Abstract
Sepsis induced by bacteria or viruses can result in multiorgan dysfunction, which is a major cause of death in intensive care units. Current treatments are only supportive, and there are no treatments that reverse the pathophysiological effects of sepsis. Vitamin C has antioxidant, anti‐inflammatory, anticoagulant and immune modulatory actions, so it is a rational treatment for sepsis. Here, we summarise data that support the use of megadose vitamin C as a treatment for sepsis and COVID‐19. Megadose intravenous sodium ascorbate (150 g per 40 kg over 7 h) dramatically improved the clinical state and cardiovascular, pulmonary, hepatic and renal function and decreased body temperature, in a clinically relevant ovine model of Gram‐negative bacteria‐induced sepsis. In a critically ill COVID‐19 patient, intravenous sodium ascorbate (60 g) restored arterial pressure, improved renal function and increased arterial blood oxygen levels. These findings suggest that megadose vitamin C should be trialled as a treatment for sepsis and COVID‐19.
Keywords: acute kidney injury, COVID‐19, hypotension, hypoxia, inflammation, oxidative stress, sepsis, sodium ascorbate, vitamin C
Abbreviations
- AKI
acute kidney injury
- COVID‐19
Coronovirus disease 2019
- eNOS
endothelial nitric oxide synthase
- RCTs
randomised clinical trials
- SARS‐CoV‐2
Severe acute respiratory syndrome coronavirus 2
- SOFA
sequential organ failure assessment
1. INTRODUCTION
Sepsis is a pathophysiological syndrome characterised by an overwhelming immune response to a bacterial, viral or fungal infection that can lead to multiorgan dysfunction and death (Singer et al., 2016). Sepsis is the leading cause of mortality in intensive care units with an annual global incidence of 49 million cases and 11 million deaths (Rudd et al., 2020). Standard of care treatment for sepsis consists of antibiotics, fluid resuscitation and vasopressors (Rhodes et al., 2017), with continuous renal replacement therapy being increasingly used in critically ill patients (Bellomo et al., 2021). These interventions are mostly aimed towards keeping the patient alive in the expectation that organ function should recover following resolution of the infection. However, patients who recover from severe sepsis frequently exhibit a degree of chronic organ dysfunction. Currently, there are no treatments that reverse sepsis‐induced organ dysfunction.
2. PATHOPHYSIOLOGY OF SEPSIS‐INDUCED CARDIOVASCULAR AND RENAL DYSFUNCTION
The factors causing sepsis‐induced organ dysfunction remain unclear due to the complex pathophysiology of sepsis that changes as the response to the infection progresses. There is evidence that redox homeostasis is disrupted in sepsis resulting in oxidative stress, which together with excessive inflammation is thought to cause mitochondrial, endothelial and microvascular dysfunction, resulting in vasoplegia, inflammation‐mediated tissue injury, tissue hypoxia and multi‐organ dysfunction (Joffre & Hellman, 2021; Lankadeva et al., 2019) (Figure 1).
FIGURE 1.
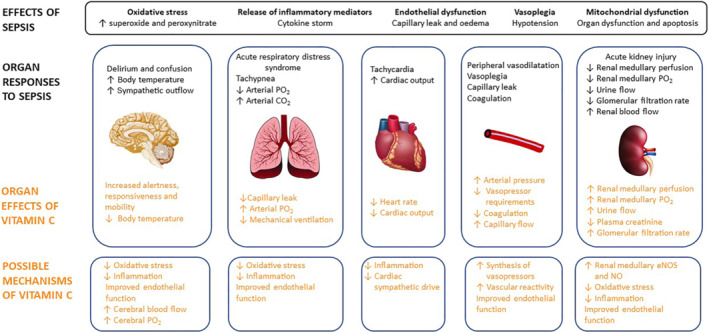
Overview of the systemic and organ specific effects of ovine sepsis, the effects of treatment we have observed with intravenous megadose vitamin C and the possible mechanisms of action that require investigation
Hypotension secondary to peripheral vasodilatation is a hallmark of sepsis that is treated with aggressive fluid resuscitation and vasopressor therapy to restore target mean arterial pressure (Rhodes et al., 2017). Reduced vascular responsiveness to noradrenaline, the primary vasopressor used clinically, is common in sepsis resulting in persistent and sometimes refractory hypotension (Annane et al., 1998), which, in itself, can result in tissue hypoperfusion and hypoxia, mitochondrial dysfunction and multi‐organ failure.
Indeed, tissue hypoperfusion and hypoxia in the renal medulla are critical pathophysiological features of ovine hyperdynamic sepsis that precede the development of acute kidney injury (AKI) by 8 to 12 h (Calzavacca et al., 2015). Renal medullary hypoxia can lead to mitochondrial dysfunction, initiating a progressive loss of renal function culminating in AKI (Lankadeva et al., 2019; Nourbakhsh & Singh, 2014). Therapies that target these sepsis‐induced pathophysiological processes may confer better circulatory management and mitigate AKI.
3. ANTIOXIDANTS AS A THERAPY FOR SEPSIS‐INDUCED ORGAN DYSFUNCTION
In view of the damaging effects of tissue hypoxia and oxidative stress, there has been interest over many decades in the use of antioxidants as a treatment for sepsis. The antioxidant N‐acetylcysteine, which has both antioxidant and anti‐inflammatory properties, showed some promise in experimental studies, but clinical studies have yielded largely disappointing findings (Chertoff, 2018). The antioxidant tempol has been shown to reduce the level of AKI in a porcine model of sepsis (Matejovic et al., 2005), but its effects have not been examined clinically. Recently, however, there has been increasing interest in the effects of vitamin C as a treatment for sepsis.
4. RATIONALE FOR VITAMIN C THERAPY IN SEPSIS
Vitamin C, ascorbic acid, is an essential vitamin in humans that must be obtained in the diet, as it cannot be synthesised because of mutations in the gene for gluconolactone oxidase, the final biosynthetic enzyme in its synthesis. Vitamin C is essential for collagen synthesis, accounting for the symptoms of scurvy caused by its deficiency. Vitamin C also has numerous pleiotropic effects that would be predicted to be of benefit in sepsis, including as an anti‐oxidant, anti‐inflammatory, anticoagulant, immune modulator and stimulant of noradrenaline and vasopressin synthesis (Holford et al., 2020) (Figure 1). Furthermore, critically ill patients have low plasma vitamin C levels (11 vs. 62 μmol·L−1 in healthy subjects) (Schorah et al., 1996), most likely due to increased metabolic turnover and downregulation of cellular sodium‐dependent vitamin C transporters (Subramanian et al., 2018), which is compounded by the inability of humans to synthesise vitamin C. Importantly, intravenous administration is required to produce high plasma levels of vitamin C, as there is a limit on its absorption from the intestine (Padayatty et al., 2004). These observations provided the impetus for clinical trials examining the effects of intravenous vitamin C in sepsis.
5. CONTROVERSIES WITH HIGH‐DOSE VITAMIN C THERAPY IN HUMAN SEPSIS
Single‐centre controlled randomised clinical trials (RCTs) showed that intravenous vitamin C reduced inflammatory biomarkers and reduced sequential organ failure assessment (SOFA) scores (50, 100 and 200 mg·kg‐1·day−1, n = 24) (Fowler et al., 2014) and improved vasopressor sensitivity (2 g four times per day, n = 28) (Zabet et al., 2016). A widely publicised single‐centre before and after study (n = 47), using a combination therapy of vitamin C (1.5 g four times per day) with hydrocortisone and thiamine, reduced organ failure and mortality from 40.4% to 8.5% (Marik et al., 2017). However, subsequent multicentre RCTs, VITAMINS (Fujii et al., 2020), ACTS (Moskowitz et al., 2020) and ATESS (Hwang et al., 2020) that trialled a maximum dose of vitamin C of 6 g·day−1 for up to 10 days with thiamine ± corticosteroid, had no significant benefit above placebo treatment. The CITRIS‐ALI trial, that used 200 mg·kg−1·day−1 (16 g·day−1 in an 80‐kg patient) of intravenous vitamin C for 4 days, found no improvement in the primary end point of change in mean modified SOFA score. However, 28‐day mortality was reduced from 46% to 30% (Fowler et al., 2019).
We hypothesised that the lack of consistent benefit in the clinical trials of vitamin C in sepsis might be due to the use of inadequate doses. In view of these findings, and the fact that very high doses of intravenous vitamin C have been shown to be safe in burns and cancer patients (Yanase et al., 2020), we recently investigated the safety and efficacy of a much larger dose (megadose) of vitamin C in a clinically relevant large animal model of sepsis. Such a preclinical safety study was essential as the very high plasma levels of vitamin C can have pro‐oxidant effects (Chen et al., 2008), which may be detrimental in critical illness.
6. A PRECLINICAL RANDOMISED CONTROLLED TRIAL OF MEGADOSE VITAMIN C IN OVINE SEPSIS
Intravenous administration of a megadose of sodium ascorbate (150 g per ~40 kg, 7 h) in established ovine hyperdynamic sepsis, induced by infusion of live Escherichia coli, caused a remarkable improvement in the clinical state from malaise, lethargy and somnolence to an alert, responsive, mobile state (Lankadeva et al., 2021). MAP was restored to pre‐septic levels with reduced noradrenaline requirements, which decreased to zero in four of five cases. Megadose vitamin C increased arterial blood oxygen levels, indicative of improvements in lung function, restored body temperature from febrile to normal levels and reduced arterial blood lactate indicating improved metabolic function. The treatment also reversed renal medullary hypoperfusion and hypoxia, accompanied by a reversal in septic AKI, as shown by dramatic increases in urine flow and creatinine clearance leading to a normalisation of plasma creatinine levels (Figure 1).
The redistribution of intrarenal perfusion in ovine septic AKI (Calzavacca et al., 2015) is accompanied by reduced gene expression of renal medullary endothelial nitric oxide synthase (eNOS) (Langenberg et al., 2008). It is important to determine if the reversal of renal medullary microcirculatory dysfunction by vitamin C results from its ability to increase eNOS activity and thus nitric oxide bioavailability (Ladurner et al., 2012).
7. TREATMENT OF COVID‐19 WITH VITAMIN C
Critically ill COVID‐19 patients develop an excessive inflammatory response, disseminated intravascular coagulation and multiorgan dysfunction. Immunosuppressive agents such as tocilizumab (a humanised monoclonal antibody against the IL‐6 receptor) (Salama et al., 2021) and dexamethasone (RECOVERY Collaborative Group, 2021) have been shown to be beneficial treatments for such patients. The known actions of vitamin C indicate that it would also be a possible adjunct treatment for COVID‐19. The findings that plasma vitamin C levels are low in COVID‐19 patients (Chiscano‐Camon et al., 2020), and that vitamin C lowers expression of angiotensin converting enzyme 2, the entry point for SARS‐CoV‐2 into cells (Ivanov et al., 2021), further indicate that it may have beneficial actions in COVID‐19. Intravenous vitamin C (1.5–14.0 g) has been investigated in COVID‐19 patients with mild beneficial effects (Jamalimoghadamsiahkali et al., 2021; Zhao et al., 2021), but the effects of megadoses have not been studied.
Following our finding of beneficial effects of megadose vitamin C in ovine sepsis, a critically ill patient with COVID‐19‐induced acute respiratory distress syndrome, hypotension and AKI was treated with intravenous sodium ascorbate (60 g over 7 h) (Lankadeva et al., 2021). As in septic sheep, sodium ascorbate restored arterial pressure in the face of complete withdrawal of noradrenaline. Plasma creatinine decreased from 118 to 84 μmol·L−1, whereas urine flow increased from 10 to 400 ml·h−1. There were decreases in heart rate from 130–105 beats per minute and in blood lactate, from 2.6 to 1.9 mmol·L−1. Arterial blood oxygen levels improved while fractional inspired oxygen was reduced. The patient was subsequently extubated on intensive care Day 15 and discharged from hospital 22 days after megadose vitamin C treatment.
8. CLINICAL TRIALS OF MEGADOSE VITAMIN C
Two pilot placebo controlled RCTs are currently underway examining the effects of intravenous megadose vitamin C treatment (sodium ascorbate, 60 and 120 g) in septic patients across three tertiary hospitals in Australia (ACTRN12620000651987p; NCT04796636).
9. CONCLUSIONS
Our findings demonstrate that intravenous megadose intravenous vitamin C reversed organ dysfunction and improved the clinical state in a clinically relevant ovine model of sepsis. We also demonstrated the safety and benefit of this treatment in one critically ill COVID‐19 patient. It is now critical to complete dose–response studies in which the plasma levels of vitamin C are measured to determine the optimum dosing regimen. Further studies are necessary to determine the mechanisms by which megadose intravenous vitamin C improves the clinical state and reverses multi‐organ dysfunction in sepsis. Such studies are essential to provide the scientific rationale for the design of large double‐blinded multicentre RCTs.
10. NOMENCLATURE OF TARGETS AND LIGANDS
Key protein targets and ligands in this article are hyperlinked to corresponding entries in the IUPHAR/BPS Guide to PHARMACOLOGY (http://www.guidetopharmacology.org) and are permanently archived in the Concise Guide to PHARMACOLOGY 2019/20 (Alexander, Fabbro et al., 2019a, 2019b; Alexander, Kelly et al., 2019).
CONFLICT OF INTEREST
YRL, RB and CNM have a provisional patent on vitamin C use in sepsis (2020901120).
ACKNOWLEDGEMENTS
This study was supported by a grant from the National Health and Medical Research Council of Australia (NHMRC, GNT1188514) and by funding from the Victorian Government Operational Infrastructure Support Grant. YRL was supported by a Future Leader Fellowship from the National Heart Foundation of Australia (NHF, 101853).
May, C. N. , Bellomo, R. , & Lankadeva, Y. R. (2021). Therapeutic potential of megadose vitamin C to reverse organ dysfunction in sepsis and COVID‐19. British Journal of Pharmacology, 178(19), 3864–3868. 10.1111/bph.15579
Funding information National Health and Medical Research Council of Australlia, Grant/Award Number: GNT1188514; National Heart Foundation of Australia, Grant/Award Number: 101853; Victorian Government Operational Infrastructure Support Grant
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request. Some data may not be made available because of privacy or ethical restrictions.
REFERENCES
- Alexander, S. P. H. , Fabbro, D. , Kelly, E. , Mathie, A. , Peters, J. A. , Veale, E. L. , Armstrong, J. F. , Faccenda, E. , Harding, S. D. , Pawson, A. J. , & Sharman, J. L. (2019a). The concise guide to PHARMACOLOGY 2019/20: Catalytic receptors. British Journal of Pharmacology, 176, S247–S296. 10.1111/bph.14751 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexander, S. P. H. , Fabbro, D. , Kelly, E. , Mathie, A. , Peters, J. A. , Veale, E. L. , Armstrong, J. F. , Faccenda, E. , Harding, S. D. , Pawson, A. J. , & Sharman, J. L. (2019b). The concise guide to PHARMACOLOGY 2019/20: Enzymes. British Journal of Pharmacology, 176, S297–S396. 10.1111/bph.14752 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexander, S. P. H. , Kelly, E. , Mathie, A. , Peters, J. A. , Veale, E. L. , Armstrong, J. F. , Faccenda, E. , Harding, S. D. , Pawson, A. J. , Sharman, J. L. , & Southan, C. (2019). The concise guide to PHARMACOLOGY 2019/20: Transporters. British Journal of Pharmacology, 176, S397–S493. 10.1111/bph.14753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Annane, D. , Bellissant, E. , Sebille, V. , Lesieur, O. , Mathieu, B. , Raphael, J. C. , & Gajdos, P. (1998). Impaired pressor sensitivity to noradrenaline in septic shock patients with and without impaired adrenal function reserve. British Journal of Clinical Pharmacology, 46, 589–597. 10.1046/j.1365-2125.1998.00833.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bellomo, R. , Baldwin, I. , Ronco, C. , & Kellum, J. A. (2021). ICU‐based renal replacement therapy. Critical Care Medicine, 49, 406–418. 10.1097/CCM.0000000000004831 [DOI] [PubMed] [Google Scholar]
- Calzavacca, P. , Evans, R. G. , Bailey, M. , Bellomo, R. , & May, C. N. (2015). Cortical and medullary tissue perfusion and oxygenation in experimental septic acute kidney injury. Critical Care Medicine, 43, e431–e439. 10.1097/CCM.0000000000001198 [DOI] [PubMed] [Google Scholar]
- Chen, Q. , Espey, M. G. , Sun, A. Y. , Pooput, C. , Kirk, K. L. , Krishna, M. C. , Khosh, D. B. , Drisko, J. , & Levine, M. (2008). Pharmacologic doses of ascorbate act as a prooxidant and decrease growth of aggressive tumor xenografts in mice. Proceedings of the National Academy of Sciences of the United States of America, 105, 11105–11109. 10.1073/pnas.0804226105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chertoff, J. (2018). N‐Acetylcysteine's role in sepsis and potential benefit in patients with microcirculatory derangements. Journal of Intensive Care Medicine, 33, 87–96. 10.1177/0885066617696850 [DOI] [PubMed] [Google Scholar]
- Chiscano‐Camon, L. , Ruiz‐Rodriguez, J. C. , Ruiz‐Sanmartin, A. , Roca, O. , & Ferrer, R. (2020). Vitamin C levels in patients with SARS‐CoV‐2‐associated acute respiratory distress syndrome. Critical Care, 24, 522. 10.1186/s13054-020-03249-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fowler, A. A. 3rd , Syed, A. A. , Knowlson, S. , Sculthorpe, R. , Farthing, D. , DeWilde, C. , Farthing, C. A. , Larus, T. L. , Martin, E. , Brophy, D. F. , Gupta, S. , & Fisher, B. J. (2014). Phase I safety trial of intravenous ascorbic acid in patients with severe sepsis. Journal of Translational Medicine, 12, 32. 10.1186/1479-5876-12-32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fowler, A. A. 3rd , Truwit, J. D. , Hite, R. D. , Morris, P. E. , DeWilde, C. , Priday, A. , Fisher, B. , Thacker, L. R. 2nd , Natarajan, R. , Brophy, D. F. , Sculthorpe, R. , Nanchal, R. , Syed, A. , Sturgill, J. , Martin, G. S. , Sevransky, J. , Kashiouris, M. , Hamman, S. , Egan, K. F. , … Nanchal, R. (2019). Effect of vitamin C infusion on organ failure and biomarkers of inflammation and vascular injury in patients with sepsis and severe acute respiratory failure: The CITRIS‐ALI randomized clinical trial. JAMA: The Journal of the American Medical Association, 322, 1261–1270. 10.1001/jama.2019.11825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujii, T. , Luethi, N. , Young, P. J. , Frei, D. R. , Eastwood, G. M. , French, C. J. , Deane, A. M. , Shehabi, Y. , Hajjar, L. A. , Oliveira, G. , Udy, A. A. , & for the VITAMINS Trial Investigators . (2020). Effect of vitamin C, hydrocortisone, and thiamine vs hydrocortisone alone on time alive and free of vasopressor support among patients with septic shock: The VITAMINS randomized clinical trial. JAMA: The Journal of the American Medical Association, 323, 423–431. 10.1001/jama.2019.22176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holford, P. , Carr, A. C. , Jovic, T. H. , Ali, S. R. , Whitaker, I. S. , Marik, P. E. , & Smith, A. D. (2020). Vitamin C—An adjunctive therapy for respiratory infection, sepsis and COVID‐19. Nutrients, 12. 10.3390/nu12123760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hwang, S. Y. , Ryoo, S. M. , Park, J. E. , Jo, Y. H. , Jang, D. H. , Suh, G. J. , Kim, T. , Kim, Y. J. , Kim, S. , Cho, H. , & Jo, I. J. (2020). Combination therapy of vitamin C and thiamine for septic shock: A multi‐Centre, double‐blinded randomized, controlled study. Intensive Care Medicine, 46, 2015–2025. 10.1007/s00134-020-06191-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ivanov, V. , Goc, A. , Ivanova, S. , Niedzwiecki, A. , & Rath, M. (2021). Inhibition of ACE2 expression by ascorbic acid alone and its combinations with other natural compounds. Infectious Diseases: Research and Treatment, 14, 1178633721994605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jamalimoghadamsiahkali, S. , Zarezade, B. , Koolaji, S. , SeyedAlinaghi, S. , Zendehdel, A. , Tabarestani, M. , … Ghiasvand, F. (2021). Safety and effectiveness of high‐dose vitamin C in patients with COVID‐19: A randomized open‐label clinical trial. European Journal of Medical Research, 26, 20. 10.1186/s40001-021-00490-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joffre, J. , & Hellman, J. (2021). Oxidative stress and endothelial dysfunction in sepsis and acute inflammation. Antioxidants & Redox Signaling. 10.1089/ars.2021.0027 [DOI] [PubMed] [Google Scholar]
- Ladurner, A. , Schmitt, C. A. , Schachner, D. , Atanasov, A. G. , Werner, E. R. , Dirsch, V. M. , & Heiss, E. H. (2012). Ascorbate stimulates endothelial nitric oxide synthase enzyme activity by rapid modulation of its phosphorylation status. Free Radical Biology & Medicine, 52, 2082–2090. 10.1016/j.freeradbiomed.2012.03.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langenberg, C. , Bagshaw, S. M. , May, C. N. , & Bellomo, R. (2008). The histopathology of septic acute kidney injury: A systematic review. Critical Care, 12, R38. 10.1186/cc6823 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lankadeva, Y. R. , Okazaki, N. , Evans, R. G. , Bellomo, R. , & May, C. N. (2019). Renal medullary hypoxia: A new therapeutic target for septic acute kidney injury? Seminars in Nephrology, 39, 543–553. 10.1016/j.semnephrol.2019.10.004 [DOI] [PubMed] [Google Scholar]
- Lankadeva, Y. R. , Peiris, R. M. , Okazaki, N. , Birchall, I. E. , Trask‐Marino, A. , Dornom, A. , Vale, T. A. , Evans, R. G. , Yanase, F. , Bellomo, R. , & May, C. N. (2021). Reversal of the pathophysiological responses to gram‐negative sepsis by megadose vitamin C. Critical Care Medicine, 49, e179–e190. 10.1097/CCM.0000000000004770 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marik, P. E. , Khangoora, V. , Rivera, R. , Hooper, M. H. , & Catravas, J. (2017). Hydrocortisone, vitamin C, and thiamine for the treatment of severe sepsis and septic shock: A retrospective before‐after study. Chest, 151, 1229–1238. 10.1016/j.chest.2016.11.036 [DOI] [PubMed] [Google Scholar]
- Matejovic, M. , Krouzecky, A. , Martinkova, V. , Rokyta, R. Jr. , Radej, J. , Kralova, H. , Treska, V. , Radermacher, P. , & Novak, I. (2005). Effects of tempol, a free radical scavenger, on long‐term hyperdynamic porcine bacteremia. Critical Care Medicine, 33, 1057–1063. 10.1097/01.CCM.0000162927.94753.63 [DOI] [PubMed] [Google Scholar]
- Moskowitz, A. , Huang, D. T. , Hou, P. C. , Gong, J. , Doshi, P. B. , Grossestreuer, A. V. , Andersen, L. W. , Ngo, L. , Sherwin, R. L. , Berg, K. M. , Chase, M. , & for the ACTS Clinical Trial Investigators . (2020). Effect of ascorbic acid, corticosteroids, and thiamine on organ injury in septic shock: The ACTS randomized clinical trial. JAMA: The Journal of the American Medical Association, 324, 642–650. 10.1001/jama.2020.11946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nourbakhsh, N. , & Singh, P. (2014). Role of renal oxygenation and mitochondrial function in the pathophysiology of acute kidney injury. Nephron. Clinical Practice, 127, 149–152. 10.1159/000363545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Padayatty, S. J. , Sun, H. , Wang, Y. , Riordan, H. D. , Hewitt, S. M. , Katz, A. , Wesley, R. A. , & Levine, M. (2004). Vitamin C pharmacokinetics: Implications for oral and intravenous use. Annals of Internal Medicine, 140, 533–537. 10.7326/0003-4819-140-7-200404060-00010 [DOI] [PubMed] [Google Scholar]
- RECOVERY Collaborative Group . (2021). Dexamethasone in hospitalized patients with Covid‐19. The New England Journal of Medicine, 384, 693–704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhodes, A. , Evans, L. E. , Alhazzani, W. , Levy, M. M. , Antonelli, M. , Ferrer, R. , & Dellinger, R. P. (2017). Surviving sepsis campaign: International guidelines for management of sepsis and septic shock: 2016. Critical Care Medicine, 45, 486–552. 10.1097/CCM.0000000000002255 [DOI] [PubMed] [Google Scholar]
- Rudd, K. E. , Johnson, S. C. , Agesa, K. M. , Shackelford, K. A. , Tsoi, D. , Kievlan, D. R. , Colombara, D. V. , Ikuta, K. S. , Kissoon, N. , Finfer, S. , & Fleischmann‐Struzek, C. (2020). Global, regional, and national sepsis incidence and mortality, 1990‐2017: Analysis for the global burden of disease study. Lancet, 395, 200–211. 10.1016/S0140-6736(19)32989-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salama, C. , Han, J. , Yau, L. , Reiss, W. G. , Kramer, B. , Neidhart, J. D. , Criner, G. J. , Kaplan‐Lewis, E. , Baden, R. , Pandit, L. , & Cameron, M. L. (2021). Tocilizumab in patients hospitalized with Covid‐19 pneumonia. The New England Journal of Medicine, 384, 20–30. 10.1056/NEJMoa2030340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schorah, C. J. , Downing, C. , Piripitsi, A. , Gallivan, L. , Al‐Hazaa, A. H. , Sanderson, M. J. , & Bodenham, A. (1996). Total vitamin C, ascorbic acid, and dehydroascorbic acid concentrations in plasma of critically ill patients. The American Journal of Clinical Nutrition, 63, 760–765. 10.1093/ajcn/63.5.760 [DOI] [PubMed] [Google Scholar]
- Singer, M. , Deutschman, C. S. , Seymour, C. W. , Shankar‐Hari, M. , Annane, D. , Bauer, M. , Bellomo, R. , Bernard, G. R. , Chiche, J. D. , Coopersmith, C. M. , & Hotchkiss, R. S. (2016). The third international consensus definitions for sepsis and septic shock (Sepsis‐3). JAMA: The Journal of the American Medical Association, 315, 801–810. 10.1001/jama.2016.0287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Subramanian, V. S. , Sabui, S. , Moradi, H. , Marchant, J. S. , & Said, H. M. (2018). Inhibition of intestinal ascorbic acid uptake by lipopolysaccharide is mediated via transcriptional mechanisms. Biochimica et Biophysica Acta ‐ Biomembranes, 1860, 556–565. 10.1016/j.bbamem.2017.10.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanase, F. , Fujii, T. , Naorungroj, T. , Belletti, A. , Luethi, N. , Carr, A. C. , Young, P. J. , & Bellomo, R. (2020). Harm of intravenous high dose vitamin C therapy in adult patients: A scoping review. Critical Care Medicine, 48, e620–e628. 10.1097/CCM.0000000000004396 [DOI] [PubMed] [Google Scholar]
- Zabet, M. H. , Mohammadi, M. , Ramezani, M. , & Khalili, H. (2016). Effect of high‐dose ascorbic acid on vasopressor's requirement in septic shock. Journal of Research in Pharmacy Practice, 5, 94–100. 10.4103/2279-042X.179569 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao, B. , Ling, Y. , Li, J. , Peng, Y. , Huang, J. , Wang, Y. , Qu, H. , Gao, Y. , Li, Y. , Hu, B. , & Lu, S. (2021). Beneficial aspects of high dose intravenous vitamin C on patients with COVID‐19 pneumonia in severe condition: A retrospective case series study. Annals of Palliative Medicine, 10, 1599–1609. 10.21037/apm-20-1387 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request. Some data may not be made available because of privacy or ethical restrictions.


