risk is higher than with maintenance therapy, with acute liver failure cases described in patients after withdrawal of treatment[72]. Although DPA has teratogenic potential, a clear increase in risk has not been observed in patients with this treatment, similar to trientine and zinc salts[73,74]. On the other hand, copper deficiency could have a teratogenic effect, so it is advised to reduce chelating therapy by 25%-50% during pregnancy.
PROGNOSIS
In the absence of adequate treatment, the prognosis of WD is fatal[7], but with treatment, this entity has an excellent prognosis. However, we should consider that severe neurological alterations may not be improved, although most patients show significantly improved neurological involvement. Similarly, psychiatric manifestations also improve and can even disappear. On the other hand, patients with cirrhosis often remain compensated and do not have cirrhosis complications, although patients with WD and liver cirrosis should be screened for HCC[54].
FUTURE TREATMENT APPROACHES FOR WD
To date, treatments for WD are based on removing excess copper from the body or LT. Currently, many clinical trials are investigating new treatments with higher efficacy and tolerance, but only a few studies have focused on copper metabolism restoration.
Liver-targeted gene therapy represents an attractive treatment option for many liver conditions[75,76]. Recently, Murillo et al[77] demonstrated that the use of recombinant adeno-associated viral vector (rAAV8), containing complementary DNA encoding copper transporting ATPase2, normalized soluble haloceruloplasmin, and hepatic parenchymal copper levels for more than six months after a single administration, in an animal model[77]. Related to these results, a phase I/II study in sixteen adult WD patients will start in 2021 (clinicalgov. Identifier: NCT04537377), where a single intravenous dose of a rAAV liver tropic capsid containing a single-stranded DNA genome carrying a shortened version of the ATP7B gene will be used.
The regenerative medicine field has progressed in the past two decades. The role of hepatocytes in liver repair is well known. In fact, hepatocyte transplantation has been proposed as an alternative approach to LT, but has some disadvantages such as weak viability in cell culture, the complexity of hepatocyte source, and the vulnerability to cryopreservation[78]. In this sense, stem-cell therapy has been shown to be a potential therapeutic approach in several liver diseases[79,80]. The differentiation potential of mesenchymal cells into hepatocytes has been demonstrated in several studies[81,82]. Indeed, mesenchymal cells can be easily isolated from visceral fat or bone marrow, expanded without losing their differentiation potential, and can migrate to injured areas[83]. The potential to ameliorate liver injury in preclinical and clinical studies has been previously described[84,85]. Recently, induced Pluripotent Stem Cells (iPSCs) have dominated the field of regenerative medicine. These cells have been isolated from patients with different liver diseases showing specific genotypes[86,87]. IPSCs can be isolated by non-invasive methods[88], providing a hepatocyte source for genetic disorders, protein dysfunction, and subsequent cellular defects responsible for specific diseases. A previous study described the generation of iPSCs from WD donor fibroblasts (skin samples) that bear the R778L mutation in the ATP7B gene and their differentiation into hepatocyte-like cells with defective copper transport[89]. They reported gene correction using a lentiviral vector. In the future, hepatocyte-like cells from similarly genetically corrected iPSCs could be an option for autologous transplantation in WD patients. In summary, the expanding tools of gene editing and cell therapy with promising results in other monogenic liver diseases provide a new approach in WD, which could improve the quality of life of these patients by restoring copper metabolism.
CONCLUSION
The knowledge on WD is increasing. The diagnosis of this entity is based on clinical features, biochemical parameters and genetic testing, although new biomarkers are on the horizon. The development of new and effective treatments, including gene therapy, is promising for the future treatment of this disease.
Footnotes
Conflict-of-interest statement: Authors declare no conflict of interests for this article.
Manuscript source: Invited manuscript
Peer-review started: January 24, 2021
First decision: March 8, 2021
Article in press: May 8, 2021
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Spain
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B, B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: El-Shabrawi MH, Janicko M S-Editor: Fan JR L-Editor: Webster JR P-Editor: Wang LL
Contributor Information
Ana Lucena-Valera, Department of Gastroenterology, Hospital Universitario Virgen del Rocio, Sevilla 41013, Spain.
Domingo Perez-Palacios, Department of Gastroenterology, Hospital Universitario Virgen del Rocio, Sevilla 41013, Spain.
Rocio Muñoz-Hernandez, SeLiver Group, Instituto de Biomedicina de Sevilla (IBiS), Hospital Universitario Virgen del Rocío/CSIC/Universidad de Sevilla, Sevilla, España.
Manuel Romero-Gómez, Department of Unit of Digestive Diseases, Hospital Universitario Virgen del Rocio, Sevilla 41014, Spain.
Javier Ampuero, Department of Unit of Digestive Diseases, Hospital Universitario Virgen del Rocio, Sevilla 41014, Spain. jampuero-ibis@us.es.
References
- 1.Liu J, Luan J, Zhou X, Cui Y, Han J. Epidemiology, diagnosis, and treatment of Wilson's disease. Intractable Rare Dis Res. 2017;6:249–255. doi: 10.5582/irdr.2017.01057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Poujois A, Woimant F. Wilson's disease: A 2017 update. Clin Res Hepatol Gastroenterol. 2018;42:512–520. doi: 10.1016/j.clinre.2018.03.007. [DOI] [PubMed] [Google Scholar]
- 3.Camarata MA, Gottfried M, Rule JA, Ala A, Lee WM, Todd Stravitz R, Schilsky ML. Outcomes of Acute Liver Injury in Adults Due to Wilson's Disease: Is Survival Without Transplant Possible? Liver Transpl. 2020;26:330–336. doi: 10.1002/lt.25703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bennett J, Hahn SH. Clinical molecular diagnosis of Wilson disease. Semin Liver Dis. 2011;31:233–238. doi: 10.1055/s-0031-1286054. [DOI] [PubMed] [Google Scholar]
- 5.Chang IJ, Hahn SH. The genetics of Wilson disease. 1st ed. Elsevier . 2017 doi: 10.1016/B978-0-444-63625-6.00003-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.HGMD The Human Gene Mutation Database. [cited 10 January 2021]. Available from: http://www.hgmd.cf.ac.uk/ac/gene.php?gene=ATP7B .
- 7.European Association for Study of Liver. EASL Clinical Practice Guidelines: Wilson's disease. J Hepatol. 2012;56:671–685. doi: 10.1016/j.jhep.2011.11.007. [DOI] [PubMed] [Google Scholar]
- 8.Mulligan C, Bronstein JM. Wilson Disease: An Overview and Approach to Management. Neurol Clin. 2020;38:417–432. doi: 10.1016/j.ncl.2020.01.005. [DOI] [PubMed] [Google Scholar]
- 9.Stättermayer AF, Entenmann A, Gschwantler M, Zoller H, Hofer H, Ferenci P. The dilemma to diagnose Wilson disease by genetic testing alone. Eur J Clin Invest. 2019;49:e13147. doi: 10.1111/eci.13147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schilsky ML. Wilson Disease: Diagnosis, Treatment, and Follow-up. Clin Liver Dis. 2017;21:755–767. doi: 10.1016/j.cld.2017.06.011. [DOI] [PubMed] [Google Scholar]
- 11.Medici V, Shibata NM, Kharbanda KK, Islam MS, Keen CL, Kim K, Tillman B, French SW, Halsted CH, LaSalle JM. Maternal choline modifies fetal liver copper, gene expression, DNA methylation, and neonatal growth in the tx-j mouse model of Wilson disease. Epigenetics. 2014;9:286–296. doi: 10.4161/epi.27110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li H, Tao R, Liu L, Shang S. Population screening and diagnostic strategies in screening family members of Wilson's disease patients. Ann Transl Med. 2019;7:S59. doi: 10.21037/atm.2019.03.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Roberts EA, Schilsky ML American Association for Study of Liver Diseases (AASLD) Diagnosis and treatment of Wilson disease: an update. Hepatology. 2008;47:2089–2111. doi: 10.1002/hep.22261. [DOI] [PubMed] [Google Scholar]
- 14.Shribman S, Warner TT, Dooley JS. Clinical presentations of Wilson disease. Ann Transl Med. 2019;7:S60. doi: 10.21037/atm.2019.04.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nagral A, Sarma MS, Matthai J, Kukkle PL, Devarbhavi H, Sinha S, Alam S, Bavdekar A, Dhiman RK, Eapen CE, Goyal V, Mohan N, Kandadai RM, Sathiyasekaran M, Poddar U, Sibal A, Sankaranarayanan S, Srivastava A, Thapa BR, Wadia PM, Yachha SK, Dhawan A. Wilson's Disease: Clinical Practice Guidelines of the Indian National Association for Study of the Liver, the Indian Society of Pediatric Gastroenterology, Hepatology and Nutrition, and the Movement Disorders Society of India. J Clin Exp Hepatol. 2019;9:74–98. doi: 10.1016/j.jceh.2018.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Merle U, Schaefer M, Ferenci P, Stremmel W. Clinical presentation, diagnosis and long-term outcome of Wilson's disease: a cohort study. Gut. 2007;56:115–120. doi: 10.1136/gut.2005.087262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ferenci P, Stremmel W, Członkowska A, Szalay F, Viveiros A, Stättermayer AF, Bruha R, Houwen R, Pop TL, Stauber R, Gschwantler M, Pfeiffenberger J, Yurdaydin C, Aigner E, Steindl-Munda P, Dienes HP, Zoller H, Weiss KH. Age and Sex but Not ATP7B Genotype Effectively Influence the Clinical Phenotype of Wilson Disease. Hepatology. 2019;69:1464–1476. doi: 10.1002/hep.30280. [DOI] [PubMed] [Google Scholar]
- 18.González Torre JA, Cruz-Gómez ÁJ, Belenguer A, Sanchis-Segura C, Ávila C, Forn C. Hippocampal dysfunction is associated with memory impairment in multiple sclerosis: A volumetric and functional connectivity study. Mult Scler. 2017;23:1854–1863. doi: 10.1177/1352458516688349. [DOI] [PubMed] [Google Scholar]
- 19.Svetel M, Potrebić A, Pekmezović T, Tomić A, Kresojević N, Jesić R, Dragasević N, Kostić VS. Neuropsychiatric aspects of treated Wilson's disease. Parkinsonism Relat Disord. 2009;15:772–775. doi: 10.1016/j.parkreldis.2009.01.010. [DOI] [PubMed] [Google Scholar]
- 20.Sridhar MS, Pineda R. Anterior segment optical coherence tomography to look for Kayser-Fleischer rings. Pract Neurol. 2017;17:222–223. doi: 10.1136/practneurol-2017-001605. [DOI] [PubMed] [Google Scholar]
- 21.Nagral A, Jhaveri A, Nalawade S, Momaya N, Chakkarwar V, Malde P. Kayser-Fleischer rings or bile pigment rings? Indian J Gastroenterol. 2015;34:410–412. doi: 10.1007/s12664-015-0603-2. [DOI] [PubMed] [Google Scholar]
- 22.Golding DN, Walshe JM. Arthropathy of Wilson's disease. Study of clinical and radiological features in 32 patients. Ann Rheum Dis. 1977;36:99–111. doi: 10.1136/ard.36.2.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chu CC, Huang CC, Chu NS. Recurrent hypokalemic muscle weakness as an initial manifestation of Wilson's disease. Nephron. 1996;73:477–479. doi: 10.1159/000189114. [DOI] [PubMed] [Google Scholar]
- 24.Hlubocká Z, Marecek Z, Linhart A, Kejková E, Pospísilová L, Martásek P, Aschermann M. Cardiac involvement in Wilson disease. J Inherit Metab Dis. 2002;25:269–277. doi: 10.1023/a:1016546223327. [DOI] [PubMed] [Google Scholar]
- 25.Nakada SY, Brown MR, Rabinowitz R. Wilson's disease presenting as symptomatic urolithiasis: a case report and review of the literature. J Urol. 1994;152:978–979. doi: 10.1016/s0022-5347(17)32635-6. [DOI] [PubMed] [Google Scholar]
- 26.Weizman Z, Picard E, Barki Y, Moses S. Wilson's disease associated with pancreatitis. J Pediatr Gastroenterol Nutr. 1988;7:931–933. doi: 10.1097/00005176-198811000-00024. [DOI] [PubMed] [Google Scholar]
- 27.Carpenter TO, Carnes DL Jr, Anast CS. Hypoparathyroidism in Wilson's disease. N Engl J Med. 1983;309:873–877. doi: 10.1056/NEJM198310133091501. [DOI] [PubMed] [Google Scholar]
- 28.Klee JG. Undiagnosed Wilson's disease as cause of unexplained miscarriage. Lancet. 1979;2:423. doi: 10.1016/s0140-6736(79)90440-9. [DOI] [PubMed] [Google Scholar]
- 29.Tarnacka B, Rodo M, Cichy S, Członkowska A. Procreation ability in Wilson's disease. Acta Neurol Scand. 2000;101:395–398. doi: 10.1034/j.1600-0404.2000.90140a.x. [DOI] [PubMed] [Google Scholar]
- 30.Guindi M. Wilson disease. Semin Diagn Pathol. 2019;36:415–422. doi: 10.1053/j.semdp.2019.07.008. [DOI] [PubMed] [Google Scholar]
- 31.Macintyre G, Gutfreund KS, Martin WR, Camicioli R, Cox DW. Value of an enzymatic assay for the determination of serum ceruloplasmin. J Lab Clin Med. 2004;144:294–301. doi: 10.1016/j.lab.2004.08.005. [DOI] [PubMed] [Google Scholar]
- 32.Członkowska A, Litwin T, Dusek P, Ferenci P, Lutsenko S, Medici V, Rybakowski JK, Weiss KH, Schilsky ML. Wilson disease. Nat Rev Dis Primers. 2018;4:21. doi: 10.1038/s41572-018-0018-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Harris ZL, Klomp LW, Gitlin JD. Aceruloplasminemia: an inherited neurodegenerative disease with impairment of iron homeostasis. Am J Clin Nutr. 1998;67:972S–977S. doi: 10.1093/ajcn/67.5.972S. [DOI] [PubMed] [Google Scholar]
- 34.Poujois A, Woimant F. Biochemical Markers. In: Wilson Disease. Elsevier. 2019:115–124. [Google Scholar]
- 35.Bruguera M, Abraldes JG. [Common problems in the diagnosis and treatment of Wilson's disease] Gastroenterol Hepatol. 2013;36:316–325. doi: 10.1016/j.gastrohep.2012.10.003. [DOI] [PubMed] [Google Scholar]
- 36.Gross JB Jr, Ludwig J, Wiesner RH, McCall JT, LaRusso NF. Abnormalities in tests of copper metabolism in primary sclerosing cholangitis. Gastroenterology. 1985;89:272–278. doi: 10.1016/0016-5085(85)90326-9. [DOI] [PubMed] [Google Scholar]
- 37.Duncan A, Yacoubian C, Beetham R, Catchpole A, Bullock D. The role of calculated non-caeruloplasmin-bound copper in Wilson's disease. Ann Clin Biochem. 2017;54:649–654. doi: 10.1177/0004563216676843. [DOI] [PubMed] [Google Scholar]
- 38.Woimant F, Djebrani-Oussedik N, Poujois A. New tools for Wilson's disease diagnosis: exchangeable copper fraction. Ann Transl Med. 2019;7:S70. doi: 10.21037/atm.2019.03.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Poujois A, Trocello JM, Djebrani-Oussedik N, Poupon J, Collet C, Girardot-Tinant N, Sobesky R, Habès D, Debray D, Vanlemmens C, Fluchère F, Ory-Magne F, Labreuche J, Preda C, Woimant F. Exchangeable copper: a reflection of the neurological severity in Wilson's disease. Eur J Neurol. 2017;24:154–160. doi: 10.1111/ene.13171. [DOI] [PubMed] [Google Scholar]
- 40.El Balkhi S, Trocello JM, Poupon J, Chappuis P, Massicot F, Girardot-Tinant N, Woimant F. Relative exchangeable copper: a new highly sensitive and highly specific biomarker for Wilson's disease diagnosis. Clin Chim Acta. 2011;412:2254–2260. doi: 10.1016/j.cca.2011.08.019. [DOI] [PubMed] [Google Scholar]
- 41.Schmitt F, Podevin G, Poupon J, Roux J, Legras P, Trocello JM, Woimant F, Laprévote O, Nguyen TH, El Balkhi S. Evolution of exchangeable copper and relative exchangeable copper through the course of Wilson's disease in the Long Evans Cinnamon rat. PLoS One. 2013;8:e82323. doi: 10.1371/journal.pone.0082323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Trocello JM, El Balkhi S, Woimant F, Girardot-Tinant N, Chappuis P, Lloyd C, Poupon J. Relative exchangeable copper: a promising tool for family screening in Wilson disease. Mov Disord. 2014;29:558–562. doi: 10.1002/mds.25763. [DOI] [PubMed] [Google Scholar]
- 43.Steindl P, Ferenci P, Dienes HP, Grimm G, Pabinger I, Madl C, Maier-Dobersberger T, Herneth A, Dragosics B, Meryn S, Knoflach P, Granditsch G, Gangl A. Wilson's disease in patients presenting with liver disease: a diagnostic challenge. Gastroenterology. 1997;113:212–218. doi: 10.1016/s0016-5085(97)70097-0. [DOI] [PubMed] [Google Scholar]
- 44.Martins da Costa C, Baldwin D, Portmann B, Lolin Y, Mowat AP, Mieli-Vergani G. Value of urinary copper excretion after penicillamine challenge in the diagnosis of Wilson's disease. Hepatology. 1992;15:609–615. doi: 10.1002/hep.1840150410. [DOI] [PubMed] [Google Scholar]
- 45.Frommer DJ. Urinary copper excretion and hepatic copper concentrations in liver disease. Digestion. 1981;21:169–178. doi: 10.1159/000198559. [DOI] [PubMed] [Google Scholar]
- 46.Nemolato S, Serra S, Saccani S, Faa G. Deparaffination time: a crucial point in histochemical detection of tissue copper. Eur J Histochem. 2008;52:175–178. doi: 10.4081/1209. [DOI] [PubMed] [Google Scholar]
- 47.Tanner MS. Indian childhood cirrhosis and tyrolean childhood cirrhosis: Disorders of a copper transport gene? In: Advances in Experimental Medicine and Biology. Kluwer Academic/Plenum Publishers, 1999: 127–137. [PubMed] [Google Scholar]
- 48.Dusek P, Litwin T, Czlonkowska A. Wilson disease and other neurodegenerations with metal accumulations. Neurol Clin. 2015;33:175–204. doi: 10.1016/j.ncl.2014.09.006. [DOI] [PubMed] [Google Scholar]
- 49.Prashanth LK, Sinha S, Taly AB, Vasudev MK. Do MRI features distinguish Wilson's disease from other early onset extrapyramidal disorders? Mov Disord. 2010;25:672–678. doi: 10.1002/mds.22689. [DOI] [PubMed] [Google Scholar]
- 50.Durand F. Wilson's disease: an old disease keeps its old secrets. Eur J Gastroenterol Hepatol. 2007;19:97–99. doi: 10.1097/MEG.0b013e32800fef34. [DOI] [PubMed] [Google Scholar]
- 51.Ferenci P, Caca K, Loudianos G, Mieli-Vergani G, Tanner S, Sternlieb I, Schilsky M, Cox D, Berr F. Diagnosis and phenotypic classification of Wilson disease. Liver Int. 2003;23:139–142. doi: 10.1034/j.1600-0676.2003.00824.x. [DOI] [PubMed] [Google Scholar]
- 52.Anheim M, Chamouard P, Rudolf G, Ellero B, Vercueil L, Goichot B, Marescaux C, Tranchant C. Unexpected combination of inherited chorea-acanthocytosis with MDR3 (ABCB4) defect mimicking Wilson's disease. Clin Genet. 2010;78:294–295. doi: 10.1111/j.1399-0004.2010.01386.x. [DOI] [PubMed] [Google Scholar]
- 53.Ramraj R, Finegold MJ, Karpen SJ. Progressive familial intrahepatic cholestasis type 3: overlapping presentation with Wilson disease. Clin Pediatr (Phila) 2012;51:689–691. doi: 10.1177/0009922812451076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kathawala M, Hirschfield GM. Insights into the management of Wilson's disease. Therap Adv Gastroenterol. 2017;10:889–905. doi: 10.1177/1756283X17731520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Weiss KH, Thurik F, Gotthardt DN, Schäfer M, Teufel U, Wiegand F, Merle U, Ferenci-Foerster D, Maieron A, Stauber R, Zoller H, Schmidt HH, Reuner U, Hefter H, Trocello JM, Houwen RH, Ferenci P, Stremmel W EUROWILSON Consortium. Efficacy and safety of oral chelators in treatment of patients with Wilson disease. Clin Gastroenterol Hepatol 2013; 11: 1028-35. :e1–2. doi: 10.1016/j.cgh.2013.03.012. [DOI] [PubMed] [Google Scholar]
- 56.Brewer GJ. Neurologically presenting Wilson's disease: epidemiology, pathophysiology and treatment. CNS Drugs. 2005;19:185–192. doi: 10.2165/00023210-200519030-00001. [DOI] [PubMed] [Google Scholar]
- 57.Litwin T, Dzieżyc K, Karliński M, Chabik G, Czepiel W, Członkowska A. Early neurological worsening in patients with Wilson's disease. J Neurol Sci. 2015;355:162–167. doi: 10.1016/j.jns.2015.06.010. [DOI] [PubMed] [Google Scholar]
- 58.Ala A, Aliu E, Schilsky ML. Prospective pilot study of a single daily dosage of trientine for the treatment of Wilson disease. Dig Dis Sci. 2015;60:1433–1439. doi: 10.1007/s10620-014-3495-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hoogenraad TU, Koevoet R, de Ruyter Korver EG. Oral zinc sulphate as long-term treatment in Wilson's disease (hepatolenticular degeneration) Eur Neurol. 1979;18:205–211. doi: 10.1159/000115077. [DOI] [PubMed] [Google Scholar]
- 60.den Hamer CJA, Hoogeraad TU, Klompjan ERK. Persistence of the antagonistic influence of zinc on copper absorption after cessation of zinc supplementation for more than five days. Biol Trace Elem Res . 1984;1:99–106. [Google Scholar]
- 61.Weiss KH, Gotthardt DN, Klemm D, Merle U, Ferenci-Foerster D, Schaefer M, Ferenci P, Stremmel W. Zinc monotherapy is not as effective as chelating agents in treatment of Wilson disease. Gastroenterology 2011; 140: 1189-1198. :e1. doi: 10.1053/j.gastro.2010.12.034. [DOI] [PubMed] [Google Scholar]
- 62.Appenzeller-Herzog C, Mathes T, Heeres MLS, Weiss KH, Houwen RHJ, Ewald H. Comparative effectiveness of common therapies for Wilson disease: A systematic review and meta-analysis of controlled studies. Liver Int. 2019;39:2136–2152. doi: 10.1111/liv.14179. [DOI] [PubMed] [Google Scholar]
- 63.Brewer GJ, Askari F, Lorincz MT, Carlson M, Schilsky M, Kluin KJ, Hedera P, Moretti P, Fink JK, Tankanow R, Dick RB, Sitterly J. Treatment of Wilson disease with ammonium tetrathiomolybdate: IV. Comparison of tetrathiomolybdate and trientine in a double-blind study of treatment of the neurologic presentation of Wilson disease. Arch Neurol. 2006;63:521–527. doi: 10.1001/archneur.63.4.521. [DOI] [PubMed] [Google Scholar]
- 64.Weiss KH, Askari FK, Czlonkowska A, Ferenci P, Bronstein JM, Bega D, Ala A, Nicholl D, Flint S, Olsson L, Plitz T, Bjartmar C, Schilsky ML. Bis-choline tetrathiomolybdate in patients with Wilson's disease: an open-label, multicentre, phase 2 study. Lancet Gastroenterol Hepatol. 2017;2:869–876. doi: 10.1016/S2468-1253(17)30293-5. [DOI] [PubMed] [Google Scholar]
- 65.Lichtmannegger J, Leitzinger C, Wimmer R, Schmitt S, Schulz S, Kabiri Y, Eberhagen C, Rieder T, Janik D, Neff F, Straub BK, Schirmacher P, DiSpirito AA, Bandow N, Baral BS, Flatley A, Kremmer E, Denk G, Reiter FP, Hohenester S, Eckardt-Schupp F, Dencher NA, Adamski J, Sauer V, Niemietz C, Schmidt HH, Merle U, Gotthardt DN, Kroemer G, Weiss KH, Zischka H. Methanobactin reverses acute liver failure in a rat model of Wilson disease. J Clin Invest. 2016;126:2721–2735. doi: 10.1172/JCI85226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Turnlund JR, Keyes WR, Kim SK, Domek JM. Long-term high copper intake: effects on copper absorption, retention, and homeostasis in men. Am J Clin Nutr. 2005;81:822–828. doi: 10.1093/ajcn/81.4.822. [DOI] [PubMed] [Google Scholar]
- 67.Guillaud O, Dumortier J, Sobesky R, Debray D, Wolf P, Vanlemmens C, Durand F, Calmus Y, Duvoux C, Dharancy S, Kamar N, Boudjema K, Bernard PH, Pageaux GP, Salamé E, Gugenheim J, Lachaux A, Habes D, Radenne S, Hardwigsen J, Chazouillères O, Trocello JM, Woimant F, Ichai P, Branchereau S, Soubrane O, Castaing D, Jacquemin E, Samuel D, Duclos-Vallée JC. Long term results of liver transplantation for Wilson's disease: experience in France. J Hepatol. 2014;60:579–589. doi: 10.1016/j.jhep.2013.10.025. [DOI] [PubMed] [Google Scholar]
- 68.Nazer H, Ede RJ, Mowat AP, Williams R. Wilson's disease: clinical presentation and use of prognostic index. Gut. 1986;27:1377–1381. doi: 10.1136/gut.27.11.1377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Dhawan A, Taylor RM, Cheeseman P, De Silva P, Katsiyiannakis L, Mieli-Vergani G. Wilson's disease in children: 37-year experience and revised King's score for liver transplantation. Liver Transpl. 2005;11:441–448. doi: 10.1002/lt.20352. [DOI] [PubMed] [Google Scholar]
- 70.Laurencin C, Brunet AS, Dumortier J, Lion-Francois L, Thobois S, Mabrut JY, Dubois R, Woimant F, Poujois A, Guillaud O, Lachaux A, Broussolle E. Liver Transplantation in Wilson's Disease with Neurological Impairment: Evaluation in 4 Patients. Eur Neurol. 2017;77:5–15. doi: 10.1159/000452658. [DOI] [PubMed] [Google Scholar]
- 71.Poujois A, Sobesky R, Meissner W, de Medeiros E, Vanlemmens C, Brunet AS, Broussolle E, Duclos-Vallée FW. The French experience of liver transplantation for severe neurological forms of Wilson disease [abstract] Mov Disord. 2016;31 Suppl 2 [Google Scholar]
- 72.Shimono N, Ishibashi H, Ikematsu H, Kudo J, Shirahama M, Inaba S, Maeda K, Yamasaki K, Niho Y. Fulminant hepatic failure during perinatal period in a pregnant woman with Wilson's disease. Gastroenterol Jpn. 1991;26:69–73. doi: 10.1007/BF02779512. [DOI] [PubMed] [Google Scholar]
- 73.Brewer GJ, Johnson VD, Dick RD, Hedera P, Fink JK, Kluin KJ. Treatment of Wilson's disease with zinc. XVII: treatment during pregnancy. Hepatology. 2000;31:364–370. doi: 10.1002/hep.510310216. [DOI] [PubMed] [Google Scholar]
- 74.Walshe JM. The management of pregnancy in Wilson's disease treated with trientine. Q J Med. 1986;58:81–87. [PubMed] [Google Scholar]
- 75.Jarrett KE, Lee CM, Yeh YH, Hsu RH, Gupta R, Zhang M, Rodriguez PJ, Lee CS, Gillard BK, Bissig KD, Pownall HJ, Martin JF, Bao G, Lagor WR. Somatic genome editing with CRISPR/Cas9 generates and corrects a metabolic disease. Sci Rep. 2017;7:44624. doi: 10.1038/srep44624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Stephens CJ, Kashentseva E, Everett W, Kaliberova L, Curiel DT. Targeted in vivo knock-in of human alpha-1-antitrypsin cDNA using adenoviral delivery of CRISPR/Cas9. Gene Ther. 2018;25:139–156. doi: 10.1038/s41434-018-0003-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Murillo O, Luqui DM, Gazquez C, Martinez-Espartosa D, Navarro-Blasco I, Monreal JI, Guembe L, Moreno-Cermeño A, Corrales FJ, Prieto J, Hernandez-Alcoceba R, Gonzalez-Aseguinolaza G. Long-term metabolic correction of Wilson's disease in a murine model by gene therapy. J Hepatol. 2016;64:419–426. doi: 10.1016/j.jhep.2015.09.014. [DOI] [PubMed] [Google Scholar]
- 78.Puppi J, Strom SC, Hughes RD, Bansal S, Castell JV, Dagher I, Ellis EC, Nowak G, Ericzon BG, Fox IJ, Gómez-Lechón MJ, Guha C, Gupta S, Mitry RR, Ohashi K, Ott M, Reid LM, Roy-Chowdhury J, Sokal E, Weber A, Dhawan A. Improving the techniques for human hepatocyte transplantation: report from a consensus meeting in London. Cell Transplant. 2012;21:1–10. doi: 10.3727/096368911X566208. [DOI] [PubMed] [Google Scholar]
- 79.Mohamadnejad M, Alimoghaddam K, Bagheri M, Ashrafi M, Abdollahzadeh L, Akhlaghpoor S, Bashtar M, Ghavamzadeh A, Malekzadeh R. Randomized placebo-controlled trial of mesenchymal stem cell transplantation in decompensated cirrhosis. Liver Int. 2013;33:1490–1496. doi: 10.1111/liv.12228. [DOI] [PubMed] [Google Scholar]
- 80.Hay DC, Fletcher J, Payne C, Terrace JD, Gallagher RC, Snoeys J, Black JR, Wojtacha D, Samuel K, Hannoun Z, Pryde A, Filippi C, Currie IS, Forbes SJ, Ross JA, Newsome PN, Iredale JP. Highly efficient differentiation of hESCs to functional hepatic endoderm requires ActivinA and Wnt3a signaling. Proc Natl Acad Sci USA. 2008;105:12301–12306. doi: 10.1073/pnas.0806522105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Shu SN, Wei L, Wang JH, Zhan YT, Chen HS, Wang Y. Hepatic differentiation capability of rat bone marrow-derived mesenchymal stem cells and hematopoietic stem cells. World J Gastroenterol. 2004;10:2818–2822. doi: 10.3748/wjg.v10.i19.2818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Si-Tayeb K, Noto FK, Nagaoka M, Li J, Battle MA, Duris C, North PE, Dalton S, Duncan SA. Highly efficient generation of human hepatocyte-like cells from induced pluripotent stem cells. Hepatology. 2010;51:297–305. doi: 10.1002/hep.23354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Eom YW, Shim KY, Baik SK. Mesenchymal stem cell therapy for liver fibrosis. Korean J Intern Med. 2015;30:580–589. doi: 10.3904/kjim.2015.30.5.580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Abdel Aziz MT, Atta HM, Mahfouz S, Fouad HH, Roshdy NK, Ahmed HH, Rashed LA, Sabry D, Hassouna AA, Hasan NM. Therapeutic potential of bone marrow-derived mesenchymal stem cells on experimental liver fibrosis. Clin Biochem. 2007;40:893–899. doi: 10.1016/j.clinbiochem.2007.04.017. [DOI] [PubMed] [Google Scholar]
- 85.Mohamadnejad M, Vosough M, Moossavi S, Nikfam S, Mardpour S, Akhlaghpoor S, Ashrafi M, Azimian V, Jarughi N, Hosseini SE, Moeininia F, Bagheri M, Sharafkhah M, Aghdami N, Malekzadeh R, Baharvand H. Intraportal Infusion of Bone Marrow Mononuclear or CD133+ Cells in Patients With Decompensated Cirrhosis: A Double-Blind Randomized Controlled Trial. Stem Cells Transl Med. 2016;5:87–94. doi: 10.5966/sctm.2015-0004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Rashid ST, Corbineau S, Hannan N, Marciniak SJ, Miranda E, Alexander G, Huang-Doran I, Griffin J, Ahrlund-Richter L, Skepper J, Semple R, Weber A, Lomas DA, Vallier L. Modeling inherited metabolic disorders of the liver using human induced pluripotent stem cells. J Clin Invest. 2010;120:3127–3136. doi: 10.1172/JCI43122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Sullivan GJ, Hay DC, Park IH, Fletcher J, Hannoun Z, Payne CM, Dalgetty D, Black JR, Ross JA, Samuel K, Wang G, Daley GQ, Lee JH, Church GM, Forbes SJ, Iredale JP, Wilmut I. Generation of functional human hepatic endoderm from human induced pluripotent stem cells. Hepatology. 2010;51:329–335. doi: 10.1002/hep.23335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhou T, Benda C, Dunzinger S, Huang Y, Ho JC, Yang J, Wang Y, Zhang Y, Zhuang Q, Li Y, Bao X, Tse HF, Grillari J, Grillari-Voglauer R, Pei D, Esteban MA. Generation of human induced pluripotent stem cells from urine samples. Nat Protoc. 2012;7:2080–2089. doi: 10.1038/nprot.2012.115. [DOI] [PubMed] [Google Scholar]
- 89.Zhang S, Chen S, Li W, Guo X, Zhao P, Xu J, Chen Y, Pan Q, Liu X, Zychlinski D, Lu H, Tortorella MD, Schambach A, Wang Y, Pei D, Esteban MA. Rescue of ATP7B function in hepatocyte-like cells from Wilson's disease induced pluripotent stem cells using gene therapy or the chaperone drug curcumin. Hum Mol Genet. 2011;20:3176–3187. doi: 10.1093/hmg/ddr223. [DOI] [PubMed] [Google Scholar]